Article

2023 Update on menopause
- Author:
- Andrew M. Kaunitz, MD, NCMP
- JoAnn V. Pinkerton, MD, NCMP
Article
The perimenopausal period and the benefits of progestin IUDs
- Author:
- JoAnn V. Pinkerton, MD, NCMP
- Barbara S. Levy, MD
- Andrew M. Kaunitz, MD, NCMP
- Steven R. Goldstein, MD, NCMP, CCD
Although the use is off label, a progestin IUD can provide contraception not only in the reproductive years but through the perimenopause, to...
Article
Management considerations for women with von Willebrand disease
- Author:
- Andrew M. Kaunitz, MD, NCMP
- Andra H. James, MD, MPH
A review and commentary on strategies for managing heavy menstrual bleeding, intrapartum use of neuraxial anesthesia, and postpartum hemorrhage in...
Article

2022 Update on menopause
- Author:
- Andrew M. Kaunitz, MD, NCMP
- JoAnn Pinkerton, MD, NCMP
A review of new evidence on risks of synthetic progestins in menopausal women at average risk for breast cancer, advantages of...
Article
Appropriate cancer screening for women with dense breasts
- Author:
- Barbara Levy, MD
- Andrew M. Kaunitz, MD, NCMP
The data do not support mandating supplemental MRI as part of breast cancer screening for women with extremely dense breasts
...Article

HT for women who have had BSO before the age of natural menopause: Discerning the nuances
- Author:
- Stephanie Faubion, MD, MBA, NCMP
- Andrew M. Kaunitz, MD, NCMP
- Ekta Kapoor, MBBS, NCMP
Hormone therapy (HT) helps mitigate BSO’s adverse health effects; treatment recommendations are based mainly on observational data and society...
Article
Do recent data on use of menopausal HT and subsequent risk of dementia indicate an association?
- Author:
- Andrew M. Kaunitz, MD, NCMP
Article
Is vaginal laser therapy more efficacious in improving vaginal menopausal symptoms compared with sham therapy?
- Author:
- Andrew M. Kaunitz, MD, NCMP
- Cheryl B. Iglesia, MD
No. According to a randomized controlled trial in which 78 postmenopausal women with bothersome vaginal symptoms received either...
Article
4 new short-acting hormonal contraceptives offer enhancement over earlier options
- Author:
- Anita L. Nelson, MD
- Andrew M. Kaunitz, MD, NCMP
A review of recently marketed contraceptive products that provide important incremental improvements over existing formulations...
Article
2021 Update on menopause
- Author:
- Andrew M. Kaunitz, MD, NCMP
- Deanna C. McCullough, MD
Studies clarify menopausal HT’s impact on breast cancer risk
Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal...
Article
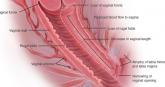
2020 Update on Menopause
- Author:
- Andrew M. Kaunitz, MD, NCMP
- JoAnn V. Pinkerton, MD, NCMP
- JoAnn E. Manson, MD, DrPH, NCMP
Recent studies show low-dose vaginal estrogen to be a highly effective, and safe, treatment for genitourinary syndrome of...
Article
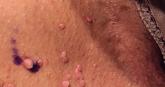
Can you identify these numerous papules in the groin area?
- Author:
- Penelope J. Kallis, MD
- Stephanie J. Carstens, MD
- Andrew M. Kaunitz, MD, NCMP
A case of multiple asymptomatic “pink skin tags”
Article
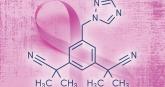
Breast cancer chemoprophylaxis in high-risk women: How persistent is the impact of an aromatase inhibitor after 5 years of use?
- Author:
- Andrew M. Kaunitz, MD, NCMP
Among postmenopausal women at high risk for breast cancer, those treated with anastrozole (N = 1,920) compared with placebo (N = 1,944)...
Article

Can the office visit interval for routine pessary care be extended safely?
- Author:
- Andrew M. Kaunitz, MD, NCMP
Yes, according to results of a randomized trial of 130 pessary users in which a 24-week pessary care visit was found to be...
Article
Persistent vulvar itch: What is the diagnosis?
- Author:
- Kerrie G. Satcher, MD
- Stephanie J. Carstens, MD
- Andrew M. Kaunitz, MD, NCMP
A case of pink, symmetrical bilateral plaques on the labia majora, without evidence of atrophy or scarring and with scant white...
