User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
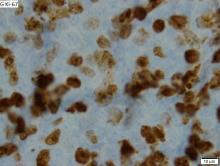
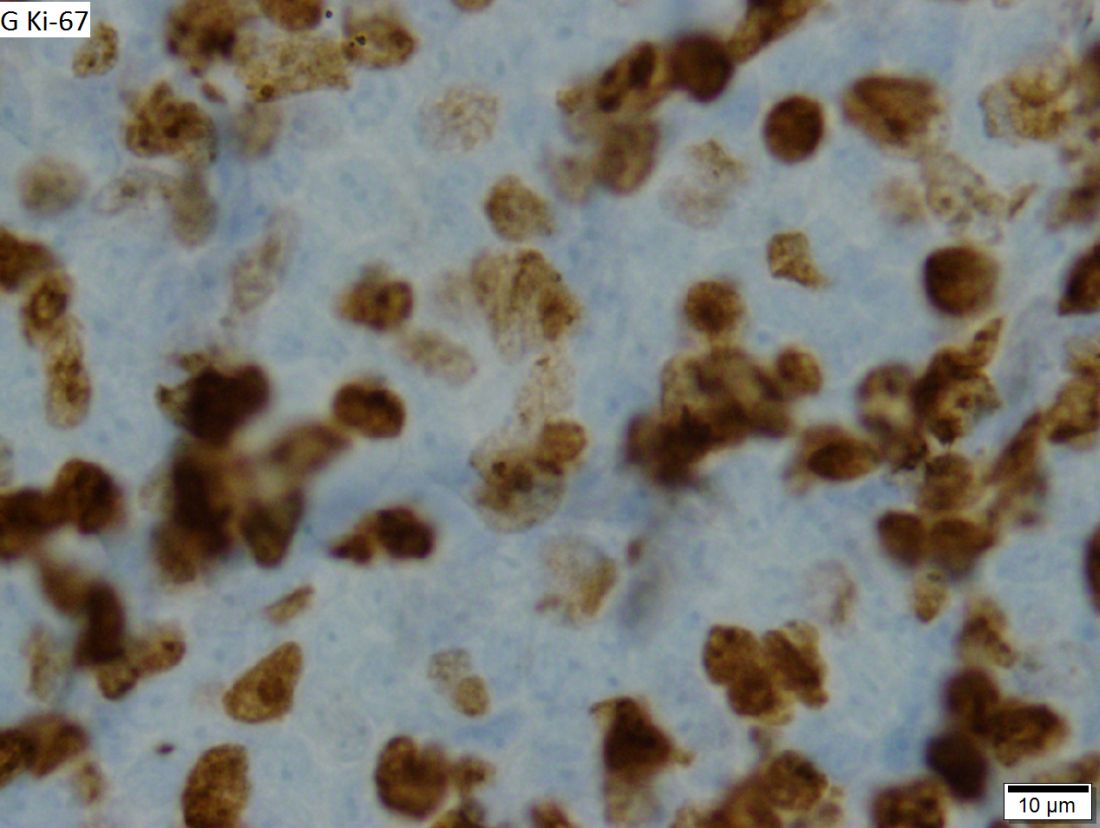
VcR-CVAD yields high responses, ‘excellent’ survival in MCL
Adding rituximab and bortezomib to a moderate-intensity chemotherapy regimen and following it up with maintenance rituximab produced high response rates and “excellent” survival outcomes for adults with previously untreated mantle cell lymphoma (MCL), investigators reported in long-term follow-up of a small study.
The objective response rate (ORR) among 30 patients with MCL treated with VcR-CVAD – bortezomib (Velcade), rituximab, and hyperCVAD (hyper-fractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone) followed by rituximab maintenance – was 90%, including a high proportion of complete responses (CR) or unconfirmed complete responses.
After a median follow-up of 7.8 years, the rates of 6-year progression-free and overall survival (PFS and OS) were 53% and 70%, respectively, with patients older and younger than 60 years having equally good outcomes, according to Julie E. Chang, MD, of the Wisconsin Institute of Medical Research in Madison, and her colleagues.
VcR-CVAD is a moderate-intensity regimen with a favorable toxicity profile that allowed tolerability even in an older population, the investigators noted. “An important lesson illustrated by VcR-CVAD is that long-term remissions are achievable in some patients without intensive inductions or consolidation,” they wrote in Clinical Lymphoma, Myeloma & Leukemia.
The investigators previously reported that after a median follow-up of 42 months, the 3-year PFS and OS were 63% and 86%, respectively, and that these outcomes were comparable to those reported with more intensive regimens (Br J Haematol. 2011 Oct;155[2]:190-7).
As noted, the ORR was 90%, including 77% CR/unconfirmed CR, 6-year PFS was 53%, and 6-year OS was 70%.
A univariate analysis showed a significant interaction between lactate dehydrogenase levels and age for PFS, and a trend, albeit not significant, toward an interaction with LDH levels and age for OS.
In multivariate analysis, worse Eastern Cooperative Oncology Group (ECOG) performance status at baseline showed a nonsignificant trend toward worse OS. In contrast, an increase of one in the number of extranodal disease sites was associated with better OS (relative risk 0.66, 95% confidence interval 0.01-0.66).
The investigators noted that the advent of new agents with activity against MCL and the use of prognostic information, such as minimal residual disease measurements, could help clinicians develop induction and maintenance strategies with better efficacy and lower toxicity than VcR-CVAD.
The study was supported by the National Institutes of Health, Millennium Pharmaceuticals, and the University of Wisconsin Forward Lymphoma Research Fund. Dr. Chang reported research funding from Genentech. One coauthor disclosed consulting work for Genentech and Millennium and research funding from Genentech.
hematologynews@frontlinemedcom.com
SOURCE: Chang J et al. Clin Lymphoma Myeloma Leuk. 2018 Jan;18(1):e61-e67. doi: 10.1016/j.clml.2017.10.006.
Adding rituximab and bortezomib to a moderate-intensity chemotherapy regimen and following it up with maintenance rituximab produced high response rates and “excellent” survival outcomes for adults with previously untreated mantle cell lymphoma (MCL), investigators reported in long-term follow-up of a small study.
The objective response rate (ORR) among 30 patients with MCL treated with VcR-CVAD – bortezomib (Velcade), rituximab, and hyperCVAD (hyper-fractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone) followed by rituximab maintenance – was 90%, including a high proportion of complete responses (CR) or unconfirmed complete responses.
After a median follow-up of 7.8 years, the rates of 6-year progression-free and overall survival (PFS and OS) were 53% and 70%, respectively, with patients older and younger than 60 years having equally good outcomes, according to Julie E. Chang, MD, of the Wisconsin Institute of Medical Research in Madison, and her colleagues.
VcR-CVAD is a moderate-intensity regimen with a favorable toxicity profile that allowed tolerability even in an older population, the investigators noted. “An important lesson illustrated by VcR-CVAD is that long-term remissions are achievable in some patients without intensive inductions or consolidation,” they wrote in Clinical Lymphoma, Myeloma & Leukemia.
The investigators previously reported that after a median follow-up of 42 months, the 3-year PFS and OS were 63% and 86%, respectively, and that these outcomes were comparable to those reported with more intensive regimens (Br J Haematol. 2011 Oct;155[2]:190-7).
As noted, the ORR was 90%, including 77% CR/unconfirmed CR, 6-year PFS was 53%, and 6-year OS was 70%.
A univariate analysis showed a significant interaction between lactate dehydrogenase levels and age for PFS, and a trend, albeit not significant, toward an interaction with LDH levels and age for OS.
In multivariate analysis, worse Eastern Cooperative Oncology Group (ECOG) performance status at baseline showed a nonsignificant trend toward worse OS. In contrast, an increase of one in the number of extranodal disease sites was associated with better OS (relative risk 0.66, 95% confidence interval 0.01-0.66).
The investigators noted that the advent of new agents with activity against MCL and the use of prognostic information, such as minimal residual disease measurements, could help clinicians develop induction and maintenance strategies with better efficacy and lower toxicity than VcR-CVAD.
The study was supported by the National Institutes of Health, Millennium Pharmaceuticals, and the University of Wisconsin Forward Lymphoma Research Fund. Dr. Chang reported research funding from Genentech. One coauthor disclosed consulting work for Genentech and Millennium and research funding from Genentech.
hematologynews@frontlinemedcom.com
SOURCE: Chang J et al. Clin Lymphoma Myeloma Leuk. 2018 Jan;18(1):e61-e67. doi: 10.1016/j.clml.2017.10.006.
Adding rituximab and bortezomib to a moderate-intensity chemotherapy regimen and following it up with maintenance rituximab produced high response rates and “excellent” survival outcomes for adults with previously untreated mantle cell lymphoma (MCL), investigators reported in long-term follow-up of a small study.
The objective response rate (ORR) among 30 patients with MCL treated with VcR-CVAD – bortezomib (Velcade), rituximab, and hyperCVAD (hyper-fractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone) followed by rituximab maintenance – was 90%, including a high proportion of complete responses (CR) or unconfirmed complete responses.
After a median follow-up of 7.8 years, the rates of 6-year progression-free and overall survival (PFS and OS) were 53% and 70%, respectively, with patients older and younger than 60 years having equally good outcomes, according to Julie E. Chang, MD, of the Wisconsin Institute of Medical Research in Madison, and her colleagues.
VcR-CVAD is a moderate-intensity regimen with a favorable toxicity profile that allowed tolerability even in an older population, the investigators noted. “An important lesson illustrated by VcR-CVAD is that long-term remissions are achievable in some patients without intensive inductions or consolidation,” they wrote in Clinical Lymphoma, Myeloma & Leukemia.
The investigators previously reported that after a median follow-up of 42 months, the 3-year PFS and OS were 63% and 86%, respectively, and that these outcomes were comparable to those reported with more intensive regimens (Br J Haematol. 2011 Oct;155[2]:190-7).
As noted, the ORR was 90%, including 77% CR/unconfirmed CR, 6-year PFS was 53%, and 6-year OS was 70%.
A univariate analysis showed a significant interaction between lactate dehydrogenase levels and age for PFS, and a trend, albeit not significant, toward an interaction with LDH levels and age for OS.
In multivariate analysis, worse Eastern Cooperative Oncology Group (ECOG) performance status at baseline showed a nonsignificant trend toward worse OS. In contrast, an increase of one in the number of extranodal disease sites was associated with better OS (relative risk 0.66, 95% confidence interval 0.01-0.66).
The investigators noted that the advent of new agents with activity against MCL and the use of prognostic information, such as minimal residual disease measurements, could help clinicians develop induction and maintenance strategies with better efficacy and lower toxicity than VcR-CVAD.
The study was supported by the National Institutes of Health, Millennium Pharmaceuticals, and the University of Wisconsin Forward Lymphoma Research Fund. Dr. Chang reported research funding from Genentech. One coauthor disclosed consulting work for Genentech and Millennium and research funding from Genentech.
hematologynews@frontlinemedcom.com
SOURCE: Chang J et al. Clin Lymphoma Myeloma Leuk. 2018 Jan;18(1):e61-e67. doi: 10.1016/j.clml.2017.10.006.
FROM CLINICAL LYMPHOMA, MYELOMA & LEUKEMIA
Key clinical point:
Major finding: The objective response rate was 90%, including 77% complete or unconfirmed complete responses.
Study details: Open-label study of 30 patients with previously untreated MCL.
Disclosures: The study was supported by the National Institutes of Health, Millennium Pharmaceuticals, and the University of Wisconsin Forward Lymphoma Research Fund. Dr. Chang reported research funding from Genentech. A coauthor reported consulting work for Genentech and Millennium and research funding from Genentech.
Source: Chang J et al. Clin Lymphoma Myeloma Leuk. 2018 Jan;18(1):e61-e67. doi: 10.1016/j.clml.2017.10.006.
BTK inhibitor zanubrutinib active in non-Hodgkin lymphomas
ATLANTA – , according to data presented at the annual meeting of the American Society of Hematology.
Response rates ranged from 31% to 88% depending on the lymphoma subtype. Overall, approximately 10% of patients discontinued the drug because of adverse events, reported Constantine S. Tam, MBBS, MD, of Peter MacCallum Cancer Centre & St. Vincent’s Hospital, Melbourne.
“There was encouraging activity against all the spectrum of indolent and aggressive NHL subtypes … and durable responses were observed across a variety of histologies,” Dr. Tam said.
Zanubrutinib is a second-generation BTK inhibitor that, based on biochemical assays, has higher selectivity against BTK than ibrutinib, Dr. Tam said.
He presented results of an open-label, multicenter, phase 1b study of daily or twice-daily zanubrutinib in patients with B-cell malignancies, most of them relapsed or refractory to prior therapies. The lymphoma subtypes he presented included diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), mantle cell lymphoma (MCL), and marginal zone lymphoma (MZL).
For 34 patients with indolent lymphomas (FL and MZL), the most frequent adverse events were petechiae/purpura/contusion and upper respiratory tract infection. Eleven grade 3-5 adverse events were reported, including neutropenia, infection, nausea, urinary tract infection, and abdominal pain.
Atrial fibrillation was observed in two patients in the aggressive NHL cohort, for an overall AF rate of approximately 2%, Dr. Tam said.
For 65 patients with aggressive lymphomas (DLBCL and MCL), the most frequent adverse events were petechiae/purpura/contusion and diarrhea; 27 grade 3-5 adverse events were reported, including neutropenia, pneumonia, and anemia.
The highest overall response rate reported was for MCL, at 88% (28 of 32 patients) followed by MZL at 78% (7 of 9 patients), FL at 41% (7 of 17 patients), and DLBCL 31% (8 of 26 patients).
The recommended phase 2 dose for zanubrutinib is either 320 mg/day once daily or a split dose of 160 mg twice daily, Dr. Tam said.
Based on this experience, investigators started a registration trial of zanubrutinib in combination with obinutuzumab for FL, and additional trials are planned, according to Dr. Tam.
There are also registration trials in Waldenstrom macroglobulinemia and chronic lymphocytic leukemia based on other data suggesting activity of zanubrutinib in those disease types, he added.
Zanubrutinib is a product of BeiGene. Dr. Tam reported disclosures related to Roche, Janssen Cilag, Abbvie, Celgene, Pharmacyclics, Onyx, and Amgen.
SOURCE: Tam C et al, ASH 2017, Abstract 152
ATLANTA – , according to data presented at the annual meeting of the American Society of Hematology.
Response rates ranged from 31% to 88% depending on the lymphoma subtype. Overall, approximately 10% of patients discontinued the drug because of adverse events, reported Constantine S. Tam, MBBS, MD, of Peter MacCallum Cancer Centre & St. Vincent’s Hospital, Melbourne.
“There was encouraging activity against all the spectrum of indolent and aggressive NHL subtypes … and durable responses were observed across a variety of histologies,” Dr. Tam said.
Zanubrutinib is a second-generation BTK inhibitor that, based on biochemical assays, has higher selectivity against BTK than ibrutinib, Dr. Tam said.
He presented results of an open-label, multicenter, phase 1b study of daily or twice-daily zanubrutinib in patients with B-cell malignancies, most of them relapsed or refractory to prior therapies. The lymphoma subtypes he presented included diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), mantle cell lymphoma (MCL), and marginal zone lymphoma (MZL).
For 34 patients with indolent lymphomas (FL and MZL), the most frequent adverse events were petechiae/purpura/contusion and upper respiratory tract infection. Eleven grade 3-5 adverse events were reported, including neutropenia, infection, nausea, urinary tract infection, and abdominal pain.
Atrial fibrillation was observed in two patients in the aggressive NHL cohort, for an overall AF rate of approximately 2%, Dr. Tam said.
For 65 patients with aggressive lymphomas (DLBCL and MCL), the most frequent adverse events were petechiae/purpura/contusion and diarrhea; 27 grade 3-5 adverse events were reported, including neutropenia, pneumonia, and anemia.
The highest overall response rate reported was for MCL, at 88% (28 of 32 patients) followed by MZL at 78% (7 of 9 patients), FL at 41% (7 of 17 patients), and DLBCL 31% (8 of 26 patients).
The recommended phase 2 dose for zanubrutinib is either 320 mg/day once daily or a split dose of 160 mg twice daily, Dr. Tam said.
Based on this experience, investigators started a registration trial of zanubrutinib in combination with obinutuzumab for FL, and additional trials are planned, according to Dr. Tam.
There are also registration trials in Waldenstrom macroglobulinemia and chronic lymphocytic leukemia based on other data suggesting activity of zanubrutinib in those disease types, he added.
Zanubrutinib is a product of BeiGene. Dr. Tam reported disclosures related to Roche, Janssen Cilag, Abbvie, Celgene, Pharmacyclics, Onyx, and Amgen.
SOURCE: Tam C et al, ASH 2017, Abstract 152
ATLANTA – , according to data presented at the annual meeting of the American Society of Hematology.
Response rates ranged from 31% to 88% depending on the lymphoma subtype. Overall, approximately 10% of patients discontinued the drug because of adverse events, reported Constantine S. Tam, MBBS, MD, of Peter MacCallum Cancer Centre & St. Vincent’s Hospital, Melbourne.
“There was encouraging activity against all the spectrum of indolent and aggressive NHL subtypes … and durable responses were observed across a variety of histologies,” Dr. Tam said.
Zanubrutinib is a second-generation BTK inhibitor that, based on biochemical assays, has higher selectivity against BTK than ibrutinib, Dr. Tam said.
He presented results of an open-label, multicenter, phase 1b study of daily or twice-daily zanubrutinib in patients with B-cell malignancies, most of them relapsed or refractory to prior therapies. The lymphoma subtypes he presented included diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), mantle cell lymphoma (MCL), and marginal zone lymphoma (MZL).
For 34 patients with indolent lymphomas (FL and MZL), the most frequent adverse events were petechiae/purpura/contusion and upper respiratory tract infection. Eleven grade 3-5 adverse events were reported, including neutropenia, infection, nausea, urinary tract infection, and abdominal pain.
Atrial fibrillation was observed in two patients in the aggressive NHL cohort, for an overall AF rate of approximately 2%, Dr. Tam said.
For 65 patients with aggressive lymphomas (DLBCL and MCL), the most frequent adverse events were petechiae/purpura/contusion and diarrhea; 27 grade 3-5 adverse events were reported, including neutropenia, pneumonia, and anemia.
The highest overall response rate reported was for MCL, at 88% (28 of 32 patients) followed by MZL at 78% (7 of 9 patients), FL at 41% (7 of 17 patients), and DLBCL 31% (8 of 26 patients).
The recommended phase 2 dose for zanubrutinib is either 320 mg/day once daily or a split dose of 160 mg twice daily, Dr. Tam said.
Based on this experience, investigators started a registration trial of zanubrutinib in combination with obinutuzumab for FL, and additional trials are planned, according to Dr. Tam.
There are also registration trials in Waldenstrom macroglobulinemia and chronic lymphocytic leukemia based on other data suggesting activity of zanubrutinib in those disease types, he added.
Zanubrutinib is a product of BeiGene. Dr. Tam reported disclosures related to Roche, Janssen Cilag, Abbvie, Celgene, Pharmacyclics, Onyx, and Amgen.
SOURCE: Tam C et al, ASH 2017, Abstract 152
REPORTING FROM ASH 2017
Key clinical point: Monotherapy with the BTK inhibitor zanubrutinib (BGB-3111) was active and well tolerated in patients with a variety of non-Hodgkin lymphoma (NHL) subtypes.
Major finding: Response rates ranged from 31% to 88% depending on the lymphoma subtype.
Data source: Preliminary results of an open-label, multicenter, phase 1b study including 99 patients with relapsed or refractory diffuse large B-cell lymphoma, follicular lymphoma, mantle cell lymphoma, or marginal zone lymphoma.
Disclosures: Zanubrutinib is a product of BeiGene. Constantine S. Tam, MBBS, MD, reported disclosures related to Roche, Janssen Cilag, Abbvie, Celgene, Pharmacyclics, Onyx, and Amgen.
Source: Tam C et al. ASH 2017, Abstract 152.
Study identifies predictors of acquired von Willebrand disease
ATLANTA – Waldenström macroglobulinemia can present as acquired von Willebrand disease (VWD), and when it does, the finding strongly correlates with high serum IgM levels and the presence of CXCR4 mutations, according to the results of a large, single-center retrospective study.
Further, successfully treating Waldenström macroglobulinemia often resolves acquired VWD, and the depth of treatment response predicts the degree of improvement, Jorge J. Castillo, MD, and his associates wrote in a poster presented at the annual meeting of the American Society of Hematology.
Acquired VWD is an uncommon, poorly understood presentation of Waldenström macroglobulinemia. Because affected patients require treatment, better characterizing this subgroup is important, the investigators noted.
At the Bing Center for Waldenström Macroglobulinemia at Dana-Farber Cancer Institute in Boston, the researchers retrospectively studied 320 individuals with newly diagnosed Waldenström macroglobulinemia and used logistic regression analysis to seek predictors of acquired VWD, which they evaluated by measuring levels of VW factor antigen, VW factor activity, and factor VIII. Levels under 30% were considered VWD and levels between 30% and 50% were considered low-level VWD.
In all, 49 individuals had acquired VWD while 271 patients did not. These two groups were similar in terms of age, sex, hemoglobin level, platelet count, and bone marrow involvement. However, 45% of patients with acquired VWD had serum IgM levels above 6,000 mg/dL versus 6% of patients without acquired VWD (P less than .001), and 47% of patients with acquired VWD had serum IgM levels between 3,000 and 5,999 versus 31% of patients without acquired VWD (P less than .001). Also, 77% of patients with acquired VWD tested positive for CXCR4 mutation versus 37% of patients without acquired VWD (P less than .001).
A significantly higher proportion of patients without acquired VWD had white blood cell concentrations above 6,000/mcL (29% vs. 50%; P = .006). This finding lost statistical significance in the logistic regression model, but all the other variables remained significantly associated. Serum IgM levels above 6,000 mg/dL conferred a 55-fold increase in the odds of having acquired VWD (95% confidence interval, 17-177; P less than .001), and serum IgM levels between 3,000 and 5,999 mg/dL led to an 11-fold increase in these odds (95% CI, 4-34). The presence of CXCR4 mutations was associated with a sixfold increased odds of acquired VWD (95% CI, 2-15). The P value for each of these three associations was at or below .001.
Therapy for Waldenström macroglobulinemia led to statistically significant increases in levels of factor VIII, VW factor antigen, and VW factor activity (P less than .001) and the median of each level improved by at least 35% after treatment. After treatment, 78% of patients with acquired VWD had levels of all three measures above 50% (versus 0% before treatment; P less than .001). Patients with acquired VWD with the best responses to treatment had about a 90% decrease in IgM levels, while those with a partial response had about a two-thirds decrease and patients with stable disease had about a 20% decrease. A linear regression model confirmed that depth of treatment response, based on change in IgM level, correlated with degree of improvement in VWD – that is, the extent of improvement in levels of VW factor antigen, VW factor activity, and factor VIII.
No external funding sources were reported. Dr. Castillo disclosed consulting ties and research funding from Pharmacyclics and Janssen, and research funding from Millenium and Abbvie.
SOURCE: Castillo J, et al. ASH 2017 Abstract 1088.
ATLANTA – Waldenström macroglobulinemia can present as acquired von Willebrand disease (VWD), and when it does, the finding strongly correlates with high serum IgM levels and the presence of CXCR4 mutations, according to the results of a large, single-center retrospective study.
Further, successfully treating Waldenström macroglobulinemia often resolves acquired VWD, and the depth of treatment response predicts the degree of improvement, Jorge J. Castillo, MD, and his associates wrote in a poster presented at the annual meeting of the American Society of Hematology.
Acquired VWD is an uncommon, poorly understood presentation of Waldenström macroglobulinemia. Because affected patients require treatment, better characterizing this subgroup is important, the investigators noted.
At the Bing Center for Waldenström Macroglobulinemia at Dana-Farber Cancer Institute in Boston, the researchers retrospectively studied 320 individuals with newly diagnosed Waldenström macroglobulinemia and used logistic regression analysis to seek predictors of acquired VWD, which they evaluated by measuring levels of VW factor antigen, VW factor activity, and factor VIII. Levels under 30% were considered VWD and levels between 30% and 50% were considered low-level VWD.
In all, 49 individuals had acquired VWD while 271 patients did not. These two groups were similar in terms of age, sex, hemoglobin level, platelet count, and bone marrow involvement. However, 45% of patients with acquired VWD had serum IgM levels above 6,000 mg/dL versus 6% of patients without acquired VWD (P less than .001), and 47% of patients with acquired VWD had serum IgM levels between 3,000 and 5,999 versus 31% of patients without acquired VWD (P less than .001). Also, 77% of patients with acquired VWD tested positive for CXCR4 mutation versus 37% of patients without acquired VWD (P less than .001).
A significantly higher proportion of patients without acquired VWD had white blood cell concentrations above 6,000/mcL (29% vs. 50%; P = .006). This finding lost statistical significance in the logistic regression model, but all the other variables remained significantly associated. Serum IgM levels above 6,000 mg/dL conferred a 55-fold increase in the odds of having acquired VWD (95% confidence interval, 17-177; P less than .001), and serum IgM levels between 3,000 and 5,999 mg/dL led to an 11-fold increase in these odds (95% CI, 4-34). The presence of CXCR4 mutations was associated with a sixfold increased odds of acquired VWD (95% CI, 2-15). The P value for each of these three associations was at or below .001.
Therapy for Waldenström macroglobulinemia led to statistically significant increases in levels of factor VIII, VW factor antigen, and VW factor activity (P less than .001) and the median of each level improved by at least 35% after treatment. After treatment, 78% of patients with acquired VWD had levels of all three measures above 50% (versus 0% before treatment; P less than .001). Patients with acquired VWD with the best responses to treatment had about a 90% decrease in IgM levels, while those with a partial response had about a two-thirds decrease and patients with stable disease had about a 20% decrease. A linear regression model confirmed that depth of treatment response, based on change in IgM level, correlated with degree of improvement in VWD – that is, the extent of improvement in levels of VW factor antigen, VW factor activity, and factor VIII.
No external funding sources were reported. Dr. Castillo disclosed consulting ties and research funding from Pharmacyclics and Janssen, and research funding from Millenium and Abbvie.
SOURCE: Castillo J, et al. ASH 2017 Abstract 1088.
ATLANTA – Waldenström macroglobulinemia can present as acquired von Willebrand disease (VWD), and when it does, the finding strongly correlates with high serum IgM levels and the presence of CXCR4 mutations, according to the results of a large, single-center retrospective study.
Further, successfully treating Waldenström macroglobulinemia often resolves acquired VWD, and the depth of treatment response predicts the degree of improvement, Jorge J. Castillo, MD, and his associates wrote in a poster presented at the annual meeting of the American Society of Hematology.
Acquired VWD is an uncommon, poorly understood presentation of Waldenström macroglobulinemia. Because affected patients require treatment, better characterizing this subgroup is important, the investigators noted.
At the Bing Center for Waldenström Macroglobulinemia at Dana-Farber Cancer Institute in Boston, the researchers retrospectively studied 320 individuals with newly diagnosed Waldenström macroglobulinemia and used logistic regression analysis to seek predictors of acquired VWD, which they evaluated by measuring levels of VW factor antigen, VW factor activity, and factor VIII. Levels under 30% were considered VWD and levels between 30% and 50% were considered low-level VWD.
In all, 49 individuals had acquired VWD while 271 patients did not. These two groups were similar in terms of age, sex, hemoglobin level, platelet count, and bone marrow involvement. However, 45% of patients with acquired VWD had serum IgM levels above 6,000 mg/dL versus 6% of patients without acquired VWD (P less than .001), and 47% of patients with acquired VWD had serum IgM levels between 3,000 and 5,999 versus 31% of patients without acquired VWD (P less than .001). Also, 77% of patients with acquired VWD tested positive for CXCR4 mutation versus 37% of patients without acquired VWD (P less than .001).
A significantly higher proportion of patients without acquired VWD had white blood cell concentrations above 6,000/mcL (29% vs. 50%; P = .006). This finding lost statistical significance in the logistic regression model, but all the other variables remained significantly associated. Serum IgM levels above 6,000 mg/dL conferred a 55-fold increase in the odds of having acquired VWD (95% confidence interval, 17-177; P less than .001), and serum IgM levels between 3,000 and 5,999 mg/dL led to an 11-fold increase in these odds (95% CI, 4-34). The presence of CXCR4 mutations was associated with a sixfold increased odds of acquired VWD (95% CI, 2-15). The P value for each of these three associations was at or below .001.
Therapy for Waldenström macroglobulinemia led to statistically significant increases in levels of factor VIII, VW factor antigen, and VW factor activity (P less than .001) and the median of each level improved by at least 35% after treatment. After treatment, 78% of patients with acquired VWD had levels of all three measures above 50% (versus 0% before treatment; P less than .001). Patients with acquired VWD with the best responses to treatment had about a 90% decrease in IgM levels, while those with a partial response had about a two-thirds decrease and patients with stable disease had about a 20% decrease. A linear regression model confirmed that depth of treatment response, based on change in IgM level, correlated with degree of improvement in VWD – that is, the extent of improvement in levels of VW factor antigen, VW factor activity, and factor VIII.
No external funding sources were reported. Dr. Castillo disclosed consulting ties and research funding from Pharmacyclics and Janssen, and research funding from Millenium and Abbvie.
SOURCE: Castillo J, et al. ASH 2017 Abstract 1088.
AT ASH 2017
Key clinical point: Successfully treating Waldenström macroglobulinemia often resolves acquired von Willebrand disease.
Major finding: Therapy for Waldenström macroglobulinemia led to statistically significant increases in levels of factor VIII, VW factor antigen, and VW factor activity (P less than .001) and the median of each level improved by at least 35% after treatment.
Data source: A single-center retrospective study of 320 patients with newly diagnosed Waldenström macroglobulinemia.
Disclosures: No external funding sources were reported. Dr. Castillo disclosed consultancy and research funding from Pharmacyclics and Janssen. He also disclosed research funding from Millenium and Abbvie.
Source: Castillo J, et al. ASH 2017 Abstract 1088.
Rare neurological complication linked to Waldenstrom disease
Bilateral facial nerve palsy has been associated with underlying Waldenstrom disease in only one other known published case report, which was published in 2014. In a more recent case report published in the Journal of Clinical Neuroscience, Gabriel Torrealba-Acosta, MD, and colleagues in the department of neurology at Massachusetts General Hospital, Boston, described a second case involving a 67-year-old Hispanic man with a history of Waldenstrom disease who presented with subacute onset of bilateral facial weakness.
The patient, who had longstanding painful neuropathy, had presented to urgent care with a new-onset left facial nerve palsy, was then diagnosed with left Bell’s palsy, and began treatment with valacyclovir and prednisone.
The left-sided facial weakness gradually progressed to total paralysis of the left lower face and inability to close the left eye, and 2 weeks later, he developed right facial weakness that ran a similar course. The patient had a complicated clinical course that included symptomatic acute-on-chronic subdural hematoma, among other complications; eventually the patient’s symptoms stabilized and cranial neuropathies gradually improved, according to the report.
Bilateral facial nerve palsy is an extremely rare condition, occurring in just 0.3%-2% of all facial nerve palsy cases, according to the authors. By contrast, unilateral facial nerve palsy (or Bell’s palsy) is far more common, but it still occurs in only 25 patients per 100,000 population, they said.
Most cases of bilateral facial nerve palsy are caused by underlying Guillain-Barré syndrome, though some are congenital, related to trauma, or caused by etiologies that are metabolic, immunologic, or neoplastic in nature. While various types of neurological disturbances – from ischemic and hemorrhagic events to meningoencephalitis – have been documented to occur in up to a quarter of patients with Waldenstrom disease.
“Given the large differential that comprises the assessment of a bilateral facial nerve palsy, it warrants for an extensive work-up, and Waldenstrom’s macroglobulinemia should be sought as an additional possible etiology,” the authors wrote.
Dr. Torrealba-Acosta and coauthors reported having no financial disclosures.
SOURCE: Torrealba-Acosta G et al. J Clin Neurosci. 2017. doi: 10.1016/j.jocn.2017.10.081.
Bilateral facial nerve palsy has been associated with underlying Waldenstrom disease in only one other known published case report, which was published in 2014. In a more recent case report published in the Journal of Clinical Neuroscience, Gabriel Torrealba-Acosta, MD, and colleagues in the department of neurology at Massachusetts General Hospital, Boston, described a second case involving a 67-year-old Hispanic man with a history of Waldenstrom disease who presented with subacute onset of bilateral facial weakness.
The patient, who had longstanding painful neuropathy, had presented to urgent care with a new-onset left facial nerve palsy, was then diagnosed with left Bell’s palsy, and began treatment with valacyclovir and prednisone.
The left-sided facial weakness gradually progressed to total paralysis of the left lower face and inability to close the left eye, and 2 weeks later, he developed right facial weakness that ran a similar course. The patient had a complicated clinical course that included symptomatic acute-on-chronic subdural hematoma, among other complications; eventually the patient’s symptoms stabilized and cranial neuropathies gradually improved, according to the report.
Bilateral facial nerve palsy is an extremely rare condition, occurring in just 0.3%-2% of all facial nerve palsy cases, according to the authors. By contrast, unilateral facial nerve palsy (or Bell’s palsy) is far more common, but it still occurs in only 25 patients per 100,000 population, they said.
Most cases of bilateral facial nerve palsy are caused by underlying Guillain-Barré syndrome, though some are congenital, related to trauma, or caused by etiologies that are metabolic, immunologic, or neoplastic in nature. While various types of neurological disturbances – from ischemic and hemorrhagic events to meningoencephalitis – have been documented to occur in up to a quarter of patients with Waldenstrom disease.
“Given the large differential that comprises the assessment of a bilateral facial nerve palsy, it warrants for an extensive work-up, and Waldenstrom’s macroglobulinemia should be sought as an additional possible etiology,” the authors wrote.
Dr. Torrealba-Acosta and coauthors reported having no financial disclosures.
SOURCE: Torrealba-Acosta G et al. J Clin Neurosci. 2017. doi: 10.1016/j.jocn.2017.10.081.
Bilateral facial nerve palsy has been associated with underlying Waldenstrom disease in only one other known published case report, which was published in 2014. In a more recent case report published in the Journal of Clinical Neuroscience, Gabriel Torrealba-Acosta, MD, and colleagues in the department of neurology at Massachusetts General Hospital, Boston, described a second case involving a 67-year-old Hispanic man with a history of Waldenstrom disease who presented with subacute onset of bilateral facial weakness.
The patient, who had longstanding painful neuropathy, had presented to urgent care with a new-onset left facial nerve palsy, was then diagnosed with left Bell’s palsy, and began treatment with valacyclovir and prednisone.
The left-sided facial weakness gradually progressed to total paralysis of the left lower face and inability to close the left eye, and 2 weeks later, he developed right facial weakness that ran a similar course. The patient had a complicated clinical course that included symptomatic acute-on-chronic subdural hematoma, among other complications; eventually the patient’s symptoms stabilized and cranial neuropathies gradually improved, according to the report.
Bilateral facial nerve palsy is an extremely rare condition, occurring in just 0.3%-2% of all facial nerve palsy cases, according to the authors. By contrast, unilateral facial nerve palsy (or Bell’s palsy) is far more common, but it still occurs in only 25 patients per 100,000 population, they said.
Most cases of bilateral facial nerve palsy are caused by underlying Guillain-Barré syndrome, though some are congenital, related to trauma, or caused by etiologies that are metabolic, immunologic, or neoplastic in nature. While various types of neurological disturbances – from ischemic and hemorrhagic events to meningoencephalitis – have been documented to occur in up to a quarter of patients with Waldenstrom disease.
“Given the large differential that comprises the assessment of a bilateral facial nerve palsy, it warrants for an extensive work-up, and Waldenstrom’s macroglobulinemia should be sought as an additional possible etiology,” the authors wrote.
Dr. Torrealba-Acosta and coauthors reported having no financial disclosures.
SOURCE: Torrealba-Acosta G et al. J Clin Neurosci. 2017. doi: 10.1016/j.jocn.2017.10.081.
FROM THE JOURNAL OF CLINICAL NEUROSCIENCE
Rituximab may be best choice for splenic MZL
For patients with splenic marginal zone lymphoma (MZL) requiring treatment, rituximab may be a superior choice, compared with splenectomy, according to authors of a recent review article.
Although treatment with splenectomy and rituximab both are associated with high rates of 10-year survival, splenectomy also is associated with acute surgical complications and late toxicities, mainly from infections, Christina Kalpadakis, MD, of the department of hematology at Heraklion University Hospital, University of Crete, Greece, reported in Best Practice & Research Clinical Haematology.
The review article aims to answer the question of whether rituximab monotherapy should replace splenectomy as the treatment of choice, according to the authors, who said treatment for this rare, low-grade B-cell lymphoma has not been standardized due to a lack of large, randomized trials.
Asymptomatic splenic MZL without significant cytopenias can be managed with a watch-and-wait approach, but when treatment is warranted, the choice is often between splenectomy, rituximab monotherapy, or chemoimmunotherapy.
“Based on the existing retrospective series of patients, rituximab monotherapy appears to be the best choice since it combines high efficacy with the lowest toxicity,” Dr. Kalpadakis and her colleagues wrote.
Until the early 2000s, splenectomy was the standard treatment modality for splenic MZL, offering quick amelioration of symptoms related to splenomegaly in more than 90% of patients, as well as improvements in hypersplenism-related cytopenias, the researchers reported. Among 100 patients with splenic MZL, the median progression-free survival with splenectomy was 8.25 years, and median 10-year overall survival was 67%.
However, responses to splenectomy are not complete because some patients are not good candidates for splenectomy, including those with lymphadenopathy or heavy bone marrow infiltration, according to the authors. Moreover, since it is a major surgical procedure that can be associated with complications and infections, splenectomy is not appropriate for elderly patients or patients with comorbidities with a high surgical risk, they wrote.
“Furthermore, splenectomy cannot eradicate bone marrow disease and has no impact on other extrasplenic disease localization such as lymphadenopathy,” they added.
Rituximab monotherapy, by contrast, has “minimal toxicity” with high efficacy, including complete response rates of around 50% and a 10-year overall survival of 85% reported in a series of 104 patients.
Maintenance with rituximab may further improve the quality of responses, although data are limited. One study, conducted by the review article authors, showed that rituximab maintenance increased the complete response rate from 42% after induction treatment to 71% after maintenance, along with a significant improvement in response duration, though no differences in overall survival have been observed thus far.
Rituximab plus chemotherapy has been evaluated, but with “significant toxicity without improvement in the outcome” versus rituximab monotherapy, the researchers wrote. Splenectomy is “effective” but should be reserved for patients refractory to rituximab, Dr. Kalpadakis and her colleagues wrote.
The researchers reported having no relevant financial disclosures.
SOURCE: Kalpadakis C et al. Best Pract Res Clin Haematol. Mar-Jun 2017. doi:10.1016/j.beha.2017.10.011.
For patients with splenic marginal zone lymphoma (MZL) requiring treatment, rituximab may be a superior choice, compared with splenectomy, according to authors of a recent review article.
Although treatment with splenectomy and rituximab both are associated with high rates of 10-year survival, splenectomy also is associated with acute surgical complications and late toxicities, mainly from infections, Christina Kalpadakis, MD, of the department of hematology at Heraklion University Hospital, University of Crete, Greece, reported in Best Practice & Research Clinical Haematology.
The review article aims to answer the question of whether rituximab monotherapy should replace splenectomy as the treatment of choice, according to the authors, who said treatment for this rare, low-grade B-cell lymphoma has not been standardized due to a lack of large, randomized trials.
Asymptomatic splenic MZL without significant cytopenias can be managed with a watch-and-wait approach, but when treatment is warranted, the choice is often between splenectomy, rituximab monotherapy, or chemoimmunotherapy.
“Based on the existing retrospective series of patients, rituximab monotherapy appears to be the best choice since it combines high efficacy with the lowest toxicity,” Dr. Kalpadakis and her colleagues wrote.
Until the early 2000s, splenectomy was the standard treatment modality for splenic MZL, offering quick amelioration of symptoms related to splenomegaly in more than 90% of patients, as well as improvements in hypersplenism-related cytopenias, the researchers reported. Among 100 patients with splenic MZL, the median progression-free survival with splenectomy was 8.25 years, and median 10-year overall survival was 67%.
However, responses to splenectomy are not complete because some patients are not good candidates for splenectomy, including those with lymphadenopathy or heavy bone marrow infiltration, according to the authors. Moreover, since it is a major surgical procedure that can be associated with complications and infections, splenectomy is not appropriate for elderly patients or patients with comorbidities with a high surgical risk, they wrote.
“Furthermore, splenectomy cannot eradicate bone marrow disease and has no impact on other extrasplenic disease localization such as lymphadenopathy,” they added.
Rituximab monotherapy, by contrast, has “minimal toxicity” with high efficacy, including complete response rates of around 50% and a 10-year overall survival of 85% reported in a series of 104 patients.
Maintenance with rituximab may further improve the quality of responses, although data are limited. One study, conducted by the review article authors, showed that rituximab maintenance increased the complete response rate from 42% after induction treatment to 71% after maintenance, along with a significant improvement in response duration, though no differences in overall survival have been observed thus far.
Rituximab plus chemotherapy has been evaluated, but with “significant toxicity without improvement in the outcome” versus rituximab monotherapy, the researchers wrote. Splenectomy is “effective” but should be reserved for patients refractory to rituximab, Dr. Kalpadakis and her colleagues wrote.
The researchers reported having no relevant financial disclosures.
SOURCE: Kalpadakis C et al. Best Pract Res Clin Haematol. Mar-Jun 2017. doi:10.1016/j.beha.2017.10.011.
For patients with splenic marginal zone lymphoma (MZL) requiring treatment, rituximab may be a superior choice, compared with splenectomy, according to authors of a recent review article.
Although treatment with splenectomy and rituximab both are associated with high rates of 10-year survival, splenectomy also is associated with acute surgical complications and late toxicities, mainly from infections, Christina Kalpadakis, MD, of the department of hematology at Heraklion University Hospital, University of Crete, Greece, reported in Best Practice & Research Clinical Haematology.
The review article aims to answer the question of whether rituximab monotherapy should replace splenectomy as the treatment of choice, according to the authors, who said treatment for this rare, low-grade B-cell lymphoma has not been standardized due to a lack of large, randomized trials.
Asymptomatic splenic MZL without significant cytopenias can be managed with a watch-and-wait approach, but when treatment is warranted, the choice is often between splenectomy, rituximab monotherapy, or chemoimmunotherapy.
“Based on the existing retrospective series of patients, rituximab monotherapy appears to be the best choice since it combines high efficacy with the lowest toxicity,” Dr. Kalpadakis and her colleagues wrote.
Until the early 2000s, splenectomy was the standard treatment modality for splenic MZL, offering quick amelioration of symptoms related to splenomegaly in more than 90% of patients, as well as improvements in hypersplenism-related cytopenias, the researchers reported. Among 100 patients with splenic MZL, the median progression-free survival with splenectomy was 8.25 years, and median 10-year overall survival was 67%.
However, responses to splenectomy are not complete because some patients are not good candidates for splenectomy, including those with lymphadenopathy or heavy bone marrow infiltration, according to the authors. Moreover, since it is a major surgical procedure that can be associated with complications and infections, splenectomy is not appropriate for elderly patients or patients with comorbidities with a high surgical risk, they wrote.
“Furthermore, splenectomy cannot eradicate bone marrow disease and has no impact on other extrasplenic disease localization such as lymphadenopathy,” they added.
Rituximab monotherapy, by contrast, has “minimal toxicity” with high efficacy, including complete response rates of around 50% and a 10-year overall survival of 85% reported in a series of 104 patients.
Maintenance with rituximab may further improve the quality of responses, although data are limited. One study, conducted by the review article authors, showed that rituximab maintenance increased the complete response rate from 42% after induction treatment to 71% after maintenance, along with a significant improvement in response duration, though no differences in overall survival have been observed thus far.
Rituximab plus chemotherapy has been evaluated, but with “significant toxicity without improvement in the outcome” versus rituximab monotherapy, the researchers wrote. Splenectomy is “effective” but should be reserved for patients refractory to rituximab, Dr. Kalpadakis and her colleagues wrote.
The researchers reported having no relevant financial disclosures.
SOURCE: Kalpadakis C et al. Best Pract Res Clin Haematol. Mar-Jun 2017. doi:10.1016/j.beha.2017.10.011.
FROM BEST PRACTICE & RESEARCH CLINICAL HAEMATOLOGY
Key clinical point:
Major finding: Both splenectomy and rituximab are associated with high rates of 10-year overalls survival, but splenectomy has higher rates of surgical complications and infection.
Study details: Review article of 63 publications, mostly retrospective studies of marginal zone lymphoma.
Disclosures: The researchers reported having no relevant financial disclosures.
Source: Kalpadakis C et al. Best Pract Res Clin Haematol. 2017 Mar-Jun. doi:10.1016/j.beha.2017.10.011.
VIDEO - New lymphoma drug approvals: Clinical use, future directions
ATLANTA – 2017 was a banner year for the approval of new drugs to treat hematologic disorders.
At a special interest session at the annual meeting of American Society of Hematology, representatives from the Food and Drug Administration joined forces with clinicians to discuss the use of the newly approved treatments in the real-world setting.
In this video interview,
“This is extremely exciting,” she said regarding the pace of new approvals for hematologic malignancies.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Axicabtagene ciloleucel, a CAR T-cell product approved in October for the treatment of relapsed/refractory large B-cell lymphoma in adults, is particularly interesting, she said.
“The data shows that if you look at a population of diffuse large B-cell lymphoma patients, that historically have a very poor outcome, there is definitely an impressive response rate and improved survival, compared to the natural history cohort,” said Dr. Heslop of Baylor College of Medicine, Houston.
However, while the findings are encouraging, only 30%-40% are having a durable response, she added.
“So I think there’ll be lots of efforts to try and improve the response rate by combination with other agents such as checkpoint inhibitors or other immunomodulators,” she said.
With respect to the second-generation Bruton’s tyrosine kinase inhibitor acalabrutinib, which was approved in October for adults with mantle cell lymphoma who have been treated with at least one prior therapy, she discussed the potential for improved outcomes and the importance of looking further into its use in patients who have failed ibrutinib therapy, as well as its use in combination with other agents, such as bendamustine and rituximab early in the course of disease.
Copanlisib, a PI3 kinase inhibitor approved in September, is an addition to the armamentarium for adult patients with relapsed follicular lymphoma after two lines of previous therapy.
“It still does have some side effects, as do other drugs in this class, so I think it’s place will still need to be defined,” Dr. Heslop said.
She reported having no relevant financial disclosures.
ATLANTA – 2017 was a banner year for the approval of new drugs to treat hematologic disorders.
At a special interest session at the annual meeting of American Society of Hematology, representatives from the Food and Drug Administration joined forces with clinicians to discuss the use of the newly approved treatments in the real-world setting.
In this video interview,
“This is extremely exciting,” she said regarding the pace of new approvals for hematologic malignancies.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Axicabtagene ciloleucel, a CAR T-cell product approved in October for the treatment of relapsed/refractory large B-cell lymphoma in adults, is particularly interesting, she said.
“The data shows that if you look at a population of diffuse large B-cell lymphoma patients, that historically have a very poor outcome, there is definitely an impressive response rate and improved survival, compared to the natural history cohort,” said Dr. Heslop of Baylor College of Medicine, Houston.
However, while the findings are encouraging, only 30%-40% are having a durable response, she added.
“So I think there’ll be lots of efforts to try and improve the response rate by combination with other agents such as checkpoint inhibitors or other immunomodulators,” she said.
With respect to the second-generation Bruton’s tyrosine kinase inhibitor acalabrutinib, which was approved in October for adults with mantle cell lymphoma who have been treated with at least one prior therapy, she discussed the potential for improved outcomes and the importance of looking further into its use in patients who have failed ibrutinib therapy, as well as its use in combination with other agents, such as bendamustine and rituximab early in the course of disease.
Copanlisib, a PI3 kinase inhibitor approved in September, is an addition to the armamentarium for adult patients with relapsed follicular lymphoma after two lines of previous therapy.
“It still does have some side effects, as do other drugs in this class, so I think it’s place will still need to be defined,” Dr. Heslop said.
She reported having no relevant financial disclosures.
ATLANTA – 2017 was a banner year for the approval of new drugs to treat hematologic disorders.
At a special interest session at the annual meeting of American Society of Hematology, representatives from the Food and Drug Administration joined forces with clinicians to discuss the use of the newly approved treatments in the real-world setting.
In this video interview,
“This is extremely exciting,” she said regarding the pace of new approvals for hematologic malignancies.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Axicabtagene ciloleucel, a CAR T-cell product approved in October for the treatment of relapsed/refractory large B-cell lymphoma in adults, is particularly interesting, she said.
“The data shows that if you look at a population of diffuse large B-cell lymphoma patients, that historically have a very poor outcome, there is definitely an impressive response rate and improved survival, compared to the natural history cohort,” said Dr. Heslop of Baylor College of Medicine, Houston.
However, while the findings are encouraging, only 30%-40% are having a durable response, she added.
“So I think there’ll be lots of efforts to try and improve the response rate by combination with other agents such as checkpoint inhibitors or other immunomodulators,” she said.
With respect to the second-generation Bruton’s tyrosine kinase inhibitor acalabrutinib, which was approved in October for adults with mantle cell lymphoma who have been treated with at least one prior therapy, she discussed the potential for improved outcomes and the importance of looking further into its use in patients who have failed ibrutinib therapy, as well as its use in combination with other agents, such as bendamustine and rituximab early in the course of disease.
Copanlisib, a PI3 kinase inhibitor approved in September, is an addition to the armamentarium for adult patients with relapsed follicular lymphoma after two lines of previous therapy.
“It still does have some side effects, as do other drugs in this class, so I think it’s place will still need to be defined,” Dr. Heslop said.
She reported having no relevant financial disclosures.
REPORTING FROM ASH 2017
VIDEO: Ibrutinib PFS is nearly 3 years in MCL patients who had one prior therapy
ATLANTA – Ibrutinib yielded a median progression-free survival (PFS) of nearly 3 years for patients with relapsed or refractory mantle cell lymphoma (MCL) treated with the agent after just one prior line of therapy, according to pooled long-term follow-up data presented at the annual meeting of the American Society of Hematology.
With a 3.5-year median follow-up, a median PFS of 33.6 months was reported for MCL patients with one prior line of therapy, compared to 8.4 months for patients who had two or more prior lines of therapy, reported lead study author Simon Rule, MD, of Plymouth (England) University Medical School.
“I think the take-home message from the ibrutinib data is the earlier you use the drug in the relapse setting, the better the outcomes you’re going to get,” Dr. Rule said in an interview. “It is quite dramatic, the difference between one prior line of therapy and subsequent lines of therapy.”
Response rates were also higher in MCL patients who had only one prior line of therapy, Dr. Rule said.
The overall and complete response rates for that group were 77.8% and 36.4%, respectively, according to data Dr. Rule presented in an oral presentation. For patients receiving more than one line of therapy prior to ibrutinib, overall and complete response rates were 66.8% and 22.9%.
The pooled analysis presented by Dr. Rule included 370 patients enrolled in ibrutinib trials between 2011 and 2013. Patients in those trials received oral ibrutinib 560 mg daily until progressive disease or unacceptable toxicity.
Patients achieving a CR had an “extraordinary” PFS of nearly 4 years and a median duration of response of 55 months, Dr. Rule said.
Atrial fibrillation (AF) rates were reported to be 5.7% (21/370 patients). A total of 53 patients in the analysis had ongoing controlled AF/arrhythmia or had a history of it. Over the course of follow-up, 70% of them (37 patients) had no recurrences, according to Dr. Rule and his colleagues.
No patients discontinued ibrutinib because of grade 3-4 AF, they reported. Moreover, less than 2% of the 370 patients discontinued or reduced dose of ibrutinib because of grade 3-4 bleeding or AF.
New onset grade 3-4 adverse events were more common in the first year of treatment and generally were less frequent over time, Dr. Rule said.
Patients with only one prior line of therapy were less likely to have grade 3-4 adverse events, suggesting again that “the earlier you use [ibrutinib], the fewer side effects you get, particularly with hematologic toxicity,” Dr. Rule said.
Sponsorship for the research came from Janssen, and funding for writing assistance came from Janssen Global Services. Dr. Rule reported financial relationships with Janssen and several other companies.
SOURCE: Rule S et al. ASH 2017 Abstract 151.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – Ibrutinib yielded a median progression-free survival (PFS) of nearly 3 years for patients with relapsed or refractory mantle cell lymphoma (MCL) treated with the agent after just one prior line of therapy, according to pooled long-term follow-up data presented at the annual meeting of the American Society of Hematology.
With a 3.5-year median follow-up, a median PFS of 33.6 months was reported for MCL patients with one prior line of therapy, compared to 8.4 months for patients who had two or more prior lines of therapy, reported lead study author Simon Rule, MD, of Plymouth (England) University Medical School.
“I think the take-home message from the ibrutinib data is the earlier you use the drug in the relapse setting, the better the outcomes you’re going to get,” Dr. Rule said in an interview. “It is quite dramatic, the difference between one prior line of therapy and subsequent lines of therapy.”
Response rates were also higher in MCL patients who had only one prior line of therapy, Dr. Rule said.
The overall and complete response rates for that group were 77.8% and 36.4%, respectively, according to data Dr. Rule presented in an oral presentation. For patients receiving more than one line of therapy prior to ibrutinib, overall and complete response rates were 66.8% and 22.9%.
The pooled analysis presented by Dr. Rule included 370 patients enrolled in ibrutinib trials between 2011 and 2013. Patients in those trials received oral ibrutinib 560 mg daily until progressive disease or unacceptable toxicity.
Patients achieving a CR had an “extraordinary” PFS of nearly 4 years and a median duration of response of 55 months, Dr. Rule said.
Atrial fibrillation (AF) rates were reported to be 5.7% (21/370 patients). A total of 53 patients in the analysis had ongoing controlled AF/arrhythmia or had a history of it. Over the course of follow-up, 70% of them (37 patients) had no recurrences, according to Dr. Rule and his colleagues.
No patients discontinued ibrutinib because of grade 3-4 AF, they reported. Moreover, less than 2% of the 370 patients discontinued or reduced dose of ibrutinib because of grade 3-4 bleeding or AF.
New onset grade 3-4 adverse events were more common in the first year of treatment and generally were less frequent over time, Dr. Rule said.
Patients with only one prior line of therapy were less likely to have grade 3-4 adverse events, suggesting again that “the earlier you use [ibrutinib], the fewer side effects you get, particularly with hematologic toxicity,” Dr. Rule said.
Sponsorship for the research came from Janssen, and funding for writing assistance came from Janssen Global Services. Dr. Rule reported financial relationships with Janssen and several other companies.
SOURCE: Rule S et al. ASH 2017 Abstract 151.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – Ibrutinib yielded a median progression-free survival (PFS) of nearly 3 years for patients with relapsed or refractory mantle cell lymphoma (MCL) treated with the agent after just one prior line of therapy, according to pooled long-term follow-up data presented at the annual meeting of the American Society of Hematology.
With a 3.5-year median follow-up, a median PFS of 33.6 months was reported for MCL patients with one prior line of therapy, compared to 8.4 months for patients who had two or more prior lines of therapy, reported lead study author Simon Rule, MD, of Plymouth (England) University Medical School.
“I think the take-home message from the ibrutinib data is the earlier you use the drug in the relapse setting, the better the outcomes you’re going to get,” Dr. Rule said in an interview. “It is quite dramatic, the difference between one prior line of therapy and subsequent lines of therapy.”
Response rates were also higher in MCL patients who had only one prior line of therapy, Dr. Rule said.
The overall and complete response rates for that group were 77.8% and 36.4%, respectively, according to data Dr. Rule presented in an oral presentation. For patients receiving more than one line of therapy prior to ibrutinib, overall and complete response rates were 66.8% and 22.9%.
The pooled analysis presented by Dr. Rule included 370 patients enrolled in ibrutinib trials between 2011 and 2013. Patients in those trials received oral ibrutinib 560 mg daily until progressive disease or unacceptable toxicity.
Patients achieving a CR had an “extraordinary” PFS of nearly 4 years and a median duration of response of 55 months, Dr. Rule said.
Atrial fibrillation (AF) rates were reported to be 5.7% (21/370 patients). A total of 53 patients in the analysis had ongoing controlled AF/arrhythmia or had a history of it. Over the course of follow-up, 70% of them (37 patients) had no recurrences, according to Dr. Rule and his colleagues.
No patients discontinued ibrutinib because of grade 3-4 AF, they reported. Moreover, less than 2% of the 370 patients discontinued or reduced dose of ibrutinib because of grade 3-4 bleeding or AF.
New onset grade 3-4 adverse events were more common in the first year of treatment and generally were less frequent over time, Dr. Rule said.
Patients with only one prior line of therapy were less likely to have grade 3-4 adverse events, suggesting again that “the earlier you use [ibrutinib], the fewer side effects you get, particularly with hematologic toxicity,” Dr. Rule said.
Sponsorship for the research came from Janssen, and funding for writing assistance came from Janssen Global Services. Dr. Rule reported financial relationships with Janssen and several other companies.
SOURCE: Rule S et al. ASH 2017 Abstract 151.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
REPORTING FROM ASH 2017
Key clinical point:
Major finding: Median PFS was 33.6 months for MCL patients with one prior line of therapy, versus 8.4 months for patients who had two or more prior lines of therapy.
Study details: A pooled analysis of 370 patients enrolled in ibrutinib clinical trials with a median 3.5-year follow-up.
Disclosures: Janssen sponsored the research and Janssen Global Services funded writing assistance. Lead author Simon Rule, MD, reported financial relationships with Janssen and several other companies.
Source: Rule S et al. ASH 2017 Abstract 151.
VTE rates in lenalidomide-treated NHL may warrant prophylaxis
ATLANTA – The rate of venous thromboembolism (VTE) in patients with non-Hodgkin lymphoma (NHL) treated with lenalidomide is similar to that seen in multiple myeloma, according to results of recent systematic review and meta-analysis of trials representing more than 10,000 treatment cycles.
Although rates of VTE for NHL and myeloma could not be directly compared statistically, the finding may have clinical implications for NHL patients, said lead study author Samuel Yamshon, MD, an internal medicine resident at Cornell University, New York.
“Although outpatient VTE prophylaxis is not currently recommended, it should be carefully considered in patients with lymphoma being treated with lenalidomide, especially those receiving lenalidomide as a single agent,” Dr. Yamshon said in a presentation of the results at the annual meeting of the American Society of Hematology.
The rate of thrombosis in patients with B cell NHL who received lenalidomide treatment was 0.75 events per 100 patient-cycles, according to results of the meta-analysis, which was based on 28 articles including 10,332 cycles of lenalidomide received by patients with B-cell NHL.
Reported rates of thrombosis in previously untreated myeloma patients treated with lenalidomide are between 0.7 and 0.8 per 100 patient-cycles, Dr. Yamshon said in his presentation.
Notably, single-agent lenalidomide was linked with a significantly increased risk of thrombosis compared with lenalidomide treatment in combinations. The relative risk of VTE for lenalidomide as a single agent versus lenalidomide in combination was 2.01 (95% confidence interval, 1.28-3.16; P = .002), according to the presented data.
The investigators were unsure why single-agent lenalidomide appeared to have caused increased rates of thrombosis compared to lenalidomide in combinations. “Perhaps patients treated with additional agents have a lower tumor burden, leading to less venous obstruction causing clots, or perhaps there’s a direct interaction between lenalidomide and tumor leading to effects on the vasculature and mediators of coagulation,” Dr. Yamshon said.
Chemotherapy and biologic combinations had somewhat different VTE rates when compared to single-agent lenalidomide. The rate in patients receiving lenalidomide alone was 1.06 events per 100 patient-cycles, compared with 0.73 and 0.41 events per 100 patient-cycles, respectively, for lenalidomide plus chemotherapy and lenalidomide plus biologics.
However, the lower event rate with lenalidomide and biologics compared with lenalidomide and chemotherapy was a “nonsignificant trend” that was likely caused by differences in patient characteristics between the two cohorts, according to Dr. Yamshon.
None of the studies included in the meta-analysis were prospectively designed to measure VTE as a primary or secondary outcome, Dr. Yamshon noted in a discussion of the study’s limitations.
Further studies are warranted to determine lenalidomide’s effect on the vasculature and how it effects mediators of coagulation, he added.
Based on the current results, Dr. Yamshon said it may be reasonable to consider VTE prophylaxis in NHL patients receiving lenalidomide.
“If we’re going to be recommending outpatient VTE prophylaxis in everyone on lenalidomide in multiple myeloma, and the rates (of VTE) are the same, I think it certainly makes sense based on the data to recommend it,” he said in a question-and-answer session.
Dr. Yamshon reported no conflicts related to the study. Coauthors reported disclosures related to Roche, Celgene, Seattle Genetics, Pharmacyclics, Cell Medica, Janssen, and AstraZeneca.
SOURCE: Yamshon S et al. Abstract 677.
ATLANTA – The rate of venous thromboembolism (VTE) in patients with non-Hodgkin lymphoma (NHL) treated with lenalidomide is similar to that seen in multiple myeloma, according to results of recent systematic review and meta-analysis of trials representing more than 10,000 treatment cycles.
Although rates of VTE for NHL and myeloma could not be directly compared statistically, the finding may have clinical implications for NHL patients, said lead study author Samuel Yamshon, MD, an internal medicine resident at Cornell University, New York.
“Although outpatient VTE prophylaxis is not currently recommended, it should be carefully considered in patients with lymphoma being treated with lenalidomide, especially those receiving lenalidomide as a single agent,” Dr. Yamshon said in a presentation of the results at the annual meeting of the American Society of Hematology.
The rate of thrombosis in patients with B cell NHL who received lenalidomide treatment was 0.75 events per 100 patient-cycles, according to results of the meta-analysis, which was based on 28 articles including 10,332 cycles of lenalidomide received by patients with B-cell NHL.
Reported rates of thrombosis in previously untreated myeloma patients treated with lenalidomide are between 0.7 and 0.8 per 100 patient-cycles, Dr. Yamshon said in his presentation.
Notably, single-agent lenalidomide was linked with a significantly increased risk of thrombosis compared with lenalidomide treatment in combinations. The relative risk of VTE for lenalidomide as a single agent versus lenalidomide in combination was 2.01 (95% confidence interval, 1.28-3.16; P = .002), according to the presented data.
The investigators were unsure why single-agent lenalidomide appeared to have caused increased rates of thrombosis compared to lenalidomide in combinations. “Perhaps patients treated with additional agents have a lower tumor burden, leading to less venous obstruction causing clots, or perhaps there’s a direct interaction between lenalidomide and tumor leading to effects on the vasculature and mediators of coagulation,” Dr. Yamshon said.
Chemotherapy and biologic combinations had somewhat different VTE rates when compared to single-agent lenalidomide. The rate in patients receiving lenalidomide alone was 1.06 events per 100 patient-cycles, compared with 0.73 and 0.41 events per 100 patient-cycles, respectively, for lenalidomide plus chemotherapy and lenalidomide plus biologics.
However, the lower event rate with lenalidomide and biologics compared with lenalidomide and chemotherapy was a “nonsignificant trend” that was likely caused by differences in patient characteristics between the two cohorts, according to Dr. Yamshon.
None of the studies included in the meta-analysis were prospectively designed to measure VTE as a primary or secondary outcome, Dr. Yamshon noted in a discussion of the study’s limitations.
Further studies are warranted to determine lenalidomide’s effect on the vasculature and how it effects mediators of coagulation, he added.
Based on the current results, Dr. Yamshon said it may be reasonable to consider VTE prophylaxis in NHL patients receiving lenalidomide.
“If we’re going to be recommending outpatient VTE prophylaxis in everyone on lenalidomide in multiple myeloma, and the rates (of VTE) are the same, I think it certainly makes sense based on the data to recommend it,” he said in a question-and-answer session.
Dr. Yamshon reported no conflicts related to the study. Coauthors reported disclosures related to Roche, Celgene, Seattle Genetics, Pharmacyclics, Cell Medica, Janssen, and AstraZeneca.
SOURCE: Yamshon S et al. Abstract 677.
ATLANTA – The rate of venous thromboembolism (VTE) in patients with non-Hodgkin lymphoma (NHL) treated with lenalidomide is similar to that seen in multiple myeloma, according to results of recent systematic review and meta-analysis of trials representing more than 10,000 treatment cycles.
Although rates of VTE for NHL and myeloma could not be directly compared statistically, the finding may have clinical implications for NHL patients, said lead study author Samuel Yamshon, MD, an internal medicine resident at Cornell University, New York.
“Although outpatient VTE prophylaxis is not currently recommended, it should be carefully considered in patients with lymphoma being treated with lenalidomide, especially those receiving lenalidomide as a single agent,” Dr. Yamshon said in a presentation of the results at the annual meeting of the American Society of Hematology.
The rate of thrombosis in patients with B cell NHL who received lenalidomide treatment was 0.75 events per 100 patient-cycles, according to results of the meta-analysis, which was based on 28 articles including 10,332 cycles of lenalidomide received by patients with B-cell NHL.
Reported rates of thrombosis in previously untreated myeloma patients treated with lenalidomide are between 0.7 and 0.8 per 100 patient-cycles, Dr. Yamshon said in his presentation.
Notably, single-agent lenalidomide was linked with a significantly increased risk of thrombosis compared with lenalidomide treatment in combinations. The relative risk of VTE for lenalidomide as a single agent versus lenalidomide in combination was 2.01 (95% confidence interval, 1.28-3.16; P = .002), according to the presented data.
The investigators were unsure why single-agent lenalidomide appeared to have caused increased rates of thrombosis compared to lenalidomide in combinations. “Perhaps patients treated with additional agents have a lower tumor burden, leading to less venous obstruction causing clots, or perhaps there’s a direct interaction between lenalidomide and tumor leading to effects on the vasculature and mediators of coagulation,” Dr. Yamshon said.
Chemotherapy and biologic combinations had somewhat different VTE rates when compared to single-agent lenalidomide. The rate in patients receiving lenalidomide alone was 1.06 events per 100 patient-cycles, compared with 0.73 and 0.41 events per 100 patient-cycles, respectively, for lenalidomide plus chemotherapy and lenalidomide plus biologics.
However, the lower event rate with lenalidomide and biologics compared with lenalidomide and chemotherapy was a “nonsignificant trend” that was likely caused by differences in patient characteristics between the two cohorts, according to Dr. Yamshon.
None of the studies included in the meta-analysis were prospectively designed to measure VTE as a primary or secondary outcome, Dr. Yamshon noted in a discussion of the study’s limitations.
Further studies are warranted to determine lenalidomide’s effect on the vasculature and how it effects mediators of coagulation, he added.
Based on the current results, Dr. Yamshon said it may be reasonable to consider VTE prophylaxis in NHL patients receiving lenalidomide.
“If we’re going to be recommending outpatient VTE prophylaxis in everyone on lenalidomide in multiple myeloma, and the rates (of VTE) are the same, I think it certainly makes sense based on the data to recommend it,” he said in a question-and-answer session.
Dr. Yamshon reported no conflicts related to the study. Coauthors reported disclosures related to Roche, Celgene, Seattle Genetics, Pharmacyclics, Cell Medica, Janssen, and AstraZeneca.
SOURCE: Yamshon S et al. Abstract 677.
AT ASH 2017
Key clinical point: The rates of VTE in patients on lenalidomide are similar whether they’re being treated for B cell non-Hodgkin lymphoma (NHL) or multiple myeloma, which suggests that VTE prophylaxis should be more carefully considered in B cell NHL patients.
Major finding: The rate of thrombosis in patients with B cell NHL who received lenalidomide treatment was 0.75 events per 100 patient-cycles.
Data source: A systematic review and meta-analysis of 28 articles including 10,332 cycles of lenalidomide received by patients with B cell NHL.
Disclosures: Authors of the study reported disclosures related to Roche, Celgene, Seattle Genetics, Pharmacyclics, Cell Medica, Janssen, and AstraZeneca.
Source: Yamshon S et al. Abstract 677.
VIDEO: Venetoclax/rituximab prolongs PFS in relapsed/refractory CLL
ATLANTA – Relapsed or refractory chronic lymphocytic leukemia (CLL) often has a suboptimal response to conventional chemotherapy, because of adverse biological features that can accumulate in cells.
The combination of bendamustine (Treanda) and rituximab has been associated with about 60% overall responses rates, median progression-free survival of approximately 15 months, and overall survival of nearly 3 years in patients with CLL, and there is now evidence that substituting venetoclax (Venclexta) for bendamustine could improve outcomes even further.
In a video interview at the annual meeting of the American Society of Hematology, comparing bendamustine plus rituximab with venetoclax plus rituximab in patients with relapsed/refractory CLL.
Venetoclax/rituximab was superior to bendamustine/rituximab for prolonging progression-free survival, with effects consistent across subgroups, regardless of mutation status, and for having a clinically meaningful improvement in overall survival.
The MURANO trial was funded by AbbVie. Dr. Seymour reported honoraria, research funding, and advisory committee and speakers bureau participation for AbbVie and other companies.
ATLANTA – Relapsed or refractory chronic lymphocytic leukemia (CLL) often has a suboptimal response to conventional chemotherapy, because of adverse biological features that can accumulate in cells.
The combination of bendamustine (Treanda) and rituximab has been associated with about 60% overall responses rates, median progression-free survival of approximately 15 months, and overall survival of nearly 3 years in patients with CLL, and there is now evidence that substituting venetoclax (Venclexta) for bendamustine could improve outcomes even further.
In a video interview at the annual meeting of the American Society of Hematology, comparing bendamustine plus rituximab with venetoclax plus rituximab in patients with relapsed/refractory CLL.
Venetoclax/rituximab was superior to bendamustine/rituximab for prolonging progression-free survival, with effects consistent across subgroups, regardless of mutation status, and for having a clinically meaningful improvement in overall survival.
The MURANO trial was funded by AbbVie. Dr. Seymour reported honoraria, research funding, and advisory committee and speakers bureau participation for AbbVie and other companies.
ATLANTA – Relapsed or refractory chronic lymphocytic leukemia (CLL) often has a suboptimal response to conventional chemotherapy, because of adverse biological features that can accumulate in cells.
The combination of bendamustine (Treanda) and rituximab has been associated with about 60% overall responses rates, median progression-free survival of approximately 15 months, and overall survival of nearly 3 years in patients with CLL, and there is now evidence that substituting venetoclax (Venclexta) for bendamustine could improve outcomes even further.
In a video interview at the annual meeting of the American Society of Hematology, comparing bendamustine plus rituximab with venetoclax plus rituximab in patients with relapsed/refractory CLL.
Venetoclax/rituximab was superior to bendamustine/rituximab for prolonging progression-free survival, with effects consistent across subgroups, regardless of mutation status, and for having a clinically meaningful improvement in overall survival.
The MURANO trial was funded by AbbVie. Dr. Seymour reported honoraria, research funding, and advisory committee and speakers bureau participation for AbbVie and other companies.
REPORTING FROM ASH 2017
VIDEO: CAR T cell axi-cel drives B-cell lymphomas into remission
ATLANTA – In the ZUMA-1 trial, more than one-third of patients with refractory large B-cell lymphomas treated with the chimeric antigen receptor (CAR) T-cell product axicabtagene ciloleucel (Yescarta; axi-cel) had durable responses, with some patients having complete responses lasting more than 1 year after a single infusion.
Updated combined phase 1 and 2 results in 108 patients with diffuse large B-cell lymphoma, primary mediastinal B-cell lymphoma, or transformed follicular lymphoma showed an objective response rate of 82% of patients – including 58% showing complete responses – after a median follow-up of 15.4 months.
In a video interview at the annual meeting of the American Society of Hematology, Sattva S. Neelapu, MD, from the University of Texas MD Anderson Cancer Center in Houston discusses the use of CAR T cells directed against the CD19 antigen in patients with relapsed/refractory B-cell lymphomas and describes efforts to improve responses while managing adverse events common to CAR T-cell therapies, notably cytokine release syndrome.
ZUMA-1 is supported by Kite Pharma, which developed axicabtagene ciloleucel, and the Leukemia & Lymphoma Society’s Therapy Acceleration Program. Dr. Neelapu reported receiving advisory board fees from the company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – In the ZUMA-1 trial, more than one-third of patients with refractory large B-cell lymphomas treated with the chimeric antigen receptor (CAR) T-cell product axicabtagene ciloleucel (Yescarta; axi-cel) had durable responses, with some patients having complete responses lasting more than 1 year after a single infusion.
Updated combined phase 1 and 2 results in 108 patients with diffuse large B-cell lymphoma, primary mediastinal B-cell lymphoma, or transformed follicular lymphoma showed an objective response rate of 82% of patients – including 58% showing complete responses – after a median follow-up of 15.4 months.
In a video interview at the annual meeting of the American Society of Hematology, Sattva S. Neelapu, MD, from the University of Texas MD Anderson Cancer Center in Houston discusses the use of CAR T cells directed against the CD19 antigen in patients with relapsed/refractory B-cell lymphomas and describes efforts to improve responses while managing adverse events common to CAR T-cell therapies, notably cytokine release syndrome.
ZUMA-1 is supported by Kite Pharma, which developed axicabtagene ciloleucel, and the Leukemia & Lymphoma Society’s Therapy Acceleration Program. Dr. Neelapu reported receiving advisory board fees from the company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – In the ZUMA-1 trial, more than one-third of patients with refractory large B-cell lymphomas treated with the chimeric antigen receptor (CAR) T-cell product axicabtagene ciloleucel (Yescarta; axi-cel) had durable responses, with some patients having complete responses lasting more than 1 year after a single infusion.
Updated combined phase 1 and 2 results in 108 patients with diffuse large B-cell lymphoma, primary mediastinal B-cell lymphoma, or transformed follicular lymphoma showed an objective response rate of 82% of patients – including 58% showing complete responses – after a median follow-up of 15.4 months.
In a video interview at the annual meeting of the American Society of Hematology, Sattva S. Neelapu, MD, from the University of Texas MD Anderson Cancer Center in Houston discusses the use of CAR T cells directed against the CD19 antigen in patients with relapsed/refractory B-cell lymphomas and describes efforts to improve responses while managing adverse events common to CAR T-cell therapies, notably cytokine release syndrome.
ZUMA-1 is supported by Kite Pharma, which developed axicabtagene ciloleucel, and the Leukemia & Lymphoma Society’s Therapy Acceleration Program. Dr. Neelapu reported receiving advisory board fees from the company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
REPORTING FROM ASH 2017