Article
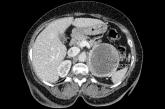
64-year-old woman • hot flashes, facial flushing, excessive sweating, and palpitations • daily headaches • history of hypertension • Dx?
- Author:
- Nicole O. Vietor, MD
- Andrew J. Spiro, MD
- Mohamed K. M. Shakir, MD
- Mitchell Harrison, MD
- Robert C. Vietor, MD
- Thanh D. Hoang, DO
► Hot flashes, facial flushing, excessive sweating, and palpitations
► Daily headaches
► History of hypertension
