User login
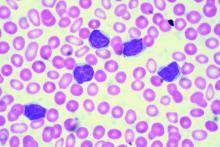
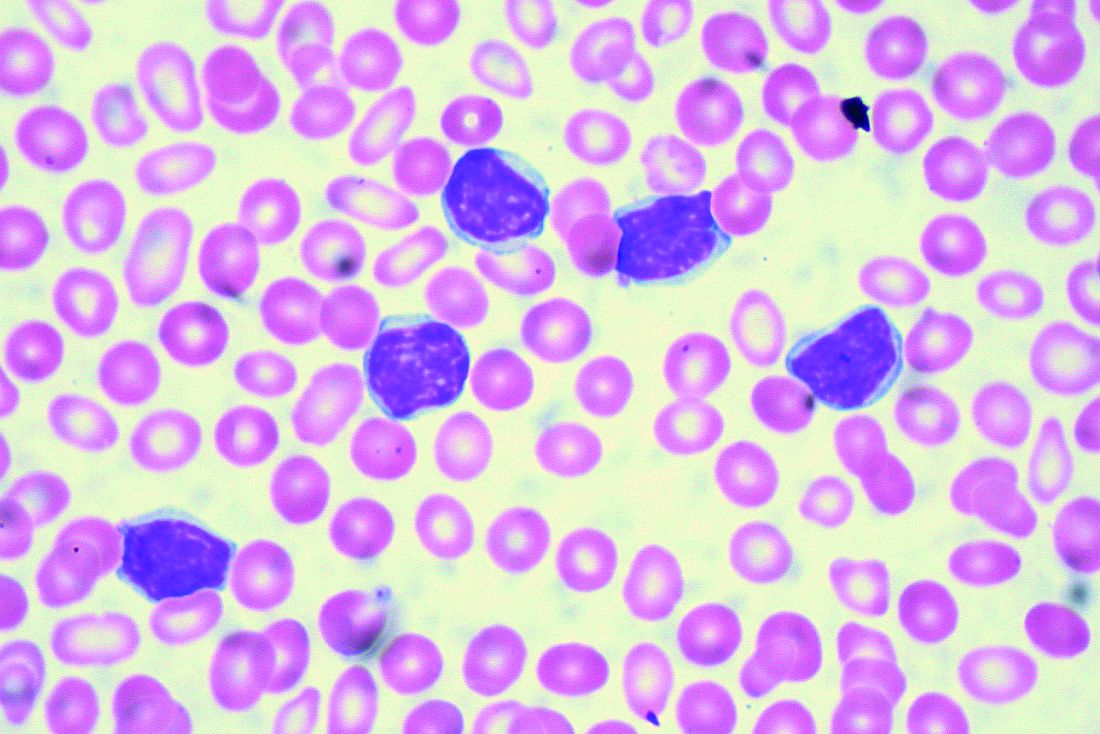
TKI discontinuation appears safe in CML
NEW YORK – Despite initial concerns that stopping tyrosine kinase inhibitor treatment would be ill-advised in patients with chronic myeloid leukemia (CML), clinical trial data suggest it is a safe and reasonable strategy, according to a leading expert.
“About 95% of people in all of these trials will regain their original response when they start off on therapy again,” said Jerald P. Radich, MD, of Fred Hutchinson Cancer Research Center/Seattle Cancer Care Alliance.
“There’s been a few that don’t, but blast crisis has been very, very rare, thank goodness, so it looks to be fairly safe for now,” Dr. Radich said at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
That being said, careful follow up is still required, Dr. Radich cautioned, noting that there was still an excess of CML in Hiroshima and Nagasaki atomic bomb survivors evident decades after radiation exposure.
“CML is a very strange disease,” he said. “You can’t eliminate the possibility of some slow-growing clone that, once you take [a patient] off tyrosine kinase therapy, is going into an accelerated phase and might take years to manifest itself.”
In one of the latest reports to shed light on what happens after discontinuation, investigators for the ENESTop study reported that treatment-free remission “seems achievable” in patients who have sustained, deep remissions after discontinuing nilotinib second-line therapy (Ann Intern Med. 2018 Apr 3;168[7]:461-70).
In ENESTop, chronic phase CML patients on tyrosine kinase inhibitors for at least 3 years were eligible to discontinue therapy if they achieved MR4.5 (BCR-ABL1IS of 0.0032% or less) and maintained that response level during a 1-year consolidation phase.
Out of 163 patients in the study, 126 met the criteria to enter the treatment-free remission phase; of that subset, 58% maintained treatment-free remission at 48 weeks, while 53% maintained it at 96 weeks, investigators said.
For 56 patients who restarted nilotinib, 55 regained at least major molecular response (MMR), and 52 regained MR4.5, while none had progression to accelerated phase or blast crisis, according to the report.
Similarly, earlier reported results from the ENESTfreedom trial showed that, of 190 patients entering the treatment-free remission phase after a median duration of 43.5 months on nilotinib, more than half remained in MMR or better at 48 weeks (Leukemia. 2017 Jul;31[7]:1525-31).
Of 86 patients who started nilotinib again after losing MMR, 98.8% regained MMR and 88.4% regained MR4.5 by the data cutoff date for the trial.
Duration and depth of response may make a “little bit of difference” in likelihood of relapse, Dr. Radich added.
In an interim analysis of a prospective multicenter, nonrandomized European discontinuation trial (EURO-SKI), investigators found that patients achieving deep molecular responses had good molecular relapse-free survival (Lancet Oncol. 2018 Jun;19[6]:747-57).
Based on that, investigators suggested that patients with deep molecular responses should be considered for discontinuation to spare them from side effects and to reduce health expenditures.
Results of these and other trials are “pretty much unbelievable,” Dr. Radich said. That’s in part because mathematical modeling – extrapolated from early trials – had suggested it could take nearly 50 years to completely eradicate minimal residual disease with tyrosine kinase inhibitors, and that the cumulative cure rate after 30 years of treatment could be as low as 31%.
Dr. Radich reported financial disclosures related to Amgen, Novartis, and Seattle Genetics.
NEW YORK – Despite initial concerns that stopping tyrosine kinase inhibitor treatment would be ill-advised in patients with chronic myeloid leukemia (CML), clinical trial data suggest it is a safe and reasonable strategy, according to a leading expert.
“About 95% of people in all of these trials will regain their original response when they start off on therapy again,” said Jerald P. Radich, MD, of Fred Hutchinson Cancer Research Center/Seattle Cancer Care Alliance.
“There’s been a few that don’t, but blast crisis has been very, very rare, thank goodness, so it looks to be fairly safe for now,” Dr. Radich said at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
That being said, careful follow up is still required, Dr. Radich cautioned, noting that there was still an excess of CML in Hiroshima and Nagasaki atomic bomb survivors evident decades after radiation exposure.
“CML is a very strange disease,” he said. “You can’t eliminate the possibility of some slow-growing clone that, once you take [a patient] off tyrosine kinase therapy, is going into an accelerated phase and might take years to manifest itself.”
In one of the latest reports to shed light on what happens after discontinuation, investigators for the ENESTop study reported that treatment-free remission “seems achievable” in patients who have sustained, deep remissions after discontinuing nilotinib second-line therapy (Ann Intern Med. 2018 Apr 3;168[7]:461-70).
In ENESTop, chronic phase CML patients on tyrosine kinase inhibitors for at least 3 years were eligible to discontinue therapy if they achieved MR4.5 (BCR-ABL1IS of 0.0032% or less) and maintained that response level during a 1-year consolidation phase.
Out of 163 patients in the study, 126 met the criteria to enter the treatment-free remission phase; of that subset, 58% maintained treatment-free remission at 48 weeks, while 53% maintained it at 96 weeks, investigators said.
For 56 patients who restarted nilotinib, 55 regained at least major molecular response (MMR), and 52 regained MR4.5, while none had progression to accelerated phase or blast crisis, according to the report.
Similarly, earlier reported results from the ENESTfreedom trial showed that, of 190 patients entering the treatment-free remission phase after a median duration of 43.5 months on nilotinib, more than half remained in MMR or better at 48 weeks (Leukemia. 2017 Jul;31[7]:1525-31).
Of 86 patients who started nilotinib again after losing MMR, 98.8% regained MMR and 88.4% regained MR4.5 by the data cutoff date for the trial.
Duration and depth of response may make a “little bit of difference” in likelihood of relapse, Dr. Radich added.
In an interim analysis of a prospective multicenter, nonrandomized European discontinuation trial (EURO-SKI), investigators found that patients achieving deep molecular responses had good molecular relapse-free survival (Lancet Oncol. 2018 Jun;19[6]:747-57).
Based on that, investigators suggested that patients with deep molecular responses should be considered for discontinuation to spare them from side effects and to reduce health expenditures.
Results of these and other trials are “pretty much unbelievable,” Dr. Radich said. That’s in part because mathematical modeling – extrapolated from early trials – had suggested it could take nearly 50 years to completely eradicate minimal residual disease with tyrosine kinase inhibitors, and that the cumulative cure rate after 30 years of treatment could be as low as 31%.
Dr. Radich reported financial disclosures related to Amgen, Novartis, and Seattle Genetics.
NEW YORK – Despite initial concerns that stopping tyrosine kinase inhibitor treatment would be ill-advised in patients with chronic myeloid leukemia (CML), clinical trial data suggest it is a safe and reasonable strategy, according to a leading expert.
“About 95% of people in all of these trials will regain their original response when they start off on therapy again,” said Jerald P. Radich, MD, of Fred Hutchinson Cancer Research Center/Seattle Cancer Care Alliance.
“There’s been a few that don’t, but blast crisis has been very, very rare, thank goodness, so it looks to be fairly safe for now,” Dr. Radich said at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
That being said, careful follow up is still required, Dr. Radich cautioned, noting that there was still an excess of CML in Hiroshima and Nagasaki atomic bomb survivors evident decades after radiation exposure.
“CML is a very strange disease,” he said. “You can’t eliminate the possibility of some slow-growing clone that, once you take [a patient] off tyrosine kinase therapy, is going into an accelerated phase and might take years to manifest itself.”
In one of the latest reports to shed light on what happens after discontinuation, investigators for the ENESTop study reported that treatment-free remission “seems achievable” in patients who have sustained, deep remissions after discontinuing nilotinib second-line therapy (Ann Intern Med. 2018 Apr 3;168[7]:461-70).
In ENESTop, chronic phase CML patients on tyrosine kinase inhibitors for at least 3 years were eligible to discontinue therapy if they achieved MR4.5 (BCR-ABL1IS of 0.0032% or less) and maintained that response level during a 1-year consolidation phase.
Out of 163 patients in the study, 126 met the criteria to enter the treatment-free remission phase; of that subset, 58% maintained treatment-free remission at 48 weeks, while 53% maintained it at 96 weeks, investigators said.
For 56 patients who restarted nilotinib, 55 regained at least major molecular response (MMR), and 52 regained MR4.5, while none had progression to accelerated phase or blast crisis, according to the report.
Similarly, earlier reported results from the ENESTfreedom trial showed that, of 190 patients entering the treatment-free remission phase after a median duration of 43.5 months on nilotinib, more than half remained in MMR or better at 48 weeks (Leukemia. 2017 Jul;31[7]:1525-31).
Of 86 patients who started nilotinib again after losing MMR, 98.8% regained MMR and 88.4% regained MR4.5 by the data cutoff date for the trial.
Duration and depth of response may make a “little bit of difference” in likelihood of relapse, Dr. Radich added.
In an interim analysis of a prospective multicenter, nonrandomized European discontinuation trial (EURO-SKI), investigators found that patients achieving deep molecular responses had good molecular relapse-free survival (Lancet Oncol. 2018 Jun;19[6]:747-57).
Based on that, investigators suggested that patients with deep molecular responses should be considered for discontinuation to spare them from side effects and to reduce health expenditures.
Results of these and other trials are “pretty much unbelievable,” Dr. Radich said. That’s in part because mathematical modeling – extrapolated from early trials – had suggested it could take nearly 50 years to completely eradicate minimal residual disease with tyrosine kinase inhibitors, and that the cumulative cure rate after 30 years of treatment could be as low as 31%.
Dr. Radich reported financial disclosures related to Amgen, Novartis, and Seattle Genetics.
EXPERT ANALYSIS FROM NCCN HEMATOLOGIC MALIGNANCIES
Will quad therapy become the new standard in myeloma?
NEW YORK – , though data from additional randomized trials are needed to define their role in clinical practice, according to Natalie S. Callander, MD, of the University of Wisconsin Carbone Cancer Center, Madison.
“The outlook for myeloma patients is quite good,” Dr. Callander said at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
“Triplet therapy is the standard, and quad therapy may be in the future.”
The study that set the standard for triplets in myeloma, according to Dr. Callander, is SWOG 0777, an open-label, phase 3 trial that compared bortezomib with lenalidomide and dexamethasone (VRd) to lenalidomide and dexamethasone alone in patients with newly diagnosed myeloma.
Adding bortezomib to lenalidomide and dexamethasone significantly improved both progression-free and overall survival in the 525-patient trial, with a risk-benefit profile that was acceptable (Lancet. 2017 Feb 4;389[10068]:519-27).
The median progression-free survival was 43 months for the triplet, versus 30 months for the two-drug regimen (P = .0018); likewise, median overall survival was significantly improved, at 75 months versus 64 months for triplet versus doublet therapy (P = .025).
“Very convincingly, just receiving that short exposure to bortezomib ended up causing a substantial increase of progression-free and overall survival,” Dr. Callander said.
The efficacy of multiple triplet regimens has been documented, including the combination of carfilzomib, lenalidomide, and dexamethasone (KRd); cyclophosphamide, bortezomib, and dexamethasone (CyBorD); and more recently, ixazomib, lenalidomide, and dexamethasone (IRd). These regimens have “excellent” response rates and survival data, Dr. Callander said.
Data is now emerging on the potential role of four-drug combinations, she added. The combination of elotuzumab plus VRd produced high response rates that were even higher after transplant, with reasonable toxicity, Dr. Callander said of phase 2 trial data presented at the 2017 annual meeting of the American Society of Clinical Oncology.
Similarly, the combination of daratumumab plus KRd had a 100% rate of partial response or better in phase 2 data presented at ASCO in 2017, with rates of very good partial response and complete response that improved with successive cycles of therapy, she said.
Even so, “it remains to be seen whether four drugs will be the new standard,” Dr. Callander told the NCCN attendees.
Four- versus three-drug strategies are being evaluated in ongoing randomized clinical trials, including patients with previously untreated myeloma, she said. Those studies include Cassiopeia, which is evaluating bortezomib, thalidomide, and dexamethasone (with or without daratumumab), and GRIFFIN, which is looking at VRd (with or without daratumumab).
Daratumumab recently received an additional indication in the treatment of myeloma, this time as part of a four-drug regimen, Dr. Callander added in a discussion on treatment options for elderly myeloma patients.
The Food and Drug Administration approved the monoclonal antibody in combination with bortezomib, melphalan, and prednisone (VMP) for treatment of newly diagnosed myeloma patients who are transplant ineligible.
That approval was based on results of the multicenter phase 3 ALCYONE study, showing an 18-month progression-free survival rate of 71.6% for the four-drug combination versus 50.2% for VMP alone (N Engl J Med. 2018;378:518-28).
Dr. Callander reported having no relevant financial disclosures.
NEW YORK – , though data from additional randomized trials are needed to define their role in clinical practice, according to Natalie S. Callander, MD, of the University of Wisconsin Carbone Cancer Center, Madison.
“The outlook for myeloma patients is quite good,” Dr. Callander said at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
“Triplet therapy is the standard, and quad therapy may be in the future.”
The study that set the standard for triplets in myeloma, according to Dr. Callander, is SWOG 0777, an open-label, phase 3 trial that compared bortezomib with lenalidomide and dexamethasone (VRd) to lenalidomide and dexamethasone alone in patients with newly diagnosed myeloma.
Adding bortezomib to lenalidomide and dexamethasone significantly improved both progression-free and overall survival in the 525-patient trial, with a risk-benefit profile that was acceptable (Lancet. 2017 Feb 4;389[10068]:519-27).
The median progression-free survival was 43 months for the triplet, versus 30 months for the two-drug regimen (P = .0018); likewise, median overall survival was significantly improved, at 75 months versus 64 months for triplet versus doublet therapy (P = .025).
“Very convincingly, just receiving that short exposure to bortezomib ended up causing a substantial increase of progression-free and overall survival,” Dr. Callander said.
The efficacy of multiple triplet regimens has been documented, including the combination of carfilzomib, lenalidomide, and dexamethasone (KRd); cyclophosphamide, bortezomib, and dexamethasone (CyBorD); and more recently, ixazomib, lenalidomide, and dexamethasone (IRd). These regimens have “excellent” response rates and survival data, Dr. Callander said.
Data is now emerging on the potential role of four-drug combinations, she added. The combination of elotuzumab plus VRd produced high response rates that were even higher after transplant, with reasonable toxicity, Dr. Callander said of phase 2 trial data presented at the 2017 annual meeting of the American Society of Clinical Oncology.
Similarly, the combination of daratumumab plus KRd had a 100% rate of partial response or better in phase 2 data presented at ASCO in 2017, with rates of very good partial response and complete response that improved with successive cycles of therapy, she said.
Even so, “it remains to be seen whether four drugs will be the new standard,” Dr. Callander told the NCCN attendees.
Four- versus three-drug strategies are being evaluated in ongoing randomized clinical trials, including patients with previously untreated myeloma, she said. Those studies include Cassiopeia, which is evaluating bortezomib, thalidomide, and dexamethasone (with or without daratumumab), and GRIFFIN, which is looking at VRd (with or without daratumumab).
Daratumumab recently received an additional indication in the treatment of myeloma, this time as part of a four-drug regimen, Dr. Callander added in a discussion on treatment options for elderly myeloma patients.
The Food and Drug Administration approved the monoclonal antibody in combination with bortezomib, melphalan, and prednisone (VMP) for treatment of newly diagnosed myeloma patients who are transplant ineligible.
That approval was based on results of the multicenter phase 3 ALCYONE study, showing an 18-month progression-free survival rate of 71.6% for the four-drug combination versus 50.2% for VMP alone (N Engl J Med. 2018;378:518-28).
Dr. Callander reported having no relevant financial disclosures.
NEW YORK – , though data from additional randomized trials are needed to define their role in clinical practice, according to Natalie S. Callander, MD, of the University of Wisconsin Carbone Cancer Center, Madison.
“The outlook for myeloma patients is quite good,” Dr. Callander said at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
“Triplet therapy is the standard, and quad therapy may be in the future.”
The study that set the standard for triplets in myeloma, according to Dr. Callander, is SWOG 0777, an open-label, phase 3 trial that compared bortezomib with lenalidomide and dexamethasone (VRd) to lenalidomide and dexamethasone alone in patients with newly diagnosed myeloma.
Adding bortezomib to lenalidomide and dexamethasone significantly improved both progression-free and overall survival in the 525-patient trial, with a risk-benefit profile that was acceptable (Lancet. 2017 Feb 4;389[10068]:519-27).
The median progression-free survival was 43 months for the triplet, versus 30 months for the two-drug regimen (P = .0018); likewise, median overall survival was significantly improved, at 75 months versus 64 months for triplet versus doublet therapy (P = .025).
“Very convincingly, just receiving that short exposure to bortezomib ended up causing a substantial increase of progression-free and overall survival,” Dr. Callander said.
The efficacy of multiple triplet regimens has been documented, including the combination of carfilzomib, lenalidomide, and dexamethasone (KRd); cyclophosphamide, bortezomib, and dexamethasone (CyBorD); and more recently, ixazomib, lenalidomide, and dexamethasone (IRd). These regimens have “excellent” response rates and survival data, Dr. Callander said.
Data is now emerging on the potential role of four-drug combinations, she added. The combination of elotuzumab plus VRd produced high response rates that were even higher after transplant, with reasonable toxicity, Dr. Callander said of phase 2 trial data presented at the 2017 annual meeting of the American Society of Clinical Oncology.
Similarly, the combination of daratumumab plus KRd had a 100% rate of partial response or better in phase 2 data presented at ASCO in 2017, with rates of very good partial response and complete response that improved with successive cycles of therapy, she said.
Even so, “it remains to be seen whether four drugs will be the new standard,” Dr. Callander told the NCCN attendees.
Four- versus three-drug strategies are being evaluated in ongoing randomized clinical trials, including patients with previously untreated myeloma, she said. Those studies include Cassiopeia, which is evaluating bortezomib, thalidomide, and dexamethasone (with or without daratumumab), and GRIFFIN, which is looking at VRd (with or without daratumumab).
Daratumumab recently received an additional indication in the treatment of myeloma, this time as part of a four-drug regimen, Dr. Callander added in a discussion on treatment options for elderly myeloma patients.
The Food and Drug Administration approved the monoclonal antibody in combination with bortezomib, melphalan, and prednisone (VMP) for treatment of newly diagnosed myeloma patients who are transplant ineligible.
That approval was based on results of the multicenter phase 3 ALCYONE study, showing an 18-month progression-free survival rate of 71.6% for the four-drug combination versus 50.2% for VMP alone (N Engl J Med. 2018;378:518-28).
Dr. Callander reported having no relevant financial disclosures.
EXPERT ANALYSIS FROM NCCN HEMATOLOGIC MALIGNANCIES
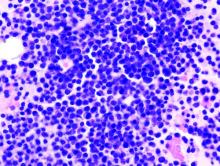
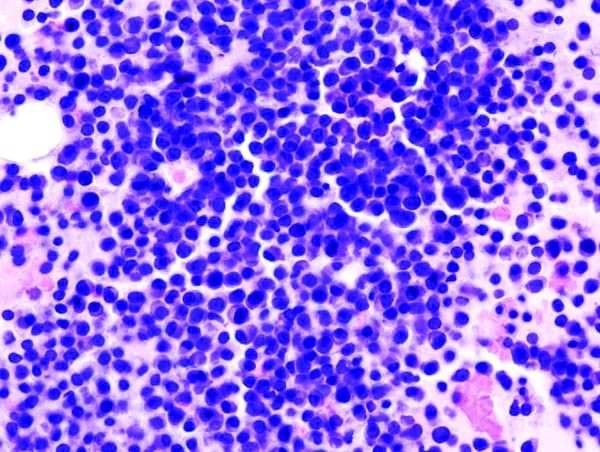
Novel options for treating hairy cell leukemia
NEW YORK – Ibrutinib, and now moxetumomab pasudotox, are two novel therapies that can be tried in patients with previously treated hairy cell leukemia, although data and experience with them are so far limited in this rare disease, experts said during a panel discussion at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
Since there are so few patients, data on the BTK inhibitor ibrutinib in hairy cell leukemia is largely “anecdotal,” said Andrew D. Zelenetz, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York.
The anti-CD22 monoclonal antibody moxetumomab pasudotox – approved for hairy cell leukemia in September – isn’t yet on the formulary at Memorial Sloan Kettering, Dr. Zelenetz added in a panel discussion of treatment options for a patient previously treated with purine analogueues and vemurafenib.
Between the two agents, moxetumomab pasudotox has more robust data in this disease, said John N. Allan, MD, of Weill Cornell Medicine, New York.
“I think if you can get access to the drug, that’s probably the best answer,” Dr. Allan said in the case discussion.
Hairy cell leukemia is an indolent B-cell lymphoma that makes up just 2% of all lymphoid leukemias, according to NCCN guidelines.
It is a chronic disease that requires long-term management, according to Dr. Allan.
First-line treatment is usually a purine analogue, either cladribine or pentostatin, and multiple treatments are possible as long as responses of greater than 2 years are achieved, he told attendees at the NCCN conference.
For relapses more than 2 years after first-line treatment, patients can be retreated with the same purine analogue, with or without rituximab, or can be switched to the alternative purine analogue, he said.
Vemurafenib, the BRAF inhibitor, is “surprisingly” effective in 90% of classic hairy cell leukemia patients with the BRAF V600E mutation, Dr. Allan added, though only about 40% of patients achieve complete response.
In discussing therapy options for a hairy cell leukemia patient previously treated with purine analogues and vemurafenib, Dr. Allan noted that the data behind ibrutinib includes case reports and early clinical investigations.
Several phase 1 studies with small numbers of patients show response rates “in the 50% range,” he said.
“This is an option,” he said. “It’s in the guidelines, and it’s something to consider.”
Moxetumomab pasudotox was recently approved for intravenous use in adults with relapsed or refractory hairy cell leukemia who have had at least two previous systemic treatments, including a purine nucleoside analogue. The CD22-directed cytotoxin is the first of its kind for treating patients with hairy cell leukemia, according to the Food and Drug Administration.
In a single-arm, open-label clinical trial including 80 patients with hairy cell leukemia who had previous treatment in line with that indication, 75% had a partial or complete response, of whom 30% had a durable complete response (CR), defined as maintaining hematologic remission for at least 180 days following CR.
Following the FDA’s approval of moxetumomab pasudotox, the NCCN updated its hairy cell leukemia clinical practice guidelines to include the drug as a category 2A recommendation for relapsed/refractory treatment. Other category 2A options in that setting include ibrutinib, vemurafenib with or without rituximab, or a clinical trial.
Along with that, NCCN guideline authors added a full page on special considerations for use of moxetumomab pasudotox. That includes advice on monitoring for capillary leak syndrome and hemolytic uremic syndrome, along with guidance on capillary leak syndrome grading and management by grade.
Dr. Zelenetz reported financial disclosures related to Adaptive Biotechnology, Amgen, AstraZeneca, Celgene, Genentech, Gilead, Hoffman La Roche, MEI Pharma, MorphoSys AG, Novartis, Pfizer, Pharmacyclics, Roche, and Verastem Oncology. Dr. Allan reported disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis, and Verastem Oncology.
NEW YORK – Ibrutinib, and now moxetumomab pasudotox, are two novel therapies that can be tried in patients with previously treated hairy cell leukemia, although data and experience with them are so far limited in this rare disease, experts said during a panel discussion at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
Since there are so few patients, data on the BTK inhibitor ibrutinib in hairy cell leukemia is largely “anecdotal,” said Andrew D. Zelenetz, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York.
The anti-CD22 monoclonal antibody moxetumomab pasudotox – approved for hairy cell leukemia in September – isn’t yet on the formulary at Memorial Sloan Kettering, Dr. Zelenetz added in a panel discussion of treatment options for a patient previously treated with purine analogueues and vemurafenib.
Between the two agents, moxetumomab pasudotox has more robust data in this disease, said John N. Allan, MD, of Weill Cornell Medicine, New York.
“I think if you can get access to the drug, that’s probably the best answer,” Dr. Allan said in the case discussion.
Hairy cell leukemia is an indolent B-cell lymphoma that makes up just 2% of all lymphoid leukemias, according to NCCN guidelines.
It is a chronic disease that requires long-term management, according to Dr. Allan.
First-line treatment is usually a purine analogue, either cladribine or pentostatin, and multiple treatments are possible as long as responses of greater than 2 years are achieved, he told attendees at the NCCN conference.
For relapses more than 2 years after first-line treatment, patients can be retreated with the same purine analogue, with or without rituximab, or can be switched to the alternative purine analogue, he said.
Vemurafenib, the BRAF inhibitor, is “surprisingly” effective in 90% of classic hairy cell leukemia patients with the BRAF V600E mutation, Dr. Allan added, though only about 40% of patients achieve complete response.
In discussing therapy options for a hairy cell leukemia patient previously treated with purine analogues and vemurafenib, Dr. Allan noted that the data behind ibrutinib includes case reports and early clinical investigations.
Several phase 1 studies with small numbers of patients show response rates “in the 50% range,” he said.
“This is an option,” he said. “It’s in the guidelines, and it’s something to consider.”
Moxetumomab pasudotox was recently approved for intravenous use in adults with relapsed or refractory hairy cell leukemia who have had at least two previous systemic treatments, including a purine nucleoside analogue. The CD22-directed cytotoxin is the first of its kind for treating patients with hairy cell leukemia, according to the Food and Drug Administration.
In a single-arm, open-label clinical trial including 80 patients with hairy cell leukemia who had previous treatment in line with that indication, 75% had a partial or complete response, of whom 30% had a durable complete response (CR), defined as maintaining hematologic remission for at least 180 days following CR.
Following the FDA’s approval of moxetumomab pasudotox, the NCCN updated its hairy cell leukemia clinical practice guidelines to include the drug as a category 2A recommendation for relapsed/refractory treatment. Other category 2A options in that setting include ibrutinib, vemurafenib with or without rituximab, or a clinical trial.
Along with that, NCCN guideline authors added a full page on special considerations for use of moxetumomab pasudotox. That includes advice on monitoring for capillary leak syndrome and hemolytic uremic syndrome, along with guidance on capillary leak syndrome grading and management by grade.
Dr. Zelenetz reported financial disclosures related to Adaptive Biotechnology, Amgen, AstraZeneca, Celgene, Genentech, Gilead, Hoffman La Roche, MEI Pharma, MorphoSys AG, Novartis, Pfizer, Pharmacyclics, Roche, and Verastem Oncology. Dr. Allan reported disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis, and Verastem Oncology.
NEW YORK – Ibrutinib, and now moxetumomab pasudotox, are two novel therapies that can be tried in patients with previously treated hairy cell leukemia, although data and experience with them are so far limited in this rare disease, experts said during a panel discussion at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
Since there are so few patients, data on the BTK inhibitor ibrutinib in hairy cell leukemia is largely “anecdotal,” said Andrew D. Zelenetz, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York.
The anti-CD22 monoclonal antibody moxetumomab pasudotox – approved for hairy cell leukemia in September – isn’t yet on the formulary at Memorial Sloan Kettering, Dr. Zelenetz added in a panel discussion of treatment options for a patient previously treated with purine analogueues and vemurafenib.
Between the two agents, moxetumomab pasudotox has more robust data in this disease, said John N. Allan, MD, of Weill Cornell Medicine, New York.
“I think if you can get access to the drug, that’s probably the best answer,” Dr. Allan said in the case discussion.
Hairy cell leukemia is an indolent B-cell lymphoma that makes up just 2% of all lymphoid leukemias, according to NCCN guidelines.
It is a chronic disease that requires long-term management, according to Dr. Allan.
First-line treatment is usually a purine analogue, either cladribine or pentostatin, and multiple treatments are possible as long as responses of greater than 2 years are achieved, he told attendees at the NCCN conference.
For relapses more than 2 years after first-line treatment, patients can be retreated with the same purine analogue, with or without rituximab, or can be switched to the alternative purine analogue, he said.
Vemurafenib, the BRAF inhibitor, is “surprisingly” effective in 90% of classic hairy cell leukemia patients with the BRAF V600E mutation, Dr. Allan added, though only about 40% of patients achieve complete response.
In discussing therapy options for a hairy cell leukemia patient previously treated with purine analogues and vemurafenib, Dr. Allan noted that the data behind ibrutinib includes case reports and early clinical investigations.
Several phase 1 studies with small numbers of patients show response rates “in the 50% range,” he said.
“This is an option,” he said. “It’s in the guidelines, and it’s something to consider.”
Moxetumomab pasudotox was recently approved for intravenous use in adults with relapsed or refractory hairy cell leukemia who have had at least two previous systemic treatments, including a purine nucleoside analogue. The CD22-directed cytotoxin is the first of its kind for treating patients with hairy cell leukemia, according to the Food and Drug Administration.
In a single-arm, open-label clinical trial including 80 patients with hairy cell leukemia who had previous treatment in line with that indication, 75% had a partial or complete response, of whom 30% had a durable complete response (CR), defined as maintaining hematologic remission for at least 180 days following CR.
Following the FDA’s approval of moxetumomab pasudotox, the NCCN updated its hairy cell leukemia clinical practice guidelines to include the drug as a category 2A recommendation for relapsed/refractory treatment. Other category 2A options in that setting include ibrutinib, vemurafenib with or without rituximab, or a clinical trial.
Along with that, NCCN guideline authors added a full page on special considerations for use of moxetumomab pasudotox. That includes advice on monitoring for capillary leak syndrome and hemolytic uremic syndrome, along with guidance on capillary leak syndrome grading and management by grade.
Dr. Zelenetz reported financial disclosures related to Adaptive Biotechnology, Amgen, AstraZeneca, Celgene, Genentech, Gilead, Hoffman La Roche, MEI Pharma, MorphoSys AG, Novartis, Pfizer, Pharmacyclics, Roche, and Verastem Oncology. Dr. Allan reported disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis, and Verastem Oncology.
EXPERT ANALYSIS FROM NCCN HEMATOLOGIC MALIGNANCIES
B-cell maturation antigen targeted in myeloma trials
NEW YORK – Three novel treatment strategies that target B-cell maturation antigen (BCMA) have shown promise in recent multiple myeloma clinical trials, according to Shaji K. Kumar, MD, of the Mayo Clinic Cancer Center in Rochester, Minn.
These strategies include B-cell maturation antigen (BCMA)–specific chimeric antigen receptor (CAR) T-cell therapies, bispecific T-cell engagers (BiTEs), and a BCMA antibody–drug conjugate, Dr. Kumar said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
“Clearly, there are a lot of exciting drugs that are currently in clinical trials, but these three platforms appear to be much more advanced than the others, and hopefully we will see that in the clinic in the near future,” Dr. Kumar said.
The antibody-drug conjugate, GSK2857916, is a humanized IgG1 anti-BCMA antibody conjugated to a microtubule-disrupting agent that has produced an overall response rate in 67% in a group of myeloma patients who had previously received multiple standard-of-care agents.
“Some of the responses were quite durable, lasting several months,” he said.
Now, GSK2857916 is being evaluated in a variety of different combinations, including in a phase 2 study of the antibody-drug conjugate in combination with lenalidomide plus dexamethasone, or bortezomib plus dexamethasone, in patients with relapsed or refractory disease.
Some of the most “exciting” data with anti-BCMA CAR T-cell therapy in myeloma involves bb2121, which showed durable clinical responses in heavily pretreated patients, according to data presented at the 2017 annual meeting of the American Society of Hematology.
“The overall response rate is quite significant,” said Dr. Kumar, who related a 94% rate of overall response that was even higher in patients treated with doses of 150 x 106 CAR+ T cells or more. Many of the response were lasting, he said, with five patients in ongoing response for more than 1 year.
“The results are exciting enough that this is actually moving forward with registration trials,” Dr. Kumar said.
Additionally, promising results have been presented on a novel CAR T-cell product, LCAR-B38M, which principally targets BCMA and led to a significant number of patients who achieved stringent complete response that lasted beyond 1 year.
Multiple BCMA-targeting CAR T-cell products that use different vectors and costimulatory molecules are currently undergoing clinical trials, Dr. Kumar said.
In contrast to CAR T-cell products that must be customized to each patient in a process that takes weeks, BiTEs are a ready-made approach to allow T cells to engage with tumor cells.
“In patients with advanced disease, a lot can change in that short time frame, so having an approach that is off the shelf, which is not patient specific, is quite attractive,” Dr. Kumar said.
BCMA-directed BiTE therapies under investigation include AMG 420 and PF-06863135, he added.
Dr. Kumar reported one disclosure related to Dr. Reddy’s Laboratories.
NEW YORK – Three novel treatment strategies that target B-cell maturation antigen (BCMA) have shown promise in recent multiple myeloma clinical trials, according to Shaji K. Kumar, MD, of the Mayo Clinic Cancer Center in Rochester, Minn.
These strategies include B-cell maturation antigen (BCMA)–specific chimeric antigen receptor (CAR) T-cell therapies, bispecific T-cell engagers (BiTEs), and a BCMA antibody–drug conjugate, Dr. Kumar said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
“Clearly, there are a lot of exciting drugs that are currently in clinical trials, but these three platforms appear to be much more advanced than the others, and hopefully we will see that in the clinic in the near future,” Dr. Kumar said.
The antibody-drug conjugate, GSK2857916, is a humanized IgG1 anti-BCMA antibody conjugated to a microtubule-disrupting agent that has produced an overall response rate in 67% in a group of myeloma patients who had previously received multiple standard-of-care agents.
“Some of the responses were quite durable, lasting several months,” he said.
Now, GSK2857916 is being evaluated in a variety of different combinations, including in a phase 2 study of the antibody-drug conjugate in combination with lenalidomide plus dexamethasone, or bortezomib plus dexamethasone, in patients with relapsed or refractory disease.
Some of the most “exciting” data with anti-BCMA CAR T-cell therapy in myeloma involves bb2121, which showed durable clinical responses in heavily pretreated patients, according to data presented at the 2017 annual meeting of the American Society of Hematology.
“The overall response rate is quite significant,” said Dr. Kumar, who related a 94% rate of overall response that was even higher in patients treated with doses of 150 x 106 CAR+ T cells or more. Many of the response were lasting, he said, with five patients in ongoing response for more than 1 year.
“The results are exciting enough that this is actually moving forward with registration trials,” Dr. Kumar said.
Additionally, promising results have been presented on a novel CAR T-cell product, LCAR-B38M, which principally targets BCMA and led to a significant number of patients who achieved stringent complete response that lasted beyond 1 year.
Multiple BCMA-targeting CAR T-cell products that use different vectors and costimulatory molecules are currently undergoing clinical trials, Dr. Kumar said.
In contrast to CAR T-cell products that must be customized to each patient in a process that takes weeks, BiTEs are a ready-made approach to allow T cells to engage with tumor cells.
“In patients with advanced disease, a lot can change in that short time frame, so having an approach that is off the shelf, which is not patient specific, is quite attractive,” Dr. Kumar said.
BCMA-directed BiTE therapies under investigation include AMG 420 and PF-06863135, he added.
Dr. Kumar reported one disclosure related to Dr. Reddy’s Laboratories.
NEW YORK – Three novel treatment strategies that target B-cell maturation antigen (BCMA) have shown promise in recent multiple myeloma clinical trials, according to Shaji K. Kumar, MD, of the Mayo Clinic Cancer Center in Rochester, Minn.
These strategies include B-cell maturation antigen (BCMA)–specific chimeric antigen receptor (CAR) T-cell therapies, bispecific T-cell engagers (BiTEs), and a BCMA antibody–drug conjugate, Dr. Kumar said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
“Clearly, there are a lot of exciting drugs that are currently in clinical trials, but these three platforms appear to be much more advanced than the others, and hopefully we will see that in the clinic in the near future,” Dr. Kumar said.
The antibody-drug conjugate, GSK2857916, is a humanized IgG1 anti-BCMA antibody conjugated to a microtubule-disrupting agent that has produced an overall response rate in 67% in a group of myeloma patients who had previously received multiple standard-of-care agents.
“Some of the responses were quite durable, lasting several months,” he said.
Now, GSK2857916 is being evaluated in a variety of different combinations, including in a phase 2 study of the antibody-drug conjugate in combination with lenalidomide plus dexamethasone, or bortezomib plus dexamethasone, in patients with relapsed or refractory disease.
Some of the most “exciting” data with anti-BCMA CAR T-cell therapy in myeloma involves bb2121, which showed durable clinical responses in heavily pretreated patients, according to data presented at the 2017 annual meeting of the American Society of Hematology.
“The overall response rate is quite significant,” said Dr. Kumar, who related a 94% rate of overall response that was even higher in patients treated with doses of 150 x 106 CAR+ T cells or more. Many of the response were lasting, he said, with five patients in ongoing response for more than 1 year.
“The results are exciting enough that this is actually moving forward with registration trials,” Dr. Kumar said.
Additionally, promising results have been presented on a novel CAR T-cell product, LCAR-B38M, which principally targets BCMA and led to a significant number of patients who achieved stringent complete response that lasted beyond 1 year.
Multiple BCMA-targeting CAR T-cell products that use different vectors and costimulatory molecules are currently undergoing clinical trials, Dr. Kumar said.
In contrast to CAR T-cell products that must be customized to each patient in a process that takes weeks, BiTEs are a ready-made approach to allow T cells to engage with tumor cells.
“In patients with advanced disease, a lot can change in that short time frame, so having an approach that is off the shelf, which is not patient specific, is quite attractive,” Dr. Kumar said.
BCMA-directed BiTE therapies under investigation include AMG 420 and PF-06863135, he added.
Dr. Kumar reported one disclosure related to Dr. Reddy’s Laboratories.
EXPERT ANALYSIS FROM THE NCCN HEMATOLOGIC MALIGNANCIES CONGRESS
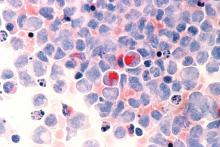
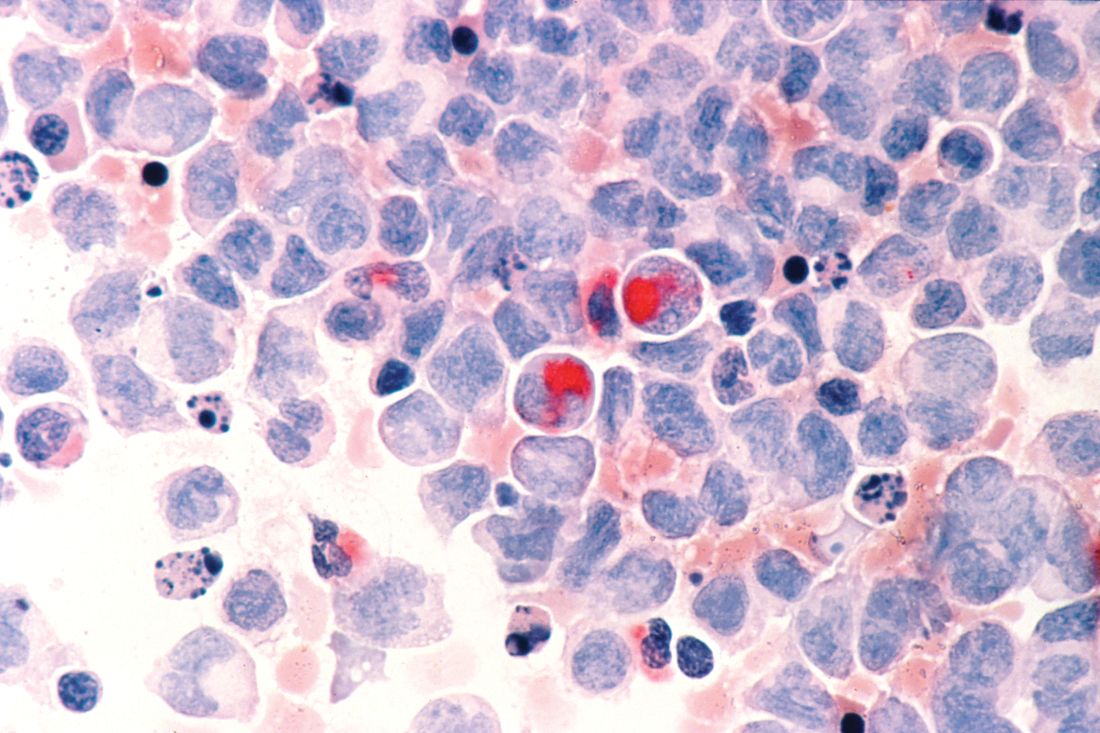
Venetoclax promising in unfit elderly AML patients
New York – For unfit elderly patients with acute myeloid leukemia (AML), venetoclax may be one of the most promising potential options that is emerging, according to an expert in the field.
The oral B-cell lymphoma 2 (BCL-2) inhibitor is the treatment that some in the AML community are “most excited about” for this population, Eunice S. Wang, MD, of Roswell Park Comprehensive Cancer Center in Buffalo, N.Y., said at the National Comprehensive Cancer Network (NCCN) Annual Congress: Hematologic Malignancies.
Venetoclax is currently approved by the Food and Drug Administration for previously treated chronic lymphocytic leukemia (CLL), alone or in combination with rituximab. It has been granted four Breakthrough Therapy designations from the FDA in AML. In July 2018, AbbVie submitted a Supplemental New Drug Application to the FDA for its use in combination with a hypomethylating agent (HMA) or in combination with low-dose cytarabine (LDAC) for the treatment of newly diagnosed AML patients who are ineligible for intensive chemotherapy.
“This agent doesn’t work on its own but had worked in the refractory setting and can be a great option upfront,” Dr. Wang said.
About half the patients were alive at 1 year following treatment with venetoclax plus low-dose chemotherapy, whether that was LDAC or an HMA, she said, commenting on recently reported results.
In a phase 1b trial, venetoclax was evaluated in combination with either azacitidine or decitabine. In recently reported preliminary results that included 57 patients aged 65 years or older who were ineligible for standard induction therapy, the combination was well tolerated and had promising activity (Lancet Oncol. 2018 Feb;19[2]:216-28).
Overall, 35 patients (61%) had complete remission (CR) or complete remission with incomplete marrow recovery (CRi).
In another report on this same trial, which included 33 patients from a single participating center who received venetoclax and azacytidine, the overall response rate was 91%, including 19 (58%) with CR and 9 (27%) with CRi (Blood. 2017 Dec;130 [Suppl 1]:181).
A separate phase 1/2 trial examined venetoclax plus LDAC in treatment-naive elderly patients unfit for intensive chemotherapy. In the 1-year outcomes that have been reported, the observed CR/CRi rate was 62%, median overall survival was an “encouraging” 11.4 months, and the observed 12-month overall survival was 46% in 61 patients treated at a venetoclax dose of 600 mg (Blood. 2017 Dec;130 [Suppl 1]:890).
In those 61 patients, treatment-related grade 3/4 adverse events included thrombocytopenia in 59%, neutropenia in 46%, febrile neutropenia in 36%, anemia in 28%, decreased white blood cell count in 26%, and one case of tumor lysis syndrome, according to the report.
Based on those findings in a cohort of patients with poor risk features, venetoclax 600 mg plus LDAC was carried forward to be evaluated in an ongoing phase 3 study, investigators noted at the time.
Dr. Wang reported financial relationships with AbbVie, Amgen, ImmunoGen, Incyte, Novartis, and Otsuka.
New York – For unfit elderly patients with acute myeloid leukemia (AML), venetoclax may be one of the most promising potential options that is emerging, according to an expert in the field.
The oral B-cell lymphoma 2 (BCL-2) inhibitor is the treatment that some in the AML community are “most excited about” for this population, Eunice S. Wang, MD, of Roswell Park Comprehensive Cancer Center in Buffalo, N.Y., said at the National Comprehensive Cancer Network (NCCN) Annual Congress: Hematologic Malignancies.
Venetoclax is currently approved by the Food and Drug Administration for previously treated chronic lymphocytic leukemia (CLL), alone or in combination with rituximab. It has been granted four Breakthrough Therapy designations from the FDA in AML. In July 2018, AbbVie submitted a Supplemental New Drug Application to the FDA for its use in combination with a hypomethylating agent (HMA) or in combination with low-dose cytarabine (LDAC) for the treatment of newly diagnosed AML patients who are ineligible for intensive chemotherapy.
“This agent doesn’t work on its own but had worked in the refractory setting and can be a great option upfront,” Dr. Wang said.
About half the patients were alive at 1 year following treatment with venetoclax plus low-dose chemotherapy, whether that was LDAC or an HMA, she said, commenting on recently reported results.
In a phase 1b trial, venetoclax was evaluated in combination with either azacitidine or decitabine. In recently reported preliminary results that included 57 patients aged 65 years or older who were ineligible for standard induction therapy, the combination was well tolerated and had promising activity (Lancet Oncol. 2018 Feb;19[2]:216-28).
Overall, 35 patients (61%) had complete remission (CR) or complete remission with incomplete marrow recovery (CRi).
In another report on this same trial, which included 33 patients from a single participating center who received venetoclax and azacytidine, the overall response rate was 91%, including 19 (58%) with CR and 9 (27%) with CRi (Blood. 2017 Dec;130 [Suppl 1]:181).
A separate phase 1/2 trial examined venetoclax plus LDAC in treatment-naive elderly patients unfit for intensive chemotherapy. In the 1-year outcomes that have been reported, the observed CR/CRi rate was 62%, median overall survival was an “encouraging” 11.4 months, and the observed 12-month overall survival was 46% in 61 patients treated at a venetoclax dose of 600 mg (Blood. 2017 Dec;130 [Suppl 1]:890).
In those 61 patients, treatment-related grade 3/4 adverse events included thrombocytopenia in 59%, neutropenia in 46%, febrile neutropenia in 36%, anemia in 28%, decreased white blood cell count in 26%, and one case of tumor lysis syndrome, according to the report.
Based on those findings in a cohort of patients with poor risk features, venetoclax 600 mg plus LDAC was carried forward to be evaluated in an ongoing phase 3 study, investigators noted at the time.
Dr. Wang reported financial relationships with AbbVie, Amgen, ImmunoGen, Incyte, Novartis, and Otsuka.
New York – For unfit elderly patients with acute myeloid leukemia (AML), venetoclax may be one of the most promising potential options that is emerging, according to an expert in the field.
The oral B-cell lymphoma 2 (BCL-2) inhibitor is the treatment that some in the AML community are “most excited about” for this population, Eunice S. Wang, MD, of Roswell Park Comprehensive Cancer Center in Buffalo, N.Y., said at the National Comprehensive Cancer Network (NCCN) Annual Congress: Hematologic Malignancies.
Venetoclax is currently approved by the Food and Drug Administration for previously treated chronic lymphocytic leukemia (CLL), alone or in combination with rituximab. It has been granted four Breakthrough Therapy designations from the FDA in AML. In July 2018, AbbVie submitted a Supplemental New Drug Application to the FDA for its use in combination with a hypomethylating agent (HMA) or in combination with low-dose cytarabine (LDAC) for the treatment of newly diagnosed AML patients who are ineligible for intensive chemotherapy.
“This agent doesn’t work on its own but had worked in the refractory setting and can be a great option upfront,” Dr. Wang said.
About half the patients were alive at 1 year following treatment with venetoclax plus low-dose chemotherapy, whether that was LDAC or an HMA, she said, commenting on recently reported results.
In a phase 1b trial, venetoclax was evaluated in combination with either azacitidine or decitabine. In recently reported preliminary results that included 57 patients aged 65 years or older who were ineligible for standard induction therapy, the combination was well tolerated and had promising activity (Lancet Oncol. 2018 Feb;19[2]:216-28).
Overall, 35 patients (61%) had complete remission (CR) or complete remission with incomplete marrow recovery (CRi).
In another report on this same trial, which included 33 patients from a single participating center who received venetoclax and azacytidine, the overall response rate was 91%, including 19 (58%) with CR and 9 (27%) with CRi (Blood. 2017 Dec;130 [Suppl 1]:181).
A separate phase 1/2 trial examined venetoclax plus LDAC in treatment-naive elderly patients unfit for intensive chemotherapy. In the 1-year outcomes that have been reported, the observed CR/CRi rate was 62%, median overall survival was an “encouraging” 11.4 months, and the observed 12-month overall survival was 46% in 61 patients treated at a venetoclax dose of 600 mg (Blood. 2017 Dec;130 [Suppl 1]:890).
In those 61 patients, treatment-related grade 3/4 adverse events included thrombocytopenia in 59%, neutropenia in 46%, febrile neutropenia in 36%, anemia in 28%, decreased white blood cell count in 26%, and one case of tumor lysis syndrome, according to the report.
Based on those findings in a cohort of patients with poor risk features, venetoclax 600 mg plus LDAC was carried forward to be evaluated in an ongoing phase 3 study, investigators noted at the time.
Dr. Wang reported financial relationships with AbbVie, Amgen, ImmunoGen, Incyte, Novartis, and Otsuka.
EXPERT ANALYSIS FROM THE NCCN ANNUAL CONGRESS: HEMATOLOGIC MALIGNANCIES
CAR T-cell studies dominate ongoing cellular therapy trials
NEW YORK – The cell therapy landscape increasingly involves strategies beyond chimeric antigen receptor (CAR) T-cell therapy, but those studies still predominate among investigational trials, according to Frederick L. Locke, MD, of Moffitt Cancer Center in Tampa.
Researchers are looking at CAR T-cell therapy for earlier lines of treatment, especially in patients with aggressive lymphomas, Dr. Locke said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
Of 753 trials examining cell therapies and listed at ClinicalTrials.gov as of March 30, 2018, about half (404) were CAR T-cell therapies. The others included T-cell receptor therapies, tumor infiltrating lymphocyte therapies, dendritic cell vaccines, and natural killer cell–based therapies, according to an article in Nature Reviews.
“The development isn’t just here in the United States,” Dr. Locke said. “It’s really global. We see a lot of activity in Europe, but also in China. We’re seeing medical advances across the world through molecular biology and gene engineering of T cells and other immune cells which can be adoptively transferred into patients.”
That activity includes studies seeking to move CAR T-cell therapy earlier in the treatment paradigm for some diseases, he added. “CAR T-cell therapy in non-Hodgkin lymphoma is really beginning a paradigm shift, at least in my mind.”
Several large, randomized trials that are now comparing CD19 CAR T-cell therapy with second-line standard-of-care therapies for patients with aggressive B-cell lymphomas. Among those trials is ZUMA-7, a phase 3, randomized trial comparing axicabtagene ciloleucel with standard-of-care treatment in patients with relapsed or refractory diffuse large B-cell lymphoma.
While prognosis remains poor for relapsed or progressing aggressive B-cell lymphomas treated with chemotherapy, data to date suggest CAR T-cell therapy produces durable, long-term remissions in about 40% of patients at “a year out and counting,” Dr. Locke said.
He presented a proposed treatment algorithm that included R-CHOP chemotherapy up front and CAR T-cell therapy in later lines of treatment, an approach that Dr. Locke speculated could result in a cure rate of perhaps 80% in large-cell lymphomas.
Encouraging longer-term data is emerging, with some patients with aggressive T-cell lymphomas now without recurrence for 5 years or more following a single infusion of CAR T-cell therapy, he said.
Dr. Locke reported a financial disclosure related to Cellular Biomedicine Group.
NEW YORK – The cell therapy landscape increasingly involves strategies beyond chimeric antigen receptor (CAR) T-cell therapy, but those studies still predominate among investigational trials, according to Frederick L. Locke, MD, of Moffitt Cancer Center in Tampa.
Researchers are looking at CAR T-cell therapy for earlier lines of treatment, especially in patients with aggressive lymphomas, Dr. Locke said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
Of 753 trials examining cell therapies and listed at ClinicalTrials.gov as of March 30, 2018, about half (404) were CAR T-cell therapies. The others included T-cell receptor therapies, tumor infiltrating lymphocyte therapies, dendritic cell vaccines, and natural killer cell–based therapies, according to an article in Nature Reviews.
“The development isn’t just here in the United States,” Dr. Locke said. “It’s really global. We see a lot of activity in Europe, but also in China. We’re seeing medical advances across the world through molecular biology and gene engineering of T cells and other immune cells which can be adoptively transferred into patients.”
That activity includes studies seeking to move CAR T-cell therapy earlier in the treatment paradigm for some diseases, he added. “CAR T-cell therapy in non-Hodgkin lymphoma is really beginning a paradigm shift, at least in my mind.”
Several large, randomized trials that are now comparing CD19 CAR T-cell therapy with second-line standard-of-care therapies for patients with aggressive B-cell lymphomas. Among those trials is ZUMA-7, a phase 3, randomized trial comparing axicabtagene ciloleucel with standard-of-care treatment in patients with relapsed or refractory diffuse large B-cell lymphoma.
While prognosis remains poor for relapsed or progressing aggressive B-cell lymphomas treated with chemotherapy, data to date suggest CAR T-cell therapy produces durable, long-term remissions in about 40% of patients at “a year out and counting,” Dr. Locke said.
He presented a proposed treatment algorithm that included R-CHOP chemotherapy up front and CAR T-cell therapy in later lines of treatment, an approach that Dr. Locke speculated could result in a cure rate of perhaps 80% in large-cell lymphomas.
Encouraging longer-term data is emerging, with some patients with aggressive T-cell lymphomas now without recurrence for 5 years or more following a single infusion of CAR T-cell therapy, he said.
Dr. Locke reported a financial disclosure related to Cellular Biomedicine Group.
NEW YORK – The cell therapy landscape increasingly involves strategies beyond chimeric antigen receptor (CAR) T-cell therapy, but those studies still predominate among investigational trials, according to Frederick L. Locke, MD, of Moffitt Cancer Center in Tampa.
Researchers are looking at CAR T-cell therapy for earlier lines of treatment, especially in patients with aggressive lymphomas, Dr. Locke said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
Of 753 trials examining cell therapies and listed at ClinicalTrials.gov as of March 30, 2018, about half (404) were CAR T-cell therapies. The others included T-cell receptor therapies, tumor infiltrating lymphocyte therapies, dendritic cell vaccines, and natural killer cell–based therapies, according to an article in Nature Reviews.
“The development isn’t just here in the United States,” Dr. Locke said. “It’s really global. We see a lot of activity in Europe, but also in China. We’re seeing medical advances across the world through molecular biology and gene engineering of T cells and other immune cells which can be adoptively transferred into patients.”
That activity includes studies seeking to move CAR T-cell therapy earlier in the treatment paradigm for some diseases, he added. “CAR T-cell therapy in non-Hodgkin lymphoma is really beginning a paradigm shift, at least in my mind.”
Several large, randomized trials that are now comparing CD19 CAR T-cell therapy with second-line standard-of-care therapies for patients with aggressive B-cell lymphomas. Among those trials is ZUMA-7, a phase 3, randomized trial comparing axicabtagene ciloleucel with standard-of-care treatment in patients with relapsed or refractory diffuse large B-cell lymphoma.
While prognosis remains poor for relapsed or progressing aggressive B-cell lymphomas treated with chemotherapy, data to date suggest CAR T-cell therapy produces durable, long-term remissions in about 40% of patients at “a year out and counting,” Dr. Locke said.
He presented a proposed treatment algorithm that included R-CHOP chemotherapy up front and CAR T-cell therapy in later lines of treatment, an approach that Dr. Locke speculated could result in a cure rate of perhaps 80% in large-cell lymphomas.
Encouraging longer-term data is emerging, with some patients with aggressive T-cell lymphomas now without recurrence for 5 years or more following a single infusion of CAR T-cell therapy, he said.
Dr. Locke reported a financial disclosure related to Cellular Biomedicine Group.
EXPERT ANALYSIS FROM THE NCCN HEMATOLOGIC MALIGNANCIES CONGRESS
Real-world clues for optimal sequencing of CLL novel agents
NEW YORK – Although optimal sequencing strategies in chronic lymphocytic leukemia are still unclear, real-world data suggest an alternate kinase inhibitor or venetoclax is the best approach for a patient who has received ibrutinib or idelalisib, according to John N. Allan, MD, of Cornell University, New York.
“I think for the most part, there’s enough evidence,” Dr. Allan said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
“If you had one to two lines of therapy, it still favors the novel agents rather than the chemotherapy arms in all these studies,” said Dr. Allan, referring to some of the pivotal trials supporting approval of novel agents in chronic lymphocytic leukemia (CLL). “The earlier we get to these drugs, I believe, the better.”
While venetoclax after ibrutinib is supported by multiple studies, “vice versa is unknown, but there’s seemingly no reason to think it wouldn’t work – different mechanisms of actions, different pathways,” Dr. Allan said.
What is clear, he added, is that retreating those patients with chemoimmunotherapy is not optimal.
In support of that, he cited a multicenter retrospective analysis, which is believed to be the largest real-world experience to date of novel agents in CLL looking at post–kinase inhibitor salvage strategies (Ann Oncol. 2017 May 1;28[5]:1050-6).
Using an alternate kinase inhibitor or venetoclax resulted in superior progression-free survival versus chemoimmunotherapy at the time of initial kinase inhibitor failure in that study, which looked at treatment strategies and outcomes for 683 patients.
Ibrutinib appeared to be superior to idelalisib as a first kinase inhibitor, with significantly better progression-free survival in both frontline and relapsed/refractory settings, and in both complex karyotype and del17p patients, according to the report. Additionally, the response rate to venetoclax seemed superior to that of idelalisib in patients who discontinued ibrutinib because of progression or toxicity.
All of that supports the need for trials to test various sequencing strategies and establish clear treatment algorithms, according to Dr. Allan. “Optimal sequencing is unknown, but real-world data gives us some idea.”
For relapsed/refractory patients, ibrutinib, idelalisib, and venetoclax all have lengthened responses, improved survival, and are approved by the Food and Drug Administration, he added, noting that the toxicity profiles vary and must be understood when dosing and prescribing these agents.
More novel treatments are on the way. On Sept. 24, just days after Dr. Allan’s NCCN presentation, the FDA granted approval to duvelisib for adults with relapsed or refractory CLL or small lymphocytic lymphoma following two or more previous lines of therapy.
Dr. Allan reported financial disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis Pharmaceuticals, and Verastem Oncology.
NEW YORK – Although optimal sequencing strategies in chronic lymphocytic leukemia are still unclear, real-world data suggest an alternate kinase inhibitor or venetoclax is the best approach for a patient who has received ibrutinib or idelalisib, according to John N. Allan, MD, of Cornell University, New York.
“I think for the most part, there’s enough evidence,” Dr. Allan said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
“If you had one to two lines of therapy, it still favors the novel agents rather than the chemotherapy arms in all these studies,” said Dr. Allan, referring to some of the pivotal trials supporting approval of novel agents in chronic lymphocytic leukemia (CLL). “The earlier we get to these drugs, I believe, the better.”
While venetoclax after ibrutinib is supported by multiple studies, “vice versa is unknown, but there’s seemingly no reason to think it wouldn’t work – different mechanisms of actions, different pathways,” Dr. Allan said.
What is clear, he added, is that retreating those patients with chemoimmunotherapy is not optimal.
In support of that, he cited a multicenter retrospective analysis, which is believed to be the largest real-world experience to date of novel agents in CLL looking at post–kinase inhibitor salvage strategies (Ann Oncol. 2017 May 1;28[5]:1050-6).
Using an alternate kinase inhibitor or venetoclax resulted in superior progression-free survival versus chemoimmunotherapy at the time of initial kinase inhibitor failure in that study, which looked at treatment strategies and outcomes for 683 patients.
Ibrutinib appeared to be superior to idelalisib as a first kinase inhibitor, with significantly better progression-free survival in both frontline and relapsed/refractory settings, and in both complex karyotype and del17p patients, according to the report. Additionally, the response rate to venetoclax seemed superior to that of idelalisib in patients who discontinued ibrutinib because of progression or toxicity.
All of that supports the need for trials to test various sequencing strategies and establish clear treatment algorithms, according to Dr. Allan. “Optimal sequencing is unknown, but real-world data gives us some idea.”
For relapsed/refractory patients, ibrutinib, idelalisib, and venetoclax all have lengthened responses, improved survival, and are approved by the Food and Drug Administration, he added, noting that the toxicity profiles vary and must be understood when dosing and prescribing these agents.
More novel treatments are on the way. On Sept. 24, just days after Dr. Allan’s NCCN presentation, the FDA granted approval to duvelisib for adults with relapsed or refractory CLL or small lymphocytic lymphoma following two or more previous lines of therapy.
Dr. Allan reported financial disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis Pharmaceuticals, and Verastem Oncology.
NEW YORK – Although optimal sequencing strategies in chronic lymphocytic leukemia are still unclear, real-world data suggest an alternate kinase inhibitor or venetoclax is the best approach for a patient who has received ibrutinib or idelalisib, according to John N. Allan, MD, of Cornell University, New York.
“I think for the most part, there’s enough evidence,” Dr. Allan said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
“If you had one to two lines of therapy, it still favors the novel agents rather than the chemotherapy arms in all these studies,” said Dr. Allan, referring to some of the pivotal trials supporting approval of novel agents in chronic lymphocytic leukemia (CLL). “The earlier we get to these drugs, I believe, the better.”
While venetoclax after ibrutinib is supported by multiple studies, “vice versa is unknown, but there’s seemingly no reason to think it wouldn’t work – different mechanisms of actions, different pathways,” Dr. Allan said.
What is clear, he added, is that retreating those patients with chemoimmunotherapy is not optimal.
In support of that, he cited a multicenter retrospective analysis, which is believed to be the largest real-world experience to date of novel agents in CLL looking at post–kinase inhibitor salvage strategies (Ann Oncol. 2017 May 1;28[5]:1050-6).
Using an alternate kinase inhibitor or venetoclax resulted in superior progression-free survival versus chemoimmunotherapy at the time of initial kinase inhibitor failure in that study, which looked at treatment strategies and outcomes for 683 patients.
Ibrutinib appeared to be superior to idelalisib as a first kinase inhibitor, with significantly better progression-free survival in both frontline and relapsed/refractory settings, and in both complex karyotype and del17p patients, according to the report. Additionally, the response rate to venetoclax seemed superior to that of idelalisib in patients who discontinued ibrutinib because of progression or toxicity.
All of that supports the need for trials to test various sequencing strategies and establish clear treatment algorithms, according to Dr. Allan. “Optimal sequencing is unknown, but real-world data gives us some idea.”
For relapsed/refractory patients, ibrutinib, idelalisib, and venetoclax all have lengthened responses, improved survival, and are approved by the Food and Drug Administration, he added, noting that the toxicity profiles vary and must be understood when dosing and prescribing these agents.
More novel treatments are on the way. On Sept. 24, just days after Dr. Allan’s NCCN presentation, the FDA granted approval to duvelisib for adults with relapsed or refractory CLL or small lymphocytic lymphoma following two or more previous lines of therapy.
Dr. Allan reported financial disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis Pharmaceuticals, and Verastem Oncology.
EXPERT ANALYSIS FROM NCCN HEMATOLOGIC MALIGNANCIES CONGRESS
CAR T may have curative potential in Hodgkin lymphoma
NEW YORK – Although the data set is small and not yet mature, chimeric antigen receptor (CAR) T-cell therapy appears to be a promising approach for Hodgkin lymphoma, according to Philippe Armand, MD, PhD, of Dana-Farber/Brigham and Women’s Cancer Center and the Massachusetts General Hospital Cancer Center in Boston.
While based on a handful of patients, the data do suggest this approach may play a role by targeting CD30 or Epstein Barr virus (EBV), Dr. Armand said at the NCCN Annual Congress: Hematologic Malignancies.
“Most importantly perhaps, like its experience outside of Hodgkin lymphoma, it may really have curative potential, based on the long [complete response] rates that have been already exhibited,” he said.
Much of the published clinical experience to date is with CD30-directed CAR Ts, Dr. Armand said, noting that in Hodgkin lymphoma, results so far show promise for this particular approach.
In a recent phase 1 dose escalation study, nine patients with relapsed/refractory Hodgkin lymphoma or anaplastic large-cell lymphoma (ALCL) received infusions of autologous T cells modified to express CD30-specific CAR T cells encoding the CD28 costimulatory domain, with no conditioning regimen.
Out of seven relapsed Hodgkin lymphoma patients, one had a complete response (CR) lasting beyond 2.5 years following a second infusion. Another patient had a CR persisting almost 2 years, and three patients had transient stable disease. One of the two ALCL patients had a CR lasting 9 months after a fourth infusion.
No toxicities attributable to the therapy were seen, according to the investigators.
The CD30 CAR T cells are being evaluated with a conditioning regimen in the phase 1 RELY-30 trial. Preliminary results presented at the 2018 European Society for Blood and Marrow Transplantation meeting in Lisbon showed better expansion of CAR T cells and responses in three out of five patients, including two CRs, according to Dr. Armand.
A CD30-directed CAR T-cell therapy with a 4-1BB costimulatory domain has also been tested in a small group of Hodgkin patients with a response rate of 35% – including some CRs – with an apparent lower response rate in patients with extranodal involvement. Dr. Armand noted that those findings need to be validated in additional studies.
Among non CD-30 targeted products, a CD19 CAR-T approach has been tried in Hodgkin lymphoma, though preliminary results suggest only transient activity, according to the presenter.
One interesting approach has been the targeting of EBV, he added. Recently reported results showed that two doses of T cells with specificity for EBV-derived tumor antigens induced clinical responses in patients with EBV-positive Hodgkin lymphoma.
The cells were engineered to express dominant-negative TGF-beta receptor type 2, according to the report. “We know that TGF-beta provides a strong immunosuppressant signal in the tumor microenvironment,” Dr. Armand said, noting that some of the responses in the seven evaluable patients lasted 4 years or more.
Dr. Armand reported financial disclosures related to Adaptive Biotechnologies/Sequenta, Affimed, Bristol-Myers Squibb, Merck, and Roche.
NEW YORK – Although the data set is small and not yet mature, chimeric antigen receptor (CAR) T-cell therapy appears to be a promising approach for Hodgkin lymphoma, according to Philippe Armand, MD, PhD, of Dana-Farber/Brigham and Women’s Cancer Center and the Massachusetts General Hospital Cancer Center in Boston.
While based on a handful of patients, the data do suggest this approach may play a role by targeting CD30 or Epstein Barr virus (EBV), Dr. Armand said at the NCCN Annual Congress: Hematologic Malignancies.
“Most importantly perhaps, like its experience outside of Hodgkin lymphoma, it may really have curative potential, based on the long [complete response] rates that have been already exhibited,” he said.
Much of the published clinical experience to date is with CD30-directed CAR Ts, Dr. Armand said, noting that in Hodgkin lymphoma, results so far show promise for this particular approach.
In a recent phase 1 dose escalation study, nine patients with relapsed/refractory Hodgkin lymphoma or anaplastic large-cell lymphoma (ALCL) received infusions of autologous T cells modified to express CD30-specific CAR T cells encoding the CD28 costimulatory domain, with no conditioning regimen.
Out of seven relapsed Hodgkin lymphoma patients, one had a complete response (CR) lasting beyond 2.5 years following a second infusion. Another patient had a CR persisting almost 2 years, and three patients had transient stable disease. One of the two ALCL patients had a CR lasting 9 months after a fourth infusion.
No toxicities attributable to the therapy were seen, according to the investigators.
The CD30 CAR T cells are being evaluated with a conditioning regimen in the phase 1 RELY-30 trial. Preliminary results presented at the 2018 European Society for Blood and Marrow Transplantation meeting in Lisbon showed better expansion of CAR T cells and responses in three out of five patients, including two CRs, according to Dr. Armand.
A CD30-directed CAR T-cell therapy with a 4-1BB costimulatory domain has also been tested in a small group of Hodgkin patients with a response rate of 35% – including some CRs – with an apparent lower response rate in patients with extranodal involvement. Dr. Armand noted that those findings need to be validated in additional studies.
Among non CD-30 targeted products, a CD19 CAR-T approach has been tried in Hodgkin lymphoma, though preliminary results suggest only transient activity, according to the presenter.
One interesting approach has been the targeting of EBV, he added. Recently reported results showed that two doses of T cells with specificity for EBV-derived tumor antigens induced clinical responses in patients with EBV-positive Hodgkin lymphoma.
The cells were engineered to express dominant-negative TGF-beta receptor type 2, according to the report. “We know that TGF-beta provides a strong immunosuppressant signal in the tumor microenvironment,” Dr. Armand said, noting that some of the responses in the seven evaluable patients lasted 4 years or more.
Dr. Armand reported financial disclosures related to Adaptive Biotechnologies/Sequenta, Affimed, Bristol-Myers Squibb, Merck, and Roche.
NEW YORK – Although the data set is small and not yet mature, chimeric antigen receptor (CAR) T-cell therapy appears to be a promising approach for Hodgkin lymphoma, according to Philippe Armand, MD, PhD, of Dana-Farber/Brigham and Women’s Cancer Center and the Massachusetts General Hospital Cancer Center in Boston.
While based on a handful of patients, the data do suggest this approach may play a role by targeting CD30 or Epstein Barr virus (EBV), Dr. Armand said at the NCCN Annual Congress: Hematologic Malignancies.
“Most importantly perhaps, like its experience outside of Hodgkin lymphoma, it may really have curative potential, based on the long [complete response] rates that have been already exhibited,” he said.
Much of the published clinical experience to date is with CD30-directed CAR Ts, Dr. Armand said, noting that in Hodgkin lymphoma, results so far show promise for this particular approach.
In a recent phase 1 dose escalation study, nine patients with relapsed/refractory Hodgkin lymphoma or anaplastic large-cell lymphoma (ALCL) received infusions of autologous T cells modified to express CD30-specific CAR T cells encoding the CD28 costimulatory domain, with no conditioning regimen.
Out of seven relapsed Hodgkin lymphoma patients, one had a complete response (CR) lasting beyond 2.5 years following a second infusion. Another patient had a CR persisting almost 2 years, and three patients had transient stable disease. One of the two ALCL patients had a CR lasting 9 months after a fourth infusion.
No toxicities attributable to the therapy were seen, according to the investigators.
The CD30 CAR T cells are being evaluated with a conditioning regimen in the phase 1 RELY-30 trial. Preliminary results presented at the 2018 European Society for Blood and Marrow Transplantation meeting in Lisbon showed better expansion of CAR T cells and responses in three out of five patients, including two CRs, according to Dr. Armand.
A CD30-directed CAR T-cell therapy with a 4-1BB costimulatory domain has also been tested in a small group of Hodgkin patients with a response rate of 35% – including some CRs – with an apparent lower response rate in patients with extranodal involvement. Dr. Armand noted that those findings need to be validated in additional studies.
Among non CD-30 targeted products, a CD19 CAR-T approach has been tried in Hodgkin lymphoma, though preliminary results suggest only transient activity, according to the presenter.
One interesting approach has been the targeting of EBV, he added. Recently reported results showed that two doses of T cells with specificity for EBV-derived tumor antigens induced clinical responses in patients with EBV-positive Hodgkin lymphoma.
The cells were engineered to express dominant-negative TGF-beta receptor type 2, according to the report. “We know that TGF-beta provides a strong immunosuppressant signal in the tumor microenvironment,” Dr. Armand said, noting that some of the responses in the seven evaluable patients lasted 4 years or more.
Dr. Armand reported financial disclosures related to Adaptive Biotechnologies/Sequenta, Affimed, Bristol-Myers Squibb, Merck, and Roche.
EXPERT ANALYSIS FROM NCCN ANNUAL CONGRESS: HEMATOLOGIC MALIGNANCIES