Article
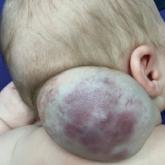
Vascular Mass on the Posterior Neck in a Newborn
- Author:
- Roksana Hesari, DO
- Mandy Alhajj, DO
- Howard D. Wang, MD
- Sonal D. Shah, MD
A newborn male was delivered via cesarean section at 38 weeks 5 days’ gestation with a large vascular mass on the posterior neck. The mass...
