User login
Mediterranean diet improves cognition in MS
due to a potential neuroprotective mechanism, according to findings of a study that was released early, ahead of presentation at the annual meting of the American Academy of Neurology.
“We were most surprised by the magnitude of the results,” said Ilana Katz Sand, MD, associate professor of neurology at the Corinne Goldsmith Dickinson Center for MS at Mount Sinai in New York. “We hypothesized a significant association between Mediterranean diet and cognition in MS, but we did not anticipate the 20% absolute difference, particularly because we rigorously controlled the demographic and health-related factors, like socioeconomic status, body mass index, and exercise habits.”
The Mediterranean diet consists of predominately vegetables, fruits, legumes, fish, and healthy fats while minimizing the consumption of dairy products, meats, and saturated acids. Previous literature has drawn an association between diet and MS symptomology, notably with regard to the Mediterranean diet. These studies indicated a connection between thalamic volume in patients with early MS as well as objectively captured MS-related disability. In this study, researchers have continued their investigation by exploring how the Mediterranean diet affects cognition.
In this cross-sectional observational study, investigators evaluated 563 people with MS ranging in age from 18 to 65 years (n = 563; 71% women; aged 44.2 ± 11.3 years). To accomplish this task, researchers conducted a retrospective chart review capturing data from patients with MS who had undergone neurobehavioral screenings. Qualifying subjects completed the Mediterranean Diet Adherence Screener (MEDAS) to determine the extent to which they adhered to the Mediterranean diet. A 14-item questionnaire, MEDAS assess a person’s usual intake of healthful foods such as vegetables and olive oil, as well as minimization of unhealthy foods such as butter and red meat. They also completed an analogue of the CICMAS cognitive battery comprised of a composite of Symbol Digit Modalities Test, Hopkins Verbal Learning Test, Revised, and CANTAB Paired Associate Learning.
Researchers evaluated patient-reported outcomes adjusted based on demographics (i.e., age, sex, race, ethnicity, and socioeconomic status) and health-related factors. These elements included body mass index, exercise, sleep disturbance, hypertension, diabetes, hyperlipidemia, and smoking.
The study excluded patients who had another primary neurological condition in addition to MS (n = 24), serious psychiatric illness such as schizophrenia (n = 5) or clinical relapse within 6 weeks (n = 2), or missing data (n = 13).
Based on the diet scores, investigators stratified participants into four groups. Those with the scores ranging from 0 to 4 were classified into the lowest group, while scores of 9 or greater qualified participants for the high group.
Investigators observed a significant association between a higher Mediterranean diet score and condition in the population sampled. They found a mean z-score of –0.67 (0.95). In addition, a higher MEDAS proved an independent indicator of better cognition (B = 0.08 [95% confidence interval (CI), 0.05, 0.11], beta = 0.20, P < .001). In fact, a high MEDAS independently correlated to a 20% lower risk for cognitive impairment (odds ratio [OR] = .80 {95% CI, 0.73, 0.89}, P < .001). Ultimately, the study’s findings demonstrated MEDAS served as the strongest health-related indicator of z-score and cognitive impairment. Moreover, dietary modification based on effect suggested stronger associations between diet and cognition with progressive disease as opposed to relapsing disease, as noted by the relationship between the z-score and cognition.
“Further research is needed,” Dr. Katz Sand said. “But because the progressive phenotype reflects more prominent neurodegeneration, the greater observed effect size in those with progressive MS suggests a potential neuroprotective mechanism.”
This study was funded in part by an Irma T. Hirschl/Monique Weill-Caulier Research Award to Dr. Katz Sand. Dr. Katz Sand and coauthors also received funding from the National Multiple Sclerosis Society.
due to a potential neuroprotective mechanism, according to findings of a study that was released early, ahead of presentation at the annual meting of the American Academy of Neurology.
“We were most surprised by the magnitude of the results,” said Ilana Katz Sand, MD, associate professor of neurology at the Corinne Goldsmith Dickinson Center for MS at Mount Sinai in New York. “We hypothesized a significant association between Mediterranean diet and cognition in MS, but we did not anticipate the 20% absolute difference, particularly because we rigorously controlled the demographic and health-related factors, like socioeconomic status, body mass index, and exercise habits.”
The Mediterranean diet consists of predominately vegetables, fruits, legumes, fish, and healthy fats while minimizing the consumption of dairy products, meats, and saturated acids. Previous literature has drawn an association between diet and MS symptomology, notably with regard to the Mediterranean diet. These studies indicated a connection between thalamic volume in patients with early MS as well as objectively captured MS-related disability. In this study, researchers have continued their investigation by exploring how the Mediterranean diet affects cognition.
In this cross-sectional observational study, investigators evaluated 563 people with MS ranging in age from 18 to 65 years (n = 563; 71% women; aged 44.2 ± 11.3 years). To accomplish this task, researchers conducted a retrospective chart review capturing data from patients with MS who had undergone neurobehavioral screenings. Qualifying subjects completed the Mediterranean Diet Adherence Screener (MEDAS) to determine the extent to which they adhered to the Mediterranean diet. A 14-item questionnaire, MEDAS assess a person’s usual intake of healthful foods such as vegetables and olive oil, as well as minimization of unhealthy foods such as butter and red meat. They also completed an analogue of the CICMAS cognitive battery comprised of a composite of Symbol Digit Modalities Test, Hopkins Verbal Learning Test, Revised, and CANTAB Paired Associate Learning.
Researchers evaluated patient-reported outcomes adjusted based on demographics (i.e., age, sex, race, ethnicity, and socioeconomic status) and health-related factors. These elements included body mass index, exercise, sleep disturbance, hypertension, diabetes, hyperlipidemia, and smoking.
The study excluded patients who had another primary neurological condition in addition to MS (n = 24), serious psychiatric illness such as schizophrenia (n = 5) or clinical relapse within 6 weeks (n = 2), or missing data (n = 13).
Based on the diet scores, investigators stratified participants into four groups. Those with the scores ranging from 0 to 4 were classified into the lowest group, while scores of 9 or greater qualified participants for the high group.
Investigators observed a significant association between a higher Mediterranean diet score and condition in the population sampled. They found a mean z-score of –0.67 (0.95). In addition, a higher MEDAS proved an independent indicator of better cognition (B = 0.08 [95% confidence interval (CI), 0.05, 0.11], beta = 0.20, P < .001). In fact, a high MEDAS independently correlated to a 20% lower risk for cognitive impairment (odds ratio [OR] = .80 {95% CI, 0.73, 0.89}, P < .001). Ultimately, the study’s findings demonstrated MEDAS served as the strongest health-related indicator of z-score and cognitive impairment. Moreover, dietary modification based on effect suggested stronger associations between diet and cognition with progressive disease as opposed to relapsing disease, as noted by the relationship between the z-score and cognition.
“Further research is needed,” Dr. Katz Sand said. “But because the progressive phenotype reflects more prominent neurodegeneration, the greater observed effect size in those with progressive MS suggests a potential neuroprotective mechanism.”
This study was funded in part by an Irma T. Hirschl/Monique Weill-Caulier Research Award to Dr. Katz Sand. Dr. Katz Sand and coauthors also received funding from the National Multiple Sclerosis Society.
due to a potential neuroprotective mechanism, according to findings of a study that was released early, ahead of presentation at the annual meting of the American Academy of Neurology.
“We were most surprised by the magnitude of the results,” said Ilana Katz Sand, MD, associate professor of neurology at the Corinne Goldsmith Dickinson Center for MS at Mount Sinai in New York. “We hypothesized a significant association between Mediterranean diet and cognition in MS, but we did not anticipate the 20% absolute difference, particularly because we rigorously controlled the demographic and health-related factors, like socioeconomic status, body mass index, and exercise habits.”
The Mediterranean diet consists of predominately vegetables, fruits, legumes, fish, and healthy fats while minimizing the consumption of dairy products, meats, and saturated acids. Previous literature has drawn an association between diet and MS symptomology, notably with regard to the Mediterranean diet. These studies indicated a connection between thalamic volume in patients with early MS as well as objectively captured MS-related disability. In this study, researchers have continued their investigation by exploring how the Mediterranean diet affects cognition.
In this cross-sectional observational study, investigators evaluated 563 people with MS ranging in age from 18 to 65 years (n = 563; 71% women; aged 44.2 ± 11.3 years). To accomplish this task, researchers conducted a retrospective chart review capturing data from patients with MS who had undergone neurobehavioral screenings. Qualifying subjects completed the Mediterranean Diet Adherence Screener (MEDAS) to determine the extent to which they adhered to the Mediterranean diet. A 14-item questionnaire, MEDAS assess a person’s usual intake of healthful foods such as vegetables and olive oil, as well as minimization of unhealthy foods such as butter and red meat. They also completed an analogue of the CICMAS cognitive battery comprised of a composite of Symbol Digit Modalities Test, Hopkins Verbal Learning Test, Revised, and CANTAB Paired Associate Learning.
Researchers evaluated patient-reported outcomes adjusted based on demographics (i.e., age, sex, race, ethnicity, and socioeconomic status) and health-related factors. These elements included body mass index, exercise, sleep disturbance, hypertension, diabetes, hyperlipidemia, and smoking.
The study excluded patients who had another primary neurological condition in addition to MS (n = 24), serious psychiatric illness such as schizophrenia (n = 5) or clinical relapse within 6 weeks (n = 2), or missing data (n = 13).
Based on the diet scores, investigators stratified participants into four groups. Those with the scores ranging from 0 to 4 were classified into the lowest group, while scores of 9 or greater qualified participants for the high group.
Investigators observed a significant association between a higher Mediterranean diet score and condition in the population sampled. They found a mean z-score of –0.67 (0.95). In addition, a higher MEDAS proved an independent indicator of better cognition (B = 0.08 [95% confidence interval (CI), 0.05, 0.11], beta = 0.20, P < .001). In fact, a high MEDAS independently correlated to a 20% lower risk for cognitive impairment (odds ratio [OR] = .80 {95% CI, 0.73, 0.89}, P < .001). Ultimately, the study’s findings demonstrated MEDAS served as the strongest health-related indicator of z-score and cognitive impairment. Moreover, dietary modification based on effect suggested stronger associations between diet and cognition with progressive disease as opposed to relapsing disease, as noted by the relationship between the z-score and cognition.
“Further research is needed,” Dr. Katz Sand said. “But because the progressive phenotype reflects more prominent neurodegeneration, the greater observed effect size in those with progressive MS suggests a potential neuroprotective mechanism.”
This study was funded in part by an Irma T. Hirschl/Monique Weill-Caulier Research Award to Dr. Katz Sand. Dr. Katz Sand and coauthors also received funding from the National Multiple Sclerosis Society.
FROM AAN 2023
Obstructive sleep apnea linked to early cognitive decline
In a pilot study out of King’s College London, participants with severe OSA experienced worse executive functioning as well as social and emotional recognition versus healthy controls.
Major risk factors for OSA include obesity, high blood pressure, smoking, high cholesterol, and being middle-aged or older. Because some researchers have hypothesized that cognitive deficits could be driven by such comorbidities, the study investigators recruited middle-aged men with no medical comorbidities.
“Traditionally, we were more concerned with sleep apnea’s metabolic and cardiovascular comorbidities, and indeed, when cognitive deficits were demonstrated, most were attributed to them, and yet, our patients and their partners/families commonly tell us differently,” lead investigator Ivana Rosenzweig, MD, PhD, of King’s College London, who is also a consultant in sleep medicine and neuropsychiatry at Guy’s and St Thomas’ Hospital, London, said in an interview.
“Our findings provide a very important first step towards challenging the long-standing dogma that sleep apnea has little to do with the brain – apart from causing sleepiness – and that it is a predominantly nonneuro/psychiatric illness,” added Dr. Rosenzweig.
The findings were published online in Frontiers in Sleep.
Brain changes
The researchers wanted to understand how OSA may be linked to cognitive decline in the absence of cardiovascular and metabolic conditions.
To accomplish this, the investigators studied 27 men between the ages of 35 and 70 with a new diagnosis of mild to severe OSA without any comorbidities (16 with mild OSA and 11 with severe OSA). They also studied a control group of seven men matched for age, body mass index, and education level.
The team tested participants’ cognitive performance using the Cambridge Neuropsychological Test Automated Battery and found that the most significant deficits for the OSA group, compared with controls, were in areas of visual matching ability (P < .0001), short-term visual recognition memory, nonverbal patterns, executive functioning and attentional set-shifting (P < .001), psychomotor functioning, and social cognition and emotional recognition (P < .05).
On the latter two tests, impaired participants were less likely to accurately identify the emotion on computer-generated faces. Those with mild OSA performed better than those with severe OSA on these tasks, but rarely worse than controls.
Dr. Rosenzweig noted that the findings were one-of-a-kind because of the recruitment of patients with OSA who were otherwise healthy and nonobese, “something one rarely sees in the sleep clinic, where we commonly encounter patients with already developed comorbidities.
“In order to truly revolutionize the treatment for our patients, it is important to understand how much the accompanying comorbidities, such as systemic hypertension, obesity, diabetes, hyperlipidemia, and other various serious cardiovascular and metabolic diseases and how much the illness itself may shape the demonstrated cognitive deficits,” she said.
She also said that “it is widely agreed that medical problems in middle age may predispose to increased prevalence of dementia in later years.
Moreover, the very link between sleep apnea and Alzheimer’s, vascular and mixed dementia is increasingly demonstrated,” said Dr. Rosenzweig.
Although women typically have a lower prevalence of OSA than men, Dr. Rosenzweig said women were not included in the study “because we are too complex. As a lifelong feminist it pains me to say this, but to get any authoritative answer on our physiology, we need decent funding in place so that we can take into account all the intricacies of the changes of our sleep, physiology, and metabolism.
“While there is always lots of noise about how important it is to answer these questions, there are only very limited funds available for the sleep research,” she added.
Dr. Rosenzweig’s future research will focus on the potential link between OSA and neuroinflammation.
In a comment, Liza Ashbrook, MD, associate professor of neurology at the University of California, San Francisco, said the findings “add to the growing list of negative health consequences associated with sleep apnea.”
She said that, if the cognitive changes found in the study are, in fact, caused by OSA, it is unclear whether they are the beginning of long-term cognitive changes or a symptom of fragmented sleep that may be reversible.
Dr. Ashbrook said she would be interested in seeing research on understanding the effect of OSA treatment on the affected cognitive domains.
The study was funded by the Wellcome Trust. No relevant financial relationships were reported.
A version of this article originally appeared on Medscape.com.
In a pilot study out of King’s College London, participants with severe OSA experienced worse executive functioning as well as social and emotional recognition versus healthy controls.
Major risk factors for OSA include obesity, high blood pressure, smoking, high cholesterol, and being middle-aged or older. Because some researchers have hypothesized that cognitive deficits could be driven by such comorbidities, the study investigators recruited middle-aged men with no medical comorbidities.
“Traditionally, we were more concerned with sleep apnea’s metabolic and cardiovascular comorbidities, and indeed, when cognitive deficits were demonstrated, most were attributed to them, and yet, our patients and their partners/families commonly tell us differently,” lead investigator Ivana Rosenzweig, MD, PhD, of King’s College London, who is also a consultant in sleep medicine and neuropsychiatry at Guy’s and St Thomas’ Hospital, London, said in an interview.
“Our findings provide a very important first step towards challenging the long-standing dogma that sleep apnea has little to do with the brain – apart from causing sleepiness – and that it is a predominantly nonneuro/psychiatric illness,” added Dr. Rosenzweig.
The findings were published online in Frontiers in Sleep.
Brain changes
The researchers wanted to understand how OSA may be linked to cognitive decline in the absence of cardiovascular and metabolic conditions.
To accomplish this, the investigators studied 27 men between the ages of 35 and 70 with a new diagnosis of mild to severe OSA without any comorbidities (16 with mild OSA and 11 with severe OSA). They also studied a control group of seven men matched for age, body mass index, and education level.
The team tested participants’ cognitive performance using the Cambridge Neuropsychological Test Automated Battery and found that the most significant deficits for the OSA group, compared with controls, were in areas of visual matching ability (P < .0001), short-term visual recognition memory, nonverbal patterns, executive functioning and attentional set-shifting (P < .001), psychomotor functioning, and social cognition and emotional recognition (P < .05).
On the latter two tests, impaired participants were less likely to accurately identify the emotion on computer-generated faces. Those with mild OSA performed better than those with severe OSA on these tasks, but rarely worse than controls.
Dr. Rosenzweig noted that the findings were one-of-a-kind because of the recruitment of patients with OSA who were otherwise healthy and nonobese, “something one rarely sees in the sleep clinic, where we commonly encounter patients with already developed comorbidities.
“In order to truly revolutionize the treatment for our patients, it is important to understand how much the accompanying comorbidities, such as systemic hypertension, obesity, diabetes, hyperlipidemia, and other various serious cardiovascular and metabolic diseases and how much the illness itself may shape the demonstrated cognitive deficits,” she said.
She also said that “it is widely agreed that medical problems in middle age may predispose to increased prevalence of dementia in later years.
Moreover, the very link between sleep apnea and Alzheimer’s, vascular and mixed dementia is increasingly demonstrated,” said Dr. Rosenzweig.
Although women typically have a lower prevalence of OSA than men, Dr. Rosenzweig said women were not included in the study “because we are too complex. As a lifelong feminist it pains me to say this, but to get any authoritative answer on our physiology, we need decent funding in place so that we can take into account all the intricacies of the changes of our sleep, physiology, and metabolism.
“While there is always lots of noise about how important it is to answer these questions, there are only very limited funds available for the sleep research,” she added.
Dr. Rosenzweig’s future research will focus on the potential link between OSA and neuroinflammation.
In a comment, Liza Ashbrook, MD, associate professor of neurology at the University of California, San Francisco, said the findings “add to the growing list of negative health consequences associated with sleep apnea.”
She said that, if the cognitive changes found in the study are, in fact, caused by OSA, it is unclear whether they are the beginning of long-term cognitive changes or a symptom of fragmented sleep that may be reversible.
Dr. Ashbrook said she would be interested in seeing research on understanding the effect of OSA treatment on the affected cognitive domains.
The study was funded by the Wellcome Trust. No relevant financial relationships were reported.
A version of this article originally appeared on Medscape.com.
In a pilot study out of King’s College London, participants with severe OSA experienced worse executive functioning as well as social and emotional recognition versus healthy controls.
Major risk factors for OSA include obesity, high blood pressure, smoking, high cholesterol, and being middle-aged or older. Because some researchers have hypothesized that cognitive deficits could be driven by such comorbidities, the study investigators recruited middle-aged men with no medical comorbidities.
“Traditionally, we were more concerned with sleep apnea’s metabolic and cardiovascular comorbidities, and indeed, when cognitive deficits were demonstrated, most were attributed to them, and yet, our patients and their partners/families commonly tell us differently,” lead investigator Ivana Rosenzweig, MD, PhD, of King’s College London, who is also a consultant in sleep medicine and neuropsychiatry at Guy’s and St Thomas’ Hospital, London, said in an interview.
“Our findings provide a very important first step towards challenging the long-standing dogma that sleep apnea has little to do with the brain – apart from causing sleepiness – and that it is a predominantly nonneuro/psychiatric illness,” added Dr. Rosenzweig.
The findings were published online in Frontiers in Sleep.
Brain changes
The researchers wanted to understand how OSA may be linked to cognitive decline in the absence of cardiovascular and metabolic conditions.
To accomplish this, the investigators studied 27 men between the ages of 35 and 70 with a new diagnosis of mild to severe OSA without any comorbidities (16 with mild OSA and 11 with severe OSA). They also studied a control group of seven men matched for age, body mass index, and education level.
The team tested participants’ cognitive performance using the Cambridge Neuropsychological Test Automated Battery and found that the most significant deficits for the OSA group, compared with controls, were in areas of visual matching ability (P < .0001), short-term visual recognition memory, nonverbal patterns, executive functioning and attentional set-shifting (P < .001), psychomotor functioning, and social cognition and emotional recognition (P < .05).
On the latter two tests, impaired participants were less likely to accurately identify the emotion on computer-generated faces. Those with mild OSA performed better than those with severe OSA on these tasks, but rarely worse than controls.
Dr. Rosenzweig noted that the findings were one-of-a-kind because of the recruitment of patients with OSA who were otherwise healthy and nonobese, “something one rarely sees in the sleep clinic, where we commonly encounter patients with already developed comorbidities.
“In order to truly revolutionize the treatment for our patients, it is important to understand how much the accompanying comorbidities, such as systemic hypertension, obesity, diabetes, hyperlipidemia, and other various serious cardiovascular and metabolic diseases and how much the illness itself may shape the demonstrated cognitive deficits,” she said.
She also said that “it is widely agreed that medical problems in middle age may predispose to increased prevalence of dementia in later years.
Moreover, the very link between sleep apnea and Alzheimer’s, vascular and mixed dementia is increasingly demonstrated,” said Dr. Rosenzweig.
Although women typically have a lower prevalence of OSA than men, Dr. Rosenzweig said women were not included in the study “because we are too complex. As a lifelong feminist it pains me to say this, but to get any authoritative answer on our physiology, we need decent funding in place so that we can take into account all the intricacies of the changes of our sleep, physiology, and metabolism.
“While there is always lots of noise about how important it is to answer these questions, there are only very limited funds available for the sleep research,” she added.
Dr. Rosenzweig’s future research will focus on the potential link between OSA and neuroinflammation.
In a comment, Liza Ashbrook, MD, associate professor of neurology at the University of California, San Francisco, said the findings “add to the growing list of negative health consequences associated with sleep apnea.”
She said that, if the cognitive changes found in the study are, in fact, caused by OSA, it is unclear whether they are the beginning of long-term cognitive changes or a symptom of fragmented sleep that may be reversible.
Dr. Ashbrook said she would be interested in seeing research on understanding the effect of OSA treatment on the affected cognitive domains.
The study was funded by the Wellcome Trust. No relevant financial relationships were reported.
A version of this article originally appeared on Medscape.com.
FROM FRONTIERS IN SLEEP
Food insecurity linked to more rapid cognitive decline in seniors
Food insecurity is linked to a more rapid decline in executive function in older adults, a new study shows.
The findings were reported just weeks after a pandemic-era expansion in Supplemental Nutrition Assistance Program benefits ended, leading to less food assistance for about 5 million people over age 60 who participate in the program.
“Even though we found only a very small association between food insecurity and executive function, it’s still meaningful, because food insecurity is something we can prevent,” lead investigator Boeun Kim, PhD, MPH, RN, postdoctoral fellow at Johns Hopkins University School of Nursing, Baltimore, told this news organization.
The findings were published online in JAMA Network Open.
National data
The number of Americans over 60 with food insecurity has more than doubled since 2007, with an estimated 5.2 million older adults reporting food insecurity in 2020.
Prior studies have linked malnutrition and food insecurity to a decline in cognitive function. Participating in food assistance programs such as SNAP is associated with slower memory decline in older adults.
However, to date, there has been no longitudinal study that has used data from a nationally representative sample of older Americans, which, Dr. Kim said, could limit generalizability of the findings.
To address that issue, investigators analyzed data from 3,037 participants in the National Health and Aging Trends Study, which includes community dwellers age 65 and older who receive Medicare.
Participants reported food insecurity over 7 years, from 2012 to 2019. Data on immediate memory, delayed memory, and executive function were from 2013 to 2020.
Food insecurity was defined as going without groceries due to limited ability or social support; a lack of hot meals related to functional limitation or no help; going without eating because of the inability to feed oneself or no available support; skipping meals due to insufficient food or money; or skipping meals for 5 days or more.
Immediate and delayed recall were assessed using a 10-item word-list memory task, and executive function was measured using a clock drawing test. Each year’s cognitive functions were linked to the prior year’s food insecurity data.
Over 7 years, 417 people, or 12.1%, experienced food insecurity at least once.
Those with food insecurity were more likely to be older, female, part of racial and ethnic minority groups, living alone, obese, and have a lower income and educational attainment, depressive symptoms, social isolation and disability, compared with those without food insecurity.
After adjusting for age, sex, race/ethnicity, educational level, income, marital status, body mass index, functional disability, social isolation, and other potential confounders, researchers found that food insecurity was associated with a more rapid decline in executive function (mean difference in annual change in executive function score, −0.04; 95% confidence interval, −0.09 to −0.003).
Food insecurity was not associated with baseline cognitive function scores or changes in immediate or delayed recall.
“Clinicians should be aware of the experience of food insecurity and the higher risk of cognitive decline so maybe they could do universal screening and refer people with food insecurity to programs that can help them access nutritious meals,” Dr. Kim said.
A sign of other problems?
Thomas Vidic, MD, said food insecurity often goes hand-in-hand with lack of medication adherence, lack of regular medical care, and a host of other issues. Dr. Vidic is a neurologist at the Elkhart Clinic, Ind., and an adjunct clinical professor of neurology at Indiana University.
“When a person has food insecurity, they likely have other problems, and they’re going to degenerate faster,” said Dr. Vidic, who was not part of the study. “This is one important component, and it’s one more way of getting a handle on people who are failing.”
Dr. Vidic, who has dealt with the issue of food insecurity with his own patients, said he suspects the self-report nature of the study may hide the true scale of the problem.
“I suspect the numbers might actually be higher,” he said, adding that the study fills a gap in the literature with a large, nationally representative sample.
“We’re looking for issues to help with the elderly as far as what can we do to keep dementia from progressing,” he said. “There are some things that make sense, but we’ve never had this kind of data before.”
The study was funded by the National Institute on Aging. Dr. Kim and Dr. Vidic have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Food insecurity is linked to a more rapid decline in executive function in older adults, a new study shows.
The findings were reported just weeks after a pandemic-era expansion in Supplemental Nutrition Assistance Program benefits ended, leading to less food assistance for about 5 million people over age 60 who participate in the program.
“Even though we found only a very small association between food insecurity and executive function, it’s still meaningful, because food insecurity is something we can prevent,” lead investigator Boeun Kim, PhD, MPH, RN, postdoctoral fellow at Johns Hopkins University School of Nursing, Baltimore, told this news organization.
The findings were published online in JAMA Network Open.
National data
The number of Americans over 60 with food insecurity has more than doubled since 2007, with an estimated 5.2 million older adults reporting food insecurity in 2020.
Prior studies have linked malnutrition and food insecurity to a decline in cognitive function. Participating in food assistance programs such as SNAP is associated with slower memory decline in older adults.
However, to date, there has been no longitudinal study that has used data from a nationally representative sample of older Americans, which, Dr. Kim said, could limit generalizability of the findings.
To address that issue, investigators analyzed data from 3,037 participants in the National Health and Aging Trends Study, which includes community dwellers age 65 and older who receive Medicare.
Participants reported food insecurity over 7 years, from 2012 to 2019. Data on immediate memory, delayed memory, and executive function were from 2013 to 2020.
Food insecurity was defined as going without groceries due to limited ability or social support; a lack of hot meals related to functional limitation or no help; going without eating because of the inability to feed oneself or no available support; skipping meals due to insufficient food or money; or skipping meals for 5 days or more.
Immediate and delayed recall were assessed using a 10-item word-list memory task, and executive function was measured using a clock drawing test. Each year’s cognitive functions were linked to the prior year’s food insecurity data.
Over 7 years, 417 people, or 12.1%, experienced food insecurity at least once.
Those with food insecurity were more likely to be older, female, part of racial and ethnic minority groups, living alone, obese, and have a lower income and educational attainment, depressive symptoms, social isolation and disability, compared with those without food insecurity.
After adjusting for age, sex, race/ethnicity, educational level, income, marital status, body mass index, functional disability, social isolation, and other potential confounders, researchers found that food insecurity was associated with a more rapid decline in executive function (mean difference in annual change in executive function score, −0.04; 95% confidence interval, −0.09 to −0.003).
Food insecurity was not associated with baseline cognitive function scores or changes in immediate or delayed recall.
“Clinicians should be aware of the experience of food insecurity and the higher risk of cognitive decline so maybe they could do universal screening and refer people with food insecurity to programs that can help them access nutritious meals,” Dr. Kim said.
A sign of other problems?
Thomas Vidic, MD, said food insecurity often goes hand-in-hand with lack of medication adherence, lack of regular medical care, and a host of other issues. Dr. Vidic is a neurologist at the Elkhart Clinic, Ind., and an adjunct clinical professor of neurology at Indiana University.
“When a person has food insecurity, they likely have other problems, and they’re going to degenerate faster,” said Dr. Vidic, who was not part of the study. “This is one important component, and it’s one more way of getting a handle on people who are failing.”
Dr. Vidic, who has dealt with the issue of food insecurity with his own patients, said he suspects the self-report nature of the study may hide the true scale of the problem.
“I suspect the numbers might actually be higher,” he said, adding that the study fills a gap in the literature with a large, nationally representative sample.
“We’re looking for issues to help with the elderly as far as what can we do to keep dementia from progressing,” he said. “There are some things that make sense, but we’ve never had this kind of data before.”
The study was funded by the National Institute on Aging. Dr. Kim and Dr. Vidic have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Food insecurity is linked to a more rapid decline in executive function in older adults, a new study shows.
The findings were reported just weeks after a pandemic-era expansion in Supplemental Nutrition Assistance Program benefits ended, leading to less food assistance for about 5 million people over age 60 who participate in the program.
“Even though we found only a very small association between food insecurity and executive function, it’s still meaningful, because food insecurity is something we can prevent,” lead investigator Boeun Kim, PhD, MPH, RN, postdoctoral fellow at Johns Hopkins University School of Nursing, Baltimore, told this news organization.
The findings were published online in JAMA Network Open.
National data
The number of Americans over 60 with food insecurity has more than doubled since 2007, with an estimated 5.2 million older adults reporting food insecurity in 2020.
Prior studies have linked malnutrition and food insecurity to a decline in cognitive function. Participating in food assistance programs such as SNAP is associated with slower memory decline in older adults.
However, to date, there has been no longitudinal study that has used data from a nationally representative sample of older Americans, which, Dr. Kim said, could limit generalizability of the findings.
To address that issue, investigators analyzed data from 3,037 participants in the National Health and Aging Trends Study, which includes community dwellers age 65 and older who receive Medicare.
Participants reported food insecurity over 7 years, from 2012 to 2019. Data on immediate memory, delayed memory, and executive function were from 2013 to 2020.
Food insecurity was defined as going without groceries due to limited ability or social support; a lack of hot meals related to functional limitation or no help; going without eating because of the inability to feed oneself or no available support; skipping meals due to insufficient food or money; or skipping meals for 5 days or more.
Immediate and delayed recall were assessed using a 10-item word-list memory task, and executive function was measured using a clock drawing test. Each year’s cognitive functions were linked to the prior year’s food insecurity data.
Over 7 years, 417 people, or 12.1%, experienced food insecurity at least once.
Those with food insecurity were more likely to be older, female, part of racial and ethnic minority groups, living alone, obese, and have a lower income and educational attainment, depressive symptoms, social isolation and disability, compared with those without food insecurity.
After adjusting for age, sex, race/ethnicity, educational level, income, marital status, body mass index, functional disability, social isolation, and other potential confounders, researchers found that food insecurity was associated with a more rapid decline in executive function (mean difference in annual change in executive function score, −0.04; 95% confidence interval, −0.09 to −0.003).
Food insecurity was not associated with baseline cognitive function scores or changes in immediate or delayed recall.
“Clinicians should be aware of the experience of food insecurity and the higher risk of cognitive decline so maybe they could do universal screening and refer people with food insecurity to programs that can help them access nutritious meals,” Dr. Kim said.
A sign of other problems?
Thomas Vidic, MD, said food insecurity often goes hand-in-hand with lack of medication adherence, lack of regular medical care, and a host of other issues. Dr. Vidic is a neurologist at the Elkhart Clinic, Ind., and an adjunct clinical professor of neurology at Indiana University.
“When a person has food insecurity, they likely have other problems, and they’re going to degenerate faster,” said Dr. Vidic, who was not part of the study. “This is one important component, and it’s one more way of getting a handle on people who are failing.”
Dr. Vidic, who has dealt with the issue of food insecurity with his own patients, said he suspects the self-report nature of the study may hide the true scale of the problem.
“I suspect the numbers might actually be higher,” he said, adding that the study fills a gap in the literature with a large, nationally representative sample.
“We’re looking for issues to help with the elderly as far as what can we do to keep dementia from progressing,” he said. “There are some things that make sense, but we’ve never had this kind of data before.”
The study was funded by the National Institute on Aging. Dr. Kim and Dr. Vidic have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Urban green and blue spaces linked to less psychological distress
The findings of the study, which was released ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology, build on a growing understanding of the relationship between types and qualities of urban environments and dementia risk.
Adithya Vegaraju, a student at Washington State University, Spokane, led the study, which looked at data from the Washington State Behavioral Risk Factor Surveillance System to assess prevalence of serious psychological distress among 42,980 Washington state residents aged 65 and over.
The data, collected between 2011 and 2019, used a self-reported questionnaire to determine serious psychological distress, which is defined as a level of mental distress considered debilitating enough to warrant treatment.
Mr. Vegaraju and his coauthor Solmaz Amiri, DDes, also of Washington State University, used ZIP codes, along with U.S. census data, to approximate the urban adults’ proximity to green and blue spaces.
After controlling for potential confounders of age, sex, ethnicity, education, and marital status, the investigators found that people living within half a mile of green or blue spaces had a 17% lower risk of experiencing serious psychological distress, compared with people living farther from these spaces, the investigators said in a news release.
Implications for cognitive decline and dementia?
Psychological distress in adults has been linked in population-based longitudinal studies to later cognitive decline and dementia. One study in older adults found the risk of dementia to be more than 50% higher among adults aged 50-70 with persistent depression. Blue and green spaces have also been investigated in relation to neurodegenerative disease among older adults; a 2022 study looking at data from some 62 million Medicare beneficiaries found those living in areas with more vegetation saw lower risk of hospitalizations for Alzheimer’s disease and related dementias.
“Since we lack effective prevention methods or treatments for mild cognitive impairment and dementia, we need to get creative in how we look at these issues,” Dr. Amiri commented in a press statement about her and Mr. Vegaraju’s findings. “Our hope is that this study showing better mental health among people living close to parks and water will trigger other studies about how these benefits work and whether this proximity can help prevent or delay mild cognitive impairment and dementia.”
The investigators acknowledged that their findings were limited by reliance on a self-reported measure of psychological distress.
A bidirectional connection with depression and dementia
In a comment, Anjum Hajat, PhD, an epidemiologist at University of Washington School of Public Health in Seattle who has also studied the relationship between green space and dementia risk in older adults, noted some further apparent limitations of the new study, for which only an abstract was available at publication.
“It has been shown that people with depression are at higher risk for dementia, but the opposite is also true,” Dr. Hajat commented. “Those with dementia are more likely to develop depression. This bidirectionality makes this study abstract difficult to interpret since the study is based on cross-sectional data: Individuals are not followed over time to see which develops first, dementia or depression.”
Additionally, Dr. Hajat noted, the data used to determine proximity to green and blue spaces did not allow for the calculation of precise distances between subjects’ homes and these spaces.
Mr. Vegaraju and Dr. Amiri’s study had no outside support, and the investigators declared no conflicts of interest. Dr. Hajat declared no conflicts of interest.
The findings of the study, which was released ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology, build on a growing understanding of the relationship between types and qualities of urban environments and dementia risk.
Adithya Vegaraju, a student at Washington State University, Spokane, led the study, which looked at data from the Washington State Behavioral Risk Factor Surveillance System to assess prevalence of serious psychological distress among 42,980 Washington state residents aged 65 and over.
The data, collected between 2011 and 2019, used a self-reported questionnaire to determine serious psychological distress, which is defined as a level of mental distress considered debilitating enough to warrant treatment.
Mr. Vegaraju and his coauthor Solmaz Amiri, DDes, also of Washington State University, used ZIP codes, along with U.S. census data, to approximate the urban adults’ proximity to green and blue spaces.
After controlling for potential confounders of age, sex, ethnicity, education, and marital status, the investigators found that people living within half a mile of green or blue spaces had a 17% lower risk of experiencing serious psychological distress, compared with people living farther from these spaces, the investigators said in a news release.
Implications for cognitive decline and dementia?
Psychological distress in adults has been linked in population-based longitudinal studies to later cognitive decline and dementia. One study in older adults found the risk of dementia to be more than 50% higher among adults aged 50-70 with persistent depression. Blue and green spaces have also been investigated in relation to neurodegenerative disease among older adults; a 2022 study looking at data from some 62 million Medicare beneficiaries found those living in areas with more vegetation saw lower risk of hospitalizations for Alzheimer’s disease and related dementias.
“Since we lack effective prevention methods or treatments for mild cognitive impairment and dementia, we need to get creative in how we look at these issues,” Dr. Amiri commented in a press statement about her and Mr. Vegaraju’s findings. “Our hope is that this study showing better mental health among people living close to parks and water will trigger other studies about how these benefits work and whether this proximity can help prevent or delay mild cognitive impairment and dementia.”
The investigators acknowledged that their findings were limited by reliance on a self-reported measure of psychological distress.
A bidirectional connection with depression and dementia
In a comment, Anjum Hajat, PhD, an epidemiologist at University of Washington School of Public Health in Seattle who has also studied the relationship between green space and dementia risk in older adults, noted some further apparent limitations of the new study, for which only an abstract was available at publication.
“It has been shown that people with depression are at higher risk for dementia, but the opposite is also true,” Dr. Hajat commented. “Those with dementia are more likely to develop depression. This bidirectionality makes this study abstract difficult to interpret since the study is based on cross-sectional data: Individuals are not followed over time to see which develops first, dementia or depression.”
Additionally, Dr. Hajat noted, the data used to determine proximity to green and blue spaces did not allow for the calculation of precise distances between subjects’ homes and these spaces.
Mr. Vegaraju and Dr. Amiri’s study had no outside support, and the investigators declared no conflicts of interest. Dr. Hajat declared no conflicts of interest.
The findings of the study, which was released ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology, build on a growing understanding of the relationship between types and qualities of urban environments and dementia risk.
Adithya Vegaraju, a student at Washington State University, Spokane, led the study, which looked at data from the Washington State Behavioral Risk Factor Surveillance System to assess prevalence of serious psychological distress among 42,980 Washington state residents aged 65 and over.
The data, collected between 2011 and 2019, used a self-reported questionnaire to determine serious psychological distress, which is defined as a level of mental distress considered debilitating enough to warrant treatment.
Mr. Vegaraju and his coauthor Solmaz Amiri, DDes, also of Washington State University, used ZIP codes, along with U.S. census data, to approximate the urban adults’ proximity to green and blue spaces.
After controlling for potential confounders of age, sex, ethnicity, education, and marital status, the investigators found that people living within half a mile of green or blue spaces had a 17% lower risk of experiencing serious psychological distress, compared with people living farther from these spaces, the investigators said in a news release.
Implications for cognitive decline and dementia?
Psychological distress in adults has been linked in population-based longitudinal studies to later cognitive decline and dementia. One study in older adults found the risk of dementia to be more than 50% higher among adults aged 50-70 with persistent depression. Blue and green spaces have also been investigated in relation to neurodegenerative disease among older adults; a 2022 study looking at data from some 62 million Medicare beneficiaries found those living in areas with more vegetation saw lower risk of hospitalizations for Alzheimer’s disease and related dementias.
“Since we lack effective prevention methods or treatments for mild cognitive impairment and dementia, we need to get creative in how we look at these issues,” Dr. Amiri commented in a press statement about her and Mr. Vegaraju’s findings. “Our hope is that this study showing better mental health among people living close to parks and water will trigger other studies about how these benefits work and whether this proximity can help prevent or delay mild cognitive impairment and dementia.”
The investigators acknowledged that their findings were limited by reliance on a self-reported measure of psychological distress.
A bidirectional connection with depression and dementia
In a comment, Anjum Hajat, PhD, an epidemiologist at University of Washington School of Public Health in Seattle who has also studied the relationship between green space and dementia risk in older adults, noted some further apparent limitations of the new study, for which only an abstract was available at publication.
“It has been shown that people with depression are at higher risk for dementia, but the opposite is also true,” Dr. Hajat commented. “Those with dementia are more likely to develop depression. This bidirectionality makes this study abstract difficult to interpret since the study is based on cross-sectional data: Individuals are not followed over time to see which develops first, dementia or depression.”
Additionally, Dr. Hajat noted, the data used to determine proximity to green and blue spaces did not allow for the calculation of precise distances between subjects’ homes and these spaces.
Mr. Vegaraju and Dr. Amiri’s study had no outside support, and the investigators declared no conflicts of interest. Dr. Hajat declared no conflicts of interest.
FROM AAN 2023
Phototherapy a safe, effective, inexpensive new option for dementia?
It may be “one of the most promising interventions for improving core symptoms” of the disease.
A new meta-analysis shows that patients with dementia who received phototherapy experienced significant cognitive improvement, compared with those who received usual treatment. However, there were no differences between study groups in terms of improved depression, agitation, or sleep problems.
“Our meta-analysis indicates that phototherapy improved cognitive function in patients with dementia. ... This suggests that phototherapy may be one of the most promising non-pharmacological interventions for improving core symptoms of dementia,” wrote the investigators, led by Xinlian Lu, Peking University, Beijing.
The study was published online in Brain and Behavior.
A new treatment option?
“As drug treatment for dementia has limitations such as medical contraindications, limited efficacy, and adverse effects, nonpharmacological therapy has been increasingly regarded as a critical part of comprehensive dementia care,” the investigators noted.
Phototherapy, which utilizes full-spectrum bright light (usually > 600 lux) or wavelength-specific light (for example, blue-enriched or blue-green), is a “promising nonpharmacological therapy” that is noninvasive, inexpensive, and safe.
Most studies of phototherapy have focused on sleep. Findings have shown “high heterogeneity” among the interventions and the populations in the studies, and results have been “inconsistent.” In addition, the effect of phototherapy on cognitive function and behavioral and psychological symptoms of dementia (BPSD) “still need to be clarified.”
In the systematic review and meta-analysis, the investigators examined the effects of phototherapy on cognitive function, BPSD, and sleep in older adults with dementia.
They searched several databases for randomized controlled trials that investigated phototherapy interventions for elderly patients. The primary outcome was cognitive function, which was assessed via the Mini-Mental State Examination (MMSE).
Secondary outcomes included BPSD, including agitation, anxiety, irritability, depression, anxiety, and sleep disturbances, as assessed by the Cornell Scale for Depression in Dementia (CSDD), the Cohen-Mansfield Agitation Inventory (CMAI), the Neuropsychiatric Inventory (NPI), and measures of sleep, including total sleep time (TST), sleep efficiency (SE), and sleep disorders, as assessed by the Sleep Disorder Inventory (SDI).
To be included in the analysis, individual studies had to focus on elderly adults who had some form of dementia. In addition, a group receiving a phototherapy intervention had to be compared with a nonintervention group, and the study had to specify one of the above-defined outcomes.
The review included phototherapy interventions of all forms, frequencies, and durations, including use of bright light, LED light, and blue or blue-green light.
Regulating circadian rhythm
Twelve studies met the researchers’ criteria. They included a total of 766 patients with dementia – 426 in the intervention group and 340 in the control group. The mean ages ranged from 73.73 to 85.9 years, and there was a greater number of female than male participants.
Of the studies, seven employed routine daily light in the control group, while the others used either dim light (≤ 50 lux) or devices without light.
The researchers found “significant positive intervention effects” for global cognitive function. Improvements in postintervention MMSE scores differed significantly between the experimental groups and control groups (mean difference, 2.68; 95% confidence interval, 1.38-3.98; I2 = 0%).
No significant differences were found in the effects of intervention on depression symptoms, as evidenced in CSDD scores (MD, −0.70; 95% CI, −3.10 to 1.70; I2 = 81%).
Among patients with higher CMAI scores, which indicate more severe agitation behaviors, there was a “trend of decreasing CMAI scores” after phototherapy (MD, −3.12; 95% CI, −8.05 to 1.82; I2 = 0%). No significant difference in NPI scores was observed between the two groups.
Similarly, no significant difference was found between the two groups in TST, SE, or SDI scores.
Adverse effects were infrequent and were not severe. Two of the 426 patients in the intervention group experienced mild ocular irritation, and one experienced slight transient redness of the forehead.
Light “may compensate for the reduction in the visual sensory input of patients with dementia and stimulate specific neurons in the suprachiasmatic nucleus of the hypothalamus to regulate circadian rhythm,” the researchers suggested.
“As circadian rhythms are involved in optimal brain function, light supplementation may act on the synchronizing/phase-shifting effects of circadian rhythms to improve cognitive function,” they added.
They note that the light box is the “most commonly used device in phototherapy.” Light boxes provide full-spectrum bright light, usually greater than 2,500 lux. The duration is 30 minutes in the daytime, and treatment lasts 4-8 weeks.
The investigators cautioned that the light box should be placed 60 cm away from the patient or above the patient’s eye level. They said that a ceiling-mounted light is a “good choice” for providing whole-day phototherapy, since such lights do not interfere with the patient’s daily routine, reduce the demand on staff, and contribute to better adherence.
Phototherapy helmets and glasses are also available. These portable devices “allow for better control of light intensity and are ergonomic without interfering with patients’ normal activities.”
The researchers noted that “further well-designed studies are needed to explore the most effective clinical implementation conditions, including device type, duration, frequency, and time.”
Easy to use
Mariana Figueiro, PhD, professor and director of the Light and Health Research Center, department of population health medicine, Icahn School of Medicine at Mount Sinai, New York, said light is the “major stimulus for the circadian system, and a robust light-dark pattern daily (which can be given by light therapy during the day) improves sleep and behavior and reduces depression and agitation.”
Dr. Figueiro, who was not involved with the current study, noted that patients with dementia “have sleep issues, which can further affect their cognition; improvement in sleep leads to improvement in cognition,” and this may be an underlying mechanism associated with these results.
The clinical significance of the study “is that this is a nonpharmacological intervention and can be easily applied in the homes or controlled facilities, and it can be used with any other medication,” she pointed out.
“More importantly, sleep medications have negative side effects, so the use of nonpharmacological interventions improving sleep and cognition is great for clinical practice,” she added.
However, she took issue with the finding that phototherapy was not effective for depression and agitation, noting that there were “too few studies to say for sure that light therapy is ineffective at improving these outcomes.”
The research received no external funding. The authors and Dr. Figueiro disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
It may be “one of the most promising interventions for improving core symptoms” of the disease.
A new meta-analysis shows that patients with dementia who received phototherapy experienced significant cognitive improvement, compared with those who received usual treatment. However, there were no differences between study groups in terms of improved depression, agitation, or sleep problems.
“Our meta-analysis indicates that phototherapy improved cognitive function in patients with dementia. ... This suggests that phototherapy may be one of the most promising non-pharmacological interventions for improving core symptoms of dementia,” wrote the investigators, led by Xinlian Lu, Peking University, Beijing.
The study was published online in Brain and Behavior.
A new treatment option?
“As drug treatment for dementia has limitations such as medical contraindications, limited efficacy, and adverse effects, nonpharmacological therapy has been increasingly regarded as a critical part of comprehensive dementia care,” the investigators noted.
Phototherapy, which utilizes full-spectrum bright light (usually > 600 lux) or wavelength-specific light (for example, blue-enriched or blue-green), is a “promising nonpharmacological therapy” that is noninvasive, inexpensive, and safe.
Most studies of phototherapy have focused on sleep. Findings have shown “high heterogeneity” among the interventions and the populations in the studies, and results have been “inconsistent.” In addition, the effect of phototherapy on cognitive function and behavioral and psychological symptoms of dementia (BPSD) “still need to be clarified.”
In the systematic review and meta-analysis, the investigators examined the effects of phototherapy on cognitive function, BPSD, and sleep in older adults with dementia.
They searched several databases for randomized controlled trials that investigated phototherapy interventions for elderly patients. The primary outcome was cognitive function, which was assessed via the Mini-Mental State Examination (MMSE).
Secondary outcomes included BPSD, including agitation, anxiety, irritability, depression, anxiety, and sleep disturbances, as assessed by the Cornell Scale for Depression in Dementia (CSDD), the Cohen-Mansfield Agitation Inventory (CMAI), the Neuropsychiatric Inventory (NPI), and measures of sleep, including total sleep time (TST), sleep efficiency (SE), and sleep disorders, as assessed by the Sleep Disorder Inventory (SDI).
To be included in the analysis, individual studies had to focus on elderly adults who had some form of dementia. In addition, a group receiving a phototherapy intervention had to be compared with a nonintervention group, and the study had to specify one of the above-defined outcomes.
The review included phototherapy interventions of all forms, frequencies, and durations, including use of bright light, LED light, and blue or blue-green light.
Regulating circadian rhythm
Twelve studies met the researchers’ criteria. They included a total of 766 patients with dementia – 426 in the intervention group and 340 in the control group. The mean ages ranged from 73.73 to 85.9 years, and there was a greater number of female than male participants.
Of the studies, seven employed routine daily light in the control group, while the others used either dim light (≤ 50 lux) or devices without light.
The researchers found “significant positive intervention effects” for global cognitive function. Improvements in postintervention MMSE scores differed significantly between the experimental groups and control groups (mean difference, 2.68; 95% confidence interval, 1.38-3.98; I2 = 0%).
No significant differences were found in the effects of intervention on depression symptoms, as evidenced in CSDD scores (MD, −0.70; 95% CI, −3.10 to 1.70; I2 = 81%).
Among patients with higher CMAI scores, which indicate more severe agitation behaviors, there was a “trend of decreasing CMAI scores” after phototherapy (MD, −3.12; 95% CI, −8.05 to 1.82; I2 = 0%). No significant difference in NPI scores was observed between the two groups.
Similarly, no significant difference was found between the two groups in TST, SE, or SDI scores.
Adverse effects were infrequent and were not severe. Two of the 426 patients in the intervention group experienced mild ocular irritation, and one experienced slight transient redness of the forehead.
Light “may compensate for the reduction in the visual sensory input of patients with dementia and stimulate specific neurons in the suprachiasmatic nucleus of the hypothalamus to regulate circadian rhythm,” the researchers suggested.
“As circadian rhythms are involved in optimal brain function, light supplementation may act on the synchronizing/phase-shifting effects of circadian rhythms to improve cognitive function,” they added.
They note that the light box is the “most commonly used device in phototherapy.” Light boxes provide full-spectrum bright light, usually greater than 2,500 lux. The duration is 30 minutes in the daytime, and treatment lasts 4-8 weeks.
The investigators cautioned that the light box should be placed 60 cm away from the patient or above the patient’s eye level. They said that a ceiling-mounted light is a “good choice” for providing whole-day phototherapy, since such lights do not interfere with the patient’s daily routine, reduce the demand on staff, and contribute to better adherence.
Phototherapy helmets and glasses are also available. These portable devices “allow for better control of light intensity and are ergonomic without interfering with patients’ normal activities.”
The researchers noted that “further well-designed studies are needed to explore the most effective clinical implementation conditions, including device type, duration, frequency, and time.”
Easy to use
Mariana Figueiro, PhD, professor and director of the Light and Health Research Center, department of population health medicine, Icahn School of Medicine at Mount Sinai, New York, said light is the “major stimulus for the circadian system, and a robust light-dark pattern daily (which can be given by light therapy during the day) improves sleep and behavior and reduces depression and agitation.”
Dr. Figueiro, who was not involved with the current study, noted that patients with dementia “have sleep issues, which can further affect their cognition; improvement in sleep leads to improvement in cognition,” and this may be an underlying mechanism associated with these results.
The clinical significance of the study “is that this is a nonpharmacological intervention and can be easily applied in the homes or controlled facilities, and it can be used with any other medication,” she pointed out.
“More importantly, sleep medications have negative side effects, so the use of nonpharmacological interventions improving sleep and cognition is great for clinical practice,” she added.
However, she took issue with the finding that phototherapy was not effective for depression and agitation, noting that there were “too few studies to say for sure that light therapy is ineffective at improving these outcomes.”
The research received no external funding. The authors and Dr. Figueiro disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
It may be “one of the most promising interventions for improving core symptoms” of the disease.
A new meta-analysis shows that patients with dementia who received phototherapy experienced significant cognitive improvement, compared with those who received usual treatment. However, there were no differences between study groups in terms of improved depression, agitation, or sleep problems.
“Our meta-analysis indicates that phototherapy improved cognitive function in patients with dementia. ... This suggests that phototherapy may be one of the most promising non-pharmacological interventions for improving core symptoms of dementia,” wrote the investigators, led by Xinlian Lu, Peking University, Beijing.
The study was published online in Brain and Behavior.
A new treatment option?
“As drug treatment for dementia has limitations such as medical contraindications, limited efficacy, and adverse effects, nonpharmacological therapy has been increasingly regarded as a critical part of comprehensive dementia care,” the investigators noted.
Phototherapy, which utilizes full-spectrum bright light (usually > 600 lux) or wavelength-specific light (for example, blue-enriched or blue-green), is a “promising nonpharmacological therapy” that is noninvasive, inexpensive, and safe.
Most studies of phototherapy have focused on sleep. Findings have shown “high heterogeneity” among the interventions and the populations in the studies, and results have been “inconsistent.” In addition, the effect of phototherapy on cognitive function and behavioral and psychological symptoms of dementia (BPSD) “still need to be clarified.”
In the systematic review and meta-analysis, the investigators examined the effects of phototherapy on cognitive function, BPSD, and sleep in older adults with dementia.
They searched several databases for randomized controlled trials that investigated phototherapy interventions for elderly patients. The primary outcome was cognitive function, which was assessed via the Mini-Mental State Examination (MMSE).
Secondary outcomes included BPSD, including agitation, anxiety, irritability, depression, anxiety, and sleep disturbances, as assessed by the Cornell Scale for Depression in Dementia (CSDD), the Cohen-Mansfield Agitation Inventory (CMAI), the Neuropsychiatric Inventory (NPI), and measures of sleep, including total sleep time (TST), sleep efficiency (SE), and sleep disorders, as assessed by the Sleep Disorder Inventory (SDI).
To be included in the analysis, individual studies had to focus on elderly adults who had some form of dementia. In addition, a group receiving a phototherapy intervention had to be compared with a nonintervention group, and the study had to specify one of the above-defined outcomes.
The review included phototherapy interventions of all forms, frequencies, and durations, including use of bright light, LED light, and blue or blue-green light.
Regulating circadian rhythm
Twelve studies met the researchers’ criteria. They included a total of 766 patients with dementia – 426 in the intervention group and 340 in the control group. The mean ages ranged from 73.73 to 85.9 years, and there was a greater number of female than male participants.
Of the studies, seven employed routine daily light in the control group, while the others used either dim light (≤ 50 lux) or devices without light.
The researchers found “significant positive intervention effects” for global cognitive function. Improvements in postintervention MMSE scores differed significantly between the experimental groups and control groups (mean difference, 2.68; 95% confidence interval, 1.38-3.98; I2 = 0%).
No significant differences were found in the effects of intervention on depression symptoms, as evidenced in CSDD scores (MD, −0.70; 95% CI, −3.10 to 1.70; I2 = 81%).
Among patients with higher CMAI scores, which indicate more severe agitation behaviors, there was a “trend of decreasing CMAI scores” after phototherapy (MD, −3.12; 95% CI, −8.05 to 1.82; I2 = 0%). No significant difference in NPI scores was observed between the two groups.
Similarly, no significant difference was found between the two groups in TST, SE, or SDI scores.
Adverse effects were infrequent and were not severe. Two of the 426 patients in the intervention group experienced mild ocular irritation, and one experienced slight transient redness of the forehead.
Light “may compensate for the reduction in the visual sensory input of patients with dementia and stimulate specific neurons in the suprachiasmatic nucleus of the hypothalamus to regulate circadian rhythm,” the researchers suggested.
“As circadian rhythms are involved in optimal brain function, light supplementation may act on the synchronizing/phase-shifting effects of circadian rhythms to improve cognitive function,” they added.
They note that the light box is the “most commonly used device in phototherapy.” Light boxes provide full-spectrum bright light, usually greater than 2,500 lux. The duration is 30 minutes in the daytime, and treatment lasts 4-8 weeks.
The investigators cautioned that the light box should be placed 60 cm away from the patient or above the patient’s eye level. They said that a ceiling-mounted light is a “good choice” for providing whole-day phototherapy, since such lights do not interfere with the patient’s daily routine, reduce the demand on staff, and contribute to better adherence.
Phototherapy helmets and glasses are also available. These portable devices “allow for better control of light intensity and are ergonomic without interfering with patients’ normal activities.”
The researchers noted that “further well-designed studies are needed to explore the most effective clinical implementation conditions, including device type, duration, frequency, and time.”
Easy to use
Mariana Figueiro, PhD, professor and director of the Light and Health Research Center, department of population health medicine, Icahn School of Medicine at Mount Sinai, New York, said light is the “major stimulus for the circadian system, and a robust light-dark pattern daily (which can be given by light therapy during the day) improves sleep and behavior and reduces depression and agitation.”
Dr. Figueiro, who was not involved with the current study, noted that patients with dementia “have sleep issues, which can further affect their cognition; improvement in sleep leads to improvement in cognition,” and this may be an underlying mechanism associated with these results.
The clinical significance of the study “is that this is a nonpharmacological intervention and can be easily applied in the homes or controlled facilities, and it can be used with any other medication,” she pointed out.
“More importantly, sleep medications have negative side effects, so the use of nonpharmacological interventions improving sleep and cognition is great for clinical practice,” she added.
However, she took issue with the finding that phototherapy was not effective for depression and agitation, noting that there were “too few studies to say for sure that light therapy is ineffective at improving these outcomes.”
The research received no external funding. The authors and Dr. Figueiro disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM BRAIN AND BEHAVIOR
Seven ‘simple’ cardiovascular health measures linked to reduced dementia risk in women
according to results of a study that was released early, ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology.
Epidemiologist Pamela M. Rist, ScD, assistant professor of medicine at Harvard Medical School and associate epidemiologist at Brigham and Women’s Hospital, both in Boston, and colleagues, used data from 13,720 women whose mean age was 54 when they enrolled in the Harvard-based Women’s Health Study between 1992 and 1995. Subjects in that study were followed up in 2004.
Putting ‘Life’s Simple 7’ to the test
Dr. Rist and colleagues used the Harvard data to discern how well closely women conformed, during the initial study period and at 10-year follow up, to what the American Heart Association describes as “Life’s Simple 7,” a list of behavioral and biometric measures that indicate and predict cardiovascular health. The measures include four modifiable behaviors – not smoking, healthy weight, a healthy diet, and being physically active – along with three biometric measures of blood pressure, cholesterol, and blood sugar (AHA has since added a sleep component).
Researchers assigned women one point for each desirable habit or measure on the list, with subjects’ average Simple 7 score at baseline 4.3, and 4.2 at 10 years’ follow-up.
The investigators then looked at Medicare data for the study subjects from 2011 to 2018 – approximately 20 years after their enrollment in the Women’s Health Study – seeking dementia diagnoses. Some 13% of the study cohort (n = 1,771) had gone on to develop dementia.
Each point on the Simple 7 score at baseline corresponded with a 6% reduction in later dementia risk, Dr. Rist and her colleagues found after adjusting for variables including age and education (odds ratio per one unit change in score, 0.94; 95% CI, 0.90-0.98). This effect was similar for Simple 7 scores measured at 10 years of follow-up (OR, 0.95; 95% CI, 0.91-1.00).
“It can be empowering for people to know that by taking steps such as exercising for a half an hour a day or keeping their blood pressure under control, they can reduce their risk of dementia,” Dr. Rist said in a statement on the findings.
‘A simple take-home message’
Reached for comment, Andrew E. Budson, MD, chief of cognitive-behavioral neurology at the VA Boston Healthcare System, praised Dr. Rist and colleagues’ study as one that “builds on existing knowledge to provide a simple take-home message that empowers women to take control of their dementia risk.”
Each of the seven known risk factors – being active, eating better, maintaining a healthy weight, not smoking, maintaining a healthy blood pressure, controlling cholesterol, and having low blood sugar – “was associated with a 6% reduced risk of dementia,” Dr. Budson continued. “So, women who work to address all seven risk factors can reduce their risk of developing dementia by 42%: a huge amount. Moreover, although this study only looked at women, I am confident that if men follow this same advice they will also be able to reduce their risk of dementia, although we don’t know if the size of the effect will be the same.”
Dr. Rist and colleagues’ study was supported by the National Institutes of Health. None of the study authors reported conflicts of interest. Dr. Budson has reported receiving past compensation as a speaker for Eli Lilly.
according to results of a study that was released early, ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology.
Epidemiologist Pamela M. Rist, ScD, assistant professor of medicine at Harvard Medical School and associate epidemiologist at Brigham and Women’s Hospital, both in Boston, and colleagues, used data from 13,720 women whose mean age was 54 when they enrolled in the Harvard-based Women’s Health Study between 1992 and 1995. Subjects in that study were followed up in 2004.
Putting ‘Life’s Simple 7’ to the test
Dr. Rist and colleagues used the Harvard data to discern how well closely women conformed, during the initial study period and at 10-year follow up, to what the American Heart Association describes as “Life’s Simple 7,” a list of behavioral and biometric measures that indicate and predict cardiovascular health. The measures include four modifiable behaviors – not smoking, healthy weight, a healthy diet, and being physically active – along with three biometric measures of blood pressure, cholesterol, and blood sugar (AHA has since added a sleep component).
Researchers assigned women one point for each desirable habit or measure on the list, with subjects’ average Simple 7 score at baseline 4.3, and 4.2 at 10 years’ follow-up.
The investigators then looked at Medicare data for the study subjects from 2011 to 2018 – approximately 20 years after their enrollment in the Women’s Health Study – seeking dementia diagnoses. Some 13% of the study cohort (n = 1,771) had gone on to develop dementia.
Each point on the Simple 7 score at baseline corresponded with a 6% reduction in later dementia risk, Dr. Rist and her colleagues found after adjusting for variables including age and education (odds ratio per one unit change in score, 0.94; 95% CI, 0.90-0.98). This effect was similar for Simple 7 scores measured at 10 years of follow-up (OR, 0.95; 95% CI, 0.91-1.00).
“It can be empowering for people to know that by taking steps such as exercising for a half an hour a day or keeping their blood pressure under control, they can reduce their risk of dementia,” Dr. Rist said in a statement on the findings.
‘A simple take-home message’
Reached for comment, Andrew E. Budson, MD, chief of cognitive-behavioral neurology at the VA Boston Healthcare System, praised Dr. Rist and colleagues’ study as one that “builds on existing knowledge to provide a simple take-home message that empowers women to take control of their dementia risk.”
Each of the seven known risk factors – being active, eating better, maintaining a healthy weight, not smoking, maintaining a healthy blood pressure, controlling cholesterol, and having low blood sugar – “was associated with a 6% reduced risk of dementia,” Dr. Budson continued. “So, women who work to address all seven risk factors can reduce their risk of developing dementia by 42%: a huge amount. Moreover, although this study only looked at women, I am confident that if men follow this same advice they will also be able to reduce their risk of dementia, although we don’t know if the size of the effect will be the same.”
Dr. Rist and colleagues’ study was supported by the National Institutes of Health. None of the study authors reported conflicts of interest. Dr. Budson has reported receiving past compensation as a speaker for Eli Lilly.
according to results of a study that was released early, ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology.
Epidemiologist Pamela M. Rist, ScD, assistant professor of medicine at Harvard Medical School and associate epidemiologist at Brigham and Women’s Hospital, both in Boston, and colleagues, used data from 13,720 women whose mean age was 54 when they enrolled in the Harvard-based Women’s Health Study between 1992 and 1995. Subjects in that study were followed up in 2004.
Putting ‘Life’s Simple 7’ to the test
Dr. Rist and colleagues used the Harvard data to discern how well closely women conformed, during the initial study period and at 10-year follow up, to what the American Heart Association describes as “Life’s Simple 7,” a list of behavioral and biometric measures that indicate and predict cardiovascular health. The measures include four modifiable behaviors – not smoking, healthy weight, a healthy diet, and being physically active – along with three biometric measures of blood pressure, cholesterol, and blood sugar (AHA has since added a sleep component).
Researchers assigned women one point for each desirable habit or measure on the list, with subjects’ average Simple 7 score at baseline 4.3, and 4.2 at 10 years’ follow-up.
The investigators then looked at Medicare data for the study subjects from 2011 to 2018 – approximately 20 years after their enrollment in the Women’s Health Study – seeking dementia diagnoses. Some 13% of the study cohort (n = 1,771) had gone on to develop dementia.
Each point on the Simple 7 score at baseline corresponded with a 6% reduction in later dementia risk, Dr. Rist and her colleagues found after adjusting for variables including age and education (odds ratio per one unit change in score, 0.94; 95% CI, 0.90-0.98). This effect was similar for Simple 7 scores measured at 10 years of follow-up (OR, 0.95; 95% CI, 0.91-1.00).
“It can be empowering for people to know that by taking steps such as exercising for a half an hour a day or keeping their blood pressure under control, they can reduce their risk of dementia,” Dr. Rist said in a statement on the findings.
‘A simple take-home message’
Reached for comment, Andrew E. Budson, MD, chief of cognitive-behavioral neurology at the VA Boston Healthcare System, praised Dr. Rist and colleagues’ study as one that “builds on existing knowledge to provide a simple take-home message that empowers women to take control of their dementia risk.”
Each of the seven known risk factors – being active, eating better, maintaining a healthy weight, not smoking, maintaining a healthy blood pressure, controlling cholesterol, and having low blood sugar – “was associated with a 6% reduced risk of dementia,” Dr. Budson continued. “So, women who work to address all seven risk factors can reduce their risk of developing dementia by 42%: a huge amount. Moreover, although this study only looked at women, I am confident that if men follow this same advice they will also be able to reduce their risk of dementia, although we don’t know if the size of the effect will be the same.”
Dr. Rist and colleagues’ study was supported by the National Institutes of Health. None of the study authors reported conflicts of interest. Dr. Budson has reported receiving past compensation as a speaker for Eli Lilly.
FROM AAN 2023
Antiamyloids linked to accelerated brain atrophy
a comprehensive meta-analysis of MRI data from clinical trials suggests.
Depending on the anti–amyloid-beta drug class, these agents can accelerate loss of whole brain and hippocampal volume and increase ventricular volume. This has been shown for some of the beta-secretase inhibitors and with several of the antiamyloid monoclonal antibodies, researchers noted.
“These data warrant concern, but we can’t make any firm conclusions yet. It is possible that the finding is not detrimental, but the usual interpretation of this finding is that volume changes are a surrogate for disease progression,” study investigator Scott Ayton, PhD, of the Florey Institute of Neuroscience and Mental Health, University of Melbourne, said in an interview.
“These data should be factored into the decisions by clinicians when they consider prescribing antiamyloid therapies. Like any side effect, clinicians should inform patients regarding the risk of brain atrophy. Patients should be actively monitored for this side effect,” Dr. Ayton said.
The study was published online in Neurology.
Earlier progression from MCI to AD?
Dr. Ayton and colleagues evaluated brain volume changes in 31 clinical trials of anti–amyloid-beta drugs that demonstrated a favorable change in at least one biomarker of pathological amyloid-beta and included detailed MRI data sufficient to assess the volumetric changes in at least one brain region.
A meta-analysis on the highest dose in each trial on the hippocampus, ventricles, and whole brain showed drug-induced acceleration of volume changes that varied by anti–amyloid-beta drug class.
Secretase inhibitors accelerated atrophy in the hippocampus (mean difference –37.1 mcL; –19.6% relative to change in placebo) and whole brain (mean difference –3.3 mL; –21.8% relative to change in placebo), but not ventricles.
Conversely, monoclonal antibodies caused accelerated ventricular enlargement (mean difference +1.3 mL; +23.8% relative to change in placebo), which was driven by the subset of monoclonal antibodies that induce amyloid-related imaging abnormalities (ARIA) (+2.1 mL; +38.7% relative to change in placebo). There was a “striking correlation between ventricular volume and ARIA frequency,” the investigators reported.
The effect of ARIA-inducing monoclonal antibodies on whole brain volume varied, with accelerated whole brain volume loss caused by donanemab (mean difference –4.6 mL; +23% relative to change in placebo) and lecanemab (–5.2 mL; +36.4% relative to change in placebo). This was not observed with aducanumab and bapineuzumab.
Monoclonal antibodies did not cause accelerated volume loss to the hippocampus regardless of whether they caused ARIA.
The researchers also modeled the effect of anti–amyloid-beta drugs on brain volume changes. In this analysis, participants with mild cognitive impairment (MCI) treated with anti–amyloid-beta drugs were projected to have a “material regression” toward brain volumes typical of AD roughly 8 months earlier than untreated peers.
The data, they note, “permit robust conclusions regarding the effect of [anti–amyloid-beta] drug classes on different brain structures, but the lack of individual patient data (which has yet to be released) limits the interpretations of our findings.”
“Questions like which brain regions are impacted by [anti–amyloid-beta] drugs and whether the volume changes are related to ARIA, plaque loss, cognitive/noncognitive outcomes, or clinical factors such as age, sex, and apoE4 genotype can and should be addressed with available data,” said Dr. Ayton.
Dr. Ayton and colleagues called on data safety monitoring boards (DSMBs) for current clinical trials of anti–amyloid-beta drugs to review volumetric data to determine if patient safety is at risk, particularly in patients who develop ARIA.
In addition, they noted ethics boards that approve trials for anti–amyloid-beta drugs “should request that volume changes be actively monitored. Long-term follow-up of brain volumes should be factored into the trial designs to determine if brain atrophy is progressive, particularly in patients who develop ARIA.”
Finally, they added that drug companies that have conducted trials of anti–amyloid-beta drugs should interrogate prior data on brain volume, report the findings, and release the data for researchers to investigate.
“I have been banging on about this for years,” said Dr. Ayton. “Unfortunately, my raising of this issue has not led to any response. The data are not available, and the basic questions haven’t been asked (publicly).”
Commendable research
In an accompanying editorial, Frederik Barkhof, MD, PhD, with Amsterdam University Medical Centers, and David Knopman, MD, with Mayo Clinic Alzheimer’s Disease Research Center, Rochester, Minn., wrote that the investigators should be “commended” for their analysis.
“The reality in 2023 is that the relevance of brain volume reductions in this therapeutic context remains uncertain,” they wrote.
“Longer periods of observation will be needed to know whether the brain volume losses continue at an accelerated rate or if they attenuate or disappear. Ultimately, it’s the clinical outcomes that matter, regardless of the MRI changes,” Barkhof and Knopman concluded.
The research was supported by funds from the Australian National Health & Medical Research Council. Dr. Ayton reported being a consultant for Eisai in the past 3 years. Dr. Barkhof reported serving on the data and safety monitoring board for Prothena and the A45-AHEAD studies; being a steering committee member for Merck, Bayer, and Biogen; and being a consultant for IXICO, Roche, Celltrion, Rewind Therapeutics, and Combinostics. Dr. Knopman reported serving on the DSMB for the Dominantly Inherited Alzheimer Network Treatment Unit study; serving on a DSMB for a tau therapeutic for Biogen; being an investigator for clinical trials sponsored by Biogen, Lilly Pharmaceuticals, and the University of Southern California. He reported consulting with Roche, Samus Therapeutics, Magellan Health, BioVie, and Alzeca Biosciences.
A version of this article first appeared on Medscape.com.
a comprehensive meta-analysis of MRI data from clinical trials suggests.
Depending on the anti–amyloid-beta drug class, these agents can accelerate loss of whole brain and hippocampal volume and increase ventricular volume. This has been shown for some of the beta-secretase inhibitors and with several of the antiamyloid monoclonal antibodies, researchers noted.
“These data warrant concern, but we can’t make any firm conclusions yet. It is possible that the finding is not detrimental, but the usual interpretation of this finding is that volume changes are a surrogate for disease progression,” study investigator Scott Ayton, PhD, of the Florey Institute of Neuroscience and Mental Health, University of Melbourne, said in an interview.
“These data should be factored into the decisions by clinicians when they consider prescribing antiamyloid therapies. Like any side effect, clinicians should inform patients regarding the risk of brain atrophy. Patients should be actively monitored for this side effect,” Dr. Ayton said.
The study was published online in Neurology.
Earlier progression from MCI to AD?
Dr. Ayton and colleagues evaluated brain volume changes in 31 clinical trials of anti–amyloid-beta drugs that demonstrated a favorable change in at least one biomarker of pathological amyloid-beta and included detailed MRI data sufficient to assess the volumetric changes in at least one brain region.
A meta-analysis on the highest dose in each trial on the hippocampus, ventricles, and whole brain showed drug-induced acceleration of volume changes that varied by anti–amyloid-beta drug class.
Secretase inhibitors accelerated atrophy in the hippocampus (mean difference –37.1 mcL; –19.6% relative to change in placebo) and whole brain (mean difference –3.3 mL; –21.8% relative to change in placebo), but not ventricles.
Conversely, monoclonal antibodies caused accelerated ventricular enlargement (mean difference +1.3 mL; +23.8% relative to change in placebo), which was driven by the subset of monoclonal antibodies that induce amyloid-related imaging abnormalities (ARIA) (+2.1 mL; +38.7% relative to change in placebo). There was a “striking correlation between ventricular volume and ARIA frequency,” the investigators reported.
The effect of ARIA-inducing monoclonal antibodies on whole brain volume varied, with accelerated whole brain volume loss caused by donanemab (mean difference –4.6 mL; +23% relative to change in placebo) and lecanemab (–5.2 mL; +36.4% relative to change in placebo). This was not observed with aducanumab and bapineuzumab.
Monoclonal antibodies did not cause accelerated volume loss to the hippocampus regardless of whether they caused ARIA.
The researchers also modeled the effect of anti–amyloid-beta drugs on brain volume changes. In this analysis, participants with mild cognitive impairment (MCI) treated with anti–amyloid-beta drugs were projected to have a “material regression” toward brain volumes typical of AD roughly 8 months earlier than untreated peers.
The data, they note, “permit robust conclusions regarding the effect of [anti–amyloid-beta] drug classes on different brain structures, but the lack of individual patient data (which has yet to be released) limits the interpretations of our findings.”
“Questions like which brain regions are impacted by [anti–amyloid-beta] drugs and whether the volume changes are related to ARIA, plaque loss, cognitive/noncognitive outcomes, or clinical factors such as age, sex, and apoE4 genotype can and should be addressed with available data,” said Dr. Ayton.
Dr. Ayton and colleagues called on data safety monitoring boards (DSMBs) for current clinical trials of anti–amyloid-beta drugs to review volumetric data to determine if patient safety is at risk, particularly in patients who develop ARIA.
In addition, they noted ethics boards that approve trials for anti–amyloid-beta drugs “should request that volume changes be actively monitored. Long-term follow-up of brain volumes should be factored into the trial designs to determine if brain atrophy is progressive, particularly in patients who develop ARIA.”
Finally, they added that drug companies that have conducted trials of anti–amyloid-beta drugs should interrogate prior data on brain volume, report the findings, and release the data for researchers to investigate.
“I have been banging on about this for years,” said Dr. Ayton. “Unfortunately, my raising of this issue has not led to any response. The data are not available, and the basic questions haven’t been asked (publicly).”
Commendable research
In an accompanying editorial, Frederik Barkhof, MD, PhD, with Amsterdam University Medical Centers, and David Knopman, MD, with Mayo Clinic Alzheimer’s Disease Research Center, Rochester, Minn., wrote that the investigators should be “commended” for their analysis.
“The reality in 2023 is that the relevance of brain volume reductions in this therapeutic context remains uncertain,” they wrote.
“Longer periods of observation will be needed to know whether the brain volume losses continue at an accelerated rate or if they attenuate or disappear. Ultimately, it’s the clinical outcomes that matter, regardless of the MRI changes,” Barkhof and Knopman concluded.
The research was supported by funds from the Australian National Health & Medical Research Council. Dr. Ayton reported being a consultant for Eisai in the past 3 years. Dr. Barkhof reported serving on the data and safety monitoring board for Prothena and the A45-AHEAD studies; being a steering committee member for Merck, Bayer, and Biogen; and being a consultant for IXICO, Roche, Celltrion, Rewind Therapeutics, and Combinostics. Dr. Knopman reported serving on the DSMB for the Dominantly Inherited Alzheimer Network Treatment Unit study; serving on a DSMB for a tau therapeutic for Biogen; being an investigator for clinical trials sponsored by Biogen, Lilly Pharmaceuticals, and the University of Southern California. He reported consulting with Roche, Samus Therapeutics, Magellan Health, BioVie, and Alzeca Biosciences.
A version of this article first appeared on Medscape.com.
a comprehensive meta-analysis of MRI data from clinical trials suggests.
Depending on the anti–amyloid-beta drug class, these agents can accelerate loss of whole brain and hippocampal volume and increase ventricular volume. This has been shown for some of the beta-secretase inhibitors and with several of the antiamyloid monoclonal antibodies, researchers noted.
“These data warrant concern, but we can’t make any firm conclusions yet. It is possible that the finding is not detrimental, but the usual interpretation of this finding is that volume changes are a surrogate for disease progression,” study investigator Scott Ayton, PhD, of the Florey Institute of Neuroscience and Mental Health, University of Melbourne, said in an interview.
“These data should be factored into the decisions by clinicians when they consider prescribing antiamyloid therapies. Like any side effect, clinicians should inform patients regarding the risk of brain atrophy. Patients should be actively monitored for this side effect,” Dr. Ayton said.
The study was published online in Neurology.
Earlier progression from MCI to AD?
Dr. Ayton and colleagues evaluated brain volume changes in 31 clinical trials of anti–amyloid-beta drugs that demonstrated a favorable change in at least one biomarker of pathological amyloid-beta and included detailed MRI data sufficient to assess the volumetric changes in at least one brain region.
A meta-analysis on the highest dose in each trial on the hippocampus, ventricles, and whole brain showed drug-induced acceleration of volume changes that varied by anti–amyloid-beta drug class.
Secretase inhibitors accelerated atrophy in the hippocampus (mean difference –37.1 mcL; –19.6% relative to change in placebo) and whole brain (mean difference –3.3 mL; –21.8% relative to change in placebo), but not ventricles.
Conversely, monoclonal antibodies caused accelerated ventricular enlargement (mean difference +1.3 mL; +23.8% relative to change in placebo), which was driven by the subset of monoclonal antibodies that induce amyloid-related imaging abnormalities (ARIA) (+2.1 mL; +38.7% relative to change in placebo). There was a “striking correlation between ventricular volume and ARIA frequency,” the investigators reported.
The effect of ARIA-inducing monoclonal antibodies on whole brain volume varied, with accelerated whole brain volume loss caused by donanemab (mean difference –4.6 mL; +23% relative to change in placebo) and lecanemab (–5.2 mL; +36.4% relative to change in placebo). This was not observed with aducanumab and bapineuzumab.
Monoclonal antibodies did not cause accelerated volume loss to the hippocampus regardless of whether they caused ARIA.
The researchers also modeled the effect of anti–amyloid-beta drugs on brain volume changes. In this analysis, participants with mild cognitive impairment (MCI) treated with anti–amyloid-beta drugs were projected to have a “material regression” toward brain volumes typical of AD roughly 8 months earlier than untreated peers.
The data, they note, “permit robust conclusions regarding the effect of [anti–amyloid-beta] drug classes on different brain structures, but the lack of individual patient data (which has yet to be released) limits the interpretations of our findings.”
“Questions like which brain regions are impacted by [anti–amyloid-beta] drugs and whether the volume changes are related to ARIA, plaque loss, cognitive/noncognitive outcomes, or clinical factors such as age, sex, and apoE4 genotype can and should be addressed with available data,” said Dr. Ayton.
Dr. Ayton and colleagues called on data safety monitoring boards (DSMBs) for current clinical trials of anti–amyloid-beta drugs to review volumetric data to determine if patient safety is at risk, particularly in patients who develop ARIA.
In addition, they noted ethics boards that approve trials for anti–amyloid-beta drugs “should request that volume changes be actively monitored. Long-term follow-up of brain volumes should be factored into the trial designs to determine if brain atrophy is progressive, particularly in patients who develop ARIA.”
Finally, they added that drug companies that have conducted trials of anti–amyloid-beta drugs should interrogate prior data on brain volume, report the findings, and release the data for researchers to investigate.
“I have been banging on about this for years,” said Dr. Ayton. “Unfortunately, my raising of this issue has not led to any response. The data are not available, and the basic questions haven’t been asked (publicly).”
Commendable research
In an accompanying editorial, Frederik Barkhof, MD, PhD, with Amsterdam University Medical Centers, and David Knopman, MD, with Mayo Clinic Alzheimer’s Disease Research Center, Rochester, Minn., wrote that the investigators should be “commended” for their analysis.
“The reality in 2023 is that the relevance of brain volume reductions in this therapeutic context remains uncertain,” they wrote.
“Longer periods of observation will be needed to know whether the brain volume losses continue at an accelerated rate or if they attenuate or disappear. Ultimately, it’s the clinical outcomes that matter, regardless of the MRI changes,” Barkhof and Knopman concluded.
The research was supported by funds from the Australian National Health & Medical Research Council. Dr. Ayton reported being a consultant for Eisai in the past 3 years. Dr. Barkhof reported serving on the data and safety monitoring board for Prothena and the A45-AHEAD studies; being a steering committee member for Merck, Bayer, and Biogen; and being a consultant for IXICO, Roche, Celltrion, Rewind Therapeutics, and Combinostics. Dr. Knopman reported serving on the DSMB for the Dominantly Inherited Alzheimer Network Treatment Unit study; serving on a DSMB for a tau therapeutic for Biogen; being an investigator for clinical trials sponsored by Biogen, Lilly Pharmaceuticals, and the University of Southern California. He reported consulting with Roche, Samus Therapeutics, Magellan Health, BioVie, and Alzeca Biosciences.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Kidney disease skews Alzheimer’s biomarker testing
New research provides more evidence that
In a cross-sectional study of adults with and those without cognitive impairment, chronic kidney disease was associated with increased plasma concentrations of phosphorylated tau (p-tau) 217 and 181.
However, there were no associations between chronic kidney disease and the ratio of p-tau217 to unphosphorylated tau 217 (pT217/T217), and the associations with p-tau181 to unphosphorylated tau 181 (pT181/T181) were attenuated in patients with cognitive impairment.
“These novel findings suggest that plasma measures of the phosphorylated to unphosphorylated tau ratios are more accurate than p-tau forms alone as they correlate less with individual difference in glomerular filtration rate or impaired kidney function,” reported an investigative team led by Oskar Hansson, MD, PhD, with Lund University in Sweden.
“Thus, to mitigate the effects of non-Alzheimer’s–related comorbidities like chronic kidney disease on the performance of plasma Alzheimer’s disease biomarkers, certain tau ratios, and specifically pT217/T217, should be considered for implementation in clinical practice and drug trials,” they added.
The study was published online in JAMA Neurology to coincide with a presentation at the International Conference on Alzheimer’s and Parkinson’s Diseases in Gothenburg, Sweden.
Skewed tau levels
Plasma biomarkers of amyloid-beta (Abeta) and tau pathologies, and in particular variants in p-tau217 and p-tau181, have shown promise for use in Alzheimer’s disease diagnosis and prognosis. However, previous reports have suggested that chronic kidney disease might influence plasma p-tau217 and p-tau181 concentrations, potentially decreasing their usefulness in the diagnostic workup of dementia.
Researchers investigated associations of chronic kidney disease with plasma ratios of p-tau217 and p-tau181 to the corresponding unphosphorylated peptides in Alzheimer’s disease.
The data came from 473 older adults, with and without cognitive impairment, from the Swedish BioFinder-2 study who had plasma tau assessments and chronic kidney disease status established within 6 months of plasma collection.
The researchers found that lower estimated glomerular filtration rate levels (indicative of kidney dysfunction) were associated with higher plasma concentrations of phosphorylated and unphosphorylated tau peptides measured simultaneously using the tau immunoprecipitation mass spectrometry assay.
However, the correlations with estimated glomerular filtration rate were nonsignificant for the pT217/T217 ratio in individuals with cognitive impairment and were significantly attenuated for pT217/T217 in cognitively unimpaired individuals and for pT181/T181 in both cognitively unimpaired and impaired individuals.
“Importantly, we demonstrate that there were no significant associations between chronic kidney disease and the pT217/T217 ratio and changes in plasma pT181/T181 associated with chronic kidney disease were small or nonsignificant,” the researchers noted.
“Our results indicate that by using p-tau/tau ratios we may be able to reduce the variability in plasma p-tau levels driven by impaired kidney function and consequently such ratios are more robust measures of brain p-tau pathology in individuals with both early- and later-stage Alzheimer’s disease,” they added.
The researchers believe this is likely true for the ratios of other related proteins – a view that is supported by findings of attenuated associations of chronic kidney disease with Abeta42/40, compared with Abeta42 and Abeta40 in the current study and in previous studies.
Important clinical implications
Reached for comment, Rebecca Edelmayer, PhD, Alzheimer’s Association senior director of scientific engagement, noted that research continues to indicate that blood biomarkers have “promise for improving, and possibly even redefining, the clinical workup for Alzheimer’s disease.
“Many of the Alzheimer’s blood tests in development today have focused on core blood biomarkers associated with amyloid accumulation and tau tangle formation in the brain,” Dr. Edelmayer said.
“Before these tests can be used more broadly in the clinic, we need to understand all of the variables that may impact the results of various blood biomarkers, including differences that may be driven by race, ethnicity, sex, and underlying health conditions, such as chronic kidney disease.
“This study corroborates other research suggesting that some Alzheimer’s-associated markers can be affected by chronic kidney disease, but by using ratios of amyloid or tau markers, we may be able to minimize these differences in results caused by underlying disease,” Dr. Edelmayer said.
Howard Fillit, MD, cofounder and chief science officer at the Alzheimer’s Drug Discovery Foundation, said, “Using these ratios makes a lot of sense because the ratios wouldn’t change with kidney function. I think this is an important advance in clinical utilization of these tests.”
Dr. Fillit noted that older people often have declining kidney function, which can be easily measured by glomerular filtration rate. Changes in blood levels of these markers with declining kidney function are “not unexpected, but [it’s] important that it’s been demonstrated.
“As we go forward, maybe the future utilization of these tests will be not only recording the ratios but also reporting the ratios in the context of somebody’s glomerular filtration rate. You could imagine a scenario where when the test is done, it’s automatically done alongside of glomerular filtration rate,” Dr. Fillit said in an interview.
The study was supported by Coins for Alzheimer’s Research Trust, the Tracy Family Stable Isotope Labeling Quantitation Center, and the National Institute of Neurological Disorders and Stroke. Dr. Hansson has received support to his institution from ADx, AVID Radiopharmaceuticals, Biogen, Eli Lilly, Eisai, Fujirebio, GE Healthcare, Pfizer, and Roche; consultancy/speaker fees from Amylyx, Alzpath, Biogen, Cerveau, Fujirebio, Genentech, Novartis, Roche, and Siemens; and personal fees from Eli Lilly, Eisai, Bioarctic, and Biogen outside the submitted work. Dr. Edelmayer and Dr. Fillit have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New research provides more evidence that
In a cross-sectional study of adults with and those without cognitive impairment, chronic kidney disease was associated with increased plasma concentrations of phosphorylated tau (p-tau) 217 and 181.
However, there were no associations between chronic kidney disease and the ratio of p-tau217 to unphosphorylated tau 217 (pT217/T217), and the associations with p-tau181 to unphosphorylated tau 181 (pT181/T181) were attenuated in patients with cognitive impairment.
“These novel findings suggest that plasma measures of the phosphorylated to unphosphorylated tau ratios are more accurate than p-tau forms alone as they correlate less with individual difference in glomerular filtration rate or impaired kidney function,” reported an investigative team led by Oskar Hansson, MD, PhD, with Lund University in Sweden.
“Thus, to mitigate the effects of non-Alzheimer’s–related comorbidities like chronic kidney disease on the performance of plasma Alzheimer’s disease biomarkers, certain tau ratios, and specifically pT217/T217, should be considered for implementation in clinical practice and drug trials,” they added.
The study was published online in JAMA Neurology to coincide with a presentation at the International Conference on Alzheimer’s and Parkinson’s Diseases in Gothenburg, Sweden.
Skewed tau levels
Plasma biomarkers of amyloid-beta (Abeta) and tau pathologies, and in particular variants in p-tau217 and p-tau181, have shown promise for use in Alzheimer’s disease diagnosis and prognosis. However, previous reports have suggested that chronic kidney disease might influence plasma p-tau217 and p-tau181 concentrations, potentially decreasing their usefulness in the diagnostic workup of dementia.
Researchers investigated associations of chronic kidney disease with plasma ratios of p-tau217 and p-tau181 to the corresponding unphosphorylated peptides in Alzheimer’s disease.
The data came from 473 older adults, with and without cognitive impairment, from the Swedish BioFinder-2 study who had plasma tau assessments and chronic kidney disease status established within 6 months of plasma collection.
The researchers found that lower estimated glomerular filtration rate levels (indicative of kidney dysfunction) were associated with higher plasma concentrations of phosphorylated and unphosphorylated tau peptides measured simultaneously using the tau immunoprecipitation mass spectrometry assay.
However, the correlations with estimated glomerular filtration rate were nonsignificant for the pT217/T217 ratio in individuals with cognitive impairment and were significantly attenuated for pT217/T217 in cognitively unimpaired individuals and for pT181/T181 in both cognitively unimpaired and impaired individuals.
“Importantly, we demonstrate that there were no significant associations between chronic kidney disease and the pT217/T217 ratio and changes in plasma pT181/T181 associated with chronic kidney disease were small or nonsignificant,” the researchers noted.
“Our results indicate that by using p-tau/tau ratios we may be able to reduce the variability in plasma p-tau levels driven by impaired kidney function and consequently such ratios are more robust measures of brain p-tau pathology in individuals with both early- and later-stage Alzheimer’s disease,” they added.
The researchers believe this is likely true for the ratios of other related proteins – a view that is supported by findings of attenuated associations of chronic kidney disease with Abeta42/40, compared with Abeta42 and Abeta40 in the current study and in previous studies.
Important clinical implications
Reached for comment, Rebecca Edelmayer, PhD, Alzheimer’s Association senior director of scientific engagement, noted that research continues to indicate that blood biomarkers have “promise for improving, and possibly even redefining, the clinical workup for Alzheimer’s disease.
“Many of the Alzheimer’s blood tests in development today have focused on core blood biomarkers associated with amyloid accumulation and tau tangle formation in the brain,” Dr. Edelmayer said.
“Before these tests can be used more broadly in the clinic, we need to understand all of the variables that may impact the results of various blood biomarkers, including differences that may be driven by race, ethnicity, sex, and underlying health conditions, such as chronic kidney disease.
“This study corroborates other research suggesting that some Alzheimer’s-associated markers can be affected by chronic kidney disease, but by using ratios of amyloid or tau markers, we may be able to minimize these differences in results caused by underlying disease,” Dr. Edelmayer said.
Howard Fillit, MD, cofounder and chief science officer at the Alzheimer’s Drug Discovery Foundation, said, “Using these ratios makes a lot of sense because the ratios wouldn’t change with kidney function. I think this is an important advance in clinical utilization of these tests.”
Dr. Fillit noted that older people often have declining kidney function, which can be easily measured by glomerular filtration rate. Changes in blood levels of these markers with declining kidney function are “not unexpected, but [it’s] important that it’s been demonstrated.
“As we go forward, maybe the future utilization of these tests will be not only recording the ratios but also reporting the ratios in the context of somebody’s glomerular filtration rate. You could imagine a scenario where when the test is done, it’s automatically done alongside of glomerular filtration rate,” Dr. Fillit said in an interview.
The study was supported by Coins for Alzheimer’s Research Trust, the Tracy Family Stable Isotope Labeling Quantitation Center, and the National Institute of Neurological Disorders and Stroke. Dr. Hansson has received support to his institution from ADx, AVID Radiopharmaceuticals, Biogen, Eli Lilly, Eisai, Fujirebio, GE Healthcare, Pfizer, and Roche; consultancy/speaker fees from Amylyx, Alzpath, Biogen, Cerveau, Fujirebio, Genentech, Novartis, Roche, and Siemens; and personal fees from Eli Lilly, Eisai, Bioarctic, and Biogen outside the submitted work. Dr. Edelmayer and Dr. Fillit have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New research provides more evidence that
In a cross-sectional study of adults with and those without cognitive impairment, chronic kidney disease was associated with increased plasma concentrations of phosphorylated tau (p-tau) 217 and 181.
However, there were no associations between chronic kidney disease and the ratio of p-tau217 to unphosphorylated tau 217 (pT217/T217), and the associations with p-tau181 to unphosphorylated tau 181 (pT181/T181) were attenuated in patients with cognitive impairment.
“These novel findings suggest that plasma measures of the phosphorylated to unphosphorylated tau ratios are more accurate than p-tau forms alone as they correlate less with individual difference in glomerular filtration rate or impaired kidney function,” reported an investigative team led by Oskar Hansson, MD, PhD, with Lund University in Sweden.
“Thus, to mitigate the effects of non-Alzheimer’s–related comorbidities like chronic kidney disease on the performance of plasma Alzheimer’s disease biomarkers, certain tau ratios, and specifically pT217/T217, should be considered for implementation in clinical practice and drug trials,” they added.
The study was published online in JAMA Neurology to coincide with a presentation at the International Conference on Alzheimer’s and Parkinson’s Diseases in Gothenburg, Sweden.
Skewed tau levels
Plasma biomarkers of amyloid-beta (Abeta) and tau pathologies, and in particular variants in p-tau217 and p-tau181, have shown promise for use in Alzheimer’s disease diagnosis and prognosis. However, previous reports have suggested that chronic kidney disease might influence plasma p-tau217 and p-tau181 concentrations, potentially decreasing their usefulness in the diagnostic workup of dementia.
Researchers investigated associations of chronic kidney disease with plasma ratios of p-tau217 and p-tau181 to the corresponding unphosphorylated peptides in Alzheimer’s disease.
The data came from 473 older adults, with and without cognitive impairment, from the Swedish BioFinder-2 study who had plasma tau assessments and chronic kidney disease status established within 6 months of plasma collection.
The researchers found that lower estimated glomerular filtration rate levels (indicative of kidney dysfunction) were associated with higher plasma concentrations of phosphorylated and unphosphorylated tau peptides measured simultaneously using the tau immunoprecipitation mass spectrometry assay.
However, the correlations with estimated glomerular filtration rate were nonsignificant for the pT217/T217 ratio in individuals with cognitive impairment and were significantly attenuated for pT217/T217 in cognitively unimpaired individuals and for pT181/T181 in both cognitively unimpaired and impaired individuals.
“Importantly, we demonstrate that there were no significant associations between chronic kidney disease and the pT217/T217 ratio and changes in plasma pT181/T181 associated with chronic kidney disease were small or nonsignificant,” the researchers noted.
“Our results indicate that by using p-tau/tau ratios we may be able to reduce the variability in plasma p-tau levels driven by impaired kidney function and consequently such ratios are more robust measures of brain p-tau pathology in individuals with both early- and later-stage Alzheimer’s disease,” they added.
The researchers believe this is likely true for the ratios of other related proteins – a view that is supported by findings of attenuated associations of chronic kidney disease with Abeta42/40, compared with Abeta42 and Abeta40 in the current study and in previous studies.
Important clinical implications
Reached for comment, Rebecca Edelmayer, PhD, Alzheimer’s Association senior director of scientific engagement, noted that research continues to indicate that blood biomarkers have “promise for improving, and possibly even redefining, the clinical workup for Alzheimer’s disease.
“Many of the Alzheimer’s blood tests in development today have focused on core blood biomarkers associated with amyloid accumulation and tau tangle formation in the brain,” Dr. Edelmayer said.
“Before these tests can be used more broadly in the clinic, we need to understand all of the variables that may impact the results of various blood biomarkers, including differences that may be driven by race, ethnicity, sex, and underlying health conditions, such as chronic kidney disease.
“This study corroborates other research suggesting that some Alzheimer’s-associated markers can be affected by chronic kidney disease, but by using ratios of amyloid or tau markers, we may be able to minimize these differences in results caused by underlying disease,” Dr. Edelmayer said.
Howard Fillit, MD, cofounder and chief science officer at the Alzheimer’s Drug Discovery Foundation, said, “Using these ratios makes a lot of sense because the ratios wouldn’t change with kidney function. I think this is an important advance in clinical utilization of these tests.”
Dr. Fillit noted that older people often have declining kidney function, which can be easily measured by glomerular filtration rate. Changes in blood levels of these markers with declining kidney function are “not unexpected, but [it’s] important that it’s been demonstrated.
“As we go forward, maybe the future utilization of these tests will be not only recording the ratios but also reporting the ratios in the context of somebody’s glomerular filtration rate. You could imagine a scenario where when the test is done, it’s automatically done alongside of glomerular filtration rate,” Dr. Fillit said in an interview.
The study was supported by Coins for Alzheimer’s Research Trust, the Tracy Family Stable Isotope Labeling Quantitation Center, and the National Institute of Neurological Disorders and Stroke. Dr. Hansson has received support to his institution from ADx, AVID Radiopharmaceuticals, Biogen, Eli Lilly, Eisai, Fujirebio, GE Healthcare, Pfizer, and Roche; consultancy/speaker fees from Amylyx, Alzpath, Biogen, Cerveau, Fujirebio, Genentech, Novartis, Roche, and Siemens; and personal fees from Eli Lilly, Eisai, Bioarctic, and Biogen outside the submitted work. Dr. Edelmayer and Dr. Fillit have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AD/PD 2023
Magnesium-rich diet linked to lower dementia risk
Investigators studied more than 6,000 cognitively healthy individuals, aged 40-73, and found that those who consumed more than 550 mg of magnesium daily had a brain age approximately 1 year younger by age 55 years, compared with a person who consumed a normal magnesium intake (~360 mg per day).
“This research highlights the potential benefits of a diet high in magnesium and the role it plays in promoting good brain health,” lead author Khawlah Alateeq, a PhD candidate in neuroscience at Australian National University’s National Centre for Epidemiology and Population Health, said in an interview.
Clinicians “can use [the findings] to counsel patients on the benefits of increasing magnesium intake through a healthy diet and monitoring magnesium levels to prevent deficiencies,” she stated.
The study was published online in the European Journal of Nutrition.
Promising target
The researchers were motivated to conduct the study because of “the growing concern over the increasing prevalence of dementia,” Ms. Alateeq said.
“Since there is no cure for dementia, and the development of pharmacological treatment for dementia has been unsuccessful over the last 30 years, prevention has been suggested as an effective approach to address the issue,” she added.
Nutrition, Ms. Alateeq said, is a “modifiable risk factor that can influence brain health and is highly amenable to scalable and cost-effective interventions.” It represents “a promising target” for risk reduction at a population level.
Previous research shows individuals with lower magnesium levels are at higher risk for AD, while those with higher dietary magnesium intake may be at lower risk of progressing from normal aging to cognitive impairment.
Most previous studies, however, included participants older than age 60 years, and it’s “unclear when the neuroprotective effects of dietary magnesium become detectable,” the researchers note.
Moreover, dietary patterns change and fluctuate, potentially leading to changes in magnesium intake over time. These changes may have as much impact as absolute magnesium at any point in time.
In light of the “current lack of understanding of when and to what extent dietary magnesium exerts its protective effects on the brain,” the researchers examined the association between magnesium trajectories over time, brain matter, and white matter lesions.
They also examined the association between magnesium and several different blood pressure measures (mean arterial pressure, systolic blood pressure, diastolic blood pressure, and pulse pressure).
Since cardiovascular health, neurodegeneration, and brain shrinkage patterns differ between men and women, the researchers stratified their analyses by sex.
Brain volume differences
The researchers analyzed the dietary magnesium intake of 6,001 individuals (mean age, 55.3 years) selected from the UK Biobank – a prospective cohort study of participants aged 37-73 at baseline, who were assessed between 2005 and 2023.
For the current study, only participants with baseline DBP and SBP measurements and structural MRI scans were included. Participants were also required to be free of neurologic disorders and to have an available record of dietary magnesium intake.
Covariates included age, sex, education, health conditions, smoking status, body mass index, amount of physical activity, smoking status, and alcohol intake.
Over a 16-month period, participants completed an online questionnaire five times. Their responses were used to calculate daily magnesium intake. Foods of particular interest included leafy green vegetables, legumes, nuts, seeds, and whole grains, all of which are magnesium rich.
They used latent class analysis (LCA) to “identify mutually exclusive subgroup (classes) of magnesium intake trajectory separately for men and women.”
Men had a slightly higher prevalence of BP medication and diabetes, compared with women, and postmenopausal women had a higher prevalence of BP medication and diabetes, compared with premenopausal women.
Compared with lower baseline magnesium intake, higher baseline dietary intake of magnesium was associated with larger brain volumes in several regions in both men and women.
The latent class analysis identified three classes of magnesium intake:
In women in particular, the “high-decreasing” trajectory was significantly associated with larger brain volumes, compared with the “normal-stable” trajectory, while the “low-increasing” trajectory was associated with smaller brain volumes.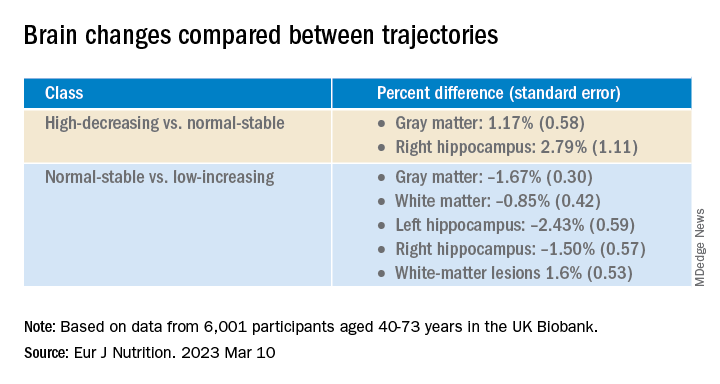
Even an increase of 1 mg of magnesium per day (above 350 mg/day) made a difference in brain volume, especially in women. The changes associated with every 1-mg increase are found in the table below: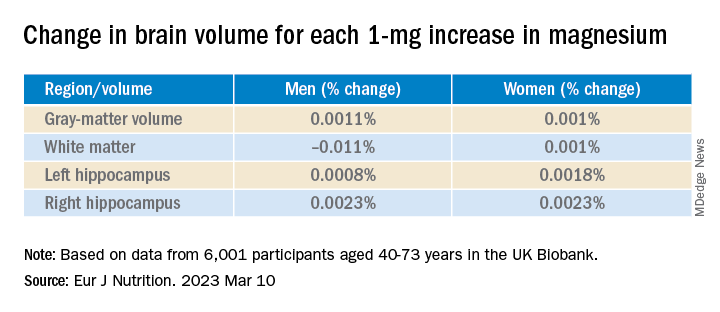
Associations between magnesium and BP measures were “mostly nonsignificant,” the researchers say, and the neuroprotective effect of higher magnesium intake in the high-decreasing trajectory was greater in postmenopausal versus premenopausal women.
“Our models indicate that compared to somebody with a normal magnesium intake (~350 mg per day), somebody in the top quartile of magnesium intake (≥ 550 mg per day) would be predicted to have a ~0.20% larger GM and ~0.46% larger RHC,” the authors summarize.
“In a population with an average age of 55 years, this effect corresponds to ~1 year of typical aging,” they note. “In other words, if this effect is generalizable to other populations, a 41% increase in magnesium intake may lead to significantly better brain health.”
Although the exact mechanisms underlying magnesium’s protective effects are “not yet clearly understood, there’s considerable evidence that magnesium levels are related to better cardiovascular health. Magnesium supplementation has been found to decrease blood pressure – and high blood pressure is a well-established risk factor for dementia,” said Ms. Alateeq.
Association, not causation
Yuko Hara, PhD, director of Aging and Prevention, Alzheimer’s Drug Discovery Foundation, noted that the study is observational and therefore shows an association, not causation.
“People eating a high-magnesium diet may also be eating a brain-healthy diet and getting high levels of nutrients/minerals other than magnesium alone,” suggested Dr. Hara, who was not involved with the study.
She noted that many foods are good sources of magnesium, including spinach, almonds, cashews, legumes, yogurt, brown rice, and avocados.
“Eating a brain-healthy diet (for example, the Mediterranean diet) is one of the Seven Steps to Protect Your Cognitive Vitality that ADDF’s Cognitive Vitality promotes,” she said.
Open Access funding was enabled and organized by the Council of Australian University Librarians and its Member Institutions. Ms. Alateeq, her co-authors, and Dr. Hara declare no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Investigators studied more than 6,000 cognitively healthy individuals, aged 40-73, and found that those who consumed more than 550 mg of magnesium daily had a brain age approximately 1 year younger by age 55 years, compared with a person who consumed a normal magnesium intake (~360 mg per day).
“This research highlights the potential benefits of a diet high in magnesium and the role it plays in promoting good brain health,” lead author Khawlah Alateeq, a PhD candidate in neuroscience at Australian National University’s National Centre for Epidemiology and Population Health, said in an interview.
Clinicians “can use [the findings] to counsel patients on the benefits of increasing magnesium intake through a healthy diet and monitoring magnesium levels to prevent deficiencies,” she stated.
The study was published online in the European Journal of Nutrition.
Promising target
The researchers were motivated to conduct the study because of “the growing concern over the increasing prevalence of dementia,” Ms. Alateeq said.
“Since there is no cure for dementia, and the development of pharmacological treatment for dementia has been unsuccessful over the last 30 years, prevention has been suggested as an effective approach to address the issue,” she added.
Nutrition, Ms. Alateeq said, is a “modifiable risk factor that can influence brain health and is highly amenable to scalable and cost-effective interventions.” It represents “a promising target” for risk reduction at a population level.
Previous research shows individuals with lower magnesium levels are at higher risk for AD, while those with higher dietary magnesium intake may be at lower risk of progressing from normal aging to cognitive impairment.
Most previous studies, however, included participants older than age 60 years, and it’s “unclear when the neuroprotective effects of dietary magnesium become detectable,” the researchers note.
Moreover, dietary patterns change and fluctuate, potentially leading to changes in magnesium intake over time. These changes may have as much impact as absolute magnesium at any point in time.
In light of the “current lack of understanding of when and to what extent dietary magnesium exerts its protective effects on the brain,” the researchers examined the association between magnesium trajectories over time, brain matter, and white matter lesions.
They also examined the association between magnesium and several different blood pressure measures (mean arterial pressure, systolic blood pressure, diastolic blood pressure, and pulse pressure).
Since cardiovascular health, neurodegeneration, and brain shrinkage patterns differ between men and women, the researchers stratified their analyses by sex.
Brain volume differences
The researchers analyzed the dietary magnesium intake of 6,001 individuals (mean age, 55.3 years) selected from the UK Biobank – a prospective cohort study of participants aged 37-73 at baseline, who were assessed between 2005 and 2023.
For the current study, only participants with baseline DBP and SBP measurements and structural MRI scans were included. Participants were also required to be free of neurologic disorders and to have an available record of dietary magnesium intake.
Covariates included age, sex, education, health conditions, smoking status, body mass index, amount of physical activity, smoking status, and alcohol intake.
Over a 16-month period, participants completed an online questionnaire five times. Their responses were used to calculate daily magnesium intake. Foods of particular interest included leafy green vegetables, legumes, nuts, seeds, and whole grains, all of which are magnesium rich.
They used latent class analysis (LCA) to “identify mutually exclusive subgroup (classes) of magnesium intake trajectory separately for men and women.”
Men had a slightly higher prevalence of BP medication and diabetes, compared with women, and postmenopausal women had a higher prevalence of BP medication and diabetes, compared with premenopausal women.
Compared with lower baseline magnesium intake, higher baseline dietary intake of magnesium was associated with larger brain volumes in several regions in both men and women.
The latent class analysis identified three classes of magnesium intake:
In women in particular, the “high-decreasing” trajectory was significantly associated with larger brain volumes, compared with the “normal-stable” trajectory, while the “low-increasing” trajectory was associated with smaller brain volumes.
Even an increase of 1 mg of magnesium per day (above 350 mg/day) made a difference in brain volume, especially in women. The changes associated with every 1-mg increase are found in the table below:
Associations between magnesium and BP measures were “mostly nonsignificant,” the researchers say, and the neuroprotective effect of higher magnesium intake in the high-decreasing trajectory was greater in postmenopausal versus premenopausal women.
“Our models indicate that compared to somebody with a normal magnesium intake (~350 mg per day), somebody in the top quartile of magnesium intake (≥ 550 mg per day) would be predicted to have a ~0.20% larger GM and ~0.46% larger RHC,” the authors summarize.
“In a population with an average age of 55 years, this effect corresponds to ~1 year of typical aging,” they note. “In other words, if this effect is generalizable to other populations, a 41% increase in magnesium intake may lead to significantly better brain health.”
Although the exact mechanisms underlying magnesium’s protective effects are “not yet clearly understood, there’s considerable evidence that magnesium levels are related to better cardiovascular health. Magnesium supplementation has been found to decrease blood pressure – and high blood pressure is a well-established risk factor for dementia,” said Ms. Alateeq.
Association, not causation
Yuko Hara, PhD, director of Aging and Prevention, Alzheimer’s Drug Discovery Foundation, noted that the study is observational and therefore shows an association, not causation.
“People eating a high-magnesium diet may also be eating a brain-healthy diet and getting high levels of nutrients/minerals other than magnesium alone,” suggested Dr. Hara, who was not involved with the study.
She noted that many foods are good sources of magnesium, including spinach, almonds, cashews, legumes, yogurt, brown rice, and avocados.
“Eating a brain-healthy diet (for example, the Mediterranean diet) is one of the Seven Steps to Protect Your Cognitive Vitality that ADDF’s Cognitive Vitality promotes,” she said.
Open Access funding was enabled and organized by the Council of Australian University Librarians and its Member Institutions. Ms. Alateeq, her co-authors, and Dr. Hara declare no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Investigators studied more than 6,000 cognitively healthy individuals, aged 40-73, and found that those who consumed more than 550 mg of magnesium daily had a brain age approximately 1 year younger by age 55 years, compared with a person who consumed a normal magnesium intake (~360 mg per day).
“This research highlights the potential benefits of a diet high in magnesium and the role it plays in promoting good brain health,” lead author Khawlah Alateeq, a PhD candidate in neuroscience at Australian National University’s National Centre for Epidemiology and Population Health, said in an interview.
Clinicians “can use [the findings] to counsel patients on the benefits of increasing magnesium intake through a healthy diet and monitoring magnesium levels to prevent deficiencies,” she stated.
The study was published online in the European Journal of Nutrition.
Promising target
The researchers were motivated to conduct the study because of “the growing concern over the increasing prevalence of dementia,” Ms. Alateeq said.
“Since there is no cure for dementia, and the development of pharmacological treatment for dementia has been unsuccessful over the last 30 years, prevention has been suggested as an effective approach to address the issue,” she added.
Nutrition, Ms. Alateeq said, is a “modifiable risk factor that can influence brain health and is highly amenable to scalable and cost-effective interventions.” It represents “a promising target” for risk reduction at a population level.
Previous research shows individuals with lower magnesium levels are at higher risk for AD, while those with higher dietary magnesium intake may be at lower risk of progressing from normal aging to cognitive impairment.
Most previous studies, however, included participants older than age 60 years, and it’s “unclear when the neuroprotective effects of dietary magnesium become detectable,” the researchers note.
Moreover, dietary patterns change and fluctuate, potentially leading to changes in magnesium intake over time. These changes may have as much impact as absolute magnesium at any point in time.
In light of the “current lack of understanding of when and to what extent dietary magnesium exerts its protective effects on the brain,” the researchers examined the association between magnesium trajectories over time, brain matter, and white matter lesions.
They also examined the association between magnesium and several different blood pressure measures (mean arterial pressure, systolic blood pressure, diastolic blood pressure, and pulse pressure).
Since cardiovascular health, neurodegeneration, and brain shrinkage patterns differ between men and women, the researchers stratified their analyses by sex.
Brain volume differences
The researchers analyzed the dietary magnesium intake of 6,001 individuals (mean age, 55.3 years) selected from the UK Biobank – a prospective cohort study of participants aged 37-73 at baseline, who were assessed between 2005 and 2023.
For the current study, only participants with baseline DBP and SBP measurements and structural MRI scans were included. Participants were also required to be free of neurologic disorders and to have an available record of dietary magnesium intake.
Covariates included age, sex, education, health conditions, smoking status, body mass index, amount of physical activity, smoking status, and alcohol intake.
Over a 16-month period, participants completed an online questionnaire five times. Their responses were used to calculate daily magnesium intake. Foods of particular interest included leafy green vegetables, legumes, nuts, seeds, and whole grains, all of which are magnesium rich.
They used latent class analysis (LCA) to “identify mutually exclusive subgroup (classes) of magnesium intake trajectory separately for men and women.”
Men had a slightly higher prevalence of BP medication and diabetes, compared with women, and postmenopausal women had a higher prevalence of BP medication and diabetes, compared with premenopausal women.
Compared with lower baseline magnesium intake, higher baseline dietary intake of magnesium was associated with larger brain volumes in several regions in both men and women.
The latent class analysis identified three classes of magnesium intake:
In women in particular, the “high-decreasing” trajectory was significantly associated with larger brain volumes, compared with the “normal-stable” trajectory, while the “low-increasing” trajectory was associated with smaller brain volumes.
Even an increase of 1 mg of magnesium per day (above 350 mg/day) made a difference in brain volume, especially in women. The changes associated with every 1-mg increase are found in the table below:
Associations between magnesium and BP measures were “mostly nonsignificant,” the researchers say, and the neuroprotective effect of higher magnesium intake in the high-decreasing trajectory was greater in postmenopausal versus premenopausal women.
“Our models indicate that compared to somebody with a normal magnesium intake (~350 mg per day), somebody in the top quartile of magnesium intake (≥ 550 mg per day) would be predicted to have a ~0.20% larger GM and ~0.46% larger RHC,” the authors summarize.
“In a population with an average age of 55 years, this effect corresponds to ~1 year of typical aging,” they note. “In other words, if this effect is generalizable to other populations, a 41% increase in magnesium intake may lead to significantly better brain health.”
Although the exact mechanisms underlying magnesium’s protective effects are “not yet clearly understood, there’s considerable evidence that magnesium levels are related to better cardiovascular health. Magnesium supplementation has been found to decrease blood pressure – and high blood pressure is a well-established risk factor for dementia,” said Ms. Alateeq.
Association, not causation
Yuko Hara, PhD, director of Aging and Prevention, Alzheimer’s Drug Discovery Foundation, noted that the study is observational and therefore shows an association, not causation.
“People eating a high-magnesium diet may also be eating a brain-healthy diet and getting high levels of nutrients/minerals other than magnesium alone,” suggested Dr. Hara, who was not involved with the study.
She noted that many foods are good sources of magnesium, including spinach, almonds, cashews, legumes, yogurt, brown rice, and avocados.
“Eating a brain-healthy diet (for example, the Mediterranean diet) is one of the Seven Steps to Protect Your Cognitive Vitality that ADDF’s Cognitive Vitality promotes,” she said.
Open Access funding was enabled and organized by the Council of Australian University Librarians and its Member Institutions. Ms. Alateeq, her co-authors, and Dr. Hara declare no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM EUROPEAN JOURNAL OF NUTRITION
Specific brain damage links hypertension to cognitive impairment
Researchers have identified specific regions of the brain that appear to be damaged by high blood pressure. The finding may explain the link between hypertension and cognitive impairment.
They used genetic information from genome-wide association studies (GWASs) and MRI scans of the brain to study the relationship between hypertension, changes in brain structures, and cognitive impairment. Using Mendelian randomization techniques, they identified nine brain structures related to cognitive impairment that are affected by blood pressure.
“We knew before that raised blood pressure was related to changes in the brain, but our research has narrowed down the changes to those that appear to be potentially causally related to cognitive impairment,” senior author Tomasz Guzik, professor of cardiovascular medicine, at the University of Edinburgh and of the Jagiellonian University, Krakow, Poland, told this news organization.
“Our study confirms a potentially causal relationship between raised blood pressure and cognitive impairment, emphasizing the importance of preventing and treating hypertension,” Prof. Guzik noted.
“But it also identifies the brain culprits of this relationship,” he added.
In the future, it may be possible to assess these nine brain structures in people with high blood pressure to identify those at increased risk of developing cognitive impairment, he said. “These patients may need more intensive care for their blood pressure. We can also investigate these brain structures for potential signaling pathways and molecular changes to see if we can find new targets for treatment to prevent cognitive impairment.”
For this report, the investigators married together different research datasets to identify brain structures potentially responsible for the effects of blood pressure on cognitive function, using results from previous GWASs and observational data from 39,000 people in the UK Biobank registry for whom brain MRI data were available.
First, they mapped brain structures potentially influenced by blood pressure in midlife using MRI scans from people in the UK Biobank registry. Then they examined the relationship between blood pressure and cognitive function in the UK Biobank registry. Next, of the brain structures affected by blood pressure, they identified those that are causally linked to cognitive impairment.
This was possible thanks to genetic markers coding for increased blood pressure, brain structure imaging phenotypes, and those coding for cognitive impairment that could be used in Mendelian randomization studies.
“We looked at 3935 brain magnetic resonance imaging–derived phenotypes in the brain and cognitive function defined by fluid intelligence score to identify genetically predicted causal relationships,” Prof. Guzik said.
They identified 200 brain structures that were causally affected by systolic blood pressure. Of these, nine were also causally related to cognitive impairment. The results were validated in a second prospective cohort of patients with hypertension.
“Some of these structures, including putamen and the white matter regions spanning between the anterior corona radiata, anterior thalamic radiation, and anterior limb of the internal capsule, may represent the target brain regions at which systolic blood pressure acts on cognitive function,” the authors comment.
In an accompanying editorial, Ernesto Schiffrin, MD, and James Engert, PhD, McGill University, Montreal, say that further mechanistic studies of the effects of blood pressure on cognitive function are required to determine precise causal pathways and the roles of relevant brain regions.
“Eventually, biomarkers could be developed to inform antihypertensive trials. Whether clinical trials targeting the specific brain structures will be feasible or if specific antihypertensives could be found that target specific structures remains to be demonstrated,” they write.
“Thus, these new studies could lead to an understanding of the signaling pathways that explain how these structures relate vascular damage to cognitive impairment in hypertension, and contribute to the development of novel interventions to more successfully address the scourge of cognitive decline and dementia in the future,” the editorialists conclude.
The study was funded by the European Research Council, the British Heart Foundation, and the Italian Ministry of Health.
A version of this article first appeared on Medscape.com.
Researchers have identified specific regions of the brain that appear to be damaged by high blood pressure. The finding may explain the link between hypertension and cognitive impairment.
They used genetic information from genome-wide association studies (GWASs) and MRI scans of the brain to study the relationship between hypertension, changes in brain structures, and cognitive impairment. Using Mendelian randomization techniques, they identified nine brain structures related to cognitive impairment that are affected by blood pressure.
“We knew before that raised blood pressure was related to changes in the brain, but our research has narrowed down the changes to those that appear to be potentially causally related to cognitive impairment,” senior author Tomasz Guzik, professor of cardiovascular medicine, at the University of Edinburgh and of the Jagiellonian University, Krakow, Poland, told this news organization.
“Our study confirms a potentially causal relationship between raised blood pressure and cognitive impairment, emphasizing the importance of preventing and treating hypertension,” Prof. Guzik noted.
“But it also identifies the brain culprits of this relationship,” he added.
In the future, it may be possible to assess these nine brain structures in people with high blood pressure to identify those at increased risk of developing cognitive impairment, he said. “These patients may need more intensive care for their blood pressure. We can also investigate these brain structures for potential signaling pathways and molecular changes to see if we can find new targets for treatment to prevent cognitive impairment.”
For this report, the investigators married together different research datasets to identify brain structures potentially responsible for the effects of blood pressure on cognitive function, using results from previous GWASs and observational data from 39,000 people in the UK Biobank registry for whom brain MRI data were available.
First, they mapped brain structures potentially influenced by blood pressure in midlife using MRI scans from people in the UK Biobank registry. Then they examined the relationship between blood pressure and cognitive function in the UK Biobank registry. Next, of the brain structures affected by blood pressure, they identified those that are causally linked to cognitive impairment.
This was possible thanks to genetic markers coding for increased blood pressure, brain structure imaging phenotypes, and those coding for cognitive impairment that could be used in Mendelian randomization studies.
“We looked at 3935 brain magnetic resonance imaging–derived phenotypes in the brain and cognitive function defined by fluid intelligence score to identify genetically predicted causal relationships,” Prof. Guzik said.
They identified 200 brain structures that were causally affected by systolic blood pressure. Of these, nine were also causally related to cognitive impairment. The results were validated in a second prospective cohort of patients with hypertension.
“Some of these structures, including putamen and the white matter regions spanning between the anterior corona radiata, anterior thalamic radiation, and anterior limb of the internal capsule, may represent the target brain regions at which systolic blood pressure acts on cognitive function,” the authors comment.
In an accompanying editorial, Ernesto Schiffrin, MD, and James Engert, PhD, McGill University, Montreal, say that further mechanistic studies of the effects of blood pressure on cognitive function are required to determine precise causal pathways and the roles of relevant brain regions.
“Eventually, biomarkers could be developed to inform antihypertensive trials. Whether clinical trials targeting the specific brain structures will be feasible or if specific antihypertensives could be found that target specific structures remains to be demonstrated,” they write.
“Thus, these new studies could lead to an understanding of the signaling pathways that explain how these structures relate vascular damage to cognitive impairment in hypertension, and contribute to the development of novel interventions to more successfully address the scourge of cognitive decline and dementia in the future,” the editorialists conclude.
The study was funded by the European Research Council, the British Heart Foundation, and the Italian Ministry of Health.
A version of this article first appeared on Medscape.com.
Researchers have identified specific regions of the brain that appear to be damaged by high blood pressure. The finding may explain the link between hypertension and cognitive impairment.
They used genetic information from genome-wide association studies (GWASs) and MRI scans of the brain to study the relationship between hypertension, changes in brain structures, and cognitive impairment. Using Mendelian randomization techniques, they identified nine brain structures related to cognitive impairment that are affected by blood pressure.
“We knew before that raised blood pressure was related to changes in the brain, but our research has narrowed down the changes to those that appear to be potentially causally related to cognitive impairment,” senior author Tomasz Guzik, professor of cardiovascular medicine, at the University of Edinburgh and of the Jagiellonian University, Krakow, Poland, told this news organization.
“Our study confirms a potentially causal relationship between raised blood pressure and cognitive impairment, emphasizing the importance of preventing and treating hypertension,” Prof. Guzik noted.
“But it also identifies the brain culprits of this relationship,” he added.
In the future, it may be possible to assess these nine brain structures in people with high blood pressure to identify those at increased risk of developing cognitive impairment, he said. “These patients may need more intensive care for their blood pressure. We can also investigate these brain structures for potential signaling pathways and molecular changes to see if we can find new targets for treatment to prevent cognitive impairment.”
For this report, the investigators married together different research datasets to identify brain structures potentially responsible for the effects of blood pressure on cognitive function, using results from previous GWASs and observational data from 39,000 people in the UK Biobank registry for whom brain MRI data were available.
First, they mapped brain structures potentially influenced by blood pressure in midlife using MRI scans from people in the UK Biobank registry. Then they examined the relationship between blood pressure and cognitive function in the UK Biobank registry. Next, of the brain structures affected by blood pressure, they identified those that are causally linked to cognitive impairment.
This was possible thanks to genetic markers coding for increased blood pressure, brain structure imaging phenotypes, and those coding for cognitive impairment that could be used in Mendelian randomization studies.
“We looked at 3935 brain magnetic resonance imaging–derived phenotypes in the brain and cognitive function defined by fluid intelligence score to identify genetically predicted causal relationships,” Prof. Guzik said.
They identified 200 brain structures that were causally affected by systolic blood pressure. Of these, nine were also causally related to cognitive impairment. The results were validated in a second prospective cohort of patients with hypertension.
“Some of these structures, including putamen and the white matter regions spanning between the anterior corona radiata, anterior thalamic radiation, and anterior limb of the internal capsule, may represent the target brain regions at which systolic blood pressure acts on cognitive function,” the authors comment.
In an accompanying editorial, Ernesto Schiffrin, MD, and James Engert, PhD, McGill University, Montreal, say that further mechanistic studies of the effects of blood pressure on cognitive function are required to determine precise causal pathways and the roles of relevant brain regions.
“Eventually, biomarkers could be developed to inform antihypertensive trials. Whether clinical trials targeting the specific brain structures will be feasible or if specific antihypertensives could be found that target specific structures remains to be demonstrated,” they write.
“Thus, these new studies could lead to an understanding of the signaling pathways that explain how these structures relate vascular damage to cognitive impairment in hypertension, and contribute to the development of novel interventions to more successfully address the scourge of cognitive decline and dementia in the future,” the editorialists conclude.
The study was funded by the European Research Council, the British Heart Foundation, and the Italian Ministry of Health.
A version of this article first appeared on Medscape.com.