User login
CLL at ASH: A ‘mountain of data’ for targeted therapies
SAN DIEGO – There was a mountain of data presented at the annual meeting of the American Society of Hematology on the use of novel agents – both as frontline therapy and in combination – for the treatment of chronic lymphocytic leukemia (CLL).
In a video interview at the meeting, Brian T. Hill, MD, PhD, of the Cleveland Clinic and Anthony Mato, MD, of Memorial Sloan Kettering Cancer Center, New York, summed up the key studies and what they mean in practice. They also looked ahead at what data are still missing that could aid in making important treatment decisions.
Dr. Hill highlighted the late-breaking abstract on the ECOG-ACRIN Cancer Research Group E1912 trial comparing ibrutinib-rituximab to a chemotherapy regimen of fludarabine, cyclophosphamide, and rituximab (FCR) in previously untreated patients under age 70 years (Abstract LBA-4). Not only was there a progression-free survival benefit with the use of the ibrutinib regimen, but there was an overall survival benefit as well, he noted.
Dr. Mato pointed to notable results from the Alliance A041202 trial of older patients with previously untreated disease that compared ibrutinib alone or in combination with rituximab, with bendamustine plus rituximab (Abstract #6). The ibrutinib-containing regimens resulted in superior progression-free survival.
The two trials taken together show a movement away from chemotherapy in the frontline setting and toward targeted agents for CLL, Dr. Mato said. “What that agent or combination of agents will be, remains to be seen,” he said. “We have now a real message about the fact that we’re ending, potentially, the era of chemotherapy for patients with CLL, which is a very welcome change.”
Dr. Mato and Dr. Hill will be discussing these trials and more CLL data during a Twitter chat on Jan. 31, 2019, from 7 p.m. to 8 p.m. EST. Join in the conversation by using and following #MDedgeChats.
SAN DIEGO – There was a mountain of data presented at the annual meeting of the American Society of Hematology on the use of novel agents – both as frontline therapy and in combination – for the treatment of chronic lymphocytic leukemia (CLL).
In a video interview at the meeting, Brian T. Hill, MD, PhD, of the Cleveland Clinic and Anthony Mato, MD, of Memorial Sloan Kettering Cancer Center, New York, summed up the key studies and what they mean in practice. They also looked ahead at what data are still missing that could aid in making important treatment decisions.
Dr. Hill highlighted the late-breaking abstract on the ECOG-ACRIN Cancer Research Group E1912 trial comparing ibrutinib-rituximab to a chemotherapy regimen of fludarabine, cyclophosphamide, and rituximab (FCR) in previously untreated patients under age 70 years (Abstract LBA-4). Not only was there a progression-free survival benefit with the use of the ibrutinib regimen, but there was an overall survival benefit as well, he noted.
Dr. Mato pointed to notable results from the Alliance A041202 trial of older patients with previously untreated disease that compared ibrutinib alone or in combination with rituximab, with bendamustine plus rituximab (Abstract #6). The ibrutinib-containing regimens resulted in superior progression-free survival.
The two trials taken together show a movement away from chemotherapy in the frontline setting and toward targeted agents for CLL, Dr. Mato said. “What that agent or combination of agents will be, remains to be seen,” he said. “We have now a real message about the fact that we’re ending, potentially, the era of chemotherapy for patients with CLL, which is a very welcome change.”
Dr. Mato and Dr. Hill will be discussing these trials and more CLL data during a Twitter chat on Jan. 31, 2019, from 7 p.m. to 8 p.m. EST. Join in the conversation by using and following #MDedgeChats.
SAN DIEGO – There was a mountain of data presented at the annual meeting of the American Society of Hematology on the use of novel agents – both as frontline therapy and in combination – for the treatment of chronic lymphocytic leukemia (CLL).
In a video interview at the meeting, Brian T. Hill, MD, PhD, of the Cleveland Clinic and Anthony Mato, MD, of Memorial Sloan Kettering Cancer Center, New York, summed up the key studies and what they mean in practice. They also looked ahead at what data are still missing that could aid in making important treatment decisions.
Dr. Hill highlighted the late-breaking abstract on the ECOG-ACRIN Cancer Research Group E1912 trial comparing ibrutinib-rituximab to a chemotherapy regimen of fludarabine, cyclophosphamide, and rituximab (FCR) in previously untreated patients under age 70 years (Abstract LBA-4). Not only was there a progression-free survival benefit with the use of the ibrutinib regimen, but there was an overall survival benefit as well, he noted.
Dr. Mato pointed to notable results from the Alliance A041202 trial of older patients with previously untreated disease that compared ibrutinib alone or in combination with rituximab, with bendamustine plus rituximab (Abstract #6). The ibrutinib-containing regimens resulted in superior progression-free survival.
The two trials taken together show a movement away from chemotherapy in the frontline setting and toward targeted agents for CLL, Dr. Mato said. “What that agent or combination of agents will be, remains to be seen,” he said. “We have now a real message about the fact that we’re ending, potentially, the era of chemotherapy for patients with CLL, which is a very welcome change.”
Dr. Mato and Dr. Hill will be discussing these trials and more CLL data during a Twitter chat on Jan. 31, 2019, from 7 p.m. to 8 p.m. EST. Join in the conversation by using and following #MDedgeChats.
REPORTING FROM ASH 2018
Social media can help doctors stay up to date
SAN DIEGO –
“People want to see that you’re a person and not strictly a physician,” said Dr. Yates, a pediatric hematologist at Texas Children’s Hospital in Houston, who has had an active Twitter presence for the last few years.
Dr. Yates – whose Twitter handle is @sicklecelldoc – said she dipped a toe in the social media waters because she wanted to bring accurate medical information to patients in the arena where they are seeking information.
“I want families to understand their condition as well as they can on whatever level they can, and so I just found this to be another way to do that ... outside of my clinic,” she said during an interview at the annual meeting of the American Society of Hematology.
But beyond correcting misinformation and serving as an advocate for patients, Dr. Yates said she gets professional benefits from being on Twitter. For instance, she uses the platform to find relevant articles as soon as they publish, without wading through all the journals.
“It’s allowed me to kind of streamline what I read,” she said.
Dr. Yates said Twitter is her social media platform of choice because it provides a simple, succinct way to communicate and provide links to more in-depth resources.
While social media can be fun and rewarding for physicians, Dr. Yates said think before you post. Ask yourself, “would you tell your chairperson this?”
Dr. Yates reported having no relevant financial disclosures.
SAN DIEGO –
“People want to see that you’re a person and not strictly a physician,” said Dr. Yates, a pediatric hematologist at Texas Children’s Hospital in Houston, who has had an active Twitter presence for the last few years.
Dr. Yates – whose Twitter handle is @sicklecelldoc – said she dipped a toe in the social media waters because she wanted to bring accurate medical information to patients in the arena where they are seeking information.
“I want families to understand their condition as well as they can on whatever level they can, and so I just found this to be another way to do that ... outside of my clinic,” she said during an interview at the annual meeting of the American Society of Hematology.
But beyond correcting misinformation and serving as an advocate for patients, Dr. Yates said she gets professional benefits from being on Twitter. For instance, she uses the platform to find relevant articles as soon as they publish, without wading through all the journals.
“It’s allowed me to kind of streamline what I read,” she said.
Dr. Yates said Twitter is her social media platform of choice because it provides a simple, succinct way to communicate and provide links to more in-depth resources.
While social media can be fun and rewarding for physicians, Dr. Yates said think before you post. Ask yourself, “would you tell your chairperson this?”
Dr. Yates reported having no relevant financial disclosures.
SAN DIEGO –
“People want to see that you’re a person and not strictly a physician,” said Dr. Yates, a pediatric hematologist at Texas Children’s Hospital in Houston, who has had an active Twitter presence for the last few years.
Dr. Yates – whose Twitter handle is @sicklecelldoc – said she dipped a toe in the social media waters because she wanted to bring accurate medical information to patients in the arena where they are seeking information.
“I want families to understand their condition as well as they can on whatever level they can, and so I just found this to be another way to do that ... outside of my clinic,” she said during an interview at the annual meeting of the American Society of Hematology.
But beyond correcting misinformation and serving as an advocate for patients, Dr. Yates said she gets professional benefits from being on Twitter. For instance, she uses the platform to find relevant articles as soon as they publish, without wading through all the journals.
“It’s allowed me to kind of streamline what I read,” she said.
Dr. Yates said Twitter is her social media platform of choice because it provides a simple, succinct way to communicate and provide links to more in-depth resources.
While social media can be fun and rewarding for physicians, Dr. Yates said think before you post. Ask yourself, “would you tell your chairperson this?”
Dr. Yates reported having no relevant financial disclosures.
REPORTING FROM ASH 2018
LCAR-B38M CAR T therapy appears durable in myeloma
SAN DIEGO – The chimeric antigen receptor (CAR) T-cell therapy LCAR-B38M is in the race for approval in multiple myeloma following encouraging phase 1 results reported at the annual meeting of the American Society of Hematology.
In the LEGEND-2 phase 1/2 open study of 57 patients with advanced relapsed/refractory multiple myeloma treated with the investigational CAR T therapy, the overall response rate was 88% and the complete response rate was 74%. Among 42 patients who achieved complete response, 39 (68%) were negative for minimal residual disease (MRD).
With a median follow-up of 12 months, the median duration of response was 16 months and progression-free survival was 15 months. But in patients who achieved MRD-negative complete response, the median progression-free survival was extended to 24 months.
Pyrexia and cytokine release syndrome were reported in 90% or more of patients. Thrombocytopenia and leukopenia were reported in nearly half of patients.
The phase 1 study was conducted by researchers from the Second Affiliated Hospital of Xi’an Jiaotong University in Xi’an, China. The B-cell maturation antigen (BCMA)–directed CAR T-cell therapy is being jointly developed by Nanjing Legend Biotech and Janssen. A phase 2 study is currently being planned in China for LCAR-B38M. In parallel, Janssen and Legend are enrolling patients in a phase 1b/2 trial of the agent (also known as JNJ-68284528) in the United States.
The therapy joins a growing field of anti-BCMA CAR T-cell agents with promising initial trial results, including bb2121.
In a video interview at ASH, Sen Zhuang, MD, PhD, vice president of oncology clinical development at Janssen Research & Development, said this class of CAR T agents offers the potential for “very long remissions” and possibly even a “cure” for myeloma.
The LEGEND-2 study is sponsored by Nanjing Legend Biotech and two of the investigators reported employment with the company.
SAN DIEGO – The chimeric antigen receptor (CAR) T-cell therapy LCAR-B38M is in the race for approval in multiple myeloma following encouraging phase 1 results reported at the annual meeting of the American Society of Hematology.
In the LEGEND-2 phase 1/2 open study of 57 patients with advanced relapsed/refractory multiple myeloma treated with the investigational CAR T therapy, the overall response rate was 88% and the complete response rate was 74%. Among 42 patients who achieved complete response, 39 (68%) were negative for minimal residual disease (MRD).
With a median follow-up of 12 months, the median duration of response was 16 months and progression-free survival was 15 months. But in patients who achieved MRD-negative complete response, the median progression-free survival was extended to 24 months.
Pyrexia and cytokine release syndrome were reported in 90% or more of patients. Thrombocytopenia and leukopenia were reported in nearly half of patients.
The phase 1 study was conducted by researchers from the Second Affiliated Hospital of Xi’an Jiaotong University in Xi’an, China. The B-cell maturation antigen (BCMA)–directed CAR T-cell therapy is being jointly developed by Nanjing Legend Biotech and Janssen. A phase 2 study is currently being planned in China for LCAR-B38M. In parallel, Janssen and Legend are enrolling patients in a phase 1b/2 trial of the agent (also known as JNJ-68284528) in the United States.
The therapy joins a growing field of anti-BCMA CAR T-cell agents with promising initial trial results, including bb2121.
In a video interview at ASH, Sen Zhuang, MD, PhD, vice president of oncology clinical development at Janssen Research & Development, said this class of CAR T agents offers the potential for “very long remissions” and possibly even a “cure” for myeloma.
The LEGEND-2 study is sponsored by Nanjing Legend Biotech and two of the investigators reported employment with the company.
SAN DIEGO – The chimeric antigen receptor (CAR) T-cell therapy LCAR-B38M is in the race for approval in multiple myeloma following encouraging phase 1 results reported at the annual meeting of the American Society of Hematology.
In the LEGEND-2 phase 1/2 open study of 57 patients with advanced relapsed/refractory multiple myeloma treated with the investigational CAR T therapy, the overall response rate was 88% and the complete response rate was 74%. Among 42 patients who achieved complete response, 39 (68%) were negative for minimal residual disease (MRD).
With a median follow-up of 12 months, the median duration of response was 16 months and progression-free survival was 15 months. But in patients who achieved MRD-negative complete response, the median progression-free survival was extended to 24 months.
Pyrexia and cytokine release syndrome were reported in 90% or more of patients. Thrombocytopenia and leukopenia were reported in nearly half of patients.
The phase 1 study was conducted by researchers from the Second Affiliated Hospital of Xi’an Jiaotong University in Xi’an, China. The B-cell maturation antigen (BCMA)–directed CAR T-cell therapy is being jointly developed by Nanjing Legend Biotech and Janssen. A phase 2 study is currently being planned in China for LCAR-B38M. In parallel, Janssen and Legend are enrolling patients in a phase 1b/2 trial of the agent (also known as JNJ-68284528) in the United States.
The therapy joins a growing field of anti-BCMA CAR T-cell agents with promising initial trial results, including bb2121.
In a video interview at ASH, Sen Zhuang, MD, PhD, vice president of oncology clinical development at Janssen Research & Development, said this class of CAR T agents offers the potential for “very long remissions” and possibly even a “cure” for myeloma.
The LEGEND-2 study is sponsored by Nanjing Legend Biotech and two of the investigators reported employment with the company.
REPORTING FROM ASH 2018
Key clinical point:
Major finding: The complete response rate was 74% with median progression-free survival of 15 months.
Study details: A phase 1/2 study of 57 patients with advanced relapsed/refractory multiple myeloma.
Disclosures: The study is sponsored by Nanjing Legend Biotech. Two of the investigators reported employment with the company.
FLT3 inhibitor moves forward in newly diagnosed AML
SAN DIEGO – In an ongoing phase 1 study, the oral FMS-like tyrosine kinase 3
The dose escalation/expansion study coupled the oral agent with induction and consolidation chemotherapy and was aimed at establishing the dosing and safety of gilteritinib.
The findings – reported at the annual meeting of the American Society of Hematology – mean that the FLT3 inhibitor will next be compared with the current standard of care, which has a 60%-65% remission rate, according to Keith W. Pratz, MD, of Johns Hopkins University in Baltimore.
“The later-phase clinical studies that we will be doing with gilteritinib will look to compare midostaurin-based chemotherapy with gilteritinib and looking for outcomes, with hopes of improving upon this 60%-65% remission rate,” Dr. Pratz said in a video interview.
Gilteritinib was recently approved by the Food and Drug Administration for relapsed/refractory AML patients with FLT3 mutations.
Dr. Pratz said the approval will provide a needed new treatment option in that patient population, which has had a low response rate to conventional therapy, in the range of 10%-15%.
“There really wasn’t a standard approved therapy prior to this and this will be what is given to most patients who have a FLT3 mutation and relapse,” he said.
Dr. Pratz reported consultancy and research funding from Astellas, which markets gilteritinib, as well as other companies.
SAN DIEGO – In an ongoing phase 1 study, the oral FMS-like tyrosine kinase 3
The dose escalation/expansion study coupled the oral agent with induction and consolidation chemotherapy and was aimed at establishing the dosing and safety of gilteritinib.
The findings – reported at the annual meeting of the American Society of Hematology – mean that the FLT3 inhibitor will next be compared with the current standard of care, which has a 60%-65% remission rate, according to Keith W. Pratz, MD, of Johns Hopkins University in Baltimore.
“The later-phase clinical studies that we will be doing with gilteritinib will look to compare midostaurin-based chemotherapy with gilteritinib and looking for outcomes, with hopes of improving upon this 60%-65% remission rate,” Dr. Pratz said in a video interview.
Gilteritinib was recently approved by the Food and Drug Administration for relapsed/refractory AML patients with FLT3 mutations.
Dr. Pratz said the approval will provide a needed new treatment option in that patient population, which has had a low response rate to conventional therapy, in the range of 10%-15%.
“There really wasn’t a standard approved therapy prior to this and this will be what is given to most patients who have a FLT3 mutation and relapse,” he said.
Dr. Pratz reported consultancy and research funding from Astellas, which markets gilteritinib, as well as other companies.
SAN DIEGO – In an ongoing phase 1 study, the oral FMS-like tyrosine kinase 3
The dose escalation/expansion study coupled the oral agent with induction and consolidation chemotherapy and was aimed at establishing the dosing and safety of gilteritinib.
The findings – reported at the annual meeting of the American Society of Hematology – mean that the FLT3 inhibitor will next be compared with the current standard of care, which has a 60%-65% remission rate, according to Keith W. Pratz, MD, of Johns Hopkins University in Baltimore.
“The later-phase clinical studies that we will be doing with gilteritinib will look to compare midostaurin-based chemotherapy with gilteritinib and looking for outcomes, with hopes of improving upon this 60%-65% remission rate,” Dr. Pratz said in a video interview.
Gilteritinib was recently approved by the Food and Drug Administration for relapsed/refractory AML patients with FLT3 mutations.
Dr. Pratz said the approval will provide a needed new treatment option in that patient population, which has had a low response rate to conventional therapy, in the range of 10%-15%.
“There really wasn’t a standard approved therapy prior to this and this will be what is given to most patients who have a FLT3 mutation and relapse,” he said.
Dr. Pratz reported consultancy and research funding from Astellas, which markets gilteritinib, as well as other companies.
REPORTING FROM ASH 2018
Beat AML trial delivers genomic results in 7 days
SAN DIEGO – Investigators demonstrated the feasibility of delivering genomic results in 7 days in a population of older, newly diagnosed patients with acute myeloid leukemia (AML).
The Beat AML Master Trial is an ongoing umbrella study that harnesses cytogenetic information and next generation sequencing to match patients with targeted therapies across a number of substudies or outside of the trial’s multicenter network.
The researchers chose AML for this precision-medicine study because of its rapid onset and lethal nature, its heterogeneity, and the availability of more-targeted therapies, said Amy Burd, PhD, of the Leukemia & Lymphoma Society, which is sponsoring the study.
Initial data from the trial showed that more than 95% of patients were assigned to treatment in 7 days or less, based on their personalized genomic information.
Overall, 285 patients had usable genomic screening data and were assigned to treatment. Of those patients, 273 were assigned to a treatment within 7 days, Dr. Burd reported at the annual meeting of the American Society of Hematology.
The speed of delivering these results is critical, said Joseph Mikhael, MD, chief medical officer for the International Myeloma Foundation in Phoenix, who moderated a media briefing on personalized medicine.
“One of the greatest challenges we faced in the concept of personalized medicine is by the time you’ve determined what is best for that patient ... the horse is already out of the barn,” Dr. Mikhael said. “You have to have started the patient on treatment already or else their disease could have progressed quite rapidly.”
In the past, genomic results might come back a month after the patient started therapy. “It was really almost academic,” he said.
In the Beat AML study, more than half (146 patients) were treated based on their AML subtype. The remaining patients (139) were not treated: 2.5% of patients died within 7 days, 7% of patients chose an alternative treatment prior to assignment, 20% chose standard of care, 9.1% chose an alternative trial after assignment, 8.1% chose palliative care, and the remainder had a reason that was not specified.
“The treatment decisions are made for what’s best for the patient even if that means a study outside of Beat AML,” Dr. Burd said.
Currently, there are 11 substudies offering treatment to trial participants across 13 clinical sites. There has been promising efficacy in many of the treatment arms, Dr. Burd said.
In the future, the researchers are looking to expand the substudies to look into novel drug combinations for certain AML subtypes, specifically isocitrate dehydrogenase 2–mutated groups.
Dr. Burd is an employee of the Leukemia & Lymphoma Society. Other coinvestigators reported financial relationships with the pharmaceutical industry. Dr. Mikhael reported research funding from AbbVie, Celgene, Onyx Pharmaceuticals, and Sanofi.
SOURCE: Burd A et al. ASH 2018, Abstract 559.
SAN DIEGO – Investigators demonstrated the feasibility of delivering genomic results in 7 days in a population of older, newly diagnosed patients with acute myeloid leukemia (AML).
The Beat AML Master Trial is an ongoing umbrella study that harnesses cytogenetic information and next generation sequencing to match patients with targeted therapies across a number of substudies or outside of the trial’s multicenter network.
The researchers chose AML for this precision-medicine study because of its rapid onset and lethal nature, its heterogeneity, and the availability of more-targeted therapies, said Amy Burd, PhD, of the Leukemia & Lymphoma Society, which is sponsoring the study.
Initial data from the trial showed that more than 95% of patients were assigned to treatment in 7 days or less, based on their personalized genomic information.
Overall, 285 patients had usable genomic screening data and were assigned to treatment. Of those patients, 273 were assigned to a treatment within 7 days, Dr. Burd reported at the annual meeting of the American Society of Hematology.
The speed of delivering these results is critical, said Joseph Mikhael, MD, chief medical officer for the International Myeloma Foundation in Phoenix, who moderated a media briefing on personalized medicine.
“One of the greatest challenges we faced in the concept of personalized medicine is by the time you’ve determined what is best for that patient ... the horse is already out of the barn,” Dr. Mikhael said. “You have to have started the patient on treatment already or else their disease could have progressed quite rapidly.”
In the past, genomic results might come back a month after the patient started therapy. “It was really almost academic,” he said.
In the Beat AML study, more than half (146 patients) were treated based on their AML subtype. The remaining patients (139) were not treated: 2.5% of patients died within 7 days, 7% of patients chose an alternative treatment prior to assignment, 20% chose standard of care, 9.1% chose an alternative trial after assignment, 8.1% chose palliative care, and the remainder had a reason that was not specified.
“The treatment decisions are made for what’s best for the patient even if that means a study outside of Beat AML,” Dr. Burd said.
Currently, there are 11 substudies offering treatment to trial participants across 13 clinical sites. There has been promising efficacy in many of the treatment arms, Dr. Burd said.
In the future, the researchers are looking to expand the substudies to look into novel drug combinations for certain AML subtypes, specifically isocitrate dehydrogenase 2–mutated groups.
Dr. Burd is an employee of the Leukemia & Lymphoma Society. Other coinvestigators reported financial relationships with the pharmaceutical industry. Dr. Mikhael reported research funding from AbbVie, Celgene, Onyx Pharmaceuticals, and Sanofi.
SOURCE: Burd A et al. ASH 2018, Abstract 559.
SAN DIEGO – Investigators demonstrated the feasibility of delivering genomic results in 7 days in a population of older, newly diagnosed patients with acute myeloid leukemia (AML).
The Beat AML Master Trial is an ongoing umbrella study that harnesses cytogenetic information and next generation sequencing to match patients with targeted therapies across a number of substudies or outside of the trial’s multicenter network.
The researchers chose AML for this precision-medicine study because of its rapid onset and lethal nature, its heterogeneity, and the availability of more-targeted therapies, said Amy Burd, PhD, of the Leukemia & Lymphoma Society, which is sponsoring the study.
Initial data from the trial showed that more than 95% of patients were assigned to treatment in 7 days or less, based on their personalized genomic information.
Overall, 285 patients had usable genomic screening data and were assigned to treatment. Of those patients, 273 were assigned to a treatment within 7 days, Dr. Burd reported at the annual meeting of the American Society of Hematology.
The speed of delivering these results is critical, said Joseph Mikhael, MD, chief medical officer for the International Myeloma Foundation in Phoenix, who moderated a media briefing on personalized medicine.
“One of the greatest challenges we faced in the concept of personalized medicine is by the time you’ve determined what is best for that patient ... the horse is already out of the barn,” Dr. Mikhael said. “You have to have started the patient on treatment already or else their disease could have progressed quite rapidly.”
In the past, genomic results might come back a month after the patient started therapy. “It was really almost academic,” he said.
In the Beat AML study, more than half (146 patients) were treated based on their AML subtype. The remaining patients (139) were not treated: 2.5% of patients died within 7 days, 7% of patients chose an alternative treatment prior to assignment, 20% chose standard of care, 9.1% chose an alternative trial after assignment, 8.1% chose palliative care, and the remainder had a reason that was not specified.
“The treatment decisions are made for what’s best for the patient even if that means a study outside of Beat AML,” Dr. Burd said.
Currently, there are 11 substudies offering treatment to trial participants across 13 clinical sites. There has been promising efficacy in many of the treatment arms, Dr. Burd said.
In the future, the researchers are looking to expand the substudies to look into novel drug combinations for certain AML subtypes, specifically isocitrate dehydrogenase 2–mutated groups.
Dr. Burd is an employee of the Leukemia & Lymphoma Society. Other coinvestigators reported financial relationships with the pharmaceutical industry. Dr. Mikhael reported research funding from AbbVie, Celgene, Onyx Pharmaceuticals, and Sanofi.
SOURCE: Burd A et al. ASH 2018, Abstract 559.
REPORTING FROM ASH 2018
Key clinical point:
Major finding: More than 95% of patients in the trial were assigned to treatment within 7 days based on results of their genomic screening.
Study details: An umbrella study of 285 patients aged 60 years and older with newly diagnosed acute myeloid leukemia.
Disclosures: The study is sponsored by the Leukemia & Lymphoma Society. Dr. Burd is an employee of the Society and other investigators reported funding from multiple pharmaceutical companies.
Source: Burd A et al. ASH 2018, Abstract 559.
Opioids appear safe for sickle cell pain
SAN DIEGO – Inpatient deaths from opioid overdose in the U.S. population has risen dramatically in recent years, but this is essentially a “never event” among patients with sickle cell disease (SCD).
These findings come from an analysis of hospital discharge data in the National Inpatient Sample from 1998 to 2013. Oladimeji Akinola Akinboro, MBBS, who led the study, said the findings suggest that the current patterns of opioid use in SCD patients are safe and that opioids should not be withheld when they are appropriate.
From 1998 to 2013, the rate of inpatient deaths from opioid overdose in the United States rose 350% overall and approximately 8% annually. In contrast, deaths among SCD patients were flat, with a rate at or near zero throughout the same time period.
Over the 16-year period, there were just nine deaths reported among SCD patients because of opioids in the inpatient setting, Dr. Akinboro of Boston University Medical Center reported at the annual meeting of the American Society of Hematology.
While the reasons behind the difference were not explored in the study, Dr. Akinboro suggested that the sickle cell community has greater experience with opioids and that patients and physicians typically have long-standing clinical relationships that make mitigation of opioid misuse easier to manage.
Opioid-related hospitalizations, however, were comparable among the general U.S. population and SCD patients. And for both groups the rates remained relatively steady throughout the study period, with the exception of a drop among SCD patients in 2002, he noted.
The study also revealed age-related trends in hospitalizations overall. Hospitalizations among sickle cell patients were stable from 1998 to 2013. However, when the researchers broke the data down by age they found that adults aged 18-44 years had an increase in hospitalizations, with the steepest rise in patients aged 65 years and older.
In total, there were more than 1.7 million hospitalizations among SCD patients in the United States from 1998 to 2013. The rate declined by 9.9% each year from 1998 to 2002, then remained flat at around 27 per 100,000 persons from 2002 to 2013.
However, for adults aged 18-44 years, hospitalizations increased from 43 per 100,000 persons in 2002 to 71 per 100,000 persons in 2013 – an annual increase of 3.8%. Patients aged 65 years and older saw their rate of hospitalization increase from 2.7 to 5.4 per 100,000 persons from 1998 to 2013 – a 6.5% increase for each year.
The study did not explore the causes behind the age-related trends but Dr. Akinboro suggested it may be because of fragmentation of adult SCD care and age- and pain-related medical comorbidities.
The researchers reported having no relevant financial disclosures.
SOURCE: Akinboro OA et al. ASH 2018, Abstract 315.
SAN DIEGO – Inpatient deaths from opioid overdose in the U.S. population has risen dramatically in recent years, but this is essentially a “never event” among patients with sickle cell disease (SCD).
These findings come from an analysis of hospital discharge data in the National Inpatient Sample from 1998 to 2013. Oladimeji Akinola Akinboro, MBBS, who led the study, said the findings suggest that the current patterns of opioid use in SCD patients are safe and that opioids should not be withheld when they are appropriate.
From 1998 to 2013, the rate of inpatient deaths from opioid overdose in the United States rose 350% overall and approximately 8% annually. In contrast, deaths among SCD patients were flat, with a rate at or near zero throughout the same time period.
Over the 16-year period, there were just nine deaths reported among SCD patients because of opioids in the inpatient setting, Dr. Akinboro of Boston University Medical Center reported at the annual meeting of the American Society of Hematology.
While the reasons behind the difference were not explored in the study, Dr. Akinboro suggested that the sickle cell community has greater experience with opioids and that patients and physicians typically have long-standing clinical relationships that make mitigation of opioid misuse easier to manage.
Opioid-related hospitalizations, however, were comparable among the general U.S. population and SCD patients. And for both groups the rates remained relatively steady throughout the study period, with the exception of a drop among SCD patients in 2002, he noted.
The study also revealed age-related trends in hospitalizations overall. Hospitalizations among sickle cell patients were stable from 1998 to 2013. However, when the researchers broke the data down by age they found that adults aged 18-44 years had an increase in hospitalizations, with the steepest rise in patients aged 65 years and older.
In total, there were more than 1.7 million hospitalizations among SCD patients in the United States from 1998 to 2013. The rate declined by 9.9% each year from 1998 to 2002, then remained flat at around 27 per 100,000 persons from 2002 to 2013.
However, for adults aged 18-44 years, hospitalizations increased from 43 per 100,000 persons in 2002 to 71 per 100,000 persons in 2013 – an annual increase of 3.8%. Patients aged 65 years and older saw their rate of hospitalization increase from 2.7 to 5.4 per 100,000 persons from 1998 to 2013 – a 6.5% increase for each year.
The study did not explore the causes behind the age-related trends but Dr. Akinboro suggested it may be because of fragmentation of adult SCD care and age- and pain-related medical comorbidities.
The researchers reported having no relevant financial disclosures.
SOURCE: Akinboro OA et al. ASH 2018, Abstract 315.
SAN DIEGO – Inpatient deaths from opioid overdose in the U.S. population has risen dramatically in recent years, but this is essentially a “never event” among patients with sickle cell disease (SCD).
These findings come from an analysis of hospital discharge data in the National Inpatient Sample from 1998 to 2013. Oladimeji Akinola Akinboro, MBBS, who led the study, said the findings suggest that the current patterns of opioid use in SCD patients are safe and that opioids should not be withheld when they are appropriate.
From 1998 to 2013, the rate of inpatient deaths from opioid overdose in the United States rose 350% overall and approximately 8% annually. In contrast, deaths among SCD patients were flat, with a rate at or near zero throughout the same time period.
Over the 16-year period, there were just nine deaths reported among SCD patients because of opioids in the inpatient setting, Dr. Akinboro of Boston University Medical Center reported at the annual meeting of the American Society of Hematology.
While the reasons behind the difference were not explored in the study, Dr. Akinboro suggested that the sickle cell community has greater experience with opioids and that patients and physicians typically have long-standing clinical relationships that make mitigation of opioid misuse easier to manage.
Opioid-related hospitalizations, however, were comparable among the general U.S. population and SCD patients. And for both groups the rates remained relatively steady throughout the study period, with the exception of a drop among SCD patients in 2002, he noted.
The study also revealed age-related trends in hospitalizations overall. Hospitalizations among sickle cell patients were stable from 1998 to 2013. However, when the researchers broke the data down by age they found that adults aged 18-44 years had an increase in hospitalizations, with the steepest rise in patients aged 65 years and older.
In total, there were more than 1.7 million hospitalizations among SCD patients in the United States from 1998 to 2013. The rate declined by 9.9% each year from 1998 to 2002, then remained flat at around 27 per 100,000 persons from 2002 to 2013.
However, for adults aged 18-44 years, hospitalizations increased from 43 per 100,000 persons in 2002 to 71 per 100,000 persons in 2013 – an annual increase of 3.8%. Patients aged 65 years and older saw their rate of hospitalization increase from 2.7 to 5.4 per 100,000 persons from 1998 to 2013 – a 6.5% increase for each year.
The study did not explore the causes behind the age-related trends but Dr. Akinboro suggested it may be because of fragmentation of adult SCD care and age- and pain-related medical comorbidities.
The researchers reported having no relevant financial disclosures.
SOURCE: Akinboro OA et al. ASH 2018, Abstract 315.
REPORTING FROM ASH 2018
Key clinical point:
Major finding: While the rate of inpatient death from opioid overdose rose 350% among the general U.S. population from 1998 to 2013, it remained essentially at zero among patients with sickle cell disease.
Study details: A retrospective analysis of discharge diagnoses from the National Inpatient Sample from 1998 to 2013.
Disclosures: The researchers reported having no relevant financial disclosures.
Source: Akinboro OA et al. ASH 2018, Abstract 315.
New treatments promise sickle cell “cure” for all ages
SAN DIEGO –
“There is an opportunity to cure your disease no matter what age you are,” Dr. Osunkwo, medical director of the sickle cell program at Levine Cancer Institute at Atrium Health in Charlotte, N.C., said in a video interview at the annual meeting of the American Society of Hematology. “Sickle cell disease is now a disease of all ages and the treatments have to be treatments for everybody of all ages, not just for children.”
Dr. Osunkwo was the moderator of a press conference highlighting top research in sickle cell disease at ASH 2018. She pointed to findings from first-in-human trials of gene therapy using a lentiviral vector targeting BCL11A to reverse the sickle cell phenotype, as well as a study examining familial haploidentical stem cell transplantation with CD34 enrichment and mononuclear add-back in high-risk patients.
These two studies show parallel progress in curative therapies and are complementary, Dr. Osunkwo said. Improvements in transplants, and specifically in how patients are prepared and managed for them, will have a benefit in gene therapy.
But there are many other sickle cell disease studies being presented at ASH this year, she noted.
“There’s a recognition that sickle cell has been an understudied, underresourced, underexposed population,” she said. “And the suffering and the magnitude of medical problems is huge and it finally has bubbled up to the surface.”
Dr. Osunkwo reported being on advisory committees for Novartis and Pfizer and on the speaker’s bureau for Novartis. She has received honoraria from Terumo BCT and funding from the Health Resources and Services Administration and the Patient-Centered Outcomes Research Institute.
SAN DIEGO –
“There is an opportunity to cure your disease no matter what age you are,” Dr. Osunkwo, medical director of the sickle cell program at Levine Cancer Institute at Atrium Health in Charlotte, N.C., said in a video interview at the annual meeting of the American Society of Hematology. “Sickle cell disease is now a disease of all ages and the treatments have to be treatments for everybody of all ages, not just for children.”
Dr. Osunkwo was the moderator of a press conference highlighting top research in sickle cell disease at ASH 2018. She pointed to findings from first-in-human trials of gene therapy using a lentiviral vector targeting BCL11A to reverse the sickle cell phenotype, as well as a study examining familial haploidentical stem cell transplantation with CD34 enrichment and mononuclear add-back in high-risk patients.
These two studies show parallel progress in curative therapies and are complementary, Dr. Osunkwo said. Improvements in transplants, and specifically in how patients are prepared and managed for them, will have a benefit in gene therapy.
But there are many other sickle cell disease studies being presented at ASH this year, she noted.
“There’s a recognition that sickle cell has been an understudied, underresourced, underexposed population,” she said. “And the suffering and the magnitude of medical problems is huge and it finally has bubbled up to the surface.”
Dr. Osunkwo reported being on advisory committees for Novartis and Pfizer and on the speaker’s bureau for Novartis. She has received honoraria from Terumo BCT and funding from the Health Resources and Services Administration and the Patient-Centered Outcomes Research Institute.
SAN DIEGO –
“There is an opportunity to cure your disease no matter what age you are,” Dr. Osunkwo, medical director of the sickle cell program at Levine Cancer Institute at Atrium Health in Charlotte, N.C., said in a video interview at the annual meeting of the American Society of Hematology. “Sickle cell disease is now a disease of all ages and the treatments have to be treatments for everybody of all ages, not just for children.”
Dr. Osunkwo was the moderator of a press conference highlighting top research in sickle cell disease at ASH 2018. She pointed to findings from first-in-human trials of gene therapy using a lentiviral vector targeting BCL11A to reverse the sickle cell phenotype, as well as a study examining familial haploidentical stem cell transplantation with CD34 enrichment and mononuclear add-back in high-risk patients.
These two studies show parallel progress in curative therapies and are complementary, Dr. Osunkwo said. Improvements in transplants, and specifically in how patients are prepared and managed for them, will have a benefit in gene therapy.
But there are many other sickle cell disease studies being presented at ASH this year, she noted.
“There’s a recognition that sickle cell has been an understudied, underresourced, underexposed population,” she said. “And the suffering and the magnitude of medical problems is huge and it finally has bubbled up to the surface.”
Dr. Osunkwo reported being on advisory committees for Novartis and Pfizer and on the speaker’s bureau for Novartis. She has received honoraria from Terumo BCT and funding from the Health Resources and Services Administration and the Patient-Centered Outcomes Research Institute.
REPORTING FROM ASH 2018
Your guide to ASH 2018: Abstracts to watch
With more than 3,000 scientific abstracts at the 2018 annual meeting of the American Society of Hematology, it can be tough to figure out what research is most relevant to practice. But the editorial advisory board of Hematology News is making it easier this year with their picks for what to watch and why.
Lymphomas
Brian T. Hill, MD, of the Cleveland Clinic, offered his top picks in lymphoma research. Results of the phase 3 international Alliance North American Intergroup Study A041202 will be presented during the ASH plenary session at 2 p.m. PT on Sunday, Dec. 2 in Hall AB of the San Diego Convention Center (Abstract 6). The study compared bendamustine plus rituximab with ibrutinib and the combination of ibrutinib plus rituximab to see if the ibrutinib-containing therapies would have superior progression-free survival (PFS) in chronic lymphocytic leukemia (CLL), compared with chemoimmunotherapy. Results indicate that ibrutinib had superior PFS in older patients with CLL and could be a standard of care in this population.
The study is worth watching because it is the first report of a head-to-head trial of chemotherapy versus ibrutinib for first-line treatment of CLL, Dr. Hill said.
Two more studies offer important reports of “real world” experiences with chimeric antigen receptor (CAR) T-cell therapy.
In one multicenter retrospective study, researchers evaluated the outcomes of axicabtagene ciloleucel (axi-cel) CAR T-cell therapy for relapsed/refractory aggressive B-cell lymphoma when it is used a standard care. The researchers will report that 30-day responses in the real-world setting were comparable to the best responses seen in the ZUMA-1 trial. The full results will be reported at 9:30 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 91).
Another retrospective analysis looked at the use of axi-cell and revealed some critical differences from ZUMA-1, specifically the overall response rate (ORR) and complete response (CR) rate were lower than those reported in the pivotal clinical trial. The findings will be reported at 9:45 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 92).
Researchers will also present the unblinded results from the ECHELON-2 study, which compared the efficacy and safety of brentuximab vedotin in combination with cyclophosphamide, doxorubicin, and prednisone (CHP) versus standard CHOP for the treatment of patients with peripheral T-cell lymphoma. The results will be presented at 6:15 p.m. PT on Monday, Dec. 3 in room 6F of the San Diego convention center (Abstract 997).
Previously reported blinded pooled data showed that the treatment was well tolerated with 3-year PFS of 53% and OS of 73%.
“This should be a new standard of care for T-cell lymphomas,” Dr. Hill said.
CAR T-cell therapy
There are a number of abstracts featuring the latest results on CAR T-cell therapy. Helen Heslop, MD, of Baylor College of Medicine, Houston, recommended an updated analysis from the ELIANA study, which looked at the efficacy and safety of tisagenlecleucel in for children and young adults with relapsed/refractory acute lymphoblastic leukemia (ALL).
“Longer-term follow-up of the ELIANA study shows encouraging remission-duration data in pediatric and young adults with ALL without additional therapy,” Dr. Heslop said.
The findings will be presented at 4:30 p.m. PT on Monday, Dec. 3 in room 6A at the San Diego Convention Center (Abstract 895).
Another notable presentation will feature results from a phase 1B/2 trial evaluating infusion of CAR T cells targeting the CD30 molecule and encoding the CD28 endodomain (CD30.CAR-Ts) after lymphodepleting chemotherapy in patients with relapsed or refractory CD30+ Hodgkin lymphoma and non-Hodgkin lymphoma.
The researchers will report that there was a significant PFS advances for who received the highest dose level of the CAR T treatment, combined with bendamustine and fludarabine.
The study will be presented at 11 a.m. PT on Monday, Dec. 3 in room 6F at the San Diego Convention Center (Abstract 681).
Dr. Heslop also recommends another study being presented in the same session, which also shows encouraging results with CD30.CAR-Ts. Dr. Heslop is one of the co-investigators on the phase 1 RELY-30 trial, which is evaluating the efficacy of CD30.CAR-Ts after lymphodepleting chemotherapy. Preliminary results suggest a substantial improvement in efficacy. The findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in room 6F of the San Diego Convention Center (Abstract 680).
MDS/MPN
Vikas Gupta, MD, of Princess Margaret Cancer Center in Toronto, highlighted three abstracts to watch in the areas of myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN).
The phase 3 Medalist trial is a randomized double-blind placebo controlled study of luspatercept to treatment anemia in patients with MDS with ring sideroblasts who require red blood cell transfusion. The researchers will report significantly reduced transfusion burdens for luspatercept, compared with placebo.
“This is a practice-changing, pivotal trial in the field of MDS for the treatment of anemia,” Dr. Gupta said.
The findings will be presented at 2 p.m. PT on Sunday, Dec. 2 during the plenary session in Hall AB in the San Diego Convention Center (Abstract 1).
Also during the Sunday plenary session is a presentation on MPN therapy (Abstract 4). Researchers will present data on secreted mutant calreticulins as rogue cytokines trigger thrombopoietin receptor (TpoR) activation, specifically in CALR-mutated cells.
“This study investigates in to the mechanistic oncogenetic aspects of mutant calreticulin, and has potential for therapeutic approaches in the future,” Dr. Gupta said.
The ASH meeting will also feature the final analysis of the MPN-RC 112 consortium trial of pegylated interferon alfa-2a versus hydroxyurea for the treatment of high-risk polycythemia vera (PV) and essential thrombocythemia (ET). The researchers will report that the CR rates at 12 and 24 months were similar in patients treated with pegylated interferon alfa-2a and hydroxyurea, but pegylated interferon alfa-2a was associated with a higher rate of serious toxicities.
“There is a continuous debate on optimal first-line cytoreductive therapy for high risk PV/ET, and this is one of the first randomized study to answer this question,” Dr. Gupta said.
The findings will be presented at 7 a.m. PT on Monday, Dec. 3 in Grand Hall D at the Manchester Grand Hyatt San Diego (Abstract 577).
AML
For attendees interested in the latest developments in acute myeloid leukemia, Thomas Fischer, MD, of Otto-von-Guericke-University Magdeburg (Germany), highlighted three don’t-miss sessions.
In an analysis of a large cohort of FLT3-ITD mutated AML patients in the RATIFY trial, researchers looked at the prognostic impact of ITD insertion site.
“Interestingly, in this large cohort of 452 FLT3-ITD mutated AML, the negative prognostic impact of beta1-sheet insertion site of FLT3-ITD could be confirmed,” Dr. Fischer said. “Further analysis of a potential predictive effect on outcome of midostaurin treatment is ongoing and will be very interesting.”
The findings will be presented at 5 p.m. PT on Sunday, Dec. 2 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 435).
Another notable presentation features results from the phase 2 RADIUS trial, a randomized study comparing standard of care, with and without midostaurin, after allogeneic stem cell transplant in FLT3-ITD–mutated AML.
“Here, efficacy and toxicity of midostaurin was investigated in a [minimal residual disease] situation post-alloSCT,” Dr. Fischer said. “Interestingly, adding midostaurin to standard of care reduced the risk of relapse at 18 months post-alloSCT by 46%.”
The complete findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 662).
Dr. Fischer singled out another study looking at the efficacy and safety of single-agent quizartinib in patients with FLT3-ITD mutated AML. In this large, randomized trial the researchers noted a significant improvement in CR rates and survival benefit with the single agent FLT3 inhibitors, compared with salvage chemotherapy for patients with relapsed/refractory mutated AML.
The findings will be presented at 8 a.m. on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 563).
Notable posters
Iberia Romina Sosa, MD, PhD, of Baylor College of Medicine in Houston, suggested several posters worth visiting in the areas of thrombosis and bleeding.
Poster 1134 looks at the TNF-alpha driven inflammation and mitochondrial dysfunction in the platelet hyperreactivity of aging and MPN.
How do you know if your therapy for thrombotic thrombocytopenic purpura is working? Poster 3736 examines the measurement of cell-derived microparticles as a possible tool to monitor response to therapy.
You don’t have to be taking aspirin to have a bleeding profile characteristic with consumption of a cyclooxygenase inhibitor. Poster 1156 provides a first report of a platelet function disorder caused by autosomal recessive inheritance of PTGS1.
Poster 2477 takes a closer look at fitusiran, an antithrombin inhibitor, which improves thrombin generation in patients with hemophilia A or B. Protocol amendments for safety monitoring move fitusiran to phase 3 trials, Dr. Sosa said.
With more than 3,000 scientific abstracts at the 2018 annual meeting of the American Society of Hematology, it can be tough to figure out what research is most relevant to practice. But the editorial advisory board of Hematology News is making it easier this year with their picks for what to watch and why.
Lymphomas
Brian T. Hill, MD, of the Cleveland Clinic, offered his top picks in lymphoma research. Results of the phase 3 international Alliance North American Intergroup Study A041202 will be presented during the ASH plenary session at 2 p.m. PT on Sunday, Dec. 2 in Hall AB of the San Diego Convention Center (Abstract 6). The study compared bendamustine plus rituximab with ibrutinib and the combination of ibrutinib plus rituximab to see if the ibrutinib-containing therapies would have superior progression-free survival (PFS) in chronic lymphocytic leukemia (CLL), compared with chemoimmunotherapy. Results indicate that ibrutinib had superior PFS in older patients with CLL and could be a standard of care in this population.
The study is worth watching because it is the first report of a head-to-head trial of chemotherapy versus ibrutinib for first-line treatment of CLL, Dr. Hill said.
Two more studies offer important reports of “real world” experiences with chimeric antigen receptor (CAR) T-cell therapy.
In one multicenter retrospective study, researchers evaluated the outcomes of axicabtagene ciloleucel (axi-cel) CAR T-cell therapy for relapsed/refractory aggressive B-cell lymphoma when it is used a standard care. The researchers will report that 30-day responses in the real-world setting were comparable to the best responses seen in the ZUMA-1 trial. The full results will be reported at 9:30 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 91).
Another retrospective analysis looked at the use of axi-cell and revealed some critical differences from ZUMA-1, specifically the overall response rate (ORR) and complete response (CR) rate were lower than those reported in the pivotal clinical trial. The findings will be reported at 9:45 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 92).
Researchers will also present the unblinded results from the ECHELON-2 study, which compared the efficacy and safety of brentuximab vedotin in combination with cyclophosphamide, doxorubicin, and prednisone (CHP) versus standard CHOP for the treatment of patients with peripheral T-cell lymphoma. The results will be presented at 6:15 p.m. PT on Monday, Dec. 3 in room 6F of the San Diego convention center (Abstract 997).
Previously reported blinded pooled data showed that the treatment was well tolerated with 3-year PFS of 53% and OS of 73%.
“This should be a new standard of care for T-cell lymphomas,” Dr. Hill said.
CAR T-cell therapy
There are a number of abstracts featuring the latest results on CAR T-cell therapy. Helen Heslop, MD, of Baylor College of Medicine, Houston, recommended an updated analysis from the ELIANA study, which looked at the efficacy and safety of tisagenlecleucel in for children and young adults with relapsed/refractory acute lymphoblastic leukemia (ALL).
“Longer-term follow-up of the ELIANA study shows encouraging remission-duration data in pediatric and young adults with ALL without additional therapy,” Dr. Heslop said.
The findings will be presented at 4:30 p.m. PT on Monday, Dec. 3 in room 6A at the San Diego Convention Center (Abstract 895).
Another notable presentation will feature results from a phase 1B/2 trial evaluating infusion of CAR T cells targeting the CD30 molecule and encoding the CD28 endodomain (CD30.CAR-Ts) after lymphodepleting chemotherapy in patients with relapsed or refractory CD30+ Hodgkin lymphoma and non-Hodgkin lymphoma.
The researchers will report that there was a significant PFS advances for who received the highest dose level of the CAR T treatment, combined with bendamustine and fludarabine.
The study will be presented at 11 a.m. PT on Monday, Dec. 3 in room 6F at the San Diego Convention Center (Abstract 681).
Dr. Heslop also recommends another study being presented in the same session, which also shows encouraging results with CD30.CAR-Ts. Dr. Heslop is one of the co-investigators on the phase 1 RELY-30 trial, which is evaluating the efficacy of CD30.CAR-Ts after lymphodepleting chemotherapy. Preliminary results suggest a substantial improvement in efficacy. The findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in room 6F of the San Diego Convention Center (Abstract 680).
MDS/MPN
Vikas Gupta, MD, of Princess Margaret Cancer Center in Toronto, highlighted three abstracts to watch in the areas of myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN).
The phase 3 Medalist trial is a randomized double-blind placebo controlled study of luspatercept to treatment anemia in patients with MDS with ring sideroblasts who require red blood cell transfusion. The researchers will report significantly reduced transfusion burdens for luspatercept, compared with placebo.
“This is a practice-changing, pivotal trial in the field of MDS for the treatment of anemia,” Dr. Gupta said.
The findings will be presented at 2 p.m. PT on Sunday, Dec. 2 during the plenary session in Hall AB in the San Diego Convention Center (Abstract 1).
Also during the Sunday plenary session is a presentation on MPN therapy (Abstract 4). Researchers will present data on secreted mutant calreticulins as rogue cytokines trigger thrombopoietin receptor (TpoR) activation, specifically in CALR-mutated cells.
“This study investigates in to the mechanistic oncogenetic aspects of mutant calreticulin, and has potential for therapeutic approaches in the future,” Dr. Gupta said.
The ASH meeting will also feature the final analysis of the MPN-RC 112 consortium trial of pegylated interferon alfa-2a versus hydroxyurea for the treatment of high-risk polycythemia vera (PV) and essential thrombocythemia (ET). The researchers will report that the CR rates at 12 and 24 months were similar in patients treated with pegylated interferon alfa-2a and hydroxyurea, but pegylated interferon alfa-2a was associated with a higher rate of serious toxicities.
“There is a continuous debate on optimal first-line cytoreductive therapy for high risk PV/ET, and this is one of the first randomized study to answer this question,” Dr. Gupta said.
The findings will be presented at 7 a.m. PT on Monday, Dec. 3 in Grand Hall D at the Manchester Grand Hyatt San Diego (Abstract 577).
AML
For attendees interested in the latest developments in acute myeloid leukemia, Thomas Fischer, MD, of Otto-von-Guericke-University Magdeburg (Germany), highlighted three don’t-miss sessions.
In an analysis of a large cohort of FLT3-ITD mutated AML patients in the RATIFY trial, researchers looked at the prognostic impact of ITD insertion site.
“Interestingly, in this large cohort of 452 FLT3-ITD mutated AML, the negative prognostic impact of beta1-sheet insertion site of FLT3-ITD could be confirmed,” Dr. Fischer said. “Further analysis of a potential predictive effect on outcome of midostaurin treatment is ongoing and will be very interesting.”
The findings will be presented at 5 p.m. PT on Sunday, Dec. 2 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 435).
Another notable presentation features results from the phase 2 RADIUS trial, a randomized study comparing standard of care, with and without midostaurin, after allogeneic stem cell transplant in FLT3-ITD–mutated AML.
“Here, efficacy and toxicity of midostaurin was investigated in a [minimal residual disease] situation post-alloSCT,” Dr. Fischer said. “Interestingly, adding midostaurin to standard of care reduced the risk of relapse at 18 months post-alloSCT by 46%.”
The complete findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 662).
Dr. Fischer singled out another study looking at the efficacy and safety of single-agent quizartinib in patients with FLT3-ITD mutated AML. In this large, randomized trial the researchers noted a significant improvement in CR rates and survival benefit with the single agent FLT3 inhibitors, compared with salvage chemotherapy for patients with relapsed/refractory mutated AML.
The findings will be presented at 8 a.m. on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 563).
Notable posters
Iberia Romina Sosa, MD, PhD, of Baylor College of Medicine in Houston, suggested several posters worth visiting in the areas of thrombosis and bleeding.
Poster 1134 looks at the TNF-alpha driven inflammation and mitochondrial dysfunction in the platelet hyperreactivity of aging and MPN.
How do you know if your therapy for thrombotic thrombocytopenic purpura is working? Poster 3736 examines the measurement of cell-derived microparticles as a possible tool to monitor response to therapy.
You don’t have to be taking aspirin to have a bleeding profile characteristic with consumption of a cyclooxygenase inhibitor. Poster 1156 provides a first report of a platelet function disorder caused by autosomal recessive inheritance of PTGS1.
Poster 2477 takes a closer look at fitusiran, an antithrombin inhibitor, which improves thrombin generation in patients with hemophilia A or B. Protocol amendments for safety monitoring move fitusiran to phase 3 trials, Dr. Sosa said.
With more than 3,000 scientific abstracts at the 2018 annual meeting of the American Society of Hematology, it can be tough to figure out what research is most relevant to practice. But the editorial advisory board of Hematology News is making it easier this year with their picks for what to watch and why.
Lymphomas
Brian T. Hill, MD, of the Cleveland Clinic, offered his top picks in lymphoma research. Results of the phase 3 international Alliance North American Intergroup Study A041202 will be presented during the ASH plenary session at 2 p.m. PT on Sunday, Dec. 2 in Hall AB of the San Diego Convention Center (Abstract 6). The study compared bendamustine plus rituximab with ibrutinib and the combination of ibrutinib plus rituximab to see if the ibrutinib-containing therapies would have superior progression-free survival (PFS) in chronic lymphocytic leukemia (CLL), compared with chemoimmunotherapy. Results indicate that ibrutinib had superior PFS in older patients with CLL and could be a standard of care in this population.
The study is worth watching because it is the first report of a head-to-head trial of chemotherapy versus ibrutinib for first-line treatment of CLL, Dr. Hill said.
Two more studies offer important reports of “real world” experiences with chimeric antigen receptor (CAR) T-cell therapy.
In one multicenter retrospective study, researchers evaluated the outcomes of axicabtagene ciloleucel (axi-cel) CAR T-cell therapy for relapsed/refractory aggressive B-cell lymphoma when it is used a standard care. The researchers will report that 30-day responses in the real-world setting were comparable to the best responses seen in the ZUMA-1 trial. The full results will be reported at 9:30 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 91).
Another retrospective analysis looked at the use of axi-cell and revealed some critical differences from ZUMA-1, specifically the overall response rate (ORR) and complete response (CR) rate were lower than those reported in the pivotal clinical trial. The findings will be reported at 9:45 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 92).
Researchers will also present the unblinded results from the ECHELON-2 study, which compared the efficacy and safety of brentuximab vedotin in combination with cyclophosphamide, doxorubicin, and prednisone (CHP) versus standard CHOP for the treatment of patients with peripheral T-cell lymphoma. The results will be presented at 6:15 p.m. PT on Monday, Dec. 3 in room 6F of the San Diego convention center (Abstract 997).
Previously reported blinded pooled data showed that the treatment was well tolerated with 3-year PFS of 53% and OS of 73%.
“This should be a new standard of care for T-cell lymphomas,” Dr. Hill said.
CAR T-cell therapy
There are a number of abstracts featuring the latest results on CAR T-cell therapy. Helen Heslop, MD, of Baylor College of Medicine, Houston, recommended an updated analysis from the ELIANA study, which looked at the efficacy and safety of tisagenlecleucel in for children and young adults with relapsed/refractory acute lymphoblastic leukemia (ALL).
“Longer-term follow-up of the ELIANA study shows encouraging remission-duration data in pediatric and young adults with ALL without additional therapy,” Dr. Heslop said.
The findings will be presented at 4:30 p.m. PT on Monday, Dec. 3 in room 6A at the San Diego Convention Center (Abstract 895).
Another notable presentation will feature results from a phase 1B/2 trial evaluating infusion of CAR T cells targeting the CD30 molecule and encoding the CD28 endodomain (CD30.CAR-Ts) after lymphodepleting chemotherapy in patients with relapsed or refractory CD30+ Hodgkin lymphoma and non-Hodgkin lymphoma.
The researchers will report that there was a significant PFS advances for who received the highest dose level of the CAR T treatment, combined with bendamustine and fludarabine.
The study will be presented at 11 a.m. PT on Monday, Dec. 3 in room 6F at the San Diego Convention Center (Abstract 681).
Dr. Heslop also recommends another study being presented in the same session, which also shows encouraging results with CD30.CAR-Ts. Dr. Heslop is one of the co-investigators on the phase 1 RELY-30 trial, which is evaluating the efficacy of CD30.CAR-Ts after lymphodepleting chemotherapy. Preliminary results suggest a substantial improvement in efficacy. The findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in room 6F of the San Diego Convention Center (Abstract 680).
MDS/MPN
Vikas Gupta, MD, of Princess Margaret Cancer Center in Toronto, highlighted three abstracts to watch in the areas of myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN).
The phase 3 Medalist trial is a randomized double-blind placebo controlled study of luspatercept to treatment anemia in patients with MDS with ring sideroblasts who require red blood cell transfusion. The researchers will report significantly reduced transfusion burdens for luspatercept, compared with placebo.
“This is a practice-changing, pivotal trial in the field of MDS for the treatment of anemia,” Dr. Gupta said.
The findings will be presented at 2 p.m. PT on Sunday, Dec. 2 during the plenary session in Hall AB in the San Diego Convention Center (Abstract 1).
Also during the Sunday plenary session is a presentation on MPN therapy (Abstract 4). Researchers will present data on secreted mutant calreticulins as rogue cytokines trigger thrombopoietin receptor (TpoR) activation, specifically in CALR-mutated cells.
“This study investigates in to the mechanistic oncogenetic aspects of mutant calreticulin, and has potential for therapeutic approaches in the future,” Dr. Gupta said.
The ASH meeting will also feature the final analysis of the MPN-RC 112 consortium trial of pegylated interferon alfa-2a versus hydroxyurea for the treatment of high-risk polycythemia vera (PV) and essential thrombocythemia (ET). The researchers will report that the CR rates at 12 and 24 months were similar in patients treated with pegylated interferon alfa-2a and hydroxyurea, but pegylated interferon alfa-2a was associated with a higher rate of serious toxicities.
“There is a continuous debate on optimal first-line cytoreductive therapy for high risk PV/ET, and this is one of the first randomized study to answer this question,” Dr. Gupta said.
The findings will be presented at 7 a.m. PT on Monday, Dec. 3 in Grand Hall D at the Manchester Grand Hyatt San Diego (Abstract 577).
AML
For attendees interested in the latest developments in acute myeloid leukemia, Thomas Fischer, MD, of Otto-von-Guericke-University Magdeburg (Germany), highlighted three don’t-miss sessions.
In an analysis of a large cohort of FLT3-ITD mutated AML patients in the RATIFY trial, researchers looked at the prognostic impact of ITD insertion site.
“Interestingly, in this large cohort of 452 FLT3-ITD mutated AML, the negative prognostic impact of beta1-sheet insertion site of FLT3-ITD could be confirmed,” Dr. Fischer said. “Further analysis of a potential predictive effect on outcome of midostaurin treatment is ongoing and will be very interesting.”
The findings will be presented at 5 p.m. PT on Sunday, Dec. 2 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 435).
Another notable presentation features results from the phase 2 RADIUS trial, a randomized study comparing standard of care, with and without midostaurin, after allogeneic stem cell transplant in FLT3-ITD–mutated AML.
“Here, efficacy and toxicity of midostaurin was investigated in a [minimal residual disease] situation post-alloSCT,” Dr. Fischer said. “Interestingly, adding midostaurin to standard of care reduced the risk of relapse at 18 months post-alloSCT by 46%.”
The complete findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 662).
Dr. Fischer singled out another study looking at the efficacy and safety of single-agent quizartinib in patients with FLT3-ITD mutated AML. In this large, randomized trial the researchers noted a significant improvement in CR rates and survival benefit with the single agent FLT3 inhibitors, compared with salvage chemotherapy for patients with relapsed/refractory mutated AML.
The findings will be presented at 8 a.m. on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 563).
Notable posters
Iberia Romina Sosa, MD, PhD, of Baylor College of Medicine in Houston, suggested several posters worth visiting in the areas of thrombosis and bleeding.
Poster 1134 looks at the TNF-alpha driven inflammation and mitochondrial dysfunction in the platelet hyperreactivity of aging and MPN.
How do you know if your therapy for thrombotic thrombocytopenic purpura is working? Poster 3736 examines the measurement of cell-derived microparticles as a possible tool to monitor response to therapy.
You don’t have to be taking aspirin to have a bleeding profile characteristic with consumption of a cyclooxygenase inhibitor. Poster 1156 provides a first report of a platelet function disorder caused by autosomal recessive inheritance of PTGS1.
Poster 2477 takes a closer look at fitusiran, an antithrombin inhibitor, which improves thrombin generation in patients with hemophilia A or B. Protocol amendments for safety monitoring move fitusiran to phase 3 trials, Dr. Sosa said.
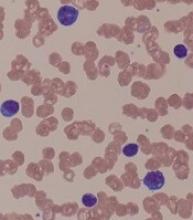
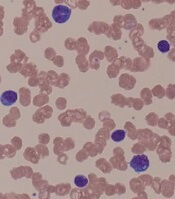
Bortezomib may overcome resistance in WM
Bortezomib may help overcome treatment resistance in patients with Waldenström’s macroglobulinemia (WM) and CXCR4 mutations, according to a new study.
Researchers assessed the impact of treatment with bortezomib and rituximab in patients with WM, based on their CXCR4 mutation status.
The team found no significant difference in progression-free survival or overall survival between patients with CXCR4 mutations and those with wild-type CXCR4.
Romanos Sklavenitis-Pistofidis, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts, and his colleagues reported this discovery in Blood.
The researchers’ main analysis included 43 patients with WM, 17 (39.5%) of whom had a CXCR4 mutation.
All patients who carried a CXCR4 mutation also had MYD88 L265P. Ten patients had frameshift mutations, one patient had a nonsense mutation, and six patients had missense mutations.
The patients were treated with bortezomib and rituximab, either upfront (n=14) or in the relapsed/refractory (n=29) setting, as part of a phase 2 trial.
Bortezomib was given at 1.6 mg/m2 on days 1, 8, and 15 every 28 days for six cycles, and rituximab was given at 375 mg/m2 on days 1, 8, 15, and 22 during cycles one and four. Patients were taken off therapy after two cycles if they had progressive disease.
The median follow-up was 90.7 months.
The researchers found no significant difference between CXCR4-mutated and wild-type patients when it came to progression-free survival (P=0.994) or overall survival (P=0.407).
The researchers repeated their analysis after excluding six patients with missense mutations and accounting for different treatment settings and found that survival remained unchanged.
“We report, for the first time, that a bortezomib-based combination is impervious to the impact of CXCR4 mutations in a cohort of patients with WM,” the researchers wrote.
“Previously, we had shown this to be true in WM cell lines, whereby genetically engineering BCWM.1 and MWCL-1 to overexpress CXCR4 had no impact on bortezomib resistance.”
The researchers noted, however, that the mechanism at work here may be different than what is seen with bortezomib in other cancers.
“Different experiments have linked CXCR4 expression and bortezomib in a variety of ways in other hematological malignancies, including multiple myeloma,” the researchers wrote.
“However, despite the complicated association in those cancer types, in WM, there seems to be a consistently neutral effect of CXCR4 mutations on bortezomib resistance in both cell line and patient data.”
The researchers recommended that this theory be tested in a prospective trial of bortezomib-based therapy in WM patients with CXCR4 mutations.
Another thing to be determined, they said, is the role of rituximab in the survival results seen in the current analysis.
This study was supported by the National Institutes of Health, the Leukemia and Lymphoma Society, and the International Waldenström Macroglobulinemia Foundation. One of the authors reported consulting and research funding from Takeda, which markets bortezomib, and other companies.
Bortezomib may help overcome treatment resistance in patients with Waldenström’s macroglobulinemia (WM) and CXCR4 mutations, according to a new study.
Researchers assessed the impact of treatment with bortezomib and rituximab in patients with WM, based on their CXCR4 mutation status.
The team found no significant difference in progression-free survival or overall survival between patients with CXCR4 mutations and those with wild-type CXCR4.
Romanos Sklavenitis-Pistofidis, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts, and his colleagues reported this discovery in Blood.
The researchers’ main analysis included 43 patients with WM, 17 (39.5%) of whom had a CXCR4 mutation.
All patients who carried a CXCR4 mutation also had MYD88 L265P. Ten patients had frameshift mutations, one patient had a nonsense mutation, and six patients had missense mutations.
The patients were treated with bortezomib and rituximab, either upfront (n=14) or in the relapsed/refractory (n=29) setting, as part of a phase 2 trial.
Bortezomib was given at 1.6 mg/m2 on days 1, 8, and 15 every 28 days for six cycles, and rituximab was given at 375 mg/m2 on days 1, 8, 15, and 22 during cycles one and four. Patients were taken off therapy after two cycles if they had progressive disease.
The median follow-up was 90.7 months.
The researchers found no significant difference between CXCR4-mutated and wild-type patients when it came to progression-free survival (P=0.994) or overall survival (P=0.407).
The researchers repeated their analysis after excluding six patients with missense mutations and accounting for different treatment settings and found that survival remained unchanged.
“We report, for the first time, that a bortezomib-based combination is impervious to the impact of CXCR4 mutations in a cohort of patients with WM,” the researchers wrote.
“Previously, we had shown this to be true in WM cell lines, whereby genetically engineering BCWM.1 and MWCL-1 to overexpress CXCR4 had no impact on bortezomib resistance.”
The researchers noted, however, that the mechanism at work here may be different than what is seen with bortezomib in other cancers.
“Different experiments have linked CXCR4 expression and bortezomib in a variety of ways in other hematological malignancies, including multiple myeloma,” the researchers wrote.
“However, despite the complicated association in those cancer types, in WM, there seems to be a consistently neutral effect of CXCR4 mutations on bortezomib resistance in both cell line and patient data.”
The researchers recommended that this theory be tested in a prospective trial of bortezomib-based therapy in WM patients with CXCR4 mutations.
Another thing to be determined, they said, is the role of rituximab in the survival results seen in the current analysis.
This study was supported by the National Institutes of Health, the Leukemia and Lymphoma Society, and the International Waldenström Macroglobulinemia Foundation. One of the authors reported consulting and research funding from Takeda, which markets bortezomib, and other companies.
Bortezomib may help overcome treatment resistance in patients with Waldenström’s macroglobulinemia (WM) and CXCR4 mutations, according to a new study.
Researchers assessed the impact of treatment with bortezomib and rituximab in patients with WM, based on their CXCR4 mutation status.
The team found no significant difference in progression-free survival or overall survival between patients with CXCR4 mutations and those with wild-type CXCR4.
Romanos Sklavenitis-Pistofidis, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts, and his colleagues reported this discovery in Blood.
The researchers’ main analysis included 43 patients with WM, 17 (39.5%) of whom had a CXCR4 mutation.
All patients who carried a CXCR4 mutation also had MYD88 L265P. Ten patients had frameshift mutations, one patient had a nonsense mutation, and six patients had missense mutations.
The patients were treated with bortezomib and rituximab, either upfront (n=14) or in the relapsed/refractory (n=29) setting, as part of a phase 2 trial.
Bortezomib was given at 1.6 mg/m2 on days 1, 8, and 15 every 28 days for six cycles, and rituximab was given at 375 mg/m2 on days 1, 8, 15, and 22 during cycles one and four. Patients were taken off therapy after two cycles if they had progressive disease.
The median follow-up was 90.7 months.
The researchers found no significant difference between CXCR4-mutated and wild-type patients when it came to progression-free survival (P=0.994) or overall survival (P=0.407).
The researchers repeated their analysis after excluding six patients with missense mutations and accounting for different treatment settings and found that survival remained unchanged.
“We report, for the first time, that a bortezomib-based combination is impervious to the impact of CXCR4 mutations in a cohort of patients with WM,” the researchers wrote.
“Previously, we had shown this to be true in WM cell lines, whereby genetically engineering BCWM.1 and MWCL-1 to overexpress CXCR4 had no impact on bortezomib resistance.”
The researchers noted, however, that the mechanism at work here may be different than what is seen with bortezomib in other cancers.
“Different experiments have linked CXCR4 expression and bortezomib in a variety of ways in other hematological malignancies, including multiple myeloma,” the researchers wrote.
“However, despite the complicated association in those cancer types, in WM, there seems to be a consistently neutral effect of CXCR4 mutations on bortezomib resistance in both cell line and patient data.”
The researchers recommended that this theory be tested in a prospective trial of bortezomib-based therapy in WM patients with CXCR4 mutations.
Another thing to be determined, they said, is the role of rituximab in the survival results seen in the current analysis.
This study was supported by the National Institutes of Health, the Leukemia and Lymphoma Society, and the International Waldenström Macroglobulinemia Foundation. One of the authors reported consulting and research funding from Takeda, which markets bortezomib, and other companies.
NHL patients report fear, isolation during chemo
Patients undergoing chemotherapy for non-Hodgkin lymphoma (NHL) report feeling isolated and uncertain, and in some cases suicidal, according to a small qualitative study.
Daren Chircop, MS, and Josianne Scerri, PhD, of the University of Malta performed in-depth interviews with four men and two women with aggressive NHL who were midway through CHOP chemotherapy at a cancer center in Malta.
The findings, which were published in the European Journal of Oncology Nursing, highlight three overarching themes. Patients reported that they felt like they were living on an “emotional roller coaster, they were becoming dependent on others, and they were facing an uncertain future.
Specifically, being admitted to a hospital for chemotherapy brought on a fear of the unknown. While being released from the hospital caused anxiety about what would happen once they left the around-the-clock care of doctors and nurses.
The side effects of the chemotherapy – particularly fatigue – led to feelings of being dependent of others and a fear of becoming a burden to families, and even to nurses.
For four of the patients, who were not allowed to leave their hospital rooms during treatment, they reported feeling “imprisoned” and isolated.
Uncertainty was another common theme, with all of the patients reporting that they did not know if they would recover from their cancer and feeling that they had no control over the future.
Additionally, two of the patients experienced suicidal thoughts related to the side effects of treatment.
While many of these feelings are similar to reports by patients with other types of cancer, the researchers suggested that NHL patients experience a greater sense of isolation because they may not be able to leave their hospital rooms while undergoing chemotherapy due to a higher risk of infection.
“There is the need for ongoing psychological support to be available throughout the treatment period,” the researchers wrote. “Moreover, as NHL patients are affected both physically and emotionally whilst undergoing chemotherapy, the engagement of these individuals in some physical activity could be of benefit to them.”
There was no outside funding for the study and the researchers reported having no financial disclosures.
SOURCE: Chircop D et al. Eur J Oncol Nurs. 2018 Aug;35:117-21.
Patients undergoing chemotherapy for non-Hodgkin lymphoma (NHL) report feeling isolated and uncertain, and in some cases suicidal, according to a small qualitative study.
Daren Chircop, MS, and Josianne Scerri, PhD, of the University of Malta performed in-depth interviews with four men and two women with aggressive NHL who were midway through CHOP chemotherapy at a cancer center in Malta.
The findings, which were published in the European Journal of Oncology Nursing, highlight three overarching themes. Patients reported that they felt like they were living on an “emotional roller coaster, they were becoming dependent on others, and they were facing an uncertain future.
Specifically, being admitted to a hospital for chemotherapy brought on a fear of the unknown. While being released from the hospital caused anxiety about what would happen once they left the around-the-clock care of doctors and nurses.
The side effects of the chemotherapy – particularly fatigue – led to feelings of being dependent of others and a fear of becoming a burden to families, and even to nurses.
For four of the patients, who were not allowed to leave their hospital rooms during treatment, they reported feeling “imprisoned” and isolated.
Uncertainty was another common theme, with all of the patients reporting that they did not know if they would recover from their cancer and feeling that they had no control over the future.
Additionally, two of the patients experienced suicidal thoughts related to the side effects of treatment.
While many of these feelings are similar to reports by patients with other types of cancer, the researchers suggested that NHL patients experience a greater sense of isolation because they may not be able to leave their hospital rooms while undergoing chemotherapy due to a higher risk of infection.
“There is the need for ongoing psychological support to be available throughout the treatment period,” the researchers wrote. “Moreover, as NHL patients are affected both physically and emotionally whilst undergoing chemotherapy, the engagement of these individuals in some physical activity could be of benefit to them.”
There was no outside funding for the study and the researchers reported having no financial disclosures.
SOURCE: Chircop D et al. Eur J Oncol Nurs. 2018 Aug;35:117-21.
Patients undergoing chemotherapy for non-Hodgkin lymphoma (NHL) report feeling isolated and uncertain, and in some cases suicidal, according to a small qualitative study.
Daren Chircop, MS, and Josianne Scerri, PhD, of the University of Malta performed in-depth interviews with four men and two women with aggressive NHL who were midway through CHOP chemotherapy at a cancer center in Malta.
The findings, which were published in the European Journal of Oncology Nursing, highlight three overarching themes. Patients reported that they felt like they were living on an “emotional roller coaster, they were becoming dependent on others, and they were facing an uncertain future.
Specifically, being admitted to a hospital for chemotherapy brought on a fear of the unknown. While being released from the hospital caused anxiety about what would happen once they left the around-the-clock care of doctors and nurses.
The side effects of the chemotherapy – particularly fatigue – led to feelings of being dependent of others and a fear of becoming a burden to families, and even to nurses.
For four of the patients, who were not allowed to leave their hospital rooms during treatment, they reported feeling “imprisoned” and isolated.
Uncertainty was another common theme, with all of the patients reporting that they did not know if they would recover from their cancer and feeling that they had no control over the future.
Additionally, two of the patients experienced suicidal thoughts related to the side effects of treatment.
While many of these feelings are similar to reports by patients with other types of cancer, the researchers suggested that NHL patients experience a greater sense of isolation because they may not be able to leave their hospital rooms while undergoing chemotherapy due to a higher risk of infection.
“There is the need for ongoing psychological support to be available throughout the treatment period,” the researchers wrote. “Moreover, as NHL patients are affected both physically and emotionally whilst undergoing chemotherapy, the engagement of these individuals in some physical activity could be of benefit to them.”
There was no outside funding for the study and the researchers reported having no financial disclosures.
SOURCE: Chircop D et al. Eur J Oncol Nurs. 2018 Aug;35:117-21.
FROM EUROPEAN JOURNAL OF ONCOLOGY NURSING
Key clinical point:
Major finding: Patients reported three themes while undergoing chemotherapy: living an emotional roller coaster, becoming dependent on others, and facing an uncertain future.
Study details: A qualitative study of six adults patients with non-Hodgkin lymphoma who were undergoing chemotherapy.
Disclosures: There was no outside funding for the study and the researchers reported having no financial disclosures.
Source: Chircop D et al. Eur J Oncol Nurs. 2018 Aug;35:117-21.