User login
Angiography in patients with prior CABG does better when planned with CT
BOSTON – Coronary angiography in patients who have previously undergone cardiac artery bypass grafting (CABG) is challenging, but the procedure can be streamlined and made safer when preprocedural CT coronary angiography (CTCA) is performed to plan the intervention, according to a randomized controlled trial.
In this study, all three endpoints, including a reduction in the incidence of contrast-induced nephropathy (CIN) and duration of the procedure, were met, according to Daniel Jones, MBBS, PhD.
Preprocedural CTCA was also associated with about a 40% improvement in patient satisfaction.
“When logistically possible, CTCA should be considered for any stable postbypass patient undergoing coronary angiography,” said Dr. Jones, who supported this assertion with data presented at the Transcatheter Cardiovascular Therapeutics annual meeting.
In this study, called BYPASS-CTCA, 688 patients with a prior CABG scheduled for invasive coronary angiography were randomized to a preprocedural CTCA or no preprocedural CTCA. Patients with stable angina and those with a non–ST elevated acute coronary syndrome were eligible. Those with ST-segment elevated MI or severe renal impairment (eGFR < 20 mL/min) were excluded.
All three co–primary endpoints favor CTCA
CTCA relative to no CTCA provided a significant advantage for all three of the coprimary endpoints, which were procedure duration, CIN as defined by KDIGO criteria, and patient satisfaction as measured by questionnaire.
The procedure duration was reduced by almost 21 minutes, cutting the time from nearly 39 minutes to less than 18 minutes (P < .001). This relative reduction was of similar magnitude across groups, such as those with or without acute coronary syndrome and procedures performed by a senior or a junior operator.
“Even when you include the preprocedural CTCA evaluation time, there was still a significant reduction [P < .001] in duration for those in the CTCA arm,” reported Dr. Jones, honorary consultant cardiologist, Barts Heart Centre, Queen Mary University, London.
The rates of CIN following the procedure in this study, which had a follow-up of 12 months, were 3.4% versus 27.9% (P < .0001) in the preprocedural CTCA and non-CTCA groups, respectively. Again, a sensitivity analysis showed a similar magnitude of risk reduction across all subgroups evaluated.
CTCA planning reduced contrast exposure
The reduced risk of CIN was consistent with a large reduction in contrast exposure for those in the CTCA group (77.4 vs. 173.0 mL; P < .001). The advantage narrowed substantially when adding in contrast exposure from CTCA, but still remained statistically significant (148.9 vs. 173.0 mL; P < .001).
Dr. Jones did not speculate about the specific reasons for the 40% improvement in patient satisfaction among those who underwent preprocedural CTCA relative to those who did not, but, again, a sensitivity analysis showed consistency across subgroups defined by race, operator experience, and underlying diagnosis.
Numerous secondary endpoints also favored CTCA over no CTCA. This included fewer catheters used to complete the procedure (three vs. four; P < .001), a greater likelihood that the procedure was performed with radial access (76.9% vs. 56.7%), and lower rates of procedural complications (2.3% vs. 10.8%; P < .001). This latter category included fewer vascular access complications such as bleeding (0.6 % vs. 4.4%; P = .007) and periprocedural MI (0.6% vs. 6.4%; P < 0.001).
In a graph of time to first major adverse cardiovascular event (MACE), the curves separated almost immediately with a consistently lower rate maintained in the CTCA arm over the 12 months of follow-up, but this is observational. Dr. Jones acknowledged that this trial was not powered to show a difference in MACE.
Study intriguing but not definitive
In a panel discussion that followed the presentation of these results at the meeting, sponsored by the Cardiovascular Research Foundation, some reservations with this study were expressed. In particular, several of the panelists, including Jeffrey W. Moses, MD, director of interventional cardiovascular therapeutics, Columbia University Medical Center, New York, expressed surprise at the 27% rate of CIN, which he considered uncommonly high even in a high-risk population.
The unusual rate of CIN was also considered problematic given that it was the most significant clinical outcome among the three co–primary endpoints. Procedural times and patient satisfaction, while valid endpoints, are important subjects of study, but Dr. Moses was not alone in suggesting this study deserves validation.
In particular, there appeared to be a consensus among panelists that a larger multicenter study looking at hard endpoints, such as MACE, would be more compelling. They indicated that even if CTCA poses a very low risk of meaningful complications, it does add expense and an extra step.
Dr. Jones reported no potential conflicts of interest. Dr. Moses reported financial relationships with Covanos, Orchestra Biomed, Ostial, and Xenter.
BOSTON – Coronary angiography in patients who have previously undergone cardiac artery bypass grafting (CABG) is challenging, but the procedure can be streamlined and made safer when preprocedural CT coronary angiography (CTCA) is performed to plan the intervention, according to a randomized controlled trial.
In this study, all three endpoints, including a reduction in the incidence of contrast-induced nephropathy (CIN) and duration of the procedure, were met, according to Daniel Jones, MBBS, PhD.
Preprocedural CTCA was also associated with about a 40% improvement in patient satisfaction.
“When logistically possible, CTCA should be considered for any stable postbypass patient undergoing coronary angiography,” said Dr. Jones, who supported this assertion with data presented at the Transcatheter Cardiovascular Therapeutics annual meeting.
In this study, called BYPASS-CTCA, 688 patients with a prior CABG scheduled for invasive coronary angiography were randomized to a preprocedural CTCA or no preprocedural CTCA. Patients with stable angina and those with a non–ST elevated acute coronary syndrome were eligible. Those with ST-segment elevated MI or severe renal impairment (eGFR < 20 mL/min) were excluded.
All three co–primary endpoints favor CTCA
CTCA relative to no CTCA provided a significant advantage for all three of the coprimary endpoints, which were procedure duration, CIN as defined by KDIGO criteria, and patient satisfaction as measured by questionnaire.
The procedure duration was reduced by almost 21 minutes, cutting the time from nearly 39 minutes to less than 18 minutes (P < .001). This relative reduction was of similar magnitude across groups, such as those with or without acute coronary syndrome and procedures performed by a senior or a junior operator.
“Even when you include the preprocedural CTCA evaluation time, there was still a significant reduction [P < .001] in duration for those in the CTCA arm,” reported Dr. Jones, honorary consultant cardiologist, Barts Heart Centre, Queen Mary University, London.
The rates of CIN following the procedure in this study, which had a follow-up of 12 months, were 3.4% versus 27.9% (P < .0001) in the preprocedural CTCA and non-CTCA groups, respectively. Again, a sensitivity analysis showed a similar magnitude of risk reduction across all subgroups evaluated.
CTCA planning reduced contrast exposure
The reduced risk of CIN was consistent with a large reduction in contrast exposure for those in the CTCA group (77.4 vs. 173.0 mL; P < .001). The advantage narrowed substantially when adding in contrast exposure from CTCA, but still remained statistically significant (148.9 vs. 173.0 mL; P < .001).
Dr. Jones did not speculate about the specific reasons for the 40% improvement in patient satisfaction among those who underwent preprocedural CTCA relative to those who did not, but, again, a sensitivity analysis showed consistency across subgroups defined by race, operator experience, and underlying diagnosis.
Numerous secondary endpoints also favored CTCA over no CTCA. This included fewer catheters used to complete the procedure (three vs. four; P < .001), a greater likelihood that the procedure was performed with radial access (76.9% vs. 56.7%), and lower rates of procedural complications (2.3% vs. 10.8%; P < .001). This latter category included fewer vascular access complications such as bleeding (0.6 % vs. 4.4%; P = .007) and periprocedural MI (0.6% vs. 6.4%; P < 0.001).
In a graph of time to first major adverse cardiovascular event (MACE), the curves separated almost immediately with a consistently lower rate maintained in the CTCA arm over the 12 months of follow-up, but this is observational. Dr. Jones acknowledged that this trial was not powered to show a difference in MACE.
Study intriguing but not definitive
In a panel discussion that followed the presentation of these results at the meeting, sponsored by the Cardiovascular Research Foundation, some reservations with this study were expressed. In particular, several of the panelists, including Jeffrey W. Moses, MD, director of interventional cardiovascular therapeutics, Columbia University Medical Center, New York, expressed surprise at the 27% rate of CIN, which he considered uncommonly high even in a high-risk population.
The unusual rate of CIN was also considered problematic given that it was the most significant clinical outcome among the three co–primary endpoints. Procedural times and patient satisfaction, while valid endpoints, are important subjects of study, but Dr. Moses was not alone in suggesting this study deserves validation.
In particular, there appeared to be a consensus among panelists that a larger multicenter study looking at hard endpoints, such as MACE, would be more compelling. They indicated that even if CTCA poses a very low risk of meaningful complications, it does add expense and an extra step.
Dr. Jones reported no potential conflicts of interest. Dr. Moses reported financial relationships with Covanos, Orchestra Biomed, Ostial, and Xenter.
BOSTON – Coronary angiography in patients who have previously undergone cardiac artery bypass grafting (CABG) is challenging, but the procedure can be streamlined and made safer when preprocedural CT coronary angiography (CTCA) is performed to plan the intervention, according to a randomized controlled trial.
In this study, all three endpoints, including a reduction in the incidence of contrast-induced nephropathy (CIN) and duration of the procedure, were met, according to Daniel Jones, MBBS, PhD.
Preprocedural CTCA was also associated with about a 40% improvement in patient satisfaction.
“When logistically possible, CTCA should be considered for any stable postbypass patient undergoing coronary angiography,” said Dr. Jones, who supported this assertion with data presented at the Transcatheter Cardiovascular Therapeutics annual meeting.
In this study, called BYPASS-CTCA, 688 patients with a prior CABG scheduled for invasive coronary angiography were randomized to a preprocedural CTCA or no preprocedural CTCA. Patients with stable angina and those with a non–ST elevated acute coronary syndrome were eligible. Those with ST-segment elevated MI or severe renal impairment (eGFR < 20 mL/min) were excluded.
All three co–primary endpoints favor CTCA
CTCA relative to no CTCA provided a significant advantage for all three of the coprimary endpoints, which were procedure duration, CIN as defined by KDIGO criteria, and patient satisfaction as measured by questionnaire.
The procedure duration was reduced by almost 21 minutes, cutting the time from nearly 39 minutes to less than 18 minutes (P < .001). This relative reduction was of similar magnitude across groups, such as those with or without acute coronary syndrome and procedures performed by a senior or a junior operator.
“Even when you include the preprocedural CTCA evaluation time, there was still a significant reduction [P < .001] in duration for those in the CTCA arm,” reported Dr. Jones, honorary consultant cardiologist, Barts Heart Centre, Queen Mary University, London.
The rates of CIN following the procedure in this study, which had a follow-up of 12 months, were 3.4% versus 27.9% (P < .0001) in the preprocedural CTCA and non-CTCA groups, respectively. Again, a sensitivity analysis showed a similar magnitude of risk reduction across all subgroups evaluated.
CTCA planning reduced contrast exposure
The reduced risk of CIN was consistent with a large reduction in contrast exposure for those in the CTCA group (77.4 vs. 173.0 mL; P < .001). The advantage narrowed substantially when adding in contrast exposure from CTCA, but still remained statistically significant (148.9 vs. 173.0 mL; P < .001).
Dr. Jones did not speculate about the specific reasons for the 40% improvement in patient satisfaction among those who underwent preprocedural CTCA relative to those who did not, but, again, a sensitivity analysis showed consistency across subgroups defined by race, operator experience, and underlying diagnosis.
Numerous secondary endpoints also favored CTCA over no CTCA. This included fewer catheters used to complete the procedure (three vs. four; P < .001), a greater likelihood that the procedure was performed with radial access (76.9% vs. 56.7%), and lower rates of procedural complications (2.3% vs. 10.8%; P < .001). This latter category included fewer vascular access complications such as bleeding (0.6 % vs. 4.4%; P = .007) and periprocedural MI (0.6% vs. 6.4%; P < 0.001).
In a graph of time to first major adverse cardiovascular event (MACE), the curves separated almost immediately with a consistently lower rate maintained in the CTCA arm over the 12 months of follow-up, but this is observational. Dr. Jones acknowledged that this trial was not powered to show a difference in MACE.
Study intriguing but not definitive
In a panel discussion that followed the presentation of these results at the meeting, sponsored by the Cardiovascular Research Foundation, some reservations with this study were expressed. In particular, several of the panelists, including Jeffrey W. Moses, MD, director of interventional cardiovascular therapeutics, Columbia University Medical Center, New York, expressed surprise at the 27% rate of CIN, which he considered uncommonly high even in a high-risk population.
The unusual rate of CIN was also considered problematic given that it was the most significant clinical outcome among the three co–primary endpoints. Procedural times and patient satisfaction, while valid endpoints, are important subjects of study, but Dr. Moses was not alone in suggesting this study deserves validation.
In particular, there appeared to be a consensus among panelists that a larger multicenter study looking at hard endpoints, such as MACE, would be more compelling. They indicated that even if CTCA poses a very low risk of meaningful complications, it does add expense and an extra step.
Dr. Jones reported no potential conflicts of interest. Dr. Moses reported financial relationships with Covanos, Orchestra Biomed, Ostial, and Xenter.
AT TCT 2022
PASCAL for MV repair noninferior to MitraClip in CLASP IID; FDA took notice
A newly available transcatheter device for edge-to-edge mitral valve (MV) repair, named for a famed scientist-inventor, is similar to the long-available MitraClip (Abbott) for short-term efficacy and safety, suggests an interim but prespecified analysis from a randomized trial.
In its comparison with MitraClip, the PASCAL transcatheter valve repair system (Edwards Lifesciences) was noninferior with respect to 30-day major adverse events and to success at achieving mitral regurgitation (MR) of no more than moderate severity within 6 months. The trial had entered patients with significant, symptomatic degenerative MR considered too high-risk for surgical repair or replacement.
The interim analysis covers 180 of the 300 patients followed in the study, of whom 117 received the PASCAL device and 63 were given MitraClip. Both groups showed significant gains in functional class, symptom status, and quality of life over 6 months, reported D. Scott Lim, MD, University of Virginia Health System Hospital, Charlottesville, and Konstantinos Koulogiannis, MD, Morristown Medical Center, N.J., jointly on Sept. 17 at the Transcatheter Cardiovascular Therapeutics (TCT) 2022 annual meeting in Boston.
Dr. Lim, one of the trial’s principal investigators, is also lead author on its same-day publication in JACC: Cardiovascular Interventions.
Based largely on those results from the CLASP IID pivotal trial, the U.S. Food and Drug Administration recently approved the PASCAL system for use in patients with degenerative MR, Edwards announced on Sept. 15. The device was approved in the European Union on Aug. 17.
MitraClip has been available in various iterations in the United States since 2013 and in Europe since 2008.
“It’s good for the field to be able to say we have two devices that are comparable,” giving clinicians more options, Vinod H. Thourani, MD, Piedmont Heart Institute, Atlanta, told this news organization.
The current analysis shows that “we’ve yet to figure out what patient pathologies will be beneficial” for each of the devices, Dr. Thourani said. “The goal will be to find out if there are certain anatomical considerations where one device is better than the other.”
It will be necessary to study “more patients, a larger cohort, with longer follow-up to allow us to see their true benefits,” he said, as well as to conduct more subgroup analyses. For now, the choice of device will probably be “operator-specific, which they feel comfortable with.”
Dr. Thourani, not an author on the current study, is the U.S. principal investigator for the CLASP IIF study looking at clinical outcomes with the two devices and says he consults for both Edwards and Abbott.
The findings are “preliminary for now,” said Michael Young, MD, Dartmouth-Hitchcock Medical Center, Lebanon, N.H., in part because, like most randomized trials, CLASP IID entered a select, not broadly representative population.
“They want to make, as best as they could, an apples-to-apples comparison, without confounding that might make it more difficult to interpret it afterwards,” Dr. Young, not associated with the trial, told this news organization.
But CLASP IID “did enroll patients that we do see and treat, so undoubtedly it’s a compelling study. We now have another device that is shown to be safe and effective. How we’re going to extrapolate it to all the patients that are being referred to our practices will, I think, be under debate and deliberation.”
The PASCAL and MitraClip devices each may be more suitable for different patients with varying mitral valve pathologies because of differences in their designs, Dr. Lim said. The PASCAL’s relative flexibility might make it preferable in patients with smaller mitral valves, and its ability to elongate during delivery could make it more suitable for patients with chordal-dense areas around the valve, he speculated.
MitraClip, Dr. Lim told this news organization, has a mechanical closure system for anchoring that may make it more appropriate for “more complicated, thicker leaflets with calcium.”
CLASP IID enrolled patients with grade 3+ or 4+ degenerative MR considered to be “at prohibitive surgical risk” at 43 sites in North America and Europe. It randomly assigned them 2-to-1 to receive the PASCAL device or MitraClip.
Either of two PASCAL versions were used, the original device or the “smaller, narrower” PASCAL Ace, Dr. Lim observed. Both versions are covered by the PASCAL Precision System FDA approval. About 40% of patients assigned to MitraClip received older versions of the device and about 60%, more recent versions, as they were entered into practice.
The mean procedure times were 88 minutes for PASCAL and 79 minutes for MitraClip (P = .023), with much of the difference attributable to the earliest PASCAL procedures. Procedure times for the device declined with greater operator experience, the published report states.
Rates of the primary safety endpoint of major adverse events at 30 days were 3.4% for PASCAL and 4.8% for MitraClip. The endpoint was a composite of cardiovascular mortality, stroke, myocardial infarction, new need for renal replacement therapy, severe bleeding, or nonelective MV reintervention.
The proportion of patients with MR grade 2+ or lower at 6 months, the primary effectiveness endpoint, assessed at a core laboratory, was 96.5% for the PASCAL group over a median follow-up of 179.5 days and 96.8% over a median of 184.5 days for those who received MitraClip.
Comparisons for both primary endpoints met the prespecified criteria for PASCAL noninferiority.
In a secondary analysis, the proportion of PASCAL patients with MR grade 1+ or less held about steady from postprocedure discharge out to 6 months, at 87.2% and 83.7%, respectively (P = .317).
But whereas 88.5% of MitraClip patients had MR grade 1+ or better at discharge, 71.2% were at that grade by 6 months (P = .003). That apparent hemodynamic deterioration raised some eyebrows at the TCT sessions as a potential sign that PASCAL functional results are more durable.
That sort of judgment is premature, offered Anita W. Asgar, MD, MSc, Montreal Heart Institute, Quebec City, as an invited discussant after the CLASP IID trial’s formal presentation at the meeting, which was sponsored by the Cardiovascular Research Foundation.
The trial is notable in part for “showing how safe this procedure is and how successful it is for these patients – this is phenomenal,” she said, but “I would caution comparing one device being better than another with such a small number of patients.”
MitraClip, Dr. Young observed, “has been, up to this point, our only option for edge-to-edge repair of the mitral valve. And many of us have years of experience and a lot of patients that we treat with that device.” His center hasn’t yet used PASCAL, but that may change as the field gains more familiarity with the device. Operators may use either device in different cases, he said.
“Depending on the program, and depending on the volume of mitral patients that you see and edge-to-edge repair that you do, it could be that you stick with one, or switch to another, or you integrate both of them and try to decide which patients might be better suited for one or the other.”
CLASP IID was sponsored by Edwards Lifesciences. Dr. Lim discloses consulting for Philips, Venus, and Valgen and receiving research grants from Abbott, Boston Scientific, Edwards Lifesciences, and Medtronic. Dr. Koulogiannis discloses consulting and serving on an advisory board for Edwards Lifesciences and as a speaker for Abbott and discloses holding equity, stocks, or stock options in 4C. Dr. Thourani discloses serving as a consultant to both Abbott and Edwards Lifesciences. Dr. Young discloses receiving consulting fees or honoraria or serving on a speaker’s bureau for Medtronic. Dr. Asgar discloses receiving research support from or holding a research contract with Abbott Vascular and receiving consulting fees or honoraria or serving on a speaker’s bureau for Medtronic, Edwards Lifesciences, and W. Gore & Associates.
A version of this article first appeared on Medscape.com.
A newly available transcatheter device for edge-to-edge mitral valve (MV) repair, named for a famed scientist-inventor, is similar to the long-available MitraClip (Abbott) for short-term efficacy and safety, suggests an interim but prespecified analysis from a randomized trial.
In its comparison with MitraClip, the PASCAL transcatheter valve repair system (Edwards Lifesciences) was noninferior with respect to 30-day major adverse events and to success at achieving mitral regurgitation (MR) of no more than moderate severity within 6 months. The trial had entered patients with significant, symptomatic degenerative MR considered too high-risk for surgical repair or replacement.
The interim analysis covers 180 of the 300 patients followed in the study, of whom 117 received the PASCAL device and 63 were given MitraClip. Both groups showed significant gains in functional class, symptom status, and quality of life over 6 months, reported D. Scott Lim, MD, University of Virginia Health System Hospital, Charlottesville, and Konstantinos Koulogiannis, MD, Morristown Medical Center, N.J., jointly on Sept. 17 at the Transcatheter Cardiovascular Therapeutics (TCT) 2022 annual meeting in Boston.
Dr. Lim, one of the trial’s principal investigators, is also lead author on its same-day publication in JACC: Cardiovascular Interventions.
Based largely on those results from the CLASP IID pivotal trial, the U.S. Food and Drug Administration recently approved the PASCAL system for use in patients with degenerative MR, Edwards announced on Sept. 15. The device was approved in the European Union on Aug. 17.
MitraClip has been available in various iterations in the United States since 2013 and in Europe since 2008.
“It’s good for the field to be able to say we have two devices that are comparable,” giving clinicians more options, Vinod H. Thourani, MD, Piedmont Heart Institute, Atlanta, told this news organization.
The current analysis shows that “we’ve yet to figure out what patient pathologies will be beneficial” for each of the devices, Dr. Thourani said. “The goal will be to find out if there are certain anatomical considerations where one device is better than the other.”
It will be necessary to study “more patients, a larger cohort, with longer follow-up to allow us to see their true benefits,” he said, as well as to conduct more subgroup analyses. For now, the choice of device will probably be “operator-specific, which they feel comfortable with.”
Dr. Thourani, not an author on the current study, is the U.S. principal investigator for the CLASP IIF study looking at clinical outcomes with the two devices and says he consults for both Edwards and Abbott.
The findings are “preliminary for now,” said Michael Young, MD, Dartmouth-Hitchcock Medical Center, Lebanon, N.H., in part because, like most randomized trials, CLASP IID entered a select, not broadly representative population.
“They want to make, as best as they could, an apples-to-apples comparison, without confounding that might make it more difficult to interpret it afterwards,” Dr. Young, not associated with the trial, told this news organization.
But CLASP IID “did enroll patients that we do see and treat, so undoubtedly it’s a compelling study. We now have another device that is shown to be safe and effective. How we’re going to extrapolate it to all the patients that are being referred to our practices will, I think, be under debate and deliberation.”
The PASCAL and MitraClip devices each may be more suitable for different patients with varying mitral valve pathologies because of differences in their designs, Dr. Lim said. The PASCAL’s relative flexibility might make it preferable in patients with smaller mitral valves, and its ability to elongate during delivery could make it more suitable for patients with chordal-dense areas around the valve, he speculated.
MitraClip, Dr. Lim told this news organization, has a mechanical closure system for anchoring that may make it more appropriate for “more complicated, thicker leaflets with calcium.”
CLASP IID enrolled patients with grade 3+ or 4+ degenerative MR considered to be “at prohibitive surgical risk” at 43 sites in North America and Europe. It randomly assigned them 2-to-1 to receive the PASCAL device or MitraClip.
Either of two PASCAL versions were used, the original device or the “smaller, narrower” PASCAL Ace, Dr. Lim observed. Both versions are covered by the PASCAL Precision System FDA approval. About 40% of patients assigned to MitraClip received older versions of the device and about 60%, more recent versions, as they were entered into practice.
The mean procedure times were 88 minutes for PASCAL and 79 minutes for MitraClip (P = .023), with much of the difference attributable to the earliest PASCAL procedures. Procedure times for the device declined with greater operator experience, the published report states.
Rates of the primary safety endpoint of major adverse events at 30 days were 3.4% for PASCAL and 4.8% for MitraClip. The endpoint was a composite of cardiovascular mortality, stroke, myocardial infarction, new need for renal replacement therapy, severe bleeding, or nonelective MV reintervention.
The proportion of patients with MR grade 2+ or lower at 6 months, the primary effectiveness endpoint, assessed at a core laboratory, was 96.5% for the PASCAL group over a median follow-up of 179.5 days and 96.8% over a median of 184.5 days for those who received MitraClip.
Comparisons for both primary endpoints met the prespecified criteria for PASCAL noninferiority.
In a secondary analysis, the proportion of PASCAL patients with MR grade 1+ or less held about steady from postprocedure discharge out to 6 months, at 87.2% and 83.7%, respectively (P = .317).
But whereas 88.5% of MitraClip patients had MR grade 1+ or better at discharge, 71.2% were at that grade by 6 months (P = .003). That apparent hemodynamic deterioration raised some eyebrows at the TCT sessions as a potential sign that PASCAL functional results are more durable.
That sort of judgment is premature, offered Anita W. Asgar, MD, MSc, Montreal Heart Institute, Quebec City, as an invited discussant after the CLASP IID trial’s formal presentation at the meeting, which was sponsored by the Cardiovascular Research Foundation.
The trial is notable in part for “showing how safe this procedure is and how successful it is for these patients – this is phenomenal,” she said, but “I would caution comparing one device being better than another with such a small number of patients.”
MitraClip, Dr. Young observed, “has been, up to this point, our only option for edge-to-edge repair of the mitral valve. And many of us have years of experience and a lot of patients that we treat with that device.” His center hasn’t yet used PASCAL, but that may change as the field gains more familiarity with the device. Operators may use either device in different cases, he said.
“Depending on the program, and depending on the volume of mitral patients that you see and edge-to-edge repair that you do, it could be that you stick with one, or switch to another, or you integrate both of them and try to decide which patients might be better suited for one or the other.”
CLASP IID was sponsored by Edwards Lifesciences. Dr. Lim discloses consulting for Philips, Venus, and Valgen and receiving research grants from Abbott, Boston Scientific, Edwards Lifesciences, and Medtronic. Dr. Koulogiannis discloses consulting and serving on an advisory board for Edwards Lifesciences and as a speaker for Abbott and discloses holding equity, stocks, or stock options in 4C. Dr. Thourani discloses serving as a consultant to both Abbott and Edwards Lifesciences. Dr. Young discloses receiving consulting fees or honoraria or serving on a speaker’s bureau for Medtronic. Dr. Asgar discloses receiving research support from or holding a research contract with Abbott Vascular and receiving consulting fees or honoraria or serving on a speaker’s bureau for Medtronic, Edwards Lifesciences, and W. Gore & Associates.
A version of this article first appeared on Medscape.com.
A newly available transcatheter device for edge-to-edge mitral valve (MV) repair, named for a famed scientist-inventor, is similar to the long-available MitraClip (Abbott) for short-term efficacy and safety, suggests an interim but prespecified analysis from a randomized trial.
In its comparison with MitraClip, the PASCAL transcatheter valve repair system (Edwards Lifesciences) was noninferior with respect to 30-day major adverse events and to success at achieving mitral regurgitation (MR) of no more than moderate severity within 6 months. The trial had entered patients with significant, symptomatic degenerative MR considered too high-risk for surgical repair or replacement.
The interim analysis covers 180 of the 300 patients followed in the study, of whom 117 received the PASCAL device and 63 were given MitraClip. Both groups showed significant gains in functional class, symptom status, and quality of life over 6 months, reported D. Scott Lim, MD, University of Virginia Health System Hospital, Charlottesville, and Konstantinos Koulogiannis, MD, Morristown Medical Center, N.J., jointly on Sept. 17 at the Transcatheter Cardiovascular Therapeutics (TCT) 2022 annual meeting in Boston.
Dr. Lim, one of the trial’s principal investigators, is also lead author on its same-day publication in JACC: Cardiovascular Interventions.
Based largely on those results from the CLASP IID pivotal trial, the U.S. Food and Drug Administration recently approved the PASCAL system for use in patients with degenerative MR, Edwards announced on Sept. 15. The device was approved in the European Union on Aug. 17.
MitraClip has been available in various iterations in the United States since 2013 and in Europe since 2008.
“It’s good for the field to be able to say we have two devices that are comparable,” giving clinicians more options, Vinod H. Thourani, MD, Piedmont Heart Institute, Atlanta, told this news organization.
The current analysis shows that “we’ve yet to figure out what patient pathologies will be beneficial” for each of the devices, Dr. Thourani said. “The goal will be to find out if there are certain anatomical considerations where one device is better than the other.”
It will be necessary to study “more patients, a larger cohort, with longer follow-up to allow us to see their true benefits,” he said, as well as to conduct more subgroup analyses. For now, the choice of device will probably be “operator-specific, which they feel comfortable with.”
Dr. Thourani, not an author on the current study, is the U.S. principal investigator for the CLASP IIF study looking at clinical outcomes with the two devices and says he consults for both Edwards and Abbott.
The findings are “preliminary for now,” said Michael Young, MD, Dartmouth-Hitchcock Medical Center, Lebanon, N.H., in part because, like most randomized trials, CLASP IID entered a select, not broadly representative population.
“They want to make, as best as they could, an apples-to-apples comparison, without confounding that might make it more difficult to interpret it afterwards,” Dr. Young, not associated with the trial, told this news organization.
But CLASP IID “did enroll patients that we do see and treat, so undoubtedly it’s a compelling study. We now have another device that is shown to be safe and effective. How we’re going to extrapolate it to all the patients that are being referred to our practices will, I think, be under debate and deliberation.”
The PASCAL and MitraClip devices each may be more suitable for different patients with varying mitral valve pathologies because of differences in their designs, Dr. Lim said. The PASCAL’s relative flexibility might make it preferable in patients with smaller mitral valves, and its ability to elongate during delivery could make it more suitable for patients with chordal-dense areas around the valve, he speculated.
MitraClip, Dr. Lim told this news organization, has a mechanical closure system for anchoring that may make it more appropriate for “more complicated, thicker leaflets with calcium.”
CLASP IID enrolled patients with grade 3+ or 4+ degenerative MR considered to be “at prohibitive surgical risk” at 43 sites in North America and Europe. It randomly assigned them 2-to-1 to receive the PASCAL device or MitraClip.
Either of two PASCAL versions were used, the original device or the “smaller, narrower” PASCAL Ace, Dr. Lim observed. Both versions are covered by the PASCAL Precision System FDA approval. About 40% of patients assigned to MitraClip received older versions of the device and about 60%, more recent versions, as they were entered into practice.
The mean procedure times were 88 minutes for PASCAL and 79 minutes for MitraClip (P = .023), with much of the difference attributable to the earliest PASCAL procedures. Procedure times for the device declined with greater operator experience, the published report states.
Rates of the primary safety endpoint of major adverse events at 30 days were 3.4% for PASCAL and 4.8% for MitraClip. The endpoint was a composite of cardiovascular mortality, stroke, myocardial infarction, new need for renal replacement therapy, severe bleeding, or nonelective MV reintervention.
The proportion of patients with MR grade 2+ or lower at 6 months, the primary effectiveness endpoint, assessed at a core laboratory, was 96.5% for the PASCAL group over a median follow-up of 179.5 days and 96.8% over a median of 184.5 days for those who received MitraClip.
Comparisons for both primary endpoints met the prespecified criteria for PASCAL noninferiority.
In a secondary analysis, the proportion of PASCAL patients with MR grade 1+ or less held about steady from postprocedure discharge out to 6 months, at 87.2% and 83.7%, respectively (P = .317).
But whereas 88.5% of MitraClip patients had MR grade 1+ or better at discharge, 71.2% were at that grade by 6 months (P = .003). That apparent hemodynamic deterioration raised some eyebrows at the TCT sessions as a potential sign that PASCAL functional results are more durable.
That sort of judgment is premature, offered Anita W. Asgar, MD, MSc, Montreal Heart Institute, Quebec City, as an invited discussant after the CLASP IID trial’s formal presentation at the meeting, which was sponsored by the Cardiovascular Research Foundation.
The trial is notable in part for “showing how safe this procedure is and how successful it is for these patients – this is phenomenal,” she said, but “I would caution comparing one device being better than another with such a small number of patients.”
MitraClip, Dr. Young observed, “has been, up to this point, our only option for edge-to-edge repair of the mitral valve. And many of us have years of experience and a lot of patients that we treat with that device.” His center hasn’t yet used PASCAL, but that may change as the field gains more familiarity with the device. Operators may use either device in different cases, he said.
“Depending on the program, and depending on the volume of mitral patients that you see and edge-to-edge repair that you do, it could be that you stick with one, or switch to another, or you integrate both of them and try to decide which patients might be better suited for one or the other.”
CLASP IID was sponsored by Edwards Lifesciences. Dr. Lim discloses consulting for Philips, Venus, and Valgen and receiving research grants from Abbott, Boston Scientific, Edwards Lifesciences, and Medtronic. Dr. Koulogiannis discloses consulting and serving on an advisory board for Edwards Lifesciences and as a speaker for Abbott and discloses holding equity, stocks, or stock options in 4C. Dr. Thourani discloses serving as a consultant to both Abbott and Edwards Lifesciences. Dr. Young discloses receiving consulting fees or honoraria or serving on a speaker’s bureau for Medtronic. Dr. Asgar discloses receiving research support from or holding a research contract with Abbott Vascular and receiving consulting fees or honoraria or serving on a speaker’s bureau for Medtronic, Edwards Lifesciences, and W. Gore & Associates.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
A week of anticoagulation halves post-PCI radial occlusion rate
Serious bleeding is not increased
BOSTON – Following transradial access for angiography or a percutaneous coronary intervention (PCI), a low dose of the factor Xa inhibitor rivaroxaban for 7 days reduces the risk of an access-site occlusion by 50%, according to results of the randomized RIVARAD trial.
Of two multicenter, randomized trials to address this question it is the larger, according to Rania Hammami, MD, who presented the results at the Transcatheter Cardiovascular Therapeutics annual meeting.
In the open-label RIVARAD trial, 538 patients were randomized to 10 mg rivaroxaban or standard care alone. Standard care at the beginning of the procedure included unfractionated heparin in a dose of 50 IU/kg for angiography and up to 100 IU/kg for PCI. Manual compression was applied at the end of the procedure followed by an evaluation for complications, such as hematoma or aneurysm.
For the primary outcome of radial access occlusion at 30 days, the lower rate in the rivaroxaban arm (6.9% vs. 13.0%) translated into a statistically significant 50% reduction (odds ratio, 0.50; P = .011).
Rivaroxaban preserves radial pulse
Rivaroxaban was also favored for the endpoint of inability at 30 days to find a radial pulse (5.8% vs. 12.2%; P = .01). Interestingly, there was some disparity for this endpoint for clinical examination and ultrasound.
“In 12 patients, we were able to palpate a radial pulse, but the ultrasound showed an occlusion of the vessel, while in 7 patients we could not find a radial pulse even though the radial artery was patent on ultrasound,” Dr. Hammami, of the department of cardiology, Hedi Chaker Hospital, Sfax, Tunisia, said at the meeting, sponsored by the Cardiovascular Research Foundation.
The incidence of hemorrhagic complications was higher in the rivaroxaban group (2.7% vs. 1.9%), but the difference did not approach statistical significance (OR, 1.5; P = .54). Moreover, all of the bleeding complications were minor (Bleeding Academic Research Consortium level 1), and none of the bleeding complications were observed in patients receiving rivaroxaban alone. Rather, all patients with bleeding were taking one or more antiplatelet drugs along with rivaroxaban.
On univariate analysis, several baseline characteristics were associated with subsequent radial occlusion, including female sex (P = .02), current smoking (P = .03), renal failure (P = .004), and PCI for acute coronary syndrome (P = .02). On multivariate analysis, female sex (P = .001) and current smoking (P < .0001) became even stronger predictors of occlusion on a statistical basis, while a prior procedure involving radial access was also a significant predictor (P = .029).
“One woman out of two developed radial access occlusion if she had a history of smoking and had a history of a transradial puncture,” Dr. Hammami reported.
In a subgroup analysis, relative protection from radial artery occlusion from a 7-day course of rivaroxaban was particularly pronounced in those with diabetes, renal failure, or hypertension relative to those without these conditions, but the protective effect appeared to be about the same regardless of body mass index, age, sheath size, or current use of statins.
These findings are consistent with other studies evaluating the risk of radial access occlusion, according to Dr. Hammami. While different studies she cited reported incidences ranging from less than 1% to more than 30%, the risk has typically been highest in populations with increased susceptibility for thrombus formation, such as smokers and patients with diabetes.
Preventing radial artery occlusion has several benefits, not least of which is preserving this access point for future interventions, according to Dr. Hammami.
RIVARAD is the largest study to evaluate an anticoagulant for the prevention of radial artery occlusion, but it is not the first. Earlier in 2022, a Chinese trial called RESTORE was published in Circulation: Cardiovascular Interventions. In that placebo-controlled study of 382 patients, 7 days of 10 mg rivaroxaban was also linked to a significant reduction in radial artery occlusion at 30 days (3.8% vs. 11.5%; P = .011).
“We don’t know if a higher dose of rivaroxaban would be more effective and equally safe,” said Dr. Hammami, but added that a Canadian trial called CAPITAL RAPTOR will test this premise. In this trial, there is a planned enrollment of 1,800 patients who will be randomized to 15 mg rivaroxaban or standard treatment.
Occlusion risk appears underappreciated
The risk of radial artery occlusion might be underappreciated. According to data cited by Dr. Hammami, only about half of interventionalists in the United States and fewer than 10% outside of the United States routinely assess radial artery patency in conjunction with radial-access PCI. The data from this trial suggest that the risk can be substantially reduced, particularly in high-risk patients, with anticoagulant therapy.
Agreeing that this is a potentially avoidable complication, Roxanna Mehran, MD, director of interventional cardiovascular research and clinical trials, Icahn School of Medicine at Mount Sinai, New York, called the RIVARAD study “a clinically meaningful trial,” and valuable for identifying risk factors as well as for showing a treatment effect and acceptable safety from a short course of a factor Xa inhibitor.
“This is very important work,” said Dr. Mehran, who praised the quality of the study and the contribution it makes for considering how and when prophylaxis is needed.
Dr. Hammami reported no potential conflicts of interest. Dr. Mehran has financial relationships with more than 25 pharmaceutical companies but none with the sponsor of this trial, which was funded by Philadelphia Pharma, a drug company based in Tunisia.
Serious bleeding is not increased
Serious bleeding is not increased
BOSTON – Following transradial access for angiography or a percutaneous coronary intervention (PCI), a low dose of the factor Xa inhibitor rivaroxaban for 7 days reduces the risk of an access-site occlusion by 50%, according to results of the randomized RIVARAD trial.
Of two multicenter, randomized trials to address this question it is the larger, according to Rania Hammami, MD, who presented the results at the Transcatheter Cardiovascular Therapeutics annual meeting.
In the open-label RIVARAD trial, 538 patients were randomized to 10 mg rivaroxaban or standard care alone. Standard care at the beginning of the procedure included unfractionated heparin in a dose of 50 IU/kg for angiography and up to 100 IU/kg for PCI. Manual compression was applied at the end of the procedure followed by an evaluation for complications, such as hematoma or aneurysm.
For the primary outcome of radial access occlusion at 30 days, the lower rate in the rivaroxaban arm (6.9% vs. 13.0%) translated into a statistically significant 50% reduction (odds ratio, 0.50; P = .011).
Rivaroxaban preserves radial pulse
Rivaroxaban was also favored for the endpoint of inability at 30 days to find a radial pulse (5.8% vs. 12.2%; P = .01). Interestingly, there was some disparity for this endpoint for clinical examination and ultrasound.
“In 12 patients, we were able to palpate a radial pulse, but the ultrasound showed an occlusion of the vessel, while in 7 patients we could not find a radial pulse even though the radial artery was patent on ultrasound,” Dr. Hammami, of the department of cardiology, Hedi Chaker Hospital, Sfax, Tunisia, said at the meeting, sponsored by the Cardiovascular Research Foundation.
The incidence of hemorrhagic complications was higher in the rivaroxaban group (2.7% vs. 1.9%), but the difference did not approach statistical significance (OR, 1.5; P = .54). Moreover, all of the bleeding complications were minor (Bleeding Academic Research Consortium level 1), and none of the bleeding complications were observed in patients receiving rivaroxaban alone. Rather, all patients with bleeding were taking one or more antiplatelet drugs along with rivaroxaban.
On univariate analysis, several baseline characteristics were associated with subsequent radial occlusion, including female sex (P = .02), current smoking (P = .03), renal failure (P = .004), and PCI for acute coronary syndrome (P = .02). On multivariate analysis, female sex (P = .001) and current smoking (P < .0001) became even stronger predictors of occlusion on a statistical basis, while a prior procedure involving radial access was also a significant predictor (P = .029).
“One woman out of two developed radial access occlusion if she had a history of smoking and had a history of a transradial puncture,” Dr. Hammami reported.
In a subgroup analysis, relative protection from radial artery occlusion from a 7-day course of rivaroxaban was particularly pronounced in those with diabetes, renal failure, or hypertension relative to those without these conditions, but the protective effect appeared to be about the same regardless of body mass index, age, sheath size, or current use of statins.
These findings are consistent with other studies evaluating the risk of radial access occlusion, according to Dr. Hammami. While different studies she cited reported incidences ranging from less than 1% to more than 30%, the risk has typically been highest in populations with increased susceptibility for thrombus formation, such as smokers and patients with diabetes.
Preventing radial artery occlusion has several benefits, not least of which is preserving this access point for future interventions, according to Dr. Hammami.
RIVARAD is the largest study to evaluate an anticoagulant for the prevention of radial artery occlusion, but it is not the first. Earlier in 2022, a Chinese trial called RESTORE was published in Circulation: Cardiovascular Interventions. In that placebo-controlled study of 382 patients, 7 days of 10 mg rivaroxaban was also linked to a significant reduction in radial artery occlusion at 30 days (3.8% vs. 11.5%; P = .011).
“We don’t know if a higher dose of rivaroxaban would be more effective and equally safe,” said Dr. Hammami, but added that a Canadian trial called CAPITAL RAPTOR will test this premise. In this trial, there is a planned enrollment of 1,800 patients who will be randomized to 15 mg rivaroxaban or standard treatment.
Occlusion risk appears underappreciated
The risk of radial artery occlusion might be underappreciated. According to data cited by Dr. Hammami, only about half of interventionalists in the United States and fewer than 10% outside of the United States routinely assess radial artery patency in conjunction with radial-access PCI. The data from this trial suggest that the risk can be substantially reduced, particularly in high-risk patients, with anticoagulant therapy.
Agreeing that this is a potentially avoidable complication, Roxanna Mehran, MD, director of interventional cardiovascular research and clinical trials, Icahn School of Medicine at Mount Sinai, New York, called the RIVARAD study “a clinically meaningful trial,” and valuable for identifying risk factors as well as for showing a treatment effect and acceptable safety from a short course of a factor Xa inhibitor.
“This is very important work,” said Dr. Mehran, who praised the quality of the study and the contribution it makes for considering how and when prophylaxis is needed.
Dr. Hammami reported no potential conflicts of interest. Dr. Mehran has financial relationships with more than 25 pharmaceutical companies but none with the sponsor of this trial, which was funded by Philadelphia Pharma, a drug company based in Tunisia.
BOSTON – Following transradial access for angiography or a percutaneous coronary intervention (PCI), a low dose of the factor Xa inhibitor rivaroxaban for 7 days reduces the risk of an access-site occlusion by 50%, according to results of the randomized RIVARAD trial.
Of two multicenter, randomized trials to address this question it is the larger, according to Rania Hammami, MD, who presented the results at the Transcatheter Cardiovascular Therapeutics annual meeting.
In the open-label RIVARAD trial, 538 patients were randomized to 10 mg rivaroxaban or standard care alone. Standard care at the beginning of the procedure included unfractionated heparin in a dose of 50 IU/kg for angiography and up to 100 IU/kg for PCI. Manual compression was applied at the end of the procedure followed by an evaluation for complications, such as hematoma or aneurysm.
For the primary outcome of radial access occlusion at 30 days, the lower rate in the rivaroxaban arm (6.9% vs. 13.0%) translated into a statistically significant 50% reduction (odds ratio, 0.50; P = .011).
Rivaroxaban preserves radial pulse
Rivaroxaban was also favored for the endpoint of inability at 30 days to find a radial pulse (5.8% vs. 12.2%; P = .01). Interestingly, there was some disparity for this endpoint for clinical examination and ultrasound.
“In 12 patients, we were able to palpate a radial pulse, but the ultrasound showed an occlusion of the vessel, while in 7 patients we could not find a radial pulse even though the radial artery was patent on ultrasound,” Dr. Hammami, of the department of cardiology, Hedi Chaker Hospital, Sfax, Tunisia, said at the meeting, sponsored by the Cardiovascular Research Foundation.
The incidence of hemorrhagic complications was higher in the rivaroxaban group (2.7% vs. 1.9%), but the difference did not approach statistical significance (OR, 1.5; P = .54). Moreover, all of the bleeding complications were minor (Bleeding Academic Research Consortium level 1), and none of the bleeding complications were observed in patients receiving rivaroxaban alone. Rather, all patients with bleeding were taking one or more antiplatelet drugs along with rivaroxaban.
On univariate analysis, several baseline characteristics were associated with subsequent radial occlusion, including female sex (P = .02), current smoking (P = .03), renal failure (P = .004), and PCI for acute coronary syndrome (P = .02). On multivariate analysis, female sex (P = .001) and current smoking (P < .0001) became even stronger predictors of occlusion on a statistical basis, while a prior procedure involving radial access was also a significant predictor (P = .029).
“One woman out of two developed radial access occlusion if she had a history of smoking and had a history of a transradial puncture,” Dr. Hammami reported.
In a subgroup analysis, relative protection from radial artery occlusion from a 7-day course of rivaroxaban was particularly pronounced in those with diabetes, renal failure, or hypertension relative to those without these conditions, but the protective effect appeared to be about the same regardless of body mass index, age, sheath size, or current use of statins.
These findings are consistent with other studies evaluating the risk of radial access occlusion, according to Dr. Hammami. While different studies she cited reported incidences ranging from less than 1% to more than 30%, the risk has typically been highest in populations with increased susceptibility for thrombus formation, such as smokers and patients with diabetes.
Preventing radial artery occlusion has several benefits, not least of which is preserving this access point for future interventions, according to Dr. Hammami.
RIVARAD is the largest study to evaluate an anticoagulant for the prevention of radial artery occlusion, but it is not the first. Earlier in 2022, a Chinese trial called RESTORE was published in Circulation: Cardiovascular Interventions. In that placebo-controlled study of 382 patients, 7 days of 10 mg rivaroxaban was also linked to a significant reduction in radial artery occlusion at 30 days (3.8% vs. 11.5%; P = .011).
“We don’t know if a higher dose of rivaroxaban would be more effective and equally safe,” said Dr. Hammami, but added that a Canadian trial called CAPITAL RAPTOR will test this premise. In this trial, there is a planned enrollment of 1,800 patients who will be randomized to 15 mg rivaroxaban or standard treatment.
Occlusion risk appears underappreciated
The risk of radial artery occlusion might be underappreciated. According to data cited by Dr. Hammami, only about half of interventionalists in the United States and fewer than 10% outside of the United States routinely assess radial artery patency in conjunction with radial-access PCI. The data from this trial suggest that the risk can be substantially reduced, particularly in high-risk patients, with anticoagulant therapy.
Agreeing that this is a potentially avoidable complication, Roxanna Mehran, MD, director of interventional cardiovascular research and clinical trials, Icahn School of Medicine at Mount Sinai, New York, called the RIVARAD study “a clinically meaningful trial,” and valuable for identifying risk factors as well as for showing a treatment effect and acceptable safety from a short course of a factor Xa inhibitor.
“This is very important work,” said Dr. Mehran, who praised the quality of the study and the contribution it makes for considering how and when prophylaxis is needed.
Dr. Hammami reported no potential conflicts of interest. Dr. Mehran has financial relationships with more than 25 pharmaceutical companies but none with the sponsor of this trial, which was funded by Philadelphia Pharma, a drug company based in Tunisia.
AT TCT 2022
Ultrasonic renal denervation passes 2-month test in uncontrolled HTN: RADIANCE II
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.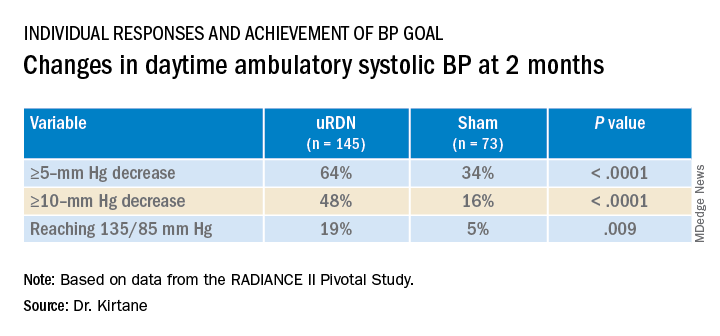
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).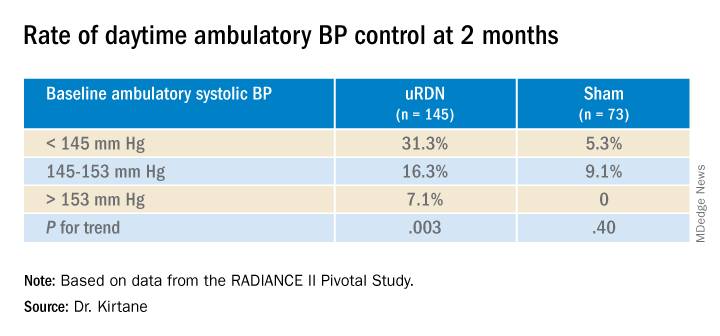
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
TAVR now used in almost 50% of younger severe aortic stenosis patients
Among patients with severe isolated aortic stenosis younger than 65, the rate of transcatheter aortic valve replacement (TAVR) now almost matches that of surgical aortic valve replacement (SAVR), despite guideline recommendations to the contrary, a study in a national U.S. population shows.
The 2020 American Heart Association/American College of Cardiology (AHA/ACC) valve guideline recommends SAVR for patients younger than 65 with severe aortic stenosis, the researchers note, but their study showed “near equal utilization between TAVR and SAVR in these younger patients by 2021,” at 48% and 52% respectively.
Toishi Sharma, MD, and colleagues presented these findings in an oral poster session at Transcatheter Cardiovascular Therapeutics 2022, and the study was simultaneously published as a Research Letter in the Journal of the American College of Cardiology (JACC).
“To our knowledge, the current findings represent the first national temporal trends study stratifying [aortic stenosis] therapies according to guideline-recommended age groups: our observations demonstrate the dramatic growth of TAVR in all age groups, including young patients,” the researchers conclude.
They analyzed changes in rates of TAVR and SAVR in a U.S. sample stratified by age: younger than 65 years, 65-80, and older than 80 years.
These findings have implications for lifetime management of younger patients who undergo TAVR, they write, “including issues related to lifetime coronary access, valve durability, and the potential for subsequent TAVR procedures over time.”
Three age groups
In a study published in JACC, this group examined changes in uptake of TAVR versus SAVR in 4,161 patients with aortic stenosis in Vermont, New Hampshire, and Maine, senior author Harold L. Dauerman, MD, said in an interview.
The greatest rate of rise of TAVR was in the group younger than 65, but that study ended in 2019, said Dr. Dauerman, from the University of Vermont Health Network, Burlington.
The 2020 guideline stratifies TAVR and SAVR recommendations such that “less than 65 should primarily be a surgical approach and greater than 80 primarily a TAVR approach, while 65 to 80 is a gray zone, and shared decision-making becomes important,” he noted.
The group hypothesized that recent trials and technology have led to a national increase in TAVR in people younger than 65.
From the Vizient clinical database, including more than 250 U.S. academic centers that perform both TAVR and SAVR, the researchers identified 142,953 patients who underwent TAVR or SAVR for isolated aortic stenosis from Oct. 1, 2015, to Dec. 31, 2021. From 2015 to 2021, the valve replacement rates in the three age groups changed as follows:
- Age less than 65: TAVR rose from 17% to 48%; SAVR fell from 83% to 52%.
- Age 65-80: TAVR rose from 46% to 87%; SAVR fell from 54% to 12%.
- Age greater than 80: TAVR rose from 83% to 99%; SAVR fell from 16% to 1%.
“All ages have grown in the last 7 years in TAVR,” Dr. Dauerman summarized. “The one that’s surprising, and in contradiction to the guideline, is the growth of TAVR in young patients less than 65.”
Among patients younger than 65, prior bypass surgery and congestive heart failure predicted the use of TAVR instead of surgery, whereas bicuspid aortic valve disease was the biggest predictor of surgery instead of TAVR.
Most studies on TAVR valve durability are limited to patients in the randomized trials who were primarily in their mid-70s to mid-80s, some of whom died before a 10-year follow-up, Dr. Dauerman noted.
European guidelines recommend surgery for patients younger than 70, and it would be interesting to see if clinicians there follow this recommendation or if TAVR is now the preferred approach, he added.
There is a need for further, longer study of TAVR in younger patients, he said, to determine whether there are long-term clinical issues of concern.
Strategy depends on more than age
The “findings are not too surprising,” John Carroll, MD, who was not involved in this research, said in an email.
“Age is only one of multiple patient characteristics that enter into consideration of TAVR versus SAVR,” said Dr. Carroll, from Anschutz Medical Campus, University of Colorado, Aurora.
“As the article reports,” he noted, “those less than 65 having TAVR are more likely to have comorbid conditions that likely made the risk of SAVR higher.”
Dr. Carroll was lead author of a review article published in 2020 based on data from the ACC–Society of Thoracic Surgeons (STS)–Transcatheter Valve Therapy (TVT) registry on 276,316 patients who had TAVR in the United States from 2011-2019.
He pointed out that Figure 2 in that review shows that “SAVR is often performed in conjunction with other surgical procedures – another major reason why SAVR remains an important treatment for valvular heart disease.”
“We are awaiting long-term data comparing TAVR to SAVR durability,” Dr. Carroll added, echoing Dr. Dauerman. “So far [there are] no major differences, but it remains a key need to fully understand TAVR and the various models in commercial use.”
“Both TAVR and SAVR used in adults are tissue valves (SAVR with mechanical valves is used in younger patients),” Dr. Carroll noted, “and all tissue valves will eventually fail if the patient lives long enough.”
Patient management strategies need to consider what treatment options exist when the first valve fails. “If the first valve is SAVR, there is now extensive experience with placing a TAVR valve inside a failing SAVR valve, so called Valve-in-Valve or TAVR-in-SAVR. This is the preferred treatment in most patients with failing SAVR valves,” he said.
“On the other hand,” he continued, “we are just beginning to see more and more patients with failing TAVR valves, and the TAVR-in-TAVR procedure is less well understood.”
“Issues such as acute coronary occlusion and long-term difficulty in accessing coronary arteries are being encountered in some patients having TAVR-in-TAVR,” Dr. Carroll noted, which he discusses in a recent editorial he coauthored about the complexities of redo TAVR, published in JACC: Cardiovascular Interventions.
The study received no funding. Dr. Dauerman has research grants and is a consultant for Medtronic and Boston Scientific. Dr. Carroll is a local principal investigator in trials sponsored by Medtronic, Abbott, and Edwards Lifesciences.
A version of this article first appeared on Medscape.com.
Among patients with severe isolated aortic stenosis younger than 65, the rate of transcatheter aortic valve replacement (TAVR) now almost matches that of surgical aortic valve replacement (SAVR), despite guideline recommendations to the contrary, a study in a national U.S. population shows.
The 2020 American Heart Association/American College of Cardiology (AHA/ACC) valve guideline recommends SAVR for patients younger than 65 with severe aortic stenosis, the researchers note, but their study showed “near equal utilization between TAVR and SAVR in these younger patients by 2021,” at 48% and 52% respectively.
Toishi Sharma, MD, and colleagues presented these findings in an oral poster session at Transcatheter Cardiovascular Therapeutics 2022, and the study was simultaneously published as a Research Letter in the Journal of the American College of Cardiology (JACC).
“To our knowledge, the current findings represent the first national temporal trends study stratifying [aortic stenosis] therapies according to guideline-recommended age groups: our observations demonstrate the dramatic growth of TAVR in all age groups, including young patients,” the researchers conclude.
They analyzed changes in rates of TAVR and SAVR in a U.S. sample stratified by age: younger than 65 years, 65-80, and older than 80 years.
These findings have implications for lifetime management of younger patients who undergo TAVR, they write, “including issues related to lifetime coronary access, valve durability, and the potential for subsequent TAVR procedures over time.”
Three age groups
In a study published in JACC, this group examined changes in uptake of TAVR versus SAVR in 4,161 patients with aortic stenosis in Vermont, New Hampshire, and Maine, senior author Harold L. Dauerman, MD, said in an interview.
The greatest rate of rise of TAVR was in the group younger than 65, but that study ended in 2019, said Dr. Dauerman, from the University of Vermont Health Network, Burlington.
The 2020 guideline stratifies TAVR and SAVR recommendations such that “less than 65 should primarily be a surgical approach and greater than 80 primarily a TAVR approach, while 65 to 80 is a gray zone, and shared decision-making becomes important,” he noted.
The group hypothesized that recent trials and technology have led to a national increase in TAVR in people younger than 65.
From the Vizient clinical database, including more than 250 U.S. academic centers that perform both TAVR and SAVR, the researchers identified 142,953 patients who underwent TAVR or SAVR for isolated aortic stenosis from Oct. 1, 2015, to Dec. 31, 2021. From 2015 to 2021, the valve replacement rates in the three age groups changed as follows:
- Age less than 65: TAVR rose from 17% to 48%; SAVR fell from 83% to 52%.
- Age 65-80: TAVR rose from 46% to 87%; SAVR fell from 54% to 12%.
- Age greater than 80: TAVR rose from 83% to 99%; SAVR fell from 16% to 1%.
“All ages have grown in the last 7 years in TAVR,” Dr. Dauerman summarized. “The one that’s surprising, and in contradiction to the guideline, is the growth of TAVR in young patients less than 65.”
Among patients younger than 65, prior bypass surgery and congestive heart failure predicted the use of TAVR instead of surgery, whereas bicuspid aortic valve disease was the biggest predictor of surgery instead of TAVR.
Most studies on TAVR valve durability are limited to patients in the randomized trials who were primarily in their mid-70s to mid-80s, some of whom died before a 10-year follow-up, Dr. Dauerman noted.
European guidelines recommend surgery for patients younger than 70, and it would be interesting to see if clinicians there follow this recommendation or if TAVR is now the preferred approach, he added.
There is a need for further, longer study of TAVR in younger patients, he said, to determine whether there are long-term clinical issues of concern.
Strategy depends on more than age
The “findings are not too surprising,” John Carroll, MD, who was not involved in this research, said in an email.
“Age is only one of multiple patient characteristics that enter into consideration of TAVR versus SAVR,” said Dr. Carroll, from Anschutz Medical Campus, University of Colorado, Aurora.
“As the article reports,” he noted, “those less than 65 having TAVR are more likely to have comorbid conditions that likely made the risk of SAVR higher.”
Dr. Carroll was lead author of a review article published in 2020 based on data from the ACC–Society of Thoracic Surgeons (STS)–Transcatheter Valve Therapy (TVT) registry on 276,316 patients who had TAVR in the United States from 2011-2019.
He pointed out that Figure 2 in that review shows that “SAVR is often performed in conjunction with other surgical procedures – another major reason why SAVR remains an important treatment for valvular heart disease.”
“We are awaiting long-term data comparing TAVR to SAVR durability,” Dr. Carroll added, echoing Dr. Dauerman. “So far [there are] no major differences, but it remains a key need to fully understand TAVR and the various models in commercial use.”
“Both TAVR and SAVR used in adults are tissue valves (SAVR with mechanical valves is used in younger patients),” Dr. Carroll noted, “and all tissue valves will eventually fail if the patient lives long enough.”
Patient management strategies need to consider what treatment options exist when the first valve fails. “If the first valve is SAVR, there is now extensive experience with placing a TAVR valve inside a failing SAVR valve, so called Valve-in-Valve or TAVR-in-SAVR. This is the preferred treatment in most patients with failing SAVR valves,” he said.
“On the other hand,” he continued, “we are just beginning to see more and more patients with failing TAVR valves, and the TAVR-in-TAVR procedure is less well understood.”
“Issues such as acute coronary occlusion and long-term difficulty in accessing coronary arteries are being encountered in some patients having TAVR-in-TAVR,” Dr. Carroll noted, which he discusses in a recent editorial he coauthored about the complexities of redo TAVR, published in JACC: Cardiovascular Interventions.
The study received no funding. Dr. Dauerman has research grants and is a consultant for Medtronic and Boston Scientific. Dr. Carroll is a local principal investigator in trials sponsored by Medtronic, Abbott, and Edwards Lifesciences.
A version of this article first appeared on Medscape.com.
Among patients with severe isolated aortic stenosis younger than 65, the rate of transcatheter aortic valve replacement (TAVR) now almost matches that of surgical aortic valve replacement (SAVR), despite guideline recommendations to the contrary, a study in a national U.S. population shows.
The 2020 American Heart Association/American College of Cardiology (AHA/ACC) valve guideline recommends SAVR for patients younger than 65 with severe aortic stenosis, the researchers note, but their study showed “near equal utilization between TAVR and SAVR in these younger patients by 2021,” at 48% and 52% respectively.
Toishi Sharma, MD, and colleagues presented these findings in an oral poster session at Transcatheter Cardiovascular Therapeutics 2022, and the study was simultaneously published as a Research Letter in the Journal of the American College of Cardiology (JACC).
“To our knowledge, the current findings represent the first national temporal trends study stratifying [aortic stenosis] therapies according to guideline-recommended age groups: our observations demonstrate the dramatic growth of TAVR in all age groups, including young patients,” the researchers conclude.
They analyzed changes in rates of TAVR and SAVR in a U.S. sample stratified by age: younger than 65 years, 65-80, and older than 80 years.
These findings have implications for lifetime management of younger patients who undergo TAVR, they write, “including issues related to lifetime coronary access, valve durability, and the potential for subsequent TAVR procedures over time.”
Three age groups
In a study published in JACC, this group examined changes in uptake of TAVR versus SAVR in 4,161 patients with aortic stenosis in Vermont, New Hampshire, and Maine, senior author Harold L. Dauerman, MD, said in an interview.
The greatest rate of rise of TAVR was in the group younger than 65, but that study ended in 2019, said Dr. Dauerman, from the University of Vermont Health Network, Burlington.
The 2020 guideline stratifies TAVR and SAVR recommendations such that “less than 65 should primarily be a surgical approach and greater than 80 primarily a TAVR approach, while 65 to 80 is a gray zone, and shared decision-making becomes important,” he noted.
The group hypothesized that recent trials and technology have led to a national increase in TAVR in people younger than 65.
From the Vizient clinical database, including more than 250 U.S. academic centers that perform both TAVR and SAVR, the researchers identified 142,953 patients who underwent TAVR or SAVR for isolated aortic stenosis from Oct. 1, 2015, to Dec. 31, 2021. From 2015 to 2021, the valve replacement rates in the three age groups changed as follows:
- Age less than 65: TAVR rose from 17% to 48%; SAVR fell from 83% to 52%.
- Age 65-80: TAVR rose from 46% to 87%; SAVR fell from 54% to 12%.
- Age greater than 80: TAVR rose from 83% to 99%; SAVR fell from 16% to 1%.
“All ages have grown in the last 7 years in TAVR,” Dr. Dauerman summarized. “The one that’s surprising, and in contradiction to the guideline, is the growth of TAVR in young patients less than 65.”
Among patients younger than 65, prior bypass surgery and congestive heart failure predicted the use of TAVR instead of surgery, whereas bicuspid aortic valve disease was the biggest predictor of surgery instead of TAVR.
Most studies on TAVR valve durability are limited to patients in the randomized trials who were primarily in their mid-70s to mid-80s, some of whom died before a 10-year follow-up, Dr. Dauerman noted.
European guidelines recommend surgery for patients younger than 70, and it would be interesting to see if clinicians there follow this recommendation or if TAVR is now the preferred approach, he added.
There is a need for further, longer study of TAVR in younger patients, he said, to determine whether there are long-term clinical issues of concern.
Strategy depends on more than age
The “findings are not too surprising,” John Carroll, MD, who was not involved in this research, said in an email.
“Age is only one of multiple patient characteristics that enter into consideration of TAVR versus SAVR,” said Dr. Carroll, from Anschutz Medical Campus, University of Colorado, Aurora.
“As the article reports,” he noted, “those less than 65 having TAVR are more likely to have comorbid conditions that likely made the risk of SAVR higher.”
Dr. Carroll was lead author of a review article published in 2020 based on data from the ACC–Society of Thoracic Surgeons (STS)–Transcatheter Valve Therapy (TVT) registry on 276,316 patients who had TAVR in the United States from 2011-2019.
He pointed out that Figure 2 in that review shows that “SAVR is often performed in conjunction with other surgical procedures – another major reason why SAVR remains an important treatment for valvular heart disease.”
“We are awaiting long-term data comparing TAVR to SAVR durability,” Dr. Carroll added, echoing Dr. Dauerman. “So far [there are] no major differences, but it remains a key need to fully understand TAVR and the various models in commercial use.”
“Both TAVR and SAVR used in adults are tissue valves (SAVR with mechanical valves is used in younger patients),” Dr. Carroll noted, “and all tissue valves will eventually fail if the patient lives long enough.”
Patient management strategies need to consider what treatment options exist when the first valve fails. “If the first valve is SAVR, there is now extensive experience with placing a TAVR valve inside a failing SAVR valve, so called Valve-in-Valve or TAVR-in-SAVR. This is the preferred treatment in most patients with failing SAVR valves,” he said.
“On the other hand,” he continued, “we are just beginning to see more and more patients with failing TAVR valves, and the TAVR-in-TAVR procedure is less well understood.”
“Issues such as acute coronary occlusion and long-term difficulty in accessing coronary arteries are being encountered in some patients having TAVR-in-TAVR,” Dr. Carroll noted, which he discusses in a recent editorial he coauthored about the complexities of redo TAVR, published in JACC: Cardiovascular Interventions.
The study received no funding. Dr. Dauerman has research grants and is a consultant for Medtronic and Boston Scientific. Dr. Carroll is a local principal investigator in trials sponsored by Medtronic, Abbott, and Edwards Lifesciences.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
In cardiogenic shock, edge-to-edge mitral valve repair improves outcome
In patients with severe mitral regurgitation (MR) and cardiogenic shock, successful transcatheter edge-to-edge repair (TEER) is associated with a substantial reduction in all-cause mortality and lower morbidity at 1 year, according to an analysis of registry data.
The data from this analysis also confirm that “successful reduction of MR is achievable with TEER in most patients with cardiogenic shock,” reported Mohamad A. Alkhouli, MD, an interventional cardiologist and professor of medicine at the Mayo Clinic, Rochester, Minn.
In those with device success, achieved in 85.6% of patients, all-cause mortality was about 21% lower (34.6% vs. 55.5%; P < .001) at 1 year than in those who were not successfully repaired, according to Dr. Alkhouli, who presented the findings at the Transcatheter Cardiovascular Therapeutics annual meeting in Boston. This translated into a reduction in the hazard ratio for death of nearly 50% (hazard ratio, 0.52; 95% confidence interval, 0.43-0.63).
A similar relative benefit was found for the composite endpoint of mortality and heart failure admissions at 1 year. Whether unadjusted (HR, 0.54; 95% CI, 0.45-0.66) or adjusted (HR, 0.51; 95% CI, 0.42-0.62), risk reductions with successful MR reduction, defined as greater than or equal to 1 grade improvement and a final MR grade of less than or equal to 2+, indicated that major adverse outcomes are reduced by about half.
STS/ACC TCT registry data queried
Drawn from the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry, 3,797 patients with cardiogenic shock underwent MR repair between November 2013 and December 2021. Outcomes at 1 year were evaluable in 2,773 of these patients. For inclusion, all had to meet at least one of the definitions of cardiogenic shock, such as inotrope use or mechanical circulatory support.
At baseline, 94.5% had a MR severity of at least 3+, and most of these had 4+. Thirty days after treatment, 88.8% had MR severity of 2+ or less, the majority of which had a severity of 1+.
These data address an important question not previously well studied, according to Dr. Alkhouli. In MR patients, cardiogenic shock is associated with a high risk of death, but there has been little evidence that valve repair does not exacerbate, let alone modify, this risk.
These data support the value of intervention, which was performed in almost all patients with MitraClipä (Abbott), the only device available for most of the period in which the registry was queried. However, Dr. Alkhouli cautioned that his data are best considered “hypothesis generating.”
“We need a randomized trial,” he said at the meeting sponsored by the Cardiovascular Research Foundation. He pointed out that this is a complex population for which multiple variables might have skewed results when data are analyzed retrospectively. Not least, those MR patients with cardiogenic shock in the database considered for TEER might well have been relatively healthy and not representative of an unselected population with both MR and cardiogenic shock.
The question might be better answered by the multicenter Canadian trial CAPITAL MINOS, which has just started. Described in an article in the American Heart Journal, it has a planned enrollment of about 150 MR patients with cardiogenic shock randomized to TEER or medical therapy. Results are expected in about 1 year, according to Dr. Alkhouli.
But regarding the present analysis, Dr. Alkhouli did note that sensitivity analyses conducted within his data across risk factors, such as degenerative versus nondegenerative MR, low (< 30%) versus higher left ventricular ejection fraction (LVEF), and presence or absence of an acute coronary syndrome (ACS), consistently supported a benefit from intervention.
Also, cardiogenic shock did not appear to be a factor in device failure, according to Dr. Alkhouli, addressing a potential criticism that cardiogenic shock was an underlying reason for device failure.
More than 90% in NYHA class III or IV heart failure
In this study, the mean age was 73 years. More than 90% were in class III or IV heart failure in the 2 weeks prior to TEER. More than half had established coronary artery disease. Other concomitant cardiovascular morbidities, including atrial fibrillation or flutter (65%), prior MI (39%), and prior stroke or transient ischemic attach (> 10%) were well represented.
When those with device success were compared with those with device failure, the risk profile was comparable. The predicted STS (Society of Thoracic Surgeons) mortality for mitral valve repair among these two groups was 14.8% versus 15% (P = 0.97), respectively.
However, those with device failure did have a lower baseline left ventricular ejection fraction (40.7% vs. 42.9%; P = .009) and a greater prevalence of moderate-to-severe or severe MR (96.1% vs. 84.9%; P < 0.001).
The growing experience with TEER means that benefit has now been shown in several complicated MR groups, such as those with severe ventricular dysfunction, renal insufficiency, and obstructive lung disease. This was a rationale for looking at the impact or repairing MR in patients with cardiogenic shock.
It is a pressing question, according to Dr. Alkhouli. He cited studies suggesting that up to 20% of patients hospitalized for cardiogenic shock have at least moderate-to-severe MR. Conversely, cardiogenic shock is not an uncommon finding in patients with MR.
While Dr. Alkhouli acknowledged that the many variables influencing outcome in patients with MR and cardiogenic shock will make a randomized trial “challenging,” many experts echoed this concern and even expressed some skepticism about the potential for an unbiased trial.
Data confirm MR repair is safe during shock
“These data do show that repair of MR is safe in patients safe in patients with cardiogenic shock,” said Anita W. Asgar, MD, an interventional cardiologist associated with the Montreal Heart Institute. She noted that there was a 5- to 6-day delay among the cardiogenic shock patients prior to undergoing MR repair in this analysis, potentially reflecting an elimination of those at very high risk. Similarly, she suggested that many interventionalists are likely to consider multiple variables before proceeding.
As a result, MR repair may not be amenable to randomization in a cardiogenic shock population, given that this decision is not typically undertaken out of the context of multiple variables.
“I am not sure that a clinical trial is ethical,” she said. She would expect that clinicians enrolling patients would only do so on a selective basis.
Alexandra J. Lansky, MD, Director of the Yale Heart and Vascular Research Program, Yale University, New Haven, Conn., also emphasized the difficulty of controlling for variables, such as the duration of cardiogenic shock, that influence decision-making.
Nevertheless, she called the data “very important” in that they at least lend some objective data for deciding whether to intervene a group of “challenging” patients not uncommonly faced in clinical practice.
Dr. Alkhouli reports financial relationships with Abbott Vascular, Boston Scientific, Johnson & Johnson, and Phillips. Dr. Asgar reports financial relationships with Abbott Vascular, Edwards Lifesciences, W.L. Gore & Associates, and Medtronic. Dr. Lasky reports no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
In patients with severe mitral regurgitation (MR) and cardiogenic shock, successful transcatheter edge-to-edge repair (TEER) is associated with a substantial reduction in all-cause mortality and lower morbidity at 1 year, according to an analysis of registry data.
The data from this analysis also confirm that “successful reduction of MR is achievable with TEER in most patients with cardiogenic shock,” reported Mohamad A. Alkhouli, MD, an interventional cardiologist and professor of medicine at the Mayo Clinic, Rochester, Minn.
In those with device success, achieved in 85.6% of patients, all-cause mortality was about 21% lower (34.6% vs. 55.5%; P < .001) at 1 year than in those who were not successfully repaired, according to Dr. Alkhouli, who presented the findings at the Transcatheter Cardiovascular Therapeutics annual meeting in Boston. This translated into a reduction in the hazard ratio for death of nearly 50% (hazard ratio, 0.52; 95% confidence interval, 0.43-0.63).
A similar relative benefit was found for the composite endpoint of mortality and heart failure admissions at 1 year. Whether unadjusted (HR, 0.54; 95% CI, 0.45-0.66) or adjusted (HR, 0.51; 95% CI, 0.42-0.62), risk reductions with successful MR reduction, defined as greater than or equal to 1 grade improvement and a final MR grade of less than or equal to 2+, indicated that major adverse outcomes are reduced by about half.
STS/ACC TCT registry data queried
Drawn from the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry, 3,797 patients with cardiogenic shock underwent MR repair between November 2013 and December 2021. Outcomes at 1 year were evaluable in 2,773 of these patients. For inclusion, all had to meet at least one of the definitions of cardiogenic shock, such as inotrope use or mechanical circulatory support.
At baseline, 94.5% had a MR severity of at least 3+, and most of these had 4+. Thirty days after treatment, 88.8% had MR severity of 2+ or less, the majority of which had a severity of 1+.
These data address an important question not previously well studied, according to Dr. Alkhouli. In MR patients, cardiogenic shock is associated with a high risk of death, but there has been little evidence that valve repair does not exacerbate, let alone modify, this risk.
These data support the value of intervention, which was performed in almost all patients with MitraClipä (Abbott), the only device available for most of the period in which the registry was queried. However, Dr. Alkhouli cautioned that his data are best considered “hypothesis generating.”
“We need a randomized trial,” he said at the meeting sponsored by the Cardiovascular Research Foundation. He pointed out that this is a complex population for which multiple variables might have skewed results when data are analyzed retrospectively. Not least, those MR patients with cardiogenic shock in the database considered for TEER might well have been relatively healthy and not representative of an unselected population with both MR and cardiogenic shock.
The question might be better answered by the multicenter Canadian trial CAPITAL MINOS, which has just started. Described in an article in the American Heart Journal, it has a planned enrollment of about 150 MR patients with cardiogenic shock randomized to TEER or medical therapy. Results are expected in about 1 year, according to Dr. Alkhouli.
But regarding the present analysis, Dr. Alkhouli did note that sensitivity analyses conducted within his data across risk factors, such as degenerative versus nondegenerative MR, low (< 30%) versus higher left ventricular ejection fraction (LVEF), and presence or absence of an acute coronary syndrome (ACS), consistently supported a benefit from intervention.
Also, cardiogenic shock did not appear to be a factor in device failure, according to Dr. Alkhouli, addressing a potential criticism that cardiogenic shock was an underlying reason for device failure.
More than 90% in NYHA class III or IV heart failure
In this study, the mean age was 73 years. More than 90% were in class III or IV heart failure in the 2 weeks prior to TEER. More than half had established coronary artery disease. Other concomitant cardiovascular morbidities, including atrial fibrillation or flutter (65%), prior MI (39%), and prior stroke or transient ischemic attach (> 10%) were well represented.
When those with device success were compared with those with device failure, the risk profile was comparable. The predicted STS (Society of Thoracic Surgeons) mortality for mitral valve repair among these two groups was 14.8% versus 15% (P = 0.97), respectively.
However, those with device failure did have a lower baseline left ventricular ejection fraction (40.7% vs. 42.9%; P = .009) and a greater prevalence of moderate-to-severe or severe MR (96.1% vs. 84.9%; P < 0.001).
The growing experience with TEER means that benefit has now been shown in several complicated MR groups, such as those with severe ventricular dysfunction, renal insufficiency, and obstructive lung disease. This was a rationale for looking at the impact or repairing MR in patients with cardiogenic shock.
It is a pressing question, according to Dr. Alkhouli. He cited studies suggesting that up to 20% of patients hospitalized for cardiogenic shock have at least moderate-to-severe MR. Conversely, cardiogenic shock is not an uncommon finding in patients with MR.
While Dr. Alkhouli acknowledged that the many variables influencing outcome in patients with MR and cardiogenic shock will make a randomized trial “challenging,” many experts echoed this concern and even expressed some skepticism about the potential for an unbiased trial.
Data confirm MR repair is safe during shock
“These data do show that repair of MR is safe in patients safe in patients with cardiogenic shock,” said Anita W. Asgar, MD, an interventional cardiologist associated with the Montreal Heart Institute. She noted that there was a 5- to 6-day delay among the cardiogenic shock patients prior to undergoing MR repair in this analysis, potentially reflecting an elimination of those at very high risk. Similarly, she suggested that many interventionalists are likely to consider multiple variables before proceeding.
As a result, MR repair may not be amenable to randomization in a cardiogenic shock population, given that this decision is not typically undertaken out of the context of multiple variables.
“I am not sure that a clinical trial is ethical,” she said. She would expect that clinicians enrolling patients would only do so on a selective basis.
Alexandra J. Lansky, MD, Director of the Yale Heart and Vascular Research Program, Yale University, New Haven, Conn., also emphasized the difficulty of controlling for variables, such as the duration of cardiogenic shock, that influence decision-making.
Nevertheless, she called the data “very important” in that they at least lend some objective data for deciding whether to intervene a group of “challenging” patients not uncommonly faced in clinical practice.
Dr. Alkhouli reports financial relationships with Abbott Vascular, Boston Scientific, Johnson & Johnson, and Phillips. Dr. Asgar reports financial relationships with Abbott Vascular, Edwards Lifesciences, W.L. Gore & Associates, and Medtronic. Dr. Lasky reports no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
In patients with severe mitral regurgitation (MR) and cardiogenic shock, successful transcatheter edge-to-edge repair (TEER) is associated with a substantial reduction in all-cause mortality and lower morbidity at 1 year, according to an analysis of registry data.
The data from this analysis also confirm that “successful reduction of MR is achievable with TEER in most patients with cardiogenic shock,” reported Mohamad A. Alkhouli, MD, an interventional cardiologist and professor of medicine at the Mayo Clinic, Rochester, Minn.
In those with device success, achieved in 85.6% of patients, all-cause mortality was about 21% lower (34.6% vs. 55.5%; P < .001) at 1 year than in those who were not successfully repaired, according to Dr. Alkhouli, who presented the findings at the Transcatheter Cardiovascular Therapeutics annual meeting in Boston. This translated into a reduction in the hazard ratio for death of nearly 50% (hazard ratio, 0.52; 95% confidence interval, 0.43-0.63).
A similar relative benefit was found for the composite endpoint of mortality and heart failure admissions at 1 year. Whether unadjusted (HR, 0.54; 95% CI, 0.45-0.66) or adjusted (HR, 0.51; 95% CI, 0.42-0.62), risk reductions with successful MR reduction, defined as greater than or equal to 1 grade improvement and a final MR grade of less than or equal to 2+, indicated that major adverse outcomes are reduced by about half.
STS/ACC TCT registry data queried
Drawn from the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry, 3,797 patients with cardiogenic shock underwent MR repair between November 2013 and December 2021. Outcomes at 1 year were evaluable in 2,773 of these patients. For inclusion, all had to meet at least one of the definitions of cardiogenic shock, such as inotrope use or mechanical circulatory support.
At baseline, 94.5% had a MR severity of at least 3+, and most of these had 4+. Thirty days after treatment, 88.8% had MR severity of 2+ or less, the majority of which had a severity of 1+.
These data address an important question not previously well studied, according to Dr. Alkhouli. In MR patients, cardiogenic shock is associated with a high risk of death, but there has been little evidence that valve repair does not exacerbate, let alone modify, this risk.
These data support the value of intervention, which was performed in almost all patients with MitraClipä (Abbott), the only device available for most of the period in which the registry was queried. However, Dr. Alkhouli cautioned that his data are best considered “hypothesis generating.”
“We need a randomized trial,” he said at the meeting sponsored by the Cardiovascular Research Foundation. He pointed out that this is a complex population for which multiple variables might have skewed results when data are analyzed retrospectively. Not least, those MR patients with cardiogenic shock in the database considered for TEER might well have been relatively healthy and not representative of an unselected population with both MR and cardiogenic shock.
The question might be better answered by the multicenter Canadian trial CAPITAL MINOS, which has just started. Described in an article in the American Heart Journal, it has a planned enrollment of about 150 MR patients with cardiogenic shock randomized to TEER or medical therapy. Results are expected in about 1 year, according to Dr. Alkhouli.
But regarding the present analysis, Dr. Alkhouli did note that sensitivity analyses conducted within his data across risk factors, such as degenerative versus nondegenerative MR, low (< 30%) versus higher left ventricular ejection fraction (LVEF), and presence or absence of an acute coronary syndrome (ACS), consistently supported a benefit from intervention.
Also, cardiogenic shock did not appear to be a factor in device failure, according to Dr. Alkhouli, addressing a potential criticism that cardiogenic shock was an underlying reason for device failure.
More than 90% in NYHA class III or IV heart failure
In this study, the mean age was 73 years. More than 90% were in class III or IV heart failure in the 2 weeks prior to TEER. More than half had established coronary artery disease. Other concomitant cardiovascular morbidities, including atrial fibrillation or flutter (65%), prior MI (39%), and prior stroke or transient ischemic attach (> 10%) were well represented.
When those with device success were compared with those with device failure, the risk profile was comparable. The predicted STS (Society of Thoracic Surgeons) mortality for mitral valve repair among these two groups was 14.8% versus 15% (P = 0.97), respectively.
However, those with device failure did have a lower baseline left ventricular ejection fraction (40.7% vs. 42.9%; P = .009) and a greater prevalence of moderate-to-severe or severe MR (96.1% vs. 84.9%; P < 0.001).
The growing experience with TEER means that benefit has now been shown in several complicated MR groups, such as those with severe ventricular dysfunction, renal insufficiency, and obstructive lung disease. This was a rationale for looking at the impact or repairing MR in patients with cardiogenic shock.
It is a pressing question, according to Dr. Alkhouli. He cited studies suggesting that up to 20% of patients hospitalized for cardiogenic shock have at least moderate-to-severe MR. Conversely, cardiogenic shock is not an uncommon finding in patients with MR.
While Dr. Alkhouli acknowledged that the many variables influencing outcome in patients with MR and cardiogenic shock will make a randomized trial “challenging,” many experts echoed this concern and even expressed some skepticism about the potential for an unbiased trial.
Data confirm MR repair is safe during shock
“These data do show that repair of MR is safe in patients safe in patients with cardiogenic shock,” said Anita W. Asgar, MD, an interventional cardiologist associated with the Montreal Heart Institute. She noted that there was a 5- to 6-day delay among the cardiogenic shock patients prior to undergoing MR repair in this analysis, potentially reflecting an elimination of those at very high risk. Similarly, she suggested that many interventionalists are likely to consider multiple variables before proceeding.
As a result, MR repair may not be amenable to randomization in a cardiogenic shock population, given that this decision is not typically undertaken out of the context of multiple variables.
“I am not sure that a clinical trial is ethical,” she said. She would expect that clinicians enrolling patients would only do so on a selective basis.
Alexandra J. Lansky, MD, Director of the Yale Heart and Vascular Research Program, Yale University, New Haven, Conn., also emphasized the difficulty of controlling for variables, such as the duration of cardiogenic shock, that influence decision-making.
Nevertheless, she called the data “very important” in that they at least lend some objective data for deciding whether to intervene a group of “challenging” patients not uncommonly faced in clinical practice.
Dr. Alkhouli reports financial relationships with Abbott Vascular, Boston Scientific, Johnson & Johnson, and Phillips. Dr. Asgar reports financial relationships with Abbott Vascular, Edwards Lifesciences, W.L. Gore & Associates, and Medtronic. Dr. Lasky reports no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
Fourth-gen transcatheter mitral valve shows clinical, procedural improvements
The design improvements introduced in the fourth-generation device for transcatheter mitral valve repair, called the MitraClip G4 (Abbott), appears to yield better outcomes than previous iterations, according to a multinational postapproval study with more than 1,000 patients.
Not least, the 1.3% all-cause mortality at 30 days in this series, called EXPAND G4, “is the lowest that has been reported to date,” reported Ralph Stephan von Bardeleben, MD, at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
The evidence of relative advantages was based on comparisons with historical data and a similar study of the previous-generation device. That previous study, called EXPAND, evaluated the MitraClip NTR and ETR systems.
Device times shorter with new device
“There were shorter device times with MitraClip G4,” said Dr. von Bardeleben, referring to a more than 10-minute advantage over the previous generation device (35 minutes in EXPAND G4 vs. 46 min in EXPAND). Although the reduction in overall median procedure time was more modest (77 vs. 80 minutes), Dr. von Bardeleben said these are “the shortest device and procedural times reported to date.”
He also reported what appeared to be incremental advantages across multiple other endpoints, such as procedural success (96.2% vs. 95.8%) and a reduction in the mean clip rate (1.4 vs. 1.5).
Compared with historical outcomes with other devices employed in transcatheter edge-to-edge repair (TEER) of mitral valves, Dr. von Bardeleben contended that the results support the premise that the MitraClip G4 system is a meaningful advance by incorporating such features as an expanded choice of clip sizes, a greater coaptation area, and a more advanced gripper actuation for leaflet grasping.
Over 90% achieve MR 1+
Not least, it appears to increase the proportion of patients who achieve a mitral regurgitation grade of 1+ (MR1+) or lower, which is increasingly recognized as the goal of TEER, said Dr. von Bardeleben, head of the Centre of Structure Heart Disease Interventions, Heart Valve Centre, Mainz, Germany.
He said the rates of 91% achieving MR1+ or less and 98% achieved 2+ or lower compare favorably with most other series and exceeds levels achieved with surgery.
Dr. von Bardeleben also contended that, because of its design features, the MitraClip G4 “expands the spectrum of TEER-suitable patients.” He noted that 5% of the patients in this real-world series had a high risk of stenosis owing to such issues as severe annular or leaflet calcification and another 5% had factors that would predict inadequate MR reduction, such as Barlow’s disease, bi-leaflet prolapse, and severe leaflet degeneration.
The 1,164 patients in EXPAND G4 were enrolled from sites in the United States, Europe, Canada, and Japan. For the key outcome measure of procedural success, echocardiograms were assessed by an independent core laboratory. Of the 1,164 patients enrolled, 1,044 (91%) had complete follow-up data at 30 days.
The procedural success rates were reflected in improvements in New York Heart Association (NYHA) functional classes and in the Kansas City Cardiomyopathy Questionnaire (KCCQ), a quality of life instrument. Prior to treatment, 69% were in NYHA class III or greater. Following treatment, the proportion was 17% (P < .0001). The 18-point improvement in the KCCQ was characterized by Dr. von Bardeleben as “both clinically and statistically significant [P < .0001].”
There were no strokes in this series, and the 30-day incidence of myocardial infarction was 0.2%. The proportion requiring cardiovascular surgery within 30 days was less than 1%. The rate of bleeding episodes, all of which were nonserious, was 7%.
The “EXPAND G4 study confirms the safety and effectiveness of the next generation MitraClip G4 system,” according to Dr. von Bardeleben, and it did so “in a contemporary real-world setting.”
Outcome data characterized as ‘excellent’
Several invited panelists participating in a discussion following the presentation agreed.
“These results are excellent,” said Raj Makkar, MD, associate director of interventional technologies at Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles. While he was impressed with the fact that only 2% missed the primary endpoint of MR 2+ or lower, he indicated that the 91% achieving MR 1+ or lower might be an even more apt signal that newer-generation devices are improving.
This was echoed by other panelists who appeared to form a general consensus over the premise that the target in TEER should no longer be MR 2+ for most patients.
“We should now be aiming for MR grade of 0-1,” stated panelist Stephan Windecker, MD, chairman, department of cardiology, University of Bern (Switzerland). He indicated that this goal is increasingly reasonable given the advances in device design and greater operator experience.
Dr. von Bardeleben reported financial relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, and Neochord. Dr. Makkar reported financial relationships with Abbott Vascular, Cordis, Edwards Lifesciences, and Medtronic. Dr. Windecker reported financial relationships with more than 30 pharmaceutical companies, including Abbott Vascular, which manufactures MitraClip G4.
The design improvements introduced in the fourth-generation device for transcatheter mitral valve repair, called the MitraClip G4 (Abbott), appears to yield better outcomes than previous iterations, according to a multinational postapproval study with more than 1,000 patients.
Not least, the 1.3% all-cause mortality at 30 days in this series, called EXPAND G4, “is the lowest that has been reported to date,” reported Ralph Stephan von Bardeleben, MD, at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
The evidence of relative advantages was based on comparisons with historical data and a similar study of the previous-generation device. That previous study, called EXPAND, evaluated the MitraClip NTR and ETR systems.
Device times shorter with new device
“There were shorter device times with MitraClip G4,” said Dr. von Bardeleben, referring to a more than 10-minute advantage over the previous generation device (35 minutes in EXPAND G4 vs. 46 min in EXPAND). Although the reduction in overall median procedure time was more modest (77 vs. 80 minutes), Dr. von Bardeleben said these are “the shortest device and procedural times reported to date.”
He also reported what appeared to be incremental advantages across multiple other endpoints, such as procedural success (96.2% vs. 95.8%) and a reduction in the mean clip rate (1.4 vs. 1.5).
Compared with historical outcomes with other devices employed in transcatheter edge-to-edge repair (TEER) of mitral valves, Dr. von Bardeleben contended that the results support the premise that the MitraClip G4 system is a meaningful advance by incorporating such features as an expanded choice of clip sizes, a greater coaptation area, and a more advanced gripper actuation for leaflet grasping.
Over 90% achieve MR 1+
Not least, it appears to increase the proportion of patients who achieve a mitral regurgitation grade of 1+ (MR1+) or lower, which is increasingly recognized as the goal of TEER, said Dr. von Bardeleben, head of the Centre of Structure Heart Disease Interventions, Heart Valve Centre, Mainz, Germany.
He said the rates of 91% achieving MR1+ or less and 98% achieved 2+ or lower compare favorably with most other series and exceeds levels achieved with surgery.
Dr. von Bardeleben also contended that, because of its design features, the MitraClip G4 “expands the spectrum of TEER-suitable patients.” He noted that 5% of the patients in this real-world series had a high risk of stenosis owing to such issues as severe annular or leaflet calcification and another 5% had factors that would predict inadequate MR reduction, such as Barlow’s disease, bi-leaflet prolapse, and severe leaflet degeneration.
The 1,164 patients in EXPAND G4 were enrolled from sites in the United States, Europe, Canada, and Japan. For the key outcome measure of procedural success, echocardiograms were assessed by an independent core laboratory. Of the 1,164 patients enrolled, 1,044 (91%) had complete follow-up data at 30 days.
The procedural success rates were reflected in improvements in New York Heart Association (NYHA) functional classes and in the Kansas City Cardiomyopathy Questionnaire (KCCQ), a quality of life instrument. Prior to treatment, 69% were in NYHA class III or greater. Following treatment, the proportion was 17% (P < .0001). The 18-point improvement in the KCCQ was characterized by Dr. von Bardeleben as “both clinically and statistically significant [P < .0001].”
There were no strokes in this series, and the 30-day incidence of myocardial infarction was 0.2%. The proportion requiring cardiovascular surgery within 30 days was less than 1%. The rate of bleeding episodes, all of which were nonserious, was 7%.
The “EXPAND G4 study confirms the safety and effectiveness of the next generation MitraClip G4 system,” according to Dr. von Bardeleben, and it did so “in a contemporary real-world setting.”
Outcome data characterized as ‘excellent’
Several invited panelists participating in a discussion following the presentation agreed.
“These results are excellent,” said Raj Makkar, MD, associate director of interventional technologies at Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles. While he was impressed with the fact that only 2% missed the primary endpoint of MR 2+ or lower, he indicated that the 91% achieving MR 1+ or lower might be an even more apt signal that newer-generation devices are improving.
This was echoed by other panelists who appeared to form a general consensus over the premise that the target in TEER should no longer be MR 2+ for most patients.
“We should now be aiming for MR grade of 0-1,” stated panelist Stephan Windecker, MD, chairman, department of cardiology, University of Bern (Switzerland). He indicated that this goal is increasingly reasonable given the advances in device design and greater operator experience.
Dr. von Bardeleben reported financial relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, and Neochord. Dr. Makkar reported financial relationships with Abbott Vascular, Cordis, Edwards Lifesciences, and Medtronic. Dr. Windecker reported financial relationships with more than 30 pharmaceutical companies, including Abbott Vascular, which manufactures MitraClip G4.
The design improvements introduced in the fourth-generation device for transcatheter mitral valve repair, called the MitraClip G4 (Abbott), appears to yield better outcomes than previous iterations, according to a multinational postapproval study with more than 1,000 patients.
Not least, the 1.3% all-cause mortality at 30 days in this series, called EXPAND G4, “is the lowest that has been reported to date,” reported Ralph Stephan von Bardeleben, MD, at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
The evidence of relative advantages was based on comparisons with historical data and a similar study of the previous-generation device. That previous study, called EXPAND, evaluated the MitraClip NTR and ETR systems.
Device times shorter with new device
“There were shorter device times with MitraClip G4,” said Dr. von Bardeleben, referring to a more than 10-minute advantage over the previous generation device (35 minutes in EXPAND G4 vs. 46 min in EXPAND). Although the reduction in overall median procedure time was more modest (77 vs. 80 minutes), Dr. von Bardeleben said these are “the shortest device and procedural times reported to date.”
He also reported what appeared to be incremental advantages across multiple other endpoints, such as procedural success (96.2% vs. 95.8%) and a reduction in the mean clip rate (1.4 vs. 1.5).
Compared with historical outcomes with other devices employed in transcatheter edge-to-edge repair (TEER) of mitral valves, Dr. von Bardeleben contended that the results support the premise that the MitraClip G4 system is a meaningful advance by incorporating such features as an expanded choice of clip sizes, a greater coaptation area, and a more advanced gripper actuation for leaflet grasping.
Over 90% achieve MR 1+
Not least, it appears to increase the proportion of patients who achieve a mitral regurgitation grade of 1+ (MR1+) or lower, which is increasingly recognized as the goal of TEER, said Dr. von Bardeleben, head of the Centre of Structure Heart Disease Interventions, Heart Valve Centre, Mainz, Germany.
He said the rates of 91% achieving MR1+ or less and 98% achieved 2+ or lower compare favorably with most other series and exceeds levels achieved with surgery.
Dr. von Bardeleben also contended that, because of its design features, the MitraClip G4 “expands the spectrum of TEER-suitable patients.” He noted that 5% of the patients in this real-world series had a high risk of stenosis owing to such issues as severe annular or leaflet calcification and another 5% had factors that would predict inadequate MR reduction, such as Barlow’s disease, bi-leaflet prolapse, and severe leaflet degeneration.
The 1,164 patients in EXPAND G4 were enrolled from sites in the United States, Europe, Canada, and Japan. For the key outcome measure of procedural success, echocardiograms were assessed by an independent core laboratory. Of the 1,164 patients enrolled, 1,044 (91%) had complete follow-up data at 30 days.
The procedural success rates were reflected in improvements in New York Heart Association (NYHA) functional classes and in the Kansas City Cardiomyopathy Questionnaire (KCCQ), a quality of life instrument. Prior to treatment, 69% were in NYHA class III or greater. Following treatment, the proportion was 17% (P < .0001). The 18-point improvement in the KCCQ was characterized by Dr. von Bardeleben as “both clinically and statistically significant [P < .0001].”
There were no strokes in this series, and the 30-day incidence of myocardial infarction was 0.2%. The proportion requiring cardiovascular surgery within 30 days was less than 1%. The rate of bleeding episodes, all of which were nonserious, was 7%.
The “EXPAND G4 study confirms the safety and effectiveness of the next generation MitraClip G4 system,” according to Dr. von Bardeleben, and it did so “in a contemporary real-world setting.”
Outcome data characterized as ‘excellent’
Several invited panelists participating in a discussion following the presentation agreed.
“These results are excellent,” said Raj Makkar, MD, associate director of interventional technologies at Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles. While he was impressed with the fact that only 2% missed the primary endpoint of MR 2+ or lower, he indicated that the 91% achieving MR 1+ or lower might be an even more apt signal that newer-generation devices are improving.
This was echoed by other panelists who appeared to form a general consensus over the premise that the target in TEER should no longer be MR 2+ for most patients.
“We should now be aiming for MR grade of 0-1,” stated panelist Stephan Windecker, MD, chairman, department of cardiology, University of Bern (Switzerland). He indicated that this goal is increasingly reasonable given the advances in device design and greater operator experience.
Dr. von Bardeleben reported financial relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, and Neochord. Dr. Makkar reported financial relationships with Abbott Vascular, Cordis, Edwards Lifesciences, and Medtronic. Dr. Windecker reported financial relationships with more than 30 pharmaceutical companies, including Abbott Vascular, which manufactures MitraClip G4.
FROM TCT 2022
Extravascular ICD surpasses goals in pivotal trial
BARCELONA – A novel “extravascular” implantable cardioverter defibrillator (ICD) that uses substernally placed electrodes surpassed its prespecified efficacy and safety targets in the device’s pivotal trial with 299 patients who received an implant.
The results showed that the extravascular ICD “provides antitachycardia pacing and low energy defibrillation while avoiding the vascular space” for lead placement, Ian Crozier, MD, said at the annual congress of the European Society of Cardiology.
“The results are fantastic; they exceeded our expectations,” said Dr. Crozier in an interview, adding that he expects the new device to receive marketing approval from regulatory agencies based on the findings. “This will be the next generation of ICD going forward,” predicted Dr. Crozier, an electrophysiologist cardiologist at Christchurch (New Zealand) Hospital.
Moving beyond transvenous and subcutaneous ICDs
Traditional ICDs use transvenous leads, which can cause vascular injury, are prone to lead fracture over time, and can produce serious infections as well as other potential complications. The U.S. Food and Drug Administration first approved an alternative-design, subcutaneous ICD in 2012 that avoids the need for transvenous leads and the risks they pose. But subcutaneous ICDs have their own limitations: an inability to provide antitachycardia pacing or chronic pacing; a limited ability to provide bradycardia pacing; and an increased device size with shorter battery life, because of the high shock power needed for effective performance. These drawbacks have collectively hindered uptake, Dr. Crozier said.
This led to development of the extravascular ICD – 10 years in the making – which uses substernally placed leads that allow antitachycardia pacing and backup pacing in a device with the size of and the anticipated battery longevity of a transvenous ICD device, noted Dr. Crozier.
A 98.7% rate of arrhythmia termination at implant
The pivotal trial’s primary efficacy endpoint was successful defibrillation based on terminating an induced, sustained, shockable ventricular arrhythmia at the time of implantation. The rate was 98.7%, compared with a prespecified target of 88%. All patients had a class I or IIa indication for an ICD.
The primary safety endpoint was freedom from major system- or procedure-related complications at 6 months, which occurred at a rate of 92.6%, compared with the study’s prespecified target rate of 79%. Both targets were derived from the historical rates of ICDs with transvenous leads.
Simultaneously with Dr. Crozier’s report at the congress, the results also appeared online in the New England Journal of Medicine.
Although the pivotal study met both prespecified endpoints, the evidence has limitations that make it likely that regulatory bodies will seek additional data, commented Fred Kusumoto, MD, director of heart rhythm services for the Mayo Clinic in Jacksonville, Fla.
Short follow-up; questions remain
“Follow-up was relatively short, less than a year,” and “questions remain” about the extravascular ICD’s performance, Dr. Kusumoto said in an interview. “Inappropriate shocks occurred in nearly 10% of patients after 11 month follow-up,” he noted, and also cited the 29 patients who needed revisions including two cases with lead fractures.
“The extravascular lead strategy has an advantage over transvenous systems because of the lower risk for extraction or explant,” and it also provides the antitachycardia pacing that’s not available with subcutaneous ICDs, he granted. But in the new study, antitachycardia termination was delivered to only 10 patients and had “reasonable” effectiveness by resolving 70% of these episodes. “Wide adoption by clinicians will depend on results from larger studies with longer follow-up,” Dr. Kusumoto maintained. He also wanted to see confirmation of the ease of lead removal after longer periods of implantation.
Implantation ‘is not difficult’
The trial ran at 46 sites in 17 countries during September 2019 to October 2021. It enrolled patients with a class I or IIa indication for an ICD, excluding patients with a prior sternotomy or need for chronic pacing, and those unable to undergo defibrillation testing.
Clinicians attempted an implantation in 316 patients and had successful placement in 299 (314 had successful placement of their substernal leads), with 292 having a functional device after 6 months, and 284 completing their planned 6-month follow-up. The median procedure time was 66 minutes, including the time for defibrillation testing.
All of the cardiologists who did the implants had received a full day of training prior to performing the procedure. “This is not a difficult procedure, but it is not a region [the substernal space] that cardiologists are familiar working in,” noted Dr. Crozier, explaining the rationale behind a policy of required implantation training.
Twenty-five adverse events occurred in 23 patients. Eighteen of these events required a system revision, including nine lead dislodgments and five infections. The seven adverse events that did not require a revision included three wound-related episodes and three hospitalizations for inappropriate shock. No patients died, nor were there any cardiac injuries as result of the implant.
During average follow-up of 10.6 months, the implanted devices delivered antitachycardia pacing to 10 patients, successfully terminating 32 of 46 episodes (70%), a rate that Dr. Crozier called “very good, and very comparable to transvenous devices.” The devices also delivered 18 appropriate shocks that successfully converted all 18 episodes.
A 10% rate of inappropriate shocks
However, 29 patients (10% of the study cohort) received inappropriate shocks in 81 episodes, with a total of 118 inappropriate shocks delivered, including 34 episodes (42%) triggered by oversensing of a P wave.
“We fully acknowledge that the inappropriate shock rate is higher than what’s seen with transvenous ICDs, but the rate is comparable to what was seen in the early trials with subcutaneous ICDs,” said Dr. Crozier. “We have a number of strategies to reduce the inappropriate shock rate to what we’d expect with conventional devices,” such as making sure that P waves are not detected by the device at the time of implantation, using new algorithms to mitigate P wave sensing, and other programming changes, he added.
Two patients had lead fractures that Dr. Crozier attributed to atypical lead locations and that are likely avoidable in the future. He expressed optimism that the extravascular ICD will avoid the high lead fracture rate over time that remains a problem for ICDs with transvenous leads.
The study also followed a subgroup of 36 patients who underwent a prespecified protocol of chronic defibrillation testing that was successful in all 36.
Dr. Crozier conceded that the extravascular ICD cannot currently deliver chronic pacing, but he expressed optimism that this capability will be available in the future.
“This innovative [extravascular] ICD system would be particularly beneficial for patients with ventricular arrhythmias that can be reliably pace terminated and avoid a transvenous endocardial lead, but more information is required,” concluded Dr. Kusumoto.
The study was sponsored by Medtronic, the company that is developing the extravascular ICD. Dr. Crozier is a consultant to and has received research funding from Medtronic. Dr. Kusumoto had no disclosures.
BARCELONA – A novel “extravascular” implantable cardioverter defibrillator (ICD) that uses substernally placed electrodes surpassed its prespecified efficacy and safety targets in the device’s pivotal trial with 299 patients who received an implant.
The results showed that the extravascular ICD “provides antitachycardia pacing and low energy defibrillation while avoiding the vascular space” for lead placement, Ian Crozier, MD, said at the annual congress of the European Society of Cardiology.
“The results are fantastic; they exceeded our expectations,” said Dr. Crozier in an interview, adding that he expects the new device to receive marketing approval from regulatory agencies based on the findings. “This will be the next generation of ICD going forward,” predicted Dr. Crozier, an electrophysiologist cardiologist at Christchurch (New Zealand) Hospital.
Moving beyond transvenous and subcutaneous ICDs
Traditional ICDs use transvenous leads, which can cause vascular injury, are prone to lead fracture over time, and can produce serious infections as well as other potential complications. The U.S. Food and Drug Administration first approved an alternative-design, subcutaneous ICD in 2012 that avoids the need for transvenous leads and the risks they pose. But subcutaneous ICDs have their own limitations: an inability to provide antitachycardia pacing or chronic pacing; a limited ability to provide bradycardia pacing; and an increased device size with shorter battery life, because of the high shock power needed for effective performance. These drawbacks have collectively hindered uptake, Dr. Crozier said.
This led to development of the extravascular ICD – 10 years in the making – which uses substernally placed leads that allow antitachycardia pacing and backup pacing in a device with the size of and the anticipated battery longevity of a transvenous ICD device, noted Dr. Crozier.
A 98.7% rate of arrhythmia termination at implant
The pivotal trial’s primary efficacy endpoint was successful defibrillation based on terminating an induced, sustained, shockable ventricular arrhythmia at the time of implantation. The rate was 98.7%, compared with a prespecified target of 88%. All patients had a class I or IIa indication for an ICD.
The primary safety endpoint was freedom from major system- or procedure-related complications at 6 months, which occurred at a rate of 92.6%, compared with the study’s prespecified target rate of 79%. Both targets were derived from the historical rates of ICDs with transvenous leads.
Simultaneously with Dr. Crozier’s report at the congress, the results also appeared online in the New England Journal of Medicine.
Although the pivotal study met both prespecified endpoints, the evidence has limitations that make it likely that regulatory bodies will seek additional data, commented Fred Kusumoto, MD, director of heart rhythm services for the Mayo Clinic in Jacksonville, Fla.
Short follow-up; questions remain
“Follow-up was relatively short, less than a year,” and “questions remain” about the extravascular ICD’s performance, Dr. Kusumoto said in an interview. “Inappropriate shocks occurred in nearly 10% of patients after 11 month follow-up,” he noted, and also cited the 29 patients who needed revisions including two cases with lead fractures.
“The extravascular lead strategy has an advantage over transvenous systems because of the lower risk for extraction or explant,” and it also provides the antitachycardia pacing that’s not available with subcutaneous ICDs, he granted. But in the new study, antitachycardia termination was delivered to only 10 patients and had “reasonable” effectiveness by resolving 70% of these episodes. “Wide adoption by clinicians will depend on results from larger studies with longer follow-up,” Dr. Kusumoto maintained. He also wanted to see confirmation of the ease of lead removal after longer periods of implantation.
Implantation ‘is not difficult’
The trial ran at 46 sites in 17 countries during September 2019 to October 2021. It enrolled patients with a class I or IIa indication for an ICD, excluding patients with a prior sternotomy or need for chronic pacing, and those unable to undergo defibrillation testing.
Clinicians attempted an implantation in 316 patients and had successful placement in 299 (314 had successful placement of their substernal leads), with 292 having a functional device after 6 months, and 284 completing their planned 6-month follow-up. The median procedure time was 66 minutes, including the time for defibrillation testing.
All of the cardiologists who did the implants had received a full day of training prior to performing the procedure. “This is not a difficult procedure, but it is not a region [the substernal space] that cardiologists are familiar working in,” noted Dr. Crozier, explaining the rationale behind a policy of required implantation training.
Twenty-five adverse events occurred in 23 patients. Eighteen of these events required a system revision, including nine lead dislodgments and five infections. The seven adverse events that did not require a revision included three wound-related episodes and three hospitalizations for inappropriate shock. No patients died, nor were there any cardiac injuries as result of the implant.
During average follow-up of 10.6 months, the implanted devices delivered antitachycardia pacing to 10 patients, successfully terminating 32 of 46 episodes (70%), a rate that Dr. Crozier called “very good, and very comparable to transvenous devices.” The devices also delivered 18 appropriate shocks that successfully converted all 18 episodes.
A 10% rate of inappropriate shocks
However, 29 patients (10% of the study cohort) received inappropriate shocks in 81 episodes, with a total of 118 inappropriate shocks delivered, including 34 episodes (42%) triggered by oversensing of a P wave.
“We fully acknowledge that the inappropriate shock rate is higher than what’s seen with transvenous ICDs, but the rate is comparable to what was seen in the early trials with subcutaneous ICDs,” said Dr. Crozier. “We have a number of strategies to reduce the inappropriate shock rate to what we’d expect with conventional devices,” such as making sure that P waves are not detected by the device at the time of implantation, using new algorithms to mitigate P wave sensing, and other programming changes, he added.
Two patients had lead fractures that Dr. Crozier attributed to atypical lead locations and that are likely avoidable in the future. He expressed optimism that the extravascular ICD will avoid the high lead fracture rate over time that remains a problem for ICDs with transvenous leads.
The study also followed a subgroup of 36 patients who underwent a prespecified protocol of chronic defibrillation testing that was successful in all 36.
Dr. Crozier conceded that the extravascular ICD cannot currently deliver chronic pacing, but he expressed optimism that this capability will be available in the future.
“This innovative [extravascular] ICD system would be particularly beneficial for patients with ventricular arrhythmias that can be reliably pace terminated and avoid a transvenous endocardial lead, but more information is required,” concluded Dr. Kusumoto.
The study was sponsored by Medtronic, the company that is developing the extravascular ICD. Dr. Crozier is a consultant to and has received research funding from Medtronic. Dr. Kusumoto had no disclosures.
BARCELONA – A novel “extravascular” implantable cardioverter defibrillator (ICD) that uses substernally placed electrodes surpassed its prespecified efficacy and safety targets in the device’s pivotal trial with 299 patients who received an implant.
The results showed that the extravascular ICD “provides antitachycardia pacing and low energy defibrillation while avoiding the vascular space” for lead placement, Ian Crozier, MD, said at the annual congress of the European Society of Cardiology.
“The results are fantastic; they exceeded our expectations,” said Dr. Crozier in an interview, adding that he expects the new device to receive marketing approval from regulatory agencies based on the findings. “This will be the next generation of ICD going forward,” predicted Dr. Crozier, an electrophysiologist cardiologist at Christchurch (New Zealand) Hospital.
Moving beyond transvenous and subcutaneous ICDs
Traditional ICDs use transvenous leads, which can cause vascular injury, are prone to lead fracture over time, and can produce serious infections as well as other potential complications. The U.S. Food and Drug Administration first approved an alternative-design, subcutaneous ICD in 2012 that avoids the need for transvenous leads and the risks they pose. But subcutaneous ICDs have their own limitations: an inability to provide antitachycardia pacing or chronic pacing; a limited ability to provide bradycardia pacing; and an increased device size with shorter battery life, because of the high shock power needed for effective performance. These drawbacks have collectively hindered uptake, Dr. Crozier said.
This led to development of the extravascular ICD – 10 years in the making – which uses substernally placed leads that allow antitachycardia pacing and backup pacing in a device with the size of and the anticipated battery longevity of a transvenous ICD device, noted Dr. Crozier.
A 98.7% rate of arrhythmia termination at implant
The pivotal trial’s primary efficacy endpoint was successful defibrillation based on terminating an induced, sustained, shockable ventricular arrhythmia at the time of implantation. The rate was 98.7%, compared with a prespecified target of 88%. All patients had a class I or IIa indication for an ICD.
The primary safety endpoint was freedom from major system- or procedure-related complications at 6 months, which occurred at a rate of 92.6%, compared with the study’s prespecified target rate of 79%. Both targets were derived from the historical rates of ICDs with transvenous leads.
Simultaneously with Dr. Crozier’s report at the congress, the results also appeared online in the New England Journal of Medicine.
Although the pivotal study met both prespecified endpoints, the evidence has limitations that make it likely that regulatory bodies will seek additional data, commented Fred Kusumoto, MD, director of heart rhythm services for the Mayo Clinic in Jacksonville, Fla.
Short follow-up; questions remain
“Follow-up was relatively short, less than a year,” and “questions remain” about the extravascular ICD’s performance, Dr. Kusumoto said in an interview. “Inappropriate shocks occurred in nearly 10% of patients after 11 month follow-up,” he noted, and also cited the 29 patients who needed revisions including two cases with lead fractures.
“The extravascular lead strategy has an advantage over transvenous systems because of the lower risk for extraction or explant,” and it also provides the antitachycardia pacing that’s not available with subcutaneous ICDs, he granted. But in the new study, antitachycardia termination was delivered to only 10 patients and had “reasonable” effectiveness by resolving 70% of these episodes. “Wide adoption by clinicians will depend on results from larger studies with longer follow-up,” Dr. Kusumoto maintained. He also wanted to see confirmation of the ease of lead removal after longer periods of implantation.
Implantation ‘is not difficult’
The trial ran at 46 sites in 17 countries during September 2019 to October 2021. It enrolled patients with a class I or IIa indication for an ICD, excluding patients with a prior sternotomy or need for chronic pacing, and those unable to undergo defibrillation testing.
Clinicians attempted an implantation in 316 patients and had successful placement in 299 (314 had successful placement of their substernal leads), with 292 having a functional device after 6 months, and 284 completing their planned 6-month follow-up. The median procedure time was 66 minutes, including the time for defibrillation testing.
All of the cardiologists who did the implants had received a full day of training prior to performing the procedure. “This is not a difficult procedure, but it is not a region [the substernal space] that cardiologists are familiar working in,” noted Dr. Crozier, explaining the rationale behind a policy of required implantation training.
Twenty-five adverse events occurred in 23 patients. Eighteen of these events required a system revision, including nine lead dislodgments and five infections. The seven adverse events that did not require a revision included three wound-related episodes and three hospitalizations for inappropriate shock. No patients died, nor were there any cardiac injuries as result of the implant.
During average follow-up of 10.6 months, the implanted devices delivered antitachycardia pacing to 10 patients, successfully terminating 32 of 46 episodes (70%), a rate that Dr. Crozier called “very good, and very comparable to transvenous devices.” The devices also delivered 18 appropriate shocks that successfully converted all 18 episodes.
A 10% rate of inappropriate shocks
However, 29 patients (10% of the study cohort) received inappropriate shocks in 81 episodes, with a total of 118 inappropriate shocks delivered, including 34 episodes (42%) triggered by oversensing of a P wave.
“We fully acknowledge that the inappropriate shock rate is higher than what’s seen with transvenous ICDs, but the rate is comparable to what was seen in the early trials with subcutaneous ICDs,” said Dr. Crozier. “We have a number of strategies to reduce the inappropriate shock rate to what we’d expect with conventional devices,” such as making sure that P waves are not detected by the device at the time of implantation, using new algorithms to mitigate P wave sensing, and other programming changes, he added.
Two patients had lead fractures that Dr. Crozier attributed to atypical lead locations and that are likely avoidable in the future. He expressed optimism that the extravascular ICD will avoid the high lead fracture rate over time that remains a problem for ICDs with transvenous leads.
The study also followed a subgroup of 36 patients who underwent a prespecified protocol of chronic defibrillation testing that was successful in all 36.
Dr. Crozier conceded that the extravascular ICD cannot currently deliver chronic pacing, but he expressed optimism that this capability will be available in the future.
“This innovative [extravascular] ICD system would be particularly beneficial for patients with ventricular arrhythmias that can be reliably pace terminated and avoid a transvenous endocardial lead, but more information is required,” concluded Dr. Kusumoto.
The study was sponsored by Medtronic, the company that is developing the extravascular ICD. Dr. Crozier is a consultant to and has received research funding from Medtronic. Dr. Kusumoto had no disclosures.
AT ESC CONGRESS 2022
Heparin pretreatment may safely open arteries before STEMI cath
, suggests a large registry study.
An open infarct-related artery (IRA) at angiography on cath-lab arrival presents STEMI patients an opportunity for earlier reperfusion and a chance, in theory at least, for smaller infarcts and maybe improved clinical outcomes.
In the new analysis, which covers more than 40,000 patients with STEMI in Sweden, the 38% who received heparin before cath-lab arrival were 11% less likely to show IRA occlusion at angiography prior to direct percutaneous coronary intervention (PCI). They also showed a 13% lower 30-day mortality compared with patients who were started on heparin in the cath lab. Importantly, their risk of major bleeding in the hospital did not increase.
The “early reperfusion” associated with IRA patency at angiography “could have long-term benefit due to smaller infarct size,” potentially explaining the observed 30-day survival gain in the pretreatment group, Oskar Love Emilsson, Lund (Sweden) University, said in an interview.
Mr. Emilsson, a third-year medical student, reported the analysis at the annual congress of the European Society of Cardiology, and is lead author on its same-day publication in the journal EuroIntervention.
He mentioned a few cautions in interpreting the study, which is based primarily on data from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR). It included several sensitivity analyses that continued to back pretreatment heparin as a significant predictor of an unoccluded IRA but didn’t consistently support the 30-day mortality benefit seen in the primary analysis.
And, although the pretreatment group overall didn’t have more major bleeds, the risk did go up significantly for those older than 75 or those who weighed less than 60 kg (132 pounds) or underwent catheterization with an access route other than the radial artery. Extra caution should be exercised in such patients who receive heparin before cath-lab arrival for PCI, Mr. Emilsson observed.
“Our results suggest that heparin pretreatment might be a good option to improve patency of infarct related arteries in STEMI,” and potentially clinical outcomes, he said. “However, a definite answer would require a randomized controlled trial.”
Meanwhile, the current study may be the largest yet to look at clinical outcomes after pretreatment with unfractionated heparin before PCI for acute STEMI, the report states. There have been some observational studies, subanalyses of STEMI trials, and even a few limited randomized trials – including the HEAP trial published in 2000 – to weigh in on the subject. Some have supported the strategy, others have not.
“With rapid door-to-balloon times in STEMI, it can be challenging to show a significant difference between a prehospital heparin approach and heparin given in the lab,” observed Sunil V. Rao, MD, NYU Langone Health System, New York, who is not connected with the current study.
Many EDs in the United States have “a STEMI protocol that calls for an IV bolus of heparin. It would be tougher in the U.S. to give it in the ambulance but again, it’s not clear how much advantage that would really provide,” he told this news organization.
Support from randomized trials would be needed before the practice could be formally recommended. “The SCAAR registries have set the standard for how registries should be conducted,” Dr. Rao said. “This is a very well done observational study, but it is observational.”
The priority for STEMI patients, he added, “really should be to get them to the lab as fast as possible. If the ED protocol includes heparin before the cath lab, that’s great, but I don’t think we should delay getting these patients to the lab to accommodate pre–cath-lab heparin.”
The current analysis covered 41,631 patients with STEMI from 2008 through to 2016, of whom 38% were pretreated with heparin in an ambulance or the ED. The remaining 62% initiated heparin in the cath lab.
About one-third of the group had an open IRA at angiography. The adjusted risk ratio (RR) for IRA occlusion at angiography for patients pretreated vs. not pretreated with heparin was 0.89 (95% confidence interval [CI], 0.87-0.90).
The corresponding RR for death within 30 days was 0.87 (95% CI, 0.77-0.99), and for major in-hospital bleeding it was 1.01 (95% CI, 0.86-1.18).
The analysis was adjusted for other medications received before cath-lab arrival, especially a long list of antiplatelets and non-heparin antithrombins. That strengthens the case for heparin pretreatment as an independent predictor of an open IRA at initial angiography, Mr. Emilsson said.
Comparisons of propensity-score–matched subgroups of the total cohort, conducted separately for the IRA-occlusion endpoint and the endpoints of 30-day mortality and major bleeding, produced similar results.
Some observational data suggest that antiplatelet pretreatment with a P2Y12 inhibitor may promote IRA patency on angiography after cath lab arrival, Dr. Rao observed. “This indicates that there probably is a role of earlier antithrombotic therapy in STEMI patients, but the randomized trials have not shown a consistent benefit,” he said, referring in particular to the ATLANTIC trial.
Mr. Emilsson and Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, suggests a large registry study.
An open infarct-related artery (IRA) at angiography on cath-lab arrival presents STEMI patients an opportunity for earlier reperfusion and a chance, in theory at least, for smaller infarcts and maybe improved clinical outcomes.
In the new analysis, which covers more than 40,000 patients with STEMI in Sweden, the 38% who received heparin before cath-lab arrival were 11% less likely to show IRA occlusion at angiography prior to direct percutaneous coronary intervention (PCI). They also showed a 13% lower 30-day mortality compared with patients who were started on heparin in the cath lab. Importantly, their risk of major bleeding in the hospital did not increase.
The “early reperfusion” associated with IRA patency at angiography “could have long-term benefit due to smaller infarct size,” potentially explaining the observed 30-day survival gain in the pretreatment group, Oskar Love Emilsson, Lund (Sweden) University, said in an interview.
Mr. Emilsson, a third-year medical student, reported the analysis at the annual congress of the European Society of Cardiology, and is lead author on its same-day publication in the journal EuroIntervention.
He mentioned a few cautions in interpreting the study, which is based primarily on data from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR). It included several sensitivity analyses that continued to back pretreatment heparin as a significant predictor of an unoccluded IRA but didn’t consistently support the 30-day mortality benefit seen in the primary analysis.
And, although the pretreatment group overall didn’t have more major bleeds, the risk did go up significantly for those older than 75 or those who weighed less than 60 kg (132 pounds) or underwent catheterization with an access route other than the radial artery. Extra caution should be exercised in such patients who receive heparin before cath-lab arrival for PCI, Mr. Emilsson observed.
“Our results suggest that heparin pretreatment might be a good option to improve patency of infarct related arteries in STEMI,” and potentially clinical outcomes, he said. “However, a definite answer would require a randomized controlled trial.”
Meanwhile, the current study may be the largest yet to look at clinical outcomes after pretreatment with unfractionated heparin before PCI for acute STEMI, the report states. There have been some observational studies, subanalyses of STEMI trials, and even a few limited randomized trials – including the HEAP trial published in 2000 – to weigh in on the subject. Some have supported the strategy, others have not.
“With rapid door-to-balloon times in STEMI, it can be challenging to show a significant difference between a prehospital heparin approach and heparin given in the lab,” observed Sunil V. Rao, MD, NYU Langone Health System, New York, who is not connected with the current study.
Many EDs in the United States have “a STEMI protocol that calls for an IV bolus of heparin. It would be tougher in the U.S. to give it in the ambulance but again, it’s not clear how much advantage that would really provide,” he told this news organization.
Support from randomized trials would be needed before the practice could be formally recommended. “The SCAAR registries have set the standard for how registries should be conducted,” Dr. Rao said. “This is a very well done observational study, but it is observational.”
The priority for STEMI patients, he added, “really should be to get them to the lab as fast as possible. If the ED protocol includes heparin before the cath lab, that’s great, but I don’t think we should delay getting these patients to the lab to accommodate pre–cath-lab heparin.”
The current analysis covered 41,631 patients with STEMI from 2008 through to 2016, of whom 38% were pretreated with heparin in an ambulance or the ED. The remaining 62% initiated heparin in the cath lab.
About one-third of the group had an open IRA at angiography. The adjusted risk ratio (RR) for IRA occlusion at angiography for patients pretreated vs. not pretreated with heparin was 0.89 (95% confidence interval [CI], 0.87-0.90).
The corresponding RR for death within 30 days was 0.87 (95% CI, 0.77-0.99), and for major in-hospital bleeding it was 1.01 (95% CI, 0.86-1.18).
The analysis was adjusted for other medications received before cath-lab arrival, especially a long list of antiplatelets and non-heparin antithrombins. That strengthens the case for heparin pretreatment as an independent predictor of an open IRA at initial angiography, Mr. Emilsson said.
Comparisons of propensity-score–matched subgroups of the total cohort, conducted separately for the IRA-occlusion endpoint and the endpoints of 30-day mortality and major bleeding, produced similar results.
Some observational data suggest that antiplatelet pretreatment with a P2Y12 inhibitor may promote IRA patency on angiography after cath lab arrival, Dr. Rao observed. “This indicates that there probably is a role of earlier antithrombotic therapy in STEMI patients, but the randomized trials have not shown a consistent benefit,” he said, referring in particular to the ATLANTIC trial.
Mr. Emilsson and Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, suggests a large registry study.
An open infarct-related artery (IRA) at angiography on cath-lab arrival presents STEMI patients an opportunity for earlier reperfusion and a chance, in theory at least, for smaller infarcts and maybe improved clinical outcomes.
In the new analysis, which covers more than 40,000 patients with STEMI in Sweden, the 38% who received heparin before cath-lab arrival were 11% less likely to show IRA occlusion at angiography prior to direct percutaneous coronary intervention (PCI). They also showed a 13% lower 30-day mortality compared with patients who were started on heparin in the cath lab. Importantly, their risk of major bleeding in the hospital did not increase.
The “early reperfusion” associated with IRA patency at angiography “could have long-term benefit due to smaller infarct size,” potentially explaining the observed 30-day survival gain in the pretreatment group, Oskar Love Emilsson, Lund (Sweden) University, said in an interview.
Mr. Emilsson, a third-year medical student, reported the analysis at the annual congress of the European Society of Cardiology, and is lead author on its same-day publication in the journal EuroIntervention.
He mentioned a few cautions in interpreting the study, which is based primarily on data from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR). It included several sensitivity analyses that continued to back pretreatment heparin as a significant predictor of an unoccluded IRA but didn’t consistently support the 30-day mortality benefit seen in the primary analysis.
And, although the pretreatment group overall didn’t have more major bleeds, the risk did go up significantly for those older than 75 or those who weighed less than 60 kg (132 pounds) or underwent catheterization with an access route other than the radial artery. Extra caution should be exercised in such patients who receive heparin before cath-lab arrival for PCI, Mr. Emilsson observed.
“Our results suggest that heparin pretreatment might be a good option to improve patency of infarct related arteries in STEMI,” and potentially clinical outcomes, he said. “However, a definite answer would require a randomized controlled trial.”
Meanwhile, the current study may be the largest yet to look at clinical outcomes after pretreatment with unfractionated heparin before PCI for acute STEMI, the report states. There have been some observational studies, subanalyses of STEMI trials, and even a few limited randomized trials – including the HEAP trial published in 2000 – to weigh in on the subject. Some have supported the strategy, others have not.
“With rapid door-to-balloon times in STEMI, it can be challenging to show a significant difference between a prehospital heparin approach and heparin given in the lab,” observed Sunil V. Rao, MD, NYU Langone Health System, New York, who is not connected with the current study.
Many EDs in the United States have “a STEMI protocol that calls for an IV bolus of heparin. It would be tougher in the U.S. to give it in the ambulance but again, it’s not clear how much advantage that would really provide,” he told this news organization.
Support from randomized trials would be needed before the practice could be formally recommended. “The SCAAR registries have set the standard for how registries should be conducted,” Dr. Rao said. “This is a very well done observational study, but it is observational.”
The priority for STEMI patients, he added, “really should be to get them to the lab as fast as possible. If the ED protocol includes heparin before the cath lab, that’s great, but I don’t think we should delay getting these patients to the lab to accommodate pre–cath-lab heparin.”
The current analysis covered 41,631 patients with STEMI from 2008 through to 2016, of whom 38% were pretreated with heparin in an ambulance or the ED. The remaining 62% initiated heparin in the cath lab.
About one-third of the group had an open IRA at angiography. The adjusted risk ratio (RR) for IRA occlusion at angiography for patients pretreated vs. not pretreated with heparin was 0.89 (95% confidence interval [CI], 0.87-0.90).
The corresponding RR for death within 30 days was 0.87 (95% CI, 0.77-0.99), and for major in-hospital bleeding it was 1.01 (95% CI, 0.86-1.18).
The analysis was adjusted for other medications received before cath-lab arrival, especially a long list of antiplatelets and non-heparin antithrombins. That strengthens the case for heparin pretreatment as an independent predictor of an open IRA at initial angiography, Mr. Emilsson said.
Comparisons of propensity-score–matched subgroups of the total cohort, conducted separately for the IRA-occlusion endpoint and the endpoints of 30-day mortality and major bleeding, produced similar results.
Some observational data suggest that antiplatelet pretreatment with a P2Y12 inhibitor may promote IRA patency on angiography after cath lab arrival, Dr. Rao observed. “This indicates that there probably is a role of earlier antithrombotic therapy in STEMI patients, but the randomized trials have not shown a consistent benefit,” he said, referring in particular to the ATLANTIC trial.
Mr. Emilsson and Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ESC CONGRESS 2022
PARADISE-MI results obscured as post hoc analysis finds flaws
A post hoc analysis of the PARADISE-MI trial, although not intended to alter the conclusions generated by the published data, suggests that clinically relevant benefits were obscured, providing the basis for recommending different analyses for future studies that are more suited to capture the most clinically significant endpoints.
“What these data show us is that we need clinical trial designs moving towards more pragmatic information that better reflect clinical practice,” reported Otavio Berwanger, MD, PhD, director of the Academic Research Organization at Hospital Israelita Albert Einstein, São Paulo, Brazil.
The reevaluation of the PARADISE-MI data, presented at the annual congress of the European Society of Cardiology in Barcelona, was based on a win ratio analysis and on the inclusion of investigator-reported endpoints, not just adjudicated events. Both appear to reveal clinically meaningful benefits not reflected in the published study, according to Dr. Berwanger.
In PARADISE-MI, which was published in the New England Journal of Medicine last year, more than 5,500 patients were randomized to the angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan or the ACE inhibitor ramipril after a myocardial infarction. A reduced left ventricular ejection fraction (LVEF), pulmonary congestion, or both were required for enrollment.
For the primary composite outcomes of death from cardiovascular (CV) causes or incident heart failure, the ARNI had a 10% numerical advantage, but it did not reach statistical significance (hazard ratio [HR], 0.90; P = .17).
“PARADISE-MI was a neutral trial. This post hoc analysis will not change that result,” Dr. Berwanger emphasized. However, the post hoc analysis does provide a basis for exploring why conventional trial designs might not be providing answers that are relevant and helpful for clinical practice.
New analysis provides positive trial result
When the data from PARADISE-MI are reevaluated in a hierarchical win ratio analysis with CV death serving as the most severe and important outcome, the principal conclusion changes. Whether events are reevaluated in this format by the clinical events committee (CEC) or by investigators, there is a greater number of total wins than total losses for the ARNI. Combined, sacubitril/valsartan was associated with a win ratio of 1.17 (95% confidence interval, 1.03-1.33; P = 0.015) over ramipril.
Using a sports analogy, Dr. Berwanger explained that the win ratio analysis divides the total number of wins to the total number of losses to provide a much more clinically relevant approach to keeping score. It also used a hierarchical analysis so that the most serious and important events are considered first.
In addition to CV death, this analysis included first hospitalization for heart failure and first outpatient heart failure events. CEC-defined events and events reported by investigators were evaluated separately.
The ARNI had more wins than losses in every category for all outcomes, whether CEC adjudicated or investigator reported, but most of this benefit was generated by the endpoint of CEC-adjudicated CV deaths. This accounted for 36.9% of all events (investigator-documented CV death accounted for 0.7%). This is important because PARADISE-MI, like many standard trials, was conducted on a time-to-primary event basis.
“In this type of analysis, the first event is what counts. Usually time-to-first-event analyses are dominated by nonfatal events,” Dr. Berwanger explained. He believes that placing more weight on the most serious events results in an emphasis on what outcomes are of greatest clinical interest.
In addition, Dr. Berwanger argued that it is important to consider investigator-reported events, not just CEC-adjudicated events. While adjudicated events improve the rigor of the data, Dr. Berwanger suggested it omits outcomes with which clinicians are most concerned.
Investigator, adjudicated outcomes differ
Again, using PARADISE-MI as an example, he reevaluated the primary outcome based on investigator reports. When investigator-reported events are included, the number of events increased in both the ARNI (443 vs. 338) and ramipril (516 vs. 373) arms, but the advantage of the ARNI over the ACE inhibitors now reached statistical significance (HR, 0.85; P = .01).
“The data suggest that maybe we should find definitions for adjudication that are closer to clinical judgment in the real world and clinical practice,” Dr. Berwanger said.
One possible explanation for the neutral result in PARADISE-MI is that benefit of an ARNI over an ACE inhibitor would only be expected in those at risk for progressive left ventricular dysfunction, and it is likely that a substantial proportion of patients enrolled in this trial recovered, according to Johann Bauersachs, MD, PhD, professor and head of cardiology at Hannover (Germany) Medical School.
“You cannot predict which patients with reduced LV function following an MI will go on to chronic remodeling and which will recover,” said Dr. Bauersachs, who was an ESC-invited discussant of Dr. Berwanger’s post hoc analysis.
He agreed that Dr. Berwanger has raised several important issues in standard trial design that might have prevented PARADISE-MI from showing a benefit from an ARNI, but he pointed out that there are other potential issues, such as the low use of mineralocorticoid antagonists in PARADISE-MI, that may have skewed results.
However, he agreed generally with the premise that there is a need for trial design likely to generate more clinically useful information.
“We have now seen the win-ratio approach used in several studies,” said Dr. Bauersachs, citing in particular the EMPULSE trial presented at the 2022 meeting of the American College of Cardiology. “It is a very useful tool, and I think we will be seeing it used more in the future.”
However, he indicated that the issues raised by Dr. Berwanger are not necessarily easily resolved. Dr. Bauersachs endorsed the effort to consider trial designs that generate data that are more immediately clinically applicable but suggested that different types of designs may be required for different types of clinical questions.
Dr. Berwanger reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Pfizer, Servier, and Novartis, which provided funding for the PARADISE-MI trial. Dr. Bauersachs reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Boehringer Ingelheim, Cardior, Corvia, CVRx, Novartis, Pfizer, Vifor, and Zoll.
A post hoc analysis of the PARADISE-MI trial, although not intended to alter the conclusions generated by the published data, suggests that clinically relevant benefits were obscured, providing the basis for recommending different analyses for future studies that are more suited to capture the most clinically significant endpoints.
“What these data show us is that we need clinical trial designs moving towards more pragmatic information that better reflect clinical practice,” reported Otavio Berwanger, MD, PhD, director of the Academic Research Organization at Hospital Israelita Albert Einstein, São Paulo, Brazil.
The reevaluation of the PARADISE-MI data, presented at the annual congress of the European Society of Cardiology in Barcelona, was based on a win ratio analysis and on the inclusion of investigator-reported endpoints, not just adjudicated events. Both appear to reveal clinically meaningful benefits not reflected in the published study, according to Dr. Berwanger.
In PARADISE-MI, which was published in the New England Journal of Medicine last year, more than 5,500 patients were randomized to the angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan or the ACE inhibitor ramipril after a myocardial infarction. A reduced left ventricular ejection fraction (LVEF), pulmonary congestion, or both were required for enrollment.
For the primary composite outcomes of death from cardiovascular (CV) causes or incident heart failure, the ARNI had a 10% numerical advantage, but it did not reach statistical significance (hazard ratio [HR], 0.90; P = .17).
“PARADISE-MI was a neutral trial. This post hoc analysis will not change that result,” Dr. Berwanger emphasized. However, the post hoc analysis does provide a basis for exploring why conventional trial designs might not be providing answers that are relevant and helpful for clinical practice.
New analysis provides positive trial result
When the data from PARADISE-MI are reevaluated in a hierarchical win ratio analysis with CV death serving as the most severe and important outcome, the principal conclusion changes. Whether events are reevaluated in this format by the clinical events committee (CEC) or by investigators, there is a greater number of total wins than total losses for the ARNI. Combined, sacubitril/valsartan was associated with a win ratio of 1.17 (95% confidence interval, 1.03-1.33; P = 0.015) over ramipril.
Using a sports analogy, Dr. Berwanger explained that the win ratio analysis divides the total number of wins to the total number of losses to provide a much more clinically relevant approach to keeping score. It also used a hierarchical analysis so that the most serious and important events are considered first.
In addition to CV death, this analysis included first hospitalization for heart failure and first outpatient heart failure events. CEC-defined events and events reported by investigators were evaluated separately.
The ARNI had more wins than losses in every category for all outcomes, whether CEC adjudicated or investigator reported, but most of this benefit was generated by the endpoint of CEC-adjudicated CV deaths. This accounted for 36.9% of all events (investigator-documented CV death accounted for 0.7%). This is important because PARADISE-MI, like many standard trials, was conducted on a time-to-primary event basis.
“In this type of analysis, the first event is what counts. Usually time-to-first-event analyses are dominated by nonfatal events,” Dr. Berwanger explained. He believes that placing more weight on the most serious events results in an emphasis on what outcomes are of greatest clinical interest.
In addition, Dr. Berwanger argued that it is important to consider investigator-reported events, not just CEC-adjudicated events. While adjudicated events improve the rigor of the data, Dr. Berwanger suggested it omits outcomes with which clinicians are most concerned.
Investigator, adjudicated outcomes differ
Again, using PARADISE-MI as an example, he reevaluated the primary outcome based on investigator reports. When investigator-reported events are included, the number of events increased in both the ARNI (443 vs. 338) and ramipril (516 vs. 373) arms, but the advantage of the ARNI over the ACE inhibitors now reached statistical significance (HR, 0.85; P = .01).
“The data suggest that maybe we should find definitions for adjudication that are closer to clinical judgment in the real world and clinical practice,” Dr. Berwanger said.
One possible explanation for the neutral result in PARADISE-MI is that benefit of an ARNI over an ACE inhibitor would only be expected in those at risk for progressive left ventricular dysfunction, and it is likely that a substantial proportion of patients enrolled in this trial recovered, according to Johann Bauersachs, MD, PhD, professor and head of cardiology at Hannover (Germany) Medical School.
“You cannot predict which patients with reduced LV function following an MI will go on to chronic remodeling and which will recover,” said Dr. Bauersachs, who was an ESC-invited discussant of Dr. Berwanger’s post hoc analysis.
He agreed that Dr. Berwanger has raised several important issues in standard trial design that might have prevented PARADISE-MI from showing a benefit from an ARNI, but he pointed out that there are other potential issues, such as the low use of mineralocorticoid antagonists in PARADISE-MI, that may have skewed results.
However, he agreed generally with the premise that there is a need for trial design likely to generate more clinically useful information.
“We have now seen the win-ratio approach used in several studies,” said Dr. Bauersachs, citing in particular the EMPULSE trial presented at the 2022 meeting of the American College of Cardiology. “It is a very useful tool, and I think we will be seeing it used more in the future.”
However, he indicated that the issues raised by Dr. Berwanger are not necessarily easily resolved. Dr. Bauersachs endorsed the effort to consider trial designs that generate data that are more immediately clinically applicable but suggested that different types of designs may be required for different types of clinical questions.
Dr. Berwanger reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Pfizer, Servier, and Novartis, which provided funding for the PARADISE-MI trial. Dr. Bauersachs reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Boehringer Ingelheim, Cardior, Corvia, CVRx, Novartis, Pfizer, Vifor, and Zoll.
A post hoc analysis of the PARADISE-MI trial, although not intended to alter the conclusions generated by the published data, suggests that clinically relevant benefits were obscured, providing the basis for recommending different analyses for future studies that are more suited to capture the most clinically significant endpoints.
“What these data show us is that we need clinical trial designs moving towards more pragmatic information that better reflect clinical practice,” reported Otavio Berwanger, MD, PhD, director of the Academic Research Organization at Hospital Israelita Albert Einstein, São Paulo, Brazil.
The reevaluation of the PARADISE-MI data, presented at the annual congress of the European Society of Cardiology in Barcelona, was based on a win ratio analysis and on the inclusion of investigator-reported endpoints, not just adjudicated events. Both appear to reveal clinically meaningful benefits not reflected in the published study, according to Dr. Berwanger.
In PARADISE-MI, which was published in the New England Journal of Medicine last year, more than 5,500 patients were randomized to the angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan or the ACE inhibitor ramipril after a myocardial infarction. A reduced left ventricular ejection fraction (LVEF), pulmonary congestion, or both were required for enrollment.
For the primary composite outcomes of death from cardiovascular (CV) causes or incident heart failure, the ARNI had a 10% numerical advantage, but it did not reach statistical significance (hazard ratio [HR], 0.90; P = .17).
“PARADISE-MI was a neutral trial. This post hoc analysis will not change that result,” Dr. Berwanger emphasized. However, the post hoc analysis does provide a basis for exploring why conventional trial designs might not be providing answers that are relevant and helpful for clinical practice.
New analysis provides positive trial result
When the data from PARADISE-MI are reevaluated in a hierarchical win ratio analysis with CV death serving as the most severe and important outcome, the principal conclusion changes. Whether events are reevaluated in this format by the clinical events committee (CEC) or by investigators, there is a greater number of total wins than total losses for the ARNI. Combined, sacubitril/valsartan was associated with a win ratio of 1.17 (95% confidence interval, 1.03-1.33; P = 0.015) over ramipril.
Using a sports analogy, Dr. Berwanger explained that the win ratio analysis divides the total number of wins to the total number of losses to provide a much more clinically relevant approach to keeping score. It also used a hierarchical analysis so that the most serious and important events are considered first.
In addition to CV death, this analysis included first hospitalization for heart failure and first outpatient heart failure events. CEC-defined events and events reported by investigators were evaluated separately.
The ARNI had more wins than losses in every category for all outcomes, whether CEC adjudicated or investigator reported, but most of this benefit was generated by the endpoint of CEC-adjudicated CV deaths. This accounted for 36.9% of all events (investigator-documented CV death accounted for 0.7%). This is important because PARADISE-MI, like many standard trials, was conducted on a time-to-primary event basis.
“In this type of analysis, the first event is what counts. Usually time-to-first-event analyses are dominated by nonfatal events,” Dr. Berwanger explained. He believes that placing more weight on the most serious events results in an emphasis on what outcomes are of greatest clinical interest.
In addition, Dr. Berwanger argued that it is important to consider investigator-reported events, not just CEC-adjudicated events. While adjudicated events improve the rigor of the data, Dr. Berwanger suggested it omits outcomes with which clinicians are most concerned.
Investigator, adjudicated outcomes differ
Again, using PARADISE-MI as an example, he reevaluated the primary outcome based on investigator reports. When investigator-reported events are included, the number of events increased in both the ARNI (443 vs. 338) and ramipril (516 vs. 373) arms, but the advantage of the ARNI over the ACE inhibitors now reached statistical significance (HR, 0.85; P = .01).
“The data suggest that maybe we should find definitions for adjudication that are closer to clinical judgment in the real world and clinical practice,” Dr. Berwanger said.
One possible explanation for the neutral result in PARADISE-MI is that benefit of an ARNI over an ACE inhibitor would only be expected in those at risk for progressive left ventricular dysfunction, and it is likely that a substantial proportion of patients enrolled in this trial recovered, according to Johann Bauersachs, MD, PhD, professor and head of cardiology at Hannover (Germany) Medical School.
“You cannot predict which patients with reduced LV function following an MI will go on to chronic remodeling and which will recover,” said Dr. Bauersachs, who was an ESC-invited discussant of Dr. Berwanger’s post hoc analysis.
He agreed that Dr. Berwanger has raised several important issues in standard trial design that might have prevented PARADISE-MI from showing a benefit from an ARNI, but he pointed out that there are other potential issues, such as the low use of mineralocorticoid antagonists in PARADISE-MI, that may have skewed results.
However, he agreed generally with the premise that there is a need for trial design likely to generate more clinically useful information.
“We have now seen the win-ratio approach used in several studies,” said Dr. Bauersachs, citing in particular the EMPULSE trial presented at the 2022 meeting of the American College of Cardiology. “It is a very useful tool, and I think we will be seeing it used more in the future.”
However, he indicated that the issues raised by Dr. Berwanger are not necessarily easily resolved. Dr. Bauersachs endorsed the effort to consider trial designs that generate data that are more immediately clinically applicable but suggested that different types of designs may be required for different types of clinical questions.
Dr. Berwanger reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Pfizer, Servier, and Novartis, which provided funding for the PARADISE-MI trial. Dr. Bauersachs reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Boehringer Ingelheim, Cardior, Corvia, CVRx, Novartis, Pfizer, Vifor, and Zoll.
FROM ESC CONGRESS 2022