User login
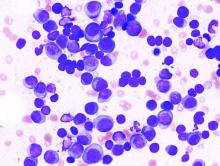
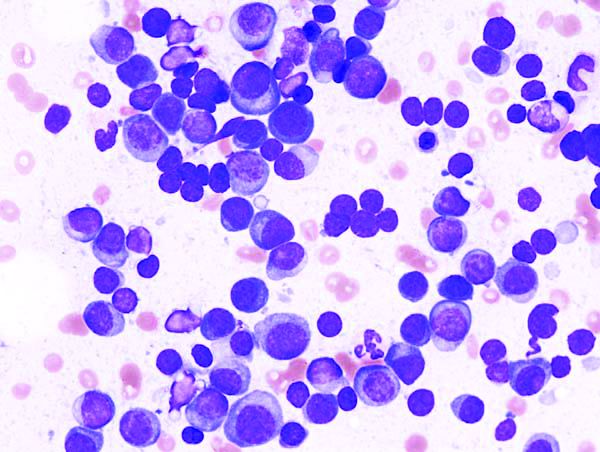
Relapsed multiple myeloma becomes increasingly aggressive and difficult to treat with each additional TP53 alteration, according to investigators.
Findings from the study help illuminate the mechanics of myeloma disease progression and demonstrate the value of clonal competition assays, reported lead author Umair Munawar of the University Hospital Würzburg (Germany) and colleagues.
“The implications of mono-allelic TP53 lesions for the clinical outcome remain controversial, but clonal selection and evolution is a common feature of myeloma progression, and patients with TP53 wild-type or mono-allelic inactivation may present a double hit on relapse,” the investigators wrote in Blood. “Here, we addressed the hypothesis that sequential acquisition of TP53 hits lead to a gain of proliferative fitness of [multiple myeloma] cancer cells, inducing the expansion and domination of the affected clones within the patient’s bone marrow.”
The investigators used sleeping beauty and CRISPR/Cas9 techniques to create double- and single-hit multiple myeloma cell lines that were stably transfected with fluorescent proteins. By observing coculture pairings of wild-type, single-hit, and double-hit cells, the investigators found a hierarchy of proliferation that depended on the number of TP53 alterations. For instance, when double-hit cells were cocultured with wild-type cells in a 1:3 ratio, it took 21 days for the double-hit cells to reach 50% of the total culture population. Similarly, single-hit cells outcompeted wild-type cells after 38 days, while double-hit cells took 35 days to overcome the single-hit population.
Further testing showed that comparatively smaller initial populations of TP53-aberrant cells required longer to outcompete larger wild-type populations, which could explain why deeper responses in the clinic are often followed by longer periods without disease progression, the investigators suggested.
A comparison of transcriptomes between wild-type cells and TP53 mutants revealed differences in about 900 genes, including 14 signaling pathways. Specifically, downregulation impacted antigen processing and presentation, chemokine signaling, and oxidative phosphorylation.
“These differences on the transcriptomic level well reflect the biology of ultra–high risk disease,” the investigators wrote, referring to increased glucose uptake on PET, resistance to immunotherapies, and extramedullary disease.
“[This study] underscores the power of clonal competition assays to decipher the effect of genomic lesions in tumors to better understand their impact on progression and disease relapse in [multiple myeloma],” the investigators concluded.
The study was funded by Deutsche Forschungsgemeinschaft, the CDW Stiftung, and the IZKF Würzburg. The investigators reported additional support from the CRIS foundation, the German Cancer Aid, and the University of Würzburg.
SOURCE: Munawar U et al. Blood. 2019 Jul 24. doi: 10.1182/blood.2019000080.
Relapsed multiple myeloma becomes increasingly aggressive and difficult to treat with each additional TP53 alteration, according to investigators.
Findings from the study help illuminate the mechanics of myeloma disease progression and demonstrate the value of clonal competition assays, reported lead author Umair Munawar of the University Hospital Würzburg (Germany) and colleagues.
“The implications of mono-allelic TP53 lesions for the clinical outcome remain controversial, but clonal selection and evolution is a common feature of myeloma progression, and patients with TP53 wild-type or mono-allelic inactivation may present a double hit on relapse,” the investigators wrote in Blood. “Here, we addressed the hypothesis that sequential acquisition of TP53 hits lead to a gain of proliferative fitness of [multiple myeloma] cancer cells, inducing the expansion and domination of the affected clones within the patient’s bone marrow.”
The investigators used sleeping beauty and CRISPR/Cas9 techniques to create double- and single-hit multiple myeloma cell lines that were stably transfected with fluorescent proteins. By observing coculture pairings of wild-type, single-hit, and double-hit cells, the investigators found a hierarchy of proliferation that depended on the number of TP53 alterations. For instance, when double-hit cells were cocultured with wild-type cells in a 1:3 ratio, it took 21 days for the double-hit cells to reach 50% of the total culture population. Similarly, single-hit cells outcompeted wild-type cells after 38 days, while double-hit cells took 35 days to overcome the single-hit population.
Further testing showed that comparatively smaller initial populations of TP53-aberrant cells required longer to outcompete larger wild-type populations, which could explain why deeper responses in the clinic are often followed by longer periods without disease progression, the investigators suggested.
A comparison of transcriptomes between wild-type cells and TP53 mutants revealed differences in about 900 genes, including 14 signaling pathways. Specifically, downregulation impacted antigen processing and presentation, chemokine signaling, and oxidative phosphorylation.
“These differences on the transcriptomic level well reflect the biology of ultra–high risk disease,” the investigators wrote, referring to increased glucose uptake on PET, resistance to immunotherapies, and extramedullary disease.
“[This study] underscores the power of clonal competition assays to decipher the effect of genomic lesions in tumors to better understand their impact on progression and disease relapse in [multiple myeloma],” the investigators concluded.
The study was funded by Deutsche Forschungsgemeinschaft, the CDW Stiftung, and the IZKF Würzburg. The investigators reported additional support from the CRIS foundation, the German Cancer Aid, and the University of Würzburg.
SOURCE: Munawar U et al. Blood. 2019 Jul 24. doi: 10.1182/blood.2019000080.
Relapsed multiple myeloma becomes increasingly aggressive and difficult to treat with each additional TP53 alteration, according to investigators.
Findings from the study help illuminate the mechanics of myeloma disease progression and demonstrate the value of clonal competition assays, reported lead author Umair Munawar of the University Hospital Würzburg (Germany) and colleagues.
“The implications of mono-allelic TP53 lesions for the clinical outcome remain controversial, but clonal selection and evolution is a common feature of myeloma progression, and patients with TP53 wild-type or mono-allelic inactivation may present a double hit on relapse,” the investigators wrote in Blood. “Here, we addressed the hypothesis that sequential acquisition of TP53 hits lead to a gain of proliferative fitness of [multiple myeloma] cancer cells, inducing the expansion and domination of the affected clones within the patient’s bone marrow.”
The investigators used sleeping beauty and CRISPR/Cas9 techniques to create double- and single-hit multiple myeloma cell lines that were stably transfected with fluorescent proteins. By observing coculture pairings of wild-type, single-hit, and double-hit cells, the investigators found a hierarchy of proliferation that depended on the number of TP53 alterations. For instance, when double-hit cells were cocultured with wild-type cells in a 1:3 ratio, it took 21 days for the double-hit cells to reach 50% of the total culture population. Similarly, single-hit cells outcompeted wild-type cells after 38 days, while double-hit cells took 35 days to overcome the single-hit population.
Further testing showed that comparatively smaller initial populations of TP53-aberrant cells required longer to outcompete larger wild-type populations, which could explain why deeper responses in the clinic are often followed by longer periods without disease progression, the investigators suggested.
A comparison of transcriptomes between wild-type cells and TP53 mutants revealed differences in about 900 genes, including 14 signaling pathways. Specifically, downregulation impacted antigen processing and presentation, chemokine signaling, and oxidative phosphorylation.
“These differences on the transcriptomic level well reflect the biology of ultra–high risk disease,” the investigators wrote, referring to increased glucose uptake on PET, resistance to immunotherapies, and extramedullary disease.
“[This study] underscores the power of clonal competition assays to decipher the effect of genomic lesions in tumors to better understand their impact on progression and disease relapse in [multiple myeloma],” the investigators concluded.
The study was funded by Deutsche Forschungsgemeinschaft, the CDW Stiftung, and the IZKF Würzburg. The investigators reported additional support from the CRIS foundation, the German Cancer Aid, and the University of Würzburg.
SOURCE: Munawar U et al. Blood. 2019 Jul 24. doi: 10.1182/blood.2019000080.
FROM BLOOD