User login
2023 Update on fertility
Total fertility rate and fertility care: Demographic shifts and changing demands
Vollset SE, Goren E, Yuan C-W, et al. Fertility, mortality, migration, and population scenarios for 195 countries and territories from 2017 to 2100: a forecasting analysis for the Global Burden of Disease Study. Lancet. 2020;396:1285-1306.
The total fertility rate (TFR) globally is decreasing rapidly, and in the United States it is now 1.8 births per woman, well below the required replacement rate of 2.1 that maintains the population.1 These reduced TFRs result in significant demographic shifts that affect the economy, workforce, society, health care needs, environment, and geopolitical standing of every country. These changes also will shift demands for the volume and type of services delivered by women’s health care clinicians.
In addition to the TFR, mortality rates and migration rates play essential roles in determining a country’s population.2 Anticipation and planning for these population and health care service changes by each country’s government, business, professionals, and other stakeholders are imperative to manage their impact and optimize quality of life.

US standings in projected population and economic growth
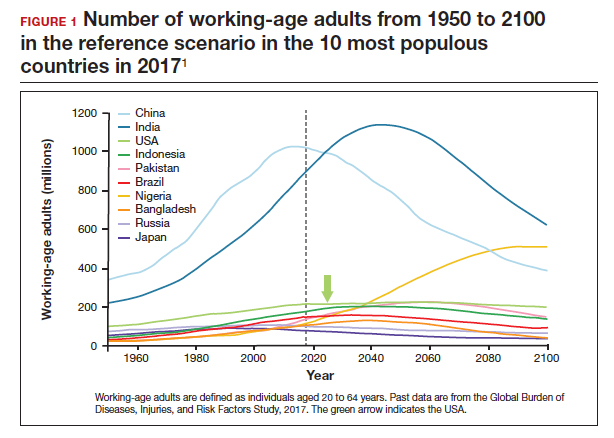
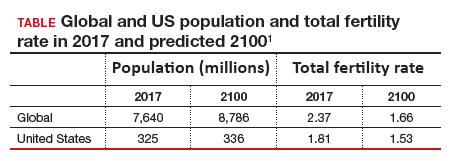
The US population is predicted to peak at 364 million in 2062 and decrease to 336 million in 2100, at which time it will be the fourth largest country in the world, according to a forecasting analysis by Vollset and colleagues.1 China is expected to become the biggest economy in the world in 2035, but this is predicted to change because of its decreasing population so that by 2098 the United States will again be the country with the largest economy (FIGURE 1).1
For the United States to maintain its economic and geopolitical standing, it is important to have policies that promote families. Other countries, especially in northern Europe, have implemented such policies. These include education of the population,economic incentives to create families, extended day care, and favorable tax policies.3 They also include increased access to family-forming fertility care. Such policies in Denmark have resulted in approximately 10% of all children being born from assisted reproductive technology (ART), compared with about 1.5% in the United States. Other countries have similar policies and success in increasing the number of children born from ART.
In the United States, the American Society for Reproductive Medicine (ASRM), RESOLVE: the National Infertility Association, the American Medical Women’s Association (AMWA), and others are promoting the need for increased access to fertility care and family-forming resources, primarily through family-forming benefits provided by companies.4 Such benefits are critical since the primary reason most people do not undergo fertility care is a lack of affordability. Only 1 person in 4 in the United States who needs fertility care receives treatment. Increased access would result in more babies being born to help address the reduced TFR.
Educational access, contraceptive goals, and access to fertility care
Continued trends in women’s educational attainment and access to contraception will hasten declines in the fertility rate and slow population growth (TABLE).1 These educational and contraceptive goals also must be pursued so that every person can achieve their individual reproductive life goals of having a family if and when they want to have a family. In addition to helping address the decreasing TFR, there is a fundamental right to found a family, as stated in the United Nations charter. It is a matter of social justice and equity that everyone who wants to have a family can access reproductive care on a nondiscriminatory basis when needed.
While the need for more and better insurance coverage for infertility has been well documented for many years, the decreasing TFR in the United States is an additional compelling reason that government, business, and other stakeholders should continue to increase access to fertility benefits and care. Women’s health care clinicians are encouraged to support these initiatives that also improve quality of life, equity, and social justice.
The decreasing global and US total fertility rate causes significant demographic changes, with major socioeconomic and health care consequences. The reduced TFR impacts women’s health care services, including the need for increased access to fertility care. Government and corporate policies, including those that improve access to fertility care, will help society adapt to these changes.
Continue to: A new comprehensive ovulatory disorders classification system developed by FIGO...
A new comprehensive ovulatory disorders classification system developed by FIGO
Munro MG, Balen AH, Cho S, et al; FIGO Committee on Menstrual Disorders and Related Health Impacts, and FIGO Committee on Reproductive Medicine, Endocrinology, and Infertility. The FIGO ovulatory disorders classification system. Fertil Steril. 2022;118:768-786.
Ovulatory disorders are well-recognized and common causes of infertility and abnormal uterine bleeding (AUB). Ovulatory disorders occur on a spectrum, with the most severe form being anovulation, and comprise a heterogeneous group that has been classically categorized based on an initial monograph published by the World Health Organization (WHO) in 1973. That classification was based on gonadotropin levels and categorized these disorders into 3 groups: 1) hypogonadotropic (such as hypothalamic amenorrhea), 2) eugonadotropic (such as polycystic ovary syndrome [PCOS]), and 3) hypergonadotropic (such as primary ovarian insufficiency). This initial classification was the subject of several subsequent iterations and modifications over the past 50 years; for example, at one point, ovulatory disorder caused by hyperprolactinemia was added as a separate fourth category. However, due to advances in endocrine assays, imaging technology, and genetics, our understanding of ovulatory disorders has expanded remarkably over the past several decades.
Previous FIGO classifications
Considering the emergent complexity of these disorders and the limitations of the original WHO classification to capture these subtleties adequately, the International Federation of Gynecology and Obstetrics (FIGO) recently developed and published a new classification system for ovulatory disorders.5 This new system was designed using a meticulously followed Delphi process with inputs from a diverse group of national and international professional organizations, subspecialty societies, specialty journals, recognized experts in the field, and lay individuals interested in the subject matter.
Of note, FIGO had previously published classification systems for nongestational normal and abnormal uterine bleeding in the reproductive years (FIGO AUB System 1),as well as a subsequent classification system that described potential causes of AUB symptoms (FIGO AUB System 2), with the 9 categories arranged under the acronym PALM-COEIN (Polyp, Adenomyosis, Leiomyoma, Malignancy–Coagulopathy, Ovulatory dysfunction, Endometrial disorders, Iatrogenic, and Not otherwise classified). This new FIGO classification of ovulatory disorders can be viewed as a continuation of the previous initiatives and aims to further categorize the subgroup of AUB-O (AUB with ovulatory disorders). However, it is important to recognize that while most ovulatory disorders manifest with the symptoms of AUB, the absence of AUB symptoms does not necessarily preclude ovulatory disorders.
New system uses a 3-tier approach
The new FIGO classification system for ovulatory disorders has adopted a 3-tier system.
The first tier is based on the anatomic components of the hypothalamic-pituitary-ovarian (HPO) axis and is referred to with the acronym HyPO, for Hypothalamic-Pituitary-Ovarian. Recognizing that PCOS refers to a distinct spectrum of conditions that share a variable combination of signs and symptoms caused to varying degrees by different pathophysiologic mechanisms that involve inherent ovarian follicular dysfunction, neuroendocrine dysfunction, insulin resistance, and androgen excess, it is categorized in a separate class of its own in the first tier, referred to with the letter P.
Adding PCOS to the anatomical categories referred to by HyPO, the first tier is overall referred to with the acronym HyPO-P (FIGURE 2).5
The second tier of stratification provides further etiologic details for any of the primary 3 anatomic classifications of hypothalamic, pituitary, and ovarian. These etiologies are arranged in 10 distinct groups under the mnemonic GAIN-FIT-PIE, which stands for Genetic, Autoimmune, Iatrogenic, Neoplasm; Functional, Infectious/inflammatory, Trauma and vascular; and Physiological, Idiopathic, Endocrine.
The third tier of the system refers to the specific clinical diagnosis. For example, an individual with Kallmann syndrome would be categorized as having type I (hypothalamic), Genetic, Kallmann syndrome, and an individual with PCOS would be categorized simply as having type IV, PCOS.
Our understanding of the etiology of ovulatory disorders has substantially increased over the past several decades. This progress has prompted the need to develop a more comprehensive classification system for these disorders. FIGO recently published a 3-tier classification system for ovulatory disorders that can be remembered with 2 mnemonics: HyPO-P and GAIN-FIT-PIE.
It is hoped that widespread adoption of this new classification system results in better and more concise communication between clinicians, researchers, and patients, ultimately leading to continued improvement in our understanding of the pathophysiology and management of ovulatory disorders.
Continue to: Live birth rate with conventional IVF shown noninferior to that with PGT-A...
Live birth rate with conventional IVF shown noninferior to that with PGT-A
Yan J, Qin Y, Zhao H, et al. Live birth with or without preimplantation genetic testing for aneuploidy. N Engl J Med. 2021;385:2047-2058.
Preimplantation genetic testing for aneuploidy (PGT-A) is increasingly used in many in vitro fertilization (IVF) cycles in the United States. Based on data from the Centers for Disease Control and Prevention, 43.8% of embryo transfers in the United States in 2019 included at least 1 PGT-A–tested embryo.6 Despite this widespread use, however, there are still no robust clinical data for PGT-A’s efficacy and safety, and the guidelines published by the ASRM do not recommend its routine use in all IVF cycles.7 In the past 2 to 3 years, several large studies have raised questions about the reported benefit of this technology.8,9
Details of the trial
In a multicenter, controlled, noninferiority trial conducted by Yan and colleagues, 1,212 subfertile women were randomly assigned to either conventional IVF with embryo selection based on morphology or embryo selection based on PGT-A with next-generation sequencing. Inclusion criteria were the diagnosis of subfertility, undergoing their first IVF cycle, female age of 20 to 37, and the availability of 3 or more good-quality blastocysts.
On day 5 of embryo culture, patients with 3 or more blastocysts were randomly assigned in a 1:1 ratio to either the PGT-A group or conventional IVF. All embryos were then frozen, and patients subsequently underwent frozen embryo transfer of a single blastocyst, selected based on either morphology or euploid result by PGT-A. If the initial transfer did not result in a live birth, and there were remaining transferable embryos (either a euploid embryo in the PGT-A group or a morphologically transferable embryo in the conventional IVF group), patients underwent successive frozen embryo transfers until either there was a live birth or no more embryos were available for transfer.
The study’s primary outcome was the cumulative live birth rate per randomly assigned patient that resulted from up to 3 frozen embryo transfer cycles within 1 year. There were 606 patients randomly assigned to the PGT-A group and 606 randomly assigned to the conventional IVF group.
In the PGT-A group, 468 women (77.2%) had live births; in the conventional IVF group, 496 women (81.8%) had live births. Women in the PGT-A group had a lower incidence of pregnancy loss compared with the conventional IVF group: 8.7% versus 12.6% (absolute difference of -3.9%; 95% confidence interval [CI], -7.5 to -0.2). There was no difference in obstetric and neonatal outcomes between the 2 groups. The authors concluded that among women with 3 or more good-quality blastocysts, conventional IVF resulted in a cumulative live birth rate that was noninferior to that of the PGT-A group.
Some benefit shown with PGT-A
Although the study by Yan and colleagues did not show any benefit, and even a possible reduction, with regard to cumulative live birth rate for PGT-A, it did show a 4% reduction in clinical pregnancy loss when PGT-A was used. Furthermore, the study design has been criticized for performing PGT-A on only 3 blastocysts in the PGT-A group. It is quite conceivable that the PGT-A group would have had more euploid embryos available for transfer if the study design had included all the available embryos instead of only 3. On the other hand, one could argue that if the authors had extended the study to include all the available embryos, the conventional group would have also had more embryos for transfer and, therefore, more chances for pregnancy and live birth.
It is also important to recognize that only patients who had at least 3 embryos available for biopsy were included in this study, and therefore the results of this study cannot be extended to patients with fewer embryos, such as those with diminished ovarian reserve.
In summary, based on this study’s results, we may conclude that for the good-prognosis patients in the age group of 20 to 37 who have at least 3 embryos available for biopsy, PGT-A may reduce the miscarriage rate by about 4%, but this benefit comes at the expense of about a 4% reduction in the cumulative live birth rate. ●
Despite the lack of robust evidence for efficacy, safety, and cost-effectiveness, PGT-A has been widely adopted into clinical IVF practice in the United States over the past several years. A large randomized controlled trial has suggested that, compared with conventional IVF, PGT-A application may actually result in a slightly lower cumulative live birth rate, while the miscarriage rate may be slightly higher with conventional IVF.
PGT-A is a novel and evolving technology with the potential to improve embryo selection in IVF; however, at this juncture, there is not enough clinical data for its universal and routine use in all IVF cycles. PGT-A can potentially be more helpful in older women (>38–40) with good ovarian reserve who are likely to have a larger cohort of embryos to select from. Patients must clearly understand this technology’s pros and cons before agreeing to incorporate it into their care plan.
- Vollset SE, Goren E, Yuan C-W, et al. Fertility, mortality, migration, and population scenarios for 195 countries and territories from 2017 to 2100: a forecasting analysis for the Global Burden of Disease Study. Lancet. 2020;396:1285-1306.
- Dao TH, Docquier F, Maurel M, et al. Global migration in the twentieth and twenty-first centuries: the unstoppable force of demography. Rev World Econ. 2021;157:417-449.
- Atlas of fertility treatment policies in Europe. December 2021. Fertility Europe. Accessed December 29, 2022. https:// fertilityeurope.eu/atlas/#:~:text=Fertility%20Europe%20 in%20conjunction%20with%20the%20European%20 Parliamentary,The%20Atlas%20describes%20the%20 current%20situation%20in%202021
- AMWA’s physician fertility initiative. June 2021. American Medical Women’s Association. Accessed December 29, 2022. https://www.amwa-doc.org/our-work/initiatives/physician -infertility/
- Munro MG, Balen AH, Cho S, et al; FIGO Committee on Menstrual Disorders and Related Health Impacts, and FIGO Committee on Reproductive Medicine, Endocrinology, and Infertility. The FIGO ovulatory disorders classification system. Fertil Steril. 2022;118:768-786.
- Centers for Disease Control and Prevention. 2019 Assisted Reproductive Technology Fertility Clinic and National Summary Report. US Dept of Health and Human Services; 2021. Accessed February 24, 2023. https://www.cdc.gov/art /reports/2019/fertility-clinic.html
- Practice Committees of the American Society for Reproductive Medicine and the Society for Assisted Reproductive Technology. The use of preimplantation genetic testing for aneuploidy (PGT-A): a committee opinion. Fertil Steril. 2018;109:429-436.
- Yan J, Qin Y, Zhao H, et al. Live birth with or without preimplantation genetic testing for aneuploidy. N Engl J Med. 2021;385:2047-2058.
- Kucherov A, Fazzari M, Lieman H, et al. PGT-A is associated with reduced cumulative live birth rate in first reported IVF stimulation cycles age ≤ 40: an analysis of 133,494 autologous cycles reported to SART CORS. J Assist Reprod Genet. 2023;40:137-149.
Total fertility rate and fertility care: Demographic shifts and changing demands
Vollset SE, Goren E, Yuan C-W, et al. Fertility, mortality, migration, and population scenarios for 195 countries and territories from 2017 to 2100: a forecasting analysis for the Global Burden of Disease Study. Lancet. 2020;396:1285-1306.
The total fertility rate (TFR) globally is decreasing rapidly, and in the United States it is now 1.8 births per woman, well below the required replacement rate of 2.1 that maintains the population.1 These reduced TFRs result in significant demographic shifts that affect the economy, workforce, society, health care needs, environment, and geopolitical standing of every country. These changes also will shift demands for the volume and type of services delivered by women’s health care clinicians.
In addition to the TFR, mortality rates and migration rates play essential roles in determining a country’s population.2 Anticipation and planning for these population and health care service changes by each country’s government, business, professionals, and other stakeholders are imperative to manage their impact and optimize quality of life.

US standings in projected population and economic growth
The US population is predicted to peak at 364 million in 2062 and decrease to 336 million in 2100, at which time it will be the fourth largest country in the world, according to a forecasting analysis by Vollset and colleagues.1 China is expected to become the biggest economy in the world in 2035, but this is predicted to change because of its decreasing population so that by 2098 the United States will again be the country with the largest economy (FIGURE 1).1

For the United States to maintain its economic and geopolitical standing, it is important to have policies that promote families. Other countries, especially in northern Europe, have implemented such policies. These include education of the population,economic incentives to create families, extended day care, and favorable tax policies.3 They also include increased access to family-forming fertility care. Such policies in Denmark have resulted in approximately 10% of all children being born from assisted reproductive technology (ART), compared with about 1.5% in the United States. Other countries have similar policies and success in increasing the number of children born from ART.
In the United States, the American Society for Reproductive Medicine (ASRM), RESOLVE: the National Infertility Association, the American Medical Women’s Association (AMWA), and others are promoting the need for increased access to fertility care and family-forming resources, primarily through family-forming benefits provided by companies.4 Such benefits are critical since the primary reason most people do not undergo fertility care is a lack of affordability. Only 1 person in 4 in the United States who needs fertility care receives treatment. Increased access would result in more babies being born to help address the reduced TFR.
Educational access, contraceptive goals, and access to fertility care
Continued trends in women’s educational attainment and access to contraception will hasten declines in the fertility rate and slow population growth (TABLE).1 These educational and contraceptive goals also must be pursued so that every person can achieve their individual reproductive life goals of having a family if and when they want to have a family. In addition to helping address the decreasing TFR, there is a fundamental right to found a family, as stated in the United Nations charter. It is a matter of social justice and equity that everyone who wants to have a family can access reproductive care on a nondiscriminatory basis when needed.

While the need for more and better insurance coverage for infertility has been well documented for many years, the decreasing TFR in the United States is an additional compelling reason that government, business, and other stakeholders should continue to increase access to fertility benefits and care. Women’s health care clinicians are encouraged to support these initiatives that also improve quality of life, equity, and social justice.
The decreasing global and US total fertility rate causes significant demographic changes, with major socioeconomic and health care consequences. The reduced TFR impacts women’s health care services, including the need for increased access to fertility care. Government and corporate policies, including those that improve access to fertility care, will help society adapt to these changes.
Continue to: A new comprehensive ovulatory disorders classification system developed by FIGO...
A new comprehensive ovulatory disorders classification system developed by FIGO
Munro MG, Balen AH, Cho S, et al; FIGO Committee on Menstrual Disorders and Related Health Impacts, and FIGO Committee on Reproductive Medicine, Endocrinology, and Infertility. The FIGO ovulatory disorders classification system. Fertil Steril. 2022;118:768-786.
Ovulatory disorders are well-recognized and common causes of infertility and abnormal uterine bleeding (AUB). Ovulatory disorders occur on a spectrum, with the most severe form being anovulation, and comprise a heterogeneous group that has been classically categorized based on an initial monograph published by the World Health Organization (WHO) in 1973. That classification was based on gonadotropin levels and categorized these disorders into 3 groups: 1) hypogonadotropic (such as hypothalamic amenorrhea), 2) eugonadotropic (such as polycystic ovary syndrome [PCOS]), and 3) hypergonadotropic (such as primary ovarian insufficiency). This initial classification was the subject of several subsequent iterations and modifications over the past 50 years; for example, at one point, ovulatory disorder caused by hyperprolactinemia was added as a separate fourth category. However, due to advances in endocrine assays, imaging technology, and genetics, our understanding of ovulatory disorders has expanded remarkably over the past several decades.
Previous FIGO classifications
Considering the emergent complexity of these disorders and the limitations of the original WHO classification to capture these subtleties adequately, the International Federation of Gynecology and Obstetrics (FIGO) recently developed and published a new classification system for ovulatory disorders.5 This new system was designed using a meticulously followed Delphi process with inputs from a diverse group of national and international professional organizations, subspecialty societies, specialty journals, recognized experts in the field, and lay individuals interested in the subject matter.
Of note, FIGO had previously published classification systems for nongestational normal and abnormal uterine bleeding in the reproductive years (FIGO AUB System 1),as well as a subsequent classification system that described potential causes of AUB symptoms (FIGO AUB System 2), with the 9 categories arranged under the acronym PALM-COEIN (Polyp, Adenomyosis, Leiomyoma, Malignancy–Coagulopathy, Ovulatory dysfunction, Endometrial disorders, Iatrogenic, and Not otherwise classified). This new FIGO classification of ovulatory disorders can be viewed as a continuation of the previous initiatives and aims to further categorize the subgroup of AUB-O (AUB with ovulatory disorders). However, it is important to recognize that while most ovulatory disorders manifest with the symptoms of AUB, the absence of AUB symptoms does not necessarily preclude ovulatory disorders.
New system uses a 3-tier approach
The new FIGO classification system for ovulatory disorders has adopted a 3-tier system.
The first tier is based on the anatomic components of the hypothalamic-pituitary-ovarian (HPO) axis and is referred to with the acronym HyPO, for Hypothalamic-Pituitary-Ovarian. Recognizing that PCOS refers to a distinct spectrum of conditions that share a variable combination of signs and symptoms caused to varying degrees by different pathophysiologic mechanisms that involve inherent ovarian follicular dysfunction, neuroendocrine dysfunction, insulin resistance, and androgen excess, it is categorized in a separate class of its own in the first tier, referred to with the letter P.
Adding PCOS to the anatomical categories referred to by HyPO, the first tier is overall referred to with the acronym HyPO-P (FIGURE 2).5
The second tier of stratification provides further etiologic details for any of the primary 3 anatomic classifications of hypothalamic, pituitary, and ovarian. These etiologies are arranged in 10 distinct groups under the mnemonic GAIN-FIT-PIE, which stands for Genetic, Autoimmune, Iatrogenic, Neoplasm; Functional, Infectious/inflammatory, Trauma and vascular; and Physiological, Idiopathic, Endocrine.
The third tier of the system refers to the specific clinical diagnosis. For example, an individual with Kallmann syndrome would be categorized as having type I (hypothalamic), Genetic, Kallmann syndrome, and an individual with PCOS would be categorized simply as having type IV, PCOS.
Our understanding of the etiology of ovulatory disorders has substantially increased over the past several decades. This progress has prompted the need to develop a more comprehensive classification system for these disorders. FIGO recently published a 3-tier classification system for ovulatory disorders that can be remembered with 2 mnemonics: HyPO-P and GAIN-FIT-PIE.
It is hoped that widespread adoption of this new classification system results in better and more concise communication between clinicians, researchers, and patients, ultimately leading to continued improvement in our understanding of the pathophysiology and management of ovulatory disorders.
Continue to: Live birth rate with conventional IVF shown noninferior to that with PGT-A...
Live birth rate with conventional IVF shown noninferior to that with PGT-A
Yan J, Qin Y, Zhao H, et al. Live birth with or without preimplantation genetic testing for aneuploidy. N Engl J Med. 2021;385:2047-2058.
Preimplantation genetic testing for aneuploidy (PGT-A) is increasingly used in many in vitro fertilization (IVF) cycles in the United States. Based on data from the Centers for Disease Control and Prevention, 43.8% of embryo transfers in the United States in 2019 included at least 1 PGT-A–tested embryo.6 Despite this widespread use, however, there are still no robust clinical data for PGT-A’s efficacy and safety, and the guidelines published by the ASRM do not recommend its routine use in all IVF cycles.7 In the past 2 to 3 years, several large studies have raised questions about the reported benefit of this technology.8,9
Details of the trial
In a multicenter, controlled, noninferiority trial conducted by Yan and colleagues, 1,212 subfertile women were randomly assigned to either conventional IVF with embryo selection based on morphology or embryo selection based on PGT-A with next-generation sequencing. Inclusion criteria were the diagnosis of subfertility, undergoing their first IVF cycle, female age of 20 to 37, and the availability of 3 or more good-quality blastocysts.
On day 5 of embryo culture, patients with 3 or more blastocysts were randomly assigned in a 1:1 ratio to either the PGT-A group or conventional IVF. All embryos were then frozen, and patients subsequently underwent frozen embryo transfer of a single blastocyst, selected based on either morphology or euploid result by PGT-A. If the initial transfer did not result in a live birth, and there were remaining transferable embryos (either a euploid embryo in the PGT-A group or a morphologically transferable embryo in the conventional IVF group), patients underwent successive frozen embryo transfers until either there was a live birth or no more embryos were available for transfer.
The study’s primary outcome was the cumulative live birth rate per randomly assigned patient that resulted from up to 3 frozen embryo transfer cycles within 1 year. There were 606 patients randomly assigned to the PGT-A group and 606 randomly assigned to the conventional IVF group.
In the PGT-A group, 468 women (77.2%) had live births; in the conventional IVF group, 496 women (81.8%) had live births. Women in the PGT-A group had a lower incidence of pregnancy loss compared with the conventional IVF group: 8.7% versus 12.6% (absolute difference of -3.9%; 95% confidence interval [CI], -7.5 to -0.2). There was no difference in obstetric and neonatal outcomes between the 2 groups. The authors concluded that among women with 3 or more good-quality blastocysts, conventional IVF resulted in a cumulative live birth rate that was noninferior to that of the PGT-A group.
Some benefit shown with PGT-A
Although the study by Yan and colleagues did not show any benefit, and even a possible reduction, with regard to cumulative live birth rate for PGT-A, it did show a 4% reduction in clinical pregnancy loss when PGT-A was used. Furthermore, the study design has been criticized for performing PGT-A on only 3 blastocysts in the PGT-A group. It is quite conceivable that the PGT-A group would have had more euploid embryos available for transfer if the study design had included all the available embryos instead of only 3. On the other hand, one could argue that if the authors had extended the study to include all the available embryos, the conventional group would have also had more embryos for transfer and, therefore, more chances for pregnancy and live birth.
It is also important to recognize that only patients who had at least 3 embryos available for biopsy were included in this study, and therefore the results of this study cannot be extended to patients with fewer embryos, such as those with diminished ovarian reserve.
In summary, based on this study’s results, we may conclude that for the good-prognosis patients in the age group of 20 to 37 who have at least 3 embryos available for biopsy, PGT-A may reduce the miscarriage rate by about 4%, but this benefit comes at the expense of about a 4% reduction in the cumulative live birth rate. ●
Despite the lack of robust evidence for efficacy, safety, and cost-effectiveness, PGT-A has been widely adopted into clinical IVF practice in the United States over the past several years. A large randomized controlled trial has suggested that, compared with conventional IVF, PGT-A application may actually result in a slightly lower cumulative live birth rate, while the miscarriage rate may be slightly higher with conventional IVF.
PGT-A is a novel and evolving technology with the potential to improve embryo selection in IVF; however, at this juncture, there is not enough clinical data for its universal and routine use in all IVF cycles. PGT-A can potentially be more helpful in older women (>38–40) with good ovarian reserve who are likely to have a larger cohort of embryos to select from. Patients must clearly understand this technology’s pros and cons before agreeing to incorporate it into their care plan.
Total fertility rate and fertility care: Demographic shifts and changing demands
Vollset SE, Goren E, Yuan C-W, et al. Fertility, mortality, migration, and population scenarios for 195 countries and territories from 2017 to 2100: a forecasting analysis for the Global Burden of Disease Study. Lancet. 2020;396:1285-1306.
The total fertility rate (TFR) globally is decreasing rapidly, and in the United States it is now 1.8 births per woman, well below the required replacement rate of 2.1 that maintains the population.1 These reduced TFRs result in significant demographic shifts that affect the economy, workforce, society, health care needs, environment, and geopolitical standing of every country. These changes also will shift demands for the volume and type of services delivered by women’s health care clinicians.
In addition to the TFR, mortality rates and migration rates play essential roles in determining a country’s population.2 Anticipation and planning for these population and health care service changes by each country’s government, business, professionals, and other stakeholders are imperative to manage their impact and optimize quality of life.

US standings in projected population and economic growth
The US population is predicted to peak at 364 million in 2062 and decrease to 336 million in 2100, at which time it will be the fourth largest country in the world, according to a forecasting analysis by Vollset and colleagues.1 China is expected to become the biggest economy in the world in 2035, but this is predicted to change because of its decreasing population so that by 2098 the United States will again be the country with the largest economy (FIGURE 1).1

For the United States to maintain its economic and geopolitical standing, it is important to have policies that promote families. Other countries, especially in northern Europe, have implemented such policies. These include education of the population,economic incentives to create families, extended day care, and favorable tax policies.3 They also include increased access to family-forming fertility care. Such policies in Denmark have resulted in approximately 10% of all children being born from assisted reproductive technology (ART), compared with about 1.5% in the United States. Other countries have similar policies and success in increasing the number of children born from ART.
In the United States, the American Society for Reproductive Medicine (ASRM), RESOLVE: the National Infertility Association, the American Medical Women’s Association (AMWA), and others are promoting the need for increased access to fertility care and family-forming resources, primarily through family-forming benefits provided by companies.4 Such benefits are critical since the primary reason most people do not undergo fertility care is a lack of affordability. Only 1 person in 4 in the United States who needs fertility care receives treatment. Increased access would result in more babies being born to help address the reduced TFR.
Educational access, contraceptive goals, and access to fertility care
Continued trends in women’s educational attainment and access to contraception will hasten declines in the fertility rate and slow population growth (TABLE).1 These educational and contraceptive goals also must be pursued so that every person can achieve their individual reproductive life goals of having a family if and when they want to have a family. In addition to helping address the decreasing TFR, there is a fundamental right to found a family, as stated in the United Nations charter. It is a matter of social justice and equity that everyone who wants to have a family can access reproductive care on a nondiscriminatory basis when needed.

While the need for more and better insurance coverage for infertility has been well documented for many years, the decreasing TFR in the United States is an additional compelling reason that government, business, and other stakeholders should continue to increase access to fertility benefits and care. Women’s health care clinicians are encouraged to support these initiatives that also improve quality of life, equity, and social justice.
The decreasing global and US total fertility rate causes significant demographic changes, with major socioeconomic and health care consequences. The reduced TFR impacts women’s health care services, including the need for increased access to fertility care. Government and corporate policies, including those that improve access to fertility care, will help society adapt to these changes.
Continue to: A new comprehensive ovulatory disorders classification system developed by FIGO...
A new comprehensive ovulatory disorders classification system developed by FIGO
Munro MG, Balen AH, Cho S, et al; FIGO Committee on Menstrual Disorders and Related Health Impacts, and FIGO Committee on Reproductive Medicine, Endocrinology, and Infertility. The FIGO ovulatory disorders classification system. Fertil Steril. 2022;118:768-786.
Ovulatory disorders are well-recognized and common causes of infertility and abnormal uterine bleeding (AUB). Ovulatory disorders occur on a spectrum, with the most severe form being anovulation, and comprise a heterogeneous group that has been classically categorized based on an initial monograph published by the World Health Organization (WHO) in 1973. That classification was based on gonadotropin levels and categorized these disorders into 3 groups: 1) hypogonadotropic (such as hypothalamic amenorrhea), 2) eugonadotropic (such as polycystic ovary syndrome [PCOS]), and 3) hypergonadotropic (such as primary ovarian insufficiency). This initial classification was the subject of several subsequent iterations and modifications over the past 50 years; for example, at one point, ovulatory disorder caused by hyperprolactinemia was added as a separate fourth category. However, due to advances in endocrine assays, imaging technology, and genetics, our understanding of ovulatory disorders has expanded remarkably over the past several decades.
Previous FIGO classifications
Considering the emergent complexity of these disorders and the limitations of the original WHO classification to capture these subtleties adequately, the International Federation of Gynecology and Obstetrics (FIGO) recently developed and published a new classification system for ovulatory disorders.5 This new system was designed using a meticulously followed Delphi process with inputs from a diverse group of national and international professional organizations, subspecialty societies, specialty journals, recognized experts in the field, and lay individuals interested in the subject matter.
Of note, FIGO had previously published classification systems for nongestational normal and abnormal uterine bleeding in the reproductive years (FIGO AUB System 1),as well as a subsequent classification system that described potential causes of AUB symptoms (FIGO AUB System 2), with the 9 categories arranged under the acronym PALM-COEIN (Polyp, Adenomyosis, Leiomyoma, Malignancy–Coagulopathy, Ovulatory dysfunction, Endometrial disorders, Iatrogenic, and Not otherwise classified). This new FIGO classification of ovulatory disorders can be viewed as a continuation of the previous initiatives and aims to further categorize the subgroup of AUB-O (AUB with ovulatory disorders). However, it is important to recognize that while most ovulatory disorders manifest with the symptoms of AUB, the absence of AUB symptoms does not necessarily preclude ovulatory disorders.
New system uses a 3-tier approach
The new FIGO classification system for ovulatory disorders has adopted a 3-tier system.
The first tier is based on the anatomic components of the hypothalamic-pituitary-ovarian (HPO) axis and is referred to with the acronym HyPO, for Hypothalamic-Pituitary-Ovarian. Recognizing that PCOS refers to a distinct spectrum of conditions that share a variable combination of signs and symptoms caused to varying degrees by different pathophysiologic mechanisms that involve inherent ovarian follicular dysfunction, neuroendocrine dysfunction, insulin resistance, and androgen excess, it is categorized in a separate class of its own in the first tier, referred to with the letter P.
Adding PCOS to the anatomical categories referred to by HyPO, the first tier is overall referred to with the acronym HyPO-P (FIGURE 2).5
The second tier of stratification provides further etiologic details for any of the primary 3 anatomic classifications of hypothalamic, pituitary, and ovarian. These etiologies are arranged in 10 distinct groups under the mnemonic GAIN-FIT-PIE, which stands for Genetic, Autoimmune, Iatrogenic, Neoplasm; Functional, Infectious/inflammatory, Trauma and vascular; and Physiological, Idiopathic, Endocrine.
The third tier of the system refers to the specific clinical diagnosis. For example, an individual with Kallmann syndrome would be categorized as having type I (hypothalamic), Genetic, Kallmann syndrome, and an individual with PCOS would be categorized simply as having type IV, PCOS.
Our understanding of the etiology of ovulatory disorders has substantially increased over the past several decades. This progress has prompted the need to develop a more comprehensive classification system for these disorders. FIGO recently published a 3-tier classification system for ovulatory disorders that can be remembered with 2 mnemonics: HyPO-P and GAIN-FIT-PIE.
It is hoped that widespread adoption of this new classification system results in better and more concise communication between clinicians, researchers, and patients, ultimately leading to continued improvement in our understanding of the pathophysiology and management of ovulatory disorders.
Continue to: Live birth rate with conventional IVF shown noninferior to that with PGT-A...
Live birth rate with conventional IVF shown noninferior to that with PGT-A
Yan J, Qin Y, Zhao H, et al. Live birth with or without preimplantation genetic testing for aneuploidy. N Engl J Med. 2021;385:2047-2058.
Preimplantation genetic testing for aneuploidy (PGT-A) is increasingly used in many in vitro fertilization (IVF) cycles in the United States. Based on data from the Centers for Disease Control and Prevention, 43.8% of embryo transfers in the United States in 2019 included at least 1 PGT-A–tested embryo.6 Despite this widespread use, however, there are still no robust clinical data for PGT-A’s efficacy and safety, and the guidelines published by the ASRM do not recommend its routine use in all IVF cycles.7 In the past 2 to 3 years, several large studies have raised questions about the reported benefit of this technology.8,9
Details of the trial
In a multicenter, controlled, noninferiority trial conducted by Yan and colleagues, 1,212 subfertile women were randomly assigned to either conventional IVF with embryo selection based on morphology or embryo selection based on PGT-A with next-generation sequencing. Inclusion criteria were the diagnosis of subfertility, undergoing their first IVF cycle, female age of 20 to 37, and the availability of 3 or more good-quality blastocysts.
On day 5 of embryo culture, patients with 3 or more blastocysts were randomly assigned in a 1:1 ratio to either the PGT-A group or conventional IVF. All embryos were then frozen, and patients subsequently underwent frozen embryo transfer of a single blastocyst, selected based on either morphology or euploid result by PGT-A. If the initial transfer did not result in a live birth, and there were remaining transferable embryos (either a euploid embryo in the PGT-A group or a morphologically transferable embryo in the conventional IVF group), patients underwent successive frozen embryo transfers until either there was a live birth or no more embryos were available for transfer.
The study’s primary outcome was the cumulative live birth rate per randomly assigned patient that resulted from up to 3 frozen embryo transfer cycles within 1 year. There were 606 patients randomly assigned to the PGT-A group and 606 randomly assigned to the conventional IVF group.
In the PGT-A group, 468 women (77.2%) had live births; in the conventional IVF group, 496 women (81.8%) had live births. Women in the PGT-A group had a lower incidence of pregnancy loss compared with the conventional IVF group: 8.7% versus 12.6% (absolute difference of -3.9%; 95% confidence interval [CI], -7.5 to -0.2). There was no difference in obstetric and neonatal outcomes between the 2 groups. The authors concluded that among women with 3 or more good-quality blastocysts, conventional IVF resulted in a cumulative live birth rate that was noninferior to that of the PGT-A group.
Some benefit shown with PGT-A
Although the study by Yan and colleagues did not show any benefit, and even a possible reduction, with regard to cumulative live birth rate for PGT-A, it did show a 4% reduction in clinical pregnancy loss when PGT-A was used. Furthermore, the study design has been criticized for performing PGT-A on only 3 blastocysts in the PGT-A group. It is quite conceivable that the PGT-A group would have had more euploid embryos available for transfer if the study design had included all the available embryos instead of only 3. On the other hand, one could argue that if the authors had extended the study to include all the available embryos, the conventional group would have also had more embryos for transfer and, therefore, more chances for pregnancy and live birth.
It is also important to recognize that only patients who had at least 3 embryos available for biopsy were included in this study, and therefore the results of this study cannot be extended to patients with fewer embryos, such as those with diminished ovarian reserve.
In summary, based on this study’s results, we may conclude that for the good-prognosis patients in the age group of 20 to 37 who have at least 3 embryos available for biopsy, PGT-A may reduce the miscarriage rate by about 4%, but this benefit comes at the expense of about a 4% reduction in the cumulative live birth rate. ●
Despite the lack of robust evidence for efficacy, safety, and cost-effectiveness, PGT-A has been widely adopted into clinical IVF practice in the United States over the past several years. A large randomized controlled trial has suggested that, compared with conventional IVF, PGT-A application may actually result in a slightly lower cumulative live birth rate, while the miscarriage rate may be slightly higher with conventional IVF.
PGT-A is a novel and evolving technology with the potential to improve embryo selection in IVF; however, at this juncture, there is not enough clinical data for its universal and routine use in all IVF cycles. PGT-A can potentially be more helpful in older women (>38–40) with good ovarian reserve who are likely to have a larger cohort of embryos to select from. Patients must clearly understand this technology’s pros and cons before agreeing to incorporate it into their care plan.
- Vollset SE, Goren E, Yuan C-W, et al. Fertility, mortality, migration, and population scenarios for 195 countries and territories from 2017 to 2100: a forecasting analysis for the Global Burden of Disease Study. Lancet. 2020;396:1285-1306.
- Dao TH, Docquier F, Maurel M, et al. Global migration in the twentieth and twenty-first centuries: the unstoppable force of demography. Rev World Econ. 2021;157:417-449.
- Atlas of fertility treatment policies in Europe. December 2021. Fertility Europe. Accessed December 29, 2022. https:// fertilityeurope.eu/atlas/#:~:text=Fertility%20Europe%20 in%20conjunction%20with%20the%20European%20 Parliamentary,The%20Atlas%20describes%20the%20 current%20situation%20in%202021
- AMWA’s physician fertility initiative. June 2021. American Medical Women’s Association. Accessed December 29, 2022. https://www.amwa-doc.org/our-work/initiatives/physician -infertility/
- Munro MG, Balen AH, Cho S, et al; FIGO Committee on Menstrual Disorders and Related Health Impacts, and FIGO Committee on Reproductive Medicine, Endocrinology, and Infertility. The FIGO ovulatory disorders classification system. Fertil Steril. 2022;118:768-786.
- Centers for Disease Control and Prevention. 2019 Assisted Reproductive Technology Fertility Clinic and National Summary Report. US Dept of Health and Human Services; 2021. Accessed February 24, 2023. https://www.cdc.gov/art /reports/2019/fertility-clinic.html
- Practice Committees of the American Society for Reproductive Medicine and the Society for Assisted Reproductive Technology. The use of preimplantation genetic testing for aneuploidy (PGT-A): a committee opinion. Fertil Steril. 2018;109:429-436.
- Yan J, Qin Y, Zhao H, et al. Live birth with or without preimplantation genetic testing for aneuploidy. N Engl J Med. 2021;385:2047-2058.
- Kucherov A, Fazzari M, Lieman H, et al. PGT-A is associated with reduced cumulative live birth rate in first reported IVF stimulation cycles age ≤ 40: an analysis of 133,494 autologous cycles reported to SART CORS. J Assist Reprod Genet. 2023;40:137-149.
- Vollset SE, Goren E, Yuan C-W, et al. Fertility, mortality, migration, and population scenarios for 195 countries and territories from 2017 to 2100: a forecasting analysis for the Global Burden of Disease Study. Lancet. 2020;396:1285-1306.
- Dao TH, Docquier F, Maurel M, et al. Global migration in the twentieth and twenty-first centuries: the unstoppable force of demography. Rev World Econ. 2021;157:417-449.
- Atlas of fertility treatment policies in Europe. December 2021. Fertility Europe. Accessed December 29, 2022. https:// fertilityeurope.eu/atlas/#:~:text=Fertility%20Europe%20 in%20conjunction%20with%20the%20European%20 Parliamentary,The%20Atlas%20describes%20the%20 current%20situation%20in%202021
- AMWA’s physician fertility initiative. June 2021. American Medical Women’s Association. Accessed December 29, 2022. https://www.amwa-doc.org/our-work/initiatives/physician -infertility/
- Munro MG, Balen AH, Cho S, et al; FIGO Committee on Menstrual Disorders and Related Health Impacts, and FIGO Committee on Reproductive Medicine, Endocrinology, and Infertility. The FIGO ovulatory disorders classification system. Fertil Steril. 2022;118:768-786.
- Centers for Disease Control and Prevention. 2019 Assisted Reproductive Technology Fertility Clinic and National Summary Report. US Dept of Health and Human Services; 2021. Accessed February 24, 2023. https://www.cdc.gov/art /reports/2019/fertility-clinic.html
- Practice Committees of the American Society for Reproductive Medicine and the Society for Assisted Reproductive Technology. The use of preimplantation genetic testing for aneuploidy (PGT-A): a committee opinion. Fertil Steril. 2018;109:429-436.
- Yan J, Qin Y, Zhao H, et al. Live birth with or without preimplantation genetic testing for aneuploidy. N Engl J Med. 2021;385:2047-2058.
- Kucherov A, Fazzari M, Lieman H, et al. PGT-A is associated with reduced cumulative live birth rate in first reported IVF stimulation cycles age ≤ 40: an analysis of 133,494 autologous cycles reported to SART CORS. J Assist Reprod Genet. 2023;40:137-149.
2022 Update on fertility
In this Update, the authors discuss 2 important areas that impact fertility. First, with in vitro fertilization (IVF), successful implantation that leads to live birth requires a normal embryo and a receptive endometrium. While research using advanced molecular array technology has resulted in a clinical test to identify the optimal window of implantation, recent evidence has questioned its clinical effectiveness. Second, recognizing the importance of endometriosis—a common disease with high burden that causes pain, infertility, and other symptoms—the World Health Organization (WHO) last year published an informative fact sheet that highlights the diagnosis, treatment options, and challenges of this significant disease.
Endometrial receptivity array and the quest for optimal endometrial preparation prior to embryo transfer in IVF
Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
A successful pregnancy requires optimal crosstalk between the embryo and the endometrium. Over the past several decades, research efforts to improve IVF outcomes have been focused mainly on the embryo factor and methods to improve embryo selection, such as extended culture to blastocyst, time-lapse imaging (morphokinetic assessment), and more notably, preimplantation genetic testing for aneuploidy (PGT-A). However, the other half of the equation, the endometrium, has not garnered the attention that it deserves. Effort has therefore been renewed to optimize the endometrial factor by better diagnosing and treating various forms of endometrial dysfunction that could lead to infertility in general and lack of success with IVF and euploid embryo transfers in particular.
Historical background on endometrial function
Progesterone has long been recognized as the main effector that transforms the estrogen-primed endometrium into a receptive state that results in successful embryo implantation. Progesterone exposure is required at appropriate levels and duration before the endometrium becomes receptive to the embryo. If implantation does not occur soon after the endometrium has attained receptive status (7–10 days after ovulation), further progesterone exposure results in progression of endometrial changes that no longer permit successful implantation.
As early as the 1950s, “luteal phase deficiency” was defined as due to inadequate progesterone secretion and resulted in a short luteal phase. In the 1970s, histologic “dating” of the endometrium became the gold standard for diagnosing luteal phase defects; this relied on a classic histologic appearance of secretory phase endometrium and its changes throughout the luteal phase. Subsequently, however, results of prospective randomized controlled trials published in 2004 cast significant doubt on the accuracy and reproducibility of these endometrial biopsies and did not show any clinical diagnostic benefit or correlation with pregnancy outcomes.
21st century advances: Endometrial dating 2.0
A decade later, with the advancement of molecular biology tools such as microarray technology, researchers were able to study endometrial gene expression patterns at different stages of the menstrual cycle. They identified different phases of endometrial development with molecular profiles, or “signatures,” for the luteal phase, endometriosis, polycystic ovary syndrome, and uterine fibroids.
In 2013, researchers in Spain introduced a diagnostic test called endometrial receptivity array (ERA) with the stated goal of being able to temporally define the receptive endometrium and identify prereceptive as well as postreceptive states.1 In other words, instead of the histologic dating of the endometrium used in the 1970s, it represented “molecular dating” of the endometrium. Although the initial studies were conducted among women who experienced prior unsuccessful embryo transfers (the so-called recurrent implantation failure, or RIF), the test’s scope was subsequently expanded to include any individual planning on a frozen embryo transfer (FET), regardless of any prior attempts. The term personalized embryo transfer (pET) was coined to suggest the ability to define the best time (up to hours) for embryo transfers on an individual basis. Despite lack of independent validation studies, ERA was then widely adopted by many clinicians (and requested by some patients) with the hope of improving IVF outcomes.
However, not unlike many other novel innovations in assisted reproductive technology, ERA regrettably did not withstand the test of time. Three independent studies in 2021, 1 randomized clinical trial and 2 observational cohort studies, did not show any benefit with regard to implantation rates, pregnancy rates, or live birth rates when ERA was performed in the general infertility population.2-4
Continue to: Study results...
Study results
The cohort study that matched 133 ERA patients with 353 non-ERA patients showed live birth rates of 49.62% for the ERA group and 54.96% for the non-ERA group (odds ratio [OR], 0.8074; 95% confidence interval [CI], 0.5424–1.2018).2 Of note, no difference occurred between subgroups based on the prior number of FETs or the receptivity status (TABLE 1).
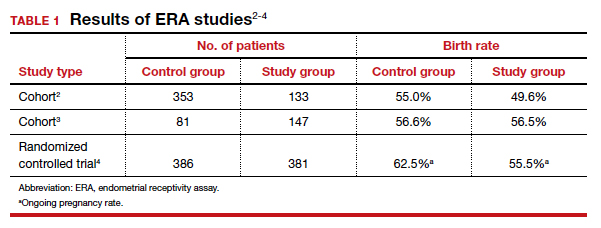
Another cohort study from the University of California, Los Angeles, published in 2021 analyzed 228 single euploid FET cycles.3 This study did not show any benefit for routine ERA testing, with a live birth rate of 56.6% in the non-ERA group and 56.5% in the ERA group.
Still, the most convincing evidence for the lack of benefit from routine ERA was noted from the results of the randomized clinical trial.4 A total of 767 patients were randomly allocated, 381 to the ERA group and 386 to the control group. There was no difference in ongoing pregnancy rates between the 2 groups. Perhaps more important, even after limiting the analysis to individuals with a nonreceptive ERA result, there was no difference in ongoing pregnancy rates between the 2 groups: 62.5% in the control group (default timing of transfer) and 55.5% in the study group (transfer timing adjusted based on ERA) (rate ratio [RR], 0.9; 95% CI, 0.70–1.14).
ERA usefulness is unsupported in general infertility population
The studies discussed collectively suggest with a high degree of certainty that there is no indication for routine ERA testing in the general infertility population prior to frozen embryo transfers.
Although these studies all were conducted in the general infertility population and did not specifically evaluate the performance of ERA in women with recurrent pregnancy loss or recurrent implantation failure, it is important to acknowledge that if ERA were truly able to define the window of receptivity, one would expect a lower implantation rate if the embryos were transferred outside of the window suggested by the ERA. This was not the case in these studies, as they all showed equivalent pregnancy rates in the control (nonadjusted) groups even when ERA suggested a nonreceptive status.
This observation seriously questions the validity of ERA regarding its ability to temporally define the window of receptivity. On the other hand, as stated earlier, there is still a possibility for ERA to be beneficial for a small subgroup of patients whose window of receptivity may not be as wide as expected in the general population. The challenging question would be how best to identify the particular group with a narrow, or displaced, window of receptivity.
The optimal timing for implantation of a normal embryo requires a receptive endometrium. The endometrial biopsy was used widely for many years before research showed it was not clinically useful. More recently, the endometrial receptivity array has been suggested to help time the frozen embryo transfer. Unfortunately, recent studies have shown that this test is not clinically useful for the general infertility population.
Continue to: WHO raises awareness of endometriosis burden and...
WHO raises awareness of endometriosis burden and highlights need to address diagnosis and treatment for women’s reproductive health
World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room /fact-sheets/detail/endometriosis. Accessed January 3, 2022.
The WHO published its first fact sheet on endometriosis in March 2021, recognizing endometriosis as a severe disease that affects almost 190 million women with life-impacting pain, infertility, other symptoms, and especially with chronic, significant emotional sequelae (TABLE 2).5 The disease’s variable and broad symptoms result in a lack of awareness and diagnosis by both women and health care providers, especially in low- and middle-income countries and in disadvantaged populations in developed countries. Increased awareness to promote earlier diagnosis, improved training for better management, expanded research for greater understanding, and policies that increase access to quality care are needed to ensure the reproductive health and rights of tens of millions of women with endometriosis.

Endometriosis characteristics and symptoms
Endometriosis is characterized by the presence of tissue resembling endometrium outside the uterus, where it causes a chronic inflammatory reaction that may result in the formation of scar tissue. Endometriotic lesions may be superficial, cystic ovarian endometriomas, or deep lesions, causing a myriad of pain and related symptoms.6.7
Chronic pain may occur because pain centers in the brain become hyperresponsive over time (central sensitization); this can occur at any point throughout the life course of endometriosis, even when endometriosis lesions are no longer visible. Sometimes, endometriosis is asymptomatic. In addition, endometriosis can cause infertility through anatomic distortion and inflammatory, endocrinologic, and other pathways.
The origins of endometriosis are thought to be multifactorial and include retrograde menstruation, cellular metaplasia, and/or stem cells that spread through blood and lymphatic vessels. Endometriosis is estrogen dependent, but lesion growth also is affected by altered or impaired immunity, localized complex hormonal influences, genetics, and possibly environmental contaminants.
Impact on public health and reproductive rights
Endometriosis has significant social, public health, and economic implications. It can decrease quality of life and prevent girls and women from attending work or school.8 Painful sex can affect sexual health. The WHO states that, “Addressing endometriosis will empower those affected by it, by supporting their human right to the highest standard of sexual and reproductive health, quality of life, and overall well-being.”5
At present, no known way is available to prevent or cure endometriosis. Early diagnosis and treatment, however, may slow or halt its natural progression and associated symptoms.
Diagnostic steps and treatment options
Early suspicion of endometriosis is the most important factor, followed by a careful history of menstrual symptoms and chronic pelvic pain, early referral to specialists for ultrasonography or other imaging, and sometimes surgical or laparoscopic visualization. Empirical treatment can be begun without histologic or laparoscopic confirmation.
Endometriosis can be treated with medications and/or surgery depending on symptoms, lesions, desired outcome, and patient choice.5,6 Common therapies include contraceptive steroids, nonsteroidal anti-inflammatory medications, and analgesics. Medical treatments focus on either lowering estrogen or increasing progesterone levels.
Surgery can remove endometriosis lesions, adhesions, and scar tissue. However, success in reducing pain symptoms and increasing pregnancy rates often depends on the extent of disease.
For infertility due to endometriosis, treatment options include laparoscopic surgical removal of endometriosis, ovarian stimulation with intrauterine insemination (IUI), and IVF. Multidisciplinary treatment addressing different symptoms and overall health often requires referral to pain experts and other specialists.9
The WHO perspective on endometriosis
Recognizing the importance of endometriosis and its impact on people’s sexual and reproductive health, quality of life, and overall well-being, the WHO is taking action to improve awareness, diagnosis, and treatment of endometriosis (TABLE 3).5 ●

Endometriosis is now recognized as a disease with significant burden for women everywhere. Widespread lack of awareness of presenting symptoms and management options means that all women’s health care clinicians need to become better informed about endometriosis so they can improve the quality of care they provide.
- Ruiz-Alonso M, Blesa D, Díaz-Gimeno P, et al. The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertil Steril. 2013;100:818-824.
- Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
- Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
- Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
- World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room/fact-sheets/detail /endometriosis. Accessed January 3, 2022.
- Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382:1244-1256.
- Johnson NP, Hummelshoj L, Adamson GD, et al. World Endometriosis Society consensus on the classification of endometriosis. Hum Reprod. 2017;32:315-324.
- Nnoaham K, Hummelshoj L, Webster P, et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil Steril. 2011;96:366-373.e8.
- Carey ET, Till SR, As-Sanie S. Pharmacological management of chronic pelvic pain in women. Drugs. 2017;77:285-301.
In this Update, the authors discuss 2 important areas that impact fertility. First, with in vitro fertilization (IVF), successful implantation that leads to live birth requires a normal embryo and a receptive endometrium. While research using advanced molecular array technology has resulted in a clinical test to identify the optimal window of implantation, recent evidence has questioned its clinical effectiveness. Second, recognizing the importance of endometriosis—a common disease with high burden that causes pain, infertility, and other symptoms—the World Health Organization (WHO) last year published an informative fact sheet that highlights the diagnosis, treatment options, and challenges of this significant disease.
Endometrial receptivity array and the quest for optimal endometrial preparation prior to embryo transfer in IVF
Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
A successful pregnancy requires optimal crosstalk between the embryo and the endometrium. Over the past several decades, research efforts to improve IVF outcomes have been focused mainly on the embryo factor and methods to improve embryo selection, such as extended culture to blastocyst, time-lapse imaging (morphokinetic assessment), and more notably, preimplantation genetic testing for aneuploidy (PGT-A). However, the other half of the equation, the endometrium, has not garnered the attention that it deserves. Effort has therefore been renewed to optimize the endometrial factor by better diagnosing and treating various forms of endometrial dysfunction that could lead to infertility in general and lack of success with IVF and euploid embryo transfers in particular.
Historical background on endometrial function
Progesterone has long been recognized as the main effector that transforms the estrogen-primed endometrium into a receptive state that results in successful embryo implantation. Progesterone exposure is required at appropriate levels and duration before the endometrium becomes receptive to the embryo. If implantation does not occur soon after the endometrium has attained receptive status (7–10 days after ovulation), further progesterone exposure results in progression of endometrial changes that no longer permit successful implantation.
As early as the 1950s, “luteal phase deficiency” was defined as due to inadequate progesterone secretion and resulted in a short luteal phase. In the 1970s, histologic “dating” of the endometrium became the gold standard for diagnosing luteal phase defects; this relied on a classic histologic appearance of secretory phase endometrium and its changes throughout the luteal phase. Subsequently, however, results of prospective randomized controlled trials published in 2004 cast significant doubt on the accuracy and reproducibility of these endometrial biopsies and did not show any clinical diagnostic benefit or correlation with pregnancy outcomes.
21st century advances: Endometrial dating 2.0
A decade later, with the advancement of molecular biology tools such as microarray technology, researchers were able to study endometrial gene expression patterns at different stages of the menstrual cycle. They identified different phases of endometrial development with molecular profiles, or “signatures,” for the luteal phase, endometriosis, polycystic ovary syndrome, and uterine fibroids.
In 2013, researchers in Spain introduced a diagnostic test called endometrial receptivity array (ERA) with the stated goal of being able to temporally define the receptive endometrium and identify prereceptive as well as postreceptive states.1 In other words, instead of the histologic dating of the endometrium used in the 1970s, it represented “molecular dating” of the endometrium. Although the initial studies were conducted among women who experienced prior unsuccessful embryo transfers (the so-called recurrent implantation failure, or RIF), the test’s scope was subsequently expanded to include any individual planning on a frozen embryo transfer (FET), regardless of any prior attempts. The term personalized embryo transfer (pET) was coined to suggest the ability to define the best time (up to hours) for embryo transfers on an individual basis. Despite lack of independent validation studies, ERA was then widely adopted by many clinicians (and requested by some patients) with the hope of improving IVF outcomes.
However, not unlike many other novel innovations in assisted reproductive technology, ERA regrettably did not withstand the test of time. Three independent studies in 2021, 1 randomized clinical trial and 2 observational cohort studies, did not show any benefit with regard to implantation rates, pregnancy rates, or live birth rates when ERA was performed in the general infertility population.2-4
Continue to: Study results...
Study results
The cohort study that matched 133 ERA patients with 353 non-ERA patients showed live birth rates of 49.62% for the ERA group and 54.96% for the non-ERA group (odds ratio [OR], 0.8074; 95% confidence interval [CI], 0.5424–1.2018).2 Of note, no difference occurred between subgroups based on the prior number of FETs or the receptivity status (TABLE 1).

Another cohort study from the University of California, Los Angeles, published in 2021 analyzed 228 single euploid FET cycles.3 This study did not show any benefit for routine ERA testing, with a live birth rate of 56.6% in the non-ERA group and 56.5% in the ERA group.
Still, the most convincing evidence for the lack of benefit from routine ERA was noted from the results of the randomized clinical trial.4 A total of 767 patients were randomly allocated, 381 to the ERA group and 386 to the control group. There was no difference in ongoing pregnancy rates between the 2 groups. Perhaps more important, even after limiting the analysis to individuals with a nonreceptive ERA result, there was no difference in ongoing pregnancy rates between the 2 groups: 62.5% in the control group (default timing of transfer) and 55.5% in the study group (transfer timing adjusted based on ERA) (rate ratio [RR], 0.9; 95% CI, 0.70–1.14).
ERA usefulness is unsupported in general infertility population
The studies discussed collectively suggest with a high degree of certainty that there is no indication for routine ERA testing in the general infertility population prior to frozen embryo transfers.
Although these studies all were conducted in the general infertility population and did not specifically evaluate the performance of ERA in women with recurrent pregnancy loss or recurrent implantation failure, it is important to acknowledge that if ERA were truly able to define the window of receptivity, one would expect a lower implantation rate if the embryos were transferred outside of the window suggested by the ERA. This was not the case in these studies, as they all showed equivalent pregnancy rates in the control (nonadjusted) groups even when ERA suggested a nonreceptive status.
This observation seriously questions the validity of ERA regarding its ability to temporally define the window of receptivity. On the other hand, as stated earlier, there is still a possibility for ERA to be beneficial for a small subgroup of patients whose window of receptivity may not be as wide as expected in the general population. The challenging question would be how best to identify the particular group with a narrow, or displaced, window of receptivity.
The optimal timing for implantation of a normal embryo requires a receptive endometrium. The endometrial biopsy was used widely for many years before research showed it was not clinically useful. More recently, the endometrial receptivity array has been suggested to help time the frozen embryo transfer. Unfortunately, recent studies have shown that this test is not clinically useful for the general infertility population.
Continue to: WHO raises awareness of endometriosis burden and...
WHO raises awareness of endometriosis burden and highlights need to address diagnosis and treatment for women’s reproductive health
World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room /fact-sheets/detail/endometriosis. Accessed January 3, 2022.
The WHO published its first fact sheet on endometriosis in March 2021, recognizing endometriosis as a severe disease that affects almost 190 million women with life-impacting pain, infertility, other symptoms, and especially with chronic, significant emotional sequelae (TABLE 2).5 The disease’s variable and broad symptoms result in a lack of awareness and diagnosis by both women and health care providers, especially in low- and middle-income countries and in disadvantaged populations in developed countries. Increased awareness to promote earlier diagnosis, improved training for better management, expanded research for greater understanding, and policies that increase access to quality care are needed to ensure the reproductive health and rights of tens of millions of women with endometriosis.

Endometriosis characteristics and symptoms
Endometriosis is characterized by the presence of tissue resembling endometrium outside the uterus, where it causes a chronic inflammatory reaction that may result in the formation of scar tissue. Endometriotic lesions may be superficial, cystic ovarian endometriomas, or deep lesions, causing a myriad of pain and related symptoms.6.7
Chronic pain may occur because pain centers in the brain become hyperresponsive over time (central sensitization); this can occur at any point throughout the life course of endometriosis, even when endometriosis lesions are no longer visible. Sometimes, endometriosis is asymptomatic. In addition, endometriosis can cause infertility through anatomic distortion and inflammatory, endocrinologic, and other pathways.
The origins of endometriosis are thought to be multifactorial and include retrograde menstruation, cellular metaplasia, and/or stem cells that spread through blood and lymphatic vessels. Endometriosis is estrogen dependent, but lesion growth also is affected by altered or impaired immunity, localized complex hormonal influences, genetics, and possibly environmental contaminants.
Impact on public health and reproductive rights
Endometriosis has significant social, public health, and economic implications. It can decrease quality of life and prevent girls and women from attending work or school.8 Painful sex can affect sexual health. The WHO states that, “Addressing endometriosis will empower those affected by it, by supporting their human right to the highest standard of sexual and reproductive health, quality of life, and overall well-being.”5
At present, no known way is available to prevent or cure endometriosis. Early diagnosis and treatment, however, may slow or halt its natural progression and associated symptoms.
Diagnostic steps and treatment options
Early suspicion of endometriosis is the most important factor, followed by a careful history of menstrual symptoms and chronic pelvic pain, early referral to specialists for ultrasonography or other imaging, and sometimes surgical or laparoscopic visualization. Empirical treatment can be begun without histologic or laparoscopic confirmation.
Endometriosis can be treated with medications and/or surgery depending on symptoms, lesions, desired outcome, and patient choice.5,6 Common therapies include contraceptive steroids, nonsteroidal anti-inflammatory medications, and analgesics. Medical treatments focus on either lowering estrogen or increasing progesterone levels.
Surgery can remove endometriosis lesions, adhesions, and scar tissue. However, success in reducing pain symptoms and increasing pregnancy rates often depends on the extent of disease.
For infertility due to endometriosis, treatment options include laparoscopic surgical removal of endometriosis, ovarian stimulation with intrauterine insemination (IUI), and IVF. Multidisciplinary treatment addressing different symptoms and overall health often requires referral to pain experts and other specialists.9
The WHO perspective on endometriosis
Recognizing the importance of endometriosis and its impact on people’s sexual and reproductive health, quality of life, and overall well-being, the WHO is taking action to improve awareness, diagnosis, and treatment of endometriosis (TABLE 3).5 ●

Endometriosis is now recognized as a disease with significant burden for women everywhere. Widespread lack of awareness of presenting symptoms and management options means that all women’s health care clinicians need to become better informed about endometriosis so they can improve the quality of care they provide.
In this Update, the authors discuss 2 important areas that impact fertility. First, with in vitro fertilization (IVF), successful implantation that leads to live birth requires a normal embryo and a receptive endometrium. While research using advanced molecular array technology has resulted in a clinical test to identify the optimal window of implantation, recent evidence has questioned its clinical effectiveness. Second, recognizing the importance of endometriosis—a common disease with high burden that causes pain, infertility, and other symptoms—the World Health Organization (WHO) last year published an informative fact sheet that highlights the diagnosis, treatment options, and challenges of this significant disease.
Endometrial receptivity array and the quest for optimal endometrial preparation prior to embryo transfer in IVF
Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
A successful pregnancy requires optimal crosstalk between the embryo and the endometrium. Over the past several decades, research efforts to improve IVF outcomes have been focused mainly on the embryo factor and methods to improve embryo selection, such as extended culture to blastocyst, time-lapse imaging (morphokinetic assessment), and more notably, preimplantation genetic testing for aneuploidy (PGT-A). However, the other half of the equation, the endometrium, has not garnered the attention that it deserves. Effort has therefore been renewed to optimize the endometrial factor by better diagnosing and treating various forms of endometrial dysfunction that could lead to infertility in general and lack of success with IVF and euploid embryo transfers in particular.
Historical background on endometrial function
Progesterone has long been recognized as the main effector that transforms the estrogen-primed endometrium into a receptive state that results in successful embryo implantation. Progesterone exposure is required at appropriate levels and duration before the endometrium becomes receptive to the embryo. If implantation does not occur soon after the endometrium has attained receptive status (7–10 days after ovulation), further progesterone exposure results in progression of endometrial changes that no longer permit successful implantation.
As early as the 1950s, “luteal phase deficiency” was defined as due to inadequate progesterone secretion and resulted in a short luteal phase. In the 1970s, histologic “dating” of the endometrium became the gold standard for diagnosing luteal phase defects; this relied on a classic histologic appearance of secretory phase endometrium and its changes throughout the luteal phase. Subsequently, however, results of prospective randomized controlled trials published in 2004 cast significant doubt on the accuracy and reproducibility of these endometrial biopsies and did not show any clinical diagnostic benefit or correlation with pregnancy outcomes.
21st century advances: Endometrial dating 2.0
A decade later, with the advancement of molecular biology tools such as microarray technology, researchers were able to study endometrial gene expression patterns at different stages of the menstrual cycle. They identified different phases of endometrial development with molecular profiles, or “signatures,” for the luteal phase, endometriosis, polycystic ovary syndrome, and uterine fibroids.
In 2013, researchers in Spain introduced a diagnostic test called endometrial receptivity array (ERA) with the stated goal of being able to temporally define the receptive endometrium and identify prereceptive as well as postreceptive states.1 In other words, instead of the histologic dating of the endometrium used in the 1970s, it represented “molecular dating” of the endometrium. Although the initial studies were conducted among women who experienced prior unsuccessful embryo transfers (the so-called recurrent implantation failure, or RIF), the test’s scope was subsequently expanded to include any individual planning on a frozen embryo transfer (FET), regardless of any prior attempts. The term personalized embryo transfer (pET) was coined to suggest the ability to define the best time (up to hours) for embryo transfers on an individual basis. Despite lack of independent validation studies, ERA was then widely adopted by many clinicians (and requested by some patients) with the hope of improving IVF outcomes.
However, not unlike many other novel innovations in assisted reproductive technology, ERA regrettably did not withstand the test of time. Three independent studies in 2021, 1 randomized clinical trial and 2 observational cohort studies, did not show any benefit with regard to implantation rates, pregnancy rates, or live birth rates when ERA was performed in the general infertility population.2-4
Continue to: Study results...
Study results
The cohort study that matched 133 ERA patients with 353 non-ERA patients showed live birth rates of 49.62% for the ERA group and 54.96% for the non-ERA group (odds ratio [OR], 0.8074; 95% confidence interval [CI], 0.5424–1.2018).2 Of note, no difference occurred between subgroups based on the prior number of FETs or the receptivity status (TABLE 1).

Another cohort study from the University of California, Los Angeles, published in 2021 analyzed 228 single euploid FET cycles.3 This study did not show any benefit for routine ERA testing, with a live birth rate of 56.6% in the non-ERA group and 56.5% in the ERA group.
Still, the most convincing evidence for the lack of benefit from routine ERA was noted from the results of the randomized clinical trial.4 A total of 767 patients were randomly allocated, 381 to the ERA group and 386 to the control group. There was no difference in ongoing pregnancy rates between the 2 groups. Perhaps more important, even after limiting the analysis to individuals with a nonreceptive ERA result, there was no difference in ongoing pregnancy rates between the 2 groups: 62.5% in the control group (default timing of transfer) and 55.5% in the study group (transfer timing adjusted based on ERA) (rate ratio [RR], 0.9; 95% CI, 0.70–1.14).
ERA usefulness is unsupported in general infertility population
The studies discussed collectively suggest with a high degree of certainty that there is no indication for routine ERA testing in the general infertility population prior to frozen embryo transfers.
Although these studies all were conducted in the general infertility population and did not specifically evaluate the performance of ERA in women with recurrent pregnancy loss or recurrent implantation failure, it is important to acknowledge that if ERA were truly able to define the window of receptivity, one would expect a lower implantation rate if the embryos were transferred outside of the window suggested by the ERA. This was not the case in these studies, as they all showed equivalent pregnancy rates in the control (nonadjusted) groups even when ERA suggested a nonreceptive status.
This observation seriously questions the validity of ERA regarding its ability to temporally define the window of receptivity. On the other hand, as stated earlier, there is still a possibility for ERA to be beneficial for a small subgroup of patients whose window of receptivity may not be as wide as expected in the general population. The challenging question would be how best to identify the particular group with a narrow, or displaced, window of receptivity.
The optimal timing for implantation of a normal embryo requires a receptive endometrium. The endometrial biopsy was used widely for many years before research showed it was not clinically useful. More recently, the endometrial receptivity array has been suggested to help time the frozen embryo transfer. Unfortunately, recent studies have shown that this test is not clinically useful for the general infertility population.
Continue to: WHO raises awareness of endometriosis burden and...
WHO raises awareness of endometriosis burden and highlights need to address diagnosis and treatment for women’s reproductive health
World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room /fact-sheets/detail/endometriosis. Accessed January 3, 2022.
The WHO published its first fact sheet on endometriosis in March 2021, recognizing endometriosis as a severe disease that affects almost 190 million women with life-impacting pain, infertility, other symptoms, and especially with chronic, significant emotional sequelae (TABLE 2).5 The disease’s variable and broad symptoms result in a lack of awareness and diagnosis by both women and health care providers, especially in low- and middle-income countries and in disadvantaged populations in developed countries. Increased awareness to promote earlier diagnosis, improved training for better management, expanded research for greater understanding, and policies that increase access to quality care are needed to ensure the reproductive health and rights of tens of millions of women with endometriosis.

Endometriosis characteristics and symptoms
Endometriosis is characterized by the presence of tissue resembling endometrium outside the uterus, where it causes a chronic inflammatory reaction that may result in the formation of scar tissue. Endometriotic lesions may be superficial, cystic ovarian endometriomas, or deep lesions, causing a myriad of pain and related symptoms.6.7
Chronic pain may occur because pain centers in the brain become hyperresponsive over time (central sensitization); this can occur at any point throughout the life course of endometriosis, even when endometriosis lesions are no longer visible. Sometimes, endometriosis is asymptomatic. In addition, endometriosis can cause infertility through anatomic distortion and inflammatory, endocrinologic, and other pathways.
The origins of endometriosis are thought to be multifactorial and include retrograde menstruation, cellular metaplasia, and/or stem cells that spread through blood and lymphatic vessels. Endometriosis is estrogen dependent, but lesion growth also is affected by altered or impaired immunity, localized complex hormonal influences, genetics, and possibly environmental contaminants.
Impact on public health and reproductive rights
Endometriosis has significant social, public health, and economic implications. It can decrease quality of life and prevent girls and women from attending work or school.8 Painful sex can affect sexual health. The WHO states that, “Addressing endometriosis will empower those affected by it, by supporting their human right to the highest standard of sexual and reproductive health, quality of life, and overall well-being.”5
At present, no known way is available to prevent or cure endometriosis. Early diagnosis and treatment, however, may slow or halt its natural progression and associated symptoms.
Diagnostic steps and treatment options
Early suspicion of endometriosis is the most important factor, followed by a careful history of menstrual symptoms and chronic pelvic pain, early referral to specialists for ultrasonography or other imaging, and sometimes surgical or laparoscopic visualization. Empirical treatment can be begun without histologic or laparoscopic confirmation.
Endometriosis can be treated with medications and/or surgery depending on symptoms, lesions, desired outcome, and patient choice.5,6 Common therapies include contraceptive steroids, nonsteroidal anti-inflammatory medications, and analgesics. Medical treatments focus on either lowering estrogen or increasing progesterone levels.
Surgery can remove endometriosis lesions, adhesions, and scar tissue. However, success in reducing pain symptoms and increasing pregnancy rates often depends on the extent of disease.
For infertility due to endometriosis, treatment options include laparoscopic surgical removal of endometriosis, ovarian stimulation with intrauterine insemination (IUI), and IVF. Multidisciplinary treatment addressing different symptoms and overall health often requires referral to pain experts and other specialists.9
The WHO perspective on endometriosis
Recognizing the importance of endometriosis and its impact on people’s sexual and reproductive health, quality of life, and overall well-being, the WHO is taking action to improve awareness, diagnosis, and treatment of endometriosis (TABLE 3).5 ●

Endometriosis is now recognized as a disease with significant burden for women everywhere. Widespread lack of awareness of presenting symptoms and management options means that all women’s health care clinicians need to become better informed about endometriosis so they can improve the quality of care they provide.
- Ruiz-Alonso M, Blesa D, Díaz-Gimeno P, et al. The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertil Steril. 2013;100:818-824.
- Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
- Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
- Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
- World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room/fact-sheets/detail /endometriosis. Accessed January 3, 2022.
- Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382:1244-1256.
- Johnson NP, Hummelshoj L, Adamson GD, et al. World Endometriosis Society consensus on the classification of endometriosis. Hum Reprod. 2017;32:315-324.
- Nnoaham K, Hummelshoj L, Webster P, et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil Steril. 2011;96:366-373.e8.
- Carey ET, Till SR, As-Sanie S. Pharmacological management of chronic pelvic pain in women. Drugs. 2017;77:285-301.
- Ruiz-Alonso M, Blesa D, Díaz-Gimeno P, et al. The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertil Steril. 2013;100:818-824.
- Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
- Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
- Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
- World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room/fact-sheets/detail /endometriosis. Accessed January 3, 2022.
- Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382:1244-1256.
- Johnson NP, Hummelshoj L, Adamson GD, et al. World Endometriosis Society consensus on the classification of endometriosis. Hum Reprod. 2017;32:315-324.
- Nnoaham K, Hummelshoj L, Webster P, et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil Steril. 2011;96:366-373.e8.
- Carey ET, Till SR, As-Sanie S. Pharmacological management of chronic pelvic pain in women. Drugs. 2017;77:285-301.
2021 Update on fertility
In this Update, we discuss several aspects of infertility and emerging technologic advances in treatment. We review an important infertility fact sheet recently issued by the World Health Organization (WHO) that provides a succinct overview of infertility causes, the rights of infertility patients, treatment challenges, and advocacy efforts. In addition, we discuss what the infertility literature reveals about reducing multiple birth rates and the technologic, financial, and social factors involved. Finally, we look at the molecular progress made in germline-editing technology and the myriad complications involved in its potential future translation to clinical phenotyping.
WHO recognizes the burden of infertility and addresses fertility care needs
World Health Organization (WHO). Infertility fact sheet. September 14, 2020. https://www.who.int/news-room/fact-sheets/detail/infertility. Accessed January 24, 2021.
The WHO published its first comprehensive infertility fact sheet in September 2020. This document is important because it validates infertility as a high-burden disease and disability that diminishes quality of life for up to 186 million individuals globally. The infertility fact sheet is a comprehensive yet focused quick read that addresses the causes of infertility, why infertility is important, challenges, and the WHO response.
Factors in infertility
Infertility is caused by different factors in women and men, yet sometimes it is unexplained, and its relative importance can vary from country to country. For women, tubal disorders (for example, postinfectious), uterine problems (fibroids, congenital), endometriosis, ovarian disorders (polycystic ovary syndrome, ovulation disorders), and endocrine imbalances are the most common factors.
For men, causes of infertility include obstruction of the reproductive tract (as after injuries or infection); hormonal disorders in the hypothalamus, pituitary, and/or testicles (for example, low testosterone); testicular failure to produce sperm (such as after cancer treatment); and abnormal sperm function and quality (low count, motility, or morphology).
Environmental and lifestyle factors— including smoking, obesity, alcohol, or toxins—can affect fertility.
Continue to: Recognizing all individuals’ fertility rights...
Recognizing all individuals’ fertility rights
The WHO infertility fact sheet makes strong statements, recognizing that individuals and couples have the right to decide the number, timing, and spacing of their children. Addressing infertility is therefore an important part of realizing the right of individuals and couples to found a family. This includes heterosexual couples, same-sex partners, older persons, individuals not in sexual relationships who might require infertility management and fertility care services, and notably marginalized populations.
Addressing infertility also can help mitigate gender inequality, which has significant negative social impacts on the lives of infertile individuals, especially women. Fertility education is important to reduce the fear of infertility and contraception use in those wanting pregnancy in the future.
In most countries the biggest challenges are availability, access, and quality of interventions to address infertility. This includes the United States, where only 1 in 4 individuals receive the fertility care they need. Lack of prioritization, ineffective public health strategies, inadequate funding, and costs are barriers. Health policies need to recognize that infertility is a disease that often can be prevented, thereby reducing future costs. Comprehensive awareness and education programs, laws and policies that regulate and ensure access and the human rights of all involved, are essential.
Advocacy efforts
To address infertility and fertility care, the WHO is committed to:
- collaborate with partners on epidemiologic and etiologic research
- facilitate policy dialogue globally to frame infertility within a legal and policy framework
- support generation of data on the burden of infertility
- develop guidelines
- produce other documents of standards
- collaborate with all stakeholders to strengthen political commitment and health system capacity, and
- provide country-level technical support to develop or strengthen policies and services.
For your practice, this means that infertility is recognized as a disease that should receive its appropriate share of health care resources. Infertility and fertility care are the right of every individual according to their desires to found a family. Besides providing the best care you can to all your patients, including referring them when necessary, all health care clinicians should advocate on behalf of their patients to payors, policy makers, and the public the need to provide equitable laws, resources, and funding for infertility and fertility care.
Every person has the right to infertility and fertility care as endorsed by the recent WHO infertility fact sheet. To address this high-burden disease, all women’s health care clinicians should be aware of, equitably diagnose and treat, refer as necessary, and advocate for infertile individuals.
Continue to: Lessons learned in reducing multiple pregnancy rates in infertility treatment...
Lessons learned in reducing multiple pregnancy rates in infertility treatment
Views and reviews section. Fertil Steril. 2020;114:671- 672; 673-679; 680-689; 690-714; 715-721.
In the October 2020 issue of Fertility and Sterility, the Views and Reviews section included 5 articles on avoiding multiple live birth rates (LBRs) in assisted reproductive technologies (ART).1-5 International experts provided a comprehensive review of global multiple LBRs and their associated negative impact on maternal and perinatal outcomes, reasons for global variability, strategies to reduce multiples, single embryo transfer, and implications of funding and reporting. These international comparisons and recommendations are helpful and applicable to infertility care in the United States.3
The rise of multiple birth rates
During the first decade of in vitro fertilization (IVF), live birth rates were low, increasing to 14% in 1990. Multiple embryos needed to be transferred so that even these LBRs could be obtained. In the 1990s, however, laboratory technology improved rapidly, with increased implantation rates and subsequent rapid increases in LBR, but also with increased multiple birth rates (MBRs).
In the United States, clinic-specific reporting helped create competition among clinics for the best LBRs, and this led to MBRs of 30% and higher. Numerous studies documented the associated significantly increased morbidity and mortality of both mothers and babies. Similar situations occurred in many other countries while some, especially Nordic nations, Australia, New Zealand, and Japan, had twin rates of less than 10% or even 5% since the early 2000s. So why the difference?
The higher MBR is due largely to the transfer of more than one embryo. The immediate solution is therefore always to perform elective single embryo transfer (eSET). However, numerous factors affect the decision to perform eSET or not, and this ideal is far from being achieved. Older women, those with longer duration of infertility and/or failed treatment, often feel a time pressure and want to transfer more embryos. Of course, biologically this is reasonable because the number and quality of their embryos is lower. While attempts have been made to assess embryo quality with preimplantation genetic testing for aneuploidy, evidence that this increases the LBR is controversial except possibly in women aged 35 to 38 years. This is especially true when the cumulative LBR, that is, the number of live births after transfer of all embryos from an egg retrieval cycle, is the measured outcome.
The major factor that determines the frequency of eSET is financial. Affordability is the out-of-pocket cost (after insurance or other subsidy) as a percentage of disposable income, and it is the most important factor that determines whether eSET is performed. Less affordable treatment creates a financial incentive to transfer more than one embryo to maximize the pregnancy rates in fewer cycles.5 Other factors include whether the effectiveness of treatment, that is, LBR, is emphasized over safety, that is, MBR. In the United States, the Society for Assisted Reproductive Technology now reports cumulative LBR, singleton and multiple LBR, and preterm births as outcomes, thereby increasing the emphasis on eSET.
Sociologic, cultural, and religious factors also can affect the frequency of eSET. Even within the United States, great variation exists in values and beliefs regarding infertility treatment. It can be challenging to determine who makes decisions: the patient alone, the physician, the payor, professional guidelines, or laws. In many countries, including the United States, it is an amalgam of these.
Setting new goals
If the goal is to reduce the MBR, what should that rate be? In the past few years, the MBR in the United States has been reduced to approximately 10%. It is reasonable now to set a goal of 5% in the next several years. To do this, we can learn from countries that have been successful. The United States already has very high-quality clinical and laboratory services, knowledgeable physicians, and a reasonable regulatory environment. Improved technology, specifically embryo selection for transfer, and focus on adherence to established embryo transfer guidelines could help.
Many would argue that eSET essentially should be performed always in women younger than age 40 and in all women of any age with a known euploid embryo. The major problem that drives multiples is the lack of affordability, which can be addressed by increased subsidies from payors. Increased subsidies can result from legislative mandates or societal pressures on employers, either of which could be associated with requirements for eSET and/or reduced MBRs.
In your practice, you can now reassure your infertility patients that cumulative LBRs are excellent in the United States and that the risk of multiple pregnancy has been reduced dramatically. This should encourage more patients to accept and take advantage of this successful technology that has resulted in the birth of millions of babies globally. Further reduction of the MBR to 5% should be possible within a few years through education and advocacy by women’s health care clinicians that results in increased subsidies and more affordable IVF.
The multiple birth rate in ART has been reduced to 10% in the United States through an increased understanding of the complex factors that affect embryo transfer practices globally. Further progress will depend primarily on increased subsidies that make ART more affordable.
Continue to: Genetics and ART...
Genetics and ART: Selection versus correction
Adashi EY, Cohen IG. The case for remedial germline editing—the long-term view. JAMA. 2020;323:1762-1763.
Rosenbaum L. The future of gene editing—toward scientific and social consensus. N Engl J Med. 2019;380:971-975.
Cyranoski D. The CRISPR-baby scandal: what’s next for human gene-editing. Nature. 2019;566:440-442.
de Wert G, Pennings G, Clarke A, et al; European Society of Human Genetics and European Society of Human Reproduction and Embryology. Human germline gene editing: recommendations of ESHG and ESHRE. Hum Reprod Open. 2018;hox025.
Following the completion of the Human Genome Project in 2003 and major technologic advancements in the subsequent years, the field of human genetics became the focal point of convergence for several distinct but interrelated disciplines: bioinformatics, computational biology, and sequencing technologies. As the result, individual human genomes can now be sequenced at a single base pair level, and with higher fidelity, at a fraction of the original cost and at a much faster speed.
This molecular progress, however, has not been accompanied by an equivalent clinical progress, because in a significant number of cases a defined and predictable clinical phenotype cannot be attributed to a detected molecular genotype. This has resulted in an overabundance of variants of uncertain significance. Variable expressivity, incomplete penetrance, epigenetics, mosaicism, and the polygenic nature of many human traits further complicate reliable interpretation and prognostication of the colossal amount of molecular genetic data that are being generated by the above-mentioned technologic advances.
Considering these limitations, at this juncture it is crucial to acknowledge that any attempts to prematurely commercialize these preclinical and research studies (such as polygenic risk scores for embryos) are perilous and have the potential to cause significant harm in terms of unnecessary stress and anxiety for intended parents as well as the potential for yet-unmapped societal and legal implications.
However, it is just a matter of time until more accurate clinical phenotyping catches up with molecular genotyping. As we get closer to this next historic milestone, precision medicine in the postnatal life (with regard to both diagnostics and therapeutics) and preimplantation genetic testing (PGT) at the prenatal stage for a much wider spectrum of conditions—including both monogenic and polygenic traits—may indeed become a reality.
The potential of germline editing
Specifically regarding PGT (which requires IVF), it is important to recognize that due to the limited and nonrenewable endowment of human oocytes (ovarian reserve), combined with the detrimental impact of advancing age on the quality of the remaining cohort as manifested by a higher risk of aneuploidy, the current clinical practice of trying to “select” a nonaffected embryo can be very inefficient. As a result, the intended parents pursuing such treatments may need to undergo multiple cycles of ovarian stimulation and oocyte retrieval.
A potential solution for genes associated with known diseases is the prospect of remedial germline editing by CRISPR–Cas9 technology or its future descendants. This would take advantage of the existing embryos to try to “correct” the defective gene instead of trying to “select” a normal embryo. These technologies are still in the early stages of development and are remotely distant from clinical applications. On the other hand, although germline gene editing, if actualized, would be a monumental breakthrough in the history of genetics and medicine, we must be cognizant of its serious legal, societal, and ethical ramifications, which are currently unknown. Furthermore, even at the biologic and technical level, the technology still is not advanced enough to reliably rule out off-target modifications, and the unintended clinical consequences of the on-target corrections have not been studied either.
Regulation of genetic modifications
Due to these myriad concerns and the lack of an existing appropriate regulatory framework and oversight for such interventions, current US law (since December 2015, through provisions in annual federal appropriations laws passed by Congress and renewed annually thereafter) bars the US Food and Drug Administration from considering any clinical trial application “in which a human embryo is intentionally created or modified to include a heritable genetic modification.” Notably, this moratorium also prohibits mitochondrial replacement technology (MRT), which is a less controversial and relatively better-studied innovation.
Mitochondrial genetic disorders caused by the mutations in mitochondrial DNA (versus nuclear DNA) are amenable to a specific treatment strategy aimed at substituting the defective maternal mitochondrial genome with the mitochondrial genome of an unaffected donor oocyte. This can be achieved via either pronuclear transfer, which involves isolation and transfer of the male and female pronuclei from an affected embryo to an enucleated normal donor embryo, or maternal spindle transfer, which involves isolation and transfer of the metaphase II spindle complex of an affected oocyte to an enucleated disease-free donor egg. It is noteworthy that in 2015 in the United Kingdom, Parliament expanded the definition of “permitted eggs and embryos” to include those “where unhealthy mitochondrial DNA is replaced by healthy mitochondrial DNA from a donor.” This thereby allows the UK Human Fertilisation and Embryology Authority to formally direct and oversee clinical trials in MRT.
Summing up
Although the future of assisted human reproduction cannot be clearly outlined at this time, it is likely to be radically different from the current state given these emerging applications at the intersection of ART and diagnostic and therapeutic genetics. To ensure that exploring this uncharted territory will ultimately be in the interest of humankind and civilization, proper regulatory oversight—after careful consideration of all ethical, societal, and legal implications—needs to be developed for all preclinical and clinical research in this field. Participatory public engagement must be an integrated part of this process. ●
The field of human genetics has already transformed medicine. However, the convergence of the interrelated disciplines of bioinformatics, computational biology, sequencing technologies, and CRISPR–Cas9 technology is creating incredible new advances that will bring great benefits but also major societal challenges.
- Farquhar C. Avoiding multiple pregnancies in assisted reproductive technologies: transferring one embryo at a time should be the norm. Fertil Steril. 2020;114:671-672.
- Bergh C, Kamath MS, Wang R, et al. Strategies to reduce multiple pregnancies during medically assisted reproduction. Fertil Steril. 2020;114:673-679.
- Adamson GD, Norman RJ. Why are multiple pregnancy rates and single embryo transfer rates so different globally, and what do we do about it? Fertil Steril. 2020;114:680-689.
- Eapen A, Ryan GL, Ten Eyck P, et al. Current evidence supporting a goal of singletons: a review of maternal and neonatal outcomes associated with twin versus singleton pregnancies after in vitro fertilization and intracytoplasmic sperm injection. Fertil Steril. 2020;114: 690-714.
- Chambers GM, Keller E, Choi S, et al. Funding and public reporting strategies for reducing multiple pregnancy from fertility treatments. Fertil Steril. 2020;114:715-721.
In this Update, we discuss several aspects of infertility and emerging technologic advances in treatment. We review an important infertility fact sheet recently issued by the World Health Organization (WHO) that provides a succinct overview of infertility causes, the rights of infertility patients, treatment challenges, and advocacy efforts. In addition, we discuss what the infertility literature reveals about reducing multiple birth rates and the technologic, financial, and social factors involved. Finally, we look at the molecular progress made in germline-editing technology and the myriad complications involved in its potential future translation to clinical phenotyping.
WHO recognizes the burden of infertility and addresses fertility care needs
World Health Organization (WHO). Infertility fact sheet. September 14, 2020. https://www.who.int/news-room/fact-sheets/detail/infertility. Accessed January 24, 2021.
The WHO published its first comprehensive infertility fact sheet in September 2020. This document is important because it validates infertility as a high-burden disease and disability that diminishes quality of life for up to 186 million individuals globally. The infertility fact sheet is a comprehensive yet focused quick read that addresses the causes of infertility, why infertility is important, challenges, and the WHO response.
Factors in infertility
Infertility is caused by different factors in women and men, yet sometimes it is unexplained, and its relative importance can vary from country to country. For women, tubal disorders (for example, postinfectious), uterine problems (fibroids, congenital), endometriosis, ovarian disorders (polycystic ovary syndrome, ovulation disorders), and endocrine imbalances are the most common factors.
For men, causes of infertility include obstruction of the reproductive tract (as after injuries or infection); hormonal disorders in the hypothalamus, pituitary, and/or testicles (for example, low testosterone); testicular failure to produce sperm (such as after cancer treatment); and abnormal sperm function and quality (low count, motility, or morphology).
Environmental and lifestyle factors— including smoking, obesity, alcohol, or toxins—can affect fertility.
Continue to: Recognizing all individuals’ fertility rights...
Recognizing all individuals’ fertility rights
The WHO infertility fact sheet makes strong statements, recognizing that individuals and couples have the right to decide the number, timing, and spacing of their children. Addressing infertility is therefore an important part of realizing the right of individuals and couples to found a family. This includes heterosexual couples, same-sex partners, older persons, individuals not in sexual relationships who might require infertility management and fertility care services, and notably marginalized populations.
Addressing infertility also can help mitigate gender inequality, which has significant negative social impacts on the lives of infertile individuals, especially women. Fertility education is important to reduce the fear of infertility and contraception use in those wanting pregnancy in the future.
In most countries the biggest challenges are availability, access, and quality of interventions to address infertility. This includes the United States, where only 1 in 4 individuals receive the fertility care they need. Lack of prioritization, ineffective public health strategies, inadequate funding, and costs are barriers. Health policies need to recognize that infertility is a disease that often can be prevented, thereby reducing future costs. Comprehensive awareness and education programs, laws and policies that regulate and ensure access and the human rights of all involved, are essential.
Advocacy efforts
To address infertility and fertility care, the WHO is committed to:
- collaborate with partners on epidemiologic and etiologic research
- facilitate policy dialogue globally to frame infertility within a legal and policy framework
- support generation of data on the burden of infertility
- develop guidelines
- produce other documents of standards
- collaborate with all stakeholders to strengthen political commitment and health system capacity, and
- provide country-level technical support to develop or strengthen policies and services.
For your practice, this means that infertility is recognized as a disease that should receive its appropriate share of health care resources. Infertility and fertility care are the right of every individual according to their desires to found a family. Besides providing the best care you can to all your patients, including referring them when necessary, all health care clinicians should advocate on behalf of their patients to payors, policy makers, and the public the need to provide equitable laws, resources, and funding for infertility and fertility care.
Every person has the right to infertility and fertility care as endorsed by the recent WHO infertility fact sheet. To address this high-burden disease, all women’s health care clinicians should be aware of, equitably diagnose and treat, refer as necessary, and advocate for infertile individuals.
Continue to: Lessons learned in reducing multiple pregnancy rates in infertility treatment...
Lessons learned in reducing multiple pregnancy rates in infertility treatment
Views and reviews section. Fertil Steril. 2020;114:671- 672; 673-679; 680-689; 690-714; 715-721.
In the October 2020 issue of Fertility and Sterility, the Views and Reviews section included 5 articles on avoiding multiple live birth rates (LBRs) in assisted reproductive technologies (ART).1-5 International experts provided a comprehensive review of global multiple LBRs and their associated negative impact on maternal and perinatal outcomes, reasons for global variability, strategies to reduce multiples, single embryo transfer, and implications of funding and reporting. These international comparisons and recommendations are helpful and applicable to infertility care in the United States.3
The rise of multiple birth rates
During the first decade of in vitro fertilization (IVF), live birth rates were low, increasing to 14% in 1990. Multiple embryos needed to be transferred so that even these LBRs could be obtained. In the 1990s, however, laboratory technology improved rapidly, with increased implantation rates and subsequent rapid increases in LBR, but also with increased multiple birth rates (MBRs).
In the United States, clinic-specific reporting helped create competition among clinics for the best LBRs, and this led to MBRs of 30% and higher. Numerous studies documented the associated significantly increased morbidity and mortality of both mothers and babies. Similar situations occurred in many other countries while some, especially Nordic nations, Australia, New Zealand, and Japan, had twin rates of less than 10% or even 5% since the early 2000s. So why the difference?
The higher MBR is due largely to the transfer of more than one embryo. The immediate solution is therefore always to perform elective single embryo transfer (eSET). However, numerous factors affect the decision to perform eSET or not, and this ideal is far from being achieved. Older women, those with longer duration of infertility and/or failed treatment, often feel a time pressure and want to transfer more embryos. Of course, biologically this is reasonable because the number and quality of their embryos is lower. While attempts have been made to assess embryo quality with preimplantation genetic testing for aneuploidy, evidence that this increases the LBR is controversial except possibly in women aged 35 to 38 years. This is especially true when the cumulative LBR, that is, the number of live births after transfer of all embryos from an egg retrieval cycle, is the measured outcome.
The major factor that determines the frequency of eSET is financial. Affordability is the out-of-pocket cost (after insurance or other subsidy) as a percentage of disposable income, and it is the most important factor that determines whether eSET is performed. Less affordable treatment creates a financial incentive to transfer more than one embryo to maximize the pregnancy rates in fewer cycles.5 Other factors include whether the effectiveness of treatment, that is, LBR, is emphasized over safety, that is, MBR. In the United States, the Society for Assisted Reproductive Technology now reports cumulative LBR, singleton and multiple LBR, and preterm births as outcomes, thereby increasing the emphasis on eSET.
Sociologic, cultural, and religious factors also can affect the frequency of eSET. Even within the United States, great variation exists in values and beliefs regarding infertility treatment. It can be challenging to determine who makes decisions: the patient alone, the physician, the payor, professional guidelines, or laws. In many countries, including the United States, it is an amalgam of these.
Setting new goals
If the goal is to reduce the MBR, what should that rate be? In the past few years, the MBR in the United States has been reduced to approximately 10%. It is reasonable now to set a goal of 5% in the next several years. To do this, we can learn from countries that have been successful. The United States already has very high-quality clinical and laboratory services, knowledgeable physicians, and a reasonable regulatory environment. Improved technology, specifically embryo selection for transfer, and focus on adherence to established embryo transfer guidelines could help.
Many would argue that eSET essentially should be performed always in women younger than age 40 and in all women of any age with a known euploid embryo. The major problem that drives multiples is the lack of affordability, which can be addressed by increased subsidies from payors. Increased subsidies can result from legislative mandates or societal pressures on employers, either of which could be associated with requirements for eSET and/or reduced MBRs.
In your practice, you can now reassure your infertility patients that cumulative LBRs are excellent in the United States and that the risk of multiple pregnancy has been reduced dramatically. This should encourage more patients to accept and take advantage of this successful technology that has resulted in the birth of millions of babies globally. Further reduction of the MBR to 5% should be possible within a few years through education and advocacy by women’s health care clinicians that results in increased subsidies and more affordable IVF.
The multiple birth rate in ART has been reduced to 10% in the United States through an increased understanding of the complex factors that affect embryo transfer practices globally. Further progress will depend primarily on increased subsidies that make ART more affordable.
Continue to: Genetics and ART...
Genetics and ART: Selection versus correction
Adashi EY, Cohen IG. The case for remedial germline editing—the long-term view. JAMA. 2020;323:1762-1763.
Rosenbaum L. The future of gene editing—toward scientific and social consensus. N Engl J Med. 2019;380:971-975.
Cyranoski D. The CRISPR-baby scandal: what’s next for human gene-editing. Nature. 2019;566:440-442.
de Wert G, Pennings G, Clarke A, et al; European Society of Human Genetics and European Society of Human Reproduction and Embryology. Human germline gene editing: recommendations of ESHG and ESHRE. Hum Reprod Open. 2018;hox025.
Following the completion of the Human Genome Project in 2003 and major technologic advancements in the subsequent years, the field of human genetics became the focal point of convergence for several distinct but interrelated disciplines: bioinformatics, computational biology, and sequencing technologies. As the result, individual human genomes can now be sequenced at a single base pair level, and with higher fidelity, at a fraction of the original cost and at a much faster speed.
This molecular progress, however, has not been accompanied by an equivalent clinical progress, because in a significant number of cases a defined and predictable clinical phenotype cannot be attributed to a detected molecular genotype. This has resulted in an overabundance of variants of uncertain significance. Variable expressivity, incomplete penetrance, epigenetics, mosaicism, and the polygenic nature of many human traits further complicate reliable interpretation and prognostication of the colossal amount of molecular genetic data that are being generated by the above-mentioned technologic advances.
Considering these limitations, at this juncture it is crucial to acknowledge that any attempts to prematurely commercialize these preclinical and research studies (such as polygenic risk scores for embryos) are perilous and have the potential to cause significant harm in terms of unnecessary stress and anxiety for intended parents as well as the potential for yet-unmapped societal and legal implications.
However, it is just a matter of time until more accurate clinical phenotyping catches up with molecular genotyping. As we get closer to this next historic milestone, precision medicine in the postnatal life (with regard to both diagnostics and therapeutics) and preimplantation genetic testing (PGT) at the prenatal stage for a much wider spectrum of conditions—including both monogenic and polygenic traits—may indeed become a reality.
The potential of germline editing
Specifically regarding PGT (which requires IVF), it is important to recognize that due to the limited and nonrenewable endowment of human oocytes (ovarian reserve), combined with the detrimental impact of advancing age on the quality of the remaining cohort as manifested by a higher risk of aneuploidy, the current clinical practice of trying to “select” a nonaffected embryo can be very inefficient. As a result, the intended parents pursuing such treatments may need to undergo multiple cycles of ovarian stimulation and oocyte retrieval.
A potential solution for genes associated with known diseases is the prospect of remedial germline editing by CRISPR–Cas9 technology or its future descendants. This would take advantage of the existing embryos to try to “correct” the defective gene instead of trying to “select” a normal embryo. These technologies are still in the early stages of development and are remotely distant from clinical applications. On the other hand, although germline gene editing, if actualized, would be a monumental breakthrough in the history of genetics and medicine, we must be cognizant of its serious legal, societal, and ethical ramifications, which are currently unknown. Furthermore, even at the biologic and technical level, the technology still is not advanced enough to reliably rule out off-target modifications, and the unintended clinical consequences of the on-target corrections have not been studied either.
Regulation of genetic modifications
Due to these myriad concerns and the lack of an existing appropriate regulatory framework and oversight for such interventions, current US law (since December 2015, through provisions in annual federal appropriations laws passed by Congress and renewed annually thereafter) bars the US Food and Drug Administration from considering any clinical trial application “in which a human embryo is intentionally created or modified to include a heritable genetic modification.” Notably, this moratorium also prohibits mitochondrial replacement technology (MRT), which is a less controversial and relatively better-studied innovation.
Mitochondrial genetic disorders caused by the mutations in mitochondrial DNA (versus nuclear DNA) are amenable to a specific treatment strategy aimed at substituting the defective maternal mitochondrial genome with the mitochondrial genome of an unaffected donor oocyte. This can be achieved via either pronuclear transfer, which involves isolation and transfer of the male and female pronuclei from an affected embryo to an enucleated normal donor embryo, or maternal spindle transfer, which involves isolation and transfer of the metaphase II spindle complex of an affected oocyte to an enucleated disease-free donor egg. It is noteworthy that in 2015 in the United Kingdom, Parliament expanded the definition of “permitted eggs and embryos” to include those “where unhealthy mitochondrial DNA is replaced by healthy mitochondrial DNA from a donor.” This thereby allows the UK Human Fertilisation and Embryology Authority to formally direct and oversee clinical trials in MRT.
Summing up
Although the future of assisted human reproduction cannot be clearly outlined at this time, it is likely to be radically different from the current state given these emerging applications at the intersection of ART and diagnostic and therapeutic genetics. To ensure that exploring this uncharted territory will ultimately be in the interest of humankind and civilization, proper regulatory oversight—after careful consideration of all ethical, societal, and legal implications—needs to be developed for all preclinical and clinical research in this field. Participatory public engagement must be an integrated part of this process. ●
The field of human genetics has already transformed medicine. However, the convergence of the interrelated disciplines of bioinformatics, computational biology, sequencing technologies, and CRISPR–Cas9 technology is creating incredible new advances that will bring great benefits but also major societal challenges.
In this Update, we discuss several aspects of infertility and emerging technologic advances in treatment. We review an important infertility fact sheet recently issued by the World Health Organization (WHO) that provides a succinct overview of infertility causes, the rights of infertility patients, treatment challenges, and advocacy efforts. In addition, we discuss what the infertility literature reveals about reducing multiple birth rates and the technologic, financial, and social factors involved. Finally, we look at the molecular progress made in germline-editing technology and the myriad complications involved in its potential future translation to clinical phenotyping.
WHO recognizes the burden of infertility and addresses fertility care needs
World Health Organization (WHO). Infertility fact sheet. September 14, 2020. https://www.who.int/news-room/fact-sheets/detail/infertility. Accessed January 24, 2021.
The WHO published its first comprehensive infertility fact sheet in September 2020. This document is important because it validates infertility as a high-burden disease and disability that diminishes quality of life for up to 186 million individuals globally. The infertility fact sheet is a comprehensive yet focused quick read that addresses the causes of infertility, why infertility is important, challenges, and the WHO response.
Factors in infertility
Infertility is caused by different factors in women and men, yet sometimes it is unexplained, and its relative importance can vary from country to country. For women, tubal disorders (for example, postinfectious), uterine problems (fibroids, congenital), endometriosis, ovarian disorders (polycystic ovary syndrome, ovulation disorders), and endocrine imbalances are the most common factors.
For men, causes of infertility include obstruction of the reproductive tract (as after injuries or infection); hormonal disorders in the hypothalamus, pituitary, and/or testicles (for example, low testosterone); testicular failure to produce sperm (such as after cancer treatment); and abnormal sperm function and quality (low count, motility, or morphology).
Environmental and lifestyle factors— including smoking, obesity, alcohol, or toxins—can affect fertility.
Continue to: Recognizing all individuals’ fertility rights...
Recognizing all individuals’ fertility rights
The WHO infertility fact sheet makes strong statements, recognizing that individuals and couples have the right to decide the number, timing, and spacing of their children. Addressing infertility is therefore an important part of realizing the right of individuals and couples to found a family. This includes heterosexual couples, same-sex partners, older persons, individuals not in sexual relationships who might require infertility management and fertility care services, and notably marginalized populations.
Addressing infertility also can help mitigate gender inequality, which has significant negative social impacts on the lives of infertile individuals, especially women. Fertility education is important to reduce the fear of infertility and contraception use in those wanting pregnancy in the future.
In most countries the biggest challenges are availability, access, and quality of interventions to address infertility. This includes the United States, where only 1 in 4 individuals receive the fertility care they need. Lack of prioritization, ineffective public health strategies, inadequate funding, and costs are barriers. Health policies need to recognize that infertility is a disease that often can be prevented, thereby reducing future costs. Comprehensive awareness and education programs, laws and policies that regulate and ensure access and the human rights of all involved, are essential.
Advocacy efforts
To address infertility and fertility care, the WHO is committed to:
- collaborate with partners on epidemiologic and etiologic research
- facilitate policy dialogue globally to frame infertility within a legal and policy framework
- support generation of data on the burden of infertility
- develop guidelines
- produce other documents of standards
- collaborate with all stakeholders to strengthen political commitment and health system capacity, and
- provide country-level technical support to develop or strengthen policies and services.
For your practice, this means that infertility is recognized as a disease that should receive its appropriate share of health care resources. Infertility and fertility care are the right of every individual according to their desires to found a family. Besides providing the best care you can to all your patients, including referring them when necessary, all health care clinicians should advocate on behalf of their patients to payors, policy makers, and the public the need to provide equitable laws, resources, and funding for infertility and fertility care.
Every person has the right to infertility and fertility care as endorsed by the recent WHO infertility fact sheet. To address this high-burden disease, all women’s health care clinicians should be aware of, equitably diagnose and treat, refer as necessary, and advocate for infertile individuals.
Continue to: Lessons learned in reducing multiple pregnancy rates in infertility treatment...
Lessons learned in reducing multiple pregnancy rates in infertility treatment
Views and reviews section. Fertil Steril. 2020;114:671- 672; 673-679; 680-689; 690-714; 715-721.
In the October 2020 issue of Fertility and Sterility, the Views and Reviews section included 5 articles on avoiding multiple live birth rates (LBRs) in assisted reproductive technologies (ART).1-5 International experts provided a comprehensive review of global multiple LBRs and their associated negative impact on maternal and perinatal outcomes, reasons for global variability, strategies to reduce multiples, single embryo transfer, and implications of funding and reporting. These international comparisons and recommendations are helpful and applicable to infertility care in the United States.3
The rise of multiple birth rates
During the first decade of in vitro fertilization (IVF), live birth rates were low, increasing to 14% in 1990. Multiple embryos needed to be transferred so that even these LBRs could be obtained. In the 1990s, however, laboratory technology improved rapidly, with increased implantation rates and subsequent rapid increases in LBR, but also with increased multiple birth rates (MBRs).
In the United States, clinic-specific reporting helped create competition among clinics for the best LBRs, and this led to MBRs of 30% and higher. Numerous studies documented the associated significantly increased morbidity and mortality of both mothers and babies. Similar situations occurred in many other countries while some, especially Nordic nations, Australia, New Zealand, and Japan, had twin rates of less than 10% or even 5% since the early 2000s. So why the difference?
The higher MBR is due largely to the transfer of more than one embryo. The immediate solution is therefore always to perform elective single embryo transfer (eSET). However, numerous factors affect the decision to perform eSET or not, and this ideal is far from being achieved. Older women, those with longer duration of infertility and/or failed treatment, often feel a time pressure and want to transfer more embryos. Of course, biologically this is reasonable because the number and quality of their embryos is lower. While attempts have been made to assess embryo quality with preimplantation genetic testing for aneuploidy, evidence that this increases the LBR is controversial except possibly in women aged 35 to 38 years. This is especially true when the cumulative LBR, that is, the number of live births after transfer of all embryos from an egg retrieval cycle, is the measured outcome.
The major factor that determines the frequency of eSET is financial. Affordability is the out-of-pocket cost (after insurance or other subsidy) as a percentage of disposable income, and it is the most important factor that determines whether eSET is performed. Less affordable treatment creates a financial incentive to transfer more than one embryo to maximize the pregnancy rates in fewer cycles.5 Other factors include whether the effectiveness of treatment, that is, LBR, is emphasized over safety, that is, MBR. In the United States, the Society for Assisted Reproductive Technology now reports cumulative LBR, singleton and multiple LBR, and preterm births as outcomes, thereby increasing the emphasis on eSET.
Sociologic, cultural, and religious factors also can affect the frequency of eSET. Even within the United States, great variation exists in values and beliefs regarding infertility treatment. It can be challenging to determine who makes decisions: the patient alone, the physician, the payor, professional guidelines, or laws. In many countries, including the United States, it is an amalgam of these.
Setting new goals
If the goal is to reduce the MBR, what should that rate be? In the past few years, the MBR in the United States has been reduced to approximately 10%. It is reasonable now to set a goal of 5% in the next several years. To do this, we can learn from countries that have been successful. The United States already has very high-quality clinical and laboratory services, knowledgeable physicians, and a reasonable regulatory environment. Improved technology, specifically embryo selection for transfer, and focus on adherence to established embryo transfer guidelines could help.
Many would argue that eSET essentially should be performed always in women younger than age 40 and in all women of any age with a known euploid embryo. The major problem that drives multiples is the lack of affordability, which can be addressed by increased subsidies from payors. Increased subsidies can result from legislative mandates or societal pressures on employers, either of which could be associated with requirements for eSET and/or reduced MBRs.
In your practice, you can now reassure your infertility patients that cumulative LBRs are excellent in the United States and that the risk of multiple pregnancy has been reduced dramatically. This should encourage more patients to accept and take advantage of this successful technology that has resulted in the birth of millions of babies globally. Further reduction of the MBR to 5% should be possible within a few years through education and advocacy by women’s health care clinicians that results in increased subsidies and more affordable IVF.
The multiple birth rate in ART has been reduced to 10% in the United States through an increased understanding of the complex factors that affect embryo transfer practices globally. Further progress will depend primarily on increased subsidies that make ART more affordable.
Continue to: Genetics and ART...
Genetics and ART: Selection versus correction
Adashi EY, Cohen IG. The case for remedial germline editing—the long-term view. JAMA. 2020;323:1762-1763.
Rosenbaum L. The future of gene editing—toward scientific and social consensus. N Engl J Med. 2019;380:971-975.
Cyranoski D. The CRISPR-baby scandal: what’s next for human gene-editing. Nature. 2019;566:440-442.
de Wert G, Pennings G, Clarke A, et al; European Society of Human Genetics and European Society of Human Reproduction and Embryology. Human germline gene editing: recommendations of ESHG and ESHRE. Hum Reprod Open. 2018;hox025.
Following the completion of the Human Genome Project in 2003 and major technologic advancements in the subsequent years, the field of human genetics became the focal point of convergence for several distinct but interrelated disciplines: bioinformatics, computational biology, and sequencing technologies. As the result, individual human genomes can now be sequenced at a single base pair level, and with higher fidelity, at a fraction of the original cost and at a much faster speed.
This molecular progress, however, has not been accompanied by an equivalent clinical progress, because in a significant number of cases a defined and predictable clinical phenotype cannot be attributed to a detected molecular genotype. This has resulted in an overabundance of variants of uncertain significance. Variable expressivity, incomplete penetrance, epigenetics, mosaicism, and the polygenic nature of many human traits further complicate reliable interpretation and prognostication of the colossal amount of molecular genetic data that are being generated by the above-mentioned technologic advances.
Considering these limitations, at this juncture it is crucial to acknowledge that any attempts to prematurely commercialize these preclinical and research studies (such as polygenic risk scores for embryos) are perilous and have the potential to cause significant harm in terms of unnecessary stress and anxiety for intended parents as well as the potential for yet-unmapped societal and legal implications.
However, it is just a matter of time until more accurate clinical phenotyping catches up with molecular genotyping. As we get closer to this next historic milestone, precision medicine in the postnatal life (with regard to both diagnostics and therapeutics) and preimplantation genetic testing (PGT) at the prenatal stage for a much wider spectrum of conditions—including both monogenic and polygenic traits—may indeed become a reality.
The potential of germline editing
Specifically regarding PGT (which requires IVF), it is important to recognize that due to the limited and nonrenewable endowment of human oocytes (ovarian reserve), combined with the detrimental impact of advancing age on the quality of the remaining cohort as manifested by a higher risk of aneuploidy, the current clinical practice of trying to “select” a nonaffected embryo can be very inefficient. As a result, the intended parents pursuing such treatments may need to undergo multiple cycles of ovarian stimulation and oocyte retrieval.
A potential solution for genes associated with known diseases is the prospect of remedial germline editing by CRISPR–Cas9 technology or its future descendants. This would take advantage of the existing embryos to try to “correct” the defective gene instead of trying to “select” a normal embryo. These technologies are still in the early stages of development and are remotely distant from clinical applications. On the other hand, although germline gene editing, if actualized, would be a monumental breakthrough in the history of genetics and medicine, we must be cognizant of its serious legal, societal, and ethical ramifications, which are currently unknown. Furthermore, even at the biologic and technical level, the technology still is not advanced enough to reliably rule out off-target modifications, and the unintended clinical consequences of the on-target corrections have not been studied either.
Regulation of genetic modifications
Due to these myriad concerns and the lack of an existing appropriate regulatory framework and oversight for such interventions, current US law (since December 2015, through provisions in annual federal appropriations laws passed by Congress and renewed annually thereafter) bars the US Food and Drug Administration from considering any clinical trial application “in which a human embryo is intentionally created or modified to include a heritable genetic modification.” Notably, this moratorium also prohibits mitochondrial replacement technology (MRT), which is a less controversial and relatively better-studied innovation.
Mitochondrial genetic disorders caused by the mutations in mitochondrial DNA (versus nuclear DNA) are amenable to a specific treatment strategy aimed at substituting the defective maternal mitochondrial genome with the mitochondrial genome of an unaffected donor oocyte. This can be achieved via either pronuclear transfer, which involves isolation and transfer of the male and female pronuclei from an affected embryo to an enucleated normal donor embryo, or maternal spindle transfer, which involves isolation and transfer of the metaphase II spindle complex of an affected oocyte to an enucleated disease-free donor egg. It is noteworthy that in 2015 in the United Kingdom, Parliament expanded the definition of “permitted eggs and embryos” to include those “where unhealthy mitochondrial DNA is replaced by healthy mitochondrial DNA from a donor.” This thereby allows the UK Human Fertilisation and Embryology Authority to formally direct and oversee clinical trials in MRT.
Summing up
Although the future of assisted human reproduction cannot be clearly outlined at this time, it is likely to be radically different from the current state given these emerging applications at the intersection of ART and diagnostic and therapeutic genetics. To ensure that exploring this uncharted territory will ultimately be in the interest of humankind and civilization, proper regulatory oversight—after careful consideration of all ethical, societal, and legal implications—needs to be developed for all preclinical and clinical research in this field. Participatory public engagement must be an integrated part of this process. ●
The field of human genetics has already transformed medicine. However, the convergence of the interrelated disciplines of bioinformatics, computational biology, sequencing technologies, and CRISPR–Cas9 technology is creating incredible new advances that will bring great benefits but also major societal challenges.
- Farquhar C. Avoiding multiple pregnancies in assisted reproductive technologies: transferring one embryo at a time should be the norm. Fertil Steril. 2020;114:671-672.
- Bergh C, Kamath MS, Wang R, et al. Strategies to reduce multiple pregnancies during medically assisted reproduction. Fertil Steril. 2020;114:673-679.
- Adamson GD, Norman RJ. Why are multiple pregnancy rates and single embryo transfer rates so different globally, and what do we do about it? Fertil Steril. 2020;114:680-689.
- Eapen A, Ryan GL, Ten Eyck P, et al. Current evidence supporting a goal of singletons: a review of maternal and neonatal outcomes associated with twin versus singleton pregnancies after in vitro fertilization and intracytoplasmic sperm injection. Fertil Steril. 2020;114: 690-714.
- Chambers GM, Keller E, Choi S, et al. Funding and public reporting strategies for reducing multiple pregnancy from fertility treatments. Fertil Steril. 2020;114:715-721.
- Farquhar C. Avoiding multiple pregnancies in assisted reproductive technologies: transferring one embryo at a time should be the norm. Fertil Steril. 2020;114:671-672.
- Bergh C, Kamath MS, Wang R, et al. Strategies to reduce multiple pregnancies during medically assisted reproduction. Fertil Steril. 2020;114:673-679.
- Adamson GD, Norman RJ. Why are multiple pregnancy rates and single embryo transfer rates so different globally, and what do we do about it? Fertil Steril. 2020;114:680-689.
- Eapen A, Ryan GL, Ten Eyck P, et al. Current evidence supporting a goal of singletons: a review of maternal and neonatal outcomes associated with twin versus singleton pregnancies after in vitro fertilization and intracytoplasmic sperm injection. Fertil Steril. 2020;114: 690-714.
- Chambers GM, Keller E, Choi S, et al. Funding and public reporting strategies for reducing multiple pregnancy from fertility treatments. Fertil Steril. 2020;114:715-721.
2020 Update on fertility
Although we are not able to cover all of the important developments in fertility medicine over the past year, there were 3 important articles published in the past 12 months that we highlight here. First, we discuss an American College of Obstetricians and Gynecologists (ACOG) committee opinion on genetic carrier screening that was reaffirmed in 2019. Second, we explore an interesting retrospective analysis of time-lapse videos and clinical outcomes of more than 10,000 embryos from 8 IVF clinics, across 4 countries. The authors assessed whether a deep learning model could predict the probability of pregnancy with fetal heart from time-lapse videos in the hopes that their research can improve prioritization of the most viable embryo for single embryo transfer. Last, we consider a review of the data on obstetric and reproductive health effects of preconception and prenatal exposure to several environmental toxicants, including heavy metals, endocrine-disrupting chemicals, pesticides, and air pollution.
Preconception genetic carrier screening: Standardize your counseling approach
American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 690: carrier screening in the age of genomic medicine. Obstet Gynecol. 2017;129:e35-e40.
With the rapid development of advanced and high throughput platforms for DNA sequencing in the past several years, the cost of genetic testing has decreased dramatically. Women's health care providers in general, and fertility specialists in particular, are uniquely positioned to take advantage of these novel and yet affordable technologies by counseling prospective parents during the preconception counseling, or early prenatal period, about the availability of genetic carrier screening and its potential to provide actionable information in a timely manner. The ultimate objective of genetic carrier screening is to enable individuals to make an informed decision regarding their reproductive choices based on their personal values. In a study by Larsen and colleagues, the uptake of genetic carrier screening was significantly higher when offered in the preconception period (68.7%), compared with during pregnancy (35.1%), which highlights the significance of early counseling.1
Based on the Centers for Disease Control and Prevention's Birth/Infant Death Data set, birth defects affect 1 in every 33 (about 3%) of all babies born in the United States each year and account for 20% of infant mortality.2 About 20% of birth defects are caused by single-gene (monogenic) disorders, and although some of these are due to dominant conditions or de novo mutations, a significant proportion are due to autosomal recessive, or X-chromosome linked conditions that are commonly assessed by genetic carrier screening.
ACOG published a committee opinion on "Carrier Screening in the Age of Genomic Medicine" in March 2017, which was reaffirmed in 2019.3
Residual risk. Several points discussed in this document are of paramount importance, including the need for pretest and posttest counseling and consent, as well as a discussion of "residual risk." Newer platforms employ sequencing techniques that potentially can detect most, if not all, of the disease-causing variants in the tested genes, such as the gene for cystic fibrosis and, therefore, have a higher detection rate compared with the older PCR-based techniques for a limited number of specific mutations included in the panel. Due to a variety of technical and biological limitations, however, such as allelic dropouts and the occurrence of de novo mutations, the detection rate is not 100%; there is always a residual risk that needs to be estimated and provided to individuals based on the existing knowledge on frequency of gene, penetrance of phenotype, and prevalence of condition in the general and specific ethnic populations.
Continue to: Expanded vs panethnic screening...
Expanded vs panethnic screening. Furthermore, although sequencing technology has made "expanded carrier screening" for several hundred conditions, simultaneous to and independent of ethnicity and family history, more easily available and affordable, ethnic-specific and panethnic screening for a more limited number of conditions are still acceptable approaches. Having said this, when the first partner screened is identified to be a carrier, his/her reproductive partners must be offered next-generation sequencing to identify less common disease-causing variants.4
A cautionary point to consider when expanded carrier screening panels are requested is the significant variability among commercial laboratories with regard to the conditions included in their panels. In addition, consider the absence of a well-defined or predictable phenotype for some of the included conditions.
Perhaps the most important matter when it comes to genetic carrier screening is to have a standard counseling approach that is persistently followed and offers the opportunity for individuals to know about their genetic testing options and available reproductive choices, including the use of donor gametes, preimplantation genetic testing for monogenic disease (PGT-M, formerly known as preimplantation genetic diagnosis, or PGD), prenatal testing, and pregnancy management options. For couples and/or individuals who decide to proceed with an affected pregnancy, earlier diagnosis can assist with postnatal management.
Medicolegal responsibility. Genetic carrier screening also is of specific relevance to the field of fertility medicine and assisted reproductive technology (ART) as a potential liability issue. Couples and individuals who are undergoing fertility treatment with in vitro fertilization (IVF) for a variety of medical or personal reasons are a specific group that certainly should be offered genetic carrier screening, as they have the option of "adding on" PGT-M (PGD) to their existing treatment plan at a fraction of the cost and treatment burden that would have otherwise been needed if they were not undergoing IVF. After counseling, some individuals and couples may ultimately opt out of genetic carrier screening. The counseling discussion needs to be clearly documented in the medical chart.
The preconception period is the perfect time to have a discussion about genetic carrier screening; it offers the opportunity for timely interventions if desired by the couples or individuals.
Continue to: Artificial intelligence and embryo selection...
Artificial intelligence and embryo selection
With continued improvements in embryo culture conditions and cryopreservation technology, there has been a tremendous amount of interest in developing better methods for embryo selection. These efforts are aimed at encouraging elective single embryo transfer (eSET) for women of all ages, thereby lowering the risk of multiple pregnancy and its associated adverse neonatal and obstetric outcomes—without compromising the pregnancy rates per transfer or lengthening the time to pregnancy.
One of the most extensively studied methods for this purpose is preimplantation genetic testing for aneuploidy (PGT-A, formerly known as PGS), but emerging data from large multicenter randomized clinical trials (RCTs) have again cast significant doubt on PGT-A's efficacy and utility.5 Meanwhile, alternative methods for embryo selection are currently under investigation, including noninvasive PGT-A and morphokinetic assessment of embryo development via analysis of images obtained by time-lapse imaging.
The potential of time-lapse imaging
Despite the initial promising results from time-lapse imaging, subsequent RCTs have not shown a significant clinical benefit.6 However, these early methods of morphokinetic assessment are mainly dependent on the embryologists' subjective assessment of individual static frames and "annotation" of observed spatial and temporal features of embryo development. In addition to being a very time-consuming task, this process is subject to significant interobserver and intraobserver variability.
Considering these limitations, even machine-based algorithms that incorporate these annotations along with such other clinical variables as parental age and prior obstetric history, have a low predictive power for the outcome of embryo transfer, with an area under the curve (AUC) of the ROC curve of 0.65 to 0.74. (An AUC of 0.5 represents completely random prediction and an AUC of 1.0 suggests perfect prediction.)7
A recent study by Tran and colleagues has employed a deep learning (neural network) model to analyze the entire raw time-lapse videos in an automated manner without prior annotation by embryologists. After analysis of 10,638 embryos from 8 different IVF clinics in 4 different countries, they have reported an AUC of 0.93 (95% confidence interval, 0.92-0.94) for prediction of fetal heart rate activity detected at 7 weeks of gestation or beyond. Although these data are very preliminary and have not yet been validated prospectively in larger datasets for live birth, it may herald the beginning of a new era for the automation and standardization of embryo assessment with artificial intelligence—similar to the rapidly increasing role of facial recognition technology for various applications.
Improved standardization of noninvasive embryo selection with growing use of artificial intelligence is a promising new tool to improve the safety and efficacy of ART.
Continue to: Environmental toxicants: The hidden danger...
Environmental toxicants: The hidden danger
Segal TR, Giudice LC. Before the beginning: environmental exposures and reproductive and obstetrical outcomes. Fertil Steril. 2019;112:613-621.
We receive news daily about the existential risk to humans of climate change. However, a risk that is likely as serious goes almost unseen by the public and most health care providers. That risk is environmental toxicants.8
More than 80,000 chemicals are registered in the United States, most in the last 75 years. These chemicals are ubiquitous. All of us are continuously exposed to and suffused with these toxicants and their metabolites. Air pollution adds insult to injury. Since this exposure has especially significant implications for fertility, infertility, pregnancy, perinatal health, childhood development, adult diseases, and later generational reproduction, it is imperative that reproductive health professionals take responsibility for helping mitigate this environmental crisis.
The problem is exceptionally complicated
The risks posed by environmental toxicants are much less visible than those for climate change, so the public, policymakers, and providers are largely unaware or may even seem uncaring. Few health professionals have sufficient knowledge to deliver care in this area, know which questions to ask, or have adequate information/medical record tools to assist them in care—and what are the possible interventions?
Addressing risk posed by individual toxicants
Addressing the problem clinically requires asking patients questions about exposure and recommending interventions. Toxicant chemicals include the neurotoxin mercury, which can be addressed by limiting intake of fish, especially certain types.
Lead was used before 1978 in paint, it also was used in gas and in water pipes. People living in older homes may be exposed, as well as those in occupations exposed to lead. Others with lead exposure risk include immigrants from areas without lead regulations and people using pica- or lead-glazed pottery. Lead exposure has been associated with multiple pregnancy complications and permanently impaired intellectual development in children. If lead testing reveals high levels, chelation therapy can help.
Cadmium is a heavy metal used in rechargeable batteries, paint pigment, and plastic production. Exposure results from food intake, smoking, and second-hand smoke. Cadmium accumulates in the liver, kidneys, testes, ovaries, and placenta. Exposure causes itai-itai disease, which is characterized by osteomalacia and renal tubular dysfunction as well as epigenetic changes in placental DNA and damage to the reproductive system. Eating organic food and reducing industrial exposure to cadmium are preventive strategies.
Pesticides are ubiquitous, with 90% of the US population having detectable levels. Exposure during the preconception period can lead to intrauterine growth restriction, low birth weight, subsequent cancers, and other problems. Eating organic food can reduce risk, as can frequent hand washing when exposed to pesticides, using protective gear, and removing shoes in the home.
Endocrine-disrupting chemicals (EDCs) are chemicals that can mimic or block endogenous hormones, which leads to adverse health outcomes. In addition to heavy metals, 3 important EDCs are bisphenol A (BPA), phthalates, and polybrominated diethyl ethers (PBDEs). Exposure is ubiquitous from industrial food processing, personal care products, cosmetics, and dust. Phthalates and BPA have short half-lives of hours to days, while PBDEs can persist in adipose tissue for months. Abnormal urogenital and neurologic development and thyroid disruption can result. Eating organic food, eating at home, and decreasing processed food intake can reduce exposure.
BPA is used in plastics, canned food liners, cash register receipts, and epoxy resins. Exposure is through inhalation, ingestion, and dermal absorption and affects semen quality, fertilization, placentation, and early reproduction. Limiting the use of plastic containers, not microwaving food in plastic, and avoiding thermal paper cash register receipts can reduce exposure.
Phthalates are synthetically derived and used as plasticizers in personal and medical products. The major source of phthalate exposure is food; exposure causes sperm, egg, and DNA damage. Phthalate avoidance involves replacing plastic bottles with glass or stainless steel, avoiding reheating food in plastic containers, and choosing "fragrance free" products.
PBDEs are used in flame retardants on upholstery, textiles, carpeting, and some electronics. Most PBDEs have been replaced by alternatives; however, their half-life is up to 12 years. Complications caused by PBDEs include thyroid disruption, resulting in abnormal fetal brain development. Avoiding dust and furniture that contain PBDEs, as well as hand washing, reduces exposure risk.
Air pollutants are associated with adverse obstetric outcomes and lower cognitive function in children. Avoiding areas with heavy traffic, staying indoors when air is heavily polluted, and using a HEPA filter in the home can reduce chemicals from air pollution.
Recommendations
The magnitude of the problem that environmental toxicant exposure creates requires health care providers to take action. The table in the publication by Segal and Giudice can be used as a tool that patients can answer first themselves before review by their provider.2 It can be added to your electronic health record and/or patient portal. Even making general comments to raise awareness, asking questions regarding exposure, and making recommendations can be helpful (TABLES 1 and 2). When possible, we also should advocate for public awareness and policy changes that address this significant health issue.


Environmental toxicants are a significant health problem that can be effectively mitigated through patient questions and recommended interventions.
- Larsen D, Ma J, Strassberg M, et al. The uptake of pan-ethnic expanded carrier screening is higher when offered during preconception or early prenatal genetic counseling. Prenat Diagn. 2019;39:319-323.
- Matthews TJ, MacDorman MF, Thoma ME. Infant Mortality Statistics From the 2013 Period Linked Birth/Infant Death Data Set. Natl Vital Stat Rep. 2015;64:1-30.
- American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 690: carrier screening in the age of genomic medicine. Obstet Gynecol. 2017;129:e35-e40.
- Gregg AR, Edwards JG. Prenatal genetic carrier screening in the genomic age. Semin Perinatol. 2018;42:303-306.
- Munné S, Kaplan B, Frattarelli JL, et al; STAR Study Group. Preimplantation genetic testing for aneuploidy versus morphology as selection criteria for single frozen-thawed embryo transfer in good-prognosis patients: a multicenter randomized clinical trial. Fertil Steril. 2019;112:1071-1079. e7.
- Goodman LR, Goldberg J, Falcone T, et al. Does the addition of time-lapse morphokinetics in the selection of embryos for transfer improve pregnancy rates? A randomized controlled trial. Fertil Steril. 2016;105:275-285.e10.
- Blank C, Wildeboer RR, DeCroo I, et al. Prediction of implantation after blastocyst transfer in in vitro fertilization: a machine-learning perspective. Fertil Steril. 2019;111:318- 326.
- The American College of Obstetricians and Gynecologists Committee on Health Care for Underserved Women; American Society for Reproductive Medicine Practice Committee; The University of California, San Francisco Program on Reproductive Health and the Environment. ACOG Committee Opinion No. 575. Exposure to environmental toxic agents. Fertil Steril. 2013;100:931-934.
Although we are not able to cover all of the important developments in fertility medicine over the past year, there were 3 important articles published in the past 12 months that we highlight here. First, we discuss an American College of Obstetricians and Gynecologists (ACOG) committee opinion on genetic carrier screening that was reaffirmed in 2019. Second, we explore an interesting retrospective analysis of time-lapse videos and clinical outcomes of more than 10,000 embryos from 8 IVF clinics, across 4 countries. The authors assessed whether a deep learning model could predict the probability of pregnancy with fetal heart from time-lapse videos in the hopes that their research can improve prioritization of the most viable embryo for single embryo transfer. Last, we consider a review of the data on obstetric and reproductive health effects of preconception and prenatal exposure to several environmental toxicants, including heavy metals, endocrine-disrupting chemicals, pesticides, and air pollution.
Preconception genetic carrier screening: Standardize your counseling approach
American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 690: carrier screening in the age of genomic medicine. Obstet Gynecol. 2017;129:e35-e40.
With the rapid development of advanced and high throughput platforms for DNA sequencing in the past several years, the cost of genetic testing has decreased dramatically. Women's health care providers in general, and fertility specialists in particular, are uniquely positioned to take advantage of these novel and yet affordable technologies by counseling prospective parents during the preconception counseling, or early prenatal period, about the availability of genetic carrier screening and its potential to provide actionable information in a timely manner. The ultimate objective of genetic carrier screening is to enable individuals to make an informed decision regarding their reproductive choices based on their personal values. In a study by Larsen and colleagues, the uptake of genetic carrier screening was significantly higher when offered in the preconception period (68.7%), compared with during pregnancy (35.1%), which highlights the significance of early counseling.1
Based on the Centers for Disease Control and Prevention's Birth/Infant Death Data set, birth defects affect 1 in every 33 (about 3%) of all babies born in the United States each year and account for 20% of infant mortality.2 About 20% of birth defects are caused by single-gene (monogenic) disorders, and although some of these are due to dominant conditions or de novo mutations, a significant proportion are due to autosomal recessive, or X-chromosome linked conditions that are commonly assessed by genetic carrier screening.
ACOG published a committee opinion on "Carrier Screening in the Age of Genomic Medicine" in March 2017, which was reaffirmed in 2019.3
Residual risk. Several points discussed in this document are of paramount importance, including the need for pretest and posttest counseling and consent, as well as a discussion of "residual risk." Newer platforms employ sequencing techniques that potentially can detect most, if not all, of the disease-causing variants in the tested genes, such as the gene for cystic fibrosis and, therefore, have a higher detection rate compared with the older PCR-based techniques for a limited number of specific mutations included in the panel. Due to a variety of technical and biological limitations, however, such as allelic dropouts and the occurrence of de novo mutations, the detection rate is not 100%; there is always a residual risk that needs to be estimated and provided to individuals based on the existing knowledge on frequency of gene, penetrance of phenotype, and prevalence of condition in the general and specific ethnic populations.
Continue to: Expanded vs panethnic screening...
Expanded vs panethnic screening. Furthermore, although sequencing technology has made "expanded carrier screening" for several hundred conditions, simultaneous to and independent of ethnicity and family history, more easily available and affordable, ethnic-specific and panethnic screening for a more limited number of conditions are still acceptable approaches. Having said this, when the first partner screened is identified to be a carrier, his/her reproductive partners must be offered next-generation sequencing to identify less common disease-causing variants.4
A cautionary point to consider when expanded carrier screening panels are requested is the significant variability among commercial laboratories with regard to the conditions included in their panels. In addition, consider the absence of a well-defined or predictable phenotype for some of the included conditions.
Perhaps the most important matter when it comes to genetic carrier screening is to have a standard counseling approach that is persistently followed and offers the opportunity for individuals to know about their genetic testing options and available reproductive choices, including the use of donor gametes, preimplantation genetic testing for monogenic disease (PGT-M, formerly known as preimplantation genetic diagnosis, or PGD), prenatal testing, and pregnancy management options. For couples and/or individuals who decide to proceed with an affected pregnancy, earlier diagnosis can assist with postnatal management.
Medicolegal responsibility. Genetic carrier screening also is of specific relevance to the field of fertility medicine and assisted reproductive technology (ART) as a potential liability issue. Couples and individuals who are undergoing fertility treatment with in vitro fertilization (IVF) for a variety of medical or personal reasons are a specific group that certainly should be offered genetic carrier screening, as they have the option of "adding on" PGT-M (PGD) to their existing treatment plan at a fraction of the cost and treatment burden that would have otherwise been needed if they were not undergoing IVF. After counseling, some individuals and couples may ultimately opt out of genetic carrier screening. The counseling discussion needs to be clearly documented in the medical chart.
The preconception period is the perfect time to have a discussion about genetic carrier screening; it offers the opportunity for timely interventions if desired by the couples or individuals.
Continue to: Artificial intelligence and embryo selection...
Artificial intelligence and embryo selection
With continued improvements in embryo culture conditions and cryopreservation technology, there has been a tremendous amount of interest in developing better methods for embryo selection. These efforts are aimed at encouraging elective single embryo transfer (eSET) for women of all ages, thereby lowering the risk of multiple pregnancy and its associated adverse neonatal and obstetric outcomes—without compromising the pregnancy rates per transfer or lengthening the time to pregnancy.
One of the most extensively studied methods for this purpose is preimplantation genetic testing for aneuploidy (PGT-A, formerly known as PGS), but emerging data from large multicenter randomized clinical trials (RCTs) have again cast significant doubt on PGT-A's efficacy and utility.5 Meanwhile, alternative methods for embryo selection are currently under investigation, including noninvasive PGT-A and morphokinetic assessment of embryo development via analysis of images obtained by time-lapse imaging.
The potential of time-lapse imaging
Despite the initial promising results from time-lapse imaging, subsequent RCTs have not shown a significant clinical benefit.6 However, these early methods of morphokinetic assessment are mainly dependent on the embryologists' subjective assessment of individual static frames and "annotation" of observed spatial and temporal features of embryo development. In addition to being a very time-consuming task, this process is subject to significant interobserver and intraobserver variability.
Considering these limitations, even machine-based algorithms that incorporate these annotations along with such other clinical variables as parental age and prior obstetric history, have a low predictive power for the outcome of embryo transfer, with an area under the curve (AUC) of the ROC curve of 0.65 to 0.74. (An AUC of 0.5 represents completely random prediction and an AUC of 1.0 suggests perfect prediction.)7
A recent study by Tran and colleagues has employed a deep learning (neural network) model to analyze the entire raw time-lapse videos in an automated manner without prior annotation by embryologists. After analysis of 10,638 embryos from 8 different IVF clinics in 4 different countries, they have reported an AUC of 0.93 (95% confidence interval, 0.92-0.94) for prediction of fetal heart rate activity detected at 7 weeks of gestation or beyond. Although these data are very preliminary and have not yet been validated prospectively in larger datasets for live birth, it may herald the beginning of a new era for the automation and standardization of embryo assessment with artificial intelligence—similar to the rapidly increasing role of facial recognition technology for various applications.
Improved standardization of noninvasive embryo selection with growing use of artificial intelligence is a promising new tool to improve the safety and efficacy of ART.
Continue to: Environmental toxicants: The hidden danger...
Environmental toxicants: The hidden danger
Segal TR, Giudice LC. Before the beginning: environmental exposures and reproductive and obstetrical outcomes. Fertil Steril. 2019;112:613-621.
We receive news daily about the existential risk to humans of climate change. However, a risk that is likely as serious goes almost unseen by the public and most health care providers. That risk is environmental toxicants.8
More than 80,000 chemicals are registered in the United States, most in the last 75 years. These chemicals are ubiquitous. All of us are continuously exposed to and suffused with these toxicants and their metabolites. Air pollution adds insult to injury. Since this exposure has especially significant implications for fertility, infertility, pregnancy, perinatal health, childhood development, adult diseases, and later generational reproduction, it is imperative that reproductive health professionals take responsibility for helping mitigate this environmental crisis.
The problem is exceptionally complicated
The risks posed by environmental toxicants are much less visible than those for climate change, so the public, policymakers, and providers are largely unaware or may even seem uncaring. Few health professionals have sufficient knowledge to deliver care in this area, know which questions to ask, or have adequate information/medical record tools to assist them in care—and what are the possible interventions?
Addressing risk posed by individual toxicants
Addressing the problem clinically requires asking patients questions about exposure and recommending interventions. Toxicant chemicals include the neurotoxin mercury, which can be addressed by limiting intake of fish, especially certain types.
Lead was used before 1978 in paint, it also was used in gas and in water pipes. People living in older homes may be exposed, as well as those in occupations exposed to lead. Others with lead exposure risk include immigrants from areas without lead regulations and people using pica- or lead-glazed pottery. Lead exposure has been associated with multiple pregnancy complications and permanently impaired intellectual development in children. If lead testing reveals high levels, chelation therapy can help.
Cadmium is a heavy metal used in rechargeable batteries, paint pigment, and plastic production. Exposure results from food intake, smoking, and second-hand smoke. Cadmium accumulates in the liver, kidneys, testes, ovaries, and placenta. Exposure causes itai-itai disease, which is characterized by osteomalacia and renal tubular dysfunction as well as epigenetic changes in placental DNA and damage to the reproductive system. Eating organic food and reducing industrial exposure to cadmium are preventive strategies.
Pesticides are ubiquitous, with 90% of the US population having detectable levels. Exposure during the preconception period can lead to intrauterine growth restriction, low birth weight, subsequent cancers, and other problems. Eating organic food can reduce risk, as can frequent hand washing when exposed to pesticides, using protective gear, and removing shoes in the home.
Endocrine-disrupting chemicals (EDCs) are chemicals that can mimic or block endogenous hormones, which leads to adverse health outcomes. In addition to heavy metals, 3 important EDCs are bisphenol A (BPA), phthalates, and polybrominated diethyl ethers (PBDEs). Exposure is ubiquitous from industrial food processing, personal care products, cosmetics, and dust. Phthalates and BPA have short half-lives of hours to days, while PBDEs can persist in adipose tissue for months. Abnormal urogenital and neurologic development and thyroid disruption can result. Eating organic food, eating at home, and decreasing processed food intake can reduce exposure.
BPA is used in plastics, canned food liners, cash register receipts, and epoxy resins. Exposure is through inhalation, ingestion, and dermal absorption and affects semen quality, fertilization, placentation, and early reproduction. Limiting the use of plastic containers, not microwaving food in plastic, and avoiding thermal paper cash register receipts can reduce exposure.
Phthalates are synthetically derived and used as plasticizers in personal and medical products. The major source of phthalate exposure is food; exposure causes sperm, egg, and DNA damage. Phthalate avoidance involves replacing plastic bottles with glass or stainless steel, avoiding reheating food in plastic containers, and choosing "fragrance free" products.
PBDEs are used in flame retardants on upholstery, textiles, carpeting, and some electronics. Most PBDEs have been replaced by alternatives; however, their half-life is up to 12 years. Complications caused by PBDEs include thyroid disruption, resulting in abnormal fetal brain development. Avoiding dust and furniture that contain PBDEs, as well as hand washing, reduces exposure risk.
Air pollutants are associated with adverse obstetric outcomes and lower cognitive function in children. Avoiding areas with heavy traffic, staying indoors when air is heavily polluted, and using a HEPA filter in the home can reduce chemicals from air pollution.
Recommendations
The magnitude of the problem that environmental toxicant exposure creates requires health care providers to take action. The table in the publication by Segal and Giudice can be used as a tool that patients can answer first themselves before review by their provider.2 It can be added to your electronic health record and/or patient portal. Even making general comments to raise awareness, asking questions regarding exposure, and making recommendations can be helpful (TABLES 1 and 2). When possible, we also should advocate for public awareness and policy changes that address this significant health issue.


Environmental toxicants are a significant health problem that can be effectively mitigated through patient questions and recommended interventions.
Although we are not able to cover all of the important developments in fertility medicine over the past year, there were 3 important articles published in the past 12 months that we highlight here. First, we discuss an American College of Obstetricians and Gynecologists (ACOG) committee opinion on genetic carrier screening that was reaffirmed in 2019. Second, we explore an interesting retrospective analysis of time-lapse videos and clinical outcomes of more than 10,000 embryos from 8 IVF clinics, across 4 countries. The authors assessed whether a deep learning model could predict the probability of pregnancy with fetal heart from time-lapse videos in the hopes that their research can improve prioritization of the most viable embryo for single embryo transfer. Last, we consider a review of the data on obstetric and reproductive health effects of preconception and prenatal exposure to several environmental toxicants, including heavy metals, endocrine-disrupting chemicals, pesticides, and air pollution.
Preconception genetic carrier screening: Standardize your counseling approach
American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 690: carrier screening in the age of genomic medicine. Obstet Gynecol. 2017;129:e35-e40.
With the rapid development of advanced and high throughput platforms for DNA sequencing in the past several years, the cost of genetic testing has decreased dramatically. Women's health care providers in general, and fertility specialists in particular, are uniquely positioned to take advantage of these novel and yet affordable technologies by counseling prospective parents during the preconception counseling, or early prenatal period, about the availability of genetic carrier screening and its potential to provide actionable information in a timely manner. The ultimate objective of genetic carrier screening is to enable individuals to make an informed decision regarding their reproductive choices based on their personal values. In a study by Larsen and colleagues, the uptake of genetic carrier screening was significantly higher when offered in the preconception period (68.7%), compared with during pregnancy (35.1%), which highlights the significance of early counseling.1
Based on the Centers for Disease Control and Prevention's Birth/Infant Death Data set, birth defects affect 1 in every 33 (about 3%) of all babies born in the United States each year and account for 20% of infant mortality.2 About 20% of birth defects are caused by single-gene (monogenic) disorders, and although some of these are due to dominant conditions or de novo mutations, a significant proportion are due to autosomal recessive, or X-chromosome linked conditions that are commonly assessed by genetic carrier screening.
ACOG published a committee opinion on "Carrier Screening in the Age of Genomic Medicine" in March 2017, which was reaffirmed in 2019.3
Residual risk. Several points discussed in this document are of paramount importance, including the need for pretest and posttest counseling and consent, as well as a discussion of "residual risk." Newer platforms employ sequencing techniques that potentially can detect most, if not all, of the disease-causing variants in the tested genes, such as the gene for cystic fibrosis and, therefore, have a higher detection rate compared with the older PCR-based techniques for a limited number of specific mutations included in the panel. Due to a variety of technical and biological limitations, however, such as allelic dropouts and the occurrence of de novo mutations, the detection rate is not 100%; there is always a residual risk that needs to be estimated and provided to individuals based on the existing knowledge on frequency of gene, penetrance of phenotype, and prevalence of condition in the general and specific ethnic populations.
Continue to: Expanded vs panethnic screening...
Expanded vs panethnic screening. Furthermore, although sequencing technology has made "expanded carrier screening" for several hundred conditions, simultaneous to and independent of ethnicity and family history, more easily available and affordable, ethnic-specific and panethnic screening for a more limited number of conditions are still acceptable approaches. Having said this, when the first partner screened is identified to be a carrier, his/her reproductive partners must be offered next-generation sequencing to identify less common disease-causing variants.4
A cautionary point to consider when expanded carrier screening panels are requested is the significant variability among commercial laboratories with regard to the conditions included in their panels. In addition, consider the absence of a well-defined or predictable phenotype for some of the included conditions.
Perhaps the most important matter when it comes to genetic carrier screening is to have a standard counseling approach that is persistently followed and offers the opportunity for individuals to know about their genetic testing options and available reproductive choices, including the use of donor gametes, preimplantation genetic testing for monogenic disease (PGT-M, formerly known as preimplantation genetic diagnosis, or PGD), prenatal testing, and pregnancy management options. For couples and/or individuals who decide to proceed with an affected pregnancy, earlier diagnosis can assist with postnatal management.
Medicolegal responsibility. Genetic carrier screening also is of specific relevance to the field of fertility medicine and assisted reproductive technology (ART) as a potential liability issue. Couples and individuals who are undergoing fertility treatment with in vitro fertilization (IVF) for a variety of medical or personal reasons are a specific group that certainly should be offered genetic carrier screening, as they have the option of "adding on" PGT-M (PGD) to their existing treatment plan at a fraction of the cost and treatment burden that would have otherwise been needed if they were not undergoing IVF. After counseling, some individuals and couples may ultimately opt out of genetic carrier screening. The counseling discussion needs to be clearly documented in the medical chart.
The preconception period is the perfect time to have a discussion about genetic carrier screening; it offers the opportunity for timely interventions if desired by the couples or individuals.
Continue to: Artificial intelligence and embryo selection...
Artificial intelligence and embryo selection
With continued improvements in embryo culture conditions and cryopreservation technology, there has been a tremendous amount of interest in developing better methods for embryo selection. These efforts are aimed at encouraging elective single embryo transfer (eSET) for women of all ages, thereby lowering the risk of multiple pregnancy and its associated adverse neonatal and obstetric outcomes—without compromising the pregnancy rates per transfer or lengthening the time to pregnancy.
One of the most extensively studied methods for this purpose is preimplantation genetic testing for aneuploidy (PGT-A, formerly known as PGS), but emerging data from large multicenter randomized clinical trials (RCTs) have again cast significant doubt on PGT-A's efficacy and utility.5 Meanwhile, alternative methods for embryo selection are currently under investigation, including noninvasive PGT-A and morphokinetic assessment of embryo development via analysis of images obtained by time-lapse imaging.
The potential of time-lapse imaging
Despite the initial promising results from time-lapse imaging, subsequent RCTs have not shown a significant clinical benefit.6 However, these early methods of morphokinetic assessment are mainly dependent on the embryologists' subjective assessment of individual static frames and "annotation" of observed spatial and temporal features of embryo development. In addition to being a very time-consuming task, this process is subject to significant interobserver and intraobserver variability.
Considering these limitations, even machine-based algorithms that incorporate these annotations along with such other clinical variables as parental age and prior obstetric history, have a low predictive power for the outcome of embryo transfer, with an area under the curve (AUC) of the ROC curve of 0.65 to 0.74. (An AUC of 0.5 represents completely random prediction and an AUC of 1.0 suggests perfect prediction.)7
A recent study by Tran and colleagues has employed a deep learning (neural network) model to analyze the entire raw time-lapse videos in an automated manner without prior annotation by embryologists. After analysis of 10,638 embryos from 8 different IVF clinics in 4 different countries, they have reported an AUC of 0.93 (95% confidence interval, 0.92-0.94) for prediction of fetal heart rate activity detected at 7 weeks of gestation or beyond. Although these data are very preliminary and have not yet been validated prospectively in larger datasets for live birth, it may herald the beginning of a new era for the automation and standardization of embryo assessment with artificial intelligence—similar to the rapidly increasing role of facial recognition technology for various applications.
Improved standardization of noninvasive embryo selection with growing use of artificial intelligence is a promising new tool to improve the safety and efficacy of ART.
Continue to: Environmental toxicants: The hidden danger...
Environmental toxicants: The hidden danger
Segal TR, Giudice LC. Before the beginning: environmental exposures and reproductive and obstetrical outcomes. Fertil Steril. 2019;112:613-621.
We receive news daily about the existential risk to humans of climate change. However, a risk that is likely as serious goes almost unseen by the public and most health care providers. That risk is environmental toxicants.8
More than 80,000 chemicals are registered in the United States, most in the last 75 years. These chemicals are ubiquitous. All of us are continuously exposed to and suffused with these toxicants and their metabolites. Air pollution adds insult to injury. Since this exposure has especially significant implications for fertility, infertility, pregnancy, perinatal health, childhood development, adult diseases, and later generational reproduction, it is imperative that reproductive health professionals take responsibility for helping mitigate this environmental crisis.
The problem is exceptionally complicated
The risks posed by environmental toxicants are much less visible than those for climate change, so the public, policymakers, and providers are largely unaware or may even seem uncaring. Few health professionals have sufficient knowledge to deliver care in this area, know which questions to ask, or have adequate information/medical record tools to assist them in care—and what are the possible interventions?
Addressing risk posed by individual toxicants
Addressing the problem clinically requires asking patients questions about exposure and recommending interventions. Toxicant chemicals include the neurotoxin mercury, which can be addressed by limiting intake of fish, especially certain types.
Lead was used before 1978 in paint, it also was used in gas and in water pipes. People living in older homes may be exposed, as well as those in occupations exposed to lead. Others with lead exposure risk include immigrants from areas without lead regulations and people using pica- or lead-glazed pottery. Lead exposure has been associated with multiple pregnancy complications and permanently impaired intellectual development in children. If lead testing reveals high levels, chelation therapy can help.
Cadmium is a heavy metal used in rechargeable batteries, paint pigment, and plastic production. Exposure results from food intake, smoking, and second-hand smoke. Cadmium accumulates in the liver, kidneys, testes, ovaries, and placenta. Exposure causes itai-itai disease, which is characterized by osteomalacia and renal tubular dysfunction as well as epigenetic changes in placental DNA and damage to the reproductive system. Eating organic food and reducing industrial exposure to cadmium are preventive strategies.
Pesticides are ubiquitous, with 90% of the US population having detectable levels. Exposure during the preconception period can lead to intrauterine growth restriction, low birth weight, subsequent cancers, and other problems. Eating organic food can reduce risk, as can frequent hand washing when exposed to pesticides, using protective gear, and removing shoes in the home.
Endocrine-disrupting chemicals (EDCs) are chemicals that can mimic or block endogenous hormones, which leads to adverse health outcomes. In addition to heavy metals, 3 important EDCs are bisphenol A (BPA), phthalates, and polybrominated diethyl ethers (PBDEs). Exposure is ubiquitous from industrial food processing, personal care products, cosmetics, and dust. Phthalates and BPA have short half-lives of hours to days, while PBDEs can persist in adipose tissue for months. Abnormal urogenital and neurologic development and thyroid disruption can result. Eating organic food, eating at home, and decreasing processed food intake can reduce exposure.
BPA is used in plastics, canned food liners, cash register receipts, and epoxy resins. Exposure is through inhalation, ingestion, and dermal absorption and affects semen quality, fertilization, placentation, and early reproduction. Limiting the use of plastic containers, not microwaving food in plastic, and avoiding thermal paper cash register receipts can reduce exposure.
Phthalates are synthetically derived and used as plasticizers in personal and medical products. The major source of phthalate exposure is food; exposure causes sperm, egg, and DNA damage. Phthalate avoidance involves replacing plastic bottles with glass or stainless steel, avoiding reheating food in plastic containers, and choosing "fragrance free" products.
PBDEs are used in flame retardants on upholstery, textiles, carpeting, and some electronics. Most PBDEs have been replaced by alternatives; however, their half-life is up to 12 years. Complications caused by PBDEs include thyroid disruption, resulting in abnormal fetal brain development. Avoiding dust and furniture that contain PBDEs, as well as hand washing, reduces exposure risk.
Air pollutants are associated with adverse obstetric outcomes and lower cognitive function in children. Avoiding areas with heavy traffic, staying indoors when air is heavily polluted, and using a HEPA filter in the home can reduce chemicals from air pollution.
Recommendations
The magnitude of the problem that environmental toxicant exposure creates requires health care providers to take action. The table in the publication by Segal and Giudice can be used as a tool that patients can answer first themselves before review by their provider.2 It can be added to your electronic health record and/or patient portal. Even making general comments to raise awareness, asking questions regarding exposure, and making recommendations can be helpful (TABLES 1 and 2). When possible, we also should advocate for public awareness and policy changes that address this significant health issue.


Environmental toxicants are a significant health problem that can be effectively mitigated through patient questions and recommended interventions.
- Larsen D, Ma J, Strassberg M, et al. The uptake of pan-ethnic expanded carrier screening is higher when offered during preconception or early prenatal genetic counseling. Prenat Diagn. 2019;39:319-323.
- Matthews TJ, MacDorman MF, Thoma ME. Infant Mortality Statistics From the 2013 Period Linked Birth/Infant Death Data Set. Natl Vital Stat Rep. 2015;64:1-30.
- American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 690: carrier screening in the age of genomic medicine. Obstet Gynecol. 2017;129:e35-e40.
- Gregg AR, Edwards JG. Prenatal genetic carrier screening in the genomic age. Semin Perinatol. 2018;42:303-306.
- Munné S, Kaplan B, Frattarelli JL, et al; STAR Study Group. Preimplantation genetic testing for aneuploidy versus morphology as selection criteria for single frozen-thawed embryo transfer in good-prognosis patients: a multicenter randomized clinical trial. Fertil Steril. 2019;112:1071-1079. e7.
- Goodman LR, Goldberg J, Falcone T, et al. Does the addition of time-lapse morphokinetics in the selection of embryos for transfer improve pregnancy rates? A randomized controlled trial. Fertil Steril. 2016;105:275-285.e10.
- Blank C, Wildeboer RR, DeCroo I, et al. Prediction of implantation after blastocyst transfer in in vitro fertilization: a machine-learning perspective. Fertil Steril. 2019;111:318- 326.
- The American College of Obstetricians and Gynecologists Committee on Health Care for Underserved Women; American Society for Reproductive Medicine Practice Committee; The University of California, San Francisco Program on Reproductive Health and the Environment. ACOG Committee Opinion No. 575. Exposure to environmental toxic agents. Fertil Steril. 2013;100:931-934.
- Larsen D, Ma J, Strassberg M, et al. The uptake of pan-ethnic expanded carrier screening is higher when offered during preconception or early prenatal genetic counseling. Prenat Diagn. 2019;39:319-323.
- Matthews TJ, MacDorman MF, Thoma ME. Infant Mortality Statistics From the 2013 Period Linked Birth/Infant Death Data Set. Natl Vital Stat Rep. 2015;64:1-30.
- American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 690: carrier screening in the age of genomic medicine. Obstet Gynecol. 2017;129:e35-e40.
- Gregg AR, Edwards JG. Prenatal genetic carrier screening in the genomic age. Semin Perinatol. 2018;42:303-306.
- Munné S, Kaplan B, Frattarelli JL, et al; STAR Study Group. Preimplantation genetic testing for aneuploidy versus morphology as selection criteria for single frozen-thawed embryo transfer in good-prognosis patients: a multicenter randomized clinical trial. Fertil Steril. 2019;112:1071-1079. e7.
- Goodman LR, Goldberg J, Falcone T, et al. Does the addition of time-lapse morphokinetics in the selection of embryos for transfer improve pregnancy rates? A randomized controlled trial. Fertil Steril. 2016;105:275-285.e10.
- Blank C, Wildeboer RR, DeCroo I, et al. Prediction of implantation after blastocyst transfer in in vitro fertilization: a machine-learning perspective. Fertil Steril. 2019;111:318- 326.
- The American College of Obstetricians and Gynecologists Committee on Health Care for Underserved Women; American Society for Reproductive Medicine Practice Committee; The University of California, San Francisco Program on Reproductive Health and the Environment. ACOG Committee Opinion No. 575. Exposure to environmental toxic agents. Fertil Steril. 2013;100:931-934.