User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Checkpoint inhibitor plus rituximab is active in non-Hodgkin lymphoma
A macrophage-activating immune checkpoint inhibitor, combined with rituximab therapy, was safe and produced durable complete responses in patients with relapsed or refractory non-Hodgkin lymphoma, according to results of a phase 1b study.
Mainly low-grade toxic effects were seen on treatment with Hu5F9-G4 (5F9) and rituximab, which induced responses in more than half of patients, of which more than one-third were complete responses, the study investigators reported.
Most of the responses were ongoing at the time of data cutoff, suggesting durable responses with the combination of rituximab and 5F9 – a humanized monoclonal antibody that blocks CD47, an antiphagocytic or “do not eat me” signal overexpressed by most cancers, Ranjana Advani, MD, of Stanford (Calif.) University, and her coauthors wrote.
“The macrophage-mediated activity of 5F9 plus rituximab may serve as an effective new immunotherapy for stimulating the innate immune system,” Dr. Advani and her colleagues reported in the New England Journal of Medicine.
The study included 22 patients, including 15 with diffuse large B-cell lymphoma (DLBCL) and 7 with follicular lymphoma, who had received a median of four prior therapies. Almost all of the non-Hodgkin lymphomas (21, or 95%) were refractory to rituximab.
All patients received intravenous 5F9 starting with a priming dose of 1 mg/kg followed by weekly maintenance doses of 10-30 mg/kg in three dose-escalation cohorts, given until disease progression or lack of clinical benefit. Intravenous rituximab at 375 mg/m2 weekly was started on the second week of the first cycle, and then monthly for cycles 2 through 6.
“Substantial antitumor activity” was seen with this chemotherapy-free regimen in a group of heavily pretreated, largely rituximab-refractory patients, Dr. Advani and her coauthors wrote in their report.
The objective response rate was 50%, including a 36% complete response rate in the intent-to-treat analysis. For DLBCL, the rates of objective and complete responses were 40% and 33%, while for follicular lymphoma, they were 71% and 43%.
The median duration of response was not reached in either disease cohort with a median follow-up of 6.2 months for DLBCL and 8.1 months for follicular lymphoma. Of the 11 patients who responded, 10 (91%) were still in response at the time of data cutoff. “Longer follow-up is needed,” the investigators wrote.
Most adverse events were seen within the first few weeks of treatment and mainly included anemia and infusion-related reactions. The anemia was an expected, on-target effect of 5F9 because of selective clearance of older red cells, which was predictable, transient, and mitigated by the maintenance dosing strategy employed in this phase 1b trial.
“As red cells age, they lose CD47 expression and gain expression of prophagocytic signals, leading to homeostatic clearance,” they wrote.
The activity of 5F9 and rituximab is “synergistic” based on the results of previous, preclinical investigations in models of lymphoma, Dr. Advani and her coauthors added.
A phase 2 trial of 5F9 plus rituximab in relapsed or refractory B-cell non-Hodgkin lymphoma is ongoing, according to their report.
The study was supported by Forty Seven and the Leukemia & Lymphoma Society. Dr. Advani reported disclosures related to Forty Seven, Bristol-Myers Squibb, Pharmacyclics, Seattle Genetics, and Roche/Genentech, among others.
SOURCE: Advani R et al. N Engl J Med. 2018;379:1711-21.
A macrophage-activating immune checkpoint inhibitor, combined with rituximab therapy, was safe and produced durable complete responses in patients with relapsed or refractory non-Hodgkin lymphoma, according to results of a phase 1b study.
Mainly low-grade toxic effects were seen on treatment with Hu5F9-G4 (5F9) and rituximab, which induced responses in more than half of patients, of which more than one-third were complete responses, the study investigators reported.
Most of the responses were ongoing at the time of data cutoff, suggesting durable responses with the combination of rituximab and 5F9 – a humanized monoclonal antibody that blocks CD47, an antiphagocytic or “do not eat me” signal overexpressed by most cancers, Ranjana Advani, MD, of Stanford (Calif.) University, and her coauthors wrote.
“The macrophage-mediated activity of 5F9 plus rituximab may serve as an effective new immunotherapy for stimulating the innate immune system,” Dr. Advani and her colleagues reported in the New England Journal of Medicine.
The study included 22 patients, including 15 with diffuse large B-cell lymphoma (DLBCL) and 7 with follicular lymphoma, who had received a median of four prior therapies. Almost all of the non-Hodgkin lymphomas (21, or 95%) were refractory to rituximab.
All patients received intravenous 5F9 starting with a priming dose of 1 mg/kg followed by weekly maintenance doses of 10-30 mg/kg in three dose-escalation cohorts, given until disease progression or lack of clinical benefit. Intravenous rituximab at 375 mg/m2 weekly was started on the second week of the first cycle, and then monthly for cycles 2 through 6.
“Substantial antitumor activity” was seen with this chemotherapy-free regimen in a group of heavily pretreated, largely rituximab-refractory patients, Dr. Advani and her coauthors wrote in their report.
The objective response rate was 50%, including a 36% complete response rate in the intent-to-treat analysis. For DLBCL, the rates of objective and complete responses were 40% and 33%, while for follicular lymphoma, they were 71% and 43%.
The median duration of response was not reached in either disease cohort with a median follow-up of 6.2 months for DLBCL and 8.1 months for follicular lymphoma. Of the 11 patients who responded, 10 (91%) were still in response at the time of data cutoff. “Longer follow-up is needed,” the investigators wrote.
Most adverse events were seen within the first few weeks of treatment and mainly included anemia and infusion-related reactions. The anemia was an expected, on-target effect of 5F9 because of selective clearance of older red cells, which was predictable, transient, and mitigated by the maintenance dosing strategy employed in this phase 1b trial.
“As red cells age, they lose CD47 expression and gain expression of prophagocytic signals, leading to homeostatic clearance,” they wrote.
The activity of 5F9 and rituximab is “synergistic” based on the results of previous, preclinical investigations in models of lymphoma, Dr. Advani and her coauthors added.
A phase 2 trial of 5F9 plus rituximab in relapsed or refractory B-cell non-Hodgkin lymphoma is ongoing, according to their report.
The study was supported by Forty Seven and the Leukemia & Lymphoma Society. Dr. Advani reported disclosures related to Forty Seven, Bristol-Myers Squibb, Pharmacyclics, Seattle Genetics, and Roche/Genentech, among others.
SOURCE: Advani R et al. N Engl J Med. 2018;379:1711-21.
A macrophage-activating immune checkpoint inhibitor, combined with rituximab therapy, was safe and produced durable complete responses in patients with relapsed or refractory non-Hodgkin lymphoma, according to results of a phase 1b study.
Mainly low-grade toxic effects were seen on treatment with Hu5F9-G4 (5F9) and rituximab, which induced responses in more than half of patients, of which more than one-third were complete responses, the study investigators reported.
Most of the responses were ongoing at the time of data cutoff, suggesting durable responses with the combination of rituximab and 5F9 – a humanized monoclonal antibody that blocks CD47, an antiphagocytic or “do not eat me” signal overexpressed by most cancers, Ranjana Advani, MD, of Stanford (Calif.) University, and her coauthors wrote.
“The macrophage-mediated activity of 5F9 plus rituximab may serve as an effective new immunotherapy for stimulating the innate immune system,” Dr. Advani and her colleagues reported in the New England Journal of Medicine.
The study included 22 patients, including 15 with diffuse large B-cell lymphoma (DLBCL) and 7 with follicular lymphoma, who had received a median of four prior therapies. Almost all of the non-Hodgkin lymphomas (21, or 95%) were refractory to rituximab.
All patients received intravenous 5F9 starting with a priming dose of 1 mg/kg followed by weekly maintenance doses of 10-30 mg/kg in three dose-escalation cohorts, given until disease progression or lack of clinical benefit. Intravenous rituximab at 375 mg/m2 weekly was started on the second week of the first cycle, and then monthly for cycles 2 through 6.
“Substantial antitumor activity” was seen with this chemotherapy-free regimen in a group of heavily pretreated, largely rituximab-refractory patients, Dr. Advani and her coauthors wrote in their report.
The objective response rate was 50%, including a 36% complete response rate in the intent-to-treat analysis. For DLBCL, the rates of objective and complete responses were 40% and 33%, while for follicular lymphoma, they were 71% and 43%.
The median duration of response was not reached in either disease cohort with a median follow-up of 6.2 months for DLBCL and 8.1 months for follicular lymphoma. Of the 11 patients who responded, 10 (91%) were still in response at the time of data cutoff. “Longer follow-up is needed,” the investigators wrote.
Most adverse events were seen within the first few weeks of treatment and mainly included anemia and infusion-related reactions. The anemia was an expected, on-target effect of 5F9 because of selective clearance of older red cells, which was predictable, transient, and mitigated by the maintenance dosing strategy employed in this phase 1b trial.
“As red cells age, they lose CD47 expression and gain expression of prophagocytic signals, leading to homeostatic clearance,” they wrote.
The activity of 5F9 and rituximab is “synergistic” based on the results of previous, preclinical investigations in models of lymphoma, Dr. Advani and her coauthors added.
A phase 2 trial of 5F9 plus rituximab in relapsed or refractory B-cell non-Hodgkin lymphoma is ongoing, according to their report.
The study was supported by Forty Seven and the Leukemia & Lymphoma Society. Dr. Advani reported disclosures related to Forty Seven, Bristol-Myers Squibb, Pharmacyclics, Seattle Genetics, and Roche/Genentech, among others.
SOURCE: Advani R et al. N Engl J Med. 2018;379:1711-21.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point:
Major finding: Rates of overall and complete responses were 50% and 36%, respectively, with most responses ongoing at the time of data cutoff.
Study details: A phase 1b study of 22 patients, including 15 with diffuse large B-cell lymphoma and 7 with follicular lymphoma.
Disclosures: The study was supported by Forty Seven and the Leukemia & Lymphoma Society. Study authors reported disclosures related to Forty Seven, Bristol-Myers Squibb, Pharmacyclics, Seattle Genetics, and Roche/Genentech, among others.
Source: Advani R et al. N Engl J Med. 2018;379:1711-21.
Line complications plague dose-adjusted EPOCH-R in non-Hodgkin lymphoma
Dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (DA-EPOCH-R), which is used to treat several types of aggressive non-Hodgkin lymphomas, is associated with high rates of line-associated complications, a new study suggests.
These findings, published in Clinical Lymphoma, Myeloma & Leukemia, confirm other recent findings that DA-EPOCH-R has a significantly greater rate of complications, compared with that of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) therapy.
The authors note that the use of DA-EPOCH-R is based on data from early phase trials, as well as retrospective data, that support its use as induction chemotherapy in high-grade B-cell lymphoma with MYC, BCL2, and/or BCL6 translocations. But currently, there are no data published from randomized trials that support the use of upfront DA-EPOCH-R therapy.
DA-EPOCH-R is an infusion-based therapy that requires a central venous catheter.
In their study, Rachel J. David, MD, of Wilmot Cancer Institute, Rochester, N.Y., and her colleagues conducted a retrospective study that included all patients treated with DA-EPOCH-R at their institution between March 2011 and July 2016, and also included a concurrent cohort of patients with diffuse large B-cell lymphoma (DLBCL) who were treated with R-CHOP. The goal was to identify the rates and predictors of line-associated complications linked with the use of DA-EPOCH-R therapy in this population.
The patient cohort comprised 43 patients who received DA-EPOCH-R and 44 patients who received RCHOP.
Patients in the DA-EPOCH-R cohort experienced a significantly higher rate of complications (P =.03), compared with the R-CHOP group.
In the DA-EPOCH-R cohort, 17 patients (39.5%) reported at least one LAC, which included venous thromboembolism, chemotherapy extravasation, and line-associated infection, during the study period. Grade 3 toxicity was observed in 41% of these patients.
In contrast, eight patients (18.2%) in the R-CHOP arm experienced at least one complication, with five of the eight patients experiencing grade 3-4 toxicity.
In a univariate analysis, body mass index of 35 kg/m2 and the use of a peripherally inserted central catheter line were both significantly associated with a higher risk of venous thromboembolism (P = .04 and P = .02, respectively).
“For patients undergoing treatment with DA-EPOCH-R in whom the use of [central venous catheters] cannot be avoided, the morbidity of [line-associated complications] should be factored in by the clinician when determining upfront treatment,” the researchers wrote.
They reported having no conflicts of interest.
SOURCE: David RJ et al. Clin Lymphoma Myeloma Leuk. 2018 Aug 29. doi: 10.1016/j.clml.2018.08.014.
Dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (DA-EPOCH-R), which is used to treat several types of aggressive non-Hodgkin lymphomas, is associated with high rates of line-associated complications, a new study suggests.
These findings, published in Clinical Lymphoma, Myeloma & Leukemia, confirm other recent findings that DA-EPOCH-R has a significantly greater rate of complications, compared with that of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) therapy.
The authors note that the use of DA-EPOCH-R is based on data from early phase trials, as well as retrospective data, that support its use as induction chemotherapy in high-grade B-cell lymphoma with MYC, BCL2, and/or BCL6 translocations. But currently, there are no data published from randomized trials that support the use of upfront DA-EPOCH-R therapy.
DA-EPOCH-R is an infusion-based therapy that requires a central venous catheter.
In their study, Rachel J. David, MD, of Wilmot Cancer Institute, Rochester, N.Y., and her colleagues conducted a retrospective study that included all patients treated with DA-EPOCH-R at their institution between March 2011 and July 2016, and also included a concurrent cohort of patients with diffuse large B-cell lymphoma (DLBCL) who were treated with R-CHOP. The goal was to identify the rates and predictors of line-associated complications linked with the use of DA-EPOCH-R therapy in this population.
The patient cohort comprised 43 patients who received DA-EPOCH-R and 44 patients who received RCHOP.
Patients in the DA-EPOCH-R cohort experienced a significantly higher rate of complications (P =.03), compared with the R-CHOP group.
In the DA-EPOCH-R cohort, 17 patients (39.5%) reported at least one LAC, which included venous thromboembolism, chemotherapy extravasation, and line-associated infection, during the study period. Grade 3 toxicity was observed in 41% of these patients.
In contrast, eight patients (18.2%) in the R-CHOP arm experienced at least one complication, with five of the eight patients experiencing grade 3-4 toxicity.
In a univariate analysis, body mass index of 35 kg/m2 and the use of a peripherally inserted central catheter line were both significantly associated with a higher risk of venous thromboembolism (P = .04 and P = .02, respectively).
“For patients undergoing treatment with DA-EPOCH-R in whom the use of [central venous catheters] cannot be avoided, the morbidity of [line-associated complications] should be factored in by the clinician when determining upfront treatment,” the researchers wrote.
They reported having no conflicts of interest.
SOURCE: David RJ et al. Clin Lymphoma Myeloma Leuk. 2018 Aug 29. doi: 10.1016/j.clml.2018.08.014.
Dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (DA-EPOCH-R), which is used to treat several types of aggressive non-Hodgkin lymphomas, is associated with high rates of line-associated complications, a new study suggests.
These findings, published in Clinical Lymphoma, Myeloma & Leukemia, confirm other recent findings that DA-EPOCH-R has a significantly greater rate of complications, compared with that of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) therapy.
The authors note that the use of DA-EPOCH-R is based on data from early phase trials, as well as retrospective data, that support its use as induction chemotherapy in high-grade B-cell lymphoma with MYC, BCL2, and/or BCL6 translocations. But currently, there are no data published from randomized trials that support the use of upfront DA-EPOCH-R therapy.
DA-EPOCH-R is an infusion-based therapy that requires a central venous catheter.
In their study, Rachel J. David, MD, of Wilmot Cancer Institute, Rochester, N.Y., and her colleagues conducted a retrospective study that included all patients treated with DA-EPOCH-R at their institution between March 2011 and July 2016, and also included a concurrent cohort of patients with diffuse large B-cell lymphoma (DLBCL) who were treated with R-CHOP. The goal was to identify the rates and predictors of line-associated complications linked with the use of DA-EPOCH-R therapy in this population.
The patient cohort comprised 43 patients who received DA-EPOCH-R and 44 patients who received RCHOP.
Patients in the DA-EPOCH-R cohort experienced a significantly higher rate of complications (P =.03), compared with the R-CHOP group.
In the DA-EPOCH-R cohort, 17 patients (39.5%) reported at least one LAC, which included venous thromboembolism, chemotherapy extravasation, and line-associated infection, during the study period. Grade 3 toxicity was observed in 41% of these patients.
In contrast, eight patients (18.2%) in the R-CHOP arm experienced at least one complication, with five of the eight patients experiencing grade 3-4 toxicity.
In a univariate analysis, body mass index of 35 kg/m2 and the use of a peripherally inserted central catheter line were both significantly associated with a higher risk of venous thromboembolism (P = .04 and P = .02, respectively).
“For patients undergoing treatment with DA-EPOCH-R in whom the use of [central venous catheters] cannot be avoided, the morbidity of [line-associated complications] should be factored in by the clinician when determining upfront treatment,” the researchers wrote.
They reported having no conflicts of interest.
SOURCE: David RJ et al. Clin Lymphoma Myeloma Leuk. 2018 Aug 29. doi: 10.1016/j.clml.2018.08.014.
FROM CLINICAL LYMPHOMA, MYELOMA & LEUKEMIA
Key clinical point:
Major finding: In all, 17 dose-adjusted R-EPOCH patients (39.5%) experienced at least one line-associated complication, versus 8 patients (18.2%) in the R-CHOP group.
Study details: A retrospective single-institution study with 87 patients.
Disclosures: The researchers reported having no conflicts of interest.
Source: David RJ et al. Clin Lymphoma Myeloma Leuk. 2018 Aug 29. doi: 10.1016/j.clml.2018.08.014.
First-line bortezomib prolongs survival in MCL
Bortezomib in combination with rituximab plus chemotherapy significantly improved overall survival in transplant-ineligible patients with newly diagnosed mantle cell lymphoma (MCL), compared with standard treatment, according to final results from the international, phase 3 LYM-3002 trial.
After a median follow-up period of 82.0 months, median overall survival was 90.7 months among participants who were given first-line bortezomib in addition to rituximab plus cyclophosphamide, doxorubicin, and prednisone (VR-CAP) versus 55.7 months in the control arm, where patients were given rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), for a hazard ratio of 0.66 (95% confidence interval, 0.51-0.85; P = .001).
Tadeusz Robak, MD, of the Medical University of Lodz in Poland, and his colleagues also reported that patients in the bortezomib arm experienced two novel adverse effects, which were different from findings reported in the primary analysis. Each case was classified as grade 4; there was one case of gastric cancer and one case of lung adenocarcinoma.
The findings were reported in the Lancet Oncology.
Among 268 patients in the follow-up analysis set, the median age was 66 years and 31% were classified as high risk based on the MCL-specific International Prognostic Index (MIPI). For those considered high risk, no significant difference was noted when comparing the two groups on the basis of overall survival.
“When analyzed according to MIPI risk category, VR-CAP was associated with significantly improved overall survival, compared with R-CHOP in the low-risk and intermediate-risk categories, but not in the high-risk category,” the investigators wrote.
The authors acknowledged a key limitation of the study was that rituximab was not given as a maintenance therapy since it was not considered standard of care at the time of study initiation.
Moving forward, Dr. Robak and his colleagues recommended that bortezomib be investigated in combination with newer targeted therapies in order to establish best practice for treating MCL.
The study was sponsored by Janssen Pharmaceuticals. The authors reported financial ties to Janssen, Celgene, Ipsen Biopharmaceuticals, Johnson & Johnson, Novartis, and others.
SOURCE: Robak T et al. Lancet Oncol. 2018 Oct 19. doi: 10.1016/S1470-2045(18)30685-5.
The proteasome inhibitor, bortezomib, represents a “substantial advance” for the treatment of newly diagnosed mantle cell lymphoma, according to Simon Rule, MD.
In an accompanying commentary, he stated that bortezomib-based VR-CAP (rituximab plus cyclophosphamide, doxorubicin, and prednisone) showed a clear survival benefit in the LYM-3002 trial, compared with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP). However, in order to use this combination in elderly patients, the administration method must be considered. Additionally, it makes sense to routinely use rituximab maintenance.
While the final analysis of the LYM-3002 trial is positive, there are caveats to consider before changing practice, particularly for elderly patients. First, the study had a somewhat younger population and fewer high-risk patients, compared with the only similar study of R-CHOP regimen in an elderly population. The bortezomib plus VR-CAP combination also had significant toxicity that could limit its widespread use in elderly patients.
Dr. Rule also noted that, internationally, bendamustine-based therapy is increasingly being chosen over R-CHOP for older patients with mantle cell lymphoma.
“Whether VR-CAP or the combination of bortezomib and bendamustine-based regimens will be the optimal approach has yet to be established. However, if R-CHOP is being considered, then the long-term survival results reported by Robak and colleagues strongly support the use of VR-CAP as an alternative,” Dr. Rule wrote.
Dr. Rule is with the University of Plymouth (England). These comments are adapted from his commentary (Lancet Oncol. 2018 Oct 19. doi: 10.1016/S1470-2045[18]30743-5). Dr. Rule reported receiving grants and personal fees from Janssen Pharmaceuticals.
The proteasome inhibitor, bortezomib, represents a “substantial advance” for the treatment of newly diagnosed mantle cell lymphoma, according to Simon Rule, MD.
In an accompanying commentary, he stated that bortezomib-based VR-CAP (rituximab plus cyclophosphamide, doxorubicin, and prednisone) showed a clear survival benefit in the LYM-3002 trial, compared with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP). However, in order to use this combination in elderly patients, the administration method must be considered. Additionally, it makes sense to routinely use rituximab maintenance.
While the final analysis of the LYM-3002 trial is positive, there are caveats to consider before changing practice, particularly for elderly patients. First, the study had a somewhat younger population and fewer high-risk patients, compared with the only similar study of R-CHOP regimen in an elderly population. The bortezomib plus VR-CAP combination also had significant toxicity that could limit its widespread use in elderly patients.
Dr. Rule also noted that, internationally, bendamustine-based therapy is increasingly being chosen over R-CHOP for older patients with mantle cell lymphoma.
“Whether VR-CAP or the combination of bortezomib and bendamustine-based regimens will be the optimal approach has yet to be established. However, if R-CHOP is being considered, then the long-term survival results reported by Robak and colleagues strongly support the use of VR-CAP as an alternative,” Dr. Rule wrote.
Dr. Rule is with the University of Plymouth (England). These comments are adapted from his commentary (Lancet Oncol. 2018 Oct 19. doi: 10.1016/S1470-2045[18]30743-5). Dr. Rule reported receiving grants and personal fees from Janssen Pharmaceuticals.
The proteasome inhibitor, bortezomib, represents a “substantial advance” for the treatment of newly diagnosed mantle cell lymphoma, according to Simon Rule, MD.
In an accompanying commentary, he stated that bortezomib-based VR-CAP (rituximab plus cyclophosphamide, doxorubicin, and prednisone) showed a clear survival benefit in the LYM-3002 trial, compared with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP). However, in order to use this combination in elderly patients, the administration method must be considered. Additionally, it makes sense to routinely use rituximab maintenance.
While the final analysis of the LYM-3002 trial is positive, there are caveats to consider before changing practice, particularly for elderly patients. First, the study had a somewhat younger population and fewer high-risk patients, compared with the only similar study of R-CHOP regimen in an elderly population. The bortezomib plus VR-CAP combination also had significant toxicity that could limit its widespread use in elderly patients.
Dr. Rule also noted that, internationally, bendamustine-based therapy is increasingly being chosen over R-CHOP for older patients with mantle cell lymphoma.
“Whether VR-CAP or the combination of bortezomib and bendamustine-based regimens will be the optimal approach has yet to be established. However, if R-CHOP is being considered, then the long-term survival results reported by Robak and colleagues strongly support the use of VR-CAP as an alternative,” Dr. Rule wrote.
Dr. Rule is with the University of Plymouth (England). These comments are adapted from his commentary (Lancet Oncol. 2018 Oct 19. doi: 10.1016/S1470-2045[18]30743-5). Dr. Rule reported receiving grants and personal fees from Janssen Pharmaceuticals.
Bortezomib in combination with rituximab plus chemotherapy significantly improved overall survival in transplant-ineligible patients with newly diagnosed mantle cell lymphoma (MCL), compared with standard treatment, according to final results from the international, phase 3 LYM-3002 trial.
After a median follow-up period of 82.0 months, median overall survival was 90.7 months among participants who were given first-line bortezomib in addition to rituximab plus cyclophosphamide, doxorubicin, and prednisone (VR-CAP) versus 55.7 months in the control arm, where patients were given rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), for a hazard ratio of 0.66 (95% confidence interval, 0.51-0.85; P = .001).
Tadeusz Robak, MD, of the Medical University of Lodz in Poland, and his colleagues also reported that patients in the bortezomib arm experienced two novel adverse effects, which were different from findings reported in the primary analysis. Each case was classified as grade 4; there was one case of gastric cancer and one case of lung adenocarcinoma.
The findings were reported in the Lancet Oncology.
Among 268 patients in the follow-up analysis set, the median age was 66 years and 31% were classified as high risk based on the MCL-specific International Prognostic Index (MIPI). For those considered high risk, no significant difference was noted when comparing the two groups on the basis of overall survival.
“When analyzed according to MIPI risk category, VR-CAP was associated with significantly improved overall survival, compared with R-CHOP in the low-risk and intermediate-risk categories, but not in the high-risk category,” the investigators wrote.
The authors acknowledged a key limitation of the study was that rituximab was not given as a maintenance therapy since it was not considered standard of care at the time of study initiation.
Moving forward, Dr. Robak and his colleagues recommended that bortezomib be investigated in combination with newer targeted therapies in order to establish best practice for treating MCL.
The study was sponsored by Janssen Pharmaceuticals. The authors reported financial ties to Janssen, Celgene, Ipsen Biopharmaceuticals, Johnson & Johnson, Novartis, and others.
SOURCE: Robak T et al. Lancet Oncol. 2018 Oct 19. doi: 10.1016/S1470-2045(18)30685-5.
Bortezomib in combination with rituximab plus chemotherapy significantly improved overall survival in transplant-ineligible patients with newly diagnosed mantle cell lymphoma (MCL), compared with standard treatment, according to final results from the international, phase 3 LYM-3002 trial.
After a median follow-up period of 82.0 months, median overall survival was 90.7 months among participants who were given first-line bortezomib in addition to rituximab plus cyclophosphamide, doxorubicin, and prednisone (VR-CAP) versus 55.7 months in the control arm, where patients were given rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), for a hazard ratio of 0.66 (95% confidence interval, 0.51-0.85; P = .001).
Tadeusz Robak, MD, of the Medical University of Lodz in Poland, and his colleagues also reported that patients in the bortezomib arm experienced two novel adverse effects, which were different from findings reported in the primary analysis. Each case was classified as grade 4; there was one case of gastric cancer and one case of lung adenocarcinoma.
The findings were reported in the Lancet Oncology.
Among 268 patients in the follow-up analysis set, the median age was 66 years and 31% were classified as high risk based on the MCL-specific International Prognostic Index (MIPI). For those considered high risk, no significant difference was noted when comparing the two groups on the basis of overall survival.
“When analyzed according to MIPI risk category, VR-CAP was associated with significantly improved overall survival, compared with R-CHOP in the low-risk and intermediate-risk categories, but not in the high-risk category,” the investigators wrote.
The authors acknowledged a key limitation of the study was that rituximab was not given as a maintenance therapy since it was not considered standard of care at the time of study initiation.
Moving forward, Dr. Robak and his colleagues recommended that bortezomib be investigated in combination with newer targeted therapies in order to establish best practice for treating MCL.
The study was sponsored by Janssen Pharmaceuticals. The authors reported financial ties to Janssen, Celgene, Ipsen Biopharmaceuticals, Johnson & Johnson, Novartis, and others.
SOURCE: Robak T et al. Lancet Oncol. 2018 Oct 19. doi: 10.1016/S1470-2045(18)30685-5.
FROM THE LANCET ONCOLOGY
Key clinical point:
Major finding: Median overall survival was 90.7 months in the intervention arm (bortezomib in addition to rituximab plus chemotherapy) versus 55.7 months in the control arm (hazard ratio, 0.66; 95% confidence interval, 0.51-0.85; P = .001).
Study details: LYM-3002 was a phase 3, randomized, open-label study of 487 transplant-ineligible patients with untreated mantle cell lymphoma.
Disclosures: The study was sponsored by Janssen Pharmaceuticals. The authors reported financial ties with Janssen, Celgene, Ipsen Biopharmaceuticals, Johnson & Johnson, Novartis, and others.
Source: Robak T et al. Lancet Oncol. 2018 Oct 19. doi: 10.1016/S1470-2045(18)30685-5.
Older age predicts mortality after alloHCT in NHL, but not relapse
Elderly patients with non-Hodgkin lymphoma (NHL) are more likely to die, but not relapse, within 1 year of allogeneic hematopoietic cell transplantation (alloHCT), compared with younger or middle-age patients, according to investigators.
Comorbidities also increased risks of nonrelapse mortality (NRM) at 1 year, but to a lesser extent than that of elderly status, reported lead author Charalampia Kyriakou, MD, PhD, of the department of haematology at University College London Hospital and London North West University Healthcare NHS Trust, and her colleagues.
“Although alloHCT is feasible and effective in very old patients, the increased NRM risk must be taken into account when assessing the indication for alloHCT for NHL in this age group,” the investigators wrote in Biology of Blood and Marrow Transplantation.
This decision is becoming more common, they noted. “With the advent of reduced-intensity conditioning (RIC) strategies and other improvements in transplantation technology, alloHCT is being increasingly considered in elderly patients with [relapsed and refractory] NHL.”
The retrospective study analyzed 3,919 patients with NHL who underwent alloHCT between 2003 and 2013. Patients were sorted into three age groups: young (18-50 years), middle age (51-65 years), or elderly (66-77 years).
Disease types also were reported: 1,461 patients had follicular lymphoma (FL; 37%), 1,192 had diffuse large B cell lymphoma (DLBCL; 30%), 823 had mantle cell lymphoma (MCL; 21%), and 443 had peripheral T cell lymphoma (PTCL; 11%).
At the time of alloHCT, about 85% of patients were chemosensitive, with the remainder being chemorefractory. The age groups had similar patient characteristics, with exceptions noted for unrelated donors, MCL, and RIC, which became increasingly overrepresented with age.
The results showed that NRM at 1 year was 13% for young patients, 20% for middle-age patients, and 33% for elderly patients (P less than .001). Overall survival at 3 years followed an inverse trend, decreasing with age from 60% in young patients to 54% in middle-age patients, before dropping more dramatically to 38% in the elderly (P less than .001).
In contrast to these significant associations between age and survival, relapse risk at 3 years remained relatively consistent, with young patients at 30%, middle-age patients at 31%, and elderly patients at 28% (P = .355).
The investigators noted that the risk of NRM increased most dramatically between middle age and old age, with less significant differences between the middle-age and young groups. They suggested that “age per se should have a limited impact on the indication for alloHCT for NHL in patients up to age 65 years.”
The increased risk with elderly status could not be fully explained by comorbidities, although these were more common in elderly patients. After analyzing information from a subset of patients, the investigators concluded that “the presence of comorbidities is a significant risk factor for NRM and survival, but this does not fully explain the outcome disadvantages in our [elderly] group.” Therefore, age remains an independent risk factor.
“The information provided in this cohort of patients with NHL, the largest reported to date, is useful and relevant, especially in the era of evolving therapies,” the investigators wrote. They added that the information is “even more relevant now with the availability of treatment with ... chimeric antigen receptor (CAR) T cells ... after relapse post-alloHCT.”
The investigators reported having no financial disclosures.
SOURCE: Kyriakou C et al. Biol Blood Marrow Transplant. 2018 Sep 13. doi: 10.1016/j.bbmt.2018.08.025.
Elderly patients with non-Hodgkin lymphoma (NHL) are more likely to die, but not relapse, within 1 year of allogeneic hematopoietic cell transplantation (alloHCT), compared with younger or middle-age patients, according to investigators.
Comorbidities also increased risks of nonrelapse mortality (NRM) at 1 year, but to a lesser extent than that of elderly status, reported lead author Charalampia Kyriakou, MD, PhD, of the department of haematology at University College London Hospital and London North West University Healthcare NHS Trust, and her colleagues.
“Although alloHCT is feasible and effective in very old patients, the increased NRM risk must be taken into account when assessing the indication for alloHCT for NHL in this age group,” the investigators wrote in Biology of Blood and Marrow Transplantation.
This decision is becoming more common, they noted. “With the advent of reduced-intensity conditioning (RIC) strategies and other improvements in transplantation technology, alloHCT is being increasingly considered in elderly patients with [relapsed and refractory] NHL.”
The retrospective study analyzed 3,919 patients with NHL who underwent alloHCT between 2003 and 2013. Patients were sorted into three age groups: young (18-50 years), middle age (51-65 years), or elderly (66-77 years).
Disease types also were reported: 1,461 patients had follicular lymphoma (FL; 37%), 1,192 had diffuse large B cell lymphoma (DLBCL; 30%), 823 had mantle cell lymphoma (MCL; 21%), and 443 had peripheral T cell lymphoma (PTCL; 11%).
At the time of alloHCT, about 85% of patients were chemosensitive, with the remainder being chemorefractory. The age groups had similar patient characteristics, with exceptions noted for unrelated donors, MCL, and RIC, which became increasingly overrepresented with age.
The results showed that NRM at 1 year was 13% for young patients, 20% for middle-age patients, and 33% for elderly patients (P less than .001). Overall survival at 3 years followed an inverse trend, decreasing with age from 60% in young patients to 54% in middle-age patients, before dropping more dramatically to 38% in the elderly (P less than .001).
In contrast to these significant associations between age and survival, relapse risk at 3 years remained relatively consistent, with young patients at 30%, middle-age patients at 31%, and elderly patients at 28% (P = .355).
The investigators noted that the risk of NRM increased most dramatically between middle age and old age, with less significant differences between the middle-age and young groups. They suggested that “age per se should have a limited impact on the indication for alloHCT for NHL in patients up to age 65 years.”
The increased risk with elderly status could not be fully explained by comorbidities, although these were more common in elderly patients. After analyzing information from a subset of patients, the investigators concluded that “the presence of comorbidities is a significant risk factor for NRM and survival, but this does not fully explain the outcome disadvantages in our [elderly] group.” Therefore, age remains an independent risk factor.
“The information provided in this cohort of patients with NHL, the largest reported to date, is useful and relevant, especially in the era of evolving therapies,” the investigators wrote. They added that the information is “even more relevant now with the availability of treatment with ... chimeric antigen receptor (CAR) T cells ... after relapse post-alloHCT.”
The investigators reported having no financial disclosures.
SOURCE: Kyriakou C et al. Biol Blood Marrow Transplant. 2018 Sep 13. doi: 10.1016/j.bbmt.2018.08.025.
Elderly patients with non-Hodgkin lymphoma (NHL) are more likely to die, but not relapse, within 1 year of allogeneic hematopoietic cell transplantation (alloHCT), compared with younger or middle-age patients, according to investigators.
Comorbidities also increased risks of nonrelapse mortality (NRM) at 1 year, but to a lesser extent than that of elderly status, reported lead author Charalampia Kyriakou, MD, PhD, of the department of haematology at University College London Hospital and London North West University Healthcare NHS Trust, and her colleagues.
“Although alloHCT is feasible and effective in very old patients, the increased NRM risk must be taken into account when assessing the indication for alloHCT for NHL in this age group,” the investigators wrote in Biology of Blood and Marrow Transplantation.
This decision is becoming more common, they noted. “With the advent of reduced-intensity conditioning (RIC) strategies and other improvements in transplantation technology, alloHCT is being increasingly considered in elderly patients with [relapsed and refractory] NHL.”
The retrospective study analyzed 3,919 patients with NHL who underwent alloHCT between 2003 and 2013. Patients were sorted into three age groups: young (18-50 years), middle age (51-65 years), or elderly (66-77 years).
Disease types also were reported: 1,461 patients had follicular lymphoma (FL; 37%), 1,192 had diffuse large B cell lymphoma (DLBCL; 30%), 823 had mantle cell lymphoma (MCL; 21%), and 443 had peripheral T cell lymphoma (PTCL; 11%).
At the time of alloHCT, about 85% of patients were chemosensitive, with the remainder being chemorefractory. The age groups had similar patient characteristics, with exceptions noted for unrelated donors, MCL, and RIC, which became increasingly overrepresented with age.
The results showed that NRM at 1 year was 13% for young patients, 20% for middle-age patients, and 33% for elderly patients (P less than .001). Overall survival at 3 years followed an inverse trend, decreasing with age from 60% in young patients to 54% in middle-age patients, before dropping more dramatically to 38% in the elderly (P less than .001).
In contrast to these significant associations between age and survival, relapse risk at 3 years remained relatively consistent, with young patients at 30%, middle-age patients at 31%, and elderly patients at 28% (P = .355).
The investigators noted that the risk of NRM increased most dramatically between middle age and old age, with less significant differences between the middle-age and young groups. They suggested that “age per se should have a limited impact on the indication for alloHCT for NHL in patients up to age 65 years.”
The increased risk with elderly status could not be fully explained by comorbidities, although these were more common in elderly patients. After analyzing information from a subset of patients, the investigators concluded that “the presence of comorbidities is a significant risk factor for NRM and survival, but this does not fully explain the outcome disadvantages in our [elderly] group.” Therefore, age remains an independent risk factor.
“The information provided in this cohort of patients with NHL, the largest reported to date, is useful and relevant, especially in the era of evolving therapies,” the investigators wrote. They added that the information is “even more relevant now with the availability of treatment with ... chimeric antigen receptor (CAR) T cells ... after relapse post-alloHCT.”
The investigators reported having no financial disclosures.
SOURCE: Kyriakou C et al. Biol Blood Marrow Transplant. 2018 Sep 13. doi: 10.1016/j.bbmt.2018.08.025.
FROM BIOLOGY OF BLOOD AND MARROW TRANSPLANTATION
Key clinical point:
Major finding: One-year nonrelapse mortality (NRM) was 13% for young patients, 20% for middle-age patients, and 33% for elderly patients (P less than .001).
Study details: A retrospective analysis of 3,919 patients with NHL who underwent alloHCT between 2003 and 2013.
Disclosures: The researchers reported having no financial disclosures.
Source: Kyriakou C et al. Biol Blood Marrow Transplant. 2018 Sep 13. doi: 10.1016/j.bbmt.2018.08.025.
When to choose stem cell transplant in PTCL
DUBROVNIK, CROATIA – , according to one expert.
The success of HSCT varies according to the subtype of PTCL and the type of transplant, Ali Bazarbachi, MD, PhD, of the American University of Beirut, Lebanon, said at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
For example, autologous (auto) HSCT given as frontline consolidation can be considered the standard of care for PTCL–not otherwise specified (NOS), angioimmunoblastic T-cell lymphoma (AITL), and certain patients with anaplastic large-cell lymphoma (ALCL), according to Dr. Bazarbachi.
On the other hand, auto-HSCT should never be used in patients with adult T-cell leukemia/lymphoma (ATLL).
Both auto-HSCT and allogeneic (allo) HSCT are options for patients with nonlocalized, extranodal natural killer T-cell lymphoma (ENKTL), nasal type, but only at certain times.
State of PTCL treatment
Patients with newly diagnosed PTCL are no longer treated like patients with B-cell lymphoma, but treatment outcomes in PTCL still leave a lot to be desired, Dr. Bazarbachi said.
He noted that, with any of the chemotherapy regimens used, typically, about a third of patients are primary refractory, a third relapse, and a quarter are cured. Only two forms of PTCL are frequently curable – localized ENKTL and anaplastic lymphoma kinase–positive (ALK-positive) ALCL.
Current treatment strategies for PTCL do include HSCT, but recommendations vary. Dr. Bazarbachi made the following recommendations, supported by evidence from clinical trials.
PTCL-NOS, AITL, and ALCL
For patients with PTCL-NOS, AITL, or ALK-negative, non-DUSP22 ALCL, auto-HSCT as frontline consolidation can be considered the standard of care in patients who responded to induction, Dr. Bazarbachi said.
In a study published in 2012, high-dose chemotherapy and auto-HSCT as consolidation improved 5-year overall survival – compared with previous results with CHOP – in patients with ALK-negative ALCL, AITL, PTCL-NOS, and enteropathy-associated T-cell lymphoma (J Clin Oncol. 2012 Sep 1;30[25]:3093-9; ISRN Hematol. 2011 Jun 16. doi: 10.5402/2011/623924).
Allo-HSCT may also be an option for frontline consolidation in patients with PTCL-NOS, AITL, or ALK-negative, non-DUSP22 ALCL, according to Dr. Bazarbachi.
“Allo-transplant is not dead in this indication,” he said. “But it should be either part of a clinical trial or [given] to some selected patients – those with persistent bone marrow involvement, very young patients, or patients with primary refractory disease.”
Results from the COMPLETE study showed improved survival in patients who received consolidation with auto- or allo-HSCT, compared with patients who did not receive a transplant (Blood. 2017;130:342).
COMPLETE patients with AITL or PTCL-NOS had improvements in progression-free and overall survival with HSCT. The survival advantage was “less evident” in patients with ALCL, the researchers said, but this trial included both ALK-negative and ALK-positive patients.
Allo- and auto-HSCT can be options after relapse in patients with PTCL-NOS, AITL, or ALK-negative, non-DUSP22 ALCL, Dr. Bazarbachi said.
However, chemosensitive patients who have relapsed should receive auto-HSCT only if they did not receive it frontline. Patients who have already undergone auto-HSCT can receive allo-HSCT, Dr. Bazarbachi said.
He added that refractory patients should not undergo auto-HSCT and should receive allo-HSCT only within the context of a clinical trial.
ATLL
ATLL has a dismal prognosis, but allo-HSCT as frontline consolidation is potentially curative, Dr. Bazarbachi said. It is most effective in patients who have achieved a complete or partial response to induction (Blood. 2012 Aug 23;120[8]:1734-41).
However, allo-HSCT should not be given as consolidation to ATLL patients who have received prior mogamulizumab. These patients have an increased risk of morbidity and mortality if they undergo allo-HSCT.
Also, allo-HSCT should not be given to refractory ATLL patients, although it may be an option for relapsed patients.
Dr. Bazarbachi stressed that ATLL patients should not receive auto-HSCT at any time, as frontline consolidation, after relapse, or if they have refractory disease.
Auto-HSCT “does not work in this disease,” he said. In a study published in 2014, all four ATLL patients who underwent auto-HSCT “rapidly” died (Bone Marrow Transplant. 2014 Oct;49[10]:1266-8).
ENKTL
Dr. Bazarbachi said frontline consolidation with auto-HSCT should be considered the standard of care for patients with non-localized ENKTL, nasal type.
Auto-HSCT has been shown to improve survival in these patients, and it is most effective when patients have achieved a complete response to induction (Biol Blood Marrow Transplant. 2008 Dec;14[12]:1356-64).
Allo-HSCT also is an option for frontline consolidation in patients with nonlocalized ENKTL, nasal type, Dr. Bazarbachi said.
He added that chemosensitive patients who have relapsed can receive allo-HSCT, but they should receive auto-HSCT only if they did not receive it in the frontline setting. Both types of transplant should take place when patients are in complete remission.
Patients with refractory, nonlocalized ENKTL, nasal type, should not receive auto-HSCT, but allo-HSCT is an option, Dr. Bazarbachi said.
Dr. Bazarbachi did not declare any conflicts of interest.
The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Associates, which is owned by the parent company of this news organization.
DUBROVNIK, CROATIA – , according to one expert.
The success of HSCT varies according to the subtype of PTCL and the type of transplant, Ali Bazarbachi, MD, PhD, of the American University of Beirut, Lebanon, said at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
For example, autologous (auto) HSCT given as frontline consolidation can be considered the standard of care for PTCL–not otherwise specified (NOS), angioimmunoblastic T-cell lymphoma (AITL), and certain patients with anaplastic large-cell lymphoma (ALCL), according to Dr. Bazarbachi.
On the other hand, auto-HSCT should never be used in patients with adult T-cell leukemia/lymphoma (ATLL).
Both auto-HSCT and allogeneic (allo) HSCT are options for patients with nonlocalized, extranodal natural killer T-cell lymphoma (ENKTL), nasal type, but only at certain times.
State of PTCL treatment
Patients with newly diagnosed PTCL are no longer treated like patients with B-cell lymphoma, but treatment outcomes in PTCL still leave a lot to be desired, Dr. Bazarbachi said.
He noted that, with any of the chemotherapy regimens used, typically, about a third of patients are primary refractory, a third relapse, and a quarter are cured. Only two forms of PTCL are frequently curable – localized ENKTL and anaplastic lymphoma kinase–positive (ALK-positive) ALCL.
Current treatment strategies for PTCL do include HSCT, but recommendations vary. Dr. Bazarbachi made the following recommendations, supported by evidence from clinical trials.
PTCL-NOS, AITL, and ALCL
For patients with PTCL-NOS, AITL, or ALK-negative, non-DUSP22 ALCL, auto-HSCT as frontline consolidation can be considered the standard of care in patients who responded to induction, Dr. Bazarbachi said.
In a study published in 2012, high-dose chemotherapy and auto-HSCT as consolidation improved 5-year overall survival – compared with previous results with CHOP – in patients with ALK-negative ALCL, AITL, PTCL-NOS, and enteropathy-associated T-cell lymphoma (J Clin Oncol. 2012 Sep 1;30[25]:3093-9; ISRN Hematol. 2011 Jun 16. doi: 10.5402/2011/623924).
Allo-HSCT may also be an option for frontline consolidation in patients with PTCL-NOS, AITL, or ALK-negative, non-DUSP22 ALCL, according to Dr. Bazarbachi.
“Allo-transplant is not dead in this indication,” he said. “But it should be either part of a clinical trial or [given] to some selected patients – those with persistent bone marrow involvement, very young patients, or patients with primary refractory disease.”
Results from the COMPLETE study showed improved survival in patients who received consolidation with auto- or allo-HSCT, compared with patients who did not receive a transplant (Blood. 2017;130:342).
COMPLETE patients with AITL or PTCL-NOS had improvements in progression-free and overall survival with HSCT. The survival advantage was “less evident” in patients with ALCL, the researchers said, but this trial included both ALK-negative and ALK-positive patients.
Allo- and auto-HSCT can be options after relapse in patients with PTCL-NOS, AITL, or ALK-negative, non-DUSP22 ALCL, Dr. Bazarbachi said.
However, chemosensitive patients who have relapsed should receive auto-HSCT only if they did not receive it frontline. Patients who have already undergone auto-HSCT can receive allo-HSCT, Dr. Bazarbachi said.
He added that refractory patients should not undergo auto-HSCT and should receive allo-HSCT only within the context of a clinical trial.
ATLL
ATLL has a dismal prognosis, but allo-HSCT as frontline consolidation is potentially curative, Dr. Bazarbachi said. It is most effective in patients who have achieved a complete or partial response to induction (Blood. 2012 Aug 23;120[8]:1734-41).
However, allo-HSCT should not be given as consolidation to ATLL patients who have received prior mogamulizumab. These patients have an increased risk of morbidity and mortality if they undergo allo-HSCT.
Also, allo-HSCT should not be given to refractory ATLL patients, although it may be an option for relapsed patients.
Dr. Bazarbachi stressed that ATLL patients should not receive auto-HSCT at any time, as frontline consolidation, after relapse, or if they have refractory disease.
Auto-HSCT “does not work in this disease,” he said. In a study published in 2014, all four ATLL patients who underwent auto-HSCT “rapidly” died (Bone Marrow Transplant. 2014 Oct;49[10]:1266-8).
ENKTL
Dr. Bazarbachi said frontline consolidation with auto-HSCT should be considered the standard of care for patients with non-localized ENKTL, nasal type.
Auto-HSCT has been shown to improve survival in these patients, and it is most effective when patients have achieved a complete response to induction (Biol Blood Marrow Transplant. 2008 Dec;14[12]:1356-64).
Allo-HSCT also is an option for frontline consolidation in patients with nonlocalized ENKTL, nasal type, Dr. Bazarbachi said.
He added that chemosensitive patients who have relapsed can receive allo-HSCT, but they should receive auto-HSCT only if they did not receive it in the frontline setting. Both types of transplant should take place when patients are in complete remission.
Patients with refractory, nonlocalized ENKTL, nasal type, should not receive auto-HSCT, but allo-HSCT is an option, Dr. Bazarbachi said.
Dr. Bazarbachi did not declare any conflicts of interest.
The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Associates, which is owned by the parent company of this news organization.
DUBROVNIK, CROATIA – , according to one expert.
The success of HSCT varies according to the subtype of PTCL and the type of transplant, Ali Bazarbachi, MD, PhD, of the American University of Beirut, Lebanon, said at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
For example, autologous (auto) HSCT given as frontline consolidation can be considered the standard of care for PTCL–not otherwise specified (NOS), angioimmunoblastic T-cell lymphoma (AITL), and certain patients with anaplastic large-cell lymphoma (ALCL), according to Dr. Bazarbachi.
On the other hand, auto-HSCT should never be used in patients with adult T-cell leukemia/lymphoma (ATLL).
Both auto-HSCT and allogeneic (allo) HSCT are options for patients with nonlocalized, extranodal natural killer T-cell lymphoma (ENKTL), nasal type, but only at certain times.
State of PTCL treatment
Patients with newly diagnosed PTCL are no longer treated like patients with B-cell lymphoma, but treatment outcomes in PTCL still leave a lot to be desired, Dr. Bazarbachi said.
He noted that, with any of the chemotherapy regimens used, typically, about a third of patients are primary refractory, a third relapse, and a quarter are cured. Only two forms of PTCL are frequently curable – localized ENKTL and anaplastic lymphoma kinase–positive (ALK-positive) ALCL.
Current treatment strategies for PTCL do include HSCT, but recommendations vary. Dr. Bazarbachi made the following recommendations, supported by evidence from clinical trials.
PTCL-NOS, AITL, and ALCL
For patients with PTCL-NOS, AITL, or ALK-negative, non-DUSP22 ALCL, auto-HSCT as frontline consolidation can be considered the standard of care in patients who responded to induction, Dr. Bazarbachi said.
In a study published in 2012, high-dose chemotherapy and auto-HSCT as consolidation improved 5-year overall survival – compared with previous results with CHOP – in patients with ALK-negative ALCL, AITL, PTCL-NOS, and enteropathy-associated T-cell lymphoma (J Clin Oncol. 2012 Sep 1;30[25]:3093-9; ISRN Hematol. 2011 Jun 16. doi: 10.5402/2011/623924).
Allo-HSCT may also be an option for frontline consolidation in patients with PTCL-NOS, AITL, or ALK-negative, non-DUSP22 ALCL, according to Dr. Bazarbachi.
“Allo-transplant is not dead in this indication,” he said. “But it should be either part of a clinical trial or [given] to some selected patients – those with persistent bone marrow involvement, very young patients, or patients with primary refractory disease.”
Results from the COMPLETE study showed improved survival in patients who received consolidation with auto- or allo-HSCT, compared with patients who did not receive a transplant (Blood. 2017;130:342).
COMPLETE patients with AITL or PTCL-NOS had improvements in progression-free and overall survival with HSCT. The survival advantage was “less evident” in patients with ALCL, the researchers said, but this trial included both ALK-negative and ALK-positive patients.
Allo- and auto-HSCT can be options after relapse in patients with PTCL-NOS, AITL, or ALK-negative, non-DUSP22 ALCL, Dr. Bazarbachi said.
However, chemosensitive patients who have relapsed should receive auto-HSCT only if they did not receive it frontline. Patients who have already undergone auto-HSCT can receive allo-HSCT, Dr. Bazarbachi said.
He added that refractory patients should not undergo auto-HSCT and should receive allo-HSCT only within the context of a clinical trial.
ATLL
ATLL has a dismal prognosis, but allo-HSCT as frontline consolidation is potentially curative, Dr. Bazarbachi said. It is most effective in patients who have achieved a complete or partial response to induction (Blood. 2012 Aug 23;120[8]:1734-41).
However, allo-HSCT should not be given as consolidation to ATLL patients who have received prior mogamulizumab. These patients have an increased risk of morbidity and mortality if they undergo allo-HSCT.
Also, allo-HSCT should not be given to refractory ATLL patients, although it may be an option for relapsed patients.
Dr. Bazarbachi stressed that ATLL patients should not receive auto-HSCT at any time, as frontline consolidation, after relapse, or if they have refractory disease.
Auto-HSCT “does not work in this disease,” he said. In a study published in 2014, all four ATLL patients who underwent auto-HSCT “rapidly” died (Bone Marrow Transplant. 2014 Oct;49[10]:1266-8).
ENKTL
Dr. Bazarbachi said frontline consolidation with auto-HSCT should be considered the standard of care for patients with non-localized ENKTL, nasal type.
Auto-HSCT has been shown to improve survival in these patients, and it is most effective when patients have achieved a complete response to induction (Biol Blood Marrow Transplant. 2008 Dec;14[12]:1356-64).
Allo-HSCT also is an option for frontline consolidation in patients with nonlocalized ENKTL, nasal type, Dr. Bazarbachi said.
He added that chemosensitive patients who have relapsed can receive allo-HSCT, but they should receive auto-HSCT only if they did not receive it in the frontline setting. Both types of transplant should take place when patients are in complete remission.
Patients with refractory, nonlocalized ENKTL, nasal type, should not receive auto-HSCT, but allo-HSCT is an option, Dr. Bazarbachi said.
Dr. Bazarbachi did not declare any conflicts of interest.
The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Associates, which is owned by the parent company of this news organization.
EXPERT ANALYSIS FROM LEUKEMIA AND LYMPHOMA 2018
Study challenges LVEF assessment before DLBCL treatment
Measuring left ventricular ejection fraction (LVEF) before administering doxorubicin-based chemotherapy doesn’t appear to add clinically meaningful information, according to an analysis of diffuse large B-cell lymphoma (DLBCL) patients.
Current guidelines recommend prescreening with either echocardiography or multiple-gated acquisition (MUGA) scan to identify asymptomatic left ventricular dysfunction before administering doxorubicin-containing chemotherapy, since doxorubicin is known for its cardiotoxicity.
But other studies have challenged the usefulness of routine LVEF screening in DLBCL patients.
In the current study, Deborah L. Enns, PhD, and her colleagues at Virginia Mason Medical Center in Seattle reviewed the medical records of 291 patients diagnosed with DLBCL between 2001 and 2013.
In total, 206 patients with normal LVEF and 8 patients with low LVEF received doxorubicin (P = .006). But while that association appears to support routine prescreening to inform clinical decision making, the link disappeared when the researchers factored out previous cardiac disease (P = .51).
“It is possible that previous [heart failure] may have played a larger role in shaping treatment decisions than did LVEF test results alone,” the researchers wrote. The report is in Mayo Clinic Proceedings: Innovations, Quality & Outcomes.
In addition, for patients who had their LVEF measured, the researchers found no difference in posttreatment incidence of heart failure based on whether patients received doxorubicin (7.0%) or did not (6.8%). The same was true for patients with LVEF values of less than 50% before treatment (13% vs. 14%).
The researchers noted that there are several reasons why LVEF prescreening may not be needed before treatment in DLBCL. For instance, DLBCL patients typically receive low cumulative doses of doxorubicin. Also, doxorubicin’s high efficacy in DLBCL should be balanced with the relatively low risk of death from heart failure due to doxorubicin treatment, at around 4% after 5 years for patients without preexisting cardiac conditions.
“We recommend that the policy of routinely performing prescreening LVEF measurements in all patients with DLBCL before administering anthracycline-based chemotherapy treatments be reevaluated,” the researchers wrote.
They reported having no competing interests.
SOURCE: Enns DL et al. Mayo Clin Proc Innov Qual Outcomes. 2018 Aug 3;2(3):277-85.
Measuring left ventricular ejection fraction (LVEF) before administering doxorubicin-based chemotherapy doesn’t appear to add clinically meaningful information, according to an analysis of diffuse large B-cell lymphoma (DLBCL) patients.
Current guidelines recommend prescreening with either echocardiography or multiple-gated acquisition (MUGA) scan to identify asymptomatic left ventricular dysfunction before administering doxorubicin-containing chemotherapy, since doxorubicin is known for its cardiotoxicity.
But other studies have challenged the usefulness of routine LVEF screening in DLBCL patients.
In the current study, Deborah L. Enns, PhD, and her colleagues at Virginia Mason Medical Center in Seattle reviewed the medical records of 291 patients diagnosed with DLBCL between 2001 and 2013.
In total, 206 patients with normal LVEF and 8 patients with low LVEF received doxorubicin (P = .006). But while that association appears to support routine prescreening to inform clinical decision making, the link disappeared when the researchers factored out previous cardiac disease (P = .51).
“It is possible that previous [heart failure] may have played a larger role in shaping treatment decisions than did LVEF test results alone,” the researchers wrote. The report is in Mayo Clinic Proceedings: Innovations, Quality & Outcomes.
In addition, for patients who had their LVEF measured, the researchers found no difference in posttreatment incidence of heart failure based on whether patients received doxorubicin (7.0%) or did not (6.8%). The same was true for patients with LVEF values of less than 50% before treatment (13% vs. 14%).
The researchers noted that there are several reasons why LVEF prescreening may not be needed before treatment in DLBCL. For instance, DLBCL patients typically receive low cumulative doses of doxorubicin. Also, doxorubicin’s high efficacy in DLBCL should be balanced with the relatively low risk of death from heart failure due to doxorubicin treatment, at around 4% after 5 years for patients without preexisting cardiac conditions.
“We recommend that the policy of routinely performing prescreening LVEF measurements in all patients with DLBCL before administering anthracycline-based chemotherapy treatments be reevaluated,” the researchers wrote.
They reported having no competing interests.
SOURCE: Enns DL et al. Mayo Clin Proc Innov Qual Outcomes. 2018 Aug 3;2(3):277-85.
Measuring left ventricular ejection fraction (LVEF) before administering doxorubicin-based chemotherapy doesn’t appear to add clinically meaningful information, according to an analysis of diffuse large B-cell lymphoma (DLBCL) patients.
Current guidelines recommend prescreening with either echocardiography or multiple-gated acquisition (MUGA) scan to identify asymptomatic left ventricular dysfunction before administering doxorubicin-containing chemotherapy, since doxorubicin is known for its cardiotoxicity.
But other studies have challenged the usefulness of routine LVEF screening in DLBCL patients.
In the current study, Deborah L. Enns, PhD, and her colleagues at Virginia Mason Medical Center in Seattle reviewed the medical records of 291 patients diagnosed with DLBCL between 2001 and 2013.
In total, 206 patients with normal LVEF and 8 patients with low LVEF received doxorubicin (P = .006). But while that association appears to support routine prescreening to inform clinical decision making, the link disappeared when the researchers factored out previous cardiac disease (P = .51).
“It is possible that previous [heart failure] may have played a larger role in shaping treatment decisions than did LVEF test results alone,” the researchers wrote. The report is in Mayo Clinic Proceedings: Innovations, Quality & Outcomes.
In addition, for patients who had their LVEF measured, the researchers found no difference in posttreatment incidence of heart failure based on whether patients received doxorubicin (7.0%) or did not (6.8%). The same was true for patients with LVEF values of less than 50% before treatment (13% vs. 14%).
The researchers noted that there are several reasons why LVEF prescreening may not be needed before treatment in DLBCL. For instance, DLBCL patients typically receive low cumulative doses of doxorubicin. Also, doxorubicin’s high efficacy in DLBCL should be balanced with the relatively low risk of death from heart failure due to doxorubicin treatment, at around 4% after 5 years for patients without preexisting cardiac conditions.
“We recommend that the policy of routinely performing prescreening LVEF measurements in all patients with DLBCL before administering anthracycline-based chemotherapy treatments be reevaluated,” the researchers wrote.
They reported having no competing interests.
SOURCE: Enns DL et al. Mayo Clin Proc Innov Qual Outcomes. 2018 Aug 3;2(3):277-85.
FROM MAYO CLINIC PROCEEDINGS: INNOVATIONS, QUALITY & OUTCOMES
Key clinical point:
Major finding: Among diffuse large B-cell lymphoma (DLBCL) patients who had LVEF measured, the incidence of heart failure post treatment did not differ between patients who received doxorubicin and those who did not (P = 1.0).
Study details: A retrospective analysis of 291 patients diagnosed with DLBCL between 2001 and 2013.
Disclosures: The researchers reported having no competing interests.
Source: Enns DL et al. Mayo Clin Proc Innov Qual Outcomes. 2018 Aug 3;2(3):277-85.
Ibrutinib discontinuation harms survival in CLL
Discontinuing ibrutinib therapy because of disease progression was associated with worse survival, according to a real-world study of ibrutinib dosing in chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma patients.
Researchers at the University of Rochester Wilmot Cancer Institute in New York, who performed the single-center study, also found that optimal dosing early on in treatment has a significant impact on disease progression.
“Treating physicians need to be aware of these outcomes when initiating therapy on patients with high-risk CLL or lymphoma, as well as those with significant comorbidities or immune deficiencies,” AnnaLynn M. Williams. MS, and her colleagues reported in Clinical Lymphoma, Myeloma and Leukemia.
The researchers examined the impact of ibrutinib discontinuation and dose adherence on overall and progression-free survival in 170 patients with non-Hodgkin lymphoma and CLL treated with the drug at the Wilmot Cancer Institute between Jan. 1, 2014, and Dec. 1, 2016.
The study comprised 115 patients with CLL, 23 patients with Waldenstrom macroglobulinemia, 21 patients with mantle cell lymphoma, and 11 patients with other non-Hodgkin lymphomas. The median age of patients who started ibrutinib was 68 years, and the median treatment duration was 14.3 months. About a third of patients were taking ibrutinib as a first-line treatment.
Overall, 51 patients (30%) permanently discontinued ibrutinib during the study period, with more than half of the discontinuations stemming from adverse events or comorbidities. About 35% of the discontinuations were due to disease progression.
Median overall survival after discontinuation due to disease progression was 1.7 months. When patients discontinued for other reasons, median overall survival was not reached, compared with stopping for disease progression (P = .0008).
The researchers reported that among patients who discontinued for nonprogression reasons, 67% were alive after 1 year. Among CLL patients, 80% were alive after 1 year.
Among 20 patients who had a dose adherence of less than 80% in the first 8 weeks, the researchers found worse progression-free survival (P = .002) and overall survival (P = .021). Among CLL patients only, progression-free survival was significantly worse (P = .043) but overall survival was not (P = .816).
The study also included five patients who reduced their ibrutinib dose in the first 8 weeks – down to 280 mg in two patients, 140 mg in two patients, and 420 mg in one patient. Again, the researchers observed worse progression-free survival (P = .004) and overall survival (P = .014), compared with patients who maintained their dosing level.
Interrupting ibrutinib dosing had an impact on survival but not as much as discontinuation. Among 10 patients who interrupted therapy for more than a week and then restarted, progression-free survival was worse, compared with those who stayed on treatment continuously (P = .047), but overall survival was not significantly worse (P = .577).
“This would suggest that the ideal treatment strategy would be to recommend initiation of therapy at standard dosing and interruption as needed as directed in the [Food and Drug Administration] label,” the researchers wrote.
The study was funded by the National Cancer Institute and the Cadregari Endowment Fund. The researchers reported having no conflicts of interest.
SOURCE: Williams AM et al. Clin Lymphoma Myeloma Leuk. 2018 Oct 12. doi: 10.1016/j.clml.2018.10.005.
Discontinuing ibrutinib therapy because of disease progression was associated with worse survival, according to a real-world study of ibrutinib dosing in chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma patients.
Researchers at the University of Rochester Wilmot Cancer Institute in New York, who performed the single-center study, also found that optimal dosing early on in treatment has a significant impact on disease progression.
“Treating physicians need to be aware of these outcomes when initiating therapy on patients with high-risk CLL or lymphoma, as well as those with significant comorbidities or immune deficiencies,” AnnaLynn M. Williams. MS, and her colleagues reported in Clinical Lymphoma, Myeloma and Leukemia.
The researchers examined the impact of ibrutinib discontinuation and dose adherence on overall and progression-free survival in 170 patients with non-Hodgkin lymphoma and CLL treated with the drug at the Wilmot Cancer Institute between Jan. 1, 2014, and Dec. 1, 2016.
The study comprised 115 patients with CLL, 23 patients with Waldenstrom macroglobulinemia, 21 patients with mantle cell lymphoma, and 11 patients with other non-Hodgkin lymphomas. The median age of patients who started ibrutinib was 68 years, and the median treatment duration was 14.3 months. About a third of patients were taking ibrutinib as a first-line treatment.
Overall, 51 patients (30%) permanently discontinued ibrutinib during the study period, with more than half of the discontinuations stemming from adverse events or comorbidities. About 35% of the discontinuations were due to disease progression.
Median overall survival after discontinuation due to disease progression was 1.7 months. When patients discontinued for other reasons, median overall survival was not reached, compared with stopping for disease progression (P = .0008).
The researchers reported that among patients who discontinued for nonprogression reasons, 67% were alive after 1 year. Among CLL patients, 80% were alive after 1 year.
Among 20 patients who had a dose adherence of less than 80% in the first 8 weeks, the researchers found worse progression-free survival (P = .002) and overall survival (P = .021). Among CLL patients only, progression-free survival was significantly worse (P = .043) but overall survival was not (P = .816).
The study also included five patients who reduced their ibrutinib dose in the first 8 weeks – down to 280 mg in two patients, 140 mg in two patients, and 420 mg in one patient. Again, the researchers observed worse progression-free survival (P = .004) and overall survival (P = .014), compared with patients who maintained their dosing level.
Interrupting ibrutinib dosing had an impact on survival but not as much as discontinuation. Among 10 patients who interrupted therapy for more than a week and then restarted, progression-free survival was worse, compared with those who stayed on treatment continuously (P = .047), but overall survival was not significantly worse (P = .577).
“This would suggest that the ideal treatment strategy would be to recommend initiation of therapy at standard dosing and interruption as needed as directed in the [Food and Drug Administration] label,” the researchers wrote.
The study was funded by the National Cancer Institute and the Cadregari Endowment Fund. The researchers reported having no conflicts of interest.
SOURCE: Williams AM et al. Clin Lymphoma Myeloma Leuk. 2018 Oct 12. doi: 10.1016/j.clml.2018.10.005.
Discontinuing ibrutinib therapy because of disease progression was associated with worse survival, according to a real-world study of ibrutinib dosing in chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma patients.
Researchers at the University of Rochester Wilmot Cancer Institute in New York, who performed the single-center study, also found that optimal dosing early on in treatment has a significant impact on disease progression.
“Treating physicians need to be aware of these outcomes when initiating therapy on patients with high-risk CLL or lymphoma, as well as those with significant comorbidities or immune deficiencies,” AnnaLynn M. Williams. MS, and her colleagues reported in Clinical Lymphoma, Myeloma and Leukemia.
The researchers examined the impact of ibrutinib discontinuation and dose adherence on overall and progression-free survival in 170 patients with non-Hodgkin lymphoma and CLL treated with the drug at the Wilmot Cancer Institute between Jan. 1, 2014, and Dec. 1, 2016.
The study comprised 115 patients with CLL, 23 patients with Waldenstrom macroglobulinemia, 21 patients with mantle cell lymphoma, and 11 patients with other non-Hodgkin lymphomas. The median age of patients who started ibrutinib was 68 years, and the median treatment duration was 14.3 months. About a third of patients were taking ibrutinib as a first-line treatment.
Overall, 51 patients (30%) permanently discontinued ibrutinib during the study period, with more than half of the discontinuations stemming from adverse events or comorbidities. About 35% of the discontinuations were due to disease progression.
Median overall survival after discontinuation due to disease progression was 1.7 months. When patients discontinued for other reasons, median overall survival was not reached, compared with stopping for disease progression (P = .0008).
The researchers reported that among patients who discontinued for nonprogression reasons, 67% were alive after 1 year. Among CLL patients, 80% were alive after 1 year.
Among 20 patients who had a dose adherence of less than 80% in the first 8 weeks, the researchers found worse progression-free survival (P = .002) and overall survival (P = .021). Among CLL patients only, progression-free survival was significantly worse (P = .043) but overall survival was not (P = .816).
The study also included five patients who reduced their ibrutinib dose in the first 8 weeks – down to 280 mg in two patients, 140 mg in two patients, and 420 mg in one patient. Again, the researchers observed worse progression-free survival (P = .004) and overall survival (P = .014), compared with patients who maintained their dosing level.
Interrupting ibrutinib dosing had an impact on survival but not as much as discontinuation. Among 10 patients who interrupted therapy for more than a week and then restarted, progression-free survival was worse, compared with those who stayed on treatment continuously (P = .047), but overall survival was not significantly worse (P = .577).
“This would suggest that the ideal treatment strategy would be to recommend initiation of therapy at standard dosing and interruption as needed as directed in the [Food and Drug Administration] label,” the researchers wrote.
The study was funded by the National Cancer Institute and the Cadregari Endowment Fund. The researchers reported having no conflicts of interest.
SOURCE: Williams AM et al. Clin Lymphoma Myeloma Leuk. 2018 Oct 12. doi: 10.1016/j.clml.2018.10.005.
FROM CLINICAL LYMPHOMA, MYELOMA AND LEUKEMIA
Key clinical point:
Major finding: Median overall survival after discontinuation of ibrutinib due to disease progression was 1.7 months.
Study details: A single-institution study of 170 patients with CLL or non-Hodgkin lymphoma who were taking ibrutinib.
Disclosures: The study was funded by the National Cancer Institute and the Cadregari Endowment Fund. The researchers reported having no conflicts of interest.
Source: Williams AM et al. Clin Lymphoma Myeloma Leuk. 2018 Oct 12. doi: 10.1016/j.clml.2018.10.005.
Ibrutinib plus obinutuzumab gets priority review in CLL/SLL
The Food and Drug Administration has granted priority review to an anti-CD20, chemotherapy-free combination – ibrutinib plus obinutuzumab – for the frontline treatment of chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL).
The agency will review the combination in previously untreated adults.
Ibrutinib (Imbruvica) is already approved as a single agent for adults with CLL/SLL for all lines of therapy and in combination with bendamustine and rituximab. Obinutuzumab (Gazyva) has been approved for patients with previously untreated CLL, in combination with chlorambucil.
The current application, which is sponsored by Janssen and Pharmacyclics, is based on results from the phase 3 iLLUMINATE trial. Preliminary results announced by Janssen and Pharmacyclics showed that ibrutinib plus obinutuzumab had statistically significant better progression-free survival, compared with chlorambucil plus obinutuzumab, as assessed by an independent review committee.
Complete results from the trial will be presented at an upcoming medical meeting, according to the sponsors.
The Food and Drug Administration has granted priority review to an anti-CD20, chemotherapy-free combination – ibrutinib plus obinutuzumab – for the frontline treatment of chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL).
The agency will review the combination in previously untreated adults.
Ibrutinib (Imbruvica) is already approved as a single agent for adults with CLL/SLL for all lines of therapy and in combination with bendamustine and rituximab. Obinutuzumab (Gazyva) has been approved for patients with previously untreated CLL, in combination with chlorambucil.
The current application, which is sponsored by Janssen and Pharmacyclics, is based on results from the phase 3 iLLUMINATE trial. Preliminary results announced by Janssen and Pharmacyclics showed that ibrutinib plus obinutuzumab had statistically significant better progression-free survival, compared with chlorambucil plus obinutuzumab, as assessed by an independent review committee.
Complete results from the trial will be presented at an upcoming medical meeting, according to the sponsors.
The Food and Drug Administration has granted priority review to an anti-CD20, chemotherapy-free combination – ibrutinib plus obinutuzumab – for the frontline treatment of chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL).
The agency will review the combination in previously untreated adults.
Ibrutinib (Imbruvica) is already approved as a single agent for adults with CLL/SLL for all lines of therapy and in combination with bendamustine and rituximab. Obinutuzumab (Gazyva) has been approved for patients with previously untreated CLL, in combination with chlorambucil.
The current application, which is sponsored by Janssen and Pharmacyclics, is based on results from the phase 3 iLLUMINATE trial. Preliminary results announced by Janssen and Pharmacyclics showed that ibrutinib plus obinutuzumab had statistically significant better progression-free survival, compared with chlorambucil plus obinutuzumab, as assessed by an independent review committee.
Complete results from the trial will be presented at an upcoming medical meeting, according to the sponsors.
Entospletinib falls short in relapsed/refractory DLBCL
Entospletinib, a selective inhibitor of spleen tyrosine kinase (Syk), showed a dismal rate of progression-free survival and a high rate of adverse events in a cohort of previously treated patients with diffuse large B-cell lymphoma (DLBCL).
Entospletinib was evaluated in an open-label, single-agent, phase 2 trial (NCT01799889) with five relapsed/refractory patient cohorts: chronic lymphocytic leukemia (CLL), follicular lymphoma, other indolent non-Hodgkin lymphomas, mantle cell lymphoma, and DLBCL.
John M. Burke, MD, of Rocky Mountain Cancer Centers in Aurora, Colo., and his colleagues reported on the current analysis, which looked specifically at the 43 patients in the trial with previously treated DLBCL. Patients received at least one starting dose of 800 mg of entospletinib orally twice daily. The findings were published in Clinical Lymphoma, Myeloma & Leukemia.
In a previous report on the relapsed/refractory CLL cohort, the investigational agent demonstrated clinical activity with acceptable toxicity (Blood. 2015 Apr 9;125[15]:2336-43).
In the current report, the rate of progression-free survival (PFS) at 16 weeks was 3.6% and the median PFS was 1.5 months. None of the patients in the study achieved a complete or partial response to treatment, and just five patients had stable disease.
All patients in the study eventually discontinued treatment and the median treatment duration was 1 month.
“The lack of activity of Syk inhibition in patients with relapsed DLBCL is in contrast to what would have been expected from preclinical data,” the investigators wrote. “Although it is unclear why entospletinib monotherapy lacked activity in the present study, it is possible that resistance to Syk inhibition played a role. Potential mechanisms of resistance of DLBCL to Syk inhibition include transcriptional upregulation of Syk mediated by FOXO1 and PTEN depletion.”
The investigators said that Syk inhibition in combination with BCL2 inhibitors could potentially overcome this resistance. Another approach, they suggested, would be to offer entospletinib in combination with Janus kinase (JAK) 1/3 inhibition.
“Based on results of the preclinical data, the efficacy of entospletinib in combination will be evaluated in future clinical trials,” the investigators wrote.
The rate of adverse events was high in the DLBCL cohort. Forty-two patients (98%) experienced an adverse event and nearly three-quarters experienced a grade 3 event. Overall, 30% of the grade 3 adverse events were related to treatment. More than 40% of patients interrupted treatment because of adverse events, and 19% discontinued. Four patients experienced an adverse event that led to death.
While the lack of clinical activity may have surprised investigators, the safety profile was in line with other patient cohorts in the phase 2 study. In the CLL and indolent non-Hodgkin lymphoma cohorts, the rates of treatment interruption were 45% and 54%, respectively.
The study was supported by Gilead Sciences. Dr. Burke reported relationships with Gilead and other companies.
SOURCE: Burke JM et al. Clin Lymphoma Myeloma Leuk. 2018 Aug;18(8):e327-e331.
Entospletinib, a selective inhibitor of spleen tyrosine kinase (Syk), showed a dismal rate of progression-free survival and a high rate of adverse events in a cohort of previously treated patients with diffuse large B-cell lymphoma (DLBCL).
Entospletinib was evaluated in an open-label, single-agent, phase 2 trial (NCT01799889) with five relapsed/refractory patient cohorts: chronic lymphocytic leukemia (CLL), follicular lymphoma, other indolent non-Hodgkin lymphomas, mantle cell lymphoma, and DLBCL.
John M. Burke, MD, of Rocky Mountain Cancer Centers in Aurora, Colo., and his colleagues reported on the current analysis, which looked specifically at the 43 patients in the trial with previously treated DLBCL. Patients received at least one starting dose of 800 mg of entospletinib orally twice daily. The findings were published in Clinical Lymphoma, Myeloma & Leukemia.
In a previous report on the relapsed/refractory CLL cohort, the investigational agent demonstrated clinical activity with acceptable toxicity (Blood. 2015 Apr 9;125[15]:2336-43).
In the current report, the rate of progression-free survival (PFS) at 16 weeks was 3.6% and the median PFS was 1.5 months. None of the patients in the study achieved a complete or partial response to treatment, and just five patients had stable disease.
All patients in the study eventually discontinued treatment and the median treatment duration was 1 month.
“The lack of activity of Syk inhibition in patients with relapsed DLBCL is in contrast to what would have been expected from preclinical data,” the investigators wrote. “Although it is unclear why entospletinib monotherapy lacked activity in the present study, it is possible that resistance to Syk inhibition played a role. Potential mechanisms of resistance of DLBCL to Syk inhibition include transcriptional upregulation of Syk mediated by FOXO1 and PTEN depletion.”
The investigators said that Syk inhibition in combination with BCL2 inhibitors could potentially overcome this resistance. Another approach, they suggested, would be to offer entospletinib in combination with Janus kinase (JAK) 1/3 inhibition.
“Based on results of the preclinical data, the efficacy of entospletinib in combination will be evaluated in future clinical trials,” the investigators wrote.
The rate of adverse events was high in the DLBCL cohort. Forty-two patients (98%) experienced an adverse event and nearly three-quarters experienced a grade 3 event. Overall, 30% of the grade 3 adverse events were related to treatment. More than 40% of patients interrupted treatment because of adverse events, and 19% discontinued. Four patients experienced an adverse event that led to death.
While the lack of clinical activity may have surprised investigators, the safety profile was in line with other patient cohorts in the phase 2 study. In the CLL and indolent non-Hodgkin lymphoma cohorts, the rates of treatment interruption were 45% and 54%, respectively.
The study was supported by Gilead Sciences. Dr. Burke reported relationships with Gilead and other companies.
SOURCE: Burke JM et al. Clin Lymphoma Myeloma Leuk. 2018 Aug;18(8):e327-e331.
Entospletinib, a selective inhibitor of spleen tyrosine kinase (Syk), showed a dismal rate of progression-free survival and a high rate of adverse events in a cohort of previously treated patients with diffuse large B-cell lymphoma (DLBCL).
Entospletinib was evaluated in an open-label, single-agent, phase 2 trial (NCT01799889) with five relapsed/refractory patient cohorts: chronic lymphocytic leukemia (CLL), follicular lymphoma, other indolent non-Hodgkin lymphomas, mantle cell lymphoma, and DLBCL.
John M. Burke, MD, of Rocky Mountain Cancer Centers in Aurora, Colo., and his colleagues reported on the current analysis, which looked specifically at the 43 patients in the trial with previously treated DLBCL. Patients received at least one starting dose of 800 mg of entospletinib orally twice daily. The findings were published in Clinical Lymphoma, Myeloma & Leukemia.
In a previous report on the relapsed/refractory CLL cohort, the investigational agent demonstrated clinical activity with acceptable toxicity (Blood. 2015 Apr 9;125[15]:2336-43).
In the current report, the rate of progression-free survival (PFS) at 16 weeks was 3.6% and the median PFS was 1.5 months. None of the patients in the study achieved a complete or partial response to treatment, and just five patients had stable disease.
All patients in the study eventually discontinued treatment and the median treatment duration was 1 month.
“The lack of activity of Syk inhibition in patients with relapsed DLBCL is in contrast to what would have been expected from preclinical data,” the investigators wrote. “Although it is unclear why entospletinib monotherapy lacked activity in the present study, it is possible that resistance to Syk inhibition played a role. Potential mechanisms of resistance of DLBCL to Syk inhibition include transcriptional upregulation of Syk mediated by FOXO1 and PTEN depletion.”
The investigators said that Syk inhibition in combination with BCL2 inhibitors could potentially overcome this resistance. Another approach, they suggested, would be to offer entospletinib in combination with Janus kinase (JAK) 1/3 inhibition.
“Based on results of the preclinical data, the efficacy of entospletinib in combination will be evaluated in future clinical trials,” the investigators wrote.
The rate of adverse events was high in the DLBCL cohort. Forty-two patients (98%) experienced an adverse event and nearly three-quarters experienced a grade 3 event. Overall, 30% of the grade 3 adverse events were related to treatment. More than 40% of patients interrupted treatment because of adverse events, and 19% discontinued. Four patients experienced an adverse event that led to death.
While the lack of clinical activity may have surprised investigators, the safety profile was in line with other patient cohorts in the phase 2 study. In the CLL and indolent non-Hodgkin lymphoma cohorts, the rates of treatment interruption were 45% and 54%, respectively.
The study was supported by Gilead Sciences. Dr. Burke reported relationships with Gilead and other companies.
SOURCE: Burke JM et al. Clin Lymphoma Myeloma Leuk. 2018 Aug;18(8):e327-e331.
FROM CLINICAL LYMPHOMA, MYELOMA & LEUKEMIA
Key clinical point:
Major finding: The rate of progression-free survival at 16 weeks was 3.6% with a median PFS of 1.5 months.
Study details: An analysis of 43 relapsed/refractory DLBCL patients who received single-agent entospletinib.
Disclosures: The study was supported by Gilead Sciences. Dr. Burke reported relationships with Gilead and other companies.
Source: Burke JM et al. Clin Lymphoma Myeloma Leuk. 2018 Aug;18(8):e327-e331.
Updated ThroLy system predicts need for thromboprophylaxis
DUBROVNIK, CROATIA – An updated scoring system can more accurately identify lymphoma patients who may require thromboprophylaxis, according to researchers.
The revised scoring system, ThroLy, proved more effective than other systems for predicting thromboembolic events in lymphoma patients, with a positive predictive value of 22%-25%, a negative predictive value of 96%, sensitivity of 56%-57%, and specificity of 85%-87%.
Darko Antic, MD, PhD, of the University of Belgrade in Serbia, presented these findings at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
Dr. Antic said that he and his colleagues developed ThroLy because other systems used to predict venous thromboembolism (VTE) are not quite right for lymphoma. He noted that the Padua score is not designed for cancer patients and the Khorana score is predominantly used for solid tumor malignancies.
The ThroLy scoring system is based on variables used in the Padua and Khorana systems, as well as variables that are specific to lymphoma patients.
In a previous study, the researchers found several variables that were independently associated with risk for VTE in lymphoma, including previous VTE, previous acute MI or stroke, mediastinal involvement, high body mass index, reduced mobility, extranodal localization, neutropenia, and hemoglobin less than 100 g/L (Am J Hematol. 2016 Oct;91[10]:1014-9).
In an initial version of the ThroLy scoring system, previous VTE, previous acute MI/stroke, obesity, and mediastinal involvement were all worth two points, and the other factors were worth a single point in the ThroLy system.
Patients with scores of 0 to 1 were considered low risk, patients with scores of 2 to 3 were considered intermediate risk, and patients with scores of 4 or greater were considered high risk.
To validate and refine ThroLy, Dr. Antic and his colleagues used it to assess 1,723 lymphoma patients treated at eight institutions in Austria, Croatia, France, Jordan, Macedonia, Spain, Switzerland, and the United States.
Patients had indolent non-Hodgkin lymphoma, aggressive non-Hodgkin lymphoma, chronic lymphocytic leukemia/small lymphocytic lymphoma, and Hodgkin lymphoma. Most subjects (84%) were outpatients. A total of 9%of patients had thrombosis, with 7% having VTE.
ThroLy had a positive predictive value of 17%, compared with 11% with Khorana and 13% with Padua. The negative predictive value was 93%, 92%, and 95%, respectively. The sensitivity was 51% with ThroLy, 42% with Khorana, and 70% with Padua; specificity was 72%, 64%, and 52%, respectively.
“The positive predictive value was low [with ThroLy] but definitely higher than the positive predictive value of the other two [scoring systems],” Dr. Antic noted.
Updated models
To further improve ThroLy, the researchers updated the system, creating two new models. Model 1 included the type of lymphoma/clinical stage (1 point), previous VTE (5 points), reduced mobility (2 points), hemoglobin less than 100 g/L (1 point), and the presence of vascular devices (1 point). Model 2 included all of the variables in Model 1 plus the thrombophilic condition, which was worth 1 point.
Patients were considered low risk if they scored 2 points or lower and high risk if they scored more than 2 points.
For Model 1, the positive predictive value was 22%, the negative predictive value was 96%, the sensitivity was 56%, and the specificity was 85%. For Model 2, the positive predictive value was 25%, the negative predictive value was 96%, the sensitivity was 57%, and the specificity was 87%.
There were no major differences in model discrimination and calibration based on the country in which a patient was treated or whether the patient was treated in an inpatient or outpatient setting.
Dr. Antic did not report any conflicts of interest. The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Association, which is owned by the parent company of this news organization.
DUBROVNIK, CROATIA – An updated scoring system can more accurately identify lymphoma patients who may require thromboprophylaxis, according to researchers.
The revised scoring system, ThroLy, proved more effective than other systems for predicting thromboembolic events in lymphoma patients, with a positive predictive value of 22%-25%, a negative predictive value of 96%, sensitivity of 56%-57%, and specificity of 85%-87%.
Darko Antic, MD, PhD, of the University of Belgrade in Serbia, presented these findings at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
Dr. Antic said that he and his colleagues developed ThroLy because other systems used to predict venous thromboembolism (VTE) are not quite right for lymphoma. He noted that the Padua score is not designed for cancer patients and the Khorana score is predominantly used for solid tumor malignancies.
The ThroLy scoring system is based on variables used in the Padua and Khorana systems, as well as variables that are specific to lymphoma patients.
In a previous study, the researchers found several variables that were independently associated with risk for VTE in lymphoma, including previous VTE, previous acute MI or stroke, mediastinal involvement, high body mass index, reduced mobility, extranodal localization, neutropenia, and hemoglobin less than 100 g/L (Am J Hematol. 2016 Oct;91[10]:1014-9).
In an initial version of the ThroLy scoring system, previous VTE, previous acute MI/stroke, obesity, and mediastinal involvement were all worth two points, and the other factors were worth a single point in the ThroLy system.
Patients with scores of 0 to 1 were considered low risk, patients with scores of 2 to 3 were considered intermediate risk, and patients with scores of 4 or greater were considered high risk.
To validate and refine ThroLy, Dr. Antic and his colleagues used it to assess 1,723 lymphoma patients treated at eight institutions in Austria, Croatia, France, Jordan, Macedonia, Spain, Switzerland, and the United States.
Patients had indolent non-Hodgkin lymphoma, aggressive non-Hodgkin lymphoma, chronic lymphocytic leukemia/small lymphocytic lymphoma, and Hodgkin lymphoma. Most subjects (84%) were outpatients. A total of 9%of patients had thrombosis, with 7% having VTE.
ThroLy had a positive predictive value of 17%, compared with 11% with Khorana and 13% with Padua. The negative predictive value was 93%, 92%, and 95%, respectively. The sensitivity was 51% with ThroLy, 42% with Khorana, and 70% with Padua; specificity was 72%, 64%, and 52%, respectively.
“The positive predictive value was low [with ThroLy] but definitely higher than the positive predictive value of the other two [scoring systems],” Dr. Antic noted.
Updated models
To further improve ThroLy, the researchers updated the system, creating two new models. Model 1 included the type of lymphoma/clinical stage (1 point), previous VTE (5 points), reduced mobility (2 points), hemoglobin less than 100 g/L (1 point), and the presence of vascular devices (1 point). Model 2 included all of the variables in Model 1 plus the thrombophilic condition, which was worth 1 point.
Patients were considered low risk if they scored 2 points or lower and high risk if they scored more than 2 points.
For Model 1, the positive predictive value was 22%, the negative predictive value was 96%, the sensitivity was 56%, and the specificity was 85%. For Model 2, the positive predictive value was 25%, the negative predictive value was 96%, the sensitivity was 57%, and the specificity was 87%.
There were no major differences in model discrimination and calibration based on the country in which a patient was treated or whether the patient was treated in an inpatient or outpatient setting.
Dr. Antic did not report any conflicts of interest. The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Association, which is owned by the parent company of this news organization.
DUBROVNIK, CROATIA – An updated scoring system can more accurately identify lymphoma patients who may require thromboprophylaxis, according to researchers.
The revised scoring system, ThroLy, proved more effective than other systems for predicting thromboembolic events in lymphoma patients, with a positive predictive value of 22%-25%, a negative predictive value of 96%, sensitivity of 56%-57%, and specificity of 85%-87%.
Darko Antic, MD, PhD, of the University of Belgrade in Serbia, presented these findings at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
Dr. Antic said that he and his colleagues developed ThroLy because other systems used to predict venous thromboembolism (VTE) are not quite right for lymphoma. He noted that the Padua score is not designed for cancer patients and the Khorana score is predominantly used for solid tumor malignancies.
The ThroLy scoring system is based on variables used in the Padua and Khorana systems, as well as variables that are specific to lymphoma patients.
In a previous study, the researchers found several variables that were independently associated with risk for VTE in lymphoma, including previous VTE, previous acute MI or stroke, mediastinal involvement, high body mass index, reduced mobility, extranodal localization, neutropenia, and hemoglobin less than 100 g/L (Am J Hematol. 2016 Oct;91[10]:1014-9).
In an initial version of the ThroLy scoring system, previous VTE, previous acute MI/stroke, obesity, and mediastinal involvement were all worth two points, and the other factors were worth a single point in the ThroLy system.
Patients with scores of 0 to 1 were considered low risk, patients with scores of 2 to 3 were considered intermediate risk, and patients with scores of 4 or greater were considered high risk.
To validate and refine ThroLy, Dr. Antic and his colleagues used it to assess 1,723 lymphoma patients treated at eight institutions in Austria, Croatia, France, Jordan, Macedonia, Spain, Switzerland, and the United States.
Patients had indolent non-Hodgkin lymphoma, aggressive non-Hodgkin lymphoma, chronic lymphocytic leukemia/small lymphocytic lymphoma, and Hodgkin lymphoma. Most subjects (84%) were outpatients. A total of 9%of patients had thrombosis, with 7% having VTE.
ThroLy had a positive predictive value of 17%, compared with 11% with Khorana and 13% with Padua. The negative predictive value was 93%, 92%, and 95%, respectively. The sensitivity was 51% with ThroLy, 42% with Khorana, and 70% with Padua; specificity was 72%, 64%, and 52%, respectively.
“The positive predictive value was low [with ThroLy] but definitely higher than the positive predictive value of the other two [scoring systems],” Dr. Antic noted.
Updated models
To further improve ThroLy, the researchers updated the system, creating two new models. Model 1 included the type of lymphoma/clinical stage (1 point), previous VTE (5 points), reduced mobility (2 points), hemoglobin less than 100 g/L (1 point), and the presence of vascular devices (1 point). Model 2 included all of the variables in Model 1 plus the thrombophilic condition, which was worth 1 point.
Patients were considered low risk if they scored 2 points or lower and high risk if they scored more than 2 points.
For Model 1, the positive predictive value was 22%, the negative predictive value was 96%, the sensitivity was 56%, and the specificity was 85%. For Model 2, the positive predictive value was 25%, the negative predictive value was 96%, the sensitivity was 57%, and the specificity was 87%.
There were no major differences in model discrimination and calibration based on the country in which a patient was treated or whether the patient was treated in an inpatient or outpatient setting.
Dr. Antic did not report any conflicts of interest. The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Association, which is owned by the parent company of this news organization.
REPORTING FROM LEUKEMIA AND LYMPHOMA 2018
Key clinical point:
Major finding: The updated ThroLy had a positive predictive value of 22%-25%, a negative predictive value of 96%, sensitivity of 56%-57%, and specificity of 85%-87%.
Study details: The scoring system was validated on 1,723 lymphoma patients treated at eight institutions worldwide.
Disclosures: Dr. Antic reported having no conflicts of interest.






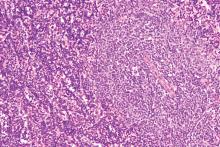
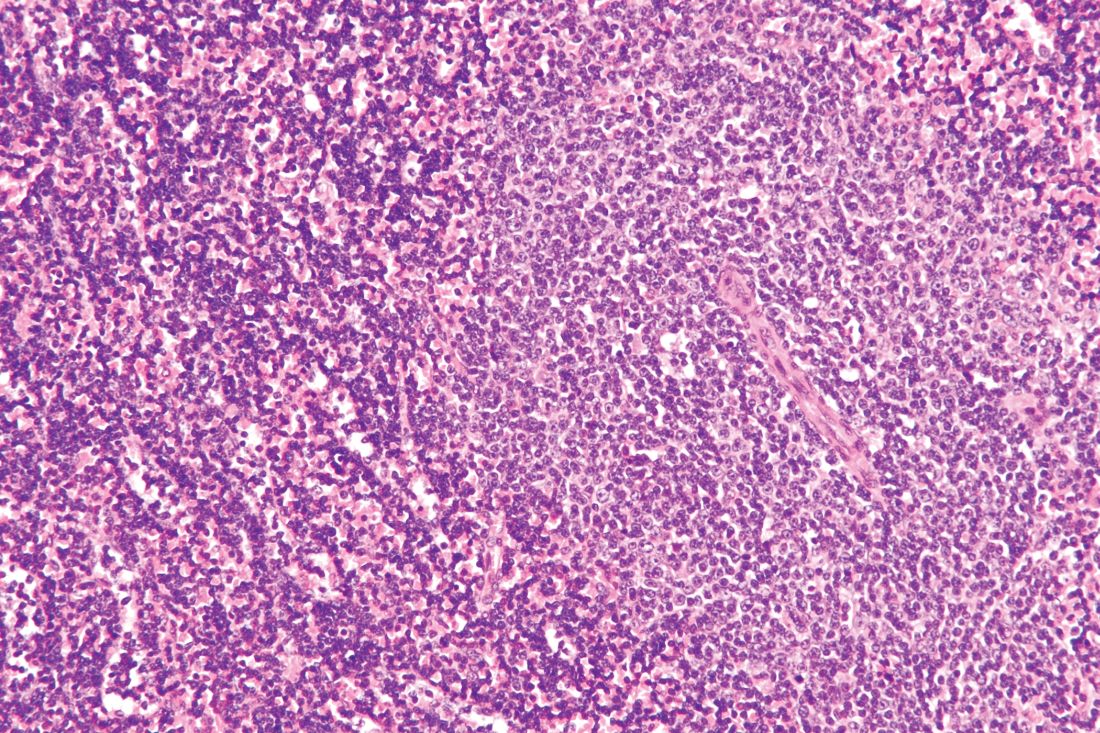
![Micrograph of a 'diffuse large B cell lymphoma. Lymph node FNA specimen. Field stain.(http://www.gnu.org/copyleft/fdl.html)], via Wikimedia Commons](https://cdn.mdedge.com/files/s3fs-public/styles/medium/public/Image/June-2017/DLBCL_web.jpg?itok=LAQhMuwC)
![Micrograph of a 'diffuse large B cell lymphoma. Lymph node FNA specimen. Field stain.(http://www.gnu.org/copyleft/fdl.html)], via Wikimedia Commons](https://cdn.mdedge.com/files/s3fs-public/Image/June-2017/DLBCL_web.jpg)