Article

Clinical Outcomes of Anatomical Total Shoulder Arthroplasty in a Young, Active Population
- Author:
- Kusnezov N
- Dunn JC
- Parada SA
- Kilcoyne K
- Waterman BR
Glenohumeral arthritis in young, active patients poses many treatment challenges, and significant concerns about component loosening and failure...
Article
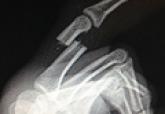
Definitive Fixation of Hand and Wrist Fractures in the Emergency Department
- Author:
- Kusnezov N
- Dunn JC
