Article
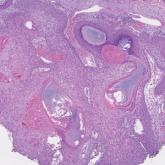
Inspection of Deep Tumor Margins for Accurate Cutaneous Squamous Cell Carcinoma Staging
- Author:
- Jeffrey Chen, BA
- Kevin T. Savage, MD
- Melissa A. Pugliano-Mauro, MD
- Ji Won Ahn, MD
To the Editor:
Histopathologic analysis of debulk specimens in Mohs micrographic surgery (MMS) may augment identification of high-risk...
Article
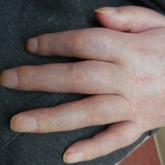
Complex Regional Pain Syndrome Type II After a Brachial Plexus and C6 Nerve Root Injury
- Author:
- Ji Won Ahn, MD
- Caroline Mann, MD
Complex regional pain syndrome (CRPS) is a neuropathic disorder of the extremities characterized by pain, a variety of autonomic and motor...
Article
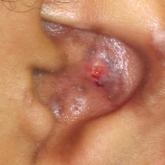
Gottron Papules Mimicking Dermatomyositis: An Unusual Manifestation of Systemic Lupus Erythematosus
- Author:
- Ji Won Ahn, MD
- Sherry Yang, MD
- Katherine Johnson, DO
- Tor Shwayder, MD
