Myocardial infarction (MI) is the leading cause of death in the United States, and schizophrenia is the leading cause of disability. But while cardiologists manage the first heart attack very aggressively to prevent a second MI, we psychiatrists generally do not manage first-episode psychosis (FEP) as aggressively to prevent the more malignant second psychotic episode. Yet abundant evidence indicates that psychiatrists must behave like cardiologists at the onset of schizophrenia and other serious psychosis.
Individuals who survive the first heart attack, which permanently destroys part of the myocardium, are at high risk for a second MI, which may lead to death or weaken the heart so much that heart transplantation becomes necessary. Only implementation of aggressive medical intervention will prevent the likelihood of death due to a second MI in a person who has already suffered a first MI.
Similarly, the FEP of schizophrenia destroys brain tissue, about 10 to 12 cc containing millions of glial cells and billions of synapses.2 This neurotoxicity of psychosis is mediated by neuroinflammation and oxidative stress.3 In most FEP patients, the risk of a second psychotic episode is high, and the tissue destruction of the brain’s gray and white matter infrastructure is even more extensive, leading to clinical deterioration, treatment resistance, and functional disability. That is the grim turning point in the trajectory of schizophrenia.
Although most FEP patients respond well to antipsychotic medications and often return to their baseline social and vocational functioning, after a second episode, they are much more likely to become disabled. Unlike physical death, the mental, cognitive, social, and vocational death of chronic schizophrenia goes on for decades with much suffering, misery, and inability to have love and work, which is what life is all about (according to Freud).
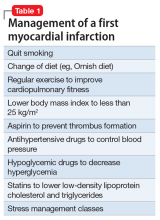
MI patients have healthy brains and minds, and they wholeheartedly agree with their cardiologists’ recommendations and religiously adhere to their cardiologists’ instructions, such as drastic lifestyle changes and a slew of medications intended to lower the risks of a second MI (Table 1).
But what is the most common psychiatric practice for a patient who suffers a FEP after he (she) is admitted to an acute inpatient ward? The patient is started on an oral antipsychotic but a long-acting injectable (LAI) antipsychotic, which is the best protection against future episodes, is never considered, let alone recommended. The patient is given a prescription for an oral antipsychotic at discharge and the family is told to find a private psychiatrist or a community mental health center for follow-up. This practice pattern will likely guarantee a relapse into a second psychotic episode for the following reasons:
- patients’ lack of insight (anosognosia) and refusal to believe they are sick or need medications
- adverse effects, especially extrapyramidal symptoms, to which FEP patients are particularly vulnerable unless they are started on small doses
- apathy and lack of motivation to take medication due to negative symptoms, which impair ability to initiate actions (avolition)
- severe memory impairment that leads to forgetting medications
- substance use, such as marijuana, stimulants, and hallucinogens, as well as alcohol, interferes with adherence.
Most patients and families are ignorant about FEP of schizophrenia and its recurrence and devastating effects.
Thus, because of the almost ubiquitous inability to adhere fully to antipsychotic medications after discharge, FEP patients are essentially destined (ie, doomed) to experience a destructive second psychotic episode, whose neurotoxicity starts the patient on a downhill journey of lifetime disability.4 LAI antipsychotics are the optimal solution to this serious problem, yet 99.99% of psychiatrists never start LAI during a FEP. This is inexplicable considering the body of evidence that supports early use of LAI to prevent relapse. Of the multipronged strategy that should be used for FEP patients to circumvent a second episode and avoid disability, starting LAI in FEP is the most important interventional tactic.5 Consider the following studies that support initiating LAIs during the FEP:
In South Africa, Emsley et al6 conducted the first study of LAI in FEP. In a 2-year follow-up, 64% of patients had complete remission and returned to their baseline functioning with restoration of insight and good quality of life. When the study ended and patients were returned to their referring psychiatrists after 2 years, all patients were switched to oral antipsychotics, because it was the standard practice among psychiatrists there. All patients relapsed within a few months due to poor adherence to oral medications. When they were placed back on the LAI they had received, a sobering (even shocking) clinical finding emerged: 16% of those who had responded so well to LAI for 2 years no longer responded!7 This rapid emergence of treatment resistance after only a second psychotic episode demonstrates how the brain changes drastically after a second episode and validates the recent adoption of “stages” in schizophrenia, similar to cancer stages.8 Many more patients will develop treatment resistance after subsequent episodes.
Subotnik et al9 compared LAI vs oral risperidone in 86 FEP patients. At the end of 1 year, they reported a 650% higher relapse rate in the oral medication group compared with the LAI group (33% vs 5%).9 This well-done study is a wake-up call for psychiatrists to help FEP patients avoid a brain-damaging second episode by using LAI as a first-line option in FEP.
In a separate study, Subotnik et al10 reported that when the blood level of a patient receiving an antipsychotic is measured at the time of discharge from FEP and every month for a year, all it took for a relapse was a drop of 25%.10 Thus, skipping an antipsychotic just 1 day out of 4 (partial nonadherence) is enough to cause a psychotic relapse.
So what should psychiatrists and nurse practitioners do to protect FEP patients from losing their lives to the permanent disability that begins with a second psychotic episode? They must simply change their attitude and their old-fashioned (antiquated?) prescribing habits that keep failing, and start administering LAI during the initial hospitalization right after a few (usually 3 or 4) days of receiving oral antipsychotics (with nursing-assured swallowing of pills) (Table 2). By starting the patient with oral antipsychotics, the presence of an allergic reaction is ruled out, and efficacy onset begins within 2 to 3 days.11 LAI can then be administered several days before discharge and continued in the outpatient setting.
However, various essential psychosocial interventions should be provided along with LAI to ensure progress toward remission and functional recovery after an FEP. The recently published National Institute of Mental Health-sponsored RAISE study12 is a prime example of the synergy between a multimodal and multidisciplinary team-based approach and antipsychotic medication to improve outcome and quality of life after emerging from FEP.
As psychiatric practitioners, we must be clinically aggressive during the “FEP window of opportunity” to avoid a second episode, thereby bending the curve of the downhill trajectory that occurs after second episodes. We must behave like cardiologists, and relentlessly protect patients who suffer a first “brain attack” from experiencing a relapse. No doubt, any psychiatrists who have a family member with FEP would channel their inner cardiologist and implement the evidence-based recommendations described above. But then, shouldn’t we apply the same standard of care to every FEP patient we see?