User login
LAS VEGAS – A novel device-based approach to the treatment of heart failure via vagal nerve stimulation resulted in improvements in objectively measurable cardiac function as well as in subjective heart failure symptoms in the ANTHEM-HF trial.
The improvements seen in ANTHEM-HF (Autonomic Neural Regulation Therapy to Enhance Myocardial Function in Heart Failure) were similar regardless of whether participants were randomized to stimulation of the left or right vagal nerve. That’s an important finding because left-sided vagal nerve stimulation (VNS) technology could readily be combined with implantable cardioverter-defibrillators and cardiac resynchronization therapy devices, which are routinely placed on the left side of the thorax, Dr. Inder S. Anand observed in presenting the study findings at the annual meeting of the Heart Failure Society of America.
“We believe that if this technology pans out and proves effective, it will be introduced into devices very easily. That’s the advantage of left-sided stimulation,” explained Dr. Anand, professor of medicine at the University of Minnesota, Minneapolis.
He was quick to add, however, that “There needs to be a lot more work before autonomic regulation therapy is ready for prime time.” That’s because ANTHEM-HF was a relatively small prospective study – just 60 patients – and it was uncontrolled and unblinded.
Left-sided VNS is a well-established treatment for epilepsy, with more than 100,000 patients having received devices in the last several decades. It’s also seeing increasing use in refractory depression.
The approach is still investigational for heart failure, where autonomic imbalance with increased sympathetic activity and decreased parasympathetic tone is associated with heart failure progression and worse clinical outcomes, the cardiologist explained.
In ANTHEM-HF, 60 patients on optimal medical therapy for heart failure with reduced ejection fraction were randomized to left- or right-sided implantation of the Cyberonics VNS device. Once activated, the VNS intensity was titrated over 10 weeks to the maximum tolerable current that remained below the threshold of heart rate change, side effects, and patient sensation. The chronic intermittent stimulation to the vagus nerve was delivered at 10 Hz, a 250 microsec pulse width, and an average stimulation current of 2.0 mA. The stimulation cycle was 14 seconds on, 66 seconds off.
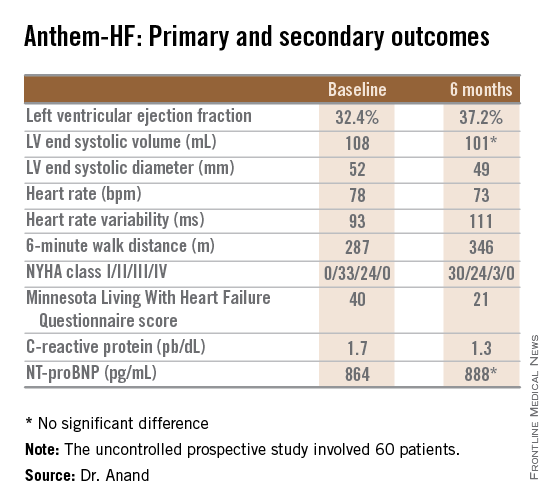
The primary study endpoint was change in LVEF over the course of 6 months. From a baseline of 32.4% it improved significantly to 37.2%, which Dr. Anand deemed “very impressive” for just 6 months of therapy. The improvement was similar regardless of whether the VNS was left or right sided.
He said the study didn’t raise any safety concerns. The only serious treatment-related adverse event was a fatal perioperative embolic stroke in a recipient of a left-sided device.
The most common adverse events were hoarseness or other voice alterations, which occurred in 19 patients. Thirteen patients reported a new cough. These side effects mirror the experience in patients treated with the VNS device for epilepsy or depression.
Discussant Jagmeet P. Singh emphasized that “it’s important to interpret the ANTHEM-HF results with some degree of caution.” That’s because the usual variability within LVEF measurements can be in the 5% range, and the 4.8% improvement seen in this study wasn’t accompanied by a significant improvement in LV end systolic volume. Moreover, the improvements seen in secondary endpoints – 6-minute walk distance, NYHA class, and the Minnesota Living With Heart Failure qualify-of-life score – are all potentially open to bias since neither patients nor physicians were blinded.
Dr. Singh noted that the NECTAR-HF study, a blinded, sham-controlled clinical trial of VNS presented earlier at the annual congress of the European Society of Cardiology, was a negative study that showed no improvement in functional measures. While NECTAR-HF caused some skeptics to question the whole concept of VNS as a useful therapeutic strategy, the stimulation protocols used in NECTAR-HF and ANTHEM-HF were quite different, and it’s likely that the stimulation amplitude employed in NECTAR-HF wasn’t sufficient to affect the target subset of vagal nerve fibers, according to Dr. Singh, director of the cardiac resynchronization therapy program and the Holter laboratory at Massachusetts General Hospital, Boston.
He considers the ANTHEM-HF finding that left-sided VNS is comparable to right to be an important advance that “really moves the field forward.” Yet many critical questions about autonomic regulation therapy for heart failure remain unanswered. These include whether it truly is safe and effective, and if so, the optimal target dose and frequency of stimulation. Answers should be forthcoming from the ongoing INOVATE-HF study, a randomized, controlled, 650-patient study, sponsored by BioControl Medical. The trial features hard clinical endpoints and higher target-dose stimulation protocols than in prior studies.
“This trial will probably put an end to the debate as to whether vagal nerve stimulation has an impact on heart failure or not,” the cardiologist predicted.
“Strategies to nonpharmacologically modulate the autonomic nervous system is an area of immense interest and investigation, and it’s going to be extended,” he observed. “I think that this is the autonomic era, and so it’s really important that we get it right. I think we’ve learned from the renal denervation experience that we need to be really careful in selecting patients for any form of autonomic manipulation. I don’t think we should select patients just off their LVEF. We should have some parameter that can quantify autonomic dysregulation prior to initiation of therapy.”
It will also be important to be able to individualize VNS therapy, just as physicians now do with beta blockers and other pharmacotherapies, he added.
The ANTHEM-HF trial was sponsored by Cyberonics. Dr. Anand reported serving as a consultant to and/or recipient of research grants from Cyberonics, Amgen, Critical Diagnostics, Novartis, and Zensun. Dr. Singh has received research support and/or served on speakers bureaus for Boston Scientific, St. Jude Medical, and the Sorin Group.
LAS VEGAS – A novel device-based approach to the treatment of heart failure via vagal nerve stimulation resulted in improvements in objectively measurable cardiac function as well as in subjective heart failure symptoms in the ANTHEM-HF trial.
The improvements seen in ANTHEM-HF (Autonomic Neural Regulation Therapy to Enhance Myocardial Function in Heart Failure) were similar regardless of whether participants were randomized to stimulation of the left or right vagal nerve. That’s an important finding because left-sided vagal nerve stimulation (VNS) technology could readily be combined with implantable cardioverter-defibrillators and cardiac resynchronization therapy devices, which are routinely placed on the left side of the thorax, Dr. Inder S. Anand observed in presenting the study findings at the annual meeting of the Heart Failure Society of America.
“We believe that if this technology pans out and proves effective, it will be introduced into devices very easily. That’s the advantage of left-sided stimulation,” explained Dr. Anand, professor of medicine at the University of Minnesota, Minneapolis.
He was quick to add, however, that “There needs to be a lot more work before autonomic regulation therapy is ready for prime time.” That’s because ANTHEM-HF was a relatively small prospective study – just 60 patients – and it was uncontrolled and unblinded.
Left-sided VNS is a well-established treatment for epilepsy, with more than 100,000 patients having received devices in the last several decades. It’s also seeing increasing use in refractory depression.
The approach is still investigational for heart failure, where autonomic imbalance with increased sympathetic activity and decreased parasympathetic tone is associated with heart failure progression and worse clinical outcomes, the cardiologist explained.
In ANTHEM-HF, 60 patients on optimal medical therapy for heart failure with reduced ejection fraction were randomized to left- or right-sided implantation of the Cyberonics VNS device. Once activated, the VNS intensity was titrated over 10 weeks to the maximum tolerable current that remained below the threshold of heart rate change, side effects, and patient sensation. The chronic intermittent stimulation to the vagus nerve was delivered at 10 Hz, a 250 microsec pulse width, and an average stimulation current of 2.0 mA. The stimulation cycle was 14 seconds on, 66 seconds off.
The primary study endpoint was change in LVEF over the course of 6 months. From a baseline of 32.4% it improved significantly to 37.2%, which Dr. Anand deemed “very impressive” for just 6 months of therapy. The improvement was similar regardless of whether the VNS was left or right sided.
He said the study didn’t raise any safety concerns. The only serious treatment-related adverse event was a fatal perioperative embolic stroke in a recipient of a left-sided device.
The most common adverse events were hoarseness or other voice alterations, which occurred in 19 patients. Thirteen patients reported a new cough. These side effects mirror the experience in patients treated with the VNS device for epilepsy or depression.
Discussant Jagmeet P. Singh emphasized that “it’s important to interpret the ANTHEM-HF results with some degree of caution.” That’s because the usual variability within LVEF measurements can be in the 5% range, and the 4.8% improvement seen in this study wasn’t accompanied by a significant improvement in LV end systolic volume. Moreover, the improvements seen in secondary endpoints – 6-minute walk distance, NYHA class, and the Minnesota Living With Heart Failure qualify-of-life score – are all potentially open to bias since neither patients nor physicians were blinded.
Dr. Singh noted that the NECTAR-HF study, a blinded, sham-controlled clinical trial of VNS presented earlier at the annual congress of the European Society of Cardiology, was a negative study that showed no improvement in functional measures. While NECTAR-HF caused some skeptics to question the whole concept of VNS as a useful therapeutic strategy, the stimulation protocols used in NECTAR-HF and ANTHEM-HF were quite different, and it’s likely that the stimulation amplitude employed in NECTAR-HF wasn’t sufficient to affect the target subset of vagal nerve fibers, according to Dr. Singh, director of the cardiac resynchronization therapy program and the Holter laboratory at Massachusetts General Hospital, Boston.
He considers the ANTHEM-HF finding that left-sided VNS is comparable to right to be an important advance that “really moves the field forward.” Yet many critical questions about autonomic regulation therapy for heart failure remain unanswered. These include whether it truly is safe and effective, and if so, the optimal target dose and frequency of stimulation. Answers should be forthcoming from the ongoing INOVATE-HF study, a randomized, controlled, 650-patient study, sponsored by BioControl Medical. The trial features hard clinical endpoints and higher target-dose stimulation protocols than in prior studies.
“This trial will probably put an end to the debate as to whether vagal nerve stimulation has an impact on heart failure or not,” the cardiologist predicted.
“Strategies to nonpharmacologically modulate the autonomic nervous system is an area of immense interest and investigation, and it’s going to be extended,” he observed. “I think that this is the autonomic era, and so it’s really important that we get it right. I think we’ve learned from the renal denervation experience that we need to be really careful in selecting patients for any form of autonomic manipulation. I don’t think we should select patients just off their LVEF. We should have some parameter that can quantify autonomic dysregulation prior to initiation of therapy.”
It will also be important to be able to individualize VNS therapy, just as physicians now do with beta blockers and other pharmacotherapies, he added.
The ANTHEM-HF trial was sponsored by Cyberonics. Dr. Anand reported serving as a consultant to and/or recipient of research grants from Cyberonics, Amgen, Critical Diagnostics, Novartis, and Zensun. Dr. Singh has received research support and/or served on speakers bureaus for Boston Scientific, St. Jude Medical, and the Sorin Group.
LAS VEGAS – A novel device-based approach to the treatment of heart failure via vagal nerve stimulation resulted in improvements in objectively measurable cardiac function as well as in subjective heart failure symptoms in the ANTHEM-HF trial.
The improvements seen in ANTHEM-HF (Autonomic Neural Regulation Therapy to Enhance Myocardial Function in Heart Failure) were similar regardless of whether participants were randomized to stimulation of the left or right vagal nerve. That’s an important finding because left-sided vagal nerve stimulation (VNS) technology could readily be combined with implantable cardioverter-defibrillators and cardiac resynchronization therapy devices, which are routinely placed on the left side of the thorax, Dr. Inder S. Anand observed in presenting the study findings at the annual meeting of the Heart Failure Society of America.
“We believe that if this technology pans out and proves effective, it will be introduced into devices very easily. That’s the advantage of left-sided stimulation,” explained Dr. Anand, professor of medicine at the University of Minnesota, Minneapolis.
He was quick to add, however, that “There needs to be a lot more work before autonomic regulation therapy is ready for prime time.” That’s because ANTHEM-HF was a relatively small prospective study – just 60 patients – and it was uncontrolled and unblinded.
Left-sided VNS is a well-established treatment for epilepsy, with more than 100,000 patients having received devices in the last several decades. It’s also seeing increasing use in refractory depression.
The approach is still investigational for heart failure, where autonomic imbalance with increased sympathetic activity and decreased parasympathetic tone is associated with heart failure progression and worse clinical outcomes, the cardiologist explained.
In ANTHEM-HF, 60 patients on optimal medical therapy for heart failure with reduced ejection fraction were randomized to left- or right-sided implantation of the Cyberonics VNS device. Once activated, the VNS intensity was titrated over 10 weeks to the maximum tolerable current that remained below the threshold of heart rate change, side effects, and patient sensation. The chronic intermittent stimulation to the vagus nerve was delivered at 10 Hz, a 250 microsec pulse width, and an average stimulation current of 2.0 mA. The stimulation cycle was 14 seconds on, 66 seconds off.
The primary study endpoint was change in LVEF over the course of 6 months. From a baseline of 32.4% it improved significantly to 37.2%, which Dr. Anand deemed “very impressive” for just 6 months of therapy. The improvement was similar regardless of whether the VNS was left or right sided.
He said the study didn’t raise any safety concerns. The only serious treatment-related adverse event was a fatal perioperative embolic stroke in a recipient of a left-sided device.
The most common adverse events were hoarseness or other voice alterations, which occurred in 19 patients. Thirteen patients reported a new cough. These side effects mirror the experience in patients treated with the VNS device for epilepsy or depression.
Discussant Jagmeet P. Singh emphasized that “it’s important to interpret the ANTHEM-HF results with some degree of caution.” That’s because the usual variability within LVEF measurements can be in the 5% range, and the 4.8% improvement seen in this study wasn’t accompanied by a significant improvement in LV end systolic volume. Moreover, the improvements seen in secondary endpoints – 6-minute walk distance, NYHA class, and the Minnesota Living With Heart Failure qualify-of-life score – are all potentially open to bias since neither patients nor physicians were blinded.
Dr. Singh noted that the NECTAR-HF study, a blinded, sham-controlled clinical trial of VNS presented earlier at the annual congress of the European Society of Cardiology, was a negative study that showed no improvement in functional measures. While NECTAR-HF caused some skeptics to question the whole concept of VNS as a useful therapeutic strategy, the stimulation protocols used in NECTAR-HF and ANTHEM-HF were quite different, and it’s likely that the stimulation amplitude employed in NECTAR-HF wasn’t sufficient to affect the target subset of vagal nerve fibers, according to Dr. Singh, director of the cardiac resynchronization therapy program and the Holter laboratory at Massachusetts General Hospital, Boston.
He considers the ANTHEM-HF finding that left-sided VNS is comparable to right to be an important advance that “really moves the field forward.” Yet many critical questions about autonomic regulation therapy for heart failure remain unanswered. These include whether it truly is safe and effective, and if so, the optimal target dose and frequency of stimulation. Answers should be forthcoming from the ongoing INOVATE-HF study, a randomized, controlled, 650-patient study, sponsored by BioControl Medical. The trial features hard clinical endpoints and higher target-dose stimulation protocols than in prior studies.
“This trial will probably put an end to the debate as to whether vagal nerve stimulation has an impact on heart failure or not,” the cardiologist predicted.
“Strategies to nonpharmacologically modulate the autonomic nervous system is an area of immense interest and investigation, and it’s going to be extended,” he observed. “I think that this is the autonomic era, and so it’s really important that we get it right. I think we’ve learned from the renal denervation experience that we need to be really careful in selecting patients for any form of autonomic manipulation. I don’t think we should select patients just off their LVEF. We should have some parameter that can quantify autonomic dysregulation prior to initiation of therapy.”
It will also be important to be able to individualize VNS therapy, just as physicians now do with beta blockers and other pharmacotherapies, he added.
The ANTHEM-HF trial was sponsored by Cyberonics. Dr. Anand reported serving as a consultant to and/or recipient of research grants from Cyberonics, Amgen, Critical Diagnostics, Novartis, and Zensun. Dr. Singh has received research support and/or served on speakers bureaus for Boston Scientific, St. Jude Medical, and the Sorin Group.
AT THE HFSA ANNUAL SCIENTIFIC MEETING
Key clinical point: Autonomic regulation therapy via a vagal nerve stimulation device shows promise for the treatment of heart failure with reduced ejection fraction.
Major finding: During 6 months of vagal nerve stimulation, patients’ mean LVEF rose significantly from 32.4% to 37.2%, with similar results regardless of whether the stimulation was left- or right-sided.
Data source: The ANTHEM-HF study was a prospective, unblinded, and uncontrolled study involving 60 patients who received vagal nerve stimulation for 6 months.
Disclosures: The study was sponsored by Cyberonics. The presenter has received research grants from and served as a consultant to that and other companies.
![[RW] Anthem-HF: Primary and secondary outcomes](https://cdn.mdedge.com/files/s3fs-public/styles/medium/public/Image/September-2017/RTEmagicC_bd9217b_106180_graphic.png.png?itok=PjlHDSzZ)