User login
Things We Do For No Reason™: Rasburicase for Adult Patients With Tumor Lysis Syndrome
Inspired by the ABIM Foundation’s Choosing Wisely ® campaign, the “Things We Do for No Reason™” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent clear-cut conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
A 35-year-old man with a history of diffuse large B-cell lymphoma (DLBCL), who most recently received treatment 12 months earlier, presents to the emergency department with abdominal pain and constipation. A computed tomography scan of the abdomen reveals retroperitoneal and mesenteric lymphadenopathy causing small bowel obstruction. The basic metabolic panel reveals a creatinine of 1.1 mg/dL, calcium of 8.5 mg/dL, phosphorus of 4 mg/dL, potassium of 4.5 mEq/L, and uric acid of 7.3 mg/dL. The admitting team contemplates using allopurinol or rasburicase for tumor lysis syndrome (TLS) prevention in the setting of recurrent DLBCL.
BACKGROUND
Tumor lysis syndrome is characterized by metabolic derangement and end-organ damage in the setting of cytotoxic chemotherapy, chemosensitive malignancy, and/or increased tumor burden.1 Risk stratification for TLS takes into account patient and disease characteristics (Table 1). Other risk factors include tumor bulk, elevated baseline serum lactate dehydrogenase, and certain types of chemotherapy (eg, cisplatin, cytarabine, etoposide, paclitaxel, cytotoxic therapies), immunotherapy, or targeted therapy.2 Elevated serum levels of uric acid, potassium, and phosphorus, as well as preexisting renal dysfunction, predispose patients to clinical TLS.3

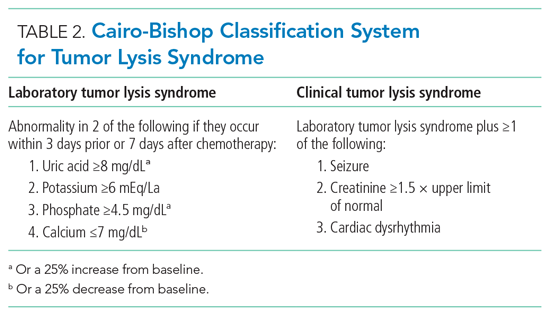
The Cairo-Bishop classification system is most frequently used to diagnose TLS (Table 2).3 Laboratory features include hyperkalemia, hyperphosphatemia, hyperuricemia, and hypocalcemia secondary to lysis of proliferating tumor cells and their nuclei. Clinical features include arrhythmias, seizures, and acute kidney injury (AKI).1 Acute kidney injury, the most common clinical complication of TLS, results from crystallization of markedly elevated plasma uric acid, leading to tubular obstruction.1,4 The development of AKI can predict morbidity (namely, the need for renal replacement therapy [RRT]) and mortality in this patient population.1
Stratifying a patient’s baseline risk of developing TLS often dictates the prevention and management plan. Therapeutic prophylaxis and management strategies for TLS include aggressive fluid resuscitation, diuresis, plasma uric acid (PUA) levels, monitoring electrolyte levels, and, in certain life-threatening situations, dialysis. Oncologists presume reducing uric acid levels prevents and treats TLS.
Current methods to reduce PUA as a means of preventing or treating TLS include xanthine oxidase inhibitors (eg, allopurinol) or urate oxidase (eg, rasburicase). Before the US Food and Drug Administration’s (FDA) approval of rasburicase to manage TLS, providers combined allopurinol (a purine analog that inhibits the enzyme xanthine oxidase, decreasing uric acid level) with aggressive fluid resuscitation. Approved by the FDA in 2002, rasburicase offers an alternative treatment for hyperuricemia by directly decreasing levels of uric acid instead of merely preventing the increased formation of uric acid. As a urate oxidase, rasburicase converts uric acid to the non-nephrotoxic, water-soluble, and freely excreted allantoin.
WHY YOU MIGHT THINK YOU SHOULD USE URATE OXIDASE IN TUMOR LYSIS SYNDROME FOR THE PREVENTION AND MANAGEMENT OF ACUTE KIDNEY INJURY
Rasburicase is often considered the standard-of-care treatment for hyperuricemia due to its ability to reduce circulating uric acid levels rapidly. The primary goal of uric acid reduction is to prevent the occurrence of AKI.
Based upon bioplausible relevance to clinically meaningful endpoints, researchers selected PUA reduction as the primary outcome in randomized controlled trials (RCTs) and observational studies to justify treatment with rasburicase. In RCTs, compassionate trials, and systematic reviews and meta-analyses, rasburicase demonstrated a more rapid reduction in uric acid levels compared to allopurinol.5 Specifically, in one study by Goldman et al,6 rasburicase decreased baseline uric acid levels in pediatric oncology patients by 86% (statistically significant) 4 hours after administration, compared to allopurinol, which only reduced baseline uric acid by 12%. According to a study by Cairo et al, allopurinol may take up to 1 day to reduce PUA.3
WHY URATE OXIDASE MAY NOT IMPROVE CLINICAL OUTCOMES IN PATIENTS AT RISK FOR OR WITH TUMOR LYSIS SYNDROME
Randomized controlled trials examining the safety, efficacy, and cost-effectiveness of rasburicase in adult patients remain sparse. Both RCTs and systematic reviews and meta-analyses rely on PUA levels as a surrogate endpoint and fail to include clinically meaningful primary endpoints (eg, change in baseline creatinine or need for RRT), raising the question as to whether rasburicase improves patient-centered outcomes.5 Since previous studies in the oncology literature show low or modest correlations between PUA reduction and patient-oriented outcomes, we must question whether PUA reduction serves as a meaningful surrogate endpoint.
Treatment of Tumor Lysis Syndrome
Two meta-analyses focusing on the treatment of TLS by Dinnel et al5 and Lopez-Olivo et al8 each included only three unique RCTs (two of the three RCTs were referenced in both meta-analyses). Moreover, both studies included only one RCT comparing rasburicase directly to allopurinol (a 2010 RCT by Cortes et al9) while the other RCTs compared the impact of different rasburicase dosing regimens. Researchers powered the head-to-head RCT by Cortes et al9 to detect a difference in PUA levels across three different arms: rasburicase, rasburicase plus allopurinol, or allopurinol alone. All three treatment arms resulted in a statistically significant reduction in serum PUA levels (87%, 78%, 66%, respectively; P = .001) without a change in the secondary, underpowered clinical outcomes such as clinical TLS or reduced renal function (defined in this study as increased creatinine, renal failure/impairment, or acute renal failure).
More recently, retrospective analyses of patients with AKI secondary to TLS found no difference in creatinine improvement, renal recovery, or prevention of RRT based on whether the patients received either rasburicase or allopurinol.10,11 While rasburicase is associated with greater PUA reduction compared to allopurinol, according to meaningful RCT and observational data as discussed previously and described further in the following section, this does not translate to clinically important risk reduction.
Prevention of Tumor Lysis Syndrome
Furthermore, there exists little compelling evidence to support the use of rasburicase for preventing AKI secondary to TLS. Even among patients at high-risk for TLS (the only group for whom rasburicase is currently recommended),5 rasburicase does not definitively prevent AKI. Data suggest that despite lowering uric acid levels, rasburicase does not consistently prevent renal injury11 or decrease the total number of subsequent inpatient days.12 The only phase 3 trial that compared the efficacy of rasburicase to allopurinol for the prevention of TLS and included clinically meaningful endpoints (eg, renal failure) found that, while rasburicase reduced uric acid levels faster than allopurinol, it did not decrease rates of clinical TLS.9
The published literature offers limited efficacy data of rasburicase in preventing TLS in low-risk patients; however, the absence of benefit of rasburicase in preventing renal failure in high-risk patients warrants skepticism as to its potential efficacy in low-risk patients.8,10
Costs-Effectiveness and Other Ethical Considerations
Rasburicase is an expensive treatment. The estimated cost of the FDA-recommended dosing is around $37,500.13 Moreover, studies comparing the cost-effectiveness of rasburicase to allopurinol focus primarily on patients at high-risk for TLS, which overestimates the cost-effectiveness of rasburicase in patients at low-to-intermediate risk for TLS.14,15 Unfortunately, some providers inappropriately prescribe rasburicase regularly to patients at low or intermediate risk for TLS. Based on observational studies of rasburicase in various clinical scenarios, including inpatient and emergency department settings, inappropriate use of rasburicase (eg, in the setting of hyperuricemia without evidence of a high-risk TLS tumor, no prior trial of allopurinol, preserved renal function, no laboratory evaluation) ranges from 32% to 70%.14,15
Finally, while <1% of patients experience rasburicase-induced anaphylaxis, 20% to 50% of patients develop gastrointestinal symptoms and viral-syndrome-like symptoms.16 Meanwhile, major side effects from allopurinol that occur with 1% to 10% frequency include maculopapular rash, pruritis, gout, nausea, vomiting, and renal failure syndrome.17 Even if the cost for rasburicase and allopurinol were similar, the lack of improved efficacy and the side-effect profiles of the two medications should make us question whether to prescribe rasburicase preferentially over allopurinol.
WHEN MIGHT URATE OXIDASE BE HELPFUL IN TUMOR LYSIS SYNDROME
While some experts recommend rasburicase prophylaxis in patients at high risk for developing TLS, such recommendations rely on low-quality evidence.2 When prescribing rasburicase, the hospitalist must ensure correct dosing. The FDA approved rasburicase for weight-based dosing at 0.2 mg/kg, though current evidence favors a single, fixed dose of 3 mg.16,17 Compared to weight-based dosing, which has an estimated cost-effectiveness ratio ranging from $27,982.77 to $119,643.59 per quality-adjusted life-year, single dosing has equivalent efficacy at approximately 50% lower cost per dose.11,17,18
WHAT YOU SHOULD DO INSTEAD
As a preventive treatment for TLS, clinicians should only consider prescribing rasburicase as a single fixed dose of 3 mg to high-risk patients.17 In the event of AKI secondary to TLS, clinicians should proceed with the mainstay treatment of resuscitation with aggressive fluid resuscitation, with a goal urine output of at least 2 mL/kg/h.1 Fluid resuscitation should be used cautiously in patients with oliguric or anuric AKI, pulmonary hypertension, congestive heart failure, and hemodynamically significant valvular disease. Clinicians should provide continuous cardiac monitoring during the initial presentation to monitor for electrocardiographic changes in the setting of hyperkalemia and hypocalcemia, and they should consult nephrology, oncology, and critical care services early in the disease course to maximize coordination of care.
RECOMMENDATIONS
Prevention
- Identify patients at high-risk of TLS (Table 1) and consider a single 3-mg dose of rasburicase.
- Manage low- and intermediate-risk patients with allopurinol and hydration.
Treatment
- Identify patients with TLS using the clinical and laboratory findings outlined in the Cairo-Bishop classification system (Table 2).
- Initiate aggressive fluid resuscitation and manage electrolyte abnormalities.
- If urate-lowering therapy is part of local hospital guidelines for TLS management, consider a single dose regimen of rasburicase utilizing shared decision-making.
CONCLUSION
Tumor lysis syndrome remains a metabolic emergency that requires rapid diagnosis and management to prevent morbidity and mortality. Current data show rasburicase rapidly decreases PUA compared to allopurinol. However, the current literature does not provide compelling evidence that rapidly lowering uric acid with rasburicase to prevent TLS or to treat AKI secondary to TLS improves patient-oriented outcomes.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason™”? Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason™” topics by emailing TWDFNR@hospitalmedicine.org
1. Howard SC, Jones DP, Pui CH. The tumor lysis syndrome. N Engl J Med.2011;364(19):1844-1854. https://doi.org/10.1056/nejmra0904569
2. Cairo MS, Coiffier B, Reiter A, Younes A; TLS Expert Panel. Recommendations for the evaluation of risk and prophylaxis of tumour lysis syndrome (TLS) in adults and children with malignant diseases: an expert TLS panel consensus. Br J Haematol. 2010;149(4):578-586. https://doi.org/10.1111/j.1365-2141.2010.08143.x
3. Cairo MS, Bishop M. Tumour lysis syndrome: new therapeutic strategies and classification. Br J Haematol.. 2004;127(1):3-11. https://doi.org/10.1111/j.1365-2141.2004.05094.x
4. Durani U, Shah ND, Go RS. In-hospital outcomes of tumor lysis syndrome: a population-based study using the National Inpatient Sample. Oncologist. 2017;22(12):1506-1509. https://doi.org/10.1634/theoncologist.2017-0147
5. Dinnel J, Moore BL, Skiver BM, Bose P. Rasburicase in the management of tumor lysis: an evidence-based review of its place in therapy. Core Evid.. 2015;10:23-38. https://doi.org/10.2147/ce.s54995
6. Goldman SC, Holcenberg JS, Finklestein JZ, et al. A randomized comparison between rasburicase and allopurinol in children with lymphoma or leukemia at high risk for tumor lysis. Blood. 2001;97(10):2998-3003. https://doi.org/10.1182/blood.v97.10.2998
7. Haslam A, Hey SP, Gill J, Prasad V. A systematic review of trial-level meta-analyses measuring the strength of association between surrogate end-points and overall survival in oncology. Eur J Cancer. 1990. 2019;106:196-211. https://doi.org/10.1016/j.ejca.2018.11.012
8. Lopez-Olivo MA, Pratt G, Palla SL, Salahudeen A. Rasburicase in tumor lysis syndrome of the adult: a systematic review and meta-analysis. Am J Kidney Dis. 2013;62(3):481-492. https://doi.org/10.1053/j.ajkd.2013.02.378
9. Cortes J, Moore JO, Maziarz RT, et al. Control of plasma uric acid in adults at risk for tumor lysis syndrome: efficacy and safety of rasburicase alone and rasburicase followed by allopurinol compared with allopurinol alone—results of a multicenter phase III study. J Clin Oncol. 2010;28(27):4207-4213. https://doi.org/10.1200/jco.2009.26.8896
10. Martens KL, Khalighi PR, Li S, et al. Comparative effectiveness of rasburicase versus allopurinol for cancer patients with renal dysfunction and hyperuricemia. Leuk Res. 2020;89:106298. https://doi.org/10.1016/j.leukres.2020.106298
11. Personett HA, Barreto EF, McCullough K, Dierkhising R, Leung N, Habermann TM. Impact of early rasburicase on incidence and outcomes of clinical tumor lysis syndrome in lymphoma. Blood. 2019;60(9)2271-2277. https://doi.org/10.1080/10428194.2019.1574000
12. Howard SC, Cockerham AR, Yvonne Barnes DN, Ryan M, Irish W, Gordan L. Real-world analysis of outpatient rasburicase to prevent and manage tumor lysis syndrome in newly diagnosed adults with leukemia or lymphoma. J Clin Pathways. 2020;6(2):46-51.
13. Abu-Hashyeh AM, Shenouda M, Al-Sharedi M. The efficacy of cost-effective fixed dose of rasburicase compared to weight-based dose in treatment and prevention of tumor lysis syndrome (TLS). J Natl Compr Canc Netw. 2020;18(3.5):QIM20-119. https://doi.org/10.6004/jnccn.2019.7516
14. Patel KK, Brown TJ, Gupta A, et al. Decreasing inappropriate use of rasburicase to promote cost-effective care. J Oncol Pract. 2019;15(2):e178-e186. https://doi.org/10.1200/jop.18.00528
15. Khalighi PR, Martens KL, White AA, et al. Utilization patterns and clinical outcomes of rasburicase administration according to tumor risk stratification. J Oncol Pharm Pract. 2020;26(3):529-535. https://doi.org/10.1177/1078155219851543
16. Elitek. Prescribing information. Sanofi-Aventis U.S., LLC; 2019. Accessed June 1, 2021. https://products.sanofi.us/elitek/Elitek.html
17. Allopurinol. Drugs & Diseases. Medscape. Accessed June 1, 2021. https://reference.medscape.com/drug/zyloprim-aloprim-allopurinol-342811
18. Jones GL, Will A, Jackson GH, Webb NJA, Rule S; British Committee for Standards in Haematology. Guidelines for the management of tumour lysis syndrome in adults and children with haematological malignancies on behalf of the British Committee for Standards in Haematology. Br J Haematol. 2015;169(5):661‐671. https://doi.org/10.1111/bjh.13403
19. Boutin A, Blackman A, O’Sullivan DM, Forcello N. The value of fixed rasburicase dosing versus weight-based dosing in the treatment and prevention of tumor lysis syndrome. J Oncol Pharm Pract. 2019;25(3):577-583. https://doi.org/10.1177/1078155217752075
Inspired by the ABIM Foundation’s Choosing Wisely ® campaign, the “Things We Do for No Reason™” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent clear-cut conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
A 35-year-old man with a history of diffuse large B-cell lymphoma (DLBCL), who most recently received treatment 12 months earlier, presents to the emergency department with abdominal pain and constipation. A computed tomography scan of the abdomen reveals retroperitoneal and mesenteric lymphadenopathy causing small bowel obstruction. The basic metabolic panel reveals a creatinine of 1.1 mg/dL, calcium of 8.5 mg/dL, phosphorus of 4 mg/dL, potassium of 4.5 mEq/L, and uric acid of 7.3 mg/dL. The admitting team contemplates using allopurinol or rasburicase for tumor lysis syndrome (TLS) prevention in the setting of recurrent DLBCL.
BACKGROUND
Tumor lysis syndrome is characterized by metabolic derangement and end-organ damage in the setting of cytotoxic chemotherapy, chemosensitive malignancy, and/or increased tumor burden.1 Risk stratification for TLS takes into account patient and disease characteristics (Table 1). Other risk factors include tumor bulk, elevated baseline serum lactate dehydrogenase, and certain types of chemotherapy (eg, cisplatin, cytarabine, etoposide, paclitaxel, cytotoxic therapies), immunotherapy, or targeted therapy.2 Elevated serum levels of uric acid, potassium, and phosphorus, as well as preexisting renal dysfunction, predispose patients to clinical TLS.3

The Cairo-Bishop classification system is most frequently used to diagnose TLS (Table 2).3 Laboratory features include hyperkalemia, hyperphosphatemia, hyperuricemia, and hypocalcemia secondary to lysis of proliferating tumor cells and their nuclei. Clinical features include arrhythmias, seizures, and acute kidney injury (AKI).1 Acute kidney injury, the most common clinical complication of TLS, results from crystallization of markedly elevated plasma uric acid, leading to tubular obstruction.1,4 The development of AKI can predict morbidity (namely, the need for renal replacement therapy [RRT]) and mortality in this patient population.1

Stratifying a patient’s baseline risk of developing TLS often dictates the prevention and management plan. Therapeutic prophylaxis and management strategies for TLS include aggressive fluid resuscitation, diuresis, plasma uric acid (PUA) levels, monitoring electrolyte levels, and, in certain life-threatening situations, dialysis. Oncologists presume reducing uric acid levels prevents and treats TLS.
Current methods to reduce PUA as a means of preventing or treating TLS include xanthine oxidase inhibitors (eg, allopurinol) or urate oxidase (eg, rasburicase). Before the US Food and Drug Administration’s (FDA) approval of rasburicase to manage TLS, providers combined allopurinol (a purine analog that inhibits the enzyme xanthine oxidase, decreasing uric acid level) with aggressive fluid resuscitation. Approved by the FDA in 2002, rasburicase offers an alternative treatment for hyperuricemia by directly decreasing levels of uric acid instead of merely preventing the increased formation of uric acid. As a urate oxidase, rasburicase converts uric acid to the non-nephrotoxic, water-soluble, and freely excreted allantoin.
WHY YOU MIGHT THINK YOU SHOULD USE URATE OXIDASE IN TUMOR LYSIS SYNDROME FOR THE PREVENTION AND MANAGEMENT OF ACUTE KIDNEY INJURY
Rasburicase is often considered the standard-of-care treatment for hyperuricemia due to its ability to reduce circulating uric acid levels rapidly. The primary goal of uric acid reduction is to prevent the occurrence of AKI.
Based upon bioplausible relevance to clinically meaningful endpoints, researchers selected PUA reduction as the primary outcome in randomized controlled trials (RCTs) and observational studies to justify treatment with rasburicase. In RCTs, compassionate trials, and systematic reviews and meta-analyses, rasburicase demonstrated a more rapid reduction in uric acid levels compared to allopurinol.5 Specifically, in one study by Goldman et al,6 rasburicase decreased baseline uric acid levels in pediatric oncology patients by 86% (statistically significant) 4 hours after administration, compared to allopurinol, which only reduced baseline uric acid by 12%. According to a study by Cairo et al, allopurinol may take up to 1 day to reduce PUA.3
WHY URATE OXIDASE MAY NOT IMPROVE CLINICAL OUTCOMES IN PATIENTS AT RISK FOR OR WITH TUMOR LYSIS SYNDROME
Randomized controlled trials examining the safety, efficacy, and cost-effectiveness of rasburicase in adult patients remain sparse. Both RCTs and systematic reviews and meta-analyses rely on PUA levels as a surrogate endpoint and fail to include clinically meaningful primary endpoints (eg, change in baseline creatinine or need for RRT), raising the question as to whether rasburicase improves patient-centered outcomes.5 Since previous studies in the oncology literature show low or modest correlations between PUA reduction and patient-oriented outcomes, we must question whether PUA reduction serves as a meaningful surrogate endpoint.
Treatment of Tumor Lysis Syndrome
Two meta-analyses focusing on the treatment of TLS by Dinnel et al5 and Lopez-Olivo et al8 each included only three unique RCTs (two of the three RCTs were referenced in both meta-analyses). Moreover, both studies included only one RCT comparing rasburicase directly to allopurinol (a 2010 RCT by Cortes et al9) while the other RCTs compared the impact of different rasburicase dosing regimens. Researchers powered the head-to-head RCT by Cortes et al9 to detect a difference in PUA levels across three different arms: rasburicase, rasburicase plus allopurinol, or allopurinol alone. All three treatment arms resulted in a statistically significant reduction in serum PUA levels (87%, 78%, 66%, respectively; P = .001) without a change in the secondary, underpowered clinical outcomes such as clinical TLS or reduced renal function (defined in this study as increased creatinine, renal failure/impairment, or acute renal failure).
More recently, retrospective analyses of patients with AKI secondary to TLS found no difference in creatinine improvement, renal recovery, or prevention of RRT based on whether the patients received either rasburicase or allopurinol.10,11 While rasburicase is associated with greater PUA reduction compared to allopurinol, according to meaningful RCT and observational data as discussed previously and described further in the following section, this does not translate to clinically important risk reduction.
Prevention of Tumor Lysis Syndrome
Furthermore, there exists little compelling evidence to support the use of rasburicase for preventing AKI secondary to TLS. Even among patients at high-risk for TLS (the only group for whom rasburicase is currently recommended),5 rasburicase does not definitively prevent AKI. Data suggest that despite lowering uric acid levels, rasburicase does not consistently prevent renal injury11 or decrease the total number of subsequent inpatient days.12 The only phase 3 trial that compared the efficacy of rasburicase to allopurinol for the prevention of TLS and included clinically meaningful endpoints (eg, renal failure) found that, while rasburicase reduced uric acid levels faster than allopurinol, it did not decrease rates of clinical TLS.9
The published literature offers limited efficacy data of rasburicase in preventing TLS in low-risk patients; however, the absence of benefit of rasburicase in preventing renal failure in high-risk patients warrants skepticism as to its potential efficacy in low-risk patients.8,10
Costs-Effectiveness and Other Ethical Considerations
Rasburicase is an expensive treatment. The estimated cost of the FDA-recommended dosing is around $37,500.13 Moreover, studies comparing the cost-effectiveness of rasburicase to allopurinol focus primarily on patients at high-risk for TLS, which overestimates the cost-effectiveness of rasburicase in patients at low-to-intermediate risk for TLS.14,15 Unfortunately, some providers inappropriately prescribe rasburicase regularly to patients at low or intermediate risk for TLS. Based on observational studies of rasburicase in various clinical scenarios, including inpatient and emergency department settings, inappropriate use of rasburicase (eg, in the setting of hyperuricemia without evidence of a high-risk TLS tumor, no prior trial of allopurinol, preserved renal function, no laboratory evaluation) ranges from 32% to 70%.14,15
Finally, while <1% of patients experience rasburicase-induced anaphylaxis, 20% to 50% of patients develop gastrointestinal symptoms and viral-syndrome-like symptoms.16 Meanwhile, major side effects from allopurinol that occur with 1% to 10% frequency include maculopapular rash, pruritis, gout, nausea, vomiting, and renal failure syndrome.17 Even if the cost for rasburicase and allopurinol were similar, the lack of improved efficacy and the side-effect profiles of the two medications should make us question whether to prescribe rasburicase preferentially over allopurinol.
WHEN MIGHT URATE OXIDASE BE HELPFUL IN TUMOR LYSIS SYNDROME
While some experts recommend rasburicase prophylaxis in patients at high risk for developing TLS, such recommendations rely on low-quality evidence.2 When prescribing rasburicase, the hospitalist must ensure correct dosing. The FDA approved rasburicase for weight-based dosing at 0.2 mg/kg, though current evidence favors a single, fixed dose of 3 mg.16,17 Compared to weight-based dosing, which has an estimated cost-effectiveness ratio ranging from $27,982.77 to $119,643.59 per quality-adjusted life-year, single dosing has equivalent efficacy at approximately 50% lower cost per dose.11,17,18
WHAT YOU SHOULD DO INSTEAD
As a preventive treatment for TLS, clinicians should only consider prescribing rasburicase as a single fixed dose of 3 mg to high-risk patients.17 In the event of AKI secondary to TLS, clinicians should proceed with the mainstay treatment of resuscitation with aggressive fluid resuscitation, with a goal urine output of at least 2 mL/kg/h.1 Fluid resuscitation should be used cautiously in patients with oliguric or anuric AKI, pulmonary hypertension, congestive heart failure, and hemodynamically significant valvular disease. Clinicians should provide continuous cardiac monitoring during the initial presentation to monitor for electrocardiographic changes in the setting of hyperkalemia and hypocalcemia, and they should consult nephrology, oncology, and critical care services early in the disease course to maximize coordination of care.
RECOMMENDATIONS
Prevention
- Identify patients at high-risk of TLS (Table 1) and consider a single 3-mg dose of rasburicase.
- Manage low- and intermediate-risk patients with allopurinol and hydration.
Treatment
- Identify patients with TLS using the clinical and laboratory findings outlined in the Cairo-Bishop classification system (Table 2).
- Initiate aggressive fluid resuscitation and manage electrolyte abnormalities.
- If urate-lowering therapy is part of local hospital guidelines for TLS management, consider a single dose regimen of rasburicase utilizing shared decision-making.
CONCLUSION
Tumor lysis syndrome remains a metabolic emergency that requires rapid diagnosis and management to prevent morbidity and mortality. Current data show rasburicase rapidly decreases PUA compared to allopurinol. However, the current literature does not provide compelling evidence that rapidly lowering uric acid with rasburicase to prevent TLS or to treat AKI secondary to TLS improves patient-oriented outcomes.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason™”? Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason™” topics by emailing TWDFNR@hospitalmedicine.org
Inspired by the ABIM Foundation’s Choosing Wisely ® campaign, the “Things We Do for No Reason™” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent clear-cut conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
A 35-year-old man with a history of diffuse large B-cell lymphoma (DLBCL), who most recently received treatment 12 months earlier, presents to the emergency department with abdominal pain and constipation. A computed tomography scan of the abdomen reveals retroperitoneal and mesenteric lymphadenopathy causing small bowel obstruction. The basic metabolic panel reveals a creatinine of 1.1 mg/dL, calcium of 8.5 mg/dL, phosphorus of 4 mg/dL, potassium of 4.5 mEq/L, and uric acid of 7.3 mg/dL. The admitting team contemplates using allopurinol or rasburicase for tumor lysis syndrome (TLS) prevention in the setting of recurrent DLBCL.
BACKGROUND
Tumor lysis syndrome is characterized by metabolic derangement and end-organ damage in the setting of cytotoxic chemotherapy, chemosensitive malignancy, and/or increased tumor burden.1 Risk stratification for TLS takes into account patient and disease characteristics (Table 1). Other risk factors include tumor bulk, elevated baseline serum lactate dehydrogenase, and certain types of chemotherapy (eg, cisplatin, cytarabine, etoposide, paclitaxel, cytotoxic therapies), immunotherapy, or targeted therapy.2 Elevated serum levels of uric acid, potassium, and phosphorus, as well as preexisting renal dysfunction, predispose patients to clinical TLS.3

The Cairo-Bishop classification system is most frequently used to diagnose TLS (Table 2).3 Laboratory features include hyperkalemia, hyperphosphatemia, hyperuricemia, and hypocalcemia secondary to lysis of proliferating tumor cells and their nuclei. Clinical features include arrhythmias, seizures, and acute kidney injury (AKI).1 Acute kidney injury, the most common clinical complication of TLS, results from crystallization of markedly elevated plasma uric acid, leading to tubular obstruction.1,4 The development of AKI can predict morbidity (namely, the need for renal replacement therapy [RRT]) and mortality in this patient population.1

Stratifying a patient’s baseline risk of developing TLS often dictates the prevention and management plan. Therapeutic prophylaxis and management strategies for TLS include aggressive fluid resuscitation, diuresis, plasma uric acid (PUA) levels, monitoring electrolyte levels, and, in certain life-threatening situations, dialysis. Oncologists presume reducing uric acid levels prevents and treats TLS.
Current methods to reduce PUA as a means of preventing or treating TLS include xanthine oxidase inhibitors (eg, allopurinol) or urate oxidase (eg, rasburicase). Before the US Food and Drug Administration’s (FDA) approval of rasburicase to manage TLS, providers combined allopurinol (a purine analog that inhibits the enzyme xanthine oxidase, decreasing uric acid level) with aggressive fluid resuscitation. Approved by the FDA in 2002, rasburicase offers an alternative treatment for hyperuricemia by directly decreasing levels of uric acid instead of merely preventing the increased formation of uric acid. As a urate oxidase, rasburicase converts uric acid to the non-nephrotoxic, water-soluble, and freely excreted allantoin.
WHY YOU MIGHT THINK YOU SHOULD USE URATE OXIDASE IN TUMOR LYSIS SYNDROME FOR THE PREVENTION AND MANAGEMENT OF ACUTE KIDNEY INJURY
Rasburicase is often considered the standard-of-care treatment for hyperuricemia due to its ability to reduce circulating uric acid levels rapidly. The primary goal of uric acid reduction is to prevent the occurrence of AKI.
Based upon bioplausible relevance to clinically meaningful endpoints, researchers selected PUA reduction as the primary outcome in randomized controlled trials (RCTs) and observational studies to justify treatment with rasburicase. In RCTs, compassionate trials, and systematic reviews and meta-analyses, rasburicase demonstrated a more rapid reduction in uric acid levels compared to allopurinol.5 Specifically, in one study by Goldman et al,6 rasburicase decreased baseline uric acid levels in pediatric oncology patients by 86% (statistically significant) 4 hours after administration, compared to allopurinol, which only reduced baseline uric acid by 12%. According to a study by Cairo et al, allopurinol may take up to 1 day to reduce PUA.3
WHY URATE OXIDASE MAY NOT IMPROVE CLINICAL OUTCOMES IN PATIENTS AT RISK FOR OR WITH TUMOR LYSIS SYNDROME
Randomized controlled trials examining the safety, efficacy, and cost-effectiveness of rasburicase in adult patients remain sparse. Both RCTs and systematic reviews and meta-analyses rely on PUA levels as a surrogate endpoint and fail to include clinically meaningful primary endpoints (eg, change in baseline creatinine or need for RRT), raising the question as to whether rasburicase improves patient-centered outcomes.5 Since previous studies in the oncology literature show low or modest correlations between PUA reduction and patient-oriented outcomes, we must question whether PUA reduction serves as a meaningful surrogate endpoint.
Treatment of Tumor Lysis Syndrome
Two meta-analyses focusing on the treatment of TLS by Dinnel et al5 and Lopez-Olivo et al8 each included only three unique RCTs (two of the three RCTs were referenced in both meta-analyses). Moreover, both studies included only one RCT comparing rasburicase directly to allopurinol (a 2010 RCT by Cortes et al9) while the other RCTs compared the impact of different rasburicase dosing regimens. Researchers powered the head-to-head RCT by Cortes et al9 to detect a difference in PUA levels across three different arms: rasburicase, rasburicase plus allopurinol, or allopurinol alone. All three treatment arms resulted in a statistically significant reduction in serum PUA levels (87%, 78%, 66%, respectively; P = .001) without a change in the secondary, underpowered clinical outcomes such as clinical TLS or reduced renal function (defined in this study as increased creatinine, renal failure/impairment, or acute renal failure).
More recently, retrospective analyses of patients with AKI secondary to TLS found no difference in creatinine improvement, renal recovery, or prevention of RRT based on whether the patients received either rasburicase or allopurinol.10,11 While rasburicase is associated with greater PUA reduction compared to allopurinol, according to meaningful RCT and observational data as discussed previously and described further in the following section, this does not translate to clinically important risk reduction.
Prevention of Tumor Lysis Syndrome
Furthermore, there exists little compelling evidence to support the use of rasburicase for preventing AKI secondary to TLS. Even among patients at high-risk for TLS (the only group for whom rasburicase is currently recommended),5 rasburicase does not definitively prevent AKI. Data suggest that despite lowering uric acid levels, rasburicase does not consistently prevent renal injury11 or decrease the total number of subsequent inpatient days.12 The only phase 3 trial that compared the efficacy of rasburicase to allopurinol for the prevention of TLS and included clinically meaningful endpoints (eg, renal failure) found that, while rasburicase reduced uric acid levels faster than allopurinol, it did not decrease rates of clinical TLS.9
The published literature offers limited efficacy data of rasburicase in preventing TLS in low-risk patients; however, the absence of benefit of rasburicase in preventing renal failure in high-risk patients warrants skepticism as to its potential efficacy in low-risk patients.8,10
Costs-Effectiveness and Other Ethical Considerations
Rasburicase is an expensive treatment. The estimated cost of the FDA-recommended dosing is around $37,500.13 Moreover, studies comparing the cost-effectiveness of rasburicase to allopurinol focus primarily on patients at high-risk for TLS, which overestimates the cost-effectiveness of rasburicase in patients at low-to-intermediate risk for TLS.14,15 Unfortunately, some providers inappropriately prescribe rasburicase regularly to patients at low or intermediate risk for TLS. Based on observational studies of rasburicase in various clinical scenarios, including inpatient and emergency department settings, inappropriate use of rasburicase (eg, in the setting of hyperuricemia without evidence of a high-risk TLS tumor, no prior trial of allopurinol, preserved renal function, no laboratory evaluation) ranges from 32% to 70%.14,15
Finally, while <1% of patients experience rasburicase-induced anaphylaxis, 20% to 50% of patients develop gastrointestinal symptoms and viral-syndrome-like symptoms.16 Meanwhile, major side effects from allopurinol that occur with 1% to 10% frequency include maculopapular rash, pruritis, gout, nausea, vomiting, and renal failure syndrome.17 Even if the cost for rasburicase and allopurinol were similar, the lack of improved efficacy and the side-effect profiles of the two medications should make us question whether to prescribe rasburicase preferentially over allopurinol.
WHEN MIGHT URATE OXIDASE BE HELPFUL IN TUMOR LYSIS SYNDROME
While some experts recommend rasburicase prophylaxis in patients at high risk for developing TLS, such recommendations rely on low-quality evidence.2 When prescribing rasburicase, the hospitalist must ensure correct dosing. The FDA approved rasburicase for weight-based dosing at 0.2 mg/kg, though current evidence favors a single, fixed dose of 3 mg.16,17 Compared to weight-based dosing, which has an estimated cost-effectiveness ratio ranging from $27,982.77 to $119,643.59 per quality-adjusted life-year, single dosing has equivalent efficacy at approximately 50% lower cost per dose.11,17,18
WHAT YOU SHOULD DO INSTEAD
As a preventive treatment for TLS, clinicians should only consider prescribing rasburicase as a single fixed dose of 3 mg to high-risk patients.17 In the event of AKI secondary to TLS, clinicians should proceed with the mainstay treatment of resuscitation with aggressive fluid resuscitation, with a goal urine output of at least 2 mL/kg/h.1 Fluid resuscitation should be used cautiously in patients with oliguric or anuric AKI, pulmonary hypertension, congestive heart failure, and hemodynamically significant valvular disease. Clinicians should provide continuous cardiac monitoring during the initial presentation to monitor for electrocardiographic changes in the setting of hyperkalemia and hypocalcemia, and they should consult nephrology, oncology, and critical care services early in the disease course to maximize coordination of care.
RECOMMENDATIONS
Prevention
- Identify patients at high-risk of TLS (Table 1) and consider a single 3-mg dose of rasburicase.
- Manage low- and intermediate-risk patients with allopurinol and hydration.
Treatment
- Identify patients with TLS using the clinical and laboratory findings outlined in the Cairo-Bishop classification system (Table 2).
- Initiate aggressive fluid resuscitation and manage electrolyte abnormalities.
- If urate-lowering therapy is part of local hospital guidelines for TLS management, consider a single dose regimen of rasburicase utilizing shared decision-making.
CONCLUSION
Tumor lysis syndrome remains a metabolic emergency that requires rapid diagnosis and management to prevent morbidity and mortality. Current data show rasburicase rapidly decreases PUA compared to allopurinol. However, the current literature does not provide compelling evidence that rapidly lowering uric acid with rasburicase to prevent TLS or to treat AKI secondary to TLS improves patient-oriented outcomes.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason™”? Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason™” topics by emailing TWDFNR@hospitalmedicine.org
1. Howard SC, Jones DP, Pui CH. The tumor lysis syndrome. N Engl J Med.2011;364(19):1844-1854. https://doi.org/10.1056/nejmra0904569
2. Cairo MS, Coiffier B, Reiter A, Younes A; TLS Expert Panel. Recommendations for the evaluation of risk and prophylaxis of tumour lysis syndrome (TLS) in adults and children with malignant diseases: an expert TLS panel consensus. Br J Haematol. 2010;149(4):578-586. https://doi.org/10.1111/j.1365-2141.2010.08143.x
3. Cairo MS, Bishop M. Tumour lysis syndrome: new therapeutic strategies and classification. Br J Haematol.. 2004;127(1):3-11. https://doi.org/10.1111/j.1365-2141.2004.05094.x
4. Durani U, Shah ND, Go RS. In-hospital outcomes of tumor lysis syndrome: a population-based study using the National Inpatient Sample. Oncologist. 2017;22(12):1506-1509. https://doi.org/10.1634/theoncologist.2017-0147
5. Dinnel J, Moore BL, Skiver BM, Bose P. Rasburicase in the management of tumor lysis: an evidence-based review of its place in therapy. Core Evid.. 2015;10:23-38. https://doi.org/10.2147/ce.s54995
6. Goldman SC, Holcenberg JS, Finklestein JZ, et al. A randomized comparison between rasburicase and allopurinol in children with lymphoma or leukemia at high risk for tumor lysis. Blood. 2001;97(10):2998-3003. https://doi.org/10.1182/blood.v97.10.2998
7. Haslam A, Hey SP, Gill J, Prasad V. A systematic review of trial-level meta-analyses measuring the strength of association between surrogate end-points and overall survival in oncology. Eur J Cancer. 1990. 2019;106:196-211. https://doi.org/10.1016/j.ejca.2018.11.012
8. Lopez-Olivo MA, Pratt G, Palla SL, Salahudeen A. Rasburicase in tumor lysis syndrome of the adult: a systematic review and meta-analysis. Am J Kidney Dis. 2013;62(3):481-492. https://doi.org/10.1053/j.ajkd.2013.02.378
9. Cortes J, Moore JO, Maziarz RT, et al. Control of plasma uric acid in adults at risk for tumor lysis syndrome: efficacy and safety of rasburicase alone and rasburicase followed by allopurinol compared with allopurinol alone—results of a multicenter phase III study. J Clin Oncol. 2010;28(27):4207-4213. https://doi.org/10.1200/jco.2009.26.8896
10. Martens KL, Khalighi PR, Li S, et al. Comparative effectiveness of rasburicase versus allopurinol for cancer patients with renal dysfunction and hyperuricemia. Leuk Res. 2020;89:106298. https://doi.org/10.1016/j.leukres.2020.106298
11. Personett HA, Barreto EF, McCullough K, Dierkhising R, Leung N, Habermann TM. Impact of early rasburicase on incidence and outcomes of clinical tumor lysis syndrome in lymphoma. Blood. 2019;60(9)2271-2277. https://doi.org/10.1080/10428194.2019.1574000
12. Howard SC, Cockerham AR, Yvonne Barnes DN, Ryan M, Irish W, Gordan L. Real-world analysis of outpatient rasburicase to prevent and manage tumor lysis syndrome in newly diagnosed adults with leukemia or lymphoma. J Clin Pathways. 2020;6(2):46-51.
13. Abu-Hashyeh AM, Shenouda M, Al-Sharedi M. The efficacy of cost-effective fixed dose of rasburicase compared to weight-based dose in treatment and prevention of tumor lysis syndrome (TLS). J Natl Compr Canc Netw. 2020;18(3.5):QIM20-119. https://doi.org/10.6004/jnccn.2019.7516
14. Patel KK, Brown TJ, Gupta A, et al. Decreasing inappropriate use of rasburicase to promote cost-effective care. J Oncol Pract. 2019;15(2):e178-e186. https://doi.org/10.1200/jop.18.00528
15. Khalighi PR, Martens KL, White AA, et al. Utilization patterns and clinical outcomes of rasburicase administration according to tumor risk stratification. J Oncol Pharm Pract. 2020;26(3):529-535. https://doi.org/10.1177/1078155219851543
16. Elitek. Prescribing information. Sanofi-Aventis U.S., LLC; 2019. Accessed June 1, 2021. https://products.sanofi.us/elitek/Elitek.html
17. Allopurinol. Drugs & Diseases. Medscape. Accessed June 1, 2021. https://reference.medscape.com/drug/zyloprim-aloprim-allopurinol-342811
18. Jones GL, Will A, Jackson GH, Webb NJA, Rule S; British Committee for Standards in Haematology. Guidelines for the management of tumour lysis syndrome in adults and children with haematological malignancies on behalf of the British Committee for Standards in Haematology. Br J Haematol. 2015;169(5):661‐671. https://doi.org/10.1111/bjh.13403
19. Boutin A, Blackman A, O’Sullivan DM, Forcello N. The value of fixed rasburicase dosing versus weight-based dosing in the treatment and prevention of tumor lysis syndrome. J Oncol Pharm Pract. 2019;25(3):577-583. https://doi.org/10.1177/1078155217752075
1. Howard SC, Jones DP, Pui CH. The tumor lysis syndrome. N Engl J Med.2011;364(19):1844-1854. https://doi.org/10.1056/nejmra0904569
2. Cairo MS, Coiffier B, Reiter A, Younes A; TLS Expert Panel. Recommendations for the evaluation of risk and prophylaxis of tumour lysis syndrome (TLS) in adults and children with malignant diseases: an expert TLS panel consensus. Br J Haematol. 2010;149(4):578-586. https://doi.org/10.1111/j.1365-2141.2010.08143.x
3. Cairo MS, Bishop M. Tumour lysis syndrome: new therapeutic strategies and classification. Br J Haematol.. 2004;127(1):3-11. https://doi.org/10.1111/j.1365-2141.2004.05094.x
4. Durani U, Shah ND, Go RS. In-hospital outcomes of tumor lysis syndrome: a population-based study using the National Inpatient Sample. Oncologist. 2017;22(12):1506-1509. https://doi.org/10.1634/theoncologist.2017-0147
5. Dinnel J, Moore BL, Skiver BM, Bose P. Rasburicase in the management of tumor lysis: an evidence-based review of its place in therapy. Core Evid.. 2015;10:23-38. https://doi.org/10.2147/ce.s54995
6. Goldman SC, Holcenberg JS, Finklestein JZ, et al. A randomized comparison between rasburicase and allopurinol in children with lymphoma or leukemia at high risk for tumor lysis. Blood. 2001;97(10):2998-3003. https://doi.org/10.1182/blood.v97.10.2998
7. Haslam A, Hey SP, Gill J, Prasad V. A systematic review of trial-level meta-analyses measuring the strength of association between surrogate end-points and overall survival in oncology. Eur J Cancer. 1990. 2019;106:196-211. https://doi.org/10.1016/j.ejca.2018.11.012
8. Lopez-Olivo MA, Pratt G, Palla SL, Salahudeen A. Rasburicase in tumor lysis syndrome of the adult: a systematic review and meta-analysis. Am J Kidney Dis. 2013;62(3):481-492. https://doi.org/10.1053/j.ajkd.2013.02.378
9. Cortes J, Moore JO, Maziarz RT, et al. Control of plasma uric acid in adults at risk for tumor lysis syndrome: efficacy and safety of rasburicase alone and rasburicase followed by allopurinol compared with allopurinol alone—results of a multicenter phase III study. J Clin Oncol. 2010;28(27):4207-4213. https://doi.org/10.1200/jco.2009.26.8896
10. Martens KL, Khalighi PR, Li S, et al. Comparative effectiveness of rasburicase versus allopurinol for cancer patients with renal dysfunction and hyperuricemia. Leuk Res. 2020;89:106298. https://doi.org/10.1016/j.leukres.2020.106298
11. Personett HA, Barreto EF, McCullough K, Dierkhising R, Leung N, Habermann TM. Impact of early rasburicase on incidence and outcomes of clinical tumor lysis syndrome in lymphoma. Blood. 2019;60(9)2271-2277. https://doi.org/10.1080/10428194.2019.1574000
12. Howard SC, Cockerham AR, Yvonne Barnes DN, Ryan M, Irish W, Gordan L. Real-world analysis of outpatient rasburicase to prevent and manage tumor lysis syndrome in newly diagnosed adults with leukemia or lymphoma. J Clin Pathways. 2020;6(2):46-51.
13. Abu-Hashyeh AM, Shenouda M, Al-Sharedi M. The efficacy of cost-effective fixed dose of rasburicase compared to weight-based dose in treatment and prevention of tumor lysis syndrome (TLS). J Natl Compr Canc Netw. 2020;18(3.5):QIM20-119. https://doi.org/10.6004/jnccn.2019.7516
14. Patel KK, Brown TJ, Gupta A, et al. Decreasing inappropriate use of rasburicase to promote cost-effective care. J Oncol Pract. 2019;15(2):e178-e186. https://doi.org/10.1200/jop.18.00528
15. Khalighi PR, Martens KL, White AA, et al. Utilization patterns and clinical outcomes of rasburicase administration according to tumor risk stratification. J Oncol Pharm Pract. 2020;26(3):529-535. https://doi.org/10.1177/1078155219851543
16. Elitek. Prescribing information. Sanofi-Aventis U.S., LLC; 2019. Accessed June 1, 2021. https://products.sanofi.us/elitek/Elitek.html
17. Allopurinol. Drugs & Diseases. Medscape. Accessed June 1, 2021. https://reference.medscape.com/drug/zyloprim-aloprim-allopurinol-342811
18. Jones GL, Will A, Jackson GH, Webb NJA, Rule S; British Committee for Standards in Haematology. Guidelines for the management of tumour lysis syndrome in adults and children with haematological malignancies on behalf of the British Committee for Standards in Haematology. Br J Haematol. 2015;169(5):661‐671. https://doi.org/10.1111/bjh.13403
19. Boutin A, Blackman A, O’Sullivan DM, Forcello N. The value of fixed rasburicase dosing versus weight-based dosing in the treatment and prevention of tumor lysis syndrome. J Oncol Pharm Pract. 2019;25(3):577-583. https://doi.org/10.1177/1078155217752075
© 2021 Society of Hospital Medicine