User login
Chronic constipation: Update on management
Chronic constipation has a variety of possible causes and mechanisms. Although traditional conservative treatments are still valid and first-line, if these fail, clinicians can choose from a growing list of new treatments, tailored to the cause in the individual patient.
This article discusses how defecation works (or doesn’t), the types of chronic constipation, the available diagnostic tools, and traditional and newer treatments, including some still in development.
THE EPIDEMIOLOGY OF CONSTIPATION
Chronic constipation is one of the most common gastrointestinal disorders, affecting about 15% of all adults and 30% of those over the age of 60.1 It can be a primary disorder or secondary to other factors.
Constipation is more prevalent in women and in institutionalized elderly people.2 It is associated with lower socioeconomic status, depression, less self-reported physical activity, certain medications, and stressful life events.3 Given its high prevalence and its impact on quality of life, it is also associated with significant utilization of healthcare resources.4
Constipation defined by Rome IV criteria
Physicians and patients may disagree about what constitutes constipation. Physicians primarily regard it as infrequent bowel movements, while patients tend to have a broader definition. According to the Rome IV criteria,5 chronic constipation is defined by the presence of the following for at least 3 months (with symptom onset at least 6 months prior to diagnosis):
(1) Two or more of the following for more than 25% of defecations:
- Straining
- Lumpy or hard stools
- Sensation of incomplete evacuation
- Sensation of anorectal obstruction or blockage
- Manual maneuvers to facilitate evacuation
- Fewer than 3 spontaneous bowel movements per week.
(2) Loose stools are rarely present without the use of laxatives.
(3) The patient does not meet the criteria for diagnosis of irritable bowel syndrome.
DEFECATION IS COMPLEX
Defecation begins when the rectum fills with stool, causing relaxation of the internal anal sphincter and the urge to defecate. The external anal sphincter, which is under voluntary control, can then either contract to delay defecation or relax to allow the stool to be expelled.6
Colonic muscles propel stool toward the rectum in repetitive localized contractions that help mix and promote absorption of the content, and larger coordinated (high-amplitude propagating) contractions that, in healthy individuals, move the stool forward from the proximal to the distal colon multiple times daily. These contractions usually occur in the morning and are accentuated by gastric distention from food and the resulting gastrocolic reflex.
Serotonin (5-HT) is released by enterochromaffin cells in response to distention of the gut wall. It mediates peristaltic movements of the gastrointestinal tract by binding to receptors (especially 5-HT4), stimulating release of neurotransmitters such as acetylcholine, causing smooth-muscle contraction behind the luminal contents and propelling them forward.
PRIMARY CONSTIPATION DISORDERS
The American Gastroenterological Association7 classifies constipation into 3 groups on the basis of colonic transit time and anorectal function:
Normal-transit constipation
Stool normally takes 20 to 72 hours to pass through the colon, with transit time affected by diet, drugs, level of physical activity, and emotional status.8
Normal-transit constipation is the most common type of constipation. The term is sometimes used interchangeably with constipation-predominant irritable bowel syndrome, but the latter is a distinct entity characterized by abdominal pain relieved by defecation as the primary symptom, as well as having occasional loose stools. These 2 conditions can be hard to tell apart, especially if the patient cannot describe the symptoms precisely.
Slow-transit constipation
Slow-transit constipation—also called delayed-transit constipation, colonoparesis, colonic inertia, and pseudo-obstruction—is defined as prolonged stool transit in the colon, ie, for more than 5 days.9 It can be the result of colonic smooth muscle dysfunction, compromised colonic neural pathways, or both, leading to slow colon peristalsis.
Factors that can affect colonic motility such as opioid use and hypothyroidism should be carefully considered in these patients. Opioids are notorious for causing constipation by decreasing bowel tone and contractility and thereby increasing colonic transit time. They also tighten up the anal sphincters, resulting in decreased rectal evacuation.10
Outlet dysfunction
Outlet dysfunction, also called pelvic floor dysfunction or defecatory disorder, is associated with incomplete rectal evacuation. It can be a consequence of weak rectal expulsion forces (slow colonic transit, rectal hyposensitivity), functional resistance to rectal evacuation (high anal resting pressure, anismus, incomplete relaxation of the anal sphincter, dyssynergic defecation), or structural outlet obstruction (excessive perineal descent, rectoceles, rectal intussusception). About 50% of patients with outlet dysfunction have concurrent slow-transit constipation.
Dyssynergic defecation is the most common outlet dysfunction disorder, accounting for about half of the cases referred to tertiary centers. It is defined as a paradoxical elevation in anal sphincter tone or less than 20% relaxation of the resting anal sphincter pressure with weak abdominal and pelvic propulsive forces.11 Anorectal biofeedback is a therapeutic option for dyssynergic defecation, as we discuss later in this article.
SECONDARY CONSTIPATION
Constipation can be secondary to several conditions and factors (Table 1), including:
- Neurologic disorders that affect gastrointestinal motility (eg, Hirschsprung disease, Parkinson disease, multiple sclerosis, spinal cord injury, stroke, spinal or ganglionic tumor, hypothyroidism, amyloidosis, diabetes mellitus, hypercalcemia)
- Drugs used to treat neurologic disorders
- Mechanical obstruction
- Diet (eg, low fiber, decreased fluid intake).
EVALUATION OF CONSTIPATION
It is crucial for physicians to efficiently use the available diagnostic tools for constipation to tailor the treatment to the patient.
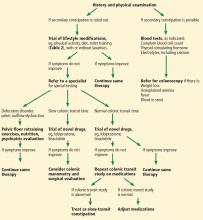
Evaluation of chronic constipation begins with a thorough history and physical examination to rule out secondary constipation (Figure 1). Red flags such as unintentional weight loss, blood in the stool, rectal pain, fever, and iron-deficiency anemia should prompt referral for colonoscopy to evaluate for malignancy, colitis, or other potential colonic abnormalities.12
A detailed perineal and rectal examination can help diagnose defecatory disorders and should include evaluation of the resting anal tone and the sphincter during simulated evacuation.
Laboratory tests of thyroid function, electrolytes, and a complete blood cell count should be ordered if clinically indicated.13
Further tests
Further diagnostic tests can be considered if symptoms persist despite conservative treatment or if a defecatory disorder is suspected. These include anorectal manometry, colonic transit studies, defecography, and colonic manometry.
Anorectal manometry and the rectal balloon expulsion test are usually done first because of their high sensitivity (88%) and specificity (89%) for defecatory disorders.14 These tests measure the function of the internal and external anal sphincters at rest and with straining and assess rectal sensitivity and compliance. Anorectal manometry is also used in biofeedback therapy in patients with dyssynergic defecation.15
Colonic transit time can be measured if anorectal manometry and the balloon expulsion test are normal. The study uses radiopaque markers, radioisotopes, or wireless motility capsules to confirm slow-transit constipation and to identify areas of delayed transit in the colon.16
Defecography is usually the next step in diagnosis if anorectal manometry and balloon expulsion tests are inconclusive or if an anatomic abnormality of the pelvic floor is suspected. It can be done with a variety of techniques. Barium defecography can identify anatomic defects, scintigraphy can quantify evacuation of artificial stools, and magnetic resonance defecography visualizes anatomic landmarks to assess pelvic floor motion without exposing the patient to radiation.17,18
Colonic manometry is most useful in patients with refractory slow-transit constipation and can identify patients with isolated colonic motor dysfunction with no pelvic floor dysfunction who may benefit from subtotal colectomy and end-ileostomy.7
TRADITIONAL TREATMENTS STILL THE MAINSTAY
Nonpharmacologic treatments are the first-line options for patients with normal-transit and slow-transit constipation and should precede diagnostic testing. Lifestyle modifications and dietary changes (Table 2) aim to augment the known factors that stimulate the gastrocolic reflex and increase intestinal motility by high-amplitude propagated contractions.
Increasing physical activity increases intestinal gas clearance, decreases bloating, and lessens constipation.19,20
Toilet training is an integral part of lifestyle modifications.21
Diet. Drinking hot caffeinated beverages, eating breakfast within an hour of waking up, and consuming fiber in the morning (25–30 g of fiber daily) have traditionally been recommended as the first-line measures for chronic constipation. Dehydrated patients with constipation also benefit from increasing their fluid intake.22
LAXATIVES
Fiber (bulk-forming laxatives) for normal-transit constipation
Fiber remains a key part of the initial management of chronic constipation, as it is cheap, available, and safe. Increasing fiber intake is effective for normal-transit constipation, but patients with slow-transit constipation or refractory outlet dysfunction are less likely to benefit.23 Other laxatives are incorporated into the regimen if first-line nonpharmacologic interventions fail (Table 3).
Bulk-forming laxatives include insoluble fiber (wheat bran) and soluble fiber (psyllium, methylcellulose, inulin, calcium polycarbophil). Insoluble fiber, though often used, has little impact on symptoms of chronic constipation after 1 month of use, and up to 60% of patients report adverse effects from it.24 On the other hand, clinical trials have shown that soluble fiber such as psyllium facilitates defecation and improves functional bowel symptoms in patients with normal-transit constipation.25
Patients should be instructed to increase their dietary fiber intake gradually to avoid adverse effects and should be told to expect significant symptomatic improvement only after a few weeks. They should also be informed that increasing dietary fiber intake can cause bloating but that the bloating is temporary. If it continues, a different fiber can be tried.
Osmotic laxatives
Osmotic laxatives are often employed as a first- line laxative treatment option for patients with constipation. They draw water into the lumen by osmosis, helping to soften stool and speed intestinal transit. They include macrogols (inert polymers of ethylene glycol), nonabsorbable carbohydrates (lactulose, sorbitol), magnesium products, and sodium phosphate products.
Polyethylene glycol, the most studied osmotic laxative, has been shown to maintain therapeutic efficacy for up to 2 years, though it is not generally used this long.26 A meta-analysis of 10 randomized clinical trials found it to be superior to lactulose in improving stool consistency and frequency, and rates of adverse effects were similar to those with placebo.27
Lactulose and sorbitol are semisynthetic disaccharides that are not absorbed from the gastrointestinal tract. Apart from the osmotic effect of the disaccharide, these sugars are metabolized by colonic bacteria to acetic acid and other short-chain fatty acids, resulting in acidification of the stool, which exerts an osmotic effect in the colonic lumen.
Lactulose and sorbitol were shown to have similar efficacy in increasing the frequency of bowel movements in a small study, though patients taking lactulose had a higher rate of nausea.28
The usual recommended dose is 15 to 30 mL once or twice daily.
Adverse effects include gas, bloating, and abdominal distention (due to fermentation by colonic bacteria) and can limit long-term use.
Magnesium citrate and magnesium hydroxide are strong osmotic laxatives, but so far no clinical trial has been done to assess their efficacy in constipation. Although the risk of hypermagnesemia is low with magnesium-based products, this group of laxatives is generally avoided in patients with renal or cardiac disease.29
Sodium phosphate enemas (Fleet enemas) are used for bowel cleansing before certain procedures but have only limited use in constipation because of potential adverse effects such as hyperphosphatemia, hypocalcemia, and the rarer but more serious complication of acute phosphate nephropathy.30
Stimulant laxatives for short-term use only
Stimulant laxatives include glycerin, bisacodyl, senna, and sodium picosulfate. Sodium piosulfate and bisacodyl have been validated for treatment of chronic constipation for up to 4 weeks.31–33
Stimulant laxative suppositories should be used 30 minutes after meals to augment the physiologic gastrocolic reflex.
As more evidence is available for osmotic laxatives such as polyethylene glycol, they tend to be preferred over stimulant agents, especially for long-term use. Clinicians have traditionally hesitated to prescribe stimulant laxatives for long-term use, as they were thought to damage the enteric nervous system.34 Although more recent studies have not shown this potential effect,35 more research is warranted on the use of stimulant laxatives for longer than 4 weeks.
STOOL SOFTENERS: LITTLE EVIDENCE
Stool softeners enhance the interaction of stool and water, leading to softer stool and easier evacuation. Docusate sodium and docusate calcium are thought to facilitate the mixing of aqueous and fatty substances, thereby softening the stool.
However, there is little evidence to support the use of docusate for constipation in hospitalized adults or in ambulatory care. A recent review reported that docusate was no better than placebo in diminishing symptoms of constipation.36
INTESTINAL SECRETAGOGUES
The secretagogues include lubiprostone, linaclotide, and plecanatide. These medications are preferred therapy for patients with normal- or slow-transit constipation once conservative therapies have failed. Even though there is no current consensus, lifestyle measures and conservative treatment options should be tried for about 8 weeks.
Lubiprostone and linaclotide are approved by the US Food and Drug Administration (FDA) for both constipation and constipation-predominant irritable bowel syndrome. They activate chloride channels on the apical surface of enterocytes, increasing intestinal secretion of chloride, which in turn increases luminal sodium efflux to maintain electroneutrality, leading to secretion of water into the intestinal lumen. This eventually facilitates intestinal transit and increases the passage of stool.
Lubiprostone
Lubiprostone, a prostaglandin E1 derivative, is approved for treating chronic constipation, constipation-predominant irritable bowel syndrome in women, and opioid-induced constipation in patients with chronic noncancer pain.
Adverse effects in clinical trials were nausea (up to 30%) and headache.37,38
Linaclotide
Linaclotide, a minimally absorbed 14-amino acid peptide, increases intestinal secretion of chloride and bicarbonate, increasing intestinal fluid and promoting intestinal transit.39 It also decreases the firing rate of the visceral afferent pain fibers and helps reduce visceral pain, especially in patients with constipation-predominant irritable bowel syndrome.40 It is approved for chronic constipation and constipation-predominant irritable bowel syndrome.41–43
Dosage starts at 145 μg/day for chronic constipation, and can be titrated up to 290 μg if there is no response or if a diagnosis of constipation-predominant irritable bowel syndrome is under consideration. Linaclotide should be taken 30 to 60 minutes before breakfast to reduce the likelihood of diarrhea.44
Adverse effects. Diarrhea led to treatment discontinuation in 4.5% of patients in one study.42
Plecanatide
Plecanatide is a guanylate cyclase-c agonist with a mode of action similar to that of linaclotide. It was recently approved by the FDA for chronic idiopathic constipation in adults. The recommended dose is 3 mg once daily.
Data from phase 2 trials in chronic constipation showed improvement in straining, abdominal discomfort, and stool frequency after 14 days of treatment.45
A phase 3 trial showed that plecanatide was more effective than placebo when used for 12 weeks in 951 patients with chronic constipation (P = .009).46 The most common adverse effect reported was diarrhea.
SEROTONIN RECEPTOR AGONISTS
Activation of serotonin 5-HT4 receptors in the gut leads to release of acetylcholine, which in turn induces mucosal secretion by activating submucosal neurons and increasing gut motility.47
Two 5-HT4 receptor agonists were withdrawn from the market (cisapride in 2000 and tegaserod in 2007) due to serious cardiovascular adverse events (fatal arrhythmias, heart attacks, and strokes) resulting from their affinity for hERG-K+ cardiac channels.
The newer agents prucalopride,48 velusetrag, and naronapride are highly selective 5-HT4 agonists with low affinity for hERG-K+ receptors and do not have proarrhythmic properties, based on extensive assessment in clinical trials.
Prucalopride
Prucalopride has been shown to accelerate gastrointestinal and colonic transit in patients with chronic constipation, with improvement in bowel movements, symptoms of chronic constipation, and quality of life.49–52
Adverse effects reported with its use have been headache, nausea, abdominal pain, and cramps.
Prucalopride is approved in Europe and Canada for chronic constipation in women but is not yet approved in the United States.
Dosage is 2 mg orally once daily. Caution is advised in elderly patients, in whom the preferred maximum dose is 1 mg daily, as there are only limited data available on the safety of this medication in the elderly.
Velusetrag
Velusetrag has been shown to increase colonic motility and improve symptoms of chronic constipation. In a phase 2 trial,53 the most effective dose was 15 mg once daily. Higher doses were associated with a higher incidence of adverse effects such as diarrhea, headache, nausea, and vomiting.
Naronapride
Naronapride (ATI-7505) is in phase 2 trials for chronic constipation. Reported adverse effects were headache, diarrhea, nausea, and vomiting.54
BILE SALT ABSORPTION INHIBITORS
Bile acids exert prosecretory and prokinetic effects by increasing colonic secretion of water and electrolytes through the activation of adenylate cyclase. This happens as a result of their deconjugation after passage into the colon.
Elobixibat is an ileal bile acid transporter inhibitor that prevents absorption of nonconjugated bile salts in the distal ileum. It has few side effects because its systemic absorption is minimal. Phase 3 trials are under way. Dosage is 5 to 20 mg daily. Adverse effects are few because systemic absorption is minimal, but include abdominal pain and diarrhea.55,56
MANAGING OPIOID-INDUCED CONSTIPATION
Opioids cause constipation by binding to mu receptors in the enteric nervous system. Activation of these receptors decreases bowel tone and contractility, which increases transit time. Stimulation of these receptors also increases anal sphincter tone, resulting in decreased rectal evacuation.57
Though underrecognized, opioid-induced constipation affects 40% of patients who take these drugs for nonmalignant pain and 90% of those taking them for cancer pain. Patients with this condition were found to take more time off work and feel more impaired in their domestic and work-related obligations than patients who did not develop constipation with use of opioids.58
Initial management of opioid-induced constipation includes increasing intake of fluids and dietary fiber (fiber alone can worsen abdominal pain in this condition by increasing stool bulk without a concomitant improvement in peristalsis) and increasing physical activity. It is common clinical practice to use a stool softener along with a stimulant laxative if lifestyle modifications are inadequate.59 If these measures are ineffective, osmotic agents can be added.
If these conventional measures fail, a peripherally acting mu-opioid receptor antagonist such as methylnaltrexone or naloxegol should be considered.
Methylnaltrexone
Methylnaltrexone60,61 is a peripherally acting mu receptor antagonist with a rapid onset of action. It does not cross the blood-brain barrier, as it contains a methyl group. It was approved by the FDA in 2008 to treat opioid-induced constipation in adults with advanced illnesses when other approaches are ineffective.
Adverse effects. Although the mu receptor antagonist alvimopan had been shown to be associated with cardiovascular events hypothesized to be a consequence of opioid withdrawal, methylnaltrexone has been deemed to have a safe cardiovascular profile without any potential effects on platelets, corrected QT interval, metabolism, heart rate, or blood pressure.61 Side effects include abdominal pain, nausea, diarrhea, hot flashes, tremor, and chills.
Contraindications. Methylnaltrexone is contraindicated in patients with structural diseases of the gastrointestinal tract, ie, peptic ulcer disease, inflammatory bowel disease, diverticulitis, stomach or intestinal cancer) since it can increase the risk of perforation.
Dosing is 1 dose subcutaneously every other day, as needed, and no more than 1 dose in a 24-hour period. Dosage is based on weight: 0.15 mg/kg/dose for patients weighing less than 38 kg or more than 114 kg; 8 mg for those weighing 38 to 62 kg; and 12 mg for those weighing 62 to 114 kg.62
Naloxegol
Naloxegol, FDA-approved for treating opioid-induced constipation in 2014, consists of naloxone conjugated with polyethylene glycol, which prevents it from crossing the blood-brain barrier and diminishing the central effects of opioid-induced analgesia. Unlike methylnaltrexone, which is given by subcutaneous injection, naloxegol is taken orally.
Adverse effects reported in clinical trials63,64 were abdominal pain, diarrhea, nausea, headache, and flatulence. No clinically relevant association with QT and corrected QT interval prolongation or cardiac repolarization was noted.64
Dosing is 25 mg by mouth once daily, which can be decreased to 12.5 mg if the initial dose is difficult to tolerate. It should be taken on an empty stomach at least 1 hour before the first meal of the day or 2 hours after the meal. In patients with renal impairment (creatinine clearance < 60 mL/min), the dose is 12.5 mg once daily.65
CONSTIPATION-PREDOMINANT IRRITABLE BOWEL SYNDROME
Irritable bowel syndrome is the reason for 3.1 million office visits and 59 million prescriptions in the United States every year, with patients equally distributed between diarrhea-predominant, constipation-predominant, and mixed subtypes.66
To be diagnosed with constipation-predominant irritable bowel syndrome, patients must meet the Rome IV criteria, more than 25% of bowel movements should have Bristol stool form types 1 or 2, and less than 25% of bowel movements should have Bristol stool form types 6 or 7. In practice, patients reporting that their bowel movements are usually constipated often suffices to make the diagnosis.5
Osmotic laxatives are often tried first, but despite improving stool frequency and consistency, they have little efficacy in satisfying complaints of bloating or abdominal pain in patients with constipation-predominant irritable syndrome.67 Stimulant laxatives have not yet been tested in clinical trials. Lubiprostone and linaclotide are FDA-approved for this condition; in women, lubiprostone is approved only for those over age 18.
Antidepressant therapy
Patients often derive additional benefit from treatment with antidepressants. A meta-analysis demonstrated a number needed to treat of 4 for selective serotonin reuptake inhibitors and tricyclic antidepressants in managing abdominal pain associated with irritable bowel syndrome.68 The major limiting factor is usually adverse effects of these drugs.
For constipation-predominant irritable bowel syndrome, selective serotonin reuptake inhibitors are preferred over tricyclics because of their additional prokinetic properties. Starting at a low dose and titrating upward slowly avoids potential adverse effects.
Cognitive behavioral therapy has also been beneficial in treating irritable bowel syndrome.69
Adjunctive therapies
Adjunctive therapies including peppermint oil, probiotics (eg, Lactobacillus, Bifidobacterium), and acupuncture have also shown promise in managing irritable bowel syndrome, but more data are needed on the use of these therapies for constipation-predominant irritable bowel syndrome before any definite conclusions can be drawn.70 Other emerging pharmacologic therapies are plecanatide (discussed earlier) and tenapanor.
Peppermint oil is an antispasmodic that inhibits calcium channels, leading to relaxation of smooth muscles in the gastrointestinal tract. Different dosages and treatment durations have been studied—450 to 900 mg daily in 2 to 3 divided doses over 1 to 3 months.71,72 The most common adverse effect reported was gastroesophageal reflux, related in part to the oil’s relaxing effect on the lower esophageal sphincter. Observation of this led to the development of enteric-coated preparations that have the potential to bypass the upper gastrointestinal tract.73
Tenapanor inhibits the sodium-hydrogen exchanger 3 channel (a regulator of sodium and water uptake in intestinal lumen), which in turn leads to a higher sodium level in the entire gastrointestinal tract (whereas linaclotide’s action is limited to the duodenum and jejunem), resulting in more fluid volume and increased luminal transit.74 It was found effective in a phase 2 clinical trial,75 and the most effective dose was 50 mg twice daily.
Since tenapanor is minimally absorbed, it has few side effects, the major ones being diarrhea (11.2% vs 0% with placebo) and urinary tract infection (5.6% vs 4.4% with placebo).75 Further study is needed to confirm these findings.
Tenapanor also has the advantage of inhibiting luminal phosphorus absorption. This has led to exploration of its use as a phosphate binder in patients with end-stage renal disease.
DYSSYNERGIC DEFECATION AND ANORECTAL BIOFEEDBACK
According to the Rome IV criteria,5 dyssynergic defecation is present if the criteria for chronic constipation are met, if a dyssynergic pattern of defecation is confirmed by manometry, imaging, or electromyography, and if 1 or more of the following are present: inability to expel an artificial stool (a 50-mL water-filled balloon) within 1 minute, prolonged colonic transit time, inability to evacuate, or 50% or more retention of barium during defecography.5
Even though biofeedback has been controversial as a treatment for dyssynergic defecation because of conflicting results in older studies,76 3 trials have shown it to be better than placebo, laxatives, and muscle relaxants, with symptomatic improvement in 70% of patients.77–79
Biofeedback therapy involves an instrument-based auditory or visual tool (using electromyographic sensors or anorectal manometry) to help patients coordinate abdominal, rectal, puborectalis, and anal sphincter muscles and produce a propulsive force using their abdominal muscles to achieve complete evacuation. Important components of this therapy include:
Proper evacuation positioning (brace-pump technique, which involves sitting on the toilet leaning forward with forearms resting on thighs, shoulders relaxed, and feet placed on a small footstool
Breathing relaxation and training exercises during defecation (no straining, keeping a normal pattern of breathing, and avoiding holding the breath while defecating)
Use of the abdominal muscles by pushing the abdomen forward, along with relaxation of the anal sphincter.80
The anorectal feedback program usually consists of 6 weekly sessions of 45 to 60 minutes each. Limitations of this therapy include unavailability, lack of trained therapists, lack of insurance coverage, and inapplicability to certain patient groups, such as those with dementia or learning disabilities.
SURGERY FOR CHRONIC CONSTIPATION
Surgery for constipation is reserved for patients who continue to have symptoms despite optimal medical therapy.
Total abdominal colectomy and ileorectal anastomosis
Total abdominal colectomy with ileorectal anastomosis is a surgical option for medically intractable slow-transit constipation. Before considering surgery, complete diagnostic testing should be done, including colonic manometry and documentation of whether the patient also has outlet dysfunction.
Even though it has shown excellent outcomes and satisfaction rates as high as 100% in patients with pure slow-transit constipation,81–83 results in older studies in patients with mixed disorders (eg, slow-transit constipation with features of outlet dysfunction) were less predictable.84 More recent studies have reported comparable long-term morbidity and postoperative satisfaction rates in those with pure slow-transit constipation and those with a mixed disorder, indicating that careful patient selection is likely the key to a favorable outcome.85
Partial colectomies based on segmental colon transit time measurements can also be considered in some patients.86
Stapled transanal resection
Stapled transanal resection involves circumferential transanal stapling of the redundant rectal mucosa. It is an option for patients with defecatory disorders, specifically large rectoceles and rectal intussusception not amenable to therapy with pelvic floor retraining exercises.87
The efficacy of this procedure in controlling symptoms and improving quality of life is around 77% to 81% at 12 months, though complication rates as high as 46% and disappointing long-term outcomes have been a deterrent to its widespread acceptance in the United States.88–91
- Mugie SM, Benninga MA, Di Lorenzo C. Epidemiology of constipation in children and adults: a systematic review. Best Pract Res Clin Gastroenterol 2011; 25:3–18.
- Kinnunen O. Study of constipation in a geriatric hospital, day hospital, old people's home and at home. Aging (Milano) 1991; 3:161–170.
- Everhart JE, Go VL, Johannes RS, Fitzsimmons SC, Roth HP, White LR. A longitudinal survey of self-reported bowel habits in the United States. Dig Dis Sci 1989; 34:1153–1162.
- Shah ND, Chitkara DK, Locke GR, Meek PD, Talley NJ. Ambulatory care for constipation in the United States, 1993-2004. Am J Gastroenterol 2008; 103:1746–1753.
- Mearin F, Lacy BE, Chang L, et al. Bowel disorders. Gastroenterology 2016; 150:1393–1407.
- Bharucha AE. Pelvic floor: anatomy and function. Neurogastroenterol Motil 2006; 18:507–519.
- Bharucha AE, Pemberton JH, Locke GR 3rd. American Gastroenterological Association technical review on constipation. Gastroenterology 2013; 144:218–238.
- Grundy D, Al-Chaer ED, Aziz Q, et al. Fundamentals of neurogastroenterology: basic science. Gastroenterology 2006; 130:1391–1411.
- Gallegos-Orozco JF, Foxx-Orenstein AE, Sterler SM, Stoa JM. Chronic constipation in the elderly. Am J Gastroenterol 2012; 107:18–26.
- Mancini I, Bruera E. Constipation in advanced cancer patients. Support Care Cancer 1998; 6:356–364.
- Bassotti G, Chistolini F, Sietchiping-Nzepa F, de Roberto G, Morelli A, Chiarioni G. Biofeedback for pelvic floor dysfunction in constipation. BMJ 2004; 328:393–396.
- American Gastroenterological Association, Bharucha AE, Dorn SD, Lembo A, Pressman A. American Gastroenterological Association medical position statement on constipation. Gastroenterology 2013; 144:211–217.
- Costilla VC, Foxx-Orenstein AE. Constipation in adults: diagnosis and management. Curr Treat Options Gastroenterol 2014; 12:310–321.
- Rao SS, Singh S. Clinical utility of colonic and anorectal manometry in chronic constipation. J Clin Gastroenterol 2010; 44:597–609.
- Minguez M, Herreros B, Sanchiz V, et al. Predictive value of the balloon expulsion test for excluding the diagnosis of pelvic floor dyssynergia in constipation. Gastroenterology 2004; 126:57–62.
- Diamant NE, Kamm MA, Wald A, Whitehead WE. AGA technical review on anorectal testing techniques. Gastroenterology 1999; 116:735–760.
- Pezim ME, Pemberton JH, Levin KE, Litchy WJ, Phillips SF. Parameters of anorectal and colonic motility in health and in severe constipation. Dis Colon Rectum 1993; 36:484–491.
- Bharucha AE, Fletcher JG, Seide B, Riederer SJ, Zinsmeister AR. Phenotypic variation in functional disorders of defecation. Gastroenterology 2005; 128:1199–1210.
- De Schryver AM, Samsom M, Smout AI. Effects of a meal and bisacodyl on colonic motility in healthy volunteers and patients with slow-transit constipation. Dig Dis Sci 2003; 48:1206–1212.
- Villoria A, Serra J, Azpiroz F, Malagelada JR. Physical activity and intestinal gas clearance in patients with bloating. Am J Gastroenterol 2006; 101:2552–2557.
- Sikirov D. Comparison of straining during defecation in three positions: results and implications for human health. Dig Dis Sci 2003; 48:1201–1205.
- Muller-Lissner SA, Kamm MA, Scarpignato C, Wald A. Myths and misconceptions about chronic constipation. Am J Gastroenterol 2005; 100:232–242.
- Voderholzer WA, Schatke W, Muhldorfer BE, Klauser AG, Birkner B, Muller-Lissner SA. Clinical response to dietary fiber treatment of chronic constipation. Am J Gastroenterol 1997; 92:95–98.
- Bijkerk CJ, de Wit NJ, Muris JW, Whorwell PJ, Knottnerus JA, Hoes AW. Soluble or insoluble fibre in irritable bowel syndrome in primary care? Randomised placebo controlled trial. BMJ 2009; 339:b3154.
- Suares NC, Ford AC. Systematic review: the effects of fibre in the management of chronic idiopathic constipation. Aliment Pharmacol Ther 2011; 33:895–901.
- Dipalma JA, Cleveland MV, McGowan J, Herrera JL. A randomized, multicenter, placebo-controlled trial of polyethylene glycol laxative for chronic treatment of chronic constipation. Am J Gastroenterol 2007; 102:1436–1441.
- Lee-Robichaud H, Thomas K, Morgan J, Nelson RL. Lactulose versus polyethylene glycol for chronic constipation. Cochrane Database Syst Rev 2010; 7:CD007570.
- Lederle FA, Busch DL, Mattox KM, West MJ, Aske DM. Cost-effective treatment of constipation in the elderly: a randomized double-blind comparison of sorbitol and lactulose. Am J Med 1990; 89:597–601.
- Nyberg C, Hendel J, Nielsen OH. The safety of osmotically acting cathartics in colonic cleansing. Nat Rev Gastroenterol Hepatol 2010; 7:557–564.
- Ainley EJ, Winwood PJ, Begley JP. Measurement of serum electrolytes and phosphate after sodium phosphate colonoscopy bowel preparation: an evaluation. Dig Dis Sci 2005; 50:1319–1323.
- Kienzle-Horn S, Vix JM, Schuijt C, Peil H, Jordan CC, Kamm MA. Efficacy and safety of bisacodyl in the acute treatment of constipation: a double-blind, randomized, placebo-controlled study. Aliment Pharmacol Ther 2006; 23:1479–1488.
- Kienzle-Horn S, Vix JM, Schuijt C, Peil H, Jordan CC, Kamm MA. Comparison of bisacodyl and sodium picosulphate in the treatment of chronic constipation. Curr Med Res Opin 2007; 23:691–699.
- Mueller-Lissner S, Kamm MA, Wald A, et al. Multicenter, 4-week, double-blind, randomized, placebo-controlled trial of sodium picosulfate in patients with chronic constipation. Am J Gastroenterol 2010; 105:897–903.
- Smith B. Pathologic changes in the colon produced by anthraquinone purgatives. Dis Colon Rectum 1973; 16:455–458.
- Kiernan JA, Heinicke EA. Sennosides do not kill myenteric neurons in the colon of the rat or mouse. Neuroscience 1989; 30:837–842.
- Ottawa (ON): Canadian Agency for Drugs and Technologies in Health. Dioctyl sulfosuccinate or docusate (calcium or sodium) for the prevention or management of constipation: a review of the clinical effectiveness. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0071207/. Accessed April 6, 2017.
- Saad R, Chey WD. Lubiprostone for chronic idiopathic constipation and irritable bowel syndrome with constipation. Expert Rev Gastroenterol Hepatol 2008; 2:497–508.
- Johanson JF, Morton D, Geenen J, Ueno R. Multicenter, 4-week, double-blind, randomized, placebo-controlled trial of lubiprostone, a locally-acting type-2 chloride channel activator, in patients with chronic constipation. Am J Gastroenterol 2008; 103:170–177.
- Harris LA, Crowell MD. Linaclotide, a new direction in the treatment of irritable bowel syndrome and chronic constipation. Curr Opin Mol Ther 2007; 9:403–410.
- Johnston JM, Kurtz CB, Macdougall JE, et al. Linaclotide improves abdominal pain and bowel habits in a phase IIb study of patients with irritable bowel syndrome with constipation. Gastroenterology 2010; 139:1877–1886.e2.
- Lembo AJ, Schneier HA, Shiff SJ, et al. Two randomized trials of linaclotide for chronic constipation. N Engl J Med 2011; 365:527–536.
- Chey WD, Lembo AJ, Lavins BJ, et al. Linaclotide for irritable bowel syndrome with constipation: a 26-week, randomized, double-blind, placebo-controlled trial to evaluate efficacy and safety. Am J Gastroenterol 2012; 107:1702–1712.
- Rao S, Lembo AJ, Shiff SJ, et al. A 12-week, randomized, controlled trial with a 4-week randomized withdrawal period to evaluate the efficacy and safety of linaclotide in irritable bowel syndrome with constipation. Am J Gastroenterol 2012; 107:1714–1725.
- Chey WD, Kurlander J, Eswaran S. Irritable bowel syndrome: a clinical review. JAMA 2015; 313:949–958.
- Shailubhai K, Talluto C, Comiskey S, Foss JA, Joslyn A, Jacob G. Phase II clinical evaluation of SP-304, a guanylate cyclase-C agonist, for treatment of chronic constipation. Am J Gastroenterol 2010; 105:S487–S488.
- Miner P, Surowitz R, Fogel R, et al. Plecanatide, a novel guanylate cyclase-C (GC-C) receptor agonist, is efficacious and safe in patients with chronic idiopathic constipation (CIC): results from a 951 patient, 12-week, multi-center trial (abstract). Gastroenterology 2013; 144:S163.
- Coss-Adame E, Rao SS. Brain and gut interactions in irritable bowel syndrome: new paradigms and new understandings. Curr Gastroenterol Rep 2014; 16:379.
- Mendzelevski B, Ausma J, Chanter DO, et al. Assessment of the cardiac safety of prucalopride in healthy volunteers: a randomized, double-blind, placebo- and positive-controlled thorough QT study. Br J Clin Pharmacol 2012; 73:203–209.
- Camilleri M, Kerstens R, Rykx A, Vandeplassche L. A placebo-controlled trial of prucalopride for severe chronic constipation. N Engl J Med 2008; 358:2344–2354.
- Tack J, van Outryve M, Beyens G, Kerstens R, Vandeplassche L. Prucalopride (Resolor) in the treatment of severe chronic constipation in patients dissatisfied with laxatives. Gut 2009; 58:357–365.
- Quigley EM, Vandeplassche L, Kerstens R, Ausma J. Clinical trial: the efficacy, impact on quality of life, and safety and tolerability of prucalopride in severe chronic constipation—a 12-week, randomized, double-blind, placebo-controlled study. Aliment Pharmacol Ther 2009; 29:315–328.
- Ford AC, Suares NC. Effect of laxatives and pharmacological therapies in chronic idiopathic constipation: systematic review and meta-analysis. Gut 2011; 60:209–218.
- Goldberg M, Li YP, Johanson JF, et al. Clinical trial: the efficacy and tolerability of velusetrag, a selective 5-HT4 agonist with high intrinsic activity, in chronic idiopathic constipation—a 4-week, randomized, double-blind, placebo-controlled, dose-response study. Aliment Pharmacol Ther 2010; 32:1102–1112.
- Palme M, Milner PG, Ellis DJ, Marmon T, Canafax DM. A novel gastrointestinal prokinetic, ATI-7505, increased spontaneous bowel movements (sbms) in a phase II, randomized, placebo-controlled study of patients with chronic idiopathic constipation (CIC). Gastroenterology 2010; 138:S-128–S-129.
- Chey WD, Camilleri M, Chang L, Rikner L, Graffner H. A randomized placebo-controlled phase IIb trial of a3309, a bile acid transporter inhibitor, for chronic idiopathic constipation. Am J Gastroenterol 2011; 106:1803–1812.
- Wong BS, Camilleri M, McKinzie S, Burton D, Graffner H, Zinsmeister AR. Effects of A3309, an ileal bile acid transporter inhibitor, on colonic transit and symptoms in females with functional constipation. Am J Gastroenterol 2011; 106:2154–2164.
- Pappagallo M. Incidence, prevalence, and management of opioid bowel dysfunction. Am J Surg 2001; 182(suppl):11S–18S.
- Bell T, Annunziata K, Leslie JB. Opioid-induced constipation negatively impacts pain management, productivity, and health-related quality of life: findings from the National Health and Wellness Survey. J Opioid Manag 2009; 5:137–144.
- Sykes NP. A volunteer model for the comparison of laxatives in opioid-related constipation. J Pain Symptom Manage 1996; 11:363–369.
- ClinicalTrials.gov. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study of oral MOA-728 for the treatment of opioid- induced bowel dysfunction in subjects with chronic nonmalignant pain. ClinicalTrials.gov Identifier: NCT00547586. https://clinicaltrials.gov/ct2/show/NCT00547586. Accessed March 22, 2017.
- ClinicalTrials.gov. An open-label study to evaluate the long-term safety of subcutaneous MOA-728 for treatment of opioid-induced constipation in subjects with nonmalignant pain. ClinicalTrials.gov Identifier: NCT00804141. https://clinicaltrials.gov/ct2/show/NCT00804141. Accessed April 6, 2017.
- Wyeth Pharmaceuticals. Relistor package insert. http://labeling.pfizer.com/showlabeling.aspx?id=499. Accessed March 22, 2017.
- Webster L, Dhar S, Eldon M, Masuoka L, Lappalainen J, Sostek M. A phase 2, double-blind, randomized, placebo-controlled, dose-escalation study to evaluate the efficacy, safety, and tolerability of naloxegol in patients with opioid-induced constipation. Pain 2013; 154:1542–1550.
- Chey WD, Webster L, Sostek M, Lappalainen J, Barker PN, Tack J. Naloxegol for opioid-induced constipation in patients with noncancer pain. N Engl J Med 2014; 370:2387–2396.
- Jones R, Prommer E, Backstedt D. Naloxegol: a novel therapy in the management of opioid-induced constipation. Am J Hosp Palliat Care 2016; 33:875–880.
- Guilera M, Balboa A, Mearin F. Bowel habit subtypes and temporal patterns in irritable bowel syndrome: systematic review. Am J Gastroenterol 2005; 100:1174–1184.
- Chapman RW, Stanghellini V, Geraint M, Halphen M. Randomized clinical trial: macrogol/PEG 3350 plus electrolytes for treatment of patients with constipation associated with irritable bowel syndrome. Am J Gastroenterol 2013; 108:1508–1515.
- Ford AC, Quigley EM, Lacy BE, et al. Effect of antidepressants and psychological therapies, including hypnotherapy, in irritable bowel syndrome: systematic review and meta-analysis. Am J Gastroenterol 2014; 109:1350–1366.
- Ballou S, Keefer L. Psychological interventions for irritable bowel syndrome and inflammatory bowel diseases. Clin Transl Gastroenterol 2017; 8:e214.
- Ford AC, Moayyedi P, Lacy BE, et al; Task Force on the Management of Functional Bowel Disorders. American College of Gastroenterology monograph on the management of irritable bowel syndrome and chronic idiopathic constipation. Am J Gastroenterol 2014; 109(suppl 1):S2–S27.
- Ford AC, Talley NJ, Spiegel BM, et al. Effect of fibre, antispasmodics, and peppermint oil in the treatment of irritable bowel syndrome: systematic review and meta-analysis. BMJ 2008; 337:a2313.
- Wall GC, Bryant GA, Bottenberg MM, Maki ED, Miesner AR. Irritable bowel syndrome: a concise review of current treatment concepts. World J Gastroenterol 2014; 20:8796–8806.
- Kligler B, Chaudhary S. Peppermint oil. Am Fam Physician 2007; 75:1027–1030.
- Spencer AG, Labonte ED, Rosenbaum DP, et al. Intestinal inhibition of the Na+/H+ exchanger 3 prevents cardiorenal damage in rats and inhibits Na+ uptake in humans. Sci Transl Med 2014; 6:227ra36.
- Rosenbaum DP. A randomized, double-blind, placebo-controlled study to assess the safety and efficacy of AZD1722 for the treatment of constipation-predominant irritable bowel syndrome (IBS-C). 2014. https://clinicaltrials.gov/ct2/show/NCT01923428. Accessed April 6, 2017.
- Rao SS. Biofeedback therapy for dyssynergic (obstructive) defecation. J Clin Gastroenterol 2000; 30:115–116.
- Cadeddu F, Salis F, De Luca E, Ciangola I, Milito G. Efficacy of biofeedback plus transanal stimulation in the management of pelvic floor dyssynergia: a randomized trial. Tech Coloproctol 2015; 19:333–338.
- Chiarioni G, Whitehead WE, Pezza V, Morelli A, Bassotti G. Biofeedback is superior to laxatives for normal transit constipation due to pelvic floor dyssynergia. Gastroenterology 2006; 130:657–664.
- Chiarioni G, Heymen S, Whitehead WE. Biofeedback therapy for dyssynergic defecation. World J Gastroenterol 2006; 12:7069–7074.
- Rao SS. Biofeedback therapy for constipation in adults. Best Pract Res Clin Gastroenterol 2011; 25:159–166.
- Hassan I, Pemberton JH, Young-Fadok TM, et al. Ileorectal anastomosis for slow transit constipation: long-term functional and quality of life results. J Gastrointest Surg 2006; 10:1330–1337.
- You YT, Wang JY, Changchien CR, et al. Segmental colectomy in the management of colonic inertia. Am Surg 1998; 64:775–777.
- Nyam DC, Pemberton JH, Ilstrup DM, Rath DM. Long-term results of surgery for chronic constipation. Dis Colon Rectum 1997; 40:273–279.
- Pemberton JH, Rath DM, Ilstrup DM. Evaluation and surgical treatment of severe chronic constipation. Ann Surg 1991; 214:403–413.
- Reshef A, Alves-Ferreira P, Zutshi M, Hull T, Gurland B. Colectomy for slow transit constipation: effective for patients with coexistent obstructed defecation. Int J Colorectal Dis 2013; 28:841–847.
- Lundin E, Karlbom U, Pahlman L, Graf W. Outcome of segmental colonic resection for slow-transit constipation. Br J Surg 2002; 89:1270–1274.
- Schwandner O, Stuto A, Jayne D, et al. Decision-making algorithm for the STARR procedure in obstructed defecation syndrome: position statement of the group of STARR pioneers. Surg Innov 2008; 15:105–109.
- Titu LV, Riyad K, Carter H, Dixon AR. Stapled transanal rectal resection for obstructed defecation: a cautionary tale. Dis Colon Rectum 2009; 52:1716–1722.
- Goede AC, Glancy D, Carter H, Mills A, Mabey K, Dixon AR. Medium-term results of stapled transanal rectal resection (STARR) for obstructed defecation and symptomatic rectal-anal intussusception. Colorectal Dis 2011; 13:1052–1057.
- Jayne DG, Schwandner O, Stuto A. Stapled transanal rectal resection for obstructed defecation syndrome: one-year results of the european STARR registry. Dis Colon Rectum 2009; 52:1205–1214.
- Madbouly KM, Abbas KS, Hussein AM. Disappointing long-term outcomes after stapled transanal rectal resection for obstructed defecation. World J Surg 2010; 34:2191–2196.
Chronic constipation has a variety of possible causes and mechanisms. Although traditional conservative treatments are still valid and first-line, if these fail, clinicians can choose from a growing list of new treatments, tailored to the cause in the individual patient.
This article discusses how defecation works (or doesn’t), the types of chronic constipation, the available diagnostic tools, and traditional and newer treatments, including some still in development.
THE EPIDEMIOLOGY OF CONSTIPATION
Chronic constipation is one of the most common gastrointestinal disorders, affecting about 15% of all adults and 30% of those over the age of 60.1 It can be a primary disorder or secondary to other factors.
Constipation is more prevalent in women and in institutionalized elderly people.2 It is associated with lower socioeconomic status, depression, less self-reported physical activity, certain medications, and stressful life events.3 Given its high prevalence and its impact on quality of life, it is also associated with significant utilization of healthcare resources.4
Constipation defined by Rome IV criteria
Physicians and patients may disagree about what constitutes constipation. Physicians primarily regard it as infrequent bowel movements, while patients tend to have a broader definition. According to the Rome IV criteria,5 chronic constipation is defined by the presence of the following for at least 3 months (with symptom onset at least 6 months prior to diagnosis):
(1) Two or more of the following for more than 25% of defecations:
- Straining
- Lumpy or hard stools
- Sensation of incomplete evacuation
- Sensation of anorectal obstruction or blockage
- Manual maneuvers to facilitate evacuation
- Fewer than 3 spontaneous bowel movements per week.
(2) Loose stools are rarely present without the use of laxatives.
(3) The patient does not meet the criteria for diagnosis of irritable bowel syndrome.
DEFECATION IS COMPLEX
Defecation begins when the rectum fills with stool, causing relaxation of the internal anal sphincter and the urge to defecate. The external anal sphincter, which is under voluntary control, can then either contract to delay defecation or relax to allow the stool to be expelled.6
Colonic muscles propel stool toward the rectum in repetitive localized contractions that help mix and promote absorption of the content, and larger coordinated (high-amplitude propagating) contractions that, in healthy individuals, move the stool forward from the proximal to the distal colon multiple times daily. These contractions usually occur in the morning and are accentuated by gastric distention from food and the resulting gastrocolic reflex.
Serotonin (5-HT) is released by enterochromaffin cells in response to distention of the gut wall. It mediates peristaltic movements of the gastrointestinal tract by binding to receptors (especially 5-HT4), stimulating release of neurotransmitters such as acetylcholine, causing smooth-muscle contraction behind the luminal contents and propelling them forward.
PRIMARY CONSTIPATION DISORDERS
The American Gastroenterological Association7 classifies constipation into 3 groups on the basis of colonic transit time and anorectal function:
Normal-transit constipation
Stool normally takes 20 to 72 hours to pass through the colon, with transit time affected by diet, drugs, level of physical activity, and emotional status.8
Normal-transit constipation is the most common type of constipation. The term is sometimes used interchangeably with constipation-predominant irritable bowel syndrome, but the latter is a distinct entity characterized by abdominal pain relieved by defecation as the primary symptom, as well as having occasional loose stools. These 2 conditions can be hard to tell apart, especially if the patient cannot describe the symptoms precisely.
Slow-transit constipation
Slow-transit constipation—also called delayed-transit constipation, colonoparesis, colonic inertia, and pseudo-obstruction—is defined as prolonged stool transit in the colon, ie, for more than 5 days.9 It can be the result of colonic smooth muscle dysfunction, compromised colonic neural pathways, or both, leading to slow colon peristalsis.
Factors that can affect colonic motility such as opioid use and hypothyroidism should be carefully considered in these patients. Opioids are notorious for causing constipation by decreasing bowel tone and contractility and thereby increasing colonic transit time. They also tighten up the anal sphincters, resulting in decreased rectal evacuation.10
Outlet dysfunction
Outlet dysfunction, also called pelvic floor dysfunction or defecatory disorder, is associated with incomplete rectal evacuation. It can be a consequence of weak rectal expulsion forces (slow colonic transit, rectal hyposensitivity), functional resistance to rectal evacuation (high anal resting pressure, anismus, incomplete relaxation of the anal sphincter, dyssynergic defecation), or structural outlet obstruction (excessive perineal descent, rectoceles, rectal intussusception). About 50% of patients with outlet dysfunction have concurrent slow-transit constipation.
Dyssynergic defecation is the most common outlet dysfunction disorder, accounting for about half of the cases referred to tertiary centers. It is defined as a paradoxical elevation in anal sphincter tone or less than 20% relaxation of the resting anal sphincter pressure with weak abdominal and pelvic propulsive forces.11 Anorectal biofeedback is a therapeutic option for dyssynergic defecation, as we discuss later in this article.
SECONDARY CONSTIPATION
Constipation can be secondary to several conditions and factors (Table 1), including:
- Neurologic disorders that affect gastrointestinal motility (eg, Hirschsprung disease, Parkinson disease, multiple sclerosis, spinal cord injury, stroke, spinal or ganglionic tumor, hypothyroidism, amyloidosis, diabetes mellitus, hypercalcemia)
- Drugs used to treat neurologic disorders
- Mechanical obstruction
- Diet (eg, low fiber, decreased fluid intake).
EVALUATION OF CONSTIPATION
It is crucial for physicians to efficiently use the available diagnostic tools for constipation to tailor the treatment to the patient.
Evaluation of chronic constipation begins with a thorough history and physical examination to rule out secondary constipation (Figure 1). Red flags such as unintentional weight loss, blood in the stool, rectal pain, fever, and iron-deficiency anemia should prompt referral for colonoscopy to evaluate for malignancy, colitis, or other potential colonic abnormalities.12
A detailed perineal and rectal examination can help diagnose defecatory disorders and should include evaluation of the resting anal tone and the sphincter during simulated evacuation.
Laboratory tests of thyroid function, electrolytes, and a complete blood cell count should be ordered if clinically indicated.13
Further tests
Further diagnostic tests can be considered if symptoms persist despite conservative treatment or if a defecatory disorder is suspected. These include anorectal manometry, colonic transit studies, defecography, and colonic manometry.
Anorectal manometry and the rectal balloon expulsion test are usually done first because of their high sensitivity (88%) and specificity (89%) for defecatory disorders.14 These tests measure the function of the internal and external anal sphincters at rest and with straining and assess rectal sensitivity and compliance. Anorectal manometry is also used in biofeedback therapy in patients with dyssynergic defecation.15
Colonic transit time can be measured if anorectal manometry and the balloon expulsion test are normal. The study uses radiopaque markers, radioisotopes, or wireless motility capsules to confirm slow-transit constipation and to identify areas of delayed transit in the colon.16
Defecography is usually the next step in diagnosis if anorectal manometry and balloon expulsion tests are inconclusive or if an anatomic abnormality of the pelvic floor is suspected. It can be done with a variety of techniques. Barium defecography can identify anatomic defects, scintigraphy can quantify evacuation of artificial stools, and magnetic resonance defecography visualizes anatomic landmarks to assess pelvic floor motion without exposing the patient to radiation.17,18
Colonic manometry is most useful in patients with refractory slow-transit constipation and can identify patients with isolated colonic motor dysfunction with no pelvic floor dysfunction who may benefit from subtotal colectomy and end-ileostomy.7
TRADITIONAL TREATMENTS STILL THE MAINSTAY
Nonpharmacologic treatments are the first-line options for patients with normal-transit and slow-transit constipation and should precede diagnostic testing. Lifestyle modifications and dietary changes (Table 2) aim to augment the known factors that stimulate the gastrocolic reflex and increase intestinal motility by high-amplitude propagated contractions.
Increasing physical activity increases intestinal gas clearance, decreases bloating, and lessens constipation.19,20
Toilet training is an integral part of lifestyle modifications.21
Diet. Drinking hot caffeinated beverages, eating breakfast within an hour of waking up, and consuming fiber in the morning (25–30 g of fiber daily) have traditionally been recommended as the first-line measures for chronic constipation. Dehydrated patients with constipation also benefit from increasing their fluid intake.22
LAXATIVES
Fiber (bulk-forming laxatives) for normal-transit constipation
Fiber remains a key part of the initial management of chronic constipation, as it is cheap, available, and safe. Increasing fiber intake is effective for normal-transit constipation, but patients with slow-transit constipation or refractory outlet dysfunction are less likely to benefit.23 Other laxatives are incorporated into the regimen if first-line nonpharmacologic interventions fail (Table 3).
Bulk-forming laxatives include insoluble fiber (wheat bran) and soluble fiber (psyllium, methylcellulose, inulin, calcium polycarbophil). Insoluble fiber, though often used, has little impact on symptoms of chronic constipation after 1 month of use, and up to 60% of patients report adverse effects from it.24 On the other hand, clinical trials have shown that soluble fiber such as psyllium facilitates defecation and improves functional bowel symptoms in patients with normal-transit constipation.25
Patients should be instructed to increase their dietary fiber intake gradually to avoid adverse effects and should be told to expect significant symptomatic improvement only after a few weeks. They should also be informed that increasing dietary fiber intake can cause bloating but that the bloating is temporary. If it continues, a different fiber can be tried.
Osmotic laxatives
Osmotic laxatives are often employed as a first- line laxative treatment option for patients with constipation. They draw water into the lumen by osmosis, helping to soften stool and speed intestinal transit. They include macrogols (inert polymers of ethylene glycol), nonabsorbable carbohydrates (lactulose, sorbitol), magnesium products, and sodium phosphate products.
Polyethylene glycol, the most studied osmotic laxative, has been shown to maintain therapeutic efficacy for up to 2 years, though it is not generally used this long.26 A meta-analysis of 10 randomized clinical trials found it to be superior to lactulose in improving stool consistency and frequency, and rates of adverse effects were similar to those with placebo.27
Lactulose and sorbitol are semisynthetic disaccharides that are not absorbed from the gastrointestinal tract. Apart from the osmotic effect of the disaccharide, these sugars are metabolized by colonic bacteria to acetic acid and other short-chain fatty acids, resulting in acidification of the stool, which exerts an osmotic effect in the colonic lumen.
Lactulose and sorbitol were shown to have similar efficacy in increasing the frequency of bowel movements in a small study, though patients taking lactulose had a higher rate of nausea.28
The usual recommended dose is 15 to 30 mL once or twice daily.
Adverse effects include gas, bloating, and abdominal distention (due to fermentation by colonic bacteria) and can limit long-term use.
Magnesium citrate and magnesium hydroxide are strong osmotic laxatives, but so far no clinical trial has been done to assess their efficacy in constipation. Although the risk of hypermagnesemia is low with magnesium-based products, this group of laxatives is generally avoided in patients with renal or cardiac disease.29
Sodium phosphate enemas (Fleet enemas) are used for bowel cleansing before certain procedures but have only limited use in constipation because of potential adverse effects such as hyperphosphatemia, hypocalcemia, and the rarer but more serious complication of acute phosphate nephropathy.30
Stimulant laxatives for short-term use only
Stimulant laxatives include glycerin, bisacodyl, senna, and sodium picosulfate. Sodium piosulfate and bisacodyl have been validated for treatment of chronic constipation for up to 4 weeks.31–33
Stimulant laxative suppositories should be used 30 minutes after meals to augment the physiologic gastrocolic reflex.
As more evidence is available for osmotic laxatives such as polyethylene glycol, they tend to be preferred over stimulant agents, especially for long-term use. Clinicians have traditionally hesitated to prescribe stimulant laxatives for long-term use, as they were thought to damage the enteric nervous system.34 Although more recent studies have not shown this potential effect,35 more research is warranted on the use of stimulant laxatives for longer than 4 weeks.
STOOL SOFTENERS: LITTLE EVIDENCE
Stool softeners enhance the interaction of stool and water, leading to softer stool and easier evacuation. Docusate sodium and docusate calcium are thought to facilitate the mixing of aqueous and fatty substances, thereby softening the stool.
However, there is little evidence to support the use of docusate for constipation in hospitalized adults or in ambulatory care. A recent review reported that docusate was no better than placebo in diminishing symptoms of constipation.36
INTESTINAL SECRETAGOGUES
The secretagogues include lubiprostone, linaclotide, and plecanatide. These medications are preferred therapy for patients with normal- or slow-transit constipation once conservative therapies have failed. Even though there is no current consensus, lifestyle measures and conservative treatment options should be tried for about 8 weeks.
Lubiprostone and linaclotide are approved by the US Food and Drug Administration (FDA) for both constipation and constipation-predominant irritable bowel syndrome. They activate chloride channels on the apical surface of enterocytes, increasing intestinal secretion of chloride, which in turn increases luminal sodium efflux to maintain electroneutrality, leading to secretion of water into the intestinal lumen. This eventually facilitates intestinal transit and increases the passage of stool.
Lubiprostone
Lubiprostone, a prostaglandin E1 derivative, is approved for treating chronic constipation, constipation-predominant irritable bowel syndrome in women, and opioid-induced constipation in patients with chronic noncancer pain.
Adverse effects in clinical trials were nausea (up to 30%) and headache.37,38
Linaclotide
Linaclotide, a minimally absorbed 14-amino acid peptide, increases intestinal secretion of chloride and bicarbonate, increasing intestinal fluid and promoting intestinal transit.39 It also decreases the firing rate of the visceral afferent pain fibers and helps reduce visceral pain, especially in patients with constipation-predominant irritable bowel syndrome.40 It is approved for chronic constipation and constipation-predominant irritable bowel syndrome.41–43
Dosage starts at 145 μg/day for chronic constipation, and can be titrated up to 290 μg if there is no response or if a diagnosis of constipation-predominant irritable bowel syndrome is under consideration. Linaclotide should be taken 30 to 60 minutes before breakfast to reduce the likelihood of diarrhea.44
Adverse effects. Diarrhea led to treatment discontinuation in 4.5% of patients in one study.42
Plecanatide
Plecanatide is a guanylate cyclase-c agonist with a mode of action similar to that of linaclotide. It was recently approved by the FDA for chronic idiopathic constipation in adults. The recommended dose is 3 mg once daily.
Data from phase 2 trials in chronic constipation showed improvement in straining, abdominal discomfort, and stool frequency after 14 days of treatment.45
A phase 3 trial showed that plecanatide was more effective than placebo when used for 12 weeks in 951 patients with chronic constipation (P = .009).46 The most common adverse effect reported was diarrhea.
SEROTONIN RECEPTOR AGONISTS
Activation of serotonin 5-HT4 receptors in the gut leads to release of acetylcholine, which in turn induces mucosal secretion by activating submucosal neurons and increasing gut motility.47
Two 5-HT4 receptor agonists were withdrawn from the market (cisapride in 2000 and tegaserod in 2007) due to serious cardiovascular adverse events (fatal arrhythmias, heart attacks, and strokes) resulting from their affinity for hERG-K+ cardiac channels.
The newer agents prucalopride,48 velusetrag, and naronapride are highly selective 5-HT4 agonists with low affinity for hERG-K+ receptors and do not have proarrhythmic properties, based on extensive assessment in clinical trials.
Prucalopride
Prucalopride has been shown to accelerate gastrointestinal and colonic transit in patients with chronic constipation, with improvement in bowel movements, symptoms of chronic constipation, and quality of life.49–52
Adverse effects reported with its use have been headache, nausea, abdominal pain, and cramps.
Prucalopride is approved in Europe and Canada for chronic constipation in women but is not yet approved in the United States.
Dosage is 2 mg orally once daily. Caution is advised in elderly patients, in whom the preferred maximum dose is 1 mg daily, as there are only limited data available on the safety of this medication in the elderly.
Velusetrag
Velusetrag has been shown to increase colonic motility and improve symptoms of chronic constipation. In a phase 2 trial,53 the most effective dose was 15 mg once daily. Higher doses were associated with a higher incidence of adverse effects such as diarrhea, headache, nausea, and vomiting.
Naronapride
Naronapride (ATI-7505) is in phase 2 trials for chronic constipation. Reported adverse effects were headache, diarrhea, nausea, and vomiting.54
BILE SALT ABSORPTION INHIBITORS
Bile acids exert prosecretory and prokinetic effects by increasing colonic secretion of water and electrolytes through the activation of adenylate cyclase. This happens as a result of their deconjugation after passage into the colon.
Elobixibat is an ileal bile acid transporter inhibitor that prevents absorption of nonconjugated bile salts in the distal ileum. It has few side effects because its systemic absorption is minimal. Phase 3 trials are under way. Dosage is 5 to 20 mg daily. Adverse effects are few because systemic absorption is minimal, but include abdominal pain and diarrhea.55,56
MANAGING OPIOID-INDUCED CONSTIPATION
Opioids cause constipation by binding to mu receptors in the enteric nervous system. Activation of these receptors decreases bowel tone and contractility, which increases transit time. Stimulation of these receptors also increases anal sphincter tone, resulting in decreased rectal evacuation.57
Though underrecognized, opioid-induced constipation affects 40% of patients who take these drugs for nonmalignant pain and 90% of those taking them for cancer pain. Patients with this condition were found to take more time off work and feel more impaired in their domestic and work-related obligations than patients who did not develop constipation with use of opioids.58
Initial management of opioid-induced constipation includes increasing intake of fluids and dietary fiber (fiber alone can worsen abdominal pain in this condition by increasing stool bulk without a concomitant improvement in peristalsis) and increasing physical activity. It is common clinical practice to use a stool softener along with a stimulant laxative if lifestyle modifications are inadequate.59 If these measures are ineffective, osmotic agents can be added.
If these conventional measures fail, a peripherally acting mu-opioid receptor antagonist such as methylnaltrexone or naloxegol should be considered.
Methylnaltrexone
Methylnaltrexone60,61 is a peripherally acting mu receptor antagonist with a rapid onset of action. It does not cross the blood-brain barrier, as it contains a methyl group. It was approved by the FDA in 2008 to treat opioid-induced constipation in adults with advanced illnesses when other approaches are ineffective.
Adverse effects. Although the mu receptor antagonist alvimopan had been shown to be associated with cardiovascular events hypothesized to be a consequence of opioid withdrawal, methylnaltrexone has been deemed to have a safe cardiovascular profile without any potential effects on platelets, corrected QT interval, metabolism, heart rate, or blood pressure.61 Side effects include abdominal pain, nausea, diarrhea, hot flashes, tremor, and chills.
Contraindications. Methylnaltrexone is contraindicated in patients with structural diseases of the gastrointestinal tract, ie, peptic ulcer disease, inflammatory bowel disease, diverticulitis, stomach or intestinal cancer) since it can increase the risk of perforation.
Dosing is 1 dose subcutaneously every other day, as needed, and no more than 1 dose in a 24-hour period. Dosage is based on weight: 0.15 mg/kg/dose for patients weighing less than 38 kg or more than 114 kg; 8 mg for those weighing 38 to 62 kg; and 12 mg for those weighing 62 to 114 kg.62
Naloxegol
Naloxegol, FDA-approved for treating opioid-induced constipation in 2014, consists of naloxone conjugated with polyethylene glycol, which prevents it from crossing the blood-brain barrier and diminishing the central effects of opioid-induced analgesia. Unlike methylnaltrexone, which is given by subcutaneous injection, naloxegol is taken orally.
Adverse effects reported in clinical trials63,64 were abdominal pain, diarrhea, nausea, headache, and flatulence. No clinically relevant association with QT and corrected QT interval prolongation or cardiac repolarization was noted.64
Dosing is 25 mg by mouth once daily, which can be decreased to 12.5 mg if the initial dose is difficult to tolerate. It should be taken on an empty stomach at least 1 hour before the first meal of the day or 2 hours after the meal. In patients with renal impairment (creatinine clearance < 60 mL/min), the dose is 12.5 mg once daily.65
CONSTIPATION-PREDOMINANT IRRITABLE BOWEL SYNDROME
Irritable bowel syndrome is the reason for 3.1 million office visits and 59 million prescriptions in the United States every year, with patients equally distributed between diarrhea-predominant, constipation-predominant, and mixed subtypes.66
To be diagnosed with constipation-predominant irritable bowel syndrome, patients must meet the Rome IV criteria, more than 25% of bowel movements should have Bristol stool form types 1 or 2, and less than 25% of bowel movements should have Bristol stool form types 6 or 7. In practice, patients reporting that their bowel movements are usually constipated often suffices to make the diagnosis.5
Osmotic laxatives are often tried first, but despite improving stool frequency and consistency, they have little efficacy in satisfying complaints of bloating or abdominal pain in patients with constipation-predominant irritable syndrome.67 Stimulant laxatives have not yet been tested in clinical trials. Lubiprostone and linaclotide are FDA-approved for this condition; in women, lubiprostone is approved only for those over age 18.
Antidepressant therapy
Patients often derive additional benefit from treatment with antidepressants. A meta-analysis demonstrated a number needed to treat of 4 for selective serotonin reuptake inhibitors and tricyclic antidepressants in managing abdominal pain associated with irritable bowel syndrome.68 The major limiting factor is usually adverse effects of these drugs.
For constipation-predominant irritable bowel syndrome, selective serotonin reuptake inhibitors are preferred over tricyclics because of their additional prokinetic properties. Starting at a low dose and titrating upward slowly avoids potential adverse effects.
Cognitive behavioral therapy has also been beneficial in treating irritable bowel syndrome.69
Adjunctive therapies
Adjunctive therapies including peppermint oil, probiotics (eg, Lactobacillus, Bifidobacterium), and acupuncture have also shown promise in managing irritable bowel syndrome, but more data are needed on the use of these therapies for constipation-predominant irritable bowel syndrome before any definite conclusions can be drawn.70 Other emerging pharmacologic therapies are plecanatide (discussed earlier) and tenapanor.
Peppermint oil is an antispasmodic that inhibits calcium channels, leading to relaxation of smooth muscles in the gastrointestinal tract. Different dosages and treatment durations have been studied—450 to 900 mg daily in 2 to 3 divided doses over 1 to 3 months.71,72 The most common adverse effect reported was gastroesophageal reflux, related in part to the oil’s relaxing effect on the lower esophageal sphincter. Observation of this led to the development of enteric-coated preparations that have the potential to bypass the upper gastrointestinal tract.73
Tenapanor inhibits the sodium-hydrogen exchanger 3 channel (a regulator of sodium and water uptake in intestinal lumen), which in turn leads to a higher sodium level in the entire gastrointestinal tract (whereas linaclotide’s action is limited to the duodenum and jejunem), resulting in more fluid volume and increased luminal transit.74 It was found effective in a phase 2 clinical trial,75 and the most effective dose was 50 mg twice daily.
Since tenapanor is minimally absorbed, it has few side effects, the major ones being diarrhea (11.2% vs 0% with placebo) and urinary tract infection (5.6% vs 4.4% with placebo).75 Further study is needed to confirm these findings.
Tenapanor also has the advantage of inhibiting luminal phosphorus absorption. This has led to exploration of its use as a phosphate binder in patients with end-stage renal disease.
DYSSYNERGIC DEFECATION AND ANORECTAL BIOFEEDBACK
According to the Rome IV criteria,5 dyssynergic defecation is present if the criteria for chronic constipation are met, if a dyssynergic pattern of defecation is confirmed by manometry, imaging, or electromyography, and if 1 or more of the following are present: inability to expel an artificial stool (a 50-mL water-filled balloon) within 1 minute, prolonged colonic transit time, inability to evacuate, or 50% or more retention of barium during defecography.5
Even though biofeedback has been controversial as a treatment for dyssynergic defecation because of conflicting results in older studies,76 3 trials have shown it to be better than placebo, laxatives, and muscle relaxants, with symptomatic improvement in 70% of patients.77–79
Biofeedback therapy involves an instrument-based auditory or visual tool (using electromyographic sensors or anorectal manometry) to help patients coordinate abdominal, rectal, puborectalis, and anal sphincter muscles and produce a propulsive force using their abdominal muscles to achieve complete evacuation. Important components of this therapy include:
Proper evacuation positioning (brace-pump technique, which involves sitting on the toilet leaning forward with forearms resting on thighs, shoulders relaxed, and feet placed on a small footstool
Breathing relaxation and training exercises during defecation (no straining, keeping a normal pattern of breathing, and avoiding holding the breath while defecating)
Use of the abdominal muscles by pushing the abdomen forward, along with relaxation of the anal sphincter.80
The anorectal feedback program usually consists of 6 weekly sessions of 45 to 60 minutes each. Limitations of this therapy include unavailability, lack of trained therapists, lack of insurance coverage, and inapplicability to certain patient groups, such as those with dementia or learning disabilities.
SURGERY FOR CHRONIC CONSTIPATION
Surgery for constipation is reserved for patients who continue to have symptoms despite optimal medical therapy.
Total abdominal colectomy and ileorectal anastomosis
Total abdominal colectomy with ileorectal anastomosis is a surgical option for medically intractable slow-transit constipation. Before considering surgery, complete diagnostic testing should be done, including colonic manometry and documentation of whether the patient also has outlet dysfunction.
Even though it has shown excellent outcomes and satisfaction rates as high as 100% in patients with pure slow-transit constipation,81–83 results in older studies in patients with mixed disorders (eg, slow-transit constipation with features of outlet dysfunction) were less predictable.84 More recent studies have reported comparable long-term morbidity and postoperative satisfaction rates in those with pure slow-transit constipation and those with a mixed disorder, indicating that careful patient selection is likely the key to a favorable outcome.85
Partial colectomies based on segmental colon transit time measurements can also be considered in some patients.86
Stapled transanal resection
Stapled transanal resection involves circumferential transanal stapling of the redundant rectal mucosa. It is an option for patients with defecatory disorders, specifically large rectoceles and rectal intussusception not amenable to therapy with pelvic floor retraining exercises.87
The efficacy of this procedure in controlling symptoms and improving quality of life is around 77% to 81% at 12 months, though complication rates as high as 46% and disappointing long-term outcomes have been a deterrent to its widespread acceptance in the United States.88–91
Chronic constipation has a variety of possible causes and mechanisms. Although traditional conservative treatments are still valid and first-line, if these fail, clinicians can choose from a growing list of new treatments, tailored to the cause in the individual patient.
This article discusses how defecation works (or doesn’t), the types of chronic constipation, the available diagnostic tools, and traditional and newer treatments, including some still in development.
THE EPIDEMIOLOGY OF CONSTIPATION
Chronic constipation is one of the most common gastrointestinal disorders, affecting about 15% of all adults and 30% of those over the age of 60.1 It can be a primary disorder or secondary to other factors.
Constipation is more prevalent in women and in institutionalized elderly people.2 It is associated with lower socioeconomic status, depression, less self-reported physical activity, certain medications, and stressful life events.3 Given its high prevalence and its impact on quality of life, it is also associated with significant utilization of healthcare resources.4
Constipation defined by Rome IV criteria
Physicians and patients may disagree about what constitutes constipation. Physicians primarily regard it as infrequent bowel movements, while patients tend to have a broader definition. According to the Rome IV criteria,5 chronic constipation is defined by the presence of the following for at least 3 months (with symptom onset at least 6 months prior to diagnosis):
(1) Two or more of the following for more than 25% of defecations:
- Straining
- Lumpy or hard stools
- Sensation of incomplete evacuation
- Sensation of anorectal obstruction or blockage
- Manual maneuvers to facilitate evacuation
- Fewer than 3 spontaneous bowel movements per week.
(2) Loose stools are rarely present without the use of laxatives.
(3) The patient does not meet the criteria for diagnosis of irritable bowel syndrome.
DEFECATION IS COMPLEX
Defecation begins when the rectum fills with stool, causing relaxation of the internal anal sphincter and the urge to defecate. The external anal sphincter, which is under voluntary control, can then either contract to delay defecation or relax to allow the stool to be expelled.6
Colonic muscles propel stool toward the rectum in repetitive localized contractions that help mix and promote absorption of the content, and larger coordinated (high-amplitude propagating) contractions that, in healthy individuals, move the stool forward from the proximal to the distal colon multiple times daily. These contractions usually occur in the morning and are accentuated by gastric distention from food and the resulting gastrocolic reflex.
Serotonin (5-HT) is released by enterochromaffin cells in response to distention of the gut wall. It mediates peristaltic movements of the gastrointestinal tract by binding to receptors (especially 5-HT4), stimulating release of neurotransmitters such as acetylcholine, causing smooth-muscle contraction behind the luminal contents and propelling them forward.
PRIMARY CONSTIPATION DISORDERS
The American Gastroenterological Association7 classifies constipation into 3 groups on the basis of colonic transit time and anorectal function:
Normal-transit constipation
Stool normally takes 20 to 72 hours to pass through the colon, with transit time affected by diet, drugs, level of physical activity, and emotional status.8
Normal-transit constipation is the most common type of constipation. The term is sometimes used interchangeably with constipation-predominant irritable bowel syndrome, but the latter is a distinct entity characterized by abdominal pain relieved by defecation as the primary symptom, as well as having occasional loose stools. These 2 conditions can be hard to tell apart, especially if the patient cannot describe the symptoms precisely.
Slow-transit constipation
Slow-transit constipation—also called delayed-transit constipation, colonoparesis, colonic inertia, and pseudo-obstruction—is defined as prolonged stool transit in the colon, ie, for more than 5 days.9 It can be the result of colonic smooth muscle dysfunction, compromised colonic neural pathways, or both, leading to slow colon peristalsis.
Factors that can affect colonic motility such as opioid use and hypothyroidism should be carefully considered in these patients. Opioids are notorious for causing constipation by decreasing bowel tone and contractility and thereby increasing colonic transit time. They also tighten up the anal sphincters, resulting in decreased rectal evacuation.10
Outlet dysfunction
Outlet dysfunction, also called pelvic floor dysfunction or defecatory disorder, is associated with incomplete rectal evacuation. It can be a consequence of weak rectal expulsion forces (slow colonic transit, rectal hyposensitivity), functional resistance to rectal evacuation (high anal resting pressure, anismus, incomplete relaxation of the anal sphincter, dyssynergic defecation), or structural outlet obstruction (excessive perineal descent, rectoceles, rectal intussusception). About 50% of patients with outlet dysfunction have concurrent slow-transit constipation.
Dyssynergic defecation is the most common outlet dysfunction disorder, accounting for about half of the cases referred to tertiary centers. It is defined as a paradoxical elevation in anal sphincter tone or less than 20% relaxation of the resting anal sphincter pressure with weak abdominal and pelvic propulsive forces.11 Anorectal biofeedback is a therapeutic option for dyssynergic defecation, as we discuss later in this article.
SECONDARY CONSTIPATION
Constipation can be secondary to several conditions and factors (Table 1), including:
- Neurologic disorders that affect gastrointestinal motility (eg, Hirschsprung disease, Parkinson disease, multiple sclerosis, spinal cord injury, stroke, spinal or ganglionic tumor, hypothyroidism, amyloidosis, diabetes mellitus, hypercalcemia)
- Drugs used to treat neurologic disorders
- Mechanical obstruction
- Diet (eg, low fiber, decreased fluid intake).
EVALUATION OF CONSTIPATION
It is crucial for physicians to efficiently use the available diagnostic tools for constipation to tailor the treatment to the patient.
Evaluation of chronic constipation begins with a thorough history and physical examination to rule out secondary constipation (Figure 1). Red flags such as unintentional weight loss, blood in the stool, rectal pain, fever, and iron-deficiency anemia should prompt referral for colonoscopy to evaluate for malignancy, colitis, or other potential colonic abnormalities.12
A detailed perineal and rectal examination can help diagnose defecatory disorders and should include evaluation of the resting anal tone and the sphincter during simulated evacuation.
Laboratory tests of thyroid function, electrolytes, and a complete blood cell count should be ordered if clinically indicated.13
Further tests
Further diagnostic tests can be considered if symptoms persist despite conservative treatment or if a defecatory disorder is suspected. These include anorectal manometry, colonic transit studies, defecography, and colonic manometry.
Anorectal manometry and the rectal balloon expulsion test are usually done first because of their high sensitivity (88%) and specificity (89%) for defecatory disorders.14 These tests measure the function of the internal and external anal sphincters at rest and with straining and assess rectal sensitivity and compliance. Anorectal manometry is also used in biofeedback therapy in patients with dyssynergic defecation.15
Colonic transit time can be measured if anorectal manometry and the balloon expulsion test are normal. The study uses radiopaque markers, radioisotopes, or wireless motility capsules to confirm slow-transit constipation and to identify areas of delayed transit in the colon.16
Defecography is usually the next step in diagnosis if anorectal manometry and balloon expulsion tests are inconclusive or if an anatomic abnormality of the pelvic floor is suspected. It can be done with a variety of techniques. Barium defecography can identify anatomic defects, scintigraphy can quantify evacuation of artificial stools, and magnetic resonance defecography visualizes anatomic landmarks to assess pelvic floor motion without exposing the patient to radiation.17,18
Colonic manometry is most useful in patients with refractory slow-transit constipation and can identify patients with isolated colonic motor dysfunction with no pelvic floor dysfunction who may benefit from subtotal colectomy and end-ileostomy.7
TRADITIONAL TREATMENTS STILL THE MAINSTAY
Nonpharmacologic treatments are the first-line options for patients with normal-transit and slow-transit constipation and should precede diagnostic testing. Lifestyle modifications and dietary changes (Table 2) aim to augment the known factors that stimulate the gastrocolic reflex and increase intestinal motility by high-amplitude propagated contractions.
Increasing physical activity increases intestinal gas clearance, decreases bloating, and lessens constipation.19,20
Toilet training is an integral part of lifestyle modifications.21
Diet. Drinking hot caffeinated beverages, eating breakfast within an hour of waking up, and consuming fiber in the morning (25–30 g of fiber daily) have traditionally been recommended as the first-line measures for chronic constipation. Dehydrated patients with constipation also benefit from increasing their fluid intake.22
LAXATIVES
Fiber (bulk-forming laxatives) for normal-transit constipation
Fiber remains a key part of the initial management of chronic constipation, as it is cheap, available, and safe. Increasing fiber intake is effective for normal-transit constipation, but patients with slow-transit constipation or refractory outlet dysfunction are less likely to benefit.23 Other laxatives are incorporated into the regimen if first-line nonpharmacologic interventions fail (Table 3).
Bulk-forming laxatives include insoluble fiber (wheat bran) and soluble fiber (psyllium, methylcellulose, inulin, calcium polycarbophil). Insoluble fiber, though often used, has little impact on symptoms of chronic constipation after 1 month of use, and up to 60% of patients report adverse effects from it.24 On the other hand, clinical trials have shown that soluble fiber such as psyllium facilitates defecation and improves functional bowel symptoms in patients with normal-transit constipation.25
Patients should be instructed to increase their dietary fiber intake gradually to avoid adverse effects and should be told to expect significant symptomatic improvement only after a few weeks. They should also be informed that increasing dietary fiber intake can cause bloating but that the bloating is temporary. If it continues, a different fiber can be tried.
Osmotic laxatives
Osmotic laxatives are often employed as a first- line laxative treatment option for patients with constipation. They draw water into the lumen by osmosis, helping to soften stool and speed intestinal transit. They include macrogols (inert polymers of ethylene glycol), nonabsorbable carbohydrates (lactulose, sorbitol), magnesium products, and sodium phosphate products.
Polyethylene glycol, the most studied osmotic laxative, has been shown to maintain therapeutic efficacy for up to 2 years, though it is not generally used this long.26 A meta-analysis of 10 randomized clinical trials found it to be superior to lactulose in improving stool consistency and frequency, and rates of adverse effects were similar to those with placebo.27
Lactulose and sorbitol are semisynthetic disaccharides that are not absorbed from the gastrointestinal tract. Apart from the osmotic effect of the disaccharide, these sugars are metabolized by colonic bacteria to acetic acid and other short-chain fatty acids, resulting in acidification of the stool, which exerts an osmotic effect in the colonic lumen.
Lactulose and sorbitol were shown to have similar efficacy in increasing the frequency of bowel movements in a small study, though patients taking lactulose had a higher rate of nausea.28
The usual recommended dose is 15 to 30 mL once or twice daily.
Adverse effects include gas, bloating, and abdominal distention (due to fermentation by colonic bacteria) and can limit long-term use.
Magnesium citrate and magnesium hydroxide are strong osmotic laxatives, but so far no clinical trial has been done to assess their efficacy in constipation. Although the risk of hypermagnesemia is low with magnesium-based products, this group of laxatives is generally avoided in patients with renal or cardiac disease.29
Sodium phosphate enemas (Fleet enemas) are used for bowel cleansing before certain procedures but have only limited use in constipation because of potential adverse effects such as hyperphosphatemia, hypocalcemia, and the rarer but more serious complication of acute phosphate nephropathy.30
Stimulant laxatives for short-term use only
Stimulant laxatives include glycerin, bisacodyl, senna, and sodium picosulfate. Sodium piosulfate and bisacodyl have been validated for treatment of chronic constipation for up to 4 weeks.31–33
Stimulant laxative suppositories should be used 30 minutes after meals to augment the physiologic gastrocolic reflex.
As more evidence is available for osmotic laxatives such as polyethylene glycol, they tend to be preferred over stimulant agents, especially for long-term use. Clinicians have traditionally hesitated to prescribe stimulant laxatives for long-term use, as they were thought to damage the enteric nervous system.34 Although more recent studies have not shown this potential effect,35 more research is warranted on the use of stimulant laxatives for longer than 4 weeks.
STOOL SOFTENERS: LITTLE EVIDENCE
Stool softeners enhance the interaction of stool and water, leading to softer stool and easier evacuation. Docusate sodium and docusate calcium are thought to facilitate the mixing of aqueous and fatty substances, thereby softening the stool.
However, there is little evidence to support the use of docusate for constipation in hospitalized adults or in ambulatory care. A recent review reported that docusate was no better than placebo in diminishing symptoms of constipation.36
INTESTINAL SECRETAGOGUES
The secretagogues include lubiprostone, linaclotide, and plecanatide. These medications are preferred therapy for patients with normal- or slow-transit constipation once conservative therapies have failed. Even though there is no current consensus, lifestyle measures and conservative treatment options should be tried for about 8 weeks.
Lubiprostone and linaclotide are approved by the US Food and Drug Administration (FDA) for both constipation and constipation-predominant irritable bowel syndrome. They activate chloride channels on the apical surface of enterocytes, increasing intestinal secretion of chloride, which in turn increases luminal sodium efflux to maintain electroneutrality, leading to secretion of water into the intestinal lumen. This eventually facilitates intestinal transit and increases the passage of stool.
Lubiprostone
Lubiprostone, a prostaglandin E1 derivative, is approved for treating chronic constipation, constipation-predominant irritable bowel syndrome in women, and opioid-induced constipation in patients with chronic noncancer pain.
Adverse effects in clinical trials were nausea (up to 30%) and headache.37,38
Linaclotide
Linaclotide, a minimally absorbed 14-amino acid peptide, increases intestinal secretion of chloride and bicarbonate, increasing intestinal fluid and promoting intestinal transit.39 It also decreases the firing rate of the visceral afferent pain fibers and helps reduce visceral pain, especially in patients with constipation-predominant irritable bowel syndrome.40 It is approved for chronic constipation and constipation-predominant irritable bowel syndrome.41–43
Dosage starts at 145 μg/day for chronic constipation, and can be titrated up to 290 μg if there is no response or if a diagnosis of constipation-predominant irritable bowel syndrome is under consideration. Linaclotide should be taken 30 to 60 minutes before breakfast to reduce the likelihood of diarrhea.44
Adverse effects. Diarrhea led to treatment discontinuation in 4.5% of patients in one study.42
Plecanatide
Plecanatide is a guanylate cyclase-c agonist with a mode of action similar to that of linaclotide. It was recently approved by the FDA for chronic idiopathic constipation in adults. The recommended dose is 3 mg once daily.
Data from phase 2 trials in chronic constipation showed improvement in straining, abdominal discomfort, and stool frequency after 14 days of treatment.45
A phase 3 trial showed that plecanatide was more effective than placebo when used for 12 weeks in 951 patients with chronic constipation (P = .009).46 The most common adverse effect reported was diarrhea.
SEROTONIN RECEPTOR AGONISTS
Activation of serotonin 5-HT4 receptors in the gut leads to release of acetylcholine, which in turn induces mucosal secretion by activating submucosal neurons and increasing gut motility.47
Two 5-HT4 receptor agonists were withdrawn from the market (cisapride in 2000 and tegaserod in 2007) due to serious cardiovascular adverse events (fatal arrhythmias, heart attacks, and strokes) resulting from their affinity for hERG-K+ cardiac channels.
The newer agents prucalopride,48 velusetrag, and naronapride are highly selective 5-HT4 agonists with low affinity for hERG-K+ receptors and do not have proarrhythmic properties, based on extensive assessment in clinical trials.
Prucalopride
Prucalopride has been shown to accelerate gastrointestinal and colonic transit in patients with chronic constipation, with improvement in bowel movements, symptoms of chronic constipation, and quality of life.49–52
Adverse effects reported with its use have been headache, nausea, abdominal pain, and cramps.
Prucalopride is approved in Europe and Canada for chronic constipation in women but is not yet approved in the United States.
Dosage is 2 mg orally once daily. Caution is advised in elderly patients, in whom the preferred maximum dose is 1 mg daily, as there are only limited data available on the safety of this medication in the elderly.
Velusetrag
Velusetrag has been shown to increase colonic motility and improve symptoms of chronic constipation. In a phase 2 trial,53 the most effective dose was 15 mg once daily. Higher doses were associated with a higher incidence of adverse effects such as diarrhea, headache, nausea, and vomiting.
Naronapride
Naronapride (ATI-7505) is in phase 2 trials for chronic constipation. Reported adverse effects were headache, diarrhea, nausea, and vomiting.54
BILE SALT ABSORPTION INHIBITORS
Bile acids exert prosecretory and prokinetic effects by increasing colonic secretion of water and electrolytes through the activation of adenylate cyclase. This happens as a result of their deconjugation after passage into the colon.
Elobixibat is an ileal bile acid transporter inhibitor that prevents absorption of nonconjugated bile salts in the distal ileum. It has few side effects because its systemic absorption is minimal. Phase 3 trials are under way. Dosage is 5 to 20 mg daily. Adverse effects are few because systemic absorption is minimal, but include abdominal pain and diarrhea.55,56
MANAGING OPIOID-INDUCED CONSTIPATION
Opioids cause constipation by binding to mu receptors in the enteric nervous system. Activation of these receptors decreases bowel tone and contractility, which increases transit time. Stimulation of these receptors also increases anal sphincter tone, resulting in decreased rectal evacuation.57
Though underrecognized, opioid-induced constipation affects 40% of patients who take these drugs for nonmalignant pain and 90% of those taking them for cancer pain. Patients with this condition were found to take more time off work and feel more impaired in their domestic and work-related obligations than patients who did not develop constipation with use of opioids.58
Initial management of opioid-induced constipation includes increasing intake of fluids and dietary fiber (fiber alone can worsen abdominal pain in this condition by increasing stool bulk without a concomitant improvement in peristalsis) and increasing physical activity. It is common clinical practice to use a stool softener along with a stimulant laxative if lifestyle modifications are inadequate.59 If these measures are ineffective, osmotic agents can be added.
If these conventional measures fail, a peripherally acting mu-opioid receptor antagonist such as methylnaltrexone or naloxegol should be considered.
Methylnaltrexone
Methylnaltrexone60,61 is a peripherally acting mu receptor antagonist with a rapid onset of action. It does not cross the blood-brain barrier, as it contains a methyl group. It was approved by the FDA in 2008 to treat opioid-induced constipation in adults with advanced illnesses when other approaches are ineffective.
Adverse effects. Although the mu receptor antagonist alvimopan had been shown to be associated with cardiovascular events hypothesized to be a consequence of opioid withdrawal, methylnaltrexone has been deemed to have a safe cardiovascular profile without any potential effects on platelets, corrected QT interval, metabolism, heart rate, or blood pressure.61 Side effects include abdominal pain, nausea, diarrhea, hot flashes, tremor, and chills.
Contraindications. Methylnaltrexone is contraindicated in patients with structural diseases of the gastrointestinal tract, ie, peptic ulcer disease, inflammatory bowel disease, diverticulitis, stomach or intestinal cancer) since it can increase the risk of perforation.
Dosing is 1 dose subcutaneously every other day, as needed, and no more than 1 dose in a 24-hour period. Dosage is based on weight: 0.15 mg/kg/dose for patients weighing less than 38 kg or more than 114 kg; 8 mg for those weighing 38 to 62 kg; and 12 mg for those weighing 62 to 114 kg.62
Naloxegol
Naloxegol, FDA-approved for treating opioid-induced constipation in 2014, consists of naloxone conjugated with polyethylene glycol, which prevents it from crossing the blood-brain barrier and diminishing the central effects of opioid-induced analgesia. Unlike methylnaltrexone, which is given by subcutaneous injection, naloxegol is taken orally.
Adverse effects reported in clinical trials63,64 were abdominal pain, diarrhea, nausea, headache, and flatulence. No clinically relevant association with QT and corrected QT interval prolongation or cardiac repolarization was noted.64
Dosing is 25 mg by mouth once daily, which can be decreased to 12.5 mg if the initial dose is difficult to tolerate. It should be taken on an empty stomach at least 1 hour before the first meal of the day or 2 hours after the meal. In patients with renal impairment (creatinine clearance < 60 mL/min), the dose is 12.5 mg once daily.65
CONSTIPATION-PREDOMINANT IRRITABLE BOWEL SYNDROME
Irritable bowel syndrome is the reason for 3.1 million office visits and 59 million prescriptions in the United States every year, with patients equally distributed between diarrhea-predominant, constipation-predominant, and mixed subtypes.66
To be diagnosed with constipation-predominant irritable bowel syndrome, patients must meet the Rome IV criteria, more than 25% of bowel movements should have Bristol stool form types 1 or 2, and less than 25% of bowel movements should have Bristol stool form types 6 or 7. In practice, patients reporting that their bowel movements are usually constipated often suffices to make the diagnosis.5
Osmotic laxatives are often tried first, but despite improving stool frequency and consistency, they have little efficacy in satisfying complaints of bloating or abdominal pain in patients with constipation-predominant irritable syndrome.67 Stimulant laxatives have not yet been tested in clinical trials. Lubiprostone and linaclotide are FDA-approved for this condition; in women, lubiprostone is approved only for those over age 18.
Antidepressant therapy
Patients often derive additional benefit from treatment with antidepressants. A meta-analysis demonstrated a number needed to treat of 4 for selective serotonin reuptake inhibitors and tricyclic antidepressants in managing abdominal pain associated with irritable bowel syndrome.68 The major limiting factor is usually adverse effects of these drugs.
For constipation-predominant irritable bowel syndrome, selective serotonin reuptake inhibitors are preferred over tricyclics because of their additional prokinetic properties. Starting at a low dose and titrating upward slowly avoids potential adverse effects.
Cognitive behavioral therapy has also been beneficial in treating irritable bowel syndrome.69
Adjunctive therapies
Adjunctive therapies including peppermint oil, probiotics (eg, Lactobacillus, Bifidobacterium), and acupuncture have also shown promise in managing irritable bowel syndrome, but more data are needed on the use of these therapies for constipation-predominant irritable bowel syndrome before any definite conclusions can be drawn.70 Other emerging pharmacologic therapies are plecanatide (discussed earlier) and tenapanor.
Peppermint oil is an antispasmodic that inhibits calcium channels, leading to relaxation of smooth muscles in the gastrointestinal tract. Different dosages and treatment durations have been studied—450 to 900 mg daily in 2 to 3 divided doses over 1 to 3 months.71,72 The most common adverse effect reported was gastroesophageal reflux, related in part to the oil’s relaxing effect on the lower esophageal sphincter. Observation of this led to the development of enteric-coated preparations that have the potential to bypass the upper gastrointestinal tract.73
Tenapanor inhibits the sodium-hydrogen exchanger 3 channel (a regulator of sodium and water uptake in intestinal lumen), which in turn leads to a higher sodium level in the entire gastrointestinal tract (whereas linaclotide’s action is limited to the duodenum and jejunem), resulting in more fluid volume and increased luminal transit.74 It was found effective in a phase 2 clinical trial,75 and the most effective dose was 50 mg twice daily.
Since tenapanor is minimally absorbed, it has few side effects, the major ones being diarrhea (11.2% vs 0% with placebo) and urinary tract infection (5.6% vs 4.4% with placebo).75 Further study is needed to confirm these findings.
Tenapanor also has the advantage of inhibiting luminal phosphorus absorption. This has led to exploration of its use as a phosphate binder in patients with end-stage renal disease.
DYSSYNERGIC DEFECATION AND ANORECTAL BIOFEEDBACK
According to the Rome IV criteria,5 dyssynergic defecation is present if the criteria for chronic constipation are met, if a dyssynergic pattern of defecation is confirmed by manometry, imaging, or electromyography, and if 1 or more of the following are present: inability to expel an artificial stool (a 50-mL water-filled balloon) within 1 minute, prolonged colonic transit time, inability to evacuate, or 50% or more retention of barium during defecography.5
Even though biofeedback has been controversial as a treatment for dyssynergic defecation because of conflicting results in older studies,76 3 trials have shown it to be better than placebo, laxatives, and muscle relaxants, with symptomatic improvement in 70% of patients.77–79
Biofeedback therapy involves an instrument-based auditory or visual tool (using electromyographic sensors or anorectal manometry) to help patients coordinate abdominal, rectal, puborectalis, and anal sphincter muscles and produce a propulsive force using their abdominal muscles to achieve complete evacuation. Important components of this therapy include:
Proper evacuation positioning (brace-pump technique, which involves sitting on the toilet leaning forward with forearms resting on thighs, shoulders relaxed, and feet placed on a small footstool
Breathing relaxation and training exercises during defecation (no straining, keeping a normal pattern of breathing, and avoiding holding the breath while defecating)
Use of the abdominal muscles by pushing the abdomen forward, along with relaxation of the anal sphincter.80
The anorectal feedback program usually consists of 6 weekly sessions of 45 to 60 minutes each. Limitations of this therapy include unavailability, lack of trained therapists, lack of insurance coverage, and inapplicability to certain patient groups, such as those with dementia or learning disabilities.
SURGERY FOR CHRONIC CONSTIPATION
Surgery for constipation is reserved for patients who continue to have symptoms despite optimal medical therapy.
Total abdominal colectomy and ileorectal anastomosis
Total abdominal colectomy with ileorectal anastomosis is a surgical option for medically intractable slow-transit constipation. Before considering surgery, complete diagnostic testing should be done, including colonic manometry and documentation of whether the patient also has outlet dysfunction.
Even though it has shown excellent outcomes and satisfaction rates as high as 100% in patients with pure slow-transit constipation,81–83 results in older studies in patients with mixed disorders (eg, slow-transit constipation with features of outlet dysfunction) were less predictable.84 More recent studies have reported comparable long-term morbidity and postoperative satisfaction rates in those with pure slow-transit constipation and those with a mixed disorder, indicating that careful patient selection is likely the key to a favorable outcome.85
Partial colectomies based on segmental colon transit time measurements can also be considered in some patients.86
Stapled transanal resection
Stapled transanal resection involves circumferential transanal stapling of the redundant rectal mucosa. It is an option for patients with defecatory disorders, specifically large rectoceles and rectal intussusception not amenable to therapy with pelvic floor retraining exercises.87
The efficacy of this procedure in controlling symptoms and improving quality of life is around 77% to 81% at 12 months, though complication rates as high as 46% and disappointing long-term outcomes have been a deterrent to its widespread acceptance in the United States.88–91
- Mugie SM, Benninga MA, Di Lorenzo C. Epidemiology of constipation in children and adults: a systematic review. Best Pract Res Clin Gastroenterol 2011; 25:3–18.
- Kinnunen O. Study of constipation in a geriatric hospital, day hospital, old people's home and at home. Aging (Milano) 1991; 3:161–170.
- Everhart JE, Go VL, Johannes RS, Fitzsimmons SC, Roth HP, White LR. A longitudinal survey of self-reported bowel habits in the United States. Dig Dis Sci 1989; 34:1153–1162.
- Shah ND, Chitkara DK, Locke GR, Meek PD, Talley NJ. Ambulatory care for constipation in the United States, 1993-2004. Am J Gastroenterol 2008; 103:1746–1753.
- Mearin F, Lacy BE, Chang L, et al. Bowel disorders. Gastroenterology 2016; 150:1393–1407.
- Bharucha AE. Pelvic floor: anatomy and function. Neurogastroenterol Motil 2006; 18:507–519.
- Bharucha AE, Pemberton JH, Locke GR 3rd. American Gastroenterological Association technical review on constipation. Gastroenterology 2013; 144:218–238.
- Grundy D, Al-Chaer ED, Aziz Q, et al. Fundamentals of neurogastroenterology: basic science. Gastroenterology 2006; 130:1391–1411.
- Gallegos-Orozco JF, Foxx-Orenstein AE, Sterler SM, Stoa JM. Chronic constipation in the elderly. Am J Gastroenterol 2012; 107:18–26.
- Mancini I, Bruera E. Constipation in advanced cancer patients. Support Care Cancer 1998; 6:356–364.
- Bassotti G, Chistolini F, Sietchiping-Nzepa F, de Roberto G, Morelli A, Chiarioni G. Biofeedback for pelvic floor dysfunction in constipation. BMJ 2004; 328:393–396.
- American Gastroenterological Association, Bharucha AE, Dorn SD, Lembo A, Pressman A. American Gastroenterological Association medical position statement on constipation. Gastroenterology 2013; 144:211–217.
- Costilla VC, Foxx-Orenstein AE. Constipation in adults: diagnosis and management. Curr Treat Options Gastroenterol 2014; 12:310–321.
- Rao SS, Singh S. Clinical utility of colonic and anorectal manometry in chronic constipation. J Clin Gastroenterol 2010; 44:597–609.
- Minguez M, Herreros B, Sanchiz V, et al. Predictive value of the balloon expulsion test for excluding the diagnosis of pelvic floor dyssynergia in constipation. Gastroenterology 2004; 126:57–62.
- Diamant NE, Kamm MA, Wald A, Whitehead WE. AGA technical review on anorectal testing techniques. Gastroenterology 1999; 116:735–760.
- Pezim ME, Pemberton JH, Levin KE, Litchy WJ, Phillips SF. Parameters of anorectal and colonic motility in health and in severe constipation. Dis Colon Rectum 1993; 36:484–491.
- Bharucha AE, Fletcher JG, Seide B, Riederer SJ, Zinsmeister AR. Phenotypic variation in functional disorders of defecation. Gastroenterology 2005; 128:1199–1210.
- De Schryver AM, Samsom M, Smout AI. Effects of a meal and bisacodyl on colonic motility in healthy volunteers and patients with slow-transit constipation. Dig Dis Sci 2003; 48:1206–1212.
- Villoria A, Serra J, Azpiroz F, Malagelada JR. Physical activity and intestinal gas clearance in patients with bloating. Am J Gastroenterol 2006; 101:2552–2557.
- Sikirov D. Comparison of straining during defecation in three positions: results and implications for human health. Dig Dis Sci 2003; 48:1201–1205.
- Muller-Lissner SA, Kamm MA, Scarpignato C, Wald A. Myths and misconceptions about chronic constipation. Am J Gastroenterol 2005; 100:232–242.
- Voderholzer WA, Schatke W, Muhldorfer BE, Klauser AG, Birkner B, Muller-Lissner SA. Clinical response to dietary fiber treatment of chronic constipation. Am J Gastroenterol 1997; 92:95–98.
- Bijkerk CJ, de Wit NJ, Muris JW, Whorwell PJ, Knottnerus JA, Hoes AW. Soluble or insoluble fibre in irritable bowel syndrome in primary care? Randomised placebo controlled trial. BMJ 2009; 339:b3154.
- Suares NC, Ford AC. Systematic review: the effects of fibre in the management of chronic idiopathic constipation. Aliment Pharmacol Ther 2011; 33:895–901.
- Dipalma JA, Cleveland MV, McGowan J, Herrera JL. A randomized, multicenter, placebo-controlled trial of polyethylene glycol laxative for chronic treatment of chronic constipation. Am J Gastroenterol 2007; 102:1436–1441.
- Lee-Robichaud H, Thomas K, Morgan J, Nelson RL. Lactulose versus polyethylene glycol for chronic constipation. Cochrane Database Syst Rev 2010; 7:CD007570.
- Lederle FA, Busch DL, Mattox KM, West MJ, Aske DM. Cost-effective treatment of constipation in the elderly: a randomized double-blind comparison of sorbitol and lactulose. Am J Med 1990; 89:597–601.
- Nyberg C, Hendel J, Nielsen OH. The safety of osmotically acting cathartics in colonic cleansing. Nat Rev Gastroenterol Hepatol 2010; 7:557–564.
- Ainley EJ, Winwood PJ, Begley JP. Measurement of serum electrolytes and phosphate after sodium phosphate colonoscopy bowel preparation: an evaluation. Dig Dis Sci 2005; 50:1319–1323.
- Kienzle-Horn S, Vix JM, Schuijt C, Peil H, Jordan CC, Kamm MA. Efficacy and safety of bisacodyl in the acute treatment of constipation: a double-blind, randomized, placebo-controlled study. Aliment Pharmacol Ther 2006; 23:1479–1488.
- Kienzle-Horn S, Vix JM, Schuijt C, Peil H, Jordan CC, Kamm MA. Comparison of bisacodyl and sodium picosulphate in the treatment of chronic constipation. Curr Med Res Opin 2007; 23:691–699.
- Mueller-Lissner S, Kamm MA, Wald A, et al. Multicenter, 4-week, double-blind, randomized, placebo-controlled trial of sodium picosulfate in patients with chronic constipation. Am J Gastroenterol 2010; 105:897–903.
- Smith B. Pathologic changes in the colon produced by anthraquinone purgatives. Dis Colon Rectum 1973; 16:455–458.
- Kiernan JA, Heinicke EA. Sennosides do not kill myenteric neurons in the colon of the rat or mouse. Neuroscience 1989; 30:837–842.
- Ottawa (ON): Canadian Agency for Drugs and Technologies in Health. Dioctyl sulfosuccinate or docusate (calcium or sodium) for the prevention or management of constipation: a review of the clinical effectiveness. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0071207/. Accessed April 6, 2017.
- Saad R, Chey WD. Lubiprostone for chronic idiopathic constipation and irritable bowel syndrome with constipation. Expert Rev Gastroenterol Hepatol 2008; 2:497–508.
- Johanson JF, Morton D, Geenen J, Ueno R. Multicenter, 4-week, double-blind, randomized, placebo-controlled trial of lubiprostone, a locally-acting type-2 chloride channel activator, in patients with chronic constipation. Am J Gastroenterol 2008; 103:170–177.
- Harris LA, Crowell MD. Linaclotide, a new direction in the treatment of irritable bowel syndrome and chronic constipation. Curr Opin Mol Ther 2007; 9:403–410.
- Johnston JM, Kurtz CB, Macdougall JE, et al. Linaclotide improves abdominal pain and bowel habits in a phase IIb study of patients with irritable bowel syndrome with constipation. Gastroenterology 2010; 139:1877–1886.e2.
- Lembo AJ, Schneier HA, Shiff SJ, et al. Two randomized trials of linaclotide for chronic constipation. N Engl J Med 2011; 365:527–536.
- Chey WD, Lembo AJ, Lavins BJ, et al. Linaclotide for irritable bowel syndrome with constipation: a 26-week, randomized, double-blind, placebo-controlled trial to evaluate efficacy and safety. Am J Gastroenterol 2012; 107:1702–1712.
- Rao S, Lembo AJ, Shiff SJ, et al. A 12-week, randomized, controlled trial with a 4-week randomized withdrawal period to evaluate the efficacy and safety of linaclotide in irritable bowel syndrome with constipation. Am J Gastroenterol 2012; 107:1714–1725.
- Chey WD, Kurlander J, Eswaran S. Irritable bowel syndrome: a clinical review. JAMA 2015; 313:949–958.
- Shailubhai K, Talluto C, Comiskey S, Foss JA, Joslyn A, Jacob G. Phase II clinical evaluation of SP-304, a guanylate cyclase-C agonist, for treatment of chronic constipation. Am J Gastroenterol 2010; 105:S487–S488.
- Miner P, Surowitz R, Fogel R, et al. Plecanatide, a novel guanylate cyclase-C (GC-C) receptor agonist, is efficacious and safe in patients with chronic idiopathic constipation (CIC): results from a 951 patient, 12-week, multi-center trial (abstract). Gastroenterology 2013; 144:S163.
- Coss-Adame E, Rao SS. Brain and gut interactions in irritable bowel syndrome: new paradigms and new understandings. Curr Gastroenterol Rep 2014; 16:379.
- Mendzelevski B, Ausma J, Chanter DO, et al. Assessment of the cardiac safety of prucalopride in healthy volunteers: a randomized, double-blind, placebo- and positive-controlled thorough QT study. Br J Clin Pharmacol 2012; 73:203–209.
- Camilleri M, Kerstens R, Rykx A, Vandeplassche L. A placebo-controlled trial of prucalopride for severe chronic constipation. N Engl J Med 2008; 358:2344–2354.
- Tack J, van Outryve M, Beyens G, Kerstens R, Vandeplassche L. Prucalopride (Resolor) in the treatment of severe chronic constipation in patients dissatisfied with laxatives. Gut 2009; 58:357–365.
- Quigley EM, Vandeplassche L, Kerstens R, Ausma J. Clinical trial: the efficacy, impact on quality of life, and safety and tolerability of prucalopride in severe chronic constipation—a 12-week, randomized, double-blind, placebo-controlled study. Aliment Pharmacol Ther 2009; 29:315–328.
- Ford AC, Suares NC. Effect of laxatives and pharmacological therapies in chronic idiopathic constipation: systematic review and meta-analysis. Gut 2011; 60:209–218.
- Goldberg M, Li YP, Johanson JF, et al. Clinical trial: the efficacy and tolerability of velusetrag, a selective 5-HT4 agonist with high intrinsic activity, in chronic idiopathic constipation—a 4-week, randomized, double-blind, placebo-controlled, dose-response study. Aliment Pharmacol Ther 2010; 32:1102–1112.
- Palme M, Milner PG, Ellis DJ, Marmon T, Canafax DM. A novel gastrointestinal prokinetic, ATI-7505, increased spontaneous bowel movements (sbms) in a phase II, randomized, placebo-controlled study of patients with chronic idiopathic constipation (CIC). Gastroenterology 2010; 138:S-128–S-129.
- Chey WD, Camilleri M, Chang L, Rikner L, Graffner H. A randomized placebo-controlled phase IIb trial of a3309, a bile acid transporter inhibitor, for chronic idiopathic constipation. Am J Gastroenterol 2011; 106:1803–1812.
- Wong BS, Camilleri M, McKinzie S, Burton D, Graffner H, Zinsmeister AR. Effects of A3309, an ileal bile acid transporter inhibitor, on colonic transit and symptoms in females with functional constipation. Am J Gastroenterol 2011; 106:2154–2164.
- Pappagallo M. Incidence, prevalence, and management of opioid bowel dysfunction. Am J Surg 2001; 182(suppl):11S–18S.
- Bell T, Annunziata K, Leslie JB. Opioid-induced constipation negatively impacts pain management, productivity, and health-related quality of life: findings from the National Health and Wellness Survey. J Opioid Manag 2009; 5:137–144.
- Sykes NP. A volunteer model for the comparison of laxatives in opioid-related constipation. J Pain Symptom Manage 1996; 11:363–369.
- ClinicalTrials.gov. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study of oral MOA-728 for the treatment of opioid- induced bowel dysfunction in subjects with chronic nonmalignant pain. ClinicalTrials.gov Identifier: NCT00547586. https://clinicaltrials.gov/ct2/show/NCT00547586. Accessed March 22, 2017.
- ClinicalTrials.gov. An open-label study to evaluate the long-term safety of subcutaneous MOA-728 for treatment of opioid-induced constipation in subjects with nonmalignant pain. ClinicalTrials.gov Identifier: NCT00804141. https://clinicaltrials.gov/ct2/show/NCT00804141. Accessed April 6, 2017.
- Wyeth Pharmaceuticals. Relistor package insert. http://labeling.pfizer.com/showlabeling.aspx?id=499. Accessed March 22, 2017.
- Webster L, Dhar S, Eldon M, Masuoka L, Lappalainen J, Sostek M. A phase 2, double-blind, randomized, placebo-controlled, dose-escalation study to evaluate the efficacy, safety, and tolerability of naloxegol in patients with opioid-induced constipation. Pain 2013; 154:1542–1550.
- Chey WD, Webster L, Sostek M, Lappalainen J, Barker PN, Tack J. Naloxegol for opioid-induced constipation in patients with noncancer pain. N Engl J Med 2014; 370:2387–2396.
- Jones R, Prommer E, Backstedt D. Naloxegol: a novel therapy in the management of opioid-induced constipation. Am J Hosp Palliat Care 2016; 33:875–880.
- Guilera M, Balboa A, Mearin F. Bowel habit subtypes and temporal patterns in irritable bowel syndrome: systematic review. Am J Gastroenterol 2005; 100:1174–1184.
- Chapman RW, Stanghellini V, Geraint M, Halphen M. Randomized clinical trial: macrogol/PEG 3350 plus electrolytes for treatment of patients with constipation associated with irritable bowel syndrome. Am J Gastroenterol 2013; 108:1508–1515.
- Ford AC, Quigley EM, Lacy BE, et al. Effect of antidepressants and psychological therapies, including hypnotherapy, in irritable bowel syndrome: systematic review and meta-analysis. Am J Gastroenterol 2014; 109:1350–1366.
- Ballou S, Keefer L. Psychological interventions for irritable bowel syndrome and inflammatory bowel diseases. Clin Transl Gastroenterol 2017; 8:e214.
- Ford AC, Moayyedi P, Lacy BE, et al; Task Force on the Management of Functional Bowel Disorders. American College of Gastroenterology monograph on the management of irritable bowel syndrome and chronic idiopathic constipation. Am J Gastroenterol 2014; 109(suppl 1):S2–S27.
- Ford AC, Talley NJ, Spiegel BM, et al. Effect of fibre, antispasmodics, and peppermint oil in the treatment of irritable bowel syndrome: systematic review and meta-analysis. BMJ 2008; 337:a2313.
- Wall GC, Bryant GA, Bottenberg MM, Maki ED, Miesner AR. Irritable bowel syndrome: a concise review of current treatment concepts. World J Gastroenterol 2014; 20:8796–8806.
- Kligler B, Chaudhary S. Peppermint oil. Am Fam Physician 2007; 75:1027–1030.
- Spencer AG, Labonte ED, Rosenbaum DP, et al. Intestinal inhibition of the Na+/H+ exchanger 3 prevents cardiorenal damage in rats and inhibits Na+ uptake in humans. Sci Transl Med 2014; 6:227ra36.
- Rosenbaum DP. A randomized, double-blind, placebo-controlled study to assess the safety and efficacy of AZD1722 for the treatment of constipation-predominant irritable bowel syndrome (IBS-C). 2014. https://clinicaltrials.gov/ct2/show/NCT01923428. Accessed April 6, 2017.
- Rao SS. Biofeedback therapy for dyssynergic (obstructive) defecation. J Clin Gastroenterol 2000; 30:115–116.
- Cadeddu F, Salis F, De Luca E, Ciangola I, Milito G. Efficacy of biofeedback plus transanal stimulation in the management of pelvic floor dyssynergia: a randomized trial. Tech Coloproctol 2015; 19:333–338.
- Chiarioni G, Whitehead WE, Pezza V, Morelli A, Bassotti G. Biofeedback is superior to laxatives for normal transit constipation due to pelvic floor dyssynergia. Gastroenterology 2006; 130:657–664.
- Chiarioni G, Heymen S, Whitehead WE. Biofeedback therapy for dyssynergic defecation. World J Gastroenterol 2006; 12:7069–7074.
- Rao SS. Biofeedback therapy for constipation in adults. Best Pract Res Clin Gastroenterol 2011; 25:159–166.
- Hassan I, Pemberton JH, Young-Fadok TM, et al. Ileorectal anastomosis for slow transit constipation: long-term functional and quality of life results. J Gastrointest Surg 2006; 10:1330–1337.
- You YT, Wang JY, Changchien CR, et al. Segmental colectomy in the management of colonic inertia. Am Surg 1998; 64:775–777.
- Nyam DC, Pemberton JH, Ilstrup DM, Rath DM. Long-term results of surgery for chronic constipation. Dis Colon Rectum 1997; 40:273–279.
- Pemberton JH, Rath DM, Ilstrup DM. Evaluation and surgical treatment of severe chronic constipation. Ann Surg 1991; 214:403–413.
- Reshef A, Alves-Ferreira P, Zutshi M, Hull T, Gurland B. Colectomy for slow transit constipation: effective for patients with coexistent obstructed defecation. Int J Colorectal Dis 2013; 28:841–847.
- Lundin E, Karlbom U, Pahlman L, Graf W. Outcome of segmental colonic resection for slow-transit constipation. Br J Surg 2002; 89:1270–1274.
- Schwandner O, Stuto A, Jayne D, et al. Decision-making algorithm for the STARR procedure in obstructed defecation syndrome: position statement of the group of STARR pioneers. Surg Innov 2008; 15:105–109.
- Titu LV, Riyad K, Carter H, Dixon AR. Stapled transanal rectal resection for obstructed defecation: a cautionary tale. Dis Colon Rectum 2009; 52:1716–1722.
- Goede AC, Glancy D, Carter H, Mills A, Mabey K, Dixon AR. Medium-term results of stapled transanal rectal resection (STARR) for obstructed defecation and symptomatic rectal-anal intussusception. Colorectal Dis 2011; 13:1052–1057.
- Jayne DG, Schwandner O, Stuto A. Stapled transanal rectal resection for obstructed defecation syndrome: one-year results of the european STARR registry. Dis Colon Rectum 2009; 52:1205–1214.
- Madbouly KM, Abbas KS, Hussein AM. Disappointing long-term outcomes after stapled transanal rectal resection for obstructed defecation. World J Surg 2010; 34:2191–2196.
- Mugie SM, Benninga MA, Di Lorenzo C. Epidemiology of constipation in children and adults: a systematic review. Best Pract Res Clin Gastroenterol 2011; 25:3–18.
- Kinnunen O. Study of constipation in a geriatric hospital, day hospital, old people's home and at home. Aging (Milano) 1991; 3:161–170.
- Everhart JE, Go VL, Johannes RS, Fitzsimmons SC, Roth HP, White LR. A longitudinal survey of self-reported bowel habits in the United States. Dig Dis Sci 1989; 34:1153–1162.
- Shah ND, Chitkara DK, Locke GR, Meek PD, Talley NJ. Ambulatory care for constipation in the United States, 1993-2004. Am J Gastroenterol 2008; 103:1746–1753.
- Mearin F, Lacy BE, Chang L, et al. Bowel disorders. Gastroenterology 2016; 150:1393–1407.
- Bharucha AE. Pelvic floor: anatomy and function. Neurogastroenterol Motil 2006; 18:507–519.
- Bharucha AE, Pemberton JH, Locke GR 3rd. American Gastroenterological Association technical review on constipation. Gastroenterology 2013; 144:218–238.
- Grundy D, Al-Chaer ED, Aziz Q, et al. Fundamentals of neurogastroenterology: basic science. Gastroenterology 2006; 130:1391–1411.
- Gallegos-Orozco JF, Foxx-Orenstein AE, Sterler SM, Stoa JM. Chronic constipation in the elderly. Am J Gastroenterol 2012; 107:18–26.
- Mancini I, Bruera E. Constipation in advanced cancer patients. Support Care Cancer 1998; 6:356–364.
- Bassotti G, Chistolini F, Sietchiping-Nzepa F, de Roberto G, Morelli A, Chiarioni G. Biofeedback for pelvic floor dysfunction in constipation. BMJ 2004; 328:393–396.
- American Gastroenterological Association, Bharucha AE, Dorn SD, Lembo A, Pressman A. American Gastroenterological Association medical position statement on constipation. Gastroenterology 2013; 144:211–217.
- Costilla VC, Foxx-Orenstein AE. Constipation in adults: diagnosis and management. Curr Treat Options Gastroenterol 2014; 12:310–321.
- Rao SS, Singh S. Clinical utility of colonic and anorectal manometry in chronic constipation. J Clin Gastroenterol 2010; 44:597–609.
- Minguez M, Herreros B, Sanchiz V, et al. Predictive value of the balloon expulsion test for excluding the diagnosis of pelvic floor dyssynergia in constipation. Gastroenterology 2004; 126:57–62.
- Diamant NE, Kamm MA, Wald A, Whitehead WE. AGA technical review on anorectal testing techniques. Gastroenterology 1999; 116:735–760.
- Pezim ME, Pemberton JH, Levin KE, Litchy WJ, Phillips SF. Parameters of anorectal and colonic motility in health and in severe constipation. Dis Colon Rectum 1993; 36:484–491.
- Bharucha AE, Fletcher JG, Seide B, Riederer SJ, Zinsmeister AR. Phenotypic variation in functional disorders of defecation. Gastroenterology 2005; 128:1199–1210.
- De Schryver AM, Samsom M, Smout AI. Effects of a meal and bisacodyl on colonic motility in healthy volunteers and patients with slow-transit constipation. Dig Dis Sci 2003; 48:1206–1212.
- Villoria A, Serra J, Azpiroz F, Malagelada JR. Physical activity and intestinal gas clearance in patients with bloating. Am J Gastroenterol 2006; 101:2552–2557.
- Sikirov D. Comparison of straining during defecation in three positions: results and implications for human health. Dig Dis Sci 2003; 48:1201–1205.
- Muller-Lissner SA, Kamm MA, Scarpignato C, Wald A. Myths and misconceptions about chronic constipation. Am J Gastroenterol 2005; 100:232–242.
- Voderholzer WA, Schatke W, Muhldorfer BE, Klauser AG, Birkner B, Muller-Lissner SA. Clinical response to dietary fiber treatment of chronic constipation. Am J Gastroenterol 1997; 92:95–98.
- Bijkerk CJ, de Wit NJ, Muris JW, Whorwell PJ, Knottnerus JA, Hoes AW. Soluble or insoluble fibre in irritable bowel syndrome in primary care? Randomised placebo controlled trial. BMJ 2009; 339:b3154.
- Suares NC, Ford AC. Systematic review: the effects of fibre in the management of chronic idiopathic constipation. Aliment Pharmacol Ther 2011; 33:895–901.
- Dipalma JA, Cleveland MV, McGowan J, Herrera JL. A randomized, multicenter, placebo-controlled trial of polyethylene glycol laxative for chronic treatment of chronic constipation. Am J Gastroenterol 2007; 102:1436–1441.
- Lee-Robichaud H, Thomas K, Morgan J, Nelson RL. Lactulose versus polyethylene glycol for chronic constipation. Cochrane Database Syst Rev 2010; 7:CD007570.
- Lederle FA, Busch DL, Mattox KM, West MJ, Aske DM. Cost-effective treatment of constipation in the elderly: a randomized double-blind comparison of sorbitol and lactulose. Am J Med 1990; 89:597–601.
- Nyberg C, Hendel J, Nielsen OH. The safety of osmotically acting cathartics in colonic cleansing. Nat Rev Gastroenterol Hepatol 2010; 7:557–564.
- Ainley EJ, Winwood PJ, Begley JP. Measurement of serum electrolytes and phosphate after sodium phosphate colonoscopy bowel preparation: an evaluation. Dig Dis Sci 2005; 50:1319–1323.
- Kienzle-Horn S, Vix JM, Schuijt C, Peil H, Jordan CC, Kamm MA. Efficacy and safety of bisacodyl in the acute treatment of constipation: a double-blind, randomized, placebo-controlled study. Aliment Pharmacol Ther 2006; 23:1479–1488.
- Kienzle-Horn S, Vix JM, Schuijt C, Peil H, Jordan CC, Kamm MA. Comparison of bisacodyl and sodium picosulphate in the treatment of chronic constipation. Curr Med Res Opin 2007; 23:691–699.
- Mueller-Lissner S, Kamm MA, Wald A, et al. Multicenter, 4-week, double-blind, randomized, placebo-controlled trial of sodium picosulfate in patients with chronic constipation. Am J Gastroenterol 2010; 105:897–903.
- Smith B. Pathologic changes in the colon produced by anthraquinone purgatives. Dis Colon Rectum 1973; 16:455–458.
- Kiernan JA, Heinicke EA. Sennosides do not kill myenteric neurons in the colon of the rat or mouse. Neuroscience 1989; 30:837–842.
- Ottawa (ON): Canadian Agency for Drugs and Technologies in Health. Dioctyl sulfosuccinate or docusate (calcium or sodium) for the prevention or management of constipation: a review of the clinical effectiveness. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0071207/. Accessed April 6, 2017.
- Saad R, Chey WD. Lubiprostone for chronic idiopathic constipation and irritable bowel syndrome with constipation. Expert Rev Gastroenterol Hepatol 2008; 2:497–508.
- Johanson JF, Morton D, Geenen J, Ueno R. Multicenter, 4-week, double-blind, randomized, placebo-controlled trial of lubiprostone, a locally-acting type-2 chloride channel activator, in patients with chronic constipation. Am J Gastroenterol 2008; 103:170–177.
- Harris LA, Crowell MD. Linaclotide, a new direction in the treatment of irritable bowel syndrome and chronic constipation. Curr Opin Mol Ther 2007; 9:403–410.
- Johnston JM, Kurtz CB, Macdougall JE, et al. Linaclotide improves abdominal pain and bowel habits in a phase IIb study of patients with irritable bowel syndrome with constipation. Gastroenterology 2010; 139:1877–1886.e2.
- Lembo AJ, Schneier HA, Shiff SJ, et al. Two randomized trials of linaclotide for chronic constipation. N Engl J Med 2011; 365:527–536.
- Chey WD, Lembo AJ, Lavins BJ, et al. Linaclotide for irritable bowel syndrome with constipation: a 26-week, randomized, double-blind, placebo-controlled trial to evaluate efficacy and safety. Am J Gastroenterol 2012; 107:1702–1712.
- Rao S, Lembo AJ, Shiff SJ, et al. A 12-week, randomized, controlled trial with a 4-week randomized withdrawal period to evaluate the efficacy and safety of linaclotide in irritable bowel syndrome with constipation. Am J Gastroenterol 2012; 107:1714–1725.
- Chey WD, Kurlander J, Eswaran S. Irritable bowel syndrome: a clinical review. JAMA 2015; 313:949–958.
- Shailubhai K, Talluto C, Comiskey S, Foss JA, Joslyn A, Jacob G. Phase II clinical evaluation of SP-304, a guanylate cyclase-C agonist, for treatment of chronic constipation. Am J Gastroenterol 2010; 105:S487–S488.
- Miner P, Surowitz R, Fogel R, et al. Plecanatide, a novel guanylate cyclase-C (GC-C) receptor agonist, is efficacious and safe in patients with chronic idiopathic constipation (CIC): results from a 951 patient, 12-week, multi-center trial (abstract). Gastroenterology 2013; 144:S163.
- Coss-Adame E, Rao SS. Brain and gut interactions in irritable bowel syndrome: new paradigms and new understandings. Curr Gastroenterol Rep 2014; 16:379.
- Mendzelevski B, Ausma J, Chanter DO, et al. Assessment of the cardiac safety of prucalopride in healthy volunteers: a randomized, double-blind, placebo- and positive-controlled thorough QT study. Br J Clin Pharmacol 2012; 73:203–209.
- Camilleri M, Kerstens R, Rykx A, Vandeplassche L. A placebo-controlled trial of prucalopride for severe chronic constipation. N Engl J Med 2008; 358:2344–2354.
- Tack J, van Outryve M, Beyens G, Kerstens R, Vandeplassche L. Prucalopride (Resolor) in the treatment of severe chronic constipation in patients dissatisfied with laxatives. Gut 2009; 58:357–365.
- Quigley EM, Vandeplassche L, Kerstens R, Ausma J. Clinical trial: the efficacy, impact on quality of life, and safety and tolerability of prucalopride in severe chronic constipation—a 12-week, randomized, double-blind, placebo-controlled study. Aliment Pharmacol Ther 2009; 29:315–328.
- Ford AC, Suares NC. Effect of laxatives and pharmacological therapies in chronic idiopathic constipation: systematic review and meta-analysis. Gut 2011; 60:209–218.
- Goldberg M, Li YP, Johanson JF, et al. Clinical trial: the efficacy and tolerability of velusetrag, a selective 5-HT4 agonist with high intrinsic activity, in chronic idiopathic constipation—a 4-week, randomized, double-blind, placebo-controlled, dose-response study. Aliment Pharmacol Ther 2010; 32:1102–1112.
- Palme M, Milner PG, Ellis DJ, Marmon T, Canafax DM. A novel gastrointestinal prokinetic, ATI-7505, increased spontaneous bowel movements (sbms) in a phase II, randomized, placebo-controlled study of patients with chronic idiopathic constipation (CIC). Gastroenterology 2010; 138:S-128–S-129.
- Chey WD, Camilleri M, Chang L, Rikner L, Graffner H. A randomized placebo-controlled phase IIb trial of a3309, a bile acid transporter inhibitor, for chronic idiopathic constipation. Am J Gastroenterol 2011; 106:1803–1812.
- Wong BS, Camilleri M, McKinzie S, Burton D, Graffner H, Zinsmeister AR. Effects of A3309, an ileal bile acid transporter inhibitor, on colonic transit and symptoms in females with functional constipation. Am J Gastroenterol 2011; 106:2154–2164.
- Pappagallo M. Incidence, prevalence, and management of opioid bowel dysfunction. Am J Surg 2001; 182(suppl):11S–18S.
- Bell T, Annunziata K, Leslie JB. Opioid-induced constipation negatively impacts pain management, productivity, and health-related quality of life: findings from the National Health and Wellness Survey. J Opioid Manag 2009; 5:137–144.
- Sykes NP. A volunteer model for the comparison of laxatives in opioid-related constipation. J Pain Symptom Manage 1996; 11:363–369.
- ClinicalTrials.gov. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study of oral MOA-728 for the treatment of opioid- induced bowel dysfunction in subjects with chronic nonmalignant pain. ClinicalTrials.gov Identifier: NCT00547586. https://clinicaltrials.gov/ct2/show/NCT00547586. Accessed March 22, 2017.
- ClinicalTrials.gov. An open-label study to evaluate the long-term safety of subcutaneous MOA-728 for treatment of opioid-induced constipation in subjects with nonmalignant pain. ClinicalTrials.gov Identifier: NCT00804141. https://clinicaltrials.gov/ct2/show/NCT00804141. Accessed April 6, 2017.
- Wyeth Pharmaceuticals. Relistor package insert. http://labeling.pfizer.com/showlabeling.aspx?id=499. Accessed March 22, 2017.
- Webster L, Dhar S, Eldon M, Masuoka L, Lappalainen J, Sostek M. A phase 2, double-blind, randomized, placebo-controlled, dose-escalation study to evaluate the efficacy, safety, and tolerability of naloxegol in patients with opioid-induced constipation. Pain 2013; 154:1542–1550.
- Chey WD, Webster L, Sostek M, Lappalainen J, Barker PN, Tack J. Naloxegol for opioid-induced constipation in patients with noncancer pain. N Engl J Med 2014; 370:2387–2396.
- Jones R, Prommer E, Backstedt D. Naloxegol: a novel therapy in the management of opioid-induced constipation. Am J Hosp Palliat Care 2016; 33:875–880.
- Guilera M, Balboa A, Mearin F. Bowel habit subtypes and temporal patterns in irritable bowel syndrome: systematic review. Am J Gastroenterol 2005; 100:1174–1184.
- Chapman RW, Stanghellini V, Geraint M, Halphen M. Randomized clinical trial: macrogol/PEG 3350 plus electrolytes for treatment of patients with constipation associated with irritable bowel syndrome. Am J Gastroenterol 2013; 108:1508–1515.
- Ford AC, Quigley EM, Lacy BE, et al. Effect of antidepressants and psychological therapies, including hypnotherapy, in irritable bowel syndrome: systematic review and meta-analysis. Am J Gastroenterol 2014; 109:1350–1366.
- Ballou S, Keefer L. Psychological interventions for irritable bowel syndrome and inflammatory bowel diseases. Clin Transl Gastroenterol 2017; 8:e214.
- Ford AC, Moayyedi P, Lacy BE, et al; Task Force on the Management of Functional Bowel Disorders. American College of Gastroenterology monograph on the management of irritable bowel syndrome and chronic idiopathic constipation. Am J Gastroenterol 2014; 109(suppl 1):S2–S27.
- Ford AC, Talley NJ, Spiegel BM, et al. Effect of fibre, antispasmodics, and peppermint oil in the treatment of irritable bowel syndrome: systematic review and meta-analysis. BMJ 2008; 337:a2313.
- Wall GC, Bryant GA, Bottenberg MM, Maki ED, Miesner AR. Irritable bowel syndrome: a concise review of current treatment concepts. World J Gastroenterol 2014; 20:8796–8806.
- Kligler B, Chaudhary S. Peppermint oil. Am Fam Physician 2007; 75:1027–1030.
- Spencer AG, Labonte ED, Rosenbaum DP, et al. Intestinal inhibition of the Na+/H+ exchanger 3 prevents cardiorenal damage in rats and inhibits Na+ uptake in humans. Sci Transl Med 2014; 6:227ra36.
- Rosenbaum DP. A randomized, double-blind, placebo-controlled study to assess the safety and efficacy of AZD1722 for the treatment of constipation-predominant irritable bowel syndrome (IBS-C). 2014. https://clinicaltrials.gov/ct2/show/NCT01923428. Accessed April 6, 2017.
- Rao SS. Biofeedback therapy for dyssynergic (obstructive) defecation. J Clin Gastroenterol 2000; 30:115–116.
- Cadeddu F, Salis F, De Luca E, Ciangola I, Milito G. Efficacy of biofeedback plus transanal stimulation in the management of pelvic floor dyssynergia: a randomized trial. Tech Coloproctol 2015; 19:333–338.
- Chiarioni G, Whitehead WE, Pezza V, Morelli A, Bassotti G. Biofeedback is superior to laxatives for normal transit constipation due to pelvic floor dyssynergia. Gastroenterology 2006; 130:657–664.
- Chiarioni G, Heymen S, Whitehead WE. Biofeedback therapy for dyssynergic defecation. World J Gastroenterol 2006; 12:7069–7074.
- Rao SS. Biofeedback therapy for constipation in adults. Best Pract Res Clin Gastroenterol 2011; 25:159–166.
- Hassan I, Pemberton JH, Young-Fadok TM, et al. Ileorectal anastomosis for slow transit constipation: long-term functional and quality of life results. J Gastrointest Surg 2006; 10:1330–1337.
- You YT, Wang JY, Changchien CR, et al. Segmental colectomy in the management of colonic inertia. Am Surg 1998; 64:775–777.
- Nyam DC, Pemberton JH, Ilstrup DM, Rath DM. Long-term results of surgery for chronic constipation. Dis Colon Rectum 1997; 40:273–279.
- Pemberton JH, Rath DM, Ilstrup DM. Evaluation and surgical treatment of severe chronic constipation. Ann Surg 1991; 214:403–413.
- Reshef A, Alves-Ferreira P, Zutshi M, Hull T, Gurland B. Colectomy for slow transit constipation: effective for patients with coexistent obstructed defecation. Int J Colorectal Dis 2013; 28:841–847.
- Lundin E, Karlbom U, Pahlman L, Graf W. Outcome of segmental colonic resection for slow-transit constipation. Br J Surg 2002; 89:1270–1274.
- Schwandner O, Stuto A, Jayne D, et al. Decision-making algorithm for the STARR procedure in obstructed defecation syndrome: position statement of the group of STARR pioneers. Surg Innov 2008; 15:105–109.
- Titu LV, Riyad K, Carter H, Dixon AR. Stapled transanal rectal resection for obstructed defecation: a cautionary tale. Dis Colon Rectum 2009; 52:1716–1722.
- Goede AC, Glancy D, Carter H, Mills A, Mabey K, Dixon AR. Medium-term results of stapled transanal rectal resection (STARR) for obstructed defecation and symptomatic rectal-anal intussusception. Colorectal Dis 2011; 13:1052–1057.
- Jayne DG, Schwandner O, Stuto A. Stapled transanal rectal resection for obstructed defecation syndrome: one-year results of the european STARR registry. Dis Colon Rectum 2009; 52:1205–1214.
- Madbouly KM, Abbas KS, Hussein AM. Disappointing long-term outcomes after stapled transanal rectal resection for obstructed defecation. World J Surg 2010; 34:2191–2196.
KEY POINTS
- Although newer drugs are available, lifestyle modifications and laxatives continue to be the treatments of choice for chronic constipation, as they have high response rates and few adverse effects and are relatively affordable.
- Chronic constipation requires different management approaches depending on whether colonic transit time is normal or prolonged and whether outlet function is abnormal.
- Surgical treatments for constipation are reserved for patients whose symptoms persist despite maximal medical therapy.
Managing irritable bowel syndrome: The low-FODMAP diet
The role of diet in controlling symptoms of irritable bowel syndrome (IBS) has gained much traction over the years,1 but until recently, diet therapy for IBS has been hindered by a lack of quality evidence, in part because of the challenges of conducting dietary clinical trials.
Several clinical trials have now been done that support a diet low in fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs) for managing IBS. Although restrictive and difficult to follow, the low-FODMAP diet is gaining popularity.
This article provides an overview of dietary interventions used to manage IBS, focusing on the low-FODMAP diet. We discuss mechanisms of malabsorption of FODMAPs and the role of FODMAPS in symptom induction; highlight clinical trials that provide evidence of benefits of the diet for IBS; and discuss the steps to implement it. We also address the nutritional adequacy of the diet and its potential effects on the gut microbiome.
IBS IS A COMMON FUNCTIONAL DISORDER
IBS is one of the most commonly diagnosed gastrointestinal disorders, and it has a significant impact on quality of life.2 It is a functional disorder characterized by chronic abdominal pain and altered bowel habits in the absence of a structural or organic cause.
The Rome IV diagnostic criteria define IBS by the following:
- Recurrent abdominal pain or discomfort at least 1 day a week in the last 3 months, associated with two or more of the following:
- Symptoms improved by defecation
- Onset associated with a change in frequency of stool
- Onset associated with a change in form or appearance of stool.
IBS mainly arises during young adulthood but can be diagnosed at any age.3
The pathophysiology of IBS involves mechanisms such as bowel distention, altered bowel motility, visceral hypersensitivity, and disruption of mucosal permeability.4 Several therapeutic modalities targeting these mechanisms have been implemented in IBS management, including antispasmodics, laxatives, antidepressants, antibiotics, and behavioral therapy. Diet is only one line of treatment and is most effective when part of a multipronged approach.
TRADITIONAL DIETARY MANAGEMENT
Diet is important in inducing the symptoms of IBS—and in controlling them. Patients identify eating as a common precipitator of symptoms, but the complex diet-symptom interaction is not fully understood and varies widely among patients. Traditional dietary advice for IBS includes adhering to a regular meal pattern, avoiding large meals, and reducing intake of fat, insoluble fibers, caffeine, spicy and gas-producing foods, and carbonated beverages.5,6
Increase soluble fiber
Fiber and fiber supplements, particularly soluble fibers such as psyllium, calcium polycarbophil, and ispaghula husk are often recommended. A meta-analysis7 found that soluble fiber but not insoluble fiber (eg, wheat bran) is associated with an improvement in IBS symptoms (relative risk [RR] 0.84, 95% confidence interval [CI] 0.73–0.94). By improving stool consistency and accelerating transit, soluble fiber is especially useful in constipation-predominant IBS while posing a low risk for adverse outcomes.7 Fiber should be started at a low dose and gradually increased over several weeks to as much as 20 to 30 g/day.
Avoid wheat
Only about 4% of patients with IBS also have celiac disease, but estimating the prevalence of nonceliac gluten sensitivity is confounded by overlapping symptoms. There is some evidence implicating gluten in IBS: celiac disease and IBS overlap in their symptoms, and symptoms are often precipitated by gluten-containing foods in patients with IBS.8 The pathogenesis of gluten-induced (or wheat-induced) symptoms in IBS is unclear, and studies have had conflicting results as to the benefits of gluten restriction in IBS.9
In a study of patients with IBS whose symptoms improved when they started a gluten-free and low-FODMAP diet, symptoms did not return when gluten was reintroduced, suggesting that it is the fructan (a FODMAP) component of wheat rather than gluten that contributes to symptoms in IBS.10
Probiotics
Probiotics are increasingly being recommended as dietary supplements for people with IBS, as awareness increases of the importance of the gut microbiota. In addition to their effects on the gut microbiota, probiotics in IBS have been shown to have anti-inflammatory effects, to alter gut motility, to modulate visceral hypersensitivity, and to restore epithelial integrity.
In a meta-analysis, Ford et al11 found that probiotics improved global IBS symptoms more than placebo (RR 0.79, 95% CI 0.70–0.89) and also reduced abdominal pain, bloating, and flatulence scores.
Which species and strains are most beneficial and the optimal dosing and duration of treatment are still unclear. Data from studies of prebiotics (nutrients that encourage the growth of probiotic bacteria) and synbiotics (combinations of prebiotics and probiotics) are limited and insufficient to draw conclusions.
FODMAPS ARE SHORT-CHAIN CARBOHYDRATES
The term FODMAPs was initially coined by researchers at Monash University in Australia to describe a collection of poorly absorbed short-chain fermentable carbohydrates that are natural components of many foods:
- Oligosaccharides, including fructans (which include inulins) and galacto-oligosaccharides
- Disaccharides, including lactose and sucrose
- Monosaccharides, including fructose
- Polyols, including sorbitol and mannitol.12
Intake of FODMAPs, especially fructose, has increased in Western diets over the past several decades from increased consumption of fruits and concentrated fruit juices, as well as from the widespread use of high-fructose corn syrup in processed foods and beverages.13
FODMAPs ARE POORLY ABSORBED
Different FODMAPs can be poorly absorbed for different reasons (Table 1). The poor absorption is related either to reduced or absent digestive enzymes (ie, hydrolases) or to slow transport across the intestinal mucosa. Excess FODMAPs in the distal small intestine and proximal colon exert osmotic pressure, drawing more water into the lumen. FODMAPs are also rapidly fermented by colonic bacteria, producing gas, bowel distention, and altered motility, all of which induce IBS symptoms.14
Fructans are fructose polymers that are not absorbed in human intestines. They have no intestinal hydrolases and no mechanisms for direct transport across the epithelium. However, a negligible amount may be absorbed after being degraded by microbes in the gut.15 Most dietary fructans are obtained from wheat and onion, which are actually low in fructans but tend to be consumed in large quantities.16
Galacto-oligosaccharides are available for colonic fermentation after ingestion due to lack of a human alpha-galactosidase. Common sources of galacto-oligosaccharides include legumes, nuts, seeds, some grains, dairy products, human milk, and some commercially produced forms added to infant formula.17,18
Lactose is poorly absorbed in people with lactase deficiency. It is mainly present in dairy products but is also added to commercial foods, including breads, cakes, and some diet products.19
Fructose is the most abundant FODMAP in the Western diet. It is either present as a free sugar or generated from the digestive breakdown of sucrose. In the intestine, it is absorbed via a direct low-capacity glucose transporter (GLUT)-5 and through GLUT-2, which is more efficient but requires the coexistence of glucose. Because of this requirement, fructose is more likely to be malabsorbed when present in excess of glucose, as in people with diminished sucrase activity. The main sources of fructose in the Western diet are fruits and fruit products, honey, and foods with added high-fructose sweeteners.13
Polyols such as sorbitol and mannitol are absorbed by slow passive diffusion because they have no active intestinal transport system. They are found in fruits and vegetables. Sugar-free chewing gum is a particularly rich source of sorbitol.20
QUANTIFYING FODMAP CONTENT
As interest in the low-FODMAP diet grew, studies were conducted to quantify FODMAPs in foods. One study used high-performance liquid chromatography to analyze FODMAP content in foods,21 and another evaluated fructan levels in a variety of fruits and vegetables using enzymatic hydrolysis.22 The Monash University low-FODMAP diet smartphone application provides patients and healthcare providers easy access to updated and detailed food analyses.23
Table 2 lists foods high in FODMAPs along with low-FODMAP alternatives. Total FODMAP intake is important, as the effects are additive.24 Readers and patients can be directed to the following websites for more information on the low-FODMAP diet: www.med.monash.edu/cecs/gastro/fodmap or www.ibsfree.net/what-is-fodmap-diet.
LOW-FODMAP DIET REDUCES SYMPTOMS
The low-FODMAP diet was inspired by the results of several studies that evaluated the role of dietary carbohydrates in inducing IBS symptoms and found improvement with their restriction.25,26
One study found that 74% of patients with IBS had less bloating, nausea, abdominal pain, and diarrhea when they restricted their intake of fructose and fructans.27
A prospective trial randomized 41 patients with IBS to 4 weeks of either a low-FODMAP diet or their habitual diet.28 The low-FODMAP diet resulted in greater improvement in overall IBS symptoms (P < .05) and stool frequency (P = .008). This study was limited by different habitual diets between patients and by lack of standardization of the low-FODMAP diet.
Halmos et al,29 in a randomized crossover trial, compared gastrointestinal symptoms in IBS patients over 3 weeks on a low-FODMAP diet vs a moderate-FODMAP (ie, regular) diet, as well as in healthy controls. Food was provided by the study and was matched for all nutrients. Up to 70% of the IBS patients had significantly lower overall symptom scores while on a low-FODMAP diet vs IBS patients on a regular diet (P < .001); bloating, abdominal pain, and flatulence were reduced. Symptoms were minimal and unaffected by either diet in the healthy controls.
A double-blind trial30 randomly assigned 25 patients with IBS who initially responded to a low-FODMAP diet to be challenged by a graduated dose of fructose alone, fructans alone, a combination of both, or glucose. The severity of overall and individual symptoms was markedly more reduced with glucose consumption than with the other carbohydrates: 70% of patients receiving fructose, 77% of those receiving fructans, and 79% of those receiving a mixture of both reported that their symptoms were not adequately controlled, compared with 14% of patients receiving glucose (P ≤ .002).30
Murray et al31 evaluated the gastrointestinal tract after a carbohydrate challenge consisting of 0.5 L of water containing 40 g of glucose, fructose, or inulin or a combination of 40 g of glucose and 40 g of fructose in 16 healthy volunteers. Magnetic resonance imaging was performed hourly for 5 hours to assess the volume of gastric contents, small-bowel water content, and colonic gas. Breath hydrogen was also measured, and symptoms were recorded after each imaging session.
Fructose significantly increased small-bowel water content compared with glucose (mean difference 28 L/min, P < .001), but combined glucose and fructose lessened the effect. Inulin had no significant effect on small-bowel water content (mean difference with glucose 2 L/min, P > .7) but led to the greatest production of colonic gas compared with glucose alone (mean difference 15 L/min, P < .05) and combined glucose and fructose (mean difference 12 L/min, P < .05). Inulin also produced the most breath hydrogen: 81% of participants had a rise after drinking inulin compared with 50% after drinking fructose. Glucose did not affect breath hydrogen concentrations, and combined glucose and fructose significantly reduced the concentration measured vs fructose alone. In patients who reported “gas” symptoms, a correlation was observed between the volume of gas in the colon and gas symptoms (r = 0.59, P < .0001).31
The authors concluded31 that long-chain carbohydrates such as inulin have a greater effect on colonic gas production and little effect on small-bowel water content, whereas small-chain FODMAPs such as fructose are likely to cause luminal distention in both the small and large intestines. The study also showed that combining equal amounts of glucose and fructose reduces malabsorption of fructose in the small bowel and reduces the effect of fructose on small-bowel water content and breath hydrogen concentration.31
PROBIOTICS HELP
A Danish study32 randomized 123 patients with IBS to one of three treatments: a low-FODMAP diet, a normal diet with probiotics containing the strain Lactobacillus rhamnosus GG (two capsules daily), or no special intervention. Symptoms were recorded weekly. IBS severity scores at week 6 were lower in patients on either the low-FODMAP diet or probiotics compared with the control group (P < .01). Subgroup analysis determined that patients with primarily diarrheal symptoms were more likely to have improved quality of life with the low-FODMAP diet.
A LOW-FODMAP DIET MAY ALSO HELP IN INFLAMMATORY BOWEL DISEASE
The low-FODMAP diet has also been studied in patients with inflammatory bowel disease with functional gut symptoms. In a retrospective pilot study,33 overall symptoms improved in about half of such patients on a low-FODMAP diet. A controlled dietary intervention trial is needed to confirm these findings and define the role of the low-FODMAP approach for patients with inflammatory bowel disease.
Marsh et al34 performed a meta-analysis of six randomized clinical trials and 16 nonrandomized interventions of a low-FODMAP diet on improving functional gastrointestinal symptoms in patients with either IBS or inflammatory bowel disease. They found significant improvements in:
- IBS Symptoms Severity Scores in the randomized trials (odds ratio [OR] 0.44, 95% CI 0.25–0.76)
- IBS Symptoms Severity Scores in the nonrandomized interventions (OR 0.03, 95% CI 0.01–0.2)
- IBS Quality of Life scores in the randomized trials (OR 1.84, 95% CI 1.12–3.03)
- IBS Quality of Life scores in the nonrandomized interventions (OR 3.18, 95% CI 1.6–6.31)
- Overall symptom severity in the randomized trials (OR 1.81, 95% CI 1.11–2.95).
DIETARY COUNSELING IS RECOMMENDED
Adherence is a major factor in the success of the low-FODMAP diet in IBS management and is strongly correlated with improved symptoms.35 Patients should be counseled on the role of food in inducing their symptoms. Haphazard dietary advice can be detrimental to outcomes, as many diets restrict food groups, impairing the consumption of essential nutrients.36 The involvement of a knowledgeable dietitian is helpful, as physicians may lack sufficient training in dietary skills and knowledge of food composition.
Access to and cost of dietary counseling can be prohibitive for some patients. Group consultation, which can decrease costs to each patient, has been found to be as effective as one-on-one sessions when administering the low-FODMAP diet in functional bowel disorders.37
ELIMINATION, THEN REINTRODUCTION
Before embarking on the low-FODMAP diet, the patient’s interest in making dietary changes should be explored, a dietary history taken, and unusual food choices or dietary behaviors assessed. The patient’s ability to adopt a restricted diet should also be gauged.
The diet should be implemented in two phases. The initial phase involves strict elimination of foods high in FODMAPs, usually over 6 to 8 weeks.38 Symptom control should be assessed: failure to control symptoms requires assessment of adherence.
If symptoms are successfully controlled, then the second phase should begin with the aim of following a less-restricted version of the diet as tolerated. Foods should gradually be phased back in and symptoms monitored. This approach minimizes unnecessary dietary restriction and ensures that a maximum variety in the diet is achieved while maintaining adequate symptom control.39
LOW-FODMAP DIET ALTERS THE GUT MICROBIOTA
Multiple putative benefits of certain bacterial species for colonic health have been reported, including the production of short-chain fatty acids. Colonic luminal concentrations of short-chain fatty acids may be important to gut health, given their role in intestinal secretion, absorption, motility, and epithelial cell structure. Because short-chain fatty acids are products of bacterial fermentation, a change in the delivery of fermentable substrates to the colon would be expected to alter the concentrations and output of fecal short-chain fatty acids.18
Several studies evaluated the effect of the low-FODMAP diet on intestinal microbiota, finding a change in the bacterial profile in the stool of patients who adopt this diet. Staudacher et al28 found a marked reduction in luminal bifidobacteria concentration after 4 weeks of a low-FODMAP diet in patients with IBS.
A single-blind randomized crossover trial40 investigated the effects of a low-FODMAP diet vs a carefully matched typical Australian diet in 27 patients with IBS and 6 healthy controls. Marked differences in absolute and relative bacterial abundance and diversity were found between the diets, but not in short-chain fatty acids or gut transit time. Compared with fecal microbiota on the typical diet, low FODMAP intake was associated with reduced absolute abundance of bacteria, and the typical FODMAP diet had evidence of stimulation of the growth of bacterial groups with putative health benefits.
The authors concluded40 that the functional significance and health implications of such changes are reasons for caution when reducing FODMAP intake in the long term and recommended liberalizing FODMAP restriction to the level of adequate symptom control in IBS patients. The study also recommended that people without symptoms not go on the low-FODMAP diet.40
Molecular approaches to characterize the gut microbiota are also being explored in an effort to identify its association with diet.
The sustainability of changes in gut microbiota and the potential long-term impact on health of following a low-FODMAP diet require further evaluation. In the meantime, patients following this diet should have FODMAP foods reintroduced based on tolerance and should consider taking probiotic supplements.41
DIETARY ADEQUACY OF THE LOW-FODMAP DIET
Continual dietary counseling should minimize nutritional inadequacies and ensure that FODMAPS are restricted only enough to control symptoms. Because no single food group is completely eliminated in this diet, patients are unlikely to experience inadequate nutrition.
Ledochowski et al26 found that in the initial, strict phase of the diet, total intake of carbohydrates (eg, starches, sugars) was reduced but intake of total energy, protein, fat, and nonstarch polysaccharides was not affected. Calcium intake was reduced in those following a low-FODMAP diet for 4 weeks.
The diet can also reduce total fiber intake and subsequently worsen constipation-predominant IBS. For those patients, lightly fermented high-fiber alternatives like oat and rice bran can be used.
ACCUMULATING EVIDENCE
The low-FODMAP diet is accumulating quality evidence for its effectiveness in controlling the functional gastrointestinal symptoms in patients with IBS. It can be difficult to adhere to over the long term due to its restrictiveness, and it is important to gradually liberalize the diet while tailoring it to the individual patient and monitoring symptoms. Further clinical trials are needed to evaluate this diet in different IBS subtypes and other gastrointestinal disorders, while defining its nutritional adequacy and effects on the intestinal microbiota profile.
- Hayes P, Corish C, O’Mahony E, Quigley EM. A dietary survey of patients with irritable bowel syndrome. J Hum Nutr Diet 2014; 27(suppl 2):36–47.
- Pare P, Gray J, Lam S, et al. Health-related quality of life, work productivity, and health care resource utilization of subjects with irritable bowel syndrome: baseline results from LOGIC (Longitudinal Outcomes Study of Gastrointestinal Symptoms in Canada), a naturalistic study. Clin Ther 2006; 28:1726–1735; discussion 1710–1711.
- Lacey BE, Mearin F, Chang L, et al. Bowel disorders. Gastroenterology 2016; 150:1393–1407.
- Drossman DA, Camilleri M, Mayer EA, Whitehead WE. AGA technical review on irritable bowel syndrome. Gastroenterology 2002; 123:2108–2131.
- Floch MH, Narayan R. Diet in the irritable bowel syndrome. J Clin Gastroenterol 2002; 35(suppl 1):S45–S52.
- Reding KW, Cain KC, Jarrett ME, Eugenio MD, Heitkemper MM. Relationship between patterns of alcohol consumption and gastrointestinal symptoms among patients with irritable bowel syndrome. Am J Gastroenterol 2013; 108:270–276.
- Moayyedi P, Quigley EM, Lacy BE, et al. The effect of fiber supplementation on irritable bowel syndrome: a systematic review and meta-analysis. Am J Gastroenterol 2014; 109:1367–1374.
- Vazquez Roque MI, Camilleri M, Smyrk T, et al. A controlled trial of gluten-free diet in patients with irritable bowel syndrome-diarrhea: effects on bowel frequency and intestinal function. Gastroenterology 2013; 144:903–911.
- Biesiekierski JR, Newnham ED, Irving PM, et al. Gluten causes gastrointestinal symptoms in subjects without celiac disease: a double-blind randomized placebo-controlled trial. Am J Gastroenterol 2011; 106:508–515.
- Biesiekierski JR, Peters SL, Newnham ED, Rosella O, Muir JG, Gibson PR. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 2013; 145:320–328.
- Ford AC, Quigley EM, Lacy BE, et al. Efficacy of prebiotics, probiotics, and synbiotics in irritable bowel syndrome and chronic idiopathic constipation: systematic review and meta-analysis. Gastroenterology 2013; 145:320–328.e1–e3.
- Central Clinical School, Monash University and The Alfred Hospital. The Monash University Low FODMAP Diet. 4th ed. Melbourne, Australia: Monash University; 2012.
- Parker K, Salas M, Nwosu VC. High fructose corn syrup: production, uses and public health concerns. Biotechnol Mol Biol Rev 2010; 5:71–78.
- Clausen MR, Jorgensen J, Mortensen PB. Comparison of diarrhea induced by ingestion of fructooligosaccharide idolax and disaccharide lactulose: role of osmolarity versus fermentation of malabsorbed carbohydrate. Dig Dis Sci 1998; 43:2696–2707.
- Barrett JS, Gearry RB, Muir JG, et al. Dietary poorly absorbed, short-chain carbohydrates increase delivery of water and fermentable substrates to the proximal colon. Aliment Pharmacol Ther 2010; 31:874–882.
- Whelan K, Abrahmsohn O, David GJ, et al. Fructan content of commonly consumed wheat, rye and gluten-free breads. Int J Food Sci Nutr 2011; 62:498–503.
- Sangwan V, Tomar SK, Singh RR, Singh AK, Ali B. Galactooligosaccharides: novel components of designer foods. J Food Sci 2011; 76:R103–R111.
- Russell DA, Ross RP, Fitzgerald GF, Stanton C. Metabolic activities and probiotic potential of bifidobacteria. Int J Food Microbiol 2011; 149:88–105.
- Lomer MC, Parkes GC, Sanderson JD. Review article: lactose intolerance in clinical practice—myths and realities. Aliment Pharmacol Ther 2008; 27:93–103.
- Langkilde AM, Andersson H, Schweizer TF, Würsch P. Digestion and absorption of sorbitol, maltitol and isomalt from the small bowel. A study in ileostomy subjects. Eur J Clin Nutr 1994; 48:768–775.
- Muir JG, Rose R, Rosella O, et al. Measurement of short-chain carbohydrates in common Australian vegetables and fruits by high-performance liquid chromatography (HPLC). J Agric Food Chem 2009; 57:554–565.
- Muir JG, Shepherd SJ, Rosella O, Rose R, Barrett JS, Gibson PR. Fructan and free fructose content of common Australian vegetables and fruit. J Agric Food Chem 2007; 55:6619–6627.
- Monash University. Monash launches Low FODMAP Diet smartphone app. http://med.monash.edu.au/news/2012/fodmap-app.html. Accessed July 13, 2016.
- Fedewa A, Rao SS. Dietary fructose intolerance, fructan intolerance and FODMAPs. Curr Gastroenterol Rep 2014; 16:370.
- Born P, Vierling T, Barina W. Fructose malabsorption and the irritable bowel syndrome. Gastroenterology 1991; 101:1454.
- Ledochowski M, Widner B, Bair H, Probst T, Fuchs D. Fructose- and sorbitol-reduced diet improves mood and gastrointestinal disturbances in fructose malabsorbers. Scand J Gastroenterol 2000; 35:1048–1052.
- Shepherd SJ, Gibson PR. Fructose malabsorption and symptoms of irritable bowel syndrome: guidelines for effective dietary management. J Am Diet Assoc 2006; 106:1631–1639.
- Staudacher HM, Lomer MC, Anderson JL, et al. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J Nutr 2012; 142:1510–1518.
- Halmos EP, Power VA, Shepherd SJ, Gibson PR, Muir JG. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 2014; 146:67–75.e5.
- Shepherd SJ, Parker FC, Muir JG, Gibson PR. Dietary triggers of abdominal symptoms in patients with irritable bowel syndrome: randomized placebo-controlled evidence. Clin Gastroenterol Hepatol 2008; 6:765–771.
- Murray K, Wilkinson-Smith V, Hoad C, et al. Differential effects of FODMAPs (fermentable oligo-, di-, mono-saccharides and polyols) on small and large intestinal contents in healthy subjects shown by MRI. Am J Gastroenterol 2014; 109:110–119.
- Pedersen N, Andersen NN, Vegh Z, et al. Ehealth: low FODMAP diet vs Lactobacillus rhamnosus GG in irritable bowel syndrome. World J Gastroenterol 2014; 20:16215–16226.
- Gearry RB, Irving PM, Barrett JS, Nathan DM, Shepherd SJ, Gibson PR. Reduction of dietary poorly absorbed short-chain carbohydrates (FODMAPs) improves abdominal symptoms in patients with inflammatory bowel disease-a pilot study. J Crohns Colitis 2009; 3:8–14.
- Marsh A, Eslick EM, Eslick GD. Does a diet low in FODMAPs reduce symptoms associated with functional gastrointestinal disorders? A comprehensive systematic review and meta-analysis. Eur J Nutr 2015 May 17. Epub ahead of print.
- de Roest RH, Dobbs BR, Chapman BA, et al. The low FODMAP diet improves gastrointestinal symptoms in patients with irritable bowel syndrome: a prospective study. Int J Clin Pract 2013; 67:895–903.
- Gibson PR, Barrett JS, Muir JG. Functional bowel symptoms and diet. Intern Med J 2013; 43:1067–1074.
- Whigham L, Joyce T, Harper G, et al. Clinical effectiveness and economic costs of group versus one-to-one education for short-chain fermentable carbohydrate restriction (low FODMAP diet) in the management of irritable bowel syndrome. J Hum Nutr Diet 2015; 28:687–696.
- Shepherd SJ, Lomer MC, Gibson PR. Short-chain carbohydrates and functional gastrointestinal disorders. Am J Gastroenterol 2013; 108:707–717.
- Shepherd SJ, Halmos E, Glance S. The role of FODMAPs in irritable bowel syndrome. Curr Opin Clin Nutr Metab Care 2014; 17:605–609.
- Halmos EP, Christophersen CT, Bird AR, Shepherd SJ, Gibson PR, Muir JG. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut 2015; 64:93–100.
- Staudacher HM, Irving PM, Lomer MC, Whelan K. Mechanisms and efficacy of dietary FODMAP restriction in IBS. Nat Rev Gastroenterol Hepatol 2014; 11:256–266.
The role of diet in controlling symptoms of irritable bowel syndrome (IBS) has gained much traction over the years,1 but until recently, diet therapy for IBS has been hindered by a lack of quality evidence, in part because of the challenges of conducting dietary clinical trials.
Several clinical trials have now been done that support a diet low in fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs) for managing IBS. Although restrictive and difficult to follow, the low-FODMAP diet is gaining popularity.
This article provides an overview of dietary interventions used to manage IBS, focusing on the low-FODMAP diet. We discuss mechanisms of malabsorption of FODMAPs and the role of FODMAPS in symptom induction; highlight clinical trials that provide evidence of benefits of the diet for IBS; and discuss the steps to implement it. We also address the nutritional adequacy of the diet and its potential effects on the gut microbiome.
IBS IS A COMMON FUNCTIONAL DISORDER
IBS is one of the most commonly diagnosed gastrointestinal disorders, and it has a significant impact on quality of life.2 It is a functional disorder characterized by chronic abdominal pain and altered bowel habits in the absence of a structural or organic cause.
The Rome IV diagnostic criteria define IBS by the following:
- Recurrent abdominal pain or discomfort at least 1 day a week in the last 3 months, associated with two or more of the following:
- Symptoms improved by defecation
- Onset associated with a change in frequency of stool
- Onset associated with a change in form or appearance of stool.
IBS mainly arises during young adulthood but can be diagnosed at any age.3
The pathophysiology of IBS involves mechanisms such as bowel distention, altered bowel motility, visceral hypersensitivity, and disruption of mucosal permeability.4 Several therapeutic modalities targeting these mechanisms have been implemented in IBS management, including antispasmodics, laxatives, antidepressants, antibiotics, and behavioral therapy. Diet is only one line of treatment and is most effective when part of a multipronged approach.
TRADITIONAL DIETARY MANAGEMENT
Diet is important in inducing the symptoms of IBS—and in controlling them. Patients identify eating as a common precipitator of symptoms, but the complex diet-symptom interaction is not fully understood and varies widely among patients. Traditional dietary advice for IBS includes adhering to a regular meal pattern, avoiding large meals, and reducing intake of fat, insoluble fibers, caffeine, spicy and gas-producing foods, and carbonated beverages.5,6
Increase soluble fiber
Fiber and fiber supplements, particularly soluble fibers such as psyllium, calcium polycarbophil, and ispaghula husk are often recommended. A meta-analysis7 found that soluble fiber but not insoluble fiber (eg, wheat bran) is associated with an improvement in IBS symptoms (relative risk [RR] 0.84, 95% confidence interval [CI] 0.73–0.94). By improving stool consistency and accelerating transit, soluble fiber is especially useful in constipation-predominant IBS while posing a low risk for adverse outcomes.7 Fiber should be started at a low dose and gradually increased over several weeks to as much as 20 to 30 g/day.
Avoid wheat
Only about 4% of patients with IBS also have celiac disease, but estimating the prevalence of nonceliac gluten sensitivity is confounded by overlapping symptoms. There is some evidence implicating gluten in IBS: celiac disease and IBS overlap in their symptoms, and symptoms are often precipitated by gluten-containing foods in patients with IBS.8 The pathogenesis of gluten-induced (or wheat-induced) symptoms in IBS is unclear, and studies have had conflicting results as to the benefits of gluten restriction in IBS.9
In a study of patients with IBS whose symptoms improved when they started a gluten-free and low-FODMAP diet, symptoms did not return when gluten was reintroduced, suggesting that it is the fructan (a FODMAP) component of wheat rather than gluten that contributes to symptoms in IBS.10
Probiotics
Probiotics are increasingly being recommended as dietary supplements for people with IBS, as awareness increases of the importance of the gut microbiota. In addition to their effects on the gut microbiota, probiotics in IBS have been shown to have anti-inflammatory effects, to alter gut motility, to modulate visceral hypersensitivity, and to restore epithelial integrity.
In a meta-analysis, Ford et al11 found that probiotics improved global IBS symptoms more than placebo (RR 0.79, 95% CI 0.70–0.89) and also reduced abdominal pain, bloating, and flatulence scores.
Which species and strains are most beneficial and the optimal dosing and duration of treatment are still unclear. Data from studies of prebiotics (nutrients that encourage the growth of probiotic bacteria) and synbiotics (combinations of prebiotics and probiotics) are limited and insufficient to draw conclusions.
FODMAPS ARE SHORT-CHAIN CARBOHYDRATES
The term FODMAPs was initially coined by researchers at Monash University in Australia to describe a collection of poorly absorbed short-chain fermentable carbohydrates that are natural components of many foods:
- Oligosaccharides, including fructans (which include inulins) and galacto-oligosaccharides
- Disaccharides, including lactose and sucrose
- Monosaccharides, including fructose
- Polyols, including sorbitol and mannitol.12
Intake of FODMAPs, especially fructose, has increased in Western diets over the past several decades from increased consumption of fruits and concentrated fruit juices, as well as from the widespread use of high-fructose corn syrup in processed foods and beverages.13
FODMAPs ARE POORLY ABSORBED
Different FODMAPs can be poorly absorbed for different reasons (Table 1). The poor absorption is related either to reduced or absent digestive enzymes (ie, hydrolases) or to slow transport across the intestinal mucosa. Excess FODMAPs in the distal small intestine and proximal colon exert osmotic pressure, drawing more water into the lumen. FODMAPs are also rapidly fermented by colonic bacteria, producing gas, bowel distention, and altered motility, all of which induce IBS symptoms.14
Fructans are fructose polymers that are not absorbed in human intestines. They have no intestinal hydrolases and no mechanisms for direct transport across the epithelium. However, a negligible amount may be absorbed after being degraded by microbes in the gut.15 Most dietary fructans are obtained from wheat and onion, which are actually low in fructans but tend to be consumed in large quantities.16
Galacto-oligosaccharides are available for colonic fermentation after ingestion due to lack of a human alpha-galactosidase. Common sources of galacto-oligosaccharides include legumes, nuts, seeds, some grains, dairy products, human milk, and some commercially produced forms added to infant formula.17,18
Lactose is poorly absorbed in people with lactase deficiency. It is mainly present in dairy products but is also added to commercial foods, including breads, cakes, and some diet products.19
Fructose is the most abundant FODMAP in the Western diet. It is either present as a free sugar or generated from the digestive breakdown of sucrose. In the intestine, it is absorbed via a direct low-capacity glucose transporter (GLUT)-5 and through GLUT-2, which is more efficient but requires the coexistence of glucose. Because of this requirement, fructose is more likely to be malabsorbed when present in excess of glucose, as in people with diminished sucrase activity. The main sources of fructose in the Western diet are fruits and fruit products, honey, and foods with added high-fructose sweeteners.13
Polyols such as sorbitol and mannitol are absorbed by slow passive diffusion because they have no active intestinal transport system. They are found in fruits and vegetables. Sugar-free chewing gum is a particularly rich source of sorbitol.20
QUANTIFYING FODMAP CONTENT
As interest in the low-FODMAP diet grew, studies were conducted to quantify FODMAPs in foods. One study used high-performance liquid chromatography to analyze FODMAP content in foods,21 and another evaluated fructan levels in a variety of fruits and vegetables using enzymatic hydrolysis.22 The Monash University low-FODMAP diet smartphone application provides patients and healthcare providers easy access to updated and detailed food analyses.23
Table 2 lists foods high in FODMAPs along with low-FODMAP alternatives. Total FODMAP intake is important, as the effects are additive.24 Readers and patients can be directed to the following websites for more information on the low-FODMAP diet: www.med.monash.edu/cecs/gastro/fodmap or www.ibsfree.net/what-is-fodmap-diet.
LOW-FODMAP DIET REDUCES SYMPTOMS
The low-FODMAP diet was inspired by the results of several studies that evaluated the role of dietary carbohydrates in inducing IBS symptoms and found improvement with their restriction.25,26
One study found that 74% of patients with IBS had less bloating, nausea, abdominal pain, and diarrhea when they restricted their intake of fructose and fructans.27
A prospective trial randomized 41 patients with IBS to 4 weeks of either a low-FODMAP diet or their habitual diet.28 The low-FODMAP diet resulted in greater improvement in overall IBS symptoms (P < .05) and stool frequency (P = .008). This study was limited by different habitual diets between patients and by lack of standardization of the low-FODMAP diet.
Halmos et al,29 in a randomized crossover trial, compared gastrointestinal symptoms in IBS patients over 3 weeks on a low-FODMAP diet vs a moderate-FODMAP (ie, regular) diet, as well as in healthy controls. Food was provided by the study and was matched for all nutrients. Up to 70% of the IBS patients had significantly lower overall symptom scores while on a low-FODMAP diet vs IBS patients on a regular diet (P < .001); bloating, abdominal pain, and flatulence were reduced. Symptoms were minimal and unaffected by either diet in the healthy controls.
A double-blind trial30 randomly assigned 25 patients with IBS who initially responded to a low-FODMAP diet to be challenged by a graduated dose of fructose alone, fructans alone, a combination of both, or glucose. The severity of overall and individual symptoms was markedly more reduced with glucose consumption than with the other carbohydrates: 70% of patients receiving fructose, 77% of those receiving fructans, and 79% of those receiving a mixture of both reported that their symptoms were not adequately controlled, compared with 14% of patients receiving glucose (P ≤ .002).30
Murray et al31 evaluated the gastrointestinal tract after a carbohydrate challenge consisting of 0.5 L of water containing 40 g of glucose, fructose, or inulin or a combination of 40 g of glucose and 40 g of fructose in 16 healthy volunteers. Magnetic resonance imaging was performed hourly for 5 hours to assess the volume of gastric contents, small-bowel water content, and colonic gas. Breath hydrogen was also measured, and symptoms were recorded after each imaging session.
Fructose significantly increased small-bowel water content compared with glucose (mean difference 28 L/min, P < .001), but combined glucose and fructose lessened the effect. Inulin had no significant effect on small-bowel water content (mean difference with glucose 2 L/min, P > .7) but led to the greatest production of colonic gas compared with glucose alone (mean difference 15 L/min, P < .05) and combined glucose and fructose (mean difference 12 L/min, P < .05). Inulin also produced the most breath hydrogen: 81% of participants had a rise after drinking inulin compared with 50% after drinking fructose. Glucose did not affect breath hydrogen concentrations, and combined glucose and fructose significantly reduced the concentration measured vs fructose alone. In patients who reported “gas” symptoms, a correlation was observed between the volume of gas in the colon and gas symptoms (r = 0.59, P < .0001).31
The authors concluded31 that long-chain carbohydrates such as inulin have a greater effect on colonic gas production and little effect on small-bowel water content, whereas small-chain FODMAPs such as fructose are likely to cause luminal distention in both the small and large intestines. The study also showed that combining equal amounts of glucose and fructose reduces malabsorption of fructose in the small bowel and reduces the effect of fructose on small-bowel water content and breath hydrogen concentration.31
PROBIOTICS HELP
A Danish study32 randomized 123 patients with IBS to one of three treatments: a low-FODMAP diet, a normal diet with probiotics containing the strain Lactobacillus rhamnosus GG (two capsules daily), or no special intervention. Symptoms were recorded weekly. IBS severity scores at week 6 were lower in patients on either the low-FODMAP diet or probiotics compared with the control group (P < .01). Subgroup analysis determined that patients with primarily diarrheal symptoms were more likely to have improved quality of life with the low-FODMAP diet.
A LOW-FODMAP DIET MAY ALSO HELP IN INFLAMMATORY BOWEL DISEASE
The low-FODMAP diet has also been studied in patients with inflammatory bowel disease with functional gut symptoms. In a retrospective pilot study,33 overall symptoms improved in about half of such patients on a low-FODMAP diet. A controlled dietary intervention trial is needed to confirm these findings and define the role of the low-FODMAP approach for patients with inflammatory bowel disease.
Marsh et al34 performed a meta-analysis of six randomized clinical trials and 16 nonrandomized interventions of a low-FODMAP diet on improving functional gastrointestinal symptoms in patients with either IBS or inflammatory bowel disease. They found significant improvements in:
- IBS Symptoms Severity Scores in the randomized trials (odds ratio [OR] 0.44, 95% CI 0.25–0.76)
- IBS Symptoms Severity Scores in the nonrandomized interventions (OR 0.03, 95% CI 0.01–0.2)
- IBS Quality of Life scores in the randomized trials (OR 1.84, 95% CI 1.12–3.03)
- IBS Quality of Life scores in the nonrandomized interventions (OR 3.18, 95% CI 1.6–6.31)
- Overall symptom severity in the randomized trials (OR 1.81, 95% CI 1.11–2.95).
DIETARY COUNSELING IS RECOMMENDED
Adherence is a major factor in the success of the low-FODMAP diet in IBS management and is strongly correlated with improved symptoms.35 Patients should be counseled on the role of food in inducing their symptoms. Haphazard dietary advice can be detrimental to outcomes, as many diets restrict food groups, impairing the consumption of essential nutrients.36 The involvement of a knowledgeable dietitian is helpful, as physicians may lack sufficient training in dietary skills and knowledge of food composition.
Access to and cost of dietary counseling can be prohibitive for some patients. Group consultation, which can decrease costs to each patient, has been found to be as effective as one-on-one sessions when administering the low-FODMAP diet in functional bowel disorders.37
ELIMINATION, THEN REINTRODUCTION
Before embarking on the low-FODMAP diet, the patient’s interest in making dietary changes should be explored, a dietary history taken, and unusual food choices or dietary behaviors assessed. The patient’s ability to adopt a restricted diet should also be gauged.
The diet should be implemented in two phases. The initial phase involves strict elimination of foods high in FODMAPs, usually over 6 to 8 weeks.38 Symptom control should be assessed: failure to control symptoms requires assessment of adherence.
If symptoms are successfully controlled, then the second phase should begin with the aim of following a less-restricted version of the diet as tolerated. Foods should gradually be phased back in and symptoms monitored. This approach minimizes unnecessary dietary restriction and ensures that a maximum variety in the diet is achieved while maintaining adequate symptom control.39
LOW-FODMAP DIET ALTERS THE GUT MICROBIOTA
Multiple putative benefits of certain bacterial species for colonic health have been reported, including the production of short-chain fatty acids. Colonic luminal concentrations of short-chain fatty acids may be important to gut health, given their role in intestinal secretion, absorption, motility, and epithelial cell structure. Because short-chain fatty acids are products of bacterial fermentation, a change in the delivery of fermentable substrates to the colon would be expected to alter the concentrations and output of fecal short-chain fatty acids.18
Several studies evaluated the effect of the low-FODMAP diet on intestinal microbiota, finding a change in the bacterial profile in the stool of patients who adopt this diet. Staudacher et al28 found a marked reduction in luminal bifidobacteria concentration after 4 weeks of a low-FODMAP diet in patients with IBS.
A single-blind randomized crossover trial40 investigated the effects of a low-FODMAP diet vs a carefully matched typical Australian diet in 27 patients with IBS and 6 healthy controls. Marked differences in absolute and relative bacterial abundance and diversity were found between the diets, but not in short-chain fatty acids or gut transit time. Compared with fecal microbiota on the typical diet, low FODMAP intake was associated with reduced absolute abundance of bacteria, and the typical FODMAP diet had evidence of stimulation of the growth of bacterial groups with putative health benefits.
The authors concluded40 that the functional significance and health implications of such changes are reasons for caution when reducing FODMAP intake in the long term and recommended liberalizing FODMAP restriction to the level of adequate symptom control in IBS patients. The study also recommended that people without symptoms not go on the low-FODMAP diet.40
Molecular approaches to characterize the gut microbiota are also being explored in an effort to identify its association with diet.
The sustainability of changes in gut microbiota and the potential long-term impact on health of following a low-FODMAP diet require further evaluation. In the meantime, patients following this diet should have FODMAP foods reintroduced based on tolerance and should consider taking probiotic supplements.41
DIETARY ADEQUACY OF THE LOW-FODMAP DIET
Continual dietary counseling should minimize nutritional inadequacies and ensure that FODMAPS are restricted only enough to control symptoms. Because no single food group is completely eliminated in this diet, patients are unlikely to experience inadequate nutrition.
Ledochowski et al26 found that in the initial, strict phase of the diet, total intake of carbohydrates (eg, starches, sugars) was reduced but intake of total energy, protein, fat, and nonstarch polysaccharides was not affected. Calcium intake was reduced in those following a low-FODMAP diet for 4 weeks.
The diet can also reduce total fiber intake and subsequently worsen constipation-predominant IBS. For those patients, lightly fermented high-fiber alternatives like oat and rice bran can be used.
ACCUMULATING EVIDENCE
The low-FODMAP diet is accumulating quality evidence for its effectiveness in controlling the functional gastrointestinal symptoms in patients with IBS. It can be difficult to adhere to over the long term due to its restrictiveness, and it is important to gradually liberalize the diet while tailoring it to the individual patient and monitoring symptoms. Further clinical trials are needed to evaluate this diet in different IBS subtypes and other gastrointestinal disorders, while defining its nutritional adequacy and effects on the intestinal microbiota profile.
The role of diet in controlling symptoms of irritable bowel syndrome (IBS) has gained much traction over the years,1 but until recently, diet therapy for IBS has been hindered by a lack of quality evidence, in part because of the challenges of conducting dietary clinical trials.
Several clinical trials have now been done that support a diet low in fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs) for managing IBS. Although restrictive and difficult to follow, the low-FODMAP diet is gaining popularity.
This article provides an overview of dietary interventions used to manage IBS, focusing on the low-FODMAP diet. We discuss mechanisms of malabsorption of FODMAPs and the role of FODMAPS in symptom induction; highlight clinical trials that provide evidence of benefits of the diet for IBS; and discuss the steps to implement it. We also address the nutritional adequacy of the diet and its potential effects on the gut microbiome.
IBS IS A COMMON FUNCTIONAL DISORDER
IBS is one of the most commonly diagnosed gastrointestinal disorders, and it has a significant impact on quality of life.2 It is a functional disorder characterized by chronic abdominal pain and altered bowel habits in the absence of a structural or organic cause.
The Rome IV diagnostic criteria define IBS by the following:
- Recurrent abdominal pain or discomfort at least 1 day a week in the last 3 months, associated with two or more of the following:
- Symptoms improved by defecation
- Onset associated with a change in frequency of stool
- Onset associated with a change in form or appearance of stool.
IBS mainly arises during young adulthood but can be diagnosed at any age.3
The pathophysiology of IBS involves mechanisms such as bowel distention, altered bowel motility, visceral hypersensitivity, and disruption of mucosal permeability.4 Several therapeutic modalities targeting these mechanisms have been implemented in IBS management, including antispasmodics, laxatives, antidepressants, antibiotics, and behavioral therapy. Diet is only one line of treatment and is most effective when part of a multipronged approach.
TRADITIONAL DIETARY MANAGEMENT
Diet is important in inducing the symptoms of IBS—and in controlling them. Patients identify eating as a common precipitator of symptoms, but the complex diet-symptom interaction is not fully understood and varies widely among patients. Traditional dietary advice for IBS includes adhering to a regular meal pattern, avoiding large meals, and reducing intake of fat, insoluble fibers, caffeine, spicy and gas-producing foods, and carbonated beverages.5,6
Increase soluble fiber
Fiber and fiber supplements, particularly soluble fibers such as psyllium, calcium polycarbophil, and ispaghula husk are often recommended. A meta-analysis7 found that soluble fiber but not insoluble fiber (eg, wheat bran) is associated with an improvement in IBS symptoms (relative risk [RR] 0.84, 95% confidence interval [CI] 0.73–0.94). By improving stool consistency and accelerating transit, soluble fiber is especially useful in constipation-predominant IBS while posing a low risk for adverse outcomes.7 Fiber should be started at a low dose and gradually increased over several weeks to as much as 20 to 30 g/day.
Avoid wheat
Only about 4% of patients with IBS also have celiac disease, but estimating the prevalence of nonceliac gluten sensitivity is confounded by overlapping symptoms. There is some evidence implicating gluten in IBS: celiac disease and IBS overlap in their symptoms, and symptoms are often precipitated by gluten-containing foods in patients with IBS.8 The pathogenesis of gluten-induced (or wheat-induced) symptoms in IBS is unclear, and studies have had conflicting results as to the benefits of gluten restriction in IBS.9
In a study of patients with IBS whose symptoms improved when they started a gluten-free and low-FODMAP diet, symptoms did not return when gluten was reintroduced, suggesting that it is the fructan (a FODMAP) component of wheat rather than gluten that contributes to symptoms in IBS.10
Probiotics
Probiotics are increasingly being recommended as dietary supplements for people with IBS, as awareness increases of the importance of the gut microbiota. In addition to their effects on the gut microbiota, probiotics in IBS have been shown to have anti-inflammatory effects, to alter gut motility, to modulate visceral hypersensitivity, and to restore epithelial integrity.
In a meta-analysis, Ford et al11 found that probiotics improved global IBS symptoms more than placebo (RR 0.79, 95% CI 0.70–0.89) and also reduced abdominal pain, bloating, and flatulence scores.
Which species and strains are most beneficial and the optimal dosing and duration of treatment are still unclear. Data from studies of prebiotics (nutrients that encourage the growth of probiotic bacteria) and synbiotics (combinations of prebiotics and probiotics) are limited and insufficient to draw conclusions.
FODMAPS ARE SHORT-CHAIN CARBOHYDRATES
The term FODMAPs was initially coined by researchers at Monash University in Australia to describe a collection of poorly absorbed short-chain fermentable carbohydrates that are natural components of many foods:
- Oligosaccharides, including fructans (which include inulins) and galacto-oligosaccharides
- Disaccharides, including lactose and sucrose
- Monosaccharides, including fructose
- Polyols, including sorbitol and mannitol.12
Intake of FODMAPs, especially fructose, has increased in Western diets over the past several decades from increased consumption of fruits and concentrated fruit juices, as well as from the widespread use of high-fructose corn syrup in processed foods and beverages.13
FODMAPs ARE POORLY ABSORBED
Different FODMAPs can be poorly absorbed for different reasons (Table 1). The poor absorption is related either to reduced or absent digestive enzymes (ie, hydrolases) or to slow transport across the intestinal mucosa. Excess FODMAPs in the distal small intestine and proximal colon exert osmotic pressure, drawing more water into the lumen. FODMAPs are also rapidly fermented by colonic bacteria, producing gas, bowel distention, and altered motility, all of which induce IBS symptoms.14
Fructans are fructose polymers that are not absorbed in human intestines. They have no intestinal hydrolases and no mechanisms for direct transport across the epithelium. However, a negligible amount may be absorbed after being degraded by microbes in the gut.15 Most dietary fructans are obtained from wheat and onion, which are actually low in fructans but tend to be consumed in large quantities.16
Galacto-oligosaccharides are available for colonic fermentation after ingestion due to lack of a human alpha-galactosidase. Common sources of galacto-oligosaccharides include legumes, nuts, seeds, some grains, dairy products, human milk, and some commercially produced forms added to infant formula.17,18
Lactose is poorly absorbed in people with lactase deficiency. It is mainly present in dairy products but is also added to commercial foods, including breads, cakes, and some diet products.19
Fructose is the most abundant FODMAP in the Western diet. It is either present as a free sugar or generated from the digestive breakdown of sucrose. In the intestine, it is absorbed via a direct low-capacity glucose transporter (GLUT)-5 and through GLUT-2, which is more efficient but requires the coexistence of glucose. Because of this requirement, fructose is more likely to be malabsorbed when present in excess of glucose, as in people with diminished sucrase activity. The main sources of fructose in the Western diet are fruits and fruit products, honey, and foods with added high-fructose sweeteners.13
Polyols such as sorbitol and mannitol are absorbed by slow passive diffusion because they have no active intestinal transport system. They are found in fruits and vegetables. Sugar-free chewing gum is a particularly rich source of sorbitol.20
QUANTIFYING FODMAP CONTENT
As interest in the low-FODMAP diet grew, studies were conducted to quantify FODMAPs in foods. One study used high-performance liquid chromatography to analyze FODMAP content in foods,21 and another evaluated fructan levels in a variety of fruits and vegetables using enzymatic hydrolysis.22 The Monash University low-FODMAP diet smartphone application provides patients and healthcare providers easy access to updated and detailed food analyses.23
Table 2 lists foods high in FODMAPs along with low-FODMAP alternatives. Total FODMAP intake is important, as the effects are additive.24 Readers and patients can be directed to the following websites for more information on the low-FODMAP diet: www.med.monash.edu/cecs/gastro/fodmap or www.ibsfree.net/what-is-fodmap-diet.
LOW-FODMAP DIET REDUCES SYMPTOMS
The low-FODMAP diet was inspired by the results of several studies that evaluated the role of dietary carbohydrates in inducing IBS symptoms and found improvement with their restriction.25,26
One study found that 74% of patients with IBS had less bloating, nausea, abdominal pain, and diarrhea when they restricted their intake of fructose and fructans.27
A prospective trial randomized 41 patients with IBS to 4 weeks of either a low-FODMAP diet or their habitual diet.28 The low-FODMAP diet resulted in greater improvement in overall IBS symptoms (P < .05) and stool frequency (P = .008). This study was limited by different habitual diets between patients and by lack of standardization of the low-FODMAP diet.
Halmos et al,29 in a randomized crossover trial, compared gastrointestinal symptoms in IBS patients over 3 weeks on a low-FODMAP diet vs a moderate-FODMAP (ie, regular) diet, as well as in healthy controls. Food was provided by the study and was matched for all nutrients. Up to 70% of the IBS patients had significantly lower overall symptom scores while on a low-FODMAP diet vs IBS patients on a regular diet (P < .001); bloating, abdominal pain, and flatulence were reduced. Symptoms were minimal and unaffected by either diet in the healthy controls.
A double-blind trial30 randomly assigned 25 patients with IBS who initially responded to a low-FODMAP diet to be challenged by a graduated dose of fructose alone, fructans alone, a combination of both, or glucose. The severity of overall and individual symptoms was markedly more reduced with glucose consumption than with the other carbohydrates: 70% of patients receiving fructose, 77% of those receiving fructans, and 79% of those receiving a mixture of both reported that their symptoms were not adequately controlled, compared with 14% of patients receiving glucose (P ≤ .002).30
Murray et al31 evaluated the gastrointestinal tract after a carbohydrate challenge consisting of 0.5 L of water containing 40 g of glucose, fructose, or inulin or a combination of 40 g of glucose and 40 g of fructose in 16 healthy volunteers. Magnetic resonance imaging was performed hourly for 5 hours to assess the volume of gastric contents, small-bowel water content, and colonic gas. Breath hydrogen was also measured, and symptoms were recorded after each imaging session.
Fructose significantly increased small-bowel water content compared with glucose (mean difference 28 L/min, P < .001), but combined glucose and fructose lessened the effect. Inulin had no significant effect on small-bowel water content (mean difference with glucose 2 L/min, P > .7) but led to the greatest production of colonic gas compared with glucose alone (mean difference 15 L/min, P < .05) and combined glucose and fructose (mean difference 12 L/min, P < .05). Inulin also produced the most breath hydrogen: 81% of participants had a rise after drinking inulin compared with 50% after drinking fructose. Glucose did not affect breath hydrogen concentrations, and combined glucose and fructose significantly reduced the concentration measured vs fructose alone. In patients who reported “gas” symptoms, a correlation was observed between the volume of gas in the colon and gas symptoms (r = 0.59, P < .0001).31
The authors concluded31 that long-chain carbohydrates such as inulin have a greater effect on colonic gas production and little effect on small-bowel water content, whereas small-chain FODMAPs such as fructose are likely to cause luminal distention in both the small and large intestines. The study also showed that combining equal amounts of glucose and fructose reduces malabsorption of fructose in the small bowel and reduces the effect of fructose on small-bowel water content and breath hydrogen concentration.31
PROBIOTICS HELP
A Danish study32 randomized 123 patients with IBS to one of three treatments: a low-FODMAP diet, a normal diet with probiotics containing the strain Lactobacillus rhamnosus GG (two capsules daily), or no special intervention. Symptoms were recorded weekly. IBS severity scores at week 6 were lower in patients on either the low-FODMAP diet or probiotics compared with the control group (P < .01). Subgroup analysis determined that patients with primarily diarrheal symptoms were more likely to have improved quality of life with the low-FODMAP diet.
A LOW-FODMAP DIET MAY ALSO HELP IN INFLAMMATORY BOWEL DISEASE
The low-FODMAP diet has also been studied in patients with inflammatory bowel disease with functional gut symptoms. In a retrospective pilot study,33 overall symptoms improved in about half of such patients on a low-FODMAP diet. A controlled dietary intervention trial is needed to confirm these findings and define the role of the low-FODMAP approach for patients with inflammatory bowel disease.
Marsh et al34 performed a meta-analysis of six randomized clinical trials and 16 nonrandomized interventions of a low-FODMAP diet on improving functional gastrointestinal symptoms in patients with either IBS or inflammatory bowel disease. They found significant improvements in:
- IBS Symptoms Severity Scores in the randomized trials (odds ratio [OR] 0.44, 95% CI 0.25–0.76)
- IBS Symptoms Severity Scores in the nonrandomized interventions (OR 0.03, 95% CI 0.01–0.2)
- IBS Quality of Life scores in the randomized trials (OR 1.84, 95% CI 1.12–3.03)
- IBS Quality of Life scores in the nonrandomized interventions (OR 3.18, 95% CI 1.6–6.31)
- Overall symptom severity in the randomized trials (OR 1.81, 95% CI 1.11–2.95).
DIETARY COUNSELING IS RECOMMENDED
Adherence is a major factor in the success of the low-FODMAP diet in IBS management and is strongly correlated with improved symptoms.35 Patients should be counseled on the role of food in inducing their symptoms. Haphazard dietary advice can be detrimental to outcomes, as many diets restrict food groups, impairing the consumption of essential nutrients.36 The involvement of a knowledgeable dietitian is helpful, as physicians may lack sufficient training in dietary skills and knowledge of food composition.
Access to and cost of dietary counseling can be prohibitive for some patients. Group consultation, which can decrease costs to each patient, has been found to be as effective as one-on-one sessions when administering the low-FODMAP diet in functional bowel disorders.37
ELIMINATION, THEN REINTRODUCTION
Before embarking on the low-FODMAP diet, the patient’s interest in making dietary changes should be explored, a dietary history taken, and unusual food choices or dietary behaviors assessed. The patient’s ability to adopt a restricted diet should also be gauged.
The diet should be implemented in two phases. The initial phase involves strict elimination of foods high in FODMAPs, usually over 6 to 8 weeks.38 Symptom control should be assessed: failure to control symptoms requires assessment of adherence.
If symptoms are successfully controlled, then the second phase should begin with the aim of following a less-restricted version of the diet as tolerated. Foods should gradually be phased back in and symptoms monitored. This approach minimizes unnecessary dietary restriction and ensures that a maximum variety in the diet is achieved while maintaining adequate symptom control.39
LOW-FODMAP DIET ALTERS THE GUT MICROBIOTA
Multiple putative benefits of certain bacterial species for colonic health have been reported, including the production of short-chain fatty acids. Colonic luminal concentrations of short-chain fatty acids may be important to gut health, given their role in intestinal secretion, absorption, motility, and epithelial cell structure. Because short-chain fatty acids are products of bacterial fermentation, a change in the delivery of fermentable substrates to the colon would be expected to alter the concentrations and output of fecal short-chain fatty acids.18
Several studies evaluated the effect of the low-FODMAP diet on intestinal microbiota, finding a change in the bacterial profile in the stool of patients who adopt this diet. Staudacher et al28 found a marked reduction in luminal bifidobacteria concentration after 4 weeks of a low-FODMAP diet in patients with IBS.
A single-blind randomized crossover trial40 investigated the effects of a low-FODMAP diet vs a carefully matched typical Australian diet in 27 patients with IBS and 6 healthy controls. Marked differences in absolute and relative bacterial abundance and diversity were found between the diets, but not in short-chain fatty acids or gut transit time. Compared with fecal microbiota on the typical diet, low FODMAP intake was associated with reduced absolute abundance of bacteria, and the typical FODMAP diet had evidence of stimulation of the growth of bacterial groups with putative health benefits.
The authors concluded40 that the functional significance and health implications of such changes are reasons for caution when reducing FODMAP intake in the long term and recommended liberalizing FODMAP restriction to the level of adequate symptom control in IBS patients. The study also recommended that people without symptoms not go on the low-FODMAP diet.40
Molecular approaches to characterize the gut microbiota are also being explored in an effort to identify its association with diet.
The sustainability of changes in gut microbiota and the potential long-term impact on health of following a low-FODMAP diet require further evaluation. In the meantime, patients following this diet should have FODMAP foods reintroduced based on tolerance and should consider taking probiotic supplements.41
DIETARY ADEQUACY OF THE LOW-FODMAP DIET
Continual dietary counseling should minimize nutritional inadequacies and ensure that FODMAPS are restricted only enough to control symptoms. Because no single food group is completely eliminated in this diet, patients are unlikely to experience inadequate nutrition.
Ledochowski et al26 found that in the initial, strict phase of the diet, total intake of carbohydrates (eg, starches, sugars) was reduced but intake of total energy, protein, fat, and nonstarch polysaccharides was not affected. Calcium intake was reduced in those following a low-FODMAP diet for 4 weeks.
The diet can also reduce total fiber intake and subsequently worsen constipation-predominant IBS. For those patients, lightly fermented high-fiber alternatives like oat and rice bran can be used.
ACCUMULATING EVIDENCE
The low-FODMAP diet is accumulating quality evidence for its effectiveness in controlling the functional gastrointestinal symptoms in patients with IBS. It can be difficult to adhere to over the long term due to its restrictiveness, and it is important to gradually liberalize the diet while tailoring it to the individual patient and monitoring symptoms. Further clinical trials are needed to evaluate this diet in different IBS subtypes and other gastrointestinal disorders, while defining its nutritional adequacy and effects on the intestinal microbiota profile.
- Hayes P, Corish C, O’Mahony E, Quigley EM. A dietary survey of patients with irritable bowel syndrome. J Hum Nutr Diet 2014; 27(suppl 2):36–47.
- Pare P, Gray J, Lam S, et al. Health-related quality of life, work productivity, and health care resource utilization of subjects with irritable bowel syndrome: baseline results from LOGIC (Longitudinal Outcomes Study of Gastrointestinal Symptoms in Canada), a naturalistic study. Clin Ther 2006; 28:1726–1735; discussion 1710–1711.
- Lacey BE, Mearin F, Chang L, et al. Bowel disorders. Gastroenterology 2016; 150:1393–1407.
- Drossman DA, Camilleri M, Mayer EA, Whitehead WE. AGA technical review on irritable bowel syndrome. Gastroenterology 2002; 123:2108–2131.
- Floch MH, Narayan R. Diet in the irritable bowel syndrome. J Clin Gastroenterol 2002; 35(suppl 1):S45–S52.
- Reding KW, Cain KC, Jarrett ME, Eugenio MD, Heitkemper MM. Relationship between patterns of alcohol consumption and gastrointestinal symptoms among patients with irritable bowel syndrome. Am J Gastroenterol 2013; 108:270–276.
- Moayyedi P, Quigley EM, Lacy BE, et al. The effect of fiber supplementation on irritable bowel syndrome: a systematic review and meta-analysis. Am J Gastroenterol 2014; 109:1367–1374.
- Vazquez Roque MI, Camilleri M, Smyrk T, et al. A controlled trial of gluten-free diet in patients with irritable bowel syndrome-diarrhea: effects on bowel frequency and intestinal function. Gastroenterology 2013; 144:903–911.
- Biesiekierski JR, Newnham ED, Irving PM, et al. Gluten causes gastrointestinal symptoms in subjects without celiac disease: a double-blind randomized placebo-controlled trial. Am J Gastroenterol 2011; 106:508–515.
- Biesiekierski JR, Peters SL, Newnham ED, Rosella O, Muir JG, Gibson PR. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 2013; 145:320–328.
- Ford AC, Quigley EM, Lacy BE, et al. Efficacy of prebiotics, probiotics, and synbiotics in irritable bowel syndrome and chronic idiopathic constipation: systematic review and meta-analysis. Gastroenterology 2013; 145:320–328.e1–e3.
- Central Clinical School, Monash University and The Alfred Hospital. The Monash University Low FODMAP Diet. 4th ed. Melbourne, Australia: Monash University; 2012.
- Parker K, Salas M, Nwosu VC. High fructose corn syrup: production, uses and public health concerns. Biotechnol Mol Biol Rev 2010; 5:71–78.
- Clausen MR, Jorgensen J, Mortensen PB. Comparison of diarrhea induced by ingestion of fructooligosaccharide idolax and disaccharide lactulose: role of osmolarity versus fermentation of malabsorbed carbohydrate. Dig Dis Sci 1998; 43:2696–2707.
- Barrett JS, Gearry RB, Muir JG, et al. Dietary poorly absorbed, short-chain carbohydrates increase delivery of water and fermentable substrates to the proximal colon. Aliment Pharmacol Ther 2010; 31:874–882.
- Whelan K, Abrahmsohn O, David GJ, et al. Fructan content of commonly consumed wheat, rye and gluten-free breads. Int J Food Sci Nutr 2011; 62:498–503.
- Sangwan V, Tomar SK, Singh RR, Singh AK, Ali B. Galactooligosaccharides: novel components of designer foods. J Food Sci 2011; 76:R103–R111.
- Russell DA, Ross RP, Fitzgerald GF, Stanton C. Metabolic activities and probiotic potential of bifidobacteria. Int J Food Microbiol 2011; 149:88–105.
- Lomer MC, Parkes GC, Sanderson JD. Review article: lactose intolerance in clinical practice—myths and realities. Aliment Pharmacol Ther 2008; 27:93–103.
- Langkilde AM, Andersson H, Schweizer TF, Würsch P. Digestion and absorption of sorbitol, maltitol and isomalt from the small bowel. A study in ileostomy subjects. Eur J Clin Nutr 1994; 48:768–775.
- Muir JG, Rose R, Rosella O, et al. Measurement of short-chain carbohydrates in common Australian vegetables and fruits by high-performance liquid chromatography (HPLC). J Agric Food Chem 2009; 57:554–565.
- Muir JG, Shepherd SJ, Rosella O, Rose R, Barrett JS, Gibson PR. Fructan and free fructose content of common Australian vegetables and fruit. J Agric Food Chem 2007; 55:6619–6627.
- Monash University. Monash launches Low FODMAP Diet smartphone app. http://med.monash.edu.au/news/2012/fodmap-app.html. Accessed July 13, 2016.
- Fedewa A, Rao SS. Dietary fructose intolerance, fructan intolerance and FODMAPs. Curr Gastroenterol Rep 2014; 16:370.
- Born P, Vierling T, Barina W. Fructose malabsorption and the irritable bowel syndrome. Gastroenterology 1991; 101:1454.
- Ledochowski M, Widner B, Bair H, Probst T, Fuchs D. Fructose- and sorbitol-reduced diet improves mood and gastrointestinal disturbances in fructose malabsorbers. Scand J Gastroenterol 2000; 35:1048–1052.
- Shepherd SJ, Gibson PR. Fructose malabsorption and symptoms of irritable bowel syndrome: guidelines for effective dietary management. J Am Diet Assoc 2006; 106:1631–1639.
- Staudacher HM, Lomer MC, Anderson JL, et al. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J Nutr 2012; 142:1510–1518.
- Halmos EP, Power VA, Shepherd SJ, Gibson PR, Muir JG. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 2014; 146:67–75.e5.
- Shepherd SJ, Parker FC, Muir JG, Gibson PR. Dietary triggers of abdominal symptoms in patients with irritable bowel syndrome: randomized placebo-controlled evidence. Clin Gastroenterol Hepatol 2008; 6:765–771.
- Murray K, Wilkinson-Smith V, Hoad C, et al. Differential effects of FODMAPs (fermentable oligo-, di-, mono-saccharides and polyols) on small and large intestinal contents in healthy subjects shown by MRI. Am J Gastroenterol 2014; 109:110–119.
- Pedersen N, Andersen NN, Vegh Z, et al. Ehealth: low FODMAP diet vs Lactobacillus rhamnosus GG in irritable bowel syndrome. World J Gastroenterol 2014; 20:16215–16226.
- Gearry RB, Irving PM, Barrett JS, Nathan DM, Shepherd SJ, Gibson PR. Reduction of dietary poorly absorbed short-chain carbohydrates (FODMAPs) improves abdominal symptoms in patients with inflammatory bowel disease-a pilot study. J Crohns Colitis 2009; 3:8–14.
- Marsh A, Eslick EM, Eslick GD. Does a diet low in FODMAPs reduce symptoms associated with functional gastrointestinal disorders? A comprehensive systematic review and meta-analysis. Eur J Nutr 2015 May 17. Epub ahead of print.
- de Roest RH, Dobbs BR, Chapman BA, et al. The low FODMAP diet improves gastrointestinal symptoms in patients with irritable bowel syndrome: a prospective study. Int J Clin Pract 2013; 67:895–903.
- Gibson PR, Barrett JS, Muir JG. Functional bowel symptoms and diet. Intern Med J 2013; 43:1067–1074.
- Whigham L, Joyce T, Harper G, et al. Clinical effectiveness and economic costs of group versus one-to-one education for short-chain fermentable carbohydrate restriction (low FODMAP diet) in the management of irritable bowel syndrome. J Hum Nutr Diet 2015; 28:687–696.
- Shepherd SJ, Lomer MC, Gibson PR. Short-chain carbohydrates and functional gastrointestinal disorders. Am J Gastroenterol 2013; 108:707–717.
- Shepherd SJ, Halmos E, Glance S. The role of FODMAPs in irritable bowel syndrome. Curr Opin Clin Nutr Metab Care 2014; 17:605–609.
- Halmos EP, Christophersen CT, Bird AR, Shepherd SJ, Gibson PR, Muir JG. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut 2015; 64:93–100.
- Staudacher HM, Irving PM, Lomer MC, Whelan K. Mechanisms and efficacy of dietary FODMAP restriction in IBS. Nat Rev Gastroenterol Hepatol 2014; 11:256–266.
- Hayes P, Corish C, O’Mahony E, Quigley EM. A dietary survey of patients with irritable bowel syndrome. J Hum Nutr Diet 2014; 27(suppl 2):36–47.
- Pare P, Gray J, Lam S, et al. Health-related quality of life, work productivity, and health care resource utilization of subjects with irritable bowel syndrome: baseline results from LOGIC (Longitudinal Outcomes Study of Gastrointestinal Symptoms in Canada), a naturalistic study. Clin Ther 2006; 28:1726–1735; discussion 1710–1711.
- Lacey BE, Mearin F, Chang L, et al. Bowel disorders. Gastroenterology 2016; 150:1393–1407.
- Drossman DA, Camilleri M, Mayer EA, Whitehead WE. AGA technical review on irritable bowel syndrome. Gastroenterology 2002; 123:2108–2131.
- Floch MH, Narayan R. Diet in the irritable bowel syndrome. J Clin Gastroenterol 2002; 35(suppl 1):S45–S52.
- Reding KW, Cain KC, Jarrett ME, Eugenio MD, Heitkemper MM. Relationship between patterns of alcohol consumption and gastrointestinal symptoms among patients with irritable bowel syndrome. Am J Gastroenterol 2013; 108:270–276.
- Moayyedi P, Quigley EM, Lacy BE, et al. The effect of fiber supplementation on irritable bowel syndrome: a systematic review and meta-analysis. Am J Gastroenterol 2014; 109:1367–1374.
- Vazquez Roque MI, Camilleri M, Smyrk T, et al. A controlled trial of gluten-free diet in patients with irritable bowel syndrome-diarrhea: effects on bowel frequency and intestinal function. Gastroenterology 2013; 144:903–911.
- Biesiekierski JR, Newnham ED, Irving PM, et al. Gluten causes gastrointestinal symptoms in subjects without celiac disease: a double-blind randomized placebo-controlled trial. Am J Gastroenterol 2011; 106:508–515.
- Biesiekierski JR, Peters SL, Newnham ED, Rosella O, Muir JG, Gibson PR. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 2013; 145:320–328.
- Ford AC, Quigley EM, Lacy BE, et al. Efficacy of prebiotics, probiotics, and synbiotics in irritable bowel syndrome and chronic idiopathic constipation: systematic review and meta-analysis. Gastroenterology 2013; 145:320–328.e1–e3.
- Central Clinical School, Monash University and The Alfred Hospital. The Monash University Low FODMAP Diet. 4th ed. Melbourne, Australia: Monash University; 2012.
- Parker K, Salas M, Nwosu VC. High fructose corn syrup: production, uses and public health concerns. Biotechnol Mol Biol Rev 2010; 5:71–78.
- Clausen MR, Jorgensen J, Mortensen PB. Comparison of diarrhea induced by ingestion of fructooligosaccharide idolax and disaccharide lactulose: role of osmolarity versus fermentation of malabsorbed carbohydrate. Dig Dis Sci 1998; 43:2696–2707.
- Barrett JS, Gearry RB, Muir JG, et al. Dietary poorly absorbed, short-chain carbohydrates increase delivery of water and fermentable substrates to the proximal colon. Aliment Pharmacol Ther 2010; 31:874–882.
- Whelan K, Abrahmsohn O, David GJ, et al. Fructan content of commonly consumed wheat, rye and gluten-free breads. Int J Food Sci Nutr 2011; 62:498–503.
- Sangwan V, Tomar SK, Singh RR, Singh AK, Ali B. Galactooligosaccharides: novel components of designer foods. J Food Sci 2011; 76:R103–R111.
- Russell DA, Ross RP, Fitzgerald GF, Stanton C. Metabolic activities and probiotic potential of bifidobacteria. Int J Food Microbiol 2011; 149:88–105.
- Lomer MC, Parkes GC, Sanderson JD. Review article: lactose intolerance in clinical practice—myths and realities. Aliment Pharmacol Ther 2008; 27:93–103.
- Langkilde AM, Andersson H, Schweizer TF, Würsch P. Digestion and absorption of sorbitol, maltitol and isomalt from the small bowel. A study in ileostomy subjects. Eur J Clin Nutr 1994; 48:768–775.
- Muir JG, Rose R, Rosella O, et al. Measurement of short-chain carbohydrates in common Australian vegetables and fruits by high-performance liquid chromatography (HPLC). J Agric Food Chem 2009; 57:554–565.
- Muir JG, Shepherd SJ, Rosella O, Rose R, Barrett JS, Gibson PR. Fructan and free fructose content of common Australian vegetables and fruit. J Agric Food Chem 2007; 55:6619–6627.
- Monash University. Monash launches Low FODMAP Diet smartphone app. http://med.monash.edu.au/news/2012/fodmap-app.html. Accessed July 13, 2016.
- Fedewa A, Rao SS. Dietary fructose intolerance, fructan intolerance and FODMAPs. Curr Gastroenterol Rep 2014; 16:370.
- Born P, Vierling T, Barina W. Fructose malabsorption and the irritable bowel syndrome. Gastroenterology 1991; 101:1454.
- Ledochowski M, Widner B, Bair H, Probst T, Fuchs D. Fructose- and sorbitol-reduced diet improves mood and gastrointestinal disturbances in fructose malabsorbers. Scand J Gastroenterol 2000; 35:1048–1052.
- Shepherd SJ, Gibson PR. Fructose malabsorption and symptoms of irritable bowel syndrome: guidelines for effective dietary management. J Am Diet Assoc 2006; 106:1631–1639.
- Staudacher HM, Lomer MC, Anderson JL, et al. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J Nutr 2012; 142:1510–1518.
- Halmos EP, Power VA, Shepherd SJ, Gibson PR, Muir JG. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 2014; 146:67–75.e5.
- Shepherd SJ, Parker FC, Muir JG, Gibson PR. Dietary triggers of abdominal symptoms in patients with irritable bowel syndrome: randomized placebo-controlled evidence. Clin Gastroenterol Hepatol 2008; 6:765–771.
- Murray K, Wilkinson-Smith V, Hoad C, et al. Differential effects of FODMAPs (fermentable oligo-, di-, mono-saccharides and polyols) on small and large intestinal contents in healthy subjects shown by MRI. Am J Gastroenterol 2014; 109:110–119.
- Pedersen N, Andersen NN, Vegh Z, et al. Ehealth: low FODMAP diet vs Lactobacillus rhamnosus GG in irritable bowel syndrome. World J Gastroenterol 2014; 20:16215–16226.
- Gearry RB, Irving PM, Barrett JS, Nathan DM, Shepherd SJ, Gibson PR. Reduction of dietary poorly absorbed short-chain carbohydrates (FODMAPs) improves abdominal symptoms in patients with inflammatory bowel disease-a pilot study. J Crohns Colitis 2009; 3:8–14.
- Marsh A, Eslick EM, Eslick GD. Does a diet low in FODMAPs reduce symptoms associated with functional gastrointestinal disorders? A comprehensive systematic review and meta-analysis. Eur J Nutr 2015 May 17. Epub ahead of print.
- de Roest RH, Dobbs BR, Chapman BA, et al. The low FODMAP diet improves gastrointestinal symptoms in patients with irritable bowel syndrome: a prospective study. Int J Clin Pract 2013; 67:895–903.
- Gibson PR, Barrett JS, Muir JG. Functional bowel symptoms and diet. Intern Med J 2013; 43:1067–1074.
- Whigham L, Joyce T, Harper G, et al. Clinical effectiveness and economic costs of group versus one-to-one education for short-chain fermentable carbohydrate restriction (low FODMAP diet) in the management of irritable bowel syndrome. J Hum Nutr Diet 2015; 28:687–696.
- Shepherd SJ, Lomer MC, Gibson PR. Short-chain carbohydrates and functional gastrointestinal disorders. Am J Gastroenterol 2013; 108:707–717.
- Shepherd SJ, Halmos E, Glance S. The role of FODMAPs in irritable bowel syndrome. Curr Opin Clin Nutr Metab Care 2014; 17:605–609.
- Halmos EP, Christophersen CT, Bird AR, Shepherd SJ, Gibson PR, Muir JG. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut 2015; 64:93–100.
- Staudacher HM, Irving PM, Lomer MC, Whelan K. Mechanisms and efficacy of dietary FODMAP restriction in IBS. Nat Rev Gastroenterol Hepatol 2014; 11:256–266.
KEY POINTS
- In clinical trials, the low-FODMAP diet has been found to improve symptoms in up to 70% of patients with IBS.
- FODMAPs are poorly absorbed for a variety of reasons.
- High-FODMAP foods include wheat, onions, legumes, dairy products, and many fruits and vegetables.
- The diet initially involves strict elimination of foods high in FODMAPs, after which they are gradually reintroduced as tolerated.
- A low-FODMAP diet may have negative effects on the gut microbiome. Therefore, we should be cautious about recommending this diet in the long term.
- Probiotics have a beneficial effect in IBS and can be taken concurrently with the diet.