Article
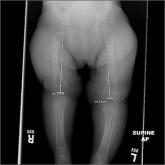
Leg-length discrepancy • asymmetric gluteal folds and popliteal fossae • positive Galeazzi test • Dx?
- Author:
- Beth P. Davis, DPT, MBA, FNAP
- Amir Barzin, DO, MS
- Cristen Page, MD, MPH
► Leg-length discrepancy
► Asymmetric gluteal folds and popliteal fossae
► Positive Galeazzi test
Article
Yeast Infection in Pregnancy? Think Twice About Fluconazole
- Author:
- Amir Barzin, DO, MS
- Anne Mounsey, MD
This study’s findings regarding the risk for miscarriage may mean it’s time to forego fluconazole in favor of topical azoles as firstline...
Article

Yeast infection in pregnancy? Think twice about fluconazole
- Author:
- Amir Barzin, DO, MS
- Anne Mounsey, MD
This study’s findings regarding the risk of miscarriage may mean it’s time to forego fluconazole in favor of topical azoles as first-line...
Article
Sterile or Nonsterile Gloves for Minor Skin Excisions?
- Author:
- Ashley Rietz, MD
- Amir Barzin, DO, MS
- Kohar Jones, MD
- Anne Mounsey, MD
Article

Sterile or non-sterile gloves for minor skin excisions?
- Author:
- Ashley Rietz, MD
- Amir Barzin, DO, MS
- Kohar Jones, MD
- Anne Mounsey, MD
Non-sterile gloves are just as effective as sterile gloves in preventing surgical site infection after minor skin surgeries.
