User login
COPD Updates From CHEST 2024
Efficacy data on ensifentrine, recently FDA-approved for moderate to severe COPD, as well as dupilumab use for symptom control, are key updates in COPD from the CHEST Annual Meeting. Dharani Narendra, MD, FCCP, CHEST Physician Editorial Board Member, from Baylor College of Medicine in Houston, Texas, reports on the findings.
Dr. Narendra begins with a trial examining the role of muscle relaxants in COPD exacerbation. The results showed that patients with COPD who were taking muscle relaxants experienced a rising risk for exacerbations over 1, 3, and 5 years, with a 20% relative risk increase by year 5.
Next, Dr. Narendra reports on ensifentrine efficacy in a subgroup analysis of the ENHANCE phase 3 trial, which provided the basis for FDA approval. Ensifentrine significantly improved peak and average FEV1 at week 12, compared with placebo.
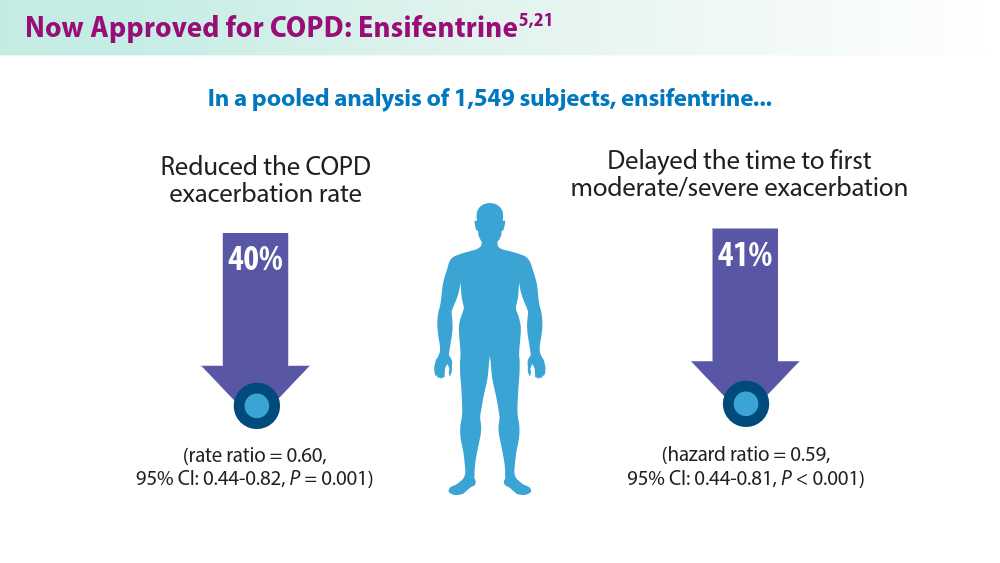
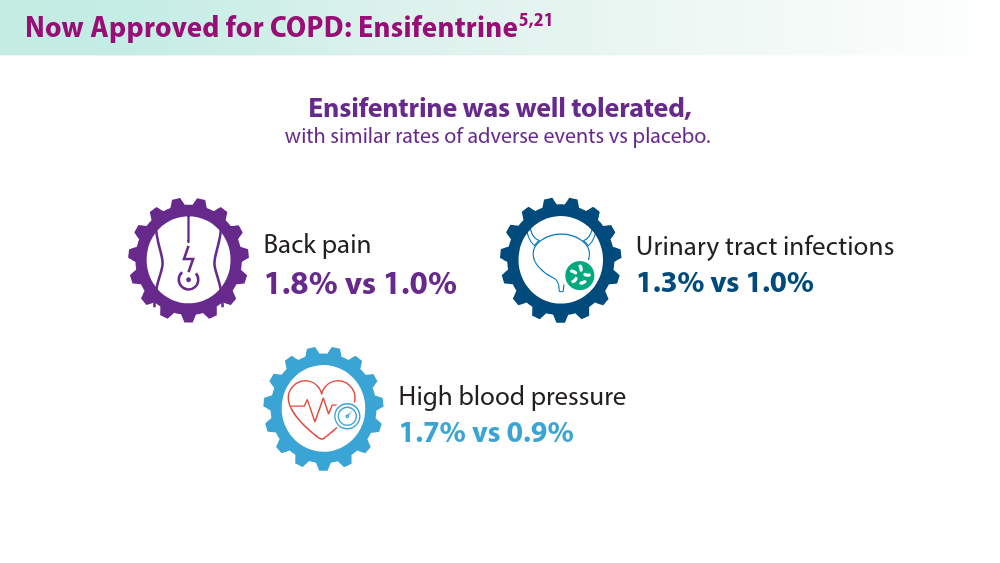
Dr. Narendra discusses a second presentation on ensifentrine — a meta-analysis of six randomized controlled trials of ensifentrine vs placebo in 2,365 patients. Significant improvements were seen in peak FEV1 and average FEV1, confirming ensifentrine efficacy in symptomatic patients with COPD with moderate to severe obstruction.
She also reviews subgroup analyses from the BOREAS and NOTUS trials, which led to the approval of dupilumab. One analysis demonstrated that dupilumab improved respiratory symptoms in patients with COPD with type 2 inflammation. Another showed significant reduction in annual exacerbation rates in patients with and without emphysema.
--
Dharani K. Narendra, MD, FCCP, Assistant Professor, Department of Pulmonary, Critical Care, and Sleep Medicine, Baylor College of Medicine, Houston, Texas
Dharani K. Narendra, MD, FCCP, has disclosed no relevant financial relationships.
Efficacy data on ensifentrine, recently FDA-approved for moderate to severe COPD, as well as dupilumab use for symptom control, are key updates in COPD from the CHEST Annual Meeting. Dharani Narendra, MD, FCCP, CHEST Physician Editorial Board Member, from Baylor College of Medicine in Houston, Texas, reports on the findings.
Dr. Narendra begins with a trial examining the role of muscle relaxants in COPD exacerbation. The results showed that patients with COPD who were taking muscle relaxants experienced a rising risk for exacerbations over 1, 3, and 5 years, with a 20% relative risk increase by year 5.
Next, Dr. Narendra reports on ensifentrine efficacy in a subgroup analysis of the ENHANCE phase 3 trial, which provided the basis for FDA approval. Ensifentrine significantly improved peak and average FEV1 at week 12, compared with placebo.
Dr. Narendra discusses a second presentation on ensifentrine — a meta-analysis of six randomized controlled trials of ensifentrine vs placebo in 2,365 patients. Significant improvements were seen in peak FEV1 and average FEV1, confirming ensifentrine efficacy in symptomatic patients with COPD with moderate to severe obstruction.
She also reviews subgroup analyses from the BOREAS and NOTUS trials, which led to the approval of dupilumab. One analysis demonstrated that dupilumab improved respiratory symptoms in patients with COPD with type 2 inflammation. Another showed significant reduction in annual exacerbation rates in patients with and without emphysema.
--
Dharani K. Narendra, MD, FCCP, Assistant Professor, Department of Pulmonary, Critical Care, and Sleep Medicine, Baylor College of Medicine, Houston, Texas
Dharani K. Narendra, MD, FCCP, has disclosed no relevant financial relationships.
Efficacy data on ensifentrine, recently FDA-approved for moderate to severe COPD, as well as dupilumab use for symptom control, are key updates in COPD from the CHEST Annual Meeting. Dharani Narendra, MD, FCCP, CHEST Physician Editorial Board Member, from Baylor College of Medicine in Houston, Texas, reports on the findings.
Dr. Narendra begins with a trial examining the role of muscle relaxants in COPD exacerbation. The results showed that patients with COPD who were taking muscle relaxants experienced a rising risk for exacerbations over 1, 3, and 5 years, with a 20% relative risk increase by year 5.
Next, Dr. Narendra reports on ensifentrine efficacy in a subgroup analysis of the ENHANCE phase 3 trial, which provided the basis for FDA approval. Ensifentrine significantly improved peak and average FEV1 at week 12, compared with placebo.
Dr. Narendra discusses a second presentation on ensifentrine — a meta-analysis of six randomized controlled trials of ensifentrine vs placebo in 2,365 patients. Significant improvements were seen in peak FEV1 and average FEV1, confirming ensifentrine efficacy in symptomatic patients with COPD with moderate to severe obstruction.
She also reviews subgroup analyses from the BOREAS and NOTUS trials, which led to the approval of dupilumab. One analysis demonstrated that dupilumab improved respiratory symptoms in patients with COPD with type 2 inflammation. Another showed significant reduction in annual exacerbation rates in patients with and without emphysema.
--
Dharani K. Narendra, MD, FCCP, Assistant Professor, Department of Pulmonary, Critical Care, and Sleep Medicine, Baylor College of Medicine, Houston, Texas
Dharani K. Narendra, MD, FCCP, has disclosed no relevant financial relationships.

Updates in COPD Guidelines and Treatment
Al Wachami N, Guennouni M, Iderdar Y, et al. Estimating the global prevalence of chronic obstructive pulmonary disease (COPD): a systematic review and meta-analysis. BMC Public Health. 2024;24(1):297. doi:10.1186/s12889-024-17686-9
COPD trends brief. American Lung Association. Accessed July 11, 2024. https://www.lung.org/research/trends-in-lung-disease/copd-trends-brief
Chronic obstructive pulmonary disease (COPD). World Health Organization. March 16, 2023. Accessed July 11, 2024. https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd)
Shalabi MS, Aqdi SW, Alfort OA, et al. Effectiveness and safety of bronchodilators and inhaled corticosteroids in the management of chronic obstructive pulmonary disease. Int J Commun Med Public Health. 2023;10(8):2955-2959. doi:10.18203/2394-6040.ijcmph20232392
McCormick B. FDA approves ensifentrine for maintenance treatment of adult patients with COPD. AJMC. June 26, 2024. Accessed July 11, 2024. https://www.ajmc.com/view/fda-approves-ensifentrine-for-maintenance-treatment-of-adult-patients-with-copd
Kersul AL, Cosio BG. Biologics in COPD. Open Resp Arch. 2024;6(2):100306. doi:10.1016/j.opresp.2024.100306
2023 GOLD Report. Global Initiative for Chronic Obstructive Lung Disease. Accessed July 11, 2024. https://goldcopd.org/2023-gold-report-2
2024 GOLD Report. Global Initiative for Chronic Obstructive Lung Disease. Accessed July 11, 2024. https://goldcopd.org/2024-gold-report/
Regeneron Pharmaceuticals Inc. Dupixent® (dupilumab) late-breaking data from NOTUS confirmatory phase 3 COPD trial presented at ATS and published in the New England Journal of Medicine [press release]. May 20, 2024. Accessed July 11, 2024. https://investor.regeneron.com/news-releases/news-release-details/dupixentr-dupilumab-late-breaking-data-notus-confirmatory-phase
Pavord ID, Chapman KR, Bafadhel M, et al. Mepolizumab for eosinophil-associated COPD: analysis of METREX and METREO. Int J Chron Obstruct Pulmon Dis. 2021;16:1755-1770. doi:10.2147/COPD.S294333
Mepolizumab as add-on treatment in participants with COPD characterized by frequent exacerbations and eosinophil level (MATINEE). Clinicaltrials.gov. Updated August 28, 2023. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT04133909
Singh D, Criner GJ, Agustí A, et al. Benralizumab prevents recurrent exacerbations in patients with chronic obstructive pulmonary disease: a post hoc analysis. Int J Chron Obstruct Pulmon Dis. 2023;18:1595-1599. doi:10.2147/COPD.S418944
Efficacy and safety of benralizumab in moderate to very severe chronic obstructive pulmonary disease (COPD) with a history of frequent exacerbations (RESOLUTE). Clinicaltrials.gov. Updated May 8, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT04053634
Efficacy and safety of tozorakimab in symptomatic chronic obstructive pulmonary disease with a history of exacerbations (TITANIA). Clinicaltrials.gov. Updated June 27, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT05158387
Efficacy and safety of tozorakimab in symptomatic chronic obstructive pulmonary disease with a history of exacerbations (OBERON). Clinicaltrials.gov. Updated June 21, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT05166889
Long-term efficacy and safety of tozorakimab in participants with chronic obstructive pulmonary disease with a history of exacerbations (PROSPERO). Clinicaltrials.gov. Updated June 20, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT05742802
Efficacy and safety of tozorakimab in symptomatic chronic obstructive pulmonary disease with a history of exacerbations (MIRANDA). Clinicaltrials.gov. Updated June 4, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT06040086
Study to assess the efficacy, safety, and tolerability of SAR440340/REGN3500/itepekimab in chronic obstructive pulmonary disease (COPD) (AERIFY-1). ClinicalTrials.gov. Updated June 21, 2024. Accessed July 11, 2024. https://classic.clinicaltrials.gov/ct2/show/NCT04701983
Study to assess the efficacy, safety, and tolerability of SAR440340/REGN3500/itepekimab in chronic obstructive pulmonary disease (COPD) (AERIFY-2). ClinicalTrials.gov. Updated May 9, 2024. Accessed July 11, 2024. https://classic.clinicaltrials.gov/ct2/show/NCT04751487
ALIENTO and ARNASA: study designs of two randomised, double-blind, placebo-controlled trials of astegolimab in patients with COPD. Medically. 2023. Accessed July 11, 2024. https://medically.gene.com/global/en/unrestricted/respiratory/ERS-2023/ers-2023-poster-brightling-aliento-and-arnasa-study-des.html
Anzueto A, Barjaktarevic IZ, Siler TM, et al. Ensifentrine, a novel phosphodiesterase 3 and 4 inhibitor for the treatment of chronic obstructive pulmonary disease: randomized, double-blind, placebo-controlled, multicenter phase III trials (the ENHANCE trials). Am J Respir Crit Care Med. 2023;208(4):406-416. doi:10.1164/rccm.202306-0944OC
US Preventive Services Taskforce. Lung cancer: screening. March 9, 2021. Accessed July 11, 2024. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening
Al Wachami N, Guennouni M, Iderdar Y, et al. Estimating the global prevalence of chronic obstructive pulmonary disease (COPD): a systematic review and meta-analysis. BMC Public Health. 2024;24(1):297. doi:10.1186/s12889-024-17686-9
COPD trends brief. American Lung Association. Accessed July 11, 2024. https://www.lung.org/research/trends-in-lung-disease/copd-trends-brief
Chronic obstructive pulmonary disease (COPD). World Health Organization. March 16, 2023. Accessed July 11, 2024. https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd)
Shalabi MS, Aqdi SW, Alfort OA, et al. Effectiveness and safety of bronchodilators and inhaled corticosteroids in the management of chronic obstructive pulmonary disease. Int J Commun Med Public Health. 2023;10(8):2955-2959. doi:10.18203/2394-6040.ijcmph20232392
McCormick B. FDA approves ensifentrine for maintenance treatment of adult patients with COPD. AJMC. June 26, 2024. Accessed July 11, 2024. https://www.ajmc.com/view/fda-approves-ensifentrine-for-maintenance-treatment-of-adult-patients-with-copd
Kersul AL, Cosio BG. Biologics in COPD. Open Resp Arch. 2024;6(2):100306. doi:10.1016/j.opresp.2024.100306
2023 GOLD Report. Global Initiative for Chronic Obstructive Lung Disease. Accessed July 11, 2024. https://goldcopd.org/2023-gold-report-2
2024 GOLD Report. Global Initiative for Chronic Obstructive Lung Disease. Accessed July 11, 2024. https://goldcopd.org/2024-gold-report/
Regeneron Pharmaceuticals Inc. Dupixent® (dupilumab) late-breaking data from NOTUS confirmatory phase 3 COPD trial presented at ATS and published in the New England Journal of Medicine [press release]. May 20, 2024. Accessed July 11, 2024. https://investor.regeneron.com/news-releases/news-release-details/dupixentr-dupilumab-late-breaking-data-notus-confirmatory-phase
Pavord ID, Chapman KR, Bafadhel M, et al. Mepolizumab for eosinophil-associated COPD: analysis of METREX and METREO. Int J Chron Obstruct Pulmon Dis. 2021;16:1755-1770. doi:10.2147/COPD.S294333
Mepolizumab as add-on treatment in participants with COPD characterized by frequent exacerbations and eosinophil level (MATINEE). Clinicaltrials.gov. Updated August 28, 2023. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT04133909
Singh D, Criner GJ, Agustí A, et al. Benralizumab prevents recurrent exacerbations in patients with chronic obstructive pulmonary disease: a post hoc analysis. Int J Chron Obstruct Pulmon Dis. 2023;18:1595-1599. doi:10.2147/COPD.S418944
Efficacy and safety of benralizumab in moderate to very severe chronic obstructive pulmonary disease (COPD) with a history of frequent exacerbations (RESOLUTE). Clinicaltrials.gov. Updated May 8, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT04053634
Efficacy and safety of tozorakimab in symptomatic chronic obstructive pulmonary disease with a history of exacerbations (TITANIA). Clinicaltrials.gov. Updated June 27, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT05158387
Efficacy and safety of tozorakimab in symptomatic chronic obstructive pulmonary disease with a history of exacerbations (OBERON). Clinicaltrials.gov. Updated June 21, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT05166889
Long-term efficacy and safety of tozorakimab in participants with chronic obstructive pulmonary disease with a history of exacerbations (PROSPERO). Clinicaltrials.gov. Updated June 20, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT05742802
Efficacy and safety of tozorakimab in symptomatic chronic obstructive pulmonary disease with a history of exacerbations (MIRANDA). Clinicaltrials.gov. Updated June 4, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT06040086
Study to assess the efficacy, safety, and tolerability of SAR440340/REGN3500/itepekimab in chronic obstructive pulmonary disease (COPD) (AERIFY-1). ClinicalTrials.gov. Updated June 21, 2024. Accessed July 11, 2024. https://classic.clinicaltrials.gov/ct2/show/NCT04701983
Study to assess the efficacy, safety, and tolerability of SAR440340/REGN3500/itepekimab in chronic obstructive pulmonary disease (COPD) (AERIFY-2). ClinicalTrials.gov. Updated May 9, 2024. Accessed July 11, 2024. https://classic.clinicaltrials.gov/ct2/show/NCT04751487
ALIENTO and ARNASA: study designs of two randomised, double-blind, placebo-controlled trials of astegolimab in patients with COPD. Medically. 2023. Accessed July 11, 2024. https://medically.gene.com/global/en/unrestricted/respiratory/ERS-2023/ers-2023-poster-brightling-aliento-and-arnasa-study-des.html
Anzueto A, Barjaktarevic IZ, Siler TM, et al. Ensifentrine, a novel phosphodiesterase 3 and 4 inhibitor for the treatment of chronic obstructive pulmonary disease: randomized, double-blind, placebo-controlled, multicenter phase III trials (the ENHANCE trials). Am J Respir Crit Care Med. 2023;208(4):406-416. doi:10.1164/rccm.202306-0944OC
US Preventive Services Taskforce. Lung cancer: screening. March 9, 2021. Accessed July 11, 2024. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening
Al Wachami N, Guennouni M, Iderdar Y, et al. Estimating the global prevalence of chronic obstructive pulmonary disease (COPD): a systematic review and meta-analysis. BMC Public Health. 2024;24(1):297. doi:10.1186/s12889-024-17686-9
COPD trends brief. American Lung Association. Accessed July 11, 2024. https://www.lung.org/research/trends-in-lung-disease/copd-trends-brief
Chronic obstructive pulmonary disease (COPD). World Health Organization. March 16, 2023. Accessed July 11, 2024. https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd)
Shalabi MS, Aqdi SW, Alfort OA, et al. Effectiveness and safety of bronchodilators and inhaled corticosteroids in the management of chronic obstructive pulmonary disease. Int J Commun Med Public Health. 2023;10(8):2955-2959. doi:10.18203/2394-6040.ijcmph20232392
McCormick B. FDA approves ensifentrine for maintenance treatment of adult patients with COPD. AJMC. June 26, 2024. Accessed July 11, 2024. https://www.ajmc.com/view/fda-approves-ensifentrine-for-maintenance-treatment-of-adult-patients-with-copd
Kersul AL, Cosio BG. Biologics in COPD. Open Resp Arch. 2024;6(2):100306. doi:10.1016/j.opresp.2024.100306
2023 GOLD Report. Global Initiative for Chronic Obstructive Lung Disease. Accessed July 11, 2024. https://goldcopd.org/2023-gold-report-2
2024 GOLD Report. Global Initiative for Chronic Obstructive Lung Disease. Accessed July 11, 2024. https://goldcopd.org/2024-gold-report/
Regeneron Pharmaceuticals Inc. Dupixent® (dupilumab) late-breaking data from NOTUS confirmatory phase 3 COPD trial presented at ATS and published in the New England Journal of Medicine [press release]. May 20, 2024. Accessed July 11, 2024. https://investor.regeneron.com/news-releases/news-release-details/dupixentr-dupilumab-late-breaking-data-notus-confirmatory-phase
Pavord ID, Chapman KR, Bafadhel M, et al. Mepolizumab for eosinophil-associated COPD: analysis of METREX and METREO. Int J Chron Obstruct Pulmon Dis. 2021;16:1755-1770. doi:10.2147/COPD.S294333
Mepolizumab as add-on treatment in participants with COPD characterized by frequent exacerbations and eosinophil level (MATINEE). Clinicaltrials.gov. Updated August 28, 2023. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT04133909
Singh D, Criner GJ, Agustí A, et al. Benralizumab prevents recurrent exacerbations in patients with chronic obstructive pulmonary disease: a post hoc analysis. Int J Chron Obstruct Pulmon Dis. 2023;18:1595-1599. doi:10.2147/COPD.S418944
Efficacy and safety of benralizumab in moderate to very severe chronic obstructive pulmonary disease (COPD) with a history of frequent exacerbations (RESOLUTE). Clinicaltrials.gov. Updated May 8, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT04053634
Efficacy and safety of tozorakimab in symptomatic chronic obstructive pulmonary disease with a history of exacerbations (TITANIA). Clinicaltrials.gov. Updated June 27, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT05158387
Efficacy and safety of tozorakimab in symptomatic chronic obstructive pulmonary disease with a history of exacerbations (OBERON). Clinicaltrials.gov. Updated June 21, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT05166889
Long-term efficacy and safety of tozorakimab in participants with chronic obstructive pulmonary disease with a history of exacerbations (PROSPERO). Clinicaltrials.gov. Updated June 20, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT05742802
Efficacy and safety of tozorakimab in symptomatic chronic obstructive pulmonary disease with a history of exacerbations (MIRANDA). Clinicaltrials.gov. Updated June 4, 2024. Accessed July 11, 2024. https://clinicaltrials.gov/study/NCT06040086
Study to assess the efficacy, safety, and tolerability of SAR440340/REGN3500/itepekimab in chronic obstructive pulmonary disease (COPD) (AERIFY-1). ClinicalTrials.gov. Updated June 21, 2024. Accessed July 11, 2024. https://classic.clinicaltrials.gov/ct2/show/NCT04701983
Study to assess the efficacy, safety, and tolerability of SAR440340/REGN3500/itepekimab in chronic obstructive pulmonary disease (COPD) (AERIFY-2). ClinicalTrials.gov. Updated May 9, 2024. Accessed July 11, 2024. https://classic.clinicaltrials.gov/ct2/show/NCT04751487
ALIENTO and ARNASA: study designs of two randomised, double-blind, placebo-controlled trials of astegolimab in patients with COPD. Medically. 2023. Accessed July 11, 2024. https://medically.gene.com/global/en/unrestricted/respiratory/ERS-2023/ers-2023-poster-brightling-aliento-and-arnasa-study-des.html
Anzueto A, Barjaktarevic IZ, Siler TM, et al. Ensifentrine, a novel phosphodiesterase 3 and 4 inhibitor for the treatment of chronic obstructive pulmonary disease: randomized, double-blind, placebo-controlled, multicenter phase III trials (the ENHANCE trials). Am J Respir Crit Care Med. 2023;208(4):406-416. doi:10.1164/rccm.202306-0944OC
US Preventive Services Taskforce. Lung cancer: screening. March 9, 2021. Accessed July 11, 2024. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening