Article
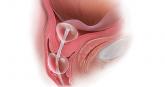
The case for outpatient cervical ripening for IOL at term for low-risk pregnancies
- Author:
- Robyn Lamar, MD, MPH
- Biftu Mengesha, MD, MAS
- Sarah Little, MD, MPH
These authors, who share their protocols, have been performing outpatient cervical ripening at their institutions for several...
