Article
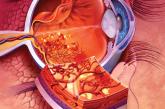
Diabetic retinopathy: The FP’s role in preserving vision
- Author:
- Bryan Farford, DO
Evaluation and Tx of this debilitating complication falls to the eye specialist, but you play a central role in assessing risk and optimizing...
Article
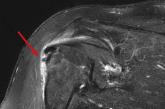
56-year-old woman • worsening pain in left upper arm • influenza vaccination in the arm a few days prior to pain onset • Dx?
- Author:
- Bryan Farford, DO
► Worsening pain in left upper arm
► Pain worsens with movement—especially abduction
► Influenza vaccination in the arm a few days prior...
Article

Osteoporosis: What About Men?
- Author:
- Bryan Farford, DO
- Jonathan Balog, DO
- Kensler Dawson Jackson, MD
Men sustain up to 40% of osteoporotic fractures, with potentially fatal results. But because osteoporosis is largely viewed as a women’s disease,...
Article
Osteoporosis: What about men?
- Author:
- Bryan Farford, DO
- Jonathan Balog, DO
- Kensler Dawson Jackson, MD
Men sustain up to 40% of osteoporotic fractures, with potentially fatal results. But because osteoporosis is largely viewed as a women’s disease,...
