User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Patient Navigators for Serious Illnesses Can Now Bill Under New Medicare Codes
In a move that acknowledges the gauntlet the US health system poses for people facing serious and fatal illnesses, Medicare will pay for a new class of workers to help patients manage treatments for conditions like cancer and heart failure.
The 2024 Medicare physician fee schedule includes new billing codes, including G0023, to pay for 60 minutes a month of care coordination by certified or trained auxiliary personnel working under the direction of a clinician.
A diagnosis of cancer or another serious illness takes a toll beyond the physical effects of the disease. Patients often scramble to make adjustments in family and work schedules to manage treatment, said Samyukta Mullangi, MD, MBA, medical director of oncology at Thyme Care, a Nashville, Tennessee–based firm that provides navigation and coordination services to oncology practices and insurers.
“It just really does create a bit of a pressure cooker for patients,” Dr. Mullangi told this news organization.
Medicare has for many years paid for medical professionals to help patients cope with the complexities of disease, such as chronic care management (CCM) provided by physicians, nurses, and physician assistants.
The new principal illness navigation (PIN) payments are intended to pay for work that to date typically has been done by people without medical degrees, including those involved in peer support networks and community health programs. The US Centers for Medicare and Medicaid Services(CMS) expects these navigators will undergo training and work under the supervision of clinicians.
The new navigators may coordinate care transitions between medical settings, follow up with patients after emergency department (ED) visits, or communicate with skilled nursing facilities regarding the psychosocial needs and functional deficits of a patient, among other functions.
CMS expects the new navigators may:
- Conduct assessments to understand a patient’s life story, strengths, needs, goals, preferences, and desired outcomes, including understanding cultural and linguistic factors.
- Provide support to accomplish the clinician’s treatment plan.
- Coordinate the receipt of needed services from healthcare facilities, home- and community-based service providers, and caregivers.
Peers as Navigators
The new navigators can be former patients who have undergone similar treatments for serious diseases, CMS said. This approach sets the new program apart from other care management services Medicare already covers, program officials wrote in the 2024 physician fee schedule.
“For some conditions, patients are best able to engage with the healthcare system and access care if they have assistance from a single, dedicated individual who has ‘lived experience,’ ” according to the rule.
The agency has taken a broad initial approach in defining what kinds of illnesses a patient may have to qualify for services. Patients must have a serious condition that is expected to last at least 3 months, such as cancer, heart failure, or substance use disorder.
But those without a definitive diagnosis may also qualify to receive navigator services.
In the rule, CMS cited a case in which a CT scan identified a suspicious mass in a patient’s colon. A clinician might decide this person would benefit from navigation services due to the potential risks for an undiagnosed illness.
“Regardless of the definitive diagnosis of the mass, presence of a colonic mass for that patient may be a serious high-risk condition that could, for example, cause obstruction and lead the patient to present to the emergency department, as well as be potentially indicative of an underlying life-threatening illness such as colon cancer,” CMS wrote in the rule.
Navigators often start their work when cancer patients are screened and guide them through initial diagnosis, potential surgery, radiation, or chemotherapy, said Sharon Gentry, MSN, RN, a former nurse navigator who is now the editor in chief of the Journal of the Academy of Oncology Nurse & Patient Navigators.
The navigators are meant to be a trusted and continual presence for patients, who otherwise might be left to start anew in finding help at each phase of care.
The navigators “see the whole picture. They see the whole journey the patient takes, from pre-diagnosis all the way through diagnosis care out through survival,” Ms. Gentry said.
Gaining a special Medicare payment for these kinds of services will elevate this work, she said.
Many newer drugs can target specific mechanisms and proteins of cancer. Often, oncology treatment involves testing to find out if mutations are allowing the cancer cells to evade a patient’s immune system.
Checking these biomarkers takes time, however. Patients sometimes become frustrated because they are anxious to begin treatment. Patients may receive inaccurate information from friends or family who went through treatment previously. Navigators can provide knowledge on the current state of care for a patient’s disease, helping them better manage anxieties.
“You have to explain to them that things have changed since the guy you drink coffee with was diagnosed with cancer, and there may be a drug that could target that,” Ms. Gentry said.
Potential Challenges
Initial uptake of the new PIN codes may be slow going, however, as clinicians and health systems may already use well-established codes. These include CCM and principal care management services, which may pay higher rates, Mullangi said.
“There might be sensitivity around not wanting to cannibalize existing programs with a new program,” Dr. Mullangi said.
In addition, many patients will have a copay for the services of principal illness navigators, Dr. Mullangi said.
While many patients have additional insurance that would cover the service, not all do. People with traditional Medicare coverage can sometimes pay 20% of the cost of some medical services.
“I think that may give patients pause, particularly if they’re already feeling the financial burden of a cancer treatment journey,” Dr. Mullangi said.
Pay rates for PIN services involve calculations of regional price differences, which are posted publicly by CMS, and potential added fees for services provided by hospital-affiliated organizations.
Consider payments for code G0023, covering 60 minutes of principal navigation services provided in a single month.
A set reimbursement for patients cared for in independent medical practices exists, with variation for local costs. Medicare’s non-facility price for G0023 would be $102.41 in some parts of Silicon Valley in California, including San Jose. In Arkansas, where costs are lower, reimbursement would be $73.14 for this same service.
Patients who get services covered by code G0023 in independent medical practices would have monthly copays of about $15-$20, depending on where they live.
The tab for patients tends to be higher for these same services if delivered through a medical practice owned by a hospital, as this would trigger the addition of facility fees to the payments made to cover the services. Facility fees are difficult for the public to ascertain before getting a treatment or service.
Dr. Mullangi and Ms. Gentry reported no relevant financial disclosures outside of their employers.
A version of this article first appeared on Medscape.com.
In a move that acknowledges the gauntlet the US health system poses for people facing serious and fatal illnesses, Medicare will pay for a new class of workers to help patients manage treatments for conditions like cancer and heart failure.
The 2024 Medicare physician fee schedule includes new billing codes, including G0023, to pay for 60 minutes a month of care coordination by certified or trained auxiliary personnel working under the direction of a clinician.
A diagnosis of cancer or another serious illness takes a toll beyond the physical effects of the disease. Patients often scramble to make adjustments in family and work schedules to manage treatment, said Samyukta Mullangi, MD, MBA, medical director of oncology at Thyme Care, a Nashville, Tennessee–based firm that provides navigation and coordination services to oncology practices and insurers.
“It just really does create a bit of a pressure cooker for patients,” Dr. Mullangi told this news organization.
Medicare has for many years paid for medical professionals to help patients cope with the complexities of disease, such as chronic care management (CCM) provided by physicians, nurses, and physician assistants.
The new principal illness navigation (PIN) payments are intended to pay for work that to date typically has been done by people without medical degrees, including those involved in peer support networks and community health programs. The US Centers for Medicare and Medicaid Services(CMS) expects these navigators will undergo training and work under the supervision of clinicians.
The new navigators may coordinate care transitions between medical settings, follow up with patients after emergency department (ED) visits, or communicate with skilled nursing facilities regarding the psychosocial needs and functional deficits of a patient, among other functions.
CMS expects the new navigators may:
- Conduct assessments to understand a patient’s life story, strengths, needs, goals, preferences, and desired outcomes, including understanding cultural and linguistic factors.
- Provide support to accomplish the clinician’s treatment plan.
- Coordinate the receipt of needed services from healthcare facilities, home- and community-based service providers, and caregivers.
Peers as Navigators
The new navigators can be former patients who have undergone similar treatments for serious diseases, CMS said. This approach sets the new program apart from other care management services Medicare already covers, program officials wrote in the 2024 physician fee schedule.
“For some conditions, patients are best able to engage with the healthcare system and access care if they have assistance from a single, dedicated individual who has ‘lived experience,’ ” according to the rule.
The agency has taken a broad initial approach in defining what kinds of illnesses a patient may have to qualify for services. Patients must have a serious condition that is expected to last at least 3 months, such as cancer, heart failure, or substance use disorder.
But those without a definitive diagnosis may also qualify to receive navigator services.
In the rule, CMS cited a case in which a CT scan identified a suspicious mass in a patient’s colon. A clinician might decide this person would benefit from navigation services due to the potential risks for an undiagnosed illness.
“Regardless of the definitive diagnosis of the mass, presence of a colonic mass for that patient may be a serious high-risk condition that could, for example, cause obstruction and lead the patient to present to the emergency department, as well as be potentially indicative of an underlying life-threatening illness such as colon cancer,” CMS wrote in the rule.
Navigators often start their work when cancer patients are screened and guide them through initial diagnosis, potential surgery, radiation, or chemotherapy, said Sharon Gentry, MSN, RN, a former nurse navigator who is now the editor in chief of the Journal of the Academy of Oncology Nurse & Patient Navigators.
The navigators are meant to be a trusted and continual presence for patients, who otherwise might be left to start anew in finding help at each phase of care.
The navigators “see the whole picture. They see the whole journey the patient takes, from pre-diagnosis all the way through diagnosis care out through survival,” Ms. Gentry said.
Gaining a special Medicare payment for these kinds of services will elevate this work, she said.
Many newer drugs can target specific mechanisms and proteins of cancer. Often, oncology treatment involves testing to find out if mutations are allowing the cancer cells to evade a patient’s immune system.
Checking these biomarkers takes time, however. Patients sometimes become frustrated because they are anxious to begin treatment. Patients may receive inaccurate information from friends or family who went through treatment previously. Navigators can provide knowledge on the current state of care for a patient’s disease, helping them better manage anxieties.
“You have to explain to them that things have changed since the guy you drink coffee with was diagnosed with cancer, and there may be a drug that could target that,” Ms. Gentry said.
Potential Challenges
Initial uptake of the new PIN codes may be slow going, however, as clinicians and health systems may already use well-established codes. These include CCM and principal care management services, which may pay higher rates, Mullangi said.
“There might be sensitivity around not wanting to cannibalize existing programs with a new program,” Dr. Mullangi said.
In addition, many patients will have a copay for the services of principal illness navigators, Dr. Mullangi said.
While many patients have additional insurance that would cover the service, not all do. People with traditional Medicare coverage can sometimes pay 20% of the cost of some medical services.
“I think that may give patients pause, particularly if they’re already feeling the financial burden of a cancer treatment journey,” Dr. Mullangi said.
Pay rates for PIN services involve calculations of regional price differences, which are posted publicly by CMS, and potential added fees for services provided by hospital-affiliated organizations.
Consider payments for code G0023, covering 60 minutes of principal navigation services provided in a single month.
A set reimbursement for patients cared for in independent medical practices exists, with variation for local costs. Medicare’s non-facility price for G0023 would be $102.41 in some parts of Silicon Valley in California, including San Jose. In Arkansas, where costs are lower, reimbursement would be $73.14 for this same service.
Patients who get services covered by code G0023 in independent medical practices would have monthly copays of about $15-$20, depending on where they live.
The tab for patients tends to be higher for these same services if delivered through a medical practice owned by a hospital, as this would trigger the addition of facility fees to the payments made to cover the services. Facility fees are difficult for the public to ascertain before getting a treatment or service.
Dr. Mullangi and Ms. Gentry reported no relevant financial disclosures outside of their employers.
A version of this article first appeared on Medscape.com.
In a move that acknowledges the gauntlet the US health system poses for people facing serious and fatal illnesses, Medicare will pay for a new class of workers to help patients manage treatments for conditions like cancer and heart failure.
The 2024 Medicare physician fee schedule includes new billing codes, including G0023, to pay for 60 minutes a month of care coordination by certified or trained auxiliary personnel working under the direction of a clinician.
A diagnosis of cancer or another serious illness takes a toll beyond the physical effects of the disease. Patients often scramble to make adjustments in family and work schedules to manage treatment, said Samyukta Mullangi, MD, MBA, medical director of oncology at Thyme Care, a Nashville, Tennessee–based firm that provides navigation and coordination services to oncology practices and insurers.
“It just really does create a bit of a pressure cooker for patients,” Dr. Mullangi told this news organization.
Medicare has for many years paid for medical professionals to help patients cope with the complexities of disease, such as chronic care management (CCM) provided by physicians, nurses, and physician assistants.
The new principal illness navigation (PIN) payments are intended to pay for work that to date typically has been done by people without medical degrees, including those involved in peer support networks and community health programs. The US Centers for Medicare and Medicaid Services(CMS) expects these navigators will undergo training and work under the supervision of clinicians.
The new navigators may coordinate care transitions between medical settings, follow up with patients after emergency department (ED) visits, or communicate with skilled nursing facilities regarding the psychosocial needs and functional deficits of a patient, among other functions.
CMS expects the new navigators may:
- Conduct assessments to understand a patient’s life story, strengths, needs, goals, preferences, and desired outcomes, including understanding cultural and linguistic factors.
- Provide support to accomplish the clinician’s treatment plan.
- Coordinate the receipt of needed services from healthcare facilities, home- and community-based service providers, and caregivers.
Peers as Navigators
The new navigators can be former patients who have undergone similar treatments for serious diseases, CMS said. This approach sets the new program apart from other care management services Medicare already covers, program officials wrote in the 2024 physician fee schedule.
“For some conditions, patients are best able to engage with the healthcare system and access care if they have assistance from a single, dedicated individual who has ‘lived experience,’ ” according to the rule.
The agency has taken a broad initial approach in defining what kinds of illnesses a patient may have to qualify for services. Patients must have a serious condition that is expected to last at least 3 months, such as cancer, heart failure, or substance use disorder.
But those without a definitive diagnosis may also qualify to receive navigator services.
In the rule, CMS cited a case in which a CT scan identified a suspicious mass in a patient’s colon. A clinician might decide this person would benefit from navigation services due to the potential risks for an undiagnosed illness.
“Regardless of the definitive diagnosis of the mass, presence of a colonic mass for that patient may be a serious high-risk condition that could, for example, cause obstruction and lead the patient to present to the emergency department, as well as be potentially indicative of an underlying life-threatening illness such as colon cancer,” CMS wrote in the rule.
Navigators often start their work when cancer patients are screened and guide them through initial diagnosis, potential surgery, radiation, or chemotherapy, said Sharon Gentry, MSN, RN, a former nurse navigator who is now the editor in chief of the Journal of the Academy of Oncology Nurse & Patient Navigators.
The navigators are meant to be a trusted and continual presence for patients, who otherwise might be left to start anew in finding help at each phase of care.
The navigators “see the whole picture. They see the whole journey the patient takes, from pre-diagnosis all the way through diagnosis care out through survival,” Ms. Gentry said.
Gaining a special Medicare payment for these kinds of services will elevate this work, she said.
Many newer drugs can target specific mechanisms and proteins of cancer. Often, oncology treatment involves testing to find out if mutations are allowing the cancer cells to evade a patient’s immune system.
Checking these biomarkers takes time, however. Patients sometimes become frustrated because they are anxious to begin treatment. Patients may receive inaccurate information from friends or family who went through treatment previously. Navigators can provide knowledge on the current state of care for a patient’s disease, helping them better manage anxieties.
“You have to explain to them that things have changed since the guy you drink coffee with was diagnosed with cancer, and there may be a drug that could target that,” Ms. Gentry said.
Potential Challenges
Initial uptake of the new PIN codes may be slow going, however, as clinicians and health systems may already use well-established codes. These include CCM and principal care management services, which may pay higher rates, Mullangi said.
“There might be sensitivity around not wanting to cannibalize existing programs with a new program,” Dr. Mullangi said.
In addition, many patients will have a copay for the services of principal illness navigators, Dr. Mullangi said.
While many patients have additional insurance that would cover the service, not all do. People with traditional Medicare coverage can sometimes pay 20% of the cost of some medical services.
“I think that may give patients pause, particularly if they’re already feeling the financial burden of a cancer treatment journey,” Dr. Mullangi said.
Pay rates for PIN services involve calculations of regional price differences, which are posted publicly by CMS, and potential added fees for services provided by hospital-affiliated organizations.
Consider payments for code G0023, covering 60 minutes of principal navigation services provided in a single month.
A set reimbursement for patients cared for in independent medical practices exists, with variation for local costs. Medicare’s non-facility price for G0023 would be $102.41 in some parts of Silicon Valley in California, including San Jose. In Arkansas, where costs are lower, reimbursement would be $73.14 for this same service.
Patients who get services covered by code G0023 in independent medical practices would have monthly copays of about $15-$20, depending on where they live.
The tab for patients tends to be higher for these same services if delivered through a medical practice owned by a hospital, as this would trigger the addition of facility fees to the payments made to cover the services. Facility fees are difficult for the public to ascertain before getting a treatment or service.
Dr. Mullangi and Ms. Gentry reported no relevant financial disclosures outside of their employers.
A version of this article first appeared on Medscape.com.

RA Prevention: A Decade of Trials Provides Insights on What’s to Come
With the discovery of autoantibodies and other risk factors for rheumatoid arthritis (RA), researchers developed clinical trials to see whether the disease can be prevented entirely. In the past 10 years, a number of these trials have concluded, with variable results.
While some trials demonstrated no effect at all, others showed that medical intervention can delay the onset of disease in certain populations and even reduce the rates of progression to RA. These completed trials also offer researchers the chance to identify opportunities to improve RA prevention trials moving forward.
“We’re looking at all that data and trying to figure out what the next step is going to be,” said Kevin Deane, MD, PhD, a professor of medicine and a rheumatologist at the University of Colorado School of Medicine, Aurora.
Key lessons include the need for improved risk stratification tools and better understanding of RA pathogenesis, he said.
The Research So Far
All RA prevention trials except for one have been completed and/or published within the past decade, bringing valuable insights to the field. (See chart below.)
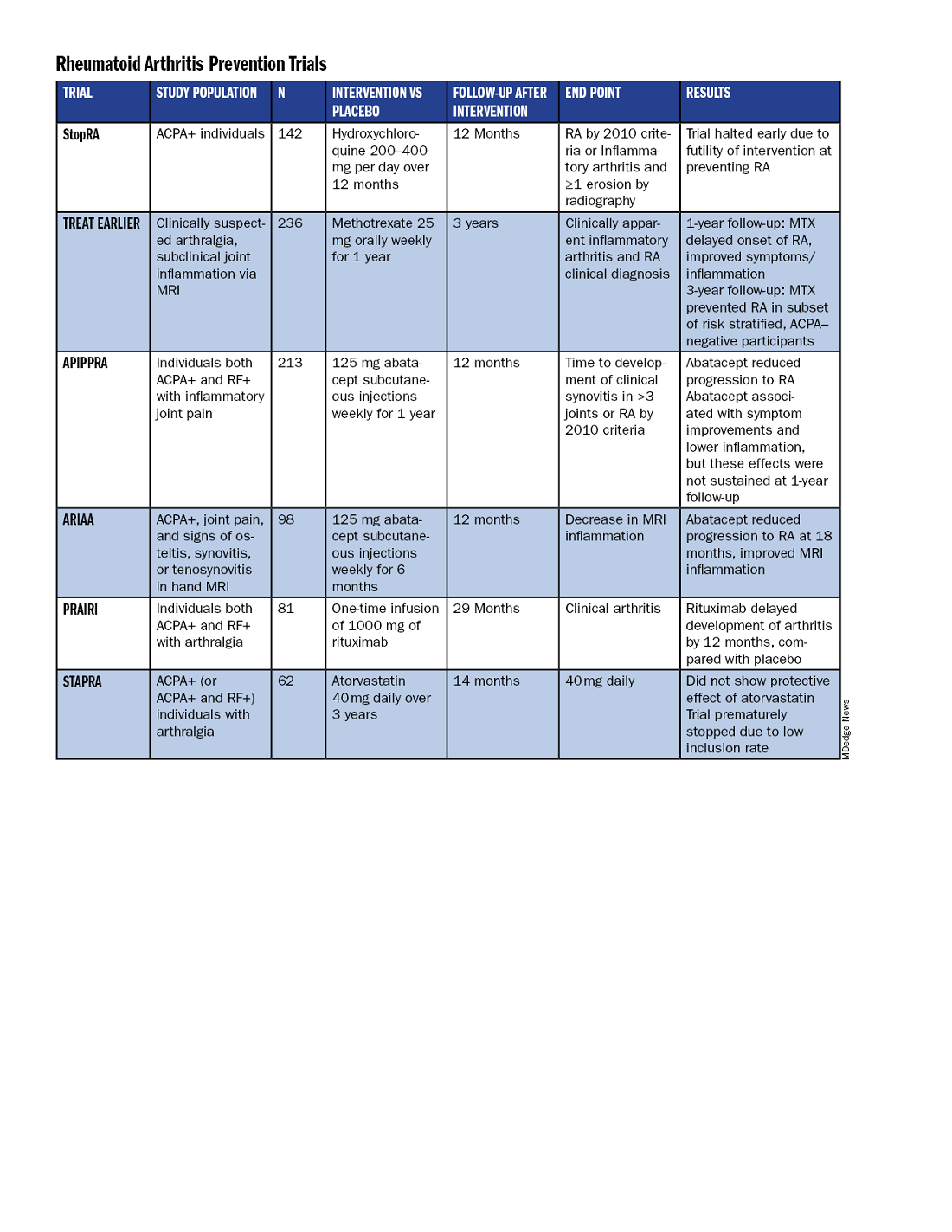
Atorvastatin (STAPRA) and hydroxychloroquine (StopRA) proved ineffective in preventing the onset of RA, and both trials were stopped early. Rituximab and methotrexate (MTX) both delayed the onset of RA, but the effect disappeared by the end of the follow-up periods.
However, the 2-year results from the TREAT EARLIER trial showed that compared with patients given placebo, those given MTX showed improved MRI-detected joint inflammation, physical functioning, and reported symptoms.
The 4-year analysis of the trial further risk stratified participants and found that MTX showed a preventive effect in anti–citrullinated protein antibody (ACPA)–negative participants at an increased risk for RA.
Abatacept also showed promise in preventing RA in two separate trials. In the ARIAA trial, compared with placebo, 6 months of treatment with abatacept reduced MRI inflammation and symptoms and lowered the rates of progression to RA. This treatment effect lessened during the 1-year follow-up period, but the difference between the two groups was still significant at 18 months.
In the APIPPRA trial, 12 months of treatment with abatacept improved subclinical inflammation and quality-of-life measures in participants and reduced the rates of progression to RA through another 12 months of observation. However, during this post-treatment follow-up period, the treatment effect began to diminish.
While there have been some promising findings — not only in disease prevention but also in disease modification — these studies all looked at different patient groups, noted Kulveer Mankia, MA, DM, an associate professor and consulting rheumatologist at the Leeds Institute of Rheumatic and Musculoskeletal Medicine, University of Leeds in England.
“You have disparate, different inclusion criteria in different studies, all of which take years to complete,” he said. For example, while the TREAT EARLIER trial recruited patients with joint pain and subclinical joint inflammation via MRI, regardless of autoantibody status, the APIPPRA trial enrolled patients that were both ACPA+ and rheumatoid factor (RF)+ with joint pain.
“You’re left extrapolating as to whether [these interventions] will work in different at-risk populations,” he said.
Even with specific inclusion criteria in each study, there can still be heterogeneity in risk within a study group, Deane said. In the TREAT EARLIER study, 18%-20% of participants ultimately developed RA over the study period, which is lower than expected.
“While it seemed like a pretty high-risk group, it wasn’t as high risk as we thought,” he said, “and that’s why we’ve gone back to the drawing board.”
Risk Stratification Efforts
There are now two ongoing joint efforts by the American College of Rheumatology (ACR) and the European Alliance of Associations for Rheumatology (EULAR) to define these populations and “bring some consensus to the field,” Mankia said.
The first aims to create a unanimous risk stratification tool for future RA prevention studies. The proposed system, devised for individuals with new joint symptoms who are at a risk for RA, was presented at the EULAR 2024 annual meeting and will be further discussed at the upcoming ACR 2024 annual meeting in Washington, DC.
The system uses a point system based on six criteria — three lab tests and three criteria commonly assessed in clinical practice:
- Morning stiffness
- Patient-reported joint swelling
- Difficulty making a fist
- Increased C-reactive protein
- RF positivity
- ACPA positivity
These criteria were picked so that the risk stratification tool can be used without imaging; however, the inclusion of MRI can further refine the score.
The ACR-EULAR task force that created the tool has emphasized that this criterion is specifically designed for research purposes and should not be used in clinical practice. Using this stratification tool should allow future clinical studies to group patients by similar risk, Deane said.
“Not that all studies have to look at exactly the same people, but each study should have similar risk stratification,” he said.
The second ACR-EULAR joint effort is taking a population-based approach to risk stratification, Deane said, to better predict RA risk in individuals without common symptoms like joint pain.
The aim is to create something analogous to the Framingham Risk Score in predicting cardiovascular disease, in which simple variables like total cholesterol, high-density lipoprotein cholesterol, systolic blood pressure, and smoking status can be used to calculate an individual’s 10-year risk for CVD, Deane explained.
The second approach could also identify patients earlier in the progression to RA, which may be easier to treat than later stages of disease.
Understanding RA Origins
However, treating an earlier stage of disease might require a different approach. Up to this point, medical interventions for RA prevention used drugs approved to treat RA, but inventions during the pre-RA stage — before any joint symptoms appear — might require targeting different immunologic pathways.
“The general concept is if there is a pre-RA stage when joints are not involved, that means all the immunologic abnormalities are probably happening somewhere else in the body,” he said. “The big question is: Where is that, and how exactly is that happening?”
One theory is that RA begins to develop in mucosal sites, such as the intestines or lungs, before it involves synovial joints.
“In the absence of resolution, these localized immune processes transition into a systemic process that targets the joints, either by direct effects of microbiota, molecular mimicry, and/or immune amplification,” wrote Deane and coauthors in a recent review article in Annals of the Rheumatic Diseases. “This, in turn, leads to inappropriate engagement of a range of effector mechanisms in both synovium and periarticular sites.”
Following this logic, the progression of the at-risk stage of RA could be considered a continuum along which there are multiple possible points for intervention. It’s also probable that the disease can develop through multiple pathways, Deane said.
“If you look at all the people who get rheumatoid arthritis, there’s probably no way those could have the same exact pathways,” he said. “There’s probably going to be different endotypes and understanding that is going to help us prevent disease in a better way.”
Looking Forward
Beyond improving risk stratification and understanding RA pathogenesis, researchers are also considering novel therapeutic approaches for future trials. Glucagon-like peptide 1 (GLP-1) receptor agonists could be worth exploring in RA prevention and treatment, said Jeffrey A. Sparks, MD, MMSc, a rheumatologist at Brigham and Women’s Hospital, Boston, Massachusetts.
These drugs — initially developed for diabetes — have already shown anti-inflammatory effects, and one study suggested that GLP-1s lowered the risk for major adverse cardiovascular events and all-cause mortality in individuals with immune-mediated inflammatory diseases. Obesity is a known risk factor for RA, so weight loss aided by GLP-1 drugs could also help reduce risk in certain patients. Clinical trials are needed to explore GLP-1s for both RA prevention and treatment, he said.
While prevention trials up to this point have used one-time, time-limited interventions, longer durations of medication or multiple rounds of therapy may be more efficacious. Even for trials that demonstrated the intervention arms had less progression to RA, this effect diminished once participants stopped the medication. In the ARIAA and APIPPRA trials using abatacept, “it wasn’t like we hit a reset button and [patients] just permanently now did not get rheumatoid arthritis,” Deane said, suggesting that alternative approaches should be explored.
“Future studies need to look at potentially longer doses of drug or lower doses of drug, or some combination that might be effective,” he said.
Deane received honoraria from Bristol-Myers Squibb, Thermo Fisher, and Werfen and grant funding from Janssen Research and Development and Gilead Sciences. Mankia received grant support from Gilead, Lilly, AstraZeneca, and Serac Life Sciences and honoraria or consultant fees from AbbVie, UCB, Lilly, Galapagos, DeepCure, Serac Life Sciences, AstraZeneca, and Zura Bio. Sparks received research support from Boehringer Ingelheim, Bristol-Myers Squibb, Janssen, and Sonoma Biotherapeutics. He consulted for AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Gilead, Inova Diagnostics, Janssen, Merck, Mustang, Optum, Pfizer, ReCor Medical, Sana, Sobi, and UCB.
A version of this article first appeared on Medscape.com.
With the discovery of autoantibodies and other risk factors for rheumatoid arthritis (RA), researchers developed clinical trials to see whether the disease can be prevented entirely. In the past 10 years, a number of these trials have concluded, with variable results.
While some trials demonstrated no effect at all, others showed that medical intervention can delay the onset of disease in certain populations and even reduce the rates of progression to RA. These completed trials also offer researchers the chance to identify opportunities to improve RA prevention trials moving forward.
“We’re looking at all that data and trying to figure out what the next step is going to be,” said Kevin Deane, MD, PhD, a professor of medicine and a rheumatologist at the University of Colorado School of Medicine, Aurora.
Key lessons include the need for improved risk stratification tools and better understanding of RA pathogenesis, he said.
The Research So Far
All RA prevention trials except for one have been completed and/or published within the past decade, bringing valuable insights to the field. (See chart below.)

Atorvastatin (STAPRA) and hydroxychloroquine (StopRA) proved ineffective in preventing the onset of RA, and both trials were stopped early. Rituximab and methotrexate (MTX) both delayed the onset of RA, but the effect disappeared by the end of the follow-up periods.
However, the 2-year results from the TREAT EARLIER trial showed that compared with patients given placebo, those given MTX showed improved MRI-detected joint inflammation, physical functioning, and reported symptoms.
The 4-year analysis of the trial further risk stratified participants and found that MTX showed a preventive effect in anti–citrullinated protein antibody (ACPA)–negative participants at an increased risk for RA.
Abatacept also showed promise in preventing RA in two separate trials. In the ARIAA trial, compared with placebo, 6 months of treatment with abatacept reduced MRI inflammation and symptoms and lowered the rates of progression to RA. This treatment effect lessened during the 1-year follow-up period, but the difference between the two groups was still significant at 18 months.
In the APIPPRA trial, 12 months of treatment with abatacept improved subclinical inflammation and quality-of-life measures in participants and reduced the rates of progression to RA through another 12 months of observation. However, during this post-treatment follow-up period, the treatment effect began to diminish.
While there have been some promising findings — not only in disease prevention but also in disease modification — these studies all looked at different patient groups, noted Kulveer Mankia, MA, DM, an associate professor and consulting rheumatologist at the Leeds Institute of Rheumatic and Musculoskeletal Medicine, University of Leeds in England.
“You have disparate, different inclusion criteria in different studies, all of which take years to complete,” he said. For example, while the TREAT EARLIER trial recruited patients with joint pain and subclinical joint inflammation via MRI, regardless of autoantibody status, the APIPPRA trial enrolled patients that were both ACPA+ and rheumatoid factor (RF)+ with joint pain.
“You’re left extrapolating as to whether [these interventions] will work in different at-risk populations,” he said.
Even with specific inclusion criteria in each study, there can still be heterogeneity in risk within a study group, Deane said. In the TREAT EARLIER study, 18%-20% of participants ultimately developed RA over the study period, which is lower than expected.
“While it seemed like a pretty high-risk group, it wasn’t as high risk as we thought,” he said, “and that’s why we’ve gone back to the drawing board.”
Risk Stratification Efforts
There are now two ongoing joint efforts by the American College of Rheumatology (ACR) and the European Alliance of Associations for Rheumatology (EULAR) to define these populations and “bring some consensus to the field,” Mankia said.
The first aims to create a unanimous risk stratification tool for future RA prevention studies. The proposed system, devised for individuals with new joint symptoms who are at a risk for RA, was presented at the EULAR 2024 annual meeting and will be further discussed at the upcoming ACR 2024 annual meeting in Washington, DC.
The system uses a point system based on six criteria — three lab tests and three criteria commonly assessed in clinical practice:
- Morning stiffness
- Patient-reported joint swelling
- Difficulty making a fist
- Increased C-reactive protein
- RF positivity
- ACPA positivity
These criteria were picked so that the risk stratification tool can be used without imaging; however, the inclusion of MRI can further refine the score.
The ACR-EULAR task force that created the tool has emphasized that this criterion is specifically designed for research purposes and should not be used in clinical practice. Using this stratification tool should allow future clinical studies to group patients by similar risk, Deane said.
“Not that all studies have to look at exactly the same people, but each study should have similar risk stratification,” he said.
The second ACR-EULAR joint effort is taking a population-based approach to risk stratification, Deane said, to better predict RA risk in individuals without common symptoms like joint pain.
The aim is to create something analogous to the Framingham Risk Score in predicting cardiovascular disease, in which simple variables like total cholesterol, high-density lipoprotein cholesterol, systolic blood pressure, and smoking status can be used to calculate an individual’s 10-year risk for CVD, Deane explained.
The second approach could also identify patients earlier in the progression to RA, which may be easier to treat than later stages of disease.
Understanding RA Origins
However, treating an earlier stage of disease might require a different approach. Up to this point, medical interventions for RA prevention used drugs approved to treat RA, but inventions during the pre-RA stage — before any joint symptoms appear — might require targeting different immunologic pathways.
“The general concept is if there is a pre-RA stage when joints are not involved, that means all the immunologic abnormalities are probably happening somewhere else in the body,” he said. “The big question is: Where is that, and how exactly is that happening?”
One theory is that RA begins to develop in mucosal sites, such as the intestines or lungs, before it involves synovial joints.
“In the absence of resolution, these localized immune processes transition into a systemic process that targets the joints, either by direct effects of microbiota, molecular mimicry, and/or immune amplification,” wrote Deane and coauthors in a recent review article in Annals of the Rheumatic Diseases. “This, in turn, leads to inappropriate engagement of a range of effector mechanisms in both synovium and periarticular sites.”
Following this logic, the progression of the at-risk stage of RA could be considered a continuum along which there are multiple possible points for intervention. It’s also probable that the disease can develop through multiple pathways, Deane said.
“If you look at all the people who get rheumatoid arthritis, there’s probably no way those could have the same exact pathways,” he said. “There’s probably going to be different endotypes and understanding that is going to help us prevent disease in a better way.”
Looking Forward
Beyond improving risk stratification and understanding RA pathogenesis, researchers are also considering novel therapeutic approaches for future trials. Glucagon-like peptide 1 (GLP-1) receptor agonists could be worth exploring in RA prevention and treatment, said Jeffrey A. Sparks, MD, MMSc, a rheumatologist at Brigham and Women’s Hospital, Boston, Massachusetts.
These drugs — initially developed for diabetes — have already shown anti-inflammatory effects, and one study suggested that GLP-1s lowered the risk for major adverse cardiovascular events and all-cause mortality in individuals with immune-mediated inflammatory diseases. Obesity is a known risk factor for RA, so weight loss aided by GLP-1 drugs could also help reduce risk in certain patients. Clinical trials are needed to explore GLP-1s for both RA prevention and treatment, he said.
While prevention trials up to this point have used one-time, time-limited interventions, longer durations of medication or multiple rounds of therapy may be more efficacious. Even for trials that demonstrated the intervention arms had less progression to RA, this effect diminished once participants stopped the medication. In the ARIAA and APIPPRA trials using abatacept, “it wasn’t like we hit a reset button and [patients] just permanently now did not get rheumatoid arthritis,” Deane said, suggesting that alternative approaches should be explored.
“Future studies need to look at potentially longer doses of drug or lower doses of drug, or some combination that might be effective,” he said.
Deane received honoraria from Bristol-Myers Squibb, Thermo Fisher, and Werfen and grant funding from Janssen Research and Development and Gilead Sciences. Mankia received grant support from Gilead, Lilly, AstraZeneca, and Serac Life Sciences and honoraria or consultant fees from AbbVie, UCB, Lilly, Galapagos, DeepCure, Serac Life Sciences, AstraZeneca, and Zura Bio. Sparks received research support from Boehringer Ingelheim, Bristol-Myers Squibb, Janssen, and Sonoma Biotherapeutics. He consulted for AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Gilead, Inova Diagnostics, Janssen, Merck, Mustang, Optum, Pfizer, ReCor Medical, Sana, Sobi, and UCB.
A version of this article first appeared on Medscape.com.
With the discovery of autoantibodies and other risk factors for rheumatoid arthritis (RA), researchers developed clinical trials to see whether the disease can be prevented entirely. In the past 10 years, a number of these trials have concluded, with variable results.
While some trials demonstrated no effect at all, others showed that medical intervention can delay the onset of disease in certain populations and even reduce the rates of progression to RA. These completed trials also offer researchers the chance to identify opportunities to improve RA prevention trials moving forward.
“We’re looking at all that data and trying to figure out what the next step is going to be,” said Kevin Deane, MD, PhD, a professor of medicine and a rheumatologist at the University of Colorado School of Medicine, Aurora.
Key lessons include the need for improved risk stratification tools and better understanding of RA pathogenesis, he said.
The Research So Far
All RA prevention trials except for one have been completed and/or published within the past decade, bringing valuable insights to the field. (See chart below.)

Atorvastatin (STAPRA) and hydroxychloroquine (StopRA) proved ineffective in preventing the onset of RA, and both trials were stopped early. Rituximab and methotrexate (MTX) both delayed the onset of RA, but the effect disappeared by the end of the follow-up periods.
However, the 2-year results from the TREAT EARLIER trial showed that compared with patients given placebo, those given MTX showed improved MRI-detected joint inflammation, physical functioning, and reported symptoms.
The 4-year analysis of the trial further risk stratified participants and found that MTX showed a preventive effect in anti–citrullinated protein antibody (ACPA)–negative participants at an increased risk for RA.
Abatacept also showed promise in preventing RA in two separate trials. In the ARIAA trial, compared with placebo, 6 months of treatment with abatacept reduced MRI inflammation and symptoms and lowered the rates of progression to RA. This treatment effect lessened during the 1-year follow-up period, but the difference between the two groups was still significant at 18 months.
In the APIPPRA trial, 12 months of treatment with abatacept improved subclinical inflammation and quality-of-life measures in participants and reduced the rates of progression to RA through another 12 months of observation. However, during this post-treatment follow-up period, the treatment effect began to diminish.
While there have been some promising findings — not only in disease prevention but also in disease modification — these studies all looked at different patient groups, noted Kulveer Mankia, MA, DM, an associate professor and consulting rheumatologist at the Leeds Institute of Rheumatic and Musculoskeletal Medicine, University of Leeds in England.
“You have disparate, different inclusion criteria in different studies, all of which take years to complete,” he said. For example, while the TREAT EARLIER trial recruited patients with joint pain and subclinical joint inflammation via MRI, regardless of autoantibody status, the APIPPRA trial enrolled patients that were both ACPA+ and rheumatoid factor (RF)+ with joint pain.
“You’re left extrapolating as to whether [these interventions] will work in different at-risk populations,” he said.
Even with specific inclusion criteria in each study, there can still be heterogeneity in risk within a study group, Deane said. In the TREAT EARLIER study, 18%-20% of participants ultimately developed RA over the study period, which is lower than expected.
“While it seemed like a pretty high-risk group, it wasn’t as high risk as we thought,” he said, “and that’s why we’ve gone back to the drawing board.”
Risk Stratification Efforts
There are now two ongoing joint efforts by the American College of Rheumatology (ACR) and the European Alliance of Associations for Rheumatology (EULAR) to define these populations and “bring some consensus to the field,” Mankia said.
The first aims to create a unanimous risk stratification tool for future RA prevention studies. The proposed system, devised for individuals with new joint symptoms who are at a risk for RA, was presented at the EULAR 2024 annual meeting and will be further discussed at the upcoming ACR 2024 annual meeting in Washington, DC.
The system uses a point system based on six criteria — three lab tests and three criteria commonly assessed in clinical practice:
- Morning stiffness
- Patient-reported joint swelling
- Difficulty making a fist
- Increased C-reactive protein
- RF positivity
- ACPA positivity
These criteria were picked so that the risk stratification tool can be used without imaging; however, the inclusion of MRI can further refine the score.
The ACR-EULAR task force that created the tool has emphasized that this criterion is specifically designed for research purposes and should not be used in clinical practice. Using this stratification tool should allow future clinical studies to group patients by similar risk, Deane said.
“Not that all studies have to look at exactly the same people, but each study should have similar risk stratification,” he said.
The second ACR-EULAR joint effort is taking a population-based approach to risk stratification, Deane said, to better predict RA risk in individuals without common symptoms like joint pain.
The aim is to create something analogous to the Framingham Risk Score in predicting cardiovascular disease, in which simple variables like total cholesterol, high-density lipoprotein cholesterol, systolic blood pressure, and smoking status can be used to calculate an individual’s 10-year risk for CVD, Deane explained.
The second approach could also identify patients earlier in the progression to RA, which may be easier to treat than later stages of disease.
Understanding RA Origins
However, treating an earlier stage of disease might require a different approach. Up to this point, medical interventions for RA prevention used drugs approved to treat RA, but inventions during the pre-RA stage — before any joint symptoms appear — might require targeting different immunologic pathways.
“The general concept is if there is a pre-RA stage when joints are not involved, that means all the immunologic abnormalities are probably happening somewhere else in the body,” he said. “The big question is: Where is that, and how exactly is that happening?”
One theory is that RA begins to develop in mucosal sites, such as the intestines or lungs, before it involves synovial joints.
“In the absence of resolution, these localized immune processes transition into a systemic process that targets the joints, either by direct effects of microbiota, molecular mimicry, and/or immune amplification,” wrote Deane and coauthors in a recent review article in Annals of the Rheumatic Diseases. “This, in turn, leads to inappropriate engagement of a range of effector mechanisms in both synovium and periarticular sites.”
Following this logic, the progression of the at-risk stage of RA could be considered a continuum along which there are multiple possible points for intervention. It’s also probable that the disease can develop through multiple pathways, Deane said.
“If you look at all the people who get rheumatoid arthritis, there’s probably no way those could have the same exact pathways,” he said. “There’s probably going to be different endotypes and understanding that is going to help us prevent disease in a better way.”
Looking Forward
Beyond improving risk stratification and understanding RA pathogenesis, researchers are also considering novel therapeutic approaches for future trials. Glucagon-like peptide 1 (GLP-1) receptor agonists could be worth exploring in RA prevention and treatment, said Jeffrey A. Sparks, MD, MMSc, a rheumatologist at Brigham and Women’s Hospital, Boston, Massachusetts.
These drugs — initially developed for diabetes — have already shown anti-inflammatory effects, and one study suggested that GLP-1s lowered the risk for major adverse cardiovascular events and all-cause mortality in individuals with immune-mediated inflammatory diseases. Obesity is a known risk factor for RA, so weight loss aided by GLP-1 drugs could also help reduce risk in certain patients. Clinical trials are needed to explore GLP-1s for both RA prevention and treatment, he said.
While prevention trials up to this point have used one-time, time-limited interventions, longer durations of medication or multiple rounds of therapy may be more efficacious. Even for trials that demonstrated the intervention arms had less progression to RA, this effect diminished once participants stopped the medication. In the ARIAA and APIPPRA trials using abatacept, “it wasn’t like we hit a reset button and [patients] just permanently now did not get rheumatoid arthritis,” Deane said, suggesting that alternative approaches should be explored.
“Future studies need to look at potentially longer doses of drug or lower doses of drug, or some combination that might be effective,” he said.
Deane received honoraria from Bristol-Myers Squibb, Thermo Fisher, and Werfen and grant funding from Janssen Research and Development and Gilead Sciences. Mankia received grant support from Gilead, Lilly, AstraZeneca, and Serac Life Sciences and honoraria or consultant fees from AbbVie, UCB, Lilly, Galapagos, DeepCure, Serac Life Sciences, AstraZeneca, and Zura Bio. Sparks received research support from Boehringer Ingelheim, Bristol-Myers Squibb, Janssen, and Sonoma Biotherapeutics. He consulted for AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Gilead, Inova Diagnostics, Janssen, Merck, Mustang, Optum, Pfizer, ReCor Medical, Sana, Sobi, and UCB.
A version of this article first appeared on Medscape.com.
Novel Treatment Promising for Cutaneous Lupus in Phase 2 Trial
TOPLINE:
particularly in subacute and chronic cases.
METHODOLOGY:
- Researchers conducted a randomized phase 2 trial to evaluate the efficacy and safety of iberdomide in 288 patients with CLE (mean age, 45 years; 97% women). Iberdomide is a cereblon modulator, which results in degradation of two transcription factors of immune cell development and homeostasis — Ikaros and Aiolos — that have been implicated in the genetic predisposition of systemic lupus.
- CLE Disease Area and Severity Index Activity (CLASI-A) endpoints included mean percent change from baseline and ≥ 50% reduction from baseline (CLASI-50), which were evaluated in all patients with baseline CLASI-A scores ≥ 8 and by CLE subtypes (acute, subacute, and chronic).
- At baseline, 56% of patients had acute CLE, 29% had chronic CLE, and 16% had subacute CLE; 28% of patients had a baseline CLASI-A score ≥ 8.
- Patients were randomly assigned to receive oral iberdomide (0.45 mg, 0.30 mg, 0.15 mg, or placebo daily) for 24 weeks while continuing standard lupus medications. At week 24, patients on placebo were rerandomized to iberdomide 0.45 mg or 0.30 mg once a day, while those on iberdomide continued their assigned dose through week 52.
TAKEAWAY:
- Among patients with baseline CLASI-A ≥ 8, the mean change in CLASI-A score from baseline at week 24 was −66.7% for those on iberdomide 0.45 mg vs −54.2% for placebo (P = .295).
- At week 24, patients with subacute CLE showed a significantly greater mean percent change from baseline in CLASI-A with iberdomide 0.45 mg vs placebo (−90.5% vs −51.2%; P = .007), while no significant differences were observed with the 0.45-mg dose vs placebo in patients with chronic or acute CLE.
- Overall, CLASI-50 responses were not significantly different among those on 0.45 mg vs placebo (55.6% vs 44.6%). The proportions of patients achieving CLASI-50 at week 24 were significantly greater for iberdomide 0.45 mg vs placebo for those with subacute CLE (91.7% vs 52.9%; P = .035) and chronic CLE (62.1% vs 27.8%; P = .029), but not for those with baseline CLASI-A ≥ 8 (66.7% vs 50%).
- More than 80% of patients had treatment-emergent adverse events (TEAEs), which were mostly mild to moderate. Over 2 years, the most common were urinary tract infections, upper respiratory tract infections, neutropenia, and nasopharyngitis. TEAEs leading to iberdomide discontinuation in one or more patients were neutropenia (n = 7), rash (n = 7), increased hepatic enzymes (n = 4), and deep vein thrombosis (n = 3).
IN PRACTICE:
“Data from this phase 2 trial of iberdomide in patients with SLE suggest that a greater proportion of patients with subacute or chronic CLE who received the higher dose of 0.45 mg iberdomide achieved CLASI-50 vs placebo. For the overall population, CLASI-50 response was not significantly different between treatment groups at week 24, partly due to a high placebo response that may have been driven by patients with acute CLE,” the authors wrote.
SOURCE:
The study was led by Victoria P. Werth, MD, of the University of Pennsylvania and the Veteran’s Administration Medical Center, both in Philadelphia, and was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The study included small patient subgroups for different CLE subtypes, which may affect the generalizability of the findings. CLE subtype was determined by the investigator without additional photographic adjudication. Additionally, the use of background lupus medications could have influenced the placebo group’s response, limiting the ability to observe the treatment effect of iberdomide monotherapy.
DISCLOSURES:
The study was funded by Bristol-Myers Squibb. Six authors reported being employed by Bristol-Myers Squibb, and several others reported consultancy and research support from various sources including Bristol-Myers Squibb.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
particularly in subacute and chronic cases.
METHODOLOGY:
- Researchers conducted a randomized phase 2 trial to evaluate the efficacy and safety of iberdomide in 288 patients with CLE (mean age, 45 years; 97% women). Iberdomide is a cereblon modulator, which results in degradation of two transcription factors of immune cell development and homeostasis — Ikaros and Aiolos — that have been implicated in the genetic predisposition of systemic lupus.
- CLE Disease Area and Severity Index Activity (CLASI-A) endpoints included mean percent change from baseline and ≥ 50% reduction from baseline (CLASI-50), which were evaluated in all patients with baseline CLASI-A scores ≥ 8 and by CLE subtypes (acute, subacute, and chronic).
- At baseline, 56% of patients had acute CLE, 29% had chronic CLE, and 16% had subacute CLE; 28% of patients had a baseline CLASI-A score ≥ 8.
- Patients were randomly assigned to receive oral iberdomide (0.45 mg, 0.30 mg, 0.15 mg, or placebo daily) for 24 weeks while continuing standard lupus medications. At week 24, patients on placebo were rerandomized to iberdomide 0.45 mg or 0.30 mg once a day, while those on iberdomide continued their assigned dose through week 52.
TAKEAWAY:
- Among patients with baseline CLASI-A ≥ 8, the mean change in CLASI-A score from baseline at week 24 was −66.7% for those on iberdomide 0.45 mg vs −54.2% for placebo (P = .295).
- At week 24, patients with subacute CLE showed a significantly greater mean percent change from baseline in CLASI-A with iberdomide 0.45 mg vs placebo (−90.5% vs −51.2%; P = .007), while no significant differences were observed with the 0.45-mg dose vs placebo in patients with chronic or acute CLE.
- Overall, CLASI-50 responses were not significantly different among those on 0.45 mg vs placebo (55.6% vs 44.6%). The proportions of patients achieving CLASI-50 at week 24 were significantly greater for iberdomide 0.45 mg vs placebo for those with subacute CLE (91.7% vs 52.9%; P = .035) and chronic CLE (62.1% vs 27.8%; P = .029), but not for those with baseline CLASI-A ≥ 8 (66.7% vs 50%).
- More than 80% of patients had treatment-emergent adverse events (TEAEs), which were mostly mild to moderate. Over 2 years, the most common were urinary tract infections, upper respiratory tract infections, neutropenia, and nasopharyngitis. TEAEs leading to iberdomide discontinuation in one or more patients were neutropenia (n = 7), rash (n = 7), increased hepatic enzymes (n = 4), and deep vein thrombosis (n = 3).
IN PRACTICE:
“Data from this phase 2 trial of iberdomide in patients with SLE suggest that a greater proportion of patients with subacute or chronic CLE who received the higher dose of 0.45 mg iberdomide achieved CLASI-50 vs placebo. For the overall population, CLASI-50 response was not significantly different between treatment groups at week 24, partly due to a high placebo response that may have been driven by patients with acute CLE,” the authors wrote.
SOURCE:
The study was led by Victoria P. Werth, MD, of the University of Pennsylvania and the Veteran’s Administration Medical Center, both in Philadelphia, and was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The study included small patient subgroups for different CLE subtypes, which may affect the generalizability of the findings. CLE subtype was determined by the investigator without additional photographic adjudication. Additionally, the use of background lupus medications could have influenced the placebo group’s response, limiting the ability to observe the treatment effect of iberdomide monotherapy.
DISCLOSURES:
The study was funded by Bristol-Myers Squibb. Six authors reported being employed by Bristol-Myers Squibb, and several others reported consultancy and research support from various sources including Bristol-Myers Squibb.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
particularly in subacute and chronic cases.
METHODOLOGY:
- Researchers conducted a randomized phase 2 trial to evaluate the efficacy and safety of iberdomide in 288 patients with CLE (mean age, 45 years; 97% women). Iberdomide is a cereblon modulator, which results in degradation of two transcription factors of immune cell development and homeostasis — Ikaros and Aiolos — that have been implicated in the genetic predisposition of systemic lupus.
- CLE Disease Area and Severity Index Activity (CLASI-A) endpoints included mean percent change from baseline and ≥ 50% reduction from baseline (CLASI-50), which were evaluated in all patients with baseline CLASI-A scores ≥ 8 and by CLE subtypes (acute, subacute, and chronic).
- At baseline, 56% of patients had acute CLE, 29% had chronic CLE, and 16% had subacute CLE; 28% of patients had a baseline CLASI-A score ≥ 8.
- Patients were randomly assigned to receive oral iberdomide (0.45 mg, 0.30 mg, 0.15 mg, or placebo daily) for 24 weeks while continuing standard lupus medications. At week 24, patients on placebo were rerandomized to iberdomide 0.45 mg or 0.30 mg once a day, while those on iberdomide continued their assigned dose through week 52.
TAKEAWAY:
- Among patients with baseline CLASI-A ≥ 8, the mean change in CLASI-A score from baseline at week 24 was −66.7% for those on iberdomide 0.45 mg vs −54.2% for placebo (P = .295).
- At week 24, patients with subacute CLE showed a significantly greater mean percent change from baseline in CLASI-A with iberdomide 0.45 mg vs placebo (−90.5% vs −51.2%; P = .007), while no significant differences were observed with the 0.45-mg dose vs placebo in patients with chronic or acute CLE.
- Overall, CLASI-50 responses were not significantly different among those on 0.45 mg vs placebo (55.6% vs 44.6%). The proportions of patients achieving CLASI-50 at week 24 were significantly greater for iberdomide 0.45 mg vs placebo for those with subacute CLE (91.7% vs 52.9%; P = .035) and chronic CLE (62.1% vs 27.8%; P = .029), but not for those with baseline CLASI-A ≥ 8 (66.7% vs 50%).
- More than 80% of patients had treatment-emergent adverse events (TEAEs), which were mostly mild to moderate. Over 2 years, the most common were urinary tract infections, upper respiratory tract infections, neutropenia, and nasopharyngitis. TEAEs leading to iberdomide discontinuation in one or more patients were neutropenia (n = 7), rash (n = 7), increased hepatic enzymes (n = 4), and deep vein thrombosis (n = 3).
IN PRACTICE:
“Data from this phase 2 trial of iberdomide in patients with SLE suggest that a greater proportion of patients with subacute or chronic CLE who received the higher dose of 0.45 mg iberdomide achieved CLASI-50 vs placebo. For the overall population, CLASI-50 response was not significantly different between treatment groups at week 24, partly due to a high placebo response that may have been driven by patients with acute CLE,” the authors wrote.
SOURCE:
The study was led by Victoria P. Werth, MD, of the University of Pennsylvania and the Veteran’s Administration Medical Center, both in Philadelphia, and was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The study included small patient subgroups for different CLE subtypes, which may affect the generalizability of the findings. CLE subtype was determined by the investigator without additional photographic adjudication. Additionally, the use of background lupus medications could have influenced the placebo group’s response, limiting the ability to observe the treatment effect of iberdomide monotherapy.
DISCLOSURES:
The study was funded by Bristol-Myers Squibb. Six authors reported being employed by Bristol-Myers Squibb, and several others reported consultancy and research support from various sources including Bristol-Myers Squibb.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
What’s the Evidence Behind Popular Supplements in Rheumatology? Experts Weigh in
Many people with rheumatologic diseases try supplements for symptom relief. Here’s what you need to know about some common picks.
Dietary supplements were a $159 billion business in the United States in 2023, and many people with rheumatologic diseases are buying in. Research suggests more than 6 in 10 people with fibromyalgia, nearly 8 in 10 people with Sjögren’s disease, and more than 8 in 10 people with rheumatoid arthritis (RA) take dietary supplements.
Whatever the symptom — pain, swelling, or fatigue — you can probably find a supplement purporting to relieve it. But do these supplements work, and are they safe? A study review in RMD Open comprising 24 systematic reviews and 150 original articles suggests more high-quality research is needed on the effects of dietary supplements on rheumatologic diseases. Most studies have focused on RA or osteoarthritis (OA), where the evidence level is moderate at best.
“The studies in this space are usually not very high quality because there’s no money to support them, among other things, plus the products are disparate,” said Janet Funk, MD, MS, professor in the School of Nutritional Sciences and Wellness at the University of Arizona, Tucson. She recommended brushing up on supplements and finding out what patients are taking so you can offer advice and watch for drug-supplement interactions.
When asked for a medication list, many patients forget to report supplements, Funk said. “You have to prompt them specifically. I think some physicians have very negative views about supplements because so little data is known, and patients might pick up on that and decide not to report their use.” She recommended saying something like: “To give you the best possible care, I want to know everything you’re taking, including supplements. The things I’m prescribing could maybe interact with the things you’re taking, so I want to make sure I know about all of it so that together we can figure out if the combination of things is safe.”
The quality of dietary supplements varies, and they aren’t regulated like drugs by the Food and Drug Administration. Funk recommended selecting products verified by NSF or ConsumerLab. They test supplements to ensure the label reflects what’s inside.
This news organization scoured the literature and asked experts to weigh in on the evidence behind popular supplements in rheumatology today.
The Essential Nutrients
Vitamin supplements are a staple in many homes — but are they helpful? “Individual vitamin supplements will not provide any benefit unless the person is deficient in a specific vitamin or mineral,” according to Elena Philippou, PhD, RD, associate professor of nutrition-dietetics at the University of Nicosia in Cyprus, and Elena Nikiphorou, MBBS, a rheumatologist at King’s College London in England. For some patients, deficiency is a reality. A retrospective cohort study in The Journal of Clinical Medicine found that people with RA were 17% more likely than age-matched control individuals to have nutrient deficiencies, perhaps because symptoms like fatigue, pain, and nausea affect their eating habits. Here’s what the science says about common vitamin supplements.
Vitamin D. This hormone-like vitamin, which attaches to receptors on immune cells to tamp down inflammation, was the most popular dietary supplement among rheumatology patients in a recent study from the United Kingdom. Vitamin D deficiency is common in people with RA, lupus, Sjögren’s disease, ankylosing spondylitis, systemic sclerosis, and fibromyalgia. In some cases, vitamin D levels track with disease activity, research suggests. Corticosteroids can also make vitamin D deficiency more likely. Can supplements help?
In RA, evidence points to small improvements. A systematic review of 11 studies including 3049 patients published in Nutrition Reviews showed that vitamin D supplements significantly reduced patients’ pain and Disease Activity Score in 28 joints (DAS28) using both C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR).
The research is mixed on the benefits of vitamin D supplementation for fibromyalgia symptoms, according to a study review in SN Comprehensive Clinical Medicine that included two studies and 80 patients on supplementation. However, researchers said it’s still worth discussing the potential benefits of taking vitamin D.
“Vitamin D supplementation is important in the context of various rheumatic diseases to prevent or treat bone disease,” said Philippou and Nikiphorou. “People with rheumatic disease should speak to their healthcare provider and ask to check their blood vitamin D concentration.” The results can help you recommend a dose.
Folate. Patients on methotrexate should take folic acid supplements under the guidance of a healthcare provider, said Philippou and Nikiphorou. The reason: Methotrexate can deplete folic acid levels, increasing the risk for side effects. An analysis of adverse event reports published showed that methotrexate users who took folic acid (or tumor necrosis factor–alpha inhibitors) had a reduced risk for hepatotoxicity or myelosuppression. A commonly recommended dose is 1 mg/d.
Vitamin B12. In a 2024 perspective paper in Rheumatology International, researchers said physicians should assess vitamin B12 levels early in the diagnostic process of rheumatologic diseases. One reason: Many symptoms of pernicious anemia, like fatigue, mimic symptoms of rheumatologic diseases. The gastrointestinal (GI) effects of systemic sclerosis could bring on vitamin B12 deficiency. In a small study in The Journal of Clinical Rheumatology, 44 of 62 patients with systemic sclerosis had low vitamin B12 levels.
Vitamin E. Vitamin E deficiency is rare in healthy adults. However, some medical conditions, like inflammatory bowel disease and malabsorption disorders, can make vitamin E deficiency more likely. In RA, a vitamin E supplement could help reduce joint swelling and sensitivity, according to a systematic review of nine studies including 39,845 patients in The European Journal of Clinical Nutrition. Researchers credit the nutrient’s role in aiding intestinal repair. Use with caution, as this supplement can increase bleeding risk in doses over 1000 mg/d.
Vitamin A. Like vitamin E, vitamin A deficiency is rare in the United States. The risk of oversupplementing is higher than undersupplementing. However, vitamin A deficiency can happen in people with chronic pancreatic, liver, or GI problems. In people with deficiency, a vitamin A supplement can help relieve dry eye symptoms common in Sjögren’s disease, suggests a narrative review published in Nutrients. Vitamin A might help reduce ocular surface changes by supporting the production of proteins that protect the outermost surfaces of the eyes. The recommended daily allowance for vitamin A is 900 μg. High-dose supplements can cause toxicity, resulting in GI symptoms and problems like lethargy, drowsiness, increased intracranial pressure, and skin changes.
The Replacements
These substances are similar to naturally occurring compounds in our bodies. The question is whether ingesting them yields benefits.
Glucosamine and chondroitin. Glucosamine and chondroitin occur naturally in our bodies and help us form and protect connective tissues. In pill form, this combo is the most popular dietary supplement for OA, according to research in the journal Maturitas. But studies of its effectiveness yield mixed results. A systematic review of 25 studies published in Inflammopharmacology showed that, in patients with knee OA, supplementation with about 1500 mg of glucosamine per day reduced tibiofemoral joint space narrowing, while supplementation with about 800 mg/d of chondroitin reduced pain intensity and improved physical function, compared with placebo. The duo of glucosamine and chondroitin did not bring significant benefits, perhaps because more studies are needed. Most side effects were mild, but some literature points to the potential for glucosamine to increase warfarin’s blood-thinning effects.
Omega-3 fatty acids. Fish oil is a top-selling supplement, and it might be helpful in inflammatory rheumatologic diseases. A systematic review of 30 studies including 710 patients published in Arthritis Research & Therapy showed that omega-3 fatty acid supplements can improve pain, swollen and tender joint count, DAS28 scores, and Health Assessment Questionnaire scores in patients with RA, psoriatic arthritis, or ankylosing spondylitis. In patients with lupus, a study review that included five studies and 284 patients in The International Journal of Environmental Research and Public Health suggested omega-3 fatty acid supplements could improve ESR, CRP, disease activity, inflammatory markers, oxidative stress, lipid levels, and endothelial function.
Omega-3 fatty acids have anti-inflammatory effects that might explain their benefits. In patients with RA, for example, fish oil supplementation was associated with elevated blood levels of resolvins and protectins, which help quell inflammation, according to a study in Prostaglandins, Leukotrienes and Essential Fatty Acids.
Philippou and Nikiphorou recommended combining food and supplements: Eat oily fish at least twice a week, regularly consume plant-based sources of omega-3s — like chia seeds, flaxseeds, or walnuts — and consider a daily supplement that contains 2 g of omega-3s from docosahexaenoic acid and eicosapentaenoic acid. Most fish oil side effects are mild, like heartburn and bad breath. Fish oil can have blood-thinning effects at high doses, so special attention is needed for patients on anticoagulants.
Probiotics. Building up the good bacteria in your gut might help you fight the effects of rheumatologic diseases. A systematic review of 80 randomized controlled trials in BMC Medicine suggested that therapies targeting the gut microbiota might improve the symptoms or inflammatory factors in celiac disease, lupus, juvenile idiopathic arthritis, psoriasis, Sjögren’s disease, multiple sclerosis, systemic sclerosis, Crohn’s disease, and ulcerative colitis. Probiotics were also shown to relieve pain in fibromyalgia, but they didn’t affect scores on the Fibromyalgia Impact Questionnaire. Probiotics were not helpful in spondyloarthritis or RA. There were no adverse events. By improving the balance of bacteria in the gut, probiotics might inhibit pro-inflammatory factors and signaling pathways and regulate CD4+ T-cell differentiation, the researchers wrote.
Not all probiotic supplements are created equal. Effects can vary by microorganism and dose. Until more high-quality studies are published, Philippou and Nikiphorou recommend daily consumption of probiotic food sources such as yogurt, kefir, sauerkraut, kimchi, tempeh, miso, and kombucha, along with prebiotic food sources such as bananas, onion, artichokes, asparagus, oats, leeks, and garlic.
Collagen. An increasingly popular supplement for hair, skin, and nails, some collagen peptide or hydrolyzed collagen supplements come with claims about joint health, too. Inside our bodies, collagen helps build joints. As a supplement, the jury is still out. A systematic review of 19 studies in The International Journal of Rheumatic Diseases suggested more research is needed to determine whether collagen supplements are harmful or helpful in OA or RA. Studies haven’t shown adverse events, and doses typically range from 2.5 to 15 g/d.
Coenzyme Q10 (CoQ10). This antioxidant occurs naturally in our cells and is produced through microbial fermentation for use in dietary supplements. A study review of 20 articles including 483 patients in Clinical Nutrition ESPEN concluded that CoQ10 supplementation up to 300 mg/d was beneficial in RA, fibromyalgia, or antiphospholipid syndrome (APS).
In RA, CoQ10 supplementation improved disease activity index, ESR, and cytokine levels and decreased malondialdehyde. CoQ10 might protect against the overproduction of reactive oxygen species that can promote inflammation and joint damage, the researchers said. In fibromyalgia, CoQ10 was linked with improvements in pain, fatigue, sleep, tender points count, mood disorders, and scores on the Fibromyalgia Impact Questionnaire in most of the included studies. CoQ10 might help in fibromyalgia by improving mitochondrial dysfunction. In APS, CoQ10 improved endothelial function and decreased prothrombotic and pro-inflammatory mediators. CoQ10 might change the expression of genes that promote atherosclerosis. A few patients had GI side effects like nausea and diarrhea, but the supplements were generally well tolerated.
Melatonin. Commonly touted as a sleep aid, this hormone has immune and anti-inflammatory activities that could benefit people with rheumatologic diseases. A study review of 13 articles including 533 patients in Clinical Nutrition ESPEN concluded that melatonin can help improve sleep, pain, and mood in fibromyalgia, OA, and osteoporosis but not in RA. Side effects were minimal, but a few people experienced nausea, drowsiness, nightmares, or headaches. Doses of 5-6 mg/d are likely safe for most adults.
The Plant-Derived Antioxidants
Many supplements used in rheumatology are antioxidants derived from herbs, spices, or other plants. When plants encounter stressors, like temperature changes or hungry insects, their secondary metabolism revs up and creates compounds with biological properties. Some of these substances influence inflammatory pathways in the human body, said Luís Silva, PhD, a medicinal chemistry researcher at the Polytechnic Institute of Guarda in Portugal. “If it is possible to reduce these kinds of anti-inflammatory processes, it is also possible that we could help people with inflammatory diseases to a good life, or a better life.”
Turmeric and curcumin. You might see this supplement labeled as turmeric, a golden spice in curry powder, or curcumin, an antioxidant compound known as a curcuminoid in turmeric. Curcuminoids might reduce inflammation by scavenging free radicals and inhibiting enzymes that make prostaglandins, Silva said.
Turmeric is the most popular herbal supplement for people with RA, according to Funk’s research. A study review of six publications including 539 patients in Frontiers in Immunology showed that curcumin supplements improved RA patients’ ESR, DAS, swollen joint count, and tender joint count. Turmeric could help patients with OA, too. Patients with OA who took 1000 mg/d of curcumin improved their pain and function, according to a systematic review including 12 studies and 1438 participants in the journal Nutrients. In lupus, small studies are promising but inconclusive, suggested a study review in Frontiers in Immunology.
Watch patients taking turmeric and methotrexate closely, Funk said. Both have been associated with liver problems. Some users also experience GI symptoms like diarrhea because turmeric doesn’t absorb well in the GI tract.
Milk thistle (silymarin). This flowering plant is often marketed as a liver-supporting supplement, but research also suggests promise in RA and OA. A systematic review of 12 studies in Current Rheumatology Reviews suggested that silymarin supplements might help relieve pain, reduce inflammation, and protect the cartilage matrix, synovial membrane, and cartilage cells in joints. This supplement might help via immunomodulatory, anti-inflammatory, antioxidant, and anti-apoptotic properties, the researchers said. Doses of 250-750 mg appear to be safe. Side effects such as gastroenteritis, diarrhea, bloating, and headache can occur.
Boswellia serrata. Sourced from the resin of a tree that grows in dry, mountainous regions of Asia and Africa, Boswellia serrata can help relieve joint pain and stiffness and improve joint function in OA, suggested a systematic review of seven trials involving 545 patients in BMC Complementary Medicine and Therapies. Users saw benefits when taking 100-250 mg/d for 4 weeks or more. Compounds in Boswellia serrata may inhibit 5-lipoxygenase, an enzyme involved in producing inflammatory leukotrienes. No adverse events were reported. In some studies, users have reported GI side effects.
Ginger. Ginger is a popular herbal supplement among people with RA, Funk’s research suggested. One small clinical trial involving 70 patients with RA in the journal Gene showed that taking 1500 mg/d of ginger for 12 weeks improved their DAS and boosted their expression of FoxP3 genes, which are linked with the function of regulatory T cells. A meta-analysis including three studies with 330 patients taking ginger published in the journal Nutrients suggested ginger can reduce pain and systemic inflammation in people with OA. Preclinical studies suggested phenolic compounds in this spicy root, such as gingerols, reduce inflammation through multiple mechanisms.
Funk’s research revealed wide variation in the quality of ginger supplements, reinforcing the importance of selecting an independently verified product. Research suggested a safe dose is up to 2-2.5 g/kg body weight.
Resveratrol. Found in red grapes and red wine, this compound is particularly good at blocking COX-2 enzymes, an important step in the inflammatory cascade, Silva said. “Because of their chemical structure, they have great affinity to these enzymes to lead to their inhibition,” he said. A study review of five articles including 481 patients in The European Journal of Rheumatology showed that people with OA, RA, or Takayasu arteritis who took 250-1000 mg/d of resveratrol saw improvements in pain, function, disease activity, joint swelling, and inflammation, with no side effects.
Cinnamon. This warming spice is gaining popularity as a supplement, reported the American Botanical Council. Cinnamon is often marketed as lowering blood sugar and supporting bone health. In a small study of 36 women with RA published in The Journal of the American College of Nutrition, participants who consumed 2 g/d of cinnamon powder had reduced DASs along with reduced pain and tender and swollen joint counts. Cinnamon may reduce pain by inhibiting prostaglandin and blunt inflammation by reducing the release of arachidonic acid from cell membranes, according to a study review in Frontiers in Pharmacology. GI problems and allergic reactions are among the most common side effects.
Funk, Nikiphorou, Philippou, and Silva all had no relevant disclosures.
A version of this article first appeared on Medscape.com.
Many people with rheumatologic diseases try supplements for symptom relief. Here’s what you need to know about some common picks.
Dietary supplements were a $159 billion business in the United States in 2023, and many people with rheumatologic diseases are buying in. Research suggests more than 6 in 10 people with fibromyalgia, nearly 8 in 10 people with Sjögren’s disease, and more than 8 in 10 people with rheumatoid arthritis (RA) take dietary supplements.
Whatever the symptom — pain, swelling, or fatigue — you can probably find a supplement purporting to relieve it. But do these supplements work, and are they safe? A study review in RMD Open comprising 24 systematic reviews and 150 original articles suggests more high-quality research is needed on the effects of dietary supplements on rheumatologic diseases. Most studies have focused on RA or osteoarthritis (OA), where the evidence level is moderate at best.
“The studies in this space are usually not very high quality because there’s no money to support them, among other things, plus the products are disparate,” said Janet Funk, MD, MS, professor in the School of Nutritional Sciences and Wellness at the University of Arizona, Tucson. She recommended brushing up on supplements and finding out what patients are taking so you can offer advice and watch for drug-supplement interactions.
When asked for a medication list, many patients forget to report supplements, Funk said. “You have to prompt them specifically. I think some physicians have very negative views about supplements because so little data is known, and patients might pick up on that and decide not to report their use.” She recommended saying something like: “To give you the best possible care, I want to know everything you’re taking, including supplements. The things I’m prescribing could maybe interact with the things you’re taking, so I want to make sure I know about all of it so that together we can figure out if the combination of things is safe.”
The quality of dietary supplements varies, and they aren’t regulated like drugs by the Food and Drug Administration. Funk recommended selecting products verified by NSF or ConsumerLab. They test supplements to ensure the label reflects what’s inside.
This news organization scoured the literature and asked experts to weigh in on the evidence behind popular supplements in rheumatology today.
The Essential Nutrients
Vitamin supplements are a staple in many homes — but are they helpful? “Individual vitamin supplements will not provide any benefit unless the person is deficient in a specific vitamin or mineral,” according to Elena Philippou, PhD, RD, associate professor of nutrition-dietetics at the University of Nicosia in Cyprus, and Elena Nikiphorou, MBBS, a rheumatologist at King’s College London in England. For some patients, deficiency is a reality. A retrospective cohort study in The Journal of Clinical Medicine found that people with RA were 17% more likely than age-matched control individuals to have nutrient deficiencies, perhaps because symptoms like fatigue, pain, and nausea affect their eating habits. Here’s what the science says about common vitamin supplements.
Vitamin D. This hormone-like vitamin, which attaches to receptors on immune cells to tamp down inflammation, was the most popular dietary supplement among rheumatology patients in a recent study from the United Kingdom. Vitamin D deficiency is common in people with RA, lupus, Sjögren’s disease, ankylosing spondylitis, systemic sclerosis, and fibromyalgia. In some cases, vitamin D levels track with disease activity, research suggests. Corticosteroids can also make vitamin D deficiency more likely. Can supplements help?
In RA, evidence points to small improvements. A systematic review of 11 studies including 3049 patients published in Nutrition Reviews showed that vitamin D supplements significantly reduced patients’ pain and Disease Activity Score in 28 joints (DAS28) using both C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR).
The research is mixed on the benefits of vitamin D supplementation for fibromyalgia symptoms, according to a study review in SN Comprehensive Clinical Medicine that included two studies and 80 patients on supplementation. However, researchers said it’s still worth discussing the potential benefits of taking vitamin D.
“Vitamin D supplementation is important in the context of various rheumatic diseases to prevent or treat bone disease,” said Philippou and Nikiphorou. “People with rheumatic disease should speak to their healthcare provider and ask to check their blood vitamin D concentration.” The results can help you recommend a dose.
Folate. Patients on methotrexate should take folic acid supplements under the guidance of a healthcare provider, said Philippou and Nikiphorou. The reason: Methotrexate can deplete folic acid levels, increasing the risk for side effects. An analysis of adverse event reports published showed that methotrexate users who took folic acid (or tumor necrosis factor–alpha inhibitors) had a reduced risk for hepatotoxicity or myelosuppression. A commonly recommended dose is 1 mg/d.
Vitamin B12. In a 2024 perspective paper in Rheumatology International, researchers said physicians should assess vitamin B12 levels early in the diagnostic process of rheumatologic diseases. One reason: Many symptoms of pernicious anemia, like fatigue, mimic symptoms of rheumatologic diseases. The gastrointestinal (GI) effects of systemic sclerosis could bring on vitamin B12 deficiency. In a small study in The Journal of Clinical Rheumatology, 44 of 62 patients with systemic sclerosis had low vitamin B12 levels.
Vitamin E. Vitamin E deficiency is rare in healthy adults. However, some medical conditions, like inflammatory bowel disease and malabsorption disorders, can make vitamin E deficiency more likely. In RA, a vitamin E supplement could help reduce joint swelling and sensitivity, according to a systematic review of nine studies including 39,845 patients in The European Journal of Clinical Nutrition. Researchers credit the nutrient’s role in aiding intestinal repair. Use with caution, as this supplement can increase bleeding risk in doses over 1000 mg/d.
Vitamin A. Like vitamin E, vitamin A deficiency is rare in the United States. The risk of oversupplementing is higher than undersupplementing. However, vitamin A deficiency can happen in people with chronic pancreatic, liver, or GI problems. In people with deficiency, a vitamin A supplement can help relieve dry eye symptoms common in Sjögren’s disease, suggests a narrative review published in Nutrients. Vitamin A might help reduce ocular surface changes by supporting the production of proteins that protect the outermost surfaces of the eyes. The recommended daily allowance for vitamin A is 900 μg. High-dose supplements can cause toxicity, resulting in GI symptoms and problems like lethargy, drowsiness, increased intracranial pressure, and skin changes.
The Replacements
These substances are similar to naturally occurring compounds in our bodies. The question is whether ingesting them yields benefits.
Glucosamine and chondroitin. Glucosamine and chondroitin occur naturally in our bodies and help us form and protect connective tissues. In pill form, this combo is the most popular dietary supplement for OA, according to research in the journal Maturitas. But studies of its effectiveness yield mixed results. A systematic review of 25 studies published in Inflammopharmacology showed that, in patients with knee OA, supplementation with about 1500 mg of glucosamine per day reduced tibiofemoral joint space narrowing, while supplementation with about 800 mg/d of chondroitin reduced pain intensity and improved physical function, compared with placebo. The duo of glucosamine and chondroitin did not bring significant benefits, perhaps because more studies are needed. Most side effects were mild, but some literature points to the potential for glucosamine to increase warfarin’s blood-thinning effects.
Omega-3 fatty acids. Fish oil is a top-selling supplement, and it might be helpful in inflammatory rheumatologic diseases. A systematic review of 30 studies including 710 patients published in Arthritis Research & Therapy showed that omega-3 fatty acid supplements can improve pain, swollen and tender joint count, DAS28 scores, and Health Assessment Questionnaire scores in patients with RA, psoriatic arthritis, or ankylosing spondylitis. In patients with lupus, a study review that included five studies and 284 patients in The International Journal of Environmental Research and Public Health suggested omega-3 fatty acid supplements could improve ESR, CRP, disease activity, inflammatory markers, oxidative stress, lipid levels, and endothelial function.
Omega-3 fatty acids have anti-inflammatory effects that might explain their benefits. In patients with RA, for example, fish oil supplementation was associated with elevated blood levels of resolvins and protectins, which help quell inflammation, according to a study in Prostaglandins, Leukotrienes and Essential Fatty Acids.
Philippou and Nikiphorou recommended combining food and supplements: Eat oily fish at least twice a week, regularly consume plant-based sources of omega-3s — like chia seeds, flaxseeds, or walnuts — and consider a daily supplement that contains 2 g of omega-3s from docosahexaenoic acid and eicosapentaenoic acid. Most fish oil side effects are mild, like heartburn and bad breath. Fish oil can have blood-thinning effects at high doses, so special attention is needed for patients on anticoagulants.
Probiotics. Building up the good bacteria in your gut might help you fight the effects of rheumatologic diseases. A systematic review of 80 randomized controlled trials in BMC Medicine suggested that therapies targeting the gut microbiota might improve the symptoms or inflammatory factors in celiac disease, lupus, juvenile idiopathic arthritis, psoriasis, Sjögren’s disease, multiple sclerosis, systemic sclerosis, Crohn’s disease, and ulcerative colitis. Probiotics were also shown to relieve pain in fibromyalgia, but they didn’t affect scores on the Fibromyalgia Impact Questionnaire. Probiotics were not helpful in spondyloarthritis or RA. There were no adverse events. By improving the balance of bacteria in the gut, probiotics might inhibit pro-inflammatory factors and signaling pathways and regulate CD4+ T-cell differentiation, the researchers wrote.
Not all probiotic supplements are created equal. Effects can vary by microorganism and dose. Until more high-quality studies are published, Philippou and Nikiphorou recommend daily consumption of probiotic food sources such as yogurt, kefir, sauerkraut, kimchi, tempeh, miso, and kombucha, along with prebiotic food sources such as bananas, onion, artichokes, asparagus, oats, leeks, and garlic.
Collagen. An increasingly popular supplement for hair, skin, and nails, some collagen peptide or hydrolyzed collagen supplements come with claims about joint health, too. Inside our bodies, collagen helps build joints. As a supplement, the jury is still out. A systematic review of 19 studies in The International Journal of Rheumatic Diseases suggested more research is needed to determine whether collagen supplements are harmful or helpful in OA or RA. Studies haven’t shown adverse events, and doses typically range from 2.5 to 15 g/d.
Coenzyme Q10 (CoQ10). This antioxidant occurs naturally in our cells and is produced through microbial fermentation for use in dietary supplements. A study review of 20 articles including 483 patients in Clinical Nutrition ESPEN concluded that CoQ10 supplementation up to 300 mg/d was beneficial in RA, fibromyalgia, or antiphospholipid syndrome (APS).
In RA, CoQ10 supplementation improved disease activity index, ESR, and cytokine levels and decreased malondialdehyde. CoQ10 might protect against the overproduction of reactive oxygen species that can promote inflammation and joint damage, the researchers said. In fibromyalgia, CoQ10 was linked with improvements in pain, fatigue, sleep, tender points count, mood disorders, and scores on the Fibromyalgia Impact Questionnaire in most of the included studies. CoQ10 might help in fibromyalgia by improving mitochondrial dysfunction. In APS, CoQ10 improved endothelial function and decreased prothrombotic and pro-inflammatory mediators. CoQ10 might change the expression of genes that promote atherosclerosis. A few patients had GI side effects like nausea and diarrhea, but the supplements were generally well tolerated.
Melatonin. Commonly touted as a sleep aid, this hormone has immune and anti-inflammatory activities that could benefit people with rheumatologic diseases. A study review of 13 articles including 533 patients in Clinical Nutrition ESPEN concluded that melatonin can help improve sleep, pain, and mood in fibromyalgia, OA, and osteoporosis but not in RA. Side effects were minimal, but a few people experienced nausea, drowsiness, nightmares, or headaches. Doses of 5-6 mg/d are likely safe for most adults.
The Plant-Derived Antioxidants
Many supplements used in rheumatology are antioxidants derived from herbs, spices, or other plants. When plants encounter stressors, like temperature changes or hungry insects, their secondary metabolism revs up and creates compounds with biological properties. Some of these substances influence inflammatory pathways in the human body, said Luís Silva, PhD, a medicinal chemistry researcher at the Polytechnic Institute of Guarda in Portugal. “If it is possible to reduce these kinds of anti-inflammatory processes, it is also possible that we could help people with inflammatory diseases to a good life, or a better life.”
Turmeric and curcumin. You might see this supplement labeled as turmeric, a golden spice in curry powder, or curcumin, an antioxidant compound known as a curcuminoid in turmeric. Curcuminoids might reduce inflammation by scavenging free radicals and inhibiting enzymes that make prostaglandins, Silva said.
Turmeric is the most popular herbal supplement for people with RA, according to Funk’s research. A study review of six publications including 539 patients in Frontiers in Immunology showed that curcumin supplements improved RA patients’ ESR, DAS, swollen joint count, and tender joint count. Turmeric could help patients with OA, too. Patients with OA who took 1000 mg/d of curcumin improved their pain and function, according to a systematic review including 12 studies and 1438 participants in the journal Nutrients. In lupus, small studies are promising but inconclusive, suggested a study review in Frontiers in Immunology.
Watch patients taking turmeric and methotrexate closely, Funk said. Both have been associated with liver problems. Some users also experience GI symptoms like diarrhea because turmeric doesn’t absorb well in the GI tract.
Milk thistle (silymarin). This flowering plant is often marketed as a liver-supporting supplement, but research also suggests promise in RA and OA. A systematic review of 12 studies in Current Rheumatology Reviews suggested that silymarin supplements might help relieve pain, reduce inflammation, and protect the cartilage matrix, synovial membrane, and cartilage cells in joints. This supplement might help via immunomodulatory, anti-inflammatory, antioxidant, and anti-apoptotic properties, the researchers said. Doses of 250-750 mg appear to be safe. Side effects such as gastroenteritis, diarrhea, bloating, and headache can occur.
Boswellia serrata. Sourced from the resin of a tree that grows in dry, mountainous regions of Asia and Africa, Boswellia serrata can help relieve joint pain and stiffness and improve joint function in OA, suggested a systematic review of seven trials involving 545 patients in BMC Complementary Medicine and Therapies. Users saw benefits when taking 100-250 mg/d for 4 weeks or more. Compounds in Boswellia serrata may inhibit 5-lipoxygenase, an enzyme involved in producing inflammatory leukotrienes. No adverse events were reported. In some studies, users have reported GI side effects.
Ginger. Ginger is a popular herbal supplement among people with RA, Funk’s research suggested. One small clinical trial involving 70 patients with RA in the journal Gene showed that taking 1500 mg/d of ginger for 12 weeks improved their DAS and boosted their expression of FoxP3 genes, which are linked with the function of regulatory T cells. A meta-analysis including three studies with 330 patients taking ginger published in the journal Nutrients suggested ginger can reduce pain and systemic inflammation in people with OA. Preclinical studies suggested phenolic compounds in this spicy root, such as gingerols, reduce inflammation through multiple mechanisms.
Funk’s research revealed wide variation in the quality of ginger supplements, reinforcing the importance of selecting an independently verified product. Research suggested a safe dose is up to 2-2.5 g/kg body weight.
Resveratrol. Found in red grapes and red wine, this compound is particularly good at blocking COX-2 enzymes, an important step in the inflammatory cascade, Silva said. “Because of their chemical structure, they have great affinity to these enzymes to lead to their inhibition,” he said. A study review of five articles including 481 patients in The European Journal of Rheumatology showed that people with OA, RA, or Takayasu arteritis who took 250-1000 mg/d of resveratrol saw improvements in pain, function, disease activity, joint swelling, and inflammation, with no side effects.
Cinnamon. This warming spice is gaining popularity as a supplement, reported the American Botanical Council. Cinnamon is often marketed as lowering blood sugar and supporting bone health. In a small study of 36 women with RA published in The Journal of the American College of Nutrition, participants who consumed 2 g/d of cinnamon powder had reduced DASs along with reduced pain and tender and swollen joint counts. Cinnamon may reduce pain by inhibiting prostaglandin and blunt inflammation by reducing the release of arachidonic acid from cell membranes, according to a study review in Frontiers in Pharmacology. GI problems and allergic reactions are among the most common side effects.
Funk, Nikiphorou, Philippou, and Silva all had no relevant disclosures.
A version of this article first appeared on Medscape.com.
Many people with rheumatologic diseases try supplements for symptom relief. Here’s what you need to know about some common picks.
Dietary supplements were a $159 billion business in the United States in 2023, and many people with rheumatologic diseases are buying in. Research suggests more than 6 in 10 people with fibromyalgia, nearly 8 in 10 people with Sjögren’s disease, and more than 8 in 10 people with rheumatoid arthritis (RA) take dietary supplements.
Whatever the symptom — pain, swelling, or fatigue — you can probably find a supplement purporting to relieve it. But do these supplements work, and are they safe? A study review in RMD Open comprising 24 systematic reviews and 150 original articles suggests more high-quality research is needed on the effects of dietary supplements on rheumatologic diseases. Most studies have focused on RA or osteoarthritis (OA), where the evidence level is moderate at best.
“The studies in this space are usually not very high quality because there’s no money to support them, among other things, plus the products are disparate,” said Janet Funk, MD, MS, professor in the School of Nutritional Sciences and Wellness at the University of Arizona, Tucson. She recommended brushing up on supplements and finding out what patients are taking so you can offer advice and watch for drug-supplement interactions.
When asked for a medication list, many patients forget to report supplements, Funk said. “You have to prompt them specifically. I think some physicians have very negative views about supplements because so little data is known, and patients might pick up on that and decide not to report their use.” She recommended saying something like: “To give you the best possible care, I want to know everything you’re taking, including supplements. The things I’m prescribing could maybe interact with the things you’re taking, so I want to make sure I know about all of it so that together we can figure out if the combination of things is safe.”
The quality of dietary supplements varies, and they aren’t regulated like drugs by the Food and Drug Administration. Funk recommended selecting products verified by NSF or ConsumerLab. They test supplements to ensure the label reflects what’s inside.
This news organization scoured the literature and asked experts to weigh in on the evidence behind popular supplements in rheumatology today.
The Essential Nutrients
Vitamin supplements are a staple in many homes — but are they helpful? “Individual vitamin supplements will not provide any benefit unless the person is deficient in a specific vitamin or mineral,” according to Elena Philippou, PhD, RD, associate professor of nutrition-dietetics at the University of Nicosia in Cyprus, and Elena Nikiphorou, MBBS, a rheumatologist at King’s College London in England. For some patients, deficiency is a reality. A retrospective cohort study in The Journal of Clinical Medicine found that people with RA were 17% more likely than age-matched control individuals to have nutrient deficiencies, perhaps because symptoms like fatigue, pain, and nausea affect their eating habits. Here’s what the science says about common vitamin supplements.
Vitamin D. This hormone-like vitamin, which attaches to receptors on immune cells to tamp down inflammation, was the most popular dietary supplement among rheumatology patients in a recent study from the United Kingdom. Vitamin D deficiency is common in people with RA, lupus, Sjögren’s disease, ankylosing spondylitis, systemic sclerosis, and fibromyalgia. In some cases, vitamin D levels track with disease activity, research suggests. Corticosteroids can also make vitamin D deficiency more likely. Can supplements help?
In RA, evidence points to small improvements. A systematic review of 11 studies including 3049 patients published in Nutrition Reviews showed that vitamin D supplements significantly reduced patients’ pain and Disease Activity Score in 28 joints (DAS28) using both C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR).
The research is mixed on the benefits of vitamin D supplementation for fibromyalgia symptoms, according to a study review in SN Comprehensive Clinical Medicine that included two studies and 80 patients on supplementation. However, researchers said it’s still worth discussing the potential benefits of taking vitamin D.
“Vitamin D supplementation is important in the context of various rheumatic diseases to prevent or treat bone disease,” said Philippou and Nikiphorou. “People with rheumatic disease should speak to their healthcare provider and ask to check their blood vitamin D concentration.” The results can help you recommend a dose.
Folate. Patients on methotrexate should take folic acid supplements under the guidance of a healthcare provider, said Philippou and Nikiphorou. The reason: Methotrexate can deplete folic acid levels, increasing the risk for side effects. An analysis of adverse event reports published showed that methotrexate users who took folic acid (or tumor necrosis factor–alpha inhibitors) had a reduced risk for hepatotoxicity or myelosuppression. A commonly recommended dose is 1 mg/d.
Vitamin B12. In a 2024 perspective paper in Rheumatology International, researchers said physicians should assess vitamin B12 levels early in the diagnostic process of rheumatologic diseases. One reason: Many symptoms of pernicious anemia, like fatigue, mimic symptoms of rheumatologic diseases. The gastrointestinal (GI) effects of systemic sclerosis could bring on vitamin B12 deficiency. In a small study in The Journal of Clinical Rheumatology, 44 of 62 patients with systemic sclerosis had low vitamin B12 levels.
Vitamin E. Vitamin E deficiency is rare in healthy adults. However, some medical conditions, like inflammatory bowel disease and malabsorption disorders, can make vitamin E deficiency more likely. In RA, a vitamin E supplement could help reduce joint swelling and sensitivity, according to a systematic review of nine studies including 39,845 patients in The European Journal of Clinical Nutrition. Researchers credit the nutrient’s role in aiding intestinal repair. Use with caution, as this supplement can increase bleeding risk in doses over 1000 mg/d.
Vitamin A. Like vitamin E, vitamin A deficiency is rare in the United States. The risk of oversupplementing is higher than undersupplementing. However, vitamin A deficiency can happen in people with chronic pancreatic, liver, or GI problems. In people with deficiency, a vitamin A supplement can help relieve dry eye symptoms common in Sjögren’s disease, suggests a narrative review published in Nutrients. Vitamin A might help reduce ocular surface changes by supporting the production of proteins that protect the outermost surfaces of the eyes. The recommended daily allowance for vitamin A is 900 μg. High-dose supplements can cause toxicity, resulting in GI symptoms and problems like lethargy, drowsiness, increased intracranial pressure, and skin changes.
The Replacements
These substances are similar to naturally occurring compounds in our bodies. The question is whether ingesting them yields benefits.
Glucosamine and chondroitin. Glucosamine and chondroitin occur naturally in our bodies and help us form and protect connective tissues. In pill form, this combo is the most popular dietary supplement for OA, according to research in the journal Maturitas. But studies of its effectiveness yield mixed results. A systematic review of 25 studies published in Inflammopharmacology showed that, in patients with knee OA, supplementation with about 1500 mg of glucosamine per day reduced tibiofemoral joint space narrowing, while supplementation with about 800 mg/d of chondroitin reduced pain intensity and improved physical function, compared with placebo. The duo of glucosamine and chondroitin did not bring significant benefits, perhaps because more studies are needed. Most side effects were mild, but some literature points to the potential for glucosamine to increase warfarin’s blood-thinning effects.
Omega-3 fatty acids. Fish oil is a top-selling supplement, and it might be helpful in inflammatory rheumatologic diseases. A systematic review of 30 studies including 710 patients published in Arthritis Research & Therapy showed that omega-3 fatty acid supplements can improve pain, swollen and tender joint count, DAS28 scores, and Health Assessment Questionnaire scores in patients with RA, psoriatic arthritis, or ankylosing spondylitis. In patients with lupus, a study review that included five studies and 284 patients in The International Journal of Environmental Research and Public Health suggested omega-3 fatty acid supplements could improve ESR, CRP, disease activity, inflammatory markers, oxidative stress, lipid levels, and endothelial function.
Omega-3 fatty acids have anti-inflammatory effects that might explain their benefits. In patients with RA, for example, fish oil supplementation was associated with elevated blood levels of resolvins and protectins, which help quell inflammation, according to a study in Prostaglandins, Leukotrienes and Essential Fatty Acids.
Philippou and Nikiphorou recommended combining food and supplements: Eat oily fish at least twice a week, regularly consume plant-based sources of omega-3s — like chia seeds, flaxseeds, or walnuts — and consider a daily supplement that contains 2 g of omega-3s from docosahexaenoic acid and eicosapentaenoic acid. Most fish oil side effects are mild, like heartburn and bad breath. Fish oil can have blood-thinning effects at high doses, so special attention is needed for patients on anticoagulants.
Probiotics. Building up the good bacteria in your gut might help you fight the effects of rheumatologic diseases. A systematic review of 80 randomized controlled trials in BMC Medicine suggested that therapies targeting the gut microbiota might improve the symptoms or inflammatory factors in celiac disease, lupus, juvenile idiopathic arthritis, psoriasis, Sjögren’s disease, multiple sclerosis, systemic sclerosis, Crohn’s disease, and ulcerative colitis. Probiotics were also shown to relieve pain in fibromyalgia, but they didn’t affect scores on the Fibromyalgia Impact Questionnaire. Probiotics were not helpful in spondyloarthritis or RA. There were no adverse events. By improving the balance of bacteria in the gut, probiotics might inhibit pro-inflammatory factors and signaling pathways and regulate CD4+ T-cell differentiation, the researchers wrote.
Not all probiotic supplements are created equal. Effects can vary by microorganism and dose. Until more high-quality studies are published, Philippou and Nikiphorou recommend daily consumption of probiotic food sources such as yogurt, kefir, sauerkraut, kimchi, tempeh, miso, and kombucha, along with prebiotic food sources such as bananas, onion, artichokes, asparagus, oats, leeks, and garlic.
Collagen. An increasingly popular supplement for hair, skin, and nails, some collagen peptide or hydrolyzed collagen supplements come with claims about joint health, too. Inside our bodies, collagen helps build joints. As a supplement, the jury is still out. A systematic review of 19 studies in The International Journal of Rheumatic Diseases suggested more research is needed to determine whether collagen supplements are harmful or helpful in OA or RA. Studies haven’t shown adverse events, and doses typically range from 2.5 to 15 g/d.
Coenzyme Q10 (CoQ10). This antioxidant occurs naturally in our cells and is produced through microbial fermentation for use in dietary supplements. A study review of 20 articles including 483 patients in Clinical Nutrition ESPEN concluded that CoQ10 supplementation up to 300 mg/d was beneficial in RA, fibromyalgia, or antiphospholipid syndrome (APS).
In RA, CoQ10 supplementation improved disease activity index, ESR, and cytokine levels and decreased malondialdehyde. CoQ10 might protect against the overproduction of reactive oxygen species that can promote inflammation and joint damage, the researchers said. In fibromyalgia, CoQ10 was linked with improvements in pain, fatigue, sleep, tender points count, mood disorders, and scores on the Fibromyalgia Impact Questionnaire in most of the included studies. CoQ10 might help in fibromyalgia by improving mitochondrial dysfunction. In APS, CoQ10 improved endothelial function and decreased prothrombotic and pro-inflammatory mediators. CoQ10 might change the expression of genes that promote atherosclerosis. A few patients had GI side effects like nausea and diarrhea, but the supplements were generally well tolerated.
Melatonin. Commonly touted as a sleep aid, this hormone has immune and anti-inflammatory activities that could benefit people with rheumatologic diseases. A study review of 13 articles including 533 patients in Clinical Nutrition ESPEN concluded that melatonin can help improve sleep, pain, and mood in fibromyalgia, OA, and osteoporosis but not in RA. Side effects were minimal, but a few people experienced nausea, drowsiness, nightmares, or headaches. Doses of 5-6 mg/d are likely safe for most adults.
The Plant-Derived Antioxidants
Many supplements used in rheumatology are antioxidants derived from herbs, spices, or other plants. When plants encounter stressors, like temperature changes or hungry insects, their secondary metabolism revs up and creates compounds with biological properties. Some of these substances influence inflammatory pathways in the human body, said Luís Silva, PhD, a medicinal chemistry researcher at the Polytechnic Institute of Guarda in Portugal. “If it is possible to reduce these kinds of anti-inflammatory processes, it is also possible that we could help people with inflammatory diseases to a good life, or a better life.”
Turmeric and curcumin. You might see this supplement labeled as turmeric, a golden spice in curry powder, or curcumin, an antioxidant compound known as a curcuminoid in turmeric. Curcuminoids might reduce inflammation by scavenging free radicals and inhibiting enzymes that make prostaglandins, Silva said.
Turmeric is the most popular herbal supplement for people with RA, according to Funk’s research. A study review of six publications including 539 patients in Frontiers in Immunology showed that curcumin supplements improved RA patients’ ESR, DAS, swollen joint count, and tender joint count. Turmeric could help patients with OA, too. Patients with OA who took 1000 mg/d of curcumin improved their pain and function, according to a systematic review including 12 studies and 1438 participants in the journal Nutrients. In lupus, small studies are promising but inconclusive, suggested a study review in Frontiers in Immunology.
Watch patients taking turmeric and methotrexate closely, Funk said. Both have been associated with liver problems. Some users also experience GI symptoms like diarrhea because turmeric doesn’t absorb well in the GI tract.
Milk thistle (silymarin). This flowering plant is often marketed as a liver-supporting supplement, but research also suggests promise in RA and OA. A systematic review of 12 studies in Current Rheumatology Reviews suggested that silymarin supplements might help relieve pain, reduce inflammation, and protect the cartilage matrix, synovial membrane, and cartilage cells in joints. This supplement might help via immunomodulatory, anti-inflammatory, antioxidant, and anti-apoptotic properties, the researchers said. Doses of 250-750 mg appear to be safe. Side effects such as gastroenteritis, diarrhea, bloating, and headache can occur.
Boswellia serrata. Sourced from the resin of a tree that grows in dry, mountainous regions of Asia and Africa, Boswellia serrata can help relieve joint pain and stiffness and improve joint function in OA, suggested a systematic review of seven trials involving 545 patients in BMC Complementary Medicine and Therapies. Users saw benefits when taking 100-250 mg/d for 4 weeks or more. Compounds in Boswellia serrata may inhibit 5-lipoxygenase, an enzyme involved in producing inflammatory leukotrienes. No adverse events were reported. In some studies, users have reported GI side effects.
Ginger. Ginger is a popular herbal supplement among people with RA, Funk’s research suggested. One small clinical trial involving 70 patients with RA in the journal Gene showed that taking 1500 mg/d of ginger for 12 weeks improved their DAS and boosted their expression of FoxP3 genes, which are linked with the function of regulatory T cells. A meta-analysis including three studies with 330 patients taking ginger published in the journal Nutrients suggested ginger can reduce pain and systemic inflammation in people with OA. Preclinical studies suggested phenolic compounds in this spicy root, such as gingerols, reduce inflammation through multiple mechanisms.
Funk’s research revealed wide variation in the quality of ginger supplements, reinforcing the importance of selecting an independently verified product. Research suggested a safe dose is up to 2-2.5 g/kg body weight.
Resveratrol. Found in red grapes and red wine, this compound is particularly good at blocking COX-2 enzymes, an important step in the inflammatory cascade, Silva said. “Because of their chemical structure, they have great affinity to these enzymes to lead to their inhibition,” he said. A study review of five articles including 481 patients in The European Journal of Rheumatology showed that people with OA, RA, or Takayasu arteritis who took 250-1000 mg/d of resveratrol saw improvements in pain, function, disease activity, joint swelling, and inflammation, with no side effects.
Cinnamon. This warming spice is gaining popularity as a supplement, reported the American Botanical Council. Cinnamon is often marketed as lowering blood sugar and supporting bone health. In a small study of 36 women with RA published in The Journal of the American College of Nutrition, participants who consumed 2 g/d of cinnamon powder had reduced DASs along with reduced pain and tender and swollen joint counts. Cinnamon may reduce pain by inhibiting prostaglandin and blunt inflammation by reducing the release of arachidonic acid from cell membranes, according to a study review in Frontiers in Pharmacology. GI problems and allergic reactions are among the most common side effects.
Funk, Nikiphorou, Philippou, and Silva all had no relevant disclosures.
A version of this article first appeared on Medscape.com.
JIA Treatment Has Increasingly Involved New DMARDs Since 2001
TOPLINE:
The use of newer biologic or targeted synthetic disease-modifying antirheumatic drugs (b/tsDMARDs) for treating juvenile idiopathic arthritis (JIA) rose sharply from 2001 to 2022, while the use of conventional synthetic DMARDs (csDMARDs) plummeted, with adalimumab becoming the most commonly used b/tsDMARD.
METHODOLOGY:
- Researchers performed a serial cross-sectional study using Merative MarketScan Commercial Claims and Encounters data from 2000 to 2022 to describe recent trends in DMARD use for children with JIA in the United States.
- They identified 20,258 new episodes of DMARD use among 13,696 children with JIA (median age, 14 years; 67.5% girls) who newly initiated at least one DMARD.
- Participants were required to have ≥ 365 days of continuous healthcare and pharmacy eligibility prior to the index date, defined as the date of DMARD initiation.
TAKEAWAY:
- The use of csDMARDs declined from 89.5% to 43.2% between 2001 and 2022 (P < .001 for trend), whereas the use of bDMARDs increased from 10.5% to 50.0% over the same period (P < .001).
- Methotrexate was the most commonly used DMARD throughout the study period ; however, as with other csDMARDs, its use declined from 42.1% in 2001 to 21.5% in 2022 (P < .001 ).
- Use of the tumor necrosis factor inhibitor adalimumab doubled from 7% in 2007 to 14% in 2008 and increased further up to 20.5% by 2022; adalimumab also became the most predominantly used b/tsDMARD after csDMARD monotherapy, accounting for 77.8% of prescriptions following csDMARDs in 2022.
- Even though the use of individual TNF inhibitors increased, their overall popularity fell in recent years as the use of newer b/tsDMARDs, such as ustekinumab and secukinumab, increased.
IN PRACTICE:
“These real-world treatment patterns give us insight into how selection of therapies for JIA has evolved with increasing availability of effective agents and help prepare for future studies on comparative DMARD safety and effectiveness,” the authors wrote.
SOURCE:
The study was led by Priyanka Yalamanchili, PharmD, MS, Center for Pharmacoepidemiology and Treatment Science, Institute for Health, Rutgers University, New Brunswick, New Jersey, and was published online October 22, 2024, in Arthritis & Rheumatology.
LIMITATIONS:
The dependence on commercial claims data may have limited the generalizability of the findings to other populations, such as those with public insurance or without insurance. The study did not have access to demographic data of the participants to investigate the presence of disparities in the use of DMARDs. Moreover, the lack of clinical details about the patients with JIA, including disease severity and specialty of prescribers, may have affected the interpretation of the results.
DISCLOSURES:
The study was supported by funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and several other institutes of the National Institutes of Health, as well as the Rheumatology Research Foundation and the Juvenile Diabetes Research Foundation. No conflicts of interest were reported by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The use of newer biologic or targeted synthetic disease-modifying antirheumatic drugs (b/tsDMARDs) for treating juvenile idiopathic arthritis (JIA) rose sharply from 2001 to 2022, while the use of conventional synthetic DMARDs (csDMARDs) plummeted, with adalimumab becoming the most commonly used b/tsDMARD.
METHODOLOGY:
- Researchers performed a serial cross-sectional study using Merative MarketScan Commercial Claims and Encounters data from 2000 to 2022 to describe recent trends in DMARD use for children with JIA in the United States.
- They identified 20,258 new episodes of DMARD use among 13,696 children with JIA (median age, 14 years; 67.5% girls) who newly initiated at least one DMARD.
- Participants were required to have ≥ 365 days of continuous healthcare and pharmacy eligibility prior to the index date, defined as the date of DMARD initiation.
TAKEAWAY:
- The use of csDMARDs declined from 89.5% to 43.2% between 2001 and 2022 (P < .001 for trend), whereas the use of bDMARDs increased from 10.5% to 50.0% over the same period (P < .001).
- Methotrexate was the most commonly used DMARD throughout the study period ; however, as with other csDMARDs, its use declined from 42.1% in 2001 to 21.5% in 2022 (P < .001 ).
- Use of the tumor necrosis factor inhibitor adalimumab doubled from 7% in 2007 to 14% in 2008 and increased further up to 20.5% by 2022; adalimumab also became the most predominantly used b/tsDMARD after csDMARD monotherapy, accounting for 77.8% of prescriptions following csDMARDs in 2022.
- Even though the use of individual TNF inhibitors increased, their overall popularity fell in recent years as the use of newer b/tsDMARDs, such as ustekinumab and secukinumab, increased.
IN PRACTICE:
“These real-world treatment patterns give us insight into how selection of therapies for JIA has evolved with increasing availability of effective agents and help prepare for future studies on comparative DMARD safety and effectiveness,” the authors wrote.
SOURCE:
The study was led by Priyanka Yalamanchili, PharmD, MS, Center for Pharmacoepidemiology and Treatment Science, Institute for Health, Rutgers University, New Brunswick, New Jersey, and was published online October 22, 2024, in Arthritis & Rheumatology.
LIMITATIONS:
The dependence on commercial claims data may have limited the generalizability of the findings to other populations, such as those with public insurance or without insurance. The study did not have access to demographic data of the participants to investigate the presence of disparities in the use of DMARDs. Moreover, the lack of clinical details about the patients with JIA, including disease severity and specialty of prescribers, may have affected the interpretation of the results.
DISCLOSURES:
The study was supported by funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and several other institutes of the National Institutes of Health, as well as the Rheumatology Research Foundation and the Juvenile Diabetes Research Foundation. No conflicts of interest were reported by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The use of newer biologic or targeted synthetic disease-modifying antirheumatic drugs (b/tsDMARDs) for treating juvenile idiopathic arthritis (JIA) rose sharply from 2001 to 2022, while the use of conventional synthetic DMARDs (csDMARDs) plummeted, with adalimumab becoming the most commonly used b/tsDMARD.
METHODOLOGY:
- Researchers performed a serial cross-sectional study using Merative MarketScan Commercial Claims and Encounters data from 2000 to 2022 to describe recent trends in DMARD use for children with JIA in the United States.
- They identified 20,258 new episodes of DMARD use among 13,696 children with JIA (median age, 14 years; 67.5% girls) who newly initiated at least one DMARD.
- Participants were required to have ≥ 365 days of continuous healthcare and pharmacy eligibility prior to the index date, defined as the date of DMARD initiation.
TAKEAWAY:
- The use of csDMARDs declined from 89.5% to 43.2% between 2001 and 2022 (P < .001 for trend), whereas the use of bDMARDs increased from 10.5% to 50.0% over the same period (P < .001).
- Methotrexate was the most commonly used DMARD throughout the study period ; however, as with other csDMARDs, its use declined from 42.1% in 2001 to 21.5% in 2022 (P < .001 ).
- Use of the tumor necrosis factor inhibitor adalimumab doubled from 7% in 2007 to 14% in 2008 and increased further up to 20.5% by 2022; adalimumab also became the most predominantly used b/tsDMARD after csDMARD monotherapy, accounting for 77.8% of prescriptions following csDMARDs in 2022.
- Even though the use of individual TNF inhibitors increased, their overall popularity fell in recent years as the use of newer b/tsDMARDs, such as ustekinumab and secukinumab, increased.
IN PRACTICE:
“These real-world treatment patterns give us insight into how selection of therapies for JIA has evolved with increasing availability of effective agents and help prepare for future studies on comparative DMARD safety and effectiveness,” the authors wrote.
SOURCE:
The study was led by Priyanka Yalamanchili, PharmD, MS, Center for Pharmacoepidemiology and Treatment Science, Institute for Health, Rutgers University, New Brunswick, New Jersey, and was published online October 22, 2024, in Arthritis & Rheumatology.
LIMITATIONS:
The dependence on commercial claims data may have limited the generalizability of the findings to other populations, such as those with public insurance or without insurance. The study did not have access to demographic data of the participants to investigate the presence of disparities in the use of DMARDs. Moreover, the lack of clinical details about the patients with JIA, including disease severity and specialty of prescribers, may have affected the interpretation of the results.
DISCLOSURES:
The study was supported by funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and several other institutes of the National Institutes of Health, as well as the Rheumatology Research Foundation and the Juvenile Diabetes Research Foundation. No conflicts of interest were reported by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
No Benefit to High-Dose IV Vs Oral Steroids in Giant Cell Arteritis
TOPLINE:
In patients with giant cell arteritis (GCA), intravenous methylprednisolone compared with oral glucocorticoids alone does not improve visual acuity and increases the risk for diabetes within the first year. Survival rates do not differ with these two treatments.
METHODOLOGY:
- Researchers conducted a population-based retrospective study at three centers in Sweden to assess the clinical characteristics, treatment-related toxicity, and mortality in patients with GCA who were receiving high-dose intravenous methylprednisolone.
- A total of 419 patients with biopsy-confirmed GCA (mean age at diagnosis, 75 years; 69% women) diagnosed from 2004 to 2019 were included.
- Patients were treated with either intravenous methylprednisolone (n = 111) at a dose of 500-1000 mg per day for 3 consecutive days or oral glucocorticoids alone (n = 308).
- Ischemic visual complications considered to indicate visual involvement were confirmed by an ophthalmologist, and data on visual acuity were collected from ophthalmologic clinic records at initial consultations and follow-up at 3-18 months.
TAKEAWAY:
- Despite a tendency toward improvement, no significant difference in visual acuity was observed with intravenous methylprednisolone compared with oral glucocorticoids.
- Patients treated with intravenous methylprednisolone had a higher risk for newly diagnosed diabetes within a year of GCA diagnosis (odds ratio [OR], 2.59; P = .01).
- The risk for diabetes remained elevated even after adjustment for the cumulative oral glucocorticoid dose at 3 months (adjusted OR, 3.30; P = .01).
- Survival rates did not significantly differ between the treatment groups over a mean follow-up of 6.6 years.
IN PRACTICE:
“In this study on the use of intravenous methylprednisolone treatment in GCA, we found no evidence of a beneficial effect in improving visual acuity or enabling more rapid tapering of the oral glucocorticoid dose,” the authors wrote. “The use of IVMP [intravenous methylprednisolone] was associated with an increased risk of diabetes during the first year compared with oral GC [glucocorticoid], raising questions about the value of IVMP in GCA treatment.”
SOURCE:
The study, led by Hampus Henningson, Department of Clinical Sciences, Rheumatology, Lund University, Lund, Sweden, was published online in Rheumatology.
LIMITATIONS:
The retrospective nature of the study may have resulted in missing data and difficulty in accurately quantifying the cumulative glucocorticoid doses. The study did not validate the diagnoses of comorbidities but relied solely on diagnostic codes.
DISCLOSURES:
This study was supported by the Swedish Research Council, Swedish Rheumatism Association, Swedish Medical Society, Alfred Österlund’s Foundation, and King Gustaf V’s 80-year foundation. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
In patients with giant cell arteritis (GCA), intravenous methylprednisolone compared with oral glucocorticoids alone does not improve visual acuity and increases the risk for diabetes within the first year. Survival rates do not differ with these two treatments.
METHODOLOGY:
- Researchers conducted a population-based retrospective study at three centers in Sweden to assess the clinical characteristics, treatment-related toxicity, and mortality in patients with GCA who were receiving high-dose intravenous methylprednisolone.
- A total of 419 patients with biopsy-confirmed GCA (mean age at diagnosis, 75 years; 69% women) diagnosed from 2004 to 2019 were included.
- Patients were treated with either intravenous methylprednisolone (n = 111) at a dose of 500-1000 mg per day for 3 consecutive days or oral glucocorticoids alone (n = 308).
- Ischemic visual complications considered to indicate visual involvement were confirmed by an ophthalmologist, and data on visual acuity were collected from ophthalmologic clinic records at initial consultations and follow-up at 3-18 months.
TAKEAWAY:
- Despite a tendency toward improvement, no significant difference in visual acuity was observed with intravenous methylprednisolone compared with oral glucocorticoids.
- Patients treated with intravenous methylprednisolone had a higher risk for newly diagnosed diabetes within a year of GCA diagnosis (odds ratio [OR], 2.59; P = .01).
- The risk for diabetes remained elevated even after adjustment for the cumulative oral glucocorticoid dose at 3 months (adjusted OR, 3.30; P = .01).
- Survival rates did not significantly differ between the treatment groups over a mean follow-up of 6.6 years.
IN PRACTICE:
“In this study on the use of intravenous methylprednisolone treatment in GCA, we found no evidence of a beneficial effect in improving visual acuity or enabling more rapid tapering of the oral glucocorticoid dose,” the authors wrote. “The use of IVMP [intravenous methylprednisolone] was associated with an increased risk of diabetes during the first year compared with oral GC [glucocorticoid], raising questions about the value of IVMP in GCA treatment.”
SOURCE:
The study, led by Hampus Henningson, Department of Clinical Sciences, Rheumatology, Lund University, Lund, Sweden, was published online in Rheumatology.
LIMITATIONS:
The retrospective nature of the study may have resulted in missing data and difficulty in accurately quantifying the cumulative glucocorticoid doses. The study did not validate the diagnoses of comorbidities but relied solely on diagnostic codes.
DISCLOSURES:
This study was supported by the Swedish Research Council, Swedish Rheumatism Association, Swedish Medical Society, Alfred Österlund’s Foundation, and King Gustaf V’s 80-year foundation. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
In patients with giant cell arteritis (GCA), intravenous methylprednisolone compared with oral glucocorticoids alone does not improve visual acuity and increases the risk for diabetes within the first year. Survival rates do not differ with these two treatments.
METHODOLOGY:
- Researchers conducted a population-based retrospective study at three centers in Sweden to assess the clinical characteristics, treatment-related toxicity, and mortality in patients with GCA who were receiving high-dose intravenous methylprednisolone.
- A total of 419 patients with biopsy-confirmed GCA (mean age at diagnosis, 75 years; 69% women) diagnosed from 2004 to 2019 were included.
- Patients were treated with either intravenous methylprednisolone (n = 111) at a dose of 500-1000 mg per day for 3 consecutive days or oral glucocorticoids alone (n = 308).
- Ischemic visual complications considered to indicate visual involvement were confirmed by an ophthalmologist, and data on visual acuity were collected from ophthalmologic clinic records at initial consultations and follow-up at 3-18 months.
TAKEAWAY:
- Despite a tendency toward improvement, no significant difference in visual acuity was observed with intravenous methylprednisolone compared with oral glucocorticoids.
- Patients treated with intravenous methylprednisolone had a higher risk for newly diagnosed diabetes within a year of GCA diagnosis (odds ratio [OR], 2.59; P = .01).
- The risk for diabetes remained elevated even after adjustment for the cumulative oral glucocorticoid dose at 3 months (adjusted OR, 3.30; P = .01).
- Survival rates did not significantly differ between the treatment groups over a mean follow-up of 6.6 years.
IN PRACTICE:
“In this study on the use of intravenous methylprednisolone treatment in GCA, we found no evidence of a beneficial effect in improving visual acuity or enabling more rapid tapering of the oral glucocorticoid dose,” the authors wrote. “The use of IVMP [intravenous methylprednisolone] was associated with an increased risk of diabetes during the first year compared with oral GC [glucocorticoid], raising questions about the value of IVMP in GCA treatment.”
SOURCE:
The study, led by Hampus Henningson, Department of Clinical Sciences, Rheumatology, Lund University, Lund, Sweden, was published online in Rheumatology.
LIMITATIONS:
The retrospective nature of the study may have resulted in missing data and difficulty in accurately quantifying the cumulative glucocorticoid doses. The study did not validate the diagnoses of comorbidities but relied solely on diagnostic codes.
DISCLOSURES:
This study was supported by the Swedish Research Council, Swedish Rheumatism Association, Swedish Medical Society, Alfred Österlund’s Foundation, and King Gustaf V’s 80-year foundation. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Minor Progress in Gender Pay Equity, But a Big Gap Persists
Despite some recent progress in compensation equity, women in medicine continue to be paid significantly lower salaries than men.
According to the Female Compensation Report 2024 by Medscape, male doctors of any kind earned an average salary of about $400,000, whereas female doctors earned approximately $309,000 — a 29% gap.
The report analyzed survey data from 7000 practicing physicians who were recruited over a 4-month period starting in October 2023. The respondents comprised roughly 60% women representing over 29 specialties.
In the 2022 report, the pay gap between the genders was 32%. But some women in the field argued substantial headway is still needed.
“You can try and pick apart the data, but I’d say we’re not really making progress,” said Susan T. Hingle, MD, an internist in Illinois and president of the American Medical Women’s Association. “A decline by a couple of percentage points is not significantly addressing this pay gap that over a lifetime is huge, can be millions of dollars.”
The gender gap was narrower among female primary care physicians (PCPs) vs medical specialists. Female PCPs earned around $253,000 per year, whereas male PCPs earned about $295,000 per year. Hingle suggested that female PCPs may enjoy more pay equity because health systems have a harder time filling these positions.
On the other hand, the gap for specialists rose from 27% in 2022 to 31% in 2023. Differences in how aggressively women and men negotiate compensation packages may play a role, said Hingle.
“Taking negotiation out of the equation would be progress to me,” said Hingle.
Pay disparity did not appear to be the result of time spent on the job — female doctors reported an average of 49 work hours per week, whereas their male counterparts reported 50 work hours per week.
Meanwhile, the pay gap progressively worsened over time. Among doctors aged 28-34 years, men earned an average of $53,000 more than women. By ages 46-49, men earned an average of $157,000 more than women.
“I had to take my employer to court to get equal compensation, sad as it is to say,” said a hospitalist in North Carolina.
Nearly 60% of women surveyed felt they were not being paid fairly for their efforts, up from less than half reported in Medscape’s 2021 report. Hingle said that this figure may not only reflect sentiments about the compensation gap, but also less support on the job, including fewer physician assistants (PAs), nurses, and administrative staff.
“At my job, I do the work of multiple people,” said a survey respondent. “Junior resident, senior resident, social worker, nurse practitioner, PA — as well as try to be a teacher, researcher, [and] an excellent doctor and have the time to make patients feel as if they are not in a rush.”
Roughly 30% of women physicians said they would not choose to go into medicine again if given the chance compared with 26% of male physicians.
“Gender inequities in our profession have a direct impact,” said Shikha Jain, MD, an oncologist in Chicago and founder of the Women in Medicine nonprofit. “I think women in general don’t feel valued in the care they’re providing.”
Jain cited bullying, harassment, and fewer opportunities for leadership and recognition as factors beyond pay that affect female physicians’ feelings of being valued.
A version of this article first appeared on Medscape.com.
Despite some recent progress in compensation equity, women in medicine continue to be paid significantly lower salaries than men.
According to the Female Compensation Report 2024 by Medscape, male doctors of any kind earned an average salary of about $400,000, whereas female doctors earned approximately $309,000 — a 29% gap.
The report analyzed survey data from 7000 practicing physicians who were recruited over a 4-month period starting in October 2023. The respondents comprised roughly 60% women representing over 29 specialties.
In the 2022 report, the pay gap between the genders was 32%. But some women in the field argued substantial headway is still needed.
“You can try and pick apart the data, but I’d say we’re not really making progress,” said Susan T. Hingle, MD, an internist in Illinois and president of the American Medical Women’s Association. “A decline by a couple of percentage points is not significantly addressing this pay gap that over a lifetime is huge, can be millions of dollars.”
The gender gap was narrower among female primary care physicians (PCPs) vs medical specialists. Female PCPs earned around $253,000 per year, whereas male PCPs earned about $295,000 per year. Hingle suggested that female PCPs may enjoy more pay equity because health systems have a harder time filling these positions.
On the other hand, the gap for specialists rose from 27% in 2022 to 31% in 2023. Differences in how aggressively women and men negotiate compensation packages may play a role, said Hingle.
“Taking negotiation out of the equation would be progress to me,” said Hingle.
Pay disparity did not appear to be the result of time spent on the job — female doctors reported an average of 49 work hours per week, whereas their male counterparts reported 50 work hours per week.
Meanwhile, the pay gap progressively worsened over time. Among doctors aged 28-34 years, men earned an average of $53,000 more than women. By ages 46-49, men earned an average of $157,000 more than women.
“I had to take my employer to court to get equal compensation, sad as it is to say,” said a hospitalist in North Carolina.
Nearly 60% of women surveyed felt they were not being paid fairly for their efforts, up from less than half reported in Medscape’s 2021 report. Hingle said that this figure may not only reflect sentiments about the compensation gap, but also less support on the job, including fewer physician assistants (PAs), nurses, and administrative staff.
“At my job, I do the work of multiple people,” said a survey respondent. “Junior resident, senior resident, social worker, nurse practitioner, PA — as well as try to be a teacher, researcher, [and] an excellent doctor and have the time to make patients feel as if they are not in a rush.”
Roughly 30% of women physicians said they would not choose to go into medicine again if given the chance compared with 26% of male physicians.
“Gender inequities in our profession have a direct impact,” said Shikha Jain, MD, an oncologist in Chicago and founder of the Women in Medicine nonprofit. “I think women in general don’t feel valued in the care they’re providing.”
Jain cited bullying, harassment, and fewer opportunities for leadership and recognition as factors beyond pay that affect female physicians’ feelings of being valued.
A version of this article first appeared on Medscape.com.
Despite some recent progress in compensation equity, women in medicine continue to be paid significantly lower salaries than men.
According to the Female Compensation Report 2024 by Medscape, male doctors of any kind earned an average salary of about $400,000, whereas female doctors earned approximately $309,000 — a 29% gap.
The report analyzed survey data from 7000 practicing physicians who were recruited over a 4-month period starting in October 2023. The respondents comprised roughly 60% women representing over 29 specialties.
In the 2022 report, the pay gap between the genders was 32%. But some women in the field argued substantial headway is still needed.
“You can try and pick apart the data, but I’d say we’re not really making progress,” said Susan T. Hingle, MD, an internist in Illinois and president of the American Medical Women’s Association. “A decline by a couple of percentage points is not significantly addressing this pay gap that over a lifetime is huge, can be millions of dollars.”
The gender gap was narrower among female primary care physicians (PCPs) vs medical specialists. Female PCPs earned around $253,000 per year, whereas male PCPs earned about $295,000 per year. Hingle suggested that female PCPs may enjoy more pay equity because health systems have a harder time filling these positions.
On the other hand, the gap for specialists rose from 27% in 2022 to 31% in 2023. Differences in how aggressively women and men negotiate compensation packages may play a role, said Hingle.
“Taking negotiation out of the equation would be progress to me,” said Hingle.
Pay disparity did not appear to be the result of time spent on the job — female doctors reported an average of 49 work hours per week, whereas their male counterparts reported 50 work hours per week.
Meanwhile, the pay gap progressively worsened over time. Among doctors aged 28-34 years, men earned an average of $53,000 more than women. By ages 46-49, men earned an average of $157,000 more than women.
“I had to take my employer to court to get equal compensation, sad as it is to say,” said a hospitalist in North Carolina.
Nearly 60% of women surveyed felt they were not being paid fairly for their efforts, up from less than half reported in Medscape’s 2021 report. Hingle said that this figure may not only reflect sentiments about the compensation gap, but also less support on the job, including fewer physician assistants (PAs), nurses, and administrative staff.
“At my job, I do the work of multiple people,” said a survey respondent. “Junior resident, senior resident, social worker, nurse practitioner, PA — as well as try to be a teacher, researcher, [and] an excellent doctor and have the time to make patients feel as if they are not in a rush.”
Roughly 30% of women physicians said they would not choose to go into medicine again if given the chance compared with 26% of male physicians.
“Gender inequities in our profession have a direct impact,” said Shikha Jain, MD, an oncologist in Chicago and founder of the Women in Medicine nonprofit. “I think women in general don’t feel valued in the care they’re providing.”
Jain cited bullying, harassment, and fewer opportunities for leadership and recognition as factors beyond pay that affect female physicians’ feelings of being valued.
A version of this article first appeared on Medscape.com.
Cannabis Often Used as a Substitute for Traditional Medications
Nearly two thirds of patients with rheumatic conditions switched to medical cannabis from medications such as nonsteroidal anti-inflammatory drugs (NSAIDs) and opioids, with the substitution being associated with greater self-reported improvement in symptoms than nonsubstitution.
METHODOLOGY:
- Researchers conducted a secondary analysis of a cross-sectional survey to investigate the prevalence of switching to medical cannabis from traditional medications in patients with rheumatic conditions from the United States and Canada.
- The survey included questions on current and past medical cannabis use, sociodemographic characteristics, medication taken and substituted, substance use, and patient-reported outcomes.
- Of the 1727 patients who completed the survey, 763 patients (mean age, 59 years; 84.1% women) reported current use of cannabis and were included in this analysis.
- Participants were asked if they had substituted any medications with medical cannabis and were sub-grouped accordingly.
- They also reported any changes in symptoms after initiating cannabis, the current and anticipated duration of medical cannabis use, methods of ingestion, cannabinoid content, and frequency of use.
TAKEAWAY:
- Overall, 62.5% reported substituting medical cannabis for certain medications, including NSAIDs (54.7%), opioids (48.6%), sleep aids (29.6%), muscle relaxants (25.2%), benzodiazepines (15.5%), and gabapentinoids (10.5%).
- The most common reasons given for substituting medical cannabis were fewer side effects (39%), better symptom control (27%), and fewer adverse effects (12%).
- Participants who substituted medical cannabis reported significant improvements in symptoms such as pain, sleep, joint stiffness, muscle spasms, and inflammation, and in overall health, compared with those who did not substitute it for medications.
- The substitution group was more likely to use inhalation methods (smoking and vaporizing) than the nonsubstitution group; they also used medical cannabis more frequently and preferred products containing delta-9-tetrahydrocannabinol.
IN PRACTICE:
“The changing legal status of cannabis has allowed a greater openness with more people willing to try cannabis for symptom relief. These encouraging results of medication reduction and favorable effect of [medical cannabis] require confirmation with more rigorous methods. At this time, survey information may be seen as a signal for effect, rather than sound evidence that could be applicable to those with musculoskeletal complaints in general,” the authors wrote.
SOURCE:
The study was led by Kevin F. Boehnke, PhD, University of Michigan Medical School, Ann Arbor, and was published online in ACR Open Rheumatology.
LIMITATIONS:
The cross-sectional nature of the study limited the determination of causality between medical cannabis use and symptom improvement. Moreover, the anonymous and self-reported nature of the survey at a single timepoint may have introduced recall bias. The sample predominantly consisted of older, White females, which may have limited the generalizability of the findings to other demographic groups.
DISCLOSURES:
Some authors received grant support from the National Institute on Drug Abuse and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Some others received payments, honoraria, grant funding, consulting fees, and travel support, and reported other ties with pharmaceutical companies and other institutions.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Nearly two thirds of patients with rheumatic conditions switched to medical cannabis from medications such as nonsteroidal anti-inflammatory drugs (NSAIDs) and opioids, with the substitution being associated with greater self-reported improvement in symptoms than nonsubstitution.
METHODOLOGY:
- Researchers conducted a secondary analysis of a cross-sectional survey to investigate the prevalence of switching to medical cannabis from traditional medications in patients with rheumatic conditions from the United States and Canada.
- The survey included questions on current and past medical cannabis use, sociodemographic characteristics, medication taken and substituted, substance use, and patient-reported outcomes.
- Of the 1727 patients who completed the survey, 763 patients (mean age, 59 years; 84.1% women) reported current use of cannabis and were included in this analysis.
- Participants were asked if they had substituted any medications with medical cannabis and were sub-grouped accordingly.
- They also reported any changes in symptoms after initiating cannabis, the current and anticipated duration of medical cannabis use, methods of ingestion, cannabinoid content, and frequency of use.
TAKEAWAY:
- Overall, 62.5% reported substituting medical cannabis for certain medications, including NSAIDs (54.7%), opioids (48.6%), sleep aids (29.6%), muscle relaxants (25.2%), benzodiazepines (15.5%), and gabapentinoids (10.5%).
- The most common reasons given for substituting medical cannabis were fewer side effects (39%), better symptom control (27%), and fewer adverse effects (12%).
- Participants who substituted medical cannabis reported significant improvements in symptoms such as pain, sleep, joint stiffness, muscle spasms, and inflammation, and in overall health, compared with those who did not substitute it for medications.
- The substitution group was more likely to use inhalation methods (smoking and vaporizing) than the nonsubstitution group; they also used medical cannabis more frequently and preferred products containing delta-9-tetrahydrocannabinol.
IN PRACTICE:
“The changing legal status of cannabis has allowed a greater openness with more people willing to try cannabis for symptom relief. These encouraging results of medication reduction and favorable effect of [medical cannabis] require confirmation with more rigorous methods. At this time, survey information may be seen as a signal for effect, rather than sound evidence that could be applicable to those with musculoskeletal complaints in general,” the authors wrote.
SOURCE:
The study was led by Kevin F. Boehnke, PhD, University of Michigan Medical School, Ann Arbor, and was published online in ACR Open Rheumatology.
LIMITATIONS:
The cross-sectional nature of the study limited the determination of causality between medical cannabis use and symptom improvement. Moreover, the anonymous and self-reported nature of the survey at a single timepoint may have introduced recall bias. The sample predominantly consisted of older, White females, which may have limited the generalizability of the findings to other demographic groups.
DISCLOSURES:
Some authors received grant support from the National Institute on Drug Abuse and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Some others received payments, honoraria, grant funding, consulting fees, and travel support, and reported other ties with pharmaceutical companies and other institutions.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Nearly two thirds of patients with rheumatic conditions switched to medical cannabis from medications such as nonsteroidal anti-inflammatory drugs (NSAIDs) and opioids, with the substitution being associated with greater self-reported improvement in symptoms than nonsubstitution.
METHODOLOGY:
- Researchers conducted a secondary analysis of a cross-sectional survey to investigate the prevalence of switching to medical cannabis from traditional medications in patients with rheumatic conditions from the United States and Canada.
- The survey included questions on current and past medical cannabis use, sociodemographic characteristics, medication taken and substituted, substance use, and patient-reported outcomes.
- Of the 1727 patients who completed the survey, 763 patients (mean age, 59 years; 84.1% women) reported current use of cannabis and were included in this analysis.
- Participants were asked if they had substituted any medications with medical cannabis and were sub-grouped accordingly.
- They also reported any changes in symptoms after initiating cannabis, the current and anticipated duration of medical cannabis use, methods of ingestion, cannabinoid content, and frequency of use.
TAKEAWAY:
- Overall, 62.5% reported substituting medical cannabis for certain medications, including NSAIDs (54.7%), opioids (48.6%), sleep aids (29.6%), muscle relaxants (25.2%), benzodiazepines (15.5%), and gabapentinoids (10.5%).
- The most common reasons given for substituting medical cannabis were fewer side effects (39%), better symptom control (27%), and fewer adverse effects (12%).
- Participants who substituted medical cannabis reported significant improvements in symptoms such as pain, sleep, joint stiffness, muscle spasms, and inflammation, and in overall health, compared with those who did not substitute it for medications.
- The substitution group was more likely to use inhalation methods (smoking and vaporizing) than the nonsubstitution group; they also used medical cannabis more frequently and preferred products containing delta-9-tetrahydrocannabinol.
IN PRACTICE:
“The changing legal status of cannabis has allowed a greater openness with more people willing to try cannabis for symptom relief. These encouraging results of medication reduction and favorable effect of [medical cannabis] require confirmation with more rigorous methods. At this time, survey information may be seen as a signal for effect, rather than sound evidence that could be applicable to those with musculoskeletal complaints in general,” the authors wrote.
SOURCE:
The study was led by Kevin F. Boehnke, PhD, University of Michigan Medical School, Ann Arbor, and was published online in ACR Open Rheumatology.
LIMITATIONS:
The cross-sectional nature of the study limited the determination of causality between medical cannabis use and symptom improvement. Moreover, the anonymous and self-reported nature of the survey at a single timepoint may have introduced recall bias. The sample predominantly consisted of older, White females, which may have limited the generalizability of the findings to other demographic groups.
DISCLOSURES:
Some authors received grant support from the National Institute on Drug Abuse and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Some others received payments, honoraria, grant funding, consulting fees, and travel support, and reported other ties with pharmaceutical companies and other institutions.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Is This Methadone’s Moment?
Methadone has been shown to be highly effective for opioid use disorder. So why is it still so difficult to prescribe in the United States and is that about to change?
This paper included more than 30,000 patients with opioid use disorder and showed those on methadone were almost 60% significantly less likely to stop treatment at 24 months than their peers assigned to buprenorphine/naloxone (adjusted hazard ratio [aHR], 1.58), with no difference in mortality risk (aHR, 0.57).
“In Canada, unlike the US, methadone and buprenorphine/naloxone are both available in office-based settings. Methadone really outperforms buprenorphine/naloxone in being able to retain people in treatment, which is our main goal and comes with a host of benefits,” Bohdan Nosyk, PhD, with Simon Fraser University in Burnaby, British Columbia, Canada, who worked on the study, said in an interview.
In addition, a recent systematic review and meta-analysis of relevant research involving more than 1 million patients with opioid use disorder also showed better treatment retention with methadone than with buprenorphine.
During the COVID-19 pandemic, relaxed methadone regulations, that included take-home medications, did not lead to an increase in overdoses. Instead, these changes improved treatment retention and patient experiences, highlighting the potential benefits of further deregulation.
‘Atrocious’ Outdated Policies
However, despite methadone’s proven efficacy and safety for opioid use disorder, it remains vastly underutilized because of outdated US policies restricting its use to opioid treatment programs (OTPs).
“It’s absolutely atrocious that methadone policies have not kept up with the evidence. If you look at other countries that have expanded their access to methadone, their overdose rates have fallen dramatically,” said Leslie Suen, MD, with the University of California, San Francisco, and coauthor of a recent JAMA Viewpoint on this topic.
“Methadone is a very good medication that’s been shown over and over to be very effective and safe,” Alan Leshner, PhD, past director of the National Institute on Drug Abuse, said in an interview.
“There is no reason why it couldn’t be administered through pharmacies or through physicians’ offices as long as it’s done in a controlled and careful way,” said Leshner.
Leshner chaired the committee that produced the 2019 report Medications for Opioid Use Disorder Save Lives.
“We learned during COVID that increasing the amount of take-home methadone and increasing access does not lead to an increase in deaths or an increase in overdose, so it’s hard to find a reason not to do it,” he said.
Change Finally on the Horizon?
Several recent and proposed policy changes could revolutionize methadone delivery in the United States.
In March 2022, in response to the pandemic, the Drug Enforcement Administration (DEA) allowed hospitals to dispense up to a 3-day supply of methadone (known as the 72-hour rule) to bridge care transitions without needing OTPs.
In April 2024, the Substance Abuse and Mental Health Services Administration and DEA codified many methadone and buprenorphine delivery flexibilities granted temporarily during the pandemic, including increased use of telehealth assessments and earlier access to take-home methadone doses.
Another contemporary policy change is expansion of the Americans with Disabilities Act mandating that patients taking medications for opioid use disorder, such as methadone, be able to continue treatment when transitioning to settings such as hospitals, jails, and skilled nursing facilities.
At the state level, California Governor Gavin Newsom recently signed a bill, effective immediately, that expands access to methadone treatment in his state.
On the horizon at the federal level is the Modernizing Opioid Treatment Access Act (MOTAA) — the bipartisan and bicameral bill introduced by Sen. Ed Markey (D-MA) and Sen. Rand Paul (R-KY), along with Rep. Donald Norcross (D-NJ) and Rep. Don Bacon, (R-NE) — that would allow methadone to be prescribed by addiction specialists and dispensed in community pharmacies.
An Ethical Imperative
“With only about 2000 OTP clinics clustered in urban areas, less than 25% of people who are diagnosed with opioid use disorder are actually able to access methadone,” Caty Simon, with the National Survivors Union, Greensboro, North Carolina, and coauthor of the JAMA Viewpoint, said in an interview.
While MOTAA represents a major step forward, limiting methadone prescribing to addiction specialists may not fully address the treatment gap, particularly in rural and underserved areas, Simon said.
To optimize methadone’s potential, she’d like to see further expansion of prescribing privileges to general healthcare providers.
“As someone with lived and living experience of opioid use and treatment, and somebody who works nationally and locally in organizations of people impacted by drug use, I know people in my area right now — marginalized people of color — who would have much better chances of survival if they were able to access methadone. If MOTAA passed tomorrow, we could save so many lives. There is an ethical imperative to pass it,” Simon said.
Leshner said he is “always very concerned about access, particularly for underserved populations, poor people, people living in rural areas. If you can access the medications you need, you’re in big trouble.”
Is this methadone’s moment? “I’m a little optimistic, but I haven’t seen the progress I would like to see,” Leshner said.
A version of this article first appeared on Medscape.com.
Methadone has been shown to be highly effective for opioid use disorder. So why is it still so difficult to prescribe in the United States and is that about to change?
This paper included more than 30,000 patients with opioid use disorder and showed those on methadone were almost 60% significantly less likely to stop treatment at 24 months than their peers assigned to buprenorphine/naloxone (adjusted hazard ratio [aHR], 1.58), with no difference in mortality risk (aHR, 0.57).
“In Canada, unlike the US, methadone and buprenorphine/naloxone are both available in office-based settings. Methadone really outperforms buprenorphine/naloxone in being able to retain people in treatment, which is our main goal and comes with a host of benefits,” Bohdan Nosyk, PhD, with Simon Fraser University in Burnaby, British Columbia, Canada, who worked on the study, said in an interview.
In addition, a recent systematic review and meta-analysis of relevant research involving more than 1 million patients with opioid use disorder also showed better treatment retention with methadone than with buprenorphine.
During the COVID-19 pandemic, relaxed methadone regulations, that included take-home medications, did not lead to an increase in overdoses. Instead, these changes improved treatment retention and patient experiences, highlighting the potential benefits of further deregulation.
‘Atrocious’ Outdated Policies
However, despite methadone’s proven efficacy and safety for opioid use disorder, it remains vastly underutilized because of outdated US policies restricting its use to opioid treatment programs (OTPs).
“It’s absolutely atrocious that methadone policies have not kept up with the evidence. If you look at other countries that have expanded their access to methadone, their overdose rates have fallen dramatically,” said Leslie Suen, MD, with the University of California, San Francisco, and coauthor of a recent JAMA Viewpoint on this topic.
“Methadone is a very good medication that’s been shown over and over to be very effective and safe,” Alan Leshner, PhD, past director of the National Institute on Drug Abuse, said in an interview.
“There is no reason why it couldn’t be administered through pharmacies or through physicians’ offices as long as it’s done in a controlled and careful way,” said Leshner.
Leshner chaired the committee that produced the 2019 report Medications for Opioid Use Disorder Save Lives.
“We learned during COVID that increasing the amount of take-home methadone and increasing access does not lead to an increase in deaths or an increase in overdose, so it’s hard to find a reason not to do it,” he said.
Change Finally on the Horizon?
Several recent and proposed policy changes could revolutionize methadone delivery in the United States.
In March 2022, in response to the pandemic, the Drug Enforcement Administration (DEA) allowed hospitals to dispense up to a 3-day supply of methadone (known as the 72-hour rule) to bridge care transitions without needing OTPs.
In April 2024, the Substance Abuse and Mental Health Services Administration and DEA codified many methadone and buprenorphine delivery flexibilities granted temporarily during the pandemic, including increased use of telehealth assessments and earlier access to take-home methadone doses.
Another contemporary policy change is expansion of the Americans with Disabilities Act mandating that patients taking medications for opioid use disorder, such as methadone, be able to continue treatment when transitioning to settings such as hospitals, jails, and skilled nursing facilities.
At the state level, California Governor Gavin Newsom recently signed a bill, effective immediately, that expands access to methadone treatment in his state.
On the horizon at the federal level is the Modernizing Opioid Treatment Access Act (MOTAA) — the bipartisan and bicameral bill introduced by Sen. Ed Markey (D-MA) and Sen. Rand Paul (R-KY), along with Rep. Donald Norcross (D-NJ) and Rep. Don Bacon, (R-NE) — that would allow methadone to be prescribed by addiction specialists and dispensed in community pharmacies.
An Ethical Imperative
“With only about 2000 OTP clinics clustered in urban areas, less than 25% of people who are diagnosed with opioid use disorder are actually able to access methadone,” Caty Simon, with the National Survivors Union, Greensboro, North Carolina, and coauthor of the JAMA Viewpoint, said in an interview.
While MOTAA represents a major step forward, limiting methadone prescribing to addiction specialists may not fully address the treatment gap, particularly in rural and underserved areas, Simon said.
To optimize methadone’s potential, she’d like to see further expansion of prescribing privileges to general healthcare providers.
“As someone with lived and living experience of opioid use and treatment, and somebody who works nationally and locally in organizations of people impacted by drug use, I know people in my area right now — marginalized people of color — who would have much better chances of survival if they were able to access methadone. If MOTAA passed tomorrow, we could save so many lives. There is an ethical imperative to pass it,” Simon said.
Leshner said he is “always very concerned about access, particularly for underserved populations, poor people, people living in rural areas. If you can access the medications you need, you’re in big trouble.”
Is this methadone’s moment? “I’m a little optimistic, but I haven’t seen the progress I would like to see,” Leshner said.
A version of this article first appeared on Medscape.com.
Methadone has been shown to be highly effective for opioid use disorder. So why is it still so difficult to prescribe in the United States and is that about to change?
This paper included more than 30,000 patients with opioid use disorder and showed those on methadone were almost 60% significantly less likely to stop treatment at 24 months than their peers assigned to buprenorphine/naloxone (adjusted hazard ratio [aHR], 1.58), with no difference in mortality risk (aHR, 0.57).
“In Canada, unlike the US, methadone and buprenorphine/naloxone are both available in office-based settings. Methadone really outperforms buprenorphine/naloxone in being able to retain people in treatment, which is our main goal and comes with a host of benefits,” Bohdan Nosyk, PhD, with Simon Fraser University in Burnaby, British Columbia, Canada, who worked on the study, said in an interview.
In addition, a recent systematic review and meta-analysis of relevant research involving more than 1 million patients with opioid use disorder also showed better treatment retention with methadone than with buprenorphine.
During the COVID-19 pandemic, relaxed methadone regulations, that included take-home medications, did not lead to an increase in overdoses. Instead, these changes improved treatment retention and patient experiences, highlighting the potential benefits of further deregulation.
‘Atrocious’ Outdated Policies
However, despite methadone’s proven efficacy and safety for opioid use disorder, it remains vastly underutilized because of outdated US policies restricting its use to opioid treatment programs (OTPs).
“It’s absolutely atrocious that methadone policies have not kept up with the evidence. If you look at other countries that have expanded their access to methadone, their overdose rates have fallen dramatically,” said Leslie Suen, MD, with the University of California, San Francisco, and coauthor of a recent JAMA Viewpoint on this topic.
“Methadone is a very good medication that’s been shown over and over to be very effective and safe,” Alan Leshner, PhD, past director of the National Institute on Drug Abuse, said in an interview.
“There is no reason why it couldn’t be administered through pharmacies or through physicians’ offices as long as it’s done in a controlled and careful way,” said Leshner.
Leshner chaired the committee that produced the 2019 report Medications for Opioid Use Disorder Save Lives.
“We learned during COVID that increasing the amount of take-home methadone and increasing access does not lead to an increase in deaths or an increase in overdose, so it’s hard to find a reason not to do it,” he said.
Change Finally on the Horizon?
Several recent and proposed policy changes could revolutionize methadone delivery in the United States.
In March 2022, in response to the pandemic, the Drug Enforcement Administration (DEA) allowed hospitals to dispense up to a 3-day supply of methadone (known as the 72-hour rule) to bridge care transitions without needing OTPs.
In April 2024, the Substance Abuse and Mental Health Services Administration and DEA codified many methadone and buprenorphine delivery flexibilities granted temporarily during the pandemic, including increased use of telehealth assessments and earlier access to take-home methadone doses.
Another contemporary policy change is expansion of the Americans with Disabilities Act mandating that patients taking medications for opioid use disorder, such as methadone, be able to continue treatment when transitioning to settings such as hospitals, jails, and skilled nursing facilities.
At the state level, California Governor Gavin Newsom recently signed a bill, effective immediately, that expands access to methadone treatment in his state.
On the horizon at the federal level is the Modernizing Opioid Treatment Access Act (MOTAA) — the bipartisan and bicameral bill introduced by Sen. Ed Markey (D-MA) and Sen. Rand Paul (R-KY), along with Rep. Donald Norcross (D-NJ) and Rep. Don Bacon, (R-NE) — that would allow methadone to be prescribed by addiction specialists and dispensed in community pharmacies.
An Ethical Imperative
“With only about 2000 OTP clinics clustered in urban areas, less than 25% of people who are diagnosed with opioid use disorder are actually able to access methadone,” Caty Simon, with the National Survivors Union, Greensboro, North Carolina, and coauthor of the JAMA Viewpoint, said in an interview.
While MOTAA represents a major step forward, limiting methadone prescribing to addiction specialists may not fully address the treatment gap, particularly in rural and underserved areas, Simon said.
To optimize methadone’s potential, she’d like to see further expansion of prescribing privileges to general healthcare providers.
“As someone with lived and living experience of opioid use and treatment, and somebody who works nationally and locally in organizations of people impacted by drug use, I know people in my area right now — marginalized people of color — who would have much better chances of survival if they were able to access methadone. If MOTAA passed tomorrow, we could save so many lives. There is an ethical imperative to pass it,” Simon said.
Leshner said he is “always very concerned about access, particularly for underserved populations, poor people, people living in rural areas. If you can access the medications you need, you’re in big trouble.”
Is this methadone’s moment? “I’m a little optimistic, but I haven’t seen the progress I would like to see,” Leshner said.
A version of this article first appeared on Medscape.com.
FDA Okays Abuse-Deterrent Opioid for Severe Pain
Roxybond, an immediate-release (IR) formulation of oxycodone hydrochloride, is made with Protega’s SentryBond technology, which makes it harder for people to crush, inject, or snort, according to the company.
In a statement from Protega, Paul Howe, the company’s chief commercial officer, said the drug meets an “unmet need for an IR opioid with abuse-deterrent technology that may reduce misuse and abuse while providing pain relief to medically appropriate patients when used as indicated.”
To determine the tablet’s ability to withstand manipulation, more than 2000 in vitro tests were conducted, according to the release. The findings indicate Roxybond reduces — but does not entirely negate — the potential for intranasal and intravenous abuse.
Roxybond was previously approved in 5-, 15-, and 30-mg doses. The 10 mg option provides clinicians with the ability to better modify side effects, manage titration, and provide precision care for patients on opioid therapy, according to Protega.
“For patients, the range of doses can provide better pain control, reduce the risk of side effects, and provide a smoother transition during dosing transitions,” the company stated.
Roxybond is contraindicated in patients with significant respiratory depression, acute or severe bronchial asthma, gastrointestinal obstruction, or hypersensitivity to oxycodone. The drug is not intended for long-term use unless otherwise determined by a clinician. Roxybond also is subject to the FDA’s Risk Evaluation and Mitigation Strategies for opioids.
“The development of Roxybond with SentryBond is a step forward in fighting the national epidemic of prescription opioid overdose,” said Eric Kinzler, PhD, vice president of medical and regulatory affairs for Protega, in a release. “Protega is dedicated to our mission to block the path to abuse and work with healthcare professionals across the continuum of care to reduce misuse and abuse.”
A version of this article first appeared on Medscape.com.
Roxybond, an immediate-release (IR) formulation of oxycodone hydrochloride, is made with Protega’s SentryBond technology, which makes it harder for people to crush, inject, or snort, according to the company.
In a statement from Protega, Paul Howe, the company’s chief commercial officer, said the drug meets an “unmet need for an IR opioid with abuse-deterrent technology that may reduce misuse and abuse while providing pain relief to medically appropriate patients when used as indicated.”
To determine the tablet’s ability to withstand manipulation, more than 2000 in vitro tests were conducted, according to the release. The findings indicate Roxybond reduces — but does not entirely negate — the potential for intranasal and intravenous abuse.
Roxybond was previously approved in 5-, 15-, and 30-mg doses. The 10 mg option provides clinicians with the ability to better modify side effects, manage titration, and provide precision care for patients on opioid therapy, according to Protega.
“For patients, the range of doses can provide better pain control, reduce the risk of side effects, and provide a smoother transition during dosing transitions,” the company stated.
Roxybond is contraindicated in patients with significant respiratory depression, acute or severe bronchial asthma, gastrointestinal obstruction, or hypersensitivity to oxycodone. The drug is not intended for long-term use unless otherwise determined by a clinician. Roxybond also is subject to the FDA’s Risk Evaluation and Mitigation Strategies for opioids.
“The development of Roxybond with SentryBond is a step forward in fighting the national epidemic of prescription opioid overdose,” said Eric Kinzler, PhD, vice president of medical and regulatory affairs for Protega, in a release. “Protega is dedicated to our mission to block the path to abuse and work with healthcare professionals across the continuum of care to reduce misuse and abuse.”
A version of this article first appeared on Medscape.com.
Roxybond, an immediate-release (IR) formulation of oxycodone hydrochloride, is made with Protega’s SentryBond technology, which makes it harder for people to crush, inject, or snort, according to the company.
In a statement from Protega, Paul Howe, the company’s chief commercial officer, said the drug meets an “unmet need for an IR opioid with abuse-deterrent technology that may reduce misuse and abuse while providing pain relief to medically appropriate patients when used as indicated.”
To determine the tablet’s ability to withstand manipulation, more than 2000 in vitro tests were conducted, according to the release. The findings indicate Roxybond reduces — but does not entirely negate — the potential for intranasal and intravenous abuse.
Roxybond was previously approved in 5-, 15-, and 30-mg doses. The 10 mg option provides clinicians with the ability to better modify side effects, manage titration, and provide precision care for patients on opioid therapy, according to Protega.
“For patients, the range of doses can provide better pain control, reduce the risk of side effects, and provide a smoother transition during dosing transitions,” the company stated.
Roxybond is contraindicated in patients with significant respiratory depression, acute or severe bronchial asthma, gastrointestinal obstruction, or hypersensitivity to oxycodone. The drug is not intended for long-term use unless otherwise determined by a clinician. Roxybond also is subject to the FDA’s Risk Evaluation and Mitigation Strategies for opioids.
“The development of Roxybond with SentryBond is a step forward in fighting the national epidemic of prescription opioid overdose,” said Eric Kinzler, PhD, vice president of medical and regulatory affairs for Protega, in a release. “Protega is dedicated to our mission to block the path to abuse and work with healthcare professionals across the continuum of care to reduce misuse and abuse.”
A version of this article first appeared on Medscape.com.








