User login
What’s the best way to manage upper extremity venous thrombosis?
STANDARD MANAGEMENT IS BEST: Start with unfractionated heparin or low-molecular-weight heparin and follow with long-term therapy with a vitamin K antagonist (strength of recommendation [SOR]: C, expert consensus and case reports). Some evidence supports thrombolytic therapy, placement of a superior vena cava filter, or surgical thrombectomy in selected patients (SOR: C, expert consensus and case reports). Whether to remove venous catheters during initial treatment for catheter-induced venous thrombosis remains unclear, because limited studies address this issue specifically (SOR: C, expert consensus and case reports).
Evidence summary
Upper extremity venous thrombosis (UEVT)—which typically refers to thrombosis of the brachial, axillary, or subclavian veins—accounts for approximately 10% of all cases of venous thromboembolism.1 UEVT can occur spontaneously (Paget-Schroetter syndrome) or develop as a complication of cancer or in-dwelling medical devices (such as long-term central venous catheters).
Although significant evidence-based data exist on treatment of lower extremity venous thrombosis, no good-quality studies specifically address management of UEVT. Review of the current literature revealed several small studies that compared various treatment strategies.
Thrombolytics may work better than anticoagulants in some patients
A small retrospective study randomized 20 patients with UEVT to either treatment with anticoagulant therapy with heparin and warfarin (n=11) or thrombolytic therapy (n=9).2 After a mean follow-up period of 81.7 months for the anticoagulation group and 52.1 months for the thrombolytic group, 4 more patients in the thrombolytic group achieved complete clinical recovery and vein patency than in the anticoagulant group (P=.04). When patients who recovered completely were added to those who showed some clinical improvement, 89% of the thrombolytic therapy group had satisfactory outcomes, compared with 36% of the anticoagulant group (P=.028).
Another small retrospective study looked at 10 consecutive patients with UEVT who were treated with either anticoagulant therapy (n=6) or thrombolytics (n=4).3 Fifty percent of patients treated with anticoagulants experienced partial or complete improvement in symptoms, whereas 100% of patients treated with thrombolytics had partial or complete resolution of both symptoms and thrombi.
Overall, both studies raise the possibility that thrombolytic therapy is more effective than anticoagulation therapy in certain patients. The studies evaluated only active patients, 23 to 58 years of age, who had no contraindications to thrombolytic therapy. Neither study reported data on long-term outcomes such as recurrences, bleeding, or post-thrombotic sequelae.
Surgery may avoid long-term anticoagulation
Two case studies evaluated treatment of UEVT with thrombolytic therapy followed by various surgical interventions (angioplasty, thrombectomy, decompression via first rib resection or anterior scalenectomy, and venous bypass).4,5 The first study reported that 8 of 9 patients who underwent first-rib resection and 1 of 2 who underwent scalenectomy were free of residual symptoms at follow-up (mean 2 years, range 6 months to 5 years).4 All patients were treated with thrombolytics before surgery.
The second study demonstrated that 50% of the patients treated with a surgical intervention without thrombolysis had complete symptom relief, while the remaining 50% reported relief from pain but still had occasional swelling.5 Although more invasive, surgery may eliminate the need for long-term anticoagulation therapy and enable a more rapid return to normal activities.
Data on vena cava filters are limited
Data supporting superior vena cava Green-field filters to treat UEVT are extremely limited. Of 6 patients with contraindications to anticoagulation therapy who were treated with a Greenfield filter, none had clinical evidence of pulmonary embolism at 14 months.6 The study reported no data regarding long-term sequelae or effects of the UEVT on the patients’ upper extremity.6
When in doubt, don’t (necessarily) take it out
For patients with central venous catheter-related deep vein thrombosis, taking out the catheter as part of treatment is controversial and should depend on clinical symptoms, the reason for the catheter, and duration of use, as well as physician judgment.7 No RCTs have studied the effects of catheter removal as part of initial treatment. Current guidelines recommend removing the catheter from patients with persistent symptoms who have failed anticoagulant or thrombolytic treatment.1
Recommendations
The American College of Chest Physicians recommends therapeutic doses of intravenous unfractionated heparin, low-molecular-weight heparin, or fondaparinux for initial treatment of UEVT, followed by at least 3 months of treatment with a vitamin K antagonist.1 The organization also suggests that surgical thrombectomy, superior vena caval filters, thrombolytic therapy, or catheter extraction may benefit selected patients.
Acknowledgement
The opinions and assertions contained herein are the private views of the author and not to be construed as official, or as reflecting the views of the US Air Force Medical Service or the US Air Force at large.
1. Hirsh J, Guyatt G, Albers GW, et al. Executive summary: American College of Chest Physicians evidence-based clinical practice guidelines, 8th ed. Chest. 2008;133(6 suppl):71S-109S.
2. Petrakis IE, Katsamouris A, Kafassis E, et al. Two different therapeutic modalities in the treatment of the upper extremity deep vein thrombosis: preliminary investigation with 20 case reports. Int J Angiol. 2000;9:46-50.
3. AbuRahma AF, Sadler D, Stuart P, et al. Conventional versus thrombolytic therapy in spontaneous (effort) axillary-subclavian vein thrombosis. Am J Surg. 1991;161:459-465.
4. Lee MC, Grassi CJ, Belkin M, et al. Early operative intervention after thrombolytic therapy for primary subclavian vein thrombosis: an effective treatment approach. J Vasc Surg. 1998;27:1101-1107.
5. Sanders RJ, Cooper MA. Surgical management of subclavian vein obstruction, including six cases of subclavian vein bypass. Surgery. 1995;118:856-863.
6. Ascer E, Gennaro M, Lorensen E, et al. Superior vena caval Green-field filters: indications, techniques, and results. J Vasc Surg. 1996;23:498-503.
7. Verso M, Agnelli G. Venous thromboembolism associated with long-term use of central venous catheters in cancer patients. J Clin Oncol. 2003;21:3665-3675.
STANDARD MANAGEMENT IS BEST: Start with unfractionated heparin or low-molecular-weight heparin and follow with long-term therapy with a vitamin K antagonist (strength of recommendation [SOR]: C, expert consensus and case reports). Some evidence supports thrombolytic therapy, placement of a superior vena cava filter, or surgical thrombectomy in selected patients (SOR: C, expert consensus and case reports). Whether to remove venous catheters during initial treatment for catheter-induced venous thrombosis remains unclear, because limited studies address this issue specifically (SOR: C, expert consensus and case reports).
Evidence summary
Upper extremity venous thrombosis (UEVT)—which typically refers to thrombosis of the brachial, axillary, or subclavian veins—accounts for approximately 10% of all cases of venous thromboembolism.1 UEVT can occur spontaneously (Paget-Schroetter syndrome) or develop as a complication of cancer or in-dwelling medical devices (such as long-term central venous catheters).
Although significant evidence-based data exist on treatment of lower extremity venous thrombosis, no good-quality studies specifically address management of UEVT. Review of the current literature revealed several small studies that compared various treatment strategies.
Thrombolytics may work better than anticoagulants in some patients
A small retrospective study randomized 20 patients with UEVT to either treatment with anticoagulant therapy with heparin and warfarin (n=11) or thrombolytic therapy (n=9).2 After a mean follow-up period of 81.7 months for the anticoagulation group and 52.1 months for the thrombolytic group, 4 more patients in the thrombolytic group achieved complete clinical recovery and vein patency than in the anticoagulant group (P=.04). When patients who recovered completely were added to those who showed some clinical improvement, 89% of the thrombolytic therapy group had satisfactory outcomes, compared with 36% of the anticoagulant group (P=.028).
Another small retrospective study looked at 10 consecutive patients with UEVT who were treated with either anticoagulant therapy (n=6) or thrombolytics (n=4).3 Fifty percent of patients treated with anticoagulants experienced partial or complete improvement in symptoms, whereas 100% of patients treated with thrombolytics had partial or complete resolution of both symptoms and thrombi.
Overall, both studies raise the possibility that thrombolytic therapy is more effective than anticoagulation therapy in certain patients. The studies evaluated only active patients, 23 to 58 years of age, who had no contraindications to thrombolytic therapy. Neither study reported data on long-term outcomes such as recurrences, bleeding, or post-thrombotic sequelae.
Surgery may avoid long-term anticoagulation
Two case studies evaluated treatment of UEVT with thrombolytic therapy followed by various surgical interventions (angioplasty, thrombectomy, decompression via first rib resection or anterior scalenectomy, and venous bypass).4,5 The first study reported that 8 of 9 patients who underwent first-rib resection and 1 of 2 who underwent scalenectomy were free of residual symptoms at follow-up (mean 2 years, range 6 months to 5 years).4 All patients were treated with thrombolytics before surgery.
The second study demonstrated that 50% of the patients treated with a surgical intervention without thrombolysis had complete symptom relief, while the remaining 50% reported relief from pain but still had occasional swelling.5 Although more invasive, surgery may eliminate the need for long-term anticoagulation therapy and enable a more rapid return to normal activities.
Data on vena cava filters are limited
Data supporting superior vena cava Green-field filters to treat UEVT are extremely limited. Of 6 patients with contraindications to anticoagulation therapy who were treated with a Greenfield filter, none had clinical evidence of pulmonary embolism at 14 months.6 The study reported no data regarding long-term sequelae or effects of the UEVT on the patients’ upper extremity.6
When in doubt, don’t (necessarily) take it out
For patients with central venous catheter-related deep vein thrombosis, taking out the catheter as part of treatment is controversial and should depend on clinical symptoms, the reason for the catheter, and duration of use, as well as physician judgment.7 No RCTs have studied the effects of catheter removal as part of initial treatment. Current guidelines recommend removing the catheter from patients with persistent symptoms who have failed anticoagulant or thrombolytic treatment.1
Recommendations
The American College of Chest Physicians recommends therapeutic doses of intravenous unfractionated heparin, low-molecular-weight heparin, or fondaparinux for initial treatment of UEVT, followed by at least 3 months of treatment with a vitamin K antagonist.1 The organization also suggests that surgical thrombectomy, superior vena caval filters, thrombolytic therapy, or catheter extraction may benefit selected patients.
Acknowledgement
The opinions and assertions contained herein are the private views of the author and not to be construed as official, or as reflecting the views of the US Air Force Medical Service or the US Air Force at large.
STANDARD MANAGEMENT IS BEST: Start with unfractionated heparin or low-molecular-weight heparin and follow with long-term therapy with a vitamin K antagonist (strength of recommendation [SOR]: C, expert consensus and case reports). Some evidence supports thrombolytic therapy, placement of a superior vena cava filter, or surgical thrombectomy in selected patients (SOR: C, expert consensus and case reports). Whether to remove venous catheters during initial treatment for catheter-induced venous thrombosis remains unclear, because limited studies address this issue specifically (SOR: C, expert consensus and case reports).
Evidence summary
Upper extremity venous thrombosis (UEVT)—which typically refers to thrombosis of the brachial, axillary, or subclavian veins—accounts for approximately 10% of all cases of venous thromboembolism.1 UEVT can occur spontaneously (Paget-Schroetter syndrome) or develop as a complication of cancer or in-dwelling medical devices (such as long-term central venous catheters).
Although significant evidence-based data exist on treatment of lower extremity venous thrombosis, no good-quality studies specifically address management of UEVT. Review of the current literature revealed several small studies that compared various treatment strategies.
Thrombolytics may work better than anticoagulants in some patients
A small retrospective study randomized 20 patients with UEVT to either treatment with anticoagulant therapy with heparin and warfarin (n=11) or thrombolytic therapy (n=9).2 After a mean follow-up period of 81.7 months for the anticoagulation group and 52.1 months for the thrombolytic group, 4 more patients in the thrombolytic group achieved complete clinical recovery and vein patency than in the anticoagulant group (P=.04). When patients who recovered completely were added to those who showed some clinical improvement, 89% of the thrombolytic therapy group had satisfactory outcomes, compared with 36% of the anticoagulant group (P=.028).
Another small retrospective study looked at 10 consecutive patients with UEVT who were treated with either anticoagulant therapy (n=6) or thrombolytics (n=4).3 Fifty percent of patients treated with anticoagulants experienced partial or complete improvement in symptoms, whereas 100% of patients treated with thrombolytics had partial or complete resolution of both symptoms and thrombi.
Overall, both studies raise the possibility that thrombolytic therapy is more effective than anticoagulation therapy in certain patients. The studies evaluated only active patients, 23 to 58 years of age, who had no contraindications to thrombolytic therapy. Neither study reported data on long-term outcomes such as recurrences, bleeding, or post-thrombotic sequelae.
Surgery may avoid long-term anticoagulation
Two case studies evaluated treatment of UEVT with thrombolytic therapy followed by various surgical interventions (angioplasty, thrombectomy, decompression via first rib resection or anterior scalenectomy, and venous bypass).4,5 The first study reported that 8 of 9 patients who underwent first-rib resection and 1 of 2 who underwent scalenectomy were free of residual symptoms at follow-up (mean 2 years, range 6 months to 5 years).4 All patients were treated with thrombolytics before surgery.
The second study demonstrated that 50% of the patients treated with a surgical intervention without thrombolysis had complete symptom relief, while the remaining 50% reported relief from pain but still had occasional swelling.5 Although more invasive, surgery may eliminate the need for long-term anticoagulation therapy and enable a more rapid return to normal activities.
Data on vena cava filters are limited
Data supporting superior vena cava Green-field filters to treat UEVT are extremely limited. Of 6 patients with contraindications to anticoagulation therapy who were treated with a Greenfield filter, none had clinical evidence of pulmonary embolism at 14 months.6 The study reported no data regarding long-term sequelae or effects of the UEVT on the patients’ upper extremity.6
When in doubt, don’t (necessarily) take it out
For patients with central venous catheter-related deep vein thrombosis, taking out the catheter as part of treatment is controversial and should depend on clinical symptoms, the reason for the catheter, and duration of use, as well as physician judgment.7 No RCTs have studied the effects of catheter removal as part of initial treatment. Current guidelines recommend removing the catheter from patients with persistent symptoms who have failed anticoagulant or thrombolytic treatment.1
Recommendations
The American College of Chest Physicians recommends therapeutic doses of intravenous unfractionated heparin, low-molecular-weight heparin, or fondaparinux for initial treatment of UEVT, followed by at least 3 months of treatment with a vitamin K antagonist.1 The organization also suggests that surgical thrombectomy, superior vena caval filters, thrombolytic therapy, or catheter extraction may benefit selected patients.
Acknowledgement
The opinions and assertions contained herein are the private views of the author and not to be construed as official, or as reflecting the views of the US Air Force Medical Service or the US Air Force at large.
1. Hirsh J, Guyatt G, Albers GW, et al. Executive summary: American College of Chest Physicians evidence-based clinical practice guidelines, 8th ed. Chest. 2008;133(6 suppl):71S-109S.
2. Petrakis IE, Katsamouris A, Kafassis E, et al. Two different therapeutic modalities in the treatment of the upper extremity deep vein thrombosis: preliminary investigation with 20 case reports. Int J Angiol. 2000;9:46-50.
3. AbuRahma AF, Sadler D, Stuart P, et al. Conventional versus thrombolytic therapy in spontaneous (effort) axillary-subclavian vein thrombosis. Am J Surg. 1991;161:459-465.
4. Lee MC, Grassi CJ, Belkin M, et al. Early operative intervention after thrombolytic therapy for primary subclavian vein thrombosis: an effective treatment approach. J Vasc Surg. 1998;27:1101-1107.
5. Sanders RJ, Cooper MA. Surgical management of subclavian vein obstruction, including six cases of subclavian vein bypass. Surgery. 1995;118:856-863.
6. Ascer E, Gennaro M, Lorensen E, et al. Superior vena caval Green-field filters: indications, techniques, and results. J Vasc Surg. 1996;23:498-503.
7. Verso M, Agnelli G. Venous thromboembolism associated with long-term use of central venous catheters in cancer patients. J Clin Oncol. 2003;21:3665-3675.
1. Hirsh J, Guyatt G, Albers GW, et al. Executive summary: American College of Chest Physicians evidence-based clinical practice guidelines, 8th ed. Chest. 2008;133(6 suppl):71S-109S.
2. Petrakis IE, Katsamouris A, Kafassis E, et al. Two different therapeutic modalities in the treatment of the upper extremity deep vein thrombosis: preliminary investigation with 20 case reports. Int J Angiol. 2000;9:46-50.
3. AbuRahma AF, Sadler D, Stuart P, et al. Conventional versus thrombolytic therapy in spontaneous (effort) axillary-subclavian vein thrombosis. Am J Surg. 1991;161:459-465.
4. Lee MC, Grassi CJ, Belkin M, et al. Early operative intervention after thrombolytic therapy for primary subclavian vein thrombosis: an effective treatment approach. J Vasc Surg. 1998;27:1101-1107.
5. Sanders RJ, Cooper MA. Surgical management of subclavian vein obstruction, including six cases of subclavian vein bypass. Surgery. 1995;118:856-863.
6. Ascer E, Gennaro M, Lorensen E, et al. Superior vena caval Green-field filters: indications, techniques, and results. J Vasc Surg. 1996;23:498-503.
7. Verso M, Agnelli G. Venous thromboembolism associated with long-term use of central venous catheters in cancer patients. J Clin Oncol. 2003;21:3665-3675.
Evidence-based answers from the Family Physicians Inquiries Network
How accurate is an MRI at diagnosing injured knee ligaments?
It is highly accurate in diagnosing injury to the anterior cruciate ligament (ACL) (strength of recommendation [SOR]: A, prospective blinded cohort studies) and posterior cruciate ligament (PCL) (SOR: B, limited number of prospective blinded cohort studies).
Insufficient data are available to evaluate the effectiveness of magnetic resonance imaging (MRI) for diagnosing injuries to the medial collateral ligament (MCL) and lateral collateral ligament (LCL).
Evidence summary
Ligamentous knee injuries from trauma are common. In 2003, patients made about 19.4 million visits to the doctor because of knee problems.1 The ACL is the most often injured knee ligament. The incidence of ACL injury is approximately 200,000 annually in the United States; 100,000 ACL reconstructions are performed each year.2,3 A complete tear of the ACL can lead to significant knee instability and, unless repaired, may limit physical activity and quality of life.
In contrast, PCL injuries don’t often cause significant instability and generally respond to nonsurgical treatment; they have less impact on a patient’s quality of life. Surgery for PCL injury is usually reserved for elite athletes and unstable injuries. MCL and LCL injuries also are generally treated nonsurgically with rehabilitation and bracing; they normally don’t require arthroscopic evaluation and repair.
An effective alternative to arthroscopy
Arthroscopy with direct visualization of the ligamentous structures is considered the gold standard for diagnosing intra-articular ligamentous knee injuries, but it’s invasive and costly. Although clinical examination is helpful in identifying injured ligaments, it may lead to unnecessary arthroscopies when used alone because of the high false-positive rate. MRI has been shown to be an effective tool for accurately diagnosing ligamentous knee injury.2,3
MRI offers high sensitivity for detecting ACL, PCL tears
Several prospective studies have compared MRI with arthroscopy for diagnosing ACL and PCL tears (TABLE).4-8 All enrolled patients had sustained knee trauma and had had a clinical exam that suggested ligamentous injury. MRI and arthroscopy were performed regardless of MRI findings. The surgeons performing arthroscopy were blinded to the MRI results.
Although MRI equipment and techniques varied in all the studies, the sensitivity and specificity remained consistently high for detecting ACL injuries. Thin-slice views, special oblique views, and a fast spin-echo technique didn’t improve either the sensitivity or specificity compared with conventional techniques or views, nor did decreasing the time interval from injury to imaging.5,9,10 Prospective studies of PCL injuries also revealed high sensitivity and specificity with MRI.
No data on MRI for MCL and LCL injuries
No prospective studies are available to assess the accuracy of MRI for suspected MCL and LCL injuries; however, MRI would likely not affect treatment or clinical outcomes, as both of these injuries are typically treated nonsurgically.
Recommendations
The American Academy of Orthopaedic Surgeons supports MRI as an effective tool for evaluating knee injury,1 and offers recommendations and guidelines for treating ligamentous knee injury based on the findings of clinical examination and MRI. The Academy states that MRI is invaluable in preventing unnecessary surgery, and recommends it whenever ligamentous injury is suspected.
Acknowledgements
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the US Air Force medical Service or the US Air Force at large.
1. American Academy of Orthopaedic Surgeons. Common knee injuries. 2007. Available at: http://orthoinfo.aaos.org/topic.cfm?topic=A00325. Accessed November 9, 2009.
2. Miyasaka KC, Daniel DM, Stone ML. The incidence of knee ligament injuries in the general population. Am J Knee Surg. 1991;4:43-48.
3. Brown CH Jr, Carson EW. Revision anterior cruciate ligament surgery. Clin Sports Med. 1999;18:109-171.
4. Rubin DA, Kettering JM, Towers JD, et al. MR imaging of knees having isolated and combined ligament injuries. AJR Am J Roentgenol. 1998;170:1207-1213.
5. Katahira K, Yamashita Y, Takahashi M, et al. MR imaging of the anterior cruciate ligament: value of thin slice direct oblique coronal technique. Radiat Med. 2001;19:1-7.
6. Munshi M, Davidson M, MacDonald PB, et al. The efficacy of magnetic resonance imaging in acute knee injuries. Clin J Sport Med. 2000;10:34-39.
7. Vaz CE, Camargo OP, Santana PJ, et al. Accuracy of magnetic resonance in identifying traumatic intraarticular knee lesions. Clinics (São Paulo). 2005;60:445-450.
8. Winters K, Tregonning R. Reliability of magnetic resonance imag- ing of the traumatic knee as determined by arthroscopy. N Z Med J. 2005;118:U1301
10. Yoon YC, Kim SS, Chung HW, et al. Diagnostic efficacy in knee MRI comparing conventional technique and multiplanar recon- struction with one-millimeter FSE PDW images. Acta Radiol. 2007;48:869-874.
It is highly accurate in diagnosing injury to the anterior cruciate ligament (ACL) (strength of recommendation [SOR]: A, prospective blinded cohort studies) and posterior cruciate ligament (PCL) (SOR: B, limited number of prospective blinded cohort studies).
Insufficient data are available to evaluate the effectiveness of magnetic resonance imaging (MRI) for diagnosing injuries to the medial collateral ligament (MCL) and lateral collateral ligament (LCL).
Evidence summary
Ligamentous knee injuries from trauma are common. In 2003, patients made about 19.4 million visits to the doctor because of knee problems.1 The ACL is the most often injured knee ligament. The incidence of ACL injury is approximately 200,000 annually in the United States; 100,000 ACL reconstructions are performed each year.2,3 A complete tear of the ACL can lead to significant knee instability and, unless repaired, may limit physical activity and quality of life.
In contrast, PCL injuries don’t often cause significant instability and generally respond to nonsurgical treatment; they have less impact on a patient’s quality of life. Surgery for PCL injury is usually reserved for elite athletes and unstable injuries. MCL and LCL injuries also are generally treated nonsurgically with rehabilitation and bracing; they normally don’t require arthroscopic evaluation and repair.
An effective alternative to arthroscopy
Arthroscopy with direct visualization of the ligamentous structures is considered the gold standard for diagnosing intra-articular ligamentous knee injuries, but it’s invasive and costly. Although clinical examination is helpful in identifying injured ligaments, it may lead to unnecessary arthroscopies when used alone because of the high false-positive rate. MRI has been shown to be an effective tool for accurately diagnosing ligamentous knee injury.2,3
MRI offers high sensitivity for detecting ACL, PCL tears
Several prospective studies have compared MRI with arthroscopy for diagnosing ACL and PCL tears (TABLE).4-8 All enrolled patients had sustained knee trauma and had had a clinical exam that suggested ligamentous injury. MRI and arthroscopy were performed regardless of MRI findings. The surgeons performing arthroscopy were blinded to the MRI results.
Although MRI equipment and techniques varied in all the studies, the sensitivity and specificity remained consistently high for detecting ACL injuries. Thin-slice views, special oblique views, and a fast spin-echo technique didn’t improve either the sensitivity or specificity compared with conventional techniques or views, nor did decreasing the time interval from injury to imaging.5,9,10 Prospective studies of PCL injuries also revealed high sensitivity and specificity with MRI.
No data on MRI for MCL and LCL injuries
No prospective studies are available to assess the accuracy of MRI for suspected MCL and LCL injuries; however, MRI would likely not affect treatment or clinical outcomes, as both of these injuries are typically treated nonsurgically.
Recommendations
The American Academy of Orthopaedic Surgeons supports MRI as an effective tool for evaluating knee injury,1 and offers recommendations and guidelines for treating ligamentous knee injury based on the findings of clinical examination and MRI. The Academy states that MRI is invaluable in preventing unnecessary surgery, and recommends it whenever ligamentous injury is suspected.
Acknowledgements
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the US Air Force medical Service or the US Air Force at large.
It is highly accurate in diagnosing injury to the anterior cruciate ligament (ACL) (strength of recommendation [SOR]: A, prospective blinded cohort studies) and posterior cruciate ligament (PCL) (SOR: B, limited number of prospective blinded cohort studies).
Insufficient data are available to evaluate the effectiveness of magnetic resonance imaging (MRI) for diagnosing injuries to the medial collateral ligament (MCL) and lateral collateral ligament (LCL).
Evidence summary
Ligamentous knee injuries from trauma are common. In 2003, patients made about 19.4 million visits to the doctor because of knee problems.1 The ACL is the most often injured knee ligament. The incidence of ACL injury is approximately 200,000 annually in the United States; 100,000 ACL reconstructions are performed each year.2,3 A complete tear of the ACL can lead to significant knee instability and, unless repaired, may limit physical activity and quality of life.
In contrast, PCL injuries don’t often cause significant instability and generally respond to nonsurgical treatment; they have less impact on a patient’s quality of life. Surgery for PCL injury is usually reserved for elite athletes and unstable injuries. MCL and LCL injuries also are generally treated nonsurgically with rehabilitation and bracing; they normally don’t require arthroscopic evaluation and repair.
An effective alternative to arthroscopy
Arthroscopy with direct visualization of the ligamentous structures is considered the gold standard for diagnosing intra-articular ligamentous knee injuries, but it’s invasive and costly. Although clinical examination is helpful in identifying injured ligaments, it may lead to unnecessary arthroscopies when used alone because of the high false-positive rate. MRI has been shown to be an effective tool for accurately diagnosing ligamentous knee injury.2,3
MRI offers high sensitivity for detecting ACL, PCL tears
Several prospective studies have compared MRI with arthroscopy for diagnosing ACL and PCL tears (TABLE).4-8 All enrolled patients had sustained knee trauma and had had a clinical exam that suggested ligamentous injury. MRI and arthroscopy were performed regardless of MRI findings. The surgeons performing arthroscopy were blinded to the MRI results.
Although MRI equipment and techniques varied in all the studies, the sensitivity and specificity remained consistently high for detecting ACL injuries. Thin-slice views, special oblique views, and a fast spin-echo technique didn’t improve either the sensitivity or specificity compared with conventional techniques or views, nor did decreasing the time interval from injury to imaging.5,9,10 Prospective studies of PCL injuries also revealed high sensitivity and specificity with MRI.
No data on MRI for MCL and LCL injuries
No prospective studies are available to assess the accuracy of MRI for suspected MCL and LCL injuries; however, MRI would likely not affect treatment or clinical outcomes, as both of these injuries are typically treated nonsurgically.
Recommendations
The American Academy of Orthopaedic Surgeons supports MRI as an effective tool for evaluating knee injury,1 and offers recommendations and guidelines for treating ligamentous knee injury based on the findings of clinical examination and MRI. The Academy states that MRI is invaluable in preventing unnecessary surgery, and recommends it whenever ligamentous injury is suspected.
Acknowledgements
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the US Air Force medical Service or the US Air Force at large.
1. American Academy of Orthopaedic Surgeons. Common knee injuries. 2007. Available at: http://orthoinfo.aaos.org/topic.cfm?topic=A00325. Accessed November 9, 2009.
2. Miyasaka KC, Daniel DM, Stone ML. The incidence of knee ligament injuries in the general population. Am J Knee Surg. 1991;4:43-48.
3. Brown CH Jr, Carson EW. Revision anterior cruciate ligament surgery. Clin Sports Med. 1999;18:109-171.
4. Rubin DA, Kettering JM, Towers JD, et al. MR imaging of knees having isolated and combined ligament injuries. AJR Am J Roentgenol. 1998;170:1207-1213.
5. Katahira K, Yamashita Y, Takahashi M, et al. MR imaging of the anterior cruciate ligament: value of thin slice direct oblique coronal technique. Radiat Med. 2001;19:1-7.
6. Munshi M, Davidson M, MacDonald PB, et al. The efficacy of magnetic resonance imaging in acute knee injuries. Clin J Sport Med. 2000;10:34-39.
7. Vaz CE, Camargo OP, Santana PJ, et al. Accuracy of magnetic resonance in identifying traumatic intraarticular knee lesions. Clinics (São Paulo). 2005;60:445-450.
8. Winters K, Tregonning R. Reliability of magnetic resonance imag- ing of the traumatic knee as determined by arthroscopy. N Z Med J. 2005;118:U1301
10. Yoon YC, Kim SS, Chung HW, et al. Diagnostic efficacy in knee MRI comparing conventional technique and multiplanar recon- struction with one-millimeter FSE PDW images. Acta Radiol. 2007;48:869-874.
1. American Academy of Orthopaedic Surgeons. Common knee injuries. 2007. Available at: http://orthoinfo.aaos.org/topic.cfm?topic=A00325. Accessed November 9, 2009.
2. Miyasaka KC, Daniel DM, Stone ML. The incidence of knee ligament injuries in the general population. Am J Knee Surg. 1991;4:43-48.
3. Brown CH Jr, Carson EW. Revision anterior cruciate ligament surgery. Clin Sports Med. 1999;18:109-171.
4. Rubin DA, Kettering JM, Towers JD, et al. MR imaging of knees having isolated and combined ligament injuries. AJR Am J Roentgenol. 1998;170:1207-1213.
5. Katahira K, Yamashita Y, Takahashi M, et al. MR imaging of the anterior cruciate ligament: value of thin slice direct oblique coronal technique. Radiat Med. 2001;19:1-7.
6. Munshi M, Davidson M, MacDonald PB, et al. The efficacy of magnetic resonance imaging in acute knee injuries. Clin J Sport Med. 2000;10:34-39.
7. Vaz CE, Camargo OP, Santana PJ, et al. Accuracy of magnetic resonance in identifying traumatic intraarticular knee lesions. Clinics (São Paulo). 2005;60:445-450.
8. Winters K, Tregonning R. Reliability of magnetic resonance imag- ing of the traumatic knee as determined by arthroscopy. N Z Med J. 2005;118:U1301
10. Yoon YC, Kim SS, Chung HW, et al. Diagnostic efficacy in knee MRI comparing conventional technique and multiplanar recon- struction with one-millimeter FSE PDW images. Acta Radiol. 2007;48:869-874.
Evidence-based answers from the Family Physicians Inquiries Network
What is the best way to screen for breast cancer in women with implants?
Mammography is best. It is considered as effective for screening women who have undergone augmentation mammoplasty as those who have not (strength of recommendation [SOR]: B, limited number of retrospective and prospective cohort studies). This question has not been well studied, however.
Evidence summary
Breast augmentation is one of the most popular plastic surgeries in the United States; an estimated 291,350 such procedures were performed in 2005.1 Breast cancer occurs in 1 of every 8 women; a projected 32,000 women who received breast implants in 2003 will develop cancer.1 Available research has focused on retrospective and prospective designs because of the ethical limitations of experimental designs. No US studies directly compare mammography with alternate screening methods, such as sonography or magnetic resonance imaging.
With implants: Lower screening sensitivity but similar prognosis
Studies show that augmentation decreases the sensitivity of screening mammography but doesn’t affect breast cancer prognosis.2 A 2004 prospective cohort study of 986,270 women found that, among asymptomatic women diagnosed with breast cancer (40 augmented, 238 nonaugmented), the sensitivity of screening mammograms was lower in women with breast implants (45%; 95% confidence interval [CI], 29.3%–61.5%) than those without (66.8%; 95% CI, 60.4%–72.8%); P=.008).2 Similarly, in symptomatic women diagnosed with breast cancer (41 augmented, 145 nonaugmented), screening sensitivity was lower in the augmented women (73.2%) than the nonaugmented women (81.4%)—although the results weren’t significant (P=.25).
Despite lower screening sensitivity, breast tumors in asymptomatic women, whether augmented or not, had similar characteristics, except for larger tumor size (3 mm) at diagnosis in augmented women. Symptomatic women with breast implants had cancers that were smaller, lower-grade, and more likely to be estrogen receptor dependent and invasive (P=.052) compared with nonaugmented women. The authors concluded that augmentation doesn’t influence the prognostic characteristics of tumors, and they recommended screening mammography at appropriate intervals.
Two other prospective cohort studies produced similar findings. A 2006 study of 4082 breast cancer patients concluded that mammography yielded a false-negative rate of 41.4% in augmented patients compared with 8.8% in nonaugmented patients (P<.0001).3 However, both augmented (n=129) and nonaugmented (n=3953) women had a comparable prognosis at diagnosis. The authors of the studies suggested diagnostic mammography for augmented patients and correlation with physical exam findings.
An earlier study of 2956 cancer patients found that mammography detected an abnormal breast mass in 66.3% of augmented women compared with 94.6% of nonaugmented women (P=.001).4 No significant differences were noted in cancer characteristics at diagnosis or survival rates (P=.78). The authors of this study concluded that mammography should be used for augmented women until a more effective screening tool is found.
Sonography vs mammography: The jury is still out
Although studies comparing screening methods have not been performed in the United States, a small Taiwanese study directly compared ultrasound to mammography in 105 women without breast implants. This retrospective cohort study found sonography to be a more useful diagnostic tool than mammography in Taiwanese women.5 Sonography had the highest sensitivity (87.5%) compared to physical examination (50.0%) and mammography (25.5%).
Sonography was recommended as the imaging tool for Asian women with smaller, denser breasts. However, it is unclear whether this result applies to US women or women who have undergone breast augmentation surgery.
Training in implant imaging is needed
Mammography appears to be the most effective screening method for women with breast implants. Despite the small differences in cancer characteristics at diagnosis between augmented and nonaugmented women, overall prognosis and survival rates are no different.1-3,6 This is true whether a screening mammogram or diagnostic mammogram is used. In any case, all available findings suggest that clinicians who perform mammography should be trained in imaging the augmented breast.6-8
Recommendations
The National Cancer Institute indicates that the best screening method for augmented women is mammography performed at a facility with employees trained in implant imaging.7 The American College of Radiology’s practice guidelines affirm that mammography is the best imaging tool available.8 The American College of Obstetrics and Gynecology and the US Preventive Services Task Force don’t comment on screening augmented women.
Acknowledgments
The opinions and assertions contained herein are the private views of the author and not to be construed as official, or as reflecting the views of the US Air Force Medical Service or the US Air Force at large.
1. Tuli R, Flynn RA, Brill KL, et al. Diagnosis, treatment, and management of breast cancer in previously augmented women. Breast J. 2006;12:343-348.
2. Miglioretti DL, Rutter CM, Gellar BM, et al. Effect of breast augmentation on the accuracy of mammography and cancer characteristics. JAMA. 2004;291:442-450.
3. Handel N, Silverstein MJ. Breast cancer diagnosis and prognosis in augmented women. Plast Reconstr Surg. 2006;118:587-593.
4. Skinner KA, Silberman H, Dougherty W, et al. Breast cancer after augmentation mammoplasty. Ann Surg Oncol. 2000;8:138-144.
5. Hou M-F, Ou-Yang F, Chuang C-H, et al. Comparison between sonography and mammography for breast cancer diagnosis in oriental women after augmentation mammaplasty. Ann Plast Surg. 2002;49:120-126.
6. Hoshaw SJ, Klein PJ, Clark BD, et al. Breast implants and cancer: causation, delayed detection, and survival. Plast Reconstr Surg. 2001;107:1393-1407.
7. National Cancer Institute. Screening Mammograms: Questions and Answers. September 4, 2007. Available at: www.cancer.gov/cancertopics/factsheet/Detection/screening-mammograms. Accessed November 2, 2007.
8. American College of Radiology. ACR Practice Guideline (amended 2006). Available at: www.acr.org/SecondaryMainMenuCategories/quality_safety/guidelines/breast/screening_mammography.aspx. Accessed November 2, 2007.
Mammography is best. It is considered as effective for screening women who have undergone augmentation mammoplasty as those who have not (strength of recommendation [SOR]: B, limited number of retrospective and prospective cohort studies). This question has not been well studied, however.
Evidence summary
Breast augmentation is one of the most popular plastic surgeries in the United States; an estimated 291,350 such procedures were performed in 2005.1 Breast cancer occurs in 1 of every 8 women; a projected 32,000 women who received breast implants in 2003 will develop cancer.1 Available research has focused on retrospective and prospective designs because of the ethical limitations of experimental designs. No US studies directly compare mammography with alternate screening methods, such as sonography or magnetic resonance imaging.
With implants: Lower screening sensitivity but similar prognosis
Studies show that augmentation decreases the sensitivity of screening mammography but doesn’t affect breast cancer prognosis.2 A 2004 prospective cohort study of 986,270 women found that, among asymptomatic women diagnosed with breast cancer (40 augmented, 238 nonaugmented), the sensitivity of screening mammograms was lower in women with breast implants (45%; 95% confidence interval [CI], 29.3%–61.5%) than those without (66.8%; 95% CI, 60.4%–72.8%); P=.008).2 Similarly, in symptomatic women diagnosed with breast cancer (41 augmented, 145 nonaugmented), screening sensitivity was lower in the augmented women (73.2%) than the nonaugmented women (81.4%)—although the results weren’t significant (P=.25).
Despite lower screening sensitivity, breast tumors in asymptomatic women, whether augmented or not, had similar characteristics, except for larger tumor size (3 mm) at diagnosis in augmented women. Symptomatic women with breast implants had cancers that were smaller, lower-grade, and more likely to be estrogen receptor dependent and invasive (P=.052) compared with nonaugmented women. The authors concluded that augmentation doesn’t influence the prognostic characteristics of tumors, and they recommended screening mammography at appropriate intervals.
Two other prospective cohort studies produced similar findings. A 2006 study of 4082 breast cancer patients concluded that mammography yielded a false-negative rate of 41.4% in augmented patients compared with 8.8% in nonaugmented patients (P<.0001).3 However, both augmented (n=129) and nonaugmented (n=3953) women had a comparable prognosis at diagnosis. The authors of the studies suggested diagnostic mammography for augmented patients and correlation with physical exam findings.
An earlier study of 2956 cancer patients found that mammography detected an abnormal breast mass in 66.3% of augmented women compared with 94.6% of nonaugmented women (P=.001).4 No significant differences were noted in cancer characteristics at diagnosis or survival rates (P=.78). The authors of this study concluded that mammography should be used for augmented women until a more effective screening tool is found.
Sonography vs mammography: The jury is still out
Although studies comparing screening methods have not been performed in the United States, a small Taiwanese study directly compared ultrasound to mammography in 105 women without breast implants. This retrospective cohort study found sonography to be a more useful diagnostic tool than mammography in Taiwanese women.5 Sonography had the highest sensitivity (87.5%) compared to physical examination (50.0%) and mammography (25.5%).
Sonography was recommended as the imaging tool for Asian women with smaller, denser breasts. However, it is unclear whether this result applies to US women or women who have undergone breast augmentation surgery.
Training in implant imaging is needed
Mammography appears to be the most effective screening method for women with breast implants. Despite the small differences in cancer characteristics at diagnosis between augmented and nonaugmented women, overall prognosis and survival rates are no different.1-3,6 This is true whether a screening mammogram or diagnostic mammogram is used. In any case, all available findings suggest that clinicians who perform mammography should be trained in imaging the augmented breast.6-8
Recommendations
The National Cancer Institute indicates that the best screening method for augmented women is mammography performed at a facility with employees trained in implant imaging.7 The American College of Radiology’s practice guidelines affirm that mammography is the best imaging tool available.8 The American College of Obstetrics and Gynecology and the US Preventive Services Task Force don’t comment on screening augmented women.
Acknowledgments
The opinions and assertions contained herein are the private views of the author and not to be construed as official, or as reflecting the views of the US Air Force Medical Service or the US Air Force at large.
Mammography is best. It is considered as effective for screening women who have undergone augmentation mammoplasty as those who have not (strength of recommendation [SOR]: B, limited number of retrospective and prospective cohort studies). This question has not been well studied, however.
Evidence summary
Breast augmentation is one of the most popular plastic surgeries in the United States; an estimated 291,350 such procedures were performed in 2005.1 Breast cancer occurs in 1 of every 8 women; a projected 32,000 women who received breast implants in 2003 will develop cancer.1 Available research has focused on retrospective and prospective designs because of the ethical limitations of experimental designs. No US studies directly compare mammography with alternate screening methods, such as sonography or magnetic resonance imaging.
With implants: Lower screening sensitivity but similar prognosis
Studies show that augmentation decreases the sensitivity of screening mammography but doesn’t affect breast cancer prognosis.2 A 2004 prospective cohort study of 986,270 women found that, among asymptomatic women diagnosed with breast cancer (40 augmented, 238 nonaugmented), the sensitivity of screening mammograms was lower in women with breast implants (45%; 95% confidence interval [CI], 29.3%–61.5%) than those without (66.8%; 95% CI, 60.4%–72.8%); P=.008).2 Similarly, in symptomatic women diagnosed with breast cancer (41 augmented, 145 nonaugmented), screening sensitivity was lower in the augmented women (73.2%) than the nonaugmented women (81.4%)—although the results weren’t significant (P=.25).
Despite lower screening sensitivity, breast tumors in asymptomatic women, whether augmented or not, had similar characteristics, except for larger tumor size (3 mm) at diagnosis in augmented women. Symptomatic women with breast implants had cancers that were smaller, lower-grade, and more likely to be estrogen receptor dependent and invasive (P=.052) compared with nonaugmented women. The authors concluded that augmentation doesn’t influence the prognostic characteristics of tumors, and they recommended screening mammography at appropriate intervals.
Two other prospective cohort studies produced similar findings. A 2006 study of 4082 breast cancer patients concluded that mammography yielded a false-negative rate of 41.4% in augmented patients compared with 8.8% in nonaugmented patients (P<.0001).3 However, both augmented (n=129) and nonaugmented (n=3953) women had a comparable prognosis at diagnosis. The authors of the studies suggested diagnostic mammography for augmented patients and correlation with physical exam findings.
An earlier study of 2956 cancer patients found that mammography detected an abnormal breast mass in 66.3% of augmented women compared with 94.6% of nonaugmented women (P=.001).4 No significant differences were noted in cancer characteristics at diagnosis or survival rates (P=.78). The authors of this study concluded that mammography should be used for augmented women until a more effective screening tool is found.
Sonography vs mammography: The jury is still out
Although studies comparing screening methods have not been performed in the United States, a small Taiwanese study directly compared ultrasound to mammography in 105 women without breast implants. This retrospective cohort study found sonography to be a more useful diagnostic tool than mammography in Taiwanese women.5 Sonography had the highest sensitivity (87.5%) compared to physical examination (50.0%) and mammography (25.5%).
Sonography was recommended as the imaging tool for Asian women with smaller, denser breasts. However, it is unclear whether this result applies to US women or women who have undergone breast augmentation surgery.
Training in implant imaging is needed
Mammography appears to be the most effective screening method for women with breast implants. Despite the small differences in cancer characteristics at diagnosis between augmented and nonaugmented women, overall prognosis and survival rates are no different.1-3,6 This is true whether a screening mammogram or diagnostic mammogram is used. In any case, all available findings suggest that clinicians who perform mammography should be trained in imaging the augmented breast.6-8
Recommendations
The National Cancer Institute indicates that the best screening method for augmented women is mammography performed at a facility with employees trained in implant imaging.7 The American College of Radiology’s practice guidelines affirm that mammography is the best imaging tool available.8 The American College of Obstetrics and Gynecology and the US Preventive Services Task Force don’t comment on screening augmented women.
Acknowledgments
The opinions and assertions contained herein are the private views of the author and not to be construed as official, or as reflecting the views of the US Air Force Medical Service or the US Air Force at large.
1. Tuli R, Flynn RA, Brill KL, et al. Diagnosis, treatment, and management of breast cancer in previously augmented women. Breast J. 2006;12:343-348.
2. Miglioretti DL, Rutter CM, Gellar BM, et al. Effect of breast augmentation on the accuracy of mammography and cancer characteristics. JAMA. 2004;291:442-450.
3. Handel N, Silverstein MJ. Breast cancer diagnosis and prognosis in augmented women. Plast Reconstr Surg. 2006;118:587-593.
4. Skinner KA, Silberman H, Dougherty W, et al. Breast cancer after augmentation mammoplasty. Ann Surg Oncol. 2000;8:138-144.
5. Hou M-F, Ou-Yang F, Chuang C-H, et al. Comparison between sonography and mammography for breast cancer diagnosis in oriental women after augmentation mammaplasty. Ann Plast Surg. 2002;49:120-126.
6. Hoshaw SJ, Klein PJ, Clark BD, et al. Breast implants and cancer: causation, delayed detection, and survival. Plast Reconstr Surg. 2001;107:1393-1407.
7. National Cancer Institute. Screening Mammograms: Questions and Answers. September 4, 2007. Available at: www.cancer.gov/cancertopics/factsheet/Detection/screening-mammograms. Accessed November 2, 2007.
8. American College of Radiology. ACR Practice Guideline (amended 2006). Available at: www.acr.org/SecondaryMainMenuCategories/quality_safety/guidelines/breast/screening_mammography.aspx. Accessed November 2, 2007.
1. Tuli R, Flynn RA, Brill KL, et al. Diagnosis, treatment, and management of breast cancer in previously augmented women. Breast J. 2006;12:343-348.
2. Miglioretti DL, Rutter CM, Gellar BM, et al. Effect of breast augmentation on the accuracy of mammography and cancer characteristics. JAMA. 2004;291:442-450.
3. Handel N, Silverstein MJ. Breast cancer diagnosis and prognosis in augmented women. Plast Reconstr Surg. 2006;118:587-593.
4. Skinner KA, Silberman H, Dougherty W, et al. Breast cancer after augmentation mammoplasty. Ann Surg Oncol. 2000;8:138-144.
5. Hou M-F, Ou-Yang F, Chuang C-H, et al. Comparison between sonography and mammography for breast cancer diagnosis in oriental women after augmentation mammaplasty. Ann Plast Surg. 2002;49:120-126.
6. Hoshaw SJ, Klein PJ, Clark BD, et al. Breast implants and cancer: causation, delayed detection, and survival. Plast Reconstr Surg. 2001;107:1393-1407.
7. National Cancer Institute. Screening Mammograms: Questions and Answers. September 4, 2007. Available at: www.cancer.gov/cancertopics/factsheet/Detection/screening-mammograms. Accessed November 2, 2007.
8. American College of Radiology. ACR Practice Guideline (amended 2006). Available at: www.acr.org/SecondaryMainMenuCategories/quality_safety/guidelines/breast/screening_mammography.aspx. Accessed November 2, 2007.
Evidence-based answers from the Family Physicians Inquiries Network
Are DMARDs effective for rheumatologic diseases besides rheumatoid arthritis?
It’s unclear whether disease-modifying antirheumatic agents (DMARDs) as first-line therapy in nonrheumatoid rheumatologic diseases are effective because the question has not been studied. As second-line therapy, the use of some DmArDs appears to be beneficial for patients with psoriatic arthritis (strength of recommendation [SOR]: A, based on systematic reviews of good-quality randomized controlled trials) and ankylosing spondylitis (SOR: B, based on systematic reviews of moderate quality trials). Data on the safety and efficacy of DMARDs as second-line therapy for other arthritic conditions is limited (SOR: C, based on small prospective cohort trials).
There are many options, but remember the risks
Richard Hoffman, MD
Chesterfield Family Medicine Residency, Richmond, Va
Traditionally, nonsteroidal anti-inflammatory agents (NSAIDs) have been the mainstay of treatment for rheumatologic disorders other than rheumatoid arthritis. methotrexate has been used in psoriatic arthritis because it also controls the skin disorder; sulfasalazine has been used in arthritis associated with inflammatory bowel disease, as it helps the bowel disorder itself. However, little evidence shows a definitive benefit for the arthritis.
The advent of tumor necrosis factor (TNF) blockers has changed the direction of research in this area; these agents are being used more and more in inflammatory arthritides. While staying up to date on the TNF antagonists, it’s important to remember the complications associated with them—particularly the increased risk of infections and increased propensity for neoplastic disorders. Consider those on TNF blockers as relatively immunosuppressed (number needed to harm [NNH]=59 for infection and 154 for malignancy).1
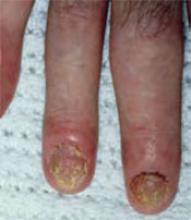
Psoriatic arthritis affecting the joints and nails
Evidence summary
The use of DMARDs has become standard of care for rheumatoid arthritis, for both therapy and prevention of progression of this debilitating disease. However, the use of DMARDs in nonrheumatoid rheumatologic disease is still under investigation, and at this point, the use of DMARDs as first-line therapy is not recommended; however, second-line therapy with DMARDs is common.
For psoriatic arthritis, DMARDs are beneficial as a second-line therapy
A Cochrane systematic review identified 13 randomized controlled trials enrolling a combined 1022 patients with psoriatic arthritis randomly assigned to receive a DMARD—methotrexate, sulfasalazine (Azulfidine), azathioprine (Imuran/ Azasan), or etretinate (Tegison; no longer available in the US)—compared with placebo.2 All agents were better than placebo; however, only 2 agents (parenteral high-dose methotrexate and sulfasalazine) had clinically important benefits for more than half the patients. The studies were too small to establish toxicity or to evaluate the other agents.
NSAIDs are still the preferred first-line therapy, concluded a recent publication on the treatment of psoriatic arthritis, which looked at 54 different studies; however, second-line therapy could include methotrexate, sulfasalazine, etanercept (Enbrel), infliximab (Remicade), cyclosporine, or combination therapy.3 Sulfasalazine appeared to be clinically beneficial for peripheral psoriatic arthritis.
Etanercept vs placebo. An initial study (60 patients) of etanercept vs placebo among patients who were permitted to stay on methotrexate or prednisone showed a response rate of 87% vs 23% (P<.0001; number needed to treat [NNT]=1.56).4
Infliximab vs placebo. A study of infliximab vs placebo involving 104 patients had similar results, with good response in 65% vs 10% (NNT=1.81) at 16 weeks; infliximab also inhibited radiographic progression by 22%.5
Cyclosporine. Although it is effective, reserve cyclosporine for patients who do not improve on other regimens, because of its nephrotoxicity.3
DMARDs show some benefit in treating ankylosing spondylitis
Two recent Cochrane systematic reviews on ankylosing spondylitis examined the use of sulfasalazine and methotrexate as second-line agents.6,7 Eleven trials were included in the sulfasalazine analysis, with a total of 895 patients. Sulfasalazine demonstrated some benefit in reducing erythrocyte sedimentation rates (ESRs) and morning stiffness, but there was no evidence that the drug reduced pain or improved physical function, spinal mobility, or rate of enthesitis. Sulfasalazine was well tolerated and may be useful in early mild disease for patients with peripheral arthritis and high ESRs. On the other hand, evidence was insufficient to determine whether methotrexate benefited patients with ankylosing spondylitis.
In other trials, infliximab and etanercept showed good potential for benefit in treating ankylosing spondylitis.
One study of infliximab vs placebo showed 61.2% vs 19.2% patients with good clinical benefit at 24 weeks and only mild or moderate adverse events (P<.001; NNT=2.38).8
Similarly, a smaller study (84 patients) showed that 60% of patients on etanercept vs 20% on placebo had good clinical benefit at only 12 weeks (P<.001, NNT=2.5).9
For other rheumatic diseases, studies are mixed
Due to cyclosporine’s toxicity, less toxic DMARDs are being evaluated to replace it for treatment of other rheumatic diseases. A recent randomized controlled trial of 100 patients with antineutrophil cytoplasmic antibody–associated systemic vasculitis showed methotrexate may be able to replace cyclosporine for both induction of remission (methotrexate=89.8% vs cyclosporine=93.5%; P=.041) and maintenance of remission (69.5% vs 46.5% at 18 months; P=.023).10
Initial trials on other rheumatic diseases have been small and have had varied results. There are mixed studies on the effectiveness of adding methotrexate to corticosteroids for giant cell arteritis.11,12
There has been no evidence of efficacy for the new TNF antagonists in either a small study on Sjögren’s syndrome (n=14)13 or a larger study on Wegener’s granulomatosis (n=180).14
The studies for use of DMARDs in lupus or scleroderma are of limited quality.
Recommendations from others
The Italian Society for Rheumatology consensus guidelines recommends TNF antagonists be considered in active psoriatic arthritis resistant to (a) NSAIDs, (b) at least 2 local steroid injections, and (c) at least 2 conventional DMARDs for patients with peripheral arthritis or enthesitis. They also recommend TNF antagonists be considered for psoriatic spondylitis resistant to NSAIDs.15
The Assessment in Ankylosing Spondylitis (ASAS) International Working Group and the European League Against Rheumatism (EULAR) recommendations for the treatment of ankylosing spondylitis, based on a systematic review of the literature and expert opinion, indicate that:
- There is good evidence for using NSAIDs and COX-2 inhibitors for symptomatic treatment.
- Conventional DMARDs are not well supported.
- TNF antagonists show a large benefit in both pain and function.
The ASAS/EULAR recommendation indicate that there is no evidence that any of these treatments actually modify the disease progression.16
Acknowledgments
The opinions and assertions contained herein are the private views of the authors and not to be construed as official, or as reflecting the views of the US Air Force medical service or the US Air Force at large.
1. Bongartz T, sutton AJ, Sweeting MJ, et al. Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA 2006;295:2275-2285.
2. Jones G, Crotty M, Brooks P. Interventions for treating psoriatic arthritis. Cochrane Database Syst Rev 2000 (3):CD000212.
3. Manadan Am, Sequeira W, Block JA. The treatment of psoriatic arthritis. Am J Ther 2006;13:72-79.
4. Mease PJ, Goffe BS, Metz J, et al. etanercept in the treatment of psoriatic arthritis and psoriasis: a randomized trial. Lancet 2000;356:385-390.
5. Kavanaugh A, Antoni CE, Gladman D, et al. The Infliximab multinational Psoriatic Arthritis Controlled Trial (ImPACT): result of radiographic analyses after 1 year. Ann Rheum Dis 2006;65:1038-1043.
6. Chen J, Liu C. Sulfasalazine for ankylosing spondylitis. Cochrane Database Syst Rev 2005;(2):CD004800.-
7. Chen J, Liu C, Lin J. Methotrexate for ankylosing spondylitis. Cochrane Database Syst Rev 2006;(4):CD004524.-
8. Heijde D, Dijkmans B, Geusens P, et al. efficacy and safety of infliximab in patients with ankylosing spondylitis. Arthritis Rheum 2005;52:582-591.
9. Calin A, Dijkmans BA, Emery P, et al. Outcomes of a multicentre randomized clinical trial of etanercept to treat ankylosing spondylitis. Ann Rheum Dis 2004;63:1594-1600.
10. Grout K, Rasmussen N, Bacon P, et al. randomized trial of cyclophosphamide versus methotrexate for induction of remission in early systemic antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheum 2005;52:2461-2469.
11. Jover JA, Hernandez-Garcia C, Morado IC, et al. Combined treatment of giant-cell arteritis with methotrexate and prednisone. a randomized, double blinded, placebo-controlled trial. Ann Intern Med 2001;134:106-114.
12. Hoffman GS, Cid MC, Hellmann DB, et al. A multicenter, randomized, double-blind, placebo-controlled trial of adjuvant methotrexate treatment for giant cell arteritis. Arthritis Rheum 2002;46:1309-1318.
13. Sankar V, Brennan MT, Kok MR, et al. etanercept in sjögren’s syndrome: a twelve-week randomized, double-blind, placebo-controlled pilot clinical trial. Arthritis Rheum 2004;50:2240-2245.
14. Wegener’s Granulomatosis etanercept Trial (WGeT) research Group. etanercept plus standard therapy for Wegener’s granulomatosis. N Engl J Med 2005;352:351-361.
15. Salvarani C, Olivieri I, Pipitone N, et al. recommendations of the Italian society for rheumatology for the use of biologic (TNF-alpha blocking) agents in the treatment of psoriatic arthritis. Clin Exp Rheumatol 2006;24:70-78.
16. Zochling J, van der Heijde D, Dougados, et al. Current evidence for the management of ankylosing spondylitis: a systematic literature review for the AsAs/eulAr management recommendations in ankylosing spondylitis. Ann Rheum Dis 2006;65:442-452.
It’s unclear whether disease-modifying antirheumatic agents (DMARDs) as first-line therapy in nonrheumatoid rheumatologic diseases are effective because the question has not been studied. As second-line therapy, the use of some DmArDs appears to be beneficial for patients with psoriatic arthritis (strength of recommendation [SOR]: A, based on systematic reviews of good-quality randomized controlled trials) and ankylosing spondylitis (SOR: B, based on systematic reviews of moderate quality trials). Data on the safety and efficacy of DMARDs as second-line therapy for other arthritic conditions is limited (SOR: C, based on small prospective cohort trials).
There are many options, but remember the risks
Richard Hoffman, MD
Chesterfield Family Medicine Residency, Richmond, Va
Traditionally, nonsteroidal anti-inflammatory agents (NSAIDs) have been the mainstay of treatment for rheumatologic disorders other than rheumatoid arthritis. methotrexate has been used in psoriatic arthritis because it also controls the skin disorder; sulfasalazine has been used in arthritis associated with inflammatory bowel disease, as it helps the bowel disorder itself. However, little evidence shows a definitive benefit for the arthritis.
The advent of tumor necrosis factor (TNF) blockers has changed the direction of research in this area; these agents are being used more and more in inflammatory arthritides. While staying up to date on the TNF antagonists, it’s important to remember the complications associated with them—particularly the increased risk of infections and increased propensity for neoplastic disorders. Consider those on TNF blockers as relatively immunosuppressed (number needed to harm [NNH]=59 for infection and 154 for malignancy).1
Psoriatic arthritis affecting the joints and nails
Evidence summary
The use of DMARDs has become standard of care for rheumatoid arthritis, for both therapy and prevention of progression of this debilitating disease. However, the use of DMARDs in nonrheumatoid rheumatologic disease is still under investigation, and at this point, the use of DMARDs as first-line therapy is not recommended; however, second-line therapy with DMARDs is common.
For psoriatic arthritis, DMARDs are beneficial as a second-line therapy
A Cochrane systematic review identified 13 randomized controlled trials enrolling a combined 1022 patients with psoriatic arthritis randomly assigned to receive a DMARD—methotrexate, sulfasalazine (Azulfidine), azathioprine (Imuran/ Azasan), or etretinate (Tegison; no longer available in the US)—compared with placebo.2 All agents were better than placebo; however, only 2 agents (parenteral high-dose methotrexate and sulfasalazine) had clinically important benefits for more than half the patients. The studies were too small to establish toxicity or to evaluate the other agents.
NSAIDs are still the preferred first-line therapy, concluded a recent publication on the treatment of psoriatic arthritis, which looked at 54 different studies; however, second-line therapy could include methotrexate, sulfasalazine, etanercept (Enbrel), infliximab (Remicade), cyclosporine, or combination therapy.3 Sulfasalazine appeared to be clinically beneficial for peripheral psoriatic arthritis.
Etanercept vs placebo. An initial study (60 patients) of etanercept vs placebo among patients who were permitted to stay on methotrexate or prednisone showed a response rate of 87% vs 23% (P<.0001; number needed to treat [NNT]=1.56).4
Infliximab vs placebo. A study of infliximab vs placebo involving 104 patients had similar results, with good response in 65% vs 10% (NNT=1.81) at 16 weeks; infliximab also inhibited radiographic progression by 22%.5
Cyclosporine. Although it is effective, reserve cyclosporine for patients who do not improve on other regimens, because of its nephrotoxicity.3
DMARDs show some benefit in treating ankylosing spondylitis
Two recent Cochrane systematic reviews on ankylosing spondylitis examined the use of sulfasalazine and methotrexate as second-line agents.6,7 Eleven trials were included in the sulfasalazine analysis, with a total of 895 patients. Sulfasalazine demonstrated some benefit in reducing erythrocyte sedimentation rates (ESRs) and morning stiffness, but there was no evidence that the drug reduced pain or improved physical function, spinal mobility, or rate of enthesitis. Sulfasalazine was well tolerated and may be useful in early mild disease for patients with peripheral arthritis and high ESRs. On the other hand, evidence was insufficient to determine whether methotrexate benefited patients with ankylosing spondylitis.
In other trials, infliximab and etanercept showed good potential for benefit in treating ankylosing spondylitis.
One study of infliximab vs placebo showed 61.2% vs 19.2% patients with good clinical benefit at 24 weeks and only mild or moderate adverse events (P<.001; NNT=2.38).8
Similarly, a smaller study (84 patients) showed that 60% of patients on etanercept vs 20% on placebo had good clinical benefit at only 12 weeks (P<.001, NNT=2.5).9
For other rheumatic diseases, studies are mixed
Due to cyclosporine’s toxicity, less toxic DMARDs are being evaluated to replace it for treatment of other rheumatic diseases. A recent randomized controlled trial of 100 patients with antineutrophil cytoplasmic antibody–associated systemic vasculitis showed methotrexate may be able to replace cyclosporine for both induction of remission (methotrexate=89.8% vs cyclosporine=93.5%; P=.041) and maintenance of remission (69.5% vs 46.5% at 18 months; P=.023).10
Initial trials on other rheumatic diseases have been small and have had varied results. There are mixed studies on the effectiveness of adding methotrexate to corticosteroids for giant cell arteritis.11,12
There has been no evidence of efficacy for the new TNF antagonists in either a small study on Sjögren’s syndrome (n=14)13 or a larger study on Wegener’s granulomatosis (n=180).14
The studies for use of DMARDs in lupus or scleroderma are of limited quality.
Recommendations from others
The Italian Society for Rheumatology consensus guidelines recommends TNF antagonists be considered in active psoriatic arthritis resistant to (a) NSAIDs, (b) at least 2 local steroid injections, and (c) at least 2 conventional DMARDs for patients with peripheral arthritis or enthesitis. They also recommend TNF antagonists be considered for psoriatic spondylitis resistant to NSAIDs.15
The Assessment in Ankylosing Spondylitis (ASAS) International Working Group and the European League Against Rheumatism (EULAR) recommendations for the treatment of ankylosing spondylitis, based on a systematic review of the literature and expert opinion, indicate that:
- There is good evidence for using NSAIDs and COX-2 inhibitors for symptomatic treatment.
- Conventional DMARDs are not well supported.
- TNF antagonists show a large benefit in both pain and function.
The ASAS/EULAR recommendation indicate that there is no evidence that any of these treatments actually modify the disease progression.16
Acknowledgments
The opinions and assertions contained herein are the private views of the authors and not to be construed as official, or as reflecting the views of the US Air Force medical service or the US Air Force at large.
It’s unclear whether disease-modifying antirheumatic agents (DMARDs) as first-line therapy in nonrheumatoid rheumatologic diseases are effective because the question has not been studied. As second-line therapy, the use of some DmArDs appears to be beneficial for patients with psoriatic arthritis (strength of recommendation [SOR]: A, based on systematic reviews of good-quality randomized controlled trials) and ankylosing spondylitis (SOR: B, based on systematic reviews of moderate quality trials). Data on the safety and efficacy of DMARDs as second-line therapy for other arthritic conditions is limited (SOR: C, based on small prospective cohort trials).
There are many options, but remember the risks
Richard Hoffman, MD
Chesterfield Family Medicine Residency, Richmond, Va
Traditionally, nonsteroidal anti-inflammatory agents (NSAIDs) have been the mainstay of treatment for rheumatologic disorders other than rheumatoid arthritis. methotrexate has been used in psoriatic arthritis because it also controls the skin disorder; sulfasalazine has been used in arthritis associated with inflammatory bowel disease, as it helps the bowel disorder itself. However, little evidence shows a definitive benefit for the arthritis.
The advent of tumor necrosis factor (TNF) blockers has changed the direction of research in this area; these agents are being used more and more in inflammatory arthritides. While staying up to date on the TNF antagonists, it’s important to remember the complications associated with them—particularly the increased risk of infections and increased propensity for neoplastic disorders. Consider those on TNF blockers as relatively immunosuppressed (number needed to harm [NNH]=59 for infection and 154 for malignancy).1
Psoriatic arthritis affecting the joints and nails
Evidence summary
The use of DMARDs has become standard of care for rheumatoid arthritis, for both therapy and prevention of progression of this debilitating disease. However, the use of DMARDs in nonrheumatoid rheumatologic disease is still under investigation, and at this point, the use of DMARDs as first-line therapy is not recommended; however, second-line therapy with DMARDs is common.
For psoriatic arthritis, DMARDs are beneficial as a second-line therapy
A Cochrane systematic review identified 13 randomized controlled trials enrolling a combined 1022 patients with psoriatic arthritis randomly assigned to receive a DMARD—methotrexate, sulfasalazine (Azulfidine), azathioprine (Imuran/ Azasan), or etretinate (Tegison; no longer available in the US)—compared with placebo.2 All agents were better than placebo; however, only 2 agents (parenteral high-dose methotrexate and sulfasalazine) had clinically important benefits for more than half the patients. The studies were too small to establish toxicity or to evaluate the other agents.
NSAIDs are still the preferred first-line therapy, concluded a recent publication on the treatment of psoriatic arthritis, which looked at 54 different studies; however, second-line therapy could include methotrexate, sulfasalazine, etanercept (Enbrel), infliximab (Remicade), cyclosporine, or combination therapy.3 Sulfasalazine appeared to be clinically beneficial for peripheral psoriatic arthritis.
Etanercept vs placebo. An initial study (60 patients) of etanercept vs placebo among patients who were permitted to stay on methotrexate or prednisone showed a response rate of 87% vs 23% (P<.0001; number needed to treat [NNT]=1.56).4
Infliximab vs placebo. A study of infliximab vs placebo involving 104 patients had similar results, with good response in 65% vs 10% (NNT=1.81) at 16 weeks; infliximab also inhibited radiographic progression by 22%.5
Cyclosporine. Although it is effective, reserve cyclosporine for patients who do not improve on other regimens, because of its nephrotoxicity.3
DMARDs show some benefit in treating ankylosing spondylitis
Two recent Cochrane systematic reviews on ankylosing spondylitis examined the use of sulfasalazine and methotrexate as second-line agents.6,7 Eleven trials were included in the sulfasalazine analysis, with a total of 895 patients. Sulfasalazine demonstrated some benefit in reducing erythrocyte sedimentation rates (ESRs) and morning stiffness, but there was no evidence that the drug reduced pain or improved physical function, spinal mobility, or rate of enthesitis. Sulfasalazine was well tolerated and may be useful in early mild disease for patients with peripheral arthritis and high ESRs. On the other hand, evidence was insufficient to determine whether methotrexate benefited patients with ankylosing spondylitis.
In other trials, infliximab and etanercept showed good potential for benefit in treating ankylosing spondylitis.
One study of infliximab vs placebo showed 61.2% vs 19.2% patients with good clinical benefit at 24 weeks and only mild or moderate adverse events (P<.001; NNT=2.38).8
Similarly, a smaller study (84 patients) showed that 60% of patients on etanercept vs 20% on placebo had good clinical benefit at only 12 weeks (P<.001, NNT=2.5).9
For other rheumatic diseases, studies are mixed
Due to cyclosporine’s toxicity, less toxic DMARDs are being evaluated to replace it for treatment of other rheumatic diseases. A recent randomized controlled trial of 100 patients with antineutrophil cytoplasmic antibody–associated systemic vasculitis showed methotrexate may be able to replace cyclosporine for both induction of remission (methotrexate=89.8% vs cyclosporine=93.5%; P=.041) and maintenance of remission (69.5% vs 46.5% at 18 months; P=.023).10
Initial trials on other rheumatic diseases have been small and have had varied results. There are mixed studies on the effectiveness of adding methotrexate to corticosteroids for giant cell arteritis.11,12
There has been no evidence of efficacy for the new TNF antagonists in either a small study on Sjögren’s syndrome (n=14)13 or a larger study on Wegener’s granulomatosis (n=180).14
The studies for use of DMARDs in lupus or scleroderma are of limited quality.
Recommendations from others
The Italian Society for Rheumatology consensus guidelines recommends TNF antagonists be considered in active psoriatic arthritis resistant to (a) NSAIDs, (b) at least 2 local steroid injections, and (c) at least 2 conventional DMARDs for patients with peripheral arthritis or enthesitis. They also recommend TNF antagonists be considered for psoriatic spondylitis resistant to NSAIDs.15
The Assessment in Ankylosing Spondylitis (ASAS) International Working Group and the European League Against Rheumatism (EULAR) recommendations for the treatment of ankylosing spondylitis, based on a systematic review of the literature and expert opinion, indicate that:
- There is good evidence for using NSAIDs and COX-2 inhibitors for symptomatic treatment.
- Conventional DMARDs are not well supported.
- TNF antagonists show a large benefit in both pain and function.
The ASAS/EULAR recommendation indicate that there is no evidence that any of these treatments actually modify the disease progression.16
Acknowledgments
The opinions and assertions contained herein are the private views of the authors and not to be construed as official, or as reflecting the views of the US Air Force medical service or the US Air Force at large.
1. Bongartz T, sutton AJ, Sweeting MJ, et al. Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA 2006;295:2275-2285.
2. Jones G, Crotty M, Brooks P. Interventions for treating psoriatic arthritis. Cochrane Database Syst Rev 2000 (3):CD000212.
3. Manadan Am, Sequeira W, Block JA. The treatment of psoriatic arthritis. Am J Ther 2006;13:72-79.
4. Mease PJ, Goffe BS, Metz J, et al. etanercept in the treatment of psoriatic arthritis and psoriasis: a randomized trial. Lancet 2000;356:385-390.
5. Kavanaugh A, Antoni CE, Gladman D, et al. The Infliximab multinational Psoriatic Arthritis Controlled Trial (ImPACT): result of radiographic analyses after 1 year. Ann Rheum Dis 2006;65:1038-1043.
6. Chen J, Liu C. Sulfasalazine for ankylosing spondylitis. Cochrane Database Syst Rev 2005;(2):CD004800.-
7. Chen J, Liu C, Lin J. Methotrexate for ankylosing spondylitis. Cochrane Database Syst Rev 2006;(4):CD004524.-
8. Heijde D, Dijkmans B, Geusens P, et al. efficacy and safety of infliximab in patients with ankylosing spondylitis. Arthritis Rheum 2005;52:582-591.
9. Calin A, Dijkmans BA, Emery P, et al. Outcomes of a multicentre randomized clinical trial of etanercept to treat ankylosing spondylitis. Ann Rheum Dis 2004;63:1594-1600.
10. Grout K, Rasmussen N, Bacon P, et al. randomized trial of cyclophosphamide versus methotrexate for induction of remission in early systemic antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheum 2005;52:2461-2469.
11. Jover JA, Hernandez-Garcia C, Morado IC, et al. Combined treatment of giant-cell arteritis with methotrexate and prednisone. a randomized, double blinded, placebo-controlled trial. Ann Intern Med 2001;134:106-114.
12. Hoffman GS, Cid MC, Hellmann DB, et al. A multicenter, randomized, double-blind, placebo-controlled trial of adjuvant methotrexate treatment for giant cell arteritis. Arthritis Rheum 2002;46:1309-1318.
13. Sankar V, Brennan MT, Kok MR, et al. etanercept in sjögren’s syndrome: a twelve-week randomized, double-blind, placebo-controlled pilot clinical trial. Arthritis Rheum 2004;50:2240-2245.
14. Wegener’s Granulomatosis etanercept Trial (WGeT) research Group. etanercept plus standard therapy for Wegener’s granulomatosis. N Engl J Med 2005;352:351-361.
15. Salvarani C, Olivieri I, Pipitone N, et al. recommendations of the Italian society for rheumatology for the use of biologic (TNF-alpha blocking) agents in the treatment of psoriatic arthritis. Clin Exp Rheumatol 2006;24:70-78.
16. Zochling J, van der Heijde D, Dougados, et al. Current evidence for the management of ankylosing spondylitis: a systematic literature review for the AsAs/eulAr management recommendations in ankylosing spondylitis. Ann Rheum Dis 2006;65:442-452.
1. Bongartz T, sutton AJ, Sweeting MJ, et al. Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA 2006;295:2275-2285.
2. Jones G, Crotty M, Brooks P. Interventions for treating psoriatic arthritis. Cochrane Database Syst Rev 2000 (3):CD000212.
3. Manadan Am, Sequeira W, Block JA. The treatment of psoriatic arthritis. Am J Ther 2006;13:72-79.
4. Mease PJ, Goffe BS, Metz J, et al. etanercept in the treatment of psoriatic arthritis and psoriasis: a randomized trial. Lancet 2000;356:385-390.
5. Kavanaugh A, Antoni CE, Gladman D, et al. The Infliximab multinational Psoriatic Arthritis Controlled Trial (ImPACT): result of radiographic analyses after 1 year. Ann Rheum Dis 2006;65:1038-1043.
6. Chen J, Liu C. Sulfasalazine for ankylosing spondylitis. Cochrane Database Syst Rev 2005;(2):CD004800.-
7. Chen J, Liu C, Lin J. Methotrexate for ankylosing spondylitis. Cochrane Database Syst Rev 2006;(4):CD004524.-
8. Heijde D, Dijkmans B, Geusens P, et al. efficacy and safety of infliximab in patients with ankylosing spondylitis. Arthritis Rheum 2005;52:582-591.
9. Calin A, Dijkmans BA, Emery P, et al. Outcomes of a multicentre randomized clinical trial of etanercept to treat ankylosing spondylitis. Ann Rheum Dis 2004;63:1594-1600.
10. Grout K, Rasmussen N, Bacon P, et al. randomized trial of cyclophosphamide versus methotrexate for induction of remission in early systemic antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheum 2005;52:2461-2469.
11. Jover JA, Hernandez-Garcia C, Morado IC, et al. Combined treatment of giant-cell arteritis with methotrexate and prednisone. a randomized, double blinded, placebo-controlled trial. Ann Intern Med 2001;134:106-114.
12. Hoffman GS, Cid MC, Hellmann DB, et al. A multicenter, randomized, double-blind, placebo-controlled trial of adjuvant methotrexate treatment for giant cell arteritis. Arthritis Rheum 2002;46:1309-1318.
13. Sankar V, Brennan MT, Kok MR, et al. etanercept in sjögren’s syndrome: a twelve-week randomized, double-blind, placebo-controlled pilot clinical trial. Arthritis Rheum 2004;50:2240-2245.
14. Wegener’s Granulomatosis etanercept Trial (WGeT) research Group. etanercept plus standard therapy for Wegener’s granulomatosis. N Engl J Med 2005;352:351-361.
15. Salvarani C, Olivieri I, Pipitone N, et al. recommendations of the Italian society for rheumatology for the use of biologic (TNF-alpha blocking) agents in the treatment of psoriatic arthritis. Clin Exp Rheumatol 2006;24:70-78.
16. Zochling J, van der Heijde D, Dougados, et al. Current evidence for the management of ankylosing spondylitis: a systematic literature review for the AsAs/eulAr management recommendations in ankylosing spondylitis. Ann Rheum Dis 2006;65:442-452.
Evidence-based answers from the Family Physicians Inquiries Network