User login
High plasma IgE predicts COPD exacerbation, mortality
COPD patients with high plasma immunoglobulin E are more likely to have exacerbations and die from any cause, based on a Danish population-cohort study.
hinting at different subsets of patients with COPD, lead author Yunus Çolak MD, PhD, of Copenhagen University Hospital, and colleagues reported.
“Additional biomarkers are necessary as blood eosinophils alone seem insufficient for risk stratification in COPD,” the investigators wrote in Annals of Allergy, Asthma & Immunology. “Since asthma and COPD share some pathophysiological mechanisms, a logical approach would be to investigate well-known biomarkers for asthma in COPD and vice versa.”
Dr. Çolak and colleagues cited previous research supporting this perspective. Specifically, IgE-targeting monoclonal antibodies have shown promise in patients with severe asthma and asthma-COPD overlap, whereas COPD with high IgE has been associated with a history of lung function decline and previous exacerbations.
The present study drew from a database of 46,598 adults enrolled in the Copenhagen General Population study. All participants underwent physical examination, completed a questionnaire, and provided blood for analysis. From this population, 1,559 individuals had COPD, among whom 446 had high plasma IgE (at least 76 IU/mL).
Over a median follow-up of 6.9 years in the COPD group, 224 severe exacerbations and 434 deaths of any cause occurred. Compared with COPD patients who had normal plasma IgE, those with high IgE were 43% more likely to have severe exacerbation (hazard ratio, 1.43; 95% confidence interval, 1.07-1.89) and 30% more likely to die of any cause (HR, 1.30; 95% CI, 1.06-1.62). These risks were similar when excluding patients with IgE of 700 IU/mL or higher.
“These findings suggest that plasma IgE concentration may be a potential prognostic biomarker and treatment target for a subset of COPD patient,” wrote Dr. Çolak and colleagues.
The above risks increased moderately when the high IgE group was trimmed to include only those with low eosinophils (less than 300 cells/mcL); in this subgroup, risk of exacerbation was increased 62% (HR, 1.62; 95% CI, 1.17-2.24), while risk of all-cause mortality was increased 47% (HR, 1.47; 95% CI, 1.14-1.88).
“We were not able to show that individuals with higher blood eosinophils further stratified by IgE had higher risk of severe exacerbation or all-cause mortality,” the investigators wrote, although they noted “the relatively low statistical power in stratified analysis,” considering the wide confidence intervals observed.
“Thus, we should be careful with interpreting the results in relation to blood eosinophils and IgE combined,” they suggested. “However, we believe that the mechanisms driving exacerbations through plasma IgE are different from those driving blood eosinophils, and we believe that plasma IgE may be a marker for a subset of COPD patients similar to blood eosinophils, which is compatible with the heterogeneity of patients with COPD.”
According to principal author Shoaib Afzal, MD, PhD, of Copenhagen University Hospital, the findings are “probably no surprise for practitioners that often observe overlap between asthma and COPD pathology.”
As smoking prevalence goes down in many countries, relatively more never-smokers are being diagnosed with COPD, Dr. Afzal said in a written comment, “which means that asthma as a risk factor for COPD is gaining importance.”
While patients with asthma can be treated with IgE-targeting omalizumab, a trial evaluating the same biologic for COPD patients with high IgE was withdrawn because of a lack of recruitment; however, Dr. Afzal suggested that this should not be the end of the story, since these new data imply that more patients could benefit than previously recognized.
“Our observational study has generated a hypothesis that needs to be tested by pulmonologists in randomized interventions trials designed with updated inclusion criteria,” he said.
Such trials are needed, Dr. Afzal went on, because they could help unlock the “huge” potential benefit that may come from characterizing COPD patients beyond “exposures, symptoms, and spirometry.”
“Sadly, the progress in establishing biomarkers in COPD for improving risk stratification and treatment allocation have been rather disappointing in the last decades, with the exception of small successes with eosinophils and perhaps FeNO,” Dr. Afzal said.
Nathaniel Marchetti, DO, professor of thoracic medicine and surgery at Temple University and medical director of the respiratory ICU at Temple University Hospital, both in Philadelphia, said the study by Dr. Afzal and colleagues is noteworthy because “biomarkers for COPD are desperately needed to help risk stratify patients for exacerbation risk and risk of disease progression and even mortality.”
In a written comment, Dr. Marchetti agreed with Dr. Afzal that the findings “open the possibility for interventional trials targeting IgE,” which could one day reshape the way patients with COPD are treated.
“I think that biomarkers will become vital in caring for patients with COPD in the future,” Dr. Marchetti said. “There will be medications that will be used to target different pathways of inflammation that drive disease progression and exacerbations. Biomarkers will be important in driving personalized medicine in COPD. We already know the disease seems to vary greatly from patient to patient.”
The study was supported by The Capital Region of Copenhagen, The Danish Lung Foundation, The Velux Foundation, and others. The investigators disclosed relationships with Boehringer Ingelheim, AstraZeneca, Sanofi Genzyme, and others. Dr. Marchetti disclosed no conflicts of interest.
COPD patients with high plasma immunoglobulin E are more likely to have exacerbations and die from any cause, based on a Danish population-cohort study.
hinting at different subsets of patients with COPD, lead author Yunus Çolak MD, PhD, of Copenhagen University Hospital, and colleagues reported.
“Additional biomarkers are necessary as blood eosinophils alone seem insufficient for risk stratification in COPD,” the investigators wrote in Annals of Allergy, Asthma & Immunology. “Since asthma and COPD share some pathophysiological mechanisms, a logical approach would be to investigate well-known biomarkers for asthma in COPD and vice versa.”
Dr. Çolak and colleagues cited previous research supporting this perspective. Specifically, IgE-targeting monoclonal antibodies have shown promise in patients with severe asthma and asthma-COPD overlap, whereas COPD with high IgE has been associated with a history of lung function decline and previous exacerbations.
The present study drew from a database of 46,598 adults enrolled in the Copenhagen General Population study. All participants underwent physical examination, completed a questionnaire, and provided blood for analysis. From this population, 1,559 individuals had COPD, among whom 446 had high plasma IgE (at least 76 IU/mL).
Over a median follow-up of 6.9 years in the COPD group, 224 severe exacerbations and 434 deaths of any cause occurred. Compared with COPD patients who had normal plasma IgE, those with high IgE were 43% more likely to have severe exacerbation (hazard ratio, 1.43; 95% confidence interval, 1.07-1.89) and 30% more likely to die of any cause (HR, 1.30; 95% CI, 1.06-1.62). These risks were similar when excluding patients with IgE of 700 IU/mL or higher.
“These findings suggest that plasma IgE concentration may be a potential prognostic biomarker and treatment target for a subset of COPD patient,” wrote Dr. Çolak and colleagues.
The above risks increased moderately when the high IgE group was trimmed to include only those with low eosinophils (less than 300 cells/mcL); in this subgroup, risk of exacerbation was increased 62% (HR, 1.62; 95% CI, 1.17-2.24), while risk of all-cause mortality was increased 47% (HR, 1.47; 95% CI, 1.14-1.88).
“We were not able to show that individuals with higher blood eosinophils further stratified by IgE had higher risk of severe exacerbation or all-cause mortality,” the investigators wrote, although they noted “the relatively low statistical power in stratified analysis,” considering the wide confidence intervals observed.
“Thus, we should be careful with interpreting the results in relation to blood eosinophils and IgE combined,” they suggested. “However, we believe that the mechanisms driving exacerbations through plasma IgE are different from those driving blood eosinophils, and we believe that plasma IgE may be a marker for a subset of COPD patients similar to blood eosinophils, which is compatible with the heterogeneity of patients with COPD.”
According to principal author Shoaib Afzal, MD, PhD, of Copenhagen University Hospital, the findings are “probably no surprise for practitioners that often observe overlap between asthma and COPD pathology.”
As smoking prevalence goes down in many countries, relatively more never-smokers are being diagnosed with COPD, Dr. Afzal said in a written comment, “which means that asthma as a risk factor for COPD is gaining importance.”
While patients with asthma can be treated with IgE-targeting omalizumab, a trial evaluating the same biologic for COPD patients with high IgE was withdrawn because of a lack of recruitment; however, Dr. Afzal suggested that this should not be the end of the story, since these new data imply that more patients could benefit than previously recognized.
“Our observational study has generated a hypothesis that needs to be tested by pulmonologists in randomized interventions trials designed with updated inclusion criteria,” he said.
Such trials are needed, Dr. Afzal went on, because they could help unlock the “huge” potential benefit that may come from characterizing COPD patients beyond “exposures, symptoms, and spirometry.”
“Sadly, the progress in establishing biomarkers in COPD for improving risk stratification and treatment allocation have been rather disappointing in the last decades, with the exception of small successes with eosinophils and perhaps FeNO,” Dr. Afzal said.
Nathaniel Marchetti, DO, professor of thoracic medicine and surgery at Temple University and medical director of the respiratory ICU at Temple University Hospital, both in Philadelphia, said the study by Dr. Afzal and colleagues is noteworthy because “biomarkers for COPD are desperately needed to help risk stratify patients for exacerbation risk and risk of disease progression and even mortality.”
In a written comment, Dr. Marchetti agreed with Dr. Afzal that the findings “open the possibility for interventional trials targeting IgE,” which could one day reshape the way patients with COPD are treated.
“I think that biomarkers will become vital in caring for patients with COPD in the future,” Dr. Marchetti said. “There will be medications that will be used to target different pathways of inflammation that drive disease progression and exacerbations. Biomarkers will be important in driving personalized medicine in COPD. We already know the disease seems to vary greatly from patient to patient.”
The study was supported by The Capital Region of Copenhagen, The Danish Lung Foundation, The Velux Foundation, and others. The investigators disclosed relationships with Boehringer Ingelheim, AstraZeneca, Sanofi Genzyme, and others. Dr. Marchetti disclosed no conflicts of interest.
COPD patients with high plasma immunoglobulin E are more likely to have exacerbations and die from any cause, based on a Danish population-cohort study.
hinting at different subsets of patients with COPD, lead author Yunus Çolak MD, PhD, of Copenhagen University Hospital, and colleagues reported.
“Additional biomarkers are necessary as blood eosinophils alone seem insufficient for risk stratification in COPD,” the investigators wrote in Annals of Allergy, Asthma & Immunology. “Since asthma and COPD share some pathophysiological mechanisms, a logical approach would be to investigate well-known biomarkers for asthma in COPD and vice versa.”
Dr. Çolak and colleagues cited previous research supporting this perspective. Specifically, IgE-targeting monoclonal antibodies have shown promise in patients with severe asthma and asthma-COPD overlap, whereas COPD with high IgE has been associated with a history of lung function decline and previous exacerbations.
The present study drew from a database of 46,598 adults enrolled in the Copenhagen General Population study. All participants underwent physical examination, completed a questionnaire, and provided blood for analysis. From this population, 1,559 individuals had COPD, among whom 446 had high plasma IgE (at least 76 IU/mL).
Over a median follow-up of 6.9 years in the COPD group, 224 severe exacerbations and 434 deaths of any cause occurred. Compared with COPD patients who had normal plasma IgE, those with high IgE were 43% more likely to have severe exacerbation (hazard ratio, 1.43; 95% confidence interval, 1.07-1.89) and 30% more likely to die of any cause (HR, 1.30; 95% CI, 1.06-1.62). These risks were similar when excluding patients with IgE of 700 IU/mL or higher.
“These findings suggest that plasma IgE concentration may be a potential prognostic biomarker and treatment target for a subset of COPD patient,” wrote Dr. Çolak and colleagues.
The above risks increased moderately when the high IgE group was trimmed to include only those with low eosinophils (less than 300 cells/mcL); in this subgroup, risk of exacerbation was increased 62% (HR, 1.62; 95% CI, 1.17-2.24), while risk of all-cause mortality was increased 47% (HR, 1.47; 95% CI, 1.14-1.88).
“We were not able to show that individuals with higher blood eosinophils further stratified by IgE had higher risk of severe exacerbation or all-cause mortality,” the investigators wrote, although they noted “the relatively low statistical power in stratified analysis,” considering the wide confidence intervals observed.
“Thus, we should be careful with interpreting the results in relation to blood eosinophils and IgE combined,” they suggested. “However, we believe that the mechanisms driving exacerbations through plasma IgE are different from those driving blood eosinophils, and we believe that plasma IgE may be a marker for a subset of COPD patients similar to blood eosinophils, which is compatible with the heterogeneity of patients with COPD.”
According to principal author Shoaib Afzal, MD, PhD, of Copenhagen University Hospital, the findings are “probably no surprise for practitioners that often observe overlap between asthma and COPD pathology.”
As smoking prevalence goes down in many countries, relatively more never-smokers are being diagnosed with COPD, Dr. Afzal said in a written comment, “which means that asthma as a risk factor for COPD is gaining importance.”
While patients with asthma can be treated with IgE-targeting omalizumab, a trial evaluating the same biologic for COPD patients with high IgE was withdrawn because of a lack of recruitment; however, Dr. Afzal suggested that this should not be the end of the story, since these new data imply that more patients could benefit than previously recognized.
“Our observational study has generated a hypothesis that needs to be tested by pulmonologists in randomized interventions trials designed with updated inclusion criteria,” he said.
Such trials are needed, Dr. Afzal went on, because they could help unlock the “huge” potential benefit that may come from characterizing COPD patients beyond “exposures, symptoms, and spirometry.”
“Sadly, the progress in establishing biomarkers in COPD for improving risk stratification and treatment allocation have been rather disappointing in the last decades, with the exception of small successes with eosinophils and perhaps FeNO,” Dr. Afzal said.
Nathaniel Marchetti, DO, professor of thoracic medicine and surgery at Temple University and medical director of the respiratory ICU at Temple University Hospital, both in Philadelphia, said the study by Dr. Afzal and colleagues is noteworthy because “biomarkers for COPD are desperately needed to help risk stratify patients for exacerbation risk and risk of disease progression and even mortality.”
In a written comment, Dr. Marchetti agreed with Dr. Afzal that the findings “open the possibility for interventional trials targeting IgE,” which could one day reshape the way patients with COPD are treated.
“I think that biomarkers will become vital in caring for patients with COPD in the future,” Dr. Marchetti said. “There will be medications that will be used to target different pathways of inflammation that drive disease progression and exacerbations. Biomarkers will be important in driving personalized medicine in COPD. We already know the disease seems to vary greatly from patient to patient.”
The study was supported by The Capital Region of Copenhagen, The Danish Lung Foundation, The Velux Foundation, and others. The investigators disclosed relationships with Boehringer Ingelheim, AstraZeneca, Sanofi Genzyme, and others. Dr. Marchetti disclosed no conflicts of interest.
FROM ANNALS OF ALLERGY, ASTHMA & IMMUNOLOGY
Parkinson’s disease: Is copper culpable?
, according to investigators. The techniques used in this research also may enable rapid identification of blood-borne cofactors driving abnormal protein development in a range of other neurodegenerative diseases, reported lead author Olena Synhaivska, MSc, of the Swiss Federal Laboratories for Materials Science and Technology, Dübendorf, Switzerland.

“While alpha‑synuclein oligomers are the known neurotoxic species in Parkinson’s disease, the development of effective anti–Parkinson’s disease drugs requires targeting of specific structures arising in the early stages of alpha‑synuclein phase transitions or the nucleation-dependent elongation of oligomers into protofibrils,” the investigators wrote in ACS Chemical Neuroscience. “In parallel, advanced methods are required to routinely characterize the size and morphology of intermediary nano- and microstructures formed during self-assembly and aggregation in the presence of aqueous metal ions to track disease progression in, for example, a blood test, to provide effective personalized patient care.”
Pathologic aggregation of alpha‑synuclein
To better understand the relationship between copper and alpha‑synuclein, the investigators used liquid-based atomic force microscopy to observe the protein in solution over 10 days as it transitioned from a simple monomer to a complex, three-dimensional aggregate. Protein aggregation occurred in the absence or presence of copper; however, when incubated in solution with Cu2+ ions, alpha‑synuclein aggregated faster, predominantly forming annular (ring-shaped) structures that were not observed in the absence of copper.

These annular oligomers are noteworthy because they are cytotoxic, and they nucleate formation of alpha‑synuclein filaments, meaning they could serve as early therapeutic targets, according to the investigators.
The above experiments were supported by Raman spectroscopy, which confirmed the various superstructures of alpha‑synuclein formed with or without copper. In addition, the investigators used molecular dynamics computer simulations to map “the dimensions, supramolecular packing interactions, and thermodynamic stabilities” involved in aggregation.
These findings “could potentially serve as guidelines for better understanding protein aggregated states in body fluids from individuals who have been exposed to environmental metals over their lifetime,” the investigators wrote. “The nanoscale imaging, chemical spectroscopy, and integrated modeling-measurement methodologies presented here may inform rapid screening of other potential blood-borne cofactors, for example, other biometals, heavy metals, physiological amino acids, and metabolites, in directing and potentially rerouting intrinsically disordered protein aggregation in the initiation and pathology of neurodegenerative diseases.”
What is copper’s role in Parkinson’s disease pathogenesis?
In a joint written comment, Vikram Khurana MD, PhD, and Richard Krolewski MD, PhD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, said, “This study is important in that it demonstrates that the presence of copper can accelerate and alter the aggregation of wild type alpha‑synuclein. We know that pathologic aggregation of alpha‑synuclein is critical for diseases like Parkinson’s disease known as synucleinopathies – so any insight into how this is happening at the biophysical level has potential implications for altering that process.”
While Dr. Khurana and Dr. Krolewski praised the elegance of the study, including the techniques used to observe alpha‑synuclein aggregation in near real-time, they suggested that more work is needed to determine relevance for patients with Parkinson’s disease.
“It is not clear whether this process is happening in cells, how alpha‑synuclein fibrils might be directly exposed to copper intracellularly (with most of the copper being bound to proteins), and the relevance of the copper concentrations used here are in question,” they said. “Substantially more cell biology and in vivo modeling would be needed to further evaluate the connection of copper specifically to synucleinopathy. All this notwithstanding, the findings are exciting and intriguing and definitely warrant follow-up.”
In the meantime, an increasing number of studies, including a recent preprint by Dr. Khurana and Dr. Krolewski, are strengthening the case for a link between copper exposure and Parkinson’s disease pathogenesis. This body of evidence, they noted, “now spans epidemiology, cell biology, and biophysics.”
Their study, which tested 53 pesticides associated with Parkinson’s disease in patient-derived pluripotent stem cells, found that 2 out of 10 pesticides causing cell death were copper compounds.
“Ongoing work will explore the mechanism of this cell death and investigate ways to mitigate it,” said Dr. Khurana and Dr. Krolewski. “Our hope is that this line of research will raise public awareness about these and other pesticides to reduce potential harm from their use and highlight protective approaches. The study by Dr. Synhaivska and colleagues now raises the possibility of new mechanisms.”
The study by Dr. Synhaivska and colleagues was supported by grants from the Swiss National Science Foundation and the Science Foundation Ireland. The investigators disclosed no conflicts of interest. Dr. Krolewski has been retained as an expert consultant for plaintiffs in a lawsuit on the role of pesticides in Parkinson’s disease causation.
, according to investigators. The techniques used in this research also may enable rapid identification of blood-borne cofactors driving abnormal protein development in a range of other neurodegenerative diseases, reported lead author Olena Synhaivska, MSc, of the Swiss Federal Laboratories for Materials Science and Technology, Dübendorf, Switzerland.

“While alpha‑synuclein oligomers are the known neurotoxic species in Parkinson’s disease, the development of effective anti–Parkinson’s disease drugs requires targeting of specific structures arising in the early stages of alpha‑synuclein phase transitions or the nucleation-dependent elongation of oligomers into protofibrils,” the investigators wrote in ACS Chemical Neuroscience. “In parallel, advanced methods are required to routinely characterize the size and morphology of intermediary nano- and microstructures formed during self-assembly and aggregation in the presence of aqueous metal ions to track disease progression in, for example, a blood test, to provide effective personalized patient care.”
Pathologic aggregation of alpha‑synuclein
To better understand the relationship between copper and alpha‑synuclein, the investigators used liquid-based atomic force microscopy to observe the protein in solution over 10 days as it transitioned from a simple monomer to a complex, three-dimensional aggregate. Protein aggregation occurred in the absence or presence of copper; however, when incubated in solution with Cu2+ ions, alpha‑synuclein aggregated faster, predominantly forming annular (ring-shaped) structures that were not observed in the absence of copper.

These annular oligomers are noteworthy because they are cytotoxic, and they nucleate formation of alpha‑synuclein filaments, meaning they could serve as early therapeutic targets, according to the investigators.
The above experiments were supported by Raman spectroscopy, which confirmed the various superstructures of alpha‑synuclein formed with or without copper. In addition, the investigators used molecular dynamics computer simulations to map “the dimensions, supramolecular packing interactions, and thermodynamic stabilities” involved in aggregation.
These findings “could potentially serve as guidelines for better understanding protein aggregated states in body fluids from individuals who have been exposed to environmental metals over their lifetime,” the investigators wrote. “The nanoscale imaging, chemical spectroscopy, and integrated modeling-measurement methodologies presented here may inform rapid screening of other potential blood-borne cofactors, for example, other biometals, heavy metals, physiological amino acids, and metabolites, in directing and potentially rerouting intrinsically disordered protein aggregation in the initiation and pathology of neurodegenerative diseases.”
What is copper’s role in Parkinson’s disease pathogenesis?
In a joint written comment, Vikram Khurana MD, PhD, and Richard Krolewski MD, PhD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, said, “This study is important in that it demonstrates that the presence of copper can accelerate and alter the aggregation of wild type alpha‑synuclein. We know that pathologic aggregation of alpha‑synuclein is critical for diseases like Parkinson’s disease known as synucleinopathies – so any insight into how this is happening at the biophysical level has potential implications for altering that process.”
While Dr. Khurana and Dr. Krolewski praised the elegance of the study, including the techniques used to observe alpha‑synuclein aggregation in near real-time, they suggested that more work is needed to determine relevance for patients with Parkinson’s disease.
“It is not clear whether this process is happening in cells, how alpha‑synuclein fibrils might be directly exposed to copper intracellularly (with most of the copper being bound to proteins), and the relevance of the copper concentrations used here are in question,” they said. “Substantially more cell biology and in vivo modeling would be needed to further evaluate the connection of copper specifically to synucleinopathy. All this notwithstanding, the findings are exciting and intriguing and definitely warrant follow-up.”
In the meantime, an increasing number of studies, including a recent preprint by Dr. Khurana and Dr. Krolewski, are strengthening the case for a link between copper exposure and Parkinson’s disease pathogenesis. This body of evidence, they noted, “now spans epidemiology, cell biology, and biophysics.”
Their study, which tested 53 pesticides associated with Parkinson’s disease in patient-derived pluripotent stem cells, found that 2 out of 10 pesticides causing cell death were copper compounds.
“Ongoing work will explore the mechanism of this cell death and investigate ways to mitigate it,” said Dr. Khurana and Dr. Krolewski. “Our hope is that this line of research will raise public awareness about these and other pesticides to reduce potential harm from their use and highlight protective approaches. The study by Dr. Synhaivska and colleagues now raises the possibility of new mechanisms.”
The study by Dr. Synhaivska and colleagues was supported by grants from the Swiss National Science Foundation and the Science Foundation Ireland. The investigators disclosed no conflicts of interest. Dr. Krolewski has been retained as an expert consultant for plaintiffs in a lawsuit on the role of pesticides in Parkinson’s disease causation.
, according to investigators. The techniques used in this research also may enable rapid identification of blood-borne cofactors driving abnormal protein development in a range of other neurodegenerative diseases, reported lead author Olena Synhaivska, MSc, of the Swiss Federal Laboratories for Materials Science and Technology, Dübendorf, Switzerland.

“While alpha‑synuclein oligomers are the known neurotoxic species in Parkinson’s disease, the development of effective anti–Parkinson’s disease drugs requires targeting of specific structures arising in the early stages of alpha‑synuclein phase transitions or the nucleation-dependent elongation of oligomers into protofibrils,” the investigators wrote in ACS Chemical Neuroscience. “In parallel, advanced methods are required to routinely characterize the size and morphology of intermediary nano- and microstructures formed during self-assembly and aggregation in the presence of aqueous metal ions to track disease progression in, for example, a blood test, to provide effective personalized patient care.”
Pathologic aggregation of alpha‑synuclein
To better understand the relationship between copper and alpha‑synuclein, the investigators used liquid-based atomic force microscopy to observe the protein in solution over 10 days as it transitioned from a simple monomer to a complex, three-dimensional aggregate. Protein aggregation occurred in the absence or presence of copper; however, when incubated in solution with Cu2+ ions, alpha‑synuclein aggregated faster, predominantly forming annular (ring-shaped) structures that were not observed in the absence of copper.

These annular oligomers are noteworthy because they are cytotoxic, and they nucleate formation of alpha‑synuclein filaments, meaning they could serve as early therapeutic targets, according to the investigators.
The above experiments were supported by Raman spectroscopy, which confirmed the various superstructures of alpha‑synuclein formed with or without copper. In addition, the investigators used molecular dynamics computer simulations to map “the dimensions, supramolecular packing interactions, and thermodynamic stabilities” involved in aggregation.
These findings “could potentially serve as guidelines for better understanding protein aggregated states in body fluids from individuals who have been exposed to environmental metals over their lifetime,” the investigators wrote. “The nanoscale imaging, chemical spectroscopy, and integrated modeling-measurement methodologies presented here may inform rapid screening of other potential blood-borne cofactors, for example, other biometals, heavy metals, physiological amino acids, and metabolites, in directing and potentially rerouting intrinsically disordered protein aggregation in the initiation and pathology of neurodegenerative diseases.”
What is copper’s role in Parkinson’s disease pathogenesis?
In a joint written comment, Vikram Khurana MD, PhD, and Richard Krolewski MD, PhD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, said, “This study is important in that it demonstrates that the presence of copper can accelerate and alter the aggregation of wild type alpha‑synuclein. We know that pathologic aggregation of alpha‑synuclein is critical for diseases like Parkinson’s disease known as synucleinopathies – so any insight into how this is happening at the biophysical level has potential implications for altering that process.”
While Dr. Khurana and Dr. Krolewski praised the elegance of the study, including the techniques used to observe alpha‑synuclein aggregation in near real-time, they suggested that more work is needed to determine relevance for patients with Parkinson’s disease.
“It is not clear whether this process is happening in cells, how alpha‑synuclein fibrils might be directly exposed to copper intracellularly (with most of the copper being bound to proteins), and the relevance of the copper concentrations used here are in question,” they said. “Substantially more cell biology and in vivo modeling would be needed to further evaluate the connection of copper specifically to synucleinopathy. All this notwithstanding, the findings are exciting and intriguing and definitely warrant follow-up.”
In the meantime, an increasing number of studies, including a recent preprint by Dr. Khurana and Dr. Krolewski, are strengthening the case for a link between copper exposure and Parkinson’s disease pathogenesis. This body of evidence, they noted, “now spans epidemiology, cell biology, and biophysics.”
Their study, which tested 53 pesticides associated with Parkinson’s disease in patient-derived pluripotent stem cells, found that 2 out of 10 pesticides causing cell death were copper compounds.
“Ongoing work will explore the mechanism of this cell death and investigate ways to mitigate it,” said Dr. Khurana and Dr. Krolewski. “Our hope is that this line of research will raise public awareness about these and other pesticides to reduce potential harm from their use and highlight protective approaches. The study by Dr. Synhaivska and colleagues now raises the possibility of new mechanisms.”
The study by Dr. Synhaivska and colleagues was supported by grants from the Swiss National Science Foundation and the Science Foundation Ireland. The investigators disclosed no conflicts of interest. Dr. Krolewski has been retained as an expert consultant for plaintiffs in a lawsuit on the role of pesticides in Parkinson’s disease causation.
FROM ACS CHEMICAL NEUROSCIENCE
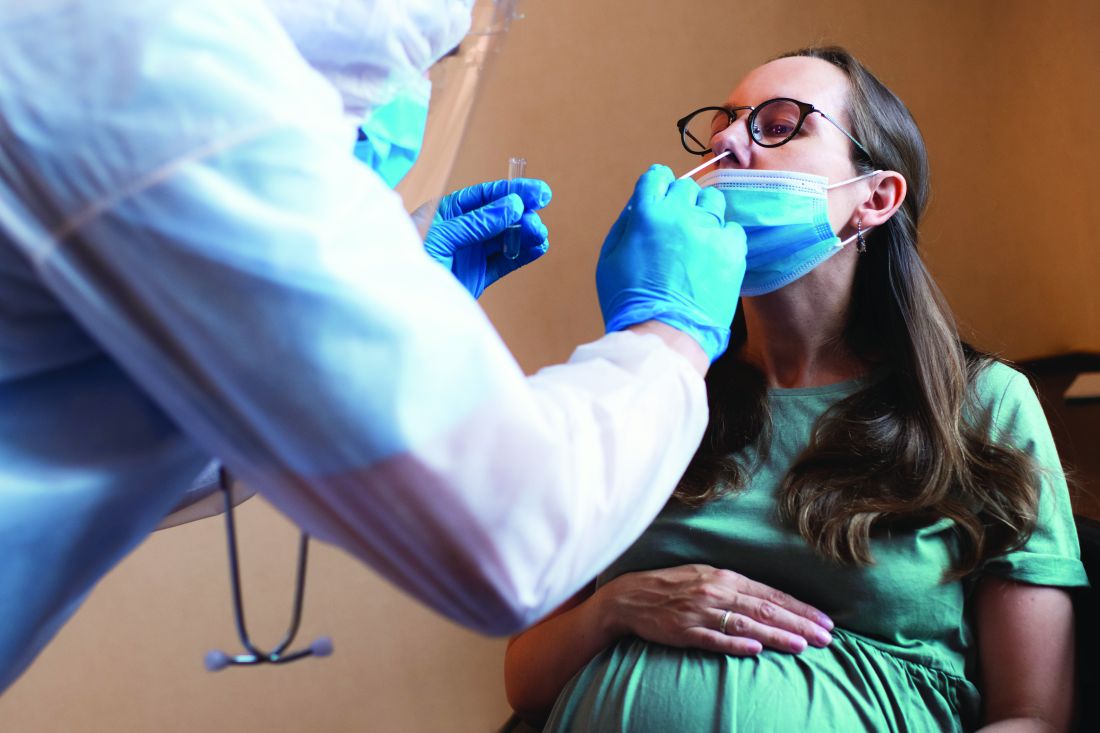
COVID-19 infection late in pregnancy linked to sevenfold risk of preterm birth
Pregnant women who get infected with SARS-CoV-2 in their third trimester are almost three times as likely to have a preterm birth, while infection after 34 weeks’ gestation raises this risk sevenfold, based on the largest matched population-based cohort study published to date.
These findings support previous studies, underscoring the need for pregnant women and their families to take preventive measures against infection, lead author Noga Fallach, MA, of the Kahn-Sagol-Maccabi Research and Innovation Center, Tel Aviv, and colleagues reported.
Past research has suggested that COVID-19 may cause low birth weights and preterm birth in pregnant women, but those studies didn’t report outcomes for each trimester, the investigators wrote in PLoS ONE, noting that “timing of viral infection during fetal development may affect birth and other health outcomes.”
To address this knowledge gap, the investigators looked back at data from 2,703 pregnant women in Israel who tested positive for SARS-CoV-2 from Feb. 21, 2020, to July 2, 2021. Pregnancy outcomes in these women were compared with outcomes in an equal number of uninfected pregnant women. Vaccination status was not reported.
Comparing the two groups showed that catching COVID-19 in the third trimester was linked with nearly triple the risk of preterm birth (odds ratio, 2.76; 95% confidence interval, 1.63-4.67), and more than quadruple the risk if COVID-19 symptoms were present (OR, 4.28; 95% CI, 1.94-9.41). Women who tested positive for SARS-CoV-2 after 34 weeks’ gestation were seven times more likely than uninfected women to deliver early (OR, 7.10; 95% CI, 2.44-20.61).
Pregnant women who caught COVID-19 in the first two trimesters were not significantly more likely to have a preterm birth. Infection was not associated with abnormally low birth rates, or pregnancy loss, in any trimester.
Tal Patalon, MD, coauthor and head of the Kahn-Sagol-Maccabi Research and Innovation Center, focused on these more optimistic findings in an interview.
“The results are encouraging, and reassuring that COVID-19 infection during pregnancy is not associated with any type of pregnancy loss,” Dr. Patalon said.
She also pointed out that the women in the study were infected with SARS-CoV-2 variants that are no longer common.
“It should be remembered that the research group tested the COVID-19 pre-Delta variants, and does not refer to the dominant variant today, which is Omicron,” Dr. Patalon said.
Still, the investigators concluded that the “results underline the importance of preventive measures taken against SARS-CoV-2 infection among pregnant women and their families.”
Sonja A. Rasmussen, MD, of the University of Florida, Gainesville, said that the issue with out-of-date variants in published research has been one of the “real challenges” in studying the ever-evolving COVID-19 pandemic; however, it’s not a good enough reason to dismiss this study.
“I think at this point, we need to assume that it applies to Omicron too,” Dr. Rasmussen said, noting that other respiratory viruses, like influenza, have also been shown to increase the risk of preterm birth when contracted in late pregnancy.
While the present findings highlight the risk of infection in the third trimester, Dr. Rasmussen advised women in all stages of pregnancy to protect themselves against COVID-19, based on the knowledge that illness in a mother can affect normal growth and development in a fetus, even if it doesn’t lead to preterm birth.
“A mom getting sick during pregnancy is not good for the baby,” Dr. Rasmussen said. “The baby’s really dependent on the mom. So you want that baby to have good nutrition throughout the pregnancy. It’s just as important earlier on as later. And you want that baby to get good oxygenation no matter what time [in the pregnancy]. I know that people want a little bit of a break [from preventive measures]. But I would emphasize that if you’re pregnant, we do all sorts of things during pregnancy to make sure that our babies are safe and healthy, and I would continue that for the whole pregnancy.”
Specifically, Dr. Rasmussen advised social distancing, use of an N95 mask, and vaccination. Getting vaccinated during pregnancy helps newborns fight off infection until 6 months of age, she added, when they become eligible for vaccination themselves. This added benefit was recently reported in a study published in the New England Journal of Medicine , for which Dr. Rasmussen cowrote an editorial .
“Vaccines have been approved for 6 months and older,” Dr. Rasmussen said. “But what do you do in those first 6 months of life? That’s a high-risk time for kids.”
Despite these risks, convincing pregnant women to get vaccinated remains a key challenge for health care providers, according to Dr. Rasmussen, even with an abundance of safety data. “Early on [in the pandemic], we said we didn’t know a lot about risks. We knew that other vaccines were safe during pregnancy, but we didn’t have a lot of information about a COVID-19 vaccine. But now we have a lot of data on safety during pregnancy, and these vaccines appear to be completely safe, based on the information we have. There have been many, many pregnant women vaccinated in the United States and in other countries.”
For reluctant expecting mothers, Dr. Rasmussen offered some words of advice: “I know that you worry about anything you do when you’re pregnant. But this is something that you can do to help your baby – now, to make a preterm birth less likely, and later, after the baby is born.
“The most important thing is for the pregnant person to hear this [vaccine recommendation] from their doctor,” she added. “If they’re going to listen to anybody, they’re going to listen to their physician. That’s what the data have shown for a long time.”
The investigators and Dr. Rasmussen disclosed no conflicts of interest.
Pregnant women who get infected with SARS-CoV-2 in their third trimester are almost three times as likely to have a preterm birth, while infection after 34 weeks’ gestation raises this risk sevenfold, based on the largest matched population-based cohort study published to date.
These findings support previous studies, underscoring the need for pregnant women and their families to take preventive measures against infection, lead author Noga Fallach, MA, of the Kahn-Sagol-Maccabi Research and Innovation Center, Tel Aviv, and colleagues reported.
Past research has suggested that COVID-19 may cause low birth weights and preterm birth in pregnant women, but those studies didn’t report outcomes for each trimester, the investigators wrote in PLoS ONE, noting that “timing of viral infection during fetal development may affect birth and other health outcomes.”
To address this knowledge gap, the investigators looked back at data from 2,703 pregnant women in Israel who tested positive for SARS-CoV-2 from Feb. 21, 2020, to July 2, 2021. Pregnancy outcomes in these women were compared with outcomes in an equal number of uninfected pregnant women. Vaccination status was not reported.
Comparing the two groups showed that catching COVID-19 in the third trimester was linked with nearly triple the risk of preterm birth (odds ratio, 2.76; 95% confidence interval, 1.63-4.67), and more than quadruple the risk if COVID-19 symptoms were present (OR, 4.28; 95% CI, 1.94-9.41). Women who tested positive for SARS-CoV-2 after 34 weeks’ gestation were seven times more likely than uninfected women to deliver early (OR, 7.10; 95% CI, 2.44-20.61).
Pregnant women who caught COVID-19 in the first two trimesters were not significantly more likely to have a preterm birth. Infection was not associated with abnormally low birth rates, or pregnancy loss, in any trimester.
Tal Patalon, MD, coauthor and head of the Kahn-Sagol-Maccabi Research and Innovation Center, focused on these more optimistic findings in an interview.
“The results are encouraging, and reassuring that COVID-19 infection during pregnancy is not associated with any type of pregnancy loss,” Dr. Patalon said.
She also pointed out that the women in the study were infected with SARS-CoV-2 variants that are no longer common.
“It should be remembered that the research group tested the COVID-19 pre-Delta variants, and does not refer to the dominant variant today, which is Omicron,” Dr. Patalon said.
Still, the investigators concluded that the “results underline the importance of preventive measures taken against SARS-CoV-2 infection among pregnant women and their families.”
Sonja A. Rasmussen, MD, of the University of Florida, Gainesville, said that the issue with out-of-date variants in published research has been one of the “real challenges” in studying the ever-evolving COVID-19 pandemic; however, it’s not a good enough reason to dismiss this study.
“I think at this point, we need to assume that it applies to Omicron too,” Dr. Rasmussen said, noting that other respiratory viruses, like influenza, have also been shown to increase the risk of preterm birth when contracted in late pregnancy.
While the present findings highlight the risk of infection in the third trimester, Dr. Rasmussen advised women in all stages of pregnancy to protect themselves against COVID-19, based on the knowledge that illness in a mother can affect normal growth and development in a fetus, even if it doesn’t lead to preterm birth.
“A mom getting sick during pregnancy is not good for the baby,” Dr. Rasmussen said. “The baby’s really dependent on the mom. So you want that baby to have good nutrition throughout the pregnancy. It’s just as important earlier on as later. And you want that baby to get good oxygenation no matter what time [in the pregnancy]. I know that people want a little bit of a break [from preventive measures]. But I would emphasize that if you’re pregnant, we do all sorts of things during pregnancy to make sure that our babies are safe and healthy, and I would continue that for the whole pregnancy.”
Specifically, Dr. Rasmussen advised social distancing, use of an N95 mask, and vaccination. Getting vaccinated during pregnancy helps newborns fight off infection until 6 months of age, she added, when they become eligible for vaccination themselves. This added benefit was recently reported in a study published in the New England Journal of Medicine , for which Dr. Rasmussen cowrote an editorial .
“Vaccines have been approved for 6 months and older,” Dr. Rasmussen said. “But what do you do in those first 6 months of life? That’s a high-risk time for kids.”
Despite these risks, convincing pregnant women to get vaccinated remains a key challenge for health care providers, according to Dr. Rasmussen, even with an abundance of safety data. “Early on [in the pandemic], we said we didn’t know a lot about risks. We knew that other vaccines were safe during pregnancy, but we didn’t have a lot of information about a COVID-19 vaccine. But now we have a lot of data on safety during pregnancy, and these vaccines appear to be completely safe, based on the information we have. There have been many, many pregnant women vaccinated in the United States and in other countries.”
For reluctant expecting mothers, Dr. Rasmussen offered some words of advice: “I know that you worry about anything you do when you’re pregnant. But this is something that you can do to help your baby – now, to make a preterm birth less likely, and later, after the baby is born.
“The most important thing is for the pregnant person to hear this [vaccine recommendation] from their doctor,” she added. “If they’re going to listen to anybody, they’re going to listen to their physician. That’s what the data have shown for a long time.”
The investigators and Dr. Rasmussen disclosed no conflicts of interest.
Pregnant women who get infected with SARS-CoV-2 in their third trimester are almost three times as likely to have a preterm birth, while infection after 34 weeks’ gestation raises this risk sevenfold, based on the largest matched population-based cohort study published to date.
These findings support previous studies, underscoring the need for pregnant women and their families to take preventive measures against infection, lead author Noga Fallach, MA, of the Kahn-Sagol-Maccabi Research and Innovation Center, Tel Aviv, and colleagues reported.
Past research has suggested that COVID-19 may cause low birth weights and preterm birth in pregnant women, but those studies didn’t report outcomes for each trimester, the investigators wrote in PLoS ONE, noting that “timing of viral infection during fetal development may affect birth and other health outcomes.”
To address this knowledge gap, the investigators looked back at data from 2,703 pregnant women in Israel who tested positive for SARS-CoV-2 from Feb. 21, 2020, to July 2, 2021. Pregnancy outcomes in these women were compared with outcomes in an equal number of uninfected pregnant women. Vaccination status was not reported.
Comparing the two groups showed that catching COVID-19 in the third trimester was linked with nearly triple the risk of preterm birth (odds ratio, 2.76; 95% confidence interval, 1.63-4.67), and more than quadruple the risk if COVID-19 symptoms were present (OR, 4.28; 95% CI, 1.94-9.41). Women who tested positive for SARS-CoV-2 after 34 weeks’ gestation were seven times more likely than uninfected women to deliver early (OR, 7.10; 95% CI, 2.44-20.61).
Pregnant women who caught COVID-19 in the first two trimesters were not significantly more likely to have a preterm birth. Infection was not associated with abnormally low birth rates, or pregnancy loss, in any trimester.
Tal Patalon, MD, coauthor and head of the Kahn-Sagol-Maccabi Research and Innovation Center, focused on these more optimistic findings in an interview.
“The results are encouraging, and reassuring that COVID-19 infection during pregnancy is not associated with any type of pregnancy loss,” Dr. Patalon said.
She also pointed out that the women in the study were infected with SARS-CoV-2 variants that are no longer common.
“It should be remembered that the research group tested the COVID-19 pre-Delta variants, and does not refer to the dominant variant today, which is Omicron,” Dr. Patalon said.
Still, the investigators concluded that the “results underline the importance of preventive measures taken against SARS-CoV-2 infection among pregnant women and their families.”
Sonja A. Rasmussen, MD, of the University of Florida, Gainesville, said that the issue with out-of-date variants in published research has been one of the “real challenges” in studying the ever-evolving COVID-19 pandemic; however, it’s not a good enough reason to dismiss this study.
“I think at this point, we need to assume that it applies to Omicron too,” Dr. Rasmussen said, noting that other respiratory viruses, like influenza, have also been shown to increase the risk of preterm birth when contracted in late pregnancy.
While the present findings highlight the risk of infection in the third trimester, Dr. Rasmussen advised women in all stages of pregnancy to protect themselves against COVID-19, based on the knowledge that illness in a mother can affect normal growth and development in a fetus, even if it doesn’t lead to preterm birth.
“A mom getting sick during pregnancy is not good for the baby,” Dr. Rasmussen said. “The baby’s really dependent on the mom. So you want that baby to have good nutrition throughout the pregnancy. It’s just as important earlier on as later. And you want that baby to get good oxygenation no matter what time [in the pregnancy]. I know that people want a little bit of a break [from preventive measures]. But I would emphasize that if you’re pregnant, we do all sorts of things during pregnancy to make sure that our babies are safe and healthy, and I would continue that for the whole pregnancy.”
Specifically, Dr. Rasmussen advised social distancing, use of an N95 mask, and vaccination. Getting vaccinated during pregnancy helps newborns fight off infection until 6 months of age, she added, when they become eligible for vaccination themselves. This added benefit was recently reported in a study published in the New England Journal of Medicine , for which Dr. Rasmussen cowrote an editorial .
“Vaccines have been approved for 6 months and older,” Dr. Rasmussen said. “But what do you do in those first 6 months of life? That’s a high-risk time for kids.”
Despite these risks, convincing pregnant women to get vaccinated remains a key challenge for health care providers, according to Dr. Rasmussen, even with an abundance of safety data. “Early on [in the pandemic], we said we didn’t know a lot about risks. We knew that other vaccines were safe during pregnancy, but we didn’t have a lot of information about a COVID-19 vaccine. But now we have a lot of data on safety during pregnancy, and these vaccines appear to be completely safe, based on the information we have. There have been many, many pregnant women vaccinated in the United States and in other countries.”
For reluctant expecting mothers, Dr. Rasmussen offered some words of advice: “I know that you worry about anything you do when you’re pregnant. But this is something that you can do to help your baby – now, to make a preterm birth less likely, and later, after the baby is born.
“The most important thing is for the pregnant person to hear this [vaccine recommendation] from their doctor,” she added. “If they’re going to listen to anybody, they’re going to listen to their physician. That’s what the data have shown for a long time.”
The investigators and Dr. Rasmussen disclosed no conflicts of interest.
FROM PLOS ONE
Strictures in Crohn’s: Balloon dilation avoids later surgery
Endoscopic balloon dilation (EBD) is an effective treatment option for strictures of the small bowel in patients with Crohn’s disease, based on a nationwide Danish cohort study.
Approximately three out of four patients who underwent an EBD were spared subsequent small bowel surgery. Similar outcomes were seen across primary and postsurgical strictures, reported lead author Mads Damsgaard Wewer, BSc, of the University of Copenhagen, and colleagues.
“Retrospective studies investigating EBD are available with variable follow-up periods; however, a nationwide study to demonstrate more precise durability of EBD in unselected patients is lacking,” the investigators wrote in European Journal of Gastroenterology & Hepatology. Their aim was to understand the use of EBD and the need for redilation and surgery. This retrospective study used a cohort of adult patients with Crohn’s disease who had strictures of the small bowel during a 19-year period.
The population comprised 9,737 patients with incident Crohn’s disease, among whom 90 (1%) underwent EBD during a median 8.2-year follow-up period. Of these 90 patients, 49 had primary strictures, while the remaining 41 had postsurgical strictures.
In the primary stricture group, 59% of patients had one EBD procedure and did not require subsequent small bowel surgery, 14% of patients required redilation but no further surgery, and 27% of patients required small bowel surgery after dilation. In this same group, the 1-, 3-, and 5-year cumulative incidence rates of EBD failure were 19%, 21%, and 25%, respectively. Of note, just 8% of patients with primary stricture who were treated with EBD ultimately required enterotomy, compared with 16% of patients with primary stricture who underwent small bowel resection without first attempting EBD.
In the postsurgical stricture group, 49% of patients underwent one EBD procedure without need for another small bowel surgery, 27% needed redilation but avoided surgery, and 24% required surgery after dilation. One-, three-, and five-year cumulative incidence rates of EBD failure in this group trended slightly higher than the primary stricture group over time, at 19%, 25%, and 29%, respectively.
The researchers noted that 25% of patients required small bowel surgery after EBD, which falls below rates of 29% to 33% reported by recent studies. They explained that this edge may be “partly explained by the careful selection of patients (with few and short strictures) receiving EBD,” as well as exclusion of patients with strictures outside the small intestine. They concluded that, “... small bowel-related EBD is an effective treatment option, and one that could be offered to more patients with Crohn’s disease in the future.”
‘Reassuring study’
David H. Bruining, MD, associate professor of medicine and section head of the inflammatory bowel disease interest group at Mayo Clinic, Rochester, Minn., called it a “reassuring study that confirms previous data regarding the efficacy of endoscopic balloon dilation of Crohn’s disease strictures.”
Dr. Bruining suggested in an interview that the findings, while drawn from Denmark, can be applied to a U.S. population; he also noted the “impressive” size of the study, as well the duration of follow-up, which extended up to 19 years.
EBD is “gaining more traction,” Dr. Bruining said, “as far as the belief among both referring physicians, and gastroenterologists, that it is effective, and it is safe. I think that body of literature is growing, and it’s more widely established at this point.”
Dr. Bruining noted that EBD should be reserved for patients who have short strictures no longer than 4-5 mm “without associated internal penetrating disease.”
In the future, such patients may have even more treatment options, Dr. Bruining predicted. New antifibrotic medications are “on the horizon,” which could one day be used with or without EBD to address fibrotic strictures in Crohn’s disease. Dr. Bruining is a part of the Stenosis Therapy and Anti-Fibrotic Research (STAR) Consortium, a group that aims to develop this emerging approach. He and his colleagues recently published a review of research into antifibrotic therapy to date.
Limiting factors
“This is an important study that really adds something to the literature,” noted Joseph Carmichael, MD, chief medical officer and chief of colon and rectal surgery at the University of California, Irvine. “The cohort is a little unusual in this area in that it encompasses a whole country. Yet this is exactly what makes the data stand apart from previous studies, since the patient population was unselected. That’s how you get a true incidence of the intervention.”
Beyond the generally favorable outcomes associated with EBD, Dr. Carmichael highlighted similar rates of success across both primary and postsurgical strictures. “Some of the previous data suggest postsurgical strictures don’t do as well with endoscopic dilation, and this [study] seems to go against that,” he said. “Which really deserves a closer look.”
Reflecting on the researchers’ call for more frequent use of EBD in patients with Crohn’s disease, Dr. Carmichael speculated that several factors may be limiting current utilization in Europe and the U.S. For one, there may not be enough interventional gastroenterologists. Also, the procedure “generally requires a high-volume provider who’s got surgical backup because these [procedures] have a 2% to 4% incidence of technical failure – including perforation or bleeding. These risks may deter patients from undergoing the procedure.
“Patients with Crohn’s disease can be pretty remarkable in their ability to endure obstruction,” he added. “If someone’s feeling their symptoms aren’t altering their quality of life, they may choose not to proceed with it.”
With all candidate patients, Dr. Carmichael recommended discussing the risks of EBD compared with the risks of declining the intervention, such as complete bowel obstruction, bowel perforation, or fistula. “That’s a question that needs to be included with every conversation when we discuss procedures,” he explained.
The researchers report that, among others, they have relationships with Janssen-Cilag, AbbVie A/S, and Celgene. Dr. Bruining and Dr. Carmichael reports no relevant conflicts of interest.
This article was updated 7/29/22.
Endoscopic balloon dilation (EBD) is an effective treatment option for strictures of the small bowel in patients with Crohn’s disease, based on a nationwide Danish cohort study.
Approximately three out of four patients who underwent an EBD were spared subsequent small bowel surgery. Similar outcomes were seen across primary and postsurgical strictures, reported lead author Mads Damsgaard Wewer, BSc, of the University of Copenhagen, and colleagues.
“Retrospective studies investigating EBD are available with variable follow-up periods; however, a nationwide study to demonstrate more precise durability of EBD in unselected patients is lacking,” the investigators wrote in European Journal of Gastroenterology & Hepatology. Their aim was to understand the use of EBD and the need for redilation and surgery. This retrospective study used a cohort of adult patients with Crohn’s disease who had strictures of the small bowel during a 19-year period.
The population comprised 9,737 patients with incident Crohn’s disease, among whom 90 (1%) underwent EBD during a median 8.2-year follow-up period. Of these 90 patients, 49 had primary strictures, while the remaining 41 had postsurgical strictures.
In the primary stricture group, 59% of patients had one EBD procedure and did not require subsequent small bowel surgery, 14% of patients required redilation but no further surgery, and 27% of patients required small bowel surgery after dilation. In this same group, the 1-, 3-, and 5-year cumulative incidence rates of EBD failure were 19%, 21%, and 25%, respectively. Of note, just 8% of patients with primary stricture who were treated with EBD ultimately required enterotomy, compared with 16% of patients with primary stricture who underwent small bowel resection without first attempting EBD.
In the postsurgical stricture group, 49% of patients underwent one EBD procedure without need for another small bowel surgery, 27% needed redilation but avoided surgery, and 24% required surgery after dilation. One-, three-, and five-year cumulative incidence rates of EBD failure in this group trended slightly higher than the primary stricture group over time, at 19%, 25%, and 29%, respectively.
The researchers noted that 25% of patients required small bowel surgery after EBD, which falls below rates of 29% to 33% reported by recent studies. They explained that this edge may be “partly explained by the careful selection of patients (with few and short strictures) receiving EBD,” as well as exclusion of patients with strictures outside the small intestine. They concluded that, “... small bowel-related EBD is an effective treatment option, and one that could be offered to more patients with Crohn’s disease in the future.”
‘Reassuring study’
David H. Bruining, MD, associate professor of medicine and section head of the inflammatory bowel disease interest group at Mayo Clinic, Rochester, Minn., called it a “reassuring study that confirms previous data regarding the efficacy of endoscopic balloon dilation of Crohn’s disease strictures.”
Dr. Bruining suggested in an interview that the findings, while drawn from Denmark, can be applied to a U.S. population; he also noted the “impressive” size of the study, as well the duration of follow-up, which extended up to 19 years.
EBD is “gaining more traction,” Dr. Bruining said, “as far as the belief among both referring physicians, and gastroenterologists, that it is effective, and it is safe. I think that body of literature is growing, and it’s more widely established at this point.”
Dr. Bruining noted that EBD should be reserved for patients who have short strictures no longer than 4-5 mm “without associated internal penetrating disease.”
In the future, such patients may have even more treatment options, Dr. Bruining predicted. New antifibrotic medications are “on the horizon,” which could one day be used with or without EBD to address fibrotic strictures in Crohn’s disease. Dr. Bruining is a part of the Stenosis Therapy and Anti-Fibrotic Research (STAR) Consortium, a group that aims to develop this emerging approach. He and his colleagues recently published a review of research into antifibrotic therapy to date.
Limiting factors
“This is an important study that really adds something to the literature,” noted Joseph Carmichael, MD, chief medical officer and chief of colon and rectal surgery at the University of California, Irvine. “The cohort is a little unusual in this area in that it encompasses a whole country. Yet this is exactly what makes the data stand apart from previous studies, since the patient population was unselected. That’s how you get a true incidence of the intervention.”
Beyond the generally favorable outcomes associated with EBD, Dr. Carmichael highlighted similar rates of success across both primary and postsurgical strictures. “Some of the previous data suggest postsurgical strictures don’t do as well with endoscopic dilation, and this [study] seems to go against that,” he said. “Which really deserves a closer look.”
Reflecting on the researchers’ call for more frequent use of EBD in patients with Crohn’s disease, Dr. Carmichael speculated that several factors may be limiting current utilization in Europe and the U.S. For one, there may not be enough interventional gastroenterologists. Also, the procedure “generally requires a high-volume provider who’s got surgical backup because these [procedures] have a 2% to 4% incidence of technical failure – including perforation or bleeding. These risks may deter patients from undergoing the procedure.
“Patients with Crohn’s disease can be pretty remarkable in their ability to endure obstruction,” he added. “If someone’s feeling their symptoms aren’t altering their quality of life, they may choose not to proceed with it.”
With all candidate patients, Dr. Carmichael recommended discussing the risks of EBD compared with the risks of declining the intervention, such as complete bowel obstruction, bowel perforation, or fistula. “That’s a question that needs to be included with every conversation when we discuss procedures,” he explained.
The researchers report that, among others, they have relationships with Janssen-Cilag, AbbVie A/S, and Celgene. Dr. Bruining and Dr. Carmichael reports no relevant conflicts of interest.
This article was updated 7/29/22.
Endoscopic balloon dilation (EBD) is an effective treatment option for strictures of the small bowel in patients with Crohn’s disease, based on a nationwide Danish cohort study.
Approximately three out of four patients who underwent an EBD were spared subsequent small bowel surgery. Similar outcomes were seen across primary and postsurgical strictures, reported lead author Mads Damsgaard Wewer, BSc, of the University of Copenhagen, and colleagues.
“Retrospective studies investigating EBD are available with variable follow-up periods; however, a nationwide study to demonstrate more precise durability of EBD in unselected patients is lacking,” the investigators wrote in European Journal of Gastroenterology & Hepatology. Their aim was to understand the use of EBD and the need for redilation and surgery. This retrospective study used a cohort of adult patients with Crohn’s disease who had strictures of the small bowel during a 19-year period.
The population comprised 9,737 patients with incident Crohn’s disease, among whom 90 (1%) underwent EBD during a median 8.2-year follow-up period. Of these 90 patients, 49 had primary strictures, while the remaining 41 had postsurgical strictures.
In the primary stricture group, 59% of patients had one EBD procedure and did not require subsequent small bowel surgery, 14% of patients required redilation but no further surgery, and 27% of patients required small bowel surgery after dilation. In this same group, the 1-, 3-, and 5-year cumulative incidence rates of EBD failure were 19%, 21%, and 25%, respectively. Of note, just 8% of patients with primary stricture who were treated with EBD ultimately required enterotomy, compared with 16% of patients with primary stricture who underwent small bowel resection without first attempting EBD.
In the postsurgical stricture group, 49% of patients underwent one EBD procedure without need for another small bowel surgery, 27% needed redilation but avoided surgery, and 24% required surgery after dilation. One-, three-, and five-year cumulative incidence rates of EBD failure in this group trended slightly higher than the primary stricture group over time, at 19%, 25%, and 29%, respectively.
The researchers noted that 25% of patients required small bowel surgery after EBD, which falls below rates of 29% to 33% reported by recent studies. They explained that this edge may be “partly explained by the careful selection of patients (with few and short strictures) receiving EBD,” as well as exclusion of patients with strictures outside the small intestine. They concluded that, “... small bowel-related EBD is an effective treatment option, and one that could be offered to more patients with Crohn’s disease in the future.”
‘Reassuring study’
David H. Bruining, MD, associate professor of medicine and section head of the inflammatory bowel disease interest group at Mayo Clinic, Rochester, Minn., called it a “reassuring study that confirms previous data regarding the efficacy of endoscopic balloon dilation of Crohn’s disease strictures.”
Dr. Bruining suggested in an interview that the findings, while drawn from Denmark, can be applied to a U.S. population; he also noted the “impressive” size of the study, as well the duration of follow-up, which extended up to 19 years.
EBD is “gaining more traction,” Dr. Bruining said, “as far as the belief among both referring physicians, and gastroenterologists, that it is effective, and it is safe. I think that body of literature is growing, and it’s more widely established at this point.”
Dr. Bruining noted that EBD should be reserved for patients who have short strictures no longer than 4-5 mm “without associated internal penetrating disease.”
In the future, such patients may have even more treatment options, Dr. Bruining predicted. New antifibrotic medications are “on the horizon,” which could one day be used with or without EBD to address fibrotic strictures in Crohn’s disease. Dr. Bruining is a part of the Stenosis Therapy and Anti-Fibrotic Research (STAR) Consortium, a group that aims to develop this emerging approach. He and his colleagues recently published a review of research into antifibrotic therapy to date.
Limiting factors
“This is an important study that really adds something to the literature,” noted Joseph Carmichael, MD, chief medical officer and chief of colon and rectal surgery at the University of California, Irvine. “The cohort is a little unusual in this area in that it encompasses a whole country. Yet this is exactly what makes the data stand apart from previous studies, since the patient population was unselected. That’s how you get a true incidence of the intervention.”
Beyond the generally favorable outcomes associated with EBD, Dr. Carmichael highlighted similar rates of success across both primary and postsurgical strictures. “Some of the previous data suggest postsurgical strictures don’t do as well with endoscopic dilation, and this [study] seems to go against that,” he said. “Which really deserves a closer look.”
Reflecting on the researchers’ call for more frequent use of EBD in patients with Crohn’s disease, Dr. Carmichael speculated that several factors may be limiting current utilization in Europe and the U.S. For one, there may not be enough interventional gastroenterologists. Also, the procedure “generally requires a high-volume provider who’s got surgical backup because these [procedures] have a 2% to 4% incidence of technical failure – including perforation or bleeding. These risks may deter patients from undergoing the procedure.
“Patients with Crohn’s disease can be pretty remarkable in their ability to endure obstruction,” he added. “If someone’s feeling their symptoms aren’t altering their quality of life, they may choose not to proceed with it.”
With all candidate patients, Dr. Carmichael recommended discussing the risks of EBD compared with the risks of declining the intervention, such as complete bowel obstruction, bowel perforation, or fistula. “That’s a question that needs to be included with every conversation when we discuss procedures,” he explained.
The researchers report that, among others, they have relationships with Janssen-Cilag, AbbVie A/S, and Celgene. Dr. Bruining and Dr. Carmichael reports no relevant conflicts of interest.
This article was updated 7/29/22.
FROM EUROPEAN JOURNAL OF GASTROENTEROLOGY & HEPATOLOGY
Does choice of biologic affect outcomes in perianal Crohn’s disease?
Choice of biologic therapy in the first line and later may impact long-term outcomes in patients with perianal Crohn’s disease (pCD), according to a retrospective study.
John Gubatan, MD, of Stanford (Calif.) University, and colleagues reported that, compared with no biologic therapy, first-line treatment with an anti–tumor necrosis factor (TNF) agent or ustekinumab significantly reduced risk of perianal abscess recurrence at 5 years, whereas vedolizumab offered no such benefit. After failure of the initial anti-TNF, switching to another anti-TNF agent is the most effective option.
“Although pCD is recognized to be an aggressive phenotype, data on whether escalating to a biologic at the time of perianal disease diagnosis may alter the natural history and long-term clinical outcomes of pCD is limited,” the researchers wrote in Journal of Clinical Gastroenterology. “This is the first study to explore how the type of biologic therapy at the time of perianal disease diagnosis and change in biologic therapy after first anti-TNF failure are associated with rates of long-term clinical outcomes.”
The study included 311 patients with pCD treated at Stanford University from 1998 to 2020. At the time of diagnosis, 168 of these patients started a biologic, most often an anti-TNF agent (n = 138), followed distantly by ustekinumab (n = 16) or vedolizumab (n = 14). Efficacy of these first-line biologics was compared with no biologic therapy in terms of five clinical outcomes at 5 years: surgical intervention, colectomy, permanent diversion, fistula closure, and perianal abscess recurrence.
Although both reduced risk of perianal abscess recurrence, it was still higher with anti-TNF therapy (hazard ratio, 0.48; 95% confidence interval, 0.32-0.74) than with ustekinumab (HR, 0.20; 95% CI, 0.07-0.56). Ustekinumab also increased the rate of perianal fistula closure by more than threefold (HR, 3.58; 95% CI, 1.04-12.35).
Vedolizumab, on the other hand, offered no significant benefit across any of the five outcomes.
None of the biologics had an impact on rates of surgical intervention, colectomy, or permanent diversion.
Further analyses explored the long-term effects of second-line biologic choice after initial failure with anti-TNF therapy. Switching to another anti-TNF agent was more effective than switching to ustekinumab at reducing risks of colectomy (HR, 0.20; 95% CI, 0.04-0.90) and permanent diversion (HR, 0.16; 95% CI, 0.03-0.94); switching to ustekinumab was more effective than switching to vedolizumab for perianal fistula closure (HR, 0.22; 95% CI, 0.05-0.96).
Switching to another anti-TNF biologic or ustekinumab may be associated with better 5-year outcomes, compared with switching to vedolizumab in patients with pCD, according to Dr. Gubatan. Other guidelines or data that might steer this sequencing decision are scant. However, the findings should be validated with prospective data, ideally from head-to-head trials.
Jordan E. Axelrad, MD, of NYU Langone Health, New York, said the present study is noteworthy for addressing a “very-difficult-to-treat condition that has limited data as well as very limited long-term outcome data for our currently available interventions.”
Dr. Axelrad appreciated how the study focused on the distribution of clinical manifestations of pCD, including ulcers (10%), fissures (23.2%), abscesses (76.1%), and fistulas (84.2%). According to Dr. Axelrad, the efficacy data provide really important insights for clinicians who choose biologic therapies. He noted that, in the absence of head-to-head clinical trials, “it’s absolutely important that we use these results to help us guide therapy” for patients with pCD.
While the biologics included in the study were efficacious to varying degrees, Dr. Axelrad pointed out that no choice was associated with a reduced risk of surgical intervention. “That really underscored for me how complex this patient population is,” he said. “Despite good medical therapies … we’re still not necessarily making a huge dent in the risk of surgical intervention requirements for this complex patient group.”
Dr. Gubatan disclosed support from a Chan Zuckerberg Biohub Physician Scientist Scholar Award, a National Institutes of Health NIDDK LRP Award, and a Doris Duke Physician Scientist Fellowship Award; his colleagues reported no conflicts of interest. Dr. Axelrad reports relationships with Janssen, AbbVie, Pfizer, and others.
Choice of biologic therapy in the first line and later may impact long-term outcomes in patients with perianal Crohn’s disease (pCD), according to a retrospective study.
John Gubatan, MD, of Stanford (Calif.) University, and colleagues reported that, compared with no biologic therapy, first-line treatment with an anti–tumor necrosis factor (TNF) agent or ustekinumab significantly reduced risk of perianal abscess recurrence at 5 years, whereas vedolizumab offered no such benefit. After failure of the initial anti-TNF, switching to another anti-TNF agent is the most effective option.
“Although pCD is recognized to be an aggressive phenotype, data on whether escalating to a biologic at the time of perianal disease diagnosis may alter the natural history and long-term clinical outcomes of pCD is limited,” the researchers wrote in Journal of Clinical Gastroenterology. “This is the first study to explore how the type of biologic therapy at the time of perianal disease diagnosis and change in biologic therapy after first anti-TNF failure are associated with rates of long-term clinical outcomes.”
The study included 311 patients with pCD treated at Stanford University from 1998 to 2020. At the time of diagnosis, 168 of these patients started a biologic, most often an anti-TNF agent (n = 138), followed distantly by ustekinumab (n = 16) or vedolizumab (n = 14). Efficacy of these first-line biologics was compared with no biologic therapy in terms of five clinical outcomes at 5 years: surgical intervention, colectomy, permanent diversion, fistula closure, and perianal abscess recurrence.
Although both reduced risk of perianal abscess recurrence, it was still higher with anti-TNF therapy (hazard ratio, 0.48; 95% confidence interval, 0.32-0.74) than with ustekinumab (HR, 0.20; 95% CI, 0.07-0.56). Ustekinumab also increased the rate of perianal fistula closure by more than threefold (HR, 3.58; 95% CI, 1.04-12.35).
Vedolizumab, on the other hand, offered no significant benefit across any of the five outcomes.
None of the biologics had an impact on rates of surgical intervention, colectomy, or permanent diversion.
Further analyses explored the long-term effects of second-line biologic choice after initial failure with anti-TNF therapy. Switching to another anti-TNF agent was more effective than switching to ustekinumab at reducing risks of colectomy (HR, 0.20; 95% CI, 0.04-0.90) and permanent diversion (HR, 0.16; 95% CI, 0.03-0.94); switching to ustekinumab was more effective than switching to vedolizumab for perianal fistula closure (HR, 0.22; 95% CI, 0.05-0.96).
Switching to another anti-TNF biologic or ustekinumab may be associated with better 5-year outcomes, compared with switching to vedolizumab in patients with pCD, according to Dr. Gubatan. Other guidelines or data that might steer this sequencing decision are scant. However, the findings should be validated with prospective data, ideally from head-to-head trials.
Jordan E. Axelrad, MD, of NYU Langone Health, New York, said the present study is noteworthy for addressing a “very-difficult-to-treat condition that has limited data as well as very limited long-term outcome data for our currently available interventions.”
Dr. Axelrad appreciated how the study focused on the distribution of clinical manifestations of pCD, including ulcers (10%), fissures (23.2%), abscesses (76.1%), and fistulas (84.2%). According to Dr. Axelrad, the efficacy data provide really important insights for clinicians who choose biologic therapies. He noted that, in the absence of head-to-head clinical trials, “it’s absolutely important that we use these results to help us guide therapy” for patients with pCD.
While the biologics included in the study were efficacious to varying degrees, Dr. Axelrad pointed out that no choice was associated with a reduced risk of surgical intervention. “That really underscored for me how complex this patient population is,” he said. “Despite good medical therapies … we’re still not necessarily making a huge dent in the risk of surgical intervention requirements for this complex patient group.”
Dr. Gubatan disclosed support from a Chan Zuckerberg Biohub Physician Scientist Scholar Award, a National Institutes of Health NIDDK LRP Award, and a Doris Duke Physician Scientist Fellowship Award; his colleagues reported no conflicts of interest. Dr. Axelrad reports relationships with Janssen, AbbVie, Pfizer, and others.
Choice of biologic therapy in the first line and later may impact long-term outcomes in patients with perianal Crohn’s disease (pCD), according to a retrospective study.
John Gubatan, MD, of Stanford (Calif.) University, and colleagues reported that, compared with no biologic therapy, first-line treatment with an anti–tumor necrosis factor (TNF) agent or ustekinumab significantly reduced risk of perianal abscess recurrence at 5 years, whereas vedolizumab offered no such benefit. After failure of the initial anti-TNF, switching to another anti-TNF agent is the most effective option.
“Although pCD is recognized to be an aggressive phenotype, data on whether escalating to a biologic at the time of perianal disease diagnosis may alter the natural history and long-term clinical outcomes of pCD is limited,” the researchers wrote in Journal of Clinical Gastroenterology. “This is the first study to explore how the type of biologic therapy at the time of perianal disease diagnosis and change in biologic therapy after first anti-TNF failure are associated with rates of long-term clinical outcomes.”
The study included 311 patients with pCD treated at Stanford University from 1998 to 2020. At the time of diagnosis, 168 of these patients started a biologic, most often an anti-TNF agent (n = 138), followed distantly by ustekinumab (n = 16) or vedolizumab (n = 14). Efficacy of these first-line biologics was compared with no biologic therapy in terms of five clinical outcomes at 5 years: surgical intervention, colectomy, permanent diversion, fistula closure, and perianal abscess recurrence.
Although both reduced risk of perianal abscess recurrence, it was still higher with anti-TNF therapy (hazard ratio, 0.48; 95% confidence interval, 0.32-0.74) than with ustekinumab (HR, 0.20; 95% CI, 0.07-0.56). Ustekinumab also increased the rate of perianal fistula closure by more than threefold (HR, 3.58; 95% CI, 1.04-12.35).
Vedolizumab, on the other hand, offered no significant benefit across any of the five outcomes.
None of the biologics had an impact on rates of surgical intervention, colectomy, or permanent diversion.
Further analyses explored the long-term effects of second-line biologic choice after initial failure with anti-TNF therapy. Switching to another anti-TNF agent was more effective than switching to ustekinumab at reducing risks of colectomy (HR, 0.20; 95% CI, 0.04-0.90) and permanent diversion (HR, 0.16; 95% CI, 0.03-0.94); switching to ustekinumab was more effective than switching to vedolizumab for perianal fistula closure (HR, 0.22; 95% CI, 0.05-0.96).
Switching to another anti-TNF biologic or ustekinumab may be associated with better 5-year outcomes, compared with switching to vedolizumab in patients with pCD, according to Dr. Gubatan. Other guidelines or data that might steer this sequencing decision are scant. However, the findings should be validated with prospective data, ideally from head-to-head trials.
Jordan E. Axelrad, MD, of NYU Langone Health, New York, said the present study is noteworthy for addressing a “very-difficult-to-treat condition that has limited data as well as very limited long-term outcome data for our currently available interventions.”
Dr. Axelrad appreciated how the study focused on the distribution of clinical manifestations of pCD, including ulcers (10%), fissures (23.2%), abscesses (76.1%), and fistulas (84.2%). According to Dr. Axelrad, the efficacy data provide really important insights for clinicians who choose biologic therapies. He noted that, in the absence of head-to-head clinical trials, “it’s absolutely important that we use these results to help us guide therapy” for patients with pCD.
While the biologics included in the study were efficacious to varying degrees, Dr. Axelrad pointed out that no choice was associated with a reduced risk of surgical intervention. “That really underscored for me how complex this patient population is,” he said. “Despite good medical therapies … we’re still not necessarily making a huge dent in the risk of surgical intervention requirements for this complex patient group.”
Dr. Gubatan disclosed support from a Chan Zuckerberg Biohub Physician Scientist Scholar Award, a National Institutes of Health NIDDK LRP Award, and a Doris Duke Physician Scientist Fellowship Award; his colleagues reported no conflicts of interest. Dr. Axelrad reports relationships with Janssen, AbbVie, Pfizer, and others.
FROM JOURNAL OF CLINICAL GASTROENTEROLOGY
Pulse oximeters lead to less oxygen supplementation for people of color
The new research suggests that skin color–related differences in pulse oximeter readings are in fact impacting clinical decision-making, lead author Eric R. Gottlieb, MD, of Brigham and Women’s Hospital and Massachusetts Institute of Technology, both in Boston, and colleagues wrote. This suggests that technology needs to updated to improve health equity, they continued, in their paper published in JAMA Internal Medicine.
“It has been known for decades that these readings are affected by various surface pigmentations, including nail polish and skin melanin, which may affect light absorption and scattering,” the investigators wrote. “This increases the risk of hidden hypoxemia [among patients with darker skin], in which patients have falsely elevated SpO2 readings, usually defined as 92% or greater, with a blood hemoglobin oxygen saturation less than 88%.”
Although published reports on this phenomenon date back to the 1980s, clinical significance has been largely discounted, they said, citing a 2008 paper on the topic, which stated that “oximetry need not have exact accuracy” to determine if a patient needs oxygen supplementation.
‘We’re not providing equal care’
Questioning the validity of this statement, Dr. Gottlieb and colleagues conducted a retrospective cohort study involving 3,069 patients admitted to intensive care at the Beth Israel Deaconess Medical Center in Boston between 2008 and 2019, thereby excluding patients treated during the COVID-19 pandemic. The population consisted of four races/ethnicities: White (87%), Black (7%), Hispanic (4%), and Asian (3%).
Aligning with previous studies, multivariable linear regression analyses showed that Asian, Black, and Hispanic patients had significantly higher SpO2 readings than White patients in relation to hemoglobin oxygen saturation values, suggesting falsely elevated readings.
Further modeling showed that these same patient groups also received lower oxygen delivery rates, which were not explained directly by race/ethnicity, but instead were mediated by the discrepancy between SpO2 and hemoglobin oxygen saturation values. In other words, physicians were responding consistently to pulse oximetry readings, rather than exhibiting a direct racial/ethnic bias in their clinical decision-making.
“We’re not providing equal care,” Dr. Gottlieb said in an interview. “It’s not that the patients are sicker, or have other socioeconomic explanations for why this happens to them. It’s us. It’s our technology. And that’s something that really has to be fixed.”
The investigators offered a cautionary view of corrective algorithms, as these “have exacerbated disparities and are subject to ethical concerns;” for example, with glomerular filtration rate estimations in Black patients.
Dr. Gottlieb also cautioned against action by individual physicians, who may now be inclined to change how they interpret pulse oximeter readings based on a patient’s race or ethnicity.
“I don’t think that we can expect physicians, every time they see a patient, to be second guessing whether the number basically reflects the truth,” he said.
Instead, Dr. Gottlieb suggested that the burden of change rests upon the shoulders of institutions, including hospitals and device manufacturers, both of which “really need to take the responsibility” for making sure that pulse oximeters are “equitable and have similar performance across races.”
While Dr. Gottlieb said that skin color likely plays the greatest role in measurement discrepancies, he encouraged stakeholders “to think broadly about this, and not just assume that it’s entirely skin color,” noting a small amount of evidence indicating that blood chemistry may also play a role. Still, he predicted that colorimetry – the direct measurement of skin color – will probably be incorporated into pulse oximeters of the future.
Black patients 3X more likely to have hidden hypoxia than White patients
Michael Sjoding, MD, of the University of Michigan, Ann Arbor, was one of the first to raise awareness of skin color–related issues with pulse oximeters during the throes of the COVID-19 pandemic. His study, which involved more than 10,000 patients, showed that Black patients were threefold more likely to have hidden hypoxia than White patients.
The present study shows that such discrepancies are indeed clinically significant, Dr. Sjoding said in an interview. And these data are needed, he added, to bring about change.
“What is being asked is potentially a big deal,” Dr. Sjoding said. “Pulse oximeters are everywhere, and it would be a big undertaking to redesign pulse oximeters and purchase new pulse oximeters. You need a compelling body of evidence to do that. I think it’s there now, clearly. So I’m hopeful that we’re going to finally move forward, towards having devices that we are confident work accurately in everyone.”
Why it has taken so long to gather this evidence, however, is a thornier topic, considering race-related discrepancies in pulse oximeter readings were first documented more than 3 decades ago.
“We sort of rediscovered something that had been known and had been described in the past,” Dr. Sjoding said. He explained how he and many of his colleagues had completed pulmonary fellowships, yet none of them knew of these potential issues with pulse oximeters until they began to observe differences in their own patients during the pandemic.
“I’ll give previous generations of researchers the benefit of the doubt,” Dr. Sjoding said, pointing out that techniques in data gathering and analysis have advanced considerably over the years. “The types of studies that were done before were very different than what we did.”
Yet Dr. Sjoding entertained the possibility that other factors may have been at play.
“I think definitely there’s a social commentary on prioritization of research,” he said.
The study was supported by grants from the National Institutes of Health. The investigators and Dr. Sjoding reported no conflicts of interest.
The new research suggests that skin color–related differences in pulse oximeter readings are in fact impacting clinical decision-making, lead author Eric R. Gottlieb, MD, of Brigham and Women’s Hospital and Massachusetts Institute of Technology, both in Boston, and colleagues wrote. This suggests that technology needs to updated to improve health equity, they continued, in their paper published in JAMA Internal Medicine.
“It has been known for decades that these readings are affected by various surface pigmentations, including nail polish and skin melanin, which may affect light absorption and scattering,” the investigators wrote. “This increases the risk of hidden hypoxemia [among patients with darker skin], in which patients have falsely elevated SpO2 readings, usually defined as 92% or greater, with a blood hemoglobin oxygen saturation less than 88%.”
Although published reports on this phenomenon date back to the 1980s, clinical significance has been largely discounted, they said, citing a 2008 paper on the topic, which stated that “oximetry need not have exact accuracy” to determine if a patient needs oxygen supplementation.
‘We’re not providing equal care’
Questioning the validity of this statement, Dr. Gottlieb and colleagues conducted a retrospective cohort study involving 3,069 patients admitted to intensive care at the Beth Israel Deaconess Medical Center in Boston between 2008 and 2019, thereby excluding patients treated during the COVID-19 pandemic. The population consisted of four races/ethnicities: White (87%), Black (7%), Hispanic (4%), and Asian (3%).
Aligning with previous studies, multivariable linear regression analyses showed that Asian, Black, and Hispanic patients had significantly higher SpO2 readings than White patients in relation to hemoglobin oxygen saturation values, suggesting falsely elevated readings.
Further modeling showed that these same patient groups also received lower oxygen delivery rates, which were not explained directly by race/ethnicity, but instead were mediated by the discrepancy between SpO2 and hemoglobin oxygen saturation values. In other words, physicians were responding consistently to pulse oximetry readings, rather than exhibiting a direct racial/ethnic bias in their clinical decision-making.
“We’re not providing equal care,” Dr. Gottlieb said in an interview. “It’s not that the patients are sicker, or have other socioeconomic explanations for why this happens to them. It’s us. It’s our technology. And that’s something that really has to be fixed.”
The investigators offered a cautionary view of corrective algorithms, as these “have exacerbated disparities and are subject to ethical concerns;” for example, with glomerular filtration rate estimations in Black patients.
Dr. Gottlieb also cautioned against action by individual physicians, who may now be inclined to change how they interpret pulse oximeter readings based on a patient’s race or ethnicity.
“I don’t think that we can expect physicians, every time they see a patient, to be second guessing whether the number basically reflects the truth,” he said.
Instead, Dr. Gottlieb suggested that the burden of change rests upon the shoulders of institutions, including hospitals and device manufacturers, both of which “really need to take the responsibility” for making sure that pulse oximeters are “equitable and have similar performance across races.”
While Dr. Gottlieb said that skin color likely plays the greatest role in measurement discrepancies, he encouraged stakeholders “to think broadly about this, and not just assume that it’s entirely skin color,” noting a small amount of evidence indicating that blood chemistry may also play a role. Still, he predicted that colorimetry – the direct measurement of skin color – will probably be incorporated into pulse oximeters of the future.
Black patients 3X more likely to have hidden hypoxia than White patients
Michael Sjoding, MD, of the University of Michigan, Ann Arbor, was one of the first to raise awareness of skin color–related issues with pulse oximeters during the throes of the COVID-19 pandemic. His study, which involved more than 10,000 patients, showed that Black patients were threefold more likely to have hidden hypoxia than White patients.
The present study shows that such discrepancies are indeed clinically significant, Dr. Sjoding said in an interview. And these data are needed, he added, to bring about change.
“What is being asked is potentially a big deal,” Dr. Sjoding said. “Pulse oximeters are everywhere, and it would be a big undertaking to redesign pulse oximeters and purchase new pulse oximeters. You need a compelling body of evidence to do that. I think it’s there now, clearly. So I’m hopeful that we’re going to finally move forward, towards having devices that we are confident work accurately in everyone.”
Why it has taken so long to gather this evidence, however, is a thornier topic, considering race-related discrepancies in pulse oximeter readings were first documented more than 3 decades ago.
“We sort of rediscovered something that had been known and had been described in the past,” Dr. Sjoding said. He explained how he and many of his colleagues had completed pulmonary fellowships, yet none of them knew of these potential issues with pulse oximeters until they began to observe differences in their own patients during the pandemic.
“I’ll give previous generations of researchers the benefit of the doubt,” Dr. Sjoding said, pointing out that techniques in data gathering and analysis have advanced considerably over the years. “The types of studies that were done before were very different than what we did.”
Yet Dr. Sjoding entertained the possibility that other factors may have been at play.
“I think definitely there’s a social commentary on prioritization of research,” he said.
The study was supported by grants from the National Institutes of Health. The investigators and Dr. Sjoding reported no conflicts of interest.
The new research suggests that skin color–related differences in pulse oximeter readings are in fact impacting clinical decision-making, lead author Eric R. Gottlieb, MD, of Brigham and Women’s Hospital and Massachusetts Institute of Technology, both in Boston, and colleagues wrote. This suggests that technology needs to updated to improve health equity, they continued, in their paper published in JAMA Internal Medicine.
“It has been known for decades that these readings are affected by various surface pigmentations, including nail polish and skin melanin, which may affect light absorption and scattering,” the investigators wrote. “This increases the risk of hidden hypoxemia [among patients with darker skin], in which patients have falsely elevated SpO2 readings, usually defined as 92% or greater, with a blood hemoglobin oxygen saturation less than 88%.”
Although published reports on this phenomenon date back to the 1980s, clinical significance has been largely discounted, they said, citing a 2008 paper on the topic, which stated that “oximetry need not have exact accuracy” to determine if a patient needs oxygen supplementation.
‘We’re not providing equal care’
Questioning the validity of this statement, Dr. Gottlieb and colleagues conducted a retrospective cohort study involving 3,069 patients admitted to intensive care at the Beth Israel Deaconess Medical Center in Boston between 2008 and 2019, thereby excluding patients treated during the COVID-19 pandemic. The population consisted of four races/ethnicities: White (87%), Black (7%), Hispanic (4%), and Asian (3%).
Aligning with previous studies, multivariable linear regression analyses showed that Asian, Black, and Hispanic patients had significantly higher SpO2 readings than White patients in relation to hemoglobin oxygen saturation values, suggesting falsely elevated readings.
Further modeling showed that these same patient groups also received lower oxygen delivery rates, which were not explained directly by race/ethnicity, but instead were mediated by the discrepancy between SpO2 and hemoglobin oxygen saturation values. In other words, physicians were responding consistently to pulse oximetry readings, rather than exhibiting a direct racial/ethnic bias in their clinical decision-making.
“We’re not providing equal care,” Dr. Gottlieb said in an interview. “It’s not that the patients are sicker, or have other socioeconomic explanations for why this happens to them. It’s us. It’s our technology. And that’s something that really has to be fixed.”
The investigators offered a cautionary view of corrective algorithms, as these “have exacerbated disparities and are subject to ethical concerns;” for example, with glomerular filtration rate estimations in Black patients.
Dr. Gottlieb also cautioned against action by individual physicians, who may now be inclined to change how they interpret pulse oximeter readings based on a patient’s race or ethnicity.
“I don’t think that we can expect physicians, every time they see a patient, to be second guessing whether the number basically reflects the truth,” he said.
Instead, Dr. Gottlieb suggested that the burden of change rests upon the shoulders of institutions, including hospitals and device manufacturers, both of which “really need to take the responsibility” for making sure that pulse oximeters are “equitable and have similar performance across races.”
While Dr. Gottlieb said that skin color likely plays the greatest role in measurement discrepancies, he encouraged stakeholders “to think broadly about this, and not just assume that it’s entirely skin color,” noting a small amount of evidence indicating that blood chemistry may also play a role. Still, he predicted that colorimetry – the direct measurement of skin color – will probably be incorporated into pulse oximeters of the future.
Black patients 3X more likely to have hidden hypoxia than White patients
Michael Sjoding, MD, of the University of Michigan, Ann Arbor, was one of the first to raise awareness of skin color–related issues with pulse oximeters during the throes of the COVID-19 pandemic. His study, which involved more than 10,000 patients, showed that Black patients were threefold more likely to have hidden hypoxia than White patients.
The present study shows that such discrepancies are indeed clinically significant, Dr. Sjoding said in an interview. And these data are needed, he added, to bring about change.
“What is being asked is potentially a big deal,” Dr. Sjoding said. “Pulse oximeters are everywhere, and it would be a big undertaking to redesign pulse oximeters and purchase new pulse oximeters. You need a compelling body of evidence to do that. I think it’s there now, clearly. So I’m hopeful that we’re going to finally move forward, towards having devices that we are confident work accurately in everyone.”
Why it has taken so long to gather this evidence, however, is a thornier topic, considering race-related discrepancies in pulse oximeter readings were first documented more than 3 decades ago.
“We sort of rediscovered something that had been known and had been described in the past,” Dr. Sjoding said. He explained how he and many of his colleagues had completed pulmonary fellowships, yet none of them knew of these potential issues with pulse oximeters until they began to observe differences in their own patients during the pandemic.
“I’ll give previous generations of researchers the benefit of the doubt,” Dr. Sjoding said, pointing out that techniques in data gathering and analysis have advanced considerably over the years. “The types of studies that were done before were very different than what we did.”
Yet Dr. Sjoding entertained the possibility that other factors may have been at play.
“I think definitely there’s a social commentary on prioritization of research,” he said.
The study was supported by grants from the National Institutes of Health. The investigators and Dr. Sjoding reported no conflicts of interest.
FROM JAMA INTERNAL MEDICINE
Medicare to cover colonoscopy after positive fecal test
Medicare will cover the full cost of colonoscopy after a positive noninvasive fecal test beginning in 2023, largely in response to a year-long advocacy campaign.
The benefit expansion is a “huge win” for patients, according to the American Gastroenterological Association, because it represents the end of out-of-pocket costs for colorectal cancer (CRC) screening.
“The continuum is complete!” said John Inadomi, MD, AGAF, past president of the AGA and a champion of the initiative within the organization.
Colonoscopy after a positive fecal test was previously considered a diagnostic procedure and therefore not considered part of the screening process by the Affordable Care Act, allowing payers to charge patients. That is, until the AGA and partners, including the American Cancer Society Cancer Action Network and Fight Colorectal Cancer, pushed back. First, the organizations successfully campaigned to ensure that private payers would cover the follow-up procedure. Now, after multiple meetings with the United States Department of Health & Human Services and Centers for Medicare & Medicaid Services, their collaborative efforts will end screening costs for patients with Medicare, pending finalization of the rule this fall. If finalized, it will take effect Jan. 2, 2023.
The policy change will “directly advance health equity” the AGA said, particularly among “rural communities and communities of color,” which are disproportionally affected by CRC.
“Cost-sharing is a well-recognized barrier to screening and has resulted in disparities,” said David Lieberman, MD, AGAF, who met with the CMS multiple times on behalf of the AGA. “Patients can now engage in CRC screening programs and be confident that they will not face unexpected cost-sharing for colonoscopy after a positive noninvasive screening test.”
AGA president John Carethers, MD, AGAF, who also met with the CMS, noted that reducing barriers to CRC screening will ultimately reduce CRC mortality.
“This is a win for all patients and should elevate our nation’s screening rates while lowering the overall cancer burden, saving lives,” he said.
Dr. Inadomi, Dr. Carethers, and Dr. Lieberman serve on the scientific advisory board of Geneoscopy; Dr. Lieberman is also on the scientific advisory board for ColoWrap.
Medicare will cover the full cost of colonoscopy after a positive noninvasive fecal test beginning in 2023, largely in response to a year-long advocacy campaign.
The benefit expansion is a “huge win” for patients, according to the American Gastroenterological Association, because it represents the end of out-of-pocket costs for colorectal cancer (CRC) screening.
“The continuum is complete!” said John Inadomi, MD, AGAF, past president of the AGA and a champion of the initiative within the organization.
Colonoscopy after a positive fecal test was previously considered a diagnostic procedure and therefore not considered part of the screening process by the Affordable Care Act, allowing payers to charge patients. That is, until the AGA and partners, including the American Cancer Society Cancer Action Network and Fight Colorectal Cancer, pushed back. First, the organizations successfully campaigned to ensure that private payers would cover the follow-up procedure. Now, after multiple meetings with the United States Department of Health & Human Services and Centers for Medicare & Medicaid Services, their collaborative efforts will end screening costs for patients with Medicare, pending finalization of the rule this fall. If finalized, it will take effect Jan. 2, 2023.
The policy change will “directly advance health equity” the AGA said, particularly among “rural communities and communities of color,” which are disproportionally affected by CRC.
“Cost-sharing is a well-recognized barrier to screening and has resulted in disparities,” said David Lieberman, MD, AGAF, who met with the CMS multiple times on behalf of the AGA. “Patients can now engage in CRC screening programs and be confident that they will not face unexpected cost-sharing for colonoscopy after a positive noninvasive screening test.”
AGA president John Carethers, MD, AGAF, who also met with the CMS, noted that reducing barriers to CRC screening will ultimately reduce CRC mortality.
“This is a win for all patients and should elevate our nation’s screening rates while lowering the overall cancer burden, saving lives,” he said.
Dr. Inadomi, Dr. Carethers, and Dr. Lieberman serve on the scientific advisory board of Geneoscopy; Dr. Lieberman is also on the scientific advisory board for ColoWrap.
Medicare will cover the full cost of colonoscopy after a positive noninvasive fecal test beginning in 2023, largely in response to a year-long advocacy campaign.
The benefit expansion is a “huge win” for patients, according to the American Gastroenterological Association, because it represents the end of out-of-pocket costs for colorectal cancer (CRC) screening.
“The continuum is complete!” said John Inadomi, MD, AGAF, past president of the AGA and a champion of the initiative within the organization.
Colonoscopy after a positive fecal test was previously considered a diagnostic procedure and therefore not considered part of the screening process by the Affordable Care Act, allowing payers to charge patients. That is, until the AGA and partners, including the American Cancer Society Cancer Action Network and Fight Colorectal Cancer, pushed back. First, the organizations successfully campaigned to ensure that private payers would cover the follow-up procedure. Now, after multiple meetings with the United States Department of Health & Human Services and Centers for Medicare & Medicaid Services, their collaborative efforts will end screening costs for patients with Medicare, pending finalization of the rule this fall. If finalized, it will take effect Jan. 2, 2023.
The policy change will “directly advance health equity” the AGA said, particularly among “rural communities and communities of color,” which are disproportionally affected by CRC.
“Cost-sharing is a well-recognized barrier to screening and has resulted in disparities,” said David Lieberman, MD, AGAF, who met with the CMS multiple times on behalf of the AGA. “Patients can now engage in CRC screening programs and be confident that they will not face unexpected cost-sharing for colonoscopy after a positive noninvasive screening test.”
AGA president John Carethers, MD, AGAF, who also met with the CMS, noted that reducing barriers to CRC screening will ultimately reduce CRC mortality.
“This is a win for all patients and should elevate our nation’s screening rates while lowering the overall cancer burden, saving lives,” he said.
Dr. Inadomi, Dr. Carethers, and Dr. Lieberman serve on the scientific advisory board of Geneoscopy; Dr. Lieberman is also on the scientific advisory board for ColoWrap.
What explains poor adherence to eosinophilic esophagitis therapy?
Almost half of adult patients with eosinophilic esophagitis (EoE) reported poor adherence to long-term medical and dietary therapy, with age younger than 40 years and low necessity beliefs being the strongest predictors, a new study finds.
Clinicians need to spend more time discussing the need for EoE therapy with their patients, especially if they are younger, according to lead author Maria L. Haasnoot, MD, of Amsterdam University Medical Center (UMC), the Netherlands, and colleagues.
“Chronic treatment is necessary to maintain suppression of the inflammation and prevent negative outcomes in the long-term,” they write.
Until the recent approval of dupilumab (Dupixent) by the U.S. Food and Drug Administration, patients with EoE relied upon off-label options, including proton pump inhibitors and swallowed topical steroids, as well as dietary interventions for ongoing suppression of inflammation. But only about 1 in 6 patients achieve complete remission at 5 years, according to Dr. Haasnoot and colleagues.
“It is uncertain to what degree limited adherence to treatment [plays] a role in the limited long-term effects of treatment,” they write.
The findings were published online in American Journal of Gastroenterology.
Addressing a knowledge gap
The cross-sectional study involved 177 adult patients with EoE treated at Amsterdam UMC, who were prescribed dietary or medical maintenance therapy. Of note, some patients were treated with budesonide, which is approved for EoE in Europe but not in the United States.
Median participant age was 43 years, with a male-skewed distribution (71% men). Patients had been on EoE treatment for 2-6 years. Most (76%) were on medical treatments. Nearly half were on diets that avoided one to five food groups, with some on both medical treatments and elimination diets.
Using a link sent by mail, participants completed the online Medication Adherence Rating Scale, along with several other questionnaires, such as the Beliefs about Medicine Questionnaire, to measure secondary outcomes, including a patient’s view of how necessary or disruptive maintenance therapy is in their life.
The overall prevalence of poor adherence to therapy was high (41.8%), including a nonsignificant difference in adherence between medical and dietary therapies.
“It might come as a surprise that dietary-treated patients are certainly not less adherent to treatment than medically treated patients,” the authors write, noting that the opposite is usually true.
Multivariate logistic regression showed that patients younger than 40 years were more than twice as likely to be poorly adherent (odds ratio, 2.571; 95% confidence interval, 1.195-5.532). Those with low necessity beliefs were more than four times as likely to be poorly adherent (OR, 4.423; 95% CI, 2.169-9.016). Other factors linked to poor adherence were patients with longer disease duration and more severe symptoms.
“Clinicians should pay more attention to treatment adherence, particularly in younger patients,” the authors conclude. “The necessity of treatment should be actively discussed, and efforts should be done to take doubts away, as this may improve treatment adherence and subsequently may improve treatment effects and long-term outcomes.”
More patient education needed
According to Jennifer L. Horsley-Silva, MD, of Mayo Clinic, Scottsdale, Ariz., “This study is important, as it is one of the first studies to investigate the rate of treatment adherence in EoE patients and attempts to identify factors associated with adherence both in medically and dietary treated patients.”
Dr. Horsley-Silva commented that the findings align with recent research she and her colleagues conducted at the Mayo Clinic, where few patients successfully completed a six-food elimination diet, even when paired with a dietitian. As with the present study, success trended lower among younger adults. “These findings highlight the need for physicians treating EoE to motivate all patients, but especially younger patients, by discussing disease pathophysiology and explaining the reason for maintenance treatment early on,” Dr. Horsley-Silva said.
Conversations should also address the discordance between symptoms and histologic disease, patient doubts and concerns, and other barriers to adherence, she noted.
“Shared decisionmaking is of utmost importance when deciding upon a maintenance treatment strategy and should be readdressed continually,” she added.
Gary W. Falk, MD, of Penn Medicine, Philadelphia, said that patients with EoE may be poorly adherent because therapies tend to be complicated and people often forget to take their medications, especially when their symptoms improve, even though this is a poor indicator of underlying disease. The discordance between symptoms and histology is “not commonly appreciated by the EoE GI community,” he noted.
Patients may benefit from knowing that untreated or undertreated EoE increases the risk for strictures and stenoses, need for dilation, and frequency of food bolus impactions, Dr. Falk said.
“The other thing we know is that once someone is induced into remission, and they stay on therapy ... long-term remission can be maintained,” he added.
The impact of Dupilumab
John Leung, MD, of Boston Food Allergy Center, also cited the complexities of EoE therapies as reason for poor adherence, though he believes this paradigm will shift now that dupilumab has been approved. Dupilumab injections are “just once a week, so it’s much easier in terms of frequency,” Dr. Leung said. “I would expect that the compliance [for dupilumab] will be better” than for older therapies.
Dr. Leung, who helped conduct the dupilumab clinical trials contributing to its approval for EoE and receives speaking honoraria from manufacturer Regeneron/Sanofi, said that dupilumab also overcomes the challenges with elimination diets while offering relief for concomitant conditions, such as “asthma, eczema, food allergies, and seasonal allergies.”
But Dr. Falk, who also worked on the dupilumab clinical trials, said the situation is “not straightforward,” even with FDA approval.
“There are going to be significant costs with [prescribing dupilumab], because it’s a biologic,” Dr. Falk said.
Dr. Falk also pointed out that prior authorization will be required, and until more studies can be conducted, the true impact of once-weekly dosing versus daily dosing remains unknown.
“I would say [dupilumab] has the potential to improve adherence, but we need to see if that’s going to be the case or not,” Dr. Falk said.
The authors disclosed relationships with Dr. Falk Pharma, AstraZeneca, and Sanofi/Regeneron (the manufacturers of Dupixent [dupilumab]), among others. Dr. Horsley-Silva, Dr. Falk, and Dr. Leung conducted clinical trials for dupilumab on behalf of Sanofi/Regeneron, with Dr. Leung also disclosing speaking honoraria from Sanofi/Regeneron. Dr. Horsley-Silva has acted as a clinical trial site principal investigator for Allakos and Celgene/Bristol-Myers Squibb.
A version of this article first appeared on Medscape.com.
Almost half of adult patients with eosinophilic esophagitis (EoE) reported poor adherence to long-term medical and dietary therapy, with age younger than 40 years and low necessity beliefs being the strongest predictors, a new study finds.
Clinicians need to spend more time discussing the need for EoE therapy with their patients, especially if they are younger, according to lead author Maria L. Haasnoot, MD, of Amsterdam University Medical Center (UMC), the Netherlands, and colleagues.
“Chronic treatment is necessary to maintain suppression of the inflammation and prevent negative outcomes in the long-term,” they write.
Until the recent approval of dupilumab (Dupixent) by the U.S. Food and Drug Administration, patients with EoE relied upon off-label options, including proton pump inhibitors and swallowed topical steroids, as well as dietary interventions for ongoing suppression of inflammation. But only about 1 in 6 patients achieve complete remission at 5 years, according to Dr. Haasnoot and colleagues.
“It is uncertain to what degree limited adherence to treatment [plays] a role in the limited long-term effects of treatment,” they write.
The findings were published online in American Journal of Gastroenterology.
Addressing a knowledge gap
The cross-sectional study involved 177 adult patients with EoE treated at Amsterdam UMC, who were prescribed dietary or medical maintenance therapy. Of note, some patients were treated with budesonide, which is approved for EoE in Europe but not in the United States.
Median participant age was 43 years, with a male-skewed distribution (71% men). Patients had been on EoE treatment for 2-6 years. Most (76%) were on medical treatments. Nearly half were on diets that avoided one to five food groups, with some on both medical treatments and elimination diets.
Using a link sent by mail, participants completed the online Medication Adherence Rating Scale, along with several other questionnaires, such as the Beliefs about Medicine Questionnaire, to measure secondary outcomes, including a patient’s view of how necessary or disruptive maintenance therapy is in their life.
The overall prevalence of poor adherence to therapy was high (41.8%), including a nonsignificant difference in adherence between medical and dietary therapies.
“It might come as a surprise that dietary-treated patients are certainly not less adherent to treatment than medically treated patients,” the authors write, noting that the opposite is usually true.
Multivariate logistic regression showed that patients younger than 40 years were more than twice as likely to be poorly adherent (odds ratio, 2.571; 95% confidence interval, 1.195-5.532). Those with low necessity beliefs were more than four times as likely to be poorly adherent (OR, 4.423; 95% CI, 2.169-9.016). Other factors linked to poor adherence were patients with longer disease duration and more severe symptoms.
“Clinicians should pay more attention to treatment adherence, particularly in younger patients,” the authors conclude. “The necessity of treatment should be actively discussed, and efforts should be done to take doubts away, as this may improve treatment adherence and subsequently may improve treatment effects and long-term outcomes.”
More patient education needed
According to Jennifer L. Horsley-Silva, MD, of Mayo Clinic, Scottsdale, Ariz., “This study is important, as it is one of the first studies to investigate the rate of treatment adherence in EoE patients and attempts to identify factors associated with adherence both in medically and dietary treated patients.”
Dr. Horsley-Silva commented that the findings align with recent research she and her colleagues conducted at the Mayo Clinic, where few patients successfully completed a six-food elimination diet, even when paired with a dietitian. As with the present study, success trended lower among younger adults. “These findings highlight the need for physicians treating EoE to motivate all patients, but especially younger patients, by discussing disease pathophysiology and explaining the reason for maintenance treatment early on,” Dr. Horsley-Silva said.
Conversations should also address the discordance between symptoms and histologic disease, patient doubts and concerns, and other barriers to adherence, she noted.
“Shared decisionmaking is of utmost importance when deciding upon a maintenance treatment strategy and should be readdressed continually,” she added.
Gary W. Falk, MD, of Penn Medicine, Philadelphia, said that patients with EoE may be poorly adherent because therapies tend to be complicated and people often forget to take their medications, especially when their symptoms improve, even though this is a poor indicator of underlying disease. The discordance between symptoms and histology is “not commonly appreciated by the EoE GI community,” he noted.
Patients may benefit from knowing that untreated or undertreated EoE increases the risk for strictures and stenoses, need for dilation, and frequency of food bolus impactions, Dr. Falk said.
“The other thing we know is that once someone is induced into remission, and they stay on therapy ... long-term remission can be maintained,” he added.
The impact of Dupilumab
John Leung, MD, of Boston Food Allergy Center, also cited the complexities of EoE therapies as reason for poor adherence, though he believes this paradigm will shift now that dupilumab has been approved. Dupilumab injections are “just once a week, so it’s much easier in terms of frequency,” Dr. Leung said. “I would expect that the compliance [for dupilumab] will be better” than for older therapies.
Dr. Leung, who helped conduct the dupilumab clinical trials contributing to its approval for EoE and receives speaking honoraria from manufacturer Regeneron/Sanofi, said that dupilumab also overcomes the challenges with elimination diets while offering relief for concomitant conditions, such as “asthma, eczema, food allergies, and seasonal allergies.”
But Dr. Falk, who also worked on the dupilumab clinical trials, said the situation is “not straightforward,” even with FDA approval.
“There are going to be significant costs with [prescribing dupilumab], because it’s a biologic,” Dr. Falk said.
Dr. Falk also pointed out that prior authorization will be required, and until more studies can be conducted, the true impact of once-weekly dosing versus daily dosing remains unknown.
“I would say [dupilumab] has the potential to improve adherence, but we need to see if that’s going to be the case or not,” Dr. Falk said.
The authors disclosed relationships with Dr. Falk Pharma, AstraZeneca, and Sanofi/Regeneron (the manufacturers of Dupixent [dupilumab]), among others. Dr. Horsley-Silva, Dr. Falk, and Dr. Leung conducted clinical trials for dupilumab on behalf of Sanofi/Regeneron, with Dr. Leung also disclosing speaking honoraria from Sanofi/Regeneron. Dr. Horsley-Silva has acted as a clinical trial site principal investigator for Allakos and Celgene/Bristol-Myers Squibb.
A version of this article first appeared on Medscape.com.
Almost half of adult patients with eosinophilic esophagitis (EoE) reported poor adherence to long-term medical and dietary therapy, with age younger than 40 years and low necessity beliefs being the strongest predictors, a new study finds.
Clinicians need to spend more time discussing the need for EoE therapy with their patients, especially if they are younger, according to lead author Maria L. Haasnoot, MD, of Amsterdam University Medical Center (UMC), the Netherlands, and colleagues.
“Chronic treatment is necessary to maintain suppression of the inflammation and prevent negative outcomes in the long-term,” they write.
Until the recent approval of dupilumab (Dupixent) by the U.S. Food and Drug Administration, patients with EoE relied upon off-label options, including proton pump inhibitors and swallowed topical steroids, as well as dietary interventions for ongoing suppression of inflammation. But only about 1 in 6 patients achieve complete remission at 5 years, according to Dr. Haasnoot and colleagues.
“It is uncertain to what degree limited adherence to treatment [plays] a role in the limited long-term effects of treatment,” they write.
The findings were published online in American Journal of Gastroenterology.
Addressing a knowledge gap
The cross-sectional study involved 177 adult patients with EoE treated at Amsterdam UMC, who were prescribed dietary or medical maintenance therapy. Of note, some patients were treated with budesonide, which is approved for EoE in Europe but not in the United States.
Median participant age was 43 years, with a male-skewed distribution (71% men). Patients had been on EoE treatment for 2-6 years. Most (76%) were on medical treatments. Nearly half were on diets that avoided one to five food groups, with some on both medical treatments and elimination diets.
Using a link sent by mail, participants completed the online Medication Adherence Rating Scale, along with several other questionnaires, such as the Beliefs about Medicine Questionnaire, to measure secondary outcomes, including a patient’s view of how necessary or disruptive maintenance therapy is in their life.
The overall prevalence of poor adherence to therapy was high (41.8%), including a nonsignificant difference in adherence between medical and dietary therapies.
“It might come as a surprise that dietary-treated patients are certainly not less adherent to treatment than medically treated patients,” the authors write, noting that the opposite is usually true.
Multivariate logistic regression showed that patients younger than 40 years were more than twice as likely to be poorly adherent (odds ratio, 2.571; 95% confidence interval, 1.195-5.532). Those with low necessity beliefs were more than four times as likely to be poorly adherent (OR, 4.423; 95% CI, 2.169-9.016). Other factors linked to poor adherence were patients with longer disease duration and more severe symptoms.
“Clinicians should pay more attention to treatment adherence, particularly in younger patients,” the authors conclude. “The necessity of treatment should be actively discussed, and efforts should be done to take doubts away, as this may improve treatment adherence and subsequently may improve treatment effects and long-term outcomes.”
More patient education needed
According to Jennifer L. Horsley-Silva, MD, of Mayo Clinic, Scottsdale, Ariz., “This study is important, as it is one of the first studies to investigate the rate of treatment adherence in EoE patients and attempts to identify factors associated with adherence both in medically and dietary treated patients.”
Dr. Horsley-Silva commented that the findings align with recent research she and her colleagues conducted at the Mayo Clinic, where few patients successfully completed a six-food elimination diet, even when paired with a dietitian. As with the present study, success trended lower among younger adults. “These findings highlight the need for physicians treating EoE to motivate all patients, but especially younger patients, by discussing disease pathophysiology and explaining the reason for maintenance treatment early on,” Dr. Horsley-Silva said.
Conversations should also address the discordance between symptoms and histologic disease, patient doubts and concerns, and other barriers to adherence, she noted.
“Shared decisionmaking is of utmost importance when deciding upon a maintenance treatment strategy and should be readdressed continually,” she added.
Gary W. Falk, MD, of Penn Medicine, Philadelphia, said that patients with EoE may be poorly adherent because therapies tend to be complicated and people often forget to take their medications, especially when their symptoms improve, even though this is a poor indicator of underlying disease. The discordance between symptoms and histology is “not commonly appreciated by the EoE GI community,” he noted.
Patients may benefit from knowing that untreated or undertreated EoE increases the risk for strictures and stenoses, need for dilation, and frequency of food bolus impactions, Dr. Falk said.
“The other thing we know is that once someone is induced into remission, and they stay on therapy ... long-term remission can be maintained,” he added.
The impact of Dupilumab
John Leung, MD, of Boston Food Allergy Center, also cited the complexities of EoE therapies as reason for poor adherence, though he believes this paradigm will shift now that dupilumab has been approved. Dupilumab injections are “just once a week, so it’s much easier in terms of frequency,” Dr. Leung said. “I would expect that the compliance [for dupilumab] will be better” than for older therapies.
Dr. Leung, who helped conduct the dupilumab clinical trials contributing to its approval for EoE and receives speaking honoraria from manufacturer Regeneron/Sanofi, said that dupilumab also overcomes the challenges with elimination diets while offering relief for concomitant conditions, such as “asthma, eczema, food allergies, and seasonal allergies.”
But Dr. Falk, who also worked on the dupilumab clinical trials, said the situation is “not straightforward,” even with FDA approval.
“There are going to be significant costs with [prescribing dupilumab], because it’s a biologic,” Dr. Falk said.
Dr. Falk also pointed out that prior authorization will be required, and until more studies can be conducted, the true impact of once-weekly dosing versus daily dosing remains unknown.
“I would say [dupilumab] has the potential to improve adherence, but we need to see if that’s going to be the case or not,” Dr. Falk said.
The authors disclosed relationships with Dr. Falk Pharma, AstraZeneca, and Sanofi/Regeneron (the manufacturers of Dupixent [dupilumab]), among others. Dr. Horsley-Silva, Dr. Falk, and Dr. Leung conducted clinical trials for dupilumab on behalf of Sanofi/Regeneron, with Dr. Leung also disclosing speaking honoraria from Sanofi/Regeneron. Dr. Horsley-Silva has acted as a clinical trial site principal investigator for Allakos and Celgene/Bristol-Myers Squibb.
A version of this article first appeared on Medscape.com.
AGA Clinical Practice Guidelines: Pharmacologic treatment of IBS
The American Gastroenterological Association has issued new guidelines for the medical treatment of irritable bowel syndrome (IBS).
The guidelines, which are separated into one publication for IBS with constipation (IBS-C) and another for IBS with diarrhea (IBS-D), are the first to advise clinicians in the usage of new, old, and over-the-counter drugs for IBS, according to a press release from the AGA.
“With more treatments available, physicians can tailor a personalized approach based on the symptoms a patient with IBS is experiencing,” AGA said.
Published simultaneously in Gastroenterology, the two guidelines describe a shared rationale for their creation, noting how the treatment landscape has changed since the AGA last issued IBS guidelines in 2014.
“New pharmacological treatments have become available and new evidence has accumulated about established treatments,” both guidelines stated. “The purpose of these guidelines is to provide evidence-based recommendations for the pharmacologic management” of individuals with IBS “based on a systematic and comprehensive synthesis of the literature.”
IBS-C
In the IBS-C guidelines, co–first authors Lin Chang, MD, AGAF, of the University of Los Angeles, and Shahnaz Sultan, MD, MHSc, AGAF, of the Minneapolis Veterans Affairs Healthcare System, noted that IBS-C accounts for “more than a third of IBS cases,” with patients frequently reporting “feeling self-conscious, avoiding sex, difficulty concentrating, [and] not feeling able to reach one’s full potential.”
They offered nine pharmacologic recommendations, eight of which are conditional, with certainty in evidence ranging from low to high.
The only strong recommendation with a high certainty in evidence is for linaclotide.
“Across four RCTs [randomized controlled trials], linaclotide improved global assessment of IBS-C symptoms (FDA responder), abdominal pain, complete spontaneous bowel movement response, as well as adequate global response,” Dr. Chang and colleagues wrote.
Conditional recommendations with moderate certainty in evidence are provided for tenapanor, plecanatide, tegaserod, and lubiprostone. Recommendations for polyethylene glycol laxatives, tricyclic antidepressants and antispasmodics are conditional and based on low-certainty evidence, as well as a conditional recommendation against selective serotonin reuptake inhibitors, also based on low-certainty evidence.
IBS-D
The IBS-D guidelines, led by co–first authors Anthony Lembo, MD, AGAF, of Beth Israel Deaconess Medical Center, Boston, and Dr. Sultan, includes eight conditional recommendations with certainty in evidence ranging from very low to moderate.
Drugs recommended based on moderate-certainty evidence include eluxadoline, alosetron, and rifaximin, with the added note that patients who respond to rifaximin but have recurrence should be treated again with rifaximin. Low-certainty evidence supported recommendations for tricyclic antidepressants, and antispasmodics. Very low–certainty evidence stands behind a recommendation for loperamide. Again, the panel made a conditional recommendation against SSRIs, also based on low-certainty evidence.
Shared decision-making
Both publications concluded with similar statements about the importance of shared decision-making, plus a practical mindset, in management of IBS.
“Acknowledging that multimodal treatments that include dietary and behavioral approaches in conjunction with drug therapy may provide maximal benefits and that treatment choices may be influenced by patient preferences, practitioners should engage in shared decision-making with patients when choosing the best therapy,” Dr. Lembo and colleagues wrote. “The importance of the patient-physician relationship is paramount in caring for individuals with IBS, and understanding patient preferences (for side-effect tolerability as well as cost) is valuable in choosing the right therapy.”
Both guidelines noted that some newer drugs for IBS have no generic alternative, and preauthorization may be required. Payer approval may depend on previous treatment failure with generic alternatives, they added.
The guidelines were commissioned and funded by the AGA Institute. The authors disclosed relationships with Ardelyx, Immunic, Protagonist, and others.
The American Gastroenterological Association has issued new guidelines for the medical treatment of irritable bowel syndrome (IBS).
The guidelines, which are separated into one publication for IBS with constipation (IBS-C) and another for IBS with diarrhea (IBS-D), are the first to advise clinicians in the usage of new, old, and over-the-counter drugs for IBS, according to a press release from the AGA.
“With more treatments available, physicians can tailor a personalized approach based on the symptoms a patient with IBS is experiencing,” AGA said.
Published simultaneously in Gastroenterology, the two guidelines describe a shared rationale for their creation, noting how the treatment landscape has changed since the AGA last issued IBS guidelines in 2014.
“New pharmacological treatments have become available and new evidence has accumulated about established treatments,” both guidelines stated. “The purpose of these guidelines is to provide evidence-based recommendations for the pharmacologic management” of individuals with IBS “based on a systematic and comprehensive synthesis of the literature.”
IBS-C
In the IBS-C guidelines, co–first authors Lin Chang, MD, AGAF, of the University of Los Angeles, and Shahnaz Sultan, MD, MHSc, AGAF, of the Minneapolis Veterans Affairs Healthcare System, noted that IBS-C accounts for “more than a third of IBS cases,” with patients frequently reporting “feeling self-conscious, avoiding sex, difficulty concentrating, [and] not feeling able to reach one’s full potential.”
They offered nine pharmacologic recommendations, eight of which are conditional, with certainty in evidence ranging from low to high.
The only strong recommendation with a high certainty in evidence is for linaclotide.
“Across four RCTs [randomized controlled trials], linaclotide improved global assessment of IBS-C symptoms (FDA responder), abdominal pain, complete spontaneous bowel movement response, as well as adequate global response,” Dr. Chang and colleagues wrote.
Conditional recommendations with moderate certainty in evidence are provided for tenapanor, plecanatide, tegaserod, and lubiprostone. Recommendations for polyethylene glycol laxatives, tricyclic antidepressants and antispasmodics are conditional and based on low-certainty evidence, as well as a conditional recommendation against selective serotonin reuptake inhibitors, also based on low-certainty evidence.
IBS-D
The IBS-D guidelines, led by co–first authors Anthony Lembo, MD, AGAF, of Beth Israel Deaconess Medical Center, Boston, and Dr. Sultan, includes eight conditional recommendations with certainty in evidence ranging from very low to moderate.
Drugs recommended based on moderate-certainty evidence include eluxadoline, alosetron, and rifaximin, with the added note that patients who respond to rifaximin but have recurrence should be treated again with rifaximin. Low-certainty evidence supported recommendations for tricyclic antidepressants, and antispasmodics. Very low–certainty evidence stands behind a recommendation for loperamide. Again, the panel made a conditional recommendation against SSRIs, also based on low-certainty evidence.
Shared decision-making
Both publications concluded with similar statements about the importance of shared decision-making, plus a practical mindset, in management of IBS.
“Acknowledging that multimodal treatments that include dietary and behavioral approaches in conjunction with drug therapy may provide maximal benefits and that treatment choices may be influenced by patient preferences, practitioners should engage in shared decision-making with patients when choosing the best therapy,” Dr. Lembo and colleagues wrote. “The importance of the patient-physician relationship is paramount in caring for individuals with IBS, and understanding patient preferences (for side-effect tolerability as well as cost) is valuable in choosing the right therapy.”
Both guidelines noted that some newer drugs for IBS have no generic alternative, and preauthorization may be required. Payer approval may depend on previous treatment failure with generic alternatives, they added.
The guidelines were commissioned and funded by the AGA Institute. The authors disclosed relationships with Ardelyx, Immunic, Protagonist, and others.
The American Gastroenterological Association has issued new guidelines for the medical treatment of irritable bowel syndrome (IBS).
The guidelines, which are separated into one publication for IBS with constipation (IBS-C) and another for IBS with diarrhea (IBS-D), are the first to advise clinicians in the usage of new, old, and over-the-counter drugs for IBS, according to a press release from the AGA.
“With more treatments available, physicians can tailor a personalized approach based on the symptoms a patient with IBS is experiencing,” AGA said.
Published simultaneously in Gastroenterology, the two guidelines describe a shared rationale for their creation, noting how the treatment landscape has changed since the AGA last issued IBS guidelines in 2014.
“New pharmacological treatments have become available and new evidence has accumulated about established treatments,” both guidelines stated. “The purpose of these guidelines is to provide evidence-based recommendations for the pharmacologic management” of individuals with IBS “based on a systematic and comprehensive synthesis of the literature.”
IBS-C
In the IBS-C guidelines, co–first authors Lin Chang, MD, AGAF, of the University of Los Angeles, and Shahnaz Sultan, MD, MHSc, AGAF, of the Minneapolis Veterans Affairs Healthcare System, noted that IBS-C accounts for “more than a third of IBS cases,” with patients frequently reporting “feeling self-conscious, avoiding sex, difficulty concentrating, [and] not feeling able to reach one’s full potential.”
They offered nine pharmacologic recommendations, eight of which are conditional, with certainty in evidence ranging from low to high.
The only strong recommendation with a high certainty in evidence is for linaclotide.
“Across four RCTs [randomized controlled trials], linaclotide improved global assessment of IBS-C symptoms (FDA responder), abdominal pain, complete spontaneous bowel movement response, as well as adequate global response,” Dr. Chang and colleagues wrote.
Conditional recommendations with moderate certainty in evidence are provided for tenapanor, plecanatide, tegaserod, and lubiprostone. Recommendations for polyethylene glycol laxatives, tricyclic antidepressants and antispasmodics are conditional and based on low-certainty evidence, as well as a conditional recommendation against selective serotonin reuptake inhibitors, also based on low-certainty evidence.
IBS-D
The IBS-D guidelines, led by co–first authors Anthony Lembo, MD, AGAF, of Beth Israel Deaconess Medical Center, Boston, and Dr. Sultan, includes eight conditional recommendations with certainty in evidence ranging from very low to moderate.
Drugs recommended based on moderate-certainty evidence include eluxadoline, alosetron, and rifaximin, with the added note that patients who respond to rifaximin but have recurrence should be treated again with rifaximin. Low-certainty evidence supported recommendations for tricyclic antidepressants, and antispasmodics. Very low–certainty evidence stands behind a recommendation for loperamide. Again, the panel made a conditional recommendation against SSRIs, also based on low-certainty evidence.
Shared decision-making
Both publications concluded with similar statements about the importance of shared decision-making, plus a practical mindset, in management of IBS.
“Acknowledging that multimodal treatments that include dietary and behavioral approaches in conjunction with drug therapy may provide maximal benefits and that treatment choices may be influenced by patient preferences, practitioners should engage in shared decision-making with patients when choosing the best therapy,” Dr. Lembo and colleagues wrote. “The importance of the patient-physician relationship is paramount in caring for individuals with IBS, and understanding patient preferences (for side-effect tolerability as well as cost) is valuable in choosing the right therapy.”
Both guidelines noted that some newer drugs for IBS have no generic alternative, and preauthorization may be required. Payer approval may depend on previous treatment failure with generic alternatives, they added.
The guidelines were commissioned and funded by the AGA Institute. The authors disclosed relationships with Ardelyx, Immunic, Protagonist, and others.
FROM GASTROENTEROLOGY
Center-based childcare associated with healthier body weight
The findings of the prospective Canadian study suggest that professional childcare centers that engage in standard practices are having a positive and lasting impact on children’s health, reported lead author Michaela Kucab, RD, MHSc, of the University of Toronto and colleagues.
“Attending center-based childcare in early childhood may influence important health behaviors including nutrition, physical activity, and routines related to child growth and weight status,” the investigators wrote in their abstract, which Ms. Kucab presented at the virtual conference sponsored by the American Society for Nutrition.
Their study involved 3,503 children who attended childcare in Canada during early childhood (mean age at baseline was 2.7 years) with follow-up from ages 4-10.
Overweight/obesity risk reduced
Children who received full-time, center-based care had a 22% lower risk of overweight/obesity and a mean body mass index z score (zBMI) that was 0.11 points lower at age 4 and 7 years than those who received non–center-based care. The benefits of center-based care were even more pronounced among children from lower-income families, who, at age 10, had a 48% lower risk of overweight/obesity and a mean zBMI that was 0.32 points lower with center-based versus non–center-based care.
In a written comment, Ms. Kucab and principal author Jonathon Maguire, MD, MSc, of the University of Toronto, explained that the former difference in zBMI translates to approximately half a pound of bodyweight in an average child, whereas the larger difference in zBMI among children from lower-income families would amount to approximately three pounds. They emphasized that these are rough estimations.
Ms. Kucab and Dr. Maguire noted that body weight differences correlated with the amount of time spent in center-based care.
“There was an observed trend, whereby the estimated mean difference [in zBMI] became slightly larger (or stronger) with a higher intensity of center-based childcare compared to non–center-based childcare,” they said.
To learn more about the earliest impacts of center-based care, the investigators are conducting a clinical trial, The Nutrition Recommendation Intervention Trials in Children’s Health Care (NuRISH), which will involve 600 children aged younger than 2 years.
Center-based childcare may reduce disadvantages of low-income children
“Although more research is needed, our findings suggest that center-based childcare may help” reduce disadvantages children from low-income families experience related to their heath,” Ms. Kucab said in a press release.
Laurent Legault, MD, an associate professor specializing in endocrinology in the department of pediatrics at McGill University, Montreal, highlighted the “quite significant” sample size of more than 3,000 participants, noting that “it’s quite tough to have numerous children” involved in a study, especially with several years of follow-up.
Dr. Legault also praised the investigators for considering socioeconomic status, “which is absolutely paramount, because, unfortunately, it’s not necessarily an even playing field for these families.”
He said the findings deserve to be promoted, as they highlight the benefits of center-based care, including ones with room for physical activity, opportunities for social interaction with other children, and a structured routine.
Still, Dr. Legault said it’s “very difficult to pinpoint specifically” what led to healthier body weights. “The problem, of course, is that obesity is very multifactorial in nature,” although “early intervention is more likely to be efficient.”
Center-based care appears to be one such intervention, he said, which should “push people to make centered care more affordable and easy to access for everyone.”The investigators and Dr. Legault reported no conflicts of interest.
The findings of the prospective Canadian study suggest that professional childcare centers that engage in standard practices are having a positive and lasting impact on children’s health, reported lead author Michaela Kucab, RD, MHSc, of the University of Toronto and colleagues.
“Attending center-based childcare in early childhood may influence important health behaviors including nutrition, physical activity, and routines related to child growth and weight status,” the investigators wrote in their abstract, which Ms. Kucab presented at the virtual conference sponsored by the American Society for Nutrition.
Their study involved 3,503 children who attended childcare in Canada during early childhood (mean age at baseline was 2.7 years) with follow-up from ages 4-10.
Overweight/obesity risk reduced
Children who received full-time, center-based care had a 22% lower risk of overweight/obesity and a mean body mass index z score (zBMI) that was 0.11 points lower at age 4 and 7 years than those who received non–center-based care. The benefits of center-based care were even more pronounced among children from lower-income families, who, at age 10, had a 48% lower risk of overweight/obesity and a mean zBMI that was 0.32 points lower with center-based versus non–center-based care.
In a written comment, Ms. Kucab and principal author Jonathon Maguire, MD, MSc, of the University of Toronto, explained that the former difference in zBMI translates to approximately half a pound of bodyweight in an average child, whereas the larger difference in zBMI among children from lower-income families would amount to approximately three pounds. They emphasized that these are rough estimations.
Ms. Kucab and Dr. Maguire noted that body weight differences correlated with the amount of time spent in center-based care.
“There was an observed trend, whereby the estimated mean difference [in zBMI] became slightly larger (or stronger) with a higher intensity of center-based childcare compared to non–center-based childcare,” they said.
To learn more about the earliest impacts of center-based care, the investigators are conducting a clinical trial, The Nutrition Recommendation Intervention Trials in Children’s Health Care (NuRISH), which will involve 600 children aged younger than 2 years.
Center-based childcare may reduce disadvantages of low-income children
“Although more research is needed, our findings suggest that center-based childcare may help” reduce disadvantages children from low-income families experience related to their heath,” Ms. Kucab said in a press release.
Laurent Legault, MD, an associate professor specializing in endocrinology in the department of pediatrics at McGill University, Montreal, highlighted the “quite significant” sample size of more than 3,000 participants, noting that “it’s quite tough to have numerous children” involved in a study, especially with several years of follow-up.
Dr. Legault also praised the investigators for considering socioeconomic status, “which is absolutely paramount, because, unfortunately, it’s not necessarily an even playing field for these families.”
He said the findings deserve to be promoted, as they highlight the benefits of center-based care, including ones with room for physical activity, opportunities for social interaction with other children, and a structured routine.
Still, Dr. Legault said it’s “very difficult to pinpoint specifically” what led to healthier body weights. “The problem, of course, is that obesity is very multifactorial in nature,” although “early intervention is more likely to be efficient.”
Center-based care appears to be one such intervention, he said, which should “push people to make centered care more affordable and easy to access for everyone.”The investigators and Dr. Legault reported no conflicts of interest.
The findings of the prospective Canadian study suggest that professional childcare centers that engage in standard practices are having a positive and lasting impact on children’s health, reported lead author Michaela Kucab, RD, MHSc, of the University of Toronto and colleagues.
“Attending center-based childcare in early childhood may influence important health behaviors including nutrition, physical activity, and routines related to child growth and weight status,” the investigators wrote in their abstract, which Ms. Kucab presented at the virtual conference sponsored by the American Society for Nutrition.
Their study involved 3,503 children who attended childcare in Canada during early childhood (mean age at baseline was 2.7 years) with follow-up from ages 4-10.
Overweight/obesity risk reduced
Children who received full-time, center-based care had a 22% lower risk of overweight/obesity and a mean body mass index z score (zBMI) that was 0.11 points lower at age 4 and 7 years than those who received non–center-based care. The benefits of center-based care were even more pronounced among children from lower-income families, who, at age 10, had a 48% lower risk of overweight/obesity and a mean zBMI that was 0.32 points lower with center-based versus non–center-based care.
In a written comment, Ms. Kucab and principal author Jonathon Maguire, MD, MSc, of the University of Toronto, explained that the former difference in zBMI translates to approximately half a pound of bodyweight in an average child, whereas the larger difference in zBMI among children from lower-income families would amount to approximately three pounds. They emphasized that these are rough estimations.
Ms. Kucab and Dr. Maguire noted that body weight differences correlated with the amount of time spent in center-based care.
“There was an observed trend, whereby the estimated mean difference [in zBMI] became slightly larger (or stronger) with a higher intensity of center-based childcare compared to non–center-based childcare,” they said.
To learn more about the earliest impacts of center-based care, the investigators are conducting a clinical trial, The Nutrition Recommendation Intervention Trials in Children’s Health Care (NuRISH), which will involve 600 children aged younger than 2 years.
Center-based childcare may reduce disadvantages of low-income children
“Although more research is needed, our findings suggest that center-based childcare may help” reduce disadvantages children from low-income families experience related to their heath,” Ms. Kucab said in a press release.
Laurent Legault, MD, an associate professor specializing in endocrinology in the department of pediatrics at McGill University, Montreal, highlighted the “quite significant” sample size of more than 3,000 participants, noting that “it’s quite tough to have numerous children” involved in a study, especially with several years of follow-up.
Dr. Legault also praised the investigators for considering socioeconomic status, “which is absolutely paramount, because, unfortunately, it’s not necessarily an even playing field for these families.”
He said the findings deserve to be promoted, as they highlight the benefits of center-based care, including ones with room for physical activity, opportunities for social interaction with other children, and a structured routine.
Still, Dr. Legault said it’s “very difficult to pinpoint specifically” what led to healthier body weights. “The problem, of course, is that obesity is very multifactorial in nature,” although “early intervention is more likely to be efficient.”
Center-based care appears to be one such intervention, he said, which should “push people to make centered care more affordable and easy to access for everyone.”The investigators and Dr. Legault reported no conflicts of interest.
FROM NUTRITION 2022