Article
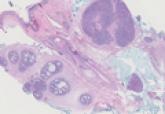
Trichoepithelioma and Spiradenoma Collision Tumor
- Author:
- Amanda F. Marsch, MD
- Jeffrey B. Shackelton, MD
- Dirk M. Elston, MD
The coexistence of more than one cutaneous adnexal neoplasm in a single biopsy specimen is unusual and is most frequently recognized in the...
Article

Lipidized Dermatofibroma
- Author:
- Amanda F. Marsch, MD
- Ramayee Periakaruppan, MD
- Marylee Braniecki, MD
Lipidized dermatofibromas most commonly are found on the ankles, which has led some authors to refer to these lesions as ankle-type fibrous...
