Article
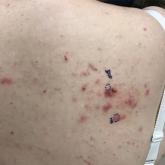
Anti–PD1 Immune Checkpoint Inhibitor–Induced Bullous Pemphigoid in Metastatic Melanoma and Non–Small Cell Lung Cancer
- Author:
- Gabrielle Schwartzman, BS
- Meagan McGinley Simpson, MD, MSc
- Ryan Jones, MD
- Kaitlyn Schiavone, DO
- Marcedes Coffman, MA
- Jon Meyerle, MD
Anti–programmed cell death 1 targeted therapies improve survival in solid and hematologic malignancies but are associated with autoimmune side...
Article
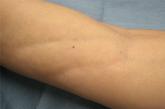
Linear Depressions and Progressive Tightening of the Extremities
- Author:
- Marsha Mitchum, MD
- Erin Spillane, MD
- Jon Meyerle, MD
- Kenneth Galeckas, MD
A 50-year-old woman presented with progressive tightening of her extremities of 2 months’ duration with eventual involvement of her trunk.
