Article
Hospital Dermatology: Review of Research in 2023-2024
- Author:
- Jenny Wei, MD
- Robert G. Micheletti, MD
Inpatient consultative dermatologists play a critical role in the care of hospitalized patients with skin disease. Our review of the 2023-2024...
Article
Inpatient Management of Hidradenitis Suppurativa: A Delphi Consensus Study
- Author:
- McKenzie Needham, BS
- Rita Pichardo, MD
- Afsaneh Alavi, MD
- Aileen Y. Chang, MD
- Steven Daveluy, MD
- Katherine L. DeNiro, MD
- Anna Dewan, MD
- Milad Eshaq, MD
- Lindy Fox, MD
- Jennifer Lin Hsiao, MD
- Benjamin Harris Kaffenberger, MD
- Joslyn S. Kirby, MD
- Daniela Kroshinsky, MD
- Alex G. Ortega-Loayza, MD
- Jennifer Brescoll Manusco, MD
- Robert G. Micheletti, MD
- Arash Mostaghimi, MD
- Caroline A. Nelson, MD
- Helena B. Pasieka, MD
- Martina L. Porter, MD
- Barry I. Resnik, MD
- Christopher J. Sayed, MD
- Vivian Y. Shi, MD
- Bridget E. Shields, MD
- Lindsay C. Strowd, MD
These recommendations serve as an important resource for providers caring for inpatients with hidradenitis suppurativa (HS) and represent a...
Article
Hospital Dermatology: Review of Research in 2022-2023
- Author:
- Juliana Berk-Krauss, MD
- Robert G. Micheletti, MD
In this review, we highlight 4 areas of focus from the published literature in 2022-2023—severe cutaneous adverse reactions, supportive...
Article
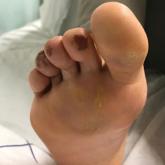
Cutaneous Manifestations of COVID-19: Characteristics, Pathogenesis, and the Role of Dermatology in the Pandemic
- Author:
- Mariam Alam, MD
- William Lewis, MD
- Joanna Harp, MD
- Katherine Steele, MD
- Misha Rosenbach, MD
- Robert G. Micheletti, MD
Cutaneous manifestations of COVID-19 may reflect the range of host immunologic responses to SARS-CoV-2.
Article
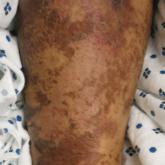
Nutritional Dermatoses in the Hospitalized Patient
- Author:
- Melissa Hoffman, MS
- Robert G. Micheletti, MD
- Bridget E. Shields, MD
This review details the risk factors for nutritional deficiency, illustrates the presentations of cutaneous disease, reviews diagnostic workups,...
Article
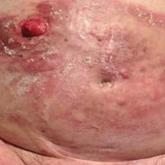
Erythematous Plaques and Nodules on the Abdomen and Groin
- Author:
- Christina C. Patrone, MD
- Caroline Nelson, MD
- Kevin J. Gaddis, MD
- Daniel Lubin, MD
- Xiaowei Xu, MD, PhD
- Ellen J. Kim, MD
- Robert G. Micheletti, MD
An 82-year-old man presented with acute abdominal pain and distension as well as an abdominal rash of 4 months' duration that was expanding...
Audio

Calciphylaxis: Diagnostic and Treatment Pearls
- Author:
- Robert G. Micheletti, MD
Article
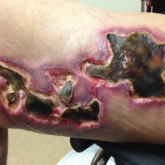
Update on Calciphylaxis Etiopathogenesis, Diagnosis, and Management
- Author:
- Urmi Khanna, MD
- Arturo Dominguez, MD
- Jesse Keller, MD
- Daniela Kroshinsky, MD, MPH
- Alex G. Ortega-Loayza, MD
- Lindsay Strowd, MD
- Robert G. Micheletti, MD
Calciphylaxis is associated with potentially severe complications and high mortality rates. This article highlights the challenges faced in...
Article
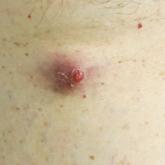
Enlarging Red Papulonodule on the Chest
- Author:
- Sheila Shaigany, MD
- Cory L. Simpson, MD, PhD
- Robert G. Micheletti, MD
A man in his 60s presented with a subcutaneous nodule on the right side of the chest. Due to impaired mental status, he was unable to describe the...
Article
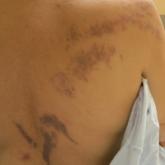
Purple Curvilinear Papules on the Back
- Author:
- Sara A. Berg, MD
- Adam I. Rubin, MD
- Robert G. Micheletti, MD
Article

An Update on the Diagnosis and Treatment of Hidradenitis Suppurativa
- Author:
- Robert G. Micheletti, MD
