User login
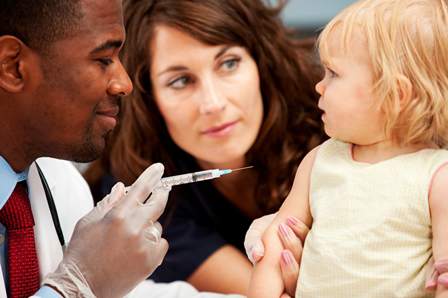
There is an increased risk of febrile seizures when children receiving certain recommended vaccinations at the same time, but that risk is low, a study found.
Dr. Jonathan Duffy from the Immunization Safety Office of the Centers for Disease Control and Prevention and his colleagues followed up on a study that showed an increased risk of febrile seizures in children vaccinated with a trivalent inactivated influenza vaccine (IIV3) and 13-valent pneumococcal conjugate vaccine (PCV) at the same time during the 2010-2011 influenza season.
The investigators wanted to assess the effect of administering other common childhood vaccines with IIV3 on the risk for febrile seizures so they examined chart records of potential cases of febrile seizures in those aged 6-23 months from the Vaccine Safety Datalink. The data were collected between the 2006-2007 through 2010-2011 influenza seasons.
The search yielded 333 chart-confirmed cases of febrile seizures. To examine the safety of each recommended vaccination administered alone or in combination, the cases were divided into two groups, one to serve as a risk interval group (n = 103) for febrile seizures on days 0 to 1 postvaccination; and one control interval comparison group (n = 230) with febrile seizures 14-20 days postvaccination. The multivariable model used for the study indicated that IIV3, PCV, and DTaP-containing vaccines were most often associated with febrile seizures in the risk interval group, but that only PCV7 showed an independent increased risk of febrile seizures (incidence rate ratio, 1.98) after the model was adjusted to strip out concomitantly administered vaccines.
Although increased risks of febrile seizures were detected for these three combinations, the overall risk of febrile seizures was quite low, on the order of 10, 24, and 38 per 100,000 vaccinated children at 6, 12, and 15 months, respectively, for the triple concomitant administration in the risk interval.
The risk of febrile seizures also was higher after receiving three different combinations of concomitantly-administered vaccinations, IIV3 plus PCV (IRR, 3.50), IIV3 plus DTaP (IRR, 3.50), and IIV3 plus PCV plus DTaP (IRR, 5.00).
“Our results suggest that the risk of [febrile seizure] is increased after certain combinations of vaccines, but the absolute risk of [febrile seizure] after these combinations is small,” Dr. Duffy and his associates noted in Pediatrics (2016;138[1]:e20160320).
The U.S. Centers for Disease Control and Prevention funded the study. Dr. Naleway and Dr. Klein reported receiving research funding/support from multiple industry sources. The remaining authors reported no financial disclosures.
Concomitant administration of influenza, DTaP, and pneumococcal conjugate vaccine (PCV) vaccines was associated with febrile seizures at a rate of up to 30 in 100,000 children immunized. This would result in one child, at most, who would be expected to experience a febrile seizure caused by the concomitant administration of these vaccines in the first 2 years of life over a 5-10 year period in an average pediatric practice, based on a patient base including 1,000 children younger than 5 years of age, which would include 3-500 patients between 6 and 24 months of age annually.
Does this mean we should stop giving these vaccines together or stop giving them at all? We say, emphatically, no.
Febrile seizures, although frightening to parents, rarely have any long-term sequelae. The risk from these diseases far outweigh the risk from the vaccines.
This study, conducted by the Vaccine Safety Datalink, and others like it, are important as we engage in dialogue with parents about the risks and benefits of vaccines.
These comments are excerpted from a commentary by Dr. Mark H. Sawyer of the University of California, San Diego, department of pediatrics and Rady Children’s Hospital, also in San Diego. Dr. Geoff Simon of Nemours duPont Pediatrics, Wilmington, Del., and Dr. Carrie Byington of the department of pediatrics, University of Utah, Salt Lake City. Dr. Sawyer and Dr. Simon are members of and Dr. Byington is the chair of the American Academy of Pediatrics Committee on Infectious Disease. Dr. Byington has intellectual property in and receives royalties from BioFire Diagnostics; Dr. Sawyer and Dr. Simon indicated they have no financial relationships relevant to this article. Funded by the National Institutes of Health. (Pediatrics. 2016 Jun 6. doi: 10.1542/peds.2016-0976 ).
Concomitant administration of influenza, DTaP, and pneumococcal conjugate vaccine (PCV) vaccines was associated with febrile seizures at a rate of up to 30 in 100,000 children immunized. This would result in one child, at most, who would be expected to experience a febrile seizure caused by the concomitant administration of these vaccines in the first 2 years of life over a 5-10 year period in an average pediatric practice, based on a patient base including 1,000 children younger than 5 years of age, which would include 3-500 patients between 6 and 24 months of age annually.
Does this mean we should stop giving these vaccines together or stop giving them at all? We say, emphatically, no.
Febrile seizures, although frightening to parents, rarely have any long-term sequelae. The risk from these diseases far outweigh the risk from the vaccines.
This study, conducted by the Vaccine Safety Datalink, and others like it, are important as we engage in dialogue with parents about the risks and benefits of vaccines.
These comments are excerpted from a commentary by Dr. Mark H. Sawyer of the University of California, San Diego, department of pediatrics and Rady Children’s Hospital, also in San Diego. Dr. Geoff Simon of Nemours duPont Pediatrics, Wilmington, Del., and Dr. Carrie Byington of the department of pediatrics, University of Utah, Salt Lake City. Dr. Sawyer and Dr. Simon are members of and Dr. Byington is the chair of the American Academy of Pediatrics Committee on Infectious Disease. Dr. Byington has intellectual property in and receives royalties from BioFire Diagnostics; Dr. Sawyer and Dr. Simon indicated they have no financial relationships relevant to this article. Funded by the National Institutes of Health. (Pediatrics. 2016 Jun 6. doi: 10.1542/peds.2016-0976 ).
Concomitant administration of influenza, DTaP, and pneumococcal conjugate vaccine (PCV) vaccines was associated with febrile seizures at a rate of up to 30 in 100,000 children immunized. This would result in one child, at most, who would be expected to experience a febrile seizure caused by the concomitant administration of these vaccines in the first 2 years of life over a 5-10 year period in an average pediatric practice, based on a patient base including 1,000 children younger than 5 years of age, which would include 3-500 patients between 6 and 24 months of age annually.
Does this mean we should stop giving these vaccines together or stop giving them at all? We say, emphatically, no.
Febrile seizures, although frightening to parents, rarely have any long-term sequelae. The risk from these diseases far outweigh the risk from the vaccines.
This study, conducted by the Vaccine Safety Datalink, and others like it, are important as we engage in dialogue with parents about the risks and benefits of vaccines.
These comments are excerpted from a commentary by Dr. Mark H. Sawyer of the University of California, San Diego, department of pediatrics and Rady Children’s Hospital, also in San Diego. Dr. Geoff Simon of Nemours duPont Pediatrics, Wilmington, Del., and Dr. Carrie Byington of the department of pediatrics, University of Utah, Salt Lake City. Dr. Sawyer and Dr. Simon are members of and Dr. Byington is the chair of the American Academy of Pediatrics Committee on Infectious Disease. Dr. Byington has intellectual property in and receives royalties from BioFire Diagnostics; Dr. Sawyer and Dr. Simon indicated they have no financial relationships relevant to this article. Funded by the National Institutes of Health. (Pediatrics. 2016 Jun 6. doi: 10.1542/peds.2016-0976 ).
There is an increased risk of febrile seizures when children receiving certain recommended vaccinations at the same time, but that risk is low, a study found.
Dr. Jonathan Duffy from the Immunization Safety Office of the Centers for Disease Control and Prevention and his colleagues followed up on a study that showed an increased risk of febrile seizures in children vaccinated with a trivalent inactivated influenza vaccine (IIV3) and 13-valent pneumococcal conjugate vaccine (PCV) at the same time during the 2010-2011 influenza season.
The investigators wanted to assess the effect of administering other common childhood vaccines with IIV3 on the risk for febrile seizures so they examined chart records of potential cases of febrile seizures in those aged 6-23 months from the Vaccine Safety Datalink. The data were collected between the 2006-2007 through 2010-2011 influenza seasons.
The search yielded 333 chart-confirmed cases of febrile seizures. To examine the safety of each recommended vaccination administered alone or in combination, the cases were divided into two groups, one to serve as a risk interval group (n = 103) for febrile seizures on days 0 to 1 postvaccination; and one control interval comparison group (n = 230) with febrile seizures 14-20 days postvaccination. The multivariable model used for the study indicated that IIV3, PCV, and DTaP-containing vaccines were most often associated with febrile seizures in the risk interval group, but that only PCV7 showed an independent increased risk of febrile seizures (incidence rate ratio, 1.98) after the model was adjusted to strip out concomitantly administered vaccines.
Although increased risks of febrile seizures were detected for these three combinations, the overall risk of febrile seizures was quite low, on the order of 10, 24, and 38 per 100,000 vaccinated children at 6, 12, and 15 months, respectively, for the triple concomitant administration in the risk interval.
The risk of febrile seizures also was higher after receiving three different combinations of concomitantly-administered vaccinations, IIV3 plus PCV (IRR, 3.50), IIV3 plus DTaP (IRR, 3.50), and IIV3 plus PCV plus DTaP (IRR, 5.00).
“Our results suggest that the risk of [febrile seizure] is increased after certain combinations of vaccines, but the absolute risk of [febrile seizure] after these combinations is small,” Dr. Duffy and his associates noted in Pediatrics (2016;138[1]:e20160320).
The U.S. Centers for Disease Control and Prevention funded the study. Dr. Naleway and Dr. Klein reported receiving research funding/support from multiple industry sources. The remaining authors reported no financial disclosures.
There is an increased risk of febrile seizures when children receiving certain recommended vaccinations at the same time, but that risk is low, a study found.
Dr. Jonathan Duffy from the Immunization Safety Office of the Centers for Disease Control and Prevention and his colleagues followed up on a study that showed an increased risk of febrile seizures in children vaccinated with a trivalent inactivated influenza vaccine (IIV3) and 13-valent pneumococcal conjugate vaccine (PCV) at the same time during the 2010-2011 influenza season.
The investigators wanted to assess the effect of administering other common childhood vaccines with IIV3 on the risk for febrile seizures so they examined chart records of potential cases of febrile seizures in those aged 6-23 months from the Vaccine Safety Datalink. The data were collected between the 2006-2007 through 2010-2011 influenza seasons.
The search yielded 333 chart-confirmed cases of febrile seizures. To examine the safety of each recommended vaccination administered alone or in combination, the cases were divided into two groups, one to serve as a risk interval group (n = 103) for febrile seizures on days 0 to 1 postvaccination; and one control interval comparison group (n = 230) with febrile seizures 14-20 days postvaccination. The multivariable model used for the study indicated that IIV3, PCV, and DTaP-containing vaccines were most often associated with febrile seizures in the risk interval group, but that only PCV7 showed an independent increased risk of febrile seizures (incidence rate ratio, 1.98) after the model was adjusted to strip out concomitantly administered vaccines.
Although increased risks of febrile seizures were detected for these three combinations, the overall risk of febrile seizures was quite low, on the order of 10, 24, and 38 per 100,000 vaccinated children at 6, 12, and 15 months, respectively, for the triple concomitant administration in the risk interval.
The risk of febrile seizures also was higher after receiving three different combinations of concomitantly-administered vaccinations, IIV3 plus PCV (IRR, 3.50), IIV3 plus DTaP (IRR, 3.50), and IIV3 plus PCV plus DTaP (IRR, 5.00).
“Our results suggest that the risk of [febrile seizure] is increased after certain combinations of vaccines, but the absolute risk of [febrile seizure] after these combinations is small,” Dr. Duffy and his associates noted in Pediatrics (2016;138[1]:e20160320).
The U.S. Centers for Disease Control and Prevention funded the study. Dr. Naleway and Dr. Klein reported receiving research funding/support from multiple industry sources. The remaining authors reported no financial disclosures.
FROM PEDIATRICS
Key clinical point: The concomitant administration of certain childhood vaccinations may slightly increase the risk of febrile seizures.
Major finding: Although the risk of febrile seizures in the population studied was low in general, it was higher for those receiving concomitantly-administered IIV3 plus PCV, IIV3 plus DTaP, and IIV3 plus PCV plus DTaP.
Data sources: Vaccine Safety Datalink repository of vaccine safety research and surveillance.
Disclosures: The Centers for Disease Control and Prevention funded the study. Dr. Naleway and Dr. Klein reported receiving research funding/support from multiple industry sources. The remaining authors reported no financial disclosures.