User login
CASE: Agitation
Mrs. M, age 39, presents to the emergency department (ED) with altered mental status. She is escorted by her husband and the police. She has a history of severe alcohol dependence, bipolar disorder (BD), anxiety, borderline personality disorder (BPD), hypothyroidism, and bulimia, and had gastric bypass surgery 4 years ago. Her husband called 911 when he could no longer manage Mrs. M’s agitated state. The police found her to be extremely paranoid, restless, and disoriented. Her husband reports that she shouted “the world is going to end” before she escaped naked into her neighborhood streets.
On several occasions Mrs. M had been admitted to the same hospital for alcohol withdrawal and dependence with subsequent liver failure, leading to jaundice, coagulopathy, and ascites. During these hospitalizations, she exhibited poor behavioral tendencies, unhealthy psychological defenses, and chronic maladaptive coping and defense mechanisms congruent with her BPD diagnosis. Specifically, she engaged in splitting of hospital staff, ranging from extreme flattery to overt devaluation and hostility. Other defense mechanisms included denial, distortion, acting out, and passive-aggressive behavior. During these admissions, Mrs. M often displayed deficits in recall and attention on Mini-Mental State Examination (MMSE), but these deficits were associated with concurrent alcohol use and improved rapidly during her stay.
In her current presentation, Mrs. M’s mental status change is more pronounced and atypical compared with earlier admissions. Her outpatient medication regimen includes lamotrigine, 100 mg/d, levothyroxine, 88 mcg/d, venlafaxine extended release (XR), 75 mg/d, clonazepam, 3 mg/d, docusate as needed for constipation, and a daily multivitamin.
The authors’ observations
Delirium is a disturbance of consciousness manifested by a reduced clarity of awareness (impairment in attention) and change in cognition (impairment in orientation, memory, and language).1,2 The disturbance develops over a short time and tends to fluctuate during the day. Delirium is a direct physiological consequence of a general medical condition, substance use (intoxication or withdrawal), or both (Table).3
Delirium generally is a reversible mental disorder but can progress to irreversible brain damage. Prompt and accurate diagnosis of delirium is essential,4 although the condition often is underdiagnosed or misdiagnosed because of lack of recognition.
Table
DSM-IV-TR diagnostic criteria for delirium
|
| Source: Reference 3 |
Patients who have convoluted histories, such as Mrs. M, are common and difficult to manage and treat. These patients become substantially more complex when they are admitted to inpatient medical or surgical services. The need to clarify between delirium (primarily medical) and depression (primarily psychiatric) becomes paramount when administering treatment and evaluating decision-making capacity.5 In Mrs. M’s case, internal medicine, neurology, and psychiatry teams each had a different approach to altered mental status. Each team’s different terminology, assessment, and objectives further complicated an already challenging case.6
EVALUATION: Confounding results
The ED physicians offer a working diagnosis of acute mental status change, administer IV lorazepam, 4 mg, and order restraints for Mrs. M’s severe agitation. Her initial vital signs reveal slightly elevated blood pressure (140/90 mm Hg) and tachycardia (115 beats per minute). Internal medicine clinicians note that Mrs. M is not in acute distress, although she refuses to speak and has a small amount of dried blood on her lips, presumably from a struggle with the police before coming to the hospital, but this is not certain. Her abdomen is not tender; she has normal bowel sounds, and no asterixis is noted on neurologic exam. Physical exam is otherwise normal. A noncontrast head CT scan shows no acute process. Initial lab values show elevations in ammonia (277 μg/dL) and γ-glutamyl transpeptidase (68 U/L). Thyroid-stimulating hormone is 1.45 mlU/L, prothrombin time is 19.5 s, partial thromboplastin time is 40.3 s, and international normalized ratio is 1.67. The internal medicine team admits Mrs. M to the intensive care unit (ICU) for further management of her mental status change with alcohol withdrawal or hepatic encephalopathy as the most likely etiologies.
Mrs. M’s husband says that his wife has not consumed alcohol in the last 4 months in preparation for a possible liver transplant; however, past interactions with Mrs. M’s family suggest they are unreliable. The Clinical Institute Withdrawal Assessment (CIWA) protocol is implemented in case her symptoms are caused by alcohol withdrawal. Her vital signs are stable and IV lorazepam, 4 mg, is administered once for agitation. Mrs. M’s husband also reports that 1 month ago his wife underwent a transjugular intrahepatic portosystemic shunt (TIPS) procedure for portal hypertension. Outpatient psychotropics (lamotrigine, 100 mg/d, and venlafaxine XR, 75 mg/d) are restarted because withdrawal from these drugs may exacerbate her symptoms. In the ICU Mrs. M experiences a tonic-clonic seizure with fecal incontinence and bitten tongue, which results in a consultation from neurology and the psychiatry consultation-liaison service.
Psychiatry recommends withholding psychotropics, stopping CIWA, and using vital sign parameters along with objective signs of diaphoresis and tremors as indicators of alcohol withdrawal for lorazepam administration. Mrs. M receives IV haloperidol, 1 mg, once during her second day in the hospital for severe agitation, but this medication is discontinued because of concern about lowering her seizure threshold.7 After treatment with lactulose, her ammonia levels trend down to 33 μg/dL, but her altered mental state persists with significant deficits in attention and orientation.
The neurology service performs an EEG that shows no slow-wave, triphasic waves, or epileptiform activity, which likely would be present in delirium or seizures. See Figure 1 for an example of triphasic waves on an EEG and Figure 2 for Mrs. M's EEG results. Subsequent lumbar puncture, MRI, and a second EEG are unremarkable. By the fifth hospital day, Mrs. M is calm and her paranoia has subsided, but she still is confused and disoriented. Psychiatry orders a third EEG while she is in this confused state; it shows no pathologic process. Based on these examinations, neurology posits that Mrs. M is not encephalopathic.
Figure 1: Representative sample of triphasic waves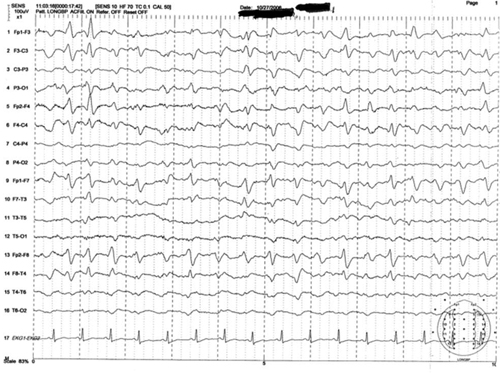
This EEG tracing is from a 54-year-old woman who underwent prolonged abdominal surgery for lysis of adhesions during which she suffered an intraoperative left subinsular stroke followed by nonconvulsive status epilepticus. The tracing demonstrates typical morphology with the positive sharp transient preceded and followed by smaller amplitude negative deflections. Symmetric, frontal predominance of findings seen is this tracing is common
Figure 2: Mrs. M’s EEG results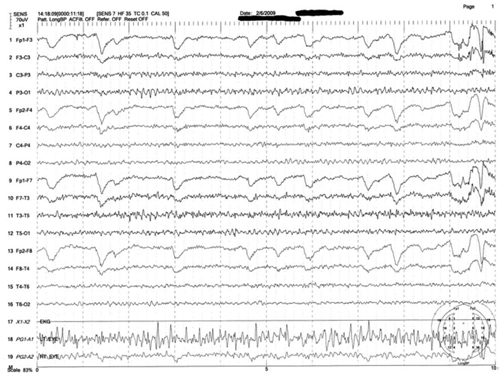
This is a representative tracing of Mrs. M’s 3 EEGs revealing an 8.5 to 9 Hz dominant alpha rhythm. There is superimposed frontally dominant beta fast activity, which is consistent with known administration of benzodiazepines
The authors’ observations
Mrs. M had repeated admissions for alcohol dependence and subsequent liver failure. Her recent hospitalization was complicated by a TIPS procedure done 1 month ago. The incidence of hepatic encephalopathy in patients undergoing TIPS is >30%, especially in the first month post-procedure, which raised suspicion that hepatic encephalopathy played a significant role in Mrs. M’s delirium.8
Because of frequent hospitalization, Mrs. M was well known to the internal medicine, neurology, and psychiatry teams, and each used different terms to describe her mental state. Internal medicine used the phrase “acute mental status change,” which covers a broad differential. Neurology used “encephalopathy,” which also is a general term. Psychiatry used “delirium,” which has narrower and more specific diagnostic criteria. Engel et al9 described the delirious patient as having “cerebral insufficiency” with universally abnormal EEG. Regardless of terminology, based on Mrs. M’s acute confusion, one would expect an abnormal EEG, but repeat EEGs were unremarkable.
Interpreting EEG
EEG is one of the few tools available for measuring acute changes in cerebral function, and an EEG slowing remains a hallmark in encephalopathic processes.10,11 Initially, the 3 specialties agreed that Mrs. M’s presentation likely was caused by underlying medical issues or substances (alcohol or others). EEG can help recognize delirium, and, in some cases, elucidate the underlying cause.10,12 It was surprising that Mrs. M’s EEGs were normal despite a clinical presentation of delirium. Because of the normal EEG findings, neurology leaned toward a primary psychiatric (“functional”) etiology as the cause of her delirium vs a general medical condition or alcohol withdrawal (“organic”).
A literature search in regards to sensitivity of EEG in delirium revealed conflicting statements and data. A standard textbook in neurology and psychiatry states that “a normal EEG virtually excludes a toxic-metabolic encephalopathy.”13 The American Psychiatric Association’s (APA) practice guidelines for delirium states: “The presence of EEG abnormalities has fairly good sensitivities for delirium (in one study, the sensitivity was found to be 75%), but the absence does not rule out the diagnosis; thus the EEG is no substitute for careful clinical observation.”6
At the beginning of Mrs. M’s care, in discussion with the neurology and internal medicine teams, we argued that Mrs. M was experiencing delirium despite her initial normal EEG. We did not expect that 2 subsequent EEGs would be normal, especially because the teams witnessed the final EEG being performed while Mrs. M was clinically evaluated and observed to be in a state of delirium.
OUTCOME: Cause still unknown
By the 6th day of hospitalization, Mrs. M’s vitals are normal and she remains hemodynamically stable. Differential diagnosis remains wide and unclear. The psychiatry team feels she could have atypical catatonia due to an underlying mood disorder. One hour after a trial of IV lorazepam, 1 mg, Mrs. M is more lucid and fully oriented, with MMSE of 28/30 (recall was 1/3), indicating normal cognition. During the exam, a psychiatry resident notes Mrs. M winks and feigns a yawn at the medical students and nurses in the room, displaying her boredom with the interview and simplicity of the mental status exam questions. Later that evening, Mrs. M exhibits bizarre sexual gestures toward male hospital staff, including licking a male nursing staff member’s hand.
Although Mrs. M’s initial confusion resolved, the severity of her comorbid psychiatric history warrants inpatient psychiatric hospitalization. She agrees to transfer to the psychiatric ward after she confesses anxiety regarding death, intense demoralization, and guilt related to her condition and her relationship with her 12-year-old daughter. She tearfully reports that she discontinued her psychotropic medications shortly after stopping alcohol 4 months ago. Shortly before her transfer, psychiatry is called back to the medicine floor because of Mrs. M’s disruptive behavior.
The team finds Mrs. M in her hospital gown, pursuing her husband in the hallway as he is leaving, yelling profanities and blaming him for her horrible experience in the hospital. Based on her demeanor, the team determines that she is back to her baseline mental state despite her mood disorder, and that her upcoming inpatient psychiatric stay likely would be too short to address her comorbid personality disorder. The next day she signs out of the hospital against medical advice.
The authors’ observations
We never clearly identified the specific etiology responsible for Mrs. M’s delirium. We assume at the initial presentation she had toxic-metabolic encephalopathy that rapidly resolved with lactulose treatment and lowering her ammonia. She then had a single tonic-clonic seizure, perhaps related to stopping and then restarting her psychotropics. Her subsequent confusion, bizarre sexual behavior, and demeanor on her final hospital days were more indicative of her psychiatric diagnoses. We now suspect that Mrs. M’s delirium was briefer than presumed and she returned to her baseline borderline personality, resulting in some factitious staging of delirium to confuse her 3 treating teams (a psychoanalyst may say this was a form of projective identification).
We felt that if Mrs. M truly was delirious due to metabolic or hepatic dysfunction or alcohol withdrawal, she would have had abnormal EEG findings. We discovered that the notion of “75% sensitivity” of EEG abnormalities cited in the APA guidelines comes from studies that include patients with “psychogenic” and “organic” delirium. Acute manias and agitated psychoses were termed “psychogenic delirium” and acute confusion due to medical conditions or substance issues was termed “organic delirium.”9,12,14-16
This poses a circular reasoning in the diagnostic criteria and clinical approach to delirium. The fallacy is that, according to DSM-IV-TR, delirium is supposed to be the result of a direct physiological consequence of a general medical condition or substance use (criterion D), and cannot be due to psychosis (eg, schizophrenia) or mania (eg, BD). We question the presumptive 75% sensitivity of EEG abnormalities in patients with delirium because it is possible that when some of these studies were conducted the definition of delirium was not solidified or fully understood. We suspect the sensitivity would be much higher if the correct definition of delirium according to DSM-IV-TR is used in future studies. To improve interdisciplinary communication and future research, it would be constructive if all disciplines could agree on a single term, with the same diagnostic criteria, when evaluating a patient with acute confusion.
Related Resources
- Meagher D. Delirium: the role of psychiatry. Advances in Psychiatric Treatment. 2001;7:433-442.
- Casey DA, DeFazio JV Jr, Vansickle K, et al. Delirium. Quick recognition, careful evaluation, and appropriate treatment. Postgrad Med. 1996;100(1):121-4, 128, 133-134.
Drug Brand Names
- Clonazepam • Klonopin
- Docusate • Surfak
- Haloperidol • Haldol
- Lamotrigine • Lamictal
- Lorazepam • Ativan
- Levothyroxine • Levoxyl, Synthtoid
- Venlafaxine XR • Effexor XR
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgment
The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, or the U.S. Government. The authors are employees of the U.S. Government. This work was prepared as part of their official duties. Title 17 U.S.C. 105 provides that “Copyright protection under this title is not available for any work of the U.S. Government.” Title 17 U.S.C. 101 defines a U.S. Government work as a work prepared by a military service member or employee of the U.S. Government as part of that person’s official duties.
1. Katz IR, Mossey J, Sussman N, et al. Bedside clinical and electrophysiological assessment: assessment of change in vulnerable patients. Int Psychogeriatr. 1991;3(2):289-300.
2. Inouye SK. Delirium in older persons. N Engl J Med. 2006;354(11):1157-1165.
3. Diagnostic and statistical manual of mental disorders, 4th ed, text rev. Washington DC: American Psychiatric Association; 2000.
4. McPhee SJ, Papadakis M, Rabow MW. CURRENT medical diagnosis and treatment. New York NY: McGraw Hill Medical; 2012.
5. Brody B. Who has capacity? N Engl J Med. 2009;361(3):232-233.
6. Practice guideline for the treatment of patients with delirium. American Psychiatric Association. Am J Psychiatry. 1999;156(5 suppl):1-20.
7. Fricchione GL, Nejad SH, Esses JA, et al. Postoperative delirium. Am J Psychiatry. 2008;165(7):803-812.
8. Sanyal AJ, Freedman AM, Shiffman ML, et al. Portosystemic encephalopathy after transjugular intrahepatic portosystemic shunt: results of a prospective controlled study. Hepatology. 1994;20(1 pt 1):46-55.
9. Engel GL, Romano J. Delirium a syndrome of cerebral insufficiency. 1959. J Neuropsychiatry Clin Neurosci. 2004;16(4):526-538.
10. Pro JD, Wells CE. The use of the electroencephalogram in the diagnosis of delirium. Dis Nerv Syst. 1977;38(10):804-808.
11. Sidhu KS, Balon R, Ajluni V, et al. Standard EEG and the difficult-to-assess mental status. Ann Clin Psychiatry. 2009;21(2):103-108.
12. Brenner RP. Utility of EEG in delirium: past views and current practice. Int Psychogeriatr. 1991;3(2):211-229.
13. Kaufman DM. Clinical neurology for psychiatrists. 5th ed. Philadelphia PA: Saunders; 2001: 230-232.
14. Bond TC. Recognition of acute delirious mania. Arch Gen Psychiatry. 1980;37(5):553-554.
15. Krauthammer C, Klerman GL. Secondary mania: manic syndromes associated with antecedent physical illness or drugs. Arch Gen Psychiatry. 1978;35(11):1333-1339.
16. Larson EW, Richelson E. Organic causes of mania. Mayo Clin Proc. 1988;63(9):906-912.
CASE: Agitation
Mrs. M, age 39, presents to the emergency department (ED) with altered mental status. She is escorted by her husband and the police. She has a history of severe alcohol dependence, bipolar disorder (BD), anxiety, borderline personality disorder (BPD), hypothyroidism, and bulimia, and had gastric bypass surgery 4 years ago. Her husband called 911 when he could no longer manage Mrs. M’s agitated state. The police found her to be extremely paranoid, restless, and disoriented. Her husband reports that she shouted “the world is going to end” before she escaped naked into her neighborhood streets.
On several occasions Mrs. M had been admitted to the same hospital for alcohol withdrawal and dependence with subsequent liver failure, leading to jaundice, coagulopathy, and ascites. During these hospitalizations, she exhibited poor behavioral tendencies, unhealthy psychological defenses, and chronic maladaptive coping and defense mechanisms congruent with her BPD diagnosis. Specifically, she engaged in splitting of hospital staff, ranging from extreme flattery to overt devaluation and hostility. Other defense mechanisms included denial, distortion, acting out, and passive-aggressive behavior. During these admissions, Mrs. M often displayed deficits in recall and attention on Mini-Mental State Examination (MMSE), but these deficits were associated with concurrent alcohol use and improved rapidly during her stay.
In her current presentation, Mrs. M’s mental status change is more pronounced and atypical compared with earlier admissions. Her outpatient medication regimen includes lamotrigine, 100 mg/d, levothyroxine, 88 mcg/d, venlafaxine extended release (XR), 75 mg/d, clonazepam, 3 mg/d, docusate as needed for constipation, and a daily multivitamin.
The authors’ observations
Delirium is a disturbance of consciousness manifested by a reduced clarity of awareness (impairment in attention) and change in cognition (impairment in orientation, memory, and language).1,2 The disturbance develops over a short time and tends to fluctuate during the day. Delirium is a direct physiological consequence of a general medical condition, substance use (intoxication or withdrawal), or both (Table).3
Delirium generally is a reversible mental disorder but can progress to irreversible brain damage. Prompt and accurate diagnosis of delirium is essential,4 although the condition often is underdiagnosed or misdiagnosed because of lack of recognition.
Table
DSM-IV-TR diagnostic criteria for delirium
|
| Source: Reference 3 |
Patients who have convoluted histories, such as Mrs. M, are common and difficult to manage and treat. These patients become substantially more complex when they are admitted to inpatient medical or surgical services. The need to clarify between delirium (primarily medical) and depression (primarily psychiatric) becomes paramount when administering treatment and evaluating decision-making capacity.5 In Mrs. M’s case, internal medicine, neurology, and psychiatry teams each had a different approach to altered mental status. Each team’s different terminology, assessment, and objectives further complicated an already challenging case.6
EVALUATION: Confounding results
The ED physicians offer a working diagnosis of acute mental status change, administer IV lorazepam, 4 mg, and order restraints for Mrs. M’s severe agitation. Her initial vital signs reveal slightly elevated blood pressure (140/90 mm Hg) and tachycardia (115 beats per minute). Internal medicine clinicians note that Mrs. M is not in acute distress, although she refuses to speak and has a small amount of dried blood on her lips, presumably from a struggle with the police before coming to the hospital, but this is not certain. Her abdomen is not tender; she has normal bowel sounds, and no asterixis is noted on neurologic exam. Physical exam is otherwise normal. A noncontrast head CT scan shows no acute process. Initial lab values show elevations in ammonia (277 μg/dL) and γ-glutamyl transpeptidase (68 U/L). Thyroid-stimulating hormone is 1.45 mlU/L, prothrombin time is 19.5 s, partial thromboplastin time is 40.3 s, and international normalized ratio is 1.67. The internal medicine team admits Mrs. M to the intensive care unit (ICU) for further management of her mental status change with alcohol withdrawal or hepatic encephalopathy as the most likely etiologies.
Mrs. M’s husband says that his wife has not consumed alcohol in the last 4 months in preparation for a possible liver transplant; however, past interactions with Mrs. M’s family suggest they are unreliable. The Clinical Institute Withdrawal Assessment (CIWA) protocol is implemented in case her symptoms are caused by alcohol withdrawal. Her vital signs are stable and IV lorazepam, 4 mg, is administered once for agitation. Mrs. M’s husband also reports that 1 month ago his wife underwent a transjugular intrahepatic portosystemic shunt (TIPS) procedure for portal hypertension. Outpatient psychotropics (lamotrigine, 100 mg/d, and venlafaxine XR, 75 mg/d) are restarted because withdrawal from these drugs may exacerbate her symptoms. In the ICU Mrs. M experiences a tonic-clonic seizure with fecal incontinence and bitten tongue, which results in a consultation from neurology and the psychiatry consultation-liaison service.
Psychiatry recommends withholding psychotropics, stopping CIWA, and using vital sign parameters along with objective signs of diaphoresis and tremors as indicators of alcohol withdrawal for lorazepam administration. Mrs. M receives IV haloperidol, 1 mg, once during her second day in the hospital for severe agitation, but this medication is discontinued because of concern about lowering her seizure threshold.7 After treatment with lactulose, her ammonia levels trend down to 33 μg/dL, but her altered mental state persists with significant deficits in attention and orientation.
The neurology service performs an EEG that shows no slow-wave, triphasic waves, or epileptiform activity, which likely would be present in delirium or seizures. See Figure 1 for an example of triphasic waves on an EEG and Figure 2 for Mrs. M's EEG results. Subsequent lumbar puncture, MRI, and a second EEG are unremarkable. By the fifth hospital day, Mrs. M is calm and her paranoia has subsided, but she still is confused and disoriented. Psychiatry orders a third EEG while she is in this confused state; it shows no pathologic process. Based on these examinations, neurology posits that Mrs. M is not encephalopathic.
Figure 1: Representative sample of triphasic waves
This EEG tracing is from a 54-year-old woman who underwent prolonged abdominal surgery for lysis of adhesions during which she suffered an intraoperative left subinsular stroke followed by nonconvulsive status epilepticus. The tracing demonstrates typical morphology with the positive sharp transient preceded and followed by smaller amplitude negative deflections. Symmetric, frontal predominance of findings seen is this tracing is common
Figure 2: Mrs. M’s EEG results
This is a representative tracing of Mrs. M’s 3 EEGs revealing an 8.5 to 9 Hz dominant alpha rhythm. There is superimposed frontally dominant beta fast activity, which is consistent with known administration of benzodiazepines
The authors’ observations
Mrs. M had repeated admissions for alcohol dependence and subsequent liver failure. Her recent hospitalization was complicated by a TIPS procedure done 1 month ago. The incidence of hepatic encephalopathy in patients undergoing TIPS is >30%, especially in the first month post-procedure, which raised suspicion that hepatic encephalopathy played a significant role in Mrs. M’s delirium.8
Because of frequent hospitalization, Mrs. M was well known to the internal medicine, neurology, and psychiatry teams, and each used different terms to describe her mental state. Internal medicine used the phrase “acute mental status change,” which covers a broad differential. Neurology used “encephalopathy,” which also is a general term. Psychiatry used “delirium,” which has narrower and more specific diagnostic criteria. Engel et al9 described the delirious patient as having “cerebral insufficiency” with universally abnormal EEG. Regardless of terminology, based on Mrs. M’s acute confusion, one would expect an abnormal EEG, but repeat EEGs were unremarkable.
Interpreting EEG
EEG is one of the few tools available for measuring acute changes in cerebral function, and an EEG slowing remains a hallmark in encephalopathic processes.10,11 Initially, the 3 specialties agreed that Mrs. M’s presentation likely was caused by underlying medical issues or substances (alcohol or others). EEG can help recognize delirium, and, in some cases, elucidate the underlying cause.10,12 It was surprising that Mrs. M’s EEGs were normal despite a clinical presentation of delirium. Because of the normal EEG findings, neurology leaned toward a primary psychiatric (“functional”) etiology as the cause of her delirium vs a general medical condition or alcohol withdrawal (“organic”).
A literature search in regards to sensitivity of EEG in delirium revealed conflicting statements and data. A standard textbook in neurology and psychiatry states that “a normal EEG virtually excludes a toxic-metabolic encephalopathy.”13 The American Psychiatric Association’s (APA) practice guidelines for delirium states: “The presence of EEG abnormalities has fairly good sensitivities for delirium (in one study, the sensitivity was found to be 75%), but the absence does not rule out the diagnosis; thus the EEG is no substitute for careful clinical observation.”6
At the beginning of Mrs. M’s care, in discussion with the neurology and internal medicine teams, we argued that Mrs. M was experiencing delirium despite her initial normal EEG. We did not expect that 2 subsequent EEGs would be normal, especially because the teams witnessed the final EEG being performed while Mrs. M was clinically evaluated and observed to be in a state of delirium.
OUTCOME: Cause still unknown
By the 6th day of hospitalization, Mrs. M’s vitals are normal and she remains hemodynamically stable. Differential diagnosis remains wide and unclear. The psychiatry team feels she could have atypical catatonia due to an underlying mood disorder. One hour after a trial of IV lorazepam, 1 mg, Mrs. M is more lucid and fully oriented, with MMSE of 28/30 (recall was 1/3), indicating normal cognition. During the exam, a psychiatry resident notes Mrs. M winks and feigns a yawn at the medical students and nurses in the room, displaying her boredom with the interview and simplicity of the mental status exam questions. Later that evening, Mrs. M exhibits bizarre sexual gestures toward male hospital staff, including licking a male nursing staff member’s hand.
Although Mrs. M’s initial confusion resolved, the severity of her comorbid psychiatric history warrants inpatient psychiatric hospitalization. She agrees to transfer to the psychiatric ward after she confesses anxiety regarding death, intense demoralization, and guilt related to her condition and her relationship with her 12-year-old daughter. She tearfully reports that she discontinued her psychotropic medications shortly after stopping alcohol 4 months ago. Shortly before her transfer, psychiatry is called back to the medicine floor because of Mrs. M’s disruptive behavior.
The team finds Mrs. M in her hospital gown, pursuing her husband in the hallway as he is leaving, yelling profanities and blaming him for her horrible experience in the hospital. Based on her demeanor, the team determines that she is back to her baseline mental state despite her mood disorder, and that her upcoming inpatient psychiatric stay likely would be too short to address her comorbid personality disorder. The next day she signs out of the hospital against medical advice.
The authors’ observations
We never clearly identified the specific etiology responsible for Mrs. M’s delirium. We assume at the initial presentation she had toxic-metabolic encephalopathy that rapidly resolved with lactulose treatment and lowering her ammonia. She then had a single tonic-clonic seizure, perhaps related to stopping and then restarting her psychotropics. Her subsequent confusion, bizarre sexual behavior, and demeanor on her final hospital days were more indicative of her psychiatric diagnoses. We now suspect that Mrs. M’s delirium was briefer than presumed and she returned to her baseline borderline personality, resulting in some factitious staging of delirium to confuse her 3 treating teams (a psychoanalyst may say this was a form of projective identification).
We felt that if Mrs. M truly was delirious due to metabolic or hepatic dysfunction or alcohol withdrawal, she would have had abnormal EEG findings. We discovered that the notion of “75% sensitivity” of EEG abnormalities cited in the APA guidelines comes from studies that include patients with “psychogenic” and “organic” delirium. Acute manias and agitated psychoses were termed “psychogenic delirium” and acute confusion due to medical conditions or substance issues was termed “organic delirium.”9,12,14-16
This poses a circular reasoning in the diagnostic criteria and clinical approach to delirium. The fallacy is that, according to DSM-IV-TR, delirium is supposed to be the result of a direct physiological consequence of a general medical condition or substance use (criterion D), and cannot be due to psychosis (eg, schizophrenia) or mania (eg, BD). We question the presumptive 75% sensitivity of EEG abnormalities in patients with delirium because it is possible that when some of these studies were conducted the definition of delirium was not solidified or fully understood. We suspect the sensitivity would be much higher if the correct definition of delirium according to DSM-IV-TR is used in future studies. To improve interdisciplinary communication and future research, it would be constructive if all disciplines could agree on a single term, with the same diagnostic criteria, when evaluating a patient with acute confusion.
Related Resources
- Meagher D. Delirium: the role of psychiatry. Advances in Psychiatric Treatment. 2001;7:433-442.
- Casey DA, DeFazio JV Jr, Vansickle K, et al. Delirium. Quick recognition, careful evaluation, and appropriate treatment. Postgrad Med. 1996;100(1):121-4, 128, 133-134.
Drug Brand Names
- Clonazepam • Klonopin
- Docusate • Surfak
- Haloperidol • Haldol
- Lamotrigine • Lamictal
- Lorazepam • Ativan
- Levothyroxine • Levoxyl, Synthtoid
- Venlafaxine XR • Effexor XR
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgment
The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, or the U.S. Government. The authors are employees of the U.S. Government. This work was prepared as part of their official duties. Title 17 U.S.C. 105 provides that “Copyright protection under this title is not available for any work of the U.S. Government.” Title 17 U.S.C. 101 defines a U.S. Government work as a work prepared by a military service member or employee of the U.S. Government as part of that person’s official duties.
CASE: Agitation
Mrs. M, age 39, presents to the emergency department (ED) with altered mental status. She is escorted by her husband and the police. She has a history of severe alcohol dependence, bipolar disorder (BD), anxiety, borderline personality disorder (BPD), hypothyroidism, and bulimia, and had gastric bypass surgery 4 years ago. Her husband called 911 when he could no longer manage Mrs. M’s agitated state. The police found her to be extremely paranoid, restless, and disoriented. Her husband reports that she shouted “the world is going to end” before she escaped naked into her neighborhood streets.
On several occasions Mrs. M had been admitted to the same hospital for alcohol withdrawal and dependence with subsequent liver failure, leading to jaundice, coagulopathy, and ascites. During these hospitalizations, she exhibited poor behavioral tendencies, unhealthy psychological defenses, and chronic maladaptive coping and defense mechanisms congruent with her BPD diagnosis. Specifically, she engaged in splitting of hospital staff, ranging from extreme flattery to overt devaluation and hostility. Other defense mechanisms included denial, distortion, acting out, and passive-aggressive behavior. During these admissions, Mrs. M often displayed deficits in recall and attention on Mini-Mental State Examination (MMSE), but these deficits were associated with concurrent alcohol use and improved rapidly during her stay.
In her current presentation, Mrs. M’s mental status change is more pronounced and atypical compared with earlier admissions. Her outpatient medication regimen includes lamotrigine, 100 mg/d, levothyroxine, 88 mcg/d, venlafaxine extended release (XR), 75 mg/d, clonazepam, 3 mg/d, docusate as needed for constipation, and a daily multivitamin.
The authors’ observations
Delirium is a disturbance of consciousness manifested by a reduced clarity of awareness (impairment in attention) and change in cognition (impairment in orientation, memory, and language).1,2 The disturbance develops over a short time and tends to fluctuate during the day. Delirium is a direct physiological consequence of a general medical condition, substance use (intoxication or withdrawal), or both (Table).3
Delirium generally is a reversible mental disorder but can progress to irreversible brain damage. Prompt and accurate diagnosis of delirium is essential,4 although the condition often is underdiagnosed or misdiagnosed because of lack of recognition.
Table
DSM-IV-TR diagnostic criteria for delirium
|
| Source: Reference 3 |
Patients who have convoluted histories, such as Mrs. M, are common and difficult to manage and treat. These patients become substantially more complex when they are admitted to inpatient medical or surgical services. The need to clarify between delirium (primarily medical) and depression (primarily psychiatric) becomes paramount when administering treatment and evaluating decision-making capacity.5 In Mrs. M’s case, internal medicine, neurology, and psychiatry teams each had a different approach to altered mental status. Each team’s different terminology, assessment, and objectives further complicated an already challenging case.6
EVALUATION: Confounding results
The ED physicians offer a working diagnosis of acute mental status change, administer IV lorazepam, 4 mg, and order restraints for Mrs. M’s severe agitation. Her initial vital signs reveal slightly elevated blood pressure (140/90 mm Hg) and tachycardia (115 beats per minute). Internal medicine clinicians note that Mrs. M is not in acute distress, although she refuses to speak and has a small amount of dried blood on her lips, presumably from a struggle with the police before coming to the hospital, but this is not certain. Her abdomen is not tender; she has normal bowel sounds, and no asterixis is noted on neurologic exam. Physical exam is otherwise normal. A noncontrast head CT scan shows no acute process. Initial lab values show elevations in ammonia (277 μg/dL) and γ-glutamyl transpeptidase (68 U/L). Thyroid-stimulating hormone is 1.45 mlU/L, prothrombin time is 19.5 s, partial thromboplastin time is 40.3 s, and international normalized ratio is 1.67. The internal medicine team admits Mrs. M to the intensive care unit (ICU) for further management of her mental status change with alcohol withdrawal or hepatic encephalopathy as the most likely etiologies.
Mrs. M’s husband says that his wife has not consumed alcohol in the last 4 months in preparation for a possible liver transplant; however, past interactions with Mrs. M’s family suggest they are unreliable. The Clinical Institute Withdrawal Assessment (CIWA) protocol is implemented in case her symptoms are caused by alcohol withdrawal. Her vital signs are stable and IV lorazepam, 4 mg, is administered once for agitation. Mrs. M’s husband also reports that 1 month ago his wife underwent a transjugular intrahepatic portosystemic shunt (TIPS) procedure for portal hypertension. Outpatient psychotropics (lamotrigine, 100 mg/d, and venlafaxine XR, 75 mg/d) are restarted because withdrawal from these drugs may exacerbate her symptoms. In the ICU Mrs. M experiences a tonic-clonic seizure with fecal incontinence and bitten tongue, which results in a consultation from neurology and the psychiatry consultation-liaison service.
Psychiatry recommends withholding psychotropics, stopping CIWA, and using vital sign parameters along with objective signs of diaphoresis and tremors as indicators of alcohol withdrawal for lorazepam administration. Mrs. M receives IV haloperidol, 1 mg, once during her second day in the hospital for severe agitation, but this medication is discontinued because of concern about lowering her seizure threshold.7 After treatment with lactulose, her ammonia levels trend down to 33 μg/dL, but her altered mental state persists with significant deficits in attention and orientation.
The neurology service performs an EEG that shows no slow-wave, triphasic waves, or epileptiform activity, which likely would be present in delirium or seizures. See Figure 1 for an example of triphasic waves on an EEG and Figure 2 for Mrs. M's EEG results. Subsequent lumbar puncture, MRI, and a second EEG are unremarkable. By the fifth hospital day, Mrs. M is calm and her paranoia has subsided, but she still is confused and disoriented. Psychiatry orders a third EEG while she is in this confused state; it shows no pathologic process. Based on these examinations, neurology posits that Mrs. M is not encephalopathic.
Figure 1: Representative sample of triphasic waves
This EEG tracing is from a 54-year-old woman who underwent prolonged abdominal surgery for lysis of adhesions during which she suffered an intraoperative left subinsular stroke followed by nonconvulsive status epilepticus. The tracing demonstrates typical morphology with the positive sharp transient preceded and followed by smaller amplitude negative deflections. Symmetric, frontal predominance of findings seen is this tracing is common
Figure 2: Mrs. M’s EEG results
This is a representative tracing of Mrs. M’s 3 EEGs revealing an 8.5 to 9 Hz dominant alpha rhythm. There is superimposed frontally dominant beta fast activity, which is consistent with known administration of benzodiazepines
The authors’ observations
Mrs. M had repeated admissions for alcohol dependence and subsequent liver failure. Her recent hospitalization was complicated by a TIPS procedure done 1 month ago. The incidence of hepatic encephalopathy in patients undergoing TIPS is >30%, especially in the first month post-procedure, which raised suspicion that hepatic encephalopathy played a significant role in Mrs. M’s delirium.8
Because of frequent hospitalization, Mrs. M was well known to the internal medicine, neurology, and psychiatry teams, and each used different terms to describe her mental state. Internal medicine used the phrase “acute mental status change,” which covers a broad differential. Neurology used “encephalopathy,” which also is a general term. Psychiatry used “delirium,” which has narrower and more specific diagnostic criteria. Engel et al9 described the delirious patient as having “cerebral insufficiency” with universally abnormal EEG. Regardless of terminology, based on Mrs. M’s acute confusion, one would expect an abnormal EEG, but repeat EEGs were unremarkable.
Interpreting EEG
EEG is one of the few tools available for measuring acute changes in cerebral function, and an EEG slowing remains a hallmark in encephalopathic processes.10,11 Initially, the 3 specialties agreed that Mrs. M’s presentation likely was caused by underlying medical issues or substances (alcohol or others). EEG can help recognize delirium, and, in some cases, elucidate the underlying cause.10,12 It was surprising that Mrs. M’s EEGs were normal despite a clinical presentation of delirium. Because of the normal EEG findings, neurology leaned toward a primary psychiatric (“functional”) etiology as the cause of her delirium vs a general medical condition or alcohol withdrawal (“organic”).
A literature search in regards to sensitivity of EEG in delirium revealed conflicting statements and data. A standard textbook in neurology and psychiatry states that “a normal EEG virtually excludes a toxic-metabolic encephalopathy.”13 The American Psychiatric Association’s (APA) practice guidelines for delirium states: “The presence of EEG abnormalities has fairly good sensitivities for delirium (in one study, the sensitivity was found to be 75%), but the absence does not rule out the diagnosis; thus the EEG is no substitute for careful clinical observation.”6
At the beginning of Mrs. M’s care, in discussion with the neurology and internal medicine teams, we argued that Mrs. M was experiencing delirium despite her initial normal EEG. We did not expect that 2 subsequent EEGs would be normal, especially because the teams witnessed the final EEG being performed while Mrs. M was clinically evaluated and observed to be in a state of delirium.
OUTCOME: Cause still unknown
By the 6th day of hospitalization, Mrs. M’s vitals are normal and she remains hemodynamically stable. Differential diagnosis remains wide and unclear. The psychiatry team feels she could have atypical catatonia due to an underlying mood disorder. One hour after a trial of IV lorazepam, 1 mg, Mrs. M is more lucid and fully oriented, with MMSE of 28/30 (recall was 1/3), indicating normal cognition. During the exam, a psychiatry resident notes Mrs. M winks and feigns a yawn at the medical students and nurses in the room, displaying her boredom with the interview and simplicity of the mental status exam questions. Later that evening, Mrs. M exhibits bizarre sexual gestures toward male hospital staff, including licking a male nursing staff member’s hand.
Although Mrs. M’s initial confusion resolved, the severity of her comorbid psychiatric history warrants inpatient psychiatric hospitalization. She agrees to transfer to the psychiatric ward after she confesses anxiety regarding death, intense demoralization, and guilt related to her condition and her relationship with her 12-year-old daughter. She tearfully reports that she discontinued her psychotropic medications shortly after stopping alcohol 4 months ago. Shortly before her transfer, psychiatry is called back to the medicine floor because of Mrs. M’s disruptive behavior.
The team finds Mrs. M in her hospital gown, pursuing her husband in the hallway as he is leaving, yelling profanities and blaming him for her horrible experience in the hospital. Based on her demeanor, the team determines that she is back to her baseline mental state despite her mood disorder, and that her upcoming inpatient psychiatric stay likely would be too short to address her comorbid personality disorder. The next day she signs out of the hospital against medical advice.
The authors’ observations
We never clearly identified the specific etiology responsible for Mrs. M’s delirium. We assume at the initial presentation she had toxic-metabolic encephalopathy that rapidly resolved with lactulose treatment and lowering her ammonia. She then had a single tonic-clonic seizure, perhaps related to stopping and then restarting her psychotropics. Her subsequent confusion, bizarre sexual behavior, and demeanor on her final hospital days were more indicative of her psychiatric diagnoses. We now suspect that Mrs. M’s delirium was briefer than presumed and she returned to her baseline borderline personality, resulting in some factitious staging of delirium to confuse her 3 treating teams (a psychoanalyst may say this was a form of projective identification).
We felt that if Mrs. M truly was delirious due to metabolic or hepatic dysfunction or alcohol withdrawal, she would have had abnormal EEG findings. We discovered that the notion of “75% sensitivity” of EEG abnormalities cited in the APA guidelines comes from studies that include patients with “psychogenic” and “organic” delirium. Acute manias and agitated psychoses were termed “psychogenic delirium” and acute confusion due to medical conditions or substance issues was termed “organic delirium.”9,12,14-16
This poses a circular reasoning in the diagnostic criteria and clinical approach to delirium. The fallacy is that, according to DSM-IV-TR, delirium is supposed to be the result of a direct physiological consequence of a general medical condition or substance use (criterion D), and cannot be due to psychosis (eg, schizophrenia) or mania (eg, BD). We question the presumptive 75% sensitivity of EEG abnormalities in patients with delirium because it is possible that when some of these studies were conducted the definition of delirium was not solidified or fully understood. We suspect the sensitivity would be much higher if the correct definition of delirium according to DSM-IV-TR is used in future studies. To improve interdisciplinary communication and future research, it would be constructive if all disciplines could agree on a single term, with the same diagnostic criteria, when evaluating a patient with acute confusion.
Related Resources
- Meagher D. Delirium: the role of psychiatry. Advances in Psychiatric Treatment. 2001;7:433-442.
- Casey DA, DeFazio JV Jr, Vansickle K, et al. Delirium. Quick recognition, careful evaluation, and appropriate treatment. Postgrad Med. 1996;100(1):121-4, 128, 133-134.
Drug Brand Names
- Clonazepam • Klonopin
- Docusate • Surfak
- Haloperidol • Haldol
- Lamotrigine • Lamictal
- Lorazepam • Ativan
- Levothyroxine • Levoxyl, Synthtoid
- Venlafaxine XR • Effexor XR
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgment
The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, or the U.S. Government. The authors are employees of the U.S. Government. This work was prepared as part of their official duties. Title 17 U.S.C. 105 provides that “Copyright protection under this title is not available for any work of the U.S. Government.” Title 17 U.S.C. 101 defines a U.S. Government work as a work prepared by a military service member or employee of the U.S. Government as part of that person’s official duties.
1. Katz IR, Mossey J, Sussman N, et al. Bedside clinical and electrophysiological assessment: assessment of change in vulnerable patients. Int Psychogeriatr. 1991;3(2):289-300.
2. Inouye SK. Delirium in older persons. N Engl J Med. 2006;354(11):1157-1165.
3. Diagnostic and statistical manual of mental disorders, 4th ed, text rev. Washington DC: American Psychiatric Association; 2000.
4. McPhee SJ, Papadakis M, Rabow MW. CURRENT medical diagnosis and treatment. New York NY: McGraw Hill Medical; 2012.
5. Brody B. Who has capacity? N Engl J Med. 2009;361(3):232-233.
6. Practice guideline for the treatment of patients with delirium. American Psychiatric Association. Am J Psychiatry. 1999;156(5 suppl):1-20.
7. Fricchione GL, Nejad SH, Esses JA, et al. Postoperative delirium. Am J Psychiatry. 2008;165(7):803-812.
8. Sanyal AJ, Freedman AM, Shiffman ML, et al. Portosystemic encephalopathy after transjugular intrahepatic portosystemic shunt: results of a prospective controlled study. Hepatology. 1994;20(1 pt 1):46-55.
9. Engel GL, Romano J. Delirium a syndrome of cerebral insufficiency. 1959. J Neuropsychiatry Clin Neurosci. 2004;16(4):526-538.
10. Pro JD, Wells CE. The use of the electroencephalogram in the diagnosis of delirium. Dis Nerv Syst. 1977;38(10):804-808.
11. Sidhu KS, Balon R, Ajluni V, et al. Standard EEG and the difficult-to-assess mental status. Ann Clin Psychiatry. 2009;21(2):103-108.
12. Brenner RP. Utility of EEG in delirium: past views and current practice. Int Psychogeriatr. 1991;3(2):211-229.
13. Kaufman DM. Clinical neurology for psychiatrists. 5th ed. Philadelphia PA: Saunders; 2001: 230-232.
14. Bond TC. Recognition of acute delirious mania. Arch Gen Psychiatry. 1980;37(5):553-554.
15. Krauthammer C, Klerman GL. Secondary mania: manic syndromes associated with antecedent physical illness or drugs. Arch Gen Psychiatry. 1978;35(11):1333-1339.
16. Larson EW, Richelson E. Organic causes of mania. Mayo Clin Proc. 1988;63(9):906-912.
1. Katz IR, Mossey J, Sussman N, et al. Bedside clinical and electrophysiological assessment: assessment of change in vulnerable patients. Int Psychogeriatr. 1991;3(2):289-300.
2. Inouye SK. Delirium in older persons. N Engl J Med. 2006;354(11):1157-1165.
3. Diagnostic and statistical manual of mental disorders, 4th ed, text rev. Washington DC: American Psychiatric Association; 2000.
4. McPhee SJ, Papadakis M, Rabow MW. CURRENT medical diagnosis and treatment. New York NY: McGraw Hill Medical; 2012.
5. Brody B. Who has capacity? N Engl J Med. 2009;361(3):232-233.
6. Practice guideline for the treatment of patients with delirium. American Psychiatric Association. Am J Psychiatry. 1999;156(5 suppl):1-20.
7. Fricchione GL, Nejad SH, Esses JA, et al. Postoperative delirium. Am J Psychiatry. 2008;165(7):803-812.
8. Sanyal AJ, Freedman AM, Shiffman ML, et al. Portosystemic encephalopathy after transjugular intrahepatic portosystemic shunt: results of a prospective controlled study. Hepatology. 1994;20(1 pt 1):46-55.
9. Engel GL, Romano J. Delirium a syndrome of cerebral insufficiency. 1959. J Neuropsychiatry Clin Neurosci. 2004;16(4):526-538.
10. Pro JD, Wells CE. The use of the electroencephalogram in the diagnosis of delirium. Dis Nerv Syst. 1977;38(10):804-808.
11. Sidhu KS, Balon R, Ajluni V, et al. Standard EEG and the difficult-to-assess mental status. Ann Clin Psychiatry. 2009;21(2):103-108.
12. Brenner RP. Utility of EEG in delirium: past views and current practice. Int Psychogeriatr. 1991;3(2):211-229.
13. Kaufman DM. Clinical neurology for psychiatrists. 5th ed. Philadelphia PA: Saunders; 2001: 230-232.
14. Bond TC. Recognition of acute delirious mania. Arch Gen Psychiatry. 1980;37(5):553-554.
15. Krauthammer C, Klerman GL. Secondary mania: manic syndromes associated with antecedent physical illness or drugs. Arch Gen Psychiatry. 1978;35(11):1333-1339.
16. Larson EW, Richelson E. Organic causes of mania. Mayo Clin Proc. 1988;63(9):906-912.