User login
2024 Rare Neurological Disease Special Report
Editor’s Note
By Glenn S. Williams
In this year’s Rare Neurological Disease Special Report, we focus on rare neurological diseases that have new therapies that have been recently approved as well as conditions for which the treatment pipeline is robust.
A Note From NORD
By Pamela Gavin
Through NORD’s collaboration with Neurology Reviews, we share cutting-edge research and insights from leading medical experts, including specialists from the NORD Rare Disease Centers of Excellence network, about the latest advances in the treatment of rare neurological conditions.
Genetic Testing for ALS, Now a Standard, Creates a Path Toward Individualized Care
By Ted Bosworth
Overall, there is a sense of progress in ALS. The hope is that clinical research is reaching a tipping point where targeted treatments may offer hope to patients with ALS.
Myasthenia Gravis: Patient Choice, Cultural Change
By John Jesitus
Used appropriately, newer treatments for myasthenia gravis can provide dramatic results faster and more safely than broad immunosuppressants.
Promise for Disease-Modifying Therapies to Tame Huntington’s Disease
By Neil Osterweil
Much progress has been made in managing the symptoms of Huntington’s disease, but the real excitement lies in the development of disease-modifying drugs and genetic therapy.
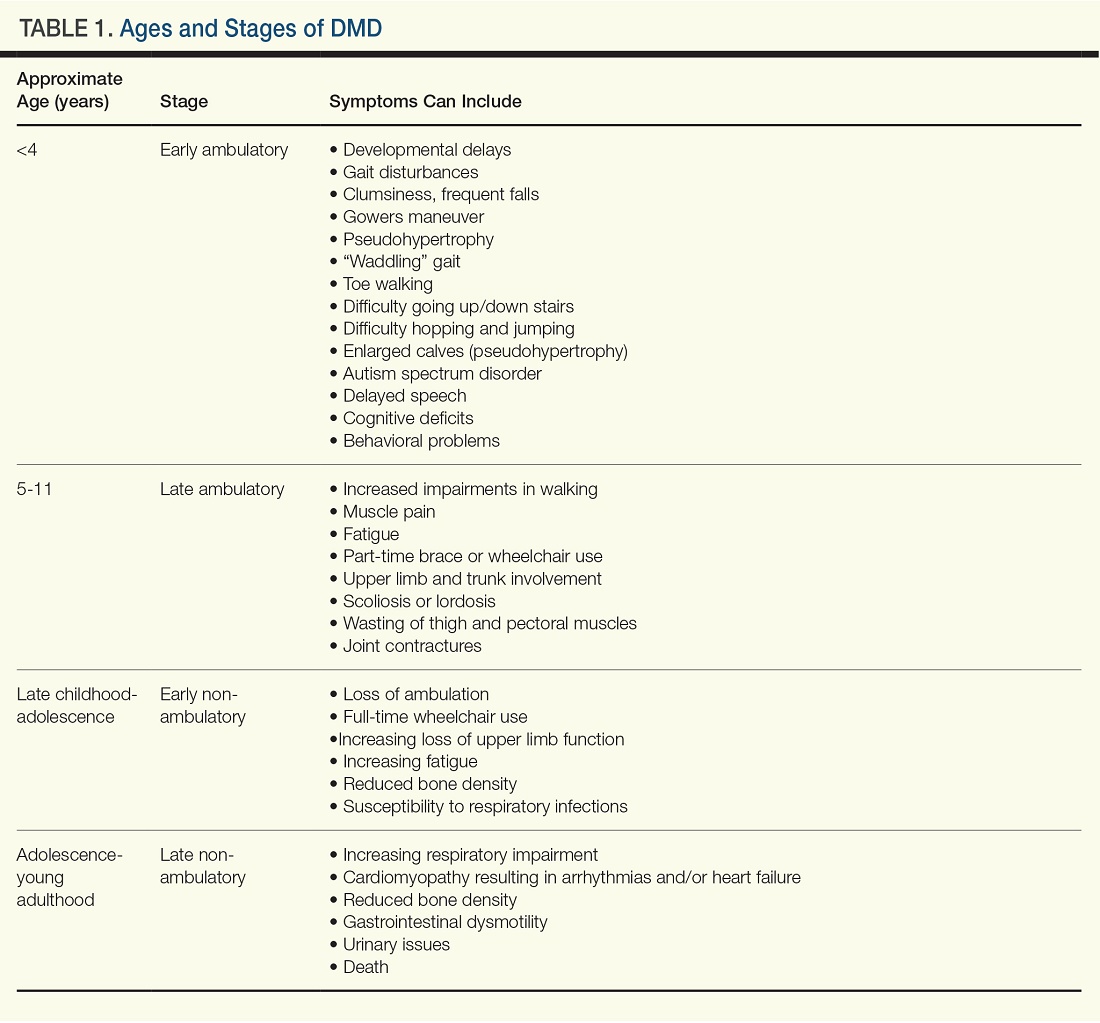
Diagnosing and Managing Duchenne Muscular Dystrophy: Tips for Practicing Clinicians
By Batya Swift Yasgur, MA, LSW
Healthcare providers should be familiar enough with Duchenne muscular dystrophy to provide timely diagnosis and early intervention as well as practical and emotional support to the patient and family/caregivers.
Neuromyelitis Optica: Historically Misdiagnosed — Now Demands Prompt Treatment
By Kate Johnson
Rapid diagnosis and treatment of NMO “means potentially preventing future devastating neurologic injury.”
Untangling CIDP
By Jennie Smith
Though a preferred biomarker remains elusive, this difficult-to-diagnose neuropathy has seen important recent advances in diagnosis and treatment.
Newborn Screening Programs: What Do Clinicians Need to Know?
By Batya Swift Yasgur, MA, LSW
The goal of newborn screening is to identify babies with genetic disorders who otherwise have no obvious symptoms.
Balancing Act: Weighing the Pros and Cons of Genetic Testing in Rare Diseases
By Frieda Wiley
While genetic testing may offer great potential for providing answers to patients and clinicians seeking insight into a rare disorder, the technology holds some pros and cons that neurologists should be aware of.
Editor’s Note
By Glenn S. Williams
In this year’s Rare Neurological Disease Special Report, we focus on rare neurological diseases that have new therapies that have been recently approved as well as conditions for which the treatment pipeline is robust.
A Note From NORD
By Pamela Gavin
Through NORD’s collaboration with Neurology Reviews, we share cutting-edge research and insights from leading medical experts, including specialists from the NORD Rare Disease Centers of Excellence network, about the latest advances in the treatment of rare neurological conditions.
Genetic Testing for ALS, Now a Standard, Creates a Path Toward Individualized Care
By Ted Bosworth
Overall, there is a sense of progress in ALS. The hope is that clinical research is reaching a tipping point where targeted treatments may offer hope to patients with ALS.
Myasthenia Gravis: Patient Choice, Cultural Change
By John Jesitus
Used appropriately, newer treatments for myasthenia gravis can provide dramatic results faster and more safely than broad immunosuppressants.
Promise for Disease-Modifying Therapies to Tame Huntington’s Disease
By Neil Osterweil
Much progress has been made in managing the symptoms of Huntington’s disease, but the real excitement lies in the development of disease-modifying drugs and genetic therapy.
Diagnosing and Managing Duchenne Muscular Dystrophy: Tips for Practicing Clinicians
By Batya Swift Yasgur, MA, LSW
Healthcare providers should be familiar enough with Duchenne muscular dystrophy to provide timely diagnosis and early intervention as well as practical and emotional support to the patient and family/caregivers.
Neuromyelitis Optica: Historically Misdiagnosed — Now Demands Prompt Treatment
By Kate Johnson
Rapid diagnosis and treatment of NMO “means potentially preventing future devastating neurologic injury.”
Untangling CIDP
By Jennie Smith
Though a preferred biomarker remains elusive, this difficult-to-diagnose neuropathy has seen important recent advances in diagnosis and treatment.
Newborn Screening Programs: What Do Clinicians Need to Know?
By Batya Swift Yasgur, MA, LSW
The goal of newborn screening is to identify babies with genetic disorders who otherwise have no obvious symptoms.
Balancing Act: Weighing the Pros and Cons of Genetic Testing in Rare Diseases
By Frieda Wiley
While genetic testing may offer great potential for providing answers to patients and clinicians seeking insight into a rare disorder, the technology holds some pros and cons that neurologists should be aware of.
Editor’s Note
By Glenn S. Williams
In this year’s Rare Neurological Disease Special Report, we focus on rare neurological diseases that have new therapies that have been recently approved as well as conditions for which the treatment pipeline is robust.
A Note From NORD
By Pamela Gavin
Through NORD’s collaboration with Neurology Reviews, we share cutting-edge research and insights from leading medical experts, including specialists from the NORD Rare Disease Centers of Excellence network, about the latest advances in the treatment of rare neurological conditions.
Genetic Testing for ALS, Now a Standard, Creates a Path Toward Individualized Care
By Ted Bosworth
Overall, there is a sense of progress in ALS. The hope is that clinical research is reaching a tipping point where targeted treatments may offer hope to patients with ALS.
Myasthenia Gravis: Patient Choice, Cultural Change
By John Jesitus
Used appropriately, newer treatments for myasthenia gravis can provide dramatic results faster and more safely than broad immunosuppressants.
Promise for Disease-Modifying Therapies to Tame Huntington’s Disease
By Neil Osterweil
Much progress has been made in managing the symptoms of Huntington’s disease, but the real excitement lies in the development of disease-modifying drugs and genetic therapy.
Diagnosing and Managing Duchenne Muscular Dystrophy: Tips for Practicing Clinicians
By Batya Swift Yasgur, MA, LSW
Healthcare providers should be familiar enough with Duchenne muscular dystrophy to provide timely diagnosis and early intervention as well as practical and emotional support to the patient and family/caregivers.
Neuromyelitis Optica: Historically Misdiagnosed — Now Demands Prompt Treatment
By Kate Johnson
Rapid diagnosis and treatment of NMO “means potentially preventing future devastating neurologic injury.”
Untangling CIDP
By Jennie Smith
Though a preferred biomarker remains elusive, this difficult-to-diagnose neuropathy has seen important recent advances in diagnosis and treatment.
Newborn Screening Programs: What Do Clinicians Need to Know?
By Batya Swift Yasgur, MA, LSW
The goal of newborn screening is to identify babies with genetic disorders who otherwise have no obvious symptoms.
Balancing Act: Weighing the Pros and Cons of Genetic Testing in Rare Diseases
By Frieda Wiley
While genetic testing may offer great potential for providing answers to patients and clinicians seeking insight into a rare disorder, the technology holds some pros and cons that neurologists should be aware of.
Editor's Note: 2024 Rare Neurological Disease Report
EDITOR’S NOTE
This year, we again focus on rare neurological diseases that have new therapies that have been recently approved as well as conditions for which the treatment pipeline is robust. Let’s hope the work of many dedicated researchers adds to the list of rare neurological diseases for which treatment is available.
This year also marks a change of leadership at NORD, our publishing partner in this annual supplement. We here at Neurology Reviews salute the leadership and accomplishments of former NORD CEO Peter Saltonstall and also welcome incoming CEO Pamela Gavin, who has spent many years in NORD leadership roles and was essential in the planning, launch, and early years of this annual supplement. I can think of no one better than Pamela Gavin to continue NORD’s mission into the future.
And finally, a recap of accolades for this annual supplement. For the second year in a row, the Rare Neurological Disease Special Report has won an Azbee award in the category of annual supplement from the American Society of Business Publication Editors. The 2023 issue won a National Gold Award and a Regional Gold Award.
—Glenn Williams, VP, Group Editor, Neurology Reviews and MDedge Neurology
A NOTE FROM NORD
Hello, and Welcome! The National Organization for Rare Disorders (NORD) is pleased to partner with Neurology Reviews to bring you the 2024 edition of the Rare Neurological Disease Report. Through this collaboration, we share cutting-edge research and insights from leading medical experts, including specialists from the NORD Rare Disease Centers of Excellence network, about the latest advances in the treatment of rare neurological conditions.
As healthcare providers, you play a key role in catalyzing advancements and bringing new hope and possibilities to the rare disease community. Your efforts can contribute to shortening the diagnostic odyssey and improving day-to-day care for people living with rare disorders in crucial ways:
Identifying patients: Healthcare providers can recognize the possible signs of a rare disease and initiate further investigation or referral to specialists. Early detection is key as it can lead to a quicker, more accurate diagnosis, better management, and improved outcomes.
Educating other physicians: Many rare diseases are not well-known or understood by the general medical community. Healthcare providers can help bridge this knowledge gap by educating other physicians about rare conditions. They can raise awareness through clinical teaching, seminars, medical literature, or continuing medical education (CME) sessions focused on rare diseases. Raising awareness and providing up-to-date information about rare diseases bolsters diagnostic and treatment capabilities within the medical field.
Providing information to patients: Once a rare disease is identified, healthcare providers can offer valuable support to patients and their families. They can provide potential treatments and management strategies. They can also connect patients with support groups, support programs, educational resources, and specialists with expertise in specific rare conditions. Clear communication and guidance on support resources can positively impact patients’ well-being, empower them to make informed decisions, and help them navigate a complex rare condition.
This issue of the Rare Disease Neurological Special Report features articles by rare disease medical experts on specific diseases with updates on clinical management. Topics include the diagnosis and management of Duchenne muscular dystrophy, the promise of disease-modifying therapies for Huntington’s disease, patient choices and cultural changes around myasthenia gravis, advances in neuromyelitis optica, and untangling chronic inflammatory demyelinating polyneuropathy. In addition, two online-only articles offer timely insights from key opinion leaders on the pros and cons of genetic testing and what clinicians need to know about newborn screening.
You will also find information about the NORD Rare Diseases and Orphan Products Breakthrough Summit. This annual event convenes thought leaders from patient advocacy organizations, industry, academia, medical and research institutions, and government to discuss critical topics facing the rare disease community.
NORD is deeply appreciative of healthcare professionals like you, who despite long hours and demanding workloads, remain committed to staying up to date on the latest medical advances for the benefit of their patients. Your dedication and hard work make a significant difference to the patients and families we serve, and your commitment does not go unnoticed. Thank you for all that you do.
—Pamela Gavin, NORD Chief Executive Officer
EDITOR’S NOTE
This year, we again focus on rare neurological diseases that have new therapies that have been recently approved as well as conditions for which the treatment pipeline is robust. Let’s hope the work of many dedicated researchers adds to the list of rare neurological diseases for which treatment is available.
This year also marks a change of leadership at NORD, our publishing partner in this annual supplement. We here at Neurology Reviews salute the leadership and accomplishments of former NORD CEO Peter Saltonstall and also welcome incoming CEO Pamela Gavin, who has spent many years in NORD leadership roles and was essential in the planning, launch, and early years of this annual supplement. I can think of no one better than Pamela Gavin to continue NORD’s mission into the future.
And finally, a recap of accolades for this annual supplement. For the second year in a row, the Rare Neurological Disease Special Report has won an Azbee award in the category of annual supplement from the American Society of Business Publication Editors. The 2023 issue won a National Gold Award and a Regional Gold Award.
—Glenn Williams, VP, Group Editor, Neurology Reviews and MDedge Neurology
A NOTE FROM NORD
Hello, and Welcome! The National Organization for Rare Disorders (NORD) is pleased to partner with Neurology Reviews to bring you the 2024 edition of the Rare Neurological Disease Report. Through this collaboration, we share cutting-edge research and insights from leading medical experts, including specialists from the NORD Rare Disease Centers of Excellence network, about the latest advances in the treatment of rare neurological conditions.
As healthcare providers, you play a key role in catalyzing advancements and bringing new hope and possibilities to the rare disease community. Your efforts can contribute to shortening the diagnostic odyssey and improving day-to-day care for people living with rare disorders in crucial ways:
Identifying patients: Healthcare providers can recognize the possible signs of a rare disease and initiate further investigation or referral to specialists. Early detection is key as it can lead to a quicker, more accurate diagnosis, better management, and improved outcomes.
Educating other physicians: Many rare diseases are not well-known or understood by the general medical community. Healthcare providers can help bridge this knowledge gap by educating other physicians about rare conditions. They can raise awareness through clinical teaching, seminars, medical literature, or continuing medical education (CME) sessions focused on rare diseases. Raising awareness and providing up-to-date information about rare diseases bolsters diagnostic and treatment capabilities within the medical field.
Providing information to patients: Once a rare disease is identified, healthcare providers can offer valuable support to patients and their families. They can provide potential treatments and management strategies. They can also connect patients with support groups, support programs, educational resources, and specialists with expertise in specific rare conditions. Clear communication and guidance on support resources can positively impact patients’ well-being, empower them to make informed decisions, and help them navigate a complex rare condition.
This issue of the Rare Disease Neurological Special Report features articles by rare disease medical experts on specific diseases with updates on clinical management. Topics include the diagnosis and management of Duchenne muscular dystrophy, the promise of disease-modifying therapies for Huntington’s disease, patient choices and cultural changes around myasthenia gravis, advances in neuromyelitis optica, and untangling chronic inflammatory demyelinating polyneuropathy. In addition, two online-only articles offer timely insights from key opinion leaders on the pros and cons of genetic testing and what clinicians need to know about newborn screening.
You will also find information about the NORD Rare Diseases and Orphan Products Breakthrough Summit. This annual event convenes thought leaders from patient advocacy organizations, industry, academia, medical and research institutions, and government to discuss critical topics facing the rare disease community.
NORD is deeply appreciative of healthcare professionals like you, who despite long hours and demanding workloads, remain committed to staying up to date on the latest medical advances for the benefit of their patients. Your dedication and hard work make a significant difference to the patients and families we serve, and your commitment does not go unnoticed. Thank you for all that you do.
—Pamela Gavin, NORD Chief Executive Officer
EDITOR’S NOTE
This year, we again focus on rare neurological diseases that have new therapies that have been recently approved as well as conditions for which the treatment pipeline is robust. Let’s hope the work of many dedicated researchers adds to the list of rare neurological diseases for which treatment is available.
This year also marks a change of leadership at NORD, our publishing partner in this annual supplement. We here at Neurology Reviews salute the leadership and accomplishments of former NORD CEO Peter Saltonstall and also welcome incoming CEO Pamela Gavin, who has spent many years in NORD leadership roles and was essential in the planning, launch, and early years of this annual supplement. I can think of no one better than Pamela Gavin to continue NORD’s mission into the future.
And finally, a recap of accolades for this annual supplement. For the second year in a row, the Rare Neurological Disease Special Report has won an Azbee award in the category of annual supplement from the American Society of Business Publication Editors. The 2023 issue won a National Gold Award and a Regional Gold Award.
—Glenn Williams, VP, Group Editor, Neurology Reviews and MDedge Neurology
A NOTE FROM NORD
Hello, and Welcome! The National Organization for Rare Disorders (NORD) is pleased to partner with Neurology Reviews to bring you the 2024 edition of the Rare Neurological Disease Report. Through this collaboration, we share cutting-edge research and insights from leading medical experts, including specialists from the NORD Rare Disease Centers of Excellence network, about the latest advances in the treatment of rare neurological conditions.
As healthcare providers, you play a key role in catalyzing advancements and bringing new hope and possibilities to the rare disease community. Your efforts can contribute to shortening the diagnostic odyssey and improving day-to-day care for people living with rare disorders in crucial ways:
Identifying patients: Healthcare providers can recognize the possible signs of a rare disease and initiate further investigation or referral to specialists. Early detection is key as it can lead to a quicker, more accurate diagnosis, better management, and improved outcomes.
Educating other physicians: Many rare diseases are not well-known or understood by the general medical community. Healthcare providers can help bridge this knowledge gap by educating other physicians about rare conditions. They can raise awareness through clinical teaching, seminars, medical literature, or continuing medical education (CME) sessions focused on rare diseases. Raising awareness and providing up-to-date information about rare diseases bolsters diagnostic and treatment capabilities within the medical field.
Providing information to patients: Once a rare disease is identified, healthcare providers can offer valuable support to patients and their families. They can provide potential treatments and management strategies. They can also connect patients with support groups, support programs, educational resources, and specialists with expertise in specific rare conditions. Clear communication and guidance on support resources can positively impact patients’ well-being, empower them to make informed decisions, and help them navigate a complex rare condition.
This issue of the Rare Disease Neurological Special Report features articles by rare disease medical experts on specific diseases with updates on clinical management. Topics include the diagnosis and management of Duchenne muscular dystrophy, the promise of disease-modifying therapies for Huntington’s disease, patient choices and cultural changes around myasthenia gravis, advances in neuromyelitis optica, and untangling chronic inflammatory demyelinating polyneuropathy. In addition, two online-only articles offer timely insights from key opinion leaders on the pros and cons of genetic testing and what clinicians need to know about newborn screening.
You will also find information about the NORD Rare Diseases and Orphan Products Breakthrough Summit. This annual event convenes thought leaders from patient advocacy organizations, industry, academia, medical and research institutions, and government to discuss critical topics facing the rare disease community.
NORD is deeply appreciative of healthcare professionals like you, who despite long hours and demanding workloads, remain committed to staying up to date on the latest medical advances for the benefit of their patients. Your dedication and hard work make a significant difference to the patients and families we serve, and your commitment does not go unnoticed. Thank you for all that you do.
—Pamela Gavin, NORD Chief Executive Officer
Balancing Act: Weighing the Pros and Cons of Genetic Testing in Rare Diseases
The overwhelming majority of rare diseases have a genetic origin, with estimates varying from 71.9% to 80% of rare diseases. Although a rare disease is defined as a condition that affects fewer than 200,000 people domestically, collectively, rare diseases impact approximately 30 million US residents, with at least one of the more than 7,000 rare genetic disorders. In fact, the population of patients with at least one rare disease mirrors the prevalence of people who have type 2 diabetes, or one in every 10 people. Despite their prevalence, most rare conditions are treated only when symptomatic, as many cases remain either misdiagnosed or undiagnosed. As with most health conditions, it is imperative to have a prompt and accurate diagnosis to improve outcomes and avoid inappropriate or unnecessary treatments that may pose severe side effects to the patient.
To that end, this report weighs the less obvious pros and cons of genetic testing in rare diseases of which neurologists should be aware.
The Path to Accurate Diagnosis Remains Long Despite Increased Genetic Testing
When it comes to identifying the greatest challenge in rare genetic disease testing for the neurology community, experts have different opinions. For Kiley Boone Quintana, MD, assistant professor of pediatrics at the University of New Mexico in Albuquerque, the greatest challenge for neurologists navigating this space lies in becoming comfortable with the unknown.
“Many neurologists think genetic testing will certainly find an answer or that the answers will be black and white — which is not true,” said Dr. Quintana. “Instead of clear answers, we often find variants of unknown significance and genetic changes like a deletion or duplication that can have reduced penetrance, so clinicians have to become comfortable with not always having an answer or not knowing exactly how the answer will impact the person.”
One reason for late diagnosis is the need for more knowledge or familiarity a clinician may have with a certain disease, given its rarity.
Perhaps the nebulous nature of genetic testing for people living with rare diseases unveils another drawback, which centers around what researchers refer to as the “diagnostic odyssey.” While the concept describing the average time to diagnosis as 5 years, the time to diagnosis can vary greatly in the rare disease community. In some cases, patients may experience diagnostic delays of only a few months. For others, the time frame could be a decade or greater. The time frame often depends on the patient’s age, phenotype, and accessibility to resources.
Despite these diagnostic challenges, Debra Regier, MD, PhD, chief, genetics and metabolism, at Children’s National Hospital in Washington, DC, sees the silver lining in identifying the underlying cause of a patient’s symptoms of illness. In some cases, a diagnosis leads a patient to access disease-specific medication. However, in the rare genetic disease space, the occurrence is low, as only approximately 10% of these diagnosed conditions have an available treatment.
Despite the small selection of disease-specific therapies for this patient population, patients may still have options, especially when it comes to palliating symptoms.
“We often look toward disease experts to consider what medications are more likely to be supportive,” Dr. Regier said. “This might mean considering a pain regimen, a seizure regimen, other type of symptomatic treatment, or even using some information learned to support the current patient from cases where other families may have preceded them in the odyssey.”
Whole Exome and Whole Genome Testing Continues Growing in Prevalence, But Neither Offers a Panacea
Historically, genetic testing was expensive, with only a few genes interrogated at a time. However, the past decade has seen prices simmer down with the introduction of next-generation sequencing — a technology that improves both the accuracy and utility of genetic testing.
One form of genetic testing, called whole exome sequencing, has proven especially helpful in recent years because it looks at all 20,000 genes and spelling changes that can cause mutations and genetic diseases. However, whole exome testing comes with its own limitations. It tests at the DNA loci that produce the actual protein blueprints but does not look at the DNA between those spaces. In addition, the medical community lacks a comprehensive understanding of all 20,000 genes, as scientists have yet to understand all their functions.
Unfortunately, the drawbacks do not stop there.
“Whole exome sequencing is not good at detecting conditions such as Huntington’s disease or Fragile X syndrome,” Dr. Quintana said. “It also fails to pick up spelling changes in DNA of noncoding regions, which we are learning do have functions in epigenetics.”
Quality also can limit reliability of both exome and genome testing. According to Dr. Regier, trustworthiness of results depends on several factors, including the lab conducting the test and the analysis performed. To help ensure quality, Dr. Regier and her colleagues use only CLIA-certified labs and labs that follow the American College of Medical Genetics (ACMG) guidelines. Furthermore, they allow only qualified experts to analyze the results, experts who hold board certifications with either the American College of Medical Genetics and Genomics or the American Board of Pathology.
Familial and Societal Stigma Surrounding Rare Diseases Engenders Emotional, Psychological, and Financial Distress
Ultimately, traversing the trajectory of delayed diagnosis and its ambiguity also leaves questions regarding how it will impact the person. All too often, these mysteries transcend the patient with the condition, affecting relatives and other loved ones, as the familial and societal stigma surrounding rare diseases engenders emotional and psychological distress.
In cases with prolonged or delayed diagnostics, Dr. Quintana said that neurologists should advise patients to prepare themselves for the potential of arduous workups — some of which may also come at a high price. Not only does a circuitous path to diagnosis impede treatment initiation, but it often results in major trauma for patients and their caregivers, who encounter significant emotional, psychological, and financial distress in the fallout. Emotional distress of misdiagnosis or lack of a diagnosis remains a significant pain point for patients and their family members alike.
Emotional distress presents the greatest drawback for the rare disease community, according to Dr. Regier. She described the cons of navigating a rare genetic disease diagnosis as “very personal” for families.
“Sometimes, there can be guilt or shame associated with a genetic illness,” Dr. Regier noted. “Understanding the ‘why’ or knowing better how to use nonspecific treatments can be incredibly important to reduce guilt and shame, but it also allows the family to feel like there is a reason and encourages inclusion in the social setting.”
Diagnosis typically results in inclusion in a patient and family group, which increases understanding while easing some of the psychological and emotional stress associated with not knowing the cause.
Establishing Social Support Networks Typically Falls on the Patient and Loved Ones
Another con in rare genetic diseases is the lack of adoption across the community.
Because of the long haul, neurologists and other clinicians should recognize the need for patients to have support. Both Dr. Regier and Dr. Quintana agreed that communal support is a critical component of managing the rare genetic disease population. However, finding one’s tribe is easier said than done. Due to the diagnostic hurdles and low number of people with confirmed diagnoses, patient communities and patient advocacy groups for people with individual rare diseases can be underdeveloped. However, the importance of family-based support groups should not be understated. The low community head counts and high level of time investment for care also contributes to poor recruitment turnouts for clinical trials and, subsequently, the sparse number of therapies for such conditions in the pipeline. However, it is also worth noting that, in the case of rare diseases, insufficient disease state knowledge, antiquated policies, lack of funding, and poor research and development diagnostic infrastructure also amplify such cons.
Patients can form communities of support by finding other families and knowing what to expect in terms of complications. While clinicians may not always have the resources to help the patient establish support systems, they can increase the patients’ awareness and encourage them to search for groups that align with their needs. Dr. Quintana reported that many of her patients find support groups of people with the same rare conditions through social media outlets such as Facebook.
Lack of Widespread Genetic Testing Adoption Remains a Barrier in Rare Diseases
As Dr. Quintana told Neurology Reviews, geneticists are more likely to order exome testing, despite the fact that genome-wide testing is slightly more likely to find a diagnosis. However, she anticipates that genome-wide testing will gain wider adoption in the future.
In terms of cost and feasibility, genetic testing can identify roughly 50% of the underlying etiology of a rare disease, including phenotyping to make a clinical diagnosis and using genetic testing, according to Dr. Regier.
Regarding the broad use of whole genome sequencing, Dr. Regier foresees that the more we learn about all the diagnostic and prognostic information rare disease testing can give us, “the more this number will grow.”
As an example of the true impact, she shared how new research indicates that changes to one’s DNA can lead to intellectual disability.
Dr. Quintana agreed that genetic testing will increase, noting an increase in genetic testing ordered from neonatal intensive care units. However, that uptick comes with the caveat of an ever-evolving landscape as genetic companies continue undergoing mergers, acquisitions, and other structural changes that can complicate service availability, provision, and acceptance.
Even if the clinician orders a comprehensive workup, he or she may still encounter resistance at the hands of insurance companies, which can prolong an accurate and prompt diagnosis while hindering families’ access to a thorough investigation.
“Genetic testing is advantageous for insurance companies as well and can prevent unnecessary lab tests to find an answer,” said Dr. Quintana.
Accessibility and Lack of Geneticists Often a Rate-Limiting Step
The paucity of geneticists also creates another hurdle. “Where I practice in New Mexico and in many other places in this country, there’s a shortage of geneticists,” Dr. Quintana said. “For 3 years, the state had only one geneticist, and that’s a lot of ground to cover.”
Dr. Quintana went on to stress the importance of neurologists and other clinicians conducting outreach in rural areas despite the logistical barriers; oftentimes, families cannot travel to big cities. Despite these geographical challenges, prenatal genetic testing is becoming more accessible for both rural and urban areas. For that reason, some babies are born with a diagnosis, allowing the parents and healthcare providers to take immediate action.
Moreover, risks and uncertainty exist around genetic testing results and access to long-term life insurance and disability insurance coverage. “Obtaining proper consent prior to genetic testing is very important,” said Dr. Quintana.
In many cases, genetic counseling may be beneficial because it offers patients some additional information and resources that help them understand not only the results of their genetic tests but also the consequences of their conditions.
Ultimately, Genetic Testing in Rare Diseases Requires All Stakeholders to Have Patience and Tenacity
Dr. Regier summarized some of the nuances of genetic testing in the rare disease community. “Families understand that you might not be able to make the diagnosis,” Dr. Regier said. “It is more important to them that you stay on the journey with them, even if there is not a diagnosis.”
Another critical element of the diagnostic voyage hinges on clinicians recognizing and honoring that every family — and patient — is different.
“Some families want to do testing while others want to take one thing at a time and start with symptom management,” Dr. Regier said. “Both of these approaches are good, and every family has the right to decide when and if genetic testing should be part of their diagnostic odyssey.”
Suggested Reading
Baynam G et al. Stigma Associated With Genetic Testing for Rare Diseases — Causes and Recommendations. Front Genet. 2024 Apr 4:15:1335768. doi: 10.3389/fgene.2024.1335768.
Marwaha S et al. A Guide for the Diagnosis of Rare and Undiagnosed Disease: Beyond the Exome. Genome Med. 2022 Feb 28;14(1):23. doi: 10.1186/s13073-022-01026-w.
The overwhelming majority of rare diseases have a genetic origin, with estimates varying from 71.9% to 80% of rare diseases. Although a rare disease is defined as a condition that affects fewer than 200,000 people domestically, collectively, rare diseases impact approximately 30 million US residents, with at least one of the more than 7,000 rare genetic disorders. In fact, the population of patients with at least one rare disease mirrors the prevalence of people who have type 2 diabetes, or one in every 10 people. Despite their prevalence, most rare conditions are treated only when symptomatic, as many cases remain either misdiagnosed or undiagnosed. As with most health conditions, it is imperative to have a prompt and accurate diagnosis to improve outcomes and avoid inappropriate or unnecessary treatments that may pose severe side effects to the patient.
To that end, this report weighs the less obvious pros and cons of genetic testing in rare diseases of which neurologists should be aware.
The Path to Accurate Diagnosis Remains Long Despite Increased Genetic Testing
When it comes to identifying the greatest challenge in rare genetic disease testing for the neurology community, experts have different opinions. For Kiley Boone Quintana, MD, assistant professor of pediatrics at the University of New Mexico in Albuquerque, the greatest challenge for neurologists navigating this space lies in becoming comfortable with the unknown.
“Many neurologists think genetic testing will certainly find an answer or that the answers will be black and white — which is not true,” said Dr. Quintana. “Instead of clear answers, we often find variants of unknown significance and genetic changes like a deletion or duplication that can have reduced penetrance, so clinicians have to become comfortable with not always having an answer or not knowing exactly how the answer will impact the person.”
One reason for late diagnosis is the need for more knowledge or familiarity a clinician may have with a certain disease, given its rarity.
Perhaps the nebulous nature of genetic testing for people living with rare diseases unveils another drawback, which centers around what researchers refer to as the “diagnostic odyssey.” While the concept describing the average time to diagnosis as 5 years, the time to diagnosis can vary greatly in the rare disease community. In some cases, patients may experience diagnostic delays of only a few months. For others, the time frame could be a decade or greater. The time frame often depends on the patient’s age, phenotype, and accessibility to resources.
Despite these diagnostic challenges, Debra Regier, MD, PhD, chief, genetics and metabolism, at Children’s National Hospital in Washington, DC, sees the silver lining in identifying the underlying cause of a patient’s symptoms of illness. In some cases, a diagnosis leads a patient to access disease-specific medication. However, in the rare genetic disease space, the occurrence is low, as only approximately 10% of these diagnosed conditions have an available treatment.
Despite the small selection of disease-specific therapies for this patient population, patients may still have options, especially when it comes to palliating symptoms.
“We often look toward disease experts to consider what medications are more likely to be supportive,” Dr. Regier said. “This might mean considering a pain regimen, a seizure regimen, other type of symptomatic treatment, or even using some information learned to support the current patient from cases where other families may have preceded them in the odyssey.”
Whole Exome and Whole Genome Testing Continues Growing in Prevalence, But Neither Offers a Panacea
Historically, genetic testing was expensive, with only a few genes interrogated at a time. However, the past decade has seen prices simmer down with the introduction of next-generation sequencing — a technology that improves both the accuracy and utility of genetic testing.
One form of genetic testing, called whole exome sequencing, has proven especially helpful in recent years because it looks at all 20,000 genes and spelling changes that can cause mutations and genetic diseases. However, whole exome testing comes with its own limitations. It tests at the DNA loci that produce the actual protein blueprints but does not look at the DNA between those spaces. In addition, the medical community lacks a comprehensive understanding of all 20,000 genes, as scientists have yet to understand all their functions.
Unfortunately, the drawbacks do not stop there.
“Whole exome sequencing is not good at detecting conditions such as Huntington’s disease or Fragile X syndrome,” Dr. Quintana said. “It also fails to pick up spelling changes in DNA of noncoding regions, which we are learning do have functions in epigenetics.”
Quality also can limit reliability of both exome and genome testing. According to Dr. Regier, trustworthiness of results depends on several factors, including the lab conducting the test and the analysis performed. To help ensure quality, Dr. Regier and her colleagues use only CLIA-certified labs and labs that follow the American College of Medical Genetics (ACMG) guidelines. Furthermore, they allow only qualified experts to analyze the results, experts who hold board certifications with either the American College of Medical Genetics and Genomics or the American Board of Pathology.
Familial and Societal Stigma Surrounding Rare Diseases Engenders Emotional, Psychological, and Financial Distress
Ultimately, traversing the trajectory of delayed diagnosis and its ambiguity also leaves questions regarding how it will impact the person. All too often, these mysteries transcend the patient with the condition, affecting relatives and other loved ones, as the familial and societal stigma surrounding rare diseases engenders emotional and psychological distress.
In cases with prolonged or delayed diagnostics, Dr. Quintana said that neurologists should advise patients to prepare themselves for the potential of arduous workups — some of which may also come at a high price. Not only does a circuitous path to diagnosis impede treatment initiation, but it often results in major trauma for patients and their caregivers, who encounter significant emotional, psychological, and financial distress in the fallout. Emotional distress of misdiagnosis or lack of a diagnosis remains a significant pain point for patients and their family members alike.
Emotional distress presents the greatest drawback for the rare disease community, according to Dr. Regier. She described the cons of navigating a rare genetic disease diagnosis as “very personal” for families.
“Sometimes, there can be guilt or shame associated with a genetic illness,” Dr. Regier noted. “Understanding the ‘why’ or knowing better how to use nonspecific treatments can be incredibly important to reduce guilt and shame, but it also allows the family to feel like there is a reason and encourages inclusion in the social setting.”
Diagnosis typically results in inclusion in a patient and family group, which increases understanding while easing some of the psychological and emotional stress associated with not knowing the cause.
Establishing Social Support Networks Typically Falls on the Patient and Loved Ones
Another con in rare genetic diseases is the lack of adoption across the community.
Because of the long haul, neurologists and other clinicians should recognize the need for patients to have support. Both Dr. Regier and Dr. Quintana agreed that communal support is a critical component of managing the rare genetic disease population. However, finding one’s tribe is easier said than done. Due to the diagnostic hurdles and low number of people with confirmed diagnoses, patient communities and patient advocacy groups for people with individual rare diseases can be underdeveloped. However, the importance of family-based support groups should not be understated. The low community head counts and high level of time investment for care also contributes to poor recruitment turnouts for clinical trials and, subsequently, the sparse number of therapies for such conditions in the pipeline. However, it is also worth noting that, in the case of rare diseases, insufficient disease state knowledge, antiquated policies, lack of funding, and poor research and development diagnostic infrastructure also amplify such cons.
Patients can form communities of support by finding other families and knowing what to expect in terms of complications. While clinicians may not always have the resources to help the patient establish support systems, they can increase the patients’ awareness and encourage them to search for groups that align with their needs. Dr. Quintana reported that many of her patients find support groups of people with the same rare conditions through social media outlets such as Facebook.
Lack of Widespread Genetic Testing Adoption Remains a Barrier in Rare Diseases
As Dr. Quintana told Neurology Reviews, geneticists are more likely to order exome testing, despite the fact that genome-wide testing is slightly more likely to find a diagnosis. However, she anticipates that genome-wide testing will gain wider adoption in the future.
In terms of cost and feasibility, genetic testing can identify roughly 50% of the underlying etiology of a rare disease, including phenotyping to make a clinical diagnosis and using genetic testing, according to Dr. Regier.
Regarding the broad use of whole genome sequencing, Dr. Regier foresees that the more we learn about all the diagnostic and prognostic information rare disease testing can give us, “the more this number will grow.”
As an example of the true impact, she shared how new research indicates that changes to one’s DNA can lead to intellectual disability.
Dr. Quintana agreed that genetic testing will increase, noting an increase in genetic testing ordered from neonatal intensive care units. However, that uptick comes with the caveat of an ever-evolving landscape as genetic companies continue undergoing mergers, acquisitions, and other structural changes that can complicate service availability, provision, and acceptance.
Even if the clinician orders a comprehensive workup, he or she may still encounter resistance at the hands of insurance companies, which can prolong an accurate and prompt diagnosis while hindering families’ access to a thorough investigation.
“Genetic testing is advantageous for insurance companies as well and can prevent unnecessary lab tests to find an answer,” said Dr. Quintana.
Accessibility and Lack of Geneticists Often a Rate-Limiting Step
The paucity of geneticists also creates another hurdle. “Where I practice in New Mexico and in many other places in this country, there’s a shortage of geneticists,” Dr. Quintana said. “For 3 years, the state had only one geneticist, and that’s a lot of ground to cover.”
Dr. Quintana went on to stress the importance of neurologists and other clinicians conducting outreach in rural areas despite the logistical barriers; oftentimes, families cannot travel to big cities. Despite these geographical challenges, prenatal genetic testing is becoming more accessible for both rural and urban areas. For that reason, some babies are born with a diagnosis, allowing the parents and healthcare providers to take immediate action.
Moreover, risks and uncertainty exist around genetic testing results and access to long-term life insurance and disability insurance coverage. “Obtaining proper consent prior to genetic testing is very important,” said Dr. Quintana.
In many cases, genetic counseling may be beneficial because it offers patients some additional information and resources that help them understand not only the results of their genetic tests but also the consequences of their conditions.
Ultimately, Genetic Testing in Rare Diseases Requires All Stakeholders to Have Patience and Tenacity
Dr. Regier summarized some of the nuances of genetic testing in the rare disease community. “Families understand that you might not be able to make the diagnosis,” Dr. Regier said. “It is more important to them that you stay on the journey with them, even if there is not a diagnosis.”
Another critical element of the diagnostic voyage hinges on clinicians recognizing and honoring that every family — and patient — is different.
“Some families want to do testing while others want to take one thing at a time and start with symptom management,” Dr. Regier said. “Both of these approaches are good, and every family has the right to decide when and if genetic testing should be part of their diagnostic odyssey.”
Suggested Reading
Baynam G et al. Stigma Associated With Genetic Testing for Rare Diseases — Causes and Recommendations. Front Genet. 2024 Apr 4:15:1335768. doi: 10.3389/fgene.2024.1335768.
Marwaha S et al. A Guide for the Diagnosis of Rare and Undiagnosed Disease: Beyond the Exome. Genome Med. 2022 Feb 28;14(1):23. doi: 10.1186/s13073-022-01026-w.
The overwhelming majority of rare diseases have a genetic origin, with estimates varying from 71.9% to 80% of rare diseases. Although a rare disease is defined as a condition that affects fewer than 200,000 people domestically, collectively, rare diseases impact approximately 30 million US residents, with at least one of the more than 7,000 rare genetic disorders. In fact, the population of patients with at least one rare disease mirrors the prevalence of people who have type 2 diabetes, or one in every 10 people. Despite their prevalence, most rare conditions are treated only when symptomatic, as many cases remain either misdiagnosed or undiagnosed. As with most health conditions, it is imperative to have a prompt and accurate diagnosis to improve outcomes and avoid inappropriate or unnecessary treatments that may pose severe side effects to the patient.
To that end, this report weighs the less obvious pros and cons of genetic testing in rare diseases of which neurologists should be aware.
The Path to Accurate Diagnosis Remains Long Despite Increased Genetic Testing
When it comes to identifying the greatest challenge in rare genetic disease testing for the neurology community, experts have different opinions. For Kiley Boone Quintana, MD, assistant professor of pediatrics at the University of New Mexico in Albuquerque, the greatest challenge for neurologists navigating this space lies in becoming comfortable with the unknown.
“Many neurologists think genetic testing will certainly find an answer or that the answers will be black and white — which is not true,” said Dr. Quintana. “Instead of clear answers, we often find variants of unknown significance and genetic changes like a deletion or duplication that can have reduced penetrance, so clinicians have to become comfortable with not always having an answer or not knowing exactly how the answer will impact the person.”
One reason for late diagnosis is the need for more knowledge or familiarity a clinician may have with a certain disease, given its rarity.
Perhaps the nebulous nature of genetic testing for people living with rare diseases unveils another drawback, which centers around what researchers refer to as the “diagnostic odyssey.” While the concept describing the average time to diagnosis as 5 years, the time to diagnosis can vary greatly in the rare disease community. In some cases, patients may experience diagnostic delays of only a few months. For others, the time frame could be a decade or greater. The time frame often depends on the patient’s age, phenotype, and accessibility to resources.
Despite these diagnostic challenges, Debra Regier, MD, PhD, chief, genetics and metabolism, at Children’s National Hospital in Washington, DC, sees the silver lining in identifying the underlying cause of a patient’s symptoms of illness. In some cases, a diagnosis leads a patient to access disease-specific medication. However, in the rare genetic disease space, the occurrence is low, as only approximately 10% of these diagnosed conditions have an available treatment.
Despite the small selection of disease-specific therapies for this patient population, patients may still have options, especially when it comes to palliating symptoms.
“We often look toward disease experts to consider what medications are more likely to be supportive,” Dr. Regier said. “This might mean considering a pain regimen, a seizure regimen, other type of symptomatic treatment, or even using some information learned to support the current patient from cases where other families may have preceded them in the odyssey.”
Whole Exome and Whole Genome Testing Continues Growing in Prevalence, But Neither Offers a Panacea
Historically, genetic testing was expensive, with only a few genes interrogated at a time. However, the past decade has seen prices simmer down with the introduction of next-generation sequencing — a technology that improves both the accuracy and utility of genetic testing.
One form of genetic testing, called whole exome sequencing, has proven especially helpful in recent years because it looks at all 20,000 genes and spelling changes that can cause mutations and genetic diseases. However, whole exome testing comes with its own limitations. It tests at the DNA loci that produce the actual protein blueprints but does not look at the DNA between those spaces. In addition, the medical community lacks a comprehensive understanding of all 20,000 genes, as scientists have yet to understand all their functions.
Unfortunately, the drawbacks do not stop there.
“Whole exome sequencing is not good at detecting conditions such as Huntington’s disease or Fragile X syndrome,” Dr. Quintana said. “It also fails to pick up spelling changes in DNA of noncoding regions, which we are learning do have functions in epigenetics.”
Quality also can limit reliability of both exome and genome testing. According to Dr. Regier, trustworthiness of results depends on several factors, including the lab conducting the test and the analysis performed. To help ensure quality, Dr. Regier and her colleagues use only CLIA-certified labs and labs that follow the American College of Medical Genetics (ACMG) guidelines. Furthermore, they allow only qualified experts to analyze the results, experts who hold board certifications with either the American College of Medical Genetics and Genomics or the American Board of Pathology.
Familial and Societal Stigma Surrounding Rare Diseases Engenders Emotional, Psychological, and Financial Distress
Ultimately, traversing the trajectory of delayed diagnosis and its ambiguity also leaves questions regarding how it will impact the person. All too often, these mysteries transcend the patient with the condition, affecting relatives and other loved ones, as the familial and societal stigma surrounding rare diseases engenders emotional and psychological distress.
In cases with prolonged or delayed diagnostics, Dr. Quintana said that neurologists should advise patients to prepare themselves for the potential of arduous workups — some of which may also come at a high price. Not only does a circuitous path to diagnosis impede treatment initiation, but it often results in major trauma for patients and their caregivers, who encounter significant emotional, psychological, and financial distress in the fallout. Emotional distress of misdiagnosis or lack of a diagnosis remains a significant pain point for patients and their family members alike.
Emotional distress presents the greatest drawback for the rare disease community, according to Dr. Regier. She described the cons of navigating a rare genetic disease diagnosis as “very personal” for families.
“Sometimes, there can be guilt or shame associated with a genetic illness,” Dr. Regier noted. “Understanding the ‘why’ or knowing better how to use nonspecific treatments can be incredibly important to reduce guilt and shame, but it also allows the family to feel like there is a reason and encourages inclusion in the social setting.”
Diagnosis typically results in inclusion in a patient and family group, which increases understanding while easing some of the psychological and emotional stress associated with not knowing the cause.
Establishing Social Support Networks Typically Falls on the Patient and Loved Ones
Another con in rare genetic diseases is the lack of adoption across the community.
Because of the long haul, neurologists and other clinicians should recognize the need for patients to have support. Both Dr. Regier and Dr. Quintana agreed that communal support is a critical component of managing the rare genetic disease population. However, finding one’s tribe is easier said than done. Due to the diagnostic hurdles and low number of people with confirmed diagnoses, patient communities and patient advocacy groups for people with individual rare diseases can be underdeveloped. However, the importance of family-based support groups should not be understated. The low community head counts and high level of time investment for care also contributes to poor recruitment turnouts for clinical trials and, subsequently, the sparse number of therapies for such conditions in the pipeline. However, it is also worth noting that, in the case of rare diseases, insufficient disease state knowledge, antiquated policies, lack of funding, and poor research and development diagnostic infrastructure also amplify such cons.
Patients can form communities of support by finding other families and knowing what to expect in terms of complications. While clinicians may not always have the resources to help the patient establish support systems, they can increase the patients’ awareness and encourage them to search for groups that align with their needs. Dr. Quintana reported that many of her patients find support groups of people with the same rare conditions through social media outlets such as Facebook.
Lack of Widespread Genetic Testing Adoption Remains a Barrier in Rare Diseases
As Dr. Quintana told Neurology Reviews, geneticists are more likely to order exome testing, despite the fact that genome-wide testing is slightly more likely to find a diagnosis. However, she anticipates that genome-wide testing will gain wider adoption in the future.
In terms of cost and feasibility, genetic testing can identify roughly 50% of the underlying etiology of a rare disease, including phenotyping to make a clinical diagnosis and using genetic testing, according to Dr. Regier.
Regarding the broad use of whole genome sequencing, Dr. Regier foresees that the more we learn about all the diagnostic and prognostic information rare disease testing can give us, “the more this number will grow.”
As an example of the true impact, she shared how new research indicates that changes to one’s DNA can lead to intellectual disability.
Dr. Quintana agreed that genetic testing will increase, noting an increase in genetic testing ordered from neonatal intensive care units. However, that uptick comes with the caveat of an ever-evolving landscape as genetic companies continue undergoing mergers, acquisitions, and other structural changes that can complicate service availability, provision, and acceptance.
Even if the clinician orders a comprehensive workup, he or she may still encounter resistance at the hands of insurance companies, which can prolong an accurate and prompt diagnosis while hindering families’ access to a thorough investigation.
“Genetic testing is advantageous for insurance companies as well and can prevent unnecessary lab tests to find an answer,” said Dr. Quintana.
Accessibility and Lack of Geneticists Often a Rate-Limiting Step
The paucity of geneticists also creates another hurdle. “Where I practice in New Mexico and in many other places in this country, there’s a shortage of geneticists,” Dr. Quintana said. “For 3 years, the state had only one geneticist, and that’s a lot of ground to cover.”
Dr. Quintana went on to stress the importance of neurologists and other clinicians conducting outreach in rural areas despite the logistical barriers; oftentimes, families cannot travel to big cities. Despite these geographical challenges, prenatal genetic testing is becoming more accessible for both rural and urban areas. For that reason, some babies are born with a diagnosis, allowing the parents and healthcare providers to take immediate action.
Moreover, risks and uncertainty exist around genetic testing results and access to long-term life insurance and disability insurance coverage. “Obtaining proper consent prior to genetic testing is very important,” said Dr. Quintana.
In many cases, genetic counseling may be beneficial because it offers patients some additional information and resources that help them understand not only the results of their genetic tests but also the consequences of their conditions.
Ultimately, Genetic Testing in Rare Diseases Requires All Stakeholders to Have Patience and Tenacity
Dr. Regier summarized some of the nuances of genetic testing in the rare disease community. “Families understand that you might not be able to make the diagnosis,” Dr. Regier said. “It is more important to them that you stay on the journey with them, even if there is not a diagnosis.”
Another critical element of the diagnostic voyage hinges on clinicians recognizing and honoring that every family — and patient — is different.
“Some families want to do testing while others want to take one thing at a time and start with symptom management,” Dr. Regier said. “Both of these approaches are good, and every family has the right to decide when and if genetic testing should be part of their diagnostic odyssey.”
Suggested Reading
Baynam G et al. Stigma Associated With Genetic Testing for Rare Diseases — Causes and Recommendations. Front Genet. 2024 Apr 4:15:1335768. doi: 10.3389/fgene.2024.1335768.
Marwaha S et al. A Guide for the Diagnosis of Rare and Undiagnosed Disease: Beyond the Exome. Genome Med. 2022 Feb 28;14(1):23. doi: 10.1186/s13073-022-01026-w.
Myasthenia Gravis: Patient Choice, Cultural Change
Used appropriately, newer treatments can provide dramatic results faster and more safely than broad immunosuppressants. However, according to experts, payers’ willingness to cover costly new therapies remains a work in progress.
The availability of more effective treatments with fewer side effects has brought about a cultural shift, said James F. Howard, Jr, MD. “The physician’s goal now is for the patient to be symptom free with grade 1 or less adverse events. And patients are demanding freedom from all the side effects that our usual course of immune therapy produces.” Dr. Howard is professor of neurology, medicine and allied health and director of the Myasthenia Gravis Clinical Trials and Translational Research Program at the University of North Carolina at Chapel Hill.
The shift has been long in coming. Although myasthenia gravis was identified in the mid-1600s, it took more than 340 years to get the first drug approved specifically for the disorder.
Worldwide prevalence estimates vary widely, from less than 200,000 to 700,000 cases.1,2 Pathophysiologically, myasthenia gravis stems from autoimmune destruction of neuromuscular junctions (NMJs), which transmit motor neuron impulses to muscle fibers.1 Symptoms include variable skeletal muscle weakness that can range from mild and transient to life-threatening.
In approximately 80% of cases, autoimmune antibodies target the postsynaptic acetylcholine receptor (AChR). Additional autoimmune targets mainly include muscle-specific kinase (MuSK) and lipoprotein receptor-related protein 4 (LRP4). However, around 10% of patients are seronegative, lacking autoantibodies detectable through conventional radioimmunoassays. Clinical disease does not always correspond with circulating antibody levels, and pathogenesis may require cooperation between multiple autoantibodies attacking the same target.3 Around 10% of MG cases are associated with thymomas.
Among myasthenia gravis treatments, immunosuppressants typically take 4-10 months to begin working and 18-36 months for maximum benefit. “Our new targeted therapies work within 1-2 weeks, with maximum improvement occurring somewhere between 8 and 12 weeks,” Dr. Howard said. Quick onset makes these drugs well suited for primary therapy in recalcitrant myasthenia gravis or as bridges to standard immunotherapy. Targeted drugs also appear to provide effective rescue therapy, although head-to-head studies are needed.
Complement Inhibition
In AChR antibody–positive myasthenia gravis, autoantibody binding with the postsynaptic AChR receptor activates complement to attack postsynaptic neuronal membrane. Complement inhibitors approved to date block activation of the terminal complement protein C5.
For many patients, complement inhibitors deliver dramatic results. Henry J. Kaminski, MD, said that the first patient for whom he prescribed a complement inhibitor outside a clinical trial went from being miserable to traveling internationally within a month. Dr. Kaminski is Meta A. Neumann Professor of Neurology at George Washington University, Washington, DC.
Eculizumab (Soliris, Alexion), earned Food and Drug Administration (FDA) approval for myasthenia gravis in 2017. Week 26 results in the phase 3 REGAIN trial showed no significant difference in Myasthenia Gravis–Activities of Daily Living (MG-ADL) scores between treatment and placebo. However, said Dr. Howard, primary investigator on the study, the negative result was a statistical aberration stemming from the FDA’s requirement to use worst-rank analysis rather than absolute change scores. What got eculizumab approved were highly positive results in the overwhelming majority of secondary endpoints.4 Subsequently, the FDA had the manufacturer rewrite the package insert using common statistical methods, which yielded positive primary results.
Ravulizumab (Ultomiris, Alexion), approved for myasthenia gravis in 2022, reduces eculizumab’s twice-monthly intravenous dosing to every 2 months (after loading doses), with very similar efficacy. The newest complement inhibitor, zilucoplan (Zilbrysq, UCB), administered once daily subcutaneously, earned FDA approval in 2023. Daily subcutaneous dosing provides patient convenience, said Dr. Howard. Because the body does not clear this small molecule as it would a full-size antibody, it is the only complement inhibitor that can be combined with a fragment crystallizable neonatal receptor (FcRn) inhibitor.
FcRn Inhibition
The FcRn exists on the surface and intracellular vesicles of many cells, including B cells, but not T cells.5FcRn inhibitors block binding of circulating IgG antibodies to the FcRn, preventing their normal recycling, significantly reducing circulating antibodies within days of treatment.
Efgartigimod (Vyvgart, Argenx), earned FDA approval in intravenous form in 2021, followed by a subcutaneous formulation that includes hyaluronidase (Vyvgart Hytrulo) in 2023. Rozanolixizumab (Rystiggo, UCB) earned FDA approval for both AChR antibody–positive and MuSK antibody–positive myasthenia gravis in 2023.
Along with rapid response, said Dr. Howard, complement inhibitors and FcRn inhibitors offer a “hugely improved” side-effect profile. In phase 3 research, the most common side effects for both classes included headache, nausea, and diarrhea.4,6,7 Because complement inhibitors increase the risk of Neisseria infection, users require immunization against meningococcal infection (or concurrent antibiotic prophylaxis) while on complement inhibitors.
Insurance Issues
With many clinicians wondering which targeted therapy to choose for a particular patient, said Dr. Howard and Dr. Kaminski, the main obstacle to wider use of these treatments is payer attitudes and practices. “While many of us would like to see these drugs used earlier in the course of disease,” Dr. Howard explained, “there are numerous restrictions placed on the physician and the patient by whatever insurance the individual has.”
Dr. Kaminski said: “There’s a lot of variability among insurance companies regarding what is expected in terms of getting approval for a certain medication. It frustrates me, thinking this patient may do well with a complement inhibitor or an FcRn inhibitor, but it takes weeks to get them approved.”
Some of his patients have been approved for, and flourished on, complement inhibitors and FcRn inhibitors, he added, and then denied a second round of treatment. Dr. Kaminski said he does not know why these patients were denied, and every time he requests reevaluation, the decision is reversed. “That’s a significant time frame for me and my staff to manage.”
When asked what can be done to address high drug prices, Dr. Howard replied, “I have no idea. I’m not an advocate of high drug prices. But I don’t think people realize the cost of doing clinical trials, which is hundreds of millions of dollars, particularly in rare diseases.”
Presently, Dr. Howard said, FcRn inhibitors are used more frequently than complement inhibitors solely because of cost. Zilucoplan will be priced below existing complement inhibitors, although it is too soon to compare its price with those of FcRn inhibitors.
When eculizumab debuted, said Dr. Howard, it cost nearly $750,000 annually. “But if you look at the number of patients treated, the cost of the drug over this population is probably less than the cost for using a cholesterol-lowering agent to treat hyperlipidemia.”
An Institute for Clinical and Economic Review (ICER) report stated that eculizumab and efgartigimod should both cost less than $20,000 annually to meet commonly used cost-effectiveness thresholds.8 However, Dr. Howard said ICER used models based on common diseases and ignored the economic impact of patients’ losing fewer workdays and avoiding long-term immunosuppressant side effects such as diabetes and osteoporosis and related treatment costs. “We’ve got to start looking at total societal cost,” he said.
Leapfrogging Ahead
Not all the new drugs work in every indicated patient, Dr. Howard said. For example, up to 30% of patients do not respond to complement inhibitors. “We don’t understand why. It’s as if we have leapfrogged way ahead in terms of therapeutics, and now we have to go back and answer all the questions – the who, what, where, and why of an individual drug and its response in folks.”
In this climate, said Dr. Kaminski, heavy direct-to-consumer advertising of newer myasthenia gravis therapies creates complications. “My patients are highly excited to see, ‘that’s my disease being advertised on Jeopardy.’ ” Many patients are frustrated with the general lack of awareness regarding myasthenia gravis, he added. “But then I’ve had patients who clearly would never qualify for a certain medication getting mailings to their homes.”
Dr. Howard countered that broader awareness of myasthenia gravis can only help. “There’s increasing recognition of the disease, not only by patients, but to some extent, by the treating clinician. Patients are coming to our offices and saying, ‘am I a candidate for this new drug?’ It’s the responsibility of the clinician to decide.”
Individual physicians’ practice patterns vary greatly, said Dr. Kaminski, and very little quantitative data exist here. But based on personal communications, academic-center neurologists tend to use targeted treatments on patients who have failed conventional treatments.
Conversely, Dr. Howard said that, because community physicians rarely see myasthenia gravis, and targeted treatments remain relatively new, many of these providers rely on prednisone, azathioprine, and mycophenolate mofetil.
B-Cell Blockers in Development
Overall, said Dr. Howard, the field of myasthenia gravis treatment development is “very rich. And pharma’s interest in myasthenia has taken off like a rocket. It’s exceptionally gratifying to those of us who take care of these patients whose life is miserable” because of adverse effects and/or nonresponse to current drugs.
“In myasthenia,” added Dr. Kaminski, “we know that T cells are promoting the activity of these auto-reactive B cells.” Many drugs currently in phase 2 or 3 development aim to eliminate B cells or signaling between T and B cells, he said. “That’s where most of the drug development is.”
Leading candidates include telitacicept (Tai’ai, RemeGen), which is both a B-lymphocyte stimulator and a proliferation-inducing ligand. A phase 3 trial (NCT05737160) is ongoing, with primary completion expected in late 2026. A second phase 3 trial (NCT06456580) recently began enrolling. Dr. Howard said that, although early results warranted phase 3 analysis, telitacicept’s phase 2 trial was open label and lacked a placebo group.9 The latter is a critical concern because placebo response rates in myasthenia gravis trials average 35%-40%.
Combined with standard care, the FcRn inhibitor nipocalimab (Johnson & Johnson) enabled patients with AChR, MuSK, and/or LRP4 autoantibodies to improve by 4.70 points on the MG-ADL vs 3.25 points for placebo (P = .002) over 24 weeks in phase 3.10All FcRn inhibitors in development can broadly reduce autoantibody levels, said Dr. Howard. “But what role they will play in myasthenia gravis when they’re several years behind leaders in the field in terms of capturing market remains to be seen.”
Additionally, batoclimab (Immunovant/Harbour BioMed) showed positive topline results in phase 3, and an elevated rate of hypercholesterolemia in treated patients that was transient and consistent with previous research.11 Subsequent to efgartigimod, Dr. Howard said, FcRn inhibitors are full-size antibodies. “I believe that contributes to the adverse events that we see. Efgartigimod is a small FcRn fragment. That’s why it’s a cleaner drug, if you will.”
FcRn inhibitors require periodic retreatment. For example, said Dr. Howard, the ADAPT phase 3 trial of efgartigimod, on which he was lead investigator, employed a cyclic dosing schedule – 4 weeks’ treatment, then observation until patients needed retreatment — because patients demanded it.12 In clinical practice, some patients have gone more than 25 weeks before needing retreatment. One of his patients went beyond 40 weeks. “Others only get around 6-9 weeks. So patient choice again enters the decision-making process.”
Rituximab targets the CD20 protein on B cells nonspecifically, producing general immunosuppression. “That’s problematic in producing significant immunosuppression,” said Dr. Kaminski. Nevertheless, he said, rituximab is very effective for most patients with MuSK-specific MG, and its application to this indication has revealed differences between the MuSK subtype and AChR antibody–positive myasthenia. Specifically, MuSK antibody–positive patients have short-lived plasmablasts, which rituximab eliminates.13
Conversely, said Dr. Kaminski, patients with AChR antibody-positive myasthenia, especially long-term, likely have long-lived plasmablasts producing antibodies. This fact, and these patients’ lack of CD20, likely explain their poor response to rituximab.
A phase 3 trial (NCT04524273) of the CD19 blocker inebilizumab (Uplinza, Amgen) reached primary completion in May. Dr. Howard said that if topline results (unreleased at press time) prove positive, inebilizumab could replace rituximab in MG — provided payers do not reject inebilizumab because of cost.
Packed Early-Development Pipeline
Regarding early-stage projects, said Dr. Howard, the pipeline is packed with compounds that target various aspects of the immune system. “The real question with those is, what’s going to be the side effect profile? All of the trials are very early. We need bigger trials with much longer observation for safety, durability, and degree of efficacy.”
The next potential B cell–targeting game changer, he said, is chimeric antigen receptor (CAR) T cell–based therapy. In a phase 2b trial of Descartes-08 (Cartesian Therapeutics), 71% of treated patients experienced clinically meaningful improvement in MG Composite score at 3 months vs 25% for placebo.14
In early clinical trials, said Dr. Howard, patients treated with Descartes-08 — which uses autologous mRNA to target B-cell maturation antigen — have shown “exceptional improvement” lasting 20 or more months. Because the drug is not ingrained permanently into the genome, Descartes-08 avoids potentially severe side effects of DNA-targeting CAR T candidates. Dr. Howard hopes a phase 3 trial will commence around January 2025.
The tolerance approach exemplified by CNP-106 (COUR Pharmaceuticals) and a myasthenia gravis tolerogen (Toleranzia) seeks to prevent the immune system from recognizing and reacting to the NMJ abnormalities that produce myasthenia gravis, potentially providing a cure. “We look forward to those trials as they come online in the next 1-2 years,” said Dr. Howard.
Unmet Needs
Historically, neurologists believed that all myasthenia gravis symptoms stemmed from muscle fatigue — the more active the muscle, the weaker it gets. However, said Dr. Kaminski, some patients might lack measurable weakness but still complain of fatigue.
Elevated levels of cytokines such as interleukin (IL)–6 or IL-17 also can produce fatigue, he noted. “With the drugs we’re using, certainly the new ones, we’re not specifically targeting this fatigue phenomenon, which has been studied in a very limited fashion.”
In the RAISE-XT zilucoplan trial, participants experienced significant improvement in fatigue scores for up to 60 weeks.15 Although zilucoplan does not address fatigue directly, said Dr. Howard, improving myasthenia gravis overall helps reduce fatigue.
The Myasthenia Gravis Symptoms Patient Reported Outcome (MG Symptoms PRO), which Dr. Kaminski helped develop, includes questions designed to distinguish muscular fatigue from overall physical fatigue.16 “I’m very interested in some of the information that’s coming out on long COVID and its effect on muscle,” Dr. Kaminski added. “We might be able to learn from there that there’s still some pathology going on beyond the neuromuscular junction.”
What the field desperately needs, said Dr. Howard, are biomarkers to identify which patients will and will not respond to certain therapeutics. “We’re not there yet.” Such biomarkers are at least 3-7 years from becoming clinical reality.
Promising antibody-independent serum markers include circulating microRNAs. For example, miRNA-150-5p and miRNA-21-5p are elevated in generalized AChR-positive myasthenia gravis and early-onset myasthenia gravis (occurring before age 50) and decline after immunosuppression and thymectomy.17
Among diagnostic modalities for patients with seronegative myasthenia gravis, said Dr. Kaminski, single-fiber EMG is the most sensitive, at approximately 95%. “It’s not perfect.” Moreover, he said, performing this test accurately requires a highly experienced expert, which many treatment centers lack.
Presently, added Dr. Kaminski, orbital MRI is neither specific nor sensitive enough to be clinically useful. “One needs to be careful with these specialized tests that are published from the best laboratory in the world that does the test, and does it repetitively.” As the search for effective myasthenia gravis biomarkers continues, avoiding false-positive results is as important as avoiding false negatives.
References
1. Bubuioc AM et al. J Med Life. 2021 Jan-Mar;14(1):7-16. doi: 10.25122/jml-2020-0145.
2. Deenen JC et al. J Neuromuscul Dis. 2015;2(1):73-85. doi: 10.3233/JND-140045.
3. Kaminski HJ et al. J Clin Invest. 2024 Jun 17;134(12):e179742. doi: 10.1172/JCI179742.
4. Howard JF Jr et al. Lancet Neurol. 2017 Dec;16(12):976-986. doi: 10.1016/S1474-4422(17)30369-1.
5. Huda R. Front Immunol. 2020 Feb 21:11:240. doi: 10.3389/fimmu.2020.00240.
6. Howard JF Jr et al. Lancet Neurol. 2023 May;22(5):395-406. doi: 10.1016/S1474-4422(23)00080-7.
7. Vu T et al. NEJM Evid. 2022 May;1(5):EVIDoa2100066. doi: 10.1056/EVIDoa2100066.
8. Tice JA et al. October 20, 2021. https://icer.org/assessment/myasthenia-gravis/.
9. Yin J et al. Eur J Neurol. 2024 Aug;31(8):e16322. doi: 10.1111/ene.16322.
10. Antozzi C et al. EAN 2024, Abstract EPR-116. https://www.neurology.org/doi/10.1212/WNL.0000000000203660.
11. Yan C et al. JAMA Neurol. 2024 Mar 4;81(4):336-345. doi: 10.1001/jamaneurol.2024.0044.
12. Howard JF Jr et al. Lancet Neurol. 2021 Jul;20(7):526-536. doi: 10.1016/S1474-4422(21)00159-9.
13. Stathopoulos P et al. JCI Insight. 2017 Sep 7;2(17):e94263. doi: 10.1172/jci.insight.94263.
14. Cartesian Therapeutics. Cartesian Therapeutics announces positive topline results from phase 2b trial of Descartes-08 in patients with myasthenia gravis. 2024 Jul 2. https://ir.cartesiantherapeutics.com/news-releases/news-release-details/cartesian-therapeutics-announces-positive-topline-results-phase.
15. Howard JF Jr et al. Ther Adv Neurol Disord. 2024 Apr 17:17:17562864241243186. doi: 10.1177/17562864241243186.
16. Cleanthous S et al. Orphanet J Rare Dis. 2021 Oct 30;16(1):457. doi: 10.1186/s13023-021-02064-0.
17. Sabre L et al. Front Immunol. 2020 Mar 4:11:213. doi: 10.3389/fimmu.2020.00213.
Used appropriately, newer treatments can provide dramatic results faster and more safely than broad immunosuppressants. However, according to experts, payers’ willingness to cover costly new therapies remains a work in progress.
The availability of more effective treatments with fewer side effects has brought about a cultural shift, said James F. Howard, Jr, MD. “The physician’s goal now is for the patient to be symptom free with grade 1 or less adverse events. And patients are demanding freedom from all the side effects that our usual course of immune therapy produces.” Dr. Howard is professor of neurology, medicine and allied health and director of the Myasthenia Gravis Clinical Trials and Translational Research Program at the University of North Carolina at Chapel Hill.
The shift has been long in coming. Although myasthenia gravis was identified in the mid-1600s, it took more than 340 years to get the first drug approved specifically for the disorder.
Worldwide prevalence estimates vary widely, from less than 200,000 to 700,000 cases.1,2 Pathophysiologically, myasthenia gravis stems from autoimmune destruction of neuromuscular junctions (NMJs), which transmit motor neuron impulses to muscle fibers.1 Symptoms include variable skeletal muscle weakness that can range from mild and transient to life-threatening.
In approximately 80% of cases, autoimmune antibodies target the postsynaptic acetylcholine receptor (AChR). Additional autoimmune targets mainly include muscle-specific kinase (MuSK) and lipoprotein receptor-related protein 4 (LRP4). However, around 10% of patients are seronegative, lacking autoantibodies detectable through conventional radioimmunoassays. Clinical disease does not always correspond with circulating antibody levels, and pathogenesis may require cooperation between multiple autoantibodies attacking the same target.3 Around 10% of MG cases are associated with thymomas.
Among myasthenia gravis treatments, immunosuppressants typically take 4-10 months to begin working and 18-36 months for maximum benefit. “Our new targeted therapies work within 1-2 weeks, with maximum improvement occurring somewhere between 8 and 12 weeks,” Dr. Howard said. Quick onset makes these drugs well suited for primary therapy in recalcitrant myasthenia gravis or as bridges to standard immunotherapy. Targeted drugs also appear to provide effective rescue therapy, although head-to-head studies are needed.
Complement Inhibition
In AChR antibody–positive myasthenia gravis, autoantibody binding with the postsynaptic AChR receptor activates complement to attack postsynaptic neuronal membrane. Complement inhibitors approved to date block activation of the terminal complement protein C5.
For many patients, complement inhibitors deliver dramatic results. Henry J. Kaminski, MD, said that the first patient for whom he prescribed a complement inhibitor outside a clinical trial went from being miserable to traveling internationally within a month. Dr. Kaminski is Meta A. Neumann Professor of Neurology at George Washington University, Washington, DC.
Eculizumab (Soliris, Alexion), earned Food and Drug Administration (FDA) approval for myasthenia gravis in 2017. Week 26 results in the phase 3 REGAIN trial showed no significant difference in Myasthenia Gravis–Activities of Daily Living (MG-ADL) scores between treatment and placebo. However, said Dr. Howard, primary investigator on the study, the negative result was a statistical aberration stemming from the FDA’s requirement to use worst-rank analysis rather than absolute change scores. What got eculizumab approved were highly positive results in the overwhelming majority of secondary endpoints.4 Subsequently, the FDA had the manufacturer rewrite the package insert using common statistical methods, which yielded positive primary results.
Ravulizumab (Ultomiris, Alexion), approved for myasthenia gravis in 2022, reduces eculizumab’s twice-monthly intravenous dosing to every 2 months (after loading doses), with very similar efficacy. The newest complement inhibitor, zilucoplan (Zilbrysq, UCB), administered once daily subcutaneously, earned FDA approval in 2023. Daily subcutaneous dosing provides patient convenience, said Dr. Howard. Because the body does not clear this small molecule as it would a full-size antibody, it is the only complement inhibitor that can be combined with a fragment crystallizable neonatal receptor (FcRn) inhibitor.
FcRn Inhibition
The FcRn exists on the surface and intracellular vesicles of many cells, including B cells, but not T cells.5FcRn inhibitors block binding of circulating IgG antibodies to the FcRn, preventing their normal recycling, significantly reducing circulating antibodies within days of treatment.
Efgartigimod (Vyvgart, Argenx), earned FDA approval in intravenous form in 2021, followed by a subcutaneous formulation that includes hyaluronidase (Vyvgart Hytrulo) in 2023. Rozanolixizumab (Rystiggo, UCB) earned FDA approval for both AChR antibody–positive and MuSK antibody–positive myasthenia gravis in 2023.
Along with rapid response, said Dr. Howard, complement inhibitors and FcRn inhibitors offer a “hugely improved” side-effect profile. In phase 3 research, the most common side effects for both classes included headache, nausea, and diarrhea.4,6,7 Because complement inhibitors increase the risk of Neisseria infection, users require immunization against meningococcal infection (or concurrent antibiotic prophylaxis) while on complement inhibitors.
Insurance Issues
With many clinicians wondering which targeted therapy to choose for a particular patient, said Dr. Howard and Dr. Kaminski, the main obstacle to wider use of these treatments is payer attitudes and practices. “While many of us would like to see these drugs used earlier in the course of disease,” Dr. Howard explained, “there are numerous restrictions placed on the physician and the patient by whatever insurance the individual has.”
Dr. Kaminski said: “There’s a lot of variability among insurance companies regarding what is expected in terms of getting approval for a certain medication. It frustrates me, thinking this patient may do well with a complement inhibitor or an FcRn inhibitor, but it takes weeks to get them approved.”
Some of his patients have been approved for, and flourished on, complement inhibitors and FcRn inhibitors, he added, and then denied a second round of treatment. Dr. Kaminski said he does not know why these patients were denied, and every time he requests reevaluation, the decision is reversed. “That’s a significant time frame for me and my staff to manage.”
When asked what can be done to address high drug prices, Dr. Howard replied, “I have no idea. I’m not an advocate of high drug prices. But I don’t think people realize the cost of doing clinical trials, which is hundreds of millions of dollars, particularly in rare diseases.”
Presently, Dr. Howard said, FcRn inhibitors are used more frequently than complement inhibitors solely because of cost. Zilucoplan will be priced below existing complement inhibitors, although it is too soon to compare its price with those of FcRn inhibitors.
When eculizumab debuted, said Dr. Howard, it cost nearly $750,000 annually. “But if you look at the number of patients treated, the cost of the drug over this population is probably less than the cost for using a cholesterol-lowering agent to treat hyperlipidemia.”
An Institute for Clinical and Economic Review (ICER) report stated that eculizumab and efgartigimod should both cost less than $20,000 annually to meet commonly used cost-effectiveness thresholds.8 However, Dr. Howard said ICER used models based on common diseases and ignored the economic impact of patients’ losing fewer workdays and avoiding long-term immunosuppressant side effects such as diabetes and osteoporosis and related treatment costs. “We’ve got to start looking at total societal cost,” he said.
Leapfrogging Ahead
Not all the new drugs work in every indicated patient, Dr. Howard said. For example, up to 30% of patients do not respond to complement inhibitors. “We don’t understand why. It’s as if we have leapfrogged way ahead in terms of therapeutics, and now we have to go back and answer all the questions – the who, what, where, and why of an individual drug and its response in folks.”
In this climate, said Dr. Kaminski, heavy direct-to-consumer advertising of newer myasthenia gravis therapies creates complications. “My patients are highly excited to see, ‘that’s my disease being advertised on Jeopardy.’ ” Many patients are frustrated with the general lack of awareness regarding myasthenia gravis, he added. “But then I’ve had patients who clearly would never qualify for a certain medication getting mailings to their homes.”
Dr. Howard countered that broader awareness of myasthenia gravis can only help. “There’s increasing recognition of the disease, not only by patients, but to some extent, by the treating clinician. Patients are coming to our offices and saying, ‘am I a candidate for this new drug?’ It’s the responsibility of the clinician to decide.”
Individual physicians’ practice patterns vary greatly, said Dr. Kaminski, and very little quantitative data exist here. But based on personal communications, academic-center neurologists tend to use targeted treatments on patients who have failed conventional treatments.
Conversely, Dr. Howard said that, because community physicians rarely see myasthenia gravis, and targeted treatments remain relatively new, many of these providers rely on prednisone, azathioprine, and mycophenolate mofetil.
B-Cell Blockers in Development
Overall, said Dr. Howard, the field of myasthenia gravis treatment development is “very rich. And pharma’s interest in myasthenia has taken off like a rocket. It’s exceptionally gratifying to those of us who take care of these patients whose life is miserable” because of adverse effects and/or nonresponse to current drugs.
“In myasthenia,” added Dr. Kaminski, “we know that T cells are promoting the activity of these auto-reactive B cells.” Many drugs currently in phase 2 or 3 development aim to eliminate B cells or signaling between T and B cells, he said. “That’s where most of the drug development is.”
Leading candidates include telitacicept (Tai’ai, RemeGen), which is both a B-lymphocyte stimulator and a proliferation-inducing ligand. A phase 3 trial (NCT05737160) is ongoing, with primary completion expected in late 2026. A second phase 3 trial (NCT06456580) recently began enrolling. Dr. Howard said that, although early results warranted phase 3 analysis, telitacicept’s phase 2 trial was open label and lacked a placebo group.9 The latter is a critical concern because placebo response rates in myasthenia gravis trials average 35%-40%.
Combined with standard care, the FcRn inhibitor nipocalimab (Johnson & Johnson) enabled patients with AChR, MuSK, and/or LRP4 autoantibodies to improve by 4.70 points on the MG-ADL vs 3.25 points for placebo (P = .002) over 24 weeks in phase 3.10All FcRn inhibitors in development can broadly reduce autoantibody levels, said Dr. Howard. “But what role they will play in myasthenia gravis when they’re several years behind leaders in the field in terms of capturing market remains to be seen.”
Additionally, batoclimab (Immunovant/Harbour BioMed) showed positive topline results in phase 3, and an elevated rate of hypercholesterolemia in treated patients that was transient and consistent with previous research.11 Subsequent to efgartigimod, Dr. Howard said, FcRn inhibitors are full-size antibodies. “I believe that contributes to the adverse events that we see. Efgartigimod is a small FcRn fragment. That’s why it’s a cleaner drug, if you will.”
FcRn inhibitors require periodic retreatment. For example, said Dr. Howard, the ADAPT phase 3 trial of efgartigimod, on which he was lead investigator, employed a cyclic dosing schedule – 4 weeks’ treatment, then observation until patients needed retreatment — because patients demanded it.12 In clinical practice, some patients have gone more than 25 weeks before needing retreatment. One of his patients went beyond 40 weeks. “Others only get around 6-9 weeks. So patient choice again enters the decision-making process.”
Rituximab targets the CD20 protein on B cells nonspecifically, producing general immunosuppression. “That’s problematic in producing significant immunosuppression,” said Dr. Kaminski. Nevertheless, he said, rituximab is very effective for most patients with MuSK-specific MG, and its application to this indication has revealed differences between the MuSK subtype and AChR antibody–positive myasthenia. Specifically, MuSK antibody–positive patients have short-lived plasmablasts, which rituximab eliminates.13
Conversely, said Dr. Kaminski, patients with AChR antibody-positive myasthenia, especially long-term, likely have long-lived plasmablasts producing antibodies. This fact, and these patients’ lack of CD20, likely explain their poor response to rituximab.
A phase 3 trial (NCT04524273) of the CD19 blocker inebilizumab (Uplinza, Amgen) reached primary completion in May. Dr. Howard said that if topline results (unreleased at press time) prove positive, inebilizumab could replace rituximab in MG — provided payers do not reject inebilizumab because of cost.
Packed Early-Development Pipeline
Regarding early-stage projects, said Dr. Howard, the pipeline is packed with compounds that target various aspects of the immune system. “The real question with those is, what’s going to be the side effect profile? All of the trials are very early. We need bigger trials with much longer observation for safety, durability, and degree of efficacy.”
The next potential B cell–targeting game changer, he said, is chimeric antigen receptor (CAR) T cell–based therapy. In a phase 2b trial of Descartes-08 (Cartesian Therapeutics), 71% of treated patients experienced clinically meaningful improvement in MG Composite score at 3 months vs 25% for placebo.14
In early clinical trials, said Dr. Howard, patients treated with Descartes-08 — which uses autologous mRNA to target B-cell maturation antigen — have shown “exceptional improvement” lasting 20 or more months. Because the drug is not ingrained permanently into the genome, Descartes-08 avoids potentially severe side effects of DNA-targeting CAR T candidates. Dr. Howard hopes a phase 3 trial will commence around January 2025.
The tolerance approach exemplified by CNP-106 (COUR Pharmaceuticals) and a myasthenia gravis tolerogen (Toleranzia) seeks to prevent the immune system from recognizing and reacting to the NMJ abnormalities that produce myasthenia gravis, potentially providing a cure. “We look forward to those trials as they come online in the next 1-2 years,” said Dr. Howard.
Unmet Needs
Historically, neurologists believed that all myasthenia gravis symptoms stemmed from muscle fatigue — the more active the muscle, the weaker it gets. However, said Dr. Kaminski, some patients might lack measurable weakness but still complain of fatigue.
Elevated levels of cytokines such as interleukin (IL)–6 or IL-17 also can produce fatigue, he noted. “With the drugs we’re using, certainly the new ones, we’re not specifically targeting this fatigue phenomenon, which has been studied in a very limited fashion.”
In the RAISE-XT zilucoplan trial, participants experienced significant improvement in fatigue scores for up to 60 weeks.15 Although zilucoplan does not address fatigue directly, said Dr. Howard, improving myasthenia gravis overall helps reduce fatigue.
The Myasthenia Gravis Symptoms Patient Reported Outcome (MG Symptoms PRO), which Dr. Kaminski helped develop, includes questions designed to distinguish muscular fatigue from overall physical fatigue.16 “I’m very interested in some of the information that’s coming out on long COVID and its effect on muscle,” Dr. Kaminski added. “We might be able to learn from there that there’s still some pathology going on beyond the neuromuscular junction.”
What the field desperately needs, said Dr. Howard, are biomarkers to identify which patients will and will not respond to certain therapeutics. “We’re not there yet.” Such biomarkers are at least 3-7 years from becoming clinical reality.
Promising antibody-independent serum markers include circulating microRNAs. For example, miRNA-150-5p and miRNA-21-5p are elevated in generalized AChR-positive myasthenia gravis and early-onset myasthenia gravis (occurring before age 50) and decline after immunosuppression and thymectomy.17
Among diagnostic modalities for patients with seronegative myasthenia gravis, said Dr. Kaminski, single-fiber EMG is the most sensitive, at approximately 95%. “It’s not perfect.” Moreover, he said, performing this test accurately requires a highly experienced expert, which many treatment centers lack.
Presently, added Dr. Kaminski, orbital MRI is neither specific nor sensitive enough to be clinically useful. “One needs to be careful with these specialized tests that are published from the best laboratory in the world that does the test, and does it repetitively.” As the search for effective myasthenia gravis biomarkers continues, avoiding false-positive results is as important as avoiding false negatives.
References
1. Bubuioc AM et al. J Med Life. 2021 Jan-Mar;14(1):7-16. doi: 10.25122/jml-2020-0145.
2. Deenen JC et al. J Neuromuscul Dis. 2015;2(1):73-85. doi: 10.3233/JND-140045.
3. Kaminski HJ et al. J Clin Invest. 2024 Jun 17;134(12):e179742. doi: 10.1172/JCI179742.
4. Howard JF Jr et al. Lancet Neurol. 2017 Dec;16(12):976-986. doi: 10.1016/S1474-4422(17)30369-1.
5. Huda R. Front Immunol. 2020 Feb 21:11:240. doi: 10.3389/fimmu.2020.00240.
6. Howard JF Jr et al. Lancet Neurol. 2023 May;22(5):395-406. doi: 10.1016/S1474-4422(23)00080-7.
7. Vu T et al. NEJM Evid. 2022 May;1(5):EVIDoa2100066. doi: 10.1056/EVIDoa2100066.
8. Tice JA et al. October 20, 2021. https://icer.org/assessment/myasthenia-gravis/.
9. Yin J et al. Eur J Neurol. 2024 Aug;31(8):e16322. doi: 10.1111/ene.16322.
10. Antozzi C et al. EAN 2024, Abstract EPR-116. https://www.neurology.org/doi/10.1212/WNL.0000000000203660.
11. Yan C et al. JAMA Neurol. 2024 Mar 4;81(4):336-345. doi: 10.1001/jamaneurol.2024.0044.
12. Howard JF Jr et al. Lancet Neurol. 2021 Jul;20(7):526-536. doi: 10.1016/S1474-4422(21)00159-9.
13. Stathopoulos P et al. JCI Insight. 2017 Sep 7;2(17):e94263. doi: 10.1172/jci.insight.94263.
14. Cartesian Therapeutics. Cartesian Therapeutics announces positive topline results from phase 2b trial of Descartes-08 in patients with myasthenia gravis. 2024 Jul 2. https://ir.cartesiantherapeutics.com/news-releases/news-release-details/cartesian-therapeutics-announces-positive-topline-results-phase.
15. Howard JF Jr et al. Ther Adv Neurol Disord. 2024 Apr 17:17:17562864241243186. doi: 10.1177/17562864241243186.
16. Cleanthous S et al. Orphanet J Rare Dis. 2021 Oct 30;16(1):457. doi: 10.1186/s13023-021-02064-0.
17. Sabre L et al. Front Immunol. 2020 Mar 4:11:213. doi: 10.3389/fimmu.2020.00213.
Used appropriately, newer treatments can provide dramatic results faster and more safely than broad immunosuppressants. However, according to experts, payers’ willingness to cover costly new therapies remains a work in progress.
The availability of more effective treatments with fewer side effects has brought about a cultural shift, said James F. Howard, Jr, MD. “The physician’s goal now is for the patient to be symptom free with grade 1 or less adverse events. And patients are demanding freedom from all the side effects that our usual course of immune therapy produces.” Dr. Howard is professor of neurology, medicine and allied health and director of the Myasthenia Gravis Clinical Trials and Translational Research Program at the University of North Carolina at Chapel Hill.
The shift has been long in coming. Although myasthenia gravis was identified in the mid-1600s, it took more than 340 years to get the first drug approved specifically for the disorder.
Worldwide prevalence estimates vary widely, from less than 200,000 to 700,000 cases.1,2 Pathophysiologically, myasthenia gravis stems from autoimmune destruction of neuromuscular junctions (NMJs), which transmit motor neuron impulses to muscle fibers.1 Symptoms include variable skeletal muscle weakness that can range from mild and transient to life-threatening.
In approximately 80% of cases, autoimmune antibodies target the postsynaptic acetylcholine receptor (AChR). Additional autoimmune targets mainly include muscle-specific kinase (MuSK) and lipoprotein receptor-related protein 4 (LRP4). However, around 10% of patients are seronegative, lacking autoantibodies detectable through conventional radioimmunoassays. Clinical disease does not always correspond with circulating antibody levels, and pathogenesis may require cooperation between multiple autoantibodies attacking the same target.3 Around 10% of MG cases are associated with thymomas.
Among myasthenia gravis treatments, immunosuppressants typically take 4-10 months to begin working and 18-36 months for maximum benefit. “Our new targeted therapies work within 1-2 weeks, with maximum improvement occurring somewhere between 8 and 12 weeks,” Dr. Howard said. Quick onset makes these drugs well suited for primary therapy in recalcitrant myasthenia gravis or as bridges to standard immunotherapy. Targeted drugs also appear to provide effective rescue therapy, although head-to-head studies are needed.
Complement Inhibition
In AChR antibody–positive myasthenia gravis, autoantibody binding with the postsynaptic AChR receptor activates complement to attack postsynaptic neuronal membrane. Complement inhibitors approved to date block activation of the terminal complement protein C5.
For many patients, complement inhibitors deliver dramatic results. Henry J. Kaminski, MD, said that the first patient for whom he prescribed a complement inhibitor outside a clinical trial went from being miserable to traveling internationally within a month. Dr. Kaminski is Meta A. Neumann Professor of Neurology at George Washington University, Washington, DC.
Eculizumab (Soliris, Alexion), earned Food and Drug Administration (FDA) approval for myasthenia gravis in 2017. Week 26 results in the phase 3 REGAIN trial showed no significant difference in Myasthenia Gravis–Activities of Daily Living (MG-ADL) scores between treatment and placebo. However, said Dr. Howard, primary investigator on the study, the negative result was a statistical aberration stemming from the FDA’s requirement to use worst-rank analysis rather than absolute change scores. What got eculizumab approved were highly positive results in the overwhelming majority of secondary endpoints.4 Subsequently, the FDA had the manufacturer rewrite the package insert using common statistical methods, which yielded positive primary results.
Ravulizumab (Ultomiris, Alexion), approved for myasthenia gravis in 2022, reduces eculizumab’s twice-monthly intravenous dosing to every 2 months (after loading doses), with very similar efficacy. The newest complement inhibitor, zilucoplan (Zilbrysq, UCB), administered once daily subcutaneously, earned FDA approval in 2023. Daily subcutaneous dosing provides patient convenience, said Dr. Howard. Because the body does not clear this small molecule as it would a full-size antibody, it is the only complement inhibitor that can be combined with a fragment crystallizable neonatal receptor (FcRn) inhibitor.
FcRn Inhibition
The FcRn exists on the surface and intracellular vesicles of many cells, including B cells, but not T cells.5FcRn inhibitors block binding of circulating IgG antibodies to the FcRn, preventing their normal recycling, significantly reducing circulating antibodies within days of treatment.
Efgartigimod (Vyvgart, Argenx), earned FDA approval in intravenous form in 2021, followed by a subcutaneous formulation that includes hyaluronidase (Vyvgart Hytrulo) in 2023. Rozanolixizumab (Rystiggo, UCB) earned FDA approval for both AChR antibody–positive and MuSK antibody–positive myasthenia gravis in 2023.
Along with rapid response, said Dr. Howard, complement inhibitors and FcRn inhibitors offer a “hugely improved” side-effect profile. In phase 3 research, the most common side effects for both classes included headache, nausea, and diarrhea.4,6,7 Because complement inhibitors increase the risk of Neisseria infection, users require immunization against meningococcal infection (or concurrent antibiotic prophylaxis) while on complement inhibitors.
Insurance Issues
With many clinicians wondering which targeted therapy to choose for a particular patient, said Dr. Howard and Dr. Kaminski, the main obstacle to wider use of these treatments is payer attitudes and practices. “While many of us would like to see these drugs used earlier in the course of disease,” Dr. Howard explained, “there are numerous restrictions placed on the physician and the patient by whatever insurance the individual has.”
Dr. Kaminski said: “There’s a lot of variability among insurance companies regarding what is expected in terms of getting approval for a certain medication. It frustrates me, thinking this patient may do well with a complement inhibitor or an FcRn inhibitor, but it takes weeks to get them approved.”
Some of his patients have been approved for, and flourished on, complement inhibitors and FcRn inhibitors, he added, and then denied a second round of treatment. Dr. Kaminski said he does not know why these patients were denied, and every time he requests reevaluation, the decision is reversed. “That’s a significant time frame for me and my staff to manage.”
When asked what can be done to address high drug prices, Dr. Howard replied, “I have no idea. I’m not an advocate of high drug prices. But I don’t think people realize the cost of doing clinical trials, which is hundreds of millions of dollars, particularly in rare diseases.”
Presently, Dr. Howard said, FcRn inhibitors are used more frequently than complement inhibitors solely because of cost. Zilucoplan will be priced below existing complement inhibitors, although it is too soon to compare its price with those of FcRn inhibitors.
When eculizumab debuted, said Dr. Howard, it cost nearly $750,000 annually. “But if you look at the number of patients treated, the cost of the drug over this population is probably less than the cost for using a cholesterol-lowering agent to treat hyperlipidemia.”
An Institute for Clinical and Economic Review (ICER) report stated that eculizumab and efgartigimod should both cost less than $20,000 annually to meet commonly used cost-effectiveness thresholds.8 However, Dr. Howard said ICER used models based on common diseases and ignored the economic impact of patients’ losing fewer workdays and avoiding long-term immunosuppressant side effects such as diabetes and osteoporosis and related treatment costs. “We’ve got to start looking at total societal cost,” he said.
Leapfrogging Ahead
Not all the new drugs work in every indicated patient, Dr. Howard said. For example, up to 30% of patients do not respond to complement inhibitors. “We don’t understand why. It’s as if we have leapfrogged way ahead in terms of therapeutics, and now we have to go back and answer all the questions – the who, what, where, and why of an individual drug and its response in folks.”
In this climate, said Dr. Kaminski, heavy direct-to-consumer advertising of newer myasthenia gravis therapies creates complications. “My patients are highly excited to see, ‘that’s my disease being advertised on Jeopardy.’ ” Many patients are frustrated with the general lack of awareness regarding myasthenia gravis, he added. “But then I’ve had patients who clearly would never qualify for a certain medication getting mailings to their homes.”
Dr. Howard countered that broader awareness of myasthenia gravis can only help. “There’s increasing recognition of the disease, not only by patients, but to some extent, by the treating clinician. Patients are coming to our offices and saying, ‘am I a candidate for this new drug?’ It’s the responsibility of the clinician to decide.”
Individual physicians’ practice patterns vary greatly, said Dr. Kaminski, and very little quantitative data exist here. But based on personal communications, academic-center neurologists tend to use targeted treatments on patients who have failed conventional treatments.
Conversely, Dr. Howard said that, because community physicians rarely see myasthenia gravis, and targeted treatments remain relatively new, many of these providers rely on prednisone, azathioprine, and mycophenolate mofetil.
B-Cell Blockers in Development
Overall, said Dr. Howard, the field of myasthenia gravis treatment development is “very rich. And pharma’s interest in myasthenia has taken off like a rocket. It’s exceptionally gratifying to those of us who take care of these patients whose life is miserable” because of adverse effects and/or nonresponse to current drugs.
“In myasthenia,” added Dr. Kaminski, “we know that T cells are promoting the activity of these auto-reactive B cells.” Many drugs currently in phase 2 or 3 development aim to eliminate B cells or signaling between T and B cells, he said. “That’s where most of the drug development is.”
Leading candidates include telitacicept (Tai’ai, RemeGen), which is both a B-lymphocyte stimulator and a proliferation-inducing ligand. A phase 3 trial (NCT05737160) is ongoing, with primary completion expected in late 2026. A second phase 3 trial (NCT06456580) recently began enrolling. Dr. Howard said that, although early results warranted phase 3 analysis, telitacicept’s phase 2 trial was open label and lacked a placebo group.9 The latter is a critical concern because placebo response rates in myasthenia gravis trials average 35%-40%.
Combined with standard care, the FcRn inhibitor nipocalimab (Johnson & Johnson) enabled patients with AChR, MuSK, and/or LRP4 autoantibodies to improve by 4.70 points on the MG-ADL vs 3.25 points for placebo (P = .002) over 24 weeks in phase 3.10All FcRn inhibitors in development can broadly reduce autoantibody levels, said Dr. Howard. “But what role they will play in myasthenia gravis when they’re several years behind leaders in the field in terms of capturing market remains to be seen.”
Additionally, batoclimab (Immunovant/Harbour BioMed) showed positive topline results in phase 3, and an elevated rate of hypercholesterolemia in treated patients that was transient and consistent with previous research.11 Subsequent to efgartigimod, Dr. Howard said, FcRn inhibitors are full-size antibodies. “I believe that contributes to the adverse events that we see. Efgartigimod is a small FcRn fragment. That’s why it’s a cleaner drug, if you will.”
FcRn inhibitors require periodic retreatment. For example, said Dr. Howard, the ADAPT phase 3 trial of efgartigimod, on which he was lead investigator, employed a cyclic dosing schedule – 4 weeks’ treatment, then observation until patients needed retreatment — because patients demanded it.12 In clinical practice, some patients have gone more than 25 weeks before needing retreatment. One of his patients went beyond 40 weeks. “Others only get around 6-9 weeks. So patient choice again enters the decision-making process.”
Rituximab targets the CD20 protein on B cells nonspecifically, producing general immunosuppression. “That’s problematic in producing significant immunosuppression,” said Dr. Kaminski. Nevertheless, he said, rituximab is very effective for most patients with MuSK-specific MG, and its application to this indication has revealed differences between the MuSK subtype and AChR antibody–positive myasthenia. Specifically, MuSK antibody–positive patients have short-lived plasmablasts, which rituximab eliminates.13
Conversely, said Dr. Kaminski, patients with AChR antibody-positive myasthenia, especially long-term, likely have long-lived plasmablasts producing antibodies. This fact, and these patients’ lack of CD20, likely explain their poor response to rituximab.
A phase 3 trial (NCT04524273) of the CD19 blocker inebilizumab (Uplinza, Amgen) reached primary completion in May. Dr. Howard said that if topline results (unreleased at press time) prove positive, inebilizumab could replace rituximab in MG — provided payers do not reject inebilizumab because of cost.
Packed Early-Development Pipeline
Regarding early-stage projects, said Dr. Howard, the pipeline is packed with compounds that target various aspects of the immune system. “The real question with those is, what’s going to be the side effect profile? All of the trials are very early. We need bigger trials with much longer observation for safety, durability, and degree of efficacy.”
The next potential B cell–targeting game changer, he said, is chimeric antigen receptor (CAR) T cell–based therapy. In a phase 2b trial of Descartes-08 (Cartesian Therapeutics), 71% of treated patients experienced clinically meaningful improvement in MG Composite score at 3 months vs 25% for placebo.14
In early clinical trials, said Dr. Howard, patients treated with Descartes-08 — which uses autologous mRNA to target B-cell maturation antigen — have shown “exceptional improvement” lasting 20 or more months. Because the drug is not ingrained permanently into the genome, Descartes-08 avoids potentially severe side effects of DNA-targeting CAR T candidates. Dr. Howard hopes a phase 3 trial will commence around January 2025.
The tolerance approach exemplified by CNP-106 (COUR Pharmaceuticals) and a myasthenia gravis tolerogen (Toleranzia) seeks to prevent the immune system from recognizing and reacting to the NMJ abnormalities that produce myasthenia gravis, potentially providing a cure. “We look forward to those trials as they come online in the next 1-2 years,” said Dr. Howard.
Unmet Needs
Historically, neurologists believed that all myasthenia gravis symptoms stemmed from muscle fatigue — the more active the muscle, the weaker it gets. However, said Dr. Kaminski, some patients might lack measurable weakness but still complain of fatigue.
Elevated levels of cytokines such as interleukin (IL)–6 or IL-17 also can produce fatigue, he noted. “With the drugs we’re using, certainly the new ones, we’re not specifically targeting this fatigue phenomenon, which has been studied in a very limited fashion.”
In the RAISE-XT zilucoplan trial, participants experienced significant improvement in fatigue scores for up to 60 weeks.15 Although zilucoplan does not address fatigue directly, said Dr. Howard, improving myasthenia gravis overall helps reduce fatigue.
The Myasthenia Gravis Symptoms Patient Reported Outcome (MG Symptoms PRO), which Dr. Kaminski helped develop, includes questions designed to distinguish muscular fatigue from overall physical fatigue.16 “I’m very interested in some of the information that’s coming out on long COVID and its effect on muscle,” Dr. Kaminski added. “We might be able to learn from there that there’s still some pathology going on beyond the neuromuscular junction.”
What the field desperately needs, said Dr. Howard, are biomarkers to identify which patients will and will not respond to certain therapeutics. “We’re not there yet.” Such biomarkers are at least 3-7 years from becoming clinical reality.
Promising antibody-independent serum markers include circulating microRNAs. For example, miRNA-150-5p and miRNA-21-5p are elevated in generalized AChR-positive myasthenia gravis and early-onset myasthenia gravis (occurring before age 50) and decline after immunosuppression and thymectomy.17
Among diagnostic modalities for patients with seronegative myasthenia gravis, said Dr. Kaminski, single-fiber EMG is the most sensitive, at approximately 95%. “It’s not perfect.” Moreover, he said, performing this test accurately requires a highly experienced expert, which many treatment centers lack.
Presently, added Dr. Kaminski, orbital MRI is neither specific nor sensitive enough to be clinically useful. “One needs to be careful with these specialized tests that are published from the best laboratory in the world that does the test, and does it repetitively.” As the search for effective myasthenia gravis biomarkers continues, avoiding false-positive results is as important as avoiding false negatives.
References
1. Bubuioc AM et al. J Med Life. 2021 Jan-Mar;14(1):7-16. doi: 10.25122/jml-2020-0145.
2. Deenen JC et al. J Neuromuscul Dis. 2015;2(1):73-85. doi: 10.3233/JND-140045.
3. Kaminski HJ et al. J Clin Invest. 2024 Jun 17;134(12):e179742. doi: 10.1172/JCI179742.
4. Howard JF Jr et al. Lancet Neurol. 2017 Dec;16(12):976-986. doi: 10.1016/S1474-4422(17)30369-1.
5. Huda R. Front Immunol. 2020 Feb 21:11:240. doi: 10.3389/fimmu.2020.00240.
6. Howard JF Jr et al. Lancet Neurol. 2023 May;22(5):395-406. doi: 10.1016/S1474-4422(23)00080-7.
7. Vu T et al. NEJM Evid. 2022 May;1(5):EVIDoa2100066. doi: 10.1056/EVIDoa2100066.
8. Tice JA et al. October 20, 2021. https://icer.org/assessment/myasthenia-gravis/.
9. Yin J et al. Eur J Neurol. 2024 Aug;31(8):e16322. doi: 10.1111/ene.16322.
10. Antozzi C et al. EAN 2024, Abstract EPR-116. https://www.neurology.org/doi/10.1212/WNL.0000000000203660.
11. Yan C et al. JAMA Neurol. 2024 Mar 4;81(4):336-345. doi: 10.1001/jamaneurol.2024.0044.
12. Howard JF Jr et al. Lancet Neurol. 2021 Jul;20(7):526-536. doi: 10.1016/S1474-4422(21)00159-9.
13. Stathopoulos P et al. JCI Insight. 2017 Sep 7;2(17):e94263. doi: 10.1172/jci.insight.94263.
14. Cartesian Therapeutics. Cartesian Therapeutics announces positive topline results from phase 2b trial of Descartes-08 in patients with myasthenia gravis. 2024 Jul 2. https://ir.cartesiantherapeutics.com/news-releases/news-release-details/cartesian-therapeutics-announces-positive-topline-results-phase.
15. Howard JF Jr et al. Ther Adv Neurol Disord. 2024 Apr 17:17:17562864241243186. doi: 10.1177/17562864241243186.
16. Cleanthous S et al. Orphanet J Rare Dis. 2021 Oct 30;16(1):457. doi: 10.1186/s13023-021-02064-0.
17. Sabre L et al. Front Immunol. 2020 Mar 4:11:213. doi: 10.3389/fimmu.2020.00213.
Untangling CIDP
Chronic inflammatory demyelinating polyradiculoneuropathy, or CIDP, is a rare immune-mediated nerve disorder characterized by progressive weakness and sensory impairment in the arms and legs, the result of an autoimmune attack on myelin.
Though some clustering of cases may occur in families, and susceptibility genes have been found, it is not considered a genetic disease. It can strike patients of either sex at any age, though most cases will occur in or after midlife.
Complicating matters further, CIDP has several variants whose symptoms differ from classical presentations.
Many patients who do not have CIDP end up being treated for it, and many CIDP patients experience delays to diagnosis and treatment that can potentially result in greater nerve damage and worse outcomes.
The good news, CIDP experts say, is that the last few years have seen important advances in diagnosis and treatment – including comprehensive new clinical guidelines and the June 2024 approval by the Food and Drug Administration of a new treatment, efgartigimod alfa and hyaluronidase-qvfc (Vyvgart, argenx). This antibody fragment represents the first non-steroid, non-immunoglobulin option for CIDP.
Despite the difficulties of recruiting patients with a tough-to-confirm disease that affects between 2 and 9 of every 100,000 people, according to the GPS-CIDP Foundation clinical trials have been successfully carried out in CIDP, and new ones continue to recruit. The experimental therapies being explored are based on a wide range of proposed disease pathways.
“It’s a very exciting time,” said Jeffrey Allen, MD, a neurologist at the University of Minnesota, Minneapolis, one of three CIDP experts who spoke about this challenging but treatable syndrome, its diagnosis and management, and the research questions that they hope to see answered.
Refining Diagnosis
In classical or typical CIDP, which accounts for most cases, patients present with progressive weakness and numbness that affects the arms and legs symmetrically, with the weakness being both proximal and distal. The disease usually evolves over a period of months, which helps distinguish it from Gullain-Barré syndrome, whose onset is more sudden and progression is less than 4 weeks.
CIDP was first described in the 1970s, and since that time more than a dozen sets of diagnostic criteria have been published. Starting about a decade ago, Dr. Allen and neurologist Richard Lewis, MD, of Cedars-Sinai Medical Center in Los Angeles, California, helped launch an effort to improve them.
“Experts in the field who were seeing patients with CIDP recognized that a lot of referrals coming to them were of people who actually didn’t have it, or they had the disease and were treated for it but didn’t need to be on treatment, or their treatment was very unconventional,” Dr. Allen said. “We wanted to try to put some data behind that.” In 2015 Dr. Allen and Dr. Lewis published a paper that found that nearly half of patients referred with a diagnosis of CIDP failed to meet basic diagnostic requirements.
Erroneous interpretation of nerve conduction studies “was a significant factor” contributing to the misdiagnoses, Dr. Lewis said. And another major problem was that patients’ response to standard treatment with intravenous immunoglobulins (current treatments have also come to include subcutaneous immunoglobulins) was not being measured objectively. Instead of evaluating patients using grip strength, walking tests, or other objective instruments, clinicians asked patients whether they felt better. “The problem is that IVIg makes people feel good,” Dr. Lewis said, “possibly by reducing normal inflammatory agents in the body.”
The 2015 paper caught the attention of neurologists and neuromuscular specialists worldwide, who reported similar problems with misdiagnosis. “And from there we did other work to try to dissect out what the more specific issues are,” Dr. Allen said. “The electrophysiology was a big one.”
Neurologist Nicholas Silvestri, MD, of the University at Buffalo in New York, one of the centers of excellence recognized by the CIDP-GBS Foundation, affirmed that nerve conduction studies, which essential to diagnosing CIDP, “are not as objective as we think they are. They’re very prone to user error and overinterpretation error. If they’re not performed appropriately, things can look like CIDP when they’re not. Very common forms of neuropathy, like diabetic neuropathy, can be misinterpreted as CIDP.”
The Challenge of Variants
After their 2015 paper on diagnostic pitfalls, Dr. Lewis and Dr. Allen, along with colleagues in the United States and Europe, started looking deeper into outcome measures and how to better follow and track patients with CIDP. In 2021 they helped create the first comprehensive clinical guidelines for CIDP in over a decade.
Much of their effort focused on atypical presentations, or what are now called variants, of CIDP — people with predominantly distal disease, asymmetrical symptomology, focal symptoms, or exclusively motor or sensory symptoms. With classical CIDP, “we don’t really have a problem with misdiagnosis,” Dr. Lewis said. With variants, however, misdiagnoses are extremely common. The 2021 guidelines try to address this, proposing differential diagnoses for each of the variants and ways to investigate them.
The guidelines also removed a subgroup of patients previously included as having CIDP. These patients, who comprise about 10% of cases, have antibodies to components of the Node of Ranvier, part of the axonal membrane, and the paranodal myelin. The autoimmune nodopathies do not respond to treatment with immunoglobulins or steroids in the way classical CIDP and its variants do. However, many patients have seen success with the immunotherapy rituximab.
“CIDP is a syndrome, not one disease,” Dr. Lewis said. “So it has been difficult to get guidelines or criteria that are sensitive to all the different forms of the disease, and yet specific for the disease and not overlapping. The nodopathies were pulled out because they don’t respond to usual treatments for CIDP. Hopefully over the years we’ll have even more specific diagnoses and can split out more patients.”
A Need for Better Biomarkers
With the neuromuscular autoimmune disease myasthenia gravis, 85% of patients have antibodies against the muscle acetylcholine receptor (AChR). Another 6% will have antibodies against muscle-specific kinase (MuSK).
Antibody profiles have long guided treatment decisions in myasthenia gravis, with AChR-positive patients responding to corticosteroids, IVIg, complement inhibitors, and other agents. MuSK-positive myasthenia gravis patients, similar to people with autoimmune nodopathies, respond poorly to IVIg but can have dramatic responses when treated with B cell–depleting therapies like rituximab.
Antibodies to nodal proteins neurofascin-155 and contactin-1 have been shown to be involved with the nodopathies. Assays for these are now commercially available, and Dr. Allen recommended that clinicians seek them for patients with a more rapid course, with tremor and ataxia, or who do not respond to standard CIDP treatments.
Still, no dominant autoantibody has been identified for the majority of presentations, including classical presentations. “I suspect it’s a heterogeneous group of multiple antibodies causing the disease,” Dr. Silvestri said. “That may explain to an extent the different manifestations and the different responses to treatment.”
Dr. Lewis said he thinks that, while more antibodies are likely to be discovered in the coming years, “we’re still identifying fewer than 20% of CIDP patients by specific antibodies, so we have a long way to go.”
Promising Trial Landscape
“CIDP is a challenging disease to study because of the diagnostic issues,” Dr. Allen said. “We know that a [nontrivial] percentage of patients ... can go into a drug-free remission. They actually don’t need treatment during that time. We don’t have any way to measure that. And if you put them in a clinical trial, it’s difficult to measure changes in the trial if they didn’t need the drug in the first place.”
In the global ADHERE trail, which looked at efgartigimod alfa and hyaluronidase-qvfc in CIDP patients, the investigators, led by Dr. Allen and Dr. Lewis, challenged patients to be off therapy for 12 weeks and allowed only those with active disease to enroll. They also used an adjudication panel of CIDP experts to review the records of each patient to assure patients had CIDP.
If two experts on the panel independently agreed that it was CIDP, Dr. Lewis said, then patients were eligible for enrollment. “If they both said they weren’t CIDP, they were not eligible. And if there was an argument between the two of them, then a third adjudicator would come in.”
About half of patients screened (n = 221) ended up included, and adjudication panels are now used in most CIDP trials.
The trial saw a positive outcome for efgartigimod alfa and hyaluronidase-qvfc, an antibody fragment that targets neonatal Fc receptor (FcRn), as a way to reduce to levels of pathogenic IgG autoantibodies. (The treatment was previously approved for myasthenia gravis.) The fact that two thirds of participants in the trial responded pointed to the likelihood that most CIDP patients have an IgG-related disease, Dr. Lewis said.
Different types of therapies are now being investigated in CIDP, among them other FcRn-inhibiting drugs and drugs inhibiting complement. Results from these trials may shed more light on the pathophysiology of the disease, which Dr. Silvestri said would be welcome.
“If I can test for antibodies, I can make a more timely diagnosis,” he said. “I’m assuming that some people with CIDP have non–antibody-driven disease. And in those cases, I want to avoid using drugs like Vyvgart, which are targeting antibodies. I want to give them a different therapy.”
Management: A Delicate Dance
Since the 1990s, the standard of care for CIDP has been IVIg and steroids. Newer subcutaneous immunoglobulin products, which take less time to administer, may be more convenient for patients than traditional IVIg and mitigate some concerning side effects.
Efgartigimod alfa and hyaluronidase-qvfc now offers an entirely different option that, while too new for clinicians to have much experience with in CIDP, represents further convenience for patients, with dosing in one 90-second subcutaneous injection per week.
In general, the sooner people are diagnosed and on therapy, the better they are likely to do, with fewer risks of irreversible axonal loss and disability. Referring to CIDP centers of excellence can help speed a definitive diagnosis.
Some patients will see a complete or near-complete recovery, while others will not. “It’s important to be up front with patients about what the benefit of treatments are, what are the expectations of treatment, what we can potentially get back, and what’s unlikely to come back,” Dr. Allen said. “We know that irreversible deficits are not uncommon in folks with CIDP. Part of that is driven by how severe their disease is or how long they’ve had it.”
Good CIDP management, according to the 2021 guidelines, involves making periodic dose reductions or withdrawing therapies on a trial basis, because people can and do experience remission. “We don’t have any test that tells us if somebody needs treatment or not. So this is the best we can do right now,” Dr. Allen said.
This process can be anxiety provoking for patients. “In my practice, there are no surprises,” he said. “We don’t typically say, ‘we’re going to stop your treatment today.’ It’s a discussion with a lead up that’s usually many months long.”
Management of CIDP also requires discussions to elicit when and whether worsening is occurring, along with a clear sense, by both patient and clinician, of what constitutes worsening.
Serial nerve conduction studies are not very useful, Dr. Lewis said, but objective disability measures are and should be more broadly adopted. These include the Medical Research Council sumscore, a test of 12 muscles that can determine weakness; a hand grip test; and functional disability scales such as Inflammatory Rasch Overall Disability Scale and Inflammatory Neuropathy Cause and Treatment scale. All are quick to administer in the office, and some can be done by the patient at home, providing the clinician useful information between visits.
“We could do a better job with educating [clinicians] on the value of different outcome measures that can really quantify disease activity,” Dr. Allen said, and pointed to the GBS-CIDP Foundation centers of excellence, which exist in most regions of the United States, as an outstanding resource for anyone wanting to know more.
“The centers are really, really helpful when you’re trying to work through some of these issues,” he said.
Suggested Reading
Allen JA and Lewis RA. Neurology. 2015 Aug 11;85(6):498-504. doi: 10.1212/WNL.0000000000001833.
Allen J et al. Neurology. 2024;102(17_supplement_1). doi: 10.1212/WNL-.0000000000206324.
Van den Bergh PYK et al. J Peripher Nerv Syst. 2021 Sep;26(3):242-268. doi: 10.1111/jns.12455.
Chronic inflammatory demyelinating polyradiculoneuropathy, or CIDP, is a rare immune-mediated nerve disorder characterized by progressive weakness and sensory impairment in the arms and legs, the result of an autoimmune attack on myelin.
Though some clustering of cases may occur in families, and susceptibility genes have been found, it is not considered a genetic disease. It can strike patients of either sex at any age, though most cases will occur in or after midlife.
Complicating matters further, CIDP has several variants whose symptoms differ from classical presentations.
Many patients who do not have CIDP end up being treated for it, and many CIDP patients experience delays to diagnosis and treatment that can potentially result in greater nerve damage and worse outcomes.
The good news, CIDP experts say, is that the last few years have seen important advances in diagnosis and treatment – including comprehensive new clinical guidelines and the June 2024 approval by the Food and Drug Administration of a new treatment, efgartigimod alfa and hyaluronidase-qvfc (Vyvgart, argenx). This antibody fragment represents the first non-steroid, non-immunoglobulin option for CIDP.
Despite the difficulties of recruiting patients with a tough-to-confirm disease that affects between 2 and 9 of every 100,000 people, according to the GPS-CIDP Foundation clinical trials have been successfully carried out in CIDP, and new ones continue to recruit. The experimental therapies being explored are based on a wide range of proposed disease pathways.
“It’s a very exciting time,” said Jeffrey Allen, MD, a neurologist at the University of Minnesota, Minneapolis, one of three CIDP experts who spoke about this challenging but treatable syndrome, its diagnosis and management, and the research questions that they hope to see answered.
Refining Diagnosis
In classical or typical CIDP, which accounts for most cases, patients present with progressive weakness and numbness that affects the arms and legs symmetrically, with the weakness being both proximal and distal. The disease usually evolves over a period of months, which helps distinguish it from Gullain-Barré syndrome, whose onset is more sudden and progression is less than 4 weeks.
CIDP was first described in the 1970s, and since that time more than a dozen sets of diagnostic criteria have been published. Starting about a decade ago, Dr. Allen and neurologist Richard Lewis, MD, of Cedars-Sinai Medical Center in Los Angeles, California, helped launch an effort to improve them.
“Experts in the field who were seeing patients with CIDP recognized that a lot of referrals coming to them were of people who actually didn’t have it, or they had the disease and were treated for it but didn’t need to be on treatment, or their treatment was very unconventional,” Dr. Allen said. “We wanted to try to put some data behind that.” In 2015 Dr. Allen and Dr. Lewis published a paper that found that nearly half of patients referred with a diagnosis of CIDP failed to meet basic diagnostic requirements.
Erroneous interpretation of nerve conduction studies “was a significant factor” contributing to the misdiagnoses, Dr. Lewis said. And another major problem was that patients’ response to standard treatment with intravenous immunoglobulins (current treatments have also come to include subcutaneous immunoglobulins) was not being measured objectively. Instead of evaluating patients using grip strength, walking tests, or other objective instruments, clinicians asked patients whether they felt better. “The problem is that IVIg makes people feel good,” Dr. Lewis said, “possibly by reducing normal inflammatory agents in the body.”
The 2015 paper caught the attention of neurologists and neuromuscular specialists worldwide, who reported similar problems with misdiagnosis. “And from there we did other work to try to dissect out what the more specific issues are,” Dr. Allen said. “The electrophysiology was a big one.”
Neurologist Nicholas Silvestri, MD, of the University at Buffalo in New York, one of the centers of excellence recognized by the CIDP-GBS Foundation, affirmed that nerve conduction studies, which essential to diagnosing CIDP, “are not as objective as we think they are. They’re very prone to user error and overinterpretation error. If they’re not performed appropriately, things can look like CIDP when they’re not. Very common forms of neuropathy, like diabetic neuropathy, can be misinterpreted as CIDP.”
The Challenge of Variants
After their 2015 paper on diagnostic pitfalls, Dr. Lewis and Dr. Allen, along with colleagues in the United States and Europe, started looking deeper into outcome measures and how to better follow and track patients with CIDP. In 2021 they helped create the first comprehensive clinical guidelines for CIDP in over a decade.
Much of their effort focused on atypical presentations, or what are now called variants, of CIDP — people with predominantly distal disease, asymmetrical symptomology, focal symptoms, or exclusively motor or sensory symptoms. With classical CIDP, “we don’t really have a problem with misdiagnosis,” Dr. Lewis said. With variants, however, misdiagnoses are extremely common. The 2021 guidelines try to address this, proposing differential diagnoses for each of the variants and ways to investigate them.
The guidelines also removed a subgroup of patients previously included as having CIDP. These patients, who comprise about 10% of cases, have antibodies to components of the Node of Ranvier, part of the axonal membrane, and the paranodal myelin. The autoimmune nodopathies do not respond to treatment with immunoglobulins or steroids in the way classical CIDP and its variants do. However, many patients have seen success with the immunotherapy rituximab.
“CIDP is a syndrome, not one disease,” Dr. Lewis said. “So it has been difficult to get guidelines or criteria that are sensitive to all the different forms of the disease, and yet specific for the disease and not overlapping. The nodopathies were pulled out because they don’t respond to usual treatments for CIDP. Hopefully over the years we’ll have even more specific diagnoses and can split out more patients.”
A Need for Better Biomarkers
With the neuromuscular autoimmune disease myasthenia gravis, 85% of patients have antibodies against the muscle acetylcholine receptor (AChR). Another 6% will have antibodies against muscle-specific kinase (MuSK).
Antibody profiles have long guided treatment decisions in myasthenia gravis, with AChR-positive patients responding to corticosteroids, IVIg, complement inhibitors, and other agents. MuSK-positive myasthenia gravis patients, similar to people with autoimmune nodopathies, respond poorly to IVIg but can have dramatic responses when treated with B cell–depleting therapies like rituximab.
Antibodies to nodal proteins neurofascin-155 and contactin-1 have been shown to be involved with the nodopathies. Assays for these are now commercially available, and Dr. Allen recommended that clinicians seek them for patients with a more rapid course, with tremor and ataxia, or who do not respond to standard CIDP treatments.
Still, no dominant autoantibody has been identified for the majority of presentations, including classical presentations. “I suspect it’s a heterogeneous group of multiple antibodies causing the disease,” Dr. Silvestri said. “That may explain to an extent the different manifestations and the different responses to treatment.”
Dr. Lewis said he thinks that, while more antibodies are likely to be discovered in the coming years, “we’re still identifying fewer than 20% of CIDP patients by specific antibodies, so we have a long way to go.”
Promising Trial Landscape
“CIDP is a challenging disease to study because of the diagnostic issues,” Dr. Allen said. “We know that a [nontrivial] percentage of patients ... can go into a drug-free remission. They actually don’t need treatment during that time. We don’t have any way to measure that. And if you put them in a clinical trial, it’s difficult to measure changes in the trial if they didn’t need the drug in the first place.”
In the global ADHERE trail, which looked at efgartigimod alfa and hyaluronidase-qvfc in CIDP patients, the investigators, led by Dr. Allen and Dr. Lewis, challenged patients to be off therapy for 12 weeks and allowed only those with active disease to enroll. They also used an adjudication panel of CIDP experts to review the records of each patient to assure patients had CIDP.
If two experts on the panel independently agreed that it was CIDP, Dr. Lewis said, then patients were eligible for enrollment. “If they both said they weren’t CIDP, they were not eligible. And if there was an argument between the two of them, then a third adjudicator would come in.”
About half of patients screened (n = 221) ended up included, and adjudication panels are now used in most CIDP trials.
The trial saw a positive outcome for efgartigimod alfa and hyaluronidase-qvfc, an antibody fragment that targets neonatal Fc receptor (FcRn), as a way to reduce to levels of pathogenic IgG autoantibodies. (The treatment was previously approved for myasthenia gravis.) The fact that two thirds of participants in the trial responded pointed to the likelihood that most CIDP patients have an IgG-related disease, Dr. Lewis said.
Different types of therapies are now being investigated in CIDP, among them other FcRn-inhibiting drugs and drugs inhibiting complement. Results from these trials may shed more light on the pathophysiology of the disease, which Dr. Silvestri said would be welcome.
“If I can test for antibodies, I can make a more timely diagnosis,” he said. “I’m assuming that some people with CIDP have non–antibody-driven disease. And in those cases, I want to avoid using drugs like Vyvgart, which are targeting antibodies. I want to give them a different therapy.”
Management: A Delicate Dance
Since the 1990s, the standard of care for CIDP has been IVIg and steroids. Newer subcutaneous immunoglobulin products, which take less time to administer, may be more convenient for patients than traditional IVIg and mitigate some concerning side effects.
Efgartigimod alfa and hyaluronidase-qvfc now offers an entirely different option that, while too new for clinicians to have much experience with in CIDP, represents further convenience for patients, with dosing in one 90-second subcutaneous injection per week.
In general, the sooner people are diagnosed and on therapy, the better they are likely to do, with fewer risks of irreversible axonal loss and disability. Referring to CIDP centers of excellence can help speed a definitive diagnosis.
Some patients will see a complete or near-complete recovery, while others will not. “It’s important to be up front with patients about what the benefit of treatments are, what are the expectations of treatment, what we can potentially get back, and what’s unlikely to come back,” Dr. Allen said. “We know that irreversible deficits are not uncommon in folks with CIDP. Part of that is driven by how severe their disease is or how long they’ve had it.”
Good CIDP management, according to the 2021 guidelines, involves making periodic dose reductions or withdrawing therapies on a trial basis, because people can and do experience remission. “We don’t have any test that tells us if somebody needs treatment or not. So this is the best we can do right now,” Dr. Allen said.
This process can be anxiety provoking for patients. “In my practice, there are no surprises,” he said. “We don’t typically say, ‘we’re going to stop your treatment today.’ It’s a discussion with a lead up that’s usually many months long.”
Management of CIDP also requires discussions to elicit when and whether worsening is occurring, along with a clear sense, by both patient and clinician, of what constitutes worsening.
Serial nerve conduction studies are not very useful, Dr. Lewis said, but objective disability measures are and should be more broadly adopted. These include the Medical Research Council sumscore, a test of 12 muscles that can determine weakness; a hand grip test; and functional disability scales such as Inflammatory Rasch Overall Disability Scale and Inflammatory Neuropathy Cause and Treatment scale. All are quick to administer in the office, and some can be done by the patient at home, providing the clinician useful information between visits.
“We could do a better job with educating [clinicians] on the value of different outcome measures that can really quantify disease activity,” Dr. Allen said, and pointed to the GBS-CIDP Foundation centers of excellence, which exist in most regions of the United States, as an outstanding resource for anyone wanting to know more.
“The centers are really, really helpful when you’re trying to work through some of these issues,” he said.
Suggested Reading
Allen JA and Lewis RA. Neurology. 2015 Aug 11;85(6):498-504. doi: 10.1212/WNL.0000000000001833.
Allen J et al. Neurology. 2024;102(17_supplement_1). doi: 10.1212/WNL-.0000000000206324.
Van den Bergh PYK et al. J Peripher Nerv Syst. 2021 Sep;26(3):242-268. doi: 10.1111/jns.12455.
Chronic inflammatory demyelinating polyradiculoneuropathy, or CIDP, is a rare immune-mediated nerve disorder characterized by progressive weakness and sensory impairment in the arms and legs, the result of an autoimmune attack on myelin.
Though some clustering of cases may occur in families, and susceptibility genes have been found, it is not considered a genetic disease. It can strike patients of either sex at any age, though most cases will occur in or after midlife.
Complicating matters further, CIDP has several variants whose symptoms differ from classical presentations.
Many patients who do not have CIDP end up being treated for it, and many CIDP patients experience delays to diagnosis and treatment that can potentially result in greater nerve damage and worse outcomes.
The good news, CIDP experts say, is that the last few years have seen important advances in diagnosis and treatment – including comprehensive new clinical guidelines and the June 2024 approval by the Food and Drug Administration of a new treatment, efgartigimod alfa and hyaluronidase-qvfc (Vyvgart, argenx). This antibody fragment represents the first non-steroid, non-immunoglobulin option for CIDP.
Despite the difficulties of recruiting patients with a tough-to-confirm disease that affects between 2 and 9 of every 100,000 people, according to the GPS-CIDP Foundation clinical trials have been successfully carried out in CIDP, and new ones continue to recruit. The experimental therapies being explored are based on a wide range of proposed disease pathways.
“It’s a very exciting time,” said Jeffrey Allen, MD, a neurologist at the University of Minnesota, Minneapolis, one of three CIDP experts who spoke about this challenging but treatable syndrome, its diagnosis and management, and the research questions that they hope to see answered.
Refining Diagnosis
In classical or typical CIDP, which accounts for most cases, patients present with progressive weakness and numbness that affects the arms and legs symmetrically, with the weakness being both proximal and distal. The disease usually evolves over a period of months, which helps distinguish it from Gullain-Barré syndrome, whose onset is more sudden and progression is less than 4 weeks.
CIDP was first described in the 1970s, and since that time more than a dozen sets of diagnostic criteria have been published. Starting about a decade ago, Dr. Allen and neurologist Richard Lewis, MD, of Cedars-Sinai Medical Center in Los Angeles, California, helped launch an effort to improve them.
“Experts in the field who were seeing patients with CIDP recognized that a lot of referrals coming to them were of people who actually didn’t have it, or they had the disease and were treated for it but didn’t need to be on treatment, or their treatment was very unconventional,” Dr. Allen said. “We wanted to try to put some data behind that.” In 2015 Dr. Allen and Dr. Lewis published a paper that found that nearly half of patients referred with a diagnosis of CIDP failed to meet basic diagnostic requirements.
Erroneous interpretation of nerve conduction studies “was a significant factor” contributing to the misdiagnoses, Dr. Lewis said. And another major problem was that patients’ response to standard treatment with intravenous immunoglobulins (current treatments have also come to include subcutaneous immunoglobulins) was not being measured objectively. Instead of evaluating patients using grip strength, walking tests, or other objective instruments, clinicians asked patients whether they felt better. “The problem is that IVIg makes people feel good,” Dr. Lewis said, “possibly by reducing normal inflammatory agents in the body.”
The 2015 paper caught the attention of neurologists and neuromuscular specialists worldwide, who reported similar problems with misdiagnosis. “And from there we did other work to try to dissect out what the more specific issues are,” Dr. Allen said. “The electrophysiology was a big one.”
Neurologist Nicholas Silvestri, MD, of the University at Buffalo in New York, one of the centers of excellence recognized by the CIDP-GBS Foundation, affirmed that nerve conduction studies, which essential to diagnosing CIDP, “are not as objective as we think they are. They’re very prone to user error and overinterpretation error. If they’re not performed appropriately, things can look like CIDP when they’re not. Very common forms of neuropathy, like diabetic neuropathy, can be misinterpreted as CIDP.”
The Challenge of Variants
After their 2015 paper on diagnostic pitfalls, Dr. Lewis and Dr. Allen, along with colleagues in the United States and Europe, started looking deeper into outcome measures and how to better follow and track patients with CIDP. In 2021 they helped create the first comprehensive clinical guidelines for CIDP in over a decade.
Much of their effort focused on atypical presentations, or what are now called variants, of CIDP — people with predominantly distal disease, asymmetrical symptomology, focal symptoms, or exclusively motor or sensory symptoms. With classical CIDP, “we don’t really have a problem with misdiagnosis,” Dr. Lewis said. With variants, however, misdiagnoses are extremely common. The 2021 guidelines try to address this, proposing differential diagnoses for each of the variants and ways to investigate them.
The guidelines also removed a subgroup of patients previously included as having CIDP. These patients, who comprise about 10% of cases, have antibodies to components of the Node of Ranvier, part of the axonal membrane, and the paranodal myelin. The autoimmune nodopathies do not respond to treatment with immunoglobulins or steroids in the way classical CIDP and its variants do. However, many patients have seen success with the immunotherapy rituximab.
“CIDP is a syndrome, not one disease,” Dr. Lewis said. “So it has been difficult to get guidelines or criteria that are sensitive to all the different forms of the disease, and yet specific for the disease and not overlapping. The nodopathies were pulled out because they don’t respond to usual treatments for CIDP. Hopefully over the years we’ll have even more specific diagnoses and can split out more patients.”
A Need for Better Biomarkers
With the neuromuscular autoimmune disease myasthenia gravis, 85% of patients have antibodies against the muscle acetylcholine receptor (AChR). Another 6% will have antibodies against muscle-specific kinase (MuSK).
Antibody profiles have long guided treatment decisions in myasthenia gravis, with AChR-positive patients responding to corticosteroids, IVIg, complement inhibitors, and other agents. MuSK-positive myasthenia gravis patients, similar to people with autoimmune nodopathies, respond poorly to IVIg but can have dramatic responses when treated with B cell–depleting therapies like rituximab.
Antibodies to nodal proteins neurofascin-155 and contactin-1 have been shown to be involved with the nodopathies. Assays for these are now commercially available, and Dr. Allen recommended that clinicians seek them for patients with a more rapid course, with tremor and ataxia, or who do not respond to standard CIDP treatments.
Still, no dominant autoantibody has been identified for the majority of presentations, including classical presentations. “I suspect it’s a heterogeneous group of multiple antibodies causing the disease,” Dr. Silvestri said. “That may explain to an extent the different manifestations and the different responses to treatment.”
Dr. Lewis said he thinks that, while more antibodies are likely to be discovered in the coming years, “we’re still identifying fewer than 20% of CIDP patients by specific antibodies, so we have a long way to go.”
Promising Trial Landscape
“CIDP is a challenging disease to study because of the diagnostic issues,” Dr. Allen said. “We know that a [nontrivial] percentage of patients ... can go into a drug-free remission. They actually don’t need treatment during that time. We don’t have any way to measure that. And if you put them in a clinical trial, it’s difficult to measure changes in the trial if they didn’t need the drug in the first place.”
In the global ADHERE trail, which looked at efgartigimod alfa and hyaluronidase-qvfc in CIDP patients, the investigators, led by Dr. Allen and Dr. Lewis, challenged patients to be off therapy for 12 weeks and allowed only those with active disease to enroll. They also used an adjudication panel of CIDP experts to review the records of each patient to assure patients had CIDP.
If two experts on the panel independently agreed that it was CIDP, Dr. Lewis said, then patients were eligible for enrollment. “If they both said they weren’t CIDP, they were not eligible. And if there was an argument between the two of them, then a third adjudicator would come in.”
About half of patients screened (n = 221) ended up included, and adjudication panels are now used in most CIDP trials.
The trial saw a positive outcome for efgartigimod alfa and hyaluronidase-qvfc, an antibody fragment that targets neonatal Fc receptor (FcRn), as a way to reduce to levels of pathogenic IgG autoantibodies. (The treatment was previously approved for myasthenia gravis.) The fact that two thirds of participants in the trial responded pointed to the likelihood that most CIDP patients have an IgG-related disease, Dr. Lewis said.
Different types of therapies are now being investigated in CIDP, among them other FcRn-inhibiting drugs and drugs inhibiting complement. Results from these trials may shed more light on the pathophysiology of the disease, which Dr. Silvestri said would be welcome.
“If I can test for antibodies, I can make a more timely diagnosis,” he said. “I’m assuming that some people with CIDP have non–antibody-driven disease. And in those cases, I want to avoid using drugs like Vyvgart, which are targeting antibodies. I want to give them a different therapy.”
Management: A Delicate Dance
Since the 1990s, the standard of care for CIDP has been IVIg and steroids. Newer subcutaneous immunoglobulin products, which take less time to administer, may be more convenient for patients than traditional IVIg and mitigate some concerning side effects.
Efgartigimod alfa and hyaluronidase-qvfc now offers an entirely different option that, while too new for clinicians to have much experience with in CIDP, represents further convenience for patients, with dosing in one 90-second subcutaneous injection per week.
In general, the sooner people are diagnosed and on therapy, the better they are likely to do, with fewer risks of irreversible axonal loss and disability. Referring to CIDP centers of excellence can help speed a definitive diagnosis.
Some patients will see a complete or near-complete recovery, while others will not. “It’s important to be up front with patients about what the benefit of treatments are, what are the expectations of treatment, what we can potentially get back, and what’s unlikely to come back,” Dr. Allen said. “We know that irreversible deficits are not uncommon in folks with CIDP. Part of that is driven by how severe their disease is or how long they’ve had it.”
Good CIDP management, according to the 2021 guidelines, involves making periodic dose reductions or withdrawing therapies on a trial basis, because people can and do experience remission. “We don’t have any test that tells us if somebody needs treatment or not. So this is the best we can do right now,” Dr. Allen said.
This process can be anxiety provoking for patients. “In my practice, there are no surprises,” he said. “We don’t typically say, ‘we’re going to stop your treatment today.’ It’s a discussion with a lead up that’s usually many months long.”
Management of CIDP also requires discussions to elicit when and whether worsening is occurring, along with a clear sense, by both patient and clinician, of what constitutes worsening.
Serial nerve conduction studies are not very useful, Dr. Lewis said, but objective disability measures are and should be more broadly adopted. These include the Medical Research Council sumscore, a test of 12 muscles that can determine weakness; a hand grip test; and functional disability scales such as Inflammatory Rasch Overall Disability Scale and Inflammatory Neuropathy Cause and Treatment scale. All are quick to administer in the office, and some can be done by the patient at home, providing the clinician useful information between visits.
“We could do a better job with educating [clinicians] on the value of different outcome measures that can really quantify disease activity,” Dr. Allen said, and pointed to the GBS-CIDP Foundation centers of excellence, which exist in most regions of the United States, as an outstanding resource for anyone wanting to know more.
“The centers are really, really helpful when you’re trying to work through some of these issues,” he said.
Suggested Reading
Allen JA and Lewis RA. Neurology. 2015 Aug 11;85(6):498-504. doi: 10.1212/WNL.0000000000001833.
Allen J et al. Neurology. 2024;102(17_supplement_1). doi: 10.1212/WNL-.0000000000206324.
Van den Bergh PYK et al. J Peripher Nerv Syst. 2021 Sep;26(3):242-268. doi: 10.1111/jns.12455.
Neuromyelitis Optica: Historically Misdiagnosed — Now Demands Prompt Treatment
Urgency of treatment is something that many physicians may not fully appreciate when it comes to neuromyelitis optica (NMO), according to experts on this rare autoimmune demyelinating disorder. This may be partly due to its similar presentation to multiple sclerosis (MS), said Michael Levy, MD, PhD, associate professor, Harvard Medical School, research director, Division of Neuroimmunology & Neuroinfectious Disease, and director, Neuroimmunology Clinic and Research Laboratory, at Massachusetts General Hospital in Boston. But while the two conditions share many clinical characteristics, “immunologically, they are about as different as can be,” he warned.
The urgency of distinction is important because where MS is known to have a relatively gradual progression, NMO is now red-flagged to potentially cause rapid and irreversible damage. While the course of MS might be described as a slow burn, NMO should be treated like a wildfire.
“That message has gotten muddled, particularly because acute treatment in MS has never been shown to affect outcome,” said Jeffrey Bennett, MD, PhD, professor of neurology and ophthalmology at the University of Colorado School of Medicine, Aurora. In contrast, rapid diagnosis and treatment of NMO “means potentially preventing future devastating neurologic injury,” he said.
First described by Dr. Eugène Devic in 1894, and sometimes known as Devic’s disease, NMO is believed to have a prevalence that varies widely depending on ethnicity and gender. A recent report suggests a prevalence of approximately1/100,000 population among Whites with an annual incidence of less than 1/million in this population, while the prevalence is higher among East Asians (approximately 3.5/100,000), and may reach as high as 10/100,000 in Blacks.1 It has a high female-to-male ratio (up to 9:1) with a mean age of onset of about 40 years, although pediatric cases are described.
It has long been recognized that NMO lacks the “neurocerebritis” of MS, with inflammation predominant in the optic and spinal nerves, but it was not until 2004 that researchers at the Mayo Clinic identified serum aquaporin-4 immunoglobulin G antibodies (AQP4-IgG) that could reliably distinguish NMO from MS. In 2015, the international consensus diagnostic criteria for neuromyelitis optica2 cited core clinical characteristics required for patients with AQP4-IgG-positive NMO spectrum disorder (NMOSD) “including clinical syndromes or MRI findings related to optic nerve, spinal cord, area postrema, other brainstem, diencephalic, or cerebral presentations.” Rarely, NMO patients can be seronegative for AQP4-IgG, but are still considered to have NMOSD for which non-opticospinal clinical and MRI characteristics findings are described. MS patients testing negative for AQP4-IgG should also be tested for the related myelin oligodendrocyte glycoprotein antibody disease (MOGAD), which has a prevalence about four to five times greater than NMO, Dr. Bennett said.
Testing
Because both NMO and MOGAD can be identified by antibodies, they are less commonly misdiagnosed as MS compared to previously. But, prior to the identification of the AQP4-IgG antibody in 2004, the misdiagnosis rate of NMO was probably about 95% said Dr. Levy.
“Of course, before we had the antibody test or clinical criteria, we couldn’t confirm a diagnosis of NMO, so basically everyone had a diagnosis of MS, and after the antibody test became commercially available in 2005/2006, we could confirm the diagnosis, with our study in 2012 showing a much lower misdiagnosis rate of 30%.”3 More recently, the misdiagnosis rates are even lower, he added. A recent study out of Argentina found a rate of only 12%.4
The specificity and sensitivity of cell binding assay serum AQP4-IgG testing is roughly 99% and 90%, respectively, better than ELIZA testing (which has a sensitivity in the 60-65% range), said Dr. Bennett. “That’s why we highly emphasize to physicians, that if you have a suspicion for NMOSD you go to a cell binding assay, and make sure that where you’re sending the serum, the lab can do that procedure.” Still, because of the risk of false positives, he urges restraint in testing for the disorder in the absence of a high suspicion for it. “If you test a lot of people indiscriminately for a rare disorder, you get a lot of false positives because the actual true positives are a very small fraction of that group. So, even with a specificity of around 99% that means 1% of the people you test are falsely positive. And if you’re testing a group of people indiscriminately, then your true positives are less than 1% by far, so then most of the people that you pick up are not truly with disease.”
Acute Treatment
While misdiagnosis of NMO as MS is less common than previously, it is still a concern, not only because of the irreversible risks associated with delayed acute treatment, but also the risks of inappropriate preventive MS therapy, which could be harmful to patients with NMO.
Acute flare-ups of NMO and MOGAD are currently all treated with the same decades-old mainstays for acute MS — intravenous steroids and plasma exchange — but the approach is more aggressive. Retrospective studies have shown that, for NMO, plasma exchange has shown an increased likelihood of improvement versus steroids alone, said Dr. Bennett, but since time is of the essence, treatment should begin before a definitive diagnosis is confirmed.
“What’s limiting our patients is, number one, recognizing NMOSD when the attack is happening in your face. You’ve got to know, hey, this is NMOSD or I’m suspicious of NMOSD and hence, I need to treat it urgently because the outcome has a high probability of not being good. You’ve got to realize that this is NMOSD before the test comes back, because by the time it comes back positive in several days, you’re probably missing the optimal window to treat. The point is to know that the presentation in front of you, the MRI pictures in front of you, the laboratory tests that you might have done in terms of spinal fluid analysis, all highly suggest NMOSD. And so hence, I’m going to take the chance that I might be wrong, but I’m going to treat as if it is and wait for the test to come back.”
Realistically, the risks associated with this approach are minor compared with the potential benefit, Dr. Bennett said. “For plasma exchange, there’s the placement of the central line, and the complications that could happen from that. Plasma exchange can lead to metabolic ionic changes in the blood, fluid shifts that might have to be watched in the hospital setting.”
While waiting for diagnostic results, one clue that may emerge from acute treatment is recovery time. “The recovery from MOGAD attacks is really distinct,” said Dr. Levy. “They get better a lot faster. So, if they’re blind in the hospital, and 3 months later they can see again with treatment, that’s MOGAD.” On the other hand, comorbidities such as lupus strongly favor NMO, he added. And another underrecognized, unique symptom of NMO is that about 10% of people may present with protracted episodes of nausea, vomiting, or hiccups, added Dr. Bennett. “What’s important is not that the neurologist recognize this per se in the emergency department, because they’re not going to be called for that patient — the GI doctors will be called for that patient. But when you’re seeing a patient who may have another presentation: a spinal cord attack, a vision attack with optic neuritis, and you ask them simply ‘have you ever had an episode of protracted nausea, vomiting, or hiccups?’ — I can’t tell you how many times I can have someone say ‘that’s weird I was just in the ED 3 months ago for that.’ And then, I know exactly what’s going on.”
Prevention of Relapse
Treatment of NMO presents some particular challenges for clinicians because the old treatment, rituximab, an anti-CD20 monoclonal antibody which has been used since 2005, has been so affordable and successful. “It’s hard to get people off,” said Dr. Levy. “It’s still the most commonly used drug for NMO in the US, even though it’s not approved. It’s cheap enough, and so people get started on that as a treatment, and then they just continue it, even as an outpatient.”
But since 2019, four new FDA-approved therapies have entered the scene with even better efficacy: the anti-CD-19 targeted medication inebilizumab (Uplizna, Viela Bio, approved in 2020), which requires two 90-minute infusions per year; the interleukin-6 (IL-6) receptor inhibitor satralizumab (Enspryng, Roche, approved in 2020), which is administered subcutaneously once monthly; and the anti-complement C5 inhibitors eculizumab (Soliris, Alexion Pharmaceuticals, approved in 2019), and ravulizumab (Ultomiris, Alexion Pharmaceuticals, approved in 2024), which require infusions every 2 weeks or every 2 months, respectively.
Both experts point to compelling clinical evidence to prescribe the Food and Drug Administration–approved drugs for newly diagnosed NMO, and to switch existing patients from rituximab to the new drugs. “The data is pretty clear that there’s about a 35% failure rate with rituximab, as opposed to less than 5% with the new drugs,” explained Dr. Levy. But ironically, where insurance companies used to balk at covering rituximab because it was not FDA approved for NMO, they are now balking at the FDA-approved options because of the cost. “Even in an academic center, where we get a discount on the drugs, the biosimilar generic of rituximab costs about $890 per dose,” he said. “So overall, it’s less than $4,000 a year for rituximab. Compare that with the most expensive FDA-approved option, which is eculizumab. That’s $715,000 per year. And then the other three drugs are below that, but none are less than about $290,000 a year.”
Patients are also hesitant to switch from rituximab if they’ve been well-controlled on it. “There’s a process to it, and I always talk my patients through it, but I would say less than half make the switch,” said Dr. Levy. “Most people want to stay. It’s a whole different schedule, and mixing two drugs. Are you going to overlap and overly immune suppress? Is the insurance going to approve it? It becomes more complicated.”
“Insurance companies are sometimes inappropriately pushing physicians, asking for patients to fail rituximab before they’ll approve an FDA-approved drug, which is like playing doctor when they’re not a licensed physician,” added Dr. Bennett. “And I think that is absolutely inappropriate, especially in light of the fact that before there were approved drugs, insurance companies used to deny rituximab because it was ‘experimental’ and ‘too expensive’ — and now it’s a cheaper alternative.”
Requiring failure on rituximab is also unethical, given the potential for irreversible damage, Dr. Levy pointed out. “With NMO we don’t tolerate a failure. That’s also how the trials of the new drugs were done. It was considered unethical to have an outcome of annualized relapse rate, like we used to in MS, where we say, OK if you have two attacks a year, then the drug has failed. With NMO, one failure, one breakthrough, and that drug is worthless. We switch.”
A Wealth of Treatment Choices
Patients opting for an FDA-approved treatment now have a “wildly effective” array of new drugs, said Dr. Levy, but choosing can be difficult when each has its own set of advantages and disadvantages. “I have equal numbers of patients on all the drugs, and I show all the data to my patients: efficacy, safety, logistics, cost, and then I ask ‘What are your priorities? Which of these things that I say really rings with you? Is it the infusion schedule? Is it the efficacy? Is it the safety concern? Is it the cost? What are you most concerned about?’ And then we start to have the conversation that way. It’s a shared decision-making process.”
There is definitely an art to finding the best fit for each patient, agreed Dr. Bennett, “both with the urgency of controlling the disease, the particular patient in front of you, their ability to adhere to certain therapies, their ability to have access to infusions, or to self-inject, or to get transported to an infusion center or have access to home infusion.”
Patient empowerment in the decision is very important, added Dr. Levy. “When people make the decision on their own, they’re much more likely to be compliant, rather than me telling them they have to do this. And that’s why I think we haven’t had a single relapse on the new drugs. There have been switches because of intolerance, and cost, and all those issues, but not because of a breakthrough attack.”
Future
Looking ahead in the field, Dr. Bennett sees the biggest potential for improvement is in the management of acute attacks, which he describes as “a major treatment gap.” Although plasma exchange is immediately effective in limiting the amount of circulating pathogenic AQP4-IgG “there are other approaches that could be even more beneficial,” he said. “A promising strategy is to use drugs that act immediately on arms of the immune response that are directly injuring brain tissue. These include serum complement and cells such as neutrophils and natural killer cells that release destructive enzymes and inflammatory mediators,” he explained. “Complement inhibitors, such as the C5 inhibitors eculizumab and ravulizumab, currently approved for NMOSD relapse prevention, act immediately to inhibit complement-mediated tissue injury. Similarly, high doses of antihistamines could be used to rapidly stop the release of the destructive enzyme elastase from neutrophils and natural killer cells, while elastase inhibitors could be given to minimize cell injury. Direct clinical studies are needed to find both the optimal treatment window and regimen.”
References
1. Hor JY et al. Epidemiology of Neuromyelitis Optica Spectrum Disorder and Its Prevalence and Incidence Worldwide. Front Neurol. 2020 Jun 26:11:501. doi: 10.3389/fneur.2020.00501.
2. Wingerchuk DM et al. International Consensus Diagnostic Criteria for Neuromyelitis Optica Spectrum Disorders. Neurology. 2015 Jul 14;85(2):177-89. doi: 10.1212/WNL.0000000000001729.
3. Mealy MA et al. Epidemiology of Neuromyelitis Optica in the United States: A Multicenter Analysis. Arch Neurol. 2012 Sep;69(9):1176-80. doi: 10.1001/archneurol.2012.314.
4. Contentti EC et al. Frequency of NMOSD Misdiagnosis in a Cohort From Latin America: Impact and Evaluation of Different Contributors. Mult Scler. 2023 Feb;29(2):277-286. doi: 10.1177/13524585221136259.
Urgency of treatment is something that many physicians may not fully appreciate when it comes to neuromyelitis optica (NMO), according to experts on this rare autoimmune demyelinating disorder. This may be partly due to its similar presentation to multiple sclerosis (MS), said Michael Levy, MD, PhD, associate professor, Harvard Medical School, research director, Division of Neuroimmunology & Neuroinfectious Disease, and director, Neuroimmunology Clinic and Research Laboratory, at Massachusetts General Hospital in Boston. But while the two conditions share many clinical characteristics, “immunologically, they are about as different as can be,” he warned.
The urgency of distinction is important because where MS is known to have a relatively gradual progression, NMO is now red-flagged to potentially cause rapid and irreversible damage. While the course of MS might be described as a slow burn, NMO should be treated like a wildfire.
“That message has gotten muddled, particularly because acute treatment in MS has never been shown to affect outcome,” said Jeffrey Bennett, MD, PhD, professor of neurology and ophthalmology at the University of Colorado School of Medicine, Aurora. In contrast, rapid diagnosis and treatment of NMO “means potentially preventing future devastating neurologic injury,” he said.
First described by Dr. Eugène Devic in 1894, and sometimes known as Devic’s disease, NMO is believed to have a prevalence that varies widely depending on ethnicity and gender. A recent report suggests a prevalence of approximately1/100,000 population among Whites with an annual incidence of less than 1/million in this population, while the prevalence is higher among East Asians (approximately 3.5/100,000), and may reach as high as 10/100,000 in Blacks.1 It has a high female-to-male ratio (up to 9:1) with a mean age of onset of about 40 years, although pediatric cases are described.
It has long been recognized that NMO lacks the “neurocerebritis” of MS, with inflammation predominant in the optic and spinal nerves, but it was not until 2004 that researchers at the Mayo Clinic identified serum aquaporin-4 immunoglobulin G antibodies (AQP4-IgG) that could reliably distinguish NMO from MS. In 2015, the international consensus diagnostic criteria for neuromyelitis optica2 cited core clinical characteristics required for patients with AQP4-IgG-positive NMO spectrum disorder (NMOSD) “including clinical syndromes or MRI findings related to optic nerve, spinal cord, area postrema, other brainstem, diencephalic, or cerebral presentations.” Rarely, NMO patients can be seronegative for AQP4-IgG, but are still considered to have NMOSD for which non-opticospinal clinical and MRI characteristics findings are described. MS patients testing negative for AQP4-IgG should also be tested for the related myelin oligodendrocyte glycoprotein antibody disease (MOGAD), which has a prevalence about four to five times greater than NMO, Dr. Bennett said.
Testing
Because both NMO and MOGAD can be identified by antibodies, they are less commonly misdiagnosed as MS compared to previously. But, prior to the identification of the AQP4-IgG antibody in 2004, the misdiagnosis rate of NMO was probably about 95% said Dr. Levy.
“Of course, before we had the antibody test or clinical criteria, we couldn’t confirm a diagnosis of NMO, so basically everyone had a diagnosis of MS, and after the antibody test became commercially available in 2005/2006, we could confirm the diagnosis, with our study in 2012 showing a much lower misdiagnosis rate of 30%.”3 More recently, the misdiagnosis rates are even lower, he added. A recent study out of Argentina found a rate of only 12%.4
The specificity and sensitivity of cell binding assay serum AQP4-IgG testing is roughly 99% and 90%, respectively, better than ELIZA testing (which has a sensitivity in the 60-65% range), said Dr. Bennett. “That’s why we highly emphasize to physicians, that if you have a suspicion for NMOSD you go to a cell binding assay, and make sure that where you’re sending the serum, the lab can do that procedure.” Still, because of the risk of false positives, he urges restraint in testing for the disorder in the absence of a high suspicion for it. “If you test a lot of people indiscriminately for a rare disorder, you get a lot of false positives because the actual true positives are a very small fraction of that group. So, even with a specificity of around 99% that means 1% of the people you test are falsely positive. And if you’re testing a group of people indiscriminately, then your true positives are less than 1% by far, so then most of the people that you pick up are not truly with disease.”
Acute Treatment
While misdiagnosis of NMO as MS is less common than previously, it is still a concern, not only because of the irreversible risks associated with delayed acute treatment, but also the risks of inappropriate preventive MS therapy, which could be harmful to patients with NMO.
Acute flare-ups of NMO and MOGAD are currently all treated with the same decades-old mainstays for acute MS — intravenous steroids and plasma exchange — but the approach is more aggressive. Retrospective studies have shown that, for NMO, plasma exchange has shown an increased likelihood of improvement versus steroids alone, said Dr. Bennett, but since time is of the essence, treatment should begin before a definitive diagnosis is confirmed.
“What’s limiting our patients is, number one, recognizing NMOSD when the attack is happening in your face. You’ve got to know, hey, this is NMOSD or I’m suspicious of NMOSD and hence, I need to treat it urgently because the outcome has a high probability of not being good. You’ve got to realize that this is NMOSD before the test comes back, because by the time it comes back positive in several days, you’re probably missing the optimal window to treat. The point is to know that the presentation in front of you, the MRI pictures in front of you, the laboratory tests that you might have done in terms of spinal fluid analysis, all highly suggest NMOSD. And so hence, I’m going to take the chance that I might be wrong, but I’m going to treat as if it is and wait for the test to come back.”
Realistically, the risks associated with this approach are minor compared with the potential benefit, Dr. Bennett said. “For plasma exchange, there’s the placement of the central line, and the complications that could happen from that. Plasma exchange can lead to metabolic ionic changes in the blood, fluid shifts that might have to be watched in the hospital setting.”
While waiting for diagnostic results, one clue that may emerge from acute treatment is recovery time. “The recovery from MOGAD attacks is really distinct,” said Dr. Levy. “They get better a lot faster. So, if they’re blind in the hospital, and 3 months later they can see again with treatment, that’s MOGAD.” On the other hand, comorbidities such as lupus strongly favor NMO, he added. And another underrecognized, unique symptom of NMO is that about 10% of people may present with protracted episodes of nausea, vomiting, or hiccups, added Dr. Bennett. “What’s important is not that the neurologist recognize this per se in the emergency department, because they’re not going to be called for that patient — the GI doctors will be called for that patient. But when you’re seeing a patient who may have another presentation: a spinal cord attack, a vision attack with optic neuritis, and you ask them simply ‘have you ever had an episode of protracted nausea, vomiting, or hiccups?’ — I can’t tell you how many times I can have someone say ‘that’s weird I was just in the ED 3 months ago for that.’ And then, I know exactly what’s going on.”
Prevention of Relapse
Treatment of NMO presents some particular challenges for clinicians because the old treatment, rituximab, an anti-CD20 monoclonal antibody which has been used since 2005, has been so affordable and successful. “It’s hard to get people off,” said Dr. Levy. “It’s still the most commonly used drug for NMO in the US, even though it’s not approved. It’s cheap enough, and so people get started on that as a treatment, and then they just continue it, even as an outpatient.”
But since 2019, four new FDA-approved therapies have entered the scene with even better efficacy: the anti-CD-19 targeted medication inebilizumab (Uplizna, Viela Bio, approved in 2020), which requires two 90-minute infusions per year; the interleukin-6 (IL-6) receptor inhibitor satralizumab (Enspryng, Roche, approved in 2020), which is administered subcutaneously once monthly; and the anti-complement C5 inhibitors eculizumab (Soliris, Alexion Pharmaceuticals, approved in 2019), and ravulizumab (Ultomiris, Alexion Pharmaceuticals, approved in 2024), which require infusions every 2 weeks or every 2 months, respectively.
Both experts point to compelling clinical evidence to prescribe the Food and Drug Administration–approved drugs for newly diagnosed NMO, and to switch existing patients from rituximab to the new drugs. “The data is pretty clear that there’s about a 35% failure rate with rituximab, as opposed to less than 5% with the new drugs,” explained Dr. Levy. But ironically, where insurance companies used to balk at covering rituximab because it was not FDA approved for NMO, they are now balking at the FDA-approved options because of the cost. “Even in an academic center, where we get a discount on the drugs, the biosimilar generic of rituximab costs about $890 per dose,” he said. “So overall, it’s less than $4,000 a year for rituximab. Compare that with the most expensive FDA-approved option, which is eculizumab. That’s $715,000 per year. And then the other three drugs are below that, but none are less than about $290,000 a year.”
Patients are also hesitant to switch from rituximab if they’ve been well-controlled on it. “There’s a process to it, and I always talk my patients through it, but I would say less than half make the switch,” said Dr. Levy. “Most people want to stay. It’s a whole different schedule, and mixing two drugs. Are you going to overlap and overly immune suppress? Is the insurance going to approve it? It becomes more complicated.”
“Insurance companies are sometimes inappropriately pushing physicians, asking for patients to fail rituximab before they’ll approve an FDA-approved drug, which is like playing doctor when they’re not a licensed physician,” added Dr. Bennett. “And I think that is absolutely inappropriate, especially in light of the fact that before there were approved drugs, insurance companies used to deny rituximab because it was ‘experimental’ and ‘too expensive’ — and now it’s a cheaper alternative.”
Requiring failure on rituximab is also unethical, given the potential for irreversible damage, Dr. Levy pointed out. “With NMO we don’t tolerate a failure. That’s also how the trials of the new drugs were done. It was considered unethical to have an outcome of annualized relapse rate, like we used to in MS, where we say, OK if you have two attacks a year, then the drug has failed. With NMO, one failure, one breakthrough, and that drug is worthless. We switch.”
A Wealth of Treatment Choices
Patients opting for an FDA-approved treatment now have a “wildly effective” array of new drugs, said Dr. Levy, but choosing can be difficult when each has its own set of advantages and disadvantages. “I have equal numbers of patients on all the drugs, and I show all the data to my patients: efficacy, safety, logistics, cost, and then I ask ‘What are your priorities? Which of these things that I say really rings with you? Is it the infusion schedule? Is it the efficacy? Is it the safety concern? Is it the cost? What are you most concerned about?’ And then we start to have the conversation that way. It’s a shared decision-making process.”
There is definitely an art to finding the best fit for each patient, agreed Dr. Bennett, “both with the urgency of controlling the disease, the particular patient in front of you, their ability to adhere to certain therapies, their ability to have access to infusions, or to self-inject, or to get transported to an infusion center or have access to home infusion.”
Patient empowerment in the decision is very important, added Dr. Levy. “When people make the decision on their own, they’re much more likely to be compliant, rather than me telling them they have to do this. And that’s why I think we haven’t had a single relapse on the new drugs. There have been switches because of intolerance, and cost, and all those issues, but not because of a breakthrough attack.”
Future
Looking ahead in the field, Dr. Bennett sees the biggest potential for improvement is in the management of acute attacks, which he describes as “a major treatment gap.” Although plasma exchange is immediately effective in limiting the amount of circulating pathogenic AQP4-IgG “there are other approaches that could be even more beneficial,” he said. “A promising strategy is to use drugs that act immediately on arms of the immune response that are directly injuring brain tissue. These include serum complement and cells such as neutrophils and natural killer cells that release destructive enzymes and inflammatory mediators,” he explained. “Complement inhibitors, such as the C5 inhibitors eculizumab and ravulizumab, currently approved for NMOSD relapse prevention, act immediately to inhibit complement-mediated tissue injury. Similarly, high doses of antihistamines could be used to rapidly stop the release of the destructive enzyme elastase from neutrophils and natural killer cells, while elastase inhibitors could be given to minimize cell injury. Direct clinical studies are needed to find both the optimal treatment window and regimen.”
References
1. Hor JY et al. Epidemiology of Neuromyelitis Optica Spectrum Disorder and Its Prevalence and Incidence Worldwide. Front Neurol. 2020 Jun 26:11:501. doi: 10.3389/fneur.2020.00501.
2. Wingerchuk DM et al. International Consensus Diagnostic Criteria for Neuromyelitis Optica Spectrum Disorders. Neurology. 2015 Jul 14;85(2):177-89. doi: 10.1212/WNL.0000000000001729.
3. Mealy MA et al. Epidemiology of Neuromyelitis Optica in the United States: A Multicenter Analysis. Arch Neurol. 2012 Sep;69(9):1176-80. doi: 10.1001/archneurol.2012.314.
4. Contentti EC et al. Frequency of NMOSD Misdiagnosis in a Cohort From Latin America: Impact and Evaluation of Different Contributors. Mult Scler. 2023 Feb;29(2):277-286. doi: 10.1177/13524585221136259.
Urgency of treatment is something that many physicians may not fully appreciate when it comes to neuromyelitis optica (NMO), according to experts on this rare autoimmune demyelinating disorder. This may be partly due to its similar presentation to multiple sclerosis (MS), said Michael Levy, MD, PhD, associate professor, Harvard Medical School, research director, Division of Neuroimmunology & Neuroinfectious Disease, and director, Neuroimmunology Clinic and Research Laboratory, at Massachusetts General Hospital in Boston. But while the two conditions share many clinical characteristics, “immunologically, they are about as different as can be,” he warned.
The urgency of distinction is important because where MS is known to have a relatively gradual progression, NMO is now red-flagged to potentially cause rapid and irreversible damage. While the course of MS might be described as a slow burn, NMO should be treated like a wildfire.
“That message has gotten muddled, particularly because acute treatment in MS has never been shown to affect outcome,” said Jeffrey Bennett, MD, PhD, professor of neurology and ophthalmology at the University of Colorado School of Medicine, Aurora. In contrast, rapid diagnosis and treatment of NMO “means potentially preventing future devastating neurologic injury,” he said.
First described by Dr. Eugène Devic in 1894, and sometimes known as Devic’s disease, NMO is believed to have a prevalence that varies widely depending on ethnicity and gender. A recent report suggests a prevalence of approximately1/100,000 population among Whites with an annual incidence of less than 1/million in this population, while the prevalence is higher among East Asians (approximately 3.5/100,000), and may reach as high as 10/100,000 in Blacks.1 It has a high female-to-male ratio (up to 9:1) with a mean age of onset of about 40 years, although pediatric cases are described.
It has long been recognized that NMO lacks the “neurocerebritis” of MS, with inflammation predominant in the optic and spinal nerves, but it was not until 2004 that researchers at the Mayo Clinic identified serum aquaporin-4 immunoglobulin G antibodies (AQP4-IgG) that could reliably distinguish NMO from MS. In 2015, the international consensus diagnostic criteria for neuromyelitis optica2 cited core clinical characteristics required for patients with AQP4-IgG-positive NMO spectrum disorder (NMOSD) “including clinical syndromes or MRI findings related to optic nerve, spinal cord, area postrema, other brainstem, diencephalic, or cerebral presentations.” Rarely, NMO patients can be seronegative for AQP4-IgG, but are still considered to have NMOSD for which non-opticospinal clinical and MRI characteristics findings are described. MS patients testing negative for AQP4-IgG should also be tested for the related myelin oligodendrocyte glycoprotein antibody disease (MOGAD), which has a prevalence about four to five times greater than NMO, Dr. Bennett said.
Testing
Because both NMO and MOGAD can be identified by antibodies, they are less commonly misdiagnosed as MS compared to previously. But, prior to the identification of the AQP4-IgG antibody in 2004, the misdiagnosis rate of NMO was probably about 95% said Dr. Levy.
“Of course, before we had the antibody test or clinical criteria, we couldn’t confirm a diagnosis of NMO, so basically everyone had a diagnosis of MS, and after the antibody test became commercially available in 2005/2006, we could confirm the diagnosis, with our study in 2012 showing a much lower misdiagnosis rate of 30%.”3 More recently, the misdiagnosis rates are even lower, he added. A recent study out of Argentina found a rate of only 12%.4
The specificity and sensitivity of cell binding assay serum AQP4-IgG testing is roughly 99% and 90%, respectively, better than ELIZA testing (which has a sensitivity in the 60-65% range), said Dr. Bennett. “That’s why we highly emphasize to physicians, that if you have a suspicion for NMOSD you go to a cell binding assay, and make sure that where you’re sending the serum, the lab can do that procedure.” Still, because of the risk of false positives, he urges restraint in testing for the disorder in the absence of a high suspicion for it. “If you test a lot of people indiscriminately for a rare disorder, you get a lot of false positives because the actual true positives are a very small fraction of that group. So, even with a specificity of around 99% that means 1% of the people you test are falsely positive. And if you’re testing a group of people indiscriminately, then your true positives are less than 1% by far, so then most of the people that you pick up are not truly with disease.”
Acute Treatment
While misdiagnosis of NMO as MS is less common than previously, it is still a concern, not only because of the irreversible risks associated with delayed acute treatment, but also the risks of inappropriate preventive MS therapy, which could be harmful to patients with NMO.
Acute flare-ups of NMO and MOGAD are currently all treated with the same decades-old mainstays for acute MS — intravenous steroids and plasma exchange — but the approach is more aggressive. Retrospective studies have shown that, for NMO, plasma exchange has shown an increased likelihood of improvement versus steroids alone, said Dr. Bennett, but since time is of the essence, treatment should begin before a definitive diagnosis is confirmed.
“What’s limiting our patients is, number one, recognizing NMOSD when the attack is happening in your face. You’ve got to know, hey, this is NMOSD or I’m suspicious of NMOSD and hence, I need to treat it urgently because the outcome has a high probability of not being good. You’ve got to realize that this is NMOSD before the test comes back, because by the time it comes back positive in several days, you’re probably missing the optimal window to treat. The point is to know that the presentation in front of you, the MRI pictures in front of you, the laboratory tests that you might have done in terms of spinal fluid analysis, all highly suggest NMOSD. And so hence, I’m going to take the chance that I might be wrong, but I’m going to treat as if it is and wait for the test to come back.”
Realistically, the risks associated with this approach are minor compared with the potential benefit, Dr. Bennett said. “For plasma exchange, there’s the placement of the central line, and the complications that could happen from that. Plasma exchange can lead to metabolic ionic changes in the blood, fluid shifts that might have to be watched in the hospital setting.”
While waiting for diagnostic results, one clue that may emerge from acute treatment is recovery time. “The recovery from MOGAD attacks is really distinct,” said Dr. Levy. “They get better a lot faster. So, if they’re blind in the hospital, and 3 months later they can see again with treatment, that’s MOGAD.” On the other hand, comorbidities such as lupus strongly favor NMO, he added. And another underrecognized, unique symptom of NMO is that about 10% of people may present with protracted episodes of nausea, vomiting, or hiccups, added Dr. Bennett. “What’s important is not that the neurologist recognize this per se in the emergency department, because they’re not going to be called for that patient — the GI doctors will be called for that patient. But when you’re seeing a patient who may have another presentation: a spinal cord attack, a vision attack with optic neuritis, and you ask them simply ‘have you ever had an episode of protracted nausea, vomiting, or hiccups?’ — I can’t tell you how many times I can have someone say ‘that’s weird I was just in the ED 3 months ago for that.’ And then, I know exactly what’s going on.”
Prevention of Relapse
Treatment of NMO presents some particular challenges for clinicians because the old treatment, rituximab, an anti-CD20 monoclonal antibody which has been used since 2005, has been so affordable and successful. “It’s hard to get people off,” said Dr. Levy. “It’s still the most commonly used drug for NMO in the US, even though it’s not approved. It’s cheap enough, and so people get started on that as a treatment, and then they just continue it, even as an outpatient.”
But since 2019, four new FDA-approved therapies have entered the scene with even better efficacy: the anti-CD-19 targeted medication inebilizumab (Uplizna, Viela Bio, approved in 2020), which requires two 90-minute infusions per year; the interleukin-6 (IL-6) receptor inhibitor satralizumab (Enspryng, Roche, approved in 2020), which is administered subcutaneously once monthly; and the anti-complement C5 inhibitors eculizumab (Soliris, Alexion Pharmaceuticals, approved in 2019), and ravulizumab (Ultomiris, Alexion Pharmaceuticals, approved in 2024), which require infusions every 2 weeks or every 2 months, respectively.
Both experts point to compelling clinical evidence to prescribe the Food and Drug Administration–approved drugs for newly diagnosed NMO, and to switch existing patients from rituximab to the new drugs. “The data is pretty clear that there’s about a 35% failure rate with rituximab, as opposed to less than 5% with the new drugs,” explained Dr. Levy. But ironically, where insurance companies used to balk at covering rituximab because it was not FDA approved for NMO, they are now balking at the FDA-approved options because of the cost. “Even in an academic center, where we get a discount on the drugs, the biosimilar generic of rituximab costs about $890 per dose,” he said. “So overall, it’s less than $4,000 a year for rituximab. Compare that with the most expensive FDA-approved option, which is eculizumab. That’s $715,000 per year. And then the other three drugs are below that, but none are less than about $290,000 a year.”
Patients are also hesitant to switch from rituximab if they’ve been well-controlled on it. “There’s a process to it, and I always talk my patients through it, but I would say less than half make the switch,” said Dr. Levy. “Most people want to stay. It’s a whole different schedule, and mixing two drugs. Are you going to overlap and overly immune suppress? Is the insurance going to approve it? It becomes more complicated.”
“Insurance companies are sometimes inappropriately pushing physicians, asking for patients to fail rituximab before they’ll approve an FDA-approved drug, which is like playing doctor when they’re not a licensed physician,” added Dr. Bennett. “And I think that is absolutely inappropriate, especially in light of the fact that before there were approved drugs, insurance companies used to deny rituximab because it was ‘experimental’ and ‘too expensive’ — and now it’s a cheaper alternative.”
Requiring failure on rituximab is also unethical, given the potential for irreversible damage, Dr. Levy pointed out. “With NMO we don’t tolerate a failure. That’s also how the trials of the new drugs were done. It was considered unethical to have an outcome of annualized relapse rate, like we used to in MS, where we say, OK if you have two attacks a year, then the drug has failed. With NMO, one failure, one breakthrough, and that drug is worthless. We switch.”
A Wealth of Treatment Choices
Patients opting for an FDA-approved treatment now have a “wildly effective” array of new drugs, said Dr. Levy, but choosing can be difficult when each has its own set of advantages and disadvantages. “I have equal numbers of patients on all the drugs, and I show all the data to my patients: efficacy, safety, logistics, cost, and then I ask ‘What are your priorities? Which of these things that I say really rings with you? Is it the infusion schedule? Is it the efficacy? Is it the safety concern? Is it the cost? What are you most concerned about?’ And then we start to have the conversation that way. It’s a shared decision-making process.”
There is definitely an art to finding the best fit for each patient, agreed Dr. Bennett, “both with the urgency of controlling the disease, the particular patient in front of you, their ability to adhere to certain therapies, their ability to have access to infusions, or to self-inject, or to get transported to an infusion center or have access to home infusion.”
Patient empowerment in the decision is very important, added Dr. Levy. “When people make the decision on their own, they’re much more likely to be compliant, rather than me telling them they have to do this. And that’s why I think we haven’t had a single relapse on the new drugs. There have been switches because of intolerance, and cost, and all those issues, but not because of a breakthrough attack.”
Future
Looking ahead in the field, Dr. Bennett sees the biggest potential for improvement is in the management of acute attacks, which he describes as “a major treatment gap.” Although plasma exchange is immediately effective in limiting the amount of circulating pathogenic AQP4-IgG “there are other approaches that could be even more beneficial,” he said. “A promising strategy is to use drugs that act immediately on arms of the immune response that are directly injuring brain tissue. These include serum complement and cells such as neutrophils and natural killer cells that release destructive enzymes and inflammatory mediators,” he explained. “Complement inhibitors, such as the C5 inhibitors eculizumab and ravulizumab, currently approved for NMOSD relapse prevention, act immediately to inhibit complement-mediated tissue injury. Similarly, high doses of antihistamines could be used to rapidly stop the release of the destructive enzyme elastase from neutrophils and natural killer cells, while elastase inhibitors could be given to minimize cell injury. Direct clinical studies are needed to find both the optimal treatment window and regimen.”
References
1. Hor JY et al. Epidemiology of Neuromyelitis Optica Spectrum Disorder and Its Prevalence and Incidence Worldwide. Front Neurol. 2020 Jun 26:11:501. doi: 10.3389/fneur.2020.00501.
2. Wingerchuk DM et al. International Consensus Diagnostic Criteria for Neuromyelitis Optica Spectrum Disorders. Neurology. 2015 Jul 14;85(2):177-89. doi: 10.1212/WNL.0000000000001729.
3. Mealy MA et al. Epidemiology of Neuromyelitis Optica in the United States: A Multicenter Analysis. Arch Neurol. 2012 Sep;69(9):1176-80. doi: 10.1001/archneurol.2012.314.
4. Contentti EC et al. Frequency of NMOSD Misdiagnosis in a Cohort From Latin America: Impact and Evaluation of Different Contributors. Mult Scler. 2023 Feb;29(2):277-286. doi: 10.1177/13524585221136259.
Diagnosing and Managing Duchenne Muscular Dystrophy: Tips for Practicing Clinicians
Duchenne muscular dystrophy (DMD) is a severe progressive inherited disease characterized by muscle wasting and ultimately culminating in death. It’s a common enough neuromuscular disorder that pediatricians and family practice physicians are likely to see at least a couple of patients with DMD over the course of their career,” John Brandsema, MD, Neuromuscular Section Head, Division of Neurology, Children’s Hospital of Philadelphia in Pennsylvania, said in an interview. Healthcare providers should therefore be familiar with the disorder so as to provide timely diagnosis and early intervention as well as practical and emotional support to the patient and family/caregivers as they traverse the challenging and often heartbreaking journey with this condition.
Pathophysiology and Disease Trajectory
DMD is caused by pathogenic variants in the X-linked DMD gene, leading to reduction in dystrophin, a protein that serves as a cytoskeletal integrator, stabilizing the plasma membrane of striated muscle cells. Dystrophin is critical for muscle membrane stability.2 In particular, mutations in the gene that encodes for dystrophin lead to dysfunction in Dp427m, which is the muscle isoform of dystrophin.3,4
DMD is one of several types of muscular dystrophies. All are progressive disorders. Over time, healthy muscle fibers disappear and are replaced by fibrotic tissue and fat, making the muscles “less able to generate force for everyday activity.”2 Ultimately, the skeletal muscle dysfunction affects not only the patient’s day-to-day mobility but other systems as well. Most patients with DMD eventually die of cardiac and/or respiratory failure between the ages of 20 and 40 years, with a median life expectancy of 22 years — although children born after 1990 have a somewhat higher median life expectancy (28 years), because of the improving standard of care.3,5
Typically, DMD first presents with developmental delays and weakness in skeletal leg muscles. As the disease goes through stages of progression, it starts involving upper extremities and other systems. (Table 1)
Genetic Causes of DMD
The DMD gene, located on the X chromosome, encodes for the production of dystrophin. Variants of this gene result in the lack of dystrophin protein, leading in turn to muscle fiber degeneration and the progressive symptoms of DMD. Because of the gene’s location on the X chromosome, males (who don’t have a second copy of the X chromosome) cannot compensate for the mutated gene, which is why the disease affects male children. Females with this mutation are carriers and typically do not develop the same severity of symptoms, although they might have milder muscle cramps, weakness, and cardiac issues.3
A female carrier with DMD (or any other X-linked disorder) has a 25% chance to have a carrier daughter, a 25% change of having a noncarrier daughter, a 25% chance of having an affected son, and a 25% chance of having a nonaffected son. A male with the disorder will pass the mutated gene on to his daughters who then become carriers. He cannot pass the disorder on to his sons because males inherit only the Y chromosome from their fathers.3
Diagnosing DMD
“It can take as long as 1-3 years for a child to be diagnosed with DMD,” Dr. Brandsema said. “Parents typically have concerns and know that something is ‘off’ about their child and they’re sent to various specialists, but it usually takes time for an accurate diagnosis to be made.” The mean age at diagnosis of DMD is between ages 4 and 5 years.6
Early identification of infants at risk for developing DMD can help move the needle toward earlier diagnosis. Newborn screening for DMD has been researched and piloted in several programs.6 In 2023, DMD was nominated for inclusion in the Recommended Universal Screening Panel (RUSP) for universal newborn screening. But in May 2024, the advisory committee on Heritable Disorders in Newborns and Children decided to postpone the vote to include DMD in the RUSP, requesting additional information to ensure an evidence-based decision.
In the absence of universal newborn screening for DMD, alternative approaches have been proposed to reduce the delay in clinical diagnosis and specialist referral, including increasing awareness among healthcare providers (eg, pediatricians, pediatric neurologists, and primary care physicians).6
The National Task Force for Early Identification of Childhood Neuromuscular Disorders delineates the steps necessary to identify pediatric muscle weakness and signs of neuromuscular disease. Primary care providers are encouraged to engage in regular developmental surveillance. A surveillance aid lays out the timetable for recommended visits, typical developmental milestones, and components of surveillance. Clinical evaluation includes a detailed patient history, family history, and physical examination.
If a neuromuscular condition is suspected, laboratory work should include creatinine phosphokinase (CK).6 Elevated serum CK points to leakage of CK through the muscle membrane, suggesting muscle damage. If CK is elevated, genetic testing should be performed; and, if negative, it should be followed by genetic sequencing that tests for small-scale mutations in the DMD gene. If that test is negative, a muscle biopsy should be performed to test for deep intronic mutations in the DMD gene.4
The diagnostic process and immediate steps after a confirmed DMD diagnosis is found in Figure 1.
Targeting Inflammation in DMD
Traditionally, corticosteroids have been the only available medical treatment for DMD and they remain a cornerstone of DMD management. A meta-analysis found “moderate evidence” that corticosteroid therapy improves muscle strength and function in the short term (12 months), and strength up to 2 years.10
The two most common corticosteroids for DMD are prednisone and deflazacort. Deflazacort (Emflaza, PTC Therapeutics) was approved in 2017 to treat patients ages 5 years and older with DMD, subsequently expanded to 2 years and older. Deflazacort has been found to be more effective than prednisone in improving functional outcomes, delaying the onset of cardiomyopathy, and improving overall survival, with fewer adverse effects.11
In 2023, vamorolone (Agamree, Catalyst Pharmaceuticals) was approved by the Food and Drug Administration (FDA) to treat DMD patients (ages 2 years and older). Vamorolone is a dissociative steroidal anti-inflammatory that reduces bone morbidities and is regarded as a safer alternative than prednisone. A clinical trial comparing two doses of vamorolone with prednisone for 24 weeks found that vamorolone 6 mg/kg per day met the primary endpoint (time to stand velocity) and four sequential secondary motor function endpoints, with less bone morbidity, compared to prednisone.12 A more recent trial found improvements in motor outcomes at 48 weeks with a dose of 6 mg/kg per day of vamorolone. Bone morbidities of prednisone were reversed when the patient transitioned to vamorolone.13
“Steroid treatment has been proven to help, usually taken daily, although other schedules have been tried,” Dr. Brandsema said. However, all steroids are fraught with adverse effects and are suboptimal in the long term in reducing the disease burden.
The anti-inflammatory agent givinostat (Duvyzat, ITF Therapeutics), an oral histone deacetylase (HDAC) inhibitor, was approved in March 2024 for the treatment of DMD in patients 6 years of age and older. It is the first nonsteroidal drug to treat patients with all genetic variants of the disease, and it has a unique mechanism of action. Deficits in dystrophin can lead to increased HDAC activity in DMD, reducing the expression of genes involved in muscle regeneration. Givinostat therefore can help to counteract the pathogenic events downstream of dystrophin deficiency by inhibiting HDAC.14
Approval for givinostat was based on the phase 3 EPIDYS trial, which randomized 179 boys with DMD to receive either givinostat or placebo. Although results of a functional task worsened in both groups over the 12-month study period, the decline was significantly smaller with givinostat versus placebo. The most common adverse events were diarrhea and vomiting.14 Dr. Brandsema noted that monitoring of triglycerides and platelet count is required, as hypertriglyceridemia and thrombocytopenia can occur. This treatment was studied in tandem with corticosteroids as a combination approach to muscle stabilization.
New Pharmacotherapeutic Options: Exon-Skipping Agents
Today’s treatments have expanded beyond corticosteroids, with newer therapeutic options that include targeted exon-skipping therapies and, more recently, gene therapies. “These new treatment paradigms have changed the face of DMD treatment,” Dr. Brandsema said.
“Exon-skipping drugs in their current form have only a modest effect, but at least they’re a step in the right direction and a breakthrough, in terms of slowing disease progression,” Dr. Brandsema said.
Current exon-skipping agents use antisense phosphorodiamidate morpholino oligomers (PMOs) to restore a DMD open reading frame. Next-generation drugs called cell-penetrating peptide-conjugated PMOs (PPMOs) are being actively researched, Dr. Brandsema said. These agents have shown enhanced cellular uptake and more efficient dystrophin restoration, compared with unconjugated PMOs.15
There are currently four FDA-approved exon-skipping agents for DMD, all of which are administered via a weekly intravenous infusion: Casimersen (Amondys-45, SRP-4045), approved by the FDA in 2021; Eteplirsen (Exondys 51), approved in 2016; Golodirsen (Vyondys 53,SRP-4053), approved in 2019; and Vitolarsen (Viltepso), approved in 2020. They can be associated with multiple side effects, depending on the drug, including upper respiratory infection, fever, cough, rash, and gastrointestinal issues.16 These agents have the potential to help 30% of DMD patients, restoring low levels of dystrophin.16
Gene Transfer Therapies
Gene transfer therapies, a new class of agents, utilize a nonpathogenic viral vector (adeno-associated virus) to transfer specific genes to patients with DMD. Gene therapy involves overexpressing the micro-dystrophin gene to restore functional dystrophin expression.16
Multiple clinical trials of gene therapy are currently in progress. In 2023, delandistrogene moxeparvovec-rokl (Elevidys, Serepta) was granted accelerated FDA approval for ambulatory individuals with DMD between the ages of 4 and 5 years of age and a confirmed mutation in the DMD gene. It received expanded approval in June 2024 to include ambulatory and nonambulatory individuals aged 4 years and older with DMD and a confirmed mutation in the DMD gene (with the exception of exon 8 or 9 mutations).
The approval was based on preliminary data from two double-blind, placebo-controlled studies and two open-label studies, which enrolled a total of 218 male patients (including those who received placebo) with a confirmed disease-causing mutation in the DMD gene.
Delandistrogene moxeparvovec-rokl is delivered as a one-time infusion and has been associated with side effects and “a lot of potential issues,” Dr. Brandsema said. “We’ve seen cardiac effects, immune system effects, increased muscle inflammation and hepatic complications, and some people who became quite unwell were hospitalized for a long time.”
Fortunately, he added, “these seem to be rare but they do happen. Once the medication has been delivered, it’s permanently in the body, so you’re managing the side effects potentially on a long-term basis.”
It is critical to discuss the risks and benefits of this treatment with the family and caregivers and with the patient as well, if he old enough and able to participate in the decision-making progress. “We don’t want to give unrealistic expectations and we want people to be aware of the potential downside of this treatment,” he said. “This is a very complex discussion because the trajectory of the disease is so devastating and this treatment does hold out hope that other therapies don’t necessarily have.”
Nonpharmacologic Interventions
Physical therapy is a mainstay in DMD treatment, addressing protection of fragile muscles, preservation of strength, and prevention of muscle contractures.16 Given the respiratory impairments that occur with DMD progression, respiratory monitoring and therapy are essential; however, the number and type of evaluations and interventions vary with the stage of the disease, intensifying as the disease progresses.16 Similarly, cardiac monitoring should begin early, with patients screened for cardiac complications, and should intensify through the stages of disease progression.16
Bone health is compromised in patients with DMD, both as a result of corticosteroid treatment and as part of the disease itself. Fractures may be asymptomatic and may go unnoticed. Thus, bone health surveillance and maintenance are critical components of DMD management.16
Patients with DMD often experience gastrointestinal issues. They may experience weight gain because of lack of mobility and corticosteroid use in early stages, or weight loss as a result of diet or fluid imbalance, low bone density, or dysphagia in later stages. Patients should be closely followed by a nutritionist, a gastroenterologist as needed, and a physical therapist.16
Psychosocial support “should be developed and implemented across the lifespan in a manner that promotes thinking about the future and sets expectations that individuals will actively participate in their care and daily activities.”9 This includes psychological care, neuropsychological evaluations, and educational support.
Assisting Patients and Families Through the DMD Journey
DMD care is best delivered in a multidisciplinary setting, where physicians of relevant specialties, physical and occupational therapists, nutritionists, social workers, and genetic counselors collaborate. At Children’s Hospital of Philadelphia, DMD care is delivered through this collaborative model.
Unfortunately, Dr. Brandsema said, many patients don’t have this type of multidisciplinary resource available. “One specialist, such as a pulmonologist or neurologist, might have to be the sole source of care.” Or parents may have to ferry their child to multiple specialists in disparate locations, placing extra stress on an already-stressed family system.
“It’s helpful to connect the family with a comprehensive care center, if possible,” Dr. Brandsema advised. If that’s not available, then he suggests recommending educational opportunities and resources through national organizations such as the Muscular Dystrophy Association; Parent Project MD; NORD; Friends, Family and Duchenne; and Cure Duchenne. Families and caregivers, along with affected individuals, can get education and support from people who understand the day-to-day reality of living with this disease.
One of the major challenges that families face is navigating the high cost of treating DMD, especially the new medications, Dr. Brandsema said. “The authorization process can be intensive and long, and the family may need to take an active role, together with the provider team, in advocating for the patient to get access.”
Among her many responsibilities, Ms. Kaschak engages in care coordination tasks and management, helps patients and caregivers understand care plans, and provides psychosocial support and education about the disease process. She assists families in completing paperwork and navigating specialty authorizations, helping families understand and navigate the complex insurance process. “My role is to bridge gaps in care,” she said.
Dr. Brandsema noted that it’s important for couples to receive genetic counseling if they’re planning to have multiple children because there is a 50% chance that their next boy will be affected. About two thirds of mothers with children who have DMD are carriers, but many are not aware of it. Receiving counseling will enable them to understand their own risks of health complications, as well as the risk to future children.
Managing DMD Across the Lifespan
Another dimension of DMD care is providing resources and help to young people with DMD as they transition into adulthood. “In the past, we had limited treatment and mortality typically took place in the early 20s, so there weren’t a lot of patients who were adults. But as medication options have expanded and management of cardiac and respiratory failure has improved, we see a more significant proportion of adults who require adult-appropriate clinics — or, at the very least, specialists who are conversant in care or can provide care across the lifespan,” Dr. Brandsema said.
The DMD Care Considerations Working Group provides recommendations regarding care across the lifespan,9 as does the Adult North Star Network, of Muscular Dystrophy UK.17,18
Dr. Brandsema emphasized that, despite their disability, many adults with DMD “still engage with the community, and live life to its fullest.” It is to be hoped that, with ongoing research, earlier diagnosis, and improved treatment options, the future will look bright for people with DMD.
Dr. Brandsema has served as a consultant for Audentes, AveXis/Novartis, Biogen, Cytokinetics, Dyne, Edgewise, Fibrogen, Genentech, Marathon, Momenta/Janssen, NS Pharma, Pfizer, PTC Therapeutics, Sarepta, Scholar Rock, Takeda, and WaVe. He is on the medical advisory council member for Cure SMA and is a site investigator for clinical trials with Alexion, Astellas, AveXis/Novartis, Biogen, Biohaven, Catabasis, CSL Behring, Cytokinetics, Dyne, Fibrogen, Genentech, Ionis, Lilly, ML Bio, Pfizer, PTC Therapeutics, Sarepta, Scholar Rock, Summit, and WaVe. Ms. Kaschak has nothing to disclose.
References
1. Venugopal V and Pavlakis S. Duchenne Muscular Dystrophy. 2023 Jul 10. In: StatPearls [Internet]. Treasure Island, Florida: StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK482346/.
2. Gao QQ and McNally EM. Compr Physiol. 2015 Jul 1;5(3):1223-39. doi: 10.1002/cphy.c140048.
3. Duan D et al. Nat Rev Dis Primers. 2021 Feb 18;7(1):13. doi: 10.1038/s41572-021-00248-3.
4. Aartsma-Rus A et al. J Pediatr. 2019 Jan:204:305-313.e14. doi: 10.1016/j.jpeds.2018.10.043.
5. Broomfield J et al. Neurology. 2021 Dec 7;97(23):e2304-e2314. doi: 10.1212/WNL.0000000000012910.
6. Mercuri E et al. Front Pediatr. 2023 Nov 10:11:1276144. doi: 10.1212/WNL.0000000000012910.
7. Birnkrant DJ et al. Lancet Neurol. 2018 Mar;17(3):251-267. doi: 10.1016/S1474-4422(18)30024-3.
8. Birnkrant DJ et al. Lancet Neurol. 2018 Apr;17(4):347-361. doi: 10.1016/S1474-4422(18)30025-5.
9. Birnkrant DJ et al. Lancet Neurol. 2018 May;17(5):445-455. doi: 10.1016/S1474-4422(18)30026-7.
10. Matthews E et al. Cochrane Database Syst Rev. 2016 May 5;2016(5):CD003725. doi: 10.1002/14651858.CD003725.pub4.
11. Bylo M et al. Ann Pharmacother. 2020 Aug;54(8):788-794. doi: 10.1177/1060028019900500.
12. Guglieri M et al. JAMA Neurol. 2022 Oct 1;79(10):1005-1014. doi: 10.1001/jamaneurol.2022.2480.
13. Dang UJ et al. Neurology. 2024 Mar 12;102(5):e208112. doi: 10.1212/WNL.0000000000208112.
14. Mercuri E et al. Lancet Neurol. 2024 Apr;23(4):393-403. doi: 10.1016/S1474-4422(24)00036-X.
15. Gushchina LV et al. Mol Ther Nucleic Acids. 2022 Nov 9:30:479-492. doi: 10.1016/j.omtn.2022.10.025.
16. Patterson G et al. Eur J Pharmacol. 2023 May 15:947:175675. doi: 10.1016/j.ejphar.2023.175675.
17. Quinlivan R et al. J Neuromuscul Dis. 2021;8(6):899-926. doi: 10.3233/JND-200609.
18. Narayan S et al. J Neuromuscul Dis. 2022;9(3):365-381. doi: 10.3233/JND-210707.
Duchenne muscular dystrophy (DMD) is a severe progressive inherited disease characterized by muscle wasting and ultimately culminating in death. It’s a common enough neuromuscular disorder that pediatricians and family practice physicians are likely to see at least a couple of patients with DMD over the course of their career,” John Brandsema, MD, Neuromuscular Section Head, Division of Neurology, Children’s Hospital of Philadelphia in Pennsylvania, said in an interview. Healthcare providers should therefore be familiar with the disorder so as to provide timely diagnosis and early intervention as well as practical and emotional support to the patient and family/caregivers as they traverse the challenging and often heartbreaking journey with this condition.
Pathophysiology and Disease Trajectory
DMD is caused by pathogenic variants in the X-linked DMD gene, leading to reduction in dystrophin, a protein that serves as a cytoskeletal integrator, stabilizing the plasma membrane of striated muscle cells. Dystrophin is critical for muscle membrane stability.2 In particular, mutations in the gene that encodes for dystrophin lead to dysfunction in Dp427m, which is the muscle isoform of dystrophin.3,4
DMD is one of several types of muscular dystrophies. All are progressive disorders. Over time, healthy muscle fibers disappear and are replaced by fibrotic tissue and fat, making the muscles “less able to generate force for everyday activity.”2 Ultimately, the skeletal muscle dysfunction affects not only the patient’s day-to-day mobility but other systems as well. Most patients with DMD eventually die of cardiac and/or respiratory failure between the ages of 20 and 40 years, with a median life expectancy of 22 years — although children born after 1990 have a somewhat higher median life expectancy (28 years), because of the improving standard of care.3,5
Typically, DMD first presents with developmental delays and weakness in skeletal leg muscles. As the disease goes through stages of progression, it starts involving upper extremities and other systems. (Table 1)
Genetic Causes of DMD
The DMD gene, located on the X chromosome, encodes for the production of dystrophin. Variants of this gene result in the lack of dystrophin protein, leading in turn to muscle fiber degeneration and the progressive symptoms of DMD. Because of the gene’s location on the X chromosome, males (who don’t have a second copy of the X chromosome) cannot compensate for the mutated gene, which is why the disease affects male children. Females with this mutation are carriers and typically do not develop the same severity of symptoms, although they might have milder muscle cramps, weakness, and cardiac issues.3
A female carrier with DMD (or any other X-linked disorder) has a 25% chance to have a carrier daughter, a 25% change of having a noncarrier daughter, a 25% chance of having an affected son, and a 25% chance of having a nonaffected son. A male with the disorder will pass the mutated gene on to his daughters who then become carriers. He cannot pass the disorder on to his sons because males inherit only the Y chromosome from their fathers.3
Diagnosing DMD
“It can take as long as 1-3 years for a child to be diagnosed with DMD,” Dr. Brandsema said. “Parents typically have concerns and know that something is ‘off’ about their child and they’re sent to various specialists, but it usually takes time for an accurate diagnosis to be made.” The mean age at diagnosis of DMD is between ages 4 and 5 years.6
Early identification of infants at risk for developing DMD can help move the needle toward earlier diagnosis. Newborn screening for DMD has been researched and piloted in several programs.6 In 2023, DMD was nominated for inclusion in the Recommended Universal Screening Panel (RUSP) for universal newborn screening. But in May 2024, the advisory committee on Heritable Disorders in Newborns and Children decided to postpone the vote to include DMD in the RUSP, requesting additional information to ensure an evidence-based decision.
In the absence of universal newborn screening for DMD, alternative approaches have been proposed to reduce the delay in clinical diagnosis and specialist referral, including increasing awareness among healthcare providers (eg, pediatricians, pediatric neurologists, and primary care physicians).6
The National Task Force for Early Identification of Childhood Neuromuscular Disorders delineates the steps necessary to identify pediatric muscle weakness and signs of neuromuscular disease. Primary care providers are encouraged to engage in regular developmental surveillance. A surveillance aid lays out the timetable for recommended visits, typical developmental milestones, and components of surveillance. Clinical evaluation includes a detailed patient history, family history, and physical examination.
If a neuromuscular condition is suspected, laboratory work should include creatinine phosphokinase (CK).6 Elevated serum CK points to leakage of CK through the muscle membrane, suggesting muscle damage. If CK is elevated, genetic testing should be performed; and, if negative, it should be followed by genetic sequencing that tests for small-scale mutations in the DMD gene. If that test is negative, a muscle biopsy should be performed to test for deep intronic mutations in the DMD gene.4
The diagnostic process and immediate steps after a confirmed DMD diagnosis is found in Figure 1.
Targeting Inflammation in DMD
Traditionally, corticosteroids have been the only available medical treatment for DMD and they remain a cornerstone of DMD management. A meta-analysis found “moderate evidence” that corticosteroid therapy improves muscle strength and function in the short term (12 months), and strength up to 2 years.10
The two most common corticosteroids for DMD are prednisone and deflazacort. Deflazacort (Emflaza, PTC Therapeutics) was approved in 2017 to treat patients ages 5 years and older with DMD, subsequently expanded to 2 years and older. Deflazacort has been found to be more effective than prednisone in improving functional outcomes, delaying the onset of cardiomyopathy, and improving overall survival, with fewer adverse effects.11
In 2023, vamorolone (Agamree, Catalyst Pharmaceuticals) was approved by the Food and Drug Administration (FDA) to treat DMD patients (ages 2 years and older). Vamorolone is a dissociative steroidal anti-inflammatory that reduces bone morbidities and is regarded as a safer alternative than prednisone. A clinical trial comparing two doses of vamorolone with prednisone for 24 weeks found that vamorolone 6 mg/kg per day met the primary endpoint (time to stand velocity) and four sequential secondary motor function endpoints, with less bone morbidity, compared to prednisone.12 A more recent trial found improvements in motor outcomes at 48 weeks with a dose of 6 mg/kg per day of vamorolone. Bone morbidities of prednisone were reversed when the patient transitioned to vamorolone.13
“Steroid treatment has been proven to help, usually taken daily, although other schedules have been tried,” Dr. Brandsema said. However, all steroids are fraught with adverse effects and are suboptimal in the long term in reducing the disease burden.
The anti-inflammatory agent givinostat (Duvyzat, ITF Therapeutics), an oral histone deacetylase (HDAC) inhibitor, was approved in March 2024 for the treatment of DMD in patients 6 years of age and older. It is the first nonsteroidal drug to treat patients with all genetic variants of the disease, and it has a unique mechanism of action. Deficits in dystrophin can lead to increased HDAC activity in DMD, reducing the expression of genes involved in muscle regeneration. Givinostat therefore can help to counteract the pathogenic events downstream of dystrophin deficiency by inhibiting HDAC.14
Approval for givinostat was based on the phase 3 EPIDYS trial, which randomized 179 boys with DMD to receive either givinostat or placebo. Although results of a functional task worsened in both groups over the 12-month study period, the decline was significantly smaller with givinostat versus placebo. The most common adverse events were diarrhea and vomiting.14 Dr. Brandsema noted that monitoring of triglycerides and platelet count is required, as hypertriglyceridemia and thrombocytopenia can occur. This treatment was studied in tandem with corticosteroids as a combination approach to muscle stabilization.
New Pharmacotherapeutic Options: Exon-Skipping Agents
Today’s treatments have expanded beyond corticosteroids, with newer therapeutic options that include targeted exon-skipping therapies and, more recently, gene therapies. “These new treatment paradigms have changed the face of DMD treatment,” Dr. Brandsema said.
“Exon-skipping drugs in their current form have only a modest effect, but at least they’re a step in the right direction and a breakthrough, in terms of slowing disease progression,” Dr. Brandsema said.
Current exon-skipping agents use antisense phosphorodiamidate morpholino oligomers (PMOs) to restore a DMD open reading frame. Next-generation drugs called cell-penetrating peptide-conjugated PMOs (PPMOs) are being actively researched, Dr. Brandsema said. These agents have shown enhanced cellular uptake and more efficient dystrophin restoration, compared with unconjugated PMOs.15
There are currently four FDA-approved exon-skipping agents for DMD, all of which are administered via a weekly intravenous infusion: Casimersen (Amondys-45, SRP-4045), approved by the FDA in 2021; Eteplirsen (Exondys 51), approved in 2016; Golodirsen (Vyondys 53,SRP-4053), approved in 2019; and Vitolarsen (Viltepso), approved in 2020. They can be associated with multiple side effects, depending on the drug, including upper respiratory infection, fever, cough, rash, and gastrointestinal issues.16 These agents have the potential to help 30% of DMD patients, restoring low levels of dystrophin.16
Gene Transfer Therapies
Gene transfer therapies, a new class of agents, utilize a nonpathogenic viral vector (adeno-associated virus) to transfer specific genes to patients with DMD. Gene therapy involves overexpressing the micro-dystrophin gene to restore functional dystrophin expression.16
Multiple clinical trials of gene therapy are currently in progress. In 2023, delandistrogene moxeparvovec-rokl (Elevidys, Serepta) was granted accelerated FDA approval for ambulatory individuals with DMD between the ages of 4 and 5 years of age and a confirmed mutation in the DMD gene. It received expanded approval in June 2024 to include ambulatory and nonambulatory individuals aged 4 years and older with DMD and a confirmed mutation in the DMD gene (with the exception of exon 8 or 9 mutations).
The approval was based on preliminary data from two double-blind, placebo-controlled studies and two open-label studies, which enrolled a total of 218 male patients (including those who received placebo) with a confirmed disease-causing mutation in the DMD gene.
Delandistrogene moxeparvovec-rokl is delivered as a one-time infusion and has been associated with side effects and “a lot of potential issues,” Dr. Brandsema said. “We’ve seen cardiac effects, immune system effects, increased muscle inflammation and hepatic complications, and some people who became quite unwell were hospitalized for a long time.”
Fortunately, he added, “these seem to be rare but they do happen. Once the medication has been delivered, it’s permanently in the body, so you’re managing the side effects potentially on a long-term basis.”
It is critical to discuss the risks and benefits of this treatment with the family and caregivers and with the patient as well, if he old enough and able to participate in the decision-making progress. “We don’t want to give unrealistic expectations and we want people to be aware of the potential downside of this treatment,” he said. “This is a very complex discussion because the trajectory of the disease is so devastating and this treatment does hold out hope that other therapies don’t necessarily have.”
Nonpharmacologic Interventions
Physical therapy is a mainstay in DMD treatment, addressing protection of fragile muscles, preservation of strength, and prevention of muscle contractures.16 Given the respiratory impairments that occur with DMD progression, respiratory monitoring and therapy are essential; however, the number and type of evaluations and interventions vary with the stage of the disease, intensifying as the disease progresses.16 Similarly, cardiac monitoring should begin early, with patients screened for cardiac complications, and should intensify through the stages of disease progression.16
Bone health is compromised in patients with DMD, both as a result of corticosteroid treatment and as part of the disease itself. Fractures may be asymptomatic and may go unnoticed. Thus, bone health surveillance and maintenance are critical components of DMD management.16
Patients with DMD often experience gastrointestinal issues. They may experience weight gain because of lack of mobility and corticosteroid use in early stages, or weight loss as a result of diet or fluid imbalance, low bone density, or dysphagia in later stages. Patients should be closely followed by a nutritionist, a gastroenterologist as needed, and a physical therapist.16
Psychosocial support “should be developed and implemented across the lifespan in a manner that promotes thinking about the future and sets expectations that individuals will actively participate in their care and daily activities.”9 This includes psychological care, neuropsychological evaluations, and educational support.
Assisting Patients and Families Through the DMD Journey
DMD care is best delivered in a multidisciplinary setting, where physicians of relevant specialties, physical and occupational therapists, nutritionists, social workers, and genetic counselors collaborate. At Children’s Hospital of Philadelphia, DMD care is delivered through this collaborative model.
Unfortunately, Dr. Brandsema said, many patients don’t have this type of multidisciplinary resource available. “One specialist, such as a pulmonologist or neurologist, might have to be the sole source of care.” Or parents may have to ferry their child to multiple specialists in disparate locations, placing extra stress on an already-stressed family system.
“It’s helpful to connect the family with a comprehensive care center, if possible,” Dr. Brandsema advised. If that’s not available, then he suggests recommending educational opportunities and resources through national organizations such as the Muscular Dystrophy Association; Parent Project MD; NORD; Friends, Family and Duchenne; and Cure Duchenne. Families and caregivers, along with affected individuals, can get education and support from people who understand the day-to-day reality of living with this disease.
One of the major challenges that families face is navigating the high cost of treating DMD, especially the new medications, Dr. Brandsema said. “The authorization process can be intensive and long, and the family may need to take an active role, together with the provider team, in advocating for the patient to get access.”
Among her many responsibilities, Ms. Kaschak engages in care coordination tasks and management, helps patients and caregivers understand care plans, and provides psychosocial support and education about the disease process. She assists families in completing paperwork and navigating specialty authorizations, helping families understand and navigate the complex insurance process. “My role is to bridge gaps in care,” she said.
Dr. Brandsema noted that it’s important for couples to receive genetic counseling if they’re planning to have multiple children because there is a 50% chance that their next boy will be affected. About two thirds of mothers with children who have DMD are carriers, but many are not aware of it. Receiving counseling will enable them to understand their own risks of health complications, as well as the risk to future children.
Managing DMD Across the Lifespan
Another dimension of DMD care is providing resources and help to young people with DMD as they transition into adulthood. “In the past, we had limited treatment and mortality typically took place in the early 20s, so there weren’t a lot of patients who were adults. But as medication options have expanded and management of cardiac and respiratory failure has improved, we see a more significant proportion of adults who require adult-appropriate clinics — or, at the very least, specialists who are conversant in care or can provide care across the lifespan,” Dr. Brandsema said.
The DMD Care Considerations Working Group provides recommendations regarding care across the lifespan,9 as does the Adult North Star Network, of Muscular Dystrophy UK.17,18
Dr. Brandsema emphasized that, despite their disability, many adults with DMD “still engage with the community, and live life to its fullest.” It is to be hoped that, with ongoing research, earlier diagnosis, and improved treatment options, the future will look bright for people with DMD.
Dr. Brandsema has served as a consultant for Audentes, AveXis/Novartis, Biogen, Cytokinetics, Dyne, Edgewise, Fibrogen, Genentech, Marathon, Momenta/Janssen, NS Pharma, Pfizer, PTC Therapeutics, Sarepta, Scholar Rock, Takeda, and WaVe. He is on the medical advisory council member for Cure SMA and is a site investigator for clinical trials with Alexion, Astellas, AveXis/Novartis, Biogen, Biohaven, Catabasis, CSL Behring, Cytokinetics, Dyne, Fibrogen, Genentech, Ionis, Lilly, ML Bio, Pfizer, PTC Therapeutics, Sarepta, Scholar Rock, Summit, and WaVe. Ms. Kaschak has nothing to disclose.
References
1. Venugopal V and Pavlakis S. Duchenne Muscular Dystrophy. 2023 Jul 10. In: StatPearls [Internet]. Treasure Island, Florida: StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK482346/.
2. Gao QQ and McNally EM. Compr Physiol. 2015 Jul 1;5(3):1223-39. doi: 10.1002/cphy.c140048.
3. Duan D et al. Nat Rev Dis Primers. 2021 Feb 18;7(1):13. doi: 10.1038/s41572-021-00248-3.
4. Aartsma-Rus A et al. J Pediatr. 2019 Jan:204:305-313.e14. doi: 10.1016/j.jpeds.2018.10.043.
5. Broomfield J et al. Neurology. 2021 Dec 7;97(23):e2304-e2314. doi: 10.1212/WNL.0000000000012910.
6. Mercuri E et al. Front Pediatr. 2023 Nov 10:11:1276144. doi: 10.1212/WNL.0000000000012910.
7. Birnkrant DJ et al. Lancet Neurol. 2018 Mar;17(3):251-267. doi: 10.1016/S1474-4422(18)30024-3.
8. Birnkrant DJ et al. Lancet Neurol. 2018 Apr;17(4):347-361. doi: 10.1016/S1474-4422(18)30025-5.
9. Birnkrant DJ et al. Lancet Neurol. 2018 May;17(5):445-455. doi: 10.1016/S1474-4422(18)30026-7.
10. Matthews E et al. Cochrane Database Syst Rev. 2016 May 5;2016(5):CD003725. doi: 10.1002/14651858.CD003725.pub4.
11. Bylo M et al. Ann Pharmacother. 2020 Aug;54(8):788-794. doi: 10.1177/1060028019900500.
12. Guglieri M et al. JAMA Neurol. 2022 Oct 1;79(10):1005-1014. doi: 10.1001/jamaneurol.2022.2480.
13. Dang UJ et al. Neurology. 2024 Mar 12;102(5):e208112. doi: 10.1212/WNL.0000000000208112.
14. Mercuri E et al. Lancet Neurol. 2024 Apr;23(4):393-403. doi: 10.1016/S1474-4422(24)00036-X.
15. Gushchina LV et al. Mol Ther Nucleic Acids. 2022 Nov 9:30:479-492. doi: 10.1016/j.omtn.2022.10.025.
16. Patterson G et al. Eur J Pharmacol. 2023 May 15:947:175675. doi: 10.1016/j.ejphar.2023.175675.
17. Quinlivan R et al. J Neuromuscul Dis. 2021;8(6):899-926. doi: 10.3233/JND-200609.
18. Narayan S et al. J Neuromuscul Dis. 2022;9(3):365-381. doi: 10.3233/JND-210707.
Duchenne muscular dystrophy (DMD) is a severe progressive inherited disease characterized by muscle wasting and ultimately culminating in death. It’s a common enough neuromuscular disorder that pediatricians and family practice physicians are likely to see at least a couple of patients with DMD over the course of their career,” John Brandsema, MD, Neuromuscular Section Head, Division of Neurology, Children’s Hospital of Philadelphia in Pennsylvania, said in an interview. Healthcare providers should therefore be familiar with the disorder so as to provide timely diagnosis and early intervention as well as practical and emotional support to the patient and family/caregivers as they traverse the challenging and often heartbreaking journey with this condition.
Pathophysiology and Disease Trajectory
DMD is caused by pathogenic variants in the X-linked DMD gene, leading to reduction in dystrophin, a protein that serves as a cytoskeletal integrator, stabilizing the plasma membrane of striated muscle cells. Dystrophin is critical for muscle membrane stability.2 In particular, mutations in the gene that encodes for dystrophin lead to dysfunction in Dp427m, which is the muscle isoform of dystrophin.3,4
DMD is one of several types of muscular dystrophies. All are progressive disorders. Over time, healthy muscle fibers disappear and are replaced by fibrotic tissue and fat, making the muscles “less able to generate force for everyday activity.”2 Ultimately, the skeletal muscle dysfunction affects not only the patient’s day-to-day mobility but other systems as well. Most patients with DMD eventually die of cardiac and/or respiratory failure between the ages of 20 and 40 years, with a median life expectancy of 22 years — although children born after 1990 have a somewhat higher median life expectancy (28 years), because of the improving standard of care.3,5
Typically, DMD first presents with developmental delays and weakness in skeletal leg muscles. As the disease goes through stages of progression, it starts involving upper extremities and other systems. (Table 1)
Genetic Causes of DMD
The DMD gene, located on the X chromosome, encodes for the production of dystrophin. Variants of this gene result in the lack of dystrophin protein, leading in turn to muscle fiber degeneration and the progressive symptoms of DMD. Because of the gene’s location on the X chromosome, males (who don’t have a second copy of the X chromosome) cannot compensate for the mutated gene, which is why the disease affects male children. Females with this mutation are carriers and typically do not develop the same severity of symptoms, although they might have milder muscle cramps, weakness, and cardiac issues.3
A female carrier with DMD (or any other X-linked disorder) has a 25% chance to have a carrier daughter, a 25% change of having a noncarrier daughter, a 25% chance of having an affected son, and a 25% chance of having a nonaffected son. A male with the disorder will pass the mutated gene on to his daughters who then become carriers. He cannot pass the disorder on to his sons because males inherit only the Y chromosome from their fathers.3
Diagnosing DMD
“It can take as long as 1-3 years for a child to be diagnosed with DMD,” Dr. Brandsema said. “Parents typically have concerns and know that something is ‘off’ about their child and they’re sent to various specialists, but it usually takes time for an accurate diagnosis to be made.” The mean age at diagnosis of DMD is between ages 4 and 5 years.6
Early identification of infants at risk for developing DMD can help move the needle toward earlier diagnosis. Newborn screening for DMD has been researched and piloted in several programs.6 In 2023, DMD was nominated for inclusion in the Recommended Universal Screening Panel (RUSP) for universal newborn screening. But in May 2024, the advisory committee on Heritable Disorders in Newborns and Children decided to postpone the vote to include DMD in the RUSP, requesting additional information to ensure an evidence-based decision.
In the absence of universal newborn screening for DMD, alternative approaches have been proposed to reduce the delay in clinical diagnosis and specialist referral, including increasing awareness among healthcare providers (eg, pediatricians, pediatric neurologists, and primary care physicians).6
The National Task Force for Early Identification of Childhood Neuromuscular Disorders delineates the steps necessary to identify pediatric muscle weakness and signs of neuromuscular disease. Primary care providers are encouraged to engage in regular developmental surveillance. A surveillance aid lays out the timetable for recommended visits, typical developmental milestones, and components of surveillance. Clinical evaluation includes a detailed patient history, family history, and physical examination.
If a neuromuscular condition is suspected, laboratory work should include creatinine phosphokinase (CK).6 Elevated serum CK points to leakage of CK through the muscle membrane, suggesting muscle damage. If CK is elevated, genetic testing should be performed; and, if negative, it should be followed by genetic sequencing that tests for small-scale mutations in the DMD gene. If that test is negative, a muscle biopsy should be performed to test for deep intronic mutations in the DMD gene.4
The diagnostic process and immediate steps after a confirmed DMD diagnosis is found in Figure 1.
Targeting Inflammation in DMD
Traditionally, corticosteroids have been the only available medical treatment for DMD and they remain a cornerstone of DMD management. A meta-analysis found “moderate evidence” that corticosteroid therapy improves muscle strength and function in the short term (12 months), and strength up to 2 years.10
The two most common corticosteroids for DMD are prednisone and deflazacort. Deflazacort (Emflaza, PTC Therapeutics) was approved in 2017 to treat patients ages 5 years and older with DMD, subsequently expanded to 2 years and older. Deflazacort has been found to be more effective than prednisone in improving functional outcomes, delaying the onset of cardiomyopathy, and improving overall survival, with fewer adverse effects.11
In 2023, vamorolone (Agamree, Catalyst Pharmaceuticals) was approved by the Food and Drug Administration (FDA) to treat DMD patients (ages 2 years and older). Vamorolone is a dissociative steroidal anti-inflammatory that reduces bone morbidities and is regarded as a safer alternative than prednisone. A clinical trial comparing two doses of vamorolone with prednisone for 24 weeks found that vamorolone 6 mg/kg per day met the primary endpoint (time to stand velocity) and four sequential secondary motor function endpoints, with less bone morbidity, compared to prednisone.12 A more recent trial found improvements in motor outcomes at 48 weeks with a dose of 6 mg/kg per day of vamorolone. Bone morbidities of prednisone were reversed when the patient transitioned to vamorolone.13
“Steroid treatment has been proven to help, usually taken daily, although other schedules have been tried,” Dr. Brandsema said. However, all steroids are fraught with adverse effects and are suboptimal in the long term in reducing the disease burden.
The anti-inflammatory agent givinostat (Duvyzat, ITF Therapeutics), an oral histone deacetylase (HDAC) inhibitor, was approved in March 2024 for the treatment of DMD in patients 6 years of age and older. It is the first nonsteroidal drug to treat patients with all genetic variants of the disease, and it has a unique mechanism of action. Deficits in dystrophin can lead to increased HDAC activity in DMD, reducing the expression of genes involved in muscle regeneration. Givinostat therefore can help to counteract the pathogenic events downstream of dystrophin deficiency by inhibiting HDAC.14
Approval for givinostat was based on the phase 3 EPIDYS trial, which randomized 179 boys with DMD to receive either givinostat or placebo. Although results of a functional task worsened in both groups over the 12-month study period, the decline was significantly smaller with givinostat versus placebo. The most common adverse events were diarrhea and vomiting.14 Dr. Brandsema noted that monitoring of triglycerides and platelet count is required, as hypertriglyceridemia and thrombocytopenia can occur. This treatment was studied in tandem with corticosteroids as a combination approach to muscle stabilization.
New Pharmacotherapeutic Options: Exon-Skipping Agents
Today’s treatments have expanded beyond corticosteroids, with newer therapeutic options that include targeted exon-skipping therapies and, more recently, gene therapies. “These new treatment paradigms have changed the face of DMD treatment,” Dr. Brandsema said.
“Exon-skipping drugs in their current form have only a modest effect, but at least they’re a step in the right direction and a breakthrough, in terms of slowing disease progression,” Dr. Brandsema said.
Current exon-skipping agents use antisense phosphorodiamidate morpholino oligomers (PMOs) to restore a DMD open reading frame. Next-generation drugs called cell-penetrating peptide-conjugated PMOs (PPMOs) are being actively researched, Dr. Brandsema said. These agents have shown enhanced cellular uptake and more efficient dystrophin restoration, compared with unconjugated PMOs.15
There are currently four FDA-approved exon-skipping agents for DMD, all of which are administered via a weekly intravenous infusion: Casimersen (Amondys-45, SRP-4045), approved by the FDA in 2021; Eteplirsen (Exondys 51), approved in 2016; Golodirsen (Vyondys 53,SRP-4053), approved in 2019; and Vitolarsen (Viltepso), approved in 2020. They can be associated with multiple side effects, depending on the drug, including upper respiratory infection, fever, cough, rash, and gastrointestinal issues.16 These agents have the potential to help 30% of DMD patients, restoring low levels of dystrophin.16
Gene Transfer Therapies
Gene transfer therapies, a new class of agents, utilize a nonpathogenic viral vector (adeno-associated virus) to transfer specific genes to patients with DMD. Gene therapy involves overexpressing the micro-dystrophin gene to restore functional dystrophin expression.16
Multiple clinical trials of gene therapy are currently in progress. In 2023, delandistrogene moxeparvovec-rokl (Elevidys, Serepta) was granted accelerated FDA approval for ambulatory individuals with DMD between the ages of 4 and 5 years of age and a confirmed mutation in the DMD gene. It received expanded approval in June 2024 to include ambulatory and nonambulatory individuals aged 4 years and older with DMD and a confirmed mutation in the DMD gene (with the exception of exon 8 or 9 mutations).
The approval was based on preliminary data from two double-blind, placebo-controlled studies and two open-label studies, which enrolled a total of 218 male patients (including those who received placebo) with a confirmed disease-causing mutation in the DMD gene.
Delandistrogene moxeparvovec-rokl is delivered as a one-time infusion and has been associated with side effects and “a lot of potential issues,” Dr. Brandsema said. “We’ve seen cardiac effects, immune system effects, increased muscle inflammation and hepatic complications, and some people who became quite unwell were hospitalized for a long time.”
Fortunately, he added, “these seem to be rare but they do happen. Once the medication has been delivered, it’s permanently in the body, so you’re managing the side effects potentially on a long-term basis.”
It is critical to discuss the risks and benefits of this treatment with the family and caregivers and with the patient as well, if he old enough and able to participate in the decision-making progress. “We don’t want to give unrealistic expectations and we want people to be aware of the potential downside of this treatment,” he said. “This is a very complex discussion because the trajectory of the disease is so devastating and this treatment does hold out hope that other therapies don’t necessarily have.”
Nonpharmacologic Interventions
Physical therapy is a mainstay in DMD treatment, addressing protection of fragile muscles, preservation of strength, and prevention of muscle contractures.16 Given the respiratory impairments that occur with DMD progression, respiratory monitoring and therapy are essential; however, the number and type of evaluations and interventions vary with the stage of the disease, intensifying as the disease progresses.16 Similarly, cardiac monitoring should begin early, with patients screened for cardiac complications, and should intensify through the stages of disease progression.16
Bone health is compromised in patients with DMD, both as a result of corticosteroid treatment and as part of the disease itself. Fractures may be asymptomatic and may go unnoticed. Thus, bone health surveillance and maintenance are critical components of DMD management.16
Patients with DMD often experience gastrointestinal issues. They may experience weight gain because of lack of mobility and corticosteroid use in early stages, or weight loss as a result of diet or fluid imbalance, low bone density, or dysphagia in later stages. Patients should be closely followed by a nutritionist, a gastroenterologist as needed, and a physical therapist.16
Psychosocial support “should be developed and implemented across the lifespan in a manner that promotes thinking about the future and sets expectations that individuals will actively participate in their care and daily activities.”9 This includes psychological care, neuropsychological evaluations, and educational support.
Assisting Patients and Families Through the DMD Journey
DMD care is best delivered in a multidisciplinary setting, where physicians of relevant specialties, physical and occupational therapists, nutritionists, social workers, and genetic counselors collaborate. At Children’s Hospital of Philadelphia, DMD care is delivered through this collaborative model.
Unfortunately, Dr. Brandsema said, many patients don’t have this type of multidisciplinary resource available. “One specialist, such as a pulmonologist or neurologist, might have to be the sole source of care.” Or parents may have to ferry their child to multiple specialists in disparate locations, placing extra stress on an already-stressed family system.
“It’s helpful to connect the family with a comprehensive care center, if possible,” Dr. Brandsema advised. If that’s not available, then he suggests recommending educational opportunities and resources through national organizations such as the Muscular Dystrophy Association; Parent Project MD; NORD; Friends, Family and Duchenne; and Cure Duchenne. Families and caregivers, along with affected individuals, can get education and support from people who understand the day-to-day reality of living with this disease.
One of the major challenges that families face is navigating the high cost of treating DMD, especially the new medications, Dr. Brandsema said. “The authorization process can be intensive and long, and the family may need to take an active role, together with the provider team, in advocating for the patient to get access.”
Among her many responsibilities, Ms. Kaschak engages in care coordination tasks and management, helps patients and caregivers understand care plans, and provides psychosocial support and education about the disease process. She assists families in completing paperwork and navigating specialty authorizations, helping families understand and navigate the complex insurance process. “My role is to bridge gaps in care,” she said.
Dr. Brandsema noted that it’s important for couples to receive genetic counseling if they’re planning to have multiple children because there is a 50% chance that their next boy will be affected. About two thirds of mothers with children who have DMD are carriers, but many are not aware of it. Receiving counseling will enable them to understand their own risks of health complications, as well as the risk to future children.
Managing DMD Across the Lifespan
Another dimension of DMD care is providing resources and help to young people with DMD as they transition into adulthood. “In the past, we had limited treatment and mortality typically took place in the early 20s, so there weren’t a lot of patients who were adults. But as medication options have expanded and management of cardiac and respiratory failure has improved, we see a more significant proportion of adults who require adult-appropriate clinics — or, at the very least, specialists who are conversant in care or can provide care across the lifespan,” Dr. Brandsema said.
The DMD Care Considerations Working Group provides recommendations regarding care across the lifespan,9 as does the Adult North Star Network, of Muscular Dystrophy UK.17,18
Dr. Brandsema emphasized that, despite their disability, many adults with DMD “still engage with the community, and live life to its fullest.” It is to be hoped that, with ongoing research, earlier diagnosis, and improved treatment options, the future will look bright for people with DMD.
Dr. Brandsema has served as a consultant for Audentes, AveXis/Novartis, Biogen, Cytokinetics, Dyne, Edgewise, Fibrogen, Genentech, Marathon, Momenta/Janssen, NS Pharma, Pfizer, PTC Therapeutics, Sarepta, Scholar Rock, Takeda, and WaVe. He is on the medical advisory council member for Cure SMA and is a site investigator for clinical trials with Alexion, Astellas, AveXis/Novartis, Biogen, Biohaven, Catabasis, CSL Behring, Cytokinetics, Dyne, Fibrogen, Genentech, Ionis, Lilly, ML Bio, Pfizer, PTC Therapeutics, Sarepta, Scholar Rock, Summit, and WaVe. Ms. Kaschak has nothing to disclose.
References
1. Venugopal V and Pavlakis S. Duchenne Muscular Dystrophy. 2023 Jul 10. In: StatPearls [Internet]. Treasure Island, Florida: StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK482346/.
2. Gao QQ and McNally EM. Compr Physiol. 2015 Jul 1;5(3):1223-39. doi: 10.1002/cphy.c140048.
3. Duan D et al. Nat Rev Dis Primers. 2021 Feb 18;7(1):13. doi: 10.1038/s41572-021-00248-3.
4. Aartsma-Rus A et al. J Pediatr. 2019 Jan:204:305-313.e14. doi: 10.1016/j.jpeds.2018.10.043.
5. Broomfield J et al. Neurology. 2021 Dec 7;97(23):e2304-e2314. doi: 10.1212/WNL.0000000000012910.
6. Mercuri E et al. Front Pediatr. 2023 Nov 10:11:1276144. doi: 10.1212/WNL.0000000000012910.
7. Birnkrant DJ et al. Lancet Neurol. 2018 Mar;17(3):251-267. doi: 10.1016/S1474-4422(18)30024-3.
8. Birnkrant DJ et al. Lancet Neurol. 2018 Apr;17(4):347-361. doi: 10.1016/S1474-4422(18)30025-5.
9. Birnkrant DJ et al. Lancet Neurol. 2018 May;17(5):445-455. doi: 10.1016/S1474-4422(18)30026-7.
10. Matthews E et al. Cochrane Database Syst Rev. 2016 May 5;2016(5):CD003725. doi: 10.1002/14651858.CD003725.pub4.
11. Bylo M et al. Ann Pharmacother. 2020 Aug;54(8):788-794. doi: 10.1177/1060028019900500.
12. Guglieri M et al. JAMA Neurol. 2022 Oct 1;79(10):1005-1014. doi: 10.1001/jamaneurol.2022.2480.
13. Dang UJ et al. Neurology. 2024 Mar 12;102(5):e208112. doi: 10.1212/WNL.0000000000208112.
14. Mercuri E et al. Lancet Neurol. 2024 Apr;23(4):393-403. doi: 10.1016/S1474-4422(24)00036-X.
15. Gushchina LV et al. Mol Ther Nucleic Acids. 2022 Nov 9:30:479-492. doi: 10.1016/j.omtn.2022.10.025.
16. Patterson G et al. Eur J Pharmacol. 2023 May 15:947:175675. doi: 10.1016/j.ejphar.2023.175675.
17. Quinlivan R et al. J Neuromuscul Dis. 2021;8(6):899-926. doi: 10.3233/JND-200609.
18. Narayan S et al. J Neuromuscul Dis. 2022;9(3):365-381. doi: 10.3233/JND-210707.
Promise for Disease-Modifying Therapies to Tame Huntington’s Disease
Much progress has been made in managing the symptoms of Huntington’s disease, but the real excitement lies in the development of disease-modifying drugs and genetic therapy.
In April 1872, The Medical and Surgical Reporter of Philadelphia published a roughly 3,000-word paper, titled “On Chorea,” by George Huntington, a 22-year-old family practice physician recently graduated from Columbia University, New York City.
“Chorea is essentially a disease of the nervous system. The name ‘chorea’ is given to the disease on account of the dancing propensities of those who are affected by it, and it is a very appropriate designation,” he wrote in the introduction.
Toward the end of the paper Dr. Huntington described a “hereditary chorea” that he had observed while on professional rounds with his father, also a physician, in towns on the eastern end of Long Island in New York.
“It is spoken of by those in whose veins the seeds of the disease are known to exist, with a kind of horror, and not at all alluded to except through dire necessity, when it is mentioned as ‘that disorder,’ ” he wrote, noting later that “I have never known a recovery or even an amelioration of symptoms in this form of chorea; when once it begins it clings to the bitter end.”1
It wasn’t until 1993 that a team of investigators identified the gene responsible for the neurodegenerative disorder we now know as Huntington’s disease.
That discovery sparked hope for better treatments and a cure, but progress over the last 31 years has been incremental. Nonetheless, recent intensive research into novel approaches for treating Huntington’s disease have considerably brightened prospects for patients and caregivers, experts say.
“This is a devastating neurodegenerative disease and in talking to families that I have had the privilege and honor of following, I hear from them that ‘in 1993 when the gene was identified and my family member — parent, grandparent, aunt, uncle — had Huntington’s, we thought that there would be a curative or disease-modifying therapy in no time,’ ” said Erin Furr Stimming, MD, FAAN, FANA professor of neurology and Memorial Hermann Endowed Chair at the McGovern Medical School at University of Texas Health Science Center in Houston.
“Here we are in 2024. I think our families are still so incredibly resilient and courageous, and they are willing and able to participate in clinical trials. Families in the Huntington’s disease community at large are really ready to have trials that do, in fact, demonstrate some evidence of slowing of disease progression. It’s an exciting time,” she said in an interview.
Repeating Nucleotides
Huntington’s disease is an autosomal dominant neurodegenerative disorder caused by an expansion of a CAG trinucleotide repeat in exon 1 of the huntingtin gene (HTT), which encodes for the huntingtin protein (HTT). The multiple repeats result in expanded expression of mutant HTT. The mutated protein disrupts normal cellular processes, alters intracellular calcium homeostasis, and interferes with gene transcription, leading to progressive degeneration of neurons, and to a hallmark Huntington’s disease triad consisting of movement disorders, cognitive decline, and mood/behavioral issues.
The number of repeats determines the severity of disease and the age of onset. In nonaffected persons the gene contains about 20 CAG repeats, but as genetics research dating from the 1990s has shown, a single HTT allele containing more than 40 CAG repeats will inevitably result in disease, whereas carriers with fewer than 36 repeats on both alleles will remain unaffected.2
The prevalence of the mutation in Western populations is estimated to be from 4 to 10 per 100,000.3
The disease usually manifests first in adults aged 30-50 years but may also occur in children or adolescents and young adults. Early symptoms often include a decline in executive function that may be noticeable to the patient’s family and friends, mood changes, and chorea.
“Because of the uncontrolled movements (chorea), a person with Huntington’s disease may lose a lot of weight without intending to, and may have trouble walking, balancing, and moving around safely. They will eventually lose the ability to work, drive, and manage tasks at home, and may qualify for disability benefits. Over time, the individual will develop difficulty with speaking and swallowing, and their movements will become slow and stiff. People with advanced Huntington’s disease need full-time care to help with their day-to-day activities, and they ultimately succumb to pneumonia, heart failure, or other complications,” according to the Huntington’s Disease Society of America.4
Managing Symptoms
say neurologists.
There are currently three medications approved for the treatment of chorea, all in the class of agents known as vesicular monoamine transporter 2 inhibitors. These agents are tetrabenazine (Xenazine, Lundbeck), deutetrabenazine (Austedo XR, Teva), and valbenazine (Ingrezza, Neurocrine Biosciences).
“These drugs can treat the chorea, but they also can help with some of the other motor features. For example, if a patient has chorea in the legs and you treat it, then maybe they’re walking will get better, or if they have chorea in the mouth and you treat the chorea, then maybe their speech and swallowing may improve,” Victor Sung, MD, professor of neurology and director of the Huntington’s Disease Clinic at the University of Alabama at Birmingham, said in an interview.
Mood and behavioral symptoms associated with Huntington’s disease – depression, anxiety, irritability, impulsivity, etc – can be managed with off-label use of antidepressants, mood stabilizers, and antipsychotic agents.
“When it comes to the cognitive symptoms, that’s a big gap where we don’t have anything that’s [Food and Drug Administration] approved for Huntington’s disease,” Dr. Sung said.
Agents used to treat Alzheimer’s disease, such as cholinesterase inhibitors and the glutamate receptor antagonist memantine, have been studied extensively for preservation of cognition in Huntington’s disease, but have not shown significant benefit.
Dr. Sung said that one promising approach to the problem of cognitive protection in Huntington’s disease is the investigational agent dalzanemdor (SAGE-718), an N-methyl-D-aspartic (NMDA) acid receptor positive allosteric modulator. The drug is in development for cognitive disorders associated with NMDA receptor dysfunction, including Huntington’s disease and Alzheimer’s disease.
In the phase 2 SURVEYOR trial, which was not powered to show efficacy of dalzanemdor over placebo, patients with Huntington’s disease reportedly tolerated the drug well, with treatment-related adverse events primarily mild or moderate in severity.
As of this writing dalzanemdor is being evaluated for efficacy, compared with placebo, in the phase 2 DIMENSION trial. The primary endpoint of this trial is a change from baseline in composite score of the Huntington’s Disease Cognitive Assessment Battery.
Disease-Modifying Therapies
As previously noted, although there is no cure for Huntington’s disease, neurology investigators are developing new strategies for delaying, stalling, or even preventing disease progression.
“There are so many different approaches, it’s hard to know where to start,” Christopher A. Ross, MD, director of the Huntington’s Disease Center at Johns Hopkins University in Baltimore, Maryland, said in an interview.
The most actively investigated approach may be huntingtin-lowering therapies, based on the supposition that mutant huntingtin protein (mHTT) is the primary toxin in Huntington’s disease.
“What you need to do is in some way lower it, and there are a number of different ways to do that: antinsense olignoclueotides, small interfering RNA, CRISPR Cas, or other gene-editing techniques, as well as gene therapy with an adenoviral vector” he said.
Gene Therapy
Dr. Ross cited as promising a phase 1/2 clinical trial of an investigational gene therapy, AMT-130 (uniQure), which consists of an adeno-associated virus vector and a gene encoding a microRNA that is designed to recognize, bind, and nonselectively lower both mHTT and wild-type HTT.
The compound is injected directly into the corpus striatum. It was demonstrated to decrease signs of Huntington’s disease in animal models, and interim data on 29 patients with Huntington’s disease followed for up to 24 months showed a statically significant, dose-dependent slowing of Huntington’s disease progression and lowering of neurofilament light protein, a marker for neuronal degeneration in Huntington’s disease, in cerebrospinal fluid (CSF).
AMT-130 targets exon 1 of the HTT gene, which appears to be an important target, Dr. Ross commented.
“The huntingtin protein is really big. The polyglutamine expansion, which is what causes the toxicity, is right at the N-terminus of exon 1, and there is increasingly good evidence that you can have an exon 1 misspliced protein product which causes the toxicity, so it may be especially important to lower Huntington by targeting exon 1,” he said.
Oral Splicing Modifier
PTC Therapeutics, based in New Jersey, is developing PTC518, an oral small molecule drug that can cross the blood-brain barrier and is reported to target mutant huntingtin protein.
The drug is a splicing modifier that promotes insertion of a premature stop codon to HTT mRNA, thereby degrading and lowering the HTT levels.5
In June 2024, the company reported that, in the phase 2a PIVOT-HD study of PTC518 in patients with Huntington’s disease, 12 months of treatment was associated with a dose-dependent lowering of mHTT in blood and CSF in an interim cohort.
“In addition, favorable trends were demonstrated on several relevant Huntington’s disease clinical assessments including Total Motor Score and Composite Unified Huntington’s Disease Rating Scale. Furthermore, following 12 months of treatment, PTC518 continues to be safe and well tolerated,” the company stated in a press release.
Antisense Oligonucleotides
Antisense oligonucleotides (ASOs) are short strands of DNA or RNA that bind to RNA sequences in faulty genes to modify production of target proteins.
One such drug, tominersen, developed jointly by Ionis and Roche, initially showed promise in a phase 1/2 trial for lowering mHTT in CSF without serious adverse events. But in a phase 3 trial, the intrathecally delivered agent was halted after an independent data monitoring committee recommended halting the trial, which Roche ended in 2021. The company reported in a letter to The New England Journal of Medicine that people in the high-dosage treatment group did measurably worse – although it remains unclear whether this was caused by excess protein lowering or an off-target effect.6 The tominersen program was the first to demonstrate that it was possible to lower HTT with an intervention, and the companies reported that they are continuing the agent’s development program.
Wave Life Sciences is developing an ASO, labeled WVE-003, designed to target a single-nucleotide polymorphism associated with the mHTT mRNA transcript within HTT. The company says that targeting the single-nucleotide polymorphism should allow lowering of expression of the mHTT will preserving wild-type HTT. This approach has the potential for therapies to prevent disease progression during the prodromal period, the company states.
Somatic Expansion
Dr. Furr Stimming and Dr. Ross both noted that there is considerable research interest into recently identified genetic modifiers that are believed to influence somatic instability, which in turn leads to somatic expansion.
“That seems to happen selectively in the neurons that are affected in Huntington’s disease. So a big puzzle for all of the neurodegenerative diseases is why are certain regions of the brain affected and other regions not? And it looks like, for the repeat expansion, this idea of somatic expansion seems to be increasingly central,” Dr. Ross said.
“The really exciting idea here is that, if somatic expansion is critical to the disease process and you could slow it down or stop it, you could go very early and potentially not just slow the progression of the disease once it starts, but conceivably even delay or possibly prevent the onset of Huntington’s disease,” he said.
Does HTT Lowering Mediate Progression?
“The next question is what does this mean clinically? Does lowering mutant huntingtin protein levels, and wild type for that matter, actually slow disease progression, which can be challenging to measure in a disease that is relatively slowly progressive?,” Dr. Furr Stimming said.
One important tool to help answer this question, she noted, is the Huntington’s Disease Integrated Staging System (HDISS), first described in 2022.7 The consensus-based system incorporates biological, clinical, and functional assessments, and characterizes patients from birth by stages, from stage 0 (persons who carry the mutation but have no detectable pathology), to stage 1 (measurable indicators of underlying pathophysiology), stage 2 (a detectable clinical phenotype), and finally to stage 3 (decline in function).
“The goal of the HDISS, which is designed for clinical research purposes, is to try to enroll individuals into clinical trials earlier rather than later, trying to get pharmaceutical companies and others to take advantage of the period prior to significant neurodegeneration occurring. Like Alzheimer’s disease and Parkinson’s disease, by the time we make a clinical diagnose significant neurodegeneration has occurred, so we really want to take advantage of the prodromal period to intervene with these potential disease-modifying therapies,” Dr. Furr Stimming said.
“At the end of the day, I suspect that we will not have just one effective disease-modifying therapy. I suspect that it will be a multifacted approach. We envision that we would hit the huntingtin protein from a few different angles. I envision that we would not only modify in some form or fashion production of the mutant huntingtin protein, but also try to influence the genetic modifiers that we think are important in somatic instability and expansion, which likely contributes to the rate of progression and symptom severity,” Dr. Furr Stimming said.
References
1. Huntington G. On Chorea. Reprinted in The Journal of Neuropsychiatry and Clinical Neurosciences. 2003;15(1):109-112. doi: 10.1176/jnp.15.1.109.
2. Kaemmerer WF and Grondin RC. Degener Neurol Neuromuscul Dis. 2019 Mar 8;9:3-17. doi: 10.2147/DNND.S163808.
3. Jimenez-Sanchez M et al. Cold Spring Harb Perspect Med. 2017 Jul 5;7(7):a024240. doi: 10.1101/cshperspect.a024240.
4. Huntington’s Disease Society of America. Overview of Huntington’s Disease. https://hdsa.org/what-is-hd/overview-of-huntingtons-disease/.
5. Beers B et al. J Neurol Neurosurg Psychiatry. 2022;93:A96. https://jnnp.bmj.com/content/93/Suppl_1/A96.1.
6. McColgan P et al. N Engl J Med. 2023 Dec 7;389(23):2203-2205. doi: 10.1056/NEJMc2300400.
7. Tabrizi SJ et al. Lancet Neurol. 2022 Jul;21(7):632-644. doi: 10.1016/S1474-4422(22)00120-X.
Much progress has been made in managing the symptoms of Huntington’s disease, but the real excitement lies in the development of disease-modifying drugs and genetic therapy.
In April 1872, The Medical and Surgical Reporter of Philadelphia published a roughly 3,000-word paper, titled “On Chorea,” by George Huntington, a 22-year-old family practice physician recently graduated from Columbia University, New York City.
“Chorea is essentially a disease of the nervous system. The name ‘chorea’ is given to the disease on account of the dancing propensities of those who are affected by it, and it is a very appropriate designation,” he wrote in the introduction.
Toward the end of the paper Dr. Huntington described a “hereditary chorea” that he had observed while on professional rounds with his father, also a physician, in towns on the eastern end of Long Island in New York.
“It is spoken of by those in whose veins the seeds of the disease are known to exist, with a kind of horror, and not at all alluded to except through dire necessity, when it is mentioned as ‘that disorder,’ ” he wrote, noting later that “I have never known a recovery or even an amelioration of symptoms in this form of chorea; when once it begins it clings to the bitter end.”1
It wasn’t until 1993 that a team of investigators identified the gene responsible for the neurodegenerative disorder we now know as Huntington’s disease.
That discovery sparked hope for better treatments and a cure, but progress over the last 31 years has been incremental. Nonetheless, recent intensive research into novel approaches for treating Huntington’s disease have considerably brightened prospects for patients and caregivers, experts say.
“This is a devastating neurodegenerative disease and in talking to families that I have had the privilege and honor of following, I hear from them that ‘in 1993 when the gene was identified and my family member — parent, grandparent, aunt, uncle — had Huntington’s, we thought that there would be a curative or disease-modifying therapy in no time,’ ” said Erin Furr Stimming, MD, FAAN, FANA professor of neurology and Memorial Hermann Endowed Chair at the McGovern Medical School at University of Texas Health Science Center in Houston.
“Here we are in 2024. I think our families are still so incredibly resilient and courageous, and they are willing and able to participate in clinical trials. Families in the Huntington’s disease community at large are really ready to have trials that do, in fact, demonstrate some evidence of slowing of disease progression. It’s an exciting time,” she said in an interview.
Repeating Nucleotides
Huntington’s disease is an autosomal dominant neurodegenerative disorder caused by an expansion of a CAG trinucleotide repeat in exon 1 of the huntingtin gene (HTT), which encodes for the huntingtin protein (HTT). The multiple repeats result in expanded expression of mutant HTT. The mutated protein disrupts normal cellular processes, alters intracellular calcium homeostasis, and interferes with gene transcription, leading to progressive degeneration of neurons, and to a hallmark Huntington’s disease triad consisting of movement disorders, cognitive decline, and mood/behavioral issues.
The number of repeats determines the severity of disease and the age of onset. In nonaffected persons the gene contains about 20 CAG repeats, but as genetics research dating from the 1990s has shown, a single HTT allele containing more than 40 CAG repeats will inevitably result in disease, whereas carriers with fewer than 36 repeats on both alleles will remain unaffected.2
The prevalence of the mutation in Western populations is estimated to be from 4 to 10 per 100,000.3
The disease usually manifests first in adults aged 30-50 years but may also occur in children or adolescents and young adults. Early symptoms often include a decline in executive function that may be noticeable to the patient’s family and friends, mood changes, and chorea.
“Because of the uncontrolled movements (chorea), a person with Huntington’s disease may lose a lot of weight without intending to, and may have trouble walking, balancing, and moving around safely. They will eventually lose the ability to work, drive, and manage tasks at home, and may qualify for disability benefits. Over time, the individual will develop difficulty with speaking and swallowing, and their movements will become slow and stiff. People with advanced Huntington’s disease need full-time care to help with their day-to-day activities, and they ultimately succumb to pneumonia, heart failure, or other complications,” according to the Huntington’s Disease Society of America.4
Managing Symptoms
say neurologists.
There are currently three medications approved for the treatment of chorea, all in the class of agents known as vesicular monoamine transporter 2 inhibitors. These agents are tetrabenazine (Xenazine, Lundbeck), deutetrabenazine (Austedo XR, Teva), and valbenazine (Ingrezza, Neurocrine Biosciences).
“These drugs can treat the chorea, but they also can help with some of the other motor features. For example, if a patient has chorea in the legs and you treat it, then maybe they’re walking will get better, or if they have chorea in the mouth and you treat the chorea, then maybe their speech and swallowing may improve,” Victor Sung, MD, professor of neurology and director of the Huntington’s Disease Clinic at the University of Alabama at Birmingham, said in an interview.
Mood and behavioral symptoms associated with Huntington’s disease – depression, anxiety, irritability, impulsivity, etc – can be managed with off-label use of antidepressants, mood stabilizers, and antipsychotic agents.
“When it comes to the cognitive symptoms, that’s a big gap where we don’t have anything that’s [Food and Drug Administration] approved for Huntington’s disease,” Dr. Sung said.
Agents used to treat Alzheimer’s disease, such as cholinesterase inhibitors and the glutamate receptor antagonist memantine, have been studied extensively for preservation of cognition in Huntington’s disease, but have not shown significant benefit.
Dr. Sung said that one promising approach to the problem of cognitive protection in Huntington’s disease is the investigational agent dalzanemdor (SAGE-718), an N-methyl-D-aspartic (NMDA) acid receptor positive allosteric modulator. The drug is in development for cognitive disorders associated with NMDA receptor dysfunction, including Huntington’s disease and Alzheimer’s disease.
In the phase 2 SURVEYOR trial, which was not powered to show efficacy of dalzanemdor over placebo, patients with Huntington’s disease reportedly tolerated the drug well, with treatment-related adverse events primarily mild or moderate in severity.
As of this writing dalzanemdor is being evaluated for efficacy, compared with placebo, in the phase 2 DIMENSION trial. The primary endpoint of this trial is a change from baseline in composite score of the Huntington’s Disease Cognitive Assessment Battery.
Disease-Modifying Therapies
As previously noted, although there is no cure for Huntington’s disease, neurology investigators are developing new strategies for delaying, stalling, or even preventing disease progression.
“There are so many different approaches, it’s hard to know where to start,” Christopher A. Ross, MD, director of the Huntington’s Disease Center at Johns Hopkins University in Baltimore, Maryland, said in an interview.
The most actively investigated approach may be huntingtin-lowering therapies, based on the supposition that mutant huntingtin protein (mHTT) is the primary toxin in Huntington’s disease.
“What you need to do is in some way lower it, and there are a number of different ways to do that: antinsense olignoclueotides, small interfering RNA, CRISPR Cas, or other gene-editing techniques, as well as gene therapy with an adenoviral vector” he said.
Gene Therapy
Dr. Ross cited as promising a phase 1/2 clinical trial of an investigational gene therapy, AMT-130 (uniQure), which consists of an adeno-associated virus vector and a gene encoding a microRNA that is designed to recognize, bind, and nonselectively lower both mHTT and wild-type HTT.
The compound is injected directly into the corpus striatum. It was demonstrated to decrease signs of Huntington’s disease in animal models, and interim data on 29 patients with Huntington’s disease followed for up to 24 months showed a statically significant, dose-dependent slowing of Huntington’s disease progression and lowering of neurofilament light protein, a marker for neuronal degeneration in Huntington’s disease, in cerebrospinal fluid (CSF).
AMT-130 targets exon 1 of the HTT gene, which appears to be an important target, Dr. Ross commented.
“The huntingtin protein is really big. The polyglutamine expansion, which is what causes the toxicity, is right at the N-terminus of exon 1, and there is increasingly good evidence that you can have an exon 1 misspliced protein product which causes the toxicity, so it may be especially important to lower Huntington by targeting exon 1,” he said.
Oral Splicing Modifier
PTC Therapeutics, based in New Jersey, is developing PTC518, an oral small molecule drug that can cross the blood-brain barrier and is reported to target mutant huntingtin protein.
The drug is a splicing modifier that promotes insertion of a premature stop codon to HTT mRNA, thereby degrading and lowering the HTT levels.5
In June 2024, the company reported that, in the phase 2a PIVOT-HD study of PTC518 in patients with Huntington’s disease, 12 months of treatment was associated with a dose-dependent lowering of mHTT in blood and CSF in an interim cohort.
“In addition, favorable trends were demonstrated on several relevant Huntington’s disease clinical assessments including Total Motor Score and Composite Unified Huntington’s Disease Rating Scale. Furthermore, following 12 months of treatment, PTC518 continues to be safe and well tolerated,” the company stated in a press release.
Antisense Oligonucleotides
Antisense oligonucleotides (ASOs) are short strands of DNA or RNA that bind to RNA sequences in faulty genes to modify production of target proteins.
One such drug, tominersen, developed jointly by Ionis and Roche, initially showed promise in a phase 1/2 trial for lowering mHTT in CSF without serious adverse events. But in a phase 3 trial, the intrathecally delivered agent was halted after an independent data monitoring committee recommended halting the trial, which Roche ended in 2021. The company reported in a letter to The New England Journal of Medicine that people in the high-dosage treatment group did measurably worse – although it remains unclear whether this was caused by excess protein lowering or an off-target effect.6 The tominersen program was the first to demonstrate that it was possible to lower HTT with an intervention, and the companies reported that they are continuing the agent’s development program.
Wave Life Sciences is developing an ASO, labeled WVE-003, designed to target a single-nucleotide polymorphism associated with the mHTT mRNA transcript within HTT. The company says that targeting the single-nucleotide polymorphism should allow lowering of expression of the mHTT will preserving wild-type HTT. This approach has the potential for therapies to prevent disease progression during the prodromal period, the company states.
Somatic Expansion
Dr. Furr Stimming and Dr. Ross both noted that there is considerable research interest into recently identified genetic modifiers that are believed to influence somatic instability, which in turn leads to somatic expansion.
“That seems to happen selectively in the neurons that are affected in Huntington’s disease. So a big puzzle for all of the neurodegenerative diseases is why are certain regions of the brain affected and other regions not? And it looks like, for the repeat expansion, this idea of somatic expansion seems to be increasingly central,” Dr. Ross said.
“The really exciting idea here is that, if somatic expansion is critical to the disease process and you could slow it down or stop it, you could go very early and potentially not just slow the progression of the disease once it starts, but conceivably even delay or possibly prevent the onset of Huntington’s disease,” he said.
Does HTT Lowering Mediate Progression?
“The next question is what does this mean clinically? Does lowering mutant huntingtin protein levels, and wild type for that matter, actually slow disease progression, which can be challenging to measure in a disease that is relatively slowly progressive?,” Dr. Furr Stimming said.
One important tool to help answer this question, she noted, is the Huntington’s Disease Integrated Staging System (HDISS), first described in 2022.7 The consensus-based system incorporates biological, clinical, and functional assessments, and characterizes patients from birth by stages, from stage 0 (persons who carry the mutation but have no detectable pathology), to stage 1 (measurable indicators of underlying pathophysiology), stage 2 (a detectable clinical phenotype), and finally to stage 3 (decline in function).
“The goal of the HDISS, which is designed for clinical research purposes, is to try to enroll individuals into clinical trials earlier rather than later, trying to get pharmaceutical companies and others to take advantage of the period prior to significant neurodegeneration occurring. Like Alzheimer’s disease and Parkinson’s disease, by the time we make a clinical diagnose significant neurodegeneration has occurred, so we really want to take advantage of the prodromal period to intervene with these potential disease-modifying therapies,” Dr. Furr Stimming said.
“At the end of the day, I suspect that we will not have just one effective disease-modifying therapy. I suspect that it will be a multifacted approach. We envision that we would hit the huntingtin protein from a few different angles. I envision that we would not only modify in some form or fashion production of the mutant huntingtin protein, but also try to influence the genetic modifiers that we think are important in somatic instability and expansion, which likely contributes to the rate of progression and symptom severity,” Dr. Furr Stimming said.
References
1. Huntington G. On Chorea. Reprinted in The Journal of Neuropsychiatry and Clinical Neurosciences. 2003;15(1):109-112. doi: 10.1176/jnp.15.1.109.
2. Kaemmerer WF and Grondin RC. Degener Neurol Neuromuscul Dis. 2019 Mar 8;9:3-17. doi: 10.2147/DNND.S163808.
3. Jimenez-Sanchez M et al. Cold Spring Harb Perspect Med. 2017 Jul 5;7(7):a024240. doi: 10.1101/cshperspect.a024240.
4. Huntington’s Disease Society of America. Overview of Huntington’s Disease. https://hdsa.org/what-is-hd/overview-of-huntingtons-disease/.
5. Beers B et al. J Neurol Neurosurg Psychiatry. 2022;93:A96. https://jnnp.bmj.com/content/93/Suppl_1/A96.1.
6. McColgan P et al. N Engl J Med. 2023 Dec 7;389(23):2203-2205. doi: 10.1056/NEJMc2300400.
7. Tabrizi SJ et al. Lancet Neurol. 2022 Jul;21(7):632-644. doi: 10.1016/S1474-4422(22)00120-X.
Much progress has been made in managing the symptoms of Huntington’s disease, but the real excitement lies in the development of disease-modifying drugs and genetic therapy.
In April 1872, The Medical and Surgical Reporter of Philadelphia published a roughly 3,000-word paper, titled “On Chorea,” by George Huntington, a 22-year-old family practice physician recently graduated from Columbia University, New York City.
“Chorea is essentially a disease of the nervous system. The name ‘chorea’ is given to the disease on account of the dancing propensities of those who are affected by it, and it is a very appropriate designation,” he wrote in the introduction.
Toward the end of the paper Dr. Huntington described a “hereditary chorea” that he had observed while on professional rounds with his father, also a physician, in towns on the eastern end of Long Island in New York.
“It is spoken of by those in whose veins the seeds of the disease are known to exist, with a kind of horror, and not at all alluded to except through dire necessity, when it is mentioned as ‘that disorder,’ ” he wrote, noting later that “I have never known a recovery or even an amelioration of symptoms in this form of chorea; when once it begins it clings to the bitter end.”1
It wasn’t until 1993 that a team of investigators identified the gene responsible for the neurodegenerative disorder we now know as Huntington’s disease.
That discovery sparked hope for better treatments and a cure, but progress over the last 31 years has been incremental. Nonetheless, recent intensive research into novel approaches for treating Huntington’s disease have considerably brightened prospects for patients and caregivers, experts say.
“This is a devastating neurodegenerative disease and in talking to families that I have had the privilege and honor of following, I hear from them that ‘in 1993 when the gene was identified and my family member — parent, grandparent, aunt, uncle — had Huntington’s, we thought that there would be a curative or disease-modifying therapy in no time,’ ” said Erin Furr Stimming, MD, FAAN, FANA professor of neurology and Memorial Hermann Endowed Chair at the McGovern Medical School at University of Texas Health Science Center in Houston.
“Here we are in 2024. I think our families are still so incredibly resilient and courageous, and they are willing and able to participate in clinical trials. Families in the Huntington’s disease community at large are really ready to have trials that do, in fact, demonstrate some evidence of slowing of disease progression. It’s an exciting time,” she said in an interview.
Repeating Nucleotides
Huntington’s disease is an autosomal dominant neurodegenerative disorder caused by an expansion of a CAG trinucleotide repeat in exon 1 of the huntingtin gene (HTT), which encodes for the huntingtin protein (HTT). The multiple repeats result in expanded expression of mutant HTT. The mutated protein disrupts normal cellular processes, alters intracellular calcium homeostasis, and interferes with gene transcription, leading to progressive degeneration of neurons, and to a hallmark Huntington’s disease triad consisting of movement disorders, cognitive decline, and mood/behavioral issues.
The number of repeats determines the severity of disease and the age of onset. In nonaffected persons the gene contains about 20 CAG repeats, but as genetics research dating from the 1990s has shown, a single HTT allele containing more than 40 CAG repeats will inevitably result in disease, whereas carriers with fewer than 36 repeats on both alleles will remain unaffected.2
The prevalence of the mutation in Western populations is estimated to be from 4 to 10 per 100,000.3
The disease usually manifests first in adults aged 30-50 years but may also occur in children or adolescents and young adults. Early symptoms often include a decline in executive function that may be noticeable to the patient’s family and friends, mood changes, and chorea.
“Because of the uncontrolled movements (chorea), a person with Huntington’s disease may lose a lot of weight without intending to, and may have trouble walking, balancing, and moving around safely. They will eventually lose the ability to work, drive, and manage tasks at home, and may qualify for disability benefits. Over time, the individual will develop difficulty with speaking and swallowing, and their movements will become slow and stiff. People with advanced Huntington’s disease need full-time care to help with their day-to-day activities, and they ultimately succumb to pneumonia, heart failure, or other complications,” according to the Huntington’s Disease Society of America.4
Managing Symptoms
say neurologists.
There are currently three medications approved for the treatment of chorea, all in the class of agents known as vesicular monoamine transporter 2 inhibitors. These agents are tetrabenazine (Xenazine, Lundbeck), deutetrabenazine (Austedo XR, Teva), and valbenazine (Ingrezza, Neurocrine Biosciences).
“These drugs can treat the chorea, but they also can help with some of the other motor features. For example, if a patient has chorea in the legs and you treat it, then maybe they’re walking will get better, or if they have chorea in the mouth and you treat the chorea, then maybe their speech and swallowing may improve,” Victor Sung, MD, professor of neurology and director of the Huntington’s Disease Clinic at the University of Alabama at Birmingham, said in an interview.
Mood and behavioral symptoms associated with Huntington’s disease – depression, anxiety, irritability, impulsivity, etc – can be managed with off-label use of antidepressants, mood stabilizers, and antipsychotic agents.
“When it comes to the cognitive symptoms, that’s a big gap where we don’t have anything that’s [Food and Drug Administration] approved for Huntington’s disease,” Dr. Sung said.
Agents used to treat Alzheimer’s disease, such as cholinesterase inhibitors and the glutamate receptor antagonist memantine, have been studied extensively for preservation of cognition in Huntington’s disease, but have not shown significant benefit.
Dr. Sung said that one promising approach to the problem of cognitive protection in Huntington’s disease is the investigational agent dalzanemdor (SAGE-718), an N-methyl-D-aspartic (NMDA) acid receptor positive allosteric modulator. The drug is in development for cognitive disorders associated with NMDA receptor dysfunction, including Huntington’s disease and Alzheimer’s disease.
In the phase 2 SURVEYOR trial, which was not powered to show efficacy of dalzanemdor over placebo, patients with Huntington’s disease reportedly tolerated the drug well, with treatment-related adverse events primarily mild or moderate in severity.
As of this writing dalzanemdor is being evaluated for efficacy, compared with placebo, in the phase 2 DIMENSION trial. The primary endpoint of this trial is a change from baseline in composite score of the Huntington’s Disease Cognitive Assessment Battery.
Disease-Modifying Therapies
As previously noted, although there is no cure for Huntington’s disease, neurology investigators are developing new strategies for delaying, stalling, or even preventing disease progression.
“There are so many different approaches, it’s hard to know where to start,” Christopher A. Ross, MD, director of the Huntington’s Disease Center at Johns Hopkins University in Baltimore, Maryland, said in an interview.
The most actively investigated approach may be huntingtin-lowering therapies, based on the supposition that mutant huntingtin protein (mHTT) is the primary toxin in Huntington’s disease.
“What you need to do is in some way lower it, and there are a number of different ways to do that: antinsense olignoclueotides, small interfering RNA, CRISPR Cas, or other gene-editing techniques, as well as gene therapy with an adenoviral vector” he said.
Gene Therapy
Dr. Ross cited as promising a phase 1/2 clinical trial of an investigational gene therapy, AMT-130 (uniQure), which consists of an adeno-associated virus vector and a gene encoding a microRNA that is designed to recognize, bind, and nonselectively lower both mHTT and wild-type HTT.
The compound is injected directly into the corpus striatum. It was demonstrated to decrease signs of Huntington’s disease in animal models, and interim data on 29 patients with Huntington’s disease followed for up to 24 months showed a statically significant, dose-dependent slowing of Huntington’s disease progression and lowering of neurofilament light protein, a marker for neuronal degeneration in Huntington’s disease, in cerebrospinal fluid (CSF).
AMT-130 targets exon 1 of the HTT gene, which appears to be an important target, Dr. Ross commented.
“The huntingtin protein is really big. The polyglutamine expansion, which is what causes the toxicity, is right at the N-terminus of exon 1, and there is increasingly good evidence that you can have an exon 1 misspliced protein product which causes the toxicity, so it may be especially important to lower Huntington by targeting exon 1,” he said.
Oral Splicing Modifier
PTC Therapeutics, based in New Jersey, is developing PTC518, an oral small molecule drug that can cross the blood-brain barrier and is reported to target mutant huntingtin protein.
The drug is a splicing modifier that promotes insertion of a premature stop codon to HTT mRNA, thereby degrading and lowering the HTT levels.5
In June 2024, the company reported that, in the phase 2a PIVOT-HD study of PTC518 in patients with Huntington’s disease, 12 months of treatment was associated with a dose-dependent lowering of mHTT in blood and CSF in an interim cohort.
“In addition, favorable trends were demonstrated on several relevant Huntington’s disease clinical assessments including Total Motor Score and Composite Unified Huntington’s Disease Rating Scale. Furthermore, following 12 months of treatment, PTC518 continues to be safe and well tolerated,” the company stated in a press release.
Antisense Oligonucleotides
Antisense oligonucleotides (ASOs) are short strands of DNA or RNA that bind to RNA sequences in faulty genes to modify production of target proteins.
One such drug, tominersen, developed jointly by Ionis and Roche, initially showed promise in a phase 1/2 trial for lowering mHTT in CSF without serious adverse events. But in a phase 3 trial, the intrathecally delivered agent was halted after an independent data monitoring committee recommended halting the trial, which Roche ended in 2021. The company reported in a letter to The New England Journal of Medicine that people in the high-dosage treatment group did measurably worse – although it remains unclear whether this was caused by excess protein lowering or an off-target effect.6 The tominersen program was the first to demonstrate that it was possible to lower HTT with an intervention, and the companies reported that they are continuing the agent’s development program.
Wave Life Sciences is developing an ASO, labeled WVE-003, designed to target a single-nucleotide polymorphism associated with the mHTT mRNA transcript within HTT. The company says that targeting the single-nucleotide polymorphism should allow lowering of expression of the mHTT will preserving wild-type HTT. This approach has the potential for therapies to prevent disease progression during the prodromal period, the company states.
Somatic Expansion
Dr. Furr Stimming and Dr. Ross both noted that there is considerable research interest into recently identified genetic modifiers that are believed to influence somatic instability, which in turn leads to somatic expansion.
“That seems to happen selectively in the neurons that are affected in Huntington’s disease. So a big puzzle for all of the neurodegenerative diseases is why are certain regions of the brain affected and other regions not? And it looks like, for the repeat expansion, this idea of somatic expansion seems to be increasingly central,” Dr. Ross said.
“The really exciting idea here is that, if somatic expansion is critical to the disease process and you could slow it down or stop it, you could go very early and potentially not just slow the progression of the disease once it starts, but conceivably even delay or possibly prevent the onset of Huntington’s disease,” he said.
Does HTT Lowering Mediate Progression?
“The next question is what does this mean clinically? Does lowering mutant huntingtin protein levels, and wild type for that matter, actually slow disease progression, which can be challenging to measure in a disease that is relatively slowly progressive?,” Dr. Furr Stimming said.
One important tool to help answer this question, she noted, is the Huntington’s Disease Integrated Staging System (HDISS), first described in 2022.7 The consensus-based system incorporates biological, clinical, and functional assessments, and characterizes patients from birth by stages, from stage 0 (persons who carry the mutation but have no detectable pathology), to stage 1 (measurable indicators of underlying pathophysiology), stage 2 (a detectable clinical phenotype), and finally to stage 3 (decline in function).
“The goal of the HDISS, which is designed for clinical research purposes, is to try to enroll individuals into clinical trials earlier rather than later, trying to get pharmaceutical companies and others to take advantage of the period prior to significant neurodegeneration occurring. Like Alzheimer’s disease and Parkinson’s disease, by the time we make a clinical diagnose significant neurodegeneration has occurred, so we really want to take advantage of the prodromal period to intervene with these potential disease-modifying therapies,” Dr. Furr Stimming said.
“At the end of the day, I suspect that we will not have just one effective disease-modifying therapy. I suspect that it will be a multifacted approach. We envision that we would hit the huntingtin protein from a few different angles. I envision that we would not only modify in some form or fashion production of the mutant huntingtin protein, but also try to influence the genetic modifiers that we think are important in somatic instability and expansion, which likely contributes to the rate of progression and symptom severity,” Dr. Furr Stimming said.
References
1. Huntington G. On Chorea. Reprinted in The Journal of Neuropsychiatry and Clinical Neurosciences. 2003;15(1):109-112. doi: 10.1176/jnp.15.1.109.
2. Kaemmerer WF and Grondin RC. Degener Neurol Neuromuscul Dis. 2019 Mar 8;9:3-17. doi: 10.2147/DNND.S163808.
3. Jimenez-Sanchez M et al. Cold Spring Harb Perspect Med. 2017 Jul 5;7(7):a024240. doi: 10.1101/cshperspect.a024240.
4. Huntington’s Disease Society of America. Overview of Huntington’s Disease. https://hdsa.org/what-is-hd/overview-of-huntingtons-disease/.
5. Beers B et al. J Neurol Neurosurg Psychiatry. 2022;93:A96. https://jnnp.bmj.com/content/93/Suppl_1/A96.1.
6. McColgan P et al. N Engl J Med. 2023 Dec 7;389(23):2203-2205. doi: 10.1056/NEJMc2300400.
7. Tabrizi SJ et al. Lancet Neurol. 2022 Jul;21(7):632-644. doi: 10.1016/S1474-4422(22)00120-X.
Newborn Screening Programs: What Do Clinicians Need to Know?
Newborn screening programs are public health services aimed at ensuring that the close to 4 million infants born each year in the United States are screened for certain serious disorders at birth. These disorders, albeit rare, are detected in roughly 12,500 newborn babies every year.
Newborn screening isn’t new, although it has expanded and transformed over the decades. The first newborn screening test was developed in the 1960s to detect phenylketonuria (PKU).1 Since then, the number of conditions screened for has increased, with programs in every US state and territory. “Newborn screening is well established now, not experimental or newfangled,” Wendy Chung, MD, PhD, professor of pediatrics, Harvard Medical School, Boston, Massachusetts, told Neurology Reviews.
In newborn screening, blood drawn from the baby’s heel is applied to specialized filter paper, which is then subjected to several analytical methods, including tandem mass spectrometry and molecular analyses to detect biomarkers for the diseases.2 More recently, genomic sequencing is being piloted as part of consented research studies.3
Newborn screening includes not only biochemical and genetic testing, but also includes noninvasive screening for hearing loss or for critical congenital heart disease using pulse oximetry. And newborn screening goes beyond analysis of a single drop of blood. Rather, “it’s an entire system, with the goal of identifying babies with genetic disorders who otherwise have no obvious symptoms,” said Dr. Chung. Left undetected and untreated, these conditions can be associated with serious adverse outcomes and even death.
Dr. Chung described newborn screening as a “one of the most successful public health programs, supporting health equity by screening almost every US baby after birth and then bringing timely treatments when relevant even before the baby develops symptoms of a disorder.” In this way, newborn screening has “saved lives and decreased disease burdens.”
There are at present 38 core conditions that the Department of Health and Human Services (HHS) regards as the most critical to screen for and 26 secondary conditions associated with these core disorders. This is called the Recommended Uniform Screening Panel (RUSP). Guidance regarding the most appropriate application of newborn screening tests, technologies and standards are provided by the Advisory Committee on Heritable Disorders in Newborns and Children (ACHDNC).
Each state “independently determines which screening tests are performed and what follow-up is provided.”4 Information about which tests are provided by which states can be found on the “Report Card” of the National Organization for Rare Diseases (NORD).
Challenges in Expanding the Current Newborn Screening
One of the major drawbacks in the current system is that “we don’t screen for enough diseases,” according to Zhanzhi Hu, PhD, of the Department of Systems Biology and the Department of Biomedical Information, Columbia University, New York City. “There are over 10,000 rare genetic diseases, but we’re currently screening for fewer than 100,” he told Neurology Reviews. Although in the United States, there are about 700-800 drugs approved for genetic diseases, “we can’t identify patients with these diseases early enough for the ideal window when treatments are most effective.”
Moreover, it’s a “lengthy process” to add new diseases to RUSP. “New conditions are added at the pace of less than one per year, on average — even for the hundreds of diseases for which there are treatments,” he said. “If we keep going at the current pace, we won’t be able to screen for those diseases for another few hundred years.”
Speeding up the pace of including new diseases in newborn screening is challenging because “we have more diseases than we have development dollars for,” Dr. Hu said. “Big pharmaceutical companies are reluctant to invest in rare diseases because the population is so small and it’s hard and expensive to develop such drugs. So if we can identify patients first, there will be more interest in developing treatments down the road.”
On the other hand, for trials to take place, these babies have to be identified in a timely manner — which requires testing. “Right now, we have a deadlock,” Dr. Hu said. “To nominate a disease, you need an approved treatment. But to get a treatment developed, you need to identify patients suitable for a clinical trial. If you have to wait for the symptoms to show up, the damage has already manifested and is irreversible. Our chance is to recognize the disease before symptom onset and then start treatment. I would call this a ‘chicken-and-egg’ problem.”
Dr. Hu is passionate about expanding newborn screening, and he has a very personal reason. Two of his children have a rare genetic disease. “My younger son, now 13 years old, was diagnosed at a much earlier age than my older son, although he had very few symptoms at the time, because his older brother was known to have the disease. As a result of this, his outcome was much better.” By contrast, Dr. Hu’s oldest son — now age 16 — wasn’t diagnosed until he became symptomatic.
His quest led him to join forces with Dr. Chung in conducting the Genomic Uniform-screening Against Rare Disease in All Newborns (Guardian) study, which screens newborns for more than 450 genetic conditions not currently screened as part of the standard newborn screening. To date, the study — which focuses on babies born in New York City — has screened about 11,000 infants.
“To accumulate enough evidence requires screening at least 100,000 babies because one requirement for nominating a disease for national inclusion in RUSP is an ‘N of 1’ study — meaning, to identify at least one positive patient using the proposed screening method in a prospective study,” Dr. Hu explained. “Most are rare diseases with an incidence rate of around one in 100,000. So getting to that magic number of 100,000 participants should enable us to hit that ‘N of 1’ for most diseases.”
The most challenging part, according to Dr. Hu, is the requirement of a prospective study, which means that you have to conduct a large-scale study enrolling tens of thousands of families and babies. If done for individual diseases (as has been the case in the past), “this is a huge cost and very inefficient.”
In reality, he added, the true incidence of these diseases is unclear. “Incidence rates are based on historical data rather than prospective studies. We’ve already seen some diseases show up more frequently than previously recorded, while others have shown up less frequently.”
For example, in the 11,000 babies screened to date, at least three girls with Rett syndrome have been identified, which is “quite a bit higher” than what has previously been identified in the literature (ie, one in 10,000-12,000 births). “This is a highly unmet need for these families because if you can initiate early treatment — at age 1, or even younger — the outcome will be better.”
He noted that there is at least one clinical trial underway for treating Rett syndrome, which has yielded “promising” data.5 “We’re hoping that by screening for diseases like Rett and identifying patients early, this will go hand-in-hand with clinical drug development. It can speed both the approval of the treatment and the addition to the newborn screening list,” Dr. Hu stated.
Screening and Drug Development Working in Tandem
Sequencing technologies have advanced and become more sophisticated as well as less costly, so interest in expanding newborn screening through newborn genome sequencing has increased. In fact, many states currently have incorporated genetic testing into newborn screening for conditions without biochemical markers. Additionally, newborn genomic sequencing is also used for further testing in infants with abnormal biochemical screening results.6
Genomic sequencing “identifies nucleotide changes that are the underlying etiology of monogenic disorders.”6 Its use could potentially enable identification of over 500 genetic disorders for which an newborn screening assay is not currently available, said Dr. Hu.
“Molecular DNA analysis has been integrated into newborn testing either as a first- or second-tier test for several conditions, including cystic fibrosis, severe combined immunodeficiency, and spinal muscular atrophy (SMA),” Dr. Hu said.
Dr. Hu pointed to SMA to illustrate the power and potential of newborn screening working hand-in-hand with the development of new treatments. SMA is a neurodegenerative disorder caused by mutations in SMN1, which encodes survival motor neuron protein (SMN).7 Deficiencies in SMN results in loss of motor neurons with muscle weakness and, often, early death.7A pilot study, on which Dr. Chung was the senior author, used both biochemical and genetic testing of close to 4000 newborns and found an SMA carrier frequency of 1.5%. One newborn was identified who had a homozygous SMN1 gene deletion and two copies of SMN2, strongly suggesting the presence of a severe type 1 SMA phenotype.8
At age 15 days, the baby was treated with nusinersen, an injection administered into the fluid surrounding the spinal cord, and the first FDA-approved genetic treatment for SMA. At the time of study publication, the baby was 12 months old, “meeting all developmental milestones and free of any respiratory issues,” the authors report.
“Screening for SMA — which was added to the RUSP in 2018 — has dramatically transformed what used to be the most common genetic cause of death in children under the age of 2,” Dr. Chung said. “Now, a once-and-done IV infusion of genetic therapy right after screening has transformed everything, taking what used to be a lethal condition and allowing children to grow up healthy.”
Advocating for Inclusion of Diseases With No Current Treatment
At present, any condition included in the RUSP is required to have a treatment, which can be dietary, surgical/procedural, or an FDA-approved drug-based agent. Unfortunately, a wide range of neurodevelopmental diseases still have no known treatments. But lack of availability of treatment shouldn’t invalidate a disease from being included in the RUSP, because even if there is no specific treatment for the condition itself, early intervention can still be initiated to prevent some of the manifestations of the condition, said Dr. Hu.
“For example, most patients with these diseases will sooner or later undergo seizures,” Dr. Hu remarked. “We know that repeated seizures can cause brain damage. If we can diagnose the disease before the seizures start to take place, we can put preventive seizure control interventions in place, even if there is no direct ‘treatment’ for the condition itself.”
Early identification can lead to early intervention, which can have other benefits, Dr. Hu noted. “If we train the brain at a young age, when the brain is most receptive, even though a disease may be progressive and will worsen, those abilities acquired earlier will last longer and remain in place longer. When these skills are acquired later, they’re forgotten sooner. This isn’t a ‘cure,’ but it will help with functional improvement.”
Moreover, parents are “interested in knowing that their child has a condition, even if no treatment is currently available for that disorder, according to our research,” Dr. Chung said. “We found that the parents we interviewed endorsed the nonmedical utility of having access to information, even in the absence of a ‘cure,’ so they could prepare for medical issues that might arise down the road and make informed choices.”9
Nina Gold, MD, director of Prenatal Medical Genetics and associate director for Research for Massachusetts General Brigham Personalized Medicine, Boston, obtained similar findings in her own research, which is currently under review for publication. “We conducted focus groups and one-on-one interviews with parents from diverse racial and socioeconomic backgrounds. At least one parent said they didn’t want to compare their child to other children if their child might have a different developmental trajectory. They stressed that the information would be helpful, even if there was no immediate clinical utility.”
Additionally, there are an “increasing number of fetal therapies for rare disorders, so information about a genetic disease in an older child can be helpful for parents who may go on to have another pregnancy,” Dr. Gold noted.
Dr. Hu detailed several other reasons for including a wider range of disorders in the RUSP. Doing so helps families avoid a “stressful and expensive diagnostic odyssey and also provides equitable access to a diagnosis.” And if these patients are identified early, “we can connect the family with clinical trials already underway or connect them to an organization such as the Accelerating Medicines Partnership (AMP) Program Bespoke Gene Therapy Consortium (AMP BGTC). Bespoke “brings together partners from the public, private, and nonprofit sectors to foster development of gene therapies intended to treat rare genetic diseases, which affect populations too small for viable commercial development.”
Next Steps Following Screening
Rebecca Sponberg, NP, of the Children’s Hospital of Orange County, UC Irvine School of Medicine, California, is part of a broader multidisciplinary team that interfaces with parents whose newborns have screened positive for a genetic disorder. The team also includes a biochemical geneticist, a pediatric neurologist, a pediatric endocrinologist, a genetic counselor, and a social worker.
Different states and locations have different procedures for receiving test results, said Dr. Chung. In some, pediatricians are the ones who receive the results, and they are tasked with the responsibility of making sure the children can start getting appropriate care. In particular, these pediatricians are associated with centers of excellence that specialize in working with families around these conditions. Other facilities have multidisciplinary teams.
Ms. Sponberg gave an example of how the process unfolded with X-linked adrenoleukodystrophy, a rare genetic disorder that affects the white matter of the nervous system and the adrenal cortex.10 “This is the most common peroxisomal disorder, affecting one in 20,000 males,” she said. “There are several different forms of the disorder, but males are most at risk for having the cerebral form, which can lead to neurological regression and hasten death. But the regression does not appear until 4 to 12 years of age.”
A baby who screens positive on the initial newborn screening has repeat testing; and if it’s confirmed, the family meets the entire team to help them understand what the disorder is, what to expect, and how it’s monitored and managed. “Children have to be followed closely with a brain MRI every 6 months to detect brain abnormalities quickly,” Ms. Sponberg explained “And we do regular bloodwork to look for adrenocortical insufficiency.”
A child who shows concerning changes on the MRI or abnormal blood test findings is immediately seen by the relevant specialist. “So far, our center has had one patient who had MRI changes consistent with the cerebral form of the disease and the patient was immediately able to receive a bone marrow transplant,” she reported. “We don’t think this child’s condition would have been picked up so quickly or treatment initiated so rapidly if we hadn’t known about it through newborn screening.”
Educating and Involving Families
Part of the role of clinicians is to provide education regarding newborn screening to families, according to Ms. Sponberg. “In my role, I have to call parents to tell them their child screened positive for a genetic condition and that we need to proceed with confirmatory testing,” she said. “We let them know if there’s a high concern that this might be a true positive for the condition, and we offer them information so they know what to expect.”
Unfortunately, Ms. Sponberg said, in the absence of education, some families are skeptical. “When I call families directly, some think it’s a scam and it can be hard to earn their trust. We need to do a better job educating families, especially our pregnant individuals, that testing will occur and if anything is abnormal, they will receive a call.”
References
1. Levy HL. Robert Guthrie and the Trials and Tribulations of Newborn Screening. Int J Neonatal Screen. 2021 Jan 19;7(1):5. doi: 10.3390/ijns7010005.
2. Chace DH et al. Clinical Chemistry and Dried Blood Spots: Increasing Laboratory Utilization by Improved Understanding of Quantitative Challenges. Bioanalysis. 2014;6(21):2791-2794. doi: 10.4155/bio.14.237.
3. Gold NB et al. Perspectives of Rare Disease Experts on Newborn Genome Sequencing. JAMA Netw Open. 2023 May 1;6(5):e2312231. doi: 10.1001/jamanetworkopen.2023.12231.
4. Weismiller DG. Expanded Newborn Screening: Information and Resources for the Family Physician. Am Fam Physician. 2017 Jun 1;95(11):703-709. https://www.aafp.org/pubs/afp/issues/2017/0601/p703.html.
5. Neul JL et al. Trofinetide for the Treatment of Rett Syndrome: A Randomized Phase 3 Study. Nat Med. 2023 Jun;29(6):1468-1475. doi: 10.1038/s41591-023-02398-1.
6. Chen T et al. Genomic Sequencing as a First-Tier Screening Test and Outcomes of Newborn Screening. JAMA Netw Open. 2023 Sep 5;6(9):e2331162. doi: 10.1001/jamanetworkopen.2023.31162.
7. Mercuri E et al. Spinal Muscular Atrophy. Nat Rev Dis Primers. 2022 Aug 4;8(1):52. doi: 10.1038/s41572-022-00380-8.
8. Kraszewski JN et al. Pilot Study of Population-Based Newborn Screening for Spinal Muscular Atrophy in New York State. Genet Med. 2018 Jun;20(6):608-613. doi: 10.1038/gim.2017.152.
9. Timmins GT et al. Diverse Parental Perspectives of the Social and Educational Needs for Expanding Newborn Screening Through Genomic Sequencing. Public Health Genomics. 2022 Sep 15:1-8. doi: 10.1159/000526382.
10. Turk BR et al. X-linked Adrenoleukodystrophy: Pathology, Pathophysiology, Diagnostic Testing, Newborn Screening and Therapies. Int J Dev Neurosci. 2020 Feb;80(1):52-72. doi: 10.1002/jdn.10003.
Newborn screening programs are public health services aimed at ensuring that the close to 4 million infants born each year in the United States are screened for certain serious disorders at birth. These disorders, albeit rare, are detected in roughly 12,500 newborn babies every year.
Newborn screening isn’t new, although it has expanded and transformed over the decades. The first newborn screening test was developed in the 1960s to detect phenylketonuria (PKU).1 Since then, the number of conditions screened for has increased, with programs in every US state and territory. “Newborn screening is well established now, not experimental or newfangled,” Wendy Chung, MD, PhD, professor of pediatrics, Harvard Medical School, Boston, Massachusetts, told Neurology Reviews.
In newborn screening, blood drawn from the baby’s heel is applied to specialized filter paper, which is then subjected to several analytical methods, including tandem mass spectrometry and molecular analyses to detect biomarkers for the diseases.2 More recently, genomic sequencing is being piloted as part of consented research studies.3
Newborn screening includes not only biochemical and genetic testing, but also includes noninvasive screening for hearing loss or for critical congenital heart disease using pulse oximetry. And newborn screening goes beyond analysis of a single drop of blood. Rather, “it’s an entire system, with the goal of identifying babies with genetic disorders who otherwise have no obvious symptoms,” said Dr. Chung. Left undetected and untreated, these conditions can be associated with serious adverse outcomes and even death.
Dr. Chung described newborn screening as a “one of the most successful public health programs, supporting health equity by screening almost every US baby after birth and then bringing timely treatments when relevant even before the baby develops symptoms of a disorder.” In this way, newborn screening has “saved lives and decreased disease burdens.”
There are at present 38 core conditions that the Department of Health and Human Services (HHS) regards as the most critical to screen for and 26 secondary conditions associated with these core disorders. This is called the Recommended Uniform Screening Panel (RUSP). Guidance regarding the most appropriate application of newborn screening tests, technologies and standards are provided by the Advisory Committee on Heritable Disorders in Newborns and Children (ACHDNC).
Each state “independently determines which screening tests are performed and what follow-up is provided.”4 Information about which tests are provided by which states can be found on the “Report Card” of the National Organization for Rare Diseases (NORD).
Challenges in Expanding the Current Newborn Screening
One of the major drawbacks in the current system is that “we don’t screen for enough diseases,” according to Zhanzhi Hu, PhD, of the Department of Systems Biology and the Department of Biomedical Information, Columbia University, New York City. “There are over 10,000 rare genetic diseases, but we’re currently screening for fewer than 100,” he told Neurology Reviews. Although in the United States, there are about 700-800 drugs approved for genetic diseases, “we can’t identify patients with these diseases early enough for the ideal window when treatments are most effective.”
Moreover, it’s a “lengthy process” to add new diseases to RUSP. “New conditions are added at the pace of less than one per year, on average — even for the hundreds of diseases for which there are treatments,” he said. “If we keep going at the current pace, we won’t be able to screen for those diseases for another few hundred years.”
Speeding up the pace of including new diseases in newborn screening is challenging because “we have more diseases than we have development dollars for,” Dr. Hu said. “Big pharmaceutical companies are reluctant to invest in rare diseases because the population is so small and it’s hard and expensive to develop such drugs. So if we can identify patients first, there will be more interest in developing treatments down the road.”
On the other hand, for trials to take place, these babies have to be identified in a timely manner — which requires testing. “Right now, we have a deadlock,” Dr. Hu said. “To nominate a disease, you need an approved treatment. But to get a treatment developed, you need to identify patients suitable for a clinical trial. If you have to wait for the symptoms to show up, the damage has already manifested and is irreversible. Our chance is to recognize the disease before symptom onset and then start treatment. I would call this a ‘chicken-and-egg’ problem.”
Dr. Hu is passionate about expanding newborn screening, and he has a very personal reason. Two of his children have a rare genetic disease. “My younger son, now 13 years old, was diagnosed at a much earlier age than my older son, although he had very few symptoms at the time, because his older brother was known to have the disease. As a result of this, his outcome was much better.” By contrast, Dr. Hu’s oldest son — now age 16 — wasn’t diagnosed until he became symptomatic.
His quest led him to join forces with Dr. Chung in conducting the Genomic Uniform-screening Against Rare Disease in All Newborns (Guardian) study, which screens newborns for more than 450 genetic conditions not currently screened as part of the standard newborn screening. To date, the study — which focuses on babies born in New York City — has screened about 11,000 infants.
“To accumulate enough evidence requires screening at least 100,000 babies because one requirement for nominating a disease for national inclusion in RUSP is an ‘N of 1’ study — meaning, to identify at least one positive patient using the proposed screening method in a prospective study,” Dr. Hu explained. “Most are rare diseases with an incidence rate of around one in 100,000. So getting to that magic number of 100,000 participants should enable us to hit that ‘N of 1’ for most diseases.”
The most challenging part, according to Dr. Hu, is the requirement of a prospective study, which means that you have to conduct a large-scale study enrolling tens of thousands of families and babies. If done for individual diseases (as has been the case in the past), “this is a huge cost and very inefficient.”
In reality, he added, the true incidence of these diseases is unclear. “Incidence rates are based on historical data rather than prospective studies. We’ve already seen some diseases show up more frequently than previously recorded, while others have shown up less frequently.”
For example, in the 11,000 babies screened to date, at least three girls with Rett syndrome have been identified, which is “quite a bit higher” than what has previously been identified in the literature (ie, one in 10,000-12,000 births). “This is a highly unmet need for these families because if you can initiate early treatment — at age 1, or even younger — the outcome will be better.”
He noted that there is at least one clinical trial underway for treating Rett syndrome, which has yielded “promising” data.5 “We’re hoping that by screening for diseases like Rett and identifying patients early, this will go hand-in-hand with clinical drug development. It can speed both the approval of the treatment and the addition to the newborn screening list,” Dr. Hu stated.
Screening and Drug Development Working in Tandem
Sequencing technologies have advanced and become more sophisticated as well as less costly, so interest in expanding newborn screening through newborn genome sequencing has increased. In fact, many states currently have incorporated genetic testing into newborn screening for conditions without biochemical markers. Additionally, newborn genomic sequencing is also used for further testing in infants with abnormal biochemical screening results.6
Genomic sequencing “identifies nucleotide changes that are the underlying etiology of monogenic disorders.”6 Its use could potentially enable identification of over 500 genetic disorders for which an newborn screening assay is not currently available, said Dr. Hu.
“Molecular DNA analysis has been integrated into newborn testing either as a first- or second-tier test for several conditions, including cystic fibrosis, severe combined immunodeficiency, and spinal muscular atrophy (SMA),” Dr. Hu said.
Dr. Hu pointed to SMA to illustrate the power and potential of newborn screening working hand-in-hand with the development of new treatments. SMA is a neurodegenerative disorder caused by mutations in SMN1, which encodes survival motor neuron protein (SMN).7 Deficiencies in SMN results in loss of motor neurons with muscle weakness and, often, early death.7A pilot study, on which Dr. Chung was the senior author, used both biochemical and genetic testing of close to 4000 newborns and found an SMA carrier frequency of 1.5%. One newborn was identified who had a homozygous SMN1 gene deletion and two copies of SMN2, strongly suggesting the presence of a severe type 1 SMA phenotype.8
At age 15 days, the baby was treated with nusinersen, an injection administered into the fluid surrounding the spinal cord, and the first FDA-approved genetic treatment for SMA. At the time of study publication, the baby was 12 months old, “meeting all developmental milestones and free of any respiratory issues,” the authors report.
“Screening for SMA — which was added to the RUSP in 2018 — has dramatically transformed what used to be the most common genetic cause of death in children under the age of 2,” Dr. Chung said. “Now, a once-and-done IV infusion of genetic therapy right after screening has transformed everything, taking what used to be a lethal condition and allowing children to grow up healthy.”
Advocating for Inclusion of Diseases With No Current Treatment
At present, any condition included in the RUSP is required to have a treatment, which can be dietary, surgical/procedural, or an FDA-approved drug-based agent. Unfortunately, a wide range of neurodevelopmental diseases still have no known treatments. But lack of availability of treatment shouldn’t invalidate a disease from being included in the RUSP, because even if there is no specific treatment for the condition itself, early intervention can still be initiated to prevent some of the manifestations of the condition, said Dr. Hu.
“For example, most patients with these diseases will sooner or later undergo seizures,” Dr. Hu remarked. “We know that repeated seizures can cause brain damage. If we can diagnose the disease before the seizures start to take place, we can put preventive seizure control interventions in place, even if there is no direct ‘treatment’ for the condition itself.”
Early identification can lead to early intervention, which can have other benefits, Dr. Hu noted. “If we train the brain at a young age, when the brain is most receptive, even though a disease may be progressive and will worsen, those abilities acquired earlier will last longer and remain in place longer. When these skills are acquired later, they’re forgotten sooner. This isn’t a ‘cure,’ but it will help with functional improvement.”
Moreover, parents are “interested in knowing that their child has a condition, even if no treatment is currently available for that disorder, according to our research,” Dr. Chung said. “We found that the parents we interviewed endorsed the nonmedical utility of having access to information, even in the absence of a ‘cure,’ so they could prepare for medical issues that might arise down the road and make informed choices.”9
Nina Gold, MD, director of Prenatal Medical Genetics and associate director for Research for Massachusetts General Brigham Personalized Medicine, Boston, obtained similar findings in her own research, which is currently under review for publication. “We conducted focus groups and one-on-one interviews with parents from diverse racial and socioeconomic backgrounds. At least one parent said they didn’t want to compare their child to other children if their child might have a different developmental trajectory. They stressed that the information would be helpful, even if there was no immediate clinical utility.”
Additionally, there are an “increasing number of fetal therapies for rare disorders, so information about a genetic disease in an older child can be helpful for parents who may go on to have another pregnancy,” Dr. Gold noted.
Dr. Hu detailed several other reasons for including a wider range of disorders in the RUSP. Doing so helps families avoid a “stressful and expensive diagnostic odyssey and also provides equitable access to a diagnosis.” And if these patients are identified early, “we can connect the family with clinical trials already underway or connect them to an organization such as the Accelerating Medicines Partnership (AMP) Program Bespoke Gene Therapy Consortium (AMP BGTC). Bespoke “brings together partners from the public, private, and nonprofit sectors to foster development of gene therapies intended to treat rare genetic diseases, which affect populations too small for viable commercial development.”
Next Steps Following Screening
Rebecca Sponberg, NP, of the Children’s Hospital of Orange County, UC Irvine School of Medicine, California, is part of a broader multidisciplinary team that interfaces with parents whose newborns have screened positive for a genetic disorder. The team also includes a biochemical geneticist, a pediatric neurologist, a pediatric endocrinologist, a genetic counselor, and a social worker.
Different states and locations have different procedures for receiving test results, said Dr. Chung. In some, pediatricians are the ones who receive the results, and they are tasked with the responsibility of making sure the children can start getting appropriate care. In particular, these pediatricians are associated with centers of excellence that specialize in working with families around these conditions. Other facilities have multidisciplinary teams.
Ms. Sponberg gave an example of how the process unfolded with X-linked adrenoleukodystrophy, a rare genetic disorder that affects the white matter of the nervous system and the adrenal cortex.10 “This is the most common peroxisomal disorder, affecting one in 20,000 males,” she said. “There are several different forms of the disorder, but males are most at risk for having the cerebral form, which can lead to neurological regression and hasten death. But the regression does not appear until 4 to 12 years of age.”
A baby who screens positive on the initial newborn screening has repeat testing; and if it’s confirmed, the family meets the entire team to help them understand what the disorder is, what to expect, and how it’s monitored and managed. “Children have to be followed closely with a brain MRI every 6 months to detect brain abnormalities quickly,” Ms. Sponberg explained “And we do regular bloodwork to look for adrenocortical insufficiency.”
A child who shows concerning changes on the MRI or abnormal blood test findings is immediately seen by the relevant specialist. “So far, our center has had one patient who had MRI changes consistent with the cerebral form of the disease and the patient was immediately able to receive a bone marrow transplant,” she reported. “We don’t think this child’s condition would have been picked up so quickly or treatment initiated so rapidly if we hadn’t known about it through newborn screening.”
Educating and Involving Families
Part of the role of clinicians is to provide education regarding newborn screening to families, according to Ms. Sponberg. “In my role, I have to call parents to tell them their child screened positive for a genetic condition and that we need to proceed with confirmatory testing,” she said. “We let them know if there’s a high concern that this might be a true positive for the condition, and we offer them information so they know what to expect.”
Unfortunately, Ms. Sponberg said, in the absence of education, some families are skeptical. “When I call families directly, some think it’s a scam and it can be hard to earn their trust. We need to do a better job educating families, especially our pregnant individuals, that testing will occur and if anything is abnormal, they will receive a call.”
References
1. Levy HL. Robert Guthrie and the Trials and Tribulations of Newborn Screening. Int J Neonatal Screen. 2021 Jan 19;7(1):5. doi: 10.3390/ijns7010005.
2. Chace DH et al. Clinical Chemistry and Dried Blood Spots: Increasing Laboratory Utilization by Improved Understanding of Quantitative Challenges. Bioanalysis. 2014;6(21):2791-2794. doi: 10.4155/bio.14.237.
3. Gold NB et al. Perspectives of Rare Disease Experts on Newborn Genome Sequencing. JAMA Netw Open. 2023 May 1;6(5):e2312231. doi: 10.1001/jamanetworkopen.2023.12231.
4. Weismiller DG. Expanded Newborn Screening: Information and Resources for the Family Physician. Am Fam Physician. 2017 Jun 1;95(11):703-709. https://www.aafp.org/pubs/afp/issues/2017/0601/p703.html.
5. Neul JL et al. Trofinetide for the Treatment of Rett Syndrome: A Randomized Phase 3 Study. Nat Med. 2023 Jun;29(6):1468-1475. doi: 10.1038/s41591-023-02398-1.
6. Chen T et al. Genomic Sequencing as a First-Tier Screening Test and Outcomes of Newborn Screening. JAMA Netw Open. 2023 Sep 5;6(9):e2331162. doi: 10.1001/jamanetworkopen.2023.31162.
7. Mercuri E et al. Spinal Muscular Atrophy. Nat Rev Dis Primers. 2022 Aug 4;8(1):52. doi: 10.1038/s41572-022-00380-8.
8. Kraszewski JN et al. Pilot Study of Population-Based Newborn Screening for Spinal Muscular Atrophy in New York State. Genet Med. 2018 Jun;20(6):608-613. doi: 10.1038/gim.2017.152.
9. Timmins GT et al. Diverse Parental Perspectives of the Social and Educational Needs for Expanding Newborn Screening Through Genomic Sequencing. Public Health Genomics. 2022 Sep 15:1-8. doi: 10.1159/000526382.
10. Turk BR et al. X-linked Adrenoleukodystrophy: Pathology, Pathophysiology, Diagnostic Testing, Newborn Screening and Therapies. Int J Dev Neurosci. 2020 Feb;80(1):52-72. doi: 10.1002/jdn.10003.
Newborn screening programs are public health services aimed at ensuring that the close to 4 million infants born each year in the United States are screened for certain serious disorders at birth. These disorders, albeit rare, are detected in roughly 12,500 newborn babies every year.
Newborn screening isn’t new, although it has expanded and transformed over the decades. The first newborn screening test was developed in the 1960s to detect phenylketonuria (PKU).1 Since then, the number of conditions screened for has increased, with programs in every US state and territory. “Newborn screening is well established now, not experimental or newfangled,” Wendy Chung, MD, PhD, professor of pediatrics, Harvard Medical School, Boston, Massachusetts, told Neurology Reviews.
In newborn screening, blood drawn from the baby’s heel is applied to specialized filter paper, which is then subjected to several analytical methods, including tandem mass spectrometry and molecular analyses to detect biomarkers for the diseases.2 More recently, genomic sequencing is being piloted as part of consented research studies.3
Newborn screening includes not only biochemical and genetic testing, but also includes noninvasive screening for hearing loss or for critical congenital heart disease using pulse oximetry. And newborn screening goes beyond analysis of a single drop of blood. Rather, “it’s an entire system, with the goal of identifying babies with genetic disorders who otherwise have no obvious symptoms,” said Dr. Chung. Left undetected and untreated, these conditions can be associated with serious adverse outcomes and even death.
Dr. Chung described newborn screening as a “one of the most successful public health programs, supporting health equity by screening almost every US baby after birth and then bringing timely treatments when relevant even before the baby develops symptoms of a disorder.” In this way, newborn screening has “saved lives and decreased disease burdens.”
There are at present 38 core conditions that the Department of Health and Human Services (HHS) regards as the most critical to screen for and 26 secondary conditions associated with these core disorders. This is called the Recommended Uniform Screening Panel (RUSP). Guidance regarding the most appropriate application of newborn screening tests, technologies and standards are provided by the Advisory Committee on Heritable Disorders in Newborns and Children (ACHDNC).
Each state “independently determines which screening tests are performed and what follow-up is provided.”4 Information about which tests are provided by which states can be found on the “Report Card” of the National Organization for Rare Diseases (NORD).
Challenges in Expanding the Current Newborn Screening
One of the major drawbacks in the current system is that “we don’t screen for enough diseases,” according to Zhanzhi Hu, PhD, of the Department of Systems Biology and the Department of Biomedical Information, Columbia University, New York City. “There are over 10,000 rare genetic diseases, but we’re currently screening for fewer than 100,” he told Neurology Reviews. Although in the United States, there are about 700-800 drugs approved for genetic diseases, “we can’t identify patients with these diseases early enough for the ideal window when treatments are most effective.”
Moreover, it’s a “lengthy process” to add new diseases to RUSP. “New conditions are added at the pace of less than one per year, on average — even for the hundreds of diseases for which there are treatments,” he said. “If we keep going at the current pace, we won’t be able to screen for those diseases for another few hundred years.”
Speeding up the pace of including new diseases in newborn screening is challenging because “we have more diseases than we have development dollars for,” Dr. Hu said. “Big pharmaceutical companies are reluctant to invest in rare diseases because the population is so small and it’s hard and expensive to develop such drugs. So if we can identify patients first, there will be more interest in developing treatments down the road.”
On the other hand, for trials to take place, these babies have to be identified in a timely manner — which requires testing. “Right now, we have a deadlock,” Dr. Hu said. “To nominate a disease, you need an approved treatment. But to get a treatment developed, you need to identify patients suitable for a clinical trial. If you have to wait for the symptoms to show up, the damage has already manifested and is irreversible. Our chance is to recognize the disease before symptom onset and then start treatment. I would call this a ‘chicken-and-egg’ problem.”
Dr. Hu is passionate about expanding newborn screening, and he has a very personal reason. Two of his children have a rare genetic disease. “My younger son, now 13 years old, was diagnosed at a much earlier age than my older son, although he had very few symptoms at the time, because his older brother was known to have the disease. As a result of this, his outcome was much better.” By contrast, Dr. Hu’s oldest son — now age 16 — wasn’t diagnosed until he became symptomatic.
His quest led him to join forces with Dr. Chung in conducting the Genomic Uniform-screening Against Rare Disease in All Newborns (Guardian) study, which screens newborns for more than 450 genetic conditions not currently screened as part of the standard newborn screening. To date, the study — which focuses on babies born in New York City — has screened about 11,000 infants.
“To accumulate enough evidence requires screening at least 100,000 babies because one requirement for nominating a disease for national inclusion in RUSP is an ‘N of 1’ study — meaning, to identify at least one positive patient using the proposed screening method in a prospective study,” Dr. Hu explained. “Most are rare diseases with an incidence rate of around one in 100,000. So getting to that magic number of 100,000 participants should enable us to hit that ‘N of 1’ for most diseases.”
The most challenging part, according to Dr. Hu, is the requirement of a prospective study, which means that you have to conduct a large-scale study enrolling tens of thousands of families and babies. If done for individual diseases (as has been the case in the past), “this is a huge cost and very inefficient.”
In reality, he added, the true incidence of these diseases is unclear. “Incidence rates are based on historical data rather than prospective studies. We’ve already seen some diseases show up more frequently than previously recorded, while others have shown up less frequently.”
For example, in the 11,000 babies screened to date, at least three girls with Rett syndrome have been identified, which is “quite a bit higher” than what has previously been identified in the literature (ie, one in 10,000-12,000 births). “This is a highly unmet need for these families because if you can initiate early treatment — at age 1, or even younger — the outcome will be better.”
He noted that there is at least one clinical trial underway for treating Rett syndrome, which has yielded “promising” data.5 “We’re hoping that by screening for diseases like Rett and identifying patients early, this will go hand-in-hand with clinical drug development. It can speed both the approval of the treatment and the addition to the newborn screening list,” Dr. Hu stated.
Screening and Drug Development Working in Tandem
Sequencing technologies have advanced and become more sophisticated as well as less costly, so interest in expanding newborn screening through newborn genome sequencing has increased. In fact, many states currently have incorporated genetic testing into newborn screening for conditions without biochemical markers. Additionally, newborn genomic sequencing is also used for further testing in infants with abnormal biochemical screening results.6
Genomic sequencing “identifies nucleotide changes that are the underlying etiology of monogenic disorders.”6 Its use could potentially enable identification of over 500 genetic disorders for which an newborn screening assay is not currently available, said Dr. Hu.
“Molecular DNA analysis has been integrated into newborn testing either as a first- or second-tier test for several conditions, including cystic fibrosis, severe combined immunodeficiency, and spinal muscular atrophy (SMA),” Dr. Hu said.
Dr. Hu pointed to SMA to illustrate the power and potential of newborn screening working hand-in-hand with the development of new treatments. SMA is a neurodegenerative disorder caused by mutations in SMN1, which encodes survival motor neuron protein (SMN).7 Deficiencies in SMN results in loss of motor neurons with muscle weakness and, often, early death.7A pilot study, on which Dr. Chung was the senior author, used both biochemical and genetic testing of close to 4000 newborns and found an SMA carrier frequency of 1.5%. One newborn was identified who had a homozygous SMN1 gene deletion and two copies of SMN2, strongly suggesting the presence of a severe type 1 SMA phenotype.8
At age 15 days, the baby was treated with nusinersen, an injection administered into the fluid surrounding the spinal cord, and the first FDA-approved genetic treatment for SMA. At the time of study publication, the baby was 12 months old, “meeting all developmental milestones and free of any respiratory issues,” the authors report.
“Screening for SMA — which was added to the RUSP in 2018 — has dramatically transformed what used to be the most common genetic cause of death in children under the age of 2,” Dr. Chung said. “Now, a once-and-done IV infusion of genetic therapy right after screening has transformed everything, taking what used to be a lethal condition and allowing children to grow up healthy.”
Advocating for Inclusion of Diseases With No Current Treatment
At present, any condition included in the RUSP is required to have a treatment, which can be dietary, surgical/procedural, or an FDA-approved drug-based agent. Unfortunately, a wide range of neurodevelopmental diseases still have no known treatments. But lack of availability of treatment shouldn’t invalidate a disease from being included in the RUSP, because even if there is no specific treatment for the condition itself, early intervention can still be initiated to prevent some of the manifestations of the condition, said Dr. Hu.
“For example, most patients with these diseases will sooner or later undergo seizures,” Dr. Hu remarked. “We know that repeated seizures can cause brain damage. If we can diagnose the disease before the seizures start to take place, we can put preventive seizure control interventions in place, even if there is no direct ‘treatment’ for the condition itself.”
Early identification can lead to early intervention, which can have other benefits, Dr. Hu noted. “If we train the brain at a young age, when the brain is most receptive, even though a disease may be progressive and will worsen, those abilities acquired earlier will last longer and remain in place longer. When these skills are acquired later, they’re forgotten sooner. This isn’t a ‘cure,’ but it will help with functional improvement.”
Moreover, parents are “interested in knowing that their child has a condition, even if no treatment is currently available for that disorder, according to our research,” Dr. Chung said. “We found that the parents we interviewed endorsed the nonmedical utility of having access to information, even in the absence of a ‘cure,’ so they could prepare for medical issues that might arise down the road and make informed choices.”9
Nina Gold, MD, director of Prenatal Medical Genetics and associate director for Research for Massachusetts General Brigham Personalized Medicine, Boston, obtained similar findings in her own research, which is currently under review for publication. “We conducted focus groups and one-on-one interviews with parents from diverse racial and socioeconomic backgrounds. At least one parent said they didn’t want to compare their child to other children if their child might have a different developmental trajectory. They stressed that the information would be helpful, even if there was no immediate clinical utility.”
Additionally, there are an “increasing number of fetal therapies for rare disorders, so information about a genetic disease in an older child can be helpful for parents who may go on to have another pregnancy,” Dr. Gold noted.
Dr. Hu detailed several other reasons for including a wider range of disorders in the RUSP. Doing so helps families avoid a “stressful and expensive diagnostic odyssey and also provides equitable access to a diagnosis.” And if these patients are identified early, “we can connect the family with clinical trials already underway or connect them to an organization such as the Accelerating Medicines Partnership (AMP) Program Bespoke Gene Therapy Consortium (AMP BGTC). Bespoke “brings together partners from the public, private, and nonprofit sectors to foster development of gene therapies intended to treat rare genetic diseases, which affect populations too small for viable commercial development.”
Next Steps Following Screening
Rebecca Sponberg, NP, of the Children’s Hospital of Orange County, UC Irvine School of Medicine, California, is part of a broader multidisciplinary team that interfaces with parents whose newborns have screened positive for a genetic disorder. The team also includes a biochemical geneticist, a pediatric neurologist, a pediatric endocrinologist, a genetic counselor, and a social worker.
Different states and locations have different procedures for receiving test results, said Dr. Chung. In some, pediatricians are the ones who receive the results, and they are tasked with the responsibility of making sure the children can start getting appropriate care. In particular, these pediatricians are associated with centers of excellence that specialize in working with families around these conditions. Other facilities have multidisciplinary teams.
Ms. Sponberg gave an example of how the process unfolded with X-linked adrenoleukodystrophy, a rare genetic disorder that affects the white matter of the nervous system and the adrenal cortex.10 “This is the most common peroxisomal disorder, affecting one in 20,000 males,” she said. “There are several different forms of the disorder, but males are most at risk for having the cerebral form, which can lead to neurological regression and hasten death. But the regression does not appear until 4 to 12 years of age.”
A baby who screens positive on the initial newborn screening has repeat testing; and if it’s confirmed, the family meets the entire team to help them understand what the disorder is, what to expect, and how it’s monitored and managed. “Children have to be followed closely with a brain MRI every 6 months to detect brain abnormalities quickly,” Ms. Sponberg explained “And we do regular bloodwork to look for adrenocortical insufficiency.”
A child who shows concerning changes on the MRI or abnormal blood test findings is immediately seen by the relevant specialist. “So far, our center has had one patient who had MRI changes consistent with the cerebral form of the disease and the patient was immediately able to receive a bone marrow transplant,” she reported. “We don’t think this child’s condition would have been picked up so quickly or treatment initiated so rapidly if we hadn’t known about it through newborn screening.”
Educating and Involving Families
Part of the role of clinicians is to provide education regarding newborn screening to families, according to Ms. Sponberg. “In my role, I have to call parents to tell them their child screened positive for a genetic condition and that we need to proceed with confirmatory testing,” she said. “We let them know if there’s a high concern that this might be a true positive for the condition, and we offer them information so they know what to expect.”
Unfortunately, Ms. Sponberg said, in the absence of education, some families are skeptical. “When I call families directly, some think it’s a scam and it can be hard to earn their trust. We need to do a better job educating families, especially our pregnant individuals, that testing will occur and if anything is abnormal, they will receive a call.”
References
1. Levy HL. Robert Guthrie and the Trials and Tribulations of Newborn Screening. Int J Neonatal Screen. 2021 Jan 19;7(1):5. doi: 10.3390/ijns7010005.
2. Chace DH et al. Clinical Chemistry and Dried Blood Spots: Increasing Laboratory Utilization by Improved Understanding of Quantitative Challenges. Bioanalysis. 2014;6(21):2791-2794. doi: 10.4155/bio.14.237.
3. Gold NB et al. Perspectives of Rare Disease Experts on Newborn Genome Sequencing. JAMA Netw Open. 2023 May 1;6(5):e2312231. doi: 10.1001/jamanetworkopen.2023.12231.
4. Weismiller DG. Expanded Newborn Screening: Information and Resources for the Family Physician. Am Fam Physician. 2017 Jun 1;95(11):703-709. https://www.aafp.org/pubs/afp/issues/2017/0601/p703.html.
5. Neul JL et al. Trofinetide for the Treatment of Rett Syndrome: A Randomized Phase 3 Study. Nat Med. 2023 Jun;29(6):1468-1475. doi: 10.1038/s41591-023-02398-1.
6. Chen T et al. Genomic Sequencing as a First-Tier Screening Test and Outcomes of Newborn Screening. JAMA Netw Open. 2023 Sep 5;6(9):e2331162. doi: 10.1001/jamanetworkopen.2023.31162.
7. Mercuri E et al. Spinal Muscular Atrophy. Nat Rev Dis Primers. 2022 Aug 4;8(1):52. doi: 10.1038/s41572-022-00380-8.
8. Kraszewski JN et al. Pilot Study of Population-Based Newborn Screening for Spinal Muscular Atrophy in New York State. Genet Med. 2018 Jun;20(6):608-613. doi: 10.1038/gim.2017.152.
9. Timmins GT et al. Diverse Parental Perspectives of the Social and Educational Needs for Expanding Newborn Screening Through Genomic Sequencing. Public Health Genomics. 2022 Sep 15:1-8. doi: 10.1159/000526382.
10. Turk BR et al. X-linked Adrenoleukodystrophy: Pathology, Pathophysiology, Diagnostic Testing, Newborn Screening and Therapies. Int J Dev Neurosci. 2020 Feb;80(1):52-72. doi: 10.1002/jdn.10003.
Genetic Testing for ALS, Now a Standard, Creates a Path Toward Individualized Care
The first therapy targeted at modifying a mutant gene associated with amyotrophic lateral sclerosis (ALS), approved in early 2023, has offered reassurance that the biology of ALS, when known, is targetable. Historically, the disease has been considered a clinical diagnosis, but the
Despite a narrow indication, the only therapy targeted at an ALS-associated gene so far, SOD1 ALS, supports the premise that the biology of ALS can be modified, according to Christina N. Fournier, MD, an associate professor in the Department of Neurology, Emory University, Atlanta, Georgia.
Rather than a single pathological entity, ALS is best understood as the end result of many different pathological processes. Each might require its own targeted therapy in order to interrupt the upstream biological pathways that drive disease.
About 15% of ALS Has An Identifiable Genetic Cause
A family history of ALS is present in about 10% of cases. A genetic cause can be identified in approximately 15%. Cases without an identifiable genetic etiology are considered sporadic. So far, the only approved therapy that modifies the function of a gene associated with ALS is tofersen (Qalsody, Biogen), an antisense oligonucleotide. Tofersen inhibits RNA transcription of the superoxide dismutase 1 (SOD1) gene to decrease production of the SOD1 protein.
This first gene therapy for ALS is a breakthrough, but it is indicated for only a small proportion of ALS patients. Even though SOD1 gene mutations represent the second most common genetic cause of ALS after the C90rf72 gene, the proportion of patients who are candidates for tofersen is low. Efficacy is expected only in about 1% of those with familial ALS and 1% of those with sporadic ALS, or about 2% of all patients with ALS.
The evidence of benefit from a treatment with a specific target has provided the basis for concluding that “we are onto something,” Dr. Fournier said. An expert in ALS, she sees reason for excitement about the prospects in treatment with the growing focus on the underlying pathways of disease rather than the downstream consequences.
“The hope is that new gene-targeted therapies will be developed in the future to treat the broader ALS population,” said Dr. Fournier, explaining that the move toward rationally targeted treatments, whether related to gene mutations or independent molecular pathways of ALS progression, has created excitement in the field.
Numerous Disease Processes Are Potentially Targetable
As treatments are developed to address nongenetic molecular processes that contribute to the risk or progression of ALS, such as neuroinflammation or abnormal protein misfolding and aggregation, individualized treatment is likely to become key. Just as not all genetic cases share mutations in the same gene, the key molecular drivers of disease are likely to differ between patients. If so, it is hoped that biomarkers reflective of this underlying biology can be identified to appropriately target treatments.
“The excitement behind the newer targets in clinical trials is based on both basic science and early clinical data that support treatment based on specific drivers of disease,” Dr. Fournier said.
In 2023 and just prior to the FDA approval of tofersen, a set of expert consensus guidelines were published calling for genetic testing to be offered to all patients with ALS. These recommendations suggested that SOD1, C9orf72, FUS, and TARDBP should be included routinely into the panel of genes evaluated, calling for additional genes to be added as they emerge as potential therapeutic targets.
Even before these guidelines were released, genetic testing was already being offered at many centers with expertise in ALS. The rationale was to differentiate ALS with a genetic etiology from that with a nongenetic etiology, as well as to counsel family members when genetic risk was identified, but genetic testing has now assumed new urgency. In addition to the potential for offering a specific treatment for SOD1-related ALS, patients with other genetic forms of disease might be candidates for genetically focused clinical trials.
Genetic testing should be performed as soon as a diagnosis of ALS is made, according to Dr. Fournier. Although not all patients have accepted genetic testing, particularly in the past when there was no immediate clinical gain from establishing the presence of a genetic mutation, she said there is no longer any controversy about clinical relevance.
Genetic Testing Is Key to Genetic Therapies
“We do not want to miss the opportunity to treat patients when we have the chance,” said Dr. Fournier, referring to both the likely advantage of an early start of the approved gene therapy as well as the opportunity to participate in a clinical trial with other gene therapies in development.
Prior to the approval of tofersen, riluzole and edaravone had been the only disease-modifying agents in widespread use, but these drugs are nonspecific. There are no established biomarkers for establishing which patients are most likely to benefit.
In the case of riluzole, a pivotal trial conducted 30 years ago showed a survival benefit relative to placebo at 12 months (74% vs. 58%; P = 0.014). In a retrospective study published in 2022 that evaluated survival in a database of 4778 ALS patients of whom 3446 received riluzole, early diagnosis of ALS and prompt treatment with riluzole was associated with longer survival than delayed treatment. The benefit of edaravone has been validated with clinical measures, such as the revised Amyotrophic Lateral Sclerosis Functional Scale (ALSFRS-R).
The retrospective study of riluzole provides the basis for predicting better benefits from disease-modifying therapies if started earlier in the course of ALS. The same premise will be explored with newer therapies that target ALS-associated genes.
Early Treatment Presumed More Effective
“We think that earlier treatment in the course of ALS is probably better for gene therapies as well,” Dr. Fournier said. She cautioned that follow-up is not yet long enough to confirm a survival benefit with tofersen, but she said it is reasonable to anticipate better and longer response when neurologic damage is limited. Citing the effect of gene therapy in spinal muscular atrophy (SMA), where progression is halted if gene therapy is initiated early in life, Dr. Fourier suggested that the emphasis on early treatment stems from the low likelihood for treatments to reverse functional impairments.
“It is conceivable that future treatments might be developed to reverse symptoms, but current drug development is largely aimed at slowing progression,” she explained. Under some circumstances, halting progression has the potential to allow some function to be regained, but as the etiologies of ALS and the pathways of progression are better understood, she believes that all targeted therapy will be started as early as possible to prevent rather than treat neurological damage.
Tofersen, the gene therapy for SOD1-ALS, has provided an opportunity to test the idea that it may be possible to prevent ALS. In a phase 3 trial called ATLAS, unaffected carriers of SOD1 variants that are associated with aggressive disease and high or complete penetrance are enrolled for a run-in phase (Part A) during which participants are followed for a rise in neurofilament light chain (NfL) levels. Based on a previous natural history study called the Pre-Symptomatic Familial ALS (Pre-fALS) study, NfL rises in the serum of unaffected SOD1 carriers prior to phenoconversion. A low NfL is an entry criterion for ATLAS.
ATLAS End Point Is Reduction in Phenoconversion to Clinically Manifest ALS
People in whom NfL rises above a predefined threshold during the run-in stage will be eligible for randomization (Part B) to receive either tofersen or placebo. Efficacy will be measured by comparing the rates of phenoconversion to clinically manifest ALS between those who receive placebo and those who receive tofersen.
Two other groups enrolled in ATLAS will be followed on open-label tofersen. One comprises people who phenoconvert during Part B and the other comprises those who develop ALS during the run-in and therefore are not enrolled in Part B. These patients, forming Parts C and D of the study, provide another set of data to evaluate whether earlier rather than later introduction of therapy provides better outcomes.
“There is a lot of interest and optimism about the trial,” said Dr. Fournier, who praised the trial design and thinks the hypothesis being explored “makes sense.”
Michael Benatar, MD, PhD, professor of neurology and public health, University of Miami School of Medicine, Miami, Florida, is the principal investigator of ATLAS and also leads the Pre-Symptomatic Familial ALS study together with a colleague, Joanne Wuu, Associate Director of Research at the University of Miami ALS Center. The hope from these initiatives, according to Dr. Fournier, is that ATLAS will offer broader learnings beyond just the SOD1 population, providing critical information about the optimal timing of treatment initiation.
The benefit from targeting genes considered causative for ALS is not yet a sure thing. A clinical trial targeting C9orf72, for example, failed to support an approvable therapy. There is a trial of a gene therapy for the FUS variant that is ongoing. Yet, the introduction of a gene therapy for SOD1 variant ALS has already established that highly targeted therapies can be effective, an important step forward after so many failed treatment trials with nonspecific drugs.
“We are seeing more and more therapies being developed to address specific ALS biology,” said Dr. Fournier, who predicts a pivot toward conceptualizing ALS as an array of pathologies rather than one disorder driven by a single mechanism. More effort is being directed to recognizing phenotypes as well as genotypes. Hopefully, more biomarkers that distinguish between ALS variants will emerge and help in individualizing treatment.
“We are not there yet, but I think many of us in the field see this as a way forward,” she said.
Multidisciplinary Care, Symptomatic Management, and Palliative Care Are Still Essential for ALS
Disease-modifying therapies are the ultimate goal in ALS, but Dr. Fournier said that the other side of the equation is multidisciplinary and palliative care. To the extent that almost all ALS therapies only modify the course of disease modestly, palliative care remains the cornerstone of day-to-day care.
“Multidisciplinary and palliative care are not necessarily novel, but they are still critically important. There are clear data to show that multidisciplinary care improves functional status and quality of life, and that this is meaningful to patients,” Dr. Fournier said.
There have been numerous improvements in the areas of multidisciplinary and palliative care, some of which can be credited to advancing technology. In centers of excellence, the multidisciplinary approach has been focused on helping patients sustain a sense of independence and self-worth.
Now robotics, devices, and software are being increasingly employed to extend patient capabilities even in relatively advanced stages of disease, according to Dr. Fournier. As one example, she cited current work in brain-computer interfaces to record electrical activity in the central nervous system to allow patients to communicate even when speech is impaired.
A focus on patient-centered clinical care is appropriate because it is the best current opportunity to improve the lives of patients with ALS. Clinically, this work is very rewarding, according to Dr. Fournier, who described ALS patients overall as generally ”very invested in advocacy and research initiatives and motivated to help others,” Dr. Fournier said.
“The diagnosis can be tough, but there is satisfaction in helping these patients navigate toward an acceptable and meaningful quality of life. They typically give a lot back,” she added.
Overall, there is a sense of progress in ALS, even though it remains a uniformly fatal disease. Dr. Fournier expressed hope that clinical research is reaching a tipping point and an emphasis on targeted treatments after a long list of failed trials over the past 30 years. However, with only one approved therapy modifying an ALS-associated gene, this approach is still in its early stages.
Dr. Fournier has financial relationships with Amylyx, Biogen, Corcept, Denali, Mitsubishi QurAlis, and Tanabe.
Suggested Reading
Benatar M et al. Design of a Randomized, Placebo-Controlled, Phase 3 Trial of Tofersen Initiated in Clinically Presymptomatic SOD1 Variant Carriers: the ATLAS Study. Neurotherapeutics. 2022 Jul;19(4):1248-1258. doi: 10.1007/s13311-022-01237-4.
Geronimo A et al. Ten Years of Riluzole Use in a Tertiary ALS Clinic. Muscle Nerve. 2022 Jun;65(6):659-666. doi: 10.1002/mus.27541.
Roggenbuck J et al. Evidence-Based Consensus Guidelines for ALS Genetic Testing and Counseling. Ann Clin Transl Neurol. 2023 Nov;10(11):2074-2091. doi: 10.1002/acn3.51895.
The first therapy targeted at modifying a mutant gene associated with amyotrophic lateral sclerosis (ALS), approved in early 2023, has offered reassurance that the biology of ALS, when known, is targetable. Historically, the disease has been considered a clinical diagnosis, but the
Despite a narrow indication, the only therapy targeted at an ALS-associated gene so far, SOD1 ALS, supports the premise that the biology of ALS can be modified, according to Christina N. Fournier, MD, an associate professor in the Department of Neurology, Emory University, Atlanta, Georgia.
Rather than a single pathological entity, ALS is best understood as the end result of many different pathological processes. Each might require its own targeted therapy in order to interrupt the upstream biological pathways that drive disease.
About 15% of ALS Has An Identifiable Genetic Cause
A family history of ALS is present in about 10% of cases. A genetic cause can be identified in approximately 15%. Cases without an identifiable genetic etiology are considered sporadic. So far, the only approved therapy that modifies the function of a gene associated with ALS is tofersen (Qalsody, Biogen), an antisense oligonucleotide. Tofersen inhibits RNA transcription of the superoxide dismutase 1 (SOD1) gene to decrease production of the SOD1 protein.
This first gene therapy for ALS is a breakthrough, but it is indicated for only a small proportion of ALS patients. Even though SOD1 gene mutations represent the second most common genetic cause of ALS after the C90rf72 gene, the proportion of patients who are candidates for tofersen is low. Efficacy is expected only in about 1% of those with familial ALS and 1% of those with sporadic ALS, or about 2% of all patients with ALS.
The evidence of benefit from a treatment with a specific target has provided the basis for concluding that “we are onto something,” Dr. Fournier said. An expert in ALS, she sees reason for excitement about the prospects in treatment with the growing focus on the underlying pathways of disease rather than the downstream consequences.
“The hope is that new gene-targeted therapies will be developed in the future to treat the broader ALS population,” said Dr. Fournier, explaining that the move toward rationally targeted treatments, whether related to gene mutations or independent molecular pathways of ALS progression, has created excitement in the field.
Numerous Disease Processes Are Potentially Targetable
As treatments are developed to address nongenetic molecular processes that contribute to the risk or progression of ALS, such as neuroinflammation or abnormal protein misfolding and aggregation, individualized treatment is likely to become key. Just as not all genetic cases share mutations in the same gene, the key molecular drivers of disease are likely to differ between patients. If so, it is hoped that biomarkers reflective of this underlying biology can be identified to appropriately target treatments.
“The excitement behind the newer targets in clinical trials is based on both basic science and early clinical data that support treatment based on specific drivers of disease,” Dr. Fournier said.
In 2023 and just prior to the FDA approval of tofersen, a set of expert consensus guidelines were published calling for genetic testing to be offered to all patients with ALS. These recommendations suggested that SOD1, C9orf72, FUS, and TARDBP should be included routinely into the panel of genes evaluated, calling for additional genes to be added as they emerge as potential therapeutic targets.
Even before these guidelines were released, genetic testing was already being offered at many centers with expertise in ALS. The rationale was to differentiate ALS with a genetic etiology from that with a nongenetic etiology, as well as to counsel family members when genetic risk was identified, but genetic testing has now assumed new urgency. In addition to the potential for offering a specific treatment for SOD1-related ALS, patients with other genetic forms of disease might be candidates for genetically focused clinical trials.
Genetic testing should be performed as soon as a diagnosis of ALS is made, according to Dr. Fournier. Although not all patients have accepted genetic testing, particularly in the past when there was no immediate clinical gain from establishing the presence of a genetic mutation, she said there is no longer any controversy about clinical relevance.
Genetic Testing Is Key to Genetic Therapies
“We do not want to miss the opportunity to treat patients when we have the chance,” said Dr. Fournier, referring to both the likely advantage of an early start of the approved gene therapy as well as the opportunity to participate in a clinical trial with other gene therapies in development.
Prior to the approval of tofersen, riluzole and edaravone had been the only disease-modifying agents in widespread use, but these drugs are nonspecific. There are no established biomarkers for establishing which patients are most likely to benefit.
In the case of riluzole, a pivotal trial conducted 30 years ago showed a survival benefit relative to placebo at 12 months (74% vs. 58%; P = 0.014). In a retrospective study published in 2022 that evaluated survival in a database of 4778 ALS patients of whom 3446 received riluzole, early diagnosis of ALS and prompt treatment with riluzole was associated with longer survival than delayed treatment. The benefit of edaravone has been validated with clinical measures, such as the revised Amyotrophic Lateral Sclerosis Functional Scale (ALSFRS-R).
The retrospective study of riluzole provides the basis for predicting better benefits from disease-modifying therapies if started earlier in the course of ALS. The same premise will be explored with newer therapies that target ALS-associated genes.
Early Treatment Presumed More Effective
“We think that earlier treatment in the course of ALS is probably better for gene therapies as well,” Dr. Fournier said. She cautioned that follow-up is not yet long enough to confirm a survival benefit with tofersen, but she said it is reasonable to anticipate better and longer response when neurologic damage is limited. Citing the effect of gene therapy in spinal muscular atrophy (SMA), where progression is halted if gene therapy is initiated early in life, Dr. Fourier suggested that the emphasis on early treatment stems from the low likelihood for treatments to reverse functional impairments.
“It is conceivable that future treatments might be developed to reverse symptoms, but current drug development is largely aimed at slowing progression,” she explained. Under some circumstances, halting progression has the potential to allow some function to be regained, but as the etiologies of ALS and the pathways of progression are better understood, she believes that all targeted therapy will be started as early as possible to prevent rather than treat neurological damage.
Tofersen, the gene therapy for SOD1-ALS, has provided an opportunity to test the idea that it may be possible to prevent ALS. In a phase 3 trial called ATLAS, unaffected carriers of SOD1 variants that are associated with aggressive disease and high or complete penetrance are enrolled for a run-in phase (Part A) during which participants are followed for a rise in neurofilament light chain (NfL) levels. Based on a previous natural history study called the Pre-Symptomatic Familial ALS (Pre-fALS) study, NfL rises in the serum of unaffected SOD1 carriers prior to phenoconversion. A low NfL is an entry criterion for ATLAS.
ATLAS End Point Is Reduction in Phenoconversion to Clinically Manifest ALS
People in whom NfL rises above a predefined threshold during the run-in stage will be eligible for randomization (Part B) to receive either tofersen or placebo. Efficacy will be measured by comparing the rates of phenoconversion to clinically manifest ALS between those who receive placebo and those who receive tofersen.
Two other groups enrolled in ATLAS will be followed on open-label tofersen. One comprises people who phenoconvert during Part B and the other comprises those who develop ALS during the run-in and therefore are not enrolled in Part B. These patients, forming Parts C and D of the study, provide another set of data to evaluate whether earlier rather than later introduction of therapy provides better outcomes.
“There is a lot of interest and optimism about the trial,” said Dr. Fournier, who praised the trial design and thinks the hypothesis being explored “makes sense.”
Michael Benatar, MD, PhD, professor of neurology and public health, University of Miami School of Medicine, Miami, Florida, is the principal investigator of ATLAS and also leads the Pre-Symptomatic Familial ALS study together with a colleague, Joanne Wuu, Associate Director of Research at the University of Miami ALS Center. The hope from these initiatives, according to Dr. Fournier, is that ATLAS will offer broader learnings beyond just the SOD1 population, providing critical information about the optimal timing of treatment initiation.
The benefit from targeting genes considered causative for ALS is not yet a sure thing. A clinical trial targeting C9orf72, for example, failed to support an approvable therapy. There is a trial of a gene therapy for the FUS variant that is ongoing. Yet, the introduction of a gene therapy for SOD1 variant ALS has already established that highly targeted therapies can be effective, an important step forward after so many failed treatment trials with nonspecific drugs.
“We are seeing more and more therapies being developed to address specific ALS biology,” said Dr. Fournier, who predicts a pivot toward conceptualizing ALS as an array of pathologies rather than one disorder driven by a single mechanism. More effort is being directed to recognizing phenotypes as well as genotypes. Hopefully, more biomarkers that distinguish between ALS variants will emerge and help in individualizing treatment.
“We are not there yet, but I think many of us in the field see this as a way forward,” she said.
Multidisciplinary Care, Symptomatic Management, and Palliative Care Are Still Essential for ALS
Disease-modifying therapies are the ultimate goal in ALS, but Dr. Fournier said that the other side of the equation is multidisciplinary and palliative care. To the extent that almost all ALS therapies only modify the course of disease modestly, palliative care remains the cornerstone of day-to-day care.
“Multidisciplinary and palliative care are not necessarily novel, but they are still critically important. There are clear data to show that multidisciplinary care improves functional status and quality of life, and that this is meaningful to patients,” Dr. Fournier said.
There have been numerous improvements in the areas of multidisciplinary and palliative care, some of which can be credited to advancing technology. In centers of excellence, the multidisciplinary approach has been focused on helping patients sustain a sense of independence and self-worth.
Now robotics, devices, and software are being increasingly employed to extend patient capabilities even in relatively advanced stages of disease, according to Dr. Fournier. As one example, she cited current work in brain-computer interfaces to record electrical activity in the central nervous system to allow patients to communicate even when speech is impaired.
A focus on patient-centered clinical care is appropriate because it is the best current opportunity to improve the lives of patients with ALS. Clinically, this work is very rewarding, according to Dr. Fournier, who described ALS patients overall as generally ”very invested in advocacy and research initiatives and motivated to help others,” Dr. Fournier said.
“The diagnosis can be tough, but there is satisfaction in helping these patients navigate toward an acceptable and meaningful quality of life. They typically give a lot back,” she added.
Overall, there is a sense of progress in ALS, even though it remains a uniformly fatal disease. Dr. Fournier expressed hope that clinical research is reaching a tipping point and an emphasis on targeted treatments after a long list of failed trials over the past 30 years. However, with only one approved therapy modifying an ALS-associated gene, this approach is still in its early stages.
Dr. Fournier has financial relationships with Amylyx, Biogen, Corcept, Denali, Mitsubishi QurAlis, and Tanabe.
Suggested Reading
Benatar M et al. Design of a Randomized, Placebo-Controlled, Phase 3 Trial of Tofersen Initiated in Clinically Presymptomatic SOD1 Variant Carriers: the ATLAS Study. Neurotherapeutics. 2022 Jul;19(4):1248-1258. doi: 10.1007/s13311-022-01237-4.
Geronimo A et al. Ten Years of Riluzole Use in a Tertiary ALS Clinic. Muscle Nerve. 2022 Jun;65(6):659-666. doi: 10.1002/mus.27541.
Roggenbuck J et al. Evidence-Based Consensus Guidelines for ALS Genetic Testing and Counseling. Ann Clin Transl Neurol. 2023 Nov;10(11):2074-2091. doi: 10.1002/acn3.51895.
The first therapy targeted at modifying a mutant gene associated with amyotrophic lateral sclerosis (ALS), approved in early 2023, has offered reassurance that the biology of ALS, when known, is targetable. Historically, the disease has been considered a clinical diagnosis, but the
Despite a narrow indication, the only therapy targeted at an ALS-associated gene so far, SOD1 ALS, supports the premise that the biology of ALS can be modified, according to Christina N. Fournier, MD, an associate professor in the Department of Neurology, Emory University, Atlanta, Georgia.
Rather than a single pathological entity, ALS is best understood as the end result of many different pathological processes. Each might require its own targeted therapy in order to interrupt the upstream biological pathways that drive disease.
About 15% of ALS Has An Identifiable Genetic Cause
A family history of ALS is present in about 10% of cases. A genetic cause can be identified in approximately 15%. Cases without an identifiable genetic etiology are considered sporadic. So far, the only approved therapy that modifies the function of a gene associated with ALS is tofersen (Qalsody, Biogen), an antisense oligonucleotide. Tofersen inhibits RNA transcription of the superoxide dismutase 1 (SOD1) gene to decrease production of the SOD1 protein.
This first gene therapy for ALS is a breakthrough, but it is indicated for only a small proportion of ALS patients. Even though SOD1 gene mutations represent the second most common genetic cause of ALS after the C90rf72 gene, the proportion of patients who are candidates for tofersen is low. Efficacy is expected only in about 1% of those with familial ALS and 1% of those with sporadic ALS, or about 2% of all patients with ALS.
The evidence of benefit from a treatment with a specific target has provided the basis for concluding that “we are onto something,” Dr. Fournier said. An expert in ALS, she sees reason for excitement about the prospects in treatment with the growing focus on the underlying pathways of disease rather than the downstream consequences.
“The hope is that new gene-targeted therapies will be developed in the future to treat the broader ALS population,” said Dr. Fournier, explaining that the move toward rationally targeted treatments, whether related to gene mutations or independent molecular pathways of ALS progression, has created excitement in the field.
Numerous Disease Processes Are Potentially Targetable
As treatments are developed to address nongenetic molecular processes that contribute to the risk or progression of ALS, such as neuroinflammation or abnormal protein misfolding and aggregation, individualized treatment is likely to become key. Just as not all genetic cases share mutations in the same gene, the key molecular drivers of disease are likely to differ between patients. If so, it is hoped that biomarkers reflective of this underlying biology can be identified to appropriately target treatments.
“The excitement behind the newer targets in clinical trials is based on both basic science and early clinical data that support treatment based on specific drivers of disease,” Dr. Fournier said.
In 2023 and just prior to the FDA approval of tofersen, a set of expert consensus guidelines were published calling for genetic testing to be offered to all patients with ALS. These recommendations suggested that SOD1, C9orf72, FUS, and TARDBP should be included routinely into the panel of genes evaluated, calling for additional genes to be added as they emerge as potential therapeutic targets.
Even before these guidelines were released, genetic testing was already being offered at many centers with expertise in ALS. The rationale was to differentiate ALS with a genetic etiology from that with a nongenetic etiology, as well as to counsel family members when genetic risk was identified, but genetic testing has now assumed new urgency. In addition to the potential for offering a specific treatment for SOD1-related ALS, patients with other genetic forms of disease might be candidates for genetically focused clinical trials.
Genetic testing should be performed as soon as a diagnosis of ALS is made, according to Dr. Fournier. Although not all patients have accepted genetic testing, particularly in the past when there was no immediate clinical gain from establishing the presence of a genetic mutation, she said there is no longer any controversy about clinical relevance.
Genetic Testing Is Key to Genetic Therapies
“We do not want to miss the opportunity to treat patients when we have the chance,” said Dr. Fournier, referring to both the likely advantage of an early start of the approved gene therapy as well as the opportunity to participate in a clinical trial with other gene therapies in development.
Prior to the approval of tofersen, riluzole and edaravone had been the only disease-modifying agents in widespread use, but these drugs are nonspecific. There are no established biomarkers for establishing which patients are most likely to benefit.
In the case of riluzole, a pivotal trial conducted 30 years ago showed a survival benefit relative to placebo at 12 months (74% vs. 58%; P = 0.014). In a retrospective study published in 2022 that evaluated survival in a database of 4778 ALS patients of whom 3446 received riluzole, early diagnosis of ALS and prompt treatment with riluzole was associated with longer survival than delayed treatment. The benefit of edaravone has been validated with clinical measures, such as the revised Amyotrophic Lateral Sclerosis Functional Scale (ALSFRS-R).
The retrospective study of riluzole provides the basis for predicting better benefits from disease-modifying therapies if started earlier in the course of ALS. The same premise will be explored with newer therapies that target ALS-associated genes.
Early Treatment Presumed More Effective
“We think that earlier treatment in the course of ALS is probably better for gene therapies as well,” Dr. Fournier said. She cautioned that follow-up is not yet long enough to confirm a survival benefit with tofersen, but she said it is reasonable to anticipate better and longer response when neurologic damage is limited. Citing the effect of gene therapy in spinal muscular atrophy (SMA), where progression is halted if gene therapy is initiated early in life, Dr. Fourier suggested that the emphasis on early treatment stems from the low likelihood for treatments to reverse functional impairments.
“It is conceivable that future treatments might be developed to reverse symptoms, but current drug development is largely aimed at slowing progression,” she explained. Under some circumstances, halting progression has the potential to allow some function to be regained, but as the etiologies of ALS and the pathways of progression are better understood, she believes that all targeted therapy will be started as early as possible to prevent rather than treat neurological damage.
Tofersen, the gene therapy for SOD1-ALS, has provided an opportunity to test the idea that it may be possible to prevent ALS. In a phase 3 trial called ATLAS, unaffected carriers of SOD1 variants that are associated with aggressive disease and high or complete penetrance are enrolled for a run-in phase (Part A) during which participants are followed for a rise in neurofilament light chain (NfL) levels. Based on a previous natural history study called the Pre-Symptomatic Familial ALS (Pre-fALS) study, NfL rises in the serum of unaffected SOD1 carriers prior to phenoconversion. A low NfL is an entry criterion for ATLAS.
ATLAS End Point Is Reduction in Phenoconversion to Clinically Manifest ALS
People in whom NfL rises above a predefined threshold during the run-in stage will be eligible for randomization (Part B) to receive either tofersen or placebo. Efficacy will be measured by comparing the rates of phenoconversion to clinically manifest ALS between those who receive placebo and those who receive tofersen.
Two other groups enrolled in ATLAS will be followed on open-label tofersen. One comprises people who phenoconvert during Part B and the other comprises those who develop ALS during the run-in and therefore are not enrolled in Part B. These patients, forming Parts C and D of the study, provide another set of data to evaluate whether earlier rather than later introduction of therapy provides better outcomes.
“There is a lot of interest and optimism about the trial,” said Dr. Fournier, who praised the trial design and thinks the hypothesis being explored “makes sense.”
Michael Benatar, MD, PhD, professor of neurology and public health, University of Miami School of Medicine, Miami, Florida, is the principal investigator of ATLAS and also leads the Pre-Symptomatic Familial ALS study together with a colleague, Joanne Wuu, Associate Director of Research at the University of Miami ALS Center. The hope from these initiatives, according to Dr. Fournier, is that ATLAS will offer broader learnings beyond just the SOD1 population, providing critical information about the optimal timing of treatment initiation.
The benefit from targeting genes considered causative for ALS is not yet a sure thing. A clinical trial targeting C9orf72, for example, failed to support an approvable therapy. There is a trial of a gene therapy for the FUS variant that is ongoing. Yet, the introduction of a gene therapy for SOD1 variant ALS has already established that highly targeted therapies can be effective, an important step forward after so many failed treatment trials with nonspecific drugs.
“We are seeing more and more therapies being developed to address specific ALS biology,” said Dr. Fournier, who predicts a pivot toward conceptualizing ALS as an array of pathologies rather than one disorder driven by a single mechanism. More effort is being directed to recognizing phenotypes as well as genotypes. Hopefully, more biomarkers that distinguish between ALS variants will emerge and help in individualizing treatment.
“We are not there yet, but I think many of us in the field see this as a way forward,” she said.
Multidisciplinary Care, Symptomatic Management, and Palliative Care Are Still Essential for ALS
Disease-modifying therapies are the ultimate goal in ALS, but Dr. Fournier said that the other side of the equation is multidisciplinary and palliative care. To the extent that almost all ALS therapies only modify the course of disease modestly, palliative care remains the cornerstone of day-to-day care.
“Multidisciplinary and palliative care are not necessarily novel, but they are still critically important. There are clear data to show that multidisciplinary care improves functional status and quality of life, and that this is meaningful to patients,” Dr. Fournier said.
There have been numerous improvements in the areas of multidisciplinary and palliative care, some of which can be credited to advancing technology. In centers of excellence, the multidisciplinary approach has been focused on helping patients sustain a sense of independence and self-worth.
Now robotics, devices, and software are being increasingly employed to extend patient capabilities even in relatively advanced stages of disease, according to Dr. Fournier. As one example, she cited current work in brain-computer interfaces to record electrical activity in the central nervous system to allow patients to communicate even when speech is impaired.
A focus on patient-centered clinical care is appropriate because it is the best current opportunity to improve the lives of patients with ALS. Clinically, this work is very rewarding, according to Dr. Fournier, who described ALS patients overall as generally ”very invested in advocacy and research initiatives and motivated to help others,” Dr. Fournier said.
“The diagnosis can be tough, but there is satisfaction in helping these patients navigate toward an acceptable and meaningful quality of life. They typically give a lot back,” she added.
Overall, there is a sense of progress in ALS, even though it remains a uniformly fatal disease. Dr. Fournier expressed hope that clinical research is reaching a tipping point and an emphasis on targeted treatments after a long list of failed trials over the past 30 years. However, with only one approved therapy modifying an ALS-associated gene, this approach is still in its early stages.
Dr. Fournier has financial relationships with Amylyx, Biogen, Corcept, Denali, Mitsubishi QurAlis, and Tanabe.
Suggested Reading
Benatar M et al. Design of a Randomized, Placebo-Controlled, Phase 3 Trial of Tofersen Initiated in Clinically Presymptomatic SOD1 Variant Carriers: the ATLAS Study. Neurotherapeutics. 2022 Jul;19(4):1248-1258. doi: 10.1007/s13311-022-01237-4.
Geronimo A et al. Ten Years of Riluzole Use in a Tertiary ALS Clinic. Muscle Nerve. 2022 Jun;65(6):659-666. doi: 10.1002/mus.27541.
Roggenbuck J et al. Evidence-Based Consensus Guidelines for ALS Genetic Testing and Counseling. Ann Clin Transl Neurol. 2023 Nov;10(11):2074-2091. doi: 10.1002/acn3.51895.