User login
with improved event-free and overall survival over intravenous cyclophosphamide, according to Keith M. Sullivan, MD, and his associates.
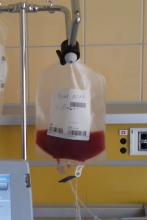
A total of 75 patients with severe scleroderma (diffuse cutaneous systemic sclerosis) underwent randomization to open-label treatment with either transplantation or intravenous cyclophosphamide at an initial dose of 500 mg/m2 of body-surface area, followed by 11 monthly infusions of 750 mg/m2 with mesna prophylaxis.
Additionally in the per-protocol population, the rate of event-free survival at 54 months was 79% in the transplantation group and 50% in the cyclophosphamide group, while the Kaplan-Meier estimated rate of event-free survival at 72 months was 74% in the transplantation group and 47% in the cyclophosphamide group (P = .03), and the rate of overall survival was 86% and 51% (P = .02). Also, in the per-protocol population at 54 months, the percentage of participants who had initiated disease-modifying antirheumatic drugs was lower in the transplantation group than in the cyclophosphamide group (9% vs. 44%; P = .001).
The researchers noted that 21 deaths occurred over a period of 72 months, including 7 participants in the transplantation group, of whom 3 died without receiving a transplant. The percentage of participants who had serious adverse events in the 72-month period was lower in the cyclophosphamide group than in the transplantation group (51% vs. 74%).
“In conclusion, at 54 months after randomization, myeloablative CD34-positive selected autologous hematopoietic stem-cell transplantation resulted in significantly better clinical outcomes than 12 months of cyclophosphamide,” the researchers wrote. “Although there was greater hematopoietic toxicity and an unquantified risk of second cancers from exposure to total body irradiation, toxic effects should be weighed against the beneficial results of treatment and the seriousness of the underlying disease.”
with improved event-free and overall survival over intravenous cyclophosphamide, according to Keith M. Sullivan, MD, and his associates.
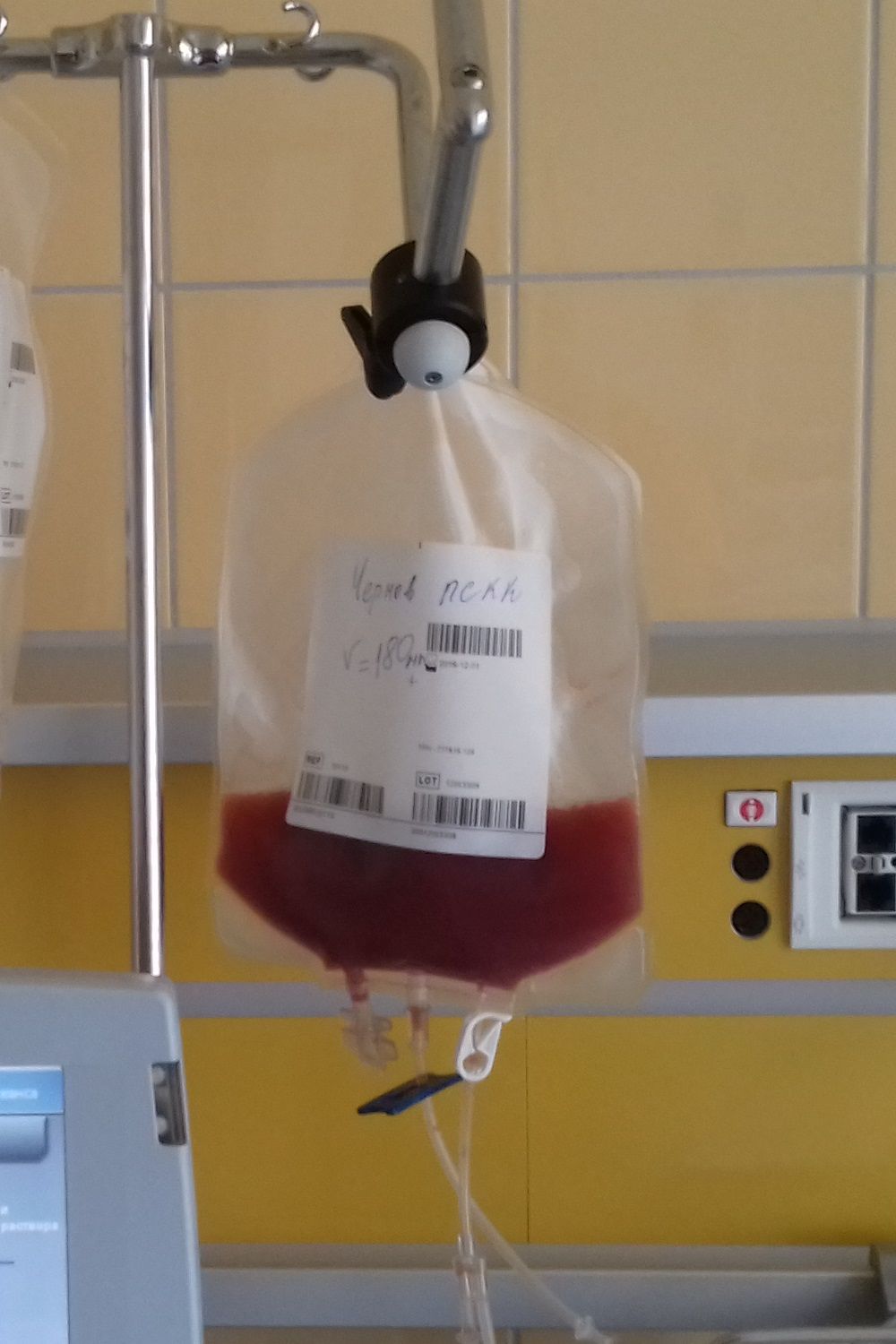
A total of 75 patients with severe scleroderma (diffuse cutaneous systemic sclerosis) underwent randomization to open-label treatment with either transplantation or intravenous cyclophosphamide at an initial dose of 500 mg/m2 of body-surface area, followed by 11 monthly infusions of 750 mg/m2 with mesna prophylaxis.
Additionally in the per-protocol population, the rate of event-free survival at 54 months was 79% in the transplantation group and 50% in the cyclophosphamide group, while the Kaplan-Meier estimated rate of event-free survival at 72 months was 74% in the transplantation group and 47% in the cyclophosphamide group (P = .03), and the rate of overall survival was 86% and 51% (P = .02). Also, in the per-protocol population at 54 months, the percentage of participants who had initiated disease-modifying antirheumatic drugs was lower in the transplantation group than in the cyclophosphamide group (9% vs. 44%; P = .001).
The researchers noted that 21 deaths occurred over a period of 72 months, including 7 participants in the transplantation group, of whom 3 died without receiving a transplant. The percentage of participants who had serious adverse events in the 72-month period was lower in the cyclophosphamide group than in the transplantation group (51% vs. 74%).
“In conclusion, at 54 months after randomization, myeloablative CD34-positive selected autologous hematopoietic stem-cell transplantation resulted in significantly better clinical outcomes than 12 months of cyclophosphamide,” the researchers wrote. “Although there was greater hematopoietic toxicity and an unquantified risk of second cancers from exposure to total body irradiation, toxic effects should be weighed against the beneficial results of treatment and the seriousness of the underlying disease.”
with improved event-free and overall survival over intravenous cyclophosphamide, according to Keith M. Sullivan, MD, and his associates.
A total of 75 patients with severe scleroderma (diffuse cutaneous systemic sclerosis) underwent randomization to open-label treatment with either transplantation or intravenous cyclophosphamide at an initial dose of 500 mg/m2 of body-surface area, followed by 11 monthly infusions of 750 mg/m2 with mesna prophylaxis.
Additionally in the per-protocol population, the rate of event-free survival at 54 months was 79% in the transplantation group and 50% in the cyclophosphamide group, while the Kaplan-Meier estimated rate of event-free survival at 72 months was 74% in the transplantation group and 47% in the cyclophosphamide group (P = .03), and the rate of overall survival was 86% and 51% (P = .02). Also, in the per-protocol population at 54 months, the percentage of participants who had initiated disease-modifying antirheumatic drugs was lower in the transplantation group than in the cyclophosphamide group (9% vs. 44%; P = .001).
The researchers noted that 21 deaths occurred over a period of 72 months, including 7 participants in the transplantation group, of whom 3 died without receiving a transplant. The percentage of participants who had serious adverse events in the 72-month period was lower in the cyclophosphamide group than in the transplantation group (51% vs. 74%).
“In conclusion, at 54 months after randomization, myeloablative CD34-positive selected autologous hematopoietic stem-cell transplantation resulted in significantly better clinical outcomes than 12 months of cyclophosphamide,” the researchers wrote. “Although there was greater hematopoietic toxicity and an unquantified risk of second cancers from exposure to total body irradiation, toxic effects should be weighed against the beneficial results of treatment and the seriousness of the underlying disease.”
FROM THE NEW ENGLAND JOURNAL OF MEDICINE