User login
2025 Crohn’s & Colitis Congress® Abstract Submissions
The 2025 Crohn’s & Colitis Congress®, a partnership of the Crohn’s & Colitis Foundation and AGA, is now accepting original inflammatory bowel disease (IBD)-research abstract submissions through Oct. 16. Abstracts are free to submit and may be selected for in-person lectures or poster presentations. Accepted abstracts will also be co-published in AGA’s Gastroenterology (https://www.gastrojournal.org/) and the Crohn’s & Colitis Foundation’s Inflammatory Bowel Diseases (https://academic.oup.com/ibdjournal).
Be sure to review the abstract submission guidelines and submit by 9 p.m. EDT, Wednesday, Oct. 16.
Presenting authors will receive notification of acceptance on Monday, Dec. 9.
The Crohn’s & Colitis Congress will take place Feb. 6-8, 2025, in San Francisco, California. It brings together the community of multidisciplinary experts and colleagues to revolutionize prevention, care and outcomes for IBD patients.
Learn alongside your colleagues and discover how to provide the absolute best care to those suffering with Crohn’s disease and ulcerative colitis.
The 2025 Crohn’s & Colitis Congress®, a partnership of the Crohn’s & Colitis Foundation and AGA, is now accepting original inflammatory bowel disease (IBD)-research abstract submissions through Oct. 16. Abstracts are free to submit and may be selected for in-person lectures or poster presentations. Accepted abstracts will also be co-published in AGA’s Gastroenterology (https://www.gastrojournal.org/) and the Crohn’s & Colitis Foundation’s Inflammatory Bowel Diseases (https://academic.oup.com/ibdjournal).
Be sure to review the abstract submission guidelines and submit by 9 p.m. EDT, Wednesday, Oct. 16.
Presenting authors will receive notification of acceptance on Monday, Dec. 9.
The Crohn’s & Colitis Congress will take place Feb. 6-8, 2025, in San Francisco, California. It brings together the community of multidisciplinary experts and colleagues to revolutionize prevention, care and outcomes for IBD patients.
Learn alongside your colleagues and discover how to provide the absolute best care to those suffering with Crohn’s disease and ulcerative colitis.
The 2025 Crohn’s & Colitis Congress®, a partnership of the Crohn’s & Colitis Foundation and AGA, is now accepting original inflammatory bowel disease (IBD)-research abstract submissions through Oct. 16. Abstracts are free to submit and may be selected for in-person lectures or poster presentations. Accepted abstracts will also be co-published in AGA’s Gastroenterology (https://www.gastrojournal.org/) and the Crohn’s & Colitis Foundation’s Inflammatory Bowel Diseases (https://academic.oup.com/ibdjournal).
Be sure to review the abstract submission guidelines and submit by 9 p.m. EDT, Wednesday, Oct. 16.
Presenting authors will receive notification of acceptance on Monday, Dec. 9.
The Crohn’s & Colitis Congress will take place Feb. 6-8, 2025, in San Francisco, California. It brings together the community of multidisciplinary experts and colleagues to revolutionize prevention, care and outcomes for IBD patients.
Learn alongside your colleagues and discover how to provide the absolute best care to those suffering with Crohn’s disease and ulcerative colitis.
Meet Our 10 Editorial Fellows
We are excited to announce the 2024-25 participants, who will gain hands-on experience and mentorship working closely with the editors and staff at the AGA journals over the next year.
The 10 editorial fellows (2 per journal) will learn about the entire editorial process, from manuscript submission to peer review to acceptance. They will participate in discussions and conferences with the boards of editors, assist with manuscript review, and help disseminate articles via their social media platforms.
Clinical Gastroenterology and Hepatology
Robyn Jordan, MD, MPH
Johns Hopkins Hospital, Baltimore | USA
Daryl Ramai, MD, MPH, MSc
Brigham and Women’s Hospital, Boston | USA
Cellular and Molecular Gastroenterology and Hepatology
Kole H. Buckley, PhD
University of Pennsylvania Perelman School of Medicine, Philadelphia | USA
Lin Y. Hung, PhD
New York University | USA
Gastroenterology
Corey J. Ketchem, MD
University of Pennsylvania, Philadelphia | USA
Rishad Khan, MD
University of Toronto | Canada
Gastro Hep Advances
Sasha Kapil, MD
UT Southwestern Medical Center, Dallas | USA
June Tome, MD
Mayo Clinic, Rochester, Minnesota | USA
Techniques and Innovations in Gastrointestinal Endoscopy
Thomas Enke, MD
University of Colorado, Aurora | USA
Sami Elamin, MD
Harvard, Beth Israel Deaconess Medical Center, Boston | USA
We are excited to announce the 2024-25 participants, who will gain hands-on experience and mentorship working closely with the editors and staff at the AGA journals over the next year.
The 10 editorial fellows (2 per journal) will learn about the entire editorial process, from manuscript submission to peer review to acceptance. They will participate in discussions and conferences with the boards of editors, assist with manuscript review, and help disseminate articles via their social media platforms.
Clinical Gastroenterology and Hepatology
Robyn Jordan, MD, MPH
Johns Hopkins Hospital, Baltimore | USA
Daryl Ramai, MD, MPH, MSc
Brigham and Women’s Hospital, Boston | USA
Cellular and Molecular Gastroenterology and Hepatology
Kole H. Buckley, PhD
University of Pennsylvania Perelman School of Medicine, Philadelphia | USA
Lin Y. Hung, PhD
New York University | USA
Gastroenterology
Corey J. Ketchem, MD
University of Pennsylvania, Philadelphia | USA
Rishad Khan, MD
University of Toronto | Canada
Gastro Hep Advances
Sasha Kapil, MD
UT Southwestern Medical Center, Dallas | USA
June Tome, MD
Mayo Clinic, Rochester, Minnesota | USA
Techniques and Innovations in Gastrointestinal Endoscopy
Thomas Enke, MD
University of Colorado, Aurora | USA
Sami Elamin, MD
Harvard, Beth Israel Deaconess Medical Center, Boston | USA
We are excited to announce the 2024-25 participants, who will gain hands-on experience and mentorship working closely with the editors and staff at the AGA journals over the next year.
The 10 editorial fellows (2 per journal) will learn about the entire editorial process, from manuscript submission to peer review to acceptance. They will participate in discussions and conferences with the boards of editors, assist with manuscript review, and help disseminate articles via their social media platforms.
Clinical Gastroenterology and Hepatology
Robyn Jordan, MD, MPH
Johns Hopkins Hospital, Baltimore | USA
Daryl Ramai, MD, MPH, MSc
Brigham and Women’s Hospital, Boston | USA
Cellular and Molecular Gastroenterology and Hepatology
Kole H. Buckley, PhD
University of Pennsylvania Perelman School of Medicine, Philadelphia | USA
Lin Y. Hung, PhD
New York University | USA
Gastroenterology
Corey J. Ketchem, MD
University of Pennsylvania, Philadelphia | USA
Rishad Khan, MD
University of Toronto | Canada
Gastro Hep Advances
Sasha Kapil, MD
UT Southwestern Medical Center, Dallas | USA
June Tome, MD
Mayo Clinic, Rochester, Minnesota | USA
Techniques and Innovations in Gastrointestinal Endoscopy
Thomas Enke, MD
University of Colorado, Aurora | USA
Sami Elamin, MD
Harvard, Beth Israel Deaconess Medical Center, Boston | USA
Creating a Planned Gift That’s Meaningful To You
The AGA Research Foundation has helped make significant strides in advancing the treatment and cure of digestive diseases by funding talented investigators.
Planning your gift to benefit AGA Research Foundation in the future is an opportunity to express what matters to you. As an AGA member, you can work with the AGA Research Foundation to ensure that your planned gift is designated for a purpose that meets your goals for leaving a legacy—such as research awards, support for specific programs, or unrestricted gifts to help meet the Foundation’s mission.
In as little as one sentence in your will and/or trust, you can complete your gift: “I give to AGA Research Foundation, a nonprofit corporation currently located at 4930 Del Ray Avenue, Bethesda, MD 20814, or its successor thereto, _________ [written amount or percentage of the estate or description of property] for its unrestricted charitable use and purpose.”
If you have named the AGA Research Foundation in your will or trust, please let us know so we can ensure that your gift is used according to your wishes. Notifying us of your plans will enable us to plan for the use of your future gift. However, if you prefer to remain anonymous, we will keep your name and gift in strict confidence.
Please contact foundation@gastro.org for more information. If you are considering a planned gift, consult with your own legal and tax advisors.
The AGA Research Foundation has helped make significant strides in advancing the treatment and cure of digestive diseases by funding talented investigators.
Planning your gift to benefit AGA Research Foundation in the future is an opportunity to express what matters to you. As an AGA member, you can work with the AGA Research Foundation to ensure that your planned gift is designated for a purpose that meets your goals for leaving a legacy—such as research awards, support for specific programs, or unrestricted gifts to help meet the Foundation’s mission.
In as little as one sentence in your will and/or trust, you can complete your gift: “I give to AGA Research Foundation, a nonprofit corporation currently located at 4930 Del Ray Avenue, Bethesda, MD 20814, or its successor thereto, _________ [written amount or percentage of the estate or description of property] for its unrestricted charitable use and purpose.”
If you have named the AGA Research Foundation in your will or trust, please let us know so we can ensure that your gift is used according to your wishes. Notifying us of your plans will enable us to plan for the use of your future gift. However, if you prefer to remain anonymous, we will keep your name and gift in strict confidence.
Please contact foundation@gastro.org for more information. If you are considering a planned gift, consult with your own legal and tax advisors.
The AGA Research Foundation has helped make significant strides in advancing the treatment and cure of digestive diseases by funding talented investigators.
Planning your gift to benefit AGA Research Foundation in the future is an opportunity to express what matters to you. As an AGA member, you can work with the AGA Research Foundation to ensure that your planned gift is designated for a purpose that meets your goals for leaving a legacy—such as research awards, support for specific programs, or unrestricted gifts to help meet the Foundation’s mission.
In as little as one sentence in your will and/or trust, you can complete your gift: “I give to AGA Research Foundation, a nonprofit corporation currently located at 4930 Del Ray Avenue, Bethesda, MD 20814, or its successor thereto, _________ [written amount or percentage of the estate or description of property] for its unrestricted charitable use and purpose.”
If you have named the AGA Research Foundation in your will or trust, please let us know so we can ensure that your gift is used according to your wishes. Notifying us of your plans will enable us to plan for the use of your future gift. However, if you prefer to remain anonymous, we will keep your name and gift in strict confidence.
Please contact foundation@gastro.org for more information. If you are considering a planned gift, consult with your own legal and tax advisors.
Support Researchers with a Donation to the AGA Research Foundation
By joining others in donating to the AGA Research Foundation, you will ensure that researchers have opportunities to continue their lifesaving work.
The AGA Research Foundation remains committed to providing young researchers with unprecedented research opportunities. Each year, we receive a caliber of nominations for AGA research awards. You can help gifted investigators as they work to advance the understanding of digestive diseases through their novel research objectives.
As an AGA member, you can help fund discoveries that will continue to improve GI practice and better patient care.
AGA grants have led to discoveries, including new approaches to down-regulate intestinal inflammation, a test for genetic predisposition to colon cancer and autoimmune liver disease treatments. The importance of these awards is evidenced by the fact that virtually every major advance leading to the understanding, prevention, treatment, and cure of digestive diseases has been made in the research laboratory of a talented young investigator.
Donate to the AGA Research Foundation to ensure that researchers have opportunities to continue their lifesaving work.
Three Easy Ways To Give
Online: Donate at www.foundation.gastro.org.
Through the mail:
AGA Research Foundation
4930 Del Ray Avenue
Bethesda, MD 20814
Over the phone: 301-222-4002
All gifts are tax-deductible to the fullest extent of U.S. law.
By joining others in donating to the AGA Research Foundation, you will ensure that researchers have opportunities to continue their lifesaving work.
The AGA Research Foundation remains committed to providing young researchers with unprecedented research opportunities. Each year, we receive a caliber of nominations for AGA research awards. You can help gifted investigators as they work to advance the understanding of digestive diseases through their novel research objectives.
As an AGA member, you can help fund discoveries that will continue to improve GI practice and better patient care.
AGA grants have led to discoveries, including new approaches to down-regulate intestinal inflammation, a test for genetic predisposition to colon cancer and autoimmune liver disease treatments. The importance of these awards is evidenced by the fact that virtually every major advance leading to the understanding, prevention, treatment, and cure of digestive diseases has been made in the research laboratory of a talented young investigator.
Donate to the AGA Research Foundation to ensure that researchers have opportunities to continue their lifesaving work.
Three Easy Ways To Give
Online: Donate at www.foundation.gastro.org.
Through the mail:
AGA Research Foundation
4930 Del Ray Avenue
Bethesda, MD 20814
Over the phone: 301-222-4002
All gifts are tax-deductible to the fullest extent of U.S. law.
By joining others in donating to the AGA Research Foundation, you will ensure that researchers have opportunities to continue their lifesaving work.
The AGA Research Foundation remains committed to providing young researchers with unprecedented research opportunities. Each year, we receive a caliber of nominations for AGA research awards. You can help gifted investigators as they work to advance the understanding of digestive diseases through their novel research objectives.
As an AGA member, you can help fund discoveries that will continue to improve GI practice and better patient care.
AGA grants have led to discoveries, including new approaches to down-regulate intestinal inflammation, a test for genetic predisposition to colon cancer and autoimmune liver disease treatments. The importance of these awards is evidenced by the fact that virtually every major advance leading to the understanding, prevention, treatment, and cure of digestive diseases has been made in the research laboratory of a talented young investigator.
Donate to the AGA Research Foundation to ensure that researchers have opportunities to continue their lifesaving work.
Three Easy Ways To Give
Online: Donate at www.foundation.gastro.org.
Through the mail:
AGA Research Foundation
4930 Del Ray Avenue
Bethesda, MD 20814
Over the phone: 301-222-4002
All gifts are tax-deductible to the fullest extent of U.S. law.
FDA Approves First Blood Test for Colorectal Cancer
In late July, the US Food and Drug Administration (FDA) approved the first use of a liquid biopsy (blood test) for colorectal cancer (CRC) screening. The test, called Shield, launched commercially the first week of August and is the first blood test to be approved by the FDA as a primary screening option for CRC that meets requirements for Medicare reimbursement.
While the convenience of a blood test could potentially encourage more people to get screened, expert consensus is that blood tests can’t prevent CRC and should not be considered a replacement for a colonoscopy. Modeling studies and expert consensus published earlier this year in Gastroenterology and in Clinical Gastroenterology and Hepatology shed light on the perils of liquid biopsy.
“Based on their current characteristics, blood tests should not be recommended to replace established colorectal cancer screening tests, since blood tests are neither as effective nor as cost-effective, and would worsen outcomes,” said David Lieberman, MD, AGAF, chair, AGA CRC Workshop chair and lead author of an expert commentary on liquid biopsy for CRC screening.
Five Key Takeaways
- A blood test for CRC that meets minimal CMS criteria for sensitivity and performed every 3 years would likely result in better outcomes than no screening.
- A blood test for CRC offers a simple process that could encourage more people to participate in screening. Patients who may have declined colonoscopy should understand the need for a colonoscopy if findings are abnormal.
- Because blood tests for CRC are predicted to be less effective and more costly than currently established screening programs, they cannot be recommended to replace established effective screening methods.
- Although blood tests would improve outcomes in currently unscreened people, substituting blood tests for a currently effective test would worsen patient outcomes and increase cost.
- Potential benchmarks that industry might use to assess an effective blood test for CRC going forward would be sensitivity for stage I-III CRC of > 90%, with sensitivity for advanced adenomas of > 40%-50%.
“Unless we have the expectation of high sensitivity and specificity, blood-based colorectal cancer tests could lead to false positive and false negative results, which are both bad for patient outcomes,” said John M. Carethers, MD, AGAF, AGA past president and vice chancellor for health sciences at the University of California San Diego.
In late July, the US Food and Drug Administration (FDA) approved the first use of a liquid biopsy (blood test) for colorectal cancer (CRC) screening. The test, called Shield, launched commercially the first week of August and is the first blood test to be approved by the FDA as a primary screening option for CRC that meets requirements for Medicare reimbursement.
While the convenience of a blood test could potentially encourage more people to get screened, expert consensus is that blood tests can’t prevent CRC and should not be considered a replacement for a colonoscopy. Modeling studies and expert consensus published earlier this year in Gastroenterology and in Clinical Gastroenterology and Hepatology shed light on the perils of liquid biopsy.
“Based on their current characteristics, blood tests should not be recommended to replace established colorectal cancer screening tests, since blood tests are neither as effective nor as cost-effective, and would worsen outcomes,” said David Lieberman, MD, AGAF, chair, AGA CRC Workshop chair and lead author of an expert commentary on liquid biopsy for CRC screening.
Five Key Takeaways
- A blood test for CRC that meets minimal CMS criteria for sensitivity and performed every 3 years would likely result in better outcomes than no screening.
- A blood test for CRC offers a simple process that could encourage more people to participate in screening. Patients who may have declined colonoscopy should understand the need for a colonoscopy if findings are abnormal.
- Because blood tests for CRC are predicted to be less effective and more costly than currently established screening programs, they cannot be recommended to replace established effective screening methods.
- Although blood tests would improve outcomes in currently unscreened people, substituting blood tests for a currently effective test would worsen patient outcomes and increase cost.
- Potential benchmarks that industry might use to assess an effective blood test for CRC going forward would be sensitivity for stage I-III CRC of > 90%, with sensitivity for advanced adenomas of > 40%-50%.
“Unless we have the expectation of high sensitivity and specificity, blood-based colorectal cancer tests could lead to false positive and false negative results, which are both bad for patient outcomes,” said John M. Carethers, MD, AGAF, AGA past president and vice chancellor for health sciences at the University of California San Diego.
In late July, the US Food and Drug Administration (FDA) approved the first use of a liquid biopsy (blood test) for colorectal cancer (CRC) screening. The test, called Shield, launched commercially the first week of August and is the first blood test to be approved by the FDA as a primary screening option for CRC that meets requirements for Medicare reimbursement.
While the convenience of a blood test could potentially encourage more people to get screened, expert consensus is that blood tests can’t prevent CRC and should not be considered a replacement for a colonoscopy. Modeling studies and expert consensus published earlier this year in Gastroenterology and in Clinical Gastroenterology and Hepatology shed light on the perils of liquid biopsy.
“Based on their current characteristics, blood tests should not be recommended to replace established colorectal cancer screening tests, since blood tests are neither as effective nor as cost-effective, and would worsen outcomes,” said David Lieberman, MD, AGAF, chair, AGA CRC Workshop chair and lead author of an expert commentary on liquid biopsy for CRC screening.
Five Key Takeaways
- A blood test for CRC that meets minimal CMS criteria for sensitivity and performed every 3 years would likely result in better outcomes than no screening.
- A blood test for CRC offers a simple process that could encourage more people to participate in screening. Patients who may have declined colonoscopy should understand the need for a colonoscopy if findings are abnormal.
- Because blood tests for CRC are predicted to be less effective and more costly than currently established screening programs, they cannot be recommended to replace established effective screening methods.
- Although blood tests would improve outcomes in currently unscreened people, substituting blood tests for a currently effective test would worsen patient outcomes and increase cost.
- Potential benchmarks that industry might use to assess an effective blood test for CRC going forward would be sensitivity for stage I-III CRC of > 90%, with sensitivity for advanced adenomas of > 40%-50%.
“Unless we have the expectation of high sensitivity and specificity, blood-based colorectal cancer tests could lead to false positive and false negative results, which are both bad for patient outcomes,” said John M. Carethers, MD, AGAF, AGA past president and vice chancellor for health sciences at the University of California San Diego.
Check Out Our New Irritable Bowel Syndrome Clinician Toolkit
AGA’s new irritable bowel syndrome toolkit gathers all our clinical guidance, continuing education materials, patient education resources, and FAQs in one convenient place. Be sure to check it out and bookmark it for easy access!
Curious about our other toolkits? Visit gastro.org/clinical-guidance to explore our toolkits on ulcerative colitis and Crohn’s disease. Keep an eye out for more coming soon!
The toolkit includes clinical guidance on:
- Pharmacological management of irritable bowel syndrome with diarrhea (IBS-D)
- Pharmacological management of irritable bowel syndrome with constipation (IBS-C)
For more resources for ulcerative colitis patients, visit the Patient Center on the AGA website. The AGA Patient Center has a variety of information that can be shared with your patients, including tips on diet, vaccine recommendations, and information on biosimilars.
AGA’s new irritable bowel syndrome toolkit gathers all our clinical guidance, continuing education materials, patient education resources, and FAQs in one convenient place. Be sure to check it out and bookmark it for easy access!
Curious about our other toolkits? Visit gastro.org/clinical-guidance to explore our toolkits on ulcerative colitis and Crohn’s disease. Keep an eye out for more coming soon!
The toolkit includes clinical guidance on:
- Pharmacological management of irritable bowel syndrome with diarrhea (IBS-D)
- Pharmacological management of irritable bowel syndrome with constipation (IBS-C)
For more resources for ulcerative colitis patients, visit the Patient Center on the AGA website. The AGA Patient Center has a variety of information that can be shared with your patients, including tips on diet, vaccine recommendations, and information on biosimilars.
AGA’s new irritable bowel syndrome toolkit gathers all our clinical guidance, continuing education materials, patient education resources, and FAQs in one convenient place. Be sure to check it out and bookmark it for easy access!
Curious about our other toolkits? Visit gastro.org/clinical-guidance to explore our toolkits on ulcerative colitis and Crohn’s disease. Keep an eye out for more coming soon!
The toolkit includes clinical guidance on:
- Pharmacological management of irritable bowel syndrome with diarrhea (IBS-D)
- Pharmacological management of irritable bowel syndrome with constipation (IBS-C)
For more resources for ulcerative colitis patients, visit the Patient Center on the AGA website. The AGA Patient Center has a variety of information that can be shared with your patients, including tips on diet, vaccine recommendations, and information on biosimilars.
Elevate Your Career: AGA Women in GI Regional Workshops Await
As a woman in a dynamic and ever-changing profession, balancing life as a powerhouse physician or scientist is no easy feat. AGA recognizes the challenges you face and is committed to addressing them directly at the AGA Women in GI Regional Workshops. The program has been expanded to six workshops in 2024.
. Participate in candid discussions regarding the challenges you face as a woman navigating the 21st century healthcare environment. Derive inspiration from your community and cultivate meaningful connections that will carry you beyond the workshop.
You may choose to join us in person or virtually, whatever fits into your busy schedule. We are also pleased to offer grants of $300 to support travel and registration fees for trainee and early career women. Additional details for the Maria Leo-Lieber Travel Award may be found in your confirmation email.
Register today for the final three workshops.
Rocky Mountain West
Saturday, Sept. 8
Colorado Springs, Colorado
Deadline to apply for a travel grant: Aug. 23
Deadline to register: Aug. 30
Southwest
Saturday, Sept. 14
Houston, Texas
Deadline to apply for a travel grant: Aug. 30
Deadline to register: Sept. 6
Southeast
Saturday, Nov. 2
Coral Gables, Florida
Deadline to apply for a travel grant: Oct. 8
Deadline to register: Oct. 25
This program is supported by Janssen.
As a woman in a dynamic and ever-changing profession, balancing life as a powerhouse physician or scientist is no easy feat. AGA recognizes the challenges you face and is committed to addressing them directly at the AGA Women in GI Regional Workshops. The program has been expanded to six workshops in 2024.
. Participate in candid discussions regarding the challenges you face as a woman navigating the 21st century healthcare environment. Derive inspiration from your community and cultivate meaningful connections that will carry you beyond the workshop.
You may choose to join us in person or virtually, whatever fits into your busy schedule. We are also pleased to offer grants of $300 to support travel and registration fees for trainee and early career women. Additional details for the Maria Leo-Lieber Travel Award may be found in your confirmation email.
Register today for the final three workshops.
Rocky Mountain West
Saturday, Sept. 8
Colorado Springs, Colorado
Deadline to apply for a travel grant: Aug. 23
Deadline to register: Aug. 30
Southwest
Saturday, Sept. 14
Houston, Texas
Deadline to apply for a travel grant: Aug. 30
Deadline to register: Sept. 6
Southeast
Saturday, Nov. 2
Coral Gables, Florida
Deadline to apply for a travel grant: Oct. 8
Deadline to register: Oct. 25
This program is supported by Janssen.
As a woman in a dynamic and ever-changing profession, balancing life as a powerhouse physician or scientist is no easy feat. AGA recognizes the challenges you face and is committed to addressing them directly at the AGA Women in GI Regional Workshops. The program has been expanded to six workshops in 2024.
. Participate in candid discussions regarding the challenges you face as a woman navigating the 21st century healthcare environment. Derive inspiration from your community and cultivate meaningful connections that will carry you beyond the workshop.
You may choose to join us in person or virtually, whatever fits into your busy schedule. We are also pleased to offer grants of $300 to support travel and registration fees for trainee and early career women. Additional details for the Maria Leo-Lieber Travel Award may be found in your confirmation email.
Register today for the final three workshops.
Rocky Mountain West
Saturday, Sept. 8
Colorado Springs, Colorado
Deadline to apply for a travel grant: Aug. 23
Deadline to register: Aug. 30
Southwest
Saturday, Sept. 14
Houston, Texas
Deadline to apply for a travel grant: Aug. 30
Deadline to register: Sept. 6
Southeast
Saturday, Nov. 2
Coral Gables, Florida
Deadline to apply for a travel grant: Oct. 8
Deadline to register: Oct. 25
This program is supported by Janssen.
How To Navigate Your First Job
In a special episode live from Digestive Disease Week® (DDW) 2024, host Dr. Matthew Whitson talks with returning guest Dr. Janice Jou. Dr. Jou is a transplant hematologist at the Portland VA and currently serves as professor of medicine and fellowship program director at Oregon Health & Science University. Don’t miss her insight as she shares advice all about what she wishes she knew when going into her first job in gastroenterology. Dr. Jou also answers questions from the audience on topics including “when to say no” and the importance of encouraging emotional transparency with fellows and faculty.
Catch up with past episodes and subscribe wherever you listen to podcasts. You can also listen by clicking on the episode name below.
- Episode 5: Janice Jou: Live from #DDW2024 with tips for your first job
- Episode 4: Loren Rabinowitz and Rachel Issaka: Building research collaborations
- Episode 3: Andy Tau: How to treat GI emergencies
- Episode 2: Laurel Fisher and Asma Khapra: Advancing and advocating for women in GI
- Episode 1: Barbara Jung: Unpacking mentorship with AGA’s president
In a special episode live from Digestive Disease Week® (DDW) 2024, host Dr. Matthew Whitson talks with returning guest Dr. Janice Jou. Dr. Jou is a transplant hematologist at the Portland VA and currently serves as professor of medicine and fellowship program director at Oregon Health & Science University. Don’t miss her insight as she shares advice all about what she wishes she knew when going into her first job in gastroenterology. Dr. Jou also answers questions from the audience on topics including “when to say no” and the importance of encouraging emotional transparency with fellows and faculty.
Catch up with past episodes and subscribe wherever you listen to podcasts. You can also listen by clicking on the episode name below.
- Episode 5: Janice Jou: Live from #DDW2024 with tips for your first job
- Episode 4: Loren Rabinowitz and Rachel Issaka: Building research collaborations
- Episode 3: Andy Tau: How to treat GI emergencies
- Episode 2: Laurel Fisher and Asma Khapra: Advancing and advocating for women in GI
- Episode 1: Barbara Jung: Unpacking mentorship with AGA’s president
In a special episode live from Digestive Disease Week® (DDW) 2024, host Dr. Matthew Whitson talks with returning guest Dr. Janice Jou. Dr. Jou is a transplant hematologist at the Portland VA and currently serves as professor of medicine and fellowship program director at Oregon Health & Science University. Don’t miss her insight as she shares advice all about what she wishes she knew when going into her first job in gastroenterology. Dr. Jou also answers questions from the audience on topics including “when to say no” and the importance of encouraging emotional transparency with fellows and faculty.
Catch up with past episodes and subscribe wherever you listen to podcasts. You can also listen by clicking on the episode name below.
- Episode 5: Janice Jou: Live from #DDW2024 with tips for your first job
- Episode 4: Loren Rabinowitz and Rachel Issaka: Building research collaborations
- Episode 3: Andy Tau: How to treat GI emergencies
- Episode 2: Laurel Fisher and Asma Khapra: Advancing and advocating for women in GI
- Episode 1: Barbara Jung: Unpacking mentorship with AGA’s president
We’re Making Progress in the Fight Against GI Cancers
The House Appropriations Committee has included AGA-provided language on gastric and esophageal cancers in the FY25 Labor, Health, and Human Services report.
Gastric and esophageal cancers rates are rising and have a low 5-year survival rate and are highly fatal due to the lack of screening — despite both cancers typically being associated with reduced mortality. Delays in diagnosis lead to poor prognoses when the cancer is already at an advanced stage.
These cancers receive disproportionately low funding and have the lowest ratio of funding to lethality to any other cancer.
That’s why it’s crucial to close the gap and increase screening for GI cancers that are less commonly screened for.
AGA’s provided language encourages NIH to develop, test and implement screening strategies for gastric and esophageal cancers using non-endoscopic screening modalities, personalized clinical risk stratification for screenings and biomarker-based risk-stratification.
Why is this important?
This submission is the first time AGA language on gastric and esophageal cancer has been requested and included in the committee’s report. This illustrates the Committee’s recognition of the importance of GI cancer detection and the work being done by NIH.
What does this mean for GI?
This is an important first step to increasing access to cancer screenings! We look forward to working with our champions on Capitol Hill to increase patient access GI cancer screenings.
The House Appropriations Committee has included AGA-provided language on gastric and esophageal cancers in the FY25 Labor, Health, and Human Services report.
Gastric and esophageal cancers rates are rising and have a low 5-year survival rate and are highly fatal due to the lack of screening — despite both cancers typically being associated with reduced mortality. Delays in diagnosis lead to poor prognoses when the cancer is already at an advanced stage.
These cancers receive disproportionately low funding and have the lowest ratio of funding to lethality to any other cancer.
That’s why it’s crucial to close the gap and increase screening for GI cancers that are less commonly screened for.
AGA’s provided language encourages NIH to develop, test and implement screening strategies for gastric and esophageal cancers using non-endoscopic screening modalities, personalized clinical risk stratification for screenings and biomarker-based risk-stratification.
Why is this important?
This submission is the first time AGA language on gastric and esophageal cancer has been requested and included in the committee’s report. This illustrates the Committee’s recognition of the importance of GI cancer detection and the work being done by NIH.
What does this mean for GI?
This is an important first step to increasing access to cancer screenings! We look forward to working with our champions on Capitol Hill to increase patient access GI cancer screenings.
The House Appropriations Committee has included AGA-provided language on gastric and esophageal cancers in the FY25 Labor, Health, and Human Services report.
Gastric and esophageal cancers rates are rising and have a low 5-year survival rate and are highly fatal due to the lack of screening — despite both cancers typically being associated with reduced mortality. Delays in diagnosis lead to poor prognoses when the cancer is already at an advanced stage.
These cancers receive disproportionately low funding and have the lowest ratio of funding to lethality to any other cancer.
That’s why it’s crucial to close the gap and increase screening for GI cancers that are less commonly screened for.
AGA’s provided language encourages NIH to develop, test and implement screening strategies for gastric and esophageal cancers using non-endoscopic screening modalities, personalized clinical risk stratification for screenings and biomarker-based risk-stratification.
Why is this important?
This submission is the first time AGA language on gastric and esophageal cancer has been requested and included in the committee’s report. This illustrates the Committee’s recognition of the importance of GI cancer detection and the work being done by NIH.
What does this mean for GI?
This is an important first step to increasing access to cancer screenings! We look forward to working with our champions on Capitol Hill to increase patient access GI cancer screenings.
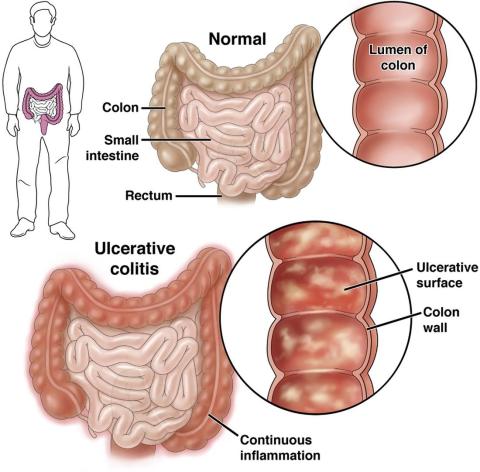
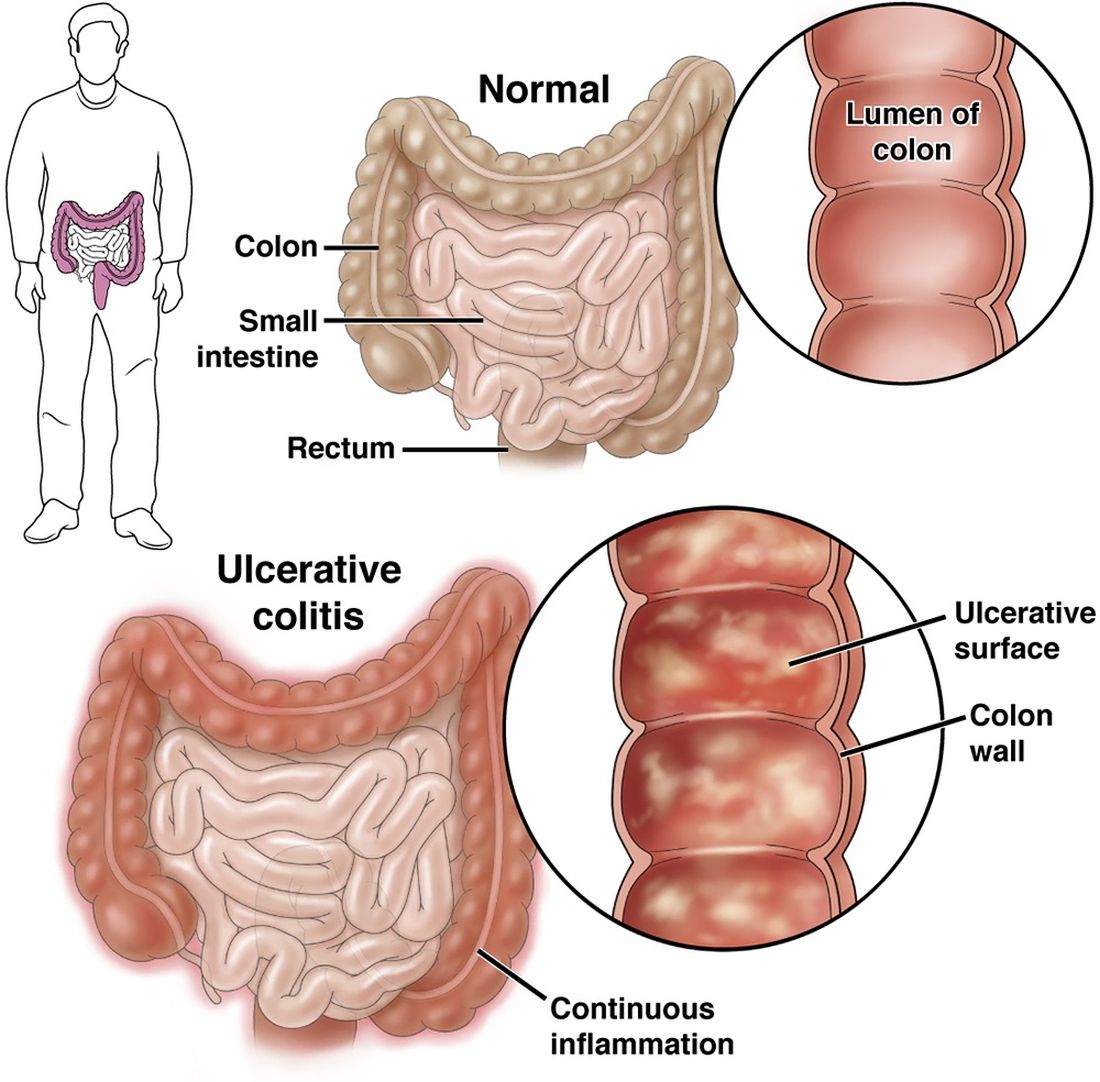
Check Out Our New Ulcerative Colitis Clinician Toolkit
Check out and bookmark AGA’s new ulcerative colitis toolkit, which compiles all our ulcerative colitis clinical guidance, continuing education resources, patient education, and FAQs into one convenient toolkit.
Curious about our other toolkits? Check out our toolkit on Crohn’s disease.
The new UC toolkit includes clinical guidance on:
- Role of biomarkers for the management of ulcerative colitis
- Medical management of moderate to severe ulcerative colitis
- Management of pouchitis and inflammatory pouch disorders
For more resources for ulcerative colitis patients, visit the Patient Center on the AGA website.
The AGA Patient Center has a variety of information that can be shared with your patients, including tips on diet, vaccine recommendations, and information on biosimilars.
Check out and bookmark AGA’s new ulcerative colitis toolkit, which compiles all our ulcerative colitis clinical guidance, continuing education resources, patient education, and FAQs into one convenient toolkit.
Curious about our other toolkits? Check out our toolkit on Crohn’s disease.
The new UC toolkit includes clinical guidance on:
- Role of biomarkers for the management of ulcerative colitis
- Medical management of moderate to severe ulcerative colitis
- Management of pouchitis and inflammatory pouch disorders
For more resources for ulcerative colitis patients, visit the Patient Center on the AGA website.
The AGA Patient Center has a variety of information that can be shared with your patients, including tips on diet, vaccine recommendations, and information on biosimilars.
Check out and bookmark AGA’s new ulcerative colitis toolkit, which compiles all our ulcerative colitis clinical guidance, continuing education resources, patient education, and FAQs into one convenient toolkit.
Curious about our other toolkits? Check out our toolkit on Crohn’s disease.
The new UC toolkit includes clinical guidance on:
- Role of biomarkers for the management of ulcerative colitis
- Medical management of moderate to severe ulcerative colitis
- Management of pouchitis and inflammatory pouch disorders
For more resources for ulcerative colitis patients, visit the Patient Center on the AGA website.
The AGA Patient Center has a variety of information that can be shared with your patients, including tips on diet, vaccine recommendations, and information on biosimilars.