Article
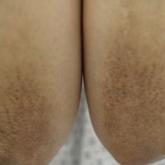
Symmetric Lichen Amyloidosis: An Atypical Location on the Bilateral Extensor Surfaces of the Arms
- Author:
- Jan M. Smogorzewski, MD
- Lina Rodriguez, MD
- Lorraine Young, MD
Lichen amyloidosis (LA) classically presents as a pruritic and papular eruption localized to the pretibial surface of the legs. This report...
Article
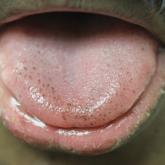
Pigmented Fungiform Papillae of the Tongue in an Indian Male
- Author:
- Jan M. Smogorzewski, MD
- Patrick Armstrong, MD
- Lorraine Young, MD
Pigmented fungiform papillae of the tongue are common lingual hyperpigmented macules in patients with skin of color. It is important to be aware...
