User login
New narcolepsy drug passes phase 3 test
SAN DIEGO – The selective dopamine and norepinephrine reuptake inhibitor solriamfetol is effective in reducing sleepiness in patients with narcolepsy, according to results of a phase 3 study.
At 150-mg and 300-mg doses, the drug had statistically significant effects on objective and subjective measures.
There are wake-promoting drugs available, such as amphetamine-related drugs that are often used off label, but addiction liability is a concern. The nonamphetamine modafinil has been approved by the Food and Drug Administration since 1998.
Jazz Pharmaceuticals is in the process of submitting solriamfetol for FDA evaluation. If approved, the drug will add to the options available for narcolepsy patients. “All of the available drugs have some limitations. Some have more abuse liability than others. Some have more robust wake-promoting properties than others. We haven’t done any head-to-head comparisons, so I can’t tell you how we will stack up,” Philip Jochelson, MD, said in an interview. Dr. Jochelson is vice president of clinical development at Jazz Pharmaceuticals and presented the results of the study at a poster session at the annual meeting of the American Neurological Association.
An earlier study showed the drug had less abuse potential than the schedule IV stimulant phentermine. That’s not surprising given the drug’s mechanism of action, Dr. Jochelson said. Amphetamine-based drugs stimulate dopamine release, which can prompt a dopamine surge that people equate with a high, he said. Solriamfetol also affects dopamine, but it is a reuptake inhibitor, so it doesn’t produce a surge.
If the drug gains approval, it remains to be seen how it will be classified on the Drug Enforcement Agency Controlled Substance scale. “Where it will fall in that spectrum is speculative at this point,” said Dr. Jochelson.
In the current study, 236 adults (aged 18-75 years) with type 1 narcolepsy were randomized to once-daily placebo, 75 mg solriamfetol, 150 mg solriamfetol, or 300 mg solriamfetol; 27.1% of patients in the 300-mg group discontinued, compared with 7.3% in the 150-mg group, 16.9% in the 75-mg group, and 10.3% in the placebo group. The mean change from baseline on the Maintenance of Wakefulness Test was statistically significant in the 300-mg group (12.3 minutes vs. 2.1 minutes for placebo, P less than .0001) and the 150-mg group (9.8 minutes vs. 2.1 minutes, P less than .0001) but not the 75-mg group (4.7 minutes vs. 2.1 minutes).
The drug also outperformed placebo at week 12 on the Epworth Sleepiness Scale. The mean change in the 300-mg group was –6.4 vs. –1.6 for placebo (P less than .001), –5.4 in the 150-mg group (P less than .0001), and –3.8 in the 75-mg group (P less than .05).
By both Maintenance of Wakefulness Test and Epworth Sleepiness Scale measures, the 150-mg and 300-mg solriamfetol groups had statistically significant differences as early as week 1.
The drug had some adverse effects, which were expected based on its pharmacologic profile. These included increases in headache (5.1% with placebo, 10.2% with 75 mg, 23.7% with 150 mg, 30.5% with 300 mg), nausea (1.7% for placebo, 5.1% for 75 mg, 10.2% for 150 mg, 16.9% for 300 mg), anxiety (1.7% with placebo, 1.7% with 75 mg, 5.1% with 150 mg, 8.5% with 300 mg), and insomnia (0% for placebo, 3.4% for 75 mg, 0% for 150 mg, 5.1% for 300 mg). Other adverse events occurring in at least 5% of patients were decreased appetite, nasopharyngitis, and dry mouth.
The study was funded by Jazz Pharmaceuticals. Dr. Jochelson is an employee of Jazz.
SAN DIEGO – The selective dopamine and norepinephrine reuptake inhibitor solriamfetol is effective in reducing sleepiness in patients with narcolepsy, according to results of a phase 3 study.
At 150-mg and 300-mg doses, the drug had statistically significant effects on objective and subjective measures.
There are wake-promoting drugs available, such as amphetamine-related drugs that are often used off label, but addiction liability is a concern. The nonamphetamine modafinil has been approved by the Food and Drug Administration since 1998.
Jazz Pharmaceuticals is in the process of submitting solriamfetol for FDA evaluation. If approved, the drug will add to the options available for narcolepsy patients. “All of the available drugs have some limitations. Some have more abuse liability than others. Some have more robust wake-promoting properties than others. We haven’t done any head-to-head comparisons, so I can’t tell you how we will stack up,” Philip Jochelson, MD, said in an interview. Dr. Jochelson is vice president of clinical development at Jazz Pharmaceuticals and presented the results of the study at a poster session at the annual meeting of the American Neurological Association.
An earlier study showed the drug had less abuse potential than the schedule IV stimulant phentermine. That’s not surprising given the drug’s mechanism of action, Dr. Jochelson said. Amphetamine-based drugs stimulate dopamine release, which can prompt a dopamine surge that people equate with a high, he said. Solriamfetol also affects dopamine, but it is a reuptake inhibitor, so it doesn’t produce a surge.
If the drug gains approval, it remains to be seen how it will be classified on the Drug Enforcement Agency Controlled Substance scale. “Where it will fall in that spectrum is speculative at this point,” said Dr. Jochelson.
In the current study, 236 adults (aged 18-75 years) with type 1 narcolepsy were randomized to once-daily placebo, 75 mg solriamfetol, 150 mg solriamfetol, or 300 mg solriamfetol; 27.1% of patients in the 300-mg group discontinued, compared with 7.3% in the 150-mg group, 16.9% in the 75-mg group, and 10.3% in the placebo group. The mean change from baseline on the Maintenance of Wakefulness Test was statistically significant in the 300-mg group (12.3 minutes vs. 2.1 minutes for placebo, P less than .0001) and the 150-mg group (9.8 minutes vs. 2.1 minutes, P less than .0001) but not the 75-mg group (4.7 minutes vs. 2.1 minutes).
The drug also outperformed placebo at week 12 on the Epworth Sleepiness Scale. The mean change in the 300-mg group was –6.4 vs. –1.6 for placebo (P less than .001), –5.4 in the 150-mg group (P less than .0001), and –3.8 in the 75-mg group (P less than .05).
By both Maintenance of Wakefulness Test and Epworth Sleepiness Scale measures, the 150-mg and 300-mg solriamfetol groups had statistically significant differences as early as week 1.
The drug had some adverse effects, which were expected based on its pharmacologic profile. These included increases in headache (5.1% with placebo, 10.2% with 75 mg, 23.7% with 150 mg, 30.5% with 300 mg), nausea (1.7% for placebo, 5.1% for 75 mg, 10.2% for 150 mg, 16.9% for 300 mg), anxiety (1.7% with placebo, 1.7% with 75 mg, 5.1% with 150 mg, 8.5% with 300 mg), and insomnia (0% for placebo, 3.4% for 75 mg, 0% for 150 mg, 5.1% for 300 mg). Other adverse events occurring in at least 5% of patients were decreased appetite, nasopharyngitis, and dry mouth.
The study was funded by Jazz Pharmaceuticals. Dr. Jochelson is an employee of Jazz.
SAN DIEGO – The selective dopamine and norepinephrine reuptake inhibitor solriamfetol is effective in reducing sleepiness in patients with narcolepsy, according to results of a phase 3 study.
At 150-mg and 300-mg doses, the drug had statistically significant effects on objective and subjective measures.
There are wake-promoting drugs available, such as amphetamine-related drugs that are often used off label, but addiction liability is a concern. The nonamphetamine modafinil has been approved by the Food and Drug Administration since 1998.
Jazz Pharmaceuticals is in the process of submitting solriamfetol for FDA evaluation. If approved, the drug will add to the options available for narcolepsy patients. “All of the available drugs have some limitations. Some have more abuse liability than others. Some have more robust wake-promoting properties than others. We haven’t done any head-to-head comparisons, so I can’t tell you how we will stack up,” Philip Jochelson, MD, said in an interview. Dr. Jochelson is vice president of clinical development at Jazz Pharmaceuticals and presented the results of the study at a poster session at the annual meeting of the American Neurological Association.
An earlier study showed the drug had less abuse potential than the schedule IV stimulant phentermine. That’s not surprising given the drug’s mechanism of action, Dr. Jochelson said. Amphetamine-based drugs stimulate dopamine release, which can prompt a dopamine surge that people equate with a high, he said. Solriamfetol also affects dopamine, but it is a reuptake inhibitor, so it doesn’t produce a surge.
If the drug gains approval, it remains to be seen how it will be classified on the Drug Enforcement Agency Controlled Substance scale. “Where it will fall in that spectrum is speculative at this point,” said Dr. Jochelson.
In the current study, 236 adults (aged 18-75 years) with type 1 narcolepsy were randomized to once-daily placebo, 75 mg solriamfetol, 150 mg solriamfetol, or 300 mg solriamfetol; 27.1% of patients in the 300-mg group discontinued, compared with 7.3% in the 150-mg group, 16.9% in the 75-mg group, and 10.3% in the placebo group. The mean change from baseline on the Maintenance of Wakefulness Test was statistically significant in the 300-mg group (12.3 minutes vs. 2.1 minutes for placebo, P less than .0001) and the 150-mg group (9.8 minutes vs. 2.1 minutes, P less than .0001) but not the 75-mg group (4.7 minutes vs. 2.1 minutes).
The drug also outperformed placebo at week 12 on the Epworth Sleepiness Scale. The mean change in the 300-mg group was –6.4 vs. –1.6 for placebo (P less than .001), –5.4 in the 150-mg group (P less than .0001), and –3.8 in the 75-mg group (P less than .05).
By both Maintenance of Wakefulness Test and Epworth Sleepiness Scale measures, the 150-mg and 300-mg solriamfetol groups had statistically significant differences as early as week 1.
The drug had some adverse effects, which were expected based on its pharmacologic profile. These included increases in headache (5.1% with placebo, 10.2% with 75 mg, 23.7% with 150 mg, 30.5% with 300 mg), nausea (1.7% for placebo, 5.1% for 75 mg, 10.2% for 150 mg, 16.9% for 300 mg), anxiety (1.7% with placebo, 1.7% with 75 mg, 5.1% with 150 mg, 8.5% with 300 mg), and insomnia (0% for placebo, 3.4% for 75 mg, 0% for 150 mg, 5.1% for 300 mg). Other adverse events occurring in at least 5% of patients were decreased appetite, nasopharyngitis, and dry mouth.
The study was funded by Jazz Pharmaceuticals. Dr. Jochelson is an employee of Jazz.
AT ANA 2017
Key clinical point: Solriamfetol outperformed placebo on both objective and subjective sleep measures.
Major finding: At 12 weeks, 150 mg increased Maintenance of Wakefulness Test score to 9.8 minutes, compared with 2.1 minutes in the placebo group.
Data source: A randomized, controlled trial (n = 236).
Disclosures: The study was funded by Jazz Pharmaceuticals. Dr. Jochelson is an employee of Jazz.
Aspirin responsiveness improved in some with OSA
Obstructive sleep apnea patients with endothelial dysfunction gained aspirin responsiveness after using continuous positive airway pressure (CPAP) therapy, according to the findings from a small study scheduled to be presented at CHEST 2017.
“Endothelial dysfunction is an important phenomenon implicated in cardiovascular morbidity in obstructive sleep apnea (OSA) patients. While it has been demonstrated that CPAP improves endothelial function, our understanding of the pathophysiologic links between CPAP therapy and cardiovascular outcomes remain limited,” researchers wrote in the study’s abstract.
The researchers examined 18 patients’ endothelial function before and after using CPAP therapy for a median of 37 days, along with the relationship between endothelial function and aspirin responsiveness in these same patients. All study participants had been recently diagnosed with moderate to severe OSA and underwent modified peripheral artery tonometry and platelet aggregometry before and after beginning CPAP therapy. Most of the patients (14) demonstrated aspirin resistance at baseline.
Endothelial dysfunction was defined as having a reactive hyperemia index (RHI) of less than or equal to 1.67, while aspirin resistance was defined as having a reading of at least 550 aspirin reaction units (ARU).
At baseline, the average RHI of patients was 1.79 (standard deviation = 0.3), with 8 of the patients having had endothelial dysfunction. Following CPAP use, patients’ mean RHI increased to 1.94 (SD = 0.36), and endothelial dysfunction was present in just 5 of the study participants.*
After using CPAP, those patients with endothelial dysfunction at baseline were responsive to aspirin, with their average ARU reading at 520 following therapy. In contrast, those patients with normal endothelial function at baseline remained resistant to aspirin following CPAP use, based on mean ARU values before and after therapy.
Lirim Krveshi, DO, of Danbury (Conn.) Hospital, is scheduled to present this study, “A Prospective Cohort Study of Endothelial Function and its Relationship to Aspirin Responsiveness in OSA Patients,” on Sunday, Oct. 29, at 1:45 p.m. in Convention Center, room 601A. This presentation is part of the Obstructive Sleep Apnea: Insights & Management session, running from 1:30 p.m. to 3:00 p.m.
The study’s authors reported no conflicts of interest.
*This article was updated Oct. 27, 2017.
Obstructive sleep apnea patients with endothelial dysfunction gained aspirin responsiveness after using continuous positive airway pressure (CPAP) therapy, according to the findings from a small study scheduled to be presented at CHEST 2017.
“Endothelial dysfunction is an important phenomenon implicated in cardiovascular morbidity in obstructive sleep apnea (OSA) patients. While it has been demonstrated that CPAP improves endothelial function, our understanding of the pathophysiologic links between CPAP therapy and cardiovascular outcomes remain limited,” researchers wrote in the study’s abstract.
The researchers examined 18 patients’ endothelial function before and after using CPAP therapy for a median of 37 days, along with the relationship between endothelial function and aspirin responsiveness in these same patients. All study participants had been recently diagnosed with moderate to severe OSA and underwent modified peripheral artery tonometry and platelet aggregometry before and after beginning CPAP therapy. Most of the patients (14) demonstrated aspirin resistance at baseline.
Endothelial dysfunction was defined as having a reactive hyperemia index (RHI) of less than or equal to 1.67, while aspirin resistance was defined as having a reading of at least 550 aspirin reaction units (ARU).
At baseline, the average RHI of patients was 1.79 (standard deviation = 0.3), with 8 of the patients having had endothelial dysfunction. Following CPAP use, patients’ mean RHI increased to 1.94 (SD = 0.36), and endothelial dysfunction was present in just 5 of the study participants.*
After using CPAP, those patients with endothelial dysfunction at baseline were responsive to aspirin, with their average ARU reading at 520 following therapy. In contrast, those patients with normal endothelial function at baseline remained resistant to aspirin following CPAP use, based on mean ARU values before and after therapy.
Lirim Krveshi, DO, of Danbury (Conn.) Hospital, is scheduled to present this study, “A Prospective Cohort Study of Endothelial Function and its Relationship to Aspirin Responsiveness in OSA Patients,” on Sunday, Oct. 29, at 1:45 p.m. in Convention Center, room 601A. This presentation is part of the Obstructive Sleep Apnea: Insights & Management session, running from 1:30 p.m. to 3:00 p.m.
The study’s authors reported no conflicts of interest.
*This article was updated Oct. 27, 2017.
Obstructive sleep apnea patients with endothelial dysfunction gained aspirin responsiveness after using continuous positive airway pressure (CPAP) therapy, according to the findings from a small study scheduled to be presented at CHEST 2017.
“Endothelial dysfunction is an important phenomenon implicated in cardiovascular morbidity in obstructive sleep apnea (OSA) patients. While it has been demonstrated that CPAP improves endothelial function, our understanding of the pathophysiologic links between CPAP therapy and cardiovascular outcomes remain limited,” researchers wrote in the study’s abstract.
The researchers examined 18 patients’ endothelial function before and after using CPAP therapy for a median of 37 days, along with the relationship between endothelial function and aspirin responsiveness in these same patients. All study participants had been recently diagnosed with moderate to severe OSA and underwent modified peripheral artery tonometry and platelet aggregometry before and after beginning CPAP therapy. Most of the patients (14) demonstrated aspirin resistance at baseline.
Endothelial dysfunction was defined as having a reactive hyperemia index (RHI) of less than or equal to 1.67, while aspirin resistance was defined as having a reading of at least 550 aspirin reaction units (ARU).
At baseline, the average RHI of patients was 1.79 (standard deviation = 0.3), with 8 of the patients having had endothelial dysfunction. Following CPAP use, patients’ mean RHI increased to 1.94 (SD = 0.36), and endothelial dysfunction was present in just 5 of the study participants.*
After using CPAP, those patients with endothelial dysfunction at baseline were responsive to aspirin, with their average ARU reading at 520 following therapy. In contrast, those patients with normal endothelial function at baseline remained resistant to aspirin following CPAP use, based on mean ARU values before and after therapy.
Lirim Krveshi, DO, of Danbury (Conn.) Hospital, is scheduled to present this study, “A Prospective Cohort Study of Endothelial Function and its Relationship to Aspirin Responsiveness in OSA Patients,” on Sunday, Oct. 29, at 1:45 p.m. in Convention Center, room 601A. This presentation is part of the Obstructive Sleep Apnea: Insights & Management session, running from 1:30 p.m. to 3:00 p.m.
The study’s authors reported no conflicts of interest.
*This article was updated Oct. 27, 2017.
FROM CHEST 2017
Key clinical point:
Major finding: The average aspirin reaction units reading for patients who had endothelial dysfunction at baseline was 520 following therapy.
Data source: A prospective cohort study of 18 patients with newly diagnosed moderate to severe OSA.
Disclosures: The study’s authors reported no conflicts of interest.
CHEST Physician’s planned coverage of CHEST 2017
CHEST Physician is providing on-site coverage of the CHEST annual meeting in Toronto from Oct. 29 through Nov. 1.
We are planning to share findings from the latest research on treating COPD, sleep apnea, pulmonary hypertension, severe asthma, and other diseases that are part of pulmonary, critical care, and sleep medicine. Any improved methods for managing an ICU and updated recommendations on screening for lung cancer will also be on our radar.
The meeting’s agenda includes presentations of hundreds of study abstracts, and we thought you would be interested in hearing which ones grabbed the attention of some of CHEST Physician’s editorial advisory board members.
Board member Susan L. Millard, MD, FCCP, suggested attendees check out presentations of the following two studies:
- Impact of Race on Quality of Life of Families of Children with Asthma when Asthma Guidelines are Followed: Long-Term Follow-Up
- Results Of A Phase 3, Multicenter, Randomized, Placebo-controlled Trial of Remimazolam: A New Ultra Short Acting Benzodiazepine for Bronchoscopy
The first study is part of a session entitled Pediatrics, scheduled to run from 3:15 to 4:15 p.m. on Sunday, Oct. 29, in Convention Center - 606. Shahid Sheikh, MD, of Nationwide Children’s Hospital in New Albany, Ohio, is scheduled to present the abstract at 4:00 p.m.
Dr. Millard, who is Therapeutic Development Network director for the Pediatric CF Care Center and director of research for pediatric pulmonary and sleep medicine at the Helen DeVos Children’s Hospital in Grand Rapids, Mich., noted that she is interested in Dr. Sheikh’s research, “because cultural diversity is such a hot topic in general.”
Her other recommendation is part of the Late Breaking Abstracts 2 session, scheduled to occur on Wednesday, Nov. 1, from 2:45 to 4:15 p.m. in Convention Center - 603. CHEST President, Gerard A. Silvestri, MD, MS, FCCP, will present the abstract at 4:00 p.m.
Dr. Millard said she is interested in this study, because new drug options are so helpful for the frequently performed bronchoscopy.
Two sleep medicine experts on CHEST Physician’s editorial advisory board also selected a few presentations they expect to be newsworthy.
David Schulman, MD, MPH, FCCP, and professor of medicine at Emory University School of Medicine in Atlanta suggested CHEST Physician cover the following studies:
- Results of a Randomized, Placebo-Controlled, Double-Blind, 12-Week, Multicenter Study of JZP-110 for the Treatment Of Excessive Sleepiness in Patients with OSA, scheduled to be presented on Sunday, Oct. 29, at 1:30 p.m. in Convention Center - 601A. Dr. Kingman Strohl, MD, FCCP, of University Hospitals Case Medical Center-Sleep Center in Shaker Heights, Ohio, will present this research during a session entitled, Obstructive Sleep Apnea: Insights & Management, running from 1:30 to 3:00 p.m.
- History of Sleep Apnea and Cardiovascular Disease may Portend Improved Mortality in Patients With Acute Ischemic Stroke, scheduled to be presented on Tuesday, Oct. 31, at 11:15 a.m., in Convention Center - 601A. Nura Festic will present this research during the session, “Sleep, Heart, Brain and More,” running from 11:00 a.m. to 12:15 p.m.
- Ischemic Preconditioning in OSA Patients Manifested after Surviving a Cardiac Arrest? John Moss, MD, of Jacksonville, Fla., will present this study on Tuesday, Oct. 31, at 11:30 a.m., in Convention Center - 601A as part of the session “Sleep, Heart, Brain and More.”
Krishna M. Sundar, MD, FCCP, also recommended that CHEST Physician cover “A Prospective Cohort Study of Endothelial Function and its Relationship to Aspirin Responsiveness in OSA Patients.” Lirim Krveshi is scheduled to present this study on Sunday, Oct. 29, at 1:45 p.m. in Convention Center - 601A. This presentation is part of the Obstructive Sleep Apnea: Insights & Management session.
Dr. Sundar is an associate clinical professor of pulmonary, critical care and sleep medicine and medical director of the Sleep-Wake Center at the University of Utah, Salt Lake City.
To view the full agenda of the CHEST annual meeting, visit: chestmeeting.chestnet.org.
Look for CHEST Physician’s coverage of CHEST 2017 on our conference coverage page.
CHEST Physician is providing on-site coverage of the CHEST annual meeting in Toronto from Oct. 29 through Nov. 1.
We are planning to share findings from the latest research on treating COPD, sleep apnea, pulmonary hypertension, severe asthma, and other diseases that are part of pulmonary, critical care, and sleep medicine. Any improved methods for managing an ICU and updated recommendations on screening for lung cancer will also be on our radar.
The meeting’s agenda includes presentations of hundreds of study abstracts, and we thought you would be interested in hearing which ones grabbed the attention of some of CHEST Physician’s editorial advisory board members.
Board member Susan L. Millard, MD, FCCP, suggested attendees check out presentations of the following two studies:
- Impact of Race on Quality of Life of Families of Children with Asthma when Asthma Guidelines are Followed: Long-Term Follow-Up
- Results Of A Phase 3, Multicenter, Randomized, Placebo-controlled Trial of Remimazolam: A New Ultra Short Acting Benzodiazepine for Bronchoscopy
The first study is part of a session entitled Pediatrics, scheduled to run from 3:15 to 4:15 p.m. on Sunday, Oct. 29, in Convention Center - 606. Shahid Sheikh, MD, of Nationwide Children’s Hospital in New Albany, Ohio, is scheduled to present the abstract at 4:00 p.m.
Dr. Millard, who is Therapeutic Development Network director for the Pediatric CF Care Center and director of research for pediatric pulmonary and sleep medicine at the Helen DeVos Children’s Hospital in Grand Rapids, Mich., noted that she is interested in Dr. Sheikh’s research, “because cultural diversity is such a hot topic in general.”
Her other recommendation is part of the Late Breaking Abstracts 2 session, scheduled to occur on Wednesday, Nov. 1, from 2:45 to 4:15 p.m. in Convention Center - 603. CHEST President, Gerard A. Silvestri, MD, MS, FCCP, will present the abstract at 4:00 p.m.
Dr. Millard said she is interested in this study, because new drug options are so helpful for the frequently performed bronchoscopy.
Two sleep medicine experts on CHEST Physician’s editorial advisory board also selected a few presentations they expect to be newsworthy.
David Schulman, MD, MPH, FCCP, and professor of medicine at Emory University School of Medicine in Atlanta suggested CHEST Physician cover the following studies:
- Results of a Randomized, Placebo-Controlled, Double-Blind, 12-Week, Multicenter Study of JZP-110 for the Treatment Of Excessive Sleepiness in Patients with OSA, scheduled to be presented on Sunday, Oct. 29, at 1:30 p.m. in Convention Center - 601A. Dr. Kingman Strohl, MD, FCCP, of University Hospitals Case Medical Center-Sleep Center in Shaker Heights, Ohio, will present this research during a session entitled, Obstructive Sleep Apnea: Insights & Management, running from 1:30 to 3:00 p.m.
- History of Sleep Apnea and Cardiovascular Disease may Portend Improved Mortality in Patients With Acute Ischemic Stroke, scheduled to be presented on Tuesday, Oct. 31, at 11:15 a.m., in Convention Center - 601A. Nura Festic will present this research during the session, “Sleep, Heart, Brain and More,” running from 11:00 a.m. to 12:15 p.m.
- Ischemic Preconditioning in OSA Patients Manifested after Surviving a Cardiac Arrest? John Moss, MD, of Jacksonville, Fla., will present this study on Tuesday, Oct. 31, at 11:30 a.m., in Convention Center - 601A as part of the session “Sleep, Heart, Brain and More.”
Krishna M. Sundar, MD, FCCP, also recommended that CHEST Physician cover “A Prospective Cohort Study of Endothelial Function and its Relationship to Aspirin Responsiveness in OSA Patients.” Lirim Krveshi is scheduled to present this study on Sunday, Oct. 29, at 1:45 p.m. in Convention Center - 601A. This presentation is part of the Obstructive Sleep Apnea: Insights & Management session.
Dr. Sundar is an associate clinical professor of pulmonary, critical care and sleep medicine and medical director of the Sleep-Wake Center at the University of Utah, Salt Lake City.
To view the full agenda of the CHEST annual meeting, visit: chestmeeting.chestnet.org.
Look for CHEST Physician’s coverage of CHEST 2017 on our conference coverage page.
CHEST Physician is providing on-site coverage of the CHEST annual meeting in Toronto from Oct. 29 through Nov. 1.
We are planning to share findings from the latest research on treating COPD, sleep apnea, pulmonary hypertension, severe asthma, and other diseases that are part of pulmonary, critical care, and sleep medicine. Any improved methods for managing an ICU and updated recommendations on screening for lung cancer will also be on our radar.
The meeting’s agenda includes presentations of hundreds of study abstracts, and we thought you would be interested in hearing which ones grabbed the attention of some of CHEST Physician’s editorial advisory board members.
Board member Susan L. Millard, MD, FCCP, suggested attendees check out presentations of the following two studies:
- Impact of Race on Quality of Life of Families of Children with Asthma when Asthma Guidelines are Followed: Long-Term Follow-Up
- Results Of A Phase 3, Multicenter, Randomized, Placebo-controlled Trial of Remimazolam: A New Ultra Short Acting Benzodiazepine for Bronchoscopy
The first study is part of a session entitled Pediatrics, scheduled to run from 3:15 to 4:15 p.m. on Sunday, Oct. 29, in Convention Center - 606. Shahid Sheikh, MD, of Nationwide Children’s Hospital in New Albany, Ohio, is scheduled to present the abstract at 4:00 p.m.
Dr. Millard, who is Therapeutic Development Network director for the Pediatric CF Care Center and director of research for pediatric pulmonary and sleep medicine at the Helen DeVos Children’s Hospital in Grand Rapids, Mich., noted that she is interested in Dr. Sheikh’s research, “because cultural diversity is such a hot topic in general.”
Her other recommendation is part of the Late Breaking Abstracts 2 session, scheduled to occur on Wednesday, Nov. 1, from 2:45 to 4:15 p.m. in Convention Center - 603. CHEST President, Gerard A. Silvestri, MD, MS, FCCP, will present the abstract at 4:00 p.m.
Dr. Millard said she is interested in this study, because new drug options are so helpful for the frequently performed bronchoscopy.
Two sleep medicine experts on CHEST Physician’s editorial advisory board also selected a few presentations they expect to be newsworthy.
David Schulman, MD, MPH, FCCP, and professor of medicine at Emory University School of Medicine in Atlanta suggested CHEST Physician cover the following studies:
- Results of a Randomized, Placebo-Controlled, Double-Blind, 12-Week, Multicenter Study of JZP-110 for the Treatment Of Excessive Sleepiness in Patients with OSA, scheduled to be presented on Sunday, Oct. 29, at 1:30 p.m. in Convention Center - 601A. Dr. Kingman Strohl, MD, FCCP, of University Hospitals Case Medical Center-Sleep Center in Shaker Heights, Ohio, will present this research during a session entitled, Obstructive Sleep Apnea: Insights & Management, running from 1:30 to 3:00 p.m.
- History of Sleep Apnea and Cardiovascular Disease may Portend Improved Mortality in Patients With Acute Ischemic Stroke, scheduled to be presented on Tuesday, Oct. 31, at 11:15 a.m., in Convention Center - 601A. Nura Festic will present this research during the session, “Sleep, Heart, Brain and More,” running from 11:00 a.m. to 12:15 p.m.
- Ischemic Preconditioning in OSA Patients Manifested after Surviving a Cardiac Arrest? John Moss, MD, of Jacksonville, Fla., will present this study on Tuesday, Oct. 31, at 11:30 a.m., in Convention Center - 601A as part of the session “Sleep, Heart, Brain and More.”
Krishna M. Sundar, MD, FCCP, also recommended that CHEST Physician cover “A Prospective Cohort Study of Endothelial Function and its Relationship to Aspirin Responsiveness in OSA Patients.” Lirim Krveshi is scheduled to present this study on Sunday, Oct. 29, at 1:45 p.m. in Convention Center - 601A. This presentation is part of the Obstructive Sleep Apnea: Insights & Management session.
Dr. Sundar is an associate clinical professor of pulmonary, critical care and sleep medicine and medical director of the Sleep-Wake Center at the University of Utah, Salt Lake City.
To view the full agenda of the CHEST annual meeting, visit: chestmeeting.chestnet.org.
Look for CHEST Physician’s coverage of CHEST 2017 on our conference coverage page.
FROM CHEST 2017
JZP-110 improves excessive sleepiness in OSA patients
The selective dopamine norepinephrine reuptake inhibitor JZP-110 was effective at treating excessive sleepiness in obstructive sleep apnea patients, according to an abstract on a study to be presented at the CHEST annual meeting.
In a 12-week, phase 3 trial, adult patients with obstructive sleep apnea were randomized to receive placebo or once-daily JZP-110 at dosages of 37.5 mg, 75 mg, 150 mg, or 300 mg. A total of 459 patients were included in the final analysis.
At the end of the trial, the mean reduction from baseline on a maintenance of wakefulness test was 0.2 minutes for patients in the placebo group, 4.7 minutes in the 37.5-mg group, 9.1 minutes in the 75-mg group, 11 minutes in the 150-mg group, and 13 minutes in the 300-mg group. Mean changes in Epworth Sleepiness Scale scores were –3.3 for patients in the placebo group, –5.1 in the 37.5-mg group, –5 in the 75-mg group, –7.7 in the 150-mg group, and –7.9 in the 300-mg group.
A significantly higher rate of patients in the 75-mg (72.4%), 150-mg (89.7%), and 300-mg (88.7%) groups improved on the Patient Global Impression of Change scale, compared with patients in the placebo (49.1%) and 37.5-mg (55.4%) groups.
Just under 68% of patients who received JZP-110 experienced at least one adverse event, compared with 47.9% of patients who received placebo. The most common adverse events were headache, nausea, decreased appetite, and anxiety. Six serious adverse events were reported over the study period, but none was related to JZP-110.
The study is scheduled to be presented on Sunday, Oct. 29, from 1:30 p.m. to 1:45 p.m. in Room 601A of the Toronto Convention Centre South Building as part of the “Obstructive Sleep Apnea: Insights & Management” session, which will run from 1:30 p.m. to 3 p.m.
The selective dopamine norepinephrine reuptake inhibitor JZP-110 was effective at treating excessive sleepiness in obstructive sleep apnea patients, according to an abstract on a study to be presented at the CHEST annual meeting.
In a 12-week, phase 3 trial, adult patients with obstructive sleep apnea were randomized to receive placebo or once-daily JZP-110 at dosages of 37.5 mg, 75 mg, 150 mg, or 300 mg. A total of 459 patients were included in the final analysis.
At the end of the trial, the mean reduction from baseline on a maintenance of wakefulness test was 0.2 minutes for patients in the placebo group, 4.7 minutes in the 37.5-mg group, 9.1 minutes in the 75-mg group, 11 minutes in the 150-mg group, and 13 minutes in the 300-mg group. Mean changes in Epworth Sleepiness Scale scores were –3.3 for patients in the placebo group, –5.1 in the 37.5-mg group, –5 in the 75-mg group, –7.7 in the 150-mg group, and –7.9 in the 300-mg group.
A significantly higher rate of patients in the 75-mg (72.4%), 150-mg (89.7%), and 300-mg (88.7%) groups improved on the Patient Global Impression of Change scale, compared with patients in the placebo (49.1%) and 37.5-mg (55.4%) groups.
Just under 68% of patients who received JZP-110 experienced at least one adverse event, compared with 47.9% of patients who received placebo. The most common adverse events were headache, nausea, decreased appetite, and anxiety. Six serious adverse events were reported over the study period, but none was related to JZP-110.
The study is scheduled to be presented on Sunday, Oct. 29, from 1:30 p.m. to 1:45 p.m. in Room 601A of the Toronto Convention Centre South Building as part of the “Obstructive Sleep Apnea: Insights & Management” session, which will run from 1:30 p.m. to 3 p.m.
The selective dopamine norepinephrine reuptake inhibitor JZP-110 was effective at treating excessive sleepiness in obstructive sleep apnea patients, according to an abstract on a study to be presented at the CHEST annual meeting.
In a 12-week, phase 3 trial, adult patients with obstructive sleep apnea were randomized to receive placebo or once-daily JZP-110 at dosages of 37.5 mg, 75 mg, 150 mg, or 300 mg. A total of 459 patients were included in the final analysis.
At the end of the trial, the mean reduction from baseline on a maintenance of wakefulness test was 0.2 minutes for patients in the placebo group, 4.7 minutes in the 37.5-mg group, 9.1 minutes in the 75-mg group, 11 minutes in the 150-mg group, and 13 minutes in the 300-mg group. Mean changes in Epworth Sleepiness Scale scores were –3.3 for patients in the placebo group, –5.1 in the 37.5-mg group, –5 in the 75-mg group, –7.7 in the 150-mg group, and –7.9 in the 300-mg group.
A significantly higher rate of patients in the 75-mg (72.4%), 150-mg (89.7%), and 300-mg (88.7%) groups improved on the Patient Global Impression of Change scale, compared with patients in the placebo (49.1%) and 37.5-mg (55.4%) groups.
Just under 68% of patients who received JZP-110 experienced at least one adverse event, compared with 47.9% of patients who received placebo. The most common adverse events were headache, nausea, decreased appetite, and anxiety. Six serious adverse events were reported over the study period, but none was related to JZP-110.
The study is scheduled to be presented on Sunday, Oct. 29, from 1:30 p.m. to 1:45 p.m. in Room 601A of the Toronto Convention Centre South Building as part of the “Obstructive Sleep Apnea: Insights & Management” session, which will run from 1:30 p.m. to 3 p.m.
FROM CHEST 2017
CBT for insomnia and hot flashes lifts mood in midlife
PHILADELPHIA – Cognitive-behavioral therapy tailored to perimenopausal and postmenopausal women who were experiencing both insomnia and vasomotor symptoms effectively improved both sleep and mood, according to a small controlled study.
When 40 women were randomized to receive either cognitive-behavioral therapy for menopausal insomnia (CBTMI) or education about menopause and sleep, those who received CBTMI had significantly reduced scores on both objective and subjective scales of depression, and their sleep also improved.
The CBTMI intervention was effective even for women with high scores on the depression scales at baseline, Sara Nowakowski, PhD, said at a top abstracts session at the annual meeting of the North American Menopause Society.
Over the 8 weeks of the study intervention, women in the CBTMI arm received four 50-minute individual sessions with either a social worker or a psychologist in a gynecology clinic outpatient setting. Counseling during the sessions focused both on hot flashes and insomnia, using evidence-based CBT techniques to address both. These included sleep restriction, changing behaviors to strengthen the association of the bed with sleep, cognitive therapy to address maladaptive beliefs about both sleep and host flashes, general sleep hygiene, hot flash coping mechanisms, and relaxation training.
Those in the menopause education control arm had a 1-hour educational session about menopausal symptoms and sleep hygiene, received written material, and were told they could make any changes desired.
Participants, whose mean age was 55 years, were included if they reported at least one nocturnal hot flash per night and met criteria for the sleep disorder of insomnia. Although patients who met criteria for major depression were not excluded, women with surgical menopause or cancer treatment–related menopause were excluded, as were those with substance use disorder, significant other psychiatric comorbidities, and those with obstructive sleep apnea or periodic limb movements/restless leg syndrome.
Dr. Nowakowski, a clinical psychologist in the department of obstetrics and gynecology, University of Texas, Galveston, and her colleagues administered the Insomnia Severity Index (ISI), the Center for Epidemiologic Studies Depression Scale (CES-D), and the Hamilton Depression Rating Scale (HDRS) both before and after the 8-week intervention.
The investigators used a mixed-models statistical analysis, finding a significant improvement over time during the study period in both patient-reported (P = .001) and clinician-assessed (P = .001) ratings of depression for the CBTMI group.
When the effect of the treatment arm was analyzed, CBTMI also offered significantly greater improvement in patient-reported (P = .009) and clinician-assessed (P = .022) depression ratings.
Patients were divided into high and low depression severity, with a score over 8 on the CES-D and 16 on the HDRS putting the participant into the high-severity category. Both groups had significant improvement on the ISI from baseline. “Treatment response for insomnia severity did not differ based on baseline depression severity,” Dr. Nowakowski said.
The efficacy of the relatively brief intervention has clinical relevance to those caring for the 39%-60% of women in midlife who have symptoms of insomnia and the 8%-40% of midlife women who report elevated depression symptoms. “Comprehensive interventions that simultaneously improve sleep and mood in midlife women are greatly needed,” she said.
The National Institutes of Health and the Hogg Foundation for Mental Health funded the study. Dr. Nowakowski reported no conflicts of interest.
koakes@frontlinemedcom.com
On Twitter @karioakes
PHILADELPHIA – Cognitive-behavioral therapy tailored to perimenopausal and postmenopausal women who were experiencing both insomnia and vasomotor symptoms effectively improved both sleep and mood, according to a small controlled study.
When 40 women were randomized to receive either cognitive-behavioral therapy for menopausal insomnia (CBTMI) or education about menopause and sleep, those who received CBTMI had significantly reduced scores on both objective and subjective scales of depression, and their sleep also improved.
The CBTMI intervention was effective even for women with high scores on the depression scales at baseline, Sara Nowakowski, PhD, said at a top abstracts session at the annual meeting of the North American Menopause Society.
Over the 8 weeks of the study intervention, women in the CBTMI arm received four 50-minute individual sessions with either a social worker or a psychologist in a gynecology clinic outpatient setting. Counseling during the sessions focused both on hot flashes and insomnia, using evidence-based CBT techniques to address both. These included sleep restriction, changing behaviors to strengthen the association of the bed with sleep, cognitive therapy to address maladaptive beliefs about both sleep and host flashes, general sleep hygiene, hot flash coping mechanisms, and relaxation training.
Those in the menopause education control arm had a 1-hour educational session about menopausal symptoms and sleep hygiene, received written material, and were told they could make any changes desired.
Participants, whose mean age was 55 years, were included if they reported at least one nocturnal hot flash per night and met criteria for the sleep disorder of insomnia. Although patients who met criteria for major depression were not excluded, women with surgical menopause or cancer treatment–related menopause were excluded, as were those with substance use disorder, significant other psychiatric comorbidities, and those with obstructive sleep apnea or periodic limb movements/restless leg syndrome.
Dr. Nowakowski, a clinical psychologist in the department of obstetrics and gynecology, University of Texas, Galveston, and her colleagues administered the Insomnia Severity Index (ISI), the Center for Epidemiologic Studies Depression Scale (CES-D), and the Hamilton Depression Rating Scale (HDRS) both before and after the 8-week intervention.
The investigators used a mixed-models statistical analysis, finding a significant improvement over time during the study period in both patient-reported (P = .001) and clinician-assessed (P = .001) ratings of depression for the CBTMI group.
When the effect of the treatment arm was analyzed, CBTMI also offered significantly greater improvement in patient-reported (P = .009) and clinician-assessed (P = .022) depression ratings.
Patients were divided into high and low depression severity, with a score over 8 on the CES-D and 16 on the HDRS putting the participant into the high-severity category. Both groups had significant improvement on the ISI from baseline. “Treatment response for insomnia severity did not differ based on baseline depression severity,” Dr. Nowakowski said.
The efficacy of the relatively brief intervention has clinical relevance to those caring for the 39%-60% of women in midlife who have symptoms of insomnia and the 8%-40% of midlife women who report elevated depression symptoms. “Comprehensive interventions that simultaneously improve sleep and mood in midlife women are greatly needed,” she said.
The National Institutes of Health and the Hogg Foundation for Mental Health funded the study. Dr. Nowakowski reported no conflicts of interest.
koakes@frontlinemedcom.com
On Twitter @karioakes
PHILADELPHIA – Cognitive-behavioral therapy tailored to perimenopausal and postmenopausal women who were experiencing both insomnia and vasomotor symptoms effectively improved both sleep and mood, according to a small controlled study.
When 40 women were randomized to receive either cognitive-behavioral therapy for menopausal insomnia (CBTMI) or education about menopause and sleep, those who received CBTMI had significantly reduced scores on both objective and subjective scales of depression, and their sleep also improved.
The CBTMI intervention was effective even for women with high scores on the depression scales at baseline, Sara Nowakowski, PhD, said at a top abstracts session at the annual meeting of the North American Menopause Society.
Over the 8 weeks of the study intervention, women in the CBTMI arm received four 50-minute individual sessions with either a social worker or a psychologist in a gynecology clinic outpatient setting. Counseling during the sessions focused both on hot flashes and insomnia, using evidence-based CBT techniques to address both. These included sleep restriction, changing behaviors to strengthen the association of the bed with sleep, cognitive therapy to address maladaptive beliefs about both sleep and host flashes, general sleep hygiene, hot flash coping mechanisms, and relaxation training.
Those in the menopause education control arm had a 1-hour educational session about menopausal symptoms and sleep hygiene, received written material, and were told they could make any changes desired.
Participants, whose mean age was 55 years, were included if they reported at least one nocturnal hot flash per night and met criteria for the sleep disorder of insomnia. Although patients who met criteria for major depression were not excluded, women with surgical menopause or cancer treatment–related menopause were excluded, as were those with substance use disorder, significant other psychiatric comorbidities, and those with obstructive sleep apnea or periodic limb movements/restless leg syndrome.
Dr. Nowakowski, a clinical psychologist in the department of obstetrics and gynecology, University of Texas, Galveston, and her colleagues administered the Insomnia Severity Index (ISI), the Center for Epidemiologic Studies Depression Scale (CES-D), and the Hamilton Depression Rating Scale (HDRS) both before and after the 8-week intervention.
The investigators used a mixed-models statistical analysis, finding a significant improvement over time during the study period in both patient-reported (P = .001) and clinician-assessed (P = .001) ratings of depression for the CBTMI group.
When the effect of the treatment arm was analyzed, CBTMI also offered significantly greater improvement in patient-reported (P = .009) and clinician-assessed (P = .022) depression ratings.
Patients were divided into high and low depression severity, with a score over 8 on the CES-D and 16 on the HDRS putting the participant into the high-severity category. Both groups had significant improvement on the ISI from baseline. “Treatment response for insomnia severity did not differ based on baseline depression severity,” Dr. Nowakowski said.
The efficacy of the relatively brief intervention has clinical relevance to those caring for the 39%-60% of women in midlife who have symptoms of insomnia and the 8%-40% of midlife women who report elevated depression symptoms. “Comprehensive interventions that simultaneously improve sleep and mood in midlife women are greatly needed,” she said.
The National Institutes of Health and the Hogg Foundation for Mental Health funded the study. Dr. Nowakowski reported no conflicts of interest.
koakes@frontlinemedcom.com
On Twitter @karioakes
AT NAMS 2017
Key clinical point:
Major finding: Patient-reported and clinician-assessed depression scores dropped after the intervention (P = .001 for both).
Data source: Randomized controlled trial of 40 midlife women with insomnia and hot flashes.
Disclosures: The National Institutes of Health and the Hogg Foundation for Mental Health funded the study. Dr. Nowakowski reported no conflicts of interest.
Slow-wave sleep linked to Parkinson’s disease cognition
SAN DIEGO – Slow-wave sleep improves cognition in Parkinson’s disease, according to University of Alabama at Birmingham investigators.
After sleep studies, they compared the cognitive performance of 16 patients who spent more than 14% of their sleep time in slow-wave (SW) sleep with the cognitive performance of 16 who spent less than 14% in SW sleep; 13 of the patients in the low SW group (81%) met the criteria for mild Parkinson’s disease (PD) cognitive impairment versus 7 of the patients in the higher SW sleep group (44%); the patients were well matched for age, sex, disease duration, and other variables.
The normative value for SW sleep is about 15%-20%, although people spend less time in SW sleep as they age. The study subjects were in their mid-60s on average, so the 14% cut point wasn’t too far off from what might be considered typical.
More SW sleep in PD was associated with better performance on measures of global function, attention/working memory, executive function, and language comprehension. Patients in the low SW group, for instance, performed 0.25 standard deviations below the normative mean on attention/working memory tests, while subjects in the high SW group performed 0.5 standard deviations above, meaning that they outperformed people in their age group who didn’t have PD. These differences were statistically significant.
It raises the question of what can be done to help PD patients sleep better. “I’m interested in exercise to improve sleep, and we have some primary data that show it’s helpful for sleep in general but also SW sleep,” said Dr. Amara. Sodium oxybate (Xyrem) might also help, but for now, it’s a controlled substance approved only for narcolepsy. “We might have to branch out and try something novel,” she said.
The next step is to “see if we can use SW sleep to predict who might have cognitive declines and find interventions to [prevent] it,” she said.
Patients in the study had been diagnosed with PD for a mean of about 7 years. People in the high SW group were on lower amounts of levodopa equivalents, however, the investigators controlled for that, as well as for the fact that women tend to spend more time in SW sleep than men. There were no differences between the groups in measures of movement problems and of subjective scores of sleep quality and daytime sleepiness.
People with high SW sleep fell asleep sooner than did those in the low SW groups, about 9 minutes versus 20 minutes. There was no correlation between visual-spatial function and SW sleep, but visual-spatial function did correlate with the amount of time spent in rapid eye movement sleep, suggesting that dreaming might be important for visual-spatial function, Dr. Amara said.
The work was funded by the National Institutes of Health. Dr. Amara had no relevant disclosures.
SAN DIEGO – Slow-wave sleep improves cognition in Parkinson’s disease, according to University of Alabama at Birmingham investigators.
After sleep studies, they compared the cognitive performance of 16 patients who spent more than 14% of their sleep time in slow-wave (SW) sleep with the cognitive performance of 16 who spent less than 14% in SW sleep; 13 of the patients in the low SW group (81%) met the criteria for mild Parkinson’s disease (PD) cognitive impairment versus 7 of the patients in the higher SW sleep group (44%); the patients were well matched for age, sex, disease duration, and other variables.
The normative value for SW sleep is about 15%-20%, although people spend less time in SW sleep as they age. The study subjects were in their mid-60s on average, so the 14% cut point wasn’t too far off from what might be considered typical.
More SW sleep in PD was associated with better performance on measures of global function, attention/working memory, executive function, and language comprehension. Patients in the low SW group, for instance, performed 0.25 standard deviations below the normative mean on attention/working memory tests, while subjects in the high SW group performed 0.5 standard deviations above, meaning that they outperformed people in their age group who didn’t have PD. These differences were statistically significant.
It raises the question of what can be done to help PD patients sleep better. “I’m interested in exercise to improve sleep, and we have some primary data that show it’s helpful for sleep in general but also SW sleep,” said Dr. Amara. Sodium oxybate (Xyrem) might also help, but for now, it’s a controlled substance approved only for narcolepsy. “We might have to branch out and try something novel,” she said.
The next step is to “see if we can use SW sleep to predict who might have cognitive declines and find interventions to [prevent] it,” she said.
Patients in the study had been diagnosed with PD for a mean of about 7 years. People in the high SW group were on lower amounts of levodopa equivalents, however, the investigators controlled for that, as well as for the fact that women tend to spend more time in SW sleep than men. There were no differences between the groups in measures of movement problems and of subjective scores of sleep quality and daytime sleepiness.
People with high SW sleep fell asleep sooner than did those in the low SW groups, about 9 minutes versus 20 minutes. There was no correlation between visual-spatial function and SW sleep, but visual-spatial function did correlate with the amount of time spent in rapid eye movement sleep, suggesting that dreaming might be important for visual-spatial function, Dr. Amara said.
The work was funded by the National Institutes of Health. Dr. Amara had no relevant disclosures.
SAN DIEGO – Slow-wave sleep improves cognition in Parkinson’s disease, according to University of Alabama at Birmingham investigators.
After sleep studies, they compared the cognitive performance of 16 patients who spent more than 14% of their sleep time in slow-wave (SW) sleep with the cognitive performance of 16 who spent less than 14% in SW sleep; 13 of the patients in the low SW group (81%) met the criteria for mild Parkinson’s disease (PD) cognitive impairment versus 7 of the patients in the higher SW sleep group (44%); the patients were well matched for age, sex, disease duration, and other variables.
The normative value for SW sleep is about 15%-20%, although people spend less time in SW sleep as they age. The study subjects were in their mid-60s on average, so the 14% cut point wasn’t too far off from what might be considered typical.
More SW sleep in PD was associated with better performance on measures of global function, attention/working memory, executive function, and language comprehension. Patients in the low SW group, for instance, performed 0.25 standard deviations below the normative mean on attention/working memory tests, while subjects in the high SW group performed 0.5 standard deviations above, meaning that they outperformed people in their age group who didn’t have PD. These differences were statistically significant.
It raises the question of what can be done to help PD patients sleep better. “I’m interested in exercise to improve sleep, and we have some primary data that show it’s helpful for sleep in general but also SW sleep,” said Dr. Amara. Sodium oxybate (Xyrem) might also help, but for now, it’s a controlled substance approved only for narcolepsy. “We might have to branch out and try something novel,” she said.
The next step is to “see if we can use SW sleep to predict who might have cognitive declines and find interventions to [prevent] it,” she said.
Patients in the study had been diagnosed with PD for a mean of about 7 years. People in the high SW group were on lower amounts of levodopa equivalents, however, the investigators controlled for that, as well as for the fact that women tend to spend more time in SW sleep than men. There were no differences between the groups in measures of movement problems and of subjective scores of sleep quality and daytime sleepiness.
People with high SW sleep fell asleep sooner than did those in the low SW groups, about 9 minutes versus 20 minutes. There was no correlation between visual-spatial function and SW sleep, but visual-spatial function did correlate with the amount of time spent in rapid eye movement sleep, suggesting that dreaming might be important for visual-spatial function, Dr. Amara said.
The work was funded by the National Institutes of Health. Dr. Amara had no relevant disclosures.
AT ANA 2017
Key clinical point:
Major finding: Thirteen patients in the low SW group (81%) met criteria for mild Parkinson’s disease (PD) cognitive impairment versus seven (44%) in the higher SW sleep group.
Data source: Cognitive testing of 32 patients with PD stratified by amount of SW sleep
Disclosures: The work was funded by the National Institutes of Health. The presenter had no relevant disclosures.
Phrenic nerve stimulator shows heart failure benefits
DALLAS– Heart failure patients with central sleep apnea who received treatment with a transvenous phrenic nerve–stimulating device showed dramatic improvement in their global self-assessment, compared with control patients, in a subgroup analysis of 80 patients enrolled in the device’s pivotal trial.
Among 35 patients with heart failure enrolled in the remedē System pivotal trial and treated for 6 months with phrenic nerve stimulation, 57% reported that they had “markedly” or “moderately” improved, compared with a 9% rate for this self-rating among 44 control heart failure patients in the trial, a statistically significant difference, Lee R. Goldberg, MD, said at the annual scientific meeting of the Heart Failure Society of America.
Further analysis focused on echocardiographic examinations after 12 months in 23 of the heart failure patients who entered the study with a left ventricular ejection fraction of 45% or less and received 12 months of phrenic nerve stimulation. The average LVEF rose in these patients from 30% at baseline to 35%, a statistically significant difference, and left ventricular end systolic volume fell by an average of almost 11 mL from baseline, a difference just short of statistical significance, findings Dr. Goldberg called “a little exciting.”
“It is very encouraging to see some evidence for ventricular remodeling,” commented Lynne W. Stevenson, MD, professor of medicine and a heart failure specialist at Vanderbilt University in Nashville, Tenn.
The FDA approved the use of this device for the treatment of moderate to severe central sleep apnea on Oct. 6. “I think we would use it” in heart failure patients with intolerable symptoms from central sleep apnea, Dr. Goldberg said in an interview during the meeting.
“There is a tight connection between sleep-disordered breathing, sleep apnea, heart failure, and cardiovascular disease, and we have been pretty aggressive in trying to treat the sleep apnea. Even if phrenic nerve stimulation just improves patients’ quality of life and is neutral for other outcomes,” it would be reasonable to offer it to patients, he said. “But many of us think there is a bigger connection that results in a therapeutic benefit [to heart failure patients] by treating their central sleep apnea.”
The pivotal trial enrolled a total of 151 patients with central sleep apnea at 31 centers in Germany, Poland, and the United States who were selected based on having an apnea-hypopnea index of at least 20 events per hour. All participants received a transvenous phrenic nerve–stimulator implant, and then randomization assigned 73 patients to have the device turned on for the first 6 months while 78 device recipients had their devices left off to serve as controls. The study’s primary efficacy endpoint was the percentage of patients having at least a 50% cut in their apnea-hypopnea index, which happened in 51% of evaluable patients in the active treatment arm and in 11% of the evaluable controls. The primary results were published last year (Lancet. 2016 Sep 3;388[10048]974-82).
“We hope this treatment will have the collateral effect of improving cardiovascular disease outcomes, but we don’t know that yet. The initial target will be patients with apnea-hypopnea episodes that affect their quality of life,” Dr. Goldberg said.
The apparent safety of this approach for treating central sleep apnea may relate to its mechanism of action, he suggested. The mortality-boosting effect of adaptive servo-ventilation may correlate with the positive pressure it creates in a patient’s chest that perhaps causes myocardial stress or hemodynamic problems. In contrast, phrenic nerve stimulation produces diaphragm motion that mimics normal breathing and creates negative chest pressure. “A lot of hypothesis generation needs to happen to better understand the underlying physiology,” Dr. Goldberg conceded.
At the end of the 6-month period that compared active treatment with control, the heart failure subgroup also showed statistically significant benefits from treatment for several sleep metrics, including apnea-hypopnea index, the central apnea index, and oxygen desaturation, and also for daytime sleepiness measured on the Epworth Sleepiness Scale. After 12 months on active treatment, patients also showed a significant improvement over baseline in their score on the Minnesota Living With Heart Failure Questionnaire, Dr. Goldberg reported.
The trial was sponsored by Respicardia, the company developing the remedē System. Dr. Goldberg has been a consultant to and has received research funding from Respicardia. Dr. Stevenson had no relevant disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
DALLAS– Heart failure patients with central sleep apnea who received treatment with a transvenous phrenic nerve–stimulating device showed dramatic improvement in their global self-assessment, compared with control patients, in a subgroup analysis of 80 patients enrolled in the device’s pivotal trial.
Among 35 patients with heart failure enrolled in the remedē System pivotal trial and treated for 6 months with phrenic nerve stimulation, 57% reported that they had “markedly” or “moderately” improved, compared with a 9% rate for this self-rating among 44 control heart failure patients in the trial, a statistically significant difference, Lee R. Goldberg, MD, said at the annual scientific meeting of the Heart Failure Society of America.
Further analysis focused on echocardiographic examinations after 12 months in 23 of the heart failure patients who entered the study with a left ventricular ejection fraction of 45% or less and received 12 months of phrenic nerve stimulation. The average LVEF rose in these patients from 30% at baseline to 35%, a statistically significant difference, and left ventricular end systolic volume fell by an average of almost 11 mL from baseline, a difference just short of statistical significance, findings Dr. Goldberg called “a little exciting.”
“It is very encouraging to see some evidence for ventricular remodeling,” commented Lynne W. Stevenson, MD, professor of medicine and a heart failure specialist at Vanderbilt University in Nashville, Tenn.
The FDA approved the use of this device for the treatment of moderate to severe central sleep apnea on Oct. 6. “I think we would use it” in heart failure patients with intolerable symptoms from central sleep apnea, Dr. Goldberg said in an interview during the meeting.
“There is a tight connection between sleep-disordered breathing, sleep apnea, heart failure, and cardiovascular disease, and we have been pretty aggressive in trying to treat the sleep apnea. Even if phrenic nerve stimulation just improves patients’ quality of life and is neutral for other outcomes,” it would be reasonable to offer it to patients, he said. “But many of us think there is a bigger connection that results in a therapeutic benefit [to heart failure patients] by treating their central sleep apnea.”
The pivotal trial enrolled a total of 151 patients with central sleep apnea at 31 centers in Germany, Poland, and the United States who were selected based on having an apnea-hypopnea index of at least 20 events per hour. All participants received a transvenous phrenic nerve–stimulator implant, and then randomization assigned 73 patients to have the device turned on for the first 6 months while 78 device recipients had their devices left off to serve as controls. The study’s primary efficacy endpoint was the percentage of patients having at least a 50% cut in their apnea-hypopnea index, which happened in 51% of evaluable patients in the active treatment arm and in 11% of the evaluable controls. The primary results were published last year (Lancet. 2016 Sep 3;388[10048]974-82).
“We hope this treatment will have the collateral effect of improving cardiovascular disease outcomes, but we don’t know that yet. The initial target will be patients with apnea-hypopnea episodes that affect their quality of life,” Dr. Goldberg said.
The apparent safety of this approach for treating central sleep apnea may relate to its mechanism of action, he suggested. The mortality-boosting effect of adaptive servo-ventilation may correlate with the positive pressure it creates in a patient’s chest that perhaps causes myocardial stress or hemodynamic problems. In contrast, phrenic nerve stimulation produces diaphragm motion that mimics normal breathing and creates negative chest pressure. “A lot of hypothesis generation needs to happen to better understand the underlying physiology,” Dr. Goldberg conceded.
At the end of the 6-month period that compared active treatment with control, the heart failure subgroup also showed statistically significant benefits from treatment for several sleep metrics, including apnea-hypopnea index, the central apnea index, and oxygen desaturation, and also for daytime sleepiness measured on the Epworth Sleepiness Scale. After 12 months on active treatment, patients also showed a significant improvement over baseline in their score on the Minnesota Living With Heart Failure Questionnaire, Dr. Goldberg reported.
The trial was sponsored by Respicardia, the company developing the remedē System. Dr. Goldberg has been a consultant to and has received research funding from Respicardia. Dr. Stevenson had no relevant disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
DALLAS– Heart failure patients with central sleep apnea who received treatment with a transvenous phrenic nerve–stimulating device showed dramatic improvement in their global self-assessment, compared with control patients, in a subgroup analysis of 80 patients enrolled in the device’s pivotal trial.
Among 35 patients with heart failure enrolled in the remedē System pivotal trial and treated for 6 months with phrenic nerve stimulation, 57% reported that they had “markedly” or “moderately” improved, compared with a 9% rate for this self-rating among 44 control heart failure patients in the trial, a statistically significant difference, Lee R. Goldberg, MD, said at the annual scientific meeting of the Heart Failure Society of America.
Further analysis focused on echocardiographic examinations after 12 months in 23 of the heart failure patients who entered the study with a left ventricular ejection fraction of 45% or less and received 12 months of phrenic nerve stimulation. The average LVEF rose in these patients from 30% at baseline to 35%, a statistically significant difference, and left ventricular end systolic volume fell by an average of almost 11 mL from baseline, a difference just short of statistical significance, findings Dr. Goldberg called “a little exciting.”
“It is very encouraging to see some evidence for ventricular remodeling,” commented Lynne W. Stevenson, MD, professor of medicine and a heart failure specialist at Vanderbilt University in Nashville, Tenn.
The FDA approved the use of this device for the treatment of moderate to severe central sleep apnea on Oct. 6. “I think we would use it” in heart failure patients with intolerable symptoms from central sleep apnea, Dr. Goldberg said in an interview during the meeting.
“There is a tight connection between sleep-disordered breathing, sleep apnea, heart failure, and cardiovascular disease, and we have been pretty aggressive in trying to treat the sleep apnea. Even if phrenic nerve stimulation just improves patients’ quality of life and is neutral for other outcomes,” it would be reasonable to offer it to patients, he said. “But many of us think there is a bigger connection that results in a therapeutic benefit [to heart failure patients] by treating their central sleep apnea.”
The pivotal trial enrolled a total of 151 patients with central sleep apnea at 31 centers in Germany, Poland, and the United States who were selected based on having an apnea-hypopnea index of at least 20 events per hour. All participants received a transvenous phrenic nerve–stimulator implant, and then randomization assigned 73 patients to have the device turned on for the first 6 months while 78 device recipients had their devices left off to serve as controls. The study’s primary efficacy endpoint was the percentage of patients having at least a 50% cut in their apnea-hypopnea index, which happened in 51% of evaluable patients in the active treatment arm and in 11% of the evaluable controls. The primary results were published last year (Lancet. 2016 Sep 3;388[10048]974-82).
“We hope this treatment will have the collateral effect of improving cardiovascular disease outcomes, but we don’t know that yet. The initial target will be patients with apnea-hypopnea episodes that affect their quality of life,” Dr. Goldberg said.
The apparent safety of this approach for treating central sleep apnea may relate to its mechanism of action, he suggested. The mortality-boosting effect of adaptive servo-ventilation may correlate with the positive pressure it creates in a patient’s chest that perhaps causes myocardial stress or hemodynamic problems. In contrast, phrenic nerve stimulation produces diaphragm motion that mimics normal breathing and creates negative chest pressure. “A lot of hypothesis generation needs to happen to better understand the underlying physiology,” Dr. Goldberg conceded.
At the end of the 6-month period that compared active treatment with control, the heart failure subgroup also showed statistically significant benefits from treatment for several sleep metrics, including apnea-hypopnea index, the central apnea index, and oxygen desaturation, and also for daytime sleepiness measured on the Epworth Sleepiness Scale. After 12 months on active treatment, patients also showed a significant improvement over baseline in their score on the Minnesota Living With Heart Failure Questionnaire, Dr. Goldberg reported.
The trial was sponsored by Respicardia, the company developing the remedē System. Dr. Goldberg has been a consultant to and has received research funding from Respicardia. Dr. Stevenson had no relevant disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE HFSA ANNUAL SCIENTIFIC MEETING
Key clinical point:
Major finding: Patient-reported global assessment improved markedly or moderately in 57% of treated patients and in 9% of controls.
Data source: Subgroup analysis of the remedē System pivotal trial.
Disclosures: The trial was sponsored by Respicardia, the company developing the remedē System. Dr. Goldberg has been a consultant to and has received research funding from Respicardia. Dr. Stevenson had no relevant disclosures.
FDA approves implantable device for central sleep apnea
The U.S. Food and Drug Administration on Oct. 6 approved an implantable device for the treatment of moderate to severe central sleep apnea.
The remedē System consists of a battery pack and small, thin wires placed under the skin in the upper chest area. The wires are inserted into the blood vessels in the chest to stimulate the phrenic nerve. The system monitors respiratory signals and, when it stimulates the nerve, the diaphragm moves to restore normal breathing.
The agency’s approval comes on the basis of study results showing that the system reduced the apnea–hypopnea index scores by 50% or more in 51% of patients studied. Control patients in the study saw an 11% reduction in their score.
Adverse events reported in the study included concomitant device interaction, implant site infection, and swelling and local tissue damage or pocket erosion. The remedē System is contraindicated for patients with active infection or who are known to require an MRI.
The U.S. Food and Drug Administration on Oct. 6 approved an implantable device for the treatment of moderate to severe central sleep apnea.
The remedē System consists of a battery pack and small, thin wires placed under the skin in the upper chest area. The wires are inserted into the blood vessels in the chest to stimulate the phrenic nerve. The system monitors respiratory signals and, when it stimulates the nerve, the diaphragm moves to restore normal breathing.
The agency’s approval comes on the basis of study results showing that the system reduced the apnea–hypopnea index scores by 50% or more in 51% of patients studied. Control patients in the study saw an 11% reduction in their score.
Adverse events reported in the study included concomitant device interaction, implant site infection, and swelling and local tissue damage or pocket erosion. The remedē System is contraindicated for patients with active infection or who are known to require an MRI.
The U.S. Food and Drug Administration on Oct. 6 approved an implantable device for the treatment of moderate to severe central sleep apnea.
The remedē System consists of a battery pack and small, thin wires placed under the skin in the upper chest area. The wires are inserted into the blood vessels in the chest to stimulate the phrenic nerve. The system monitors respiratory signals and, when it stimulates the nerve, the diaphragm moves to restore normal breathing.
The agency’s approval comes on the basis of study results showing that the system reduced the apnea–hypopnea index scores by 50% or more in 51% of patients studied. Control patients in the study saw an 11% reduction in their score.
Adverse events reported in the study included concomitant device interaction, implant site infection, and swelling and local tissue damage or pocket erosion. The remedē System is contraindicated for patients with active infection or who are known to require an MRI.
Sleep Duration Affects Likelihood of Insomnia and Depression Remission
BOSTON—Objective sleep duration moderates the probability of remission among patients with comorbid depression and insomnia, according to research presented at the 31st Annual Meeting of the Associated Professional Sleep Societies. Sleep durations of greater than five to six hours increase the likelihood that these patients will achieve insomnia remission with cognitive behavioral therapy for insomnia (CBT-I), but do not affect the likelihood of depression remission. Sleep durations of seven or more hours optimize the likelihood of insomnia remission and depression remission in response to CBT-I.
In a 2015 consensus statement, the Sleep Research Society recommended seven or more hours of sleep per night for adults younger than 60. Studies indicate that sleep durations of less than five hours and less than six hours are associated with increased morbidity and poor treatment response among patients with insomnia. “We wanted to know what [sleep-duration] cutoffs … might be better predictors of eventual insomnia and depression remission through treatment,” said Jack Edinger, PhD, Professor of Medicine at National Jewish Health in Denver.
An Analysis of the TRIAD Study
Dr. Edinger and colleagues conducted a secondary analysis of the TRIAD study, which examined whether combined treatment of depression and insomnia improves depression and sleep outcomes in participants with both disorders. Eligible participants met Diagnostic and Statistical Manual of Mental Disorders (4th ed.) criteria for major depression and primary insomnia, had a Hamilton Rating Scale for Depression (HAMD-17) score of 16 or greater, and had an Insomnia Severity Index (ISI) score of 11 or greater. People who had had psychotherapy in the previous four months, or had failed or could not tolerate previous adequate trials of the study medications, were excluded. Participants completed one night of baseline polysomnography before entering the treatment phase of the study.
The study population included 104 participants (75 women) with a mean age of 47. Mean baseline HAMD-17 score was 22, and mean baseline ISI score was 20.6. All participants received antidepressant medication (ie, citalopram, sertraline, or venlafaxine). Patients were randomized to CBT-I or sham (ie, a pseudodesensitization condition with sleep education). The investigators assessed participants biweekly with the HAMD-17 and the ISI. The treatment period lasted for 16 weeks.
CBT-I Provided Benefits
Participants with five or more hours of sleep were more likely to respond to CBT-I than participants with fewer than five hours of sleep. Among participants with sleep duration of five or more hours, insomnia remission was more likely with CBT-I than with the control condition. The five-hour cutoff had no association with depression remission.
Among participants with six or more hours of sleep, those who received CBT-I were more likely to achieve insomnia remission than controls. The six-hour cutoff did not affect the likelihood of depression remission, however.
Among participants with seven or more hours of sleep, those randomized to CBT-I were more likely to achieve insomnia remission and depression remission than controls.
“More research is needed to determine how best to achieve depression remission in those patients with less than seven hours of objective sleep duration prior to starting treatment,” Dr. Edinger concluded.
—Erik Greb
Suggested Reading
Bathgate CJ, Edinger JD, Krystal AD. Insomnia patients with objective short sleep duration have a blunted response to cognitive behavioral therapy for insomnia. Sleep. 2017;40(1).
Vgontzas AN, Liao D, Bixler EO, et al. Insomnia with objective short sleep duration is associated with a high risk for hypertension. Sleep. 2009;32(4):491-497.
Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: A joint consensus statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38(6):843-844.
BOSTON—Objective sleep duration moderates the probability of remission among patients with comorbid depression and insomnia, according to research presented at the 31st Annual Meeting of the Associated Professional Sleep Societies. Sleep durations of greater than five to six hours increase the likelihood that these patients will achieve insomnia remission with cognitive behavioral therapy for insomnia (CBT-I), but do not affect the likelihood of depression remission. Sleep durations of seven or more hours optimize the likelihood of insomnia remission and depression remission in response to CBT-I.
In a 2015 consensus statement, the Sleep Research Society recommended seven or more hours of sleep per night for adults younger than 60. Studies indicate that sleep durations of less than five hours and less than six hours are associated with increased morbidity and poor treatment response among patients with insomnia. “We wanted to know what [sleep-duration] cutoffs … might be better predictors of eventual insomnia and depression remission through treatment,” said Jack Edinger, PhD, Professor of Medicine at National Jewish Health in Denver.
An Analysis of the TRIAD Study
Dr. Edinger and colleagues conducted a secondary analysis of the TRIAD study, which examined whether combined treatment of depression and insomnia improves depression and sleep outcomes in participants with both disorders. Eligible participants met Diagnostic and Statistical Manual of Mental Disorders (4th ed.) criteria for major depression and primary insomnia, had a Hamilton Rating Scale for Depression (HAMD-17) score of 16 or greater, and had an Insomnia Severity Index (ISI) score of 11 or greater. People who had had psychotherapy in the previous four months, or had failed or could not tolerate previous adequate trials of the study medications, were excluded. Participants completed one night of baseline polysomnography before entering the treatment phase of the study.
The study population included 104 participants (75 women) with a mean age of 47. Mean baseline HAMD-17 score was 22, and mean baseline ISI score was 20.6. All participants received antidepressant medication (ie, citalopram, sertraline, or venlafaxine). Patients were randomized to CBT-I or sham (ie, a pseudodesensitization condition with sleep education). The investigators assessed participants biweekly with the HAMD-17 and the ISI. The treatment period lasted for 16 weeks.
CBT-I Provided Benefits
Participants with five or more hours of sleep were more likely to respond to CBT-I than participants with fewer than five hours of sleep. Among participants with sleep duration of five or more hours, insomnia remission was more likely with CBT-I than with the control condition. The five-hour cutoff had no association with depression remission.
Among participants with six or more hours of sleep, those who received CBT-I were more likely to achieve insomnia remission than controls. The six-hour cutoff did not affect the likelihood of depression remission, however.
Among participants with seven or more hours of sleep, those randomized to CBT-I were more likely to achieve insomnia remission and depression remission than controls.
“More research is needed to determine how best to achieve depression remission in those patients with less than seven hours of objective sleep duration prior to starting treatment,” Dr. Edinger concluded.
—Erik Greb
Suggested Reading
Bathgate CJ, Edinger JD, Krystal AD. Insomnia patients with objective short sleep duration have a blunted response to cognitive behavioral therapy for insomnia. Sleep. 2017;40(1).
Vgontzas AN, Liao D, Bixler EO, et al. Insomnia with objective short sleep duration is associated with a high risk for hypertension. Sleep. 2009;32(4):491-497.
Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: A joint consensus statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38(6):843-844.
BOSTON—Objective sleep duration moderates the probability of remission among patients with comorbid depression and insomnia, according to research presented at the 31st Annual Meeting of the Associated Professional Sleep Societies. Sleep durations of greater than five to six hours increase the likelihood that these patients will achieve insomnia remission with cognitive behavioral therapy for insomnia (CBT-I), but do not affect the likelihood of depression remission. Sleep durations of seven or more hours optimize the likelihood of insomnia remission and depression remission in response to CBT-I.
In a 2015 consensus statement, the Sleep Research Society recommended seven or more hours of sleep per night for adults younger than 60. Studies indicate that sleep durations of less than five hours and less than six hours are associated with increased morbidity and poor treatment response among patients with insomnia. “We wanted to know what [sleep-duration] cutoffs … might be better predictors of eventual insomnia and depression remission through treatment,” said Jack Edinger, PhD, Professor of Medicine at National Jewish Health in Denver.
An Analysis of the TRIAD Study
Dr. Edinger and colleagues conducted a secondary analysis of the TRIAD study, which examined whether combined treatment of depression and insomnia improves depression and sleep outcomes in participants with both disorders. Eligible participants met Diagnostic and Statistical Manual of Mental Disorders (4th ed.) criteria for major depression and primary insomnia, had a Hamilton Rating Scale for Depression (HAMD-17) score of 16 or greater, and had an Insomnia Severity Index (ISI) score of 11 or greater. People who had had psychotherapy in the previous four months, or had failed or could not tolerate previous adequate trials of the study medications, were excluded. Participants completed one night of baseline polysomnography before entering the treatment phase of the study.
The study population included 104 participants (75 women) with a mean age of 47. Mean baseline HAMD-17 score was 22, and mean baseline ISI score was 20.6. All participants received antidepressant medication (ie, citalopram, sertraline, or venlafaxine). Patients were randomized to CBT-I or sham (ie, a pseudodesensitization condition with sleep education). The investigators assessed participants biweekly with the HAMD-17 and the ISI. The treatment period lasted for 16 weeks.
CBT-I Provided Benefits
Participants with five or more hours of sleep were more likely to respond to CBT-I than participants with fewer than five hours of sleep. Among participants with sleep duration of five or more hours, insomnia remission was more likely with CBT-I than with the control condition. The five-hour cutoff had no association with depression remission.
Among participants with six or more hours of sleep, those who received CBT-I were more likely to achieve insomnia remission than controls. The six-hour cutoff did not affect the likelihood of depression remission, however.
Among participants with seven or more hours of sleep, those randomized to CBT-I were more likely to achieve insomnia remission and depression remission than controls.
“More research is needed to determine how best to achieve depression remission in those patients with less than seven hours of objective sleep duration prior to starting treatment,” Dr. Edinger concluded.
—Erik Greb
Suggested Reading
Bathgate CJ, Edinger JD, Krystal AD. Insomnia patients with objective short sleep duration have a blunted response to cognitive behavioral therapy for insomnia. Sleep. 2017;40(1).
Vgontzas AN, Liao D, Bixler EO, et al. Insomnia with objective short sleep duration is associated with a high risk for hypertension. Sleep. 2009;32(4):491-497.
Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: A joint consensus statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38(6):843-844.
Sleep issues vary by menopausal status
Perimenopausal women aged 40-59 years were less likely than were others in the same age group to average at least 7 hours’ sleep each night in 2015, according to the National Center for Health Statistics.
Among the perimenopausal women in that age group, 56% said that they slept less than 7 hours, on average, in a 24-hour period, compared with 40.5% of postmenopausal women and 32.5% of those who were premenopausal. Overall, 35.1% of women aged 40-59 did not average at least 7 hours of sleep per night, the NCHS reported in a data brief released Sept. 7.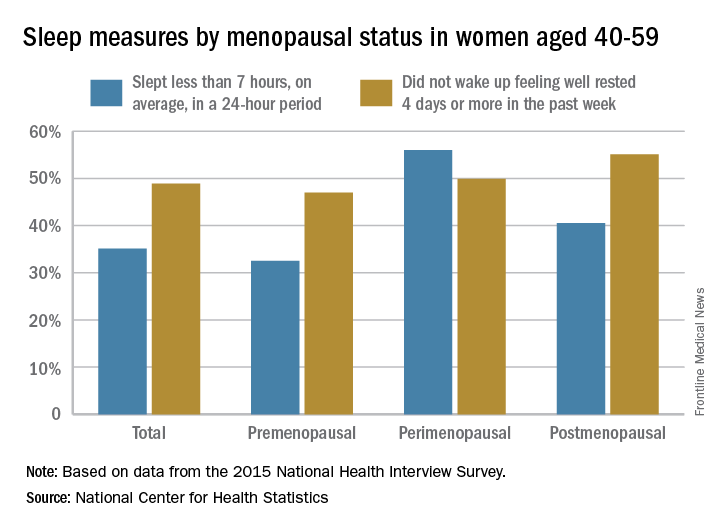
For this analysis, about 74% of the women included were premenopausal (still had a menstrual cycle), 4% were perimenopausal (last menstrual cycle was 1 year before or less), and 22% were postmenopausal (no menstrual cycle for more than 1 year or surgical menopause after removal of their ovaries).
Perimenopausal women aged 40-59 years were less likely than were others in the same age group to average at least 7 hours’ sleep each night in 2015, according to the National Center for Health Statistics.
Among the perimenopausal women in that age group, 56% said that they slept less than 7 hours, on average, in a 24-hour period, compared with 40.5% of postmenopausal women and 32.5% of those who were premenopausal. Overall, 35.1% of women aged 40-59 did not average at least 7 hours of sleep per night, the NCHS reported in a data brief released Sept. 7.
For this analysis, about 74% of the women included were premenopausal (still had a menstrual cycle), 4% were perimenopausal (last menstrual cycle was 1 year before or less), and 22% were postmenopausal (no menstrual cycle for more than 1 year or surgical menopause after removal of their ovaries).
Perimenopausal women aged 40-59 years were less likely than were others in the same age group to average at least 7 hours’ sleep each night in 2015, according to the National Center for Health Statistics.
Among the perimenopausal women in that age group, 56% said that they slept less than 7 hours, on average, in a 24-hour period, compared with 40.5% of postmenopausal women and 32.5% of those who were premenopausal. Overall, 35.1% of women aged 40-59 did not average at least 7 hours of sleep per night, the NCHS reported in a data brief released Sept. 7.
For this analysis, about 74% of the women included were premenopausal (still had a menstrual cycle), 4% were perimenopausal (last menstrual cycle was 1 year before or less), and 22% were postmenopausal (no menstrual cycle for more than 1 year or surgical menopause after removal of their ovaries).








