Article
Basal Cell Carcinoma: Analysis of Factors Associated With Incomplete Excision at a Referral Hospital in Southern Spain
- Author:
- Husein Husein-Elahmed, MD
- José Aneiros-Fernández, MD
Basal cell carcinoma (BCC) is the most prevalent malignancy, with excision as the best therapeutic approach. Incomplete excision of nonmelanoma...
Article
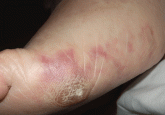
A skin lesion after cardiac catheterization
- Author:
- Salvador Arias-Santiago, MD, PhD
- José Aneiros-Fernández, MD
- Victor Carriel, PhD
- Jacinto Orgaz-Molina, MD
- Miguel Gonález-Andrades, MD, PhD
- Agustín Buendía-Eisman, MD, PhD
- Miguel Alaminos, MD, PhD
Two days after coronary angiography, the patient developed pain, cyanosis, and lesions of the sole of his foot. What is the most likely diagnosis...
Article

A nodule on a woman’s face
- Author:
- Salvador Arias-Santiago, MD
- José Aneiros-Fernández, MD
- Antonio Cutando, PhD
- Agustín Buendía-Eisman, PhD
- Ramón Naranjo-Sintes, PhD
- Antonio Campos, PhD
- Miguel Alaminos-Mingorance
It is firm, pink, 10 mm in diameter, with surrounding telangiectasias. What is it?
Article
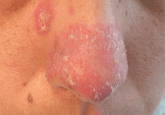
An erythematous plaque on the nose
- Author:
- Salvador Arias-Santiago, MD, PhD
- María Isabel Soriano-Hernández, MD
- José Aneiros-Fernández, MD
- Pilar Burkhardt-Pérez, PhD
- Agustín Buendía-Eisman, PhD
- Ramón Naranjo-Sintes, PhD
- Miguel Alaminos-Mingorance, PhD
A 38-year-old woman presents with a lesion that appears during periods of cold weather. What is the most likely diagnosis?
Article
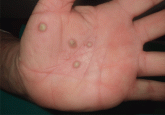
Palmoplantar eruption
- Author:
- Salvador Arias-Santiago, MD
- Husein Husein El-Ahmed, MD
- José Aneiros-Fernández, MD
- María Sierra Girón-Prieto, MD
- Ramón Naranjo-Sintes, PhD
A 38-year-old woman with a history of episodes of arthritis presents with pustules on the palms and on the soles of her feet. What is the most...
Article

An ulcerated plaque on the hand
- Author:
- Husein Husein El-Ahmed, MD
- José Aneiros-Fernández, MD
- Salvador Arias-Santiago, MD
- Rosa Ortego Del Olmo, MD
- Ramón Naranjo-Sintes, PhD
A 73-year-old farmer has a lesion on the dorsum of his hand that bleeds intermittently. What is it?
Article

Painful red nodule on the right hand
- Author:
- Salvador Arias-Santiago, MD
- José Aneiros-Fernández, MD
- Husein Husein El-Ahmed, MD
- María Sierra Girón-Prieto, MD
- Leopoldo Muñoz-Medina, PhD
- Ramón Naranjo-Sintes, PhD
A healthy man presents with a tender subcutaneous nodule on his hand that appeared after cleaning his aquarium. What is the most likely diagnosis...
Article
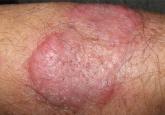
An erythematous plaque on the arm
- Author:
- Salvador Arias-Santiago, MD, PhD
- José Aneiros-Fernández, MD
- Husein Husein El-Ahmed, MD
- Pilar Burkhardt-Pérez, MD
- Ramón Naranjo-Sintes, PhD
A 68-year-old farmer presents with an asymptomatic lesion that appeared spontaneously 5 months ago and has grown progressively. What is the...
Article
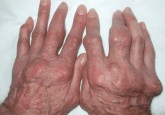
Giant nodules on the hands
- Author:
- Salvador Arias-Santiago, MD, PhD
- José Aneiros-Fernández, MD
- Husein Husein El-Ahmed, MD
- María-Sierra Girón-Prieto, MD
- María Antonia Fernández-Pugnaire, PhD
- Ramón Naranjo-Sintes, PhD
A 78-year-old man with hyperuricemia treated with allopurinol presents with asymptomatic nodules on both hands. What is the most likely diagnosis...
Article
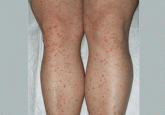
Palpable purpura
- Author:
- Salvador Arias-Santiago, MD, PhD
- José Aneiros-Fernández, MD
- María Sierra Girón-Prieto, MD
- María Antonia Fernández-Pugnaire, PhD
- Ramón Naranjo-Sintes, PhD
A healthy 47-year-old woman presents with a 3-day history of widespread asymptomatic lesions in the extremities, fever, arthralgias, and mild...
