Opinion

More tools for the COVID toolbox
- Author:
- Stephen I. Pelton, MD
The Emergency Use Authorization authorized use of specific monoclonal antibodies for individuals 12 years and above with a minimum weight of 40 kg...
Opinion

COVID-19 in children and adolescents: Disease burden and severity
- Author:
- Stephen I. Pelton, MD
Updating and summarizing the pediatric experience for the pediatric community on what children and adolescents have experienced because of SARS-...
Opinion

Direct-acting agents cure hepatitis C in children
- Author:
- Vishakha Sabharwal, MD
- Carole Moloney, PNP
- Stephen I. Pelton, MD
There now are three direct-acting antiviral agents available to treat HCV in children.
News

Consider COVID-19–associated multisystem hyperinflammatory syndrome
- Author:
- Stephen I. Pelton, MD
- Ingrid Camelo, MD
It is hypothesized that the syndrome potentially is a late-onset inflammatory process or potentially an antibody-triggered inflammatory process....
Opinion
COVID-19 in children, pregnant women: What do we know?
- Author:
- Stephen I. Pelton, MD
The remarkable observation is how few seriously ill children have been identified in the face of global spread...
Opinion
Flu vaccine: Larger impact on influenza burden than you thought?
- Author:
- Stephen I. Pelton, MD
Even the current modestly effective flu vaccines have significant benefit, protecting U.S. populations from serious disease and death.
News
Identifying CMV infection in asymptomatic newborns – one step closer?
- Author:
- Rotem Lapidot, MD
- Stephen I. Pelton, MD
A new assay may provide the way to identify asymptomatic congenital CMV infection.
Opinion
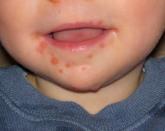
Hand, foot, and mouth disease: From self-limited to fatal
- Author:
- Stephen I. Pelton, MD
Hand, foot, and mouth disease generally is self-limited and brief, but potentially fatal complications can...
Opinion
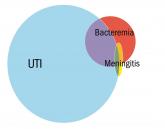
Evaluating fever in the first 90 days of life
- Author:
- Stephen I. Pelton, MD
The current approach to the febrile infant less than 90 days of age is based on risk stratification.
Opinion

A ‘game changer’ for pediatric HIV
- Author:
- Carole H. Moloney, CPNP
- Stephen I. Pelton, MD
A look at how vertical transmission of HIV was largely halted in less than 25 years.
Opinion
PANS and PANDAS – A step forward?
- Author:
- Stephen I. Pelton, MD
There’s new info to guide you in managing PANS and PANDAS.
Article

Enterovirus D68 – An emerging threat to child health
- Author:
- Stephen I. Pelton, MD
Dr. Stephen I. Pelton discusses the emerging threat of enterovirus D68.
News

Influenza: A vaccine we love to hate
- Author:
- Stephen I. Pelton, MD
News

Appendicitis, antibiotics, and surgery: An evolving trilogy
- Author:
- Stephen I. Pelton, MD
Dr. Stephen I. Pelton discusses new information about trying antibiotics rather than going straight to surgery to treat appendicitis.
News

Protecting pregnant women, infants from infections
- Author:
- Stephen I. Pelton, MD
Dr. Stephen I. Pelton emphasizes the importance of immunizing pregnant women to protect infants too young to be protected by childhood vaccines....
