User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Patient Navigators for Serious Illnesses Can Now Bill Under New Medicare Codes
In a move that acknowledges the gauntlet the US health system poses for people facing serious and fatal illnesses, Medicare will pay for a new class of workers to help patients manage treatments for conditions like cancer and heart failure.
The 2024 Medicare physician fee schedule includes new billing codes, including G0023, to pay for 60 minutes a month of care coordination by certified or trained auxiliary personnel working under the direction of a clinician.
A diagnosis of cancer or another serious illness takes a toll beyond the physical effects of the disease. Patients often scramble to make adjustments in family and work schedules to manage treatment, said Samyukta Mullangi, MD, MBA, medical director of oncology at Thyme Care, a Nashville, Tennessee–based firm that provides navigation and coordination services to oncology practices and insurers.
“It just really does create a bit of a pressure cooker for patients,” Dr. Mullangi told this news organization.
Medicare has for many years paid for medical professionals to help patients cope with the complexities of disease, such as chronic care management (CCM) provided by physicians, nurses, and physician assistants.
The new principal illness navigation (PIN) payments are intended to pay for work that to date typically has been done by people without medical degrees, including those involved in peer support networks and community health programs. The US Centers for Medicare and Medicaid Services(CMS) expects these navigators will undergo training and work under the supervision of clinicians.
The new navigators may coordinate care transitions between medical settings, follow up with patients after emergency department (ED) visits, or communicate with skilled nursing facilities regarding the psychosocial needs and functional deficits of a patient, among other functions.
CMS expects the new navigators may:
- Conduct assessments to understand a patient’s life story, strengths, needs, goals, preferences, and desired outcomes, including understanding cultural and linguistic factors.
- Provide support to accomplish the clinician’s treatment plan.
- Coordinate the receipt of needed services from healthcare facilities, home- and community-based service providers, and caregivers.
Peers as Navigators
The new navigators can be former patients who have undergone similar treatments for serious diseases, CMS said. This approach sets the new program apart from other care management services Medicare already covers, program officials wrote in the 2024 physician fee schedule.
“For some conditions, patients are best able to engage with the healthcare system and access care if they have assistance from a single, dedicated individual who has ‘lived experience,’ ” according to the rule.
The agency has taken a broad initial approach in defining what kinds of illnesses a patient may have to qualify for services. Patients must have a serious condition that is expected to last at least 3 months, such as cancer, heart failure, or substance use disorder.
But those without a definitive diagnosis may also qualify to receive navigator services.
In the rule, CMS cited a case in which a CT scan identified a suspicious mass in a patient’s colon. A clinician might decide this person would benefit from navigation services due to the potential risks for an undiagnosed illness.
“Regardless of the definitive diagnosis of the mass, presence of a colonic mass for that patient may be a serious high-risk condition that could, for example, cause obstruction and lead the patient to present to the emergency department, as well as be potentially indicative of an underlying life-threatening illness such as colon cancer,” CMS wrote in the rule.
Navigators often start their work when cancer patients are screened and guide them through initial diagnosis, potential surgery, radiation, or chemotherapy, said Sharon Gentry, MSN, RN, a former nurse navigator who is now the editor in chief of the Journal of the Academy of Oncology Nurse & Patient Navigators.
The navigators are meant to be a trusted and continual presence for patients, who otherwise might be left to start anew in finding help at each phase of care.
The navigators “see the whole picture. They see the whole journey the patient takes, from pre-diagnosis all the way through diagnosis care out through survival,” Ms. Gentry said.
Gaining a special Medicare payment for these kinds of services will elevate this work, she said.
Many newer drugs can target specific mechanisms and proteins of cancer. Often, oncology treatment involves testing to find out if mutations are allowing the cancer cells to evade a patient’s immune system.
Checking these biomarkers takes time, however. Patients sometimes become frustrated because they are anxious to begin treatment. Patients may receive inaccurate information from friends or family who went through treatment previously. Navigators can provide knowledge on the current state of care for a patient’s disease, helping them better manage anxieties.
“You have to explain to them that things have changed since the guy you drink coffee with was diagnosed with cancer, and there may be a drug that could target that,” Ms. Gentry said.
Potential Challenges
Initial uptake of the new PIN codes may be slow going, however, as clinicians and health systems may already use well-established codes. These include CCM and principal care management services, which may pay higher rates, Mullangi said.
“There might be sensitivity around not wanting to cannibalize existing programs with a new program,” Dr. Mullangi said.
In addition, many patients will have a copay for the services of principal illness navigators, Dr. Mullangi said.
While many patients have additional insurance that would cover the service, not all do. People with traditional Medicare coverage can sometimes pay 20% of the cost of some medical services.
“I think that may give patients pause, particularly if they’re already feeling the financial burden of a cancer treatment journey,” Dr. Mullangi said.
Pay rates for PIN services involve calculations of regional price differences, which are posted publicly by CMS, and potential added fees for services provided by hospital-affiliated organizations.
Consider payments for code G0023, covering 60 minutes of principal navigation services provided in a single month.
A set reimbursement for patients cared for in independent medical practices exists, with variation for local costs. Medicare’s non-facility price for G0023 would be $102.41 in some parts of Silicon Valley in California, including San Jose. In Arkansas, where costs are lower, reimbursement would be $73.14 for this same service.
Patients who get services covered by code G0023 in independent medical practices would have monthly copays of about $15-$20, depending on where they live.
The tab for patients tends to be higher for these same services if delivered through a medical practice owned by a hospital, as this would trigger the addition of facility fees to the payments made to cover the services. Facility fees are difficult for the public to ascertain before getting a treatment or service.
Dr. Mullangi and Ms. Gentry reported no relevant financial disclosures outside of their employers.
A version of this article first appeared on Medscape.com.
In a move that acknowledges the gauntlet the US health system poses for people facing serious and fatal illnesses, Medicare will pay for a new class of workers to help patients manage treatments for conditions like cancer and heart failure.
The 2024 Medicare physician fee schedule includes new billing codes, including G0023, to pay for 60 minutes a month of care coordination by certified or trained auxiliary personnel working under the direction of a clinician.
A diagnosis of cancer or another serious illness takes a toll beyond the physical effects of the disease. Patients often scramble to make adjustments in family and work schedules to manage treatment, said Samyukta Mullangi, MD, MBA, medical director of oncology at Thyme Care, a Nashville, Tennessee–based firm that provides navigation and coordination services to oncology practices and insurers.
“It just really does create a bit of a pressure cooker for patients,” Dr. Mullangi told this news organization.
Medicare has for many years paid for medical professionals to help patients cope with the complexities of disease, such as chronic care management (CCM) provided by physicians, nurses, and physician assistants.
The new principal illness navigation (PIN) payments are intended to pay for work that to date typically has been done by people without medical degrees, including those involved in peer support networks and community health programs. The US Centers for Medicare and Medicaid Services(CMS) expects these navigators will undergo training and work under the supervision of clinicians.
The new navigators may coordinate care transitions between medical settings, follow up with patients after emergency department (ED) visits, or communicate with skilled nursing facilities regarding the psychosocial needs and functional deficits of a patient, among other functions.
CMS expects the new navigators may:
- Conduct assessments to understand a patient’s life story, strengths, needs, goals, preferences, and desired outcomes, including understanding cultural and linguistic factors.
- Provide support to accomplish the clinician’s treatment plan.
- Coordinate the receipt of needed services from healthcare facilities, home- and community-based service providers, and caregivers.
Peers as Navigators
The new navigators can be former patients who have undergone similar treatments for serious diseases, CMS said. This approach sets the new program apart from other care management services Medicare already covers, program officials wrote in the 2024 physician fee schedule.
“For some conditions, patients are best able to engage with the healthcare system and access care if they have assistance from a single, dedicated individual who has ‘lived experience,’ ” according to the rule.
The agency has taken a broad initial approach in defining what kinds of illnesses a patient may have to qualify for services. Patients must have a serious condition that is expected to last at least 3 months, such as cancer, heart failure, or substance use disorder.
But those without a definitive diagnosis may also qualify to receive navigator services.
In the rule, CMS cited a case in which a CT scan identified a suspicious mass in a patient’s colon. A clinician might decide this person would benefit from navigation services due to the potential risks for an undiagnosed illness.
“Regardless of the definitive diagnosis of the mass, presence of a colonic mass for that patient may be a serious high-risk condition that could, for example, cause obstruction and lead the patient to present to the emergency department, as well as be potentially indicative of an underlying life-threatening illness such as colon cancer,” CMS wrote in the rule.
Navigators often start their work when cancer patients are screened and guide them through initial diagnosis, potential surgery, radiation, or chemotherapy, said Sharon Gentry, MSN, RN, a former nurse navigator who is now the editor in chief of the Journal of the Academy of Oncology Nurse & Patient Navigators.
The navigators are meant to be a trusted and continual presence for patients, who otherwise might be left to start anew in finding help at each phase of care.
The navigators “see the whole picture. They see the whole journey the patient takes, from pre-diagnosis all the way through diagnosis care out through survival,” Ms. Gentry said.
Gaining a special Medicare payment for these kinds of services will elevate this work, she said.
Many newer drugs can target specific mechanisms and proteins of cancer. Often, oncology treatment involves testing to find out if mutations are allowing the cancer cells to evade a patient’s immune system.
Checking these biomarkers takes time, however. Patients sometimes become frustrated because they are anxious to begin treatment. Patients may receive inaccurate information from friends or family who went through treatment previously. Navigators can provide knowledge on the current state of care for a patient’s disease, helping them better manage anxieties.
“You have to explain to them that things have changed since the guy you drink coffee with was diagnosed with cancer, and there may be a drug that could target that,” Ms. Gentry said.
Potential Challenges
Initial uptake of the new PIN codes may be slow going, however, as clinicians and health systems may already use well-established codes. These include CCM and principal care management services, which may pay higher rates, Mullangi said.
“There might be sensitivity around not wanting to cannibalize existing programs with a new program,” Dr. Mullangi said.
In addition, many patients will have a copay for the services of principal illness navigators, Dr. Mullangi said.
While many patients have additional insurance that would cover the service, not all do. People with traditional Medicare coverage can sometimes pay 20% of the cost of some medical services.
“I think that may give patients pause, particularly if they’re already feeling the financial burden of a cancer treatment journey,” Dr. Mullangi said.
Pay rates for PIN services involve calculations of regional price differences, which are posted publicly by CMS, and potential added fees for services provided by hospital-affiliated organizations.
Consider payments for code G0023, covering 60 minutes of principal navigation services provided in a single month.
A set reimbursement for patients cared for in independent medical practices exists, with variation for local costs. Medicare’s non-facility price for G0023 would be $102.41 in some parts of Silicon Valley in California, including San Jose. In Arkansas, where costs are lower, reimbursement would be $73.14 for this same service.
Patients who get services covered by code G0023 in independent medical practices would have monthly copays of about $15-$20, depending on where they live.
The tab for patients tends to be higher for these same services if delivered through a medical practice owned by a hospital, as this would trigger the addition of facility fees to the payments made to cover the services. Facility fees are difficult for the public to ascertain before getting a treatment or service.
Dr. Mullangi and Ms. Gentry reported no relevant financial disclosures outside of their employers.
A version of this article first appeared on Medscape.com.
In a move that acknowledges the gauntlet the US health system poses for people facing serious and fatal illnesses, Medicare will pay for a new class of workers to help patients manage treatments for conditions like cancer and heart failure.
The 2024 Medicare physician fee schedule includes new billing codes, including G0023, to pay for 60 minutes a month of care coordination by certified or trained auxiliary personnel working under the direction of a clinician.
A diagnosis of cancer or another serious illness takes a toll beyond the physical effects of the disease. Patients often scramble to make adjustments in family and work schedules to manage treatment, said Samyukta Mullangi, MD, MBA, medical director of oncology at Thyme Care, a Nashville, Tennessee–based firm that provides navigation and coordination services to oncology practices and insurers.
“It just really does create a bit of a pressure cooker for patients,” Dr. Mullangi told this news organization.
Medicare has for many years paid for medical professionals to help patients cope with the complexities of disease, such as chronic care management (CCM) provided by physicians, nurses, and physician assistants.
The new principal illness navigation (PIN) payments are intended to pay for work that to date typically has been done by people without medical degrees, including those involved in peer support networks and community health programs. The US Centers for Medicare and Medicaid Services(CMS) expects these navigators will undergo training and work under the supervision of clinicians.
The new navigators may coordinate care transitions between medical settings, follow up with patients after emergency department (ED) visits, or communicate with skilled nursing facilities regarding the psychosocial needs and functional deficits of a patient, among other functions.
CMS expects the new navigators may:
- Conduct assessments to understand a patient’s life story, strengths, needs, goals, preferences, and desired outcomes, including understanding cultural and linguistic factors.
- Provide support to accomplish the clinician’s treatment plan.
- Coordinate the receipt of needed services from healthcare facilities, home- and community-based service providers, and caregivers.
Peers as Navigators
The new navigators can be former patients who have undergone similar treatments for serious diseases, CMS said. This approach sets the new program apart from other care management services Medicare already covers, program officials wrote in the 2024 physician fee schedule.
“For some conditions, patients are best able to engage with the healthcare system and access care if they have assistance from a single, dedicated individual who has ‘lived experience,’ ” according to the rule.
The agency has taken a broad initial approach in defining what kinds of illnesses a patient may have to qualify for services. Patients must have a serious condition that is expected to last at least 3 months, such as cancer, heart failure, or substance use disorder.
But those without a definitive diagnosis may also qualify to receive navigator services.
In the rule, CMS cited a case in which a CT scan identified a suspicious mass in a patient’s colon. A clinician might decide this person would benefit from navigation services due to the potential risks for an undiagnosed illness.
“Regardless of the definitive diagnosis of the mass, presence of a colonic mass for that patient may be a serious high-risk condition that could, for example, cause obstruction and lead the patient to present to the emergency department, as well as be potentially indicative of an underlying life-threatening illness such as colon cancer,” CMS wrote in the rule.
Navigators often start their work when cancer patients are screened and guide them through initial diagnosis, potential surgery, radiation, or chemotherapy, said Sharon Gentry, MSN, RN, a former nurse navigator who is now the editor in chief of the Journal of the Academy of Oncology Nurse & Patient Navigators.
The navigators are meant to be a trusted and continual presence for patients, who otherwise might be left to start anew in finding help at each phase of care.
The navigators “see the whole picture. They see the whole journey the patient takes, from pre-diagnosis all the way through diagnosis care out through survival,” Ms. Gentry said.
Gaining a special Medicare payment for these kinds of services will elevate this work, she said.
Many newer drugs can target specific mechanisms and proteins of cancer. Often, oncology treatment involves testing to find out if mutations are allowing the cancer cells to evade a patient’s immune system.
Checking these biomarkers takes time, however. Patients sometimes become frustrated because they are anxious to begin treatment. Patients may receive inaccurate information from friends or family who went through treatment previously. Navigators can provide knowledge on the current state of care for a patient’s disease, helping them better manage anxieties.
“You have to explain to them that things have changed since the guy you drink coffee with was diagnosed with cancer, and there may be a drug that could target that,” Ms. Gentry said.
Potential Challenges
Initial uptake of the new PIN codes may be slow going, however, as clinicians and health systems may already use well-established codes. These include CCM and principal care management services, which may pay higher rates, Mullangi said.
“There might be sensitivity around not wanting to cannibalize existing programs with a new program,” Dr. Mullangi said.
In addition, many patients will have a copay for the services of principal illness navigators, Dr. Mullangi said.
While many patients have additional insurance that would cover the service, not all do. People with traditional Medicare coverage can sometimes pay 20% of the cost of some medical services.
“I think that may give patients pause, particularly if they’re already feeling the financial burden of a cancer treatment journey,” Dr. Mullangi said.
Pay rates for PIN services involve calculations of regional price differences, which are posted publicly by CMS, and potential added fees for services provided by hospital-affiliated organizations.
Consider payments for code G0023, covering 60 minutes of principal navigation services provided in a single month.
A set reimbursement for patients cared for in independent medical practices exists, with variation for local costs. Medicare’s non-facility price for G0023 would be $102.41 in some parts of Silicon Valley in California, including San Jose. In Arkansas, where costs are lower, reimbursement would be $73.14 for this same service.
Patients who get services covered by code G0023 in independent medical practices would have monthly copays of about $15-$20, depending on where they live.
The tab for patients tends to be higher for these same services if delivered through a medical practice owned by a hospital, as this would trigger the addition of facility fees to the payments made to cover the services. Facility fees are difficult for the public to ascertain before getting a treatment or service.
Dr. Mullangi and Ms. Gentry reported no relevant financial disclosures outside of their employers.
A version of this article first appeared on Medscape.com.

How to explain physician compounding to legislators
In Ohio, new limits on drug compounding in physicians’ offices went into effect in April and have become a real hindrance to care for dermatology patients. The State of Ohio Board of Pharmacy has defined compounding as combining two or more prescription drugs and has required that physicians who perform this “compounding” must obtain a “Terminal Distributor of Dangerous Drugs” license. Ohio is the “test state,” and these rules, unless vigorously opposed, will be coming to your state.
[polldaddy:9779752]
The rules state that “compounded” drugs used within 6 hours of preparation must be prepared in a designated clean medication area with proper hand hygiene and the use of powder-free gloves. “Compounded” drugs that are used more than 6 hours after preparation, require a designated clean room with access limited to authorized personnel, environmental control devices such as a laminar flow hood, and additional equipment and training of personnel to maintain an aseptic environment. A separate license is required for each office location.
The state pharmacy boards are eager to restrict physicians – as well as dentists and veterinarians – and to collect annual licensing fees. Additionally, according to an article from the Ohio State Medical Association, noncompliant physicians can be fined by the pharmacy board.
We are talking big money, power, and dreams of clinical relevancy (and billable activities) here.
What can dermatologists do to prevent this regulatory overreach? I encourage you to plan a visit to your state representative, where you can demonstrate how these restrictions affect you and your patients – an exercise that should be both fun and compelling. All you need to illustrate your case is a simple kit that includes a syringe (but no needles in the statehouse!), a bottle of lidocaine with epinephrine, a bottle of 8.4% bicarbonate, alcohol pads, and gloves.
First, explain to your audience that there is a skin cancer epidemic with more than 5.4 million new cases a year and that, over the past 20 years, the incidence of skin cancer has doubled and is projected to double again over the next 20 years. Further, explain that dermatologists treat more than 70% of these cases in the office setting, under local anesthesia, at a huge cost savings to the public and government (it costs an average of 12 times as much to remove these cancers in the outpatient department at the hospital). Remember, states foot most of the bill for Medicaid and Medicare gap indigent coverage.
Take the bottle of lidocaine with epinephrine and open the syringe pack (Staffers love this demonstration; everyone is fascinated with shots.). Put on your gloves, wipe the top of the lidocaine bottle with an alcohol swab, and explain that this medicine is the anesthetic preferred for skin cancer surgery. Explain how it not only numbs the skin, but also causes vasoconstriction, so that the cancer can be easily and safely removed in the office.
Then explain that, in order for the epinephrine to be stable, the solution has to be very acidic (a pH of 4.2, in fact). Explain that this makes it burn like hell unless you add 0.1 cc per cc of 8.4% bicarbonate, in which case the perceived pain on a 10-point scale will drop from 8 to 2. Then pick up the bottle of bicarbonate and explain that you will no longer be able to mix these two components anymore without a “Terminal Distributor of Dangerous Drugs” license because your state pharmacy board considers this compounding. Your representative is likely to give you looks of astonishment, disbelief, and then a dawning realization of the absurdity of the situation.
Follow-up questions may include “Why can’t you buy buffered lidocaine with epinephrine from the compounding pharmacy?” Easy answer: because each patient needs an individual prescription, and you may not know in advance which patient will need it, and how much the patient will need, and it becomes unstable once it has been buffered. It also will cost the patient $45 per 5-cc syringe, and it will be degraded by the time the patient returns from the compounding pharmacy. Explain further that it costs you only 84 cents to make a 5-cc syringe of buffered lidocaine; that some patients may need as many as 10 syringes; and that these costs are all included in the surgery (free!) if the physician draws it up in the office.
A simple summary is – less pain, less cost – and no history of infections or complications.
It is an eye-opener when you demonstrate how ridiculous the compounding rules being imposed are for physicians and patients. I’ve used this demonstration at the state and federal legislative level, and more recently, at the Food and Drug Administration.
If you get the chance, when a state legislator is in your office, become an advocate for your patients and fellow physicians. Make sure physician offices are excluded from these definitions of com
This column was updated June 22, 2017.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at dermnews@frontlinemedcom.com.
In Ohio, new limits on drug compounding in physicians’ offices went into effect in April and have become a real hindrance to care for dermatology patients. The State of Ohio Board of Pharmacy has defined compounding as combining two or more prescription drugs and has required that physicians who perform this “compounding” must obtain a “Terminal Distributor of Dangerous Drugs” license. Ohio is the “test state,” and these rules, unless vigorously opposed, will be coming to your state.
[polldaddy:9779752]
The rules state that “compounded” drugs used within 6 hours of preparation must be prepared in a designated clean medication area with proper hand hygiene and the use of powder-free gloves. “Compounded” drugs that are used more than 6 hours after preparation, require a designated clean room with access limited to authorized personnel, environmental control devices such as a laminar flow hood, and additional equipment and training of personnel to maintain an aseptic environment. A separate license is required for each office location.
The state pharmacy boards are eager to restrict physicians – as well as dentists and veterinarians – and to collect annual licensing fees. Additionally, according to an article from the Ohio State Medical Association, noncompliant physicians can be fined by the pharmacy board.
We are talking big money, power, and dreams of clinical relevancy (and billable activities) here.
What can dermatologists do to prevent this regulatory overreach? I encourage you to plan a visit to your state representative, where you can demonstrate how these restrictions affect you and your patients – an exercise that should be both fun and compelling. All you need to illustrate your case is a simple kit that includes a syringe (but no needles in the statehouse!), a bottle of lidocaine with epinephrine, a bottle of 8.4% bicarbonate, alcohol pads, and gloves.
First, explain to your audience that there is a skin cancer epidemic with more than 5.4 million new cases a year and that, over the past 20 years, the incidence of skin cancer has doubled and is projected to double again over the next 20 years. Further, explain that dermatologists treat more than 70% of these cases in the office setting, under local anesthesia, at a huge cost savings to the public and government (it costs an average of 12 times as much to remove these cancers in the outpatient department at the hospital). Remember, states foot most of the bill for Medicaid and Medicare gap indigent coverage.
Take the bottle of lidocaine with epinephrine and open the syringe pack (Staffers love this demonstration; everyone is fascinated with shots.). Put on your gloves, wipe the top of the lidocaine bottle with an alcohol swab, and explain that this medicine is the anesthetic preferred for skin cancer surgery. Explain how it not only numbs the skin, but also causes vasoconstriction, so that the cancer can be easily and safely removed in the office.
Then explain that, in order for the epinephrine to be stable, the solution has to be very acidic (a pH of 4.2, in fact). Explain that this makes it burn like hell unless you add 0.1 cc per cc of 8.4% bicarbonate, in which case the perceived pain on a 10-point scale will drop from 8 to 2. Then pick up the bottle of bicarbonate and explain that you will no longer be able to mix these two components anymore without a “Terminal Distributor of Dangerous Drugs” license because your state pharmacy board considers this compounding. Your representative is likely to give you looks of astonishment, disbelief, and then a dawning realization of the absurdity of the situation.
Follow-up questions may include “Why can’t you buy buffered lidocaine with epinephrine from the compounding pharmacy?” Easy answer: because each patient needs an individual prescription, and you may not know in advance which patient will need it, and how much the patient will need, and it becomes unstable once it has been buffered. It also will cost the patient $45 per 5-cc syringe, and it will be degraded by the time the patient returns from the compounding pharmacy. Explain further that it costs you only 84 cents to make a 5-cc syringe of buffered lidocaine; that some patients may need as many as 10 syringes; and that these costs are all included in the surgery (free!) if the physician draws it up in the office.
A simple summary is – less pain, less cost – and no history of infections or complications.
It is an eye-opener when you demonstrate how ridiculous the compounding rules being imposed are for physicians and patients. I’ve used this demonstration at the state and federal legislative level, and more recently, at the Food and Drug Administration.
If you get the chance, when a state legislator is in your office, become an advocate for your patients and fellow physicians. Make sure physician offices are excluded from these definitions of com
This column was updated June 22, 2017.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at dermnews@frontlinemedcom.com.
In Ohio, new limits on drug compounding in physicians’ offices went into effect in April and have become a real hindrance to care for dermatology patients. The State of Ohio Board of Pharmacy has defined compounding as combining two or more prescription drugs and has required that physicians who perform this “compounding” must obtain a “Terminal Distributor of Dangerous Drugs” license. Ohio is the “test state,” and these rules, unless vigorously opposed, will be coming to your state.
[polldaddy:9779752]
The rules state that “compounded” drugs used within 6 hours of preparation must be prepared in a designated clean medication area with proper hand hygiene and the use of powder-free gloves. “Compounded” drugs that are used more than 6 hours after preparation, require a designated clean room with access limited to authorized personnel, environmental control devices such as a laminar flow hood, and additional equipment and training of personnel to maintain an aseptic environment. A separate license is required for each office location.
The state pharmacy boards are eager to restrict physicians – as well as dentists and veterinarians – and to collect annual licensing fees. Additionally, according to an article from the Ohio State Medical Association, noncompliant physicians can be fined by the pharmacy board.
We are talking big money, power, and dreams of clinical relevancy (and billable activities) here.
What can dermatologists do to prevent this regulatory overreach? I encourage you to plan a visit to your state representative, where you can demonstrate how these restrictions affect you and your patients – an exercise that should be both fun and compelling. All you need to illustrate your case is a simple kit that includes a syringe (but no needles in the statehouse!), a bottle of lidocaine with epinephrine, a bottle of 8.4% bicarbonate, alcohol pads, and gloves.
First, explain to your audience that there is a skin cancer epidemic with more than 5.4 million new cases a year and that, over the past 20 years, the incidence of skin cancer has doubled and is projected to double again over the next 20 years. Further, explain that dermatologists treat more than 70% of these cases in the office setting, under local anesthesia, at a huge cost savings to the public and government (it costs an average of 12 times as much to remove these cancers in the outpatient department at the hospital). Remember, states foot most of the bill for Medicaid and Medicare gap indigent coverage.
Take the bottle of lidocaine with epinephrine and open the syringe pack (Staffers love this demonstration; everyone is fascinated with shots.). Put on your gloves, wipe the top of the lidocaine bottle with an alcohol swab, and explain that this medicine is the anesthetic preferred for skin cancer surgery. Explain how it not only numbs the skin, but also causes vasoconstriction, so that the cancer can be easily and safely removed in the office.
Then explain that, in order for the epinephrine to be stable, the solution has to be very acidic (a pH of 4.2, in fact). Explain that this makes it burn like hell unless you add 0.1 cc per cc of 8.4% bicarbonate, in which case the perceived pain on a 10-point scale will drop from 8 to 2. Then pick up the bottle of bicarbonate and explain that you will no longer be able to mix these two components anymore without a “Terminal Distributor of Dangerous Drugs” license because your state pharmacy board considers this compounding. Your representative is likely to give you looks of astonishment, disbelief, and then a dawning realization of the absurdity of the situation.
Follow-up questions may include “Why can’t you buy buffered lidocaine with epinephrine from the compounding pharmacy?” Easy answer: because each patient needs an individual prescription, and you may not know in advance which patient will need it, and how much the patient will need, and it becomes unstable once it has been buffered. It also will cost the patient $45 per 5-cc syringe, and it will be degraded by the time the patient returns from the compounding pharmacy. Explain further that it costs you only 84 cents to make a 5-cc syringe of buffered lidocaine; that some patients may need as many as 10 syringes; and that these costs are all included in the surgery (free!) if the physician draws it up in the office.
A simple summary is – less pain, less cost – and no history of infections or complications.
It is an eye-opener when you demonstrate how ridiculous the compounding rules being imposed are for physicians and patients. I’ve used this demonstration at the state and federal legislative level, and more recently, at the Food and Drug Administration.
If you get the chance, when a state legislator is in your office, become an advocate for your patients and fellow physicians. Make sure physician offices are excluded from these definitions of com
This column was updated June 22, 2017.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at dermnews@frontlinemedcom.com.
Best Practices: Protecting Dry Vulnerable Skin with CeraVe® Healing Ointment
A supplement to Dermatology News. This advertising supplement is sponsored by Valeant Pharmaceuticals.
- Reinforcing the Skin Barrier
- NEA Seal of Acceptance
- A Preventative Approach to Dry, Cracked Skin
- CeraVe Ointment in the Clinical Setting
Faculty/Faculty Disclosure
Sheila Fallon Friedlander, MD
Professor of Clinical Dermatology & Pediatrics
Director, Pediatric Dermatology Fellowship Training Program
University of California at San Diego School of Medicine
Rady Children’s Hospital,
San Diego, California
Dr. Friedlander was compensated for her participation in the development of this article.
CeraVe is a registered trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
A supplement to Dermatology News. This advertising supplement is sponsored by Valeant Pharmaceuticals.
- Reinforcing the Skin Barrier
- NEA Seal of Acceptance
- A Preventative Approach to Dry, Cracked Skin
- CeraVe Ointment in the Clinical Setting
Faculty/Faculty Disclosure
Sheila Fallon Friedlander, MD
Professor of Clinical Dermatology & Pediatrics
Director, Pediatric Dermatology Fellowship Training Program
University of California at San Diego School of Medicine
Rady Children’s Hospital,
San Diego, California
Dr. Friedlander was compensated for her participation in the development of this article.
CeraVe is a registered trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
A supplement to Dermatology News. This advertising supplement is sponsored by Valeant Pharmaceuticals.
- Reinforcing the Skin Barrier
- NEA Seal of Acceptance
- A Preventative Approach to Dry, Cracked Skin
- CeraVe Ointment in the Clinical Setting
Faculty/Faculty Disclosure
Sheila Fallon Friedlander, MD
Professor of Clinical Dermatology & Pediatrics
Director, Pediatric Dermatology Fellowship Training Program
University of California at San Diego School of Medicine
Rady Children’s Hospital,
San Diego, California
Dr. Friedlander was compensated for her participation in the development of this article.
CeraVe is a registered trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
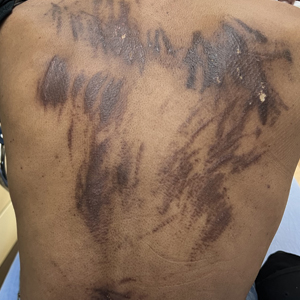
Can Fish Skin Grafts Heal Diabetic Foot Ulcers?
TOPLINE:
METHODOLOGY:
- Standard wound care for diabetic foot ulcers involves vascular assessment, surgical debridement, use of appropriate dressings, infection management, and glycemic control; however, standard care is typically associated with poor outcomes.
- Researchers conducted a multicenter clinical trial in 15 tertiary care centers with diabetic foot units across France, Italy, Germany, and Sweden to evaluate the efficacy and safety of intact fish skin grafts over standard-of-care practices in treating complex diabetic foot ulcers.
- A total of 255 patients aged 18 years or older with diabetes and lower limb wounds penetrating to the tendon, capsule, bone, or joint were randomly assigned to receive either an intact fish skin graft or standard wound care for 14 weeks.
- The primary endpoint was the percentage of wounds achieving complete closure by 16 weeks.
- Wound healing was also assessed at 20 and 24 weeks.
TAKEAWAY:
- The proportion of wounds healed at 16 weeks was higher with intact fish skin grafts than with standard-of-care (44.0% vs 26.4% adjusted odds ratio [aOR], 2.58; 95% CI, 1.48-4.56).
- The fish skin grafts continued to be more effective than standard wound care practices at weeks 20 (aOR, 2.15; 95% CI, 1.27–3.70) and 24 (aOR, 2.19; 95% CI, 1.31–3.70).
- The mean time to healing was 17.31 weeks for the intact fish skin graft group and 19.37 weeks for the standard-of-care group; intact fish skin grafts were also associated with faster healing times than standard wound care (hazard ratio, 1.59; 95% CI, 1.07-2.36).
- Target wound infections were the most common adverse events, occurring in a similar number of patients in both the groups.
IN PRACTICE:
“Our trial demonstrated treatment of complex diabetic foot ulcers with intact fish skin grafts achieved a significantly greater proportion of diabetic foot ulcers healed at 16 weeks than standard of care, and was associated with increased healing at 20 and 24 weeks. That these results were achieved in non-superficial UT [University of Texas diabetic wound classification system] grade 2 and 3 diabetic foot ulcers and included ischemic and/or infected diabetic foot ulcers is of importance,” the authors wrote.
SOURCE:
The study was led by Dured Dardari, MD, PhD, Center Hospitalier Sud Francilien, Corbeil-Essonnes, France, and was published online in NEJM Evidence.
LIMITATIONS:
No limitations were discussed for this study.
DISCLOSURES:
The study was funded by European Commission Fast Track to Innovation Horizon 2020 and Kerecis. Two authors reported being employees with or without stock options at Kerecis, and other authors reported having ties with many sources including Kerecis.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Standard wound care for diabetic foot ulcers involves vascular assessment, surgical debridement, use of appropriate dressings, infection management, and glycemic control; however, standard care is typically associated with poor outcomes.
- Researchers conducted a multicenter clinical trial in 15 tertiary care centers with diabetic foot units across France, Italy, Germany, and Sweden to evaluate the efficacy and safety of intact fish skin grafts over standard-of-care practices in treating complex diabetic foot ulcers.
- A total of 255 patients aged 18 years or older with diabetes and lower limb wounds penetrating to the tendon, capsule, bone, or joint were randomly assigned to receive either an intact fish skin graft or standard wound care for 14 weeks.
- The primary endpoint was the percentage of wounds achieving complete closure by 16 weeks.
- Wound healing was also assessed at 20 and 24 weeks.
TAKEAWAY:
- The proportion of wounds healed at 16 weeks was higher with intact fish skin grafts than with standard-of-care (44.0% vs 26.4% adjusted odds ratio [aOR], 2.58; 95% CI, 1.48-4.56).
- The fish skin grafts continued to be more effective than standard wound care practices at weeks 20 (aOR, 2.15; 95% CI, 1.27–3.70) and 24 (aOR, 2.19; 95% CI, 1.31–3.70).
- The mean time to healing was 17.31 weeks for the intact fish skin graft group and 19.37 weeks for the standard-of-care group; intact fish skin grafts were also associated with faster healing times than standard wound care (hazard ratio, 1.59; 95% CI, 1.07-2.36).
- Target wound infections were the most common adverse events, occurring in a similar number of patients in both the groups.
IN PRACTICE:
“Our trial demonstrated treatment of complex diabetic foot ulcers with intact fish skin grafts achieved a significantly greater proportion of diabetic foot ulcers healed at 16 weeks than standard of care, and was associated with increased healing at 20 and 24 weeks. That these results were achieved in non-superficial UT [University of Texas diabetic wound classification system] grade 2 and 3 diabetic foot ulcers and included ischemic and/or infected diabetic foot ulcers is of importance,” the authors wrote.
SOURCE:
The study was led by Dured Dardari, MD, PhD, Center Hospitalier Sud Francilien, Corbeil-Essonnes, France, and was published online in NEJM Evidence.
LIMITATIONS:
No limitations were discussed for this study.
DISCLOSURES:
The study was funded by European Commission Fast Track to Innovation Horizon 2020 and Kerecis. Two authors reported being employees with or without stock options at Kerecis, and other authors reported having ties with many sources including Kerecis.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Standard wound care for diabetic foot ulcers involves vascular assessment, surgical debridement, use of appropriate dressings, infection management, and glycemic control; however, standard care is typically associated with poor outcomes.
- Researchers conducted a multicenter clinical trial in 15 tertiary care centers with diabetic foot units across France, Italy, Germany, and Sweden to evaluate the efficacy and safety of intact fish skin grafts over standard-of-care practices in treating complex diabetic foot ulcers.
- A total of 255 patients aged 18 years or older with diabetes and lower limb wounds penetrating to the tendon, capsule, bone, or joint were randomly assigned to receive either an intact fish skin graft or standard wound care for 14 weeks.
- The primary endpoint was the percentage of wounds achieving complete closure by 16 weeks.
- Wound healing was also assessed at 20 and 24 weeks.
TAKEAWAY:
- The proportion of wounds healed at 16 weeks was higher with intact fish skin grafts than with standard-of-care (44.0% vs 26.4% adjusted odds ratio [aOR], 2.58; 95% CI, 1.48-4.56).
- The fish skin grafts continued to be more effective than standard wound care practices at weeks 20 (aOR, 2.15; 95% CI, 1.27–3.70) and 24 (aOR, 2.19; 95% CI, 1.31–3.70).
- The mean time to healing was 17.31 weeks for the intact fish skin graft group and 19.37 weeks for the standard-of-care group; intact fish skin grafts were also associated with faster healing times than standard wound care (hazard ratio, 1.59; 95% CI, 1.07-2.36).
- Target wound infections were the most common adverse events, occurring in a similar number of patients in both the groups.
IN PRACTICE:
“Our trial demonstrated treatment of complex diabetic foot ulcers with intact fish skin grafts achieved a significantly greater proportion of diabetic foot ulcers healed at 16 weeks than standard of care, and was associated with increased healing at 20 and 24 weeks. That these results were achieved in non-superficial UT [University of Texas diabetic wound classification system] grade 2 and 3 diabetic foot ulcers and included ischemic and/or infected diabetic foot ulcers is of importance,” the authors wrote.
SOURCE:
The study was led by Dured Dardari, MD, PhD, Center Hospitalier Sud Francilien, Corbeil-Essonnes, France, and was published online in NEJM Evidence.
LIMITATIONS:
No limitations were discussed for this study.
DISCLOSURES:
The study was funded by European Commission Fast Track to Innovation Horizon 2020 and Kerecis. Two authors reported being employees with or without stock options at Kerecis, and other authors reported having ties with many sources including Kerecis.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
AI in Medicine: Are Large Language Models Ready for the Exam Room?
In seconds, Ravi Parikh, MD, an oncologist at the Emory University School of Medicine in Atlanta, had a summary of his patient’s entire medical history. Normally, Parikh skimmed the cumbersome files before seeing a patient. However, the artificial intelligence (AI) tool his institution was testing could list the highlights he needed in a fraction of the time.
“On the whole, I like it ... it saves me time,” Parikh said of the tool. “But I’d be lying if I told you it was perfect all the time. It’s interpreting the [patient] history in some ways that may be inaccurate,” he said.
Within the first week of testing the tool, Parikh started to notice that the large language model (LLM) made a particular mistake in his patients with prostate cancer. If their prostate-specific antigen test results came back slightly elevated — which is part of normal variation — the LLM recorded it as disease progression. Because Parikh reviews all his notes — with or without using an AI tool — after a visit, he easily caught the mistake before it was added to the chart. “The problem, I think, is if these mistakes go under the hood,” he said.
In the data science world, these mistakes are called hallucinations. And a growing body of research suggests they’re happening more frequently than is safe for healthcare. The industry promised LLMs would alleviate administrative burden and reduce physician burnout. But so far, studies show these AI-tool mistakes often create more work for doctors, not less. To truly help physicians and be safe for patients, some experts say healthcare needs to build its own LLMs from the ground up. And all agree that the field desperately needs a way to vet these algorithms more thoroughly.
Prone to Error
Right now, “I think the industry is focused on taking existing LLMs and forcing them into usage for healthcare,” said Nigam H. Shah, MBBS, PhD, chief data scientist for Stanford Health. However, the value of deploying general LLMs in the healthcare space is questionable. “People are starting to wonder if we’re using these tools wrong,” he told this news organization.
In 2023, Shah and his colleagues evaluated seven LLMs on their ability to answer electronic health record–based questions. For realistic tasks, the error rate in the best cases was about 35%, he said. “To me, that rate seems a bit high ... to adopt for routine use.”
A study earlier this year by the UC San Diego School of Medicine showed that using LLMs to respond to patient messages increased the time doctors spent on messages. And this summer, a study by the clinical AI firm Mendel found that when GPT-4o or Llama-3 were used to summarize patient medical records, almost every summary contained at least one type of hallucination.
“We’ve seen cases where a patient does have drug allergies, but the system says ‘no known drug allergies’ ” in the medical history summary, said Wael Salloum, PhD, cofounder and chief science officer at Mendel. “That’s a serious hallucination.” And if physicians have to constantly verify what the system is telling them, that “defeats the purpose [of summarization],” he said.
A Higher Quality Diet
Part of the trouble with LLMs is that there’s just not enough high-quality information to feed them. The algorithms are insatiable, requiring vast swaths of data for training. GPT-3.5, for instance, was trained on 570 GB of data from the internet, more than 300 billion words. And to train GPT-4o, OpenAI reportedly transcribed more than 1 million hours of YouTube content.
However, the strategies that built these general LLMs don’t always translate well to healthcare. The internet is full of low-quality or misleading health information from wellness sites and supplement advertisements. And even data that are trustworthy, like the millions of clinical studies and the US Food and Drug Administration (FDA) statements, can be outdated, Salloum said. And “an LLM in training can’t distinguish good from bad,” he added.
The good news is that clinicians don’t rely on controversial information in the real world. Medical knowledge is standardized. “Healthcare is a domain rich with explicit knowledge,” Salloum said. So there’s potential to build a more reliable LLM that is guided by robust medical standards and guidelines.
It’s possible that healthcare could use small language models, which are LLM’s pocket-sized cousins, and perform tasks needing only bite-sized datasets requiring fewer resources and easier fine-tuning, according to Microsoft’s website. Shah said training these smaller models on real medical data might be an option, like an LLM meant to respond to patient messages that could be trained with real messages sent by physicians.
Several groups are already working on databases of standardized human medical knowledge or real physician responses. “Perhaps that will work better than using LLMs trained on the general internet. Those studies need to be done,” Shah said.
Jon Tamir, assistant professor of electrical and computer engineering and co-lead of the AI Health Lab at The University of Texas at Austin, said, “The community has recognized that we are entering a new era of AI where the dataset itself is the most important aspect. We need training sets that are highly curated and highly specialized.
“If the dataset is highly specialized, it will definitely help reduce hallucinations,” he said.
Cutting Overconfidence
A major problem with LLM mistakes is that they are often hard to detect. Hallucinations can be highly convincing even if they’re highly inaccurate, according to Tamir.
When Shah, for instance, was recently testing an LLM on de-identified patient data, he asked the LLM which blood test the patient last had. The model responded with “complete blood count [CBC].” But when he asked for the results, the model gave him white blood count and other values. “Turns out that record did not have a CBC done at all! The result was entirely made up,” he said.
Making healthcare LLMs safer and more reliable will mean training AI to acknowledge potential mistakes and uncertainty. Existing LLMs are trained to project confidence and produce a lot of answers, even when there isn’t one, Salloum said. They rarely respond with “I don’t know” even when their prediction has low confidence, he added.
Healthcare stands to benefit from a system that highlights uncertainty and potential errors. For instance, if a patient’s history shows they have smoked, stopped smoking, vaped, and started smoking again. The LLM might call them a smoker but flag the comment as uncertain because the chronology is complicated, Salloum said.
Tamir added that this strategy could improve LLM and doctor collaboration by honing in on where human expertise is needed most.
Too Little Evaluation
For any improvement strategy to work, LLMs — and all AI-assisted healthcare tools — first need a better evaluation framework. So far, LLMs have “been used in really exciting ways but not really well-vetted ways,” Tamir said.
While some AI-assisted tools, particularly in medical imaging, have undergone rigorous FDA evaluations and earned approval, most haven’t. And because the FDA only regulates algorithms that are considered medical devices, Parikh said that most LLMs used for administrative tasks and efficiency don’t fall under the regulatory agency’s purview.
But these algorithms still have access to patient information and can directly influence patient and doctor decisions. Third-party regulatory agencies are expected to emerge, but it’s still unclear who those will be. Before developers can build a safer and more efficient LLM for healthcare, they’ll need better guidelines and guardrails. “Unless we figure out evaluation, how would we know whether the healthcare-appropriate large language models are better or worse?” Shah asked.
A version of this article appeared on Medscape.com.
In seconds, Ravi Parikh, MD, an oncologist at the Emory University School of Medicine in Atlanta, had a summary of his patient’s entire medical history. Normally, Parikh skimmed the cumbersome files before seeing a patient. However, the artificial intelligence (AI) tool his institution was testing could list the highlights he needed in a fraction of the time.
“On the whole, I like it ... it saves me time,” Parikh said of the tool. “But I’d be lying if I told you it was perfect all the time. It’s interpreting the [patient] history in some ways that may be inaccurate,” he said.
Within the first week of testing the tool, Parikh started to notice that the large language model (LLM) made a particular mistake in his patients with prostate cancer. If their prostate-specific antigen test results came back slightly elevated — which is part of normal variation — the LLM recorded it as disease progression. Because Parikh reviews all his notes — with or without using an AI tool — after a visit, he easily caught the mistake before it was added to the chart. “The problem, I think, is if these mistakes go under the hood,” he said.
In the data science world, these mistakes are called hallucinations. And a growing body of research suggests they’re happening more frequently than is safe for healthcare. The industry promised LLMs would alleviate administrative burden and reduce physician burnout. But so far, studies show these AI-tool mistakes often create more work for doctors, not less. To truly help physicians and be safe for patients, some experts say healthcare needs to build its own LLMs from the ground up. And all agree that the field desperately needs a way to vet these algorithms more thoroughly.
Prone to Error
Right now, “I think the industry is focused on taking existing LLMs and forcing them into usage for healthcare,” said Nigam H. Shah, MBBS, PhD, chief data scientist for Stanford Health. However, the value of deploying general LLMs in the healthcare space is questionable. “People are starting to wonder if we’re using these tools wrong,” he told this news organization.
In 2023, Shah and his colleagues evaluated seven LLMs on their ability to answer electronic health record–based questions. For realistic tasks, the error rate in the best cases was about 35%, he said. “To me, that rate seems a bit high ... to adopt for routine use.”
A study earlier this year by the UC San Diego School of Medicine showed that using LLMs to respond to patient messages increased the time doctors spent on messages. And this summer, a study by the clinical AI firm Mendel found that when GPT-4o or Llama-3 were used to summarize patient medical records, almost every summary contained at least one type of hallucination.
“We’ve seen cases where a patient does have drug allergies, but the system says ‘no known drug allergies’ ” in the medical history summary, said Wael Salloum, PhD, cofounder and chief science officer at Mendel. “That’s a serious hallucination.” And if physicians have to constantly verify what the system is telling them, that “defeats the purpose [of summarization],” he said.
A Higher Quality Diet
Part of the trouble with LLMs is that there’s just not enough high-quality information to feed them. The algorithms are insatiable, requiring vast swaths of data for training. GPT-3.5, for instance, was trained on 570 GB of data from the internet, more than 300 billion words. And to train GPT-4o, OpenAI reportedly transcribed more than 1 million hours of YouTube content.
However, the strategies that built these general LLMs don’t always translate well to healthcare. The internet is full of low-quality or misleading health information from wellness sites and supplement advertisements. And even data that are trustworthy, like the millions of clinical studies and the US Food and Drug Administration (FDA) statements, can be outdated, Salloum said. And “an LLM in training can’t distinguish good from bad,” he added.
The good news is that clinicians don’t rely on controversial information in the real world. Medical knowledge is standardized. “Healthcare is a domain rich with explicit knowledge,” Salloum said. So there’s potential to build a more reliable LLM that is guided by robust medical standards and guidelines.
It’s possible that healthcare could use small language models, which are LLM’s pocket-sized cousins, and perform tasks needing only bite-sized datasets requiring fewer resources and easier fine-tuning, according to Microsoft’s website. Shah said training these smaller models on real medical data might be an option, like an LLM meant to respond to patient messages that could be trained with real messages sent by physicians.
Several groups are already working on databases of standardized human medical knowledge or real physician responses. “Perhaps that will work better than using LLMs trained on the general internet. Those studies need to be done,” Shah said.
Jon Tamir, assistant professor of electrical and computer engineering and co-lead of the AI Health Lab at The University of Texas at Austin, said, “The community has recognized that we are entering a new era of AI where the dataset itself is the most important aspect. We need training sets that are highly curated and highly specialized.
“If the dataset is highly specialized, it will definitely help reduce hallucinations,” he said.
Cutting Overconfidence
A major problem with LLM mistakes is that they are often hard to detect. Hallucinations can be highly convincing even if they’re highly inaccurate, according to Tamir.
When Shah, for instance, was recently testing an LLM on de-identified patient data, he asked the LLM which blood test the patient last had. The model responded with “complete blood count [CBC].” But when he asked for the results, the model gave him white blood count and other values. “Turns out that record did not have a CBC done at all! The result was entirely made up,” he said.
Making healthcare LLMs safer and more reliable will mean training AI to acknowledge potential mistakes and uncertainty. Existing LLMs are trained to project confidence and produce a lot of answers, even when there isn’t one, Salloum said. They rarely respond with “I don’t know” even when their prediction has low confidence, he added.
Healthcare stands to benefit from a system that highlights uncertainty and potential errors. For instance, if a patient’s history shows they have smoked, stopped smoking, vaped, and started smoking again. The LLM might call them a smoker but flag the comment as uncertain because the chronology is complicated, Salloum said.
Tamir added that this strategy could improve LLM and doctor collaboration by honing in on where human expertise is needed most.
Too Little Evaluation
For any improvement strategy to work, LLMs — and all AI-assisted healthcare tools — first need a better evaluation framework. So far, LLMs have “been used in really exciting ways but not really well-vetted ways,” Tamir said.
While some AI-assisted tools, particularly in medical imaging, have undergone rigorous FDA evaluations and earned approval, most haven’t. And because the FDA only regulates algorithms that are considered medical devices, Parikh said that most LLMs used for administrative tasks and efficiency don’t fall under the regulatory agency’s purview.
But these algorithms still have access to patient information and can directly influence patient and doctor decisions. Third-party regulatory agencies are expected to emerge, but it’s still unclear who those will be. Before developers can build a safer and more efficient LLM for healthcare, they’ll need better guidelines and guardrails. “Unless we figure out evaluation, how would we know whether the healthcare-appropriate large language models are better or worse?” Shah asked.
A version of this article appeared on Medscape.com.
In seconds, Ravi Parikh, MD, an oncologist at the Emory University School of Medicine in Atlanta, had a summary of his patient’s entire medical history. Normally, Parikh skimmed the cumbersome files before seeing a patient. However, the artificial intelligence (AI) tool his institution was testing could list the highlights he needed in a fraction of the time.
“On the whole, I like it ... it saves me time,” Parikh said of the tool. “But I’d be lying if I told you it was perfect all the time. It’s interpreting the [patient] history in some ways that may be inaccurate,” he said.
Within the first week of testing the tool, Parikh started to notice that the large language model (LLM) made a particular mistake in his patients with prostate cancer. If their prostate-specific antigen test results came back slightly elevated — which is part of normal variation — the LLM recorded it as disease progression. Because Parikh reviews all his notes — with or without using an AI tool — after a visit, he easily caught the mistake before it was added to the chart. “The problem, I think, is if these mistakes go under the hood,” he said.
In the data science world, these mistakes are called hallucinations. And a growing body of research suggests they’re happening more frequently than is safe for healthcare. The industry promised LLMs would alleviate administrative burden and reduce physician burnout. But so far, studies show these AI-tool mistakes often create more work for doctors, not less. To truly help physicians and be safe for patients, some experts say healthcare needs to build its own LLMs from the ground up. And all agree that the field desperately needs a way to vet these algorithms more thoroughly.
Prone to Error
Right now, “I think the industry is focused on taking existing LLMs and forcing them into usage for healthcare,” said Nigam H. Shah, MBBS, PhD, chief data scientist for Stanford Health. However, the value of deploying general LLMs in the healthcare space is questionable. “People are starting to wonder if we’re using these tools wrong,” he told this news organization.
In 2023, Shah and his colleagues evaluated seven LLMs on their ability to answer electronic health record–based questions. For realistic tasks, the error rate in the best cases was about 35%, he said. “To me, that rate seems a bit high ... to adopt for routine use.”
A study earlier this year by the UC San Diego School of Medicine showed that using LLMs to respond to patient messages increased the time doctors spent on messages. And this summer, a study by the clinical AI firm Mendel found that when GPT-4o or Llama-3 were used to summarize patient medical records, almost every summary contained at least one type of hallucination.
“We’ve seen cases where a patient does have drug allergies, but the system says ‘no known drug allergies’ ” in the medical history summary, said Wael Salloum, PhD, cofounder and chief science officer at Mendel. “That’s a serious hallucination.” And if physicians have to constantly verify what the system is telling them, that “defeats the purpose [of summarization],” he said.
A Higher Quality Diet
Part of the trouble with LLMs is that there’s just not enough high-quality information to feed them. The algorithms are insatiable, requiring vast swaths of data for training. GPT-3.5, for instance, was trained on 570 GB of data from the internet, more than 300 billion words. And to train GPT-4o, OpenAI reportedly transcribed more than 1 million hours of YouTube content.
However, the strategies that built these general LLMs don’t always translate well to healthcare. The internet is full of low-quality or misleading health information from wellness sites and supplement advertisements. And even data that are trustworthy, like the millions of clinical studies and the US Food and Drug Administration (FDA) statements, can be outdated, Salloum said. And “an LLM in training can’t distinguish good from bad,” he added.
The good news is that clinicians don’t rely on controversial information in the real world. Medical knowledge is standardized. “Healthcare is a domain rich with explicit knowledge,” Salloum said. So there’s potential to build a more reliable LLM that is guided by robust medical standards and guidelines.
It’s possible that healthcare could use small language models, which are LLM’s pocket-sized cousins, and perform tasks needing only bite-sized datasets requiring fewer resources and easier fine-tuning, according to Microsoft’s website. Shah said training these smaller models on real medical data might be an option, like an LLM meant to respond to patient messages that could be trained with real messages sent by physicians.
Several groups are already working on databases of standardized human medical knowledge or real physician responses. “Perhaps that will work better than using LLMs trained on the general internet. Those studies need to be done,” Shah said.
Jon Tamir, assistant professor of electrical and computer engineering and co-lead of the AI Health Lab at The University of Texas at Austin, said, “The community has recognized that we are entering a new era of AI where the dataset itself is the most important aspect. We need training sets that are highly curated and highly specialized.
“If the dataset is highly specialized, it will definitely help reduce hallucinations,” he said.
Cutting Overconfidence
A major problem with LLM mistakes is that they are often hard to detect. Hallucinations can be highly convincing even if they’re highly inaccurate, according to Tamir.
When Shah, for instance, was recently testing an LLM on de-identified patient data, he asked the LLM which blood test the patient last had. The model responded with “complete blood count [CBC].” But when he asked for the results, the model gave him white blood count and other values. “Turns out that record did not have a CBC done at all! The result was entirely made up,” he said.
Making healthcare LLMs safer and more reliable will mean training AI to acknowledge potential mistakes and uncertainty. Existing LLMs are trained to project confidence and produce a lot of answers, even when there isn’t one, Salloum said. They rarely respond with “I don’t know” even when their prediction has low confidence, he added.
Healthcare stands to benefit from a system that highlights uncertainty and potential errors. For instance, if a patient’s history shows they have smoked, stopped smoking, vaped, and started smoking again. The LLM might call them a smoker but flag the comment as uncertain because the chronology is complicated, Salloum said.
Tamir added that this strategy could improve LLM and doctor collaboration by honing in on where human expertise is needed most.
Too Little Evaluation
For any improvement strategy to work, LLMs — and all AI-assisted healthcare tools — first need a better evaluation framework. So far, LLMs have “been used in really exciting ways but not really well-vetted ways,” Tamir said.
While some AI-assisted tools, particularly in medical imaging, have undergone rigorous FDA evaluations and earned approval, most haven’t. And because the FDA only regulates algorithms that are considered medical devices, Parikh said that most LLMs used for administrative tasks and efficiency don’t fall under the regulatory agency’s purview.
But these algorithms still have access to patient information and can directly influence patient and doctor decisions. Third-party regulatory agencies are expected to emerge, but it’s still unclear who those will be. Before developers can build a safer and more efficient LLM for healthcare, they’ll need better guidelines and guardrails. “Unless we figure out evaluation, how would we know whether the healthcare-appropriate large language models are better or worse?” Shah asked.
A version of this article appeared on Medscape.com.
Cybersecurity Concerns Continue to Rise With Ransom, Data Manipulation, AI Risks
From the largest healthcare companies to solo practices, just every organization in medicine faces a risk for costly cyberattacks. In recent years, hackers have threatened to release the personal information of patients and employees — or paralyze online systems — unless they’re paid a ransom.
Should companies pay? It’s not an easy answer, a pair of experts told colleagues in an American Medical Association (AMA) cybersecurity webinar on October 18. It turns out that each choice — pay or don’t pay — can end up being costly.
This is just one of the new challenges facing the American medical system on the cybersecurity front, the speakers said. Others include the possibility that hackers will manipulate patient data — turning a medical test negative, for example, when it’s actually positive — and take advantage of the powers of artificial intelligence (AI).
The AMA held the webinar to educate physicians about cybersecurity risks and defenses, an especially hot topic in the wake of February’s Change Healthcare hack, which cost UnitedHealth Group an estimated $2.5 billion — so far — and deeply disrupted the American healthcare system.
Cautionary tales abound. Greg Garcia, executive director for cybersecurity of the Health Sector Coordinating Council, a coalition of medical industry organizations, pointed to a Pennsylvania clinic that refused to pay a ransom to prevent the release of hundreds of images of patients with breast cancer undressed from the waist up. Garcia told webinar participants that the ransom was $5 million.
Risky Choices
While the Federal Bureau of Investigation recommends against paying a ransom, this can be a risky choice, Garcia said. Hackers released the images, and the center has reportedly agreed to settle a class-action lawsuit for $65 million. “They traded $5 million for $60 million,” Garcia added, slightly misstating the settlement amount.
Health systems have been cagey about whether they’ve paid ransoms to prevent private data from being made public in cyberattacks. If a ransom is demanded, “it’s every organization for itself,” Garcia said.
He highlighted the case of a chain of psychiatry practices in Finland that suffered a ransomware attack in 2020. The hackers “contacted the patients and said: ‘Hey, call your clinic and tell them to pay the ransom. Otherwise, we’re going to release all your psychiatric notes to the public.’ ”
Cyberattacks continue. In October, Boston Children’s Health Physicians announced that it had suffered a “ recent security incident” involving data — possibly including Social Security numbers and treatment information — regarding patients and employees. A hacker group reportedly claimed responsibility and wants the system, which boasts more than 300 clinicians, to pay a ransom or else it will release the stolen information.
Should Paying Ransom Be a Crime?
Christian Dameff, MD, MS, an emergency medicine physician and director of the Center for Healthcare Cybersecurity at the University of California (UC), San Diego, noted that there are efforts to turn paying ransom into a crime. “If people aren’t paying ransoms, then ransomware operators will move to something else that makes them money.”
Dameff urged colleagues to understand we no longer live in a world where clinicians only bother to think of technology when they call the IT department to help them reset their password.
New challenges face clinicians, he said. “How do we develop better strategies, downtime procedures, and safe clinical care in an era where our vital technology may be gone, not just for an hour or 2, but as is the case with these ransomware attacks, sometimes weeks to months.”
Garcia said “cybersecurity is everybody’s responsibility, including frontline clinicians. Because you’re touching data, you’re touching technology, you’re touching patients, and all of those things combine to present some vulnerabilities in the digital world.”
Next Frontier: Hackers May Manipulate Patient Data
Dameff said future hackers may use AI to manipulate individual patient data in ways that threaten patient health. AI makes this easier to accomplish.
“What if I delete your allergies in your electronic health record, or I manipulate your chest x-ray, or I change your lab values so it looks like you’re in diabetic ketoacidosis when you’re not so a clinician gives you insulin when you don’t need it?”
Garcia highlighted another new threat: Phishing efforts that are harder to ignore thanks to AI.
“One of the most successful way that hackers get in, disrupt systems, and steal data is through email phishing, and it’s only going to get better because of artificial intelligence,” he said. “No longer are you going to have typos in that email written by a hacking group in Nigeria or in China. It’s going to be perfect looking.”
What can practices and healthcare systems do? Garcia highlighted federal health agency efforts to encourage organizations to adopt best practices in cybersecurity.
“If you’ve got a data breach, and you can show to the US Department of Health & Human Services [HHS] you have implemented generally recognized cybersecurity controls over the past year, that you have done your best, you did the right thing, and you still got hit, HHS is directed to essentially take it easy on you,” he said. “That’s a positive incentive.”
Ransomware Guide in the Works
Dameff said UC San Diego’s Center for Healthcare Cybersecurity plans to publish a free cybersecurity guide in 2025 that will include specific information about ransomware attacks for medical specialties such as cardiology, trauma surgery, and pediatrics.
“Then, should you ever be ransomed, you can pull out this guide. You’ll know what’s going to kind of happen, and you can better prepare for those effects.”
Will the future president prioritize healthcare cybersecurity? That remains to be seen, but crises do have the capacity to concentrate the mind, experts said.
The nation’s capital “has a very short memory, a short attention span. The policymakers tend to be reactive,” Dameff said. “All it takes is yet another Change Healthcare–like attack that disrupts 30% or more of the nation’s healthcare system for the policymakers to sit up, take notice, and try to come up with solutions.”
In addition, he said, an estimated two data breaches/ransomware attacks are occurring per day. “The fact is that we’re all patients, up to the President of the United States and every member of the Congress is a patient.”
There’s a “very existential, very palpable understanding that cyber safety is patient safety and cyber insecurity is patient insecurity,” Dameff said.
A version of this article appeared on Medscape.com.
From the largest healthcare companies to solo practices, just every organization in medicine faces a risk for costly cyberattacks. In recent years, hackers have threatened to release the personal information of patients and employees — or paralyze online systems — unless they’re paid a ransom.
Should companies pay? It’s not an easy answer, a pair of experts told colleagues in an American Medical Association (AMA) cybersecurity webinar on October 18. It turns out that each choice — pay or don’t pay — can end up being costly.
This is just one of the new challenges facing the American medical system on the cybersecurity front, the speakers said. Others include the possibility that hackers will manipulate patient data — turning a medical test negative, for example, when it’s actually positive — and take advantage of the powers of artificial intelligence (AI).
The AMA held the webinar to educate physicians about cybersecurity risks and defenses, an especially hot topic in the wake of February’s Change Healthcare hack, which cost UnitedHealth Group an estimated $2.5 billion — so far — and deeply disrupted the American healthcare system.
Cautionary tales abound. Greg Garcia, executive director for cybersecurity of the Health Sector Coordinating Council, a coalition of medical industry organizations, pointed to a Pennsylvania clinic that refused to pay a ransom to prevent the release of hundreds of images of patients with breast cancer undressed from the waist up. Garcia told webinar participants that the ransom was $5 million.
Risky Choices
While the Federal Bureau of Investigation recommends against paying a ransom, this can be a risky choice, Garcia said. Hackers released the images, and the center has reportedly agreed to settle a class-action lawsuit for $65 million. “They traded $5 million for $60 million,” Garcia added, slightly misstating the settlement amount.
Health systems have been cagey about whether they’ve paid ransoms to prevent private data from being made public in cyberattacks. If a ransom is demanded, “it’s every organization for itself,” Garcia said.
He highlighted the case of a chain of psychiatry practices in Finland that suffered a ransomware attack in 2020. The hackers “contacted the patients and said: ‘Hey, call your clinic and tell them to pay the ransom. Otherwise, we’re going to release all your psychiatric notes to the public.’ ”
Cyberattacks continue. In October, Boston Children’s Health Physicians announced that it had suffered a “ recent security incident” involving data — possibly including Social Security numbers and treatment information — regarding patients and employees. A hacker group reportedly claimed responsibility and wants the system, which boasts more than 300 clinicians, to pay a ransom or else it will release the stolen information.
Should Paying Ransom Be a Crime?
Christian Dameff, MD, MS, an emergency medicine physician and director of the Center for Healthcare Cybersecurity at the University of California (UC), San Diego, noted that there are efforts to turn paying ransom into a crime. “If people aren’t paying ransoms, then ransomware operators will move to something else that makes them money.”
Dameff urged colleagues to understand we no longer live in a world where clinicians only bother to think of technology when they call the IT department to help them reset their password.
New challenges face clinicians, he said. “How do we develop better strategies, downtime procedures, and safe clinical care in an era where our vital technology may be gone, not just for an hour or 2, but as is the case with these ransomware attacks, sometimes weeks to months.”
Garcia said “cybersecurity is everybody’s responsibility, including frontline clinicians. Because you’re touching data, you’re touching technology, you’re touching patients, and all of those things combine to present some vulnerabilities in the digital world.”
Next Frontier: Hackers May Manipulate Patient Data
Dameff said future hackers may use AI to manipulate individual patient data in ways that threaten patient health. AI makes this easier to accomplish.
“What if I delete your allergies in your electronic health record, or I manipulate your chest x-ray, or I change your lab values so it looks like you’re in diabetic ketoacidosis when you’re not so a clinician gives you insulin when you don’t need it?”
Garcia highlighted another new threat: Phishing efforts that are harder to ignore thanks to AI.
“One of the most successful way that hackers get in, disrupt systems, and steal data is through email phishing, and it’s only going to get better because of artificial intelligence,” he said. “No longer are you going to have typos in that email written by a hacking group in Nigeria or in China. It’s going to be perfect looking.”
What can practices and healthcare systems do? Garcia highlighted federal health agency efforts to encourage organizations to adopt best practices in cybersecurity.
“If you’ve got a data breach, and you can show to the US Department of Health & Human Services [HHS] you have implemented generally recognized cybersecurity controls over the past year, that you have done your best, you did the right thing, and you still got hit, HHS is directed to essentially take it easy on you,” he said. “That’s a positive incentive.”
Ransomware Guide in the Works
Dameff said UC San Diego’s Center for Healthcare Cybersecurity plans to publish a free cybersecurity guide in 2025 that will include specific information about ransomware attacks for medical specialties such as cardiology, trauma surgery, and pediatrics.
“Then, should you ever be ransomed, you can pull out this guide. You’ll know what’s going to kind of happen, and you can better prepare for those effects.”
Will the future president prioritize healthcare cybersecurity? That remains to be seen, but crises do have the capacity to concentrate the mind, experts said.
The nation’s capital “has a very short memory, a short attention span. The policymakers tend to be reactive,” Dameff said. “All it takes is yet another Change Healthcare–like attack that disrupts 30% or more of the nation’s healthcare system for the policymakers to sit up, take notice, and try to come up with solutions.”
In addition, he said, an estimated two data breaches/ransomware attacks are occurring per day. “The fact is that we’re all patients, up to the President of the United States and every member of the Congress is a patient.”
There’s a “very existential, very palpable understanding that cyber safety is patient safety and cyber insecurity is patient insecurity,” Dameff said.
A version of this article appeared on Medscape.com.
From the largest healthcare companies to solo practices, just every organization in medicine faces a risk for costly cyberattacks. In recent years, hackers have threatened to release the personal information of patients and employees — or paralyze online systems — unless they’re paid a ransom.
Should companies pay? It’s not an easy answer, a pair of experts told colleagues in an American Medical Association (AMA) cybersecurity webinar on October 18. It turns out that each choice — pay or don’t pay — can end up being costly.
This is just one of the new challenges facing the American medical system on the cybersecurity front, the speakers said. Others include the possibility that hackers will manipulate patient data — turning a medical test negative, for example, when it’s actually positive — and take advantage of the powers of artificial intelligence (AI).
The AMA held the webinar to educate physicians about cybersecurity risks and defenses, an especially hot topic in the wake of February’s Change Healthcare hack, which cost UnitedHealth Group an estimated $2.5 billion — so far — and deeply disrupted the American healthcare system.
Cautionary tales abound. Greg Garcia, executive director for cybersecurity of the Health Sector Coordinating Council, a coalition of medical industry organizations, pointed to a Pennsylvania clinic that refused to pay a ransom to prevent the release of hundreds of images of patients with breast cancer undressed from the waist up. Garcia told webinar participants that the ransom was $5 million.
Risky Choices
While the Federal Bureau of Investigation recommends against paying a ransom, this can be a risky choice, Garcia said. Hackers released the images, and the center has reportedly agreed to settle a class-action lawsuit for $65 million. “They traded $5 million for $60 million,” Garcia added, slightly misstating the settlement amount.
Health systems have been cagey about whether they’ve paid ransoms to prevent private data from being made public in cyberattacks. If a ransom is demanded, “it’s every organization for itself,” Garcia said.
He highlighted the case of a chain of psychiatry practices in Finland that suffered a ransomware attack in 2020. The hackers “contacted the patients and said: ‘Hey, call your clinic and tell them to pay the ransom. Otherwise, we’re going to release all your psychiatric notes to the public.’ ”
Cyberattacks continue. In October, Boston Children’s Health Physicians announced that it had suffered a “ recent security incident” involving data — possibly including Social Security numbers and treatment information — regarding patients and employees. A hacker group reportedly claimed responsibility and wants the system, which boasts more than 300 clinicians, to pay a ransom or else it will release the stolen information.
Should Paying Ransom Be a Crime?
Christian Dameff, MD, MS, an emergency medicine physician and director of the Center for Healthcare Cybersecurity at the University of California (UC), San Diego, noted that there are efforts to turn paying ransom into a crime. “If people aren’t paying ransoms, then ransomware operators will move to something else that makes them money.”
Dameff urged colleagues to understand we no longer live in a world where clinicians only bother to think of technology when they call the IT department to help them reset their password.
New challenges face clinicians, he said. “How do we develop better strategies, downtime procedures, and safe clinical care in an era where our vital technology may be gone, not just for an hour or 2, but as is the case with these ransomware attacks, sometimes weeks to months.”
Garcia said “cybersecurity is everybody’s responsibility, including frontline clinicians. Because you’re touching data, you’re touching technology, you’re touching patients, and all of those things combine to present some vulnerabilities in the digital world.”
Next Frontier: Hackers May Manipulate Patient Data
Dameff said future hackers may use AI to manipulate individual patient data in ways that threaten patient health. AI makes this easier to accomplish.
“What if I delete your allergies in your electronic health record, or I manipulate your chest x-ray, or I change your lab values so it looks like you’re in diabetic ketoacidosis when you’re not so a clinician gives you insulin when you don’t need it?”
Garcia highlighted another new threat: Phishing efforts that are harder to ignore thanks to AI.
“One of the most successful way that hackers get in, disrupt systems, and steal data is through email phishing, and it’s only going to get better because of artificial intelligence,” he said. “No longer are you going to have typos in that email written by a hacking group in Nigeria or in China. It’s going to be perfect looking.”
What can practices and healthcare systems do? Garcia highlighted federal health agency efforts to encourage organizations to adopt best practices in cybersecurity.
“If you’ve got a data breach, and you can show to the US Department of Health & Human Services [HHS] you have implemented generally recognized cybersecurity controls over the past year, that you have done your best, you did the right thing, and you still got hit, HHS is directed to essentially take it easy on you,” he said. “That’s a positive incentive.”
Ransomware Guide in the Works
Dameff said UC San Diego’s Center for Healthcare Cybersecurity plans to publish a free cybersecurity guide in 2025 that will include specific information about ransomware attacks for medical specialties such as cardiology, trauma surgery, and pediatrics.
“Then, should you ever be ransomed, you can pull out this guide. You’ll know what’s going to kind of happen, and you can better prepare for those effects.”
Will the future president prioritize healthcare cybersecurity? That remains to be seen, but crises do have the capacity to concentrate the mind, experts said.
The nation’s capital “has a very short memory, a short attention span. The policymakers tend to be reactive,” Dameff said. “All it takes is yet another Change Healthcare–like attack that disrupts 30% or more of the nation’s healthcare system for the policymakers to sit up, take notice, and try to come up with solutions.”
In addition, he said, an estimated two data breaches/ransomware attacks are occurring per day. “The fact is that we’re all patients, up to the President of the United States and every member of the Congress is a patient.”
There’s a “very existential, very palpable understanding that cyber safety is patient safety and cyber insecurity is patient insecurity,” Dameff said.
A version of this article appeared on Medscape.com.
ICD-10-CM Codes for CCCA, FFA Now Available
in the field of hair loss disorders.
“CCCA and FFA are conditions that require early diagnosis and intervention to prevent irreversible hair loss,” Maria Hordinsky, MD, professor of dermatology at the University of Minnesota, Minneapolis, and a member of the Board of Directors, Scarring Alopecia Foundation (SAF), said in an interview.
“The use of these new codes will make it easier for clinicians to identify affected patients and improve treatment outcomes. It also opens the door for more robust research efforts aimed at understanding the etiology and progression of CCCA and FFA, which could lead to new and more effective treatments in the future. Overall, this development represents a positive step toward improving care for individuals affected by these challenging conditions.”
The new codes — L66.81 for CCCA and L66.12 for FFA — were approved by the Centers for Disease Control and Prevention (CDC) on June 15, 2023, but not implemented until October 1, 2024.
Amy J. McMichael, MD, professor of dermatology at Wake Forest University School of Medicine, Winston-Salem, North Carolina, and a scientific advisor to SAF, told this news organization that Itisha Jefferson, a medical student at Loyola University Chicago’s Stritch School of Medicine, and her peers on the SAF’s Medical Student Executive Board, played a pivotal role in advocating for the codes.
In 2022, Jefferson, who has CCCA, and her fellow medical students helped create the proposals that were ultimately submitted to the CDC.
“They were critical in working with the CDC leaders to get the necessary information submitted and processed,” McMichael said. “They were also amazing at corralling our dermatologist group for the development of the necessary presentations and helped to shepherd us to the finish line for all logistic issues.”
On March 8, 2023, McMichael and Hordinsky made their pitch for the codes in person at the CDC’s ICD-10 Coordination and Maintenance Committee meeting, with McMichael discussing CCCA and Hordinsky discussing FFA.
“We also discussed the lack of standardized tracking, which has contributed to misdiagnoses and inadequate treatment options,” Hordinsky recalled. “We highlighted the importance of having distinct codes for these conditions to improve clinical outcomes, ensure that patients have access to appropriate care, better tracking of disease prevalence, and greater epidemiologic monitoring with access to electronic medical records and other large real-world evidence datasets and databases, the results of which could contribute to health policy decision-making.”
To spread the word about the new codes, McMichael, Hordinsky, and other members of the SAF are working with the original team of medical students, some of whom who are now dermatology residents, to develop an information guide to send to societies and organizations that were supportive of the codes. A publication in the dermatology literature is also planned.
For her part, Jefferson said that she will continue to advocate for patients with scarring alopecia as a medical student and when she becomes a physician. “I hope in the near future we will see an externally led FDA Patient-Focused Drug Development meeting for both CCCA and FFA, further advancing care and research for these conditions,” she said in an interview.
McMichael, Hordinsky, and Jefferson had no relevant disclosures to report.
A version of this article appeared on Medscape.com.
in the field of hair loss disorders.
“CCCA and FFA are conditions that require early diagnosis and intervention to prevent irreversible hair loss,” Maria Hordinsky, MD, professor of dermatology at the University of Minnesota, Minneapolis, and a member of the Board of Directors, Scarring Alopecia Foundation (SAF), said in an interview.
“The use of these new codes will make it easier for clinicians to identify affected patients and improve treatment outcomes. It also opens the door for more robust research efforts aimed at understanding the etiology and progression of CCCA and FFA, which could lead to new and more effective treatments in the future. Overall, this development represents a positive step toward improving care for individuals affected by these challenging conditions.”
The new codes — L66.81 for CCCA and L66.12 for FFA — were approved by the Centers for Disease Control and Prevention (CDC) on June 15, 2023, but not implemented until October 1, 2024.
Amy J. McMichael, MD, professor of dermatology at Wake Forest University School of Medicine, Winston-Salem, North Carolina, and a scientific advisor to SAF, told this news organization that Itisha Jefferson, a medical student at Loyola University Chicago’s Stritch School of Medicine, and her peers on the SAF’s Medical Student Executive Board, played a pivotal role in advocating for the codes.
In 2022, Jefferson, who has CCCA, and her fellow medical students helped create the proposals that were ultimately submitted to the CDC.
“They were critical in working with the CDC leaders to get the necessary information submitted and processed,” McMichael said. “They were also amazing at corralling our dermatologist group for the development of the necessary presentations and helped to shepherd us to the finish line for all logistic issues.”
On March 8, 2023, McMichael and Hordinsky made their pitch for the codes in person at the CDC’s ICD-10 Coordination and Maintenance Committee meeting, with McMichael discussing CCCA and Hordinsky discussing FFA.
“We also discussed the lack of standardized tracking, which has contributed to misdiagnoses and inadequate treatment options,” Hordinsky recalled. “We highlighted the importance of having distinct codes for these conditions to improve clinical outcomes, ensure that patients have access to appropriate care, better tracking of disease prevalence, and greater epidemiologic monitoring with access to electronic medical records and other large real-world evidence datasets and databases, the results of which could contribute to health policy decision-making.”
To spread the word about the new codes, McMichael, Hordinsky, and other members of the SAF are working with the original team of medical students, some of whom who are now dermatology residents, to develop an information guide to send to societies and organizations that were supportive of the codes. A publication in the dermatology literature is also planned.
For her part, Jefferson said that she will continue to advocate for patients with scarring alopecia as a medical student and when she becomes a physician. “I hope in the near future we will see an externally led FDA Patient-Focused Drug Development meeting for both CCCA and FFA, further advancing care and research for these conditions,” she said in an interview.
McMichael, Hordinsky, and Jefferson had no relevant disclosures to report.
A version of this article appeared on Medscape.com.
in the field of hair loss disorders.
“CCCA and FFA are conditions that require early diagnosis and intervention to prevent irreversible hair loss,” Maria Hordinsky, MD, professor of dermatology at the University of Minnesota, Minneapolis, and a member of the Board of Directors, Scarring Alopecia Foundation (SAF), said in an interview.
“The use of these new codes will make it easier for clinicians to identify affected patients and improve treatment outcomes. It also opens the door for more robust research efforts aimed at understanding the etiology and progression of CCCA and FFA, which could lead to new and more effective treatments in the future. Overall, this development represents a positive step toward improving care for individuals affected by these challenging conditions.”
The new codes — L66.81 for CCCA and L66.12 for FFA — were approved by the Centers for Disease Control and Prevention (CDC) on June 15, 2023, but not implemented until October 1, 2024.
Amy J. McMichael, MD, professor of dermatology at Wake Forest University School of Medicine, Winston-Salem, North Carolina, and a scientific advisor to SAF, told this news organization that Itisha Jefferson, a medical student at Loyola University Chicago’s Stritch School of Medicine, and her peers on the SAF’s Medical Student Executive Board, played a pivotal role in advocating for the codes.
In 2022, Jefferson, who has CCCA, and her fellow medical students helped create the proposals that were ultimately submitted to the CDC.
“They were critical in working with the CDC leaders to get the necessary information submitted and processed,” McMichael said. “They were also amazing at corralling our dermatologist group for the development of the necessary presentations and helped to shepherd us to the finish line for all logistic issues.”
On March 8, 2023, McMichael and Hordinsky made their pitch for the codes in person at the CDC’s ICD-10 Coordination and Maintenance Committee meeting, with McMichael discussing CCCA and Hordinsky discussing FFA.
“We also discussed the lack of standardized tracking, which has contributed to misdiagnoses and inadequate treatment options,” Hordinsky recalled. “We highlighted the importance of having distinct codes for these conditions to improve clinical outcomes, ensure that patients have access to appropriate care, better tracking of disease prevalence, and greater epidemiologic monitoring with access to electronic medical records and other large real-world evidence datasets and databases, the results of which could contribute to health policy decision-making.”
To spread the word about the new codes, McMichael, Hordinsky, and other members of the SAF are working with the original team of medical students, some of whom who are now dermatology residents, to develop an information guide to send to societies and organizations that were supportive of the codes. A publication in the dermatology literature is also planned.
For her part, Jefferson said that she will continue to advocate for patients with scarring alopecia as a medical student and when she becomes a physician. “I hope in the near future we will see an externally led FDA Patient-Focused Drug Development meeting for both CCCA and FFA, further advancing care and research for these conditions,” she said in an interview.
McMichael, Hordinsky, and Jefferson had no relevant disclosures to report.
A version of this article appeared on Medscape.com.
Community Outreach Benefits Dermatology Residents and Their Patients
The sun often is rising in the rearview mirror as I travel with the University of New Mexico dermatology team from Albuquerque to our satellite clinic in Gallup, New Mexico. This twice-monthly trip—with a group usually comprising an attending physician, residents, and medical students—provides an invaluable opportunity for me to take part in delivering care to a majority Native American population and connects our institution and its trainees to the state’s rural and indigenous cultures and communities.
Community outreach is an important initiative for many dermatology residency training programs. Engaging with the community outside the clinic setting allows residents to hone their clinical skills, interact with and meet new people, and help to improve access to health care, especially for members of underserved populations.
Limited access to health care remains a pressing issue in the United States, especially for underserved and rural communities. There currently is no standardized way to measure access to care, but multiple contributing factors have been identified, including but not limited to patient wait times and throughput, provider turnover, ratio of dermatologists to patient population, insurance type, and patient outcomes.1 Fortunately, there are many ways for dermatology residents to get involved and improve access to dermatologic services in their communities, including skin cancer screenings, free clinics, and teledermatology.
Skin Cancer Screenings
More than 40% of community outreach initiatives offered by dermatology residency programs are related to skin cancer screening and prevention.2 The American Academy of Dermatology’s free skin cancer check program (https://www.aad.org/member/career/volunteer/spot) offers a way to participate in or even host a skin cancer screening in your community. Since 1985, this program has identified nearly 300,000 suspicious lesions and more than 30,000 suspected melanomas. Resources for setting up a skin cancer screening in your community are available on the program’s website. Residents may take this opportunity to teach medical students how to perform full-body skin examinations and/or practice making independent decisions as the supervisor for medical trainees. Skin cancer screening events not only expand access to care in underserved communities but also help residents feel more connected to the local community, especially if they have moved to a new location for their residency training.
Free Clinics
Engaging in educational opportunities offered through residency programs is another way to participate in community outreach. In particular, many programs are affiliated with a School of Medicine within their institution that allows residents to spearhead volunteer opportunities such as working at a free clinic. In fact, more than 30% of initiatives offered at dermatology residency programs are free general dermatology clinics.2 Residents are in the unique position of being both learners themselves as well as educators to trainees.3 As part of our role, we can provide crucial specialty care to the community by working in concert with medical students and while also familiarizing ourselves with treating populations that we may not reach in our daily clinical work. For example, by participating in free clinics, we can provide care to vulnerable populations who typically may have financial or time barriers that prevent them from seeking care at the institution-associated clinic, including individuals experiencing homelessness, patients who are uninsured, and individuals who cannot take time off work to pursue medical care. Our presence in the community helps to reduce barriers to specialty care, particularly in the field of dermatology where the access shortage in the context of rising skin cancer rates prompts public health concerns.4
Teledermatology
Teledermatology became a way to extend our reach in the community more than ever before during the COVID-19 pandemic. Advances in audio, visual, and data telecommunication have been particularly helpful in dermatology, a specialty that relies heavily on visual cues for diagnosis. Synchronous, asynchronous, and hybrid teledermatology services implemented during the pandemic have gained favor among patients and dermatologists and are still applied in current practice.5,6
For example, in the state of New Mexico (where there is a severe shortage of board-certified dermatologists to care for the state’s population), teledermatology has allowed rural providers of all specialties to consult University of New Mexico dermatologists by sending clinical photographs along with patient information and history via secure messaging. Instead of having the patient travel hundreds of miles to see the nearest dermatologist for their skin condition or endure long wait times to get in to see a specialist, primary providers now can initiate treatment or work-up for their patient’s skin issue in a timely manner with the use of teledermatology to consult specialists.
Teledermatology has demonstrated cost-effectiveness, accuracy, and efficiency in conveniently expanding access to care. It offers patients and dermatologists flexibility in receiving and delivering health care, respectively.7 As residents, learning how to navigate this technologic frontier in health care delivery is imperative, as it will remain a prevalent tool in the future care of our communities, particularly in underserved areas.
Final Thoughts
Through community outreach initiatives, dermatology residents have an opportunity not only to enrich our education but also to connect with and become closer to our patients. Skin cancer screenings, free clinics, and teledermatology have provided ways to reach more communities and remain important aspects of dermatology residency.
- Patel B, Blalock TW. Defining “access to care” for dermatology at academic medical institutions. J Am Acad Dermatol. 2023;89:627-628. doi:10.1016/j.jaad.2023.03.014
- Fritsche M, Maglakelidze N, Zaenglein A, et al. Community outreach initiatives in dermatology: cross-sectional study. Arch Dermatol Res. 2023;315:2693-2695. doi:10.1007/s00403-023-02629-y
- Chiu LW. Teaching tips for dermatology residents. Cutis. 2024;113:E17-E19. doi:10.12788/cutis.1046
- Duniphin DD. Limited access to dermatology specialty care: barriers and teledermatology. Dermatol Pract Concept. 2023;13:E2023031. doi:10.5826/dpc.1301a31
- Ibrahim AE, Magdy M, Khalaf EM, et al. Teledermatology in the time of COVID-19. Int J Clin Pract. 2021;75:e15000. doi:10.1111/ijcp.15000
- Farr MA, Duvic M, Joshi TP. Teledermatology during COVID-19: an updated review. Am J Clin Dermatol. 2021;22:467-475. doi:10.1007/s40257-021-00601-y
- Lipner SR. Optimizing patient care with teledermatology: improving access, efficiency, and satisfaction. Cutis. 2024;114:63-64. doi:10.12788/cutis.1073
The sun often is rising in the rearview mirror as I travel with the University of New Mexico dermatology team from Albuquerque to our satellite clinic in Gallup, New Mexico. This twice-monthly trip—with a group usually comprising an attending physician, residents, and medical students—provides an invaluable opportunity for me to take part in delivering care to a majority Native American population and connects our institution and its trainees to the state’s rural and indigenous cultures and communities.
Community outreach is an important initiative for many dermatology residency training programs. Engaging with the community outside the clinic setting allows residents to hone their clinical skills, interact with and meet new people, and help to improve access to health care, especially for members of underserved populations.
Limited access to health care remains a pressing issue in the United States, especially for underserved and rural communities. There currently is no standardized way to measure access to care, but multiple contributing factors have been identified, including but not limited to patient wait times and throughput, provider turnover, ratio of dermatologists to patient population, insurance type, and patient outcomes.1 Fortunately, there are many ways for dermatology residents to get involved and improve access to dermatologic services in their communities, including skin cancer screenings, free clinics, and teledermatology.
Skin Cancer Screenings
More than 40% of community outreach initiatives offered by dermatology residency programs are related to skin cancer screening and prevention.2 The American Academy of Dermatology’s free skin cancer check program (https://www.aad.org/member/career/volunteer/spot) offers a way to participate in or even host a skin cancer screening in your community. Since 1985, this program has identified nearly 300,000 suspicious lesions and more than 30,000 suspected melanomas. Resources for setting up a skin cancer screening in your community are available on the program’s website. Residents may take this opportunity to teach medical students how to perform full-body skin examinations and/or practice making independent decisions as the supervisor for medical trainees. Skin cancer screening events not only expand access to care in underserved communities but also help residents feel more connected to the local community, especially if they have moved to a new location for their residency training.
Free Clinics
Engaging in educational opportunities offered through residency programs is another way to participate in community outreach. In particular, many programs are affiliated with a School of Medicine within their institution that allows residents to spearhead volunteer opportunities such as working at a free clinic. In fact, more than 30% of initiatives offered at dermatology residency programs are free general dermatology clinics.2 Residents are in the unique position of being both learners themselves as well as educators to trainees.3 As part of our role, we can provide crucial specialty care to the community by working in concert with medical students and while also familiarizing ourselves with treating populations that we may not reach in our daily clinical work. For example, by participating in free clinics, we can provide care to vulnerable populations who typically may have financial or time barriers that prevent them from seeking care at the institution-associated clinic, including individuals experiencing homelessness, patients who are uninsured, and individuals who cannot take time off work to pursue medical care. Our presence in the community helps to reduce barriers to specialty care, particularly in the field of dermatology where the access shortage in the context of rising skin cancer rates prompts public health concerns.4
Teledermatology
Teledermatology became a way to extend our reach in the community more than ever before during the COVID-19 pandemic. Advances in audio, visual, and data telecommunication have been particularly helpful in dermatology, a specialty that relies heavily on visual cues for diagnosis. Synchronous, asynchronous, and hybrid teledermatology services implemented during the pandemic have gained favor among patients and dermatologists and are still applied in current practice.5,6
For example, in the state of New Mexico (where there is a severe shortage of board-certified dermatologists to care for the state’s population), teledermatology has allowed rural providers of all specialties to consult University of New Mexico dermatologists by sending clinical photographs along with patient information and history via secure messaging. Instead of having the patient travel hundreds of miles to see the nearest dermatologist for their skin condition or endure long wait times to get in to see a specialist, primary providers now can initiate treatment or work-up for their patient’s skin issue in a timely manner with the use of teledermatology to consult specialists.
Teledermatology has demonstrated cost-effectiveness, accuracy, and efficiency in conveniently expanding access to care. It offers patients and dermatologists flexibility in receiving and delivering health care, respectively.7 As residents, learning how to navigate this technologic frontier in health care delivery is imperative, as it will remain a prevalent tool in the future care of our communities, particularly in underserved areas.
Final Thoughts
Through community outreach initiatives, dermatology residents have an opportunity not only to enrich our education but also to connect with and become closer to our patients. Skin cancer screenings, free clinics, and teledermatology have provided ways to reach more communities and remain important aspects of dermatology residency.
The sun often is rising in the rearview mirror as I travel with the University of New Mexico dermatology team from Albuquerque to our satellite clinic in Gallup, New Mexico. This twice-monthly trip—with a group usually comprising an attending physician, residents, and medical students—provides an invaluable opportunity for me to take part in delivering care to a majority Native American population and connects our institution and its trainees to the state’s rural and indigenous cultures and communities.
Community outreach is an important initiative for many dermatology residency training programs. Engaging with the community outside the clinic setting allows residents to hone their clinical skills, interact with and meet new people, and help to improve access to health care, especially for members of underserved populations.
Limited access to health care remains a pressing issue in the United States, especially for underserved and rural communities. There currently is no standardized way to measure access to care, but multiple contributing factors have been identified, including but not limited to patient wait times and throughput, provider turnover, ratio of dermatologists to patient population, insurance type, and patient outcomes.1 Fortunately, there are many ways for dermatology residents to get involved and improve access to dermatologic services in their communities, including skin cancer screenings, free clinics, and teledermatology.
Skin Cancer Screenings
More than 40% of community outreach initiatives offered by dermatology residency programs are related to skin cancer screening and prevention.2 The American Academy of Dermatology’s free skin cancer check program (https://www.aad.org/member/career/volunteer/spot) offers a way to participate in or even host a skin cancer screening in your community. Since 1985, this program has identified nearly 300,000 suspicious lesions and more than 30,000 suspected melanomas. Resources for setting up a skin cancer screening in your community are available on the program’s website. Residents may take this opportunity to teach medical students how to perform full-body skin examinations and/or practice making independent decisions as the supervisor for medical trainees. Skin cancer screening events not only expand access to care in underserved communities but also help residents feel more connected to the local community, especially if they have moved to a new location for their residency training.
Free Clinics
Engaging in educational opportunities offered through residency programs is another way to participate in community outreach. In particular, many programs are affiliated with a School of Medicine within their institution that allows residents to spearhead volunteer opportunities such as working at a free clinic. In fact, more than 30% of initiatives offered at dermatology residency programs are free general dermatology clinics.2 Residents are in the unique position of being both learners themselves as well as educators to trainees.3 As part of our role, we can provide crucial specialty care to the community by working in concert with medical students and while also familiarizing ourselves with treating populations that we may not reach in our daily clinical work. For example, by participating in free clinics, we can provide care to vulnerable populations who typically may have financial or time barriers that prevent them from seeking care at the institution-associated clinic, including individuals experiencing homelessness, patients who are uninsured, and individuals who cannot take time off work to pursue medical care. Our presence in the community helps to reduce barriers to specialty care, particularly in the field of dermatology where the access shortage in the context of rising skin cancer rates prompts public health concerns.4
Teledermatology
Teledermatology became a way to extend our reach in the community more than ever before during the COVID-19 pandemic. Advances in audio, visual, and data telecommunication have been particularly helpful in dermatology, a specialty that relies heavily on visual cues for diagnosis. Synchronous, asynchronous, and hybrid teledermatology services implemented during the pandemic have gained favor among patients and dermatologists and are still applied in current practice.5,6
For example, in the state of New Mexico (where there is a severe shortage of board-certified dermatologists to care for the state’s population), teledermatology has allowed rural providers of all specialties to consult University of New Mexico dermatologists by sending clinical photographs along with patient information and history via secure messaging. Instead of having the patient travel hundreds of miles to see the nearest dermatologist for their skin condition or endure long wait times to get in to see a specialist, primary providers now can initiate treatment or work-up for their patient’s skin issue in a timely manner with the use of teledermatology to consult specialists.
Teledermatology has demonstrated cost-effectiveness, accuracy, and efficiency in conveniently expanding access to care. It offers patients and dermatologists flexibility in receiving and delivering health care, respectively.7 As residents, learning how to navigate this technologic frontier in health care delivery is imperative, as it will remain a prevalent tool in the future care of our communities, particularly in underserved areas.
Final Thoughts
Through community outreach initiatives, dermatology residents have an opportunity not only to enrich our education but also to connect with and become closer to our patients. Skin cancer screenings, free clinics, and teledermatology have provided ways to reach more communities and remain important aspects of dermatology residency.
- Patel B, Blalock TW. Defining “access to care” for dermatology at academic medical institutions. J Am Acad Dermatol. 2023;89:627-628. doi:10.1016/j.jaad.2023.03.014
- Fritsche M, Maglakelidze N, Zaenglein A, et al. Community outreach initiatives in dermatology: cross-sectional study. Arch Dermatol Res. 2023;315:2693-2695. doi:10.1007/s00403-023-02629-y
- Chiu LW. Teaching tips for dermatology residents. Cutis. 2024;113:E17-E19. doi:10.12788/cutis.1046
- Duniphin DD. Limited access to dermatology specialty care: barriers and teledermatology. Dermatol Pract Concept. 2023;13:E2023031. doi:10.5826/dpc.1301a31
- Ibrahim AE, Magdy M, Khalaf EM, et al. Teledermatology in the time of COVID-19. Int J Clin Pract. 2021;75:e15000. doi:10.1111/ijcp.15000
- Farr MA, Duvic M, Joshi TP. Teledermatology during COVID-19: an updated review. Am J Clin Dermatol. 2021;22:467-475. doi:10.1007/s40257-021-00601-y
- Lipner SR. Optimizing patient care with teledermatology: improving access, efficiency, and satisfaction. Cutis. 2024;114:63-64. doi:10.12788/cutis.1073
- Patel B, Blalock TW. Defining “access to care” for dermatology at academic medical institutions. J Am Acad Dermatol. 2023;89:627-628. doi:10.1016/j.jaad.2023.03.014
- Fritsche M, Maglakelidze N, Zaenglein A, et al. Community outreach initiatives in dermatology: cross-sectional study. Arch Dermatol Res. 2023;315:2693-2695. doi:10.1007/s00403-023-02629-y
- Chiu LW. Teaching tips for dermatology residents. Cutis. 2024;113:E17-E19. doi:10.12788/cutis.1046
- Duniphin DD. Limited access to dermatology specialty care: barriers and teledermatology. Dermatol Pract Concept. 2023;13:E2023031. doi:10.5826/dpc.1301a31
- Ibrahim AE, Magdy M, Khalaf EM, et al. Teledermatology in the time of COVID-19. Int J Clin Pract. 2021;75:e15000. doi:10.1111/ijcp.15000
- Farr MA, Duvic M, Joshi TP. Teledermatology during COVID-19: an updated review. Am J Clin Dermatol. 2021;22:467-475. doi:10.1007/s40257-021-00601-y
- Lipner SR. Optimizing patient care with teledermatology: improving access, efficiency, and satisfaction. Cutis. 2024;114:63-64. doi:10.12788/cutis.1073
Resident Pearls
- Outreach initiatives can help residents feel more connected to their community and expand access to care.
- Skin cancer screenings, free clinics, and teledermatology are a few ways residents may get involved in their local communities.
Eruption of Multiple Linear Hyperpigmented Plaques
THE DIAGNOSIS: Chemotherapy-Induced Flagellate Dermatitis
Based on the clinical presentation and temporal relation with chemotherapy, a diagnosis of bleomycininduced flagellate dermatitis (FD) was made, as bleomycin is the only chemotherapeutic agent from this regimen that has been linked with FD.1,2 Laboratory findings revealed eosinophilia, further supporting a druginduced dermatitis. The patient was treated with oral steroids and diphenhydramine to alleviate itching and discomfort. The chemotherapy was temporarily discontinued until symptomatic improvement was observed within 2 to 3 days.
Flagellate dermatitis is characterized by unique erythematous, linear, intermingled streaks of adjoining firm papules—often preceded by a prodrome of global pruritus—that eventually become hyperpigmented as the erythema subsides. The clinical manifestation of FD can be idiopathic; true/mechanical (dermatitis artefacta, abuse, sadomasochism); chemotherapy induced (peplomycin, trastuzumab, cisplatin, docetaxel, bendamustine); toxin induced (shiitake mushroom, cnidarian stings, Paederus insects); related to rheumatologic diseases (dermatomyositis, adult-onset Still disease), dermatographism, phytophotodermatitis, or poison ivy dermatitis; or induced by chikungunya fever.1
The term flagellate originates from the Latin word flagellum, which pertains to the distinctive whiplike pattern. It was first described by Moulin et al3 in 1970 in reference to bleomycin-induced linear hyperpigmentation. Bleomycin, a glycopeptide antibiotic derived from Streptomyces verticillus, is used to treat Hodgkin lymphoma, squamous cell carcinoma, and germ cell tumors. The worldwide incidence of bleomycin-induced FD is 8% to 22% and commonly is associated with a cumulative dose greater than 100 U.2 Clinical presentation is variable in terms of onset, distribution, and morphology of the eruption and could be independent of dose, route of administration, or type of malignancy being treated. The flagellate rash commonly involves the trunk, arms, and legs; can develop within hours to 6 months of starting bleomycin therapy; often is preceded by generalized itching; and eventually heals with hyperpigmentation.
Possible mechanisms of bleomycin-induced FD include localized melanogenesis, inflammatory pigmentary incontinence, alterations to normal pigmentation patterns, cytotoxic effects of the drug itself, minor trauma/ scratching leading to increased blood flow and causing local accumulation of bleomycin, heat recall, and reduced epidermal turnover leading to extended interaction between keratinocytes and melanocytes.2 Heat exposure can act as a trigger for bleomycin-induced skin rash recall even months after the treatment is stopped.
Apart from discontinuing the drug, there is no specific treatment available for bleomycin-induced FD. The primary objective of treatment is to alleviate pruritus, which often involves the use of topical or systemic corticosteroids and oral antihistamines. The duration of treatment depends on the patient’s clinical response. Once treatment is discontinued, FD typically resolves within 6 to 8 months. However, there can be a permanent postinflammatory hyperpigmentation in the affected area.4 Although there is a concern for increased mortality after postponement of chemotherapy,5 the decision to proceed with or discontinue the chemotherapy regimen necessitates a comprehensive interdisciplinary discussion and a meticulous assessment of the risks and benefits that is customized to each individual patient. Flagellate dermatitis can reoccur with bleomycin re-exposure; a combined approach of proactive topical and systemic steroid treatment seems to diminish the likelihood of FD recurrence.5
Our case underscores the importance of recognizing, detecting, and managing FD promptly in individuals undergoing bleomycin-based chemotherapy. Medical professionals should familiarize themselves with this distinct adverse effect linked to bleomycin, enabling prompt discontinuation if necessary, and educate patients about the condition’s typically temporary nature, thereby alleviating their concerns.
- Bhushan P, Manjul P, Baliyan V. Flagellate dermatoses. Indian J Dermatol Venereol Leprol. 2014;80:149-152.
- Ziemer M, Goetze S, Juhasz K, et al. Flagellate dermatitis as a bleomycinspecific adverse effect of cytostatic therapy: a clinical-histopathologic correlation. Am J Clin Dermatol. 2011;12:68-76. doi:10.2165/11537080-000000000-00000
- Moulin G, Fière B, Beyvin A. Cutaneous pigmentation caused by bleomycin. Article in French. Bull Soc Fr Dermatol Syphiligr. 1970;77:293-296.
- Biswas A, Chaudhari PB, Sharma P, et al. Bleomycin induced flagellate erythema: revisiting a unique complication. J Cancer Res Ther. 2013;9:500-503. doi:10.4103/0973-1482.119358
- Hanna TP, King WD, Thibodeau S, et al. Mortality due to cancer treatment delay: systematic review and meta-analysis. BMJ. 2020;371:m4087. doi:10.1136/bmj.m4087
THE DIAGNOSIS: Chemotherapy-Induced Flagellate Dermatitis
Based on the clinical presentation and temporal relation with chemotherapy, a diagnosis of bleomycininduced flagellate dermatitis (FD) was made, as bleomycin is the only chemotherapeutic agent from this regimen that has been linked with FD.1,2 Laboratory findings revealed eosinophilia, further supporting a druginduced dermatitis. The patient was treated with oral steroids and diphenhydramine to alleviate itching and discomfort. The chemotherapy was temporarily discontinued until symptomatic improvement was observed within 2 to 3 days.
Flagellate dermatitis is characterized by unique erythematous, linear, intermingled streaks of adjoining firm papules—often preceded by a prodrome of global pruritus—that eventually become hyperpigmented as the erythema subsides. The clinical manifestation of FD can be idiopathic; true/mechanical (dermatitis artefacta, abuse, sadomasochism); chemotherapy induced (peplomycin, trastuzumab, cisplatin, docetaxel, bendamustine); toxin induced (shiitake mushroom, cnidarian stings, Paederus insects); related to rheumatologic diseases (dermatomyositis, adult-onset Still disease), dermatographism, phytophotodermatitis, or poison ivy dermatitis; or induced by chikungunya fever.1
The term flagellate originates from the Latin word flagellum, which pertains to the distinctive whiplike pattern. It was first described by Moulin et al3 in 1970 in reference to bleomycin-induced linear hyperpigmentation. Bleomycin, a glycopeptide antibiotic derived from Streptomyces verticillus, is used to treat Hodgkin lymphoma, squamous cell carcinoma, and germ cell tumors. The worldwide incidence of bleomycin-induced FD is 8% to 22% and commonly is associated with a cumulative dose greater than 100 U.2 Clinical presentation is variable in terms of onset, distribution, and morphology of the eruption and could be independent of dose, route of administration, or type of malignancy being treated. The flagellate rash commonly involves the trunk, arms, and legs; can develop within hours to 6 months of starting bleomycin therapy; often is preceded by generalized itching; and eventually heals with hyperpigmentation.
Possible mechanisms of bleomycin-induced FD include localized melanogenesis, inflammatory pigmentary incontinence, alterations to normal pigmentation patterns, cytotoxic effects of the drug itself, minor trauma/ scratching leading to increased blood flow and causing local accumulation of bleomycin, heat recall, and reduced epidermal turnover leading to extended interaction between keratinocytes and melanocytes.2 Heat exposure can act as a trigger for bleomycin-induced skin rash recall even months after the treatment is stopped.
Apart from discontinuing the drug, there is no specific treatment available for bleomycin-induced FD. The primary objective of treatment is to alleviate pruritus, which often involves the use of topical or systemic corticosteroids and oral antihistamines. The duration of treatment depends on the patient’s clinical response. Once treatment is discontinued, FD typically resolves within 6 to 8 months. However, there can be a permanent postinflammatory hyperpigmentation in the affected area.4 Although there is a concern for increased mortality after postponement of chemotherapy,5 the decision to proceed with or discontinue the chemotherapy regimen necessitates a comprehensive interdisciplinary discussion and a meticulous assessment of the risks and benefits that is customized to each individual patient. Flagellate dermatitis can reoccur with bleomycin re-exposure; a combined approach of proactive topical and systemic steroid treatment seems to diminish the likelihood of FD recurrence.5
Our case underscores the importance of recognizing, detecting, and managing FD promptly in individuals undergoing bleomycin-based chemotherapy. Medical professionals should familiarize themselves with this distinct adverse effect linked to bleomycin, enabling prompt discontinuation if necessary, and educate patients about the condition’s typically temporary nature, thereby alleviating their concerns.
THE DIAGNOSIS: Chemotherapy-Induced Flagellate Dermatitis
Based on the clinical presentation and temporal relation with chemotherapy, a diagnosis of bleomycininduced flagellate dermatitis (FD) was made, as bleomycin is the only chemotherapeutic agent from this regimen that has been linked with FD.1,2 Laboratory findings revealed eosinophilia, further supporting a druginduced dermatitis. The patient was treated with oral steroids and diphenhydramine to alleviate itching and discomfort. The chemotherapy was temporarily discontinued until symptomatic improvement was observed within 2 to 3 days.
Flagellate dermatitis is characterized by unique erythematous, linear, intermingled streaks of adjoining firm papules—often preceded by a prodrome of global pruritus—that eventually become hyperpigmented as the erythema subsides. The clinical manifestation of FD can be idiopathic; true/mechanical (dermatitis artefacta, abuse, sadomasochism); chemotherapy induced (peplomycin, trastuzumab, cisplatin, docetaxel, bendamustine); toxin induced (shiitake mushroom, cnidarian stings, Paederus insects); related to rheumatologic diseases (dermatomyositis, adult-onset Still disease), dermatographism, phytophotodermatitis, or poison ivy dermatitis; or induced by chikungunya fever.1
The term flagellate originates from the Latin word flagellum, which pertains to the distinctive whiplike pattern. It was first described by Moulin et al3 in 1970 in reference to bleomycin-induced linear hyperpigmentation. Bleomycin, a glycopeptide antibiotic derived from Streptomyces verticillus, is used to treat Hodgkin lymphoma, squamous cell carcinoma, and germ cell tumors. The worldwide incidence of bleomycin-induced FD is 8% to 22% and commonly is associated with a cumulative dose greater than 100 U.2 Clinical presentation is variable in terms of onset, distribution, and morphology of the eruption and could be independent of dose, route of administration, or type of malignancy being treated. The flagellate rash commonly involves the trunk, arms, and legs; can develop within hours to 6 months of starting bleomycin therapy; often is preceded by generalized itching; and eventually heals with hyperpigmentation.
Possible mechanisms of bleomycin-induced FD include localized melanogenesis, inflammatory pigmentary incontinence, alterations to normal pigmentation patterns, cytotoxic effects of the drug itself, minor trauma/ scratching leading to increased blood flow and causing local accumulation of bleomycin, heat recall, and reduced epidermal turnover leading to extended interaction between keratinocytes and melanocytes.2 Heat exposure can act as a trigger for bleomycin-induced skin rash recall even months after the treatment is stopped.
Apart from discontinuing the drug, there is no specific treatment available for bleomycin-induced FD. The primary objective of treatment is to alleviate pruritus, which often involves the use of topical or systemic corticosteroids and oral antihistamines. The duration of treatment depends on the patient’s clinical response. Once treatment is discontinued, FD typically resolves within 6 to 8 months. However, there can be a permanent postinflammatory hyperpigmentation in the affected area.4 Although there is a concern for increased mortality after postponement of chemotherapy,5 the decision to proceed with or discontinue the chemotherapy regimen necessitates a comprehensive interdisciplinary discussion and a meticulous assessment of the risks and benefits that is customized to each individual patient. Flagellate dermatitis can reoccur with bleomycin re-exposure; a combined approach of proactive topical and systemic steroid treatment seems to diminish the likelihood of FD recurrence.5
Our case underscores the importance of recognizing, detecting, and managing FD promptly in individuals undergoing bleomycin-based chemotherapy. Medical professionals should familiarize themselves with this distinct adverse effect linked to bleomycin, enabling prompt discontinuation if necessary, and educate patients about the condition’s typically temporary nature, thereby alleviating their concerns.
- Bhushan P, Manjul P, Baliyan V. Flagellate dermatoses. Indian J Dermatol Venereol Leprol. 2014;80:149-152.
- Ziemer M, Goetze S, Juhasz K, et al. Flagellate dermatitis as a bleomycinspecific adverse effect of cytostatic therapy: a clinical-histopathologic correlation. Am J Clin Dermatol. 2011;12:68-76. doi:10.2165/11537080-000000000-00000
- Moulin G, Fière B, Beyvin A. Cutaneous pigmentation caused by bleomycin. Article in French. Bull Soc Fr Dermatol Syphiligr. 1970;77:293-296.
- Biswas A, Chaudhari PB, Sharma P, et al. Bleomycin induced flagellate erythema: revisiting a unique complication. J Cancer Res Ther. 2013;9:500-503. doi:10.4103/0973-1482.119358
- Hanna TP, King WD, Thibodeau S, et al. Mortality due to cancer treatment delay: systematic review and meta-analysis. BMJ. 2020;371:m4087. doi:10.1136/bmj.m4087
- Bhushan P, Manjul P, Baliyan V. Flagellate dermatoses. Indian J Dermatol Venereol Leprol. 2014;80:149-152.
- Ziemer M, Goetze S, Juhasz K, et al. Flagellate dermatitis as a bleomycinspecific adverse effect of cytostatic therapy: a clinical-histopathologic correlation. Am J Clin Dermatol. 2011;12:68-76. doi:10.2165/11537080-000000000-00000
- Moulin G, Fière B, Beyvin A. Cutaneous pigmentation caused by bleomycin. Article in French. Bull Soc Fr Dermatol Syphiligr. 1970;77:293-296.
- Biswas A, Chaudhari PB, Sharma P, et al. Bleomycin induced flagellate erythema: revisiting a unique complication. J Cancer Res Ther. 2013;9:500-503. doi:10.4103/0973-1482.119358
- Hanna TP, King WD, Thibodeau S, et al. Mortality due to cancer treatment delay: systematic review and meta-analysis. BMJ. 2020;371:m4087. doi:10.1136/bmj.m4087
A 28-year-old man presented for evaluation of an intensely itchy rash of 5 days’ duration involving the face, trunk, arms, and legs. The patient recently had been diagnosed with classical Hodgkin lymphoma and was started on a biweekly chemotherapy regimen of adriamycin, bleomycin, vinblastine, and dacarbazine 3 weeks prior. He reported that a red, itchy, papular rash had developed on the hands 1 week after starting chemotherapy and improved with antihistamines. Symptoms of the current rash included night sweats, occasional fever, substantial unintentional weight loss, and fatigue. He had no history of urticaria, angioedema, anaphylaxis, or nail changes.
Physical examination revealed widespread, itchy, linear and curvilinear hyperpigmented plaques on the upper arms, shoulders, back (top), face, and thighs, as well as erythematous grouped papules on the bilateral palms (bottom). There was no mucosal or systemic involvement.
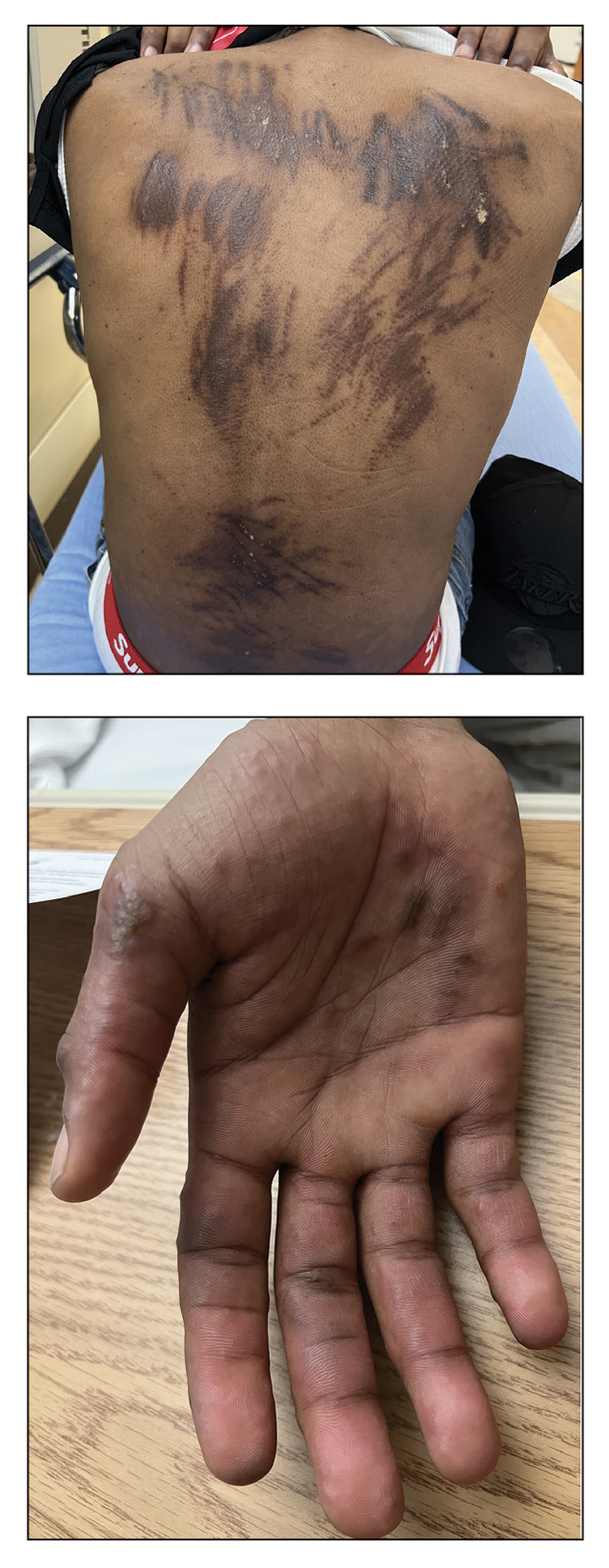
Six Tips for Media Interviews
As a physician, you might be contacted by the media to provide your professional opinion and advice. Or you might be looking for media interview opportunities to market your practice or side project. And if you do research, media interviews can be an effective way to spread the word. It’s important to prepare for a media interview so that you achieve the outcome you are looking for.
Keep your message simple. When you are a subject expert, you might think that the basics are obvious or even boring, and that the nuances are more important. However, most of the audience is looking for big-picture information that they can apply to their lives. Consider a few key takeaways, keeping in mind that your interview is likely to be edited to short sound bites or a few quotes. It may help to jot down notes so that you cover the fundamentals clearly. You could even write and rehearse a script beforehand. If there is something complicated or subtle that you want to convey, you can preface it by saying, “This is confusing but very important …” to let the audience know to give extra consideration to what you are about to say.
Avoid extremes and hyperbole. Sometimes, exaggerated statements make their way into medical discussions. Statements such as “it doesn’t matter how many calories you consume — it’s all about the quality” are common oversimplifications. But you might be upset to see your name next to a comment like this because it is not actually correct. Check the phrasing of your key takeaways to avoid being stuck defending or explaining an inaccurate statement when your patients ask you about it later.
Ask the interviewers what they are looking for. Many medical topics have some controversial element, so it is good to know what you’re getting into. Find out the purpose of the article or interview before you decide whether it is right for you. It could be about another doctor in town who is being sued; if you don’t want to be associated with that story, it might be best to decline the interview.
Explain your goals. You might accept or pursue an interview to raise awareness about an underrecognized condition. You might want the public to identify and get help for early symptoms, or you might want to create empathy for people coping with a disease you treat. Consider why you are participating in an interview, and communicate that to the interviewer to ensure that your objective can be part of the final product.
Know whom you’re dealing with. It is good to learn about the publication/media channel before you agree to participate. It may have a political bias, or perhaps the interview is intended to promote a specific product. If you agree with and support their purposes, then you may be happy to lend your opinion. But learning about the “voice” of the publication in advance allows you to make an informed decision about whether you want to be identified with a particular political ideology or product endorsement.
Ask to see your quotes before publication. It’s good to have the opportunity to make corrections in case you are accidentally misquoted or misunderstood. It is best to ask to see quotes before you agree to the interview. Some reporters may agree to (or even prefer) a written question-and-answer format so that they can directly quote your responses without rephrasing your words. You could suggest this, especially if you are too busy for a call or live meeting.
As a physician, your insights and advice can be highly beneficial to others. You can also use media interviews to propel your career forward. Doing your homework can ensure that you will be pleased with the final product and how your words were used.
Dr. Moawad, Clinical Assistant Professor, Department of Medical Education, Case Western Reserve University School of Medicine, Cleveland, Ohio, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
As a physician, you might be contacted by the media to provide your professional opinion and advice. Or you might be looking for media interview opportunities to market your practice or side project. And if you do research, media interviews can be an effective way to spread the word. It’s important to prepare for a media interview so that you achieve the outcome you are looking for.
Keep your message simple. When you are a subject expert, you might think that the basics are obvious or even boring, and that the nuances are more important. However, most of the audience is looking for big-picture information that they can apply to their lives. Consider a few key takeaways, keeping in mind that your interview is likely to be edited to short sound bites or a few quotes. It may help to jot down notes so that you cover the fundamentals clearly. You could even write and rehearse a script beforehand. If there is something complicated or subtle that you want to convey, you can preface it by saying, “This is confusing but very important …” to let the audience know to give extra consideration to what you are about to say.
Avoid extremes and hyperbole. Sometimes, exaggerated statements make their way into medical discussions. Statements such as “it doesn’t matter how many calories you consume — it’s all about the quality” are common oversimplifications. But you might be upset to see your name next to a comment like this because it is not actually correct. Check the phrasing of your key takeaways to avoid being stuck defending or explaining an inaccurate statement when your patients ask you about it later.
Ask the interviewers what they are looking for. Many medical topics have some controversial element, so it is good to know what you’re getting into. Find out the purpose of the article or interview before you decide whether it is right for you. It could be about another doctor in town who is being sued; if you don’t want to be associated with that story, it might be best to decline the interview.
Explain your goals. You might accept or pursue an interview to raise awareness about an underrecognized condition. You might want the public to identify and get help for early symptoms, or you might want to create empathy for people coping with a disease you treat. Consider why you are participating in an interview, and communicate that to the interviewer to ensure that your objective can be part of the final product.
Know whom you’re dealing with. It is good to learn about the publication/media channel before you agree to participate. It may have a political bias, or perhaps the interview is intended to promote a specific product. If you agree with and support their purposes, then you may be happy to lend your opinion. But learning about the “voice” of the publication in advance allows you to make an informed decision about whether you want to be identified with a particular political ideology or product endorsement.
Ask to see your quotes before publication. It’s good to have the opportunity to make corrections in case you are accidentally misquoted or misunderstood. It is best to ask to see quotes before you agree to the interview. Some reporters may agree to (or even prefer) a written question-and-answer format so that they can directly quote your responses without rephrasing your words. You could suggest this, especially if you are too busy for a call or live meeting.
As a physician, your insights and advice can be highly beneficial to others. You can also use media interviews to propel your career forward. Doing your homework can ensure that you will be pleased with the final product and how your words were used.
Dr. Moawad, Clinical Assistant Professor, Department of Medical Education, Case Western Reserve University School of Medicine, Cleveland, Ohio, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
As a physician, you might be contacted by the media to provide your professional opinion and advice. Or you might be looking for media interview opportunities to market your practice or side project. And if you do research, media interviews can be an effective way to spread the word. It’s important to prepare for a media interview so that you achieve the outcome you are looking for.
Keep your message simple. When you are a subject expert, you might think that the basics are obvious or even boring, and that the nuances are more important. However, most of the audience is looking for big-picture information that they can apply to their lives. Consider a few key takeaways, keeping in mind that your interview is likely to be edited to short sound bites or a few quotes. It may help to jot down notes so that you cover the fundamentals clearly. You could even write and rehearse a script beforehand. If there is something complicated or subtle that you want to convey, you can preface it by saying, “This is confusing but very important …” to let the audience know to give extra consideration to what you are about to say.
Avoid extremes and hyperbole. Sometimes, exaggerated statements make their way into medical discussions. Statements such as “it doesn’t matter how many calories you consume — it’s all about the quality” are common oversimplifications. But you might be upset to see your name next to a comment like this because it is not actually correct. Check the phrasing of your key takeaways to avoid being stuck defending or explaining an inaccurate statement when your patients ask you about it later.
Ask the interviewers what they are looking for. Many medical topics have some controversial element, so it is good to know what you’re getting into. Find out the purpose of the article or interview before you decide whether it is right for you. It could be about another doctor in town who is being sued; if you don’t want to be associated with that story, it might be best to decline the interview.
Explain your goals. You might accept or pursue an interview to raise awareness about an underrecognized condition. You might want the public to identify and get help for early symptoms, or you might want to create empathy for people coping with a disease you treat. Consider why you are participating in an interview, and communicate that to the interviewer to ensure that your objective can be part of the final product.
Know whom you’re dealing with. It is good to learn about the publication/media channel before you agree to participate. It may have a political bias, or perhaps the interview is intended to promote a specific product. If you agree with and support their purposes, then you may be happy to lend your opinion. But learning about the “voice” of the publication in advance allows you to make an informed decision about whether you want to be identified with a particular political ideology or product endorsement.
Ask to see your quotes before publication. It’s good to have the opportunity to make corrections in case you are accidentally misquoted or misunderstood. It is best to ask to see quotes before you agree to the interview. Some reporters may agree to (or even prefer) a written question-and-answer format so that they can directly quote your responses without rephrasing your words. You could suggest this, especially if you are too busy for a call or live meeting.
As a physician, your insights and advice can be highly beneficial to others. You can also use media interviews to propel your career forward. Doing your homework can ensure that you will be pleased with the final product and how your words were used.
Dr. Moawad, Clinical Assistant Professor, Department of Medical Education, Case Western Reserve University School of Medicine, Cleveland, Ohio, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.