Long-Awaited RSV Vaccines Now Available for Older Adults and Pediatric Patients
Burton L. Lesnick, MD, FCCP
Pediatric Pulmonologist
Children’s Healthcare of Atlanta
Atlanta, GA
Slideshow below.
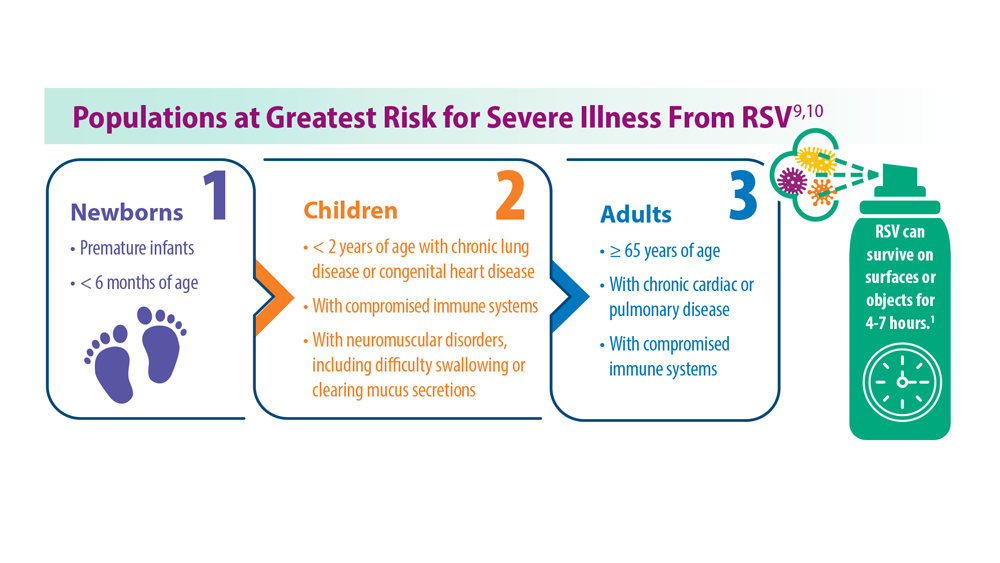
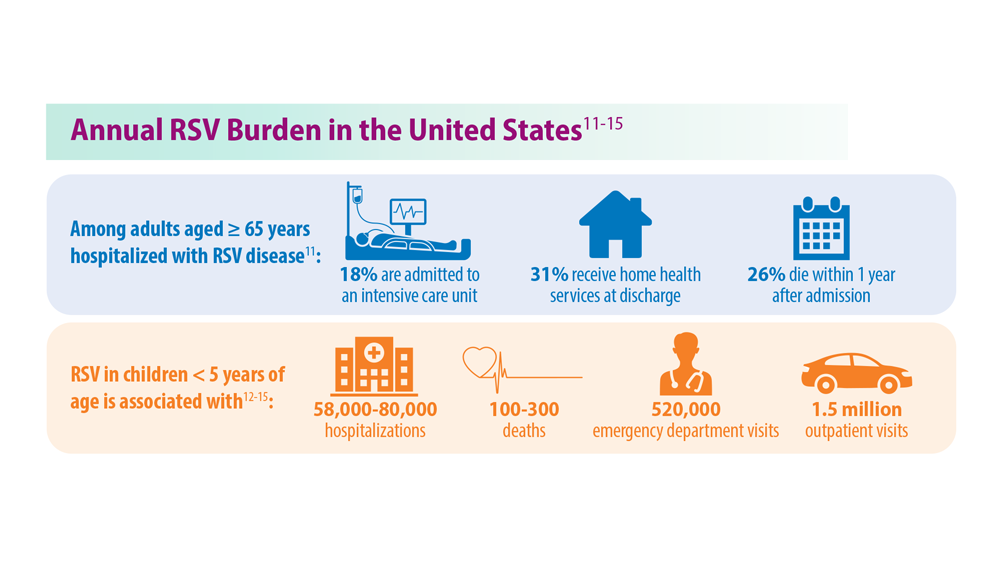
Respiratory syncytial virus (RSV) is highly contagious and transmitted by large aerosol droplets and fomites, either emitted from an infected person or by making surface-to-eye, -nose, or -mouth contact.1 Severe RSV can increase the risk of bacterial coinfections, pneumonia, and lower respiratory tract infections (LRTI)— particularly in infants and older adults.2
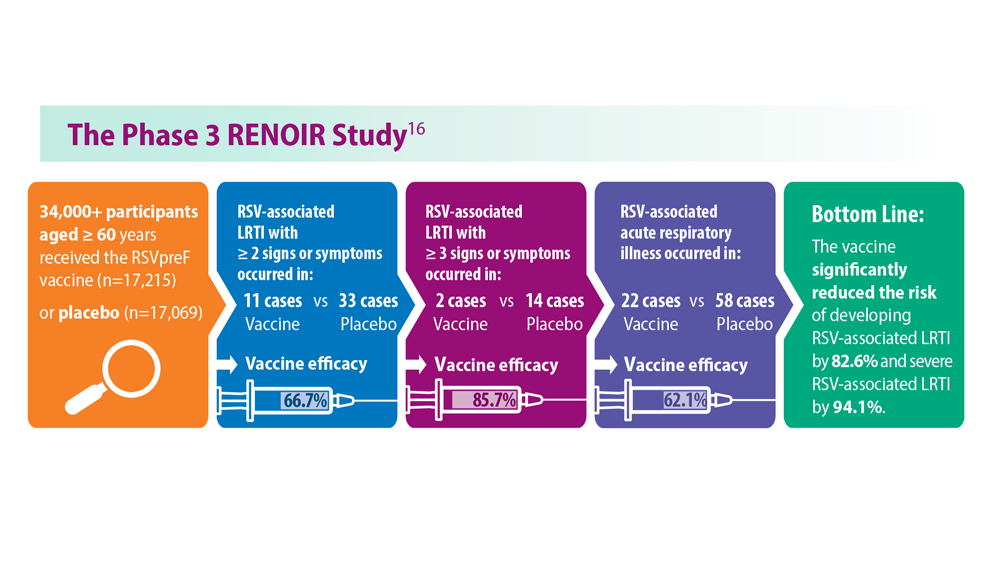
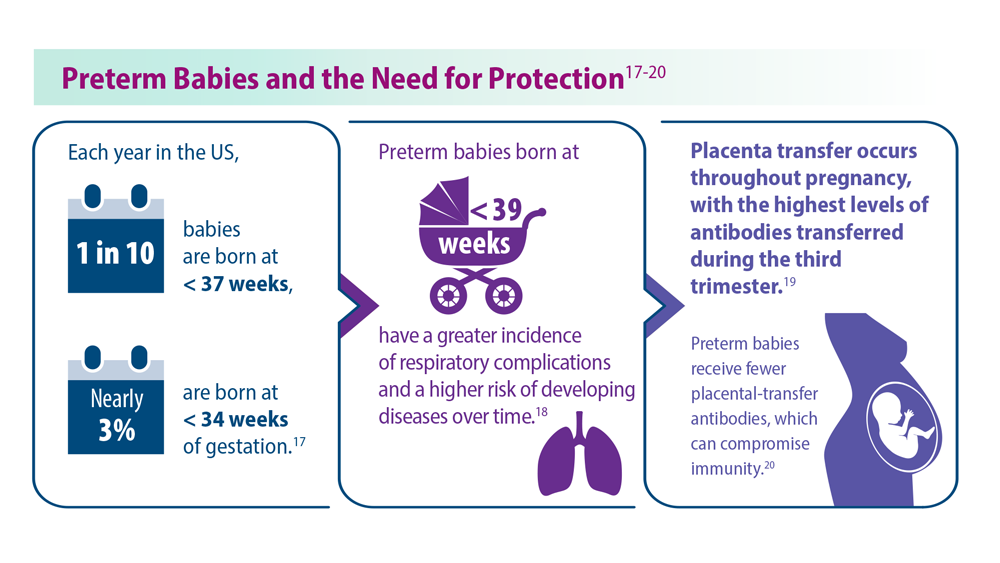
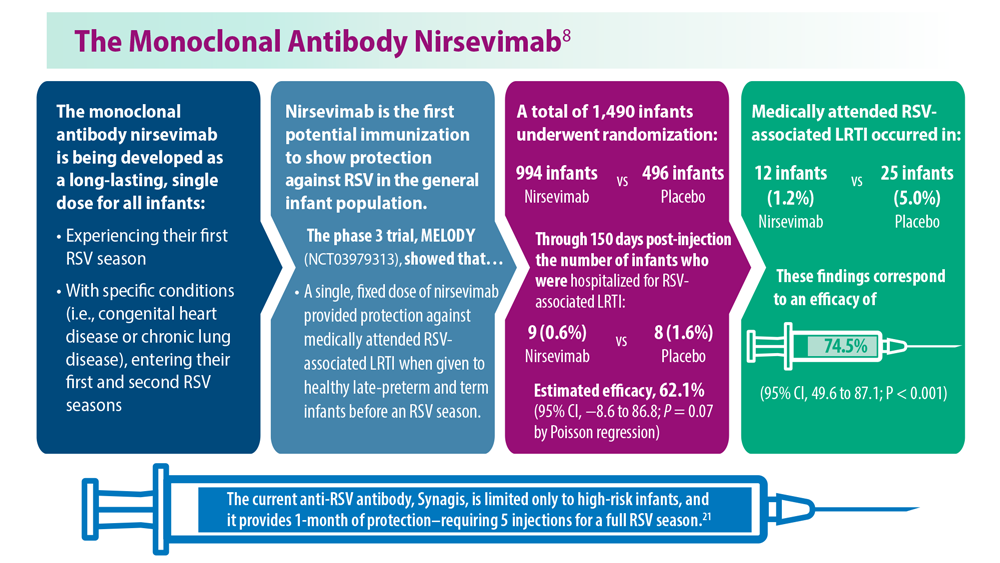
Thankfully, 2023 has been a landmark year for RSV approvals. The FDA approved its first RSV vaccine, called RSV prefusion F protein based (RSVpreF) vaccine, for people aged 60 and over in May 2023.3 In July 2023, the passive monoclonal antibody injection nirsevimab was approved as a preventative option for infants in their first and second winter seasons.4 Finally, the FDA approved the RSVpreF vaccine for pregnant individuals in late August 2023, with the goal of protecting infants.5 However, results from a recent phase 3 trial did not show significance with respect to the primary end point.6
Birth through 6 months is the leading timeframe of RSV-related death because of the low natural defenses and small airways of infants. On August 3, 2023, the CDC Advisory Committee on Immunization Practices unanimously recommended use of nirsevimab for all infants up to 8 months of age at the start of the RSV season and for infants at risk for severe RSV infection until 19 months of age.7 This decision was partly based on the MELODY and MEDLEY trials.8 In an unprecedented move, this monoclonal antibody will be made available through the Vaccines For Children program, the first monoclonal antibody to receive this designation. It is hoped that uptake of this therapy will result in fewer hospitalizations of infants with RSV bronchiolitis.
1