User login
The authors report no financial relationships relevant to this article.
CASE 1 Ovarian mass in a perimenopausal patient
A.R. is a 50-year-old gravida 3 para 3 who complains to her primary gynecologist of perimenopausal bleeding. A pelvic examination suggests an ovarian mass, and ultrasonography (US) reveals a myomatous uterus, thickened endometrium (34 mm), and a left ovarian cyst, with debris, that is 3.5×3.4×3.9 cm in size. The mass is thought to be a hemorrhagic cyst. Endometrial biopsy is benign.
Five weeks later, repeat US reveals that the mass has increased in size to 5.6×5.3×4.3 cm. It now appears complex in nature, with smooth walls and a single solid projection. The patient’s CA-125 level is 15 U/mL, which is in the normal range.
How should the mass be managed?
This scenario isn’t uncommon: Approximately one in every 10 women undergoes surgery for an adnexal mass, and an even higher percentage develop a mass that ultimately resolves or requires no surgery.1 Most of these lesions occur in women of reproductive age and are benign, often functional. The two groups at highest risk of malignancy are prepubescent and postmenopausal females.2,3 The rate of malignancy among prepubescent girls who have an adnexal mass is 35%; in postmenopausal women who have an adnexal mass, 30%.2,3
In this article, we describe how to evaluate and manage an adnexal mass in perimenopausal and postmenopausal women, as well as in the pregnant population, and outline the fundamentals of excision and surgical staging. Whenever possible, we base our observations on comprehensive guidelines and reliable data.
In the case just described, the increasing size of the mass and the complex appearance on follow-up imaging justify a surgical approach. Conservative management and definitive surgical treatment are the two treatment options for any adnexal mass.
Multipronged assessment is vital
Pelvic examination alone is insufficient to accurately assess ovarian size and internal characteristics of the mass, especially in postmenopausal and obese women.4 Nevertheless, the pelvic exam is a critical component of evaluation and often detects pathology. Pelvic examination also can assist in determining the best route of removal, depending on the mobility and size of the mass.
US yields the most information
US is the most effective tool for evaluating pelvic structures. It helps characterize masses and differentiates uterine, ovarian, and extraovarian tissues (FIGURE). If there is a high suspicion of malignancy, the next step is computed tomography (CT) to rule out metastatic disease.
US findings that suggest a malignant process are:
- solid component, not hyperechoic; usually nodular or papillary
- thick septations (2–3 mm)
- bilaterality
- positive flow to the solid component of the mass
- ascites.5
If a pregnant patient requires further evaluation for an adnexal mass, she should undergo magnetic resonance imaging (MRI) without contrast, not CT, to avoid radiation exposure. Although we lack studies of the safety of contrast agents in pregnant women, animal studies have demonstrated an increased rate of miscarriage, skeletal dysplasia, and visceral abnormalities with the use of contrast.6
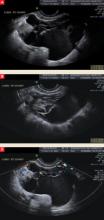
FIGURE The complex nature of a mass comes to light via ultrasonography
This complex mass of the right ovary, seen from different vantage points in A and B, contains multiple cysts and septations. Minimal blood flow is apparent in C.
An incidental mass may not require immediate surgery
A small (<5 cm in diameter), simple, asymptomatic mass may be followed conservatively (TABLE).7 Follow-up consists of repeat US, beginning within 4 to 6 weeks after detection.
Any postmenopausal woman who has a complex mass identified by US should undergo CA-125 testing and surgical excision. A mass in a premenopausal woman should also be removed if it has these characteristics—provided the mass is not functional and the complexity does not arise from hemorrhage associated with ovulation.
A program at the University of Kentucky enrolled 15,106 women older than 50 years to undergo annual transvaginal US for ovarian cancer screening.8 Of these, 18% were given a diagnosis of unilocular ovarian cysts, with an initial mean diameter of 2.7 cm; 69.4% of the cysts resolved spontaneously. No woman with an isolated unilocular cystic ovarian tumor developed ovarian cancer during the 6.3-year follow-up; the risk of malignancy was less than 0.1% with a 95% confidence interval. It therefore appears safe to follow small simple cysts in women of any age.
TABLE
When an adnexal mass is detected, possibilities are many
| EXTRAOVARIAN |
| Ectopic |
| Pedunculated fibroid |
| Hydrosalpinx |
| Tubo-ovarian abscess or diverticular abscess |
| Inclusion cyst |
| Fallopian tube cancer |
| Appendicial tumor |
| Pelvic kidney |
| OVARIAN |
Simple
|
Complex
|
Metastatic
|
Malignant, borderline, or benign
|
| * Three percent of germ-cell ovarian neoplasms are malignant; the majority are mature teratomas.7 |
Medical therapy might facilitate regression of the mass
Conservative management might also include medical therapy. Follicular cysts are very common in menarchal and perimenopausal women, and a trial of hormones, in the form of an oral contraceptive (OC) for 4 to 6 weeks, is a common strategy to prevent new cysts by suppressing ovulation. Such a trial is appropriate only for a premenopausal woman who has a simple cyst, however. A complex mass should generally not be observed unless the complexity is thought to be the result of a physiologic process. Six weeks of OC use is long enough to cause physiologic cysts to regress and to reveal which patients should proceed to surgery.9
Surgical treatment, staging
A mass that is suspicious for malignancy should be removed as soon as possible. If frozen section histology confirms the diagnosis, total abdominal hysterectomy with bilateral salpingo-oophorectomy is appropriate. If the patient desires childbearing and the cancer is of low grade and confined to the ovary, unilateral oophorectomy with ipsilateral nodes and staging is appropriate.
Surgical staging of ovarian malignancy should be carried out by a gynecologic oncologist. It involves removal of all mullerian structures, bilateral pelvic and periaortic lymph node sampling, peritoneal biopsies, and cytology or biopsy of the diaphragm. If there is no evidence of gross tumor, comprehensive staging with peritoneal biopsies, lymph node dissection, and cytology of the diaphragm is crucial.
Pathologic findings may necessitate upstaging of the patient and indicate the need for chemotherapy. Approximately 20% of patients who appear to have stage I or II localized disease have occult dissemination within the abdomen.10
If there is gross disease within the abdomen, the goal of surgery is to remove it.
When should you refer?
According to guidelines from the American College of Obstetricians and Gynecologists and the Society of Gynecologic Oncologists, a postmenopausal woman with an adnexal mass should be referred to a gynecologic oncologist when she has one or more of the following:
- nodular or fixed mass
- elevated CA-125 level (>35 U/mL)
- ascites
- evidence of metastasis on imaging
- strong family history of breast or ovarian cancer.11
A premenopausal woman should be referred if she has an adnexal mass and one or more of the following:
- elevated CA-125 (>200 U/mL)
- evidence of metastatic disease
- ascites.
Referral may also be appropriate if there is a first-degree relative with breast or ovarian cancer.11
CASE 1 Resolved
After the patient is counseled about the likelihood of malignancy, she undergoes exploratory laparotomy with frozen section. The ovary ruptures, and analysis of a frozen section is consistent with mullerian adenocarcinoma.
She then undergoes total abdominal hysterectomy and bilateral salpingooophorectomy. Gynecologic oncology is consulted, and complete staging follows, including omentectomy, peritoneal biopsies, and pelvic and periaortic lymph node dissection. Pathology reveals stage IC poorly differentiated adenocarcinoma, endometrioid type. Combination chemotherapy with carboplatin and a taxane is recommended.
CASE 2 Elderly patient with a complex mass
P.W., an 86-year-old gravida 9 para 4043, has an incidental adnexal mass detected during CT imaging. The left ovarian mass is complex and 7 cm in diameter at its largest point. The CA-125 level is 23 U/mL, and the carcinoembryonic antigen level is 4.5 ng/mL—both within normal range. A colonoscopy—performed as routine screening, not as part of the workup for the mass—is normal.
Because the mass is complex, surgery is indicated, and the physician prefers the laparoscopic approach—but is it reasonable?
Tumor markers should not be drawn reflexively with every adnexal mass. Clinical findings and diagnostic imaging must be considered to minimize false-positive test results. Do not order tumor markers without performing a thorough clinical evaluation.
A tumor isn’t the only pathology that produces elevated CA-125
Malignant epithelial tumors produce an elevated CA-125 level in 80% of cases.14 However, any disease state that causes inflammation of peritoneal surfaces will also produce an elevated CA-125 level. A few examples of disease states that cause inflammation of mesothelium-derived tissue are endometriosis, pancreatitis, colitis, pericarditis, diverticulitis, and ascites.15
Women who have an adnexal mass identified by pelvic exam should undergo US imaging. If imaging suggests that the mass is anything other than a simple cyst or functional, CA-125 measurement should follow. If imaging does not suggest malignancy, repeat US is indicated within 4 to 6 weeks to assure that the mass is resolving or is not increasing in size. Some masses in a postmenopausal woman may be followed if they are simple, less than 4 cm, and associated with a normal CA-125 level.
What level is cause for concern?
The normal CA-125 level for a postmenopausal woman is less than 30 to 35 U/mL, depending on the laboratory used. For a premenopausal woman, a normal level falls below 200 U/mL.
Young females who have a low likelihood of epithelial cancer do not need to undergo CA-125 measurement. CA-125 assessment has low sensitivity (0.5) and specificity (0.5) for epithelial cancer in premenarchal girls.16 Pubescent and prepubescent females should undergo measurement of the appropriate tumor markers for germ-cell or sex-cord tumors. Germ-cell tumor markers include α-fetoprotein, lactate dehydrogenase, and human chorionic gonadotropin. The sex-cord tumor marker is inhibin.
In cases such as this, the decision is best left to the discretion of the surgeon. If the mass is mobile and small enough to fit into a bag (to prevent spillage if it ruptures), laparoscopic removal is appropriate. As in other settings, laparoscopy speeds recovery and shortens hospitalization.
Laparoscopic removal of an adnexal mass is technically similar to an open procedure. After washings are obtained and the ureter is identified, the infundibulopelvic ligament is ligated or cauterized. The broad ligament anterior to the ureter is separated from the peritoneum. The utero-ovarian ligament is then cauterized, as is the fallopian tube, and the specimen is placed in a sealed bag. The bag is then generally removed through a 10-mm port, and the specimen is sent for pathologic evaluation.
If frozen section analysis indicates that a mass is malignant, a gynecologic oncologist can stage the patient during the same procedure. This staging can be performed laparoscopically if it is technically feasible and if the surgeon feels comfortable using this approach. If it is not possible to stage the patient at the initial surgery, staging should occur within 6 weeks after the original diagnosis.
For a discussion of the advisability of laparoscopy in a pregnant patient, see below.
CASE 2 Resolved
The patient undergoes bilateral salpingo-oophorectomy via a laparoscopic approach. During the procedure, the left ovary is placed into an endoscopic specimen bag and drained to allow adequate removal through the abdominal port site; no rupture occurs. Frozen section is benign, and the final pathology report shows the mass to be a serous cystadenoma.
The pregnant patient
CASE 3 Suspicious mass with abnormal vascularity
B.E. is a 25-year-old gravida 2 para 1001 who has a pelvic mass identified during a 20-week anomaly scan. The mass involves the left ovary and is 7.1 cm in size, well circumscribed, and solid, with multiple cystic spaces and increased flow apparent on color Doppler imaging. The mass is characterized by a large degree of abnormal vascularity, and an experienced ultrasonographer describes it as “worrisome for malignancy.” MRI is performed, and the findings are consistent with those of ultrasonography but without evidence of malignant spread. Tumor markers are within normal limits, except for ß-human chorionic gonadotropin, which is elevated for the obvious reason.
Is surgery appropriate?
Gravidas develop pelvic masses at a significant rate, with a prevalence of approximately 2.3%, according to a study of 18,391 pregnant women who underwent US imaging at Washington University between 1988 and 1993.12 The majority of patients who had an adnexal mass—76%, or 320 women—had a simple cyst that was less than 5 cm in diameter and associated with no adverse events. The other 24%, or 102 women, had a mass larger than 5 cm, either simple or complex in nature. Most masses resolved spontaneously, and only 25 required surgical removal.12 No invasive carcinomas were found.
Despite the long odds of malignancy, an adnexal mass in pregnancy warrants close evaluation and follow-up and, occasionally, surgical management.
Fine-needle aspiration of an adnexal mass is rarely appropriate. In one study, 105 ovarian specimens were removed intact and the results of cyst cytology (from fine-needle aspiration) and final ovarian histology were compared.17 (Cytologic fluid was obtained by the pathologist after intact ovary removal—not preoperatively.) Histology revealed 89 benign ovarian tumors and 13 ovarian carcinomas. The sensitivity of fine-needle aspiration was 25%, with a specificity of 90%. The false-positive rate for fine-needle aspiration was 73%, and the false-negative rate was 12%.
Biopsy is risky
Malignant cystic lesions should be biopsied only in a patient who has advanced disease confirmed, or when it is necessary to check for recurrence, to avoid spreading malignant cells in localized tumors.18
General ObGyns and primary care physicians should not make the decision to biopsy an adnexal mass. The need for such a decision is grounds for referral. Nor does a patient require a diagnosis of cancer to be referred to a gynecologic oncologist. An oncologist may elect to biopsy a woman who is a poor surgical candidate, in whom chemotherapy may be first-line therapy in the neoadjuvant setting.
When is surgery justified?
Cholecystitis, appendicitis, and ovarian torsion are common diagnoses that require operative intervention regardless of gestational age.
Otherwise, when a complex adnexal mass is identified during pregnancy and is symptomatic or large enough to require removal, the gynecologist should proceed with surgery, whenever possible, in the second trimester—the most opportune time for removal. In some women, the ideal time for surgical removal of a mass detected during pregnancy is around 18 weeks’ gestation, but certainly before 24 weeks. Given the position of the gravid uterus, exploratory laparotomy is preferred over laparoscopy at this stage of gestation.
Intraoperative evaluation of the mass by pathology with frozen section is recommended. Even though immediate staging may not be feasible, owing to the pregnancy, pathology results can reassure the patient and her family and also facilitate planning of the optimal time and route of delivery.
If the mass is determined to be malignant, the patient should undergo surgical staging after completion of the pregnancy. At 37 weeks, labor should be induced, with staging performed within the next 2 to 4 weeks, or a cesarean section should be performed, with staging carried out at that time.
Laparoscopy may be feasible in the first trimester
Because laparoscopy can be difficult to perform during pregnancy, it should be used judiciously; uterine size can limit visibility and hinder safe placement of trocars. The first trimester is the least problematic period for laparoscopy.
A large study in Sweden compared laparoscopy with laparotomy between 4 and 20 weeks’ gestation and assessed fetal outcomes.13 In the study, 2,181 women underwent laparoscopy and 1,522 underwent laparotomy. Low birth weight (<2,500 g), intrauterine growth restriction, and delivery before 37 weeks’ gestation increased among all surgical patients, with no differences attributed to the route of the procedure. Nor were there significant differences between surgical and nonsurgical patients in either infant survival at 1 year or the incidence of fetal malformation.
As long as the anesthesiologist is aware of the pregnancy, a general surgeon can safely perform either laparoscopy or laparotomy during the first or second trimester. Care should be taken not to remove a corpus luteum before the 14th week of gestation. Pregnancy should not alter the surgeon’s preferred treatment approach at this time, unless uterine size is the limiting factor.
CASE 3 Resolved
B.E. safely undergoes exploratory laparotomy and left oophorectomy at 23-4/7 weeks’ gestation. Frozen section indicates that the mass is a malignant neoplasm. The final pathology report describes a highly unusual constellation of histologic findings, including juvenile granulosa cell tumor, dysgerminoma, and gonadoblastoma. A cesarean delivery with completion of cancer staging is planned when the fetus achieves lung maturity, with preservation of the contralateral ovary and uterus nodal sampling, peritoneal biopsies, and omentectomy.
Reference
1. Hilger WS, Margina JF, Magtibay PM. Laparoscopic management of the adnexal mass. Clin Obstet Gynecol. 2006;49:535-548.
2. Hoffman M. Differential diagnosis of the adnexal mass. UpToDate Online. Available at http://utdol.com. Accessed March 10, 2007.
3. Breen JL, Maxson WS. Ovarian tumors in children and adolescents. Clin Obstet Gynecol. 1977;20:607-623.
4. Van Nagell JR, Depriest PD. Management of adnexal masses in postmenopausal women. Am J Obstet Gynecol. 2005;193:30-35.
5. Brown D. Sonographic differentiation of benign versus malignant adnexal masses. UpToDate Online. Available at http://www.utdol.com. Accessed March 10, 2007.
6. ACOG Committee on Obstetric Practice. ACOG Committee Opinion. Number 299, September 2004 (replaces Number 158, September 1995). Guidelines for diagnostic imaging during pregnancy. Obstet Gynecol. 2004;104:647-651.
7. Berek J. Novak’s Gynecology. 13th ed. Philadelphia: Lippincott, Williams & Wilkins; 2002.
8. Modesitt SC, Pavlik EJ, Ueland FR, DePriest PD, Kryscio RJ, van Nagell JR. Risk of malignancy in unilocular ovarian cystic tumors less than 10 centimeters in diameter. Obstet Gynecol. 2003;102:594-599.
9. Spanos WJ. Preoperative hormonal therapy of cystic adnexal masses. Am J Obstet Gynecol. 1973;116:551-556.
10. Young RC, Fisher RI. The staging and treatment of epithelial ovarian cancer. Can Med Assoc J. 1978;119:249-256.
11. Gostout BS, Brewer MA. Guideline for referral of the patient with an adnexal mass. Clin Obstet Gynecol. 2006;49:448-458.
12. Bernhard LM, Klebba PK, Gray DL, Mutch DG. Predictors of persistence of adnexal masses in pregnancy. Obstet Gynecol. 1999;93:585-589.
13. Reddy MB, Kallen B, Kuehl TJ. Laparoscopy during pregnancy: a study of five fetal outcome parameters with use of the Swedish Health Registry. Am J Obstet Gynecol. 1997;177:673-679.
14. Silberstein LB, Rosenthal AN, Coppack SW, Noonan K, Jacobs IJ. Ascites and a raised serum CA-125—confusing combination. J R Soc Med. 2001;94:581-582.
15. Rosenthal AN, Menon U, Jacobs IJ. Screening for ovarian cancer. Clin Obstet Gynecol. 2006;49:443-447.
16. Stankovic Z, Djuricic S, Djukic M, Jovanovic D, Vasiljevic M. Epithelial ovarian tumors and CA125 in premenarchal girls. Eur J Gynaecol Oncol. 2006;27:597-599.
17. Higgins RV, Matkins JF, Marroum MC. Comparison of fine-needle aspiration cytologic findings of ovarian cysts with ovarian histologic findings. Am J Obstet Gynecol. 1999;180(3 Pt 1):550-553.
18. Zanetta G, Trio D, Lissoni A, et al. Early and short-term complications after US-guided puncture of gynecologic lesions: evaluation after 1,000 consecutive cases. Radiology. 1993;189:161-164.
The authors report no financial relationships relevant to this article.
CASE 1 Ovarian mass in a perimenopausal patient
A.R. is a 50-year-old gravida 3 para 3 who complains to her primary gynecologist of perimenopausal bleeding. A pelvic examination suggests an ovarian mass, and ultrasonography (US) reveals a myomatous uterus, thickened endometrium (34 mm), and a left ovarian cyst, with debris, that is 3.5×3.4×3.9 cm in size. The mass is thought to be a hemorrhagic cyst. Endometrial biopsy is benign.
Five weeks later, repeat US reveals that the mass has increased in size to 5.6×5.3×4.3 cm. It now appears complex in nature, with smooth walls and a single solid projection. The patient’s CA-125 level is 15 U/mL, which is in the normal range.
How should the mass be managed?
This scenario isn’t uncommon: Approximately one in every 10 women undergoes surgery for an adnexal mass, and an even higher percentage develop a mass that ultimately resolves or requires no surgery.1 Most of these lesions occur in women of reproductive age and are benign, often functional. The two groups at highest risk of malignancy are prepubescent and postmenopausal females.2,3 The rate of malignancy among prepubescent girls who have an adnexal mass is 35%; in postmenopausal women who have an adnexal mass, 30%.2,3
In this article, we describe how to evaluate and manage an adnexal mass in perimenopausal and postmenopausal women, as well as in the pregnant population, and outline the fundamentals of excision and surgical staging. Whenever possible, we base our observations on comprehensive guidelines and reliable data.
In the case just described, the increasing size of the mass and the complex appearance on follow-up imaging justify a surgical approach. Conservative management and definitive surgical treatment are the two treatment options for any adnexal mass.
Multipronged assessment is vital
Pelvic examination alone is insufficient to accurately assess ovarian size and internal characteristics of the mass, especially in postmenopausal and obese women.4 Nevertheless, the pelvic exam is a critical component of evaluation and often detects pathology. Pelvic examination also can assist in determining the best route of removal, depending on the mobility and size of the mass.
US yields the most information
US is the most effective tool for evaluating pelvic structures. It helps characterize masses and differentiates uterine, ovarian, and extraovarian tissues (FIGURE). If there is a high suspicion of malignancy, the next step is computed tomography (CT) to rule out metastatic disease.
US findings that suggest a malignant process are:
- solid component, not hyperechoic; usually nodular or papillary
- thick septations (2–3 mm)
- bilaterality
- positive flow to the solid component of the mass
- ascites.5
If a pregnant patient requires further evaluation for an adnexal mass, she should undergo magnetic resonance imaging (MRI) without contrast, not CT, to avoid radiation exposure. Although we lack studies of the safety of contrast agents in pregnant women, animal studies have demonstrated an increased rate of miscarriage, skeletal dysplasia, and visceral abnormalities with the use of contrast.6
FIGURE The complex nature of a mass comes to light via ultrasonography
This complex mass of the right ovary, seen from different vantage points in A and B, contains multiple cysts and septations. Minimal blood flow is apparent in C.
An incidental mass may not require immediate surgery
A small (<5 cm in diameter), simple, asymptomatic mass may be followed conservatively (TABLE).7 Follow-up consists of repeat US, beginning within 4 to 6 weeks after detection.
Any postmenopausal woman who has a complex mass identified by US should undergo CA-125 testing and surgical excision. A mass in a premenopausal woman should also be removed if it has these characteristics—provided the mass is not functional and the complexity does not arise from hemorrhage associated with ovulation.
A program at the University of Kentucky enrolled 15,106 women older than 50 years to undergo annual transvaginal US for ovarian cancer screening.8 Of these, 18% were given a diagnosis of unilocular ovarian cysts, with an initial mean diameter of 2.7 cm; 69.4% of the cysts resolved spontaneously. No woman with an isolated unilocular cystic ovarian tumor developed ovarian cancer during the 6.3-year follow-up; the risk of malignancy was less than 0.1% with a 95% confidence interval. It therefore appears safe to follow small simple cysts in women of any age.
TABLE
When an adnexal mass is detected, possibilities are many
| EXTRAOVARIAN |
| Ectopic |
| Pedunculated fibroid |
| Hydrosalpinx |
| Tubo-ovarian abscess or diverticular abscess |
| Inclusion cyst |
| Fallopian tube cancer |
| Appendicial tumor |
| Pelvic kidney |
| OVARIAN |
Simple
|
Complex
|
Metastatic
|
Malignant, borderline, or benign
|
| * Three percent of germ-cell ovarian neoplasms are malignant; the majority are mature teratomas.7 |
Medical therapy might facilitate regression of the mass
Conservative management might also include medical therapy. Follicular cysts are very common in menarchal and perimenopausal women, and a trial of hormones, in the form of an oral contraceptive (OC) for 4 to 6 weeks, is a common strategy to prevent new cysts by suppressing ovulation. Such a trial is appropriate only for a premenopausal woman who has a simple cyst, however. A complex mass should generally not be observed unless the complexity is thought to be the result of a physiologic process. Six weeks of OC use is long enough to cause physiologic cysts to regress and to reveal which patients should proceed to surgery.9
Surgical treatment, staging
A mass that is suspicious for malignancy should be removed as soon as possible. If frozen section histology confirms the diagnosis, total abdominal hysterectomy with bilateral salpingo-oophorectomy is appropriate. If the patient desires childbearing and the cancer is of low grade and confined to the ovary, unilateral oophorectomy with ipsilateral nodes and staging is appropriate.
Surgical staging of ovarian malignancy should be carried out by a gynecologic oncologist. It involves removal of all mullerian structures, bilateral pelvic and periaortic lymph node sampling, peritoneal biopsies, and cytology or biopsy of the diaphragm. If there is no evidence of gross tumor, comprehensive staging with peritoneal biopsies, lymph node dissection, and cytology of the diaphragm is crucial.
Pathologic findings may necessitate upstaging of the patient and indicate the need for chemotherapy. Approximately 20% of patients who appear to have stage I or II localized disease have occult dissemination within the abdomen.10
If there is gross disease within the abdomen, the goal of surgery is to remove it.
When should you refer?
According to guidelines from the American College of Obstetricians and Gynecologists and the Society of Gynecologic Oncologists, a postmenopausal woman with an adnexal mass should be referred to a gynecologic oncologist when she has one or more of the following:
- nodular or fixed mass
- elevated CA-125 level (>35 U/mL)
- ascites
- evidence of metastasis on imaging
- strong family history of breast or ovarian cancer.11
A premenopausal woman should be referred if she has an adnexal mass and one or more of the following:
- elevated CA-125 (>200 U/mL)
- evidence of metastatic disease
- ascites.
Referral may also be appropriate if there is a first-degree relative with breast or ovarian cancer.11
CASE 1 Resolved
After the patient is counseled about the likelihood of malignancy, she undergoes exploratory laparotomy with frozen section. The ovary ruptures, and analysis of a frozen section is consistent with mullerian adenocarcinoma.
She then undergoes total abdominal hysterectomy and bilateral salpingooophorectomy. Gynecologic oncology is consulted, and complete staging follows, including omentectomy, peritoneal biopsies, and pelvic and periaortic lymph node dissection. Pathology reveals stage IC poorly differentiated adenocarcinoma, endometrioid type. Combination chemotherapy with carboplatin and a taxane is recommended.
CASE 2 Elderly patient with a complex mass
P.W., an 86-year-old gravida 9 para 4043, has an incidental adnexal mass detected during CT imaging. The left ovarian mass is complex and 7 cm in diameter at its largest point. The CA-125 level is 23 U/mL, and the carcinoembryonic antigen level is 4.5 ng/mL—both within normal range. A colonoscopy—performed as routine screening, not as part of the workup for the mass—is normal.
Because the mass is complex, surgery is indicated, and the physician prefers the laparoscopic approach—but is it reasonable?
Tumor markers should not be drawn reflexively with every adnexal mass. Clinical findings and diagnostic imaging must be considered to minimize false-positive test results. Do not order tumor markers without performing a thorough clinical evaluation.
A tumor isn’t the only pathology that produces elevated CA-125
Malignant epithelial tumors produce an elevated CA-125 level in 80% of cases.14 However, any disease state that causes inflammation of peritoneal surfaces will also produce an elevated CA-125 level. A few examples of disease states that cause inflammation of mesothelium-derived tissue are endometriosis, pancreatitis, colitis, pericarditis, diverticulitis, and ascites.15
Women who have an adnexal mass identified by pelvic exam should undergo US imaging. If imaging suggests that the mass is anything other than a simple cyst or functional, CA-125 measurement should follow. If imaging does not suggest malignancy, repeat US is indicated within 4 to 6 weeks to assure that the mass is resolving or is not increasing in size. Some masses in a postmenopausal woman may be followed if they are simple, less than 4 cm, and associated with a normal CA-125 level.
What level is cause for concern?
The normal CA-125 level for a postmenopausal woman is less than 30 to 35 U/mL, depending on the laboratory used. For a premenopausal woman, a normal level falls below 200 U/mL.
Young females who have a low likelihood of epithelial cancer do not need to undergo CA-125 measurement. CA-125 assessment has low sensitivity (0.5) and specificity (0.5) for epithelial cancer in premenarchal girls.16 Pubescent and prepubescent females should undergo measurement of the appropriate tumor markers for germ-cell or sex-cord tumors. Germ-cell tumor markers include α-fetoprotein, lactate dehydrogenase, and human chorionic gonadotropin. The sex-cord tumor marker is inhibin.
In cases such as this, the decision is best left to the discretion of the surgeon. If the mass is mobile and small enough to fit into a bag (to prevent spillage if it ruptures), laparoscopic removal is appropriate. As in other settings, laparoscopy speeds recovery and shortens hospitalization.
Laparoscopic removal of an adnexal mass is technically similar to an open procedure. After washings are obtained and the ureter is identified, the infundibulopelvic ligament is ligated or cauterized. The broad ligament anterior to the ureter is separated from the peritoneum. The utero-ovarian ligament is then cauterized, as is the fallopian tube, and the specimen is placed in a sealed bag. The bag is then generally removed through a 10-mm port, and the specimen is sent for pathologic evaluation.
If frozen section analysis indicates that a mass is malignant, a gynecologic oncologist can stage the patient during the same procedure. This staging can be performed laparoscopically if it is technically feasible and if the surgeon feels comfortable using this approach. If it is not possible to stage the patient at the initial surgery, staging should occur within 6 weeks after the original diagnosis.
For a discussion of the advisability of laparoscopy in a pregnant patient, see below.
CASE 2 Resolved
The patient undergoes bilateral salpingo-oophorectomy via a laparoscopic approach. During the procedure, the left ovary is placed into an endoscopic specimen bag and drained to allow adequate removal through the abdominal port site; no rupture occurs. Frozen section is benign, and the final pathology report shows the mass to be a serous cystadenoma.
The pregnant patient
CASE 3 Suspicious mass with abnormal vascularity
B.E. is a 25-year-old gravida 2 para 1001 who has a pelvic mass identified during a 20-week anomaly scan. The mass involves the left ovary and is 7.1 cm in size, well circumscribed, and solid, with multiple cystic spaces and increased flow apparent on color Doppler imaging. The mass is characterized by a large degree of abnormal vascularity, and an experienced ultrasonographer describes it as “worrisome for malignancy.” MRI is performed, and the findings are consistent with those of ultrasonography but without evidence of malignant spread. Tumor markers are within normal limits, except for ß-human chorionic gonadotropin, which is elevated for the obvious reason.
Is surgery appropriate?
Gravidas develop pelvic masses at a significant rate, with a prevalence of approximately 2.3%, according to a study of 18,391 pregnant women who underwent US imaging at Washington University between 1988 and 1993.12 The majority of patients who had an adnexal mass—76%, or 320 women—had a simple cyst that was less than 5 cm in diameter and associated with no adverse events. The other 24%, or 102 women, had a mass larger than 5 cm, either simple or complex in nature. Most masses resolved spontaneously, and only 25 required surgical removal.12 No invasive carcinomas were found.
Despite the long odds of malignancy, an adnexal mass in pregnancy warrants close evaluation and follow-up and, occasionally, surgical management.
Fine-needle aspiration of an adnexal mass is rarely appropriate. In one study, 105 ovarian specimens were removed intact and the results of cyst cytology (from fine-needle aspiration) and final ovarian histology were compared.17 (Cytologic fluid was obtained by the pathologist after intact ovary removal—not preoperatively.) Histology revealed 89 benign ovarian tumors and 13 ovarian carcinomas. The sensitivity of fine-needle aspiration was 25%, with a specificity of 90%. The false-positive rate for fine-needle aspiration was 73%, and the false-negative rate was 12%.
Biopsy is risky
Malignant cystic lesions should be biopsied only in a patient who has advanced disease confirmed, or when it is necessary to check for recurrence, to avoid spreading malignant cells in localized tumors.18
General ObGyns and primary care physicians should not make the decision to biopsy an adnexal mass. The need for such a decision is grounds for referral. Nor does a patient require a diagnosis of cancer to be referred to a gynecologic oncologist. An oncologist may elect to biopsy a woman who is a poor surgical candidate, in whom chemotherapy may be first-line therapy in the neoadjuvant setting.
When is surgery justified?
Cholecystitis, appendicitis, and ovarian torsion are common diagnoses that require operative intervention regardless of gestational age.
Otherwise, when a complex adnexal mass is identified during pregnancy and is symptomatic or large enough to require removal, the gynecologist should proceed with surgery, whenever possible, in the second trimester—the most opportune time for removal. In some women, the ideal time for surgical removal of a mass detected during pregnancy is around 18 weeks’ gestation, but certainly before 24 weeks. Given the position of the gravid uterus, exploratory laparotomy is preferred over laparoscopy at this stage of gestation.
Intraoperative evaluation of the mass by pathology with frozen section is recommended. Even though immediate staging may not be feasible, owing to the pregnancy, pathology results can reassure the patient and her family and also facilitate planning of the optimal time and route of delivery.
If the mass is determined to be malignant, the patient should undergo surgical staging after completion of the pregnancy. At 37 weeks, labor should be induced, with staging performed within the next 2 to 4 weeks, or a cesarean section should be performed, with staging carried out at that time.
Laparoscopy may be feasible in the first trimester
Because laparoscopy can be difficult to perform during pregnancy, it should be used judiciously; uterine size can limit visibility and hinder safe placement of trocars. The first trimester is the least problematic period for laparoscopy.
A large study in Sweden compared laparoscopy with laparotomy between 4 and 20 weeks’ gestation and assessed fetal outcomes.13 In the study, 2,181 women underwent laparoscopy and 1,522 underwent laparotomy. Low birth weight (<2,500 g), intrauterine growth restriction, and delivery before 37 weeks’ gestation increased among all surgical patients, with no differences attributed to the route of the procedure. Nor were there significant differences between surgical and nonsurgical patients in either infant survival at 1 year or the incidence of fetal malformation.
As long as the anesthesiologist is aware of the pregnancy, a general surgeon can safely perform either laparoscopy or laparotomy during the first or second trimester. Care should be taken not to remove a corpus luteum before the 14th week of gestation. Pregnancy should not alter the surgeon’s preferred treatment approach at this time, unless uterine size is the limiting factor.
CASE 3 Resolved
B.E. safely undergoes exploratory laparotomy and left oophorectomy at 23-4/7 weeks’ gestation. Frozen section indicates that the mass is a malignant neoplasm. The final pathology report describes a highly unusual constellation of histologic findings, including juvenile granulosa cell tumor, dysgerminoma, and gonadoblastoma. A cesarean delivery with completion of cancer staging is planned when the fetus achieves lung maturity, with preservation of the contralateral ovary and uterus nodal sampling, peritoneal biopsies, and omentectomy.
The authors report no financial relationships relevant to this article.
CASE 1 Ovarian mass in a perimenopausal patient
A.R. is a 50-year-old gravida 3 para 3 who complains to her primary gynecologist of perimenopausal bleeding. A pelvic examination suggests an ovarian mass, and ultrasonography (US) reveals a myomatous uterus, thickened endometrium (34 mm), and a left ovarian cyst, with debris, that is 3.5×3.4×3.9 cm in size. The mass is thought to be a hemorrhagic cyst. Endometrial biopsy is benign.
Five weeks later, repeat US reveals that the mass has increased in size to 5.6×5.3×4.3 cm. It now appears complex in nature, with smooth walls and a single solid projection. The patient’s CA-125 level is 15 U/mL, which is in the normal range.
How should the mass be managed?
This scenario isn’t uncommon: Approximately one in every 10 women undergoes surgery for an adnexal mass, and an even higher percentage develop a mass that ultimately resolves or requires no surgery.1 Most of these lesions occur in women of reproductive age and are benign, often functional. The two groups at highest risk of malignancy are prepubescent and postmenopausal females.2,3 The rate of malignancy among prepubescent girls who have an adnexal mass is 35%; in postmenopausal women who have an adnexal mass, 30%.2,3
In this article, we describe how to evaluate and manage an adnexal mass in perimenopausal and postmenopausal women, as well as in the pregnant population, and outline the fundamentals of excision and surgical staging. Whenever possible, we base our observations on comprehensive guidelines and reliable data.
In the case just described, the increasing size of the mass and the complex appearance on follow-up imaging justify a surgical approach. Conservative management and definitive surgical treatment are the two treatment options for any adnexal mass.
Multipronged assessment is vital
Pelvic examination alone is insufficient to accurately assess ovarian size and internal characteristics of the mass, especially in postmenopausal and obese women.4 Nevertheless, the pelvic exam is a critical component of evaluation and often detects pathology. Pelvic examination also can assist in determining the best route of removal, depending on the mobility and size of the mass.
US yields the most information
US is the most effective tool for evaluating pelvic structures. It helps characterize masses and differentiates uterine, ovarian, and extraovarian tissues (FIGURE). If there is a high suspicion of malignancy, the next step is computed tomography (CT) to rule out metastatic disease.
US findings that suggest a malignant process are:
- solid component, not hyperechoic; usually nodular or papillary
- thick septations (2–3 mm)
- bilaterality
- positive flow to the solid component of the mass
- ascites.5
If a pregnant patient requires further evaluation for an adnexal mass, she should undergo magnetic resonance imaging (MRI) without contrast, not CT, to avoid radiation exposure. Although we lack studies of the safety of contrast agents in pregnant women, animal studies have demonstrated an increased rate of miscarriage, skeletal dysplasia, and visceral abnormalities with the use of contrast.6
FIGURE The complex nature of a mass comes to light via ultrasonography
This complex mass of the right ovary, seen from different vantage points in A and B, contains multiple cysts and septations. Minimal blood flow is apparent in C.
An incidental mass may not require immediate surgery
A small (<5 cm in diameter), simple, asymptomatic mass may be followed conservatively (TABLE).7 Follow-up consists of repeat US, beginning within 4 to 6 weeks after detection.
Any postmenopausal woman who has a complex mass identified by US should undergo CA-125 testing and surgical excision. A mass in a premenopausal woman should also be removed if it has these characteristics—provided the mass is not functional and the complexity does not arise from hemorrhage associated with ovulation.
A program at the University of Kentucky enrolled 15,106 women older than 50 years to undergo annual transvaginal US for ovarian cancer screening.8 Of these, 18% were given a diagnosis of unilocular ovarian cysts, with an initial mean diameter of 2.7 cm; 69.4% of the cysts resolved spontaneously. No woman with an isolated unilocular cystic ovarian tumor developed ovarian cancer during the 6.3-year follow-up; the risk of malignancy was less than 0.1% with a 95% confidence interval. It therefore appears safe to follow small simple cysts in women of any age.
TABLE
When an adnexal mass is detected, possibilities are many
| EXTRAOVARIAN |
| Ectopic |
| Pedunculated fibroid |
| Hydrosalpinx |
| Tubo-ovarian abscess or diverticular abscess |
| Inclusion cyst |
| Fallopian tube cancer |
| Appendicial tumor |
| Pelvic kidney |
| OVARIAN |
Simple
|
Complex
|
Metastatic
|
Malignant, borderline, or benign
|
| * Three percent of germ-cell ovarian neoplasms are malignant; the majority are mature teratomas.7 |
Medical therapy might facilitate regression of the mass
Conservative management might also include medical therapy. Follicular cysts are very common in menarchal and perimenopausal women, and a trial of hormones, in the form of an oral contraceptive (OC) for 4 to 6 weeks, is a common strategy to prevent new cysts by suppressing ovulation. Such a trial is appropriate only for a premenopausal woman who has a simple cyst, however. A complex mass should generally not be observed unless the complexity is thought to be the result of a physiologic process. Six weeks of OC use is long enough to cause physiologic cysts to regress and to reveal which patients should proceed to surgery.9
Surgical treatment, staging
A mass that is suspicious for malignancy should be removed as soon as possible. If frozen section histology confirms the diagnosis, total abdominal hysterectomy with bilateral salpingo-oophorectomy is appropriate. If the patient desires childbearing and the cancer is of low grade and confined to the ovary, unilateral oophorectomy with ipsilateral nodes and staging is appropriate.
Surgical staging of ovarian malignancy should be carried out by a gynecologic oncologist. It involves removal of all mullerian structures, bilateral pelvic and periaortic lymph node sampling, peritoneal biopsies, and cytology or biopsy of the diaphragm. If there is no evidence of gross tumor, comprehensive staging with peritoneal biopsies, lymph node dissection, and cytology of the diaphragm is crucial.
Pathologic findings may necessitate upstaging of the patient and indicate the need for chemotherapy. Approximately 20% of patients who appear to have stage I or II localized disease have occult dissemination within the abdomen.10
If there is gross disease within the abdomen, the goal of surgery is to remove it.
When should you refer?
According to guidelines from the American College of Obstetricians and Gynecologists and the Society of Gynecologic Oncologists, a postmenopausal woman with an adnexal mass should be referred to a gynecologic oncologist when she has one or more of the following:
- nodular or fixed mass
- elevated CA-125 level (>35 U/mL)
- ascites
- evidence of metastasis on imaging
- strong family history of breast or ovarian cancer.11
A premenopausal woman should be referred if she has an adnexal mass and one or more of the following:
- elevated CA-125 (>200 U/mL)
- evidence of metastatic disease
- ascites.
Referral may also be appropriate if there is a first-degree relative with breast or ovarian cancer.11
CASE 1 Resolved
After the patient is counseled about the likelihood of malignancy, she undergoes exploratory laparotomy with frozen section. The ovary ruptures, and analysis of a frozen section is consistent with mullerian adenocarcinoma.
She then undergoes total abdominal hysterectomy and bilateral salpingooophorectomy. Gynecologic oncology is consulted, and complete staging follows, including omentectomy, peritoneal biopsies, and pelvic and periaortic lymph node dissection. Pathology reveals stage IC poorly differentiated adenocarcinoma, endometrioid type. Combination chemotherapy with carboplatin and a taxane is recommended.
CASE 2 Elderly patient with a complex mass
P.W., an 86-year-old gravida 9 para 4043, has an incidental adnexal mass detected during CT imaging. The left ovarian mass is complex and 7 cm in diameter at its largest point. The CA-125 level is 23 U/mL, and the carcinoembryonic antigen level is 4.5 ng/mL—both within normal range. A colonoscopy—performed as routine screening, not as part of the workup for the mass—is normal.
Because the mass is complex, surgery is indicated, and the physician prefers the laparoscopic approach—but is it reasonable?
Tumor markers should not be drawn reflexively with every adnexal mass. Clinical findings and diagnostic imaging must be considered to minimize false-positive test results. Do not order tumor markers without performing a thorough clinical evaluation.
A tumor isn’t the only pathology that produces elevated CA-125
Malignant epithelial tumors produce an elevated CA-125 level in 80% of cases.14 However, any disease state that causes inflammation of peritoneal surfaces will also produce an elevated CA-125 level. A few examples of disease states that cause inflammation of mesothelium-derived tissue are endometriosis, pancreatitis, colitis, pericarditis, diverticulitis, and ascites.15
Women who have an adnexal mass identified by pelvic exam should undergo US imaging. If imaging suggests that the mass is anything other than a simple cyst or functional, CA-125 measurement should follow. If imaging does not suggest malignancy, repeat US is indicated within 4 to 6 weeks to assure that the mass is resolving or is not increasing in size. Some masses in a postmenopausal woman may be followed if they are simple, less than 4 cm, and associated with a normal CA-125 level.
What level is cause for concern?
The normal CA-125 level for a postmenopausal woman is less than 30 to 35 U/mL, depending on the laboratory used. For a premenopausal woman, a normal level falls below 200 U/mL.
Young females who have a low likelihood of epithelial cancer do not need to undergo CA-125 measurement. CA-125 assessment has low sensitivity (0.5) and specificity (0.5) for epithelial cancer in premenarchal girls.16 Pubescent and prepubescent females should undergo measurement of the appropriate tumor markers for germ-cell or sex-cord tumors. Germ-cell tumor markers include α-fetoprotein, lactate dehydrogenase, and human chorionic gonadotropin. The sex-cord tumor marker is inhibin.
In cases such as this, the decision is best left to the discretion of the surgeon. If the mass is mobile and small enough to fit into a bag (to prevent spillage if it ruptures), laparoscopic removal is appropriate. As in other settings, laparoscopy speeds recovery and shortens hospitalization.
Laparoscopic removal of an adnexal mass is technically similar to an open procedure. After washings are obtained and the ureter is identified, the infundibulopelvic ligament is ligated or cauterized. The broad ligament anterior to the ureter is separated from the peritoneum. The utero-ovarian ligament is then cauterized, as is the fallopian tube, and the specimen is placed in a sealed bag. The bag is then generally removed through a 10-mm port, and the specimen is sent for pathologic evaluation.
If frozen section analysis indicates that a mass is malignant, a gynecologic oncologist can stage the patient during the same procedure. This staging can be performed laparoscopically if it is technically feasible and if the surgeon feels comfortable using this approach. If it is not possible to stage the patient at the initial surgery, staging should occur within 6 weeks after the original diagnosis.
For a discussion of the advisability of laparoscopy in a pregnant patient, see below.
CASE 2 Resolved
The patient undergoes bilateral salpingo-oophorectomy via a laparoscopic approach. During the procedure, the left ovary is placed into an endoscopic specimen bag and drained to allow adequate removal through the abdominal port site; no rupture occurs. Frozen section is benign, and the final pathology report shows the mass to be a serous cystadenoma.
The pregnant patient
CASE 3 Suspicious mass with abnormal vascularity
B.E. is a 25-year-old gravida 2 para 1001 who has a pelvic mass identified during a 20-week anomaly scan. The mass involves the left ovary and is 7.1 cm in size, well circumscribed, and solid, with multiple cystic spaces and increased flow apparent on color Doppler imaging. The mass is characterized by a large degree of abnormal vascularity, and an experienced ultrasonographer describes it as “worrisome for malignancy.” MRI is performed, and the findings are consistent with those of ultrasonography but without evidence of malignant spread. Tumor markers are within normal limits, except for ß-human chorionic gonadotropin, which is elevated for the obvious reason.
Is surgery appropriate?
Gravidas develop pelvic masses at a significant rate, with a prevalence of approximately 2.3%, according to a study of 18,391 pregnant women who underwent US imaging at Washington University between 1988 and 1993.12 The majority of patients who had an adnexal mass—76%, or 320 women—had a simple cyst that was less than 5 cm in diameter and associated with no adverse events. The other 24%, or 102 women, had a mass larger than 5 cm, either simple or complex in nature. Most masses resolved spontaneously, and only 25 required surgical removal.12 No invasive carcinomas were found.
Despite the long odds of malignancy, an adnexal mass in pregnancy warrants close evaluation and follow-up and, occasionally, surgical management.
Fine-needle aspiration of an adnexal mass is rarely appropriate. In one study, 105 ovarian specimens were removed intact and the results of cyst cytology (from fine-needle aspiration) and final ovarian histology were compared.17 (Cytologic fluid was obtained by the pathologist after intact ovary removal—not preoperatively.) Histology revealed 89 benign ovarian tumors and 13 ovarian carcinomas. The sensitivity of fine-needle aspiration was 25%, with a specificity of 90%. The false-positive rate for fine-needle aspiration was 73%, and the false-negative rate was 12%.
Biopsy is risky
Malignant cystic lesions should be biopsied only in a patient who has advanced disease confirmed, or when it is necessary to check for recurrence, to avoid spreading malignant cells in localized tumors.18
General ObGyns and primary care physicians should not make the decision to biopsy an adnexal mass. The need for such a decision is grounds for referral. Nor does a patient require a diagnosis of cancer to be referred to a gynecologic oncologist. An oncologist may elect to biopsy a woman who is a poor surgical candidate, in whom chemotherapy may be first-line therapy in the neoadjuvant setting.
When is surgery justified?
Cholecystitis, appendicitis, and ovarian torsion are common diagnoses that require operative intervention regardless of gestational age.
Otherwise, when a complex adnexal mass is identified during pregnancy and is symptomatic or large enough to require removal, the gynecologist should proceed with surgery, whenever possible, in the second trimester—the most opportune time for removal. In some women, the ideal time for surgical removal of a mass detected during pregnancy is around 18 weeks’ gestation, but certainly before 24 weeks. Given the position of the gravid uterus, exploratory laparotomy is preferred over laparoscopy at this stage of gestation.
Intraoperative evaluation of the mass by pathology with frozen section is recommended. Even though immediate staging may not be feasible, owing to the pregnancy, pathology results can reassure the patient and her family and also facilitate planning of the optimal time and route of delivery.
If the mass is determined to be malignant, the patient should undergo surgical staging after completion of the pregnancy. At 37 weeks, labor should be induced, with staging performed within the next 2 to 4 weeks, or a cesarean section should be performed, with staging carried out at that time.
Laparoscopy may be feasible in the first trimester
Because laparoscopy can be difficult to perform during pregnancy, it should be used judiciously; uterine size can limit visibility and hinder safe placement of trocars. The first trimester is the least problematic period for laparoscopy.
A large study in Sweden compared laparoscopy with laparotomy between 4 and 20 weeks’ gestation and assessed fetal outcomes.13 In the study, 2,181 women underwent laparoscopy and 1,522 underwent laparotomy. Low birth weight (<2,500 g), intrauterine growth restriction, and delivery before 37 weeks’ gestation increased among all surgical patients, with no differences attributed to the route of the procedure. Nor were there significant differences between surgical and nonsurgical patients in either infant survival at 1 year or the incidence of fetal malformation.
As long as the anesthesiologist is aware of the pregnancy, a general surgeon can safely perform either laparoscopy or laparotomy during the first or second trimester. Care should be taken not to remove a corpus luteum before the 14th week of gestation. Pregnancy should not alter the surgeon’s preferred treatment approach at this time, unless uterine size is the limiting factor.
CASE 3 Resolved
B.E. safely undergoes exploratory laparotomy and left oophorectomy at 23-4/7 weeks’ gestation. Frozen section indicates that the mass is a malignant neoplasm. The final pathology report describes a highly unusual constellation of histologic findings, including juvenile granulosa cell tumor, dysgerminoma, and gonadoblastoma. A cesarean delivery with completion of cancer staging is planned when the fetus achieves lung maturity, with preservation of the contralateral ovary and uterus nodal sampling, peritoneal biopsies, and omentectomy.
Reference
1. Hilger WS, Margina JF, Magtibay PM. Laparoscopic management of the adnexal mass. Clin Obstet Gynecol. 2006;49:535-548.
2. Hoffman M. Differential diagnosis of the adnexal mass. UpToDate Online. Available at http://utdol.com. Accessed March 10, 2007.
3. Breen JL, Maxson WS. Ovarian tumors in children and adolescents. Clin Obstet Gynecol. 1977;20:607-623.
4. Van Nagell JR, Depriest PD. Management of adnexal masses in postmenopausal women. Am J Obstet Gynecol. 2005;193:30-35.
5. Brown D. Sonographic differentiation of benign versus malignant adnexal masses. UpToDate Online. Available at http://www.utdol.com. Accessed March 10, 2007.
6. ACOG Committee on Obstetric Practice. ACOG Committee Opinion. Number 299, September 2004 (replaces Number 158, September 1995). Guidelines for diagnostic imaging during pregnancy. Obstet Gynecol. 2004;104:647-651.
7. Berek J. Novak’s Gynecology. 13th ed. Philadelphia: Lippincott, Williams & Wilkins; 2002.
8. Modesitt SC, Pavlik EJ, Ueland FR, DePriest PD, Kryscio RJ, van Nagell JR. Risk of malignancy in unilocular ovarian cystic tumors less than 10 centimeters in diameter. Obstet Gynecol. 2003;102:594-599.
9. Spanos WJ. Preoperative hormonal therapy of cystic adnexal masses. Am J Obstet Gynecol. 1973;116:551-556.
10. Young RC, Fisher RI. The staging and treatment of epithelial ovarian cancer. Can Med Assoc J. 1978;119:249-256.
11. Gostout BS, Brewer MA. Guideline for referral of the patient with an adnexal mass. Clin Obstet Gynecol. 2006;49:448-458.
12. Bernhard LM, Klebba PK, Gray DL, Mutch DG. Predictors of persistence of adnexal masses in pregnancy. Obstet Gynecol. 1999;93:585-589.
13. Reddy MB, Kallen B, Kuehl TJ. Laparoscopy during pregnancy: a study of five fetal outcome parameters with use of the Swedish Health Registry. Am J Obstet Gynecol. 1997;177:673-679.
14. Silberstein LB, Rosenthal AN, Coppack SW, Noonan K, Jacobs IJ. Ascites and a raised serum CA-125—confusing combination. J R Soc Med. 2001;94:581-582.
15. Rosenthal AN, Menon U, Jacobs IJ. Screening for ovarian cancer. Clin Obstet Gynecol. 2006;49:443-447.
16. Stankovic Z, Djuricic S, Djukic M, Jovanovic D, Vasiljevic M. Epithelial ovarian tumors and CA125 in premenarchal girls. Eur J Gynaecol Oncol. 2006;27:597-599.
17. Higgins RV, Matkins JF, Marroum MC. Comparison of fine-needle aspiration cytologic findings of ovarian cysts with ovarian histologic findings. Am J Obstet Gynecol. 1999;180(3 Pt 1):550-553.
18. Zanetta G, Trio D, Lissoni A, et al. Early and short-term complications after US-guided puncture of gynecologic lesions: evaluation after 1,000 consecutive cases. Radiology. 1993;189:161-164.
Reference
1. Hilger WS, Margina JF, Magtibay PM. Laparoscopic management of the adnexal mass. Clin Obstet Gynecol. 2006;49:535-548.
2. Hoffman M. Differential diagnosis of the adnexal mass. UpToDate Online. Available at http://utdol.com. Accessed March 10, 2007.
3. Breen JL, Maxson WS. Ovarian tumors in children and adolescents. Clin Obstet Gynecol. 1977;20:607-623.
4. Van Nagell JR, Depriest PD. Management of adnexal masses in postmenopausal women. Am J Obstet Gynecol. 2005;193:30-35.
5. Brown D. Sonographic differentiation of benign versus malignant adnexal masses. UpToDate Online. Available at http://www.utdol.com. Accessed March 10, 2007.
6. ACOG Committee on Obstetric Practice. ACOG Committee Opinion. Number 299, September 2004 (replaces Number 158, September 1995). Guidelines for diagnostic imaging during pregnancy. Obstet Gynecol. 2004;104:647-651.
7. Berek J. Novak’s Gynecology. 13th ed. Philadelphia: Lippincott, Williams & Wilkins; 2002.
8. Modesitt SC, Pavlik EJ, Ueland FR, DePriest PD, Kryscio RJ, van Nagell JR. Risk of malignancy in unilocular ovarian cystic tumors less than 10 centimeters in diameter. Obstet Gynecol. 2003;102:594-599.
9. Spanos WJ. Preoperative hormonal therapy of cystic adnexal masses. Am J Obstet Gynecol. 1973;116:551-556.
10. Young RC, Fisher RI. The staging and treatment of epithelial ovarian cancer. Can Med Assoc J. 1978;119:249-256.
11. Gostout BS, Brewer MA. Guideline for referral of the patient with an adnexal mass. Clin Obstet Gynecol. 2006;49:448-458.
12. Bernhard LM, Klebba PK, Gray DL, Mutch DG. Predictors of persistence of adnexal masses in pregnancy. Obstet Gynecol. 1999;93:585-589.
13. Reddy MB, Kallen B, Kuehl TJ. Laparoscopy during pregnancy: a study of five fetal outcome parameters with use of the Swedish Health Registry. Am J Obstet Gynecol. 1997;177:673-679.
14. Silberstein LB, Rosenthal AN, Coppack SW, Noonan K, Jacobs IJ. Ascites and a raised serum CA-125—confusing combination. J R Soc Med. 2001;94:581-582.
15. Rosenthal AN, Menon U, Jacobs IJ. Screening for ovarian cancer. Clin Obstet Gynecol. 2006;49:443-447.
16. Stankovic Z, Djuricic S, Djukic M, Jovanovic D, Vasiljevic M. Epithelial ovarian tumors and CA125 in premenarchal girls. Eur J Gynaecol Oncol. 2006;27:597-599.
17. Higgins RV, Matkins JF, Marroum MC. Comparison of fine-needle aspiration cytologic findings of ovarian cysts with ovarian histologic findings. Am J Obstet Gynecol. 1999;180(3 Pt 1):550-553.
18. Zanetta G, Trio D, Lissoni A, et al. Early and short-term complications after US-guided puncture of gynecologic lesions: evaluation after 1,000 consecutive cases. Radiology. 1993;189:161-164.