Posttraumatic stress disorder (PTSD) is a chronic and disabling psychiatric disorder. The lifetime prevalence among American adults is 6.8%.1 Management of PTSD includes treating distressing symptoms, reducing avoidant behaviors, treating comorbid conditions (eg, depression, substance use disorders, or mood dysregulation), and improving adaptive functioning, which includes restoring a psychological sense of safety and trust. PTSD can be treated using evidence-based psychotherapies, pharmacotherapy, or a combination of both modalities. For adults, evidence-based treatment guidelines recommend the use of cognitive-behavioral therapy, cognitive processing therapy, cognitive therapy, and prolonged exposure therapy.2 These guidelines also recommend (with some reservations) the use of brief eclectic psychotherapy, eye movement desensitization and reprocessing, and narrative exposure therapy.2 Although the evidence base for the use of medications is not as strong as that for the psychotherapies listed above, the guidelines recommend the use of fluoxetine, paroxetine, sertraline, and venlafaxine.2
Currently available treatments for PTSD have significant limitations. For example, trauma-focused psychotherapies can have significant rates of nonresponse, partial response, or treatment dropout.3,4 Additionally, such therapies are not widely accessible. As for pharmacotherapy, very few available options are supported by evidence, and the efficacy of these options is limited, as shown by the reports that only 60% of patients with PTSD show a response to selective serotonin reuptake inhibitors (SSRIs), and only 20% to 30% achieve complete remission.5 Additionally, it may take months for patients to achieve an acceptable level of improvement with medications. As a result, a substantial proportion of patients who seek treatment continue to remain symptomatic, with impaired levels of functioning. This lack of progress in PTSD treatment has been labeled as a national crisis, calling for an urgent need to find effective pharmacologic treatments for PTSD.6
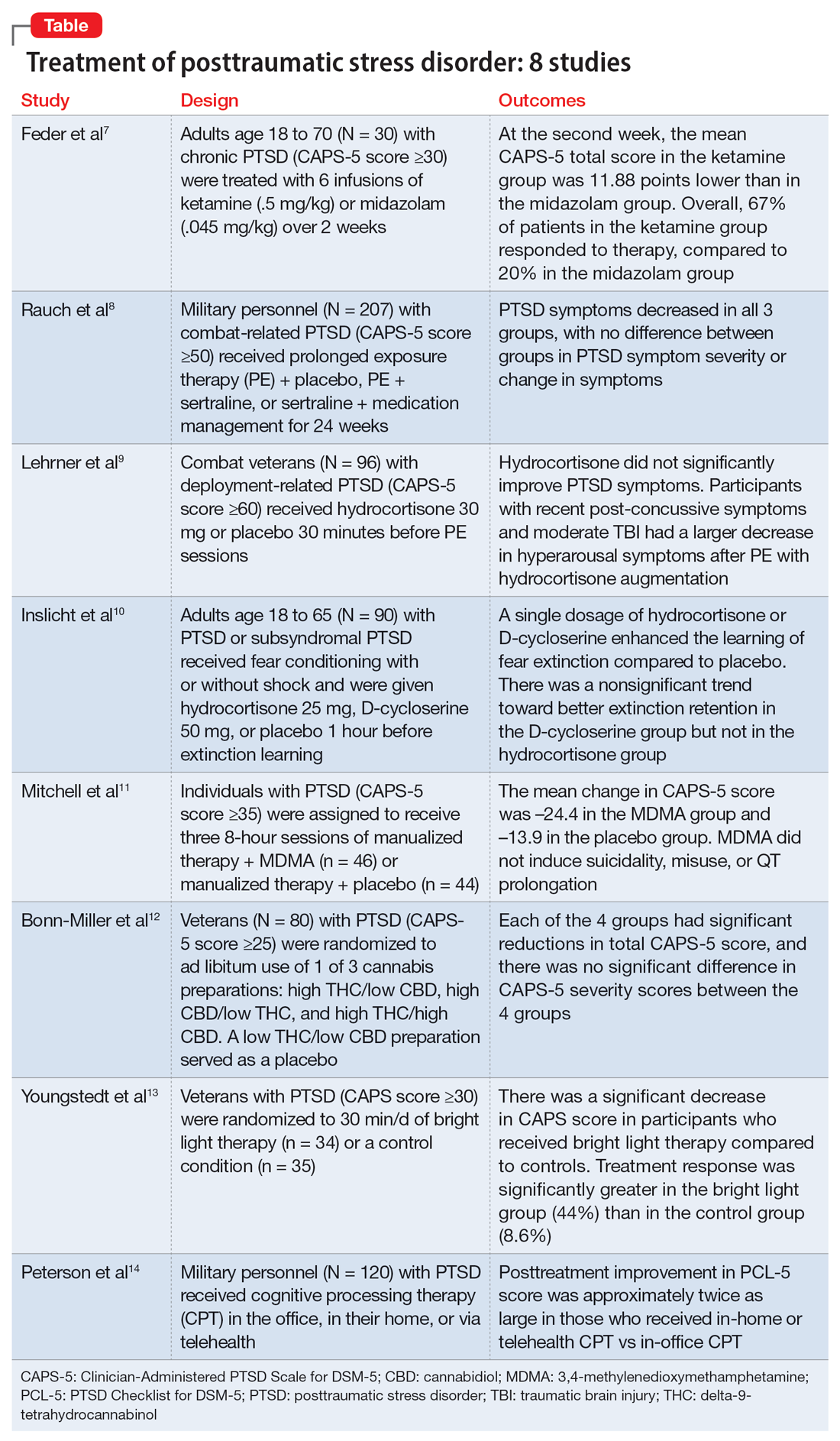
In this article, we review 8 randomized controlled trials (RCTs) of treatments for PTSD published within the last 5 years (Table7-14).
1. Feder A, Costi S, Rutter SB, et al. A randomized controlled trial of repeated ketamine administration for chronic posttraumatic stress disorder. Am J Psychiatry. 2021;178(2):193-202
Feder et al had previously found a significant and quick decrease in PTSD symptoms after a single dose of IV ketamine had. This is the first RCT to examine the effectiveness and safety of repeated IV ketamine infusions for the treatment of persistent PTSD.7
Study design
- This randomized, double-blind, parallel-arm controlled trial treated 30 individuals with chronic PTSD with 6 infusions of either ketamine (0.5 mg/kg) or midazolam (0.045 mg/kg) over 2 consecutive weeks.
- Participants were individuals age 18 to 70 with a primary diagnosis of chronic PTSD according to the DSM-5 criteria and determined by The Structure Clinical Interview for DSM-5, with a score ≥30 on the Clinician-Administered PTSD Scale for DSM-5 (CAPS-5).
- Any severe or unstable medical condition, active suicidal or homicidal ideation, lifetime history of psychotic or bipolar disorder, current anorexia nervosa or bulimia, alcohol or substance use disorder within 3 months of screening, history of recreational ketamine or phencyclidine use on more than 1 occasion or any use in the previous 2 years, and ongoing treatment with a long-acting benzodiazepine or opioid medication were all considered exclusion criteria. Individuals who took short-acting benzodiazepines had their morning doses held on infusion days. Marijuana or cannabis derivatives were allowed.
- The primary outcome measure was a change in PTSD symptom severity as measured with CAPS-5. This was administered before the first infusion and weekly thereafter. The Impact of Event Scale-Revised, the Montgomery–Åsberg Depression Rating Scale, and adverse effect measurements were used as secondary outcome measures.
- Treatment response was defined as ≥30% symptom improvement 2 weeks after the first infusion as assessed with CAPS-5.
- Individuals who responded to treatment were followed naturalistically weekly for up to 4 weeks and then monthly until loss of responder status, or up to 6 months if there was no loss of response.
Outcomes
- At the second week, the mean CAPS-5 total score in the ketamine group was 11.88 points (SE = 3.96) lower than in the midazolam group (d = 1.13; 95% CI, 0.36 to 1.91).
- In the ketamine group, 67% of patients responded to therapy, compared to 20% in the midazolam group.
- Following the 2-week course of infusions, the median period until loss of response among ketamine responders was 27.5 days.
- Ketamine infusions showed good tolerability and safety. There were no clinically significant adverse effects.
Continue to: Conclusions/limitations