Article
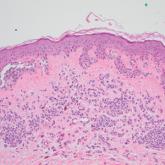
PRAME Expression in Melanocytic Proliferations in Special Sites
- Author:
- Joseph L. Maniaci, MD
- Shyam S. Raghavan, MD
Strong expression of the preferentially expressed antigen in melanoma (PRAME) via immunohistochemistry provides a reliable indicator for benignity...
