User login
Characteristics of Matched vs Nonmatched Dermatology Applicants
Dermatology residency continues to be one of the most competitive specialties, with a match rate of 84.7% for US allopathic seniors in the 2019-2020 academic year.1 In the 2019-2020 cycle, dermatology applicants were tied with plastic surgery for the highest median US Medical Licensing Examination (USMLE) Step 1 score compared with other specialties, which suggests that the top medical students are applying, yet only approximately 5 of 6 students are matching.
Factors that have been cited with successful dermatology matching include USMLE Step 1 and Step 2 Clinical Knowledge (CK) scores,2 research accomplishments,3 letters of recommendation,4 medical school performance, personal statement, grades in required clerkships, and volunteer/extracurricular experiences, among others.5
The National Resident Matching Program (NRMP) publishes data each year regarding different academic factors—USMLE scores; number of abstracts, presentations, and papers; work, volunteer, and research experiences—and compares the mean between matched and nonmatched applicants.1 However, the USMLE does not report any demographic information of the applicants and the implication it has for matching. Additionally, the number of couples participating in the couples match continues to increase each year. In the 2019-2020 cycle, 1224 couples participated in the couples match.1 However, NRMP reports only limited data regarding the couples match, and it is not specialty specific.
We aimed to determine the characteristics of matched vs nonmatched dermatology applicants. Secondarily, we aimed to determine any differences among demographics regarding matching rates, academic performance, and research publications. We also aimed to characterize the strategy and outcomes of applicants that couples matched.
Materials and Methods
The Mayo Clinic institutional review board deemed this study exempt. All applicants who applied to Mayo Clinic dermatology residency in Scottsdale, Arizona, during the 2018-2019 cycle were emailed an initial survey (N=475) before Match Day that obtained demographic information, geographic information, gap-year information, USMLE Step 1 score, publications, medical school grades, number of away rotations, and number of interviews. A follow-up survey gathering match data and couples matching data was sent to the applicants who completed the first survey on Match Day. The survey was repeated for the 2019-2020 cycle. In the second survey, Step 2 CK data were obtained. The survey was sent to 629 applicants who applied to Mayo Clinic dermatology residencies in Arizona, Minnesota, and Florida to include a broader group of applicants. For publications, applicants were asked to count only published or accepted manuscripts, not abstracts, posters, conference presentations, or submitted manuscripts. Applicants who did not respond to the second survey (match data) were not included in that part of the analysis. One survey was excluded because of implausible answers (eg, scores outside of range for USMLE Step scores).
Statistical Analysis—For statistical analyses, the applicants from both applications cycles were combined. Descriptive statistics were reported in the form of mean, median, or counts (percentages), as applicable. Means were compared using 2-sided t tests. Group comparisons were examined using χ2 tests for categorical variables. Statistical analyses were performed using the BlueSky Statistics version 6.30. P<.05 was considered significant.
Results
In 2019, a total of 149 applicants completed the initial survey (31.4% response rate), and 112 completed the follow-up survey (75.2% response rate). In 2020, a total of 142 applicants completed the initial survey (22.6% response rate), and 124 completed the follow-up survey (87.3% response rate). Combining the 2 years, after removing 1 survey with implausible answers, there were 290 respondents from the initial survey and 235 from the follow-up survey. The median (SD) age for the total applicants over both years was 27 (3.0) years, and 180 applicants were female (61.9%).
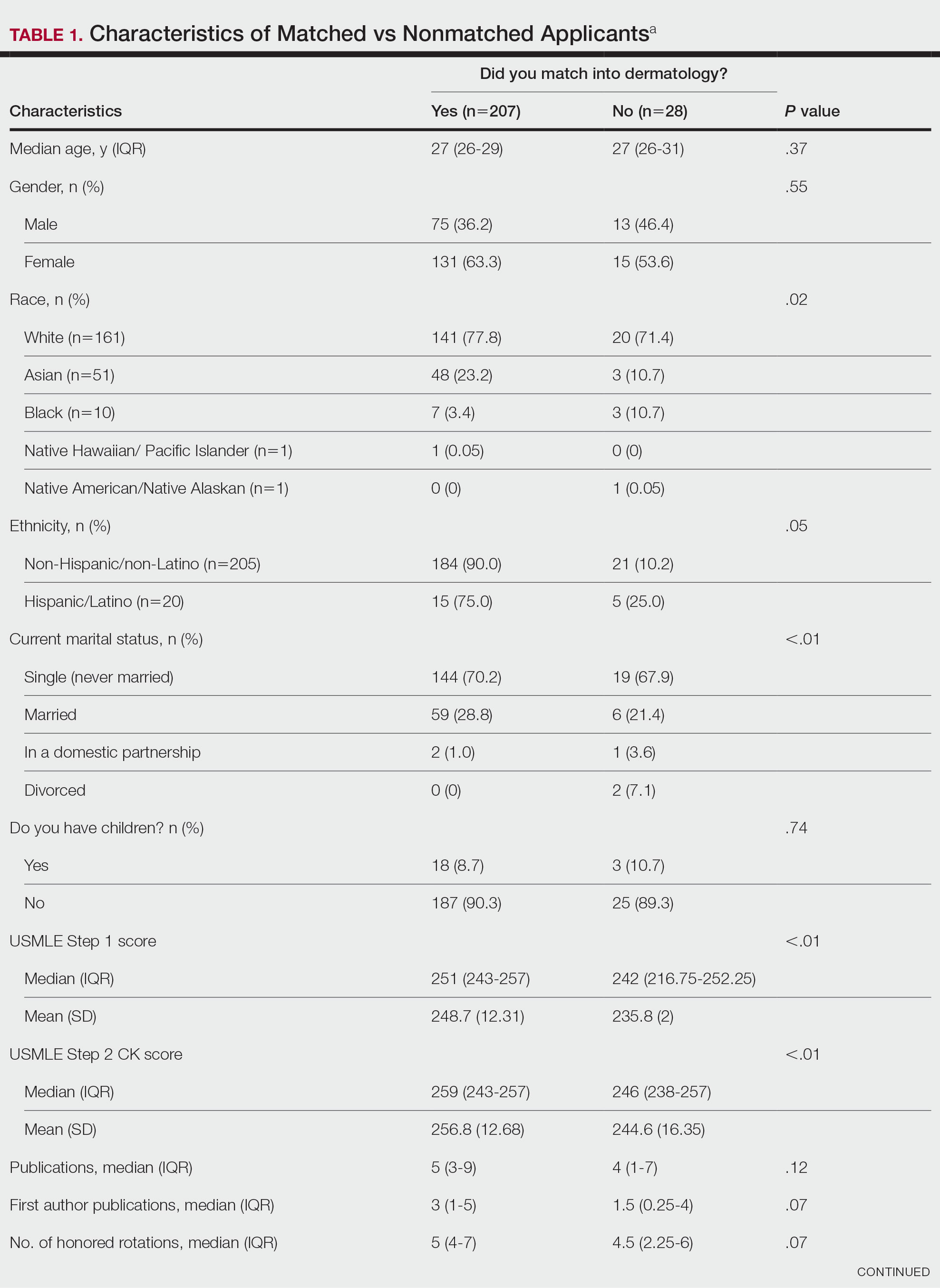
USMLE Scores—The median USMLE Step 1 score was 250, and scores ranged from 196 to 271. The median USMLE Step 2 CK score was 257, and scores ranged from 213 to 281. Higher USMLE Step 1 and Step 2 CK scores and more interviews were associated with higher match rates (Table 1). In addition, students with a dermatology program at their medical school were more likely to match than those without a home dermatology program.
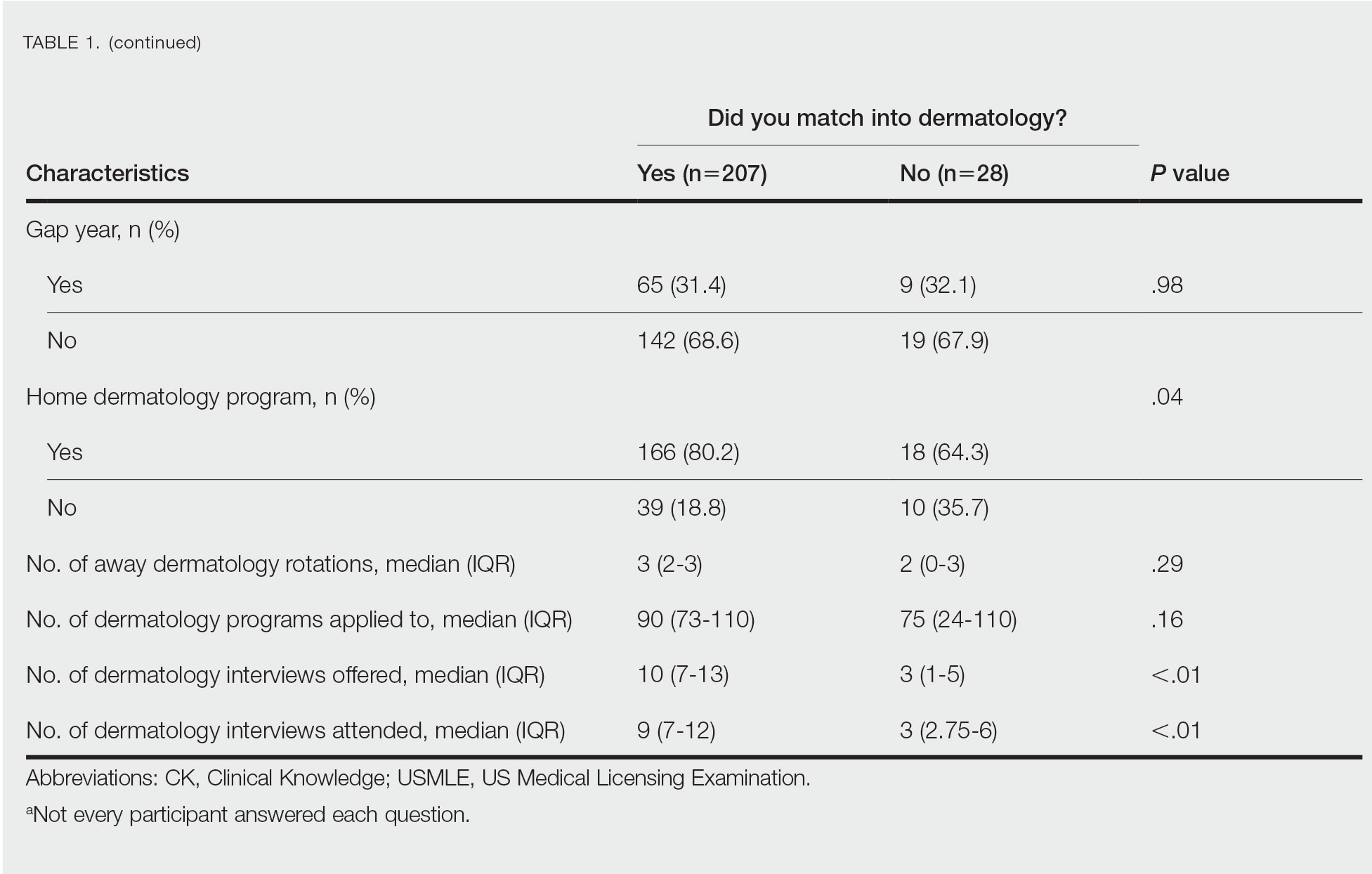
Gender Differences—There were 180 females and 110 males who completed the surveys. Males and females had similar match rates (85.2% vs 89.0%; P=.39)(Table 2).
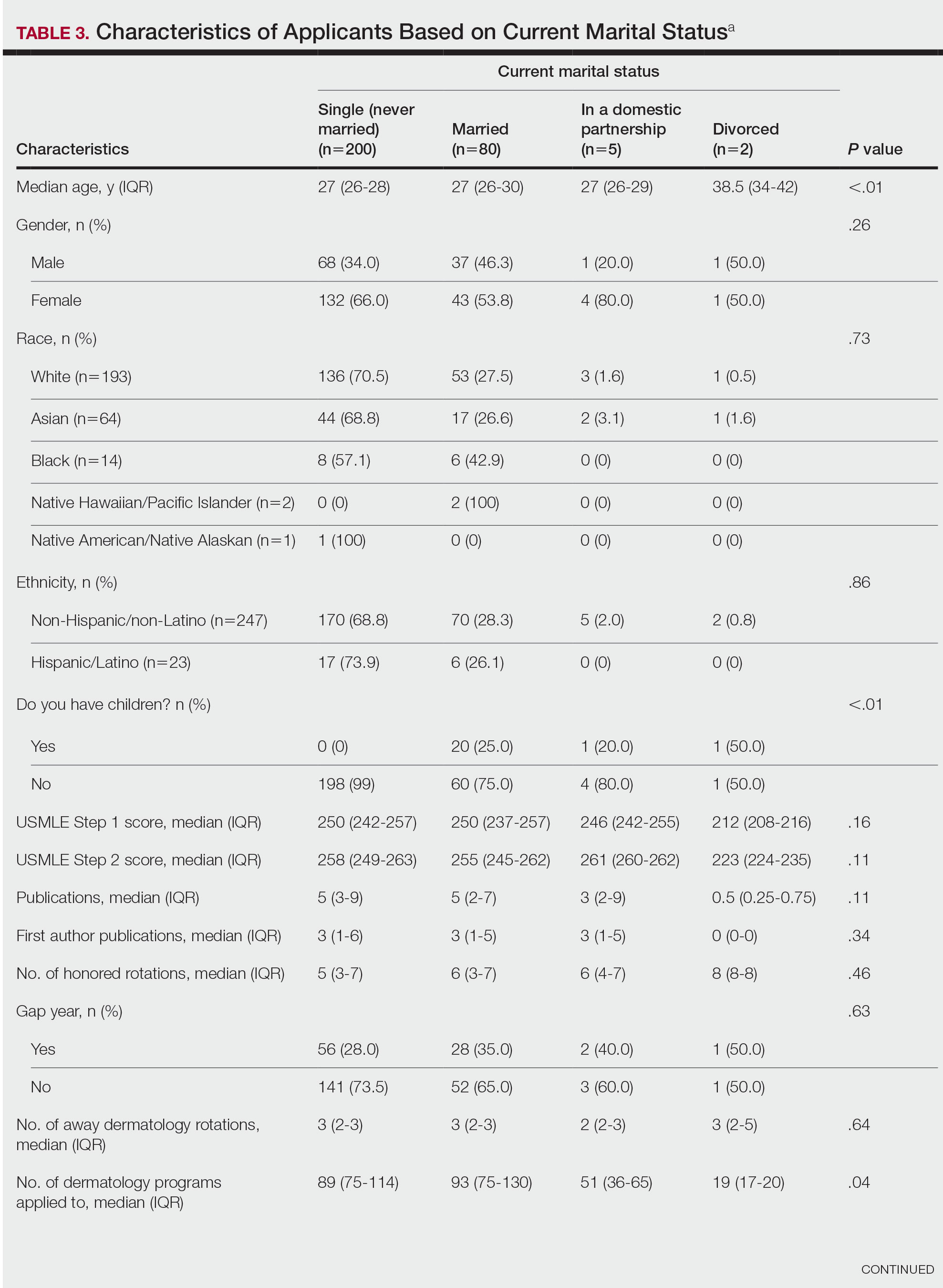
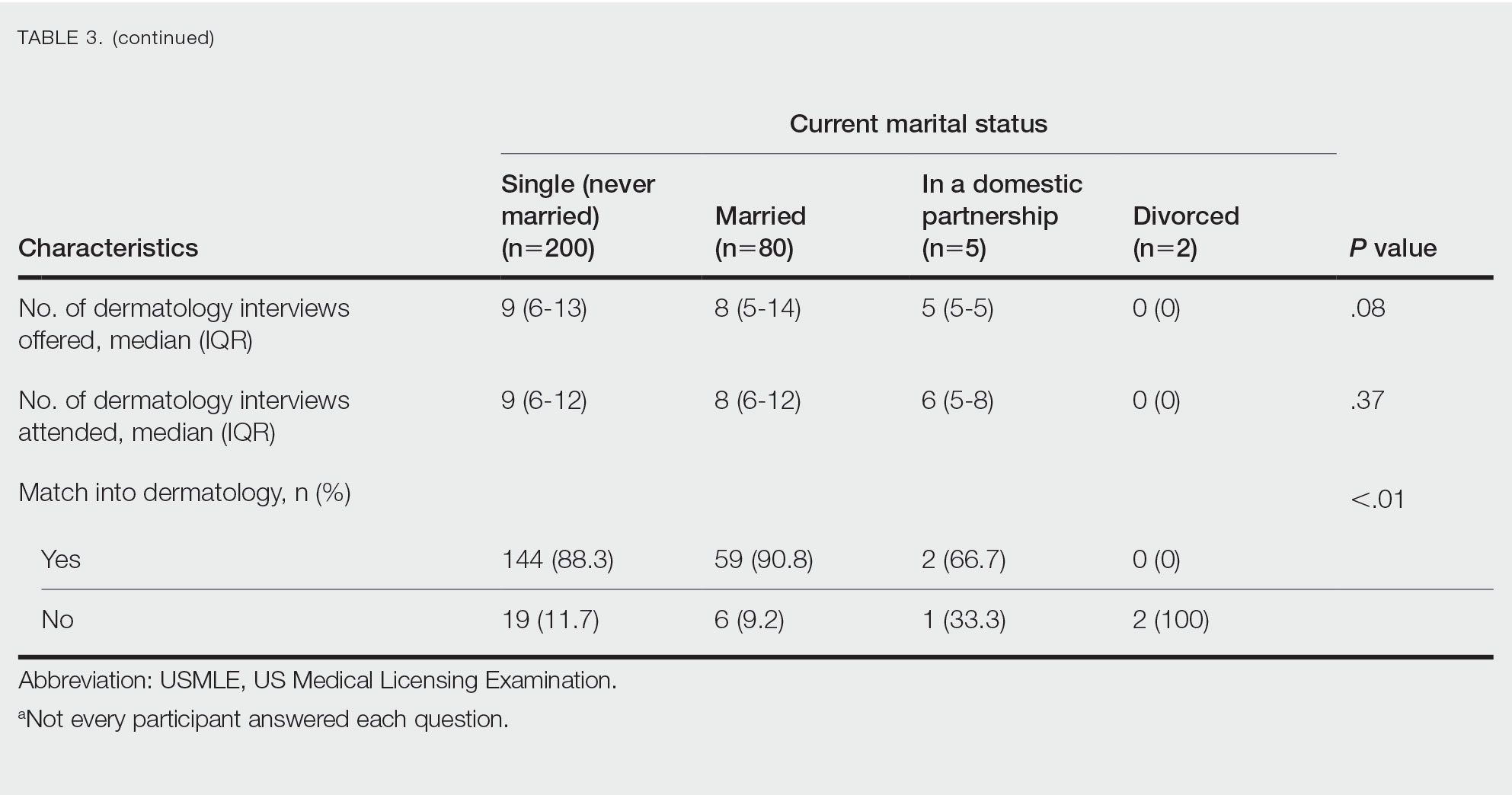
Family Life—In comparing marital status, applicants who were divorced had a higher median age (38.5 years) compared with applicants who were single, married, or in a domestic partnership (all 27 years; P<.01). Differences are outlined in Table 3.
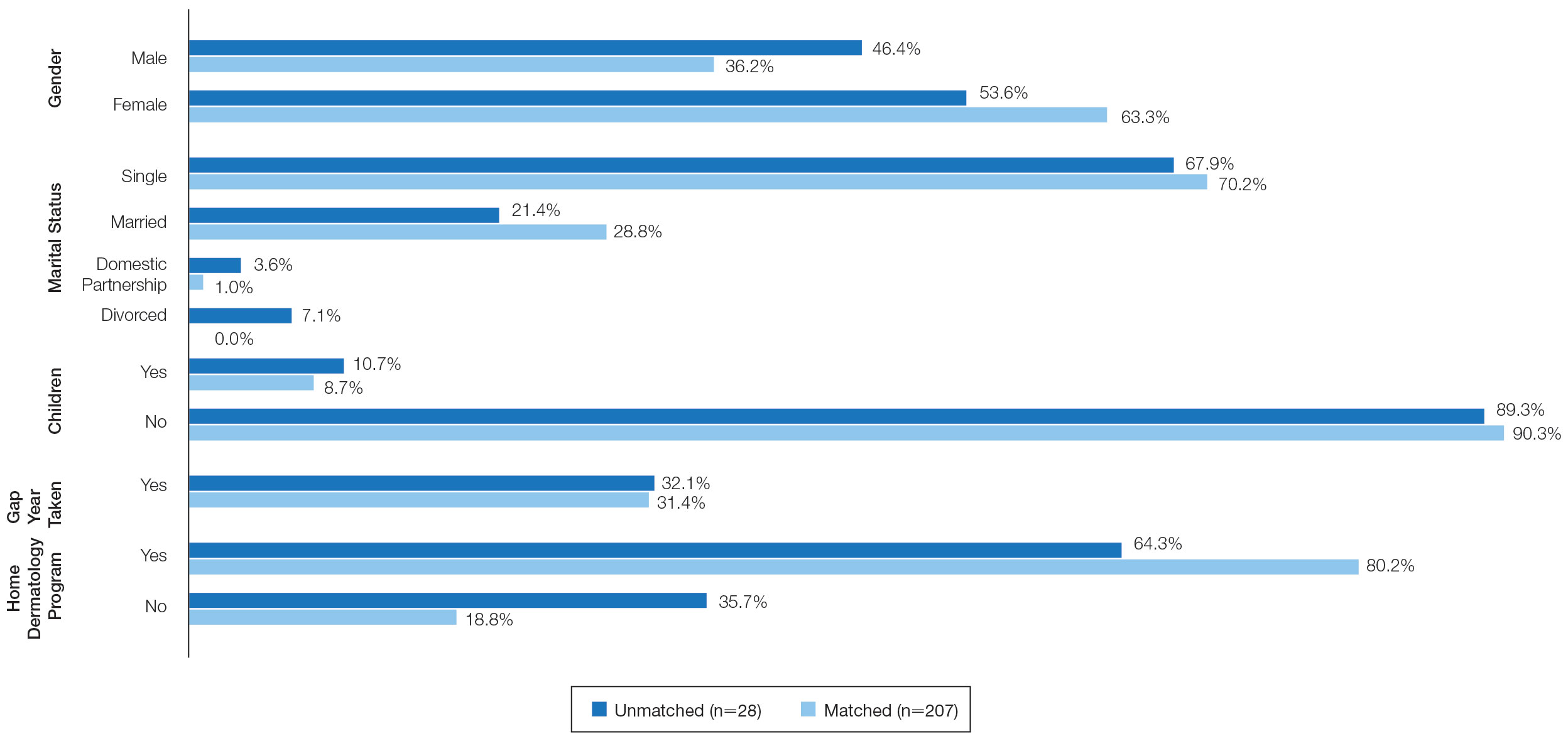
On average, applicants with children (n=27 [15 male, 12 female]; P=.13) were 3 years older than those without (30.5 vs 27; P<.01) and were more likely to be married (88.9% vs 21.5%; P<.01). Applicants with children had a mean USMLE Step 1 score of 241 compared to 251 for those without children (P=.02) and a mean USMLE Step 2 CK score of 246 compared to 258 for those without children (P<.01). Applicants with children had similar debt, number of publications, number of honored rotations, and match rates compared to applicants without children (Figure).
Couples Match—Seventeen individuals in our survey participated in the couples match (7.8%), and all 17 (100%) matched into dermatology. The mean age was 26.7 years, 12 applicants were female, 2 applicants were married, and 1 applicant had children. The mean number of interviews offered was 13.6, and the mean number of interviews attended was 11.3. This was higher than participants who were not couples matching (13.6 vs 9.8 [P=.02] and 11.3 vs 8.9 [P=.04], respectively). Applicants and their partners applied to programs and received interviews in a mean of 10 cities. Sixteen applicants reported that they contacted programs where their partner had interview offers. All participants’ rank lists included programs located in different cities than their partners’ ranked programs, and all but 1 participant ranked programs located in a different state than their partners’ ranked programs. Fifteen participants had options in their rank list for the applicant not to match, even if the partner would match. Similarly, 12 had the option for the applicant to match, even if the partner would not match. Fourteen (82.4%) matched at the same institution as their significant other. Three (17.6%) applicants matched to a program in a different state than the partner’s matched program. Two (11.8%) participants felt their relationship with their partner suffered because of the match, and 1 (5.9%) applicant was undetermined. One applicant described their relationship suffering from “unnecessary tension and anxiety” and noted “difficult conversations” about potentially matching into dermatology in a different location from their partner that could have been “devastating and not something [he or she] should have to choose.”
Comment
Factors for Matching in Dermatology—In our survey, we found the statistically significant factors of matching into dermatology included high USMLE Step 1 and Step 2 CK scores (P<.01), having a home dermatology program (P=.04), and attending a higher number of dermatology interviews (P<.01). These data are similar to NRMP results1; however, the higher likelihood of matching if the medical school has a home dermatology program has not been reported. This finding could be due to multiple factors such as students have less access to academic dermatologists for research projects, letters of recommendations, mentorship, and clinical rotations.
Gender and having children were factors that had no correlation with the match rate. There was a statistical difference of matching based on marital status (P<.01), but this is likely due to the low number of applicants in the divorced category. There were differences among demographics with USMLE Step 1 and Step 2 CK scores, which is a known factor in matching.1,2 Applicants with children had lower USMLE Step 1 and Step 2 CK scores compared to applicants without children. Females also had lower median USMLE Step 1 scores compared to males. This finding may serve as a reminder to programs when comparing USMLE Step examination scores that demographic factors may play a role. The race and ethnicity of applicants likely play a role. It has been reported that underrepresented minorities had lower match rates than White and Asian applicants in dermatology.6 There have been several published articles discussing the lack of diversity in dermatology, with a call to action.7-9
Factors for Couples Matching—The number of applicants participating in the couples match continues to increase yearly. The NMRP does publish data regarding “successful” couples matching but does not specify how many couples match together. There also is little published regarding advice for participation in the couples match. Although we had a limited number of couples that participated in the match, it is interesting to note they had similar strategies, including contacting programs at institutions that had offered interviews to their partners. This strategy may be effective, as dermatology programs offer interviews relatively late compared with other specialties.5 Additionally, this strategy may increase the number of interviews offered and received, as evidenced by the higher number of interviews offered compared with those who were not couples matching. Additionally, this survey highlights the sacrifice often needed by couples in the couples match as revealed by the inclusion of rank-list options in which the couples reside long distance or in which 1 partner does not match. This information may be helpful to applicants who are planning a strategy for the couples match in dermatology. Although this study does not encompass all dermatology applicants in the 2019-2020 cycle, we do believe it may be representative. The USMLE Step 1 scores in this study were similar to the published NRMP data.1,10 According to NRMP data from the 2019-2020 cycle, the mean USMLE Step 1 score was 248 for matched applicants and 239 for unmatched.1 The NRMP reported the mean USMLE Step 2 CK score for matched was 256 and 248 for unmatched, which also is similar to our data. The NRMP reported the mean number of programs ranked was 9.9 for matched and 4.5 for unmatched applicants.1 Again, our data were similar for number of dermatology interviews attended.
Limitations—There are limitations to this study. The main limitation is that the survey is from a single institution and had a limited number of respondents. Given the nature of the study, the accuracy of the data is dependent on the applicants’ honesty in self-reporting academic performance and other variables. There also may be a selection bias given the low response rate. The subanalyses—children and couples matching—were underpowered with the limited number of participants. Further studies that include multiple residency programs and multiple years could be helpful to provide more power and less risk of bias. We did not gather information such as the Medical Student Performance Evaluation letter, letters of recommendation, or personal statements, which do play an important role in the assessment of an applicant. However, because the applicants completed these surveys, and given these are largely blinded to applicants, we did not feel the applicants could accurately respond to those aspects of the application.
Conclusion
Our survey finds that factors associated with matching included a higher USMLE Step 1 score, having a home dermatology program, and a higher number of interviews offered and attended. Some demographics had varying USMLE Step 1 scores but similar match rates.
- National Resident Matching Program. Results and Data: 2020 Main Residency Match. National Resident Matching Program; May 2020. Accessed January 9, 2023. https://www.nrmp.org/wp-content/uploads/2021/12/MM_Results_and-Data_2020-1.pdf
- Gauer JL, Jackson JB. The association of USMLE Step 1 and Step 2 CK scores with residency match specialty and location. Med Educ Online. 2017;22:1358579.
- Wang JV, Keller M. Pressure to publish for residency applicants in dermatology. Dermatol Online J. 2016;22:13030/qt56x1t7ww.
- Wang RF, Zhang M, Kaffenberger JA. Does the dermatology standardized letter of recommendation alter applicants’ chances of matching into residency. J Am Acad Dermatol. 2017;77:e139-e140.
- National Resident Matching Program, Data Release and Research Committee: results of the 2018 NRMP Program Director Survey. Accessed December 19, 2022. https://www.nrmp.org/wp-content/uploads/2021/07/NRMP-2018-Program-Director-Survey-for-WWW.pdf
- Costello CM, Harvey JA, Besch-Stokes JG, et al. The role of race and ethnicity in the dermatology applicant match process. J Natl Med Assoc. 2022;113:666-670.
- Chen A, Shinkai K. Rethinking how we select dermatology applicants-turning the tide. JAMA Dermatol. 2017;153:259-260.
- Pandya AG, Alexis AF, Berger TG, et al. Increasing racial and ethnic diversity in dermatology: a call to action. J Am Acad Dermatol. 2016;74:584-587.
- Van Voorhees AS, Enos CW. Diversity in dermatology residency programs. J Investig Dermatol Symp Proc. 2017;18:S46-S49.
- National Resident Matching Program. Charting outcomes in the match: U.S. allopathic seniors. Characteristics of U.S. allopathic seniors who matched to their preferred specialty in the 2018 main residency match. 2nd ed. Accessed December 19, 2022. https://www.nrmp.org/wp-content/uploads/2021/07/Charting-Outcomes-in-the-Match-2018_Seniors-1.pdf
Dermatology residency continues to be one of the most competitive specialties, with a match rate of 84.7% for US allopathic seniors in the 2019-2020 academic year.1 In the 2019-2020 cycle, dermatology applicants were tied with plastic surgery for the highest median US Medical Licensing Examination (USMLE) Step 1 score compared with other specialties, which suggests that the top medical students are applying, yet only approximately 5 of 6 students are matching.
Factors that have been cited with successful dermatology matching include USMLE Step 1 and Step 2 Clinical Knowledge (CK) scores,2 research accomplishments,3 letters of recommendation,4 medical school performance, personal statement, grades in required clerkships, and volunteer/extracurricular experiences, among others.5
The National Resident Matching Program (NRMP) publishes data each year regarding different academic factors—USMLE scores; number of abstracts, presentations, and papers; work, volunteer, and research experiences—and compares the mean between matched and nonmatched applicants.1 However, the USMLE does not report any demographic information of the applicants and the implication it has for matching. Additionally, the number of couples participating in the couples match continues to increase each year. In the 2019-2020 cycle, 1224 couples participated in the couples match.1 However, NRMP reports only limited data regarding the couples match, and it is not specialty specific.
We aimed to determine the characteristics of matched vs nonmatched dermatology applicants. Secondarily, we aimed to determine any differences among demographics regarding matching rates, academic performance, and research publications. We also aimed to characterize the strategy and outcomes of applicants that couples matched.
Materials and Methods
The Mayo Clinic institutional review board deemed this study exempt. All applicants who applied to Mayo Clinic dermatology residency in Scottsdale, Arizona, during the 2018-2019 cycle were emailed an initial survey (N=475) before Match Day that obtained demographic information, geographic information, gap-year information, USMLE Step 1 score, publications, medical school grades, number of away rotations, and number of interviews. A follow-up survey gathering match data and couples matching data was sent to the applicants who completed the first survey on Match Day. The survey was repeated for the 2019-2020 cycle. In the second survey, Step 2 CK data were obtained. The survey was sent to 629 applicants who applied to Mayo Clinic dermatology residencies in Arizona, Minnesota, and Florida to include a broader group of applicants. For publications, applicants were asked to count only published or accepted manuscripts, not abstracts, posters, conference presentations, or submitted manuscripts. Applicants who did not respond to the second survey (match data) were not included in that part of the analysis. One survey was excluded because of implausible answers (eg, scores outside of range for USMLE Step scores).
Statistical Analysis—For statistical analyses, the applicants from both applications cycles were combined. Descriptive statistics were reported in the form of mean, median, or counts (percentages), as applicable. Means were compared using 2-sided t tests. Group comparisons were examined using χ2 tests for categorical variables. Statistical analyses were performed using the BlueSky Statistics version 6.30. P<.05 was considered significant.
Results
In 2019, a total of 149 applicants completed the initial survey (31.4% response rate), and 112 completed the follow-up survey (75.2% response rate). In 2020, a total of 142 applicants completed the initial survey (22.6% response rate), and 124 completed the follow-up survey (87.3% response rate). Combining the 2 years, after removing 1 survey with implausible answers, there were 290 respondents from the initial survey and 235 from the follow-up survey. The median (SD) age for the total applicants over both years was 27 (3.0) years, and 180 applicants were female (61.9%).
USMLE Scores—The median USMLE Step 1 score was 250, and scores ranged from 196 to 271. The median USMLE Step 2 CK score was 257, and scores ranged from 213 to 281. Higher USMLE Step 1 and Step 2 CK scores and more interviews were associated with higher match rates (Table 1). In addition, students with a dermatology program at their medical school were more likely to match than those without a home dermatology program.


Gender Differences—There were 180 females and 110 males who completed the surveys. Males and females had similar match rates (85.2% vs 89.0%; P=.39)(Table 2).

Family Life—In comparing marital status, applicants who were divorced had a higher median age (38.5 years) compared with applicants who were single, married, or in a domestic partnership (all 27 years; P<.01). Differences are outlined in Table 3.


On average, applicants with children (n=27 [15 male, 12 female]; P=.13) were 3 years older than those without (30.5 vs 27; P<.01) and were more likely to be married (88.9% vs 21.5%; P<.01). Applicants with children had a mean USMLE Step 1 score of 241 compared to 251 for those without children (P=.02) and a mean USMLE Step 2 CK score of 246 compared to 258 for those without children (P<.01). Applicants with children had similar debt, number of publications, number of honored rotations, and match rates compared to applicants without children (Figure).

Couples Match—Seventeen individuals in our survey participated in the couples match (7.8%), and all 17 (100%) matched into dermatology. The mean age was 26.7 years, 12 applicants were female, 2 applicants were married, and 1 applicant had children. The mean number of interviews offered was 13.6, and the mean number of interviews attended was 11.3. This was higher than participants who were not couples matching (13.6 vs 9.8 [P=.02] and 11.3 vs 8.9 [P=.04], respectively). Applicants and their partners applied to programs and received interviews in a mean of 10 cities. Sixteen applicants reported that they contacted programs where their partner had interview offers. All participants’ rank lists included programs located in different cities than their partners’ ranked programs, and all but 1 participant ranked programs located in a different state than their partners’ ranked programs. Fifteen participants had options in their rank list for the applicant not to match, even if the partner would match. Similarly, 12 had the option for the applicant to match, even if the partner would not match. Fourteen (82.4%) matched at the same institution as their significant other. Three (17.6%) applicants matched to a program in a different state than the partner’s matched program. Two (11.8%) participants felt their relationship with their partner suffered because of the match, and 1 (5.9%) applicant was undetermined. One applicant described their relationship suffering from “unnecessary tension and anxiety” and noted “difficult conversations” about potentially matching into dermatology in a different location from their partner that could have been “devastating and not something [he or she] should have to choose.”
Comment
Factors for Matching in Dermatology—In our survey, we found the statistically significant factors of matching into dermatology included high USMLE Step 1 and Step 2 CK scores (P<.01), having a home dermatology program (P=.04), and attending a higher number of dermatology interviews (P<.01). These data are similar to NRMP results1; however, the higher likelihood of matching if the medical school has a home dermatology program has not been reported. This finding could be due to multiple factors such as students have less access to academic dermatologists for research projects, letters of recommendations, mentorship, and clinical rotations.
Gender and having children were factors that had no correlation with the match rate. There was a statistical difference of matching based on marital status (P<.01), but this is likely due to the low number of applicants in the divorced category. There were differences among demographics with USMLE Step 1 and Step 2 CK scores, which is a known factor in matching.1,2 Applicants with children had lower USMLE Step 1 and Step 2 CK scores compared to applicants without children. Females also had lower median USMLE Step 1 scores compared to males. This finding may serve as a reminder to programs when comparing USMLE Step examination scores that demographic factors may play a role. The race and ethnicity of applicants likely play a role. It has been reported that underrepresented minorities had lower match rates than White and Asian applicants in dermatology.6 There have been several published articles discussing the lack of diversity in dermatology, with a call to action.7-9
Factors for Couples Matching—The number of applicants participating in the couples match continues to increase yearly. The NMRP does publish data regarding “successful” couples matching but does not specify how many couples match together. There also is little published regarding advice for participation in the couples match. Although we had a limited number of couples that participated in the match, it is interesting to note they had similar strategies, including contacting programs at institutions that had offered interviews to their partners. This strategy may be effective, as dermatology programs offer interviews relatively late compared with other specialties.5 Additionally, this strategy may increase the number of interviews offered and received, as evidenced by the higher number of interviews offered compared with those who were not couples matching. Additionally, this survey highlights the sacrifice often needed by couples in the couples match as revealed by the inclusion of rank-list options in which the couples reside long distance or in which 1 partner does not match. This information may be helpful to applicants who are planning a strategy for the couples match in dermatology. Although this study does not encompass all dermatology applicants in the 2019-2020 cycle, we do believe it may be representative. The USMLE Step 1 scores in this study were similar to the published NRMP data.1,10 According to NRMP data from the 2019-2020 cycle, the mean USMLE Step 1 score was 248 for matched applicants and 239 for unmatched.1 The NRMP reported the mean USMLE Step 2 CK score for matched was 256 and 248 for unmatched, which also is similar to our data. The NRMP reported the mean number of programs ranked was 9.9 for matched and 4.5 for unmatched applicants.1 Again, our data were similar for number of dermatology interviews attended.
Limitations—There are limitations to this study. The main limitation is that the survey is from a single institution and had a limited number of respondents. Given the nature of the study, the accuracy of the data is dependent on the applicants’ honesty in self-reporting academic performance and other variables. There also may be a selection bias given the low response rate. The subanalyses—children and couples matching—were underpowered with the limited number of participants. Further studies that include multiple residency programs and multiple years could be helpful to provide more power and less risk of bias. We did not gather information such as the Medical Student Performance Evaluation letter, letters of recommendation, or personal statements, which do play an important role in the assessment of an applicant. However, because the applicants completed these surveys, and given these are largely blinded to applicants, we did not feel the applicants could accurately respond to those aspects of the application.
Conclusion
Our survey finds that factors associated with matching included a higher USMLE Step 1 score, having a home dermatology program, and a higher number of interviews offered and attended. Some demographics had varying USMLE Step 1 scores but similar match rates.
Dermatology residency continues to be one of the most competitive specialties, with a match rate of 84.7% for US allopathic seniors in the 2019-2020 academic year.1 In the 2019-2020 cycle, dermatology applicants were tied with plastic surgery for the highest median US Medical Licensing Examination (USMLE) Step 1 score compared with other specialties, which suggests that the top medical students are applying, yet only approximately 5 of 6 students are matching.
Factors that have been cited with successful dermatology matching include USMLE Step 1 and Step 2 Clinical Knowledge (CK) scores,2 research accomplishments,3 letters of recommendation,4 medical school performance, personal statement, grades in required clerkships, and volunteer/extracurricular experiences, among others.5
The National Resident Matching Program (NRMP) publishes data each year regarding different academic factors—USMLE scores; number of abstracts, presentations, and papers; work, volunteer, and research experiences—and compares the mean between matched and nonmatched applicants.1 However, the USMLE does not report any demographic information of the applicants and the implication it has for matching. Additionally, the number of couples participating in the couples match continues to increase each year. In the 2019-2020 cycle, 1224 couples participated in the couples match.1 However, NRMP reports only limited data regarding the couples match, and it is not specialty specific.
We aimed to determine the characteristics of matched vs nonmatched dermatology applicants. Secondarily, we aimed to determine any differences among demographics regarding matching rates, academic performance, and research publications. We also aimed to characterize the strategy and outcomes of applicants that couples matched.
Materials and Methods
The Mayo Clinic institutional review board deemed this study exempt. All applicants who applied to Mayo Clinic dermatology residency in Scottsdale, Arizona, during the 2018-2019 cycle were emailed an initial survey (N=475) before Match Day that obtained demographic information, geographic information, gap-year information, USMLE Step 1 score, publications, medical school grades, number of away rotations, and number of interviews. A follow-up survey gathering match data and couples matching data was sent to the applicants who completed the first survey on Match Day. The survey was repeated for the 2019-2020 cycle. In the second survey, Step 2 CK data were obtained. The survey was sent to 629 applicants who applied to Mayo Clinic dermatology residencies in Arizona, Minnesota, and Florida to include a broader group of applicants. For publications, applicants were asked to count only published or accepted manuscripts, not abstracts, posters, conference presentations, or submitted manuscripts. Applicants who did not respond to the second survey (match data) were not included in that part of the analysis. One survey was excluded because of implausible answers (eg, scores outside of range for USMLE Step scores).
Statistical Analysis—For statistical analyses, the applicants from both applications cycles were combined. Descriptive statistics were reported in the form of mean, median, or counts (percentages), as applicable. Means were compared using 2-sided t tests. Group comparisons were examined using χ2 tests for categorical variables. Statistical analyses were performed using the BlueSky Statistics version 6.30. P<.05 was considered significant.
Results
In 2019, a total of 149 applicants completed the initial survey (31.4% response rate), and 112 completed the follow-up survey (75.2% response rate). In 2020, a total of 142 applicants completed the initial survey (22.6% response rate), and 124 completed the follow-up survey (87.3% response rate). Combining the 2 years, after removing 1 survey with implausible answers, there were 290 respondents from the initial survey and 235 from the follow-up survey. The median (SD) age for the total applicants over both years was 27 (3.0) years, and 180 applicants were female (61.9%).
USMLE Scores—The median USMLE Step 1 score was 250, and scores ranged from 196 to 271. The median USMLE Step 2 CK score was 257, and scores ranged from 213 to 281. Higher USMLE Step 1 and Step 2 CK scores and more interviews were associated with higher match rates (Table 1). In addition, students with a dermatology program at their medical school were more likely to match than those without a home dermatology program.


Gender Differences—There were 180 females and 110 males who completed the surveys. Males and females had similar match rates (85.2% vs 89.0%; P=.39)(Table 2).

Family Life—In comparing marital status, applicants who were divorced had a higher median age (38.5 years) compared with applicants who were single, married, or in a domestic partnership (all 27 years; P<.01). Differences are outlined in Table 3.


On average, applicants with children (n=27 [15 male, 12 female]; P=.13) were 3 years older than those without (30.5 vs 27; P<.01) and were more likely to be married (88.9% vs 21.5%; P<.01). Applicants with children had a mean USMLE Step 1 score of 241 compared to 251 for those without children (P=.02) and a mean USMLE Step 2 CK score of 246 compared to 258 for those without children (P<.01). Applicants with children had similar debt, number of publications, number of honored rotations, and match rates compared to applicants without children (Figure).

Couples Match—Seventeen individuals in our survey participated in the couples match (7.8%), and all 17 (100%) matched into dermatology. The mean age was 26.7 years, 12 applicants were female, 2 applicants were married, and 1 applicant had children. The mean number of interviews offered was 13.6, and the mean number of interviews attended was 11.3. This was higher than participants who were not couples matching (13.6 vs 9.8 [P=.02] and 11.3 vs 8.9 [P=.04], respectively). Applicants and their partners applied to programs and received interviews in a mean of 10 cities. Sixteen applicants reported that they contacted programs where their partner had interview offers. All participants’ rank lists included programs located in different cities than their partners’ ranked programs, and all but 1 participant ranked programs located in a different state than their partners’ ranked programs. Fifteen participants had options in their rank list for the applicant not to match, even if the partner would match. Similarly, 12 had the option for the applicant to match, even if the partner would not match. Fourteen (82.4%) matched at the same institution as their significant other. Three (17.6%) applicants matched to a program in a different state than the partner’s matched program. Two (11.8%) participants felt their relationship with their partner suffered because of the match, and 1 (5.9%) applicant was undetermined. One applicant described their relationship suffering from “unnecessary tension and anxiety” and noted “difficult conversations” about potentially matching into dermatology in a different location from their partner that could have been “devastating and not something [he or she] should have to choose.”
Comment
Factors for Matching in Dermatology—In our survey, we found the statistically significant factors of matching into dermatology included high USMLE Step 1 and Step 2 CK scores (P<.01), having a home dermatology program (P=.04), and attending a higher number of dermatology interviews (P<.01). These data are similar to NRMP results1; however, the higher likelihood of matching if the medical school has a home dermatology program has not been reported. This finding could be due to multiple factors such as students have less access to academic dermatologists for research projects, letters of recommendations, mentorship, and clinical rotations.
Gender and having children were factors that had no correlation with the match rate. There was a statistical difference of matching based on marital status (P<.01), but this is likely due to the low number of applicants in the divorced category. There were differences among demographics with USMLE Step 1 and Step 2 CK scores, which is a known factor in matching.1,2 Applicants with children had lower USMLE Step 1 and Step 2 CK scores compared to applicants without children. Females also had lower median USMLE Step 1 scores compared to males. This finding may serve as a reminder to programs when comparing USMLE Step examination scores that demographic factors may play a role. The race and ethnicity of applicants likely play a role. It has been reported that underrepresented minorities had lower match rates than White and Asian applicants in dermatology.6 There have been several published articles discussing the lack of diversity in dermatology, with a call to action.7-9
Factors for Couples Matching—The number of applicants participating in the couples match continues to increase yearly. The NMRP does publish data regarding “successful” couples matching but does not specify how many couples match together. There also is little published regarding advice for participation in the couples match. Although we had a limited number of couples that participated in the match, it is interesting to note they had similar strategies, including contacting programs at institutions that had offered interviews to their partners. This strategy may be effective, as dermatology programs offer interviews relatively late compared with other specialties.5 Additionally, this strategy may increase the number of interviews offered and received, as evidenced by the higher number of interviews offered compared with those who were not couples matching. Additionally, this survey highlights the sacrifice often needed by couples in the couples match as revealed by the inclusion of rank-list options in which the couples reside long distance or in which 1 partner does not match. This information may be helpful to applicants who are planning a strategy for the couples match in dermatology. Although this study does not encompass all dermatology applicants in the 2019-2020 cycle, we do believe it may be representative. The USMLE Step 1 scores in this study were similar to the published NRMP data.1,10 According to NRMP data from the 2019-2020 cycle, the mean USMLE Step 1 score was 248 for matched applicants and 239 for unmatched.1 The NRMP reported the mean USMLE Step 2 CK score for matched was 256 and 248 for unmatched, which also is similar to our data. The NRMP reported the mean number of programs ranked was 9.9 for matched and 4.5 for unmatched applicants.1 Again, our data were similar for number of dermatology interviews attended.
Limitations—There are limitations to this study. The main limitation is that the survey is from a single institution and had a limited number of respondents. Given the nature of the study, the accuracy of the data is dependent on the applicants’ honesty in self-reporting academic performance and other variables. There also may be a selection bias given the low response rate. The subanalyses—children and couples matching—were underpowered with the limited number of participants. Further studies that include multiple residency programs and multiple years could be helpful to provide more power and less risk of bias. We did not gather information such as the Medical Student Performance Evaluation letter, letters of recommendation, or personal statements, which do play an important role in the assessment of an applicant. However, because the applicants completed these surveys, and given these are largely blinded to applicants, we did not feel the applicants could accurately respond to those aspects of the application.
Conclusion
Our survey finds that factors associated with matching included a higher USMLE Step 1 score, having a home dermatology program, and a higher number of interviews offered and attended. Some demographics had varying USMLE Step 1 scores but similar match rates.
- National Resident Matching Program. Results and Data: 2020 Main Residency Match. National Resident Matching Program; May 2020. Accessed January 9, 2023. https://www.nrmp.org/wp-content/uploads/2021/12/MM_Results_and-Data_2020-1.pdf
- Gauer JL, Jackson JB. The association of USMLE Step 1 and Step 2 CK scores with residency match specialty and location. Med Educ Online. 2017;22:1358579.
- Wang JV, Keller M. Pressure to publish for residency applicants in dermatology. Dermatol Online J. 2016;22:13030/qt56x1t7ww.
- Wang RF, Zhang M, Kaffenberger JA. Does the dermatology standardized letter of recommendation alter applicants’ chances of matching into residency. J Am Acad Dermatol. 2017;77:e139-e140.
- National Resident Matching Program, Data Release and Research Committee: results of the 2018 NRMP Program Director Survey. Accessed December 19, 2022. https://www.nrmp.org/wp-content/uploads/2021/07/NRMP-2018-Program-Director-Survey-for-WWW.pdf
- Costello CM, Harvey JA, Besch-Stokes JG, et al. The role of race and ethnicity in the dermatology applicant match process. J Natl Med Assoc. 2022;113:666-670.
- Chen A, Shinkai K. Rethinking how we select dermatology applicants-turning the tide. JAMA Dermatol. 2017;153:259-260.
- Pandya AG, Alexis AF, Berger TG, et al. Increasing racial and ethnic diversity in dermatology: a call to action. J Am Acad Dermatol. 2016;74:584-587.
- Van Voorhees AS, Enos CW. Diversity in dermatology residency programs. J Investig Dermatol Symp Proc. 2017;18:S46-S49.
- National Resident Matching Program. Charting outcomes in the match: U.S. allopathic seniors. Characteristics of U.S. allopathic seniors who matched to their preferred specialty in the 2018 main residency match. 2nd ed. Accessed December 19, 2022. https://www.nrmp.org/wp-content/uploads/2021/07/Charting-Outcomes-in-the-Match-2018_Seniors-1.pdf
- National Resident Matching Program. Results and Data: 2020 Main Residency Match. National Resident Matching Program; May 2020. Accessed January 9, 2023. https://www.nrmp.org/wp-content/uploads/2021/12/MM_Results_and-Data_2020-1.pdf
- Gauer JL, Jackson JB. The association of USMLE Step 1 and Step 2 CK scores with residency match specialty and location. Med Educ Online. 2017;22:1358579.
- Wang JV, Keller M. Pressure to publish for residency applicants in dermatology. Dermatol Online J. 2016;22:13030/qt56x1t7ww.
- Wang RF, Zhang M, Kaffenberger JA. Does the dermatology standardized letter of recommendation alter applicants’ chances of matching into residency. J Am Acad Dermatol. 2017;77:e139-e140.
- National Resident Matching Program, Data Release and Research Committee: results of the 2018 NRMP Program Director Survey. Accessed December 19, 2022. https://www.nrmp.org/wp-content/uploads/2021/07/NRMP-2018-Program-Director-Survey-for-WWW.pdf
- Costello CM, Harvey JA, Besch-Stokes JG, et al. The role of race and ethnicity in the dermatology applicant match process. J Natl Med Assoc. 2022;113:666-670.
- Chen A, Shinkai K. Rethinking how we select dermatology applicants-turning the tide. JAMA Dermatol. 2017;153:259-260.
- Pandya AG, Alexis AF, Berger TG, et al. Increasing racial and ethnic diversity in dermatology: a call to action. J Am Acad Dermatol. 2016;74:584-587.
- Van Voorhees AS, Enos CW. Diversity in dermatology residency programs. J Investig Dermatol Symp Proc. 2017;18:S46-S49.
- National Resident Matching Program. Charting outcomes in the match: U.S. allopathic seniors. Characteristics of U.S. allopathic seniors who matched to their preferred specialty in the 2018 main residency match. 2nd ed. Accessed December 19, 2022. https://www.nrmp.org/wp-content/uploads/2021/07/Charting-Outcomes-in-the-Match-2018_Seniors-1.pdf
PRACTICE POINTS
- Dermatology residency continues to be one of the most competitive specialties, with a match rate of 84.7% in 2019.
- A high US Medical Licensing Examination (USMLE) Step 1 score and having a home dermatology program and a greater number of interviews may lead to higher likeliness of matching in dermatology.
- Most applicants (82.4%) applied to programs their partner had interviews at, suggesting this may be a helpful strategy.
Adhesive Tape to Guide Injection Depth of Botulinum Toxin for Axillary Hyperhidrosis
Practice Gap
OnabotulinumtoxinA is a US Food and Drug Administration–approved second-line treatment of axillary hyperhidrosis, with a long-term success rate greater than 80% and minimal adverse effects.1 The recommended depth and angle of injection of onabotulinumtoxinA for most cases of primary hyperhidrosis is 2 to 3 mm at a 45° angle to the skin surface.2 This small depth is difficult to accurately estimate once the needle tip is in the skin.
Injection Technique
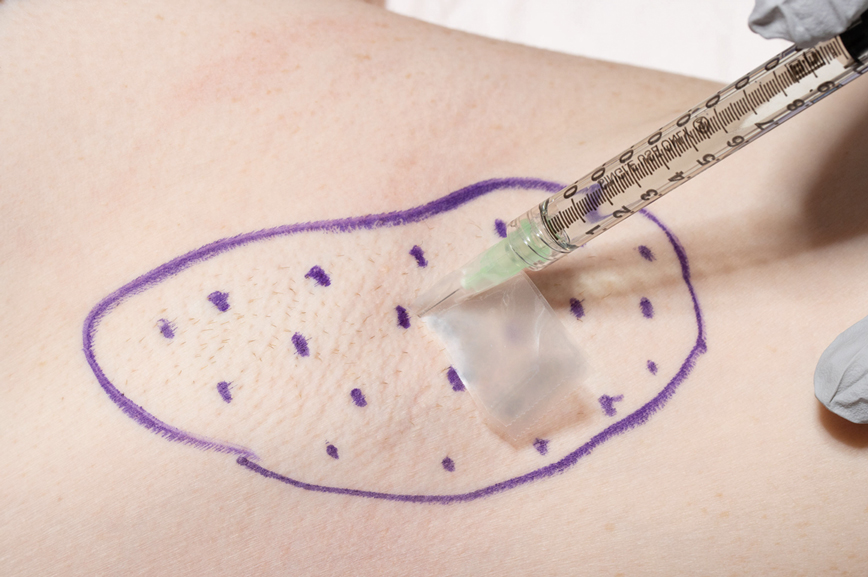
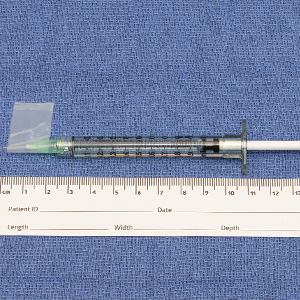
We have found that measuring 2 to 3 mm on the needle tip and then wrapping a piece of adhesive tape at that point acts as a depth guide (Figure 1). The flag shape of the tape acts as a physical barrier to prevent the needle tip from penetrating too deeply (Figure 2). This barrier also allows the injector to inject quickly to reduce the amount of pain that the patient experiences.
Practice Implications
Applying adhesive tape to a needle tip at a premeasured distance is a fast, inexpensive, and effective tool to aid accurate depth of injection for both experienced clinicians and clinicians in-training. The tape is a common office supply and the amount of tape used for a patient costs a fraction of a cent. Additionally, applying the tape takes less than 1 minute. This technique is useful for axillary hyperhidrosis injection (Figures 1 and 2) but could be used in palmar and plantar hyperhidrosis injections as well as injections other than onabotulinumtoxinA that require a specific fixed depth.
- Naumann M, Lowe NJ, Kumar CR, et al; Hyperhidrosis Clinical Investigators Group. Botulinum toxin type A is a safe and effective treatment for axillary hyperhidrosis over 16 months: a prospective study. Arch Dermatol. 2003;139:731-736. doi:10.1001/archderm.139.6.731
- Botox. Prescribing information. Allergan Pharmaceuticals Ireland;2011. Accessed May 12, 2022. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/103000s5236lbl.pdf
Practice Gap
OnabotulinumtoxinA is a US Food and Drug Administration–approved second-line treatment of axillary hyperhidrosis, with a long-term success rate greater than 80% and minimal adverse effects.1 The recommended depth and angle of injection of onabotulinumtoxinA for most cases of primary hyperhidrosis is 2 to 3 mm at a 45° angle to the skin surface.2 This small depth is difficult to accurately estimate once the needle tip is in the skin.
Injection Technique
We have found that measuring 2 to 3 mm on the needle tip and then wrapping a piece of adhesive tape at that point acts as a depth guide (Figure 1). The flag shape of the tape acts as a physical barrier to prevent the needle tip from penetrating too deeply (Figure 2). This barrier also allows the injector to inject quickly to reduce the amount of pain that the patient experiences.
Practice Implications
Applying adhesive tape to a needle tip at a premeasured distance is a fast, inexpensive, and effective tool to aid accurate depth of injection for both experienced clinicians and clinicians in-training. The tape is a common office supply and the amount of tape used for a patient costs a fraction of a cent. Additionally, applying the tape takes less than 1 minute. This technique is useful for axillary hyperhidrosis injection (Figures 1 and 2) but could be used in palmar and plantar hyperhidrosis injections as well as injections other than onabotulinumtoxinA that require a specific fixed depth.
Practice Gap
OnabotulinumtoxinA is a US Food and Drug Administration–approved second-line treatment of axillary hyperhidrosis, with a long-term success rate greater than 80% and minimal adverse effects.1 The recommended depth and angle of injection of onabotulinumtoxinA for most cases of primary hyperhidrosis is 2 to 3 mm at a 45° angle to the skin surface.2 This small depth is difficult to accurately estimate once the needle tip is in the skin.
Injection Technique
We have found that measuring 2 to 3 mm on the needle tip and then wrapping a piece of adhesive tape at that point acts as a depth guide (Figure 1). The flag shape of the tape acts as a physical barrier to prevent the needle tip from penetrating too deeply (Figure 2). This barrier also allows the injector to inject quickly to reduce the amount of pain that the patient experiences.
Practice Implications
Applying adhesive tape to a needle tip at a premeasured distance is a fast, inexpensive, and effective tool to aid accurate depth of injection for both experienced clinicians and clinicians in-training. The tape is a common office supply and the amount of tape used for a patient costs a fraction of a cent. Additionally, applying the tape takes less than 1 minute. This technique is useful for axillary hyperhidrosis injection (Figures 1 and 2) but could be used in palmar and plantar hyperhidrosis injections as well as injections other than onabotulinumtoxinA that require a specific fixed depth.
- Naumann M, Lowe NJ, Kumar CR, et al; Hyperhidrosis Clinical Investigators Group. Botulinum toxin type A is a safe and effective treatment for axillary hyperhidrosis over 16 months: a prospective study. Arch Dermatol. 2003;139:731-736. doi:10.1001/archderm.139.6.731
- Botox. Prescribing information. Allergan Pharmaceuticals Ireland;2011. Accessed May 12, 2022. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/103000s5236lbl.pdf
- Naumann M, Lowe NJ, Kumar CR, et al; Hyperhidrosis Clinical Investigators Group. Botulinum toxin type A is a safe and effective treatment for axillary hyperhidrosis over 16 months: a prospective study. Arch Dermatol. 2003;139:731-736. doi:10.1001/archderm.139.6.731
- Botox. Prescribing information. Allergan Pharmaceuticals Ireland;2011. Accessed May 12, 2022. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/103000s5236lbl.pdf
BRAF V600E Expression in Primary Melanoma and Its Association With Death: A Population-Based, Retrospective, Cross-Sectional Study
Approximately 50% of melanomas contain BRAF mutations, which occur in a greater proportion of melanomas found on sites of intermittent sun exposure.1BRAF-mutated melanomas have been associated with high levels of early-life ambient UV exposure, especially between ages 0 and 20 years.2 In addition, studies have shown that BRAF-mutated melanomas commonly are found on the trunk and extremities.1-3BRAF mutations also have been associated with younger age, superficial spreading subtype and low tumor thickness, absence of dermal melanocyte mitosis, low Ki-67 score, low phospho-histone H3 score, pigmented melanoma, advanced melanoma stage, and conjunctival melanoma.4-7BRAF mutations are found more frequently in metastatic melanoma lesions than primary melanomas, suggesting that BRAF mutations may be acquired during metastasis.8 Studies have shown different conclusions on the effect of BRAF mutation on melanoma-related death.5,9,10
The aim of this study was to identify trends in BRAF V600E–mutated melanoma according to age, sex, and melanoma-specific survival among Olmsted County, Minnesota, residents with a first diagnosis of melanoma at 18 to 60 years of age.
Methods
In total, 638 patients aged 18 to 60 years who resided in Olmsted County and had a first lifetime diagnosis of cutaneous melanoma between 1970 and 2009 were retrospectively identified as a part of the Rochester Epidemiology Project (REP). The REP is a health records linkage system that encompasses almost all sources of medical care available to the local population of Olmsted County.11 This study was approved by the Mayo Clinic Institutional Review Board (Rochester, Minnesota).
Of the 638 individuals identified in the REP, 536 had been seen at Mayo Clinic and thus potentially had tissue blocks available for the study of BRAF mutation expression. Of these 536 patients, 156 did not have sufficient residual tissue available. As a result, 380 (60%) of the original 638 patients had available blocks with sufficient tissue for immunohistochemical analysis of BRAF expression. Only primary cutaneous melanomas were included in the present study.
All specimens were reviewed by a board-certified dermatopathologist (J.S.L.) for appropriateness of inclusion, which involved confirmation of the diagnosis of melanoma, histologic type of melanoma, and presence of sufficient residual tissue for immunohistochemical stains.
All specimens were originally diagnosed as malignant melanoma at the time of clinical care by at least 2 board-certified dermatopathologists. For the purposes of this study, all specimens were rereviewed for diagnostic accuracy. We required that specimens exhibit severe cytologic and architectural atypia as well as other features favoring melanoma, such as consumption of rete pegs, pagetosis, confluence of junctional melanocytes, evidence of regression, lack of maturation of melanocytes with descent into the dermis, or mitotic figures among the dermal melanocyte population.
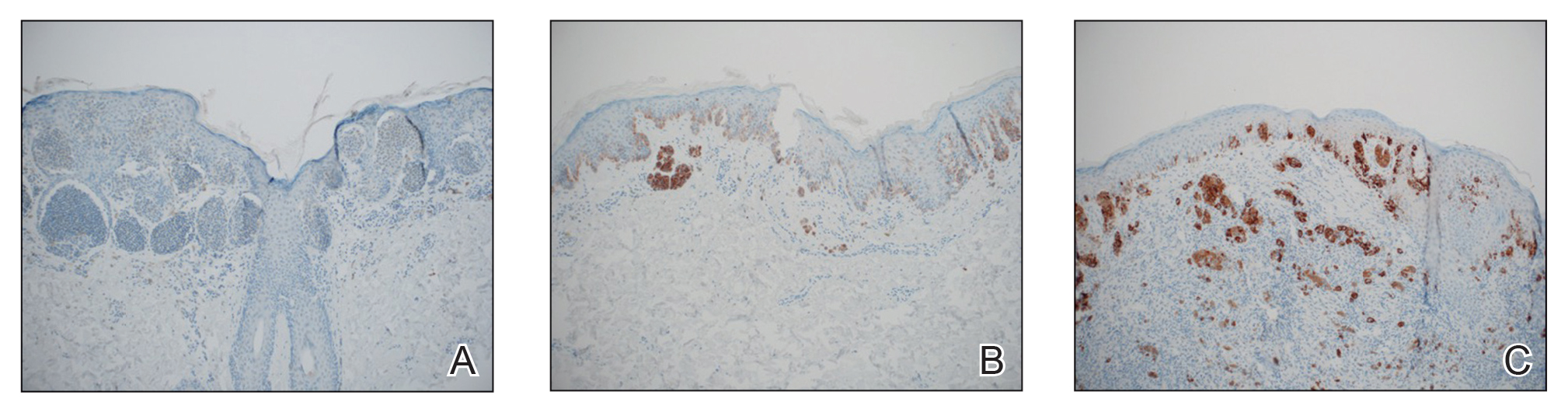
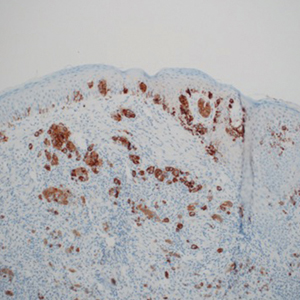
The available tissue blocks were retrieved, sectioned, confirmed as melanoma, and stained with a mouse antihuman BRAF V600E monoclonal antibody (clone VE1; Spring Bioscience) to determine the presence of a BRAF V600E mutation. BRAF staining was evaluated in conjunction with a review of the associated slides stained with hematoxylin and eosin. Cytoplasmic staining of melanocytes for BRAF was graded as negative, focal or partial positive (<50% of tumor), or diffuse positive (>50% of tumor)(Figure 1). When a melanoma arose in association with a nevus, we considered only the melanoma component for BRAF staining. We categorized the histologic type as superficial spreading, nodular, or lentigo maligna, and the location as head and neck, trunk, or extremities.
Patient characteristics and survival outcomes were gathered through the health record and included age, Breslow thickness, location, decade of diagnosis, histologic type, stage (ie, noninvasive, invasive, or advanced), and follow-up. Pathologic stage 0 was considered noninvasive; stages IA and IB, invasive; and stages IIA or higher, advanced.
Statistical Analysis—Comparisons between the group of patients in the study (n=380) and the group of patients excluded for the reasons stated above (n=258) as well as associations of mutant BRAF status (positive [partial positive and diffuse positive] vs negative) with patient age (young adults [age range, 18–39 years] and middle-aged adults [age range, 40–60 years]), sex, decade of diagnosis, location, histologic type, and stage were evaluated with Wilcoxon rank sum, χ2, Fisher exact, or Cochran-Armitage trend tests. Disease-specific survival and overall survival rates were estimated with the Kaplan-Meier method, and the duration of follow-up was calculated from the date of melanoma diagnosis to the date of death or the last follow-up. Associations of mutant BRAF expression status with death from melanoma and death from any cause were evaluated with Cox proportional hazard regression models and summarized with hazard ratio (HR) and 95% CI. Survival analyses were limited to patients with invasive or advanced disease. Statistical analyses were performed with SAS statistical software (SAS version 9.4). All tests were 2-sided, and P<.05 was considered statistically significant.
Results
Clinical and Tumor Characteristics—Of the 380 tissue specimens that underwent BRAF V600E analysis, 247 had negative staining; 106 had diffuse strong staining; and 27 had focal or partial staining. In total, 133 (35%) were positive, either partially or diffusely. The median age for patients who had negative staining was 45 years; for those with positive staining, it was 41 years (P=.07).
The patients who met inclusion criteria (n=380) were compared with those who were excluded (n=258)(eTable 1). The groups were similar on the basis of sex; age; and melanoma location, stage, and histologic subtype. However, some evidence showed that patients included in the study received the diagnosis of melanoma more recently (1970-1989, 13.2%; 1990-1999, 28.7%; 2000-2009, 58.2%) than those who were excluded (1970-1989, 24.7%; 1990-1999, 23.5%; 2000-2009, 51.8%)(P=.02).
BRAF V600E expression was more commonly found in superficial spreading (37.7%) and nodular melanomas (35.0%) than in situ melanomas (17.1%)(P=.01). Other characteristics of BRAF V600E expression are described in eTable 2. Overall, invasive and advanced melanomas were significantly more likely to harbor BRAF V600E expression than noninvasive melanomas (39.6% and 37.9%, respectively, vs 17.9%; P=.003). However, advanced melanomas more commonly expressed BRAF positivity among women, and invasive melanomas more commonly expressed BRAF positivity among men (eTable 2).
Survival—Survival analyses were limited to 297 patients with confirmed invasive or advanced disease. Of these, 180 (61%) had no BRAF V600E staining; 25 (8%) had partial staining; and 92 (31%) had diffuse positive staining. In total, 117 patients (39%) had a BRAF-mutated melanoma.
Among the patients still alive, the median (interquartile range [IQR]) duration of follow-up was 10.2 (7.0-16.8) years. Thirty-nine patients with invasive or advanced disease had died of any cause at a median (IQR) of 3.0 (1.3-10.2) years after diagnosis. In total, 26 patients died of melanoma at a median (IQR) follow-up of 2.5 (1.3-7.4) years after diagnosis. Eight women and 18 men died of malignant melanoma. Five deaths occurred because of malignant melanoma among patients aged 18 to 39 years, and 21 occurred among patients aged 40 to 60 years. In the 18- to 39-year-old group, all 5 deaths were among patients with a BRAF-positive melanoma. Estimated disease-specific survival rate (95% CI; number still at risk) at 5, 10, 15, and 20 years after diagnosis was 94% (91%-97%; 243), 91% (87%-95%; 142), 89% (85%-94%; 87), and 88% (83%-93%; 45), respectively.
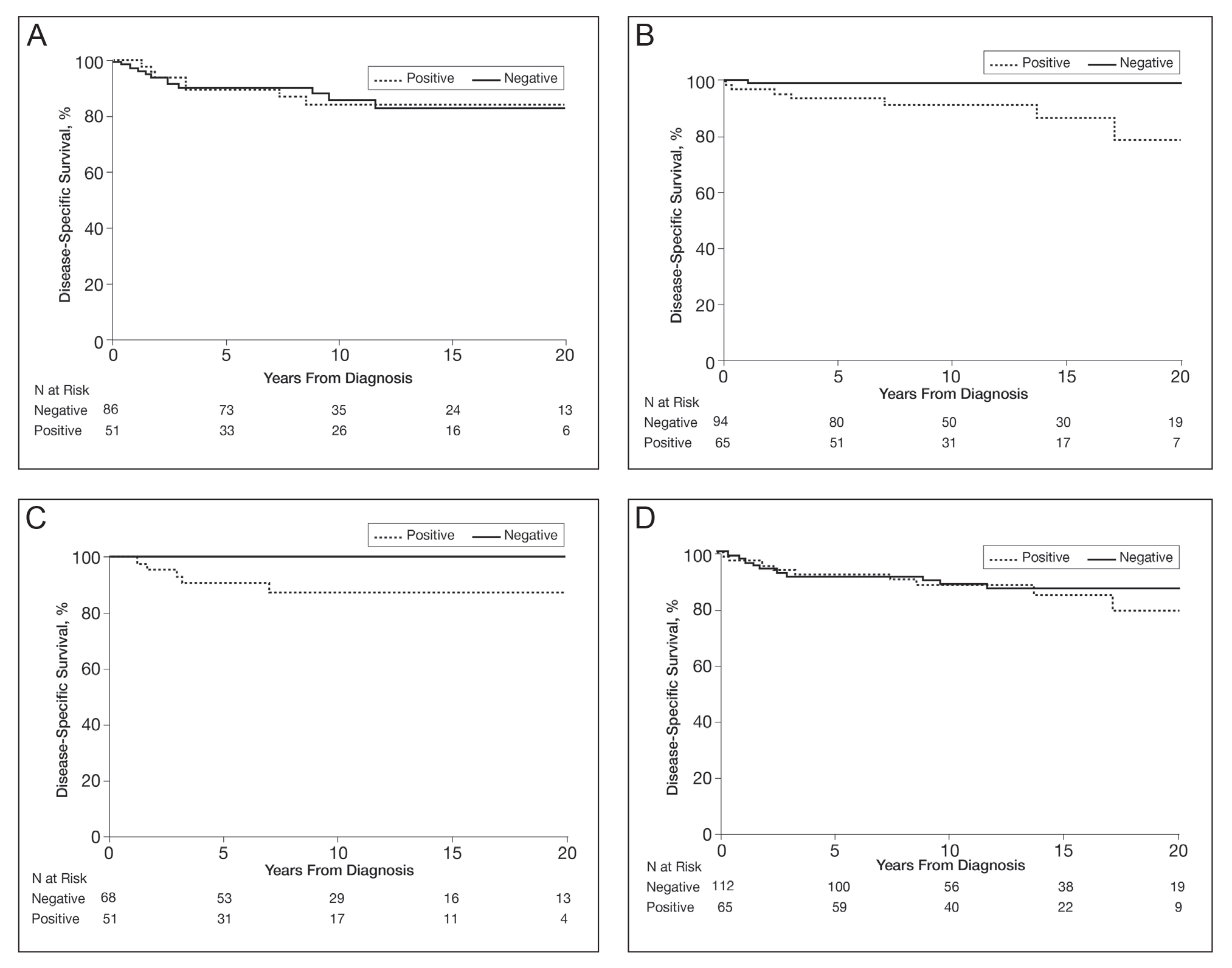
In a univariable analysis, the HR for association of positive mutant BRAF expression with death of malignant melanoma was 1.84 (95% CI, 0.85-3.98; P=.12). No statistically significant interaction was observed between decade of diagnosis and BRAF expression (P=.60). However, the interaction between sex and BRAF expression was significant (P=.04), with increased risk of death from melanoma among women with BRAF-mutated melanoma (HR, 10.88; 95% CI, 1.34-88.41; P=.026) but not among men (HR 1.02; 95% CI, 0.40-2.64; P=.97)(Figures 2A and 2B). The HR for death from malignant melanoma among young adults aged 18 to 39 years with a BRAF-mutated melanoma was 16.4 (95% CI, 0.81-330.10; P=.068), whereas the HR among adults aged 40 to 60 years with a BRAF-mutated melanoma was 1.24 (95% CI, 0.52-2.98; P=.63)(Figures 2C and 2D).
BRAF V600E expression was not significantly associated with death from any cause (HR, 1.39; 95% CI, 0.74-2.61; P=.31) or with decade of diagnosis (P=.13). Similarly, BRAF expression was not associated with death from any cause according to sex (P=.31). However, a statistically significant interaction was seen between age at diagnosis and BRAF expression (P=.003). BRAF expression was significantly associated with death from any cause for adults aged 18 to 39 years (HR, 9.60; 95% CI, 1.15-80.00; P=.04). In comparison, no association of BRAF expression with death was observed for adults aged 40 to 60 years (HR, 0.99; 95% CI, 0.48-2.03; P=.98).
Comment
We found that melanomas with BRAF mutations were more likely in advanced and invasive melanoma. The frequency of BRAF mutations among melanomas that were considered advanced was higher in women than men. Although the number of deaths was limited, women with a melanoma with BRAF expression were more likely to die of melanoma, young adults with a BRAF-mutated melanoma had an almost 10-fold increased risk of dying from any cause, and middle-aged adults showed no increased risk of death. These findings suggest that young adults who are genetically prone to a BRAF-mutated melanoma could be at a disadvantage for all-cause mortality. Although this finding was significant, the 95% CI was large, and further studies would be warranted before sound conclusions could be made.
Melanoma has been increasing in incidence across all age groups in Olmsted County over the last 4 decades.12-14 However, our results show that the percentage of BRAF-mutated melanomas in this population has been stable over time, with no statistically significant difference by age or sex. Other confounding factors may have an influence, such as increased rates of early detection and diagnosis of melanoma in contemporary times. Our data suggest that patients included in the BRAF-mutation analysis study had received the diagnosis of melanoma more recently than those who were excluded from the study, which could be due to older melanomas being less likely to have adequate tissue specimens available for immunohistochemical staining/evaluation.
Prior research has shown that BRAF-mutated melanomas typically occur on the trunk and are more likely in individuals with more than 14 nevi on the back.2 In the present cohort, BRAF-positive melanomas had a predisposition toward the trunk but also were found on the head, neck, and extremities—areas that are more likely to have long-term sun damage. One suggestion is that 2 distinct pathways for melanoma development exist: one associated with a large number of melanocytic nevi (that is more prone to genetic mutations in melanocytes) and the other associated with long-term sun exposure.15,16 The combination of these hypotheses suggests that individuals who are prone to the development of large numbers of nevi may require sun exposure for the initial insult, but the development of melanoma may be carried out by other factors after this initial sun exposure insult, whereas individuals without large numbers of nevi who may have less genetic risk may require continued long-term sun exposure for melanoma to develop.17
Our study had limitations, including the small numbers of deaths overall and cause-specific deaths of metastatic melanoma, which limited our ability to conduct more extensive multivariable modeling. Also, the retrospective nature and time frame of looking back 4 decades did not allow us to have information sufficient to categorize some patients as having dysplastic nevus syndrome or not, which would be a potentially interesting variable to include in the analysis. Because the number of deaths in the 18- to 39-year-old cohort was only 5, further statistical comparison regarding tumor type and other variables pertaining to BRAF positivity were not possible. In addition, our data were collected from patients residing in a single geographic county (Olmsted County, Minnesota), which may limit generalizability. Lastly, BRAF V600E mutations were identified through immunostaining only, not molecular data, so it is possible some patients had false-negative immunohistochemistry findings and thus were not identified.
Conclusion
BRAF-mutated melanomas were found in 35% of our cohort, with no significant change in the percentage of melanomas with BRAF V600E mutations over the last 4 decades in this population. In addition, no differences or significant trends existed according to sex and BRAF-mutated melanoma development. Women with BRAF-mutated melanomas were more likely to die of metastatic melanoma than men, and young adults with BRAF-mutated melanomas had a higher all-cause mortality risk. Further research is needed to decipher what effect BRAF-mutated melanomas have on metastasis and cause-specific death in women as well as all-cause mortality in young adults.
Acknowledgment—The authors are indebted to Scientific Publications, Mayo Clinic (Rochester, Minnesota).
- Grimaldi AM, Cassidy PB, Leachmann S, et al. Novel approaches in melanoma prevention and therapy. Cancer Treat Res. 2014;159: 443-455.
- Thomas NE, Edmiston SN, Alexander A, et al. Number of nevi and early-life ambient UV exposure are associated with BRAF-mutant melanoma. Cancer Epidemiol Biomarkers Prev. 2007;16:991-997.
- Curtin JA, Fridlyand J, Kageshita T, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005;353:2135-2147.
- Thomas NE, Edmiston SN, Alexander A, et al. Association between NRAS and BRAF mutational status and melanoma-specific survival among patients with higher-risk primary melanoma. JAMA Oncol. 2015;1:359-368.
- Liu W, Kelly JW, Trivett M, et al. Distinct clinical and pathological features are associated with the BRAF(T1799A(V600E)) mutation in primary melanoma. J Invest Dermatol. 2007;127:900-905.
- Kim SY, Kim SN, Hahn HJ, et al. Metaanalysis of BRAF mutations and clinicopathologic characteristics in primary melanoma. J Am Acad Dermatol. 2015;72:1036-1046.e2.
- Larsen AC, Dahl C, Dahmcke CM, et al. BRAF mutations in conjunctival melanoma: investigation of incidence, clinicopathological features, prognosis and paired premalignant lesions. Acta Ophthalmol. 2016;94:463-470.
- Shinozaki M, Fujimoto A, Morton DL, et al. Incidence of BRAF oncogene mutation and clinical relevance for primary cutaneous melanomas. Clin Cancer Res. 2004;10:1753-1757.
- Heppt MV, Siepmann T, Engel J, et al. Prognostic significance of BRAF and NRAS mutations in melanoma: a German study from routine care. BMC Cancer. 2017;17:536.
- Mar VJ, Liu W, Devitt B, et al. The role of BRAF mutations in primary melanoma growth rate and survival. Br J Dermatol. 2015;173:76-82.
- Rocca WA, Yawn BP, St Sauver JL, et al. History of the Rochester Epidemiology Project: half a century of medical records linkage in a US population. Mayo Clin Proc. 2012;87:1202-1213.
- Reed KB, Brewer JD, Lohse CM, et al. Increasing incidence of melanoma among young adults: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2012;87:328-334.
- Olazagasti Lourido JM, Ma JE, Lohse CM, et al. Increasing incidence of melanoma in the elderly: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2016;91:1555-1562.
- Lowe GC, Saavedra A, Reed KB, et al. Increasing incidence of melanoma among middle-aged adults: an epidemiologic study in Olmsted County, Minnesota. Mayo Clin Proc. 2014;89:52-59.
- Whiteman DC, Parsons PG, Green AC. p53 expression and risk factors for cutaneous melanoma: a case-control study. Int J Cancer. 1998;77:843-848.
- Whiteman DC, Watt P, Purdie DM, et al. Melanocytic nevi, solar keratoses, and divergent pathways to cutaneous melanoma. J Natl Cancer Inst. 2003;95:806-812.
- Olsen CM, Zens MS, Green AC, et al. Biologic markers of sun exposure and melanoma risk in women: pooled case-control analysis. Int J Cancer. 2011;129:713-723.
Approximately 50% of melanomas contain BRAF mutations, which occur in a greater proportion of melanomas found on sites of intermittent sun exposure.1BRAF-mutated melanomas have been associated with high levels of early-life ambient UV exposure, especially between ages 0 and 20 years.2 In addition, studies have shown that BRAF-mutated melanomas commonly are found on the trunk and extremities.1-3BRAF mutations also have been associated with younger age, superficial spreading subtype and low tumor thickness, absence of dermal melanocyte mitosis, low Ki-67 score, low phospho-histone H3 score, pigmented melanoma, advanced melanoma stage, and conjunctival melanoma.4-7BRAF mutations are found more frequently in metastatic melanoma lesions than primary melanomas, suggesting that BRAF mutations may be acquired during metastasis.8 Studies have shown different conclusions on the effect of BRAF mutation on melanoma-related death.5,9,10
The aim of this study was to identify trends in BRAF V600E–mutated melanoma according to age, sex, and melanoma-specific survival among Olmsted County, Minnesota, residents with a first diagnosis of melanoma at 18 to 60 years of age.
Methods
In total, 638 patients aged 18 to 60 years who resided in Olmsted County and had a first lifetime diagnosis of cutaneous melanoma between 1970 and 2009 were retrospectively identified as a part of the Rochester Epidemiology Project (REP). The REP is a health records linkage system that encompasses almost all sources of medical care available to the local population of Olmsted County.11 This study was approved by the Mayo Clinic Institutional Review Board (Rochester, Minnesota).
Of the 638 individuals identified in the REP, 536 had been seen at Mayo Clinic and thus potentially had tissue blocks available for the study of BRAF mutation expression. Of these 536 patients, 156 did not have sufficient residual tissue available. As a result, 380 (60%) of the original 638 patients had available blocks with sufficient tissue for immunohistochemical analysis of BRAF expression. Only primary cutaneous melanomas were included in the present study.
All specimens were reviewed by a board-certified dermatopathologist (J.S.L.) for appropriateness of inclusion, which involved confirmation of the diagnosis of melanoma, histologic type of melanoma, and presence of sufficient residual tissue for immunohistochemical stains.
All specimens were originally diagnosed as malignant melanoma at the time of clinical care by at least 2 board-certified dermatopathologists. For the purposes of this study, all specimens were rereviewed for diagnostic accuracy. We required that specimens exhibit severe cytologic and architectural atypia as well as other features favoring melanoma, such as consumption of rete pegs, pagetosis, confluence of junctional melanocytes, evidence of regression, lack of maturation of melanocytes with descent into the dermis, or mitotic figures among the dermal melanocyte population.
The available tissue blocks were retrieved, sectioned, confirmed as melanoma, and stained with a mouse antihuman BRAF V600E monoclonal antibody (clone VE1; Spring Bioscience) to determine the presence of a BRAF V600E mutation. BRAF staining was evaluated in conjunction with a review of the associated slides stained with hematoxylin and eosin. Cytoplasmic staining of melanocytes for BRAF was graded as negative, focal or partial positive (<50% of tumor), or diffuse positive (>50% of tumor)(Figure 1). When a melanoma arose in association with a nevus, we considered only the melanoma component for BRAF staining. We categorized the histologic type as superficial spreading, nodular, or lentigo maligna, and the location as head and neck, trunk, or extremities.
Patient characteristics and survival outcomes were gathered through the health record and included age, Breslow thickness, location, decade of diagnosis, histologic type, stage (ie, noninvasive, invasive, or advanced), and follow-up. Pathologic stage 0 was considered noninvasive; stages IA and IB, invasive; and stages IIA or higher, advanced.
Statistical Analysis—Comparisons between the group of patients in the study (n=380) and the group of patients excluded for the reasons stated above (n=258) as well as associations of mutant BRAF status (positive [partial positive and diffuse positive] vs negative) with patient age (young adults [age range, 18–39 years] and middle-aged adults [age range, 40–60 years]), sex, decade of diagnosis, location, histologic type, and stage were evaluated with Wilcoxon rank sum, χ2, Fisher exact, or Cochran-Armitage trend tests. Disease-specific survival and overall survival rates were estimated with the Kaplan-Meier method, and the duration of follow-up was calculated from the date of melanoma diagnosis to the date of death or the last follow-up. Associations of mutant BRAF expression status with death from melanoma and death from any cause were evaluated with Cox proportional hazard regression models and summarized with hazard ratio (HR) and 95% CI. Survival analyses were limited to patients with invasive or advanced disease. Statistical analyses were performed with SAS statistical software (SAS version 9.4). All tests were 2-sided, and P<.05 was considered statistically significant.
Results
Clinical and Tumor Characteristics—Of the 380 tissue specimens that underwent BRAF V600E analysis, 247 had negative staining; 106 had diffuse strong staining; and 27 had focal or partial staining. In total, 133 (35%) were positive, either partially or diffusely. The median age for patients who had negative staining was 45 years; for those with positive staining, it was 41 years (P=.07).
The patients who met inclusion criteria (n=380) were compared with those who were excluded (n=258)(eTable 1). The groups were similar on the basis of sex; age; and melanoma location, stage, and histologic subtype. However, some evidence showed that patients included in the study received the diagnosis of melanoma more recently (1970-1989, 13.2%; 1990-1999, 28.7%; 2000-2009, 58.2%) than those who were excluded (1970-1989, 24.7%; 1990-1999, 23.5%; 2000-2009, 51.8%)(P=.02).
BRAF V600E expression was more commonly found in superficial spreading (37.7%) and nodular melanomas (35.0%) than in situ melanomas (17.1%)(P=.01). Other characteristics of BRAF V600E expression are described in eTable 2. Overall, invasive and advanced melanomas were significantly more likely to harbor BRAF V600E expression than noninvasive melanomas (39.6% and 37.9%, respectively, vs 17.9%; P=.003). However, advanced melanomas more commonly expressed BRAF positivity among women, and invasive melanomas more commonly expressed BRAF positivity among men (eTable 2).
Survival—Survival analyses were limited to 297 patients with confirmed invasive or advanced disease. Of these, 180 (61%) had no BRAF V600E staining; 25 (8%) had partial staining; and 92 (31%) had diffuse positive staining. In total, 117 patients (39%) had a BRAF-mutated melanoma.
Among the patients still alive, the median (interquartile range [IQR]) duration of follow-up was 10.2 (7.0-16.8) years. Thirty-nine patients with invasive or advanced disease had died of any cause at a median (IQR) of 3.0 (1.3-10.2) years after diagnosis. In total, 26 patients died of melanoma at a median (IQR) follow-up of 2.5 (1.3-7.4) years after diagnosis. Eight women and 18 men died of malignant melanoma. Five deaths occurred because of malignant melanoma among patients aged 18 to 39 years, and 21 occurred among patients aged 40 to 60 years. In the 18- to 39-year-old group, all 5 deaths were among patients with a BRAF-positive melanoma. Estimated disease-specific survival rate (95% CI; number still at risk) at 5, 10, 15, and 20 years after diagnosis was 94% (91%-97%; 243), 91% (87%-95%; 142), 89% (85%-94%; 87), and 88% (83%-93%; 45), respectively.
In a univariable analysis, the HR for association of positive mutant BRAF expression with death of malignant melanoma was 1.84 (95% CI, 0.85-3.98; P=.12). No statistically significant interaction was observed between decade of diagnosis and BRAF expression (P=.60). However, the interaction between sex and BRAF expression was significant (P=.04), with increased risk of death from melanoma among women with BRAF-mutated melanoma (HR, 10.88; 95% CI, 1.34-88.41; P=.026) but not among men (HR 1.02; 95% CI, 0.40-2.64; P=.97)(Figures 2A and 2B). The HR for death from malignant melanoma among young adults aged 18 to 39 years with a BRAF-mutated melanoma was 16.4 (95% CI, 0.81-330.10; P=.068), whereas the HR among adults aged 40 to 60 years with a BRAF-mutated melanoma was 1.24 (95% CI, 0.52-2.98; P=.63)(Figures 2C and 2D).
BRAF V600E expression was not significantly associated with death from any cause (HR, 1.39; 95% CI, 0.74-2.61; P=.31) or with decade of diagnosis (P=.13). Similarly, BRAF expression was not associated with death from any cause according to sex (P=.31). However, a statistically significant interaction was seen between age at diagnosis and BRAF expression (P=.003). BRAF expression was significantly associated with death from any cause for adults aged 18 to 39 years (HR, 9.60; 95% CI, 1.15-80.00; P=.04). In comparison, no association of BRAF expression with death was observed for adults aged 40 to 60 years (HR, 0.99; 95% CI, 0.48-2.03; P=.98).
Comment
We found that melanomas with BRAF mutations were more likely in advanced and invasive melanoma. The frequency of BRAF mutations among melanomas that were considered advanced was higher in women than men. Although the number of deaths was limited, women with a melanoma with BRAF expression were more likely to die of melanoma, young adults with a BRAF-mutated melanoma had an almost 10-fold increased risk of dying from any cause, and middle-aged adults showed no increased risk of death. These findings suggest that young adults who are genetically prone to a BRAF-mutated melanoma could be at a disadvantage for all-cause mortality. Although this finding was significant, the 95% CI was large, and further studies would be warranted before sound conclusions could be made.
Melanoma has been increasing in incidence across all age groups in Olmsted County over the last 4 decades.12-14 However, our results show that the percentage of BRAF-mutated melanomas in this population has been stable over time, with no statistically significant difference by age or sex. Other confounding factors may have an influence, such as increased rates of early detection and diagnosis of melanoma in contemporary times. Our data suggest that patients included in the BRAF-mutation analysis study had received the diagnosis of melanoma more recently than those who were excluded from the study, which could be due to older melanomas being less likely to have adequate tissue specimens available for immunohistochemical staining/evaluation.
Prior research has shown that BRAF-mutated melanomas typically occur on the trunk and are more likely in individuals with more than 14 nevi on the back.2 In the present cohort, BRAF-positive melanomas had a predisposition toward the trunk but also were found on the head, neck, and extremities—areas that are more likely to have long-term sun damage. One suggestion is that 2 distinct pathways for melanoma development exist: one associated with a large number of melanocytic nevi (that is more prone to genetic mutations in melanocytes) and the other associated with long-term sun exposure.15,16 The combination of these hypotheses suggests that individuals who are prone to the development of large numbers of nevi may require sun exposure for the initial insult, but the development of melanoma may be carried out by other factors after this initial sun exposure insult, whereas individuals without large numbers of nevi who may have less genetic risk may require continued long-term sun exposure for melanoma to develop.17
Our study had limitations, including the small numbers of deaths overall and cause-specific deaths of metastatic melanoma, which limited our ability to conduct more extensive multivariable modeling. Also, the retrospective nature and time frame of looking back 4 decades did not allow us to have information sufficient to categorize some patients as having dysplastic nevus syndrome or not, which would be a potentially interesting variable to include in the analysis. Because the number of deaths in the 18- to 39-year-old cohort was only 5, further statistical comparison regarding tumor type and other variables pertaining to BRAF positivity were not possible. In addition, our data were collected from patients residing in a single geographic county (Olmsted County, Minnesota), which may limit generalizability. Lastly, BRAF V600E mutations were identified through immunostaining only, not molecular data, so it is possible some patients had false-negative immunohistochemistry findings and thus were not identified.
Conclusion
BRAF-mutated melanomas were found in 35% of our cohort, with no significant change in the percentage of melanomas with BRAF V600E mutations over the last 4 decades in this population. In addition, no differences or significant trends existed according to sex and BRAF-mutated melanoma development. Women with BRAF-mutated melanomas were more likely to die of metastatic melanoma than men, and young adults with BRAF-mutated melanomas had a higher all-cause mortality risk. Further research is needed to decipher what effect BRAF-mutated melanomas have on metastasis and cause-specific death in women as well as all-cause mortality in young adults.
Acknowledgment—The authors are indebted to Scientific Publications, Mayo Clinic (Rochester, Minnesota).
Approximately 50% of melanomas contain BRAF mutations, which occur in a greater proportion of melanomas found on sites of intermittent sun exposure.1BRAF-mutated melanomas have been associated with high levels of early-life ambient UV exposure, especially between ages 0 and 20 years.2 In addition, studies have shown that BRAF-mutated melanomas commonly are found on the trunk and extremities.1-3BRAF mutations also have been associated with younger age, superficial spreading subtype and low tumor thickness, absence of dermal melanocyte mitosis, low Ki-67 score, low phospho-histone H3 score, pigmented melanoma, advanced melanoma stage, and conjunctival melanoma.4-7BRAF mutations are found more frequently in metastatic melanoma lesions than primary melanomas, suggesting that BRAF mutations may be acquired during metastasis.8 Studies have shown different conclusions on the effect of BRAF mutation on melanoma-related death.5,9,10
The aim of this study was to identify trends in BRAF V600E–mutated melanoma according to age, sex, and melanoma-specific survival among Olmsted County, Minnesota, residents with a first diagnosis of melanoma at 18 to 60 years of age.
Methods
In total, 638 patients aged 18 to 60 years who resided in Olmsted County and had a first lifetime diagnosis of cutaneous melanoma between 1970 and 2009 were retrospectively identified as a part of the Rochester Epidemiology Project (REP). The REP is a health records linkage system that encompasses almost all sources of medical care available to the local population of Olmsted County.11 This study was approved by the Mayo Clinic Institutional Review Board (Rochester, Minnesota).
Of the 638 individuals identified in the REP, 536 had been seen at Mayo Clinic and thus potentially had tissue blocks available for the study of BRAF mutation expression. Of these 536 patients, 156 did not have sufficient residual tissue available. As a result, 380 (60%) of the original 638 patients had available blocks with sufficient tissue for immunohistochemical analysis of BRAF expression. Only primary cutaneous melanomas were included in the present study.
All specimens were reviewed by a board-certified dermatopathologist (J.S.L.) for appropriateness of inclusion, which involved confirmation of the diagnosis of melanoma, histologic type of melanoma, and presence of sufficient residual tissue for immunohistochemical stains.
All specimens were originally diagnosed as malignant melanoma at the time of clinical care by at least 2 board-certified dermatopathologists. For the purposes of this study, all specimens were rereviewed for diagnostic accuracy. We required that specimens exhibit severe cytologic and architectural atypia as well as other features favoring melanoma, such as consumption of rete pegs, pagetosis, confluence of junctional melanocytes, evidence of regression, lack of maturation of melanocytes with descent into the dermis, or mitotic figures among the dermal melanocyte population.
The available tissue blocks were retrieved, sectioned, confirmed as melanoma, and stained with a mouse antihuman BRAF V600E monoclonal antibody (clone VE1; Spring Bioscience) to determine the presence of a BRAF V600E mutation. BRAF staining was evaluated in conjunction with a review of the associated slides stained with hematoxylin and eosin. Cytoplasmic staining of melanocytes for BRAF was graded as negative, focal or partial positive (<50% of tumor), or diffuse positive (>50% of tumor)(Figure 1). When a melanoma arose in association with a nevus, we considered only the melanoma component for BRAF staining. We categorized the histologic type as superficial spreading, nodular, or lentigo maligna, and the location as head and neck, trunk, or extremities.
Patient characteristics and survival outcomes were gathered through the health record and included age, Breslow thickness, location, decade of diagnosis, histologic type, stage (ie, noninvasive, invasive, or advanced), and follow-up. Pathologic stage 0 was considered noninvasive; stages IA and IB, invasive; and stages IIA or higher, advanced.
Statistical Analysis—Comparisons between the group of patients in the study (n=380) and the group of patients excluded for the reasons stated above (n=258) as well as associations of mutant BRAF status (positive [partial positive and diffuse positive] vs negative) with patient age (young adults [age range, 18–39 years] and middle-aged adults [age range, 40–60 years]), sex, decade of diagnosis, location, histologic type, and stage were evaluated with Wilcoxon rank sum, χ2, Fisher exact, or Cochran-Armitage trend tests. Disease-specific survival and overall survival rates were estimated with the Kaplan-Meier method, and the duration of follow-up was calculated from the date of melanoma diagnosis to the date of death or the last follow-up. Associations of mutant BRAF expression status with death from melanoma and death from any cause were evaluated with Cox proportional hazard regression models and summarized with hazard ratio (HR) and 95% CI. Survival analyses were limited to patients with invasive or advanced disease. Statistical analyses were performed with SAS statistical software (SAS version 9.4). All tests were 2-sided, and P<.05 was considered statistically significant.
Results
Clinical and Tumor Characteristics—Of the 380 tissue specimens that underwent BRAF V600E analysis, 247 had negative staining; 106 had diffuse strong staining; and 27 had focal or partial staining. In total, 133 (35%) were positive, either partially or diffusely. The median age for patients who had negative staining was 45 years; for those with positive staining, it was 41 years (P=.07).
The patients who met inclusion criteria (n=380) were compared with those who were excluded (n=258)(eTable 1). The groups were similar on the basis of sex; age; and melanoma location, stage, and histologic subtype. However, some evidence showed that patients included in the study received the diagnosis of melanoma more recently (1970-1989, 13.2%; 1990-1999, 28.7%; 2000-2009, 58.2%) than those who were excluded (1970-1989, 24.7%; 1990-1999, 23.5%; 2000-2009, 51.8%)(P=.02).
BRAF V600E expression was more commonly found in superficial spreading (37.7%) and nodular melanomas (35.0%) than in situ melanomas (17.1%)(P=.01). Other characteristics of BRAF V600E expression are described in eTable 2. Overall, invasive and advanced melanomas were significantly more likely to harbor BRAF V600E expression than noninvasive melanomas (39.6% and 37.9%, respectively, vs 17.9%; P=.003). However, advanced melanomas more commonly expressed BRAF positivity among women, and invasive melanomas more commonly expressed BRAF positivity among men (eTable 2).
Survival—Survival analyses were limited to 297 patients with confirmed invasive or advanced disease. Of these, 180 (61%) had no BRAF V600E staining; 25 (8%) had partial staining; and 92 (31%) had diffuse positive staining. In total, 117 patients (39%) had a BRAF-mutated melanoma.
Among the patients still alive, the median (interquartile range [IQR]) duration of follow-up was 10.2 (7.0-16.8) years. Thirty-nine patients with invasive or advanced disease had died of any cause at a median (IQR) of 3.0 (1.3-10.2) years after diagnosis. In total, 26 patients died of melanoma at a median (IQR) follow-up of 2.5 (1.3-7.4) years after diagnosis. Eight women and 18 men died of malignant melanoma. Five deaths occurred because of malignant melanoma among patients aged 18 to 39 years, and 21 occurred among patients aged 40 to 60 years. In the 18- to 39-year-old group, all 5 deaths were among patients with a BRAF-positive melanoma. Estimated disease-specific survival rate (95% CI; number still at risk) at 5, 10, 15, and 20 years after diagnosis was 94% (91%-97%; 243), 91% (87%-95%; 142), 89% (85%-94%; 87), and 88% (83%-93%; 45), respectively.
In a univariable analysis, the HR for association of positive mutant BRAF expression with death of malignant melanoma was 1.84 (95% CI, 0.85-3.98; P=.12). No statistically significant interaction was observed between decade of diagnosis and BRAF expression (P=.60). However, the interaction between sex and BRAF expression was significant (P=.04), with increased risk of death from melanoma among women with BRAF-mutated melanoma (HR, 10.88; 95% CI, 1.34-88.41; P=.026) but not among men (HR 1.02; 95% CI, 0.40-2.64; P=.97)(Figures 2A and 2B). The HR for death from malignant melanoma among young adults aged 18 to 39 years with a BRAF-mutated melanoma was 16.4 (95% CI, 0.81-330.10; P=.068), whereas the HR among adults aged 40 to 60 years with a BRAF-mutated melanoma was 1.24 (95% CI, 0.52-2.98; P=.63)(Figures 2C and 2D).
BRAF V600E expression was not significantly associated with death from any cause (HR, 1.39; 95% CI, 0.74-2.61; P=.31) or with decade of diagnosis (P=.13). Similarly, BRAF expression was not associated with death from any cause according to sex (P=.31). However, a statistically significant interaction was seen between age at diagnosis and BRAF expression (P=.003). BRAF expression was significantly associated with death from any cause for adults aged 18 to 39 years (HR, 9.60; 95% CI, 1.15-80.00; P=.04). In comparison, no association of BRAF expression with death was observed for adults aged 40 to 60 years (HR, 0.99; 95% CI, 0.48-2.03; P=.98).
Comment
We found that melanomas with BRAF mutations were more likely in advanced and invasive melanoma. The frequency of BRAF mutations among melanomas that were considered advanced was higher in women than men. Although the number of deaths was limited, women with a melanoma with BRAF expression were more likely to die of melanoma, young adults with a BRAF-mutated melanoma had an almost 10-fold increased risk of dying from any cause, and middle-aged adults showed no increased risk of death. These findings suggest that young adults who are genetically prone to a BRAF-mutated melanoma could be at a disadvantage for all-cause mortality. Although this finding was significant, the 95% CI was large, and further studies would be warranted before sound conclusions could be made.
Melanoma has been increasing in incidence across all age groups in Olmsted County over the last 4 decades.12-14 However, our results show that the percentage of BRAF-mutated melanomas in this population has been stable over time, with no statistically significant difference by age or sex. Other confounding factors may have an influence, such as increased rates of early detection and diagnosis of melanoma in contemporary times. Our data suggest that patients included in the BRAF-mutation analysis study had received the diagnosis of melanoma more recently than those who were excluded from the study, which could be due to older melanomas being less likely to have adequate tissue specimens available for immunohistochemical staining/evaluation.
Prior research has shown that BRAF-mutated melanomas typically occur on the trunk and are more likely in individuals with more than 14 nevi on the back.2 In the present cohort, BRAF-positive melanomas had a predisposition toward the trunk but also were found on the head, neck, and extremities—areas that are more likely to have long-term sun damage. One suggestion is that 2 distinct pathways for melanoma development exist: one associated with a large number of melanocytic nevi (that is more prone to genetic mutations in melanocytes) and the other associated with long-term sun exposure.15,16 The combination of these hypotheses suggests that individuals who are prone to the development of large numbers of nevi may require sun exposure for the initial insult, but the development of melanoma may be carried out by other factors after this initial sun exposure insult, whereas individuals without large numbers of nevi who may have less genetic risk may require continued long-term sun exposure for melanoma to develop.17
Our study had limitations, including the small numbers of deaths overall and cause-specific deaths of metastatic melanoma, which limited our ability to conduct more extensive multivariable modeling. Also, the retrospective nature and time frame of looking back 4 decades did not allow us to have information sufficient to categorize some patients as having dysplastic nevus syndrome or not, which would be a potentially interesting variable to include in the analysis. Because the number of deaths in the 18- to 39-year-old cohort was only 5, further statistical comparison regarding tumor type and other variables pertaining to BRAF positivity were not possible. In addition, our data were collected from patients residing in a single geographic county (Olmsted County, Minnesota), which may limit generalizability. Lastly, BRAF V600E mutations were identified through immunostaining only, not molecular data, so it is possible some patients had false-negative immunohistochemistry findings and thus were not identified.
Conclusion
BRAF-mutated melanomas were found in 35% of our cohort, with no significant change in the percentage of melanomas with BRAF V600E mutations over the last 4 decades in this population. In addition, no differences or significant trends existed according to sex and BRAF-mutated melanoma development. Women with BRAF-mutated melanomas were more likely to die of metastatic melanoma than men, and young adults with BRAF-mutated melanomas had a higher all-cause mortality risk. Further research is needed to decipher what effect BRAF-mutated melanomas have on metastasis and cause-specific death in women as well as all-cause mortality in young adults.
Acknowledgment—The authors are indebted to Scientific Publications, Mayo Clinic (Rochester, Minnesota).
- Grimaldi AM, Cassidy PB, Leachmann S, et al. Novel approaches in melanoma prevention and therapy. Cancer Treat Res. 2014;159: 443-455.
- Thomas NE, Edmiston SN, Alexander A, et al. Number of nevi and early-life ambient UV exposure are associated with BRAF-mutant melanoma. Cancer Epidemiol Biomarkers Prev. 2007;16:991-997.
- Curtin JA, Fridlyand J, Kageshita T, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005;353:2135-2147.
- Thomas NE, Edmiston SN, Alexander A, et al. Association between NRAS and BRAF mutational status and melanoma-specific survival among patients with higher-risk primary melanoma. JAMA Oncol. 2015;1:359-368.
- Liu W, Kelly JW, Trivett M, et al. Distinct clinical and pathological features are associated with the BRAF(T1799A(V600E)) mutation in primary melanoma. J Invest Dermatol. 2007;127:900-905.
- Kim SY, Kim SN, Hahn HJ, et al. Metaanalysis of BRAF mutations and clinicopathologic characteristics in primary melanoma. J Am Acad Dermatol. 2015;72:1036-1046.e2.
- Larsen AC, Dahl C, Dahmcke CM, et al. BRAF mutations in conjunctival melanoma: investigation of incidence, clinicopathological features, prognosis and paired premalignant lesions. Acta Ophthalmol. 2016;94:463-470.
- Shinozaki M, Fujimoto A, Morton DL, et al. Incidence of BRAF oncogene mutation and clinical relevance for primary cutaneous melanomas. Clin Cancer Res. 2004;10:1753-1757.
- Heppt MV, Siepmann T, Engel J, et al. Prognostic significance of BRAF and NRAS mutations in melanoma: a German study from routine care. BMC Cancer. 2017;17:536.
- Mar VJ, Liu W, Devitt B, et al. The role of BRAF mutations in primary melanoma growth rate and survival. Br J Dermatol. 2015;173:76-82.
- Rocca WA, Yawn BP, St Sauver JL, et al. History of the Rochester Epidemiology Project: half a century of medical records linkage in a US population. Mayo Clin Proc. 2012;87:1202-1213.
- Reed KB, Brewer JD, Lohse CM, et al. Increasing incidence of melanoma among young adults: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2012;87:328-334.
- Olazagasti Lourido JM, Ma JE, Lohse CM, et al. Increasing incidence of melanoma in the elderly: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2016;91:1555-1562.
- Lowe GC, Saavedra A, Reed KB, et al. Increasing incidence of melanoma among middle-aged adults: an epidemiologic study in Olmsted County, Minnesota. Mayo Clin Proc. 2014;89:52-59.
- Whiteman DC, Parsons PG, Green AC. p53 expression and risk factors for cutaneous melanoma: a case-control study. Int J Cancer. 1998;77:843-848.
- Whiteman DC, Watt P, Purdie DM, et al. Melanocytic nevi, solar keratoses, and divergent pathways to cutaneous melanoma. J Natl Cancer Inst. 2003;95:806-812.
- Olsen CM, Zens MS, Green AC, et al. Biologic markers of sun exposure and melanoma risk in women: pooled case-control analysis. Int J Cancer. 2011;129:713-723.
- Grimaldi AM, Cassidy PB, Leachmann S, et al. Novel approaches in melanoma prevention and therapy. Cancer Treat Res. 2014;159: 443-455.
- Thomas NE, Edmiston SN, Alexander A, et al. Number of nevi and early-life ambient UV exposure are associated with BRAF-mutant melanoma. Cancer Epidemiol Biomarkers Prev. 2007;16:991-997.
- Curtin JA, Fridlyand J, Kageshita T, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005;353:2135-2147.
- Thomas NE, Edmiston SN, Alexander A, et al. Association between NRAS and BRAF mutational status and melanoma-specific survival among patients with higher-risk primary melanoma. JAMA Oncol. 2015;1:359-368.
- Liu W, Kelly JW, Trivett M, et al. Distinct clinical and pathological features are associated with the BRAF(T1799A(V600E)) mutation in primary melanoma. J Invest Dermatol. 2007;127:900-905.
- Kim SY, Kim SN, Hahn HJ, et al. Metaanalysis of BRAF mutations and clinicopathologic characteristics in primary melanoma. J Am Acad Dermatol. 2015;72:1036-1046.e2.
- Larsen AC, Dahl C, Dahmcke CM, et al. BRAF mutations in conjunctival melanoma: investigation of incidence, clinicopathological features, prognosis and paired premalignant lesions. Acta Ophthalmol. 2016;94:463-470.
- Shinozaki M, Fujimoto A, Morton DL, et al. Incidence of BRAF oncogene mutation and clinical relevance for primary cutaneous melanomas. Clin Cancer Res. 2004;10:1753-1757.
- Heppt MV, Siepmann T, Engel J, et al. Prognostic significance of BRAF and NRAS mutations in melanoma: a German study from routine care. BMC Cancer. 2017;17:536.
- Mar VJ, Liu W, Devitt B, et al. The role of BRAF mutations in primary melanoma growth rate and survival. Br J Dermatol. 2015;173:76-82.
- Rocca WA, Yawn BP, St Sauver JL, et al. History of the Rochester Epidemiology Project: half a century of medical records linkage in a US population. Mayo Clin Proc. 2012;87:1202-1213.
- Reed KB, Brewer JD, Lohse CM, et al. Increasing incidence of melanoma among young adults: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2012;87:328-334.
- Olazagasti Lourido JM, Ma JE, Lohse CM, et al. Increasing incidence of melanoma in the elderly: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2016;91:1555-1562.
- Lowe GC, Saavedra A, Reed KB, et al. Increasing incidence of melanoma among middle-aged adults: an epidemiologic study in Olmsted County, Minnesota. Mayo Clin Proc. 2014;89:52-59.
- Whiteman DC, Parsons PG, Green AC. p53 expression and risk factors for cutaneous melanoma: a case-control study. Int J Cancer. 1998;77:843-848.
- Whiteman DC, Watt P, Purdie DM, et al. Melanocytic nevi, solar keratoses, and divergent pathways to cutaneous melanoma. J Natl Cancer Inst. 2003;95:806-812.
- Olsen CM, Zens MS, Green AC, et al. Biologic markers of sun exposure and melanoma risk in women: pooled case-control analysis. Int J Cancer. 2011;129:713-723.
Practice Points
- Approximately 50% of melanomas contain BRAF mutations; the effects on survival are unclear.
- Women with BRAF-mutated melanoma are at increased risk for death from melanoma.
- BRAF expression is associated with death of any cause for adults aged 18 to 39 years.