User login
Antibiotic Therapy and Bacterial Resistance in Patients With Spinal Cord Injury
Nosocomial urinary tract infections (UTIs) are often associated with significant morbidity, mortality, and health care costs.1,2 Patients with spinal cord injury (SCI) often have indwelling or intermittent urinary catheters and are prone to have asymptomatic bacteriuria and UTIs. As a result, they frequently receive antimicrobial therapy and have a higher prevalence of antibiotic resistant urinary tract isolates compared with patients without SCI.3-5 Unfortunately, data are lacking to provide guidance for optimal treatment and duration for UTIs in patients with SCI.
Many studies have evaluated patient propensity for development of antibiotic resistance in UTIs. Age > 65 years, use of a urinary catheter, previous hospitalization, and prior antimicrobial use have been identified as common risk factors.6-8 Waites and colleagues evaluated antimicrobial resistance of urinary tract organisms in outpatients with SCI and found that 33% of urinary cultures isolated multidrug-resistant microorganisms. The authors demonstrated a relationship between antimicrobial resistance and broad spectrum and prophylactic use of antibiotics.3,9
This study sought to determine the incidence of resistance acquisition by comparing susceptibility profiles of the same organisms isolated from the same patient in consecutive episodes of bacteriuria. Given that prior antimicrobial use was identified as a common risk factor for antibiotic resistance in previous reports, this study also sought to determine patterns of antibiotic use in patients with SCI at the VA North Texas Health Care System (VANTHCS) in Dallas, Texas, to evaluate whether any correlations between antibiotic use and resistance acquisition exist. A secondary objective included identification of other risk factors that may increase acquisition of resistance.
Study Design
This study was a retrospective chart review approved by the Institutional Review Board at the VANTHCS. Since computerized charting was available beginning July 2003, the VA Computerized Patient Record System was queried to identify male or female adult (aged ≥ 18 years) veterans admitted to the SCI inpatient unit between July 1, 2003, and December 31, 2009, for review. Patients who had an ICD-9 code consistent with paraplegia, tetraplegia, or quadriplegia and 2 consecutive urine cultures that isolated the same organism within 6 months of each other were included. Males with a diagnosis of epididymitis or prostatitis were excluded.
The following data were collected for analysis: gender, age, weight, height, American Spinal Injury Association (ASIA) Impairment Scale Grades (A-E), duration of hospitalization in the SCI unit, the presence and type of urinary catheter, microbiology and antibiotic regimen, past medical history, previous antibiotic history, comorbidities, and concomitant drug therapy. The presence and type of urinary catheter was determined by the primary investigator and verified by the physician who oversaw care of patients with SCI.
All antimicrobial sensitivity testing was performed via the Microscan (Microscan Systems, Inc., Renton, WA) automated testing system. Acquisition of antibiotic resistance was defined as an increase of at least 2 dilutions in the breakpoint or change on the susceptibility panel from Susceptible (S) to Resistant (R) on the repeat urine culture.
Analysis of Resistance
Continuous parameters were reported as mean (standard deviation [SD]), and discrete parameters were reported as a percentage. Analyses of variance (ANOVA) were computed to evaluate the difference in the mean of the continuous parameters. The Mann-Whitney U test replaced the ANOVA when a dependent variable was not normally distributed. Associations between pairs of discrete parameters were tested with the Pearson chi-square test. Logistic regression analyses were performed to determine the associations between potential risk factors (age, ASIA grade, antibiotic duration, class of antibiotic) and antibiotic resistance. The study alpha was α < .05. All analyses were performed with SPSS 20.0 for Windows.
Three hundred fifty-five veterans admitted to the SCI unit during the study period were initially identified. Of those, 269 did not meet inclusion criteria and were excluded. The most common reason for exclusion was absence of a second positive urine culture with isolation of the same organism. Other reasons for exclusion included no urine cultures completed while admitted to the SCI unit or no diagnosis of SCI.
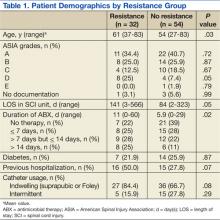
A total of 86 subjects, mean aged 56.7 years (SD, 14.2), were included in the study. Subjects were primarily men (93%) with a mean body mass index of 25.5 (SD, 7). Most of the subjects were classified Complete on the ASIA scale, meaning no motor strength or sensation below their neurologic level of injury (ASIA A; 38.4%), followed by Sensory Incomplete (ASIA B; 25.6%), Motor Incomplete-Low Muscle Strength (ASIA C; 16.3%), Motor Incomplete-High Muscle Strength (ASIA D; 14%), and Normal (ASIA E; 1.2%).
Both groups (resistance and no resistance) had similar baseline characteristics, and no differences were found for the following characteristics: ASIA grade, length of stay (LOS), presence of or control of diabetes, and presence of an indwelling urinary catheter (Table 1). However, veterans in the resistance group were significantly older than those in the no resistance group (aged 61 years vs aged 54 years; P = .03) and spent more time housed in the SCI unit with a mean LOS of 141 days vs 84 days (P = .049). Urinary pathogens developed resistance in 32 patients (37.2%, resistance group), and 54 patients (62.8%, no resistance group) did not.
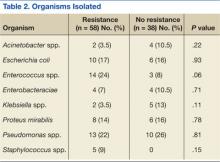
No significant differences in the types of organisms isolated were noted between the groups (Table 2). The most common pathogens isolated were Pseudomonas aeruginosa (24%), Enterococcus spp. (18%), Escherichia coli (17%), Proteus spp. (14%), Klebsiella spp. (7%), and Acinetobacter spp. (6%).
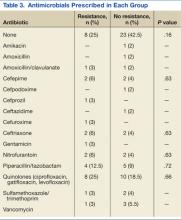
Thirty-six percent of the pathogens in the first cultures were not treated with any antibiotics, because they were considered as colonizers or contaminants. Only 61% of pathogens in the no resistance group vs 78% in the resistance group were exposed to antimicrobial treatment. In those veterans who were treated, antibiotic usage on the first urine culture was assessed to determine whether any relationship existed between receipt of a particular antimicrobial class and development of resistance. Fluoroquinolones were the most commonly prescribed antimicrobials in both the resistance and no resistance groups (Table 3).
Four risk factors (ASIA grade, antibiotic treatment duration, prior use of a cephalosporin, and prior use of penicillin) were initially identified by logistic regression analyses as being associated with resistance development. Since veterans in the resistance group were significantly older than those in the no resistance group, the analysis was repeated with age as a covariate to independently assess the association between the risk factors and resistance. After controlling for age, no significant association between the ASIA grade and resistance was identified (adjusted odds ratio [OR], 1.03; 95% confidence interval [CI]: 0.66 – 1.6). Median duration of antibiotic treatment was 6 days in all patients, 3.5 days in the no resistance group, and 9 days in the resistance group. Longer duration of treatment significantly predicted resistance (adjusted OR, 1.07; P = .03; 95% CI: 1.01 – 1.03). For every additional day the patient was on an antibiotic, he or she was 7% more likely to develop resistance.
The incidence of resistant organisms after exposure to a cephalosporin was not statistically different between groups (adjusted OR, 1.74; P = .36; 95% CI: 1.0 – 1.2). In the resistance group, 28% of the antibiotics prescribed were cephalosporins (cefuroxime, ceftriaxone, ceftazidime, and cefepime), which were used for Proteus mirabilis, Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa. In the no resistance group, 17% of the antibiotics prescribed were cephalosporins (cefepime only) and were used for Proteus mirabilis.
Organisms treated with penicillin were significantly less likely to become resistant (adjusted OR, 0.26; P = .04; 95% CI: 0.07 - 0.96). In the resistance group, 16% of the antibiotics were penicillins (piperacillin/tazobactam), which were used for Escherichia coli, Enterococcus faecalis, Acinetobacter baumannii, Pseudomonas aeruginosa, and Klebsiella pneumoniae. In the no resistance group, 22% of the antibiotics were penicillins (amoxicillin, amoxicillin/clavulanate and piperacillin/tazobactam), which were used for Proteus mirabilis, Enterococcus faecalis, and Acinetobacter baumannii.
Discussion
Longer duration of treatment significantly increased resistance on the subsequent culture in this study. For every additional day the patient was on an antibiotic, he or she was 7% more likely to develop a resistance. However, the potential impact of using a given antibiotic class on the acquisition of resistance in patients with SCI who had a UTI was not demonstrated. Surprisingly, the use of a cephalosporin was not associated with an increased incidence of resistance in this study, which was inconsistent with the findings from other studies.10 Weber and colleagues evaluated nosocomial infections in the intensive care unit. The authors suggested that restriction on the use of third-generation cephalosporins might decrease antibiotic resistance, especially in extended spectrum beta-lactamase producing gram-negative bacilli.11
The difference in this study may be explained by the lower incidence of Escherichia coli and Klebsiella pneumoniae, which are known to exhibit inducible resistance on exposure to third-generation cephalosporins. Conversely, it was found that patients treated with a penicillin were significantly less likely to develop resistant organisms from subsequent cultures. The most common penicillin used in this study’s patient population was piperacillin/tazobactam.
For complicated UTIs including pyelonephritis, the European Association of Urology (EAU) guidelines for the management of urinary and male genital tract infections recommend treatment for 3 to 5 days after defervescence or control of complicating factors.12 These recommendations could lead to much shorter treatment durations than the traditional 14-day “standard” course often prescribed. One meta-analysis recommends a 5-day course for UTIs without fever in patients with SCI vs a 14-day course for patients with fever.13 Due to the lack of data, care often varies based on the patient’s clinical status, provider experience, and opinions. The Pannek study surveyed 16 centers that specialized in SCI care. When compared with the recommendations in the EAU guidelines, the study found providers in > 50% of the responding facilities overtreated UTIs.14
Limitations
This study has several limitations. First, the sample size was much smaller than expected. Of the 355 charts reviewed, only 86 met all the criteria to be included, which limited analysis. Additionally, given the retrospective nature of the study, it was impossible to determine provider rationale for the treatment. Since a diagnosis of UTI in patients with SCI often cannot be done with conventional methods due to lack of symptoms, many investigators have emphasized the use of quantitative urinalysis to differentiate true infection vs contamination.15-17
According to the National Institute on Disability and Rehabilitation Research consensus conference recommendations, the definition of significant bacteriuria will vary, depending on the method of bladder drainage.18 While this study reviewed microbiologic cultures and the type of patient’s urinary catheter, the method of bladder drainage in the context of quantitative urinalysis was not evaluated, which limited the interpretation of microbiologic data.
It was also impossible to determine whether bacteria were cleared by the initial treatment, leading to new bacterial strains with a multidrug resistance, or whether patients relapsed. While antibiotic selection was appropriate for antimicrobial coverage, this study was not designed to detect potential inadequacies in dosing, which could also affect resistance. Last, since no genetic evaluation of the microorganisms was done, the authors cannot be sure whether the microorganisms noted on the first urine culture were of the same genetic makeup as those identified in the second urine culture.
Conclusion
Optimal duration of therapy for treatment of UTIs in patients with SCI is unclear. Despite its limitations, the study suggests exposure to longer antibiotic treatment courses may lead to increased antimicrobial resistance in the urinary tract organisms in this patient population. Further investigation with a larger sample size is required to confirm these findings.
Author disclosures
Dr. Bedimo received research grant funding from Janssen Pharmaceuticals and Merck and Company. He also serves as an ad hoc scientific advisor for Viiv Healthcare, Gilead Science, and BMD Science. All other authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
References
1. Saint S, Lipsky BA. Preventing catheter-related bacteriuria: Should we? Can we? How? Arch Intern Med. 1999;159(8):800-808.
2. Laupland KB, Bagshaw SM, Gregson DB, Kirkpatrick AW, Ross T, Church DL. Intensive care unit-acquired urinary tract infections in a regional critical care system. Crit Care. 2005;9(2):R60-R65.
3. Girard R, Mazoyer MA, Plauchu MM, Rode G. High prevalence of nosocomial infections in rehabilitation units accounted for by urinary tract infections in patients with spinal cord injury. J Hosp Infect. 2006;62(4):473-479.
4. Cardenas DD, Hooton TM. Urinary tract infection in persons with spinal cord injury. Arch Phys Med Rehabil. 1995;76(3):272-280.
5. Salomon J, Gory A, Bernard L, Ruffion A, Denys P, Chartier-Kastler E. [Urinary tract infection and neurogenic bladder]. Prog Urol. 2007;17(3):448-453.
6. Ena J, Amador C, Martinez C, Ortiz de la Tabla V. Risk factors for acquisition of urinary tract infections caused by ciprofloxacin resistant Escherichia coli. J Urol. 1995;153(1):117-120.
7. Allen UD, MacDonald N, Fuite L, Chan F, Stephens D. Risk factors for resistance to “first-line” antimicrobials among urinary tract isolates of Escherichia coli in children. CMAJ. 1999;160(10):1436-1440.
8. De Mouy D, Cavallo JD, Armengaud M, et al. [Urinary tract infection in an urban population: Etiology and antibiotic sensitivity as a function of patient history]. Presse Med. 1999;28(30):1624-1628.
9. Waites KB, Chen Y, DeVivo MJ, Canupp KC, Moser SA. Antimicrobial resistance in gram-negative bacteria isolated from the urinary tract in community-residing persons with spinal cord injury. Arch Phys Med Rehabil. 2000;81(6):764-769.
10. Shah PS, Cannon JP, Sullivan CL, Nemchausky B, Pachucki CT. Controlling antimicrobial use and decreasing microbiological laboratory tests for urinary tract infections in spinal-cord-injury patients with chronic indwelling catheters. Am J Health Syst Pharm. 2005;62(1):74-77.
11. Weber DJ, Raasch R, Rutala WA. Nosocomial infections in the ICU: The growing importance of antibiotic-resistant pathogens. Chest. 1999;115(suppl 3):34S-41S.
12. Naber KG, Bergman B, Bishop MC, et al; Urinary Tract Infection (UTI) Working Group of the Health Care Office (HCO) of the European Association of Urology (EAU). EAU guidelines for the management of urinary and male genital tract infections. Urinary Tract Infection (UTI) Working Group of the Health Care Office (HCO) of the European Association of Urology (EAU). Eur Urol. 2001;40(5):576-588.
13. Everaert K, Lumen N, Kerckhaert W, Willaert P, van Driel M. Urinary tract infections in spinal cord injury: Prevention and treatment guidelines. Acta Clin Belg. 2009;64(4):335-340.
14. Pannek J. Treatment of urinary tract infection in persons with spinal cord injury: Guidelines, evidence, and clinical practice. A questionnaire-based survey and review of the literature. J Spinal Cord Med. 2011;34(1):11-15.
15. Musher DM, Thorsteinsson SB, Airola VM II. Quantitative urinalysis. Diagnosing urinary tract infection in men. JAMA. 1976;236(18):2069-2072.
16. Deresinski SC, Perkash I. Urinary tract infections in male spinal cord injured patients. Part two: Diagnostic value of symptoms and of quantitative urinalysis. J Am Paraplegia Soc. 1985;8(1):7-10.
17. Deresinski SC, Perkash I. Urinary tract infections in male spinal cord injured patients. Part one: Bacteriologic diagnosis. J Am Paraplegia Soc. 1985;8(1):4-6.
18. Garcia Leoni ME, Esclarin De Ruz A. Management of urinary tract infection in patients with spinal cord injuries. Clin Microbiol Infect. 2003;9(8):780-785.
Nosocomial urinary tract infections (UTIs) are often associated with significant morbidity, mortality, and health care costs.1,2 Patients with spinal cord injury (SCI) often have indwelling or intermittent urinary catheters and are prone to have asymptomatic bacteriuria and UTIs. As a result, they frequently receive antimicrobial therapy and have a higher prevalence of antibiotic resistant urinary tract isolates compared with patients without SCI.3-5 Unfortunately, data are lacking to provide guidance for optimal treatment and duration for UTIs in patients with SCI.
Many studies have evaluated patient propensity for development of antibiotic resistance in UTIs. Age > 65 years, use of a urinary catheter, previous hospitalization, and prior antimicrobial use have been identified as common risk factors.6-8 Waites and colleagues evaluated antimicrobial resistance of urinary tract organisms in outpatients with SCI and found that 33% of urinary cultures isolated multidrug-resistant microorganisms. The authors demonstrated a relationship between antimicrobial resistance and broad spectrum and prophylactic use of antibiotics.3,9
This study sought to determine the incidence of resistance acquisition by comparing susceptibility profiles of the same organisms isolated from the same patient in consecutive episodes of bacteriuria. Given that prior antimicrobial use was identified as a common risk factor for antibiotic resistance in previous reports, this study also sought to determine patterns of antibiotic use in patients with SCI at the VA North Texas Health Care System (VANTHCS) in Dallas, Texas, to evaluate whether any correlations between antibiotic use and resistance acquisition exist. A secondary objective included identification of other risk factors that may increase acquisition of resistance.
Study Design
This study was a retrospective chart review approved by the Institutional Review Board at the VANTHCS. Since computerized charting was available beginning July 2003, the VA Computerized Patient Record System was queried to identify male or female adult (aged ≥ 18 years) veterans admitted to the SCI inpatient unit between July 1, 2003, and December 31, 2009, for review. Patients who had an ICD-9 code consistent with paraplegia, tetraplegia, or quadriplegia and 2 consecutive urine cultures that isolated the same organism within 6 months of each other were included. Males with a diagnosis of epididymitis or prostatitis were excluded.
The following data were collected for analysis: gender, age, weight, height, American Spinal Injury Association (ASIA) Impairment Scale Grades (A-E), duration of hospitalization in the SCI unit, the presence and type of urinary catheter, microbiology and antibiotic regimen, past medical history, previous antibiotic history, comorbidities, and concomitant drug therapy. The presence and type of urinary catheter was determined by the primary investigator and verified by the physician who oversaw care of patients with SCI.
All antimicrobial sensitivity testing was performed via the Microscan (Microscan Systems, Inc., Renton, WA) automated testing system. Acquisition of antibiotic resistance was defined as an increase of at least 2 dilutions in the breakpoint or change on the susceptibility panel from Susceptible (S) to Resistant (R) on the repeat urine culture.
Analysis of Resistance
Continuous parameters were reported as mean (standard deviation [SD]), and discrete parameters were reported as a percentage. Analyses of variance (ANOVA) were computed to evaluate the difference in the mean of the continuous parameters. The Mann-Whitney U test replaced the ANOVA when a dependent variable was not normally distributed. Associations between pairs of discrete parameters were tested with the Pearson chi-square test. Logistic regression analyses were performed to determine the associations between potential risk factors (age, ASIA grade, antibiotic duration, class of antibiotic) and antibiotic resistance. The study alpha was α < .05. All analyses were performed with SPSS 20.0 for Windows.
Three hundred fifty-five veterans admitted to the SCI unit during the study period were initially identified. Of those, 269 did not meet inclusion criteria and were excluded. The most common reason for exclusion was absence of a second positive urine culture with isolation of the same organism. Other reasons for exclusion included no urine cultures completed while admitted to the SCI unit or no diagnosis of SCI.
A total of 86 subjects, mean aged 56.7 years (SD, 14.2), were included in the study. Subjects were primarily men (93%) with a mean body mass index of 25.5 (SD, 7). Most of the subjects were classified Complete on the ASIA scale, meaning no motor strength or sensation below their neurologic level of injury (ASIA A; 38.4%), followed by Sensory Incomplete (ASIA B; 25.6%), Motor Incomplete-Low Muscle Strength (ASIA C; 16.3%), Motor Incomplete-High Muscle Strength (ASIA D; 14%), and Normal (ASIA E; 1.2%).
Both groups (resistance and no resistance) had similar baseline characteristics, and no differences were found for the following characteristics: ASIA grade, length of stay (LOS), presence of or control of diabetes, and presence of an indwelling urinary catheter (Table 1). However, veterans in the resistance group were significantly older than those in the no resistance group (aged 61 years vs aged 54 years; P = .03) and spent more time housed in the SCI unit with a mean LOS of 141 days vs 84 days (P = .049). Urinary pathogens developed resistance in 32 patients (37.2%, resistance group), and 54 patients (62.8%, no resistance group) did not.
No significant differences in the types of organisms isolated were noted between the groups (Table 2). The most common pathogens isolated were Pseudomonas aeruginosa (24%), Enterococcus spp. (18%), Escherichia coli (17%), Proteus spp. (14%), Klebsiella spp. (7%), and Acinetobacter spp. (6%).
Thirty-six percent of the pathogens in the first cultures were not treated with any antibiotics, because they were considered as colonizers or contaminants. Only 61% of pathogens in the no resistance group vs 78% in the resistance group were exposed to antimicrobial treatment. In those veterans who were treated, antibiotic usage on the first urine culture was assessed to determine whether any relationship existed between receipt of a particular antimicrobial class and development of resistance. Fluoroquinolones were the most commonly prescribed antimicrobials in both the resistance and no resistance groups (Table 3).
Four risk factors (ASIA grade, antibiotic treatment duration, prior use of a cephalosporin, and prior use of penicillin) were initially identified by logistic regression analyses as being associated with resistance development. Since veterans in the resistance group were significantly older than those in the no resistance group, the analysis was repeated with age as a covariate to independently assess the association between the risk factors and resistance. After controlling for age, no significant association between the ASIA grade and resistance was identified (adjusted odds ratio [OR], 1.03; 95% confidence interval [CI]: 0.66 – 1.6). Median duration of antibiotic treatment was 6 days in all patients, 3.5 days in the no resistance group, and 9 days in the resistance group. Longer duration of treatment significantly predicted resistance (adjusted OR, 1.07; P = .03; 95% CI: 1.01 – 1.03). For every additional day the patient was on an antibiotic, he or she was 7% more likely to develop resistance.
The incidence of resistant organisms after exposure to a cephalosporin was not statistically different between groups (adjusted OR, 1.74; P = .36; 95% CI: 1.0 – 1.2). In the resistance group, 28% of the antibiotics prescribed were cephalosporins (cefuroxime, ceftriaxone, ceftazidime, and cefepime), which were used for Proteus mirabilis, Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa. In the no resistance group, 17% of the antibiotics prescribed were cephalosporins (cefepime only) and were used for Proteus mirabilis.
Organisms treated with penicillin were significantly less likely to become resistant (adjusted OR, 0.26; P = .04; 95% CI: 0.07 - 0.96). In the resistance group, 16% of the antibiotics were penicillins (piperacillin/tazobactam), which were used for Escherichia coli, Enterococcus faecalis, Acinetobacter baumannii, Pseudomonas aeruginosa, and Klebsiella pneumoniae. In the no resistance group, 22% of the antibiotics were penicillins (amoxicillin, amoxicillin/clavulanate and piperacillin/tazobactam), which were used for Proteus mirabilis, Enterococcus faecalis, and Acinetobacter baumannii.
Discussion
Longer duration of treatment significantly increased resistance on the subsequent culture in this study. For every additional day the patient was on an antibiotic, he or she was 7% more likely to develop a resistance. However, the potential impact of using a given antibiotic class on the acquisition of resistance in patients with SCI who had a UTI was not demonstrated. Surprisingly, the use of a cephalosporin was not associated with an increased incidence of resistance in this study, which was inconsistent with the findings from other studies.10 Weber and colleagues evaluated nosocomial infections in the intensive care unit. The authors suggested that restriction on the use of third-generation cephalosporins might decrease antibiotic resistance, especially in extended spectrum beta-lactamase producing gram-negative bacilli.11
The difference in this study may be explained by the lower incidence of Escherichia coli and Klebsiella pneumoniae, which are known to exhibit inducible resistance on exposure to third-generation cephalosporins. Conversely, it was found that patients treated with a penicillin were significantly less likely to develop resistant organisms from subsequent cultures. The most common penicillin used in this study’s patient population was piperacillin/tazobactam.
For complicated UTIs including pyelonephritis, the European Association of Urology (EAU) guidelines for the management of urinary and male genital tract infections recommend treatment for 3 to 5 days after defervescence or control of complicating factors.12 These recommendations could lead to much shorter treatment durations than the traditional 14-day “standard” course often prescribed. One meta-analysis recommends a 5-day course for UTIs without fever in patients with SCI vs a 14-day course for patients with fever.13 Due to the lack of data, care often varies based on the patient’s clinical status, provider experience, and opinions. The Pannek study surveyed 16 centers that specialized in SCI care. When compared with the recommendations in the EAU guidelines, the study found providers in > 50% of the responding facilities overtreated UTIs.14
Limitations
This study has several limitations. First, the sample size was much smaller than expected. Of the 355 charts reviewed, only 86 met all the criteria to be included, which limited analysis. Additionally, given the retrospective nature of the study, it was impossible to determine provider rationale for the treatment. Since a diagnosis of UTI in patients with SCI often cannot be done with conventional methods due to lack of symptoms, many investigators have emphasized the use of quantitative urinalysis to differentiate true infection vs contamination.15-17
According to the National Institute on Disability and Rehabilitation Research consensus conference recommendations, the definition of significant bacteriuria will vary, depending on the method of bladder drainage.18 While this study reviewed microbiologic cultures and the type of patient’s urinary catheter, the method of bladder drainage in the context of quantitative urinalysis was not evaluated, which limited the interpretation of microbiologic data.
It was also impossible to determine whether bacteria were cleared by the initial treatment, leading to new bacterial strains with a multidrug resistance, or whether patients relapsed. While antibiotic selection was appropriate for antimicrobial coverage, this study was not designed to detect potential inadequacies in dosing, which could also affect resistance. Last, since no genetic evaluation of the microorganisms was done, the authors cannot be sure whether the microorganisms noted on the first urine culture were of the same genetic makeup as those identified in the second urine culture.
Conclusion
Optimal duration of therapy for treatment of UTIs in patients with SCI is unclear. Despite its limitations, the study suggests exposure to longer antibiotic treatment courses may lead to increased antimicrobial resistance in the urinary tract organisms in this patient population. Further investigation with a larger sample size is required to confirm these findings.
Author disclosures
Dr. Bedimo received research grant funding from Janssen Pharmaceuticals and Merck and Company. He also serves as an ad hoc scientific advisor for Viiv Healthcare, Gilead Science, and BMD Science. All other authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
References
1. Saint S, Lipsky BA. Preventing catheter-related bacteriuria: Should we? Can we? How? Arch Intern Med. 1999;159(8):800-808.
2. Laupland KB, Bagshaw SM, Gregson DB, Kirkpatrick AW, Ross T, Church DL. Intensive care unit-acquired urinary tract infections in a regional critical care system. Crit Care. 2005;9(2):R60-R65.
3. Girard R, Mazoyer MA, Plauchu MM, Rode G. High prevalence of nosocomial infections in rehabilitation units accounted for by urinary tract infections in patients with spinal cord injury. J Hosp Infect. 2006;62(4):473-479.
4. Cardenas DD, Hooton TM. Urinary tract infection in persons with spinal cord injury. Arch Phys Med Rehabil. 1995;76(3):272-280.
5. Salomon J, Gory A, Bernard L, Ruffion A, Denys P, Chartier-Kastler E. [Urinary tract infection and neurogenic bladder]. Prog Urol. 2007;17(3):448-453.
6. Ena J, Amador C, Martinez C, Ortiz de la Tabla V. Risk factors for acquisition of urinary tract infections caused by ciprofloxacin resistant Escherichia coli. J Urol. 1995;153(1):117-120.
7. Allen UD, MacDonald N, Fuite L, Chan F, Stephens D. Risk factors for resistance to “first-line” antimicrobials among urinary tract isolates of Escherichia coli in children. CMAJ. 1999;160(10):1436-1440.
8. De Mouy D, Cavallo JD, Armengaud M, et al. [Urinary tract infection in an urban population: Etiology and antibiotic sensitivity as a function of patient history]. Presse Med. 1999;28(30):1624-1628.
9. Waites KB, Chen Y, DeVivo MJ, Canupp KC, Moser SA. Antimicrobial resistance in gram-negative bacteria isolated from the urinary tract in community-residing persons with spinal cord injury. Arch Phys Med Rehabil. 2000;81(6):764-769.
10. Shah PS, Cannon JP, Sullivan CL, Nemchausky B, Pachucki CT. Controlling antimicrobial use and decreasing microbiological laboratory tests for urinary tract infections in spinal-cord-injury patients with chronic indwelling catheters. Am J Health Syst Pharm. 2005;62(1):74-77.
11. Weber DJ, Raasch R, Rutala WA. Nosocomial infections in the ICU: The growing importance of antibiotic-resistant pathogens. Chest. 1999;115(suppl 3):34S-41S.
12. Naber KG, Bergman B, Bishop MC, et al; Urinary Tract Infection (UTI) Working Group of the Health Care Office (HCO) of the European Association of Urology (EAU). EAU guidelines for the management of urinary and male genital tract infections. Urinary Tract Infection (UTI) Working Group of the Health Care Office (HCO) of the European Association of Urology (EAU). Eur Urol. 2001;40(5):576-588.
13. Everaert K, Lumen N, Kerckhaert W, Willaert P, van Driel M. Urinary tract infections in spinal cord injury: Prevention and treatment guidelines. Acta Clin Belg. 2009;64(4):335-340.
14. Pannek J. Treatment of urinary tract infection in persons with spinal cord injury: Guidelines, evidence, and clinical practice. A questionnaire-based survey and review of the literature. J Spinal Cord Med. 2011;34(1):11-15.
15. Musher DM, Thorsteinsson SB, Airola VM II. Quantitative urinalysis. Diagnosing urinary tract infection in men. JAMA. 1976;236(18):2069-2072.
16. Deresinski SC, Perkash I. Urinary tract infections in male spinal cord injured patients. Part two: Diagnostic value of symptoms and of quantitative urinalysis. J Am Paraplegia Soc. 1985;8(1):7-10.
17. Deresinski SC, Perkash I. Urinary tract infections in male spinal cord injured patients. Part one: Bacteriologic diagnosis. J Am Paraplegia Soc. 1985;8(1):4-6.
18. Garcia Leoni ME, Esclarin De Ruz A. Management of urinary tract infection in patients with spinal cord injuries. Clin Microbiol Infect. 2003;9(8):780-785.
Nosocomial urinary tract infections (UTIs) are often associated with significant morbidity, mortality, and health care costs.1,2 Patients with spinal cord injury (SCI) often have indwelling or intermittent urinary catheters and are prone to have asymptomatic bacteriuria and UTIs. As a result, they frequently receive antimicrobial therapy and have a higher prevalence of antibiotic resistant urinary tract isolates compared with patients without SCI.3-5 Unfortunately, data are lacking to provide guidance for optimal treatment and duration for UTIs in patients with SCI.
Many studies have evaluated patient propensity for development of antibiotic resistance in UTIs. Age > 65 years, use of a urinary catheter, previous hospitalization, and prior antimicrobial use have been identified as common risk factors.6-8 Waites and colleagues evaluated antimicrobial resistance of urinary tract organisms in outpatients with SCI and found that 33% of urinary cultures isolated multidrug-resistant microorganisms. The authors demonstrated a relationship between antimicrobial resistance and broad spectrum and prophylactic use of antibiotics.3,9
This study sought to determine the incidence of resistance acquisition by comparing susceptibility profiles of the same organisms isolated from the same patient in consecutive episodes of bacteriuria. Given that prior antimicrobial use was identified as a common risk factor for antibiotic resistance in previous reports, this study also sought to determine patterns of antibiotic use in patients with SCI at the VA North Texas Health Care System (VANTHCS) in Dallas, Texas, to evaluate whether any correlations between antibiotic use and resistance acquisition exist. A secondary objective included identification of other risk factors that may increase acquisition of resistance.
Study Design
This study was a retrospective chart review approved by the Institutional Review Board at the VANTHCS. Since computerized charting was available beginning July 2003, the VA Computerized Patient Record System was queried to identify male or female adult (aged ≥ 18 years) veterans admitted to the SCI inpatient unit between July 1, 2003, and December 31, 2009, for review. Patients who had an ICD-9 code consistent with paraplegia, tetraplegia, or quadriplegia and 2 consecutive urine cultures that isolated the same organism within 6 months of each other were included. Males with a diagnosis of epididymitis or prostatitis were excluded.
The following data were collected for analysis: gender, age, weight, height, American Spinal Injury Association (ASIA) Impairment Scale Grades (A-E), duration of hospitalization in the SCI unit, the presence and type of urinary catheter, microbiology and antibiotic regimen, past medical history, previous antibiotic history, comorbidities, and concomitant drug therapy. The presence and type of urinary catheter was determined by the primary investigator and verified by the physician who oversaw care of patients with SCI.
All antimicrobial sensitivity testing was performed via the Microscan (Microscan Systems, Inc., Renton, WA) automated testing system. Acquisition of antibiotic resistance was defined as an increase of at least 2 dilutions in the breakpoint or change on the susceptibility panel from Susceptible (S) to Resistant (R) on the repeat urine culture.
Analysis of Resistance
Continuous parameters were reported as mean (standard deviation [SD]), and discrete parameters were reported as a percentage. Analyses of variance (ANOVA) were computed to evaluate the difference in the mean of the continuous parameters. The Mann-Whitney U test replaced the ANOVA when a dependent variable was not normally distributed. Associations between pairs of discrete parameters were tested with the Pearson chi-square test. Logistic regression analyses were performed to determine the associations between potential risk factors (age, ASIA grade, antibiotic duration, class of antibiotic) and antibiotic resistance. The study alpha was α < .05. All analyses were performed with SPSS 20.0 for Windows.
Three hundred fifty-five veterans admitted to the SCI unit during the study period were initially identified. Of those, 269 did not meet inclusion criteria and were excluded. The most common reason for exclusion was absence of a second positive urine culture with isolation of the same organism. Other reasons for exclusion included no urine cultures completed while admitted to the SCI unit or no diagnosis of SCI.
A total of 86 subjects, mean aged 56.7 years (SD, 14.2), were included in the study. Subjects were primarily men (93%) with a mean body mass index of 25.5 (SD, 7). Most of the subjects were classified Complete on the ASIA scale, meaning no motor strength or sensation below their neurologic level of injury (ASIA A; 38.4%), followed by Sensory Incomplete (ASIA B; 25.6%), Motor Incomplete-Low Muscle Strength (ASIA C; 16.3%), Motor Incomplete-High Muscle Strength (ASIA D; 14%), and Normal (ASIA E; 1.2%).
Both groups (resistance and no resistance) had similar baseline characteristics, and no differences were found for the following characteristics: ASIA grade, length of stay (LOS), presence of or control of diabetes, and presence of an indwelling urinary catheter (Table 1). However, veterans in the resistance group were significantly older than those in the no resistance group (aged 61 years vs aged 54 years; P = .03) and spent more time housed in the SCI unit with a mean LOS of 141 days vs 84 days (P = .049). Urinary pathogens developed resistance in 32 patients (37.2%, resistance group), and 54 patients (62.8%, no resistance group) did not.
No significant differences in the types of organisms isolated were noted between the groups (Table 2). The most common pathogens isolated were Pseudomonas aeruginosa (24%), Enterococcus spp. (18%), Escherichia coli (17%), Proteus spp. (14%), Klebsiella spp. (7%), and Acinetobacter spp. (6%).
Thirty-six percent of the pathogens in the first cultures were not treated with any antibiotics, because they were considered as colonizers or contaminants. Only 61% of pathogens in the no resistance group vs 78% in the resistance group were exposed to antimicrobial treatment. In those veterans who were treated, antibiotic usage on the first urine culture was assessed to determine whether any relationship existed between receipt of a particular antimicrobial class and development of resistance. Fluoroquinolones were the most commonly prescribed antimicrobials in both the resistance and no resistance groups (Table 3).
Four risk factors (ASIA grade, antibiotic treatment duration, prior use of a cephalosporin, and prior use of penicillin) were initially identified by logistic regression analyses as being associated with resistance development. Since veterans in the resistance group were significantly older than those in the no resistance group, the analysis was repeated with age as a covariate to independently assess the association between the risk factors and resistance. After controlling for age, no significant association between the ASIA grade and resistance was identified (adjusted odds ratio [OR], 1.03; 95% confidence interval [CI]: 0.66 – 1.6). Median duration of antibiotic treatment was 6 days in all patients, 3.5 days in the no resistance group, and 9 days in the resistance group. Longer duration of treatment significantly predicted resistance (adjusted OR, 1.07; P = .03; 95% CI: 1.01 – 1.03). For every additional day the patient was on an antibiotic, he or she was 7% more likely to develop resistance.
The incidence of resistant organisms after exposure to a cephalosporin was not statistically different between groups (adjusted OR, 1.74; P = .36; 95% CI: 1.0 – 1.2). In the resistance group, 28% of the antibiotics prescribed were cephalosporins (cefuroxime, ceftriaxone, ceftazidime, and cefepime), which were used for Proteus mirabilis, Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa. In the no resistance group, 17% of the antibiotics prescribed were cephalosporins (cefepime only) and were used for Proteus mirabilis.
Organisms treated with penicillin were significantly less likely to become resistant (adjusted OR, 0.26; P = .04; 95% CI: 0.07 - 0.96). In the resistance group, 16% of the antibiotics were penicillins (piperacillin/tazobactam), which were used for Escherichia coli, Enterococcus faecalis, Acinetobacter baumannii, Pseudomonas aeruginosa, and Klebsiella pneumoniae. In the no resistance group, 22% of the antibiotics were penicillins (amoxicillin, amoxicillin/clavulanate and piperacillin/tazobactam), which were used for Proteus mirabilis, Enterococcus faecalis, and Acinetobacter baumannii.
Discussion
Longer duration of treatment significantly increased resistance on the subsequent culture in this study. For every additional day the patient was on an antibiotic, he or she was 7% more likely to develop a resistance. However, the potential impact of using a given antibiotic class on the acquisition of resistance in patients with SCI who had a UTI was not demonstrated. Surprisingly, the use of a cephalosporin was not associated with an increased incidence of resistance in this study, which was inconsistent with the findings from other studies.10 Weber and colleagues evaluated nosocomial infections in the intensive care unit. The authors suggested that restriction on the use of third-generation cephalosporins might decrease antibiotic resistance, especially in extended spectrum beta-lactamase producing gram-negative bacilli.11
The difference in this study may be explained by the lower incidence of Escherichia coli and Klebsiella pneumoniae, which are known to exhibit inducible resistance on exposure to third-generation cephalosporins. Conversely, it was found that patients treated with a penicillin were significantly less likely to develop resistant organisms from subsequent cultures. The most common penicillin used in this study’s patient population was piperacillin/tazobactam.
For complicated UTIs including pyelonephritis, the European Association of Urology (EAU) guidelines for the management of urinary and male genital tract infections recommend treatment for 3 to 5 days after defervescence or control of complicating factors.12 These recommendations could lead to much shorter treatment durations than the traditional 14-day “standard” course often prescribed. One meta-analysis recommends a 5-day course for UTIs without fever in patients with SCI vs a 14-day course for patients with fever.13 Due to the lack of data, care often varies based on the patient’s clinical status, provider experience, and opinions. The Pannek study surveyed 16 centers that specialized in SCI care. When compared with the recommendations in the EAU guidelines, the study found providers in > 50% of the responding facilities overtreated UTIs.14
Limitations
This study has several limitations. First, the sample size was much smaller than expected. Of the 355 charts reviewed, only 86 met all the criteria to be included, which limited analysis. Additionally, given the retrospective nature of the study, it was impossible to determine provider rationale for the treatment. Since a diagnosis of UTI in patients with SCI often cannot be done with conventional methods due to lack of symptoms, many investigators have emphasized the use of quantitative urinalysis to differentiate true infection vs contamination.15-17
According to the National Institute on Disability and Rehabilitation Research consensus conference recommendations, the definition of significant bacteriuria will vary, depending on the method of bladder drainage.18 While this study reviewed microbiologic cultures and the type of patient’s urinary catheter, the method of bladder drainage in the context of quantitative urinalysis was not evaluated, which limited the interpretation of microbiologic data.
It was also impossible to determine whether bacteria were cleared by the initial treatment, leading to new bacterial strains with a multidrug resistance, or whether patients relapsed. While antibiotic selection was appropriate for antimicrobial coverage, this study was not designed to detect potential inadequacies in dosing, which could also affect resistance. Last, since no genetic evaluation of the microorganisms was done, the authors cannot be sure whether the microorganisms noted on the first urine culture were of the same genetic makeup as those identified in the second urine culture.
Conclusion
Optimal duration of therapy for treatment of UTIs in patients with SCI is unclear. Despite its limitations, the study suggests exposure to longer antibiotic treatment courses may lead to increased antimicrobial resistance in the urinary tract organisms in this patient population. Further investigation with a larger sample size is required to confirm these findings.
Author disclosures
Dr. Bedimo received research grant funding from Janssen Pharmaceuticals and Merck and Company. He also serves as an ad hoc scientific advisor for Viiv Healthcare, Gilead Science, and BMD Science. All other authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
References
1. Saint S, Lipsky BA. Preventing catheter-related bacteriuria: Should we? Can we? How? Arch Intern Med. 1999;159(8):800-808.
2. Laupland KB, Bagshaw SM, Gregson DB, Kirkpatrick AW, Ross T, Church DL. Intensive care unit-acquired urinary tract infections in a regional critical care system. Crit Care. 2005;9(2):R60-R65.
3. Girard R, Mazoyer MA, Plauchu MM, Rode G. High prevalence of nosocomial infections in rehabilitation units accounted for by urinary tract infections in patients with spinal cord injury. J Hosp Infect. 2006;62(4):473-479.
4. Cardenas DD, Hooton TM. Urinary tract infection in persons with spinal cord injury. Arch Phys Med Rehabil. 1995;76(3):272-280.
5. Salomon J, Gory A, Bernard L, Ruffion A, Denys P, Chartier-Kastler E. [Urinary tract infection and neurogenic bladder]. Prog Urol. 2007;17(3):448-453.
6. Ena J, Amador C, Martinez C, Ortiz de la Tabla V. Risk factors for acquisition of urinary tract infections caused by ciprofloxacin resistant Escherichia coli. J Urol. 1995;153(1):117-120.
7. Allen UD, MacDonald N, Fuite L, Chan F, Stephens D. Risk factors for resistance to “first-line” antimicrobials among urinary tract isolates of Escherichia coli in children. CMAJ. 1999;160(10):1436-1440.
8. De Mouy D, Cavallo JD, Armengaud M, et al. [Urinary tract infection in an urban population: Etiology and antibiotic sensitivity as a function of patient history]. Presse Med. 1999;28(30):1624-1628.
9. Waites KB, Chen Y, DeVivo MJ, Canupp KC, Moser SA. Antimicrobial resistance in gram-negative bacteria isolated from the urinary tract in community-residing persons with spinal cord injury. Arch Phys Med Rehabil. 2000;81(6):764-769.
10. Shah PS, Cannon JP, Sullivan CL, Nemchausky B, Pachucki CT. Controlling antimicrobial use and decreasing microbiological laboratory tests for urinary tract infections in spinal-cord-injury patients with chronic indwelling catheters. Am J Health Syst Pharm. 2005;62(1):74-77.
11. Weber DJ, Raasch R, Rutala WA. Nosocomial infections in the ICU: The growing importance of antibiotic-resistant pathogens. Chest. 1999;115(suppl 3):34S-41S.
12. Naber KG, Bergman B, Bishop MC, et al; Urinary Tract Infection (UTI) Working Group of the Health Care Office (HCO) of the European Association of Urology (EAU). EAU guidelines for the management of urinary and male genital tract infections. Urinary Tract Infection (UTI) Working Group of the Health Care Office (HCO) of the European Association of Urology (EAU). Eur Urol. 2001;40(5):576-588.
13. Everaert K, Lumen N, Kerckhaert W, Willaert P, van Driel M. Urinary tract infections in spinal cord injury: Prevention and treatment guidelines. Acta Clin Belg. 2009;64(4):335-340.
14. Pannek J. Treatment of urinary tract infection in persons with spinal cord injury: Guidelines, evidence, and clinical practice. A questionnaire-based survey and review of the literature. J Spinal Cord Med. 2011;34(1):11-15.
15. Musher DM, Thorsteinsson SB, Airola VM II. Quantitative urinalysis. Diagnosing urinary tract infection in men. JAMA. 1976;236(18):2069-2072.
16. Deresinski SC, Perkash I. Urinary tract infections in male spinal cord injured patients. Part two: Diagnostic value of symptoms and of quantitative urinalysis. J Am Paraplegia Soc. 1985;8(1):7-10.
17. Deresinski SC, Perkash I. Urinary tract infections in male spinal cord injured patients. Part one: Bacteriologic diagnosis. J Am Paraplegia Soc. 1985;8(1):4-6.
18. Garcia Leoni ME, Esclarin De Ruz A. Management of urinary tract infection in patients with spinal cord injuries. Clin Microbiol Infect. 2003;9(8):780-785.