Article
Decreasing Pulmonary Embolism-Related Mortality
- Author:
- Parth Rali, MD
Publish date: October 5, 2023
Article
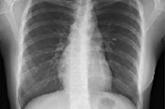
Sarcoidosis: An FP’s primer on an enigmatic disease
- Author:
- Rohit Gupta, MBBS, FCCP
- Maulin Patel, MD
- Luis Caceres, MD
- Mayur Rali, MD, FAAFP
- Parth Rali, MD
Management includes ruling out alternate diagnoses, identifying occult/overt organ involvement, determining treatment, and recognizing worrisome...
