User login
Agitation in children and adolescents: Diagnostic and treatment considerations
Managing agitation—verbal and/or motor restlessness that often is accompanied by irritability and a predisposition to aggression or violence—can be challenging in any patient, but particularly so in children and adolescents. In the United States, the prevalence of children and adolescents presenting to an emergency department (ED) for treatment of psychiatric symptoms, including agitation, has been on the rise.1,2
Similar to the multitude of causes of fever, agitation among children and adolescents has many possible causes.3 Because agitation can pose a risk for harm to others and/or self, it is important to manage it proactively. Other than studies that focus on agitation in pediatric anesthesia, there is a dearth of studies examining agitation and its treatment in children and adolescents. There is also a scarcity of training in the management of acute agitation in children and adolescents. In a 2017 survey of pediatric hospitalists and consultation-liaison psychiatrists at 38 academic children’s hospitals in North America, approximately 60% of respondents indicated that they had received no training in the evaluation or management of pediatric acute agitation.4 In addition, approximately 54% of participants said they did not screen for risk factors for pediatric agitation, even though 84% encountered the condition at least once a month, and as often as weekly.4
This article reviews evidence on the causes and treatments of agitation in children and adolescents. For the purposes of this review, child refers to a patient age 6 to 12, and adolescent refers to a patient age 13 to 17.
Identifying the cause
Addressing the underlying cause of agitation is essential. It’s also important to manage acute agitation while the underlying cause is being investigated in a way that does not jeopardize the patient’s emotional or physical safety.
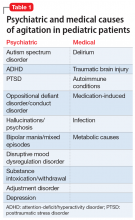
Agitation in children or teens can be due to psychiatric causes such as autism, attention-deficit/hyperactivity disorder (ADHD), or posttraumatic stress disorder (PTSD), or due to medical conditions such as delirium, traumatic brain injury, or other conditions (Table 1).
In a 2005 study of 194 children with agitation in a pediatric post-anesthesia care unit, pain (27%) and anxiety (25%) were found to be the most common causes of agitation.3 Anesthesia-related agitation was a less common cause (11%). Physiologic anomalies were found to be the underlying cause of agitation in only 3 children in this study, but were undiagnosed for a prolonged period in 2 of these 3 children, which highlights the importance of a thorough differential diagnosis in the management of agitation in children.3
Assessment of an agitated child should include a comprehensive history, physical exam, and laboratory testing as indicated. When a pediatric patient comes to the ED with a chief presentation of agitation, a thorough medical and psychiatric assessment should be performed. For patients with a history of psychiatric diagnoses, do not assume that the cause of agitation is psychiatric.
Continue to: Psychiatric causes
Psychiatric causes
Autism spectrum disorder. Children and teens with autism often feel overwhelmed due to transitions, changes, and/or sensory overload. This sensory overload may be in response to relatively subtle sensory stimuli, so it may not always be apparent to parents or others around them.
Research suggests that in general, the ability to cope effectively with emotions is difficult without optimal language development. Due to cognitive/language delays and a related lack of emotional attunement and limited skills in recognizing, expressing, or coping with emotions, difficult emotions in children and adolescents with autism can manifest as agitation.
Attention-deficit/hyperactivity disorder. Children with ADHD may be at a higher risk for agitation, in part due to poor impulse control and limited coping skills. In addition, chronic negative feedback (from parents, teachers, or both) may contribute to low self-esteem, mood symptoms, defiance, and/or other behavioral difficulties. In addition to standard pharmacotherapy for ADHD, treatment involves parent behavior modification training. Setting firm yet empathic limits, “picking battles,” and implementing a developmentally appropriate behavioral plan to manage disruptive behavior in children or adolescents with ADHD can go a long way in helping to prevent the emergence of agitation.
Posttraumatic stress disorder. In some young children, new-onset, unexplained agitation may be the only sign of abuse or trauma. Children who have undergone trauma tend to experience confusion and distress. This may manifest as agitation or aggression, or other symptoms such as increased anxiety or nightmares.5 Trauma may be in the form of witnessing violence (domestic or other); experiencing physical, sexual, and/or emotional abuse; or witnessing/experiencing other significant threats to the safety of self and/or loved ones. Re-establishing (or establishing) a sense of psychological and physical safety is paramount in such patients.6 Psychotherapy is the first-line modality of treatment in children and adolescents with PTSD.6 In general, there is a scarcity of research on medication treatments for PTSD symptoms among children and adolescents.6
Oppositional defiant disorder/conduct disorder. Oppositional defiant disorder (ODD) can be comorbid with ADHD. The diagnosis of ODD requires a pervasive pattern of anger, defiance, vindictiveness, and hostility, particularly towards authority figures. However, these symptoms need to be differentiated from the normal range of childhood behavior. Occasionally, children learn to cope maladaptively through disruptive behavior or agitation. Although a parent or caregiver may see this behavior as intentionally malevolent, in a child with limited coping skills (whether due to young age, developmental/cognitive/language/learning delays, or social communication deficits) or one who has witnessed frequent agitation or aggression in the family environment, agitation and disruptive behavior may be a maladaptive form of coping. Thus, diligence needs to be exercised in the diagnosis of ODD and in understanding the psychosocial factors affecting the child, particularly because impulsiveness and uncooperativeness on their own have been found to be linked to greater likelihood of prescription of psychotropic medications from multiple classes.7 Family-based interventions, particularly parent training, family therapy, and age-appropriate child skills training, are of prime importance in managing this condition.8 Research shows that a shortage of resources, system issues, and cultural roadblocks in implementing family-based psychosocial interventions also can contribute to the increased use of psychotropic medications for aggression in children and teens with ODD, conduct disorder, or ADHD.8 The astute clinician needs to be cognizant of this before prescribing.
Continue to: Hallucinations/psychosis
Hallucinations/psychosis. Hallucinations (whether from psychiatric or medical causes) are significantly associated with agitation.9 In particular, auditory command hallucinations have been linked to agitation. Command hallucinations in children and adolescents may be secondary to early-onset schizophrenia; however, this diagnosis is rare.10 Hallucinations can also be an adverse effect of amphetamine-based stimulant medications in children and adolescents. Visual hallucinations are most often a sign of an underlying medical disorder such as delirium, occipital lobe mass/infection, or drug intoxication or withdrawal. Hallucinations need to be distinguished from the normal, imaginative play of a young child.10
Bipolar mania. In adults, bipolar disorder is a primary psychiatric cause of agitation. In children and adolescents, the diagnosis of bipolar disorder can be complex and requires careful and nuanced history-taking. The risks of agitation are greater with bipolar disorder than with unipolar depression.11,12
Disruptive mood dysregulation disorder. Prior to DSM-5, many children and adolescents with chronic, non-episodic irritability and severe outbursts out of proportion to the situation or stimuli were given a diagnosis of bipolar disorder. These symptoms, in combination with other symptoms, are now considered part of disruptive mood dysregulation disorder when severe outbursts in a child or adolescent occur 3 to 4 times a week consistently, for at least 1 year. The diagnosis of disruptive mood dysregulation disorder requires ruling out other psychiatric and medical conditions, particularly ADHD.13
Substance intoxication/withdrawal. Intoxication or withdrawal from substances such as alcohol, stimulant medications, opioids, methamphetamines, and other agents can lead to agitation. This is more likely to occur among adolescents than children.14
Adjustment disorder. Parental divorce, especially if it is conflictual, or other life stressors, such as experiencing a move or frequent moves, may contribute to the development of agitation in children and adolescents.
Continue to: Depression
Depression. In children and adolescents, depression can manifest as anger or irritability, and occasionally as agitation.
Medical causes
Delirium. Refractory agitation is often a manifestation of delirium in children and adolescents.15 If unrecognized and untreated, delirium can be fatal.16 Therefore, it is imperative that clinicians routinely assess for delirium in any patient who presents with agitation.
Because a patient with delirium often presents with agitation and visual or auditory hallucinations, the medical team may tend to assume these symptoms are secondary to a psychiatric disorder. In this case, the role of the consultation-liaison psychiatrist is critical for guiding the medical team, particularly to continue a thorough exploration of underlying causes while avoiding polypharmacy. Noise, bright lights, frequent changes in nursing staff or caregivers, anticholinergic or benzodiazepine medications, and frequent changes in schedules should be avoided to prevent delirium from occurring or getting worse.17 A multidisciplinary team approach is key in identifying the underlying cause and managing delirium in pediatric patients.
Traumatic brain injury. Agitation may be a presenting symptom in youth with traumatic brain injury (TBI).18 Agitation may present often in the acute recovery phase.19 There is limited evidence on the efficacy and safety of pharmacotherapy for agitation in pediatric patients with TBI.18
Autoimmune conditions. In a study of 27 patients with
Continue to: Medication-induced/iatrogenic
Medication-induced/iatrogenic. Agitation can be an adverse effect of medications such as amantadine (often used for TBI),18 atypical antipsychotics,21 selective serotonin reuptake inhibitors, and serotonin-norepinephrine reuptake inhibitors.
Infection. Agitation can be a result of encephalitis, meningitis, or other infectious processes.22
Metabolic conditions. Hepatic or renal failure, diabetic ketoacidosis, and thyroid toxicosis may cause agitation in children or adolescents.22
Start with nonpharmacologic interventions
Few studies have examined de-escalation techniques in agitated children and adolescents. However, verbal de-escalation is generally viewed as the first-line technique for managing agitation in children and adolescents. When feasible, teaching and modeling developmentally appropriate stress management skills for children and teens can be a beneficial preventative strategy to reduce the incidence and worsening of agitation.23
Clinicians should refrain from using coercion.24 Coercion could harm the therapeutic alliance, thereby impeding assessment of the underlying causes of agitation, and can be particularly harmful for patients who have a history of trauma or abuse. Even in pediatric patients with no such history, coercion is discouraged due to its punitive connotations and potential to adversely impact a vulnerable child or teen.
Continue to: Establishing a therapeutic rapport...
Establishing a therapeutic rapport with the patient, when feasible, can facilitate smoother de-escalation by offering the patient an outlet to air his/her frustrations and emotions, and by helping the patient feel understood.24 To facilitate this, ensure that the patient’s basic comforts and needs are met, such as access to a warm bed, food, and safety.25
The psychiatrist’s role is to help uncover and address the underlying reason for the patient’s agony or distress. Once the child or adolescent has calmed, explore potential triggers or causes of the agitation.
There has been a significant move away from the use of restraints for managing agitation in children and adolescents.26 Restraints have a psychologically traumatizing effect,27 and have been linked to life-threatening injuries and death in children.24
Pharmacotherapy: Proceed with caution
There are no FDA-approved medications for the treatment of agitation in the general pediatric population, and any medication use in this population is off-label. There is also a dearth of research examining the safety and efficacy of using psychotropic medications for agitation in pediatric patients. Because children and adolescents are more susceptible to adverse effects and risks associated with the use of psychotropic medications, special caution is warranted. In general, pharmacologic interventions are not recommended without the use of psychotherapy-based modalities.
In the past, the aim of using medications to treat patients with agitation was to put the patient to sleep.25 This practice did not help clinicians to assess for underlying causes, and was often accompanied by a greater risk of adverse effects and reactions.24 Therefore, the goal of medication treatment for agitation is to help calm the patient instead of inducing sleep.25
Continue to: Pharmacotherapy should...
Pharmacotherapy should be used only when behavioral interventions have been unsuccessful. Key considerations for using psychotropic medications to address agitation in children and adolescents are summarized in Table 2.25
Antipsychotics, particularly second-generation antipsychotics (SGAs), have been commonly used to manage acute agitation in children and adolescents, and there has been an upswing in the use of these medications in the United States in the last several years.28 Research indicates that males, children and adolescents in foster care, and those with Medicaid have been the more frequent youth recipients of SGAs.29 Of particular concern is the prevalence of antipsychotic use among children younger than age 6. In the last few decades, there has been an increase in the prescription of antipsychotics for children younger than age 6, particularly for disruptive behavior and aggression.30 In a study of preschool-age Medicaid patients in Kentucky, 70,777 prescriptions for SGAs were given to 6,915 children <6 years of age; 73% of these prescriptions were for male patients.30 Because there is a lack of controlled studies examining the safety and efficacy of SGAs among children and adolescents, especially with long-term use, further research is needed and caution is warranted.28
The FDA has approved
Externalizing disorders among children and adolescents tend to get treated with antipsychotics.28 A Canadian study examining records of 6,916 children found that most children who had been prescribed risperidone received it for ADHD or conduct disorder, and most patients had not received laboratory testing for monitoring the antipsychotic medication they were taking.31 In a 2018 study examining medical records of 120 pediatric patients who presented to an ED in British Columbia with agitation, antipsychotics were the most commonly used medications for patients with autism spectrum disorder; most patients received at least 1 dose.14
For children and adolescents with agitation or aggression who were admitted to inpatient units, IM
Continue to: In case reports...
In case reports, a combination of olanzapine with CNS-suppressing agents has resulted in death. Therefore, do not combine olanzapine with agents such as benzodiazepines.25 In a patient with a likely medical source of agitation, insufficient evidence exists to support the use of olanzapine, and additional research is needed.25
Low-dose haloperidol has been found to be effective for delirium-related agitation in pediatric studies.15 Before initiating an antipsychotic for any child or adolescent, review the patient’s family history for reports of early cardiac death and the patient’s own history of cardiac symptoms, palpitations, syncope, or prolonged QT interval. Monitor for QT prolongation. Among commonly used antipsychotics, the risk of QT prolongation is higher with IV administration of haloperidol and with ziprasidone. Studies show that compared with oral or IM
A few studies have found risperidone to be efficacious for treating ODD and conduct disorder; however, this use is off-label, and its considerable adverse effect and risk profile needs to be weighed against the potential benefit.8
Antipsychotic polypharmacy should be avoided because of the higher risk of adverse effects and interactions, and a lack of robust, controlled studies evaluating the safety of using antipsychotics for non-FDA-approved indications in children and adolescents.7 All patients who receive antipsychotics require monitoring for extrapyramidal symptoms, tardive dyskinesia, neuroleptic malignant syndrome, orthostatic hypotension, sedation, metabolic syndrome, and other potential adverse effects. Patients receiving risperidone need to have their prolactin levels monitored periodically, and their parents should be made aware of the potential for hyperprolactinemia and other adverse effects. Aripiprazole and quetiapine may increase the risk of suicidality.
Antiepileptics. A meta-analysis of 7 randomized controlled trials examining the use of antiepileptic medications (
Continue to: In a retrospective case series...
In a retrospective case series of 30 pediatric patients with autism spectrum disorder who were given oxcarbazepine, Douglas et al35 found that 47% of participants experienced significant improvement in irritability/agitation. However, 23% of patients reported significant adverse effects leading to discontinuation. Insufficient evidence exists for the safety and efficacy of oxcarbazepine in this population.35
Benzodiazepines. The use of benzodiazepines in pediatric patients has been associated with paradoxical disinhibition reactions, particularly in children with autism and other developmental or cognitive disabilities or delays.21 There is a lack of data on the safety and efficacy of long-term use of benzodiazepines in children, especially in light of these patients’ developing brains, the risk of cognitive impairment, and the potential for dependence with long-term use. Despite this, some studies show that the use of benzodiazepines is fairly common among pediatric patients who present to the ED with agitation.14 In a recent retrospective study, Kendrick et al14 found that among pediatric patients with agitation who were brought to the ED, benzodiazepines were the most commonly prescribed medications.
Other medications. Clonidine and
Diphenhydramine, in both oral and IM forms, has been used to treat agitation in children,32 but has also been associated with a paradoxical disinhibition reaction in pediatric patients21 and therefore should be used only sparingly and with caution. Diphenhydramine has anticholinergic properties, and may worsen delirium.15 Stimulant medications can help aggressive behavior in children and adolescents with ADHD.37
Bottom Line
Agitation among children and adolescents has many possible causes. A combination of a comprehensive assessment and evidence-based, judicious treatment interventions can help prevent and manage agitation in this vulnerable population.
Related Resources
- Baker M, Carlson GA. What do we really know about PRN use in agitated children with mental health conditions: a clinical review. Evid Based Ment Health. 2018;21(4):166-170.
- Gerson R, Malas N, Mroczkowski MM. Crisis in the emergency department: the evaluation and management of acute agitation in children and adolescents. Child Adolesc Psychiatr Clin N Am. 2018;27(3):367-386.
Drug Brand Names
Amantadine • Symmetrel
Aripiprazole • Abilify
Clonidine • Catapres
Guanfacine • Intuniv, Tenex
Haloperidol • Haldol
Lamotrigine • Lamictal
Levetiracetam • Keppra, Spritam
Olanzapine • Zyprexa
Oxcarbazepine • Trileptal
Quetiapine • Seroquel
Topiramate • Topamax
Risperidone • Risperdal
Valproate • Depakene
Ziprasidone • Geodon
1. Frosch E, Kelly P. Issues in pediatric psychiatric emergency care. In: Emergency psychiatry. Cambridge, UK: Cambridge University Press; 2011:185-199.
2. American College of Emergency Physicians. Pediatric mental health emergencies in the emergency department. https://www.acep.org/patient-care/policy-statements/pediatric-mental-health-emergencies-in-the-emergency-medical-services-system/. Revised September 2018. Accessed February 23, 2019.
3. Voepel-Lewis, T, Burke C, Hadden S, et al. Nurses’ diagnoses and treatment decisions regarding care of the agitated child. J Perianesth Nurs. 2005;20(4):239-248.
4. Malas N, Spital L, Fischer J, et al. National survey on pediatric acute agitation and behavioral escalation in academic inpatient pediatric care settings. Psychosomatics. 2017;58(3):299-306.
5. Famularo R, Kinscherff R, Fenton T. Symptom differences in acute and chronic presentation of childhood post-traumatic stress disorder. Child Abuse Negl. 1990;14(3):439-444.
6. Kaminer D, Seedat S, Stein DJ. Post-traumatic stress disorder in children. World Psychiatry. 2005;4(2):121-125.
7. Ninan A, Stewart SL, Theall LA, et al. Adverse effects of psychotropic medications in children: predictive factors. J Can Acad Child Adolesc Psychiatry. 2014;23(3):218-225.
8. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 2: antipsychotics and traditional mood stabilizers. Can J Psychiatry. 2015;60(2):52-61.
9. Vareilles D, Bréhin C, Cortey C, et al. Hallucinations: Etiological analysis of children admitted to a pediatric emergency department. Arch Pediatr. 2017;24(5):445-452.
10. Bartlett J. Childhood-onset schizophrenia: what do we really know? Health Psychol Behav Med. 2014;2(1):735-747.
11. Diler RS, Goldstein TR, Hafeman D, et al. Distinguishing bipolar depression from unipolar depression in youth: Preliminary findings. J Child Adolesc Psychopharmacol. 2017;27(4):310-319.
12. Dervic K, Garcia-Amador M, Sudol K, et al. Bipolar I and II versus unipolar depression: clinical differences and impulsivity/aggression traits. Eur Psychiatry. 2015;30(1):106-113.
13. Masi L, Gignac M ADHD and DMDD comorbidities, similarities and distinctions. J Child Adolesc Behav2016;4:325.
14. Kendrick JG, Goldman RD, Carr RR. Pharmacologic management of agitation and aggression in a pediatric emergency department - a retrospective cohort study. J Pediatr Pharmacol Ther. 2018;23(6):455-459.
15. Schieveld JN, Staal M, Voogd L, et al. Refractory agitation as a marker for pediatric delirium in very young infants at a pediatric intensive care unit. Intensive Care Med. 2010;36(11):1982-1983.
16. Traube C, Silver G, Gerber LM, et al. Delirium and mortality in critically ill children: epidemiology and outcomes of pediatric delirium. Crit Care Med. 2017;45(5):891-898.
17. Bettencourt A, Mullen JE. Delirium in children: identification, prevention, and management. Crit Care Nurse. 2017;37(3):e9-e18.
18. Suskauer SJ, Trovato MK. Update on pharmaceutical intervention for disorders of consciousness and agitation after traumatic brain injury in children. PM R. 2013;5(2):142-147.
19. Nowicki M, Pearlman L, Campbell C, et al. Agitated behavior scale in pediatric traumatic brain injury. Brain Inj. 2019. doi: 10.1080/02699052.2019.1565893.
20. Mohammad SS, Jones H, Hong M, et al. Symptomatic treatment of children with anti-NMDAR encephalitis. Dev Med Child Neurol. 2016;58(4):376-384.
21. Sonnier L, Barzman D. Pharmacologic management of acutely agitated pediatric patients. Pediatr Drugs. 2011;13(1):1-10.
22. Nordstrom K, Zun LS, Wilson MP, et al. Medical evaluation and triage of the agitated patient: consensus statement of the american association for emergency psychiatry project Beta medical evaluation workgroup. West J Emerg Med. 2012;13(1):3-10.
23. Masters KJ, Bellonci C, Bernet W, et al; American Academy of Child and Adolescent Psychiatry. Practice parameter for the prevention and management of aggressive behavior in child and adolescent psychiatric institutions, with special reference to seclusion and restraint. J Am Acad Child Adolesc Psychiatry. 2002;41(2 suppl):4S-25S.
24. Croce ND, Mantovani C. Using de-escalation techniques to prevent violent behavior in pediatric psychiatric emergencies: It is possible. Pediatric Dimensions, 2017;2(1):1-2.
25. Marzullo LR. Pharmacologic management of the agitated child. Pediatr Emerg Care. 2014;30(4):269-275.
26. Caldwell B, Albert C, Azeem MW, et al. Successful seclusion and restraint prevention effort in child and adolescent programs. J Psychosoc Nurs Ment Health Serv. 2014;52(11):30-38.
27. De Hert M, Dirix N, Demunter H, et al. Prevalence and correlates of seclusion and restraint use in children and adolescents: a systematic review. Eur Child Adolesc Psychiatry. 2011;20(5):221-230.
28. Crystal S, Olfson M, Huang C, et al. Broadened use of atypical antipsychotics: safety, effectiveness, and policy challenges. Health Aff (Millwood). 2009;28(5):w770-w781.
29. American Academy of Child and Adolescent Psychiatry. Practice parameters for the use of atypical antipsychotic medication in children and adolescents. https://www.aacap.org/App_Themes/AACAP/docs/practice_parameters/Atypical_Antipsychotic_Medications_Web.pdf. Accessed March 4, 2019.
30. Lohr WD, Chowning RT, Stevenson MD, et al. Trends in atypical antipsychotics prescribed to children six years of age or less on Medicaid in Kentucky. J Child Adolesc Psychopharmacol. 2015;25(5):440-443.
31. Chen W, Cepoiu-Martin M, Stang A, et al. Antipsychotic prescribing and safety monitoring practices in children and youth: a population-based study in Alberta, Canada. Clin Drug Investig. 2018;38(5):449-455.
32. Deshmukh P, Kulkarni G, Barzman D. Recommendations for pharmacological management of inpatient aggression in children and adolescents. Psychiatry (Edgmont). 2010;7(2):32-40.
33. Haldol [package insert]. Beerse, Belgium: Janssen Pharmaceutica NV; 2005.
34. Hirota T, Veenstra-Vanderweele J, Hollander E, et al. Antiepileptic medications in autism spectrum disorder: a systematic review and meta-analysis. J Autism Dev Disord. 2014;44(4):948-957.
35. Douglas JF, Sanders KB, Benneyworth MH, et al. Brief report: retrospective case series of oxcarbazepine for irritability/agitation symptoms in autism spectrum disorder. J Autism Dev Disord. 2013;43(5):1243-1247.
36. Harmon RJ, Riggs PD. Clonidine for posttraumatic stress disorder in preschool children. J Am Acad
37. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 1: Psychostimulants, alpha-2 Agonists, and atomoxetine. Can J Psychiatry. 2015;60(2):42-51
Managing agitation—verbal and/or motor restlessness that often is accompanied by irritability and a predisposition to aggression or violence—can be challenging in any patient, but particularly so in children and adolescents. In the United States, the prevalence of children and adolescents presenting to an emergency department (ED) for treatment of psychiatric symptoms, including agitation, has been on the rise.1,2
Similar to the multitude of causes of fever, agitation among children and adolescents has many possible causes.3 Because agitation can pose a risk for harm to others and/or self, it is important to manage it proactively. Other than studies that focus on agitation in pediatric anesthesia, there is a dearth of studies examining agitation and its treatment in children and adolescents. There is also a scarcity of training in the management of acute agitation in children and adolescents. In a 2017 survey of pediatric hospitalists and consultation-liaison psychiatrists at 38 academic children’s hospitals in North America, approximately 60% of respondents indicated that they had received no training in the evaluation or management of pediatric acute agitation.4 In addition, approximately 54% of participants said they did not screen for risk factors for pediatric agitation, even though 84% encountered the condition at least once a month, and as often as weekly.4
This article reviews evidence on the causes and treatments of agitation in children and adolescents. For the purposes of this review, child refers to a patient age 6 to 12, and adolescent refers to a patient age 13 to 17.
Identifying the cause
Addressing the underlying cause of agitation is essential. It’s also important to manage acute agitation while the underlying cause is being investigated in a way that does not jeopardize the patient’s emotional or physical safety.
Agitation in children or teens can be due to psychiatric causes such as autism, attention-deficit/hyperactivity disorder (ADHD), or posttraumatic stress disorder (PTSD), or due to medical conditions such as delirium, traumatic brain injury, or other conditions (Table 1).
In a 2005 study of 194 children with agitation in a pediatric post-anesthesia care unit, pain (27%) and anxiety (25%) were found to be the most common causes of agitation.3 Anesthesia-related agitation was a less common cause (11%). Physiologic anomalies were found to be the underlying cause of agitation in only 3 children in this study, but were undiagnosed for a prolonged period in 2 of these 3 children, which highlights the importance of a thorough differential diagnosis in the management of agitation in children.3
Assessment of an agitated child should include a comprehensive history, physical exam, and laboratory testing as indicated. When a pediatric patient comes to the ED with a chief presentation of agitation, a thorough medical and psychiatric assessment should be performed. For patients with a history of psychiatric diagnoses, do not assume that the cause of agitation is psychiatric.
Continue to: Psychiatric causes
Psychiatric causes
Autism spectrum disorder. Children and teens with autism often feel overwhelmed due to transitions, changes, and/or sensory overload. This sensory overload may be in response to relatively subtle sensory stimuli, so it may not always be apparent to parents or others around them.
Research suggests that in general, the ability to cope effectively with emotions is difficult without optimal language development. Due to cognitive/language delays and a related lack of emotional attunement and limited skills in recognizing, expressing, or coping with emotions, difficult emotions in children and adolescents with autism can manifest as agitation.
Attention-deficit/hyperactivity disorder. Children with ADHD may be at a higher risk for agitation, in part due to poor impulse control and limited coping skills. In addition, chronic negative feedback (from parents, teachers, or both) may contribute to low self-esteem, mood symptoms, defiance, and/or other behavioral difficulties. In addition to standard pharmacotherapy for ADHD, treatment involves parent behavior modification training. Setting firm yet empathic limits, “picking battles,” and implementing a developmentally appropriate behavioral plan to manage disruptive behavior in children or adolescents with ADHD can go a long way in helping to prevent the emergence of agitation.
Posttraumatic stress disorder. In some young children, new-onset, unexplained agitation may be the only sign of abuse or trauma. Children who have undergone trauma tend to experience confusion and distress. This may manifest as agitation or aggression, or other symptoms such as increased anxiety or nightmares.5 Trauma may be in the form of witnessing violence (domestic or other); experiencing physical, sexual, and/or emotional abuse; or witnessing/experiencing other significant threats to the safety of self and/or loved ones. Re-establishing (or establishing) a sense of psychological and physical safety is paramount in such patients.6 Psychotherapy is the first-line modality of treatment in children and adolescents with PTSD.6 In general, there is a scarcity of research on medication treatments for PTSD symptoms among children and adolescents.6
Oppositional defiant disorder/conduct disorder. Oppositional defiant disorder (ODD) can be comorbid with ADHD. The diagnosis of ODD requires a pervasive pattern of anger, defiance, vindictiveness, and hostility, particularly towards authority figures. However, these symptoms need to be differentiated from the normal range of childhood behavior. Occasionally, children learn to cope maladaptively through disruptive behavior or agitation. Although a parent or caregiver may see this behavior as intentionally malevolent, in a child with limited coping skills (whether due to young age, developmental/cognitive/language/learning delays, or social communication deficits) or one who has witnessed frequent agitation or aggression in the family environment, agitation and disruptive behavior may be a maladaptive form of coping. Thus, diligence needs to be exercised in the diagnosis of ODD and in understanding the psychosocial factors affecting the child, particularly because impulsiveness and uncooperativeness on their own have been found to be linked to greater likelihood of prescription of psychotropic medications from multiple classes.7 Family-based interventions, particularly parent training, family therapy, and age-appropriate child skills training, are of prime importance in managing this condition.8 Research shows that a shortage of resources, system issues, and cultural roadblocks in implementing family-based psychosocial interventions also can contribute to the increased use of psychotropic medications for aggression in children and teens with ODD, conduct disorder, or ADHD.8 The astute clinician needs to be cognizant of this before prescribing.
Continue to: Hallucinations/psychosis
Hallucinations/psychosis. Hallucinations (whether from psychiatric or medical causes) are significantly associated with agitation.9 In particular, auditory command hallucinations have been linked to agitation. Command hallucinations in children and adolescents may be secondary to early-onset schizophrenia; however, this diagnosis is rare.10 Hallucinations can also be an adverse effect of amphetamine-based stimulant medications in children and adolescents. Visual hallucinations are most often a sign of an underlying medical disorder such as delirium, occipital lobe mass/infection, or drug intoxication or withdrawal. Hallucinations need to be distinguished from the normal, imaginative play of a young child.10
Bipolar mania. In adults, bipolar disorder is a primary psychiatric cause of agitation. In children and adolescents, the diagnosis of bipolar disorder can be complex and requires careful and nuanced history-taking. The risks of agitation are greater with bipolar disorder than with unipolar depression.11,12
Disruptive mood dysregulation disorder. Prior to DSM-5, many children and adolescents with chronic, non-episodic irritability and severe outbursts out of proportion to the situation or stimuli were given a diagnosis of bipolar disorder. These symptoms, in combination with other symptoms, are now considered part of disruptive mood dysregulation disorder when severe outbursts in a child or adolescent occur 3 to 4 times a week consistently, for at least 1 year. The diagnosis of disruptive mood dysregulation disorder requires ruling out other psychiatric and medical conditions, particularly ADHD.13
Substance intoxication/withdrawal. Intoxication or withdrawal from substances such as alcohol, stimulant medications, opioids, methamphetamines, and other agents can lead to agitation. This is more likely to occur among adolescents than children.14
Adjustment disorder. Parental divorce, especially if it is conflictual, or other life stressors, such as experiencing a move or frequent moves, may contribute to the development of agitation in children and adolescents.
Continue to: Depression
Depression. In children and adolescents, depression can manifest as anger or irritability, and occasionally as agitation.
Medical causes
Delirium. Refractory agitation is often a manifestation of delirium in children and adolescents.15 If unrecognized and untreated, delirium can be fatal.16 Therefore, it is imperative that clinicians routinely assess for delirium in any patient who presents with agitation.
Because a patient with delirium often presents with agitation and visual or auditory hallucinations, the medical team may tend to assume these symptoms are secondary to a psychiatric disorder. In this case, the role of the consultation-liaison psychiatrist is critical for guiding the medical team, particularly to continue a thorough exploration of underlying causes while avoiding polypharmacy. Noise, bright lights, frequent changes in nursing staff or caregivers, anticholinergic or benzodiazepine medications, and frequent changes in schedules should be avoided to prevent delirium from occurring or getting worse.17 A multidisciplinary team approach is key in identifying the underlying cause and managing delirium in pediatric patients.
Traumatic brain injury. Agitation may be a presenting symptom in youth with traumatic brain injury (TBI).18 Agitation may present often in the acute recovery phase.19 There is limited evidence on the efficacy and safety of pharmacotherapy for agitation in pediatric patients with TBI.18
Autoimmune conditions. In a study of 27 patients with
Continue to: Medication-induced/iatrogenic
Medication-induced/iatrogenic. Agitation can be an adverse effect of medications such as amantadine (often used for TBI),18 atypical antipsychotics,21 selective serotonin reuptake inhibitors, and serotonin-norepinephrine reuptake inhibitors.
Infection. Agitation can be a result of encephalitis, meningitis, or other infectious processes.22
Metabolic conditions. Hepatic or renal failure, diabetic ketoacidosis, and thyroid toxicosis may cause agitation in children or adolescents.22
Start with nonpharmacologic interventions
Few studies have examined de-escalation techniques in agitated children and adolescents. However, verbal de-escalation is generally viewed as the first-line technique for managing agitation in children and adolescents. When feasible, teaching and modeling developmentally appropriate stress management skills for children and teens can be a beneficial preventative strategy to reduce the incidence and worsening of agitation.23
Clinicians should refrain from using coercion.24 Coercion could harm the therapeutic alliance, thereby impeding assessment of the underlying causes of agitation, and can be particularly harmful for patients who have a history of trauma or abuse. Even in pediatric patients with no such history, coercion is discouraged due to its punitive connotations and potential to adversely impact a vulnerable child or teen.
Continue to: Establishing a therapeutic rapport...
Establishing a therapeutic rapport with the patient, when feasible, can facilitate smoother de-escalation by offering the patient an outlet to air his/her frustrations and emotions, and by helping the patient feel understood.24 To facilitate this, ensure that the patient’s basic comforts and needs are met, such as access to a warm bed, food, and safety.25
The psychiatrist’s role is to help uncover and address the underlying reason for the patient’s agony or distress. Once the child or adolescent has calmed, explore potential triggers or causes of the agitation.
There has been a significant move away from the use of restraints for managing agitation in children and adolescents.26 Restraints have a psychologically traumatizing effect,27 and have been linked to life-threatening injuries and death in children.24
Pharmacotherapy: Proceed with caution
There are no FDA-approved medications for the treatment of agitation in the general pediatric population, and any medication use in this population is off-label. There is also a dearth of research examining the safety and efficacy of using psychotropic medications for agitation in pediatric patients. Because children and adolescents are more susceptible to adverse effects and risks associated with the use of psychotropic medications, special caution is warranted. In general, pharmacologic interventions are not recommended without the use of psychotherapy-based modalities.
In the past, the aim of using medications to treat patients with agitation was to put the patient to sleep.25 This practice did not help clinicians to assess for underlying causes, and was often accompanied by a greater risk of adverse effects and reactions.24 Therefore, the goal of medication treatment for agitation is to help calm the patient instead of inducing sleep.25
Continue to: Pharmacotherapy should...
Pharmacotherapy should be used only when behavioral interventions have been unsuccessful. Key considerations for using psychotropic medications to address agitation in children and adolescents are summarized in Table 2.25
Antipsychotics, particularly second-generation antipsychotics (SGAs), have been commonly used to manage acute agitation in children and adolescents, and there has been an upswing in the use of these medications in the United States in the last several years.28 Research indicates that males, children and adolescents in foster care, and those with Medicaid have been the more frequent youth recipients of SGAs.29 Of particular concern is the prevalence of antipsychotic use among children younger than age 6. In the last few decades, there has been an increase in the prescription of antipsychotics for children younger than age 6, particularly for disruptive behavior and aggression.30 In a study of preschool-age Medicaid patients in Kentucky, 70,777 prescriptions for SGAs were given to 6,915 children <6 years of age; 73% of these prescriptions were for male patients.30 Because there is a lack of controlled studies examining the safety and efficacy of SGAs among children and adolescents, especially with long-term use, further research is needed and caution is warranted.28
The FDA has approved
Externalizing disorders among children and adolescents tend to get treated with antipsychotics.28 A Canadian study examining records of 6,916 children found that most children who had been prescribed risperidone received it for ADHD or conduct disorder, and most patients had not received laboratory testing for monitoring the antipsychotic medication they were taking.31 In a 2018 study examining medical records of 120 pediatric patients who presented to an ED in British Columbia with agitation, antipsychotics were the most commonly used medications for patients with autism spectrum disorder; most patients received at least 1 dose.14
For children and adolescents with agitation or aggression who were admitted to inpatient units, IM
Continue to: In case reports...
In case reports, a combination of olanzapine with CNS-suppressing agents has resulted in death. Therefore, do not combine olanzapine with agents such as benzodiazepines.25 In a patient with a likely medical source of agitation, insufficient evidence exists to support the use of olanzapine, and additional research is needed.25
Low-dose haloperidol has been found to be effective for delirium-related agitation in pediatric studies.15 Before initiating an antipsychotic for any child or adolescent, review the patient’s family history for reports of early cardiac death and the patient’s own history of cardiac symptoms, palpitations, syncope, or prolonged QT interval. Monitor for QT prolongation. Among commonly used antipsychotics, the risk of QT prolongation is higher with IV administration of haloperidol and with ziprasidone. Studies show that compared with oral or IM
A few studies have found risperidone to be efficacious for treating ODD and conduct disorder; however, this use is off-label, and its considerable adverse effect and risk profile needs to be weighed against the potential benefit.8
Antipsychotic polypharmacy should be avoided because of the higher risk of adverse effects and interactions, and a lack of robust, controlled studies evaluating the safety of using antipsychotics for non-FDA-approved indications in children and adolescents.7 All patients who receive antipsychotics require monitoring for extrapyramidal symptoms, tardive dyskinesia, neuroleptic malignant syndrome, orthostatic hypotension, sedation, metabolic syndrome, and other potential adverse effects. Patients receiving risperidone need to have their prolactin levels monitored periodically, and their parents should be made aware of the potential for hyperprolactinemia and other adverse effects. Aripiprazole and quetiapine may increase the risk of suicidality.
Antiepileptics. A meta-analysis of 7 randomized controlled trials examining the use of antiepileptic medications (
Continue to: In a retrospective case series...
In a retrospective case series of 30 pediatric patients with autism spectrum disorder who were given oxcarbazepine, Douglas et al35 found that 47% of participants experienced significant improvement in irritability/agitation. However, 23% of patients reported significant adverse effects leading to discontinuation. Insufficient evidence exists for the safety and efficacy of oxcarbazepine in this population.35
Benzodiazepines. The use of benzodiazepines in pediatric patients has been associated with paradoxical disinhibition reactions, particularly in children with autism and other developmental or cognitive disabilities or delays.21 There is a lack of data on the safety and efficacy of long-term use of benzodiazepines in children, especially in light of these patients’ developing brains, the risk of cognitive impairment, and the potential for dependence with long-term use. Despite this, some studies show that the use of benzodiazepines is fairly common among pediatric patients who present to the ED with agitation.14 In a recent retrospective study, Kendrick et al14 found that among pediatric patients with agitation who were brought to the ED, benzodiazepines were the most commonly prescribed medications.
Other medications. Clonidine and
Diphenhydramine, in both oral and IM forms, has been used to treat agitation in children,32 but has also been associated with a paradoxical disinhibition reaction in pediatric patients21 and therefore should be used only sparingly and with caution. Diphenhydramine has anticholinergic properties, and may worsen delirium.15 Stimulant medications can help aggressive behavior in children and adolescents with ADHD.37
Bottom Line
Agitation among children and adolescents has many possible causes. A combination of a comprehensive assessment and evidence-based, judicious treatment interventions can help prevent and manage agitation in this vulnerable population.
Related Resources
- Baker M, Carlson GA. What do we really know about PRN use in agitated children with mental health conditions: a clinical review. Evid Based Ment Health. 2018;21(4):166-170.
- Gerson R, Malas N, Mroczkowski MM. Crisis in the emergency department: the evaluation and management of acute agitation in children and adolescents. Child Adolesc Psychiatr Clin N Am. 2018;27(3):367-386.
Drug Brand Names
Amantadine • Symmetrel
Aripiprazole • Abilify
Clonidine • Catapres
Guanfacine • Intuniv, Tenex
Haloperidol • Haldol
Lamotrigine • Lamictal
Levetiracetam • Keppra, Spritam
Olanzapine • Zyprexa
Oxcarbazepine • Trileptal
Quetiapine • Seroquel
Topiramate • Topamax
Risperidone • Risperdal
Valproate • Depakene
Ziprasidone • Geodon
Managing agitation—verbal and/or motor restlessness that often is accompanied by irritability and a predisposition to aggression or violence—can be challenging in any patient, but particularly so in children and adolescents. In the United States, the prevalence of children and adolescents presenting to an emergency department (ED) for treatment of psychiatric symptoms, including agitation, has been on the rise.1,2
Similar to the multitude of causes of fever, agitation among children and adolescents has many possible causes.3 Because agitation can pose a risk for harm to others and/or self, it is important to manage it proactively. Other than studies that focus on agitation in pediatric anesthesia, there is a dearth of studies examining agitation and its treatment in children and adolescents. There is also a scarcity of training in the management of acute agitation in children and adolescents. In a 2017 survey of pediatric hospitalists and consultation-liaison psychiatrists at 38 academic children’s hospitals in North America, approximately 60% of respondents indicated that they had received no training in the evaluation or management of pediatric acute agitation.4 In addition, approximately 54% of participants said they did not screen for risk factors for pediatric agitation, even though 84% encountered the condition at least once a month, and as often as weekly.4
This article reviews evidence on the causes and treatments of agitation in children and adolescents. For the purposes of this review, child refers to a patient age 6 to 12, and adolescent refers to a patient age 13 to 17.
Identifying the cause
Addressing the underlying cause of agitation is essential. It’s also important to manage acute agitation while the underlying cause is being investigated in a way that does not jeopardize the patient’s emotional or physical safety.
Agitation in children or teens can be due to psychiatric causes such as autism, attention-deficit/hyperactivity disorder (ADHD), or posttraumatic stress disorder (PTSD), or due to medical conditions such as delirium, traumatic brain injury, or other conditions (Table 1).
In a 2005 study of 194 children with agitation in a pediatric post-anesthesia care unit, pain (27%) and anxiety (25%) were found to be the most common causes of agitation.3 Anesthesia-related agitation was a less common cause (11%). Physiologic anomalies were found to be the underlying cause of agitation in only 3 children in this study, but were undiagnosed for a prolonged period in 2 of these 3 children, which highlights the importance of a thorough differential diagnosis in the management of agitation in children.3
Assessment of an agitated child should include a comprehensive history, physical exam, and laboratory testing as indicated. When a pediatric patient comes to the ED with a chief presentation of agitation, a thorough medical and psychiatric assessment should be performed. For patients with a history of psychiatric diagnoses, do not assume that the cause of agitation is psychiatric.
Continue to: Psychiatric causes
Psychiatric causes
Autism spectrum disorder. Children and teens with autism often feel overwhelmed due to transitions, changes, and/or sensory overload. This sensory overload may be in response to relatively subtle sensory stimuli, so it may not always be apparent to parents or others around them.
Research suggests that in general, the ability to cope effectively with emotions is difficult without optimal language development. Due to cognitive/language delays and a related lack of emotional attunement and limited skills in recognizing, expressing, or coping with emotions, difficult emotions in children and adolescents with autism can manifest as agitation.
Attention-deficit/hyperactivity disorder. Children with ADHD may be at a higher risk for agitation, in part due to poor impulse control and limited coping skills. In addition, chronic negative feedback (from parents, teachers, or both) may contribute to low self-esteem, mood symptoms, defiance, and/or other behavioral difficulties. In addition to standard pharmacotherapy for ADHD, treatment involves parent behavior modification training. Setting firm yet empathic limits, “picking battles,” and implementing a developmentally appropriate behavioral plan to manage disruptive behavior in children or adolescents with ADHD can go a long way in helping to prevent the emergence of agitation.
Posttraumatic stress disorder. In some young children, new-onset, unexplained agitation may be the only sign of abuse or trauma. Children who have undergone trauma tend to experience confusion and distress. This may manifest as agitation or aggression, or other symptoms such as increased anxiety or nightmares.5 Trauma may be in the form of witnessing violence (domestic or other); experiencing physical, sexual, and/or emotional abuse; or witnessing/experiencing other significant threats to the safety of self and/or loved ones. Re-establishing (or establishing) a sense of psychological and physical safety is paramount in such patients.6 Psychotherapy is the first-line modality of treatment in children and adolescents with PTSD.6 In general, there is a scarcity of research on medication treatments for PTSD symptoms among children and adolescents.6
Oppositional defiant disorder/conduct disorder. Oppositional defiant disorder (ODD) can be comorbid with ADHD. The diagnosis of ODD requires a pervasive pattern of anger, defiance, vindictiveness, and hostility, particularly towards authority figures. However, these symptoms need to be differentiated from the normal range of childhood behavior. Occasionally, children learn to cope maladaptively through disruptive behavior or agitation. Although a parent or caregiver may see this behavior as intentionally malevolent, in a child with limited coping skills (whether due to young age, developmental/cognitive/language/learning delays, or social communication deficits) or one who has witnessed frequent agitation or aggression in the family environment, agitation and disruptive behavior may be a maladaptive form of coping. Thus, diligence needs to be exercised in the diagnosis of ODD and in understanding the psychosocial factors affecting the child, particularly because impulsiveness and uncooperativeness on their own have been found to be linked to greater likelihood of prescription of psychotropic medications from multiple classes.7 Family-based interventions, particularly parent training, family therapy, and age-appropriate child skills training, are of prime importance in managing this condition.8 Research shows that a shortage of resources, system issues, and cultural roadblocks in implementing family-based psychosocial interventions also can contribute to the increased use of psychotropic medications for aggression in children and teens with ODD, conduct disorder, or ADHD.8 The astute clinician needs to be cognizant of this before prescribing.
Continue to: Hallucinations/psychosis
Hallucinations/psychosis. Hallucinations (whether from psychiatric or medical causes) are significantly associated with agitation.9 In particular, auditory command hallucinations have been linked to agitation. Command hallucinations in children and adolescents may be secondary to early-onset schizophrenia; however, this diagnosis is rare.10 Hallucinations can also be an adverse effect of amphetamine-based stimulant medications in children and adolescents. Visual hallucinations are most often a sign of an underlying medical disorder such as delirium, occipital lobe mass/infection, or drug intoxication or withdrawal. Hallucinations need to be distinguished from the normal, imaginative play of a young child.10
Bipolar mania. In adults, bipolar disorder is a primary psychiatric cause of agitation. In children and adolescents, the diagnosis of bipolar disorder can be complex and requires careful and nuanced history-taking. The risks of agitation are greater with bipolar disorder than with unipolar depression.11,12
Disruptive mood dysregulation disorder. Prior to DSM-5, many children and adolescents with chronic, non-episodic irritability and severe outbursts out of proportion to the situation or stimuli were given a diagnosis of bipolar disorder. These symptoms, in combination with other symptoms, are now considered part of disruptive mood dysregulation disorder when severe outbursts in a child or adolescent occur 3 to 4 times a week consistently, for at least 1 year. The diagnosis of disruptive mood dysregulation disorder requires ruling out other psychiatric and medical conditions, particularly ADHD.13
Substance intoxication/withdrawal. Intoxication or withdrawal from substances such as alcohol, stimulant medications, opioids, methamphetamines, and other agents can lead to agitation. This is more likely to occur among adolescents than children.14
Adjustment disorder. Parental divorce, especially if it is conflictual, or other life stressors, such as experiencing a move or frequent moves, may contribute to the development of agitation in children and adolescents.
Continue to: Depression
Depression. In children and adolescents, depression can manifest as anger or irritability, and occasionally as agitation.
Medical causes
Delirium. Refractory agitation is often a manifestation of delirium in children and adolescents.15 If unrecognized and untreated, delirium can be fatal.16 Therefore, it is imperative that clinicians routinely assess for delirium in any patient who presents with agitation.
Because a patient with delirium often presents with agitation and visual or auditory hallucinations, the medical team may tend to assume these symptoms are secondary to a psychiatric disorder. In this case, the role of the consultation-liaison psychiatrist is critical for guiding the medical team, particularly to continue a thorough exploration of underlying causes while avoiding polypharmacy. Noise, bright lights, frequent changes in nursing staff or caregivers, anticholinergic or benzodiazepine medications, and frequent changes in schedules should be avoided to prevent delirium from occurring or getting worse.17 A multidisciplinary team approach is key in identifying the underlying cause and managing delirium in pediatric patients.
Traumatic brain injury. Agitation may be a presenting symptom in youth with traumatic brain injury (TBI).18 Agitation may present often in the acute recovery phase.19 There is limited evidence on the efficacy and safety of pharmacotherapy for agitation in pediatric patients with TBI.18
Autoimmune conditions. In a study of 27 patients with
Continue to: Medication-induced/iatrogenic
Medication-induced/iatrogenic. Agitation can be an adverse effect of medications such as amantadine (often used for TBI),18 atypical antipsychotics,21 selective serotonin reuptake inhibitors, and serotonin-norepinephrine reuptake inhibitors.
Infection. Agitation can be a result of encephalitis, meningitis, or other infectious processes.22
Metabolic conditions. Hepatic or renal failure, diabetic ketoacidosis, and thyroid toxicosis may cause agitation in children or adolescents.22
Start with nonpharmacologic interventions
Few studies have examined de-escalation techniques in agitated children and adolescents. However, verbal de-escalation is generally viewed as the first-line technique for managing agitation in children and adolescents. When feasible, teaching and modeling developmentally appropriate stress management skills for children and teens can be a beneficial preventative strategy to reduce the incidence and worsening of agitation.23
Clinicians should refrain from using coercion.24 Coercion could harm the therapeutic alliance, thereby impeding assessment of the underlying causes of agitation, and can be particularly harmful for patients who have a history of trauma or abuse. Even in pediatric patients with no such history, coercion is discouraged due to its punitive connotations and potential to adversely impact a vulnerable child or teen.
Continue to: Establishing a therapeutic rapport...
Establishing a therapeutic rapport with the patient, when feasible, can facilitate smoother de-escalation by offering the patient an outlet to air his/her frustrations and emotions, and by helping the patient feel understood.24 To facilitate this, ensure that the patient’s basic comforts and needs are met, such as access to a warm bed, food, and safety.25
The psychiatrist’s role is to help uncover and address the underlying reason for the patient’s agony or distress. Once the child or adolescent has calmed, explore potential triggers or causes of the agitation.
There has been a significant move away from the use of restraints for managing agitation in children and adolescents.26 Restraints have a psychologically traumatizing effect,27 and have been linked to life-threatening injuries and death in children.24
Pharmacotherapy: Proceed with caution
There are no FDA-approved medications for the treatment of agitation in the general pediatric population, and any medication use in this population is off-label. There is also a dearth of research examining the safety and efficacy of using psychotropic medications for agitation in pediatric patients. Because children and adolescents are more susceptible to adverse effects and risks associated with the use of psychotropic medications, special caution is warranted. In general, pharmacologic interventions are not recommended without the use of psychotherapy-based modalities.
In the past, the aim of using medications to treat patients with agitation was to put the patient to sleep.25 This practice did not help clinicians to assess for underlying causes, and was often accompanied by a greater risk of adverse effects and reactions.24 Therefore, the goal of medication treatment for agitation is to help calm the patient instead of inducing sleep.25
Continue to: Pharmacotherapy should...
Pharmacotherapy should be used only when behavioral interventions have been unsuccessful. Key considerations for using psychotropic medications to address agitation in children and adolescents are summarized in Table 2.25
Antipsychotics, particularly second-generation antipsychotics (SGAs), have been commonly used to manage acute agitation in children and adolescents, and there has been an upswing in the use of these medications in the United States in the last several years.28 Research indicates that males, children and adolescents in foster care, and those with Medicaid have been the more frequent youth recipients of SGAs.29 Of particular concern is the prevalence of antipsychotic use among children younger than age 6. In the last few decades, there has been an increase in the prescription of antipsychotics for children younger than age 6, particularly for disruptive behavior and aggression.30 In a study of preschool-age Medicaid patients in Kentucky, 70,777 prescriptions for SGAs were given to 6,915 children <6 years of age; 73% of these prescriptions were for male patients.30 Because there is a lack of controlled studies examining the safety and efficacy of SGAs among children and adolescents, especially with long-term use, further research is needed and caution is warranted.28
The FDA has approved
Externalizing disorders among children and adolescents tend to get treated with antipsychotics.28 A Canadian study examining records of 6,916 children found that most children who had been prescribed risperidone received it for ADHD or conduct disorder, and most patients had not received laboratory testing for monitoring the antipsychotic medication they were taking.31 In a 2018 study examining medical records of 120 pediatric patients who presented to an ED in British Columbia with agitation, antipsychotics were the most commonly used medications for patients with autism spectrum disorder; most patients received at least 1 dose.14
For children and adolescents with agitation or aggression who were admitted to inpatient units, IM
Continue to: In case reports...
In case reports, a combination of olanzapine with CNS-suppressing agents has resulted in death. Therefore, do not combine olanzapine with agents such as benzodiazepines.25 In a patient with a likely medical source of agitation, insufficient evidence exists to support the use of olanzapine, and additional research is needed.25
Low-dose haloperidol has been found to be effective for delirium-related agitation in pediatric studies.15 Before initiating an antipsychotic for any child or adolescent, review the patient’s family history for reports of early cardiac death and the patient’s own history of cardiac symptoms, palpitations, syncope, or prolonged QT interval. Monitor for QT prolongation. Among commonly used antipsychotics, the risk of QT prolongation is higher with IV administration of haloperidol and with ziprasidone. Studies show that compared with oral or IM
A few studies have found risperidone to be efficacious for treating ODD and conduct disorder; however, this use is off-label, and its considerable adverse effect and risk profile needs to be weighed against the potential benefit.8
Antipsychotic polypharmacy should be avoided because of the higher risk of adverse effects and interactions, and a lack of robust, controlled studies evaluating the safety of using antipsychotics for non-FDA-approved indications in children and adolescents.7 All patients who receive antipsychotics require monitoring for extrapyramidal symptoms, tardive dyskinesia, neuroleptic malignant syndrome, orthostatic hypotension, sedation, metabolic syndrome, and other potential adverse effects. Patients receiving risperidone need to have their prolactin levels monitored periodically, and their parents should be made aware of the potential for hyperprolactinemia and other adverse effects. Aripiprazole and quetiapine may increase the risk of suicidality.
Antiepileptics. A meta-analysis of 7 randomized controlled trials examining the use of antiepileptic medications (
Continue to: In a retrospective case series...
In a retrospective case series of 30 pediatric patients with autism spectrum disorder who were given oxcarbazepine, Douglas et al35 found that 47% of participants experienced significant improvement in irritability/agitation. However, 23% of patients reported significant adverse effects leading to discontinuation. Insufficient evidence exists for the safety and efficacy of oxcarbazepine in this population.35
Benzodiazepines. The use of benzodiazepines in pediatric patients has been associated with paradoxical disinhibition reactions, particularly in children with autism and other developmental or cognitive disabilities or delays.21 There is a lack of data on the safety and efficacy of long-term use of benzodiazepines in children, especially in light of these patients’ developing brains, the risk of cognitive impairment, and the potential for dependence with long-term use. Despite this, some studies show that the use of benzodiazepines is fairly common among pediatric patients who present to the ED with agitation.14 In a recent retrospective study, Kendrick et al14 found that among pediatric patients with agitation who were brought to the ED, benzodiazepines were the most commonly prescribed medications.
Other medications. Clonidine and
Diphenhydramine, in both oral and IM forms, has been used to treat agitation in children,32 but has also been associated with a paradoxical disinhibition reaction in pediatric patients21 and therefore should be used only sparingly and with caution. Diphenhydramine has anticholinergic properties, and may worsen delirium.15 Stimulant medications can help aggressive behavior in children and adolescents with ADHD.37
Bottom Line
Agitation among children and adolescents has many possible causes. A combination of a comprehensive assessment and evidence-based, judicious treatment interventions can help prevent and manage agitation in this vulnerable population.
Related Resources
- Baker M, Carlson GA. What do we really know about PRN use in agitated children with mental health conditions: a clinical review. Evid Based Ment Health. 2018;21(4):166-170.
- Gerson R, Malas N, Mroczkowski MM. Crisis in the emergency department: the evaluation and management of acute agitation in children and adolescents. Child Adolesc Psychiatr Clin N Am. 2018;27(3):367-386.
Drug Brand Names
Amantadine • Symmetrel
Aripiprazole • Abilify
Clonidine • Catapres
Guanfacine • Intuniv, Tenex
Haloperidol • Haldol
Lamotrigine • Lamictal
Levetiracetam • Keppra, Spritam
Olanzapine • Zyprexa
Oxcarbazepine • Trileptal
Quetiapine • Seroquel
Topiramate • Topamax
Risperidone • Risperdal
Valproate • Depakene
Ziprasidone • Geodon
1. Frosch E, Kelly P. Issues in pediatric psychiatric emergency care. In: Emergency psychiatry. Cambridge, UK: Cambridge University Press; 2011:185-199.
2. American College of Emergency Physicians. Pediatric mental health emergencies in the emergency department. https://www.acep.org/patient-care/policy-statements/pediatric-mental-health-emergencies-in-the-emergency-medical-services-system/. Revised September 2018. Accessed February 23, 2019.
3. Voepel-Lewis, T, Burke C, Hadden S, et al. Nurses’ diagnoses and treatment decisions regarding care of the agitated child. J Perianesth Nurs. 2005;20(4):239-248.
4. Malas N, Spital L, Fischer J, et al. National survey on pediatric acute agitation and behavioral escalation in academic inpatient pediatric care settings. Psychosomatics. 2017;58(3):299-306.
5. Famularo R, Kinscherff R, Fenton T. Symptom differences in acute and chronic presentation of childhood post-traumatic stress disorder. Child Abuse Negl. 1990;14(3):439-444.
6. Kaminer D, Seedat S, Stein DJ. Post-traumatic stress disorder in children. World Psychiatry. 2005;4(2):121-125.
7. Ninan A, Stewart SL, Theall LA, et al. Adverse effects of psychotropic medications in children: predictive factors. J Can Acad Child Adolesc Psychiatry. 2014;23(3):218-225.
8. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 2: antipsychotics and traditional mood stabilizers. Can J Psychiatry. 2015;60(2):52-61.
9. Vareilles D, Bréhin C, Cortey C, et al. Hallucinations: Etiological analysis of children admitted to a pediatric emergency department. Arch Pediatr. 2017;24(5):445-452.
10. Bartlett J. Childhood-onset schizophrenia: what do we really know? Health Psychol Behav Med. 2014;2(1):735-747.
11. Diler RS, Goldstein TR, Hafeman D, et al. Distinguishing bipolar depression from unipolar depression in youth: Preliminary findings. J Child Adolesc Psychopharmacol. 2017;27(4):310-319.
12. Dervic K, Garcia-Amador M, Sudol K, et al. Bipolar I and II versus unipolar depression: clinical differences and impulsivity/aggression traits. Eur Psychiatry. 2015;30(1):106-113.
13. Masi L, Gignac M ADHD and DMDD comorbidities, similarities and distinctions. J Child Adolesc Behav2016;4:325.
14. Kendrick JG, Goldman RD, Carr RR. Pharmacologic management of agitation and aggression in a pediatric emergency department - a retrospective cohort study. J Pediatr Pharmacol Ther. 2018;23(6):455-459.
15. Schieveld JN, Staal M, Voogd L, et al. Refractory agitation as a marker for pediatric delirium in very young infants at a pediatric intensive care unit. Intensive Care Med. 2010;36(11):1982-1983.
16. Traube C, Silver G, Gerber LM, et al. Delirium and mortality in critically ill children: epidemiology and outcomes of pediatric delirium. Crit Care Med. 2017;45(5):891-898.
17. Bettencourt A, Mullen JE. Delirium in children: identification, prevention, and management. Crit Care Nurse. 2017;37(3):e9-e18.
18. Suskauer SJ, Trovato MK. Update on pharmaceutical intervention for disorders of consciousness and agitation after traumatic brain injury in children. PM R. 2013;5(2):142-147.
19. Nowicki M, Pearlman L, Campbell C, et al. Agitated behavior scale in pediatric traumatic brain injury. Brain Inj. 2019. doi: 10.1080/02699052.2019.1565893.
20. Mohammad SS, Jones H, Hong M, et al. Symptomatic treatment of children with anti-NMDAR encephalitis. Dev Med Child Neurol. 2016;58(4):376-384.
21. Sonnier L, Barzman D. Pharmacologic management of acutely agitated pediatric patients. Pediatr Drugs. 2011;13(1):1-10.
22. Nordstrom K, Zun LS, Wilson MP, et al. Medical evaluation and triage of the agitated patient: consensus statement of the american association for emergency psychiatry project Beta medical evaluation workgroup. West J Emerg Med. 2012;13(1):3-10.
23. Masters KJ, Bellonci C, Bernet W, et al; American Academy of Child and Adolescent Psychiatry. Practice parameter for the prevention and management of aggressive behavior in child and adolescent psychiatric institutions, with special reference to seclusion and restraint. J Am Acad Child Adolesc Psychiatry. 2002;41(2 suppl):4S-25S.
24. Croce ND, Mantovani C. Using de-escalation techniques to prevent violent behavior in pediatric psychiatric emergencies: It is possible. Pediatric Dimensions, 2017;2(1):1-2.
25. Marzullo LR. Pharmacologic management of the agitated child. Pediatr Emerg Care. 2014;30(4):269-275.
26. Caldwell B, Albert C, Azeem MW, et al. Successful seclusion and restraint prevention effort in child and adolescent programs. J Psychosoc Nurs Ment Health Serv. 2014;52(11):30-38.
27. De Hert M, Dirix N, Demunter H, et al. Prevalence and correlates of seclusion and restraint use in children and adolescents: a systematic review. Eur Child Adolesc Psychiatry. 2011;20(5):221-230.
28. Crystal S, Olfson M, Huang C, et al. Broadened use of atypical antipsychotics: safety, effectiveness, and policy challenges. Health Aff (Millwood). 2009;28(5):w770-w781.
29. American Academy of Child and Adolescent Psychiatry. Practice parameters for the use of atypical antipsychotic medication in children and adolescents. https://www.aacap.org/App_Themes/AACAP/docs/practice_parameters/Atypical_Antipsychotic_Medications_Web.pdf. Accessed March 4, 2019.
30. Lohr WD, Chowning RT, Stevenson MD, et al. Trends in atypical antipsychotics prescribed to children six years of age or less on Medicaid in Kentucky. J Child Adolesc Psychopharmacol. 2015;25(5):440-443.
31. Chen W, Cepoiu-Martin M, Stang A, et al. Antipsychotic prescribing and safety monitoring practices in children and youth: a population-based study in Alberta, Canada. Clin Drug Investig. 2018;38(5):449-455.
32. Deshmukh P, Kulkarni G, Barzman D. Recommendations for pharmacological management of inpatient aggression in children and adolescents. Psychiatry (Edgmont). 2010;7(2):32-40.
33. Haldol [package insert]. Beerse, Belgium: Janssen Pharmaceutica NV; 2005.
34. Hirota T, Veenstra-Vanderweele J, Hollander E, et al. Antiepileptic medications in autism spectrum disorder: a systematic review and meta-analysis. J Autism Dev Disord. 2014;44(4):948-957.
35. Douglas JF, Sanders KB, Benneyworth MH, et al. Brief report: retrospective case series of oxcarbazepine for irritability/agitation symptoms in autism spectrum disorder. J Autism Dev Disord. 2013;43(5):1243-1247.
36. Harmon RJ, Riggs PD. Clonidine for posttraumatic stress disorder in preschool children. J Am Acad
37. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 1: Psychostimulants, alpha-2 Agonists, and atomoxetine. Can J Psychiatry. 2015;60(2):42-51
1. Frosch E, Kelly P. Issues in pediatric psychiatric emergency care. In: Emergency psychiatry. Cambridge, UK: Cambridge University Press; 2011:185-199.
2. American College of Emergency Physicians. Pediatric mental health emergencies in the emergency department. https://www.acep.org/patient-care/policy-statements/pediatric-mental-health-emergencies-in-the-emergency-medical-services-system/. Revised September 2018. Accessed February 23, 2019.
3. Voepel-Lewis, T, Burke C, Hadden S, et al. Nurses’ diagnoses and treatment decisions regarding care of the agitated child. J Perianesth Nurs. 2005;20(4):239-248.
4. Malas N, Spital L, Fischer J, et al. National survey on pediatric acute agitation and behavioral escalation in academic inpatient pediatric care settings. Psychosomatics. 2017;58(3):299-306.
5. Famularo R, Kinscherff R, Fenton T. Symptom differences in acute and chronic presentation of childhood post-traumatic stress disorder. Child Abuse Negl. 1990;14(3):439-444.
6. Kaminer D, Seedat S, Stein DJ. Post-traumatic stress disorder in children. World Psychiatry. 2005;4(2):121-125.
7. Ninan A, Stewart SL, Theall LA, et al. Adverse effects of psychotropic medications in children: predictive factors. J Can Acad Child Adolesc Psychiatry. 2014;23(3):218-225.
8. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 2: antipsychotics and traditional mood stabilizers. Can J Psychiatry. 2015;60(2):52-61.
9. Vareilles D, Bréhin C, Cortey C, et al. Hallucinations: Etiological analysis of children admitted to a pediatric emergency department. Arch Pediatr. 2017;24(5):445-452.
10. Bartlett J. Childhood-onset schizophrenia: what do we really know? Health Psychol Behav Med. 2014;2(1):735-747.
11. Diler RS, Goldstein TR, Hafeman D, et al. Distinguishing bipolar depression from unipolar depression in youth: Preliminary findings. J Child Adolesc Psychopharmacol. 2017;27(4):310-319.
12. Dervic K, Garcia-Amador M, Sudol K, et al. Bipolar I and II versus unipolar depression: clinical differences and impulsivity/aggression traits. Eur Psychiatry. 2015;30(1):106-113.
13. Masi L, Gignac M ADHD and DMDD comorbidities, similarities and distinctions. J Child Adolesc Behav2016;4:325.
14. Kendrick JG, Goldman RD, Carr RR. Pharmacologic management of agitation and aggression in a pediatric emergency department - a retrospective cohort study. J Pediatr Pharmacol Ther. 2018;23(6):455-459.
15. Schieveld JN, Staal M, Voogd L, et al. Refractory agitation as a marker for pediatric delirium in very young infants at a pediatric intensive care unit. Intensive Care Med. 2010;36(11):1982-1983.
16. Traube C, Silver G, Gerber LM, et al. Delirium and mortality in critically ill children: epidemiology and outcomes of pediatric delirium. Crit Care Med. 2017;45(5):891-898.
17. Bettencourt A, Mullen JE. Delirium in children: identification, prevention, and management. Crit Care Nurse. 2017;37(3):e9-e18.
18. Suskauer SJ, Trovato MK. Update on pharmaceutical intervention for disorders of consciousness and agitation after traumatic brain injury in children. PM R. 2013;5(2):142-147.
19. Nowicki M, Pearlman L, Campbell C, et al. Agitated behavior scale in pediatric traumatic brain injury. Brain Inj. 2019. doi: 10.1080/02699052.2019.1565893.
20. Mohammad SS, Jones H, Hong M, et al. Symptomatic treatment of children with anti-NMDAR encephalitis. Dev Med Child Neurol. 2016;58(4):376-384.
21. Sonnier L, Barzman D. Pharmacologic management of acutely agitated pediatric patients. Pediatr Drugs. 2011;13(1):1-10.
22. Nordstrom K, Zun LS, Wilson MP, et al. Medical evaluation and triage of the agitated patient: consensus statement of the american association for emergency psychiatry project Beta medical evaluation workgroup. West J Emerg Med. 2012;13(1):3-10.
23. Masters KJ, Bellonci C, Bernet W, et al; American Academy of Child and Adolescent Psychiatry. Practice parameter for the prevention and management of aggressive behavior in child and adolescent psychiatric institutions, with special reference to seclusion and restraint. J Am Acad Child Adolesc Psychiatry. 2002;41(2 suppl):4S-25S.
24. Croce ND, Mantovani C. Using de-escalation techniques to prevent violent behavior in pediatric psychiatric emergencies: It is possible. Pediatric Dimensions, 2017;2(1):1-2.
25. Marzullo LR. Pharmacologic management of the agitated child. Pediatr Emerg Care. 2014;30(4):269-275.
26. Caldwell B, Albert C, Azeem MW, et al. Successful seclusion and restraint prevention effort in child and adolescent programs. J Psychosoc Nurs Ment Health Serv. 2014;52(11):30-38.
27. De Hert M, Dirix N, Demunter H, et al. Prevalence and correlates of seclusion and restraint use in children and adolescents: a systematic review. Eur Child Adolesc Psychiatry. 2011;20(5):221-230.
28. Crystal S, Olfson M, Huang C, et al. Broadened use of atypical antipsychotics: safety, effectiveness, and policy challenges. Health Aff (Millwood). 2009;28(5):w770-w781.
29. American Academy of Child and Adolescent Psychiatry. Practice parameters for the use of atypical antipsychotic medication in children and adolescents. https://www.aacap.org/App_Themes/AACAP/docs/practice_parameters/Atypical_Antipsychotic_Medications_Web.pdf. Accessed March 4, 2019.
30. Lohr WD, Chowning RT, Stevenson MD, et al. Trends in atypical antipsychotics prescribed to children six years of age or less on Medicaid in Kentucky. J Child Adolesc Psychopharmacol. 2015;25(5):440-443.
31. Chen W, Cepoiu-Martin M, Stang A, et al. Antipsychotic prescribing and safety monitoring practices in children and youth: a population-based study in Alberta, Canada. Clin Drug Investig. 2018;38(5):449-455.
32. Deshmukh P, Kulkarni G, Barzman D. Recommendations for pharmacological management of inpatient aggression in children and adolescents. Psychiatry (Edgmont). 2010;7(2):32-40.
33. Haldol [package insert]. Beerse, Belgium: Janssen Pharmaceutica NV; 2005.
34. Hirota T, Veenstra-Vanderweele J, Hollander E, et al. Antiepileptic medications in autism spectrum disorder: a systematic review and meta-analysis. J Autism Dev Disord. 2014;44(4):948-957.
35. Douglas JF, Sanders KB, Benneyworth MH, et al. Brief report: retrospective case series of oxcarbazepine for irritability/agitation symptoms in autism spectrum disorder. J Autism Dev Disord. 2013;43(5):1243-1247.
36. Harmon RJ, Riggs PD. Clonidine for posttraumatic stress disorder in preschool children. J Am Acad
37. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 1: Psychostimulants, alpha-2 Agonists, and atomoxetine. Can J Psychiatry. 2015;60(2):42-51
Providing psychotherapy? Keep these principles in mind
Although the biological aspects of psychiatry are crucial, psychotherapy is an integral part of psychiatry. Unfortunately, the emphasis on psychotherapy training in psychiatry residency programs has declined compared with a decade or more ago. In an era of dwindling psychotherapy training and resources, the quality and type of psychotherapy training has become more variable. In addition to helping maintain the therapeutic alliance, nuanced psychotherapy by a trained professional can be transformational by helping patients to:
- process complex life events and emotions
- feel understood
- overcome psychological barriers to recovery
- enhance self-esteem.
When providing psychotherapy for adult patients, consider these basic, but salient points that are often overlooked.
Refrain from making life decisions for patients, except in exceptional circumstances, such as in situations of abuse and other crises.1 Telling an adult patient what to do about life decisions that he finds challenging fits more under life coaching than psychotherapy. Through therapy, patients should be helped in processing the pros and cons of certain decisions and in navigating the decision-making process to arrive at a decision that makes the most sense to them. Also, it’s not uncommon for therapeutic relationships to rupture when therapists give advice such as suggesting that a patient divorce his spouse, date a certain individual, or have children.
There are many reasons why giving advice in psychotherapy is not recommended. Giving advice can be an impediment to the therapeutic process.2 What is good advice for one patient may not be good for another. Therapists who give advice often do so from their own lens and perspective. This perspective may not only be different from the patient’s priorities and life circumstances, but the therapist also may have inadequate information about the patient’s situation,1,2 which could lead to providing advice that could even harm the patient. In addition, providing advice might prevent a patient from gaining adequate agency or self-directedness while promoting an unhealthy dependence on the therapist and reinforcing the patient’s self-doubt or lack of confidence. In these cases, the patient may later resent the therapist for the advice.
Address the ‘here and now.’1 Pay attention to immediate issues or themes that emerge, and address them with the patient gently and thoughtfully, as appropriate. Ignoring these may create risks of missing vital, underlying material that could reveal more of the patient’s inner world, as these themes can sometimes reflect other themes of the patient’s life outside of treatment.
Acknowledging and empathizing, when appropriate, are key initial steps that help decrease resistance and facilitate the therapeutic process.
Explore the affect. Paying attention to the patient’s emotional state is critical.3 This holds true for all types of psychotherapy. For example, if a patient suddenly becomes tearful when telling his story or describing recent events, this is usually a sign that the subject matter affects or holds value to the patient in a significant or meaningful way and should be further explored.
‘Meet the patient where they are.’ This doesn’t mean you should yield to the patient or give in to his demands. It implies that you should assess the patient’s readiness for a particular intervention and devise interventions from that standpoint, exploring the patient’s ambivalence, noticing resistance, and continuing to acknowledge and empathize with where the patient is in life or treatment. When utilized judiciously, this technique can help the therapist align with the patient, and help the patient move forward through resistance and ambivalence.
Be nonjudgmental and empathetic. Patients place trust in their therapists when they disclose thoughts or emotions that are sensitive, meaningful, or close to the heart. A nonjudgmental response helps the patient accept his experiences and emotions. Being empathetic requires putting oneself in another’s shoes; it does not mean agreeing with the patient. Of course, if you learn that your patient abused a child or an older adult, you are required to report it to the appropriate state agency. In addition, follow the duty to warn and protect in case of any other safety issues, as appropriate.
Do not assume. Open-ended questions and exploration are key. For example, a patient told her resident therapist that her father recently passed away. The therapist expressed to the patient how hard this must be for her. However, the patient said she was relieved by her father’s death, because he had been abusive to her for years. Because of the therapist’s comment, the patient doubted her own reaction and felt guilty for not being more upset about her father’s death.
Avoid over-identifying with your patient. If you find yourself over-identifying with a patient because you have a common background or life events, seek supervision. Over-identification not only can pose barriers to objectively identifying patterns and trends in the patient’s behavior or presentation but also can increase the risk of crossing boundaries or even minimizing the patient’s experience. Exercise caution if you find yourself wanting to be liked by your patient; this is a common mistake among beginning therapists.4
Seek supervision. If you are feeling angry, frustrated, indifferent, or overly attached toward a patient, recognize this countertransference and seek consultation or supervision from an experienced colleague or supervisor. These emotions can be valuable tools that shed light not only on the patient’s life and the session itself, but also help you identify any other factors, such as your own feelings or experiences, that might be contributing to these reactions.
1. Yalom ID. The gift of therapy: an open letter to a new generation of therapists and their patients. New York, NY: HarperCollins Publishers; 2002:46-73,142-145.
2. Bender S, Messner E. Management of impasses. In: Bender S, Messner E. Becoming a therapist: what do I say, and why? New York, NY: The Guilford Press; 2003:235-258.
3.
4. Buckley P, Karasu TB, Charles E. Common mistakes in psychotherapy. Am J Psychiatry. 1979;136(12):1578-1580.
Although the biological aspects of psychiatry are crucial, psychotherapy is an integral part of psychiatry. Unfortunately, the emphasis on psychotherapy training in psychiatry residency programs has declined compared with a decade or more ago. In an era of dwindling psychotherapy training and resources, the quality and type of psychotherapy training has become more variable. In addition to helping maintain the therapeutic alliance, nuanced psychotherapy by a trained professional can be transformational by helping patients to:
- process complex life events and emotions
- feel understood
- overcome psychological barriers to recovery
- enhance self-esteem.
When providing psychotherapy for adult patients, consider these basic, but salient points that are often overlooked.
Refrain from making life decisions for patients, except in exceptional circumstances, such as in situations of abuse and other crises.1 Telling an adult patient what to do about life decisions that he finds challenging fits more under life coaching than psychotherapy. Through therapy, patients should be helped in processing the pros and cons of certain decisions and in navigating the decision-making process to arrive at a decision that makes the most sense to them. Also, it’s not uncommon for therapeutic relationships to rupture when therapists give advice such as suggesting that a patient divorce his spouse, date a certain individual, or have children.
There are many reasons why giving advice in psychotherapy is not recommended. Giving advice can be an impediment to the therapeutic process.2 What is good advice for one patient may not be good for another. Therapists who give advice often do so from their own lens and perspective. This perspective may not only be different from the patient’s priorities and life circumstances, but the therapist also may have inadequate information about the patient’s situation,1,2 which could lead to providing advice that could even harm the patient. In addition, providing advice might prevent a patient from gaining adequate agency or self-directedness while promoting an unhealthy dependence on the therapist and reinforcing the patient’s self-doubt or lack of confidence. In these cases, the patient may later resent the therapist for the advice.
Address the ‘here and now.’1 Pay attention to immediate issues or themes that emerge, and address them with the patient gently and thoughtfully, as appropriate. Ignoring these may create risks of missing vital, underlying material that could reveal more of the patient’s inner world, as these themes can sometimes reflect other themes of the patient’s life outside of treatment.
Acknowledging and empathizing, when appropriate, are key initial steps that help decrease resistance and facilitate the therapeutic process.
Explore the affect. Paying attention to the patient’s emotional state is critical.3 This holds true for all types of psychotherapy. For example, if a patient suddenly becomes tearful when telling his story or describing recent events, this is usually a sign that the subject matter affects or holds value to the patient in a significant or meaningful way and should be further explored.
‘Meet the patient where they are.’ This doesn’t mean you should yield to the patient or give in to his demands. It implies that you should assess the patient’s readiness for a particular intervention and devise interventions from that standpoint, exploring the patient’s ambivalence, noticing resistance, and continuing to acknowledge and empathize with where the patient is in life or treatment. When utilized judiciously, this technique can help the therapist align with the patient, and help the patient move forward through resistance and ambivalence.
Be nonjudgmental and empathetic. Patients place trust in their therapists when they disclose thoughts or emotions that are sensitive, meaningful, or close to the heart. A nonjudgmental response helps the patient accept his experiences and emotions. Being empathetic requires putting oneself in another’s shoes; it does not mean agreeing with the patient. Of course, if you learn that your patient abused a child or an older adult, you are required to report it to the appropriate state agency. In addition, follow the duty to warn and protect in case of any other safety issues, as appropriate.
Do not assume. Open-ended questions and exploration are key. For example, a patient told her resident therapist that her father recently passed away. The therapist expressed to the patient how hard this must be for her. However, the patient said she was relieved by her father’s death, because he had been abusive to her for years. Because of the therapist’s comment, the patient doubted her own reaction and felt guilty for not being more upset about her father’s death.
Avoid over-identifying with your patient. If you find yourself over-identifying with a patient because you have a common background or life events, seek supervision. Over-identification not only can pose barriers to objectively identifying patterns and trends in the patient’s behavior or presentation but also can increase the risk of crossing boundaries or even minimizing the patient’s experience. Exercise caution if you find yourself wanting to be liked by your patient; this is a common mistake among beginning therapists.4
Seek supervision. If you are feeling angry, frustrated, indifferent, or overly attached toward a patient, recognize this countertransference and seek consultation or supervision from an experienced colleague or supervisor. These emotions can be valuable tools that shed light not only on the patient’s life and the session itself, but also help you identify any other factors, such as your own feelings or experiences, that might be contributing to these reactions.
Although the biological aspects of psychiatry are crucial, psychotherapy is an integral part of psychiatry. Unfortunately, the emphasis on psychotherapy training in psychiatry residency programs has declined compared with a decade or more ago. In an era of dwindling psychotherapy training and resources, the quality and type of psychotherapy training has become more variable. In addition to helping maintain the therapeutic alliance, nuanced psychotherapy by a trained professional can be transformational by helping patients to:
- process complex life events and emotions
- feel understood
- overcome psychological barriers to recovery
- enhance self-esteem.
When providing psychotherapy for adult patients, consider these basic, but salient points that are often overlooked.
Refrain from making life decisions for patients, except in exceptional circumstances, such as in situations of abuse and other crises.1 Telling an adult patient what to do about life decisions that he finds challenging fits more under life coaching than psychotherapy. Through therapy, patients should be helped in processing the pros and cons of certain decisions and in navigating the decision-making process to arrive at a decision that makes the most sense to them. Also, it’s not uncommon for therapeutic relationships to rupture when therapists give advice such as suggesting that a patient divorce his spouse, date a certain individual, or have children.
There are many reasons why giving advice in psychotherapy is not recommended. Giving advice can be an impediment to the therapeutic process.2 What is good advice for one patient may not be good for another. Therapists who give advice often do so from their own lens and perspective. This perspective may not only be different from the patient’s priorities and life circumstances, but the therapist also may have inadequate information about the patient’s situation,1,2 which could lead to providing advice that could even harm the patient. In addition, providing advice might prevent a patient from gaining adequate agency or self-directedness while promoting an unhealthy dependence on the therapist and reinforcing the patient’s self-doubt or lack of confidence. In these cases, the patient may later resent the therapist for the advice.
Address the ‘here and now.’1 Pay attention to immediate issues or themes that emerge, and address them with the patient gently and thoughtfully, as appropriate. Ignoring these may create risks of missing vital, underlying material that could reveal more of the patient’s inner world, as these themes can sometimes reflect other themes of the patient’s life outside of treatment.
Acknowledging and empathizing, when appropriate, are key initial steps that help decrease resistance and facilitate the therapeutic process.
Explore the affect. Paying attention to the patient’s emotional state is critical.3 This holds true for all types of psychotherapy. For example, if a patient suddenly becomes tearful when telling his story or describing recent events, this is usually a sign that the subject matter affects or holds value to the patient in a significant or meaningful way and should be further explored.
‘Meet the patient where they are.’ This doesn’t mean you should yield to the patient or give in to his demands. It implies that you should assess the patient’s readiness for a particular intervention and devise interventions from that standpoint, exploring the patient’s ambivalence, noticing resistance, and continuing to acknowledge and empathize with where the patient is in life or treatment. When utilized judiciously, this technique can help the therapist align with the patient, and help the patient move forward through resistance and ambivalence.
Be nonjudgmental and empathetic. Patients place trust in their therapists when they disclose thoughts or emotions that are sensitive, meaningful, or close to the heart. A nonjudgmental response helps the patient accept his experiences and emotions. Being empathetic requires putting oneself in another’s shoes; it does not mean agreeing with the patient. Of course, if you learn that your patient abused a child or an older adult, you are required to report it to the appropriate state agency. In addition, follow the duty to warn and protect in case of any other safety issues, as appropriate.
Do not assume. Open-ended questions and exploration are key. For example, a patient told her resident therapist that her father recently passed away. The therapist expressed to the patient how hard this must be for her. However, the patient said she was relieved by her father’s death, because he had been abusive to her for years. Because of the therapist’s comment, the patient doubted her own reaction and felt guilty for not being more upset about her father’s death.
Avoid over-identifying with your patient. If you find yourself over-identifying with a patient because you have a common background or life events, seek supervision. Over-identification not only can pose barriers to objectively identifying patterns and trends in the patient’s behavior or presentation but also can increase the risk of crossing boundaries or even minimizing the patient’s experience. Exercise caution if you find yourself wanting to be liked by your patient; this is a common mistake among beginning therapists.4
Seek supervision. If you are feeling angry, frustrated, indifferent, or overly attached toward a patient, recognize this countertransference and seek consultation or supervision from an experienced colleague or supervisor. These emotions can be valuable tools that shed light not only on the patient’s life and the session itself, but also help you identify any other factors, such as your own feelings or experiences, that might be contributing to these reactions.
1. Yalom ID. The gift of therapy: an open letter to a new generation of therapists and their patients. New York, NY: HarperCollins Publishers; 2002:46-73,142-145.
2. Bender S, Messner E. Management of impasses. In: Bender S, Messner E. Becoming a therapist: what do I say, and why? New York, NY: The Guilford Press; 2003:235-258.
3.
4. Buckley P, Karasu TB, Charles E. Common mistakes in psychotherapy. Am J Psychiatry. 1979;136(12):1578-1580.
1. Yalom ID. The gift of therapy: an open letter to a new generation of therapists and their patients. New York, NY: HarperCollins Publishers; 2002:46-73,142-145.
2. Bender S, Messner E. Management of impasses. In: Bender S, Messner E. Becoming a therapist: what do I say, and why? New York, NY: The Guilford Press; 2003:235-258.
3.
4. Buckley P, Karasu TB, Charles E. Common mistakes in psychotherapy. Am J Psychiatry. 1979;136(12):1578-1580.
Dozing off: Examining excessive daytime sleepiness in psychiatric patients
Excessive daytime sleepiness (EDS) is “the inability to maintain wakefulness and alertness during the major waking periods of the day, with sleep occurring unintentionally or at inappropriate times, almost daily for at least 3 months,” according to the American Academy of Sleep Medicine.1 EDS is common, with a prevalence up to 25% to 30% in the general population.1-4 The prevalence rate varies in different studies, primarily because of inconsistent definitions of EDS, and therefore differences in diagnosis and assessment.1,2,4 In a study of 300 psychiatric outpatients, 34% had EDS.3 However, studies and evidence reviewing EDS in psychiatric patients are limited.
The causes of EDS are many and varied,1,8 including medical and psychiatric etiologies. A thorough history, screening at-risk patients, and timely sleep center referral are vital to detect and appropriately manage the cause of EDS.5
This article reviews the literature on EDS, with a focus on the risks of untreated EDS, common etiologies of the condition, as well as a brief description of screening and treatment strategies.
EDS vs fatigue
Many patients describe EDS as “fatigue”1; however, a patient’s report of fatigue could be mistaken for EDS.4 Although there is overlap, it is important for physicians to distinguish between these 2 entities for accurate identification and treatment.1,4
Risk of inadequate screening
A study of 117 patients with symptomatic coronary artery disease showed that EDS is associated with significantly greater incidence of cardiovascular adverse events at 16-month follow up.2 This study had limitations such as small sample size; therefore, more studies are needed. Because of these risks, timely and accurate diagnosis not only improves the patient’s quality of life and reduces polypharmacy but also can be life-saving.
Common causes of EDS in psychiatric patients
Because of the high prevalence and severity of impairments caused by EDS, it is essential for psychiatrists to be informed about causes of EDS and thoroughly assess for the potential underlying etiology before concluding that the sleep problem is a manifestation of the psychiatric disorder and prescribing psychotropic medication for it.
Some common causes of EDS in psychiatric patients include:
Sleep-disordered breathing.8 Obstructive sleep apnea (OSA) is often underdiagnosed,6,7 and considering how common it is,6 psychiatrists likely will see many patients with OSA in their practice.5 OSA has a higher prevalence among patients with psychiatric disorders such as depression6,9 and schizophrenia. Additionally, there is evidence suggesting that patients with OSA are more likely to suffer from depression and EDS than healthy controls6,9,10; some of the proposed mechanisms are sleep fragmentation and hypoxemia.6,9-11 OSA is the most common form of sleep-disordered breathing and is a common cause of EDS.1,2,12 Also, undiagnosed and untreated OSA in patients with depression could cause refractoriness to pharmacological treatment of depression.6,9,10
When unrecognized and untreated, OSA can be life-threatening. Despite this, OSA is not regularly screened for in clinical psychiatric practice.6,10 Therefore, it is imperative that psychiatrists be well-acquainted with measures to identify at-risk patients and refer to a sleep specialist when appropriate.
OSA is accompanied by irritability, cognitive difficulties, and poor sleep, creating an overlap with symptoms of depressive disorders.6,10 Use of sedative hypnotic medications, such as benzodiazepines, which further reduces muscle tone in the airway and suppresses respiratory effort, can worsen OSA symptoms5,6,10 and pose cerebrovascular, cardiovascular, and potentially life-threatening risks, and therefore is not indicated in this population.9,13
Obesity is a risk factor for OSA.6 Patients with mood disorders or schizophrenia or other psychotic disorders are at higher risk of obesity because of psychotropic-induced weight gain, stress-induced mechanisms, and/or lower levels of self-care. When these patients have unrecognized or untreated OSA and are prescribed sedative medications at night or stimulant medications during the day, they could be at increased cardiac or respiratory risks without resolving their underlying condition. A diligent psychiatrist can dramatically reduce the risks by referring a patient for nocturnal polysomnography,1 helping the patient implement lifestyle modifications (eg, exercise, weight loss, and healthy nutrition), prescribing judiciously, and monitoring closely for such risks. An accurate diagnosis of and treatment for OSA can improve sleep6 dramatically and help depressive symptoms through better sleep, more daytime energy and concentration, and adequate oxygenation of the brain while sleeping.
Psychiatrists can screen for OSA using the STOP-Bang (Snoring, Tired, Observed apnea, Pressure, Body mass index, Age, Neck circumference, Gender) Questionnaire, which is a quick, 8-item screening scale that helps to categorize OSA risk as mild, moderate, or severe.12 Hypertension, snoring, and/or gasping for breath (“observed apnea”)—a history which often is provided by spouses or significant others—daytime dozing and/or tiredness, having a large neck circumference or volume, body mass index, male sex, and age are items on the STOP-Bang Questionnaire and also are features that should raise high clinical suspicion of OSA.12 Referral for nocturnal polysomnography in at-risk patients should be the next step1,5 in any sleep-related breathing disorder.
Treatment for OSA involves continuous positive airway pressure (CPAP) therapy, which has been shown to relieve OSA and decrease related EDS.5,6 Other treatment modalities, such as oral appliances and surgery, may be used5 in some cases, but more studies are needed for conclusive results.
Several studies have shown improved depression, mood, and cognition after administering treatment such as CPAP6,9,14 in patients with OSA and depression. Considering the significant risks of cardiovascular,8 cerebrovascular,8 and overall morbidity and mortality associated with untreated OSA,12 it is important to routinely screen for sleep-disordered breathing in patients with depression9 or other psychiatric disorders and refer for specialized sleep evaluation and treatment, when indicated.
Medications. EDS can result from some prescription and over-the-counter medications.1,2,5,7 Sedating antidepressants, antihistamines, antipsychotics, anticonvulsants,1,8 and beta blockers2 could cause sedation, which can persist during daytime, although a few studies did not find an association between antipsychotic use and EDS.3 Benzodiazepines and other sedative-hypnotics,1,7 especially long-acting agents or higher dosages,5 can lead to EDS and decreased alertness. Non-psychotropics, such as opioid pain medications,1,7 antitussives, and skeletal muscle relaxants, also can contribute to or cause daytime sedation.7 When using these agents, psychiatrists should monitor and routinely assess patients while aiming for the lowest effective dosage when feasible.
This strategy creates a framework for psychiatrists to routinely educate patients about these commonly encountered side effects, reduce polypharmacy when possible, and help patients effectively manage or prevent these adverse effects.
Depression.1 Some studies found >45% patients with depression had EDS.3,13,15 Besides an association between depression and EDS,13,16 Chellappa and Araújo13 also found a significant association between EDS and suicidal ideation. The causes of EDS in patients with depression may be varied, ranging from restless legs syndrome, residual depressive symptoms,15 to OSA. Depression is often comorbid with OSA,6 with up to 20% of patients with depression suffering from OSA,10 creating higher risk for EDS. Depressive disorders are routinely assessed during an evaluation of OSA at sleep centers, but OSA often is not screened in psychiatric practice.10
There is a strong need for regular screening for OSA in patients with depression, particularly because most studies show a link between the 2 conditions.10 Both depression and OSA have some common risk factors, such as obesity, hypertension, and metabolic syndrome.10 Patients with these conditions are at greater risk for OSA, and therefore a psychiatrist should proactively screen and refer such patients for nocturnal polysomnography when they suspect OSA. Patients with OSA and depression often present to the psychiatrist with depressive symptoms that appear to be resistant to pharmacological treatment,10 therefore underscoring the importance of screening and ruling out OSA in patients with depression.
Circadian rhythm disorders, restless legs syndrome, alcohol and other substance use, and use of prescription sedative-hypnotics are more common in patients with depression; therefore, this population is at high risk for EDS.
Circadian rhythm disorders and insufficient sleep syndrome. Insufficient sleep syndrome1,2,8 frequently causes EDS and occurs more commonly in busy people who try to get by with less sleep.8 Over time, the effect of sleep loss is cumulative and can be accompanied by mood symptoms, such as irritability, fatigue, and problems with concentration.8 Shift workers1,8 commonly experience insufficient sleep as well as circadian rhythm disorders and EDS. Modafinil is FDA-approved for EDS in shift work sleep disorder.
Geriatric patients may experience advanced sleep phase syndrome involving early awakenings.8 Adolescents, on the other hand, often suffer from delayed sleep phase syndrome, which is a type of circadian rhythm disorder, related to increasing academic and social pressures, natural pubertal shift to later sleep onset, pervading technology use, and often nebulous bedtime routines. This can be a cause of sleep persisting into daytime.8 Taking a careful history and a sleep diary may be useful because this disorder might be confused for insomnia. Treatment involves gradual shifting of the time of sleep onset through bright light exposure and other modalities.8
Adolescents might not be forthcoming about the severity of their sleep problems; therefore, psychiatrists should screen proactively through clinical interviews of patients and parents and consider this possibility when encountering an adolescent with recent-onset attention or cognitive difficulties.
Treatment for circadian rhythm disorders usually includes planned or prescribed sleep scheduling, timed light exposure,8 and occasional use of melatonin or other sedative agents.17
Hypersomnia of central origin, which includes narcolepsy, idiopathic hypersomnia, and recurrent hypersomnia, can present with EDS.1,18,19 Narcolepsy is a rare, debilitating sleep disorder that manifests as EDS or sleep attacks, with or without cataplexy, and sleep paralysis.5,8,18,19 The Multiple Sleep Latency Test and polysomnography are used for diagnosis.1,5 Shortened REM latency is a classic finding often noted on polysomnography. Treatment involves pharmacologic and behavioral strategies and education.5,8 Modafinil is FDA-approved for EDS associated with narcolepsy. Stimulant medications have been used for narcolepsy in the past; further studies are needed to establish benefit–risk ratio of use in this population.18
Kleine-Levin syndrome is a form of recurrent hypersomnia, a less common sleep disorder, characterized by episodes of excessive sleepiness accompanied by hyperphagia and hypersexuality.5,18,19
Other medical conditions,1 such as the rare familial fatal insomnia, neurological conditions1 such as encephalitis,8 epilepsy,8 Alzheimer’s disease or other types of dementia,8 Parkinson’s disease,1 or multiple sclerosis,1,18 can cause excessive daytime fatigue by causing secondary insomnia or hypersomnia.
Treating the underlying disorder is an important first step in these cases. In addition, coordinating with neurologists or other specialists involved in caring for patients with these conditions is important. Regularly reviewing and simplifying the often complex medication regimen, when possible, can go a long way in mitigating EDS in this population.
Other disorders affecting sleep. Restless legs syndrome and periodic limb movement disorder are other causes of EDS.3 Treatment involves lifestyle modifications, iron supplementation in certain patients, and use of dopaminergic agents such as ropinirole, pramipexole, and other medications, depending on severity of the condition, comorbidities, and other factors.20
Alcohol or substance use. Substance use or withdrawal can be associated with sleep disorders, such as hypersomnia,19 insomnia,19 and related EDS.5 For example, alcohol use disorder affects REM sleep, and can cause EDS. Secondary central apnea can be the result of long-standing opioid use19 and can present like EDS.
Insomnia. Primary insomnia rarely causes EDS.5 Insomnia due to a medical or psychiatric condition may be an indirect cause of EDS by causing sleep deprivation.
Steps for timely and accurate diagnosis
Utilize the following steps for facilitating timely diagnosis and treatment of EDS:
Thorough history. Patients often describe “tiredness” instead of sleepiness.8 Therefore, the astute psychiatrist should explore further when patients are presenting with this concern, especially by asking more specific questions such as the tendency to doze off during daytime.8
Family members can be vital sources for obtaining a complete history,5 especially because patients might deny,8 minimize, or not be fully aware1 of the extent of their symptoms. Asking family members about patient’s snoring, irregular breathing, or gasping at night can be particularly valuable.5 Obtaining a family history of sleep disorders can be particularly important, especially in conditions such as OSA and narcolepsy.
Asking about any history of safety issues,8 including sleepiness during driving, cooking, or other activities, is also important.
Use of scales and other screening measures. Psychiatrists can use initial screening measures in the office setting. Epworth Sleepiness Scale15,21 is a validated,2 short, self-administered measure to assess the level of daytime sleepiness; however, it has some limitations such as not being able to measure changes in sleepiness from hour to hour or day to day. Because of its limitations, the Epworth Sleepiness Scale should not be used by itself as a diagnostic tool.3 It has been commonly used for detecting OSA2 and narcolepsy. The Stanford Sleepiness Scale is a self-rating scale that measures the subjective degree of sleepiness and alertness; it has limitations as well, such as having little correlation with chronic sleep loss.8 Other tools such as visual analogue scales also could be helpful.8 For more specialized testing, such as Multiple Sleep Latency Test or polysomnography, referral to a sleep specialist is ideal.8
Education. The assessment is an opportunity for the psychiatrist to educate patients about sleep hygiene, the importance of regular bedtimes, and getting adequate sleep to avoid accumulating a sleep deficit.
Urgent referral of at-risk populations. Prompt or urgent referral of at-risk populations, such as geriatric patients or those with a history of dozing off during driving, is invaluable in preventing morbidity and mortality from untreated sleep disorders.
Patients with severe daytime sleepiness should be advised to not drive or operate heavy machinery until this condition is adequately controlled.18
Bottom Line
1. Chervin RD. Approach to the patient with excessive daytime sleepiness. http://www.uptodate.com/contents/approach-to-the-patient-with-excessive-daytime-sleepiness. Updated January 2016. Accessed June 5, 2017.
2. Lee CH, Ng WY, Hau W, et al. Excessive daytime sleepiness is associated with longer culprit lesion and adverse outcomes in patients with coronary artery disease. J Clin Sleep Med. 2013;9(12):1267-1272.
3. Hawley CJ, Gale TM, Sivakumaran T, et al. Excessive daytime sleepiness in psychiatric disorders: prevalence, correlates and clinical significance. Psychiatry Res. 2010;175(1-2):138-141.
4. Pigeon WR, Sateia MJ, Ferguson RJ. Distinguishing between excessive daytime sleepiness and fatigue: toward improved detection and treatment. J Psychosom Res. 2003;54(1):61-69.
5. Krahn LE. Excessive daytime sleepiness: diagnosing the causes. Current Psychiatry. 2002;1(1):49-57.
6. Ejaz SM, Khawaja IS, Bhatia S, et al. Obstructive sleep apnea and depression: a review. Innov Clin Neurosci. 2011;8(8):17-25.
7. Pagel JF. Excessive daytime sleepiness. Am Fam Physician. 2009;79(5):391-396.
8. Guilleminault C, Brooks SN. Excessive daytime sleepiness: a challenge for the practising neurologist. Brain. 2001;124(pt 8):1482-1491.
9. Cheng P, Casement M, Chen CF, et al. Sleep disordered breathing in major depressive disorder. J Sleep Res. 2013;22(4):459-462.
10. Schröder CM, O’Hara R. Depression and obstructive sleep apnea (OSA). Ann Gen Psychiatry. 2005;4:13.
11. Bardwell WA, Berry CC, Ancoli-Israel S, et al. Psychological correlates of sleep apnea. J Psychosom Res. 1999;47(6):583-596.
12. Chung F, Abdullah HR, Liao P. STOP-Bang Questionnaire: a practical approach to screen for obstructive sleep apnea. Chest. 2016;149(3):631-638.
13. Chellappa SL, Araújo JF. Excessive daytime sleepiness in patients with depressive disorder. Rev Bras Psiquiatr. 2006;28(2):126-129.
14. Habukawa M, Uchimura N, Kakuma T, et al. Effect of CPAP treatment on residual depressive symptoms in patients with major depression and coexisting sleep apnea: contribution of daytime sleepiness to residual depressive symptoms. Sleep Med. 2010;11(6):552-557.
15. Lundt L. Use of the Epworth Sleepiness Scale to evaluate the symptom of excessive sleepiness in major depressive disorder. Gen Hosp Psychiatry. 2005;27(2):146-148.
16. Hawley CJ. Excessive daytime sleepiness in psychiatry: a relevant focus for clinical attention and treatment? Int J Psychiatry Clin Pract. 2006;10(2):117-123.
17. Dodson ER, Zee PC. Therapeutics for circadian rhythm sleep disorders. Sleep Med Clin. 2010;5(4):701-715.
18. Morgenthaler TI, Kapur VK, Brown TM, et al; Standards of Practice Committee of the American Academy of Sleep Medicine. Practice parameters for the treatment of narcolepsy and other hypersomnias of central origin. Sleep. 2007;30(12):1705-1711.
19. Thorpy MJ. Classification of sleep disorders. Neurotherapeutics. 2012;9(4):687-701.
20. National Institute of Neurological Disorders and Stroke. Restless legs syndrome information page. https://www.ninds.nih.gov/Disorders/All-Disorders/Restless-Legs-Syndrome-Information-Page. Accessed June 2, 2017.
21. Johns MW. Reliability and factor analysis of the Epworth Sleepiness Scale. Sleep. 1992;15(4):376-381.
Excessive daytime sleepiness (EDS) is “the inability to maintain wakefulness and alertness during the major waking periods of the day, with sleep occurring unintentionally or at inappropriate times, almost daily for at least 3 months,” according to the American Academy of Sleep Medicine.1 EDS is common, with a prevalence up to 25% to 30% in the general population.1-4 The prevalence rate varies in different studies, primarily because of inconsistent definitions of EDS, and therefore differences in diagnosis and assessment.1,2,4 In a study of 300 psychiatric outpatients, 34% had EDS.3 However, studies and evidence reviewing EDS in psychiatric patients are limited.
The causes of EDS are many and varied,1,8 including medical and psychiatric etiologies. A thorough history, screening at-risk patients, and timely sleep center referral are vital to detect and appropriately manage the cause of EDS.5
This article reviews the literature on EDS, with a focus on the risks of untreated EDS, common etiologies of the condition, as well as a brief description of screening and treatment strategies.
EDS vs fatigue
Many patients describe EDS as “fatigue”1; however, a patient’s report of fatigue could be mistaken for EDS.4 Although there is overlap, it is important for physicians to distinguish between these 2 entities for accurate identification and treatment.1,4
Risk of inadequate screening
A study of 117 patients with symptomatic coronary artery disease showed that EDS is associated with significantly greater incidence of cardiovascular adverse events at 16-month follow up.2 This study had limitations such as small sample size; therefore, more studies are needed. Because of these risks, timely and accurate diagnosis not only improves the patient’s quality of life and reduces polypharmacy but also can be life-saving.
Common causes of EDS in psychiatric patients
Because of the high prevalence and severity of impairments caused by EDS, it is essential for psychiatrists to be informed about causes of EDS and thoroughly assess for the potential underlying etiology before concluding that the sleep problem is a manifestation of the psychiatric disorder and prescribing psychotropic medication for it.
Some common causes of EDS in psychiatric patients include:
Sleep-disordered breathing.8 Obstructive sleep apnea (OSA) is often underdiagnosed,6,7 and considering how common it is,6 psychiatrists likely will see many patients with OSA in their practice.5 OSA has a higher prevalence among patients with psychiatric disorders such as depression6,9 and schizophrenia. Additionally, there is evidence suggesting that patients with OSA are more likely to suffer from depression and EDS than healthy controls6,9,10; some of the proposed mechanisms are sleep fragmentation and hypoxemia.6,9-11 OSA is the most common form of sleep-disordered breathing and is a common cause of EDS.1,2,12 Also, undiagnosed and untreated OSA in patients with depression could cause refractoriness to pharmacological treatment of depression.6,9,10
When unrecognized and untreated, OSA can be life-threatening. Despite this, OSA is not regularly screened for in clinical psychiatric practice.6,10 Therefore, it is imperative that psychiatrists be well-acquainted with measures to identify at-risk patients and refer to a sleep specialist when appropriate.
OSA is accompanied by irritability, cognitive difficulties, and poor sleep, creating an overlap with symptoms of depressive disorders.6,10 Use of sedative hypnotic medications, such as benzodiazepines, which further reduces muscle tone in the airway and suppresses respiratory effort, can worsen OSA symptoms5,6,10 and pose cerebrovascular, cardiovascular, and potentially life-threatening risks, and therefore is not indicated in this population.9,13
Obesity is a risk factor for OSA.6 Patients with mood disorders or schizophrenia or other psychotic disorders are at higher risk of obesity because of psychotropic-induced weight gain, stress-induced mechanisms, and/or lower levels of self-care. When these patients have unrecognized or untreated OSA and are prescribed sedative medications at night or stimulant medications during the day, they could be at increased cardiac or respiratory risks without resolving their underlying condition. A diligent psychiatrist can dramatically reduce the risks by referring a patient for nocturnal polysomnography,1 helping the patient implement lifestyle modifications (eg, exercise, weight loss, and healthy nutrition), prescribing judiciously, and monitoring closely for such risks. An accurate diagnosis of and treatment for OSA can improve sleep6 dramatically and help depressive symptoms through better sleep, more daytime energy and concentration, and adequate oxygenation of the brain while sleeping.
Psychiatrists can screen for OSA using the STOP-Bang (Snoring, Tired, Observed apnea, Pressure, Body mass index, Age, Neck circumference, Gender) Questionnaire, which is a quick, 8-item screening scale that helps to categorize OSA risk as mild, moderate, or severe.12 Hypertension, snoring, and/or gasping for breath (“observed apnea”)—a history which often is provided by spouses or significant others—daytime dozing and/or tiredness, having a large neck circumference or volume, body mass index, male sex, and age are items on the STOP-Bang Questionnaire and also are features that should raise high clinical suspicion of OSA.12 Referral for nocturnal polysomnography in at-risk patients should be the next step1,5 in any sleep-related breathing disorder.
Treatment for OSA involves continuous positive airway pressure (CPAP) therapy, which has been shown to relieve OSA and decrease related EDS.5,6 Other treatment modalities, such as oral appliances and surgery, may be used5 in some cases, but more studies are needed for conclusive results.
Several studies have shown improved depression, mood, and cognition after administering treatment such as CPAP6,9,14 in patients with OSA and depression. Considering the significant risks of cardiovascular,8 cerebrovascular,8 and overall morbidity and mortality associated with untreated OSA,12 it is important to routinely screen for sleep-disordered breathing in patients with depression9 or other psychiatric disorders and refer for specialized sleep evaluation and treatment, when indicated.
Medications. EDS can result from some prescription and over-the-counter medications.1,2,5,7 Sedating antidepressants, antihistamines, antipsychotics, anticonvulsants,1,8 and beta blockers2 could cause sedation, which can persist during daytime, although a few studies did not find an association between antipsychotic use and EDS.3 Benzodiazepines and other sedative-hypnotics,1,7 especially long-acting agents or higher dosages,5 can lead to EDS and decreased alertness. Non-psychotropics, such as opioid pain medications,1,7 antitussives, and skeletal muscle relaxants, also can contribute to or cause daytime sedation.7 When using these agents, psychiatrists should monitor and routinely assess patients while aiming for the lowest effective dosage when feasible.
This strategy creates a framework for psychiatrists to routinely educate patients about these commonly encountered side effects, reduce polypharmacy when possible, and help patients effectively manage or prevent these adverse effects.
Depression.1 Some studies found >45% patients with depression had EDS.3,13,15 Besides an association between depression and EDS,13,16 Chellappa and Araújo13 also found a significant association between EDS and suicidal ideation. The causes of EDS in patients with depression may be varied, ranging from restless legs syndrome, residual depressive symptoms,15 to OSA. Depression is often comorbid with OSA,6 with up to 20% of patients with depression suffering from OSA,10 creating higher risk for EDS. Depressive disorders are routinely assessed during an evaluation of OSA at sleep centers, but OSA often is not screened in psychiatric practice.10
There is a strong need for regular screening for OSA in patients with depression, particularly because most studies show a link between the 2 conditions.10 Both depression and OSA have some common risk factors, such as obesity, hypertension, and metabolic syndrome.10 Patients with these conditions are at greater risk for OSA, and therefore a psychiatrist should proactively screen and refer such patients for nocturnal polysomnography when they suspect OSA. Patients with OSA and depression often present to the psychiatrist with depressive symptoms that appear to be resistant to pharmacological treatment,10 therefore underscoring the importance of screening and ruling out OSA in patients with depression.
Circadian rhythm disorders, restless legs syndrome, alcohol and other substance use, and use of prescription sedative-hypnotics are more common in patients with depression; therefore, this population is at high risk for EDS.
Circadian rhythm disorders and insufficient sleep syndrome. Insufficient sleep syndrome1,2,8 frequently causes EDS and occurs more commonly in busy people who try to get by with less sleep.8 Over time, the effect of sleep loss is cumulative and can be accompanied by mood symptoms, such as irritability, fatigue, and problems with concentration.8 Shift workers1,8 commonly experience insufficient sleep as well as circadian rhythm disorders and EDS. Modafinil is FDA-approved for EDS in shift work sleep disorder.
Geriatric patients may experience advanced sleep phase syndrome involving early awakenings.8 Adolescents, on the other hand, often suffer from delayed sleep phase syndrome, which is a type of circadian rhythm disorder, related to increasing academic and social pressures, natural pubertal shift to later sleep onset, pervading technology use, and often nebulous bedtime routines. This can be a cause of sleep persisting into daytime.8 Taking a careful history and a sleep diary may be useful because this disorder might be confused for insomnia. Treatment involves gradual shifting of the time of sleep onset through bright light exposure and other modalities.8
Adolescents might not be forthcoming about the severity of their sleep problems; therefore, psychiatrists should screen proactively through clinical interviews of patients and parents and consider this possibility when encountering an adolescent with recent-onset attention or cognitive difficulties.
Treatment for circadian rhythm disorders usually includes planned or prescribed sleep scheduling, timed light exposure,8 and occasional use of melatonin or other sedative agents.17
Hypersomnia of central origin, which includes narcolepsy, idiopathic hypersomnia, and recurrent hypersomnia, can present with EDS.1,18,19 Narcolepsy is a rare, debilitating sleep disorder that manifests as EDS or sleep attacks, with or without cataplexy, and sleep paralysis.5,8,18,19 The Multiple Sleep Latency Test and polysomnography are used for diagnosis.1,5 Shortened REM latency is a classic finding often noted on polysomnography. Treatment involves pharmacologic and behavioral strategies and education.5,8 Modafinil is FDA-approved for EDS associated with narcolepsy. Stimulant medications have been used for narcolepsy in the past; further studies are needed to establish benefit–risk ratio of use in this population.18
Kleine-Levin syndrome is a form of recurrent hypersomnia, a less common sleep disorder, characterized by episodes of excessive sleepiness accompanied by hyperphagia and hypersexuality.5,18,19
Other medical conditions,1 such as the rare familial fatal insomnia, neurological conditions1 such as encephalitis,8 epilepsy,8 Alzheimer’s disease or other types of dementia,8 Parkinson’s disease,1 or multiple sclerosis,1,18 can cause excessive daytime fatigue by causing secondary insomnia or hypersomnia.
Treating the underlying disorder is an important first step in these cases. In addition, coordinating with neurologists or other specialists involved in caring for patients with these conditions is important. Regularly reviewing and simplifying the often complex medication regimen, when possible, can go a long way in mitigating EDS in this population.
Other disorders affecting sleep. Restless legs syndrome and periodic limb movement disorder are other causes of EDS.3 Treatment involves lifestyle modifications, iron supplementation in certain patients, and use of dopaminergic agents such as ropinirole, pramipexole, and other medications, depending on severity of the condition, comorbidities, and other factors.20
Alcohol or substance use. Substance use or withdrawal can be associated with sleep disorders, such as hypersomnia,19 insomnia,19 and related EDS.5 For example, alcohol use disorder affects REM sleep, and can cause EDS. Secondary central apnea can be the result of long-standing opioid use19 and can present like EDS.
Insomnia. Primary insomnia rarely causes EDS.5 Insomnia due to a medical or psychiatric condition may be an indirect cause of EDS by causing sleep deprivation.
Steps for timely and accurate diagnosis
Utilize the following steps for facilitating timely diagnosis and treatment of EDS:
Thorough history. Patients often describe “tiredness” instead of sleepiness.8 Therefore, the astute psychiatrist should explore further when patients are presenting with this concern, especially by asking more specific questions such as the tendency to doze off during daytime.8
Family members can be vital sources for obtaining a complete history,5 especially because patients might deny,8 minimize, or not be fully aware1 of the extent of their symptoms. Asking family members about patient’s snoring, irregular breathing, or gasping at night can be particularly valuable.5 Obtaining a family history of sleep disorders can be particularly important, especially in conditions such as OSA and narcolepsy.
Asking about any history of safety issues,8 including sleepiness during driving, cooking, or other activities, is also important.
Use of scales and other screening measures. Psychiatrists can use initial screening measures in the office setting. Epworth Sleepiness Scale15,21 is a validated,2 short, self-administered measure to assess the level of daytime sleepiness; however, it has some limitations such as not being able to measure changes in sleepiness from hour to hour or day to day. Because of its limitations, the Epworth Sleepiness Scale should not be used by itself as a diagnostic tool.3 It has been commonly used for detecting OSA2 and narcolepsy. The Stanford Sleepiness Scale is a self-rating scale that measures the subjective degree of sleepiness and alertness; it has limitations as well, such as having little correlation with chronic sleep loss.8 Other tools such as visual analogue scales also could be helpful.8 For more specialized testing, such as Multiple Sleep Latency Test or polysomnography, referral to a sleep specialist is ideal.8
Education. The assessment is an opportunity for the psychiatrist to educate patients about sleep hygiene, the importance of regular bedtimes, and getting adequate sleep to avoid accumulating a sleep deficit.
Urgent referral of at-risk populations. Prompt or urgent referral of at-risk populations, such as geriatric patients or those with a history of dozing off during driving, is invaluable in preventing morbidity and mortality from untreated sleep disorders.
Patients with severe daytime sleepiness should be advised to not drive or operate heavy machinery until this condition is adequately controlled.18
Bottom Line
Excessive daytime sleepiness (EDS) is “the inability to maintain wakefulness and alertness during the major waking periods of the day, with sleep occurring unintentionally or at inappropriate times, almost daily for at least 3 months,” according to the American Academy of Sleep Medicine.1 EDS is common, with a prevalence up to 25% to 30% in the general population.1-4 The prevalence rate varies in different studies, primarily because of inconsistent definitions of EDS, and therefore differences in diagnosis and assessment.1,2,4 In a study of 300 psychiatric outpatients, 34% had EDS.3 However, studies and evidence reviewing EDS in psychiatric patients are limited.
The causes of EDS are many and varied,1,8 including medical and psychiatric etiologies. A thorough history, screening at-risk patients, and timely sleep center referral are vital to detect and appropriately manage the cause of EDS.5
This article reviews the literature on EDS, with a focus on the risks of untreated EDS, common etiologies of the condition, as well as a brief description of screening and treatment strategies.
EDS vs fatigue
Many patients describe EDS as “fatigue”1; however, a patient’s report of fatigue could be mistaken for EDS.4 Although there is overlap, it is important for physicians to distinguish between these 2 entities for accurate identification and treatment.1,4
Risk of inadequate screening
A study of 117 patients with symptomatic coronary artery disease showed that EDS is associated with significantly greater incidence of cardiovascular adverse events at 16-month follow up.2 This study had limitations such as small sample size; therefore, more studies are needed. Because of these risks, timely and accurate diagnosis not only improves the patient’s quality of life and reduces polypharmacy but also can be life-saving.
Common causes of EDS in psychiatric patients
Because of the high prevalence and severity of impairments caused by EDS, it is essential for psychiatrists to be informed about causes of EDS and thoroughly assess for the potential underlying etiology before concluding that the sleep problem is a manifestation of the psychiatric disorder and prescribing psychotropic medication for it.
Some common causes of EDS in psychiatric patients include:
Sleep-disordered breathing.8 Obstructive sleep apnea (OSA) is often underdiagnosed,6,7 and considering how common it is,6 psychiatrists likely will see many patients with OSA in their practice.5 OSA has a higher prevalence among patients with psychiatric disorders such as depression6,9 and schizophrenia. Additionally, there is evidence suggesting that patients with OSA are more likely to suffer from depression and EDS than healthy controls6,9,10; some of the proposed mechanisms are sleep fragmentation and hypoxemia.6,9-11 OSA is the most common form of sleep-disordered breathing and is a common cause of EDS.1,2,12 Also, undiagnosed and untreated OSA in patients with depression could cause refractoriness to pharmacological treatment of depression.6,9,10
When unrecognized and untreated, OSA can be life-threatening. Despite this, OSA is not regularly screened for in clinical psychiatric practice.6,10 Therefore, it is imperative that psychiatrists be well-acquainted with measures to identify at-risk patients and refer to a sleep specialist when appropriate.
OSA is accompanied by irritability, cognitive difficulties, and poor sleep, creating an overlap with symptoms of depressive disorders.6,10 Use of sedative hypnotic medications, such as benzodiazepines, which further reduces muscle tone in the airway and suppresses respiratory effort, can worsen OSA symptoms5,6,10 and pose cerebrovascular, cardiovascular, and potentially life-threatening risks, and therefore is not indicated in this population.9,13
Obesity is a risk factor for OSA.6 Patients with mood disorders or schizophrenia or other psychotic disorders are at higher risk of obesity because of psychotropic-induced weight gain, stress-induced mechanisms, and/or lower levels of self-care. When these patients have unrecognized or untreated OSA and are prescribed sedative medications at night or stimulant medications during the day, they could be at increased cardiac or respiratory risks without resolving their underlying condition. A diligent psychiatrist can dramatically reduce the risks by referring a patient for nocturnal polysomnography,1 helping the patient implement lifestyle modifications (eg, exercise, weight loss, and healthy nutrition), prescribing judiciously, and monitoring closely for such risks. An accurate diagnosis of and treatment for OSA can improve sleep6 dramatically and help depressive symptoms through better sleep, more daytime energy and concentration, and adequate oxygenation of the brain while sleeping.
Psychiatrists can screen for OSA using the STOP-Bang (Snoring, Tired, Observed apnea, Pressure, Body mass index, Age, Neck circumference, Gender) Questionnaire, which is a quick, 8-item screening scale that helps to categorize OSA risk as mild, moderate, or severe.12 Hypertension, snoring, and/or gasping for breath (“observed apnea”)—a history which often is provided by spouses or significant others—daytime dozing and/or tiredness, having a large neck circumference or volume, body mass index, male sex, and age are items on the STOP-Bang Questionnaire and also are features that should raise high clinical suspicion of OSA.12 Referral for nocturnal polysomnography in at-risk patients should be the next step1,5 in any sleep-related breathing disorder.
Treatment for OSA involves continuous positive airway pressure (CPAP) therapy, which has been shown to relieve OSA and decrease related EDS.5,6 Other treatment modalities, such as oral appliances and surgery, may be used5 in some cases, but more studies are needed for conclusive results.
Several studies have shown improved depression, mood, and cognition after administering treatment such as CPAP6,9,14 in patients with OSA and depression. Considering the significant risks of cardiovascular,8 cerebrovascular,8 and overall morbidity and mortality associated with untreated OSA,12 it is important to routinely screen for sleep-disordered breathing in patients with depression9 or other psychiatric disorders and refer for specialized sleep evaluation and treatment, when indicated.
Medications. EDS can result from some prescription and over-the-counter medications.1,2,5,7 Sedating antidepressants, antihistamines, antipsychotics, anticonvulsants,1,8 and beta blockers2 could cause sedation, which can persist during daytime, although a few studies did not find an association between antipsychotic use and EDS.3 Benzodiazepines and other sedative-hypnotics,1,7 especially long-acting agents or higher dosages,5 can lead to EDS and decreased alertness. Non-psychotropics, such as opioid pain medications,1,7 antitussives, and skeletal muscle relaxants, also can contribute to or cause daytime sedation.7 When using these agents, psychiatrists should monitor and routinely assess patients while aiming for the lowest effective dosage when feasible.
This strategy creates a framework for psychiatrists to routinely educate patients about these commonly encountered side effects, reduce polypharmacy when possible, and help patients effectively manage or prevent these adverse effects.
Depression.1 Some studies found >45% patients with depression had EDS.3,13,15 Besides an association between depression and EDS,13,16 Chellappa and Araújo13 also found a significant association between EDS and suicidal ideation. The causes of EDS in patients with depression may be varied, ranging from restless legs syndrome, residual depressive symptoms,15 to OSA. Depression is often comorbid with OSA,6 with up to 20% of patients with depression suffering from OSA,10 creating higher risk for EDS. Depressive disorders are routinely assessed during an evaluation of OSA at sleep centers, but OSA often is not screened in psychiatric practice.10
There is a strong need for regular screening for OSA in patients with depression, particularly because most studies show a link between the 2 conditions.10 Both depression and OSA have some common risk factors, such as obesity, hypertension, and metabolic syndrome.10 Patients with these conditions are at greater risk for OSA, and therefore a psychiatrist should proactively screen and refer such patients for nocturnal polysomnography when they suspect OSA. Patients with OSA and depression often present to the psychiatrist with depressive symptoms that appear to be resistant to pharmacological treatment,10 therefore underscoring the importance of screening and ruling out OSA in patients with depression.
Circadian rhythm disorders, restless legs syndrome, alcohol and other substance use, and use of prescription sedative-hypnotics are more common in patients with depression; therefore, this population is at high risk for EDS.
Circadian rhythm disorders and insufficient sleep syndrome. Insufficient sleep syndrome1,2,8 frequently causes EDS and occurs more commonly in busy people who try to get by with less sleep.8 Over time, the effect of sleep loss is cumulative and can be accompanied by mood symptoms, such as irritability, fatigue, and problems with concentration.8 Shift workers1,8 commonly experience insufficient sleep as well as circadian rhythm disorders and EDS. Modafinil is FDA-approved for EDS in shift work sleep disorder.
Geriatric patients may experience advanced sleep phase syndrome involving early awakenings.8 Adolescents, on the other hand, often suffer from delayed sleep phase syndrome, which is a type of circadian rhythm disorder, related to increasing academic and social pressures, natural pubertal shift to later sleep onset, pervading technology use, and often nebulous bedtime routines. This can be a cause of sleep persisting into daytime.8 Taking a careful history and a sleep diary may be useful because this disorder might be confused for insomnia. Treatment involves gradual shifting of the time of sleep onset through bright light exposure and other modalities.8
Adolescents might not be forthcoming about the severity of their sleep problems; therefore, psychiatrists should screen proactively through clinical interviews of patients and parents and consider this possibility when encountering an adolescent with recent-onset attention or cognitive difficulties.
Treatment for circadian rhythm disorders usually includes planned or prescribed sleep scheduling, timed light exposure,8 and occasional use of melatonin or other sedative agents.17
Hypersomnia of central origin, which includes narcolepsy, idiopathic hypersomnia, and recurrent hypersomnia, can present with EDS.1,18,19 Narcolepsy is a rare, debilitating sleep disorder that manifests as EDS or sleep attacks, with or without cataplexy, and sleep paralysis.5,8,18,19 The Multiple Sleep Latency Test and polysomnography are used for diagnosis.1,5 Shortened REM latency is a classic finding often noted on polysomnography. Treatment involves pharmacologic and behavioral strategies and education.5,8 Modafinil is FDA-approved for EDS associated with narcolepsy. Stimulant medications have been used for narcolepsy in the past; further studies are needed to establish benefit–risk ratio of use in this population.18
Kleine-Levin syndrome is a form of recurrent hypersomnia, a less common sleep disorder, characterized by episodes of excessive sleepiness accompanied by hyperphagia and hypersexuality.5,18,19
Other medical conditions,1 such as the rare familial fatal insomnia, neurological conditions1 such as encephalitis,8 epilepsy,8 Alzheimer’s disease or other types of dementia,8 Parkinson’s disease,1 or multiple sclerosis,1,18 can cause excessive daytime fatigue by causing secondary insomnia or hypersomnia.
Treating the underlying disorder is an important first step in these cases. In addition, coordinating with neurologists or other specialists involved in caring for patients with these conditions is important. Regularly reviewing and simplifying the often complex medication regimen, when possible, can go a long way in mitigating EDS in this population.
Other disorders affecting sleep. Restless legs syndrome and periodic limb movement disorder are other causes of EDS.3 Treatment involves lifestyle modifications, iron supplementation in certain patients, and use of dopaminergic agents such as ropinirole, pramipexole, and other medications, depending on severity of the condition, comorbidities, and other factors.20
Alcohol or substance use. Substance use or withdrawal can be associated with sleep disorders, such as hypersomnia,19 insomnia,19 and related EDS.5 For example, alcohol use disorder affects REM sleep, and can cause EDS. Secondary central apnea can be the result of long-standing opioid use19 and can present like EDS.
Insomnia. Primary insomnia rarely causes EDS.5 Insomnia due to a medical or psychiatric condition may be an indirect cause of EDS by causing sleep deprivation.
Steps for timely and accurate diagnosis
Utilize the following steps for facilitating timely diagnosis and treatment of EDS:
Thorough history. Patients often describe “tiredness” instead of sleepiness.8 Therefore, the astute psychiatrist should explore further when patients are presenting with this concern, especially by asking more specific questions such as the tendency to doze off during daytime.8
Family members can be vital sources for obtaining a complete history,5 especially because patients might deny,8 minimize, or not be fully aware1 of the extent of their symptoms. Asking family members about patient’s snoring, irregular breathing, or gasping at night can be particularly valuable.5 Obtaining a family history of sleep disorders can be particularly important, especially in conditions such as OSA and narcolepsy.
Asking about any history of safety issues,8 including sleepiness during driving, cooking, or other activities, is also important.
Use of scales and other screening measures. Psychiatrists can use initial screening measures in the office setting. Epworth Sleepiness Scale15,21 is a validated,2 short, self-administered measure to assess the level of daytime sleepiness; however, it has some limitations such as not being able to measure changes in sleepiness from hour to hour or day to day. Because of its limitations, the Epworth Sleepiness Scale should not be used by itself as a diagnostic tool.3 It has been commonly used for detecting OSA2 and narcolepsy. The Stanford Sleepiness Scale is a self-rating scale that measures the subjective degree of sleepiness and alertness; it has limitations as well, such as having little correlation with chronic sleep loss.8 Other tools such as visual analogue scales also could be helpful.8 For more specialized testing, such as Multiple Sleep Latency Test or polysomnography, referral to a sleep specialist is ideal.8
Education. The assessment is an opportunity for the psychiatrist to educate patients about sleep hygiene, the importance of regular bedtimes, and getting adequate sleep to avoid accumulating a sleep deficit.
Urgent referral of at-risk populations. Prompt or urgent referral of at-risk populations, such as geriatric patients or those with a history of dozing off during driving, is invaluable in preventing morbidity and mortality from untreated sleep disorders.
Patients with severe daytime sleepiness should be advised to not drive or operate heavy machinery until this condition is adequately controlled.18
Bottom Line
1. Chervin RD. Approach to the patient with excessive daytime sleepiness. http://www.uptodate.com/contents/approach-to-the-patient-with-excessive-daytime-sleepiness. Updated January 2016. Accessed June 5, 2017.
2. Lee CH, Ng WY, Hau W, et al. Excessive daytime sleepiness is associated with longer culprit lesion and adverse outcomes in patients with coronary artery disease. J Clin Sleep Med. 2013;9(12):1267-1272.
3. Hawley CJ, Gale TM, Sivakumaran T, et al. Excessive daytime sleepiness in psychiatric disorders: prevalence, correlates and clinical significance. Psychiatry Res. 2010;175(1-2):138-141.
4. Pigeon WR, Sateia MJ, Ferguson RJ. Distinguishing between excessive daytime sleepiness and fatigue: toward improved detection and treatment. J Psychosom Res. 2003;54(1):61-69.
5. Krahn LE. Excessive daytime sleepiness: diagnosing the causes. Current Psychiatry. 2002;1(1):49-57.
6. Ejaz SM, Khawaja IS, Bhatia S, et al. Obstructive sleep apnea and depression: a review. Innov Clin Neurosci. 2011;8(8):17-25.
7. Pagel JF. Excessive daytime sleepiness. Am Fam Physician. 2009;79(5):391-396.
8. Guilleminault C, Brooks SN. Excessive daytime sleepiness: a challenge for the practising neurologist. Brain. 2001;124(pt 8):1482-1491.
9. Cheng P, Casement M, Chen CF, et al. Sleep disordered breathing in major depressive disorder. J Sleep Res. 2013;22(4):459-462.
10. Schröder CM, O’Hara R. Depression and obstructive sleep apnea (OSA). Ann Gen Psychiatry. 2005;4:13.
11. Bardwell WA, Berry CC, Ancoli-Israel S, et al. Psychological correlates of sleep apnea. J Psychosom Res. 1999;47(6):583-596.
12. Chung F, Abdullah HR, Liao P. STOP-Bang Questionnaire: a practical approach to screen for obstructive sleep apnea. Chest. 2016;149(3):631-638.
13. Chellappa SL, Araújo JF. Excessive daytime sleepiness in patients with depressive disorder. Rev Bras Psiquiatr. 2006;28(2):126-129.
14. Habukawa M, Uchimura N, Kakuma T, et al. Effect of CPAP treatment on residual depressive symptoms in patients with major depression and coexisting sleep apnea: contribution of daytime sleepiness to residual depressive symptoms. Sleep Med. 2010;11(6):552-557.
15. Lundt L. Use of the Epworth Sleepiness Scale to evaluate the symptom of excessive sleepiness in major depressive disorder. Gen Hosp Psychiatry. 2005;27(2):146-148.
16. Hawley CJ. Excessive daytime sleepiness in psychiatry: a relevant focus for clinical attention and treatment? Int J Psychiatry Clin Pract. 2006;10(2):117-123.
17. Dodson ER, Zee PC. Therapeutics for circadian rhythm sleep disorders. Sleep Med Clin. 2010;5(4):701-715.
18. Morgenthaler TI, Kapur VK, Brown TM, et al; Standards of Practice Committee of the American Academy of Sleep Medicine. Practice parameters for the treatment of narcolepsy and other hypersomnias of central origin. Sleep. 2007;30(12):1705-1711.
19. Thorpy MJ. Classification of sleep disorders. Neurotherapeutics. 2012;9(4):687-701.
20. National Institute of Neurological Disorders and Stroke. Restless legs syndrome information page. https://www.ninds.nih.gov/Disorders/All-Disorders/Restless-Legs-Syndrome-Information-Page. Accessed June 2, 2017.
21. Johns MW. Reliability and factor analysis of the Epworth Sleepiness Scale. Sleep. 1992;15(4):376-381.
1. Chervin RD. Approach to the patient with excessive daytime sleepiness. http://www.uptodate.com/contents/approach-to-the-patient-with-excessive-daytime-sleepiness. Updated January 2016. Accessed June 5, 2017.
2. Lee CH, Ng WY, Hau W, et al. Excessive daytime sleepiness is associated with longer culprit lesion and adverse outcomes in patients with coronary artery disease. J Clin Sleep Med. 2013;9(12):1267-1272.
3. Hawley CJ, Gale TM, Sivakumaran T, et al. Excessive daytime sleepiness in psychiatric disorders: prevalence, correlates and clinical significance. Psychiatry Res. 2010;175(1-2):138-141.
4. Pigeon WR, Sateia MJ, Ferguson RJ. Distinguishing between excessive daytime sleepiness and fatigue: toward improved detection and treatment. J Psychosom Res. 2003;54(1):61-69.
5. Krahn LE. Excessive daytime sleepiness: diagnosing the causes. Current Psychiatry. 2002;1(1):49-57.
6. Ejaz SM, Khawaja IS, Bhatia S, et al. Obstructive sleep apnea and depression: a review. Innov Clin Neurosci. 2011;8(8):17-25.
7. Pagel JF. Excessive daytime sleepiness. Am Fam Physician. 2009;79(5):391-396.
8. Guilleminault C, Brooks SN. Excessive daytime sleepiness: a challenge for the practising neurologist. Brain. 2001;124(pt 8):1482-1491.
9. Cheng P, Casement M, Chen CF, et al. Sleep disordered breathing in major depressive disorder. J Sleep Res. 2013;22(4):459-462.
10. Schröder CM, O’Hara R. Depression and obstructive sleep apnea (OSA). Ann Gen Psychiatry. 2005;4:13.
11. Bardwell WA, Berry CC, Ancoli-Israel S, et al. Psychological correlates of sleep apnea. J Psychosom Res. 1999;47(6):583-596.
12. Chung F, Abdullah HR, Liao P. STOP-Bang Questionnaire: a practical approach to screen for obstructive sleep apnea. Chest. 2016;149(3):631-638.
13. Chellappa SL, Araújo JF. Excessive daytime sleepiness in patients with depressive disorder. Rev Bras Psiquiatr. 2006;28(2):126-129.
14. Habukawa M, Uchimura N, Kakuma T, et al. Effect of CPAP treatment on residual depressive symptoms in patients with major depression and coexisting sleep apnea: contribution of daytime sleepiness to residual depressive symptoms. Sleep Med. 2010;11(6):552-557.
15. Lundt L. Use of the Epworth Sleepiness Scale to evaluate the symptom of excessive sleepiness in major depressive disorder. Gen Hosp Psychiatry. 2005;27(2):146-148.
16. Hawley CJ. Excessive daytime sleepiness in psychiatry: a relevant focus for clinical attention and treatment? Int J Psychiatry Clin Pract. 2006;10(2):117-123.
17. Dodson ER, Zee PC. Therapeutics for circadian rhythm sleep disorders. Sleep Med Clin. 2010;5(4):701-715.
18. Morgenthaler TI, Kapur VK, Brown TM, et al; Standards of Practice Committee of the American Academy of Sleep Medicine. Practice parameters for the treatment of narcolepsy and other hypersomnias of central origin. Sleep. 2007;30(12):1705-1711.
19. Thorpy MJ. Classification of sleep disorders. Neurotherapeutics. 2012;9(4):687-701.
20. National Institute of Neurological Disorders and Stroke. Restless legs syndrome information page. https://www.ninds.nih.gov/Disorders/All-Disorders/Restless-Legs-Syndrome-Information-Page. Accessed June 2, 2017.
21. Johns MW. Reliability and factor analysis of the Epworth Sleepiness Scale. Sleep. 1992;15(4):376-381.
Excessive daytime sleepiness
Rule out these causes of inattention before diagnosing ADHD
Inattention and distractibility are highly prevalent, and can exist secondary to a number of underlying causes. When a patient (or the patient’s family) asks whether he (she) might have attention-deficit/hyperactivity disorder (ADHD), you must perform a comprehensive assessment to rule out other medical and psychiatric disorders that might be manifesting as inattention. It is important not to miss a diagnosis of ADHD, and it is vital not to mistake another medical or psychiatric condition as ADHD.
Pay attention to components of the differential diagnosis while you are evaluating a patient with possible ADHD.
Medical conditions. Several disorders can present with cognitive, attentional, and executive functioning deficits that resemble the presentation of ADHD. These include absence seizures and other types of seizures, Lyme disease, HIV infection, and encephalopathy.1
People who have completed chemotherapy (particularly children) often exhibit attentional and executive functioning deficits similar to those found in ADHD.1
Anxiety disorders, the most prevalent of psychiatric disorders, correlate highly with difficulty concentrating. Chronic stress can have negative effects on hippocampus- and prefrontal cortical-based memory and cognitive functions.2 Be cautious, therefore, when diagnosing ADHD in a patient who suffers from significant, acute, or inadequately controlled anxiety—especially one who does not have a history of a childhood onset of attentional difficulties.
On the other hand, untreated ADHD can lead to anxiety symptoms.
Drugs. A number of substances of abuse—marijuana, cocaine, ecstasy, and caffeine—can produce symptoms of poor attention or impulsivity, similar to what is seen in ADHD, through their effects on the hippocampus and prefrontal cortex.3,4 MRI studies of the brains of 8-year-olds prenatally exposed to cocaine have found changes in frontal lobes suggesting potential long-term effects on attention and impulse control in these children.5,6
Use of certain medications, such as anticholinergics, also can contribute to attentional difficulties in some patient populations.
Abuse or trauma. Difficulty concentrating is one of the core symptoms of posttraumatic stress disorder (PTSD). Rule out PTSD and recent abuse or trauma when assessing for ADHD. Children with recent trauma often present with agitation, restlessness, and behavioral disturbance—symptoms that mimic ADHD.
Mood and adjustment disorders. Difficulty concentrating also is a criterion for major depressive disorder. On the other hand, untreated ADHD also can lead to, or contribute to, development of a depressive disorder. If a patient is experiencing a major depressive episode, obtain a thorough collateral history delineating a timeline of attention difficulties, which should allow for an accurate diagnosis.
In children, ADHD and bipolar disorder can have symptom overlap; both can present with distractibility, increased energy, and mood lability—therefore making a careful history a diagnostic necessity. Furthermore, ADHD and bipolar disorder can coexist in a small percentage of ADHD patients.
Hypothyroidism. Studies show a decrease in memory, attention, and concentration in patients with overt hypothyroidism, and at least a small decrease in these domains in patients with subclinical hypothyroidism.7 Decreased cerebral blood flow in brain regions that mediate attention and executive functioning, and decreased hippocampal volume, have been observed in patients with hypothyroidism.7 Therefore, the cognitive profile in these patients can look similar to, and can be confused with, ADHD, inattentive type.
Insomnia. Sleep plays a key role in memory consolidation and maintaining attention. Sleep disorders (eg, sleep apnea, restless legs syndrome, delayed sleep phase-onset disorder) can produce chronic tiredness and significantly affect attention, concentration, and cognitive functioning in children, adolescents, and adults.8
Studies in adults have shown that sleep deprivation is linked to attentional difficulty secondary to changes in prefrontal cortex activity.9 Other studies suggest that short sleep duration in healthy children is associated with inattention and poorer academic functioning, and also was found linked to teacher reports of inattention and a cognitive profile similar to what is seen in ADHD.8
Learning disorders and developmental disabilities. Children with an undiagnosed learning disorder often present with symptoms akin to those of ADHD.1 An undiagnosed reading or mathematics disorder, for example, can have a significant impact on academic functioning, in which the child might not be paying attention because of his (her) restricted ability to grasp the subject matter.
On the other hand, keep in mind that ADHD is highly comorbid with learning disorders.10
Last, children and adults with a developmental disability can present with signs and symptoms similar to those of ADHD.1
Summing up
Comprehensive assessment and management of any underlying condition is important to address the attention deficits you observe in a patient. A collateral history from parents and significant others, school reports, relevant laboratory tests, and a full physical examination are important tools for making an accurate diagnosis.
1. Robb AS. Differential diagnosis of ADHD in school-age children. Medscape Psychiatry. http://www.medscape.com/viewarticle/544948. Published September 26, 2006. Accessed September 6, 2016.
2. Sandi C. Memory impairments associated with stress and aging. In: Bermúdez-Rattoni F, ed. Neural plasticity and memory: from genes to brain imaging. Boca Raton, FL: Taylor & Francis Group, LLC; 2007:54-55,58-59.
3. Gouzoulis-Mayfrank E, Daumann J, Tuchtenhagen F, et al. Impaired cognitive performance in drug free users of recreational ecstasy (MDMA). J Neurol Neurosurg Psychiatry. 2000;68(6):719-725.
4. Hanson KL, Winward JL, Schweinsburg AD, et al. Longitudinal study of cognition among adolescent marijuana users over three weeks of abstinence. Addict Behav. 2010;35(11):970-976.
5. Morrow CE, Culbertson JL, Accornero VH, et al. Learning disabilities and intellectual functioning in school-aged children with prenatal cocaine exposure. Dev Neuropsychol. 2006;30(3):905-931.
6. Smith LM, Chang L, Yonekura ML, et al. Brain proton magnetic resonance spectroscopy and imaging in children exposed to cocaine in utero. Pediatrics. 2001;107(2):227-231.
7. Samuels MH. Psychiatric and cognitive manifestations of hypothyroidism. Curr Opin Endocrinol Diabetes Obes. 2014;21(5):377-383.
8. Gruber R, Michaelsen S, Bergmame L, et al. Short sleep duration is associated with teacher-reported inattention and cognitive problems in healthy school-aged children. Nat Sci Sleep. 2012;4:33-40.
9. Killgore WDS. Effects of sleep deprivation on cognition. Prog Brain Res. 2010;185:105-129.
10. Czamara D, Tiesler CM, Kohlböck G, et al. Children with ADHD symptoms have a higher risk for reading, spelling and math difficulties in the GINIplus and LISAplus cohort studies. PLoS One. 2013;8(5):e63859. doi: 10.1371/journal.pone.0063859.
Inattention and distractibility are highly prevalent, and can exist secondary to a number of underlying causes. When a patient (or the patient’s family) asks whether he (she) might have attention-deficit/hyperactivity disorder (ADHD), you must perform a comprehensive assessment to rule out other medical and psychiatric disorders that might be manifesting as inattention. It is important not to miss a diagnosis of ADHD, and it is vital not to mistake another medical or psychiatric condition as ADHD.
Pay attention to components of the differential diagnosis while you are evaluating a patient with possible ADHD.
Medical conditions. Several disorders can present with cognitive, attentional, and executive functioning deficits that resemble the presentation of ADHD. These include absence seizures and other types of seizures, Lyme disease, HIV infection, and encephalopathy.1
People who have completed chemotherapy (particularly children) often exhibit attentional and executive functioning deficits similar to those found in ADHD.1
Anxiety disorders, the most prevalent of psychiatric disorders, correlate highly with difficulty concentrating. Chronic stress can have negative effects on hippocampus- and prefrontal cortical-based memory and cognitive functions.2 Be cautious, therefore, when diagnosing ADHD in a patient who suffers from significant, acute, or inadequately controlled anxiety—especially one who does not have a history of a childhood onset of attentional difficulties.
On the other hand, untreated ADHD can lead to anxiety symptoms.
Drugs. A number of substances of abuse—marijuana, cocaine, ecstasy, and caffeine—can produce symptoms of poor attention or impulsivity, similar to what is seen in ADHD, through their effects on the hippocampus and prefrontal cortex.3,4 MRI studies of the brains of 8-year-olds prenatally exposed to cocaine have found changes in frontal lobes suggesting potential long-term effects on attention and impulse control in these children.5,6
Use of certain medications, such as anticholinergics, also can contribute to attentional difficulties in some patient populations.
Abuse or trauma. Difficulty concentrating is one of the core symptoms of posttraumatic stress disorder (PTSD). Rule out PTSD and recent abuse or trauma when assessing for ADHD. Children with recent trauma often present with agitation, restlessness, and behavioral disturbance—symptoms that mimic ADHD.
Mood and adjustment disorders. Difficulty concentrating also is a criterion for major depressive disorder. On the other hand, untreated ADHD also can lead to, or contribute to, development of a depressive disorder. If a patient is experiencing a major depressive episode, obtain a thorough collateral history delineating a timeline of attention difficulties, which should allow for an accurate diagnosis.
In children, ADHD and bipolar disorder can have symptom overlap; both can present with distractibility, increased energy, and mood lability—therefore making a careful history a diagnostic necessity. Furthermore, ADHD and bipolar disorder can coexist in a small percentage of ADHD patients.
Hypothyroidism. Studies show a decrease in memory, attention, and concentration in patients with overt hypothyroidism, and at least a small decrease in these domains in patients with subclinical hypothyroidism.7 Decreased cerebral blood flow in brain regions that mediate attention and executive functioning, and decreased hippocampal volume, have been observed in patients with hypothyroidism.7 Therefore, the cognitive profile in these patients can look similar to, and can be confused with, ADHD, inattentive type.
Insomnia. Sleep plays a key role in memory consolidation and maintaining attention. Sleep disorders (eg, sleep apnea, restless legs syndrome, delayed sleep phase-onset disorder) can produce chronic tiredness and significantly affect attention, concentration, and cognitive functioning in children, adolescents, and adults.8
Studies in adults have shown that sleep deprivation is linked to attentional difficulty secondary to changes in prefrontal cortex activity.9 Other studies suggest that short sleep duration in healthy children is associated with inattention and poorer academic functioning, and also was found linked to teacher reports of inattention and a cognitive profile similar to what is seen in ADHD.8
Learning disorders and developmental disabilities. Children with an undiagnosed learning disorder often present with symptoms akin to those of ADHD.1 An undiagnosed reading or mathematics disorder, for example, can have a significant impact on academic functioning, in which the child might not be paying attention because of his (her) restricted ability to grasp the subject matter.
On the other hand, keep in mind that ADHD is highly comorbid with learning disorders.10
Last, children and adults with a developmental disability can present with signs and symptoms similar to those of ADHD.1
Summing up
Comprehensive assessment and management of any underlying condition is important to address the attention deficits you observe in a patient. A collateral history from parents and significant others, school reports, relevant laboratory tests, and a full physical examination are important tools for making an accurate diagnosis.
Inattention and distractibility are highly prevalent, and can exist secondary to a number of underlying causes. When a patient (or the patient’s family) asks whether he (she) might have attention-deficit/hyperactivity disorder (ADHD), you must perform a comprehensive assessment to rule out other medical and psychiatric disorders that might be manifesting as inattention. It is important not to miss a diagnosis of ADHD, and it is vital not to mistake another medical or psychiatric condition as ADHD.
Pay attention to components of the differential diagnosis while you are evaluating a patient with possible ADHD.
Medical conditions. Several disorders can present with cognitive, attentional, and executive functioning deficits that resemble the presentation of ADHD. These include absence seizures and other types of seizures, Lyme disease, HIV infection, and encephalopathy.1
People who have completed chemotherapy (particularly children) often exhibit attentional and executive functioning deficits similar to those found in ADHD.1
Anxiety disorders, the most prevalent of psychiatric disorders, correlate highly with difficulty concentrating. Chronic stress can have negative effects on hippocampus- and prefrontal cortical-based memory and cognitive functions.2 Be cautious, therefore, when diagnosing ADHD in a patient who suffers from significant, acute, or inadequately controlled anxiety—especially one who does not have a history of a childhood onset of attentional difficulties.
On the other hand, untreated ADHD can lead to anxiety symptoms.
Drugs. A number of substances of abuse—marijuana, cocaine, ecstasy, and caffeine—can produce symptoms of poor attention or impulsivity, similar to what is seen in ADHD, through their effects on the hippocampus and prefrontal cortex.3,4 MRI studies of the brains of 8-year-olds prenatally exposed to cocaine have found changes in frontal lobes suggesting potential long-term effects on attention and impulse control in these children.5,6
Use of certain medications, such as anticholinergics, also can contribute to attentional difficulties in some patient populations.
Abuse or trauma. Difficulty concentrating is one of the core symptoms of posttraumatic stress disorder (PTSD). Rule out PTSD and recent abuse or trauma when assessing for ADHD. Children with recent trauma often present with agitation, restlessness, and behavioral disturbance—symptoms that mimic ADHD.
Mood and adjustment disorders. Difficulty concentrating also is a criterion for major depressive disorder. On the other hand, untreated ADHD also can lead to, or contribute to, development of a depressive disorder. If a patient is experiencing a major depressive episode, obtain a thorough collateral history delineating a timeline of attention difficulties, which should allow for an accurate diagnosis.
In children, ADHD and bipolar disorder can have symptom overlap; both can present with distractibility, increased energy, and mood lability—therefore making a careful history a diagnostic necessity. Furthermore, ADHD and bipolar disorder can coexist in a small percentage of ADHD patients.
Hypothyroidism. Studies show a decrease in memory, attention, and concentration in patients with overt hypothyroidism, and at least a small decrease in these domains in patients with subclinical hypothyroidism.7 Decreased cerebral blood flow in brain regions that mediate attention and executive functioning, and decreased hippocampal volume, have been observed in patients with hypothyroidism.7 Therefore, the cognitive profile in these patients can look similar to, and can be confused with, ADHD, inattentive type.
Insomnia. Sleep plays a key role in memory consolidation and maintaining attention. Sleep disorders (eg, sleep apnea, restless legs syndrome, delayed sleep phase-onset disorder) can produce chronic tiredness and significantly affect attention, concentration, and cognitive functioning in children, adolescents, and adults.8
Studies in adults have shown that sleep deprivation is linked to attentional difficulty secondary to changes in prefrontal cortex activity.9 Other studies suggest that short sleep duration in healthy children is associated with inattention and poorer academic functioning, and also was found linked to teacher reports of inattention and a cognitive profile similar to what is seen in ADHD.8
Learning disorders and developmental disabilities. Children with an undiagnosed learning disorder often present with symptoms akin to those of ADHD.1 An undiagnosed reading or mathematics disorder, for example, can have a significant impact on academic functioning, in which the child might not be paying attention because of his (her) restricted ability to grasp the subject matter.
On the other hand, keep in mind that ADHD is highly comorbid with learning disorders.10
Last, children and adults with a developmental disability can present with signs and symptoms similar to those of ADHD.1
Summing up
Comprehensive assessment and management of any underlying condition is important to address the attention deficits you observe in a patient. A collateral history from parents and significant others, school reports, relevant laboratory tests, and a full physical examination are important tools for making an accurate diagnosis.
1. Robb AS. Differential diagnosis of ADHD in school-age children. Medscape Psychiatry. http://www.medscape.com/viewarticle/544948. Published September 26, 2006. Accessed September 6, 2016.
2. Sandi C. Memory impairments associated with stress and aging. In: Bermúdez-Rattoni F, ed. Neural plasticity and memory: from genes to brain imaging. Boca Raton, FL: Taylor & Francis Group, LLC; 2007:54-55,58-59.
3. Gouzoulis-Mayfrank E, Daumann J, Tuchtenhagen F, et al. Impaired cognitive performance in drug free users of recreational ecstasy (MDMA). J Neurol Neurosurg Psychiatry. 2000;68(6):719-725.
4. Hanson KL, Winward JL, Schweinsburg AD, et al. Longitudinal study of cognition among adolescent marijuana users over three weeks of abstinence. Addict Behav. 2010;35(11):970-976.
5. Morrow CE, Culbertson JL, Accornero VH, et al. Learning disabilities and intellectual functioning in school-aged children with prenatal cocaine exposure. Dev Neuropsychol. 2006;30(3):905-931.
6. Smith LM, Chang L, Yonekura ML, et al. Brain proton magnetic resonance spectroscopy and imaging in children exposed to cocaine in utero. Pediatrics. 2001;107(2):227-231.
7. Samuels MH. Psychiatric and cognitive manifestations of hypothyroidism. Curr Opin Endocrinol Diabetes Obes. 2014;21(5):377-383.
8. Gruber R, Michaelsen S, Bergmame L, et al. Short sleep duration is associated with teacher-reported inattention and cognitive problems in healthy school-aged children. Nat Sci Sleep. 2012;4:33-40.
9. Killgore WDS. Effects of sleep deprivation on cognition. Prog Brain Res. 2010;185:105-129.
10. Czamara D, Tiesler CM, Kohlböck G, et al. Children with ADHD symptoms have a higher risk for reading, spelling and math difficulties in the GINIplus and LISAplus cohort studies. PLoS One. 2013;8(5):e63859. doi: 10.1371/journal.pone.0063859.
1. Robb AS. Differential diagnosis of ADHD in school-age children. Medscape Psychiatry. http://www.medscape.com/viewarticle/544948. Published September 26, 2006. Accessed September 6, 2016.
2. Sandi C. Memory impairments associated with stress and aging. In: Bermúdez-Rattoni F, ed. Neural plasticity and memory: from genes to brain imaging. Boca Raton, FL: Taylor & Francis Group, LLC; 2007:54-55,58-59.
3. Gouzoulis-Mayfrank E, Daumann J, Tuchtenhagen F, et al. Impaired cognitive performance in drug free users of recreational ecstasy (MDMA). J Neurol Neurosurg Psychiatry. 2000;68(6):719-725.
4. Hanson KL, Winward JL, Schweinsburg AD, et al. Longitudinal study of cognition among adolescent marijuana users over three weeks of abstinence. Addict Behav. 2010;35(11):970-976.
5. Morrow CE, Culbertson JL, Accornero VH, et al. Learning disabilities and intellectual functioning in school-aged children with prenatal cocaine exposure. Dev Neuropsychol. 2006;30(3):905-931.
6. Smith LM, Chang L, Yonekura ML, et al. Brain proton magnetic resonance spectroscopy and imaging in children exposed to cocaine in utero. Pediatrics. 2001;107(2):227-231.
7. Samuels MH. Psychiatric and cognitive manifestations of hypothyroidism. Curr Opin Endocrinol Diabetes Obes. 2014;21(5):377-383.
8. Gruber R, Michaelsen S, Bergmame L, et al. Short sleep duration is associated with teacher-reported inattention and cognitive problems in healthy school-aged children. Nat Sci Sleep. 2012;4:33-40.
9. Killgore WDS. Effects of sleep deprivation on cognition. Prog Brain Res. 2010;185:105-129.
10. Czamara D, Tiesler CM, Kohlböck G, et al. Children with ADHD symptoms have a higher risk for reading, spelling and math difficulties in the GINIplus and LISAplus cohort studies. PLoS One. 2013;8(5):e63859. doi: 10.1371/journal.pone.0063859.