Article

An FP’s guide to exercise counseling for older adults
- Author:
- Scott T. Larson, MD
- Tyler Slayman, MD
- Lucas Carr, PhD
At what intensity and frequency should older patients exercise? This review offers guidance and resources to help you get your patients moving.
Article
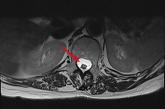
Sharp lower back pain • left-side paraspinal tenderness • anterior thigh sensory loss • Dx?
- Author:
- Srikanth Nithyanandam, MBBS, MS
- Kyle Smoot, MD, CAQ
- Kevin Schleich, PharmD
- Tyler Slayman, MD
► Acute-onset, sharp nonradiating lower back pain
► Left-side paraspinal tenderness
► Anterior thigh sensory loss
