User login
The Clinical Utility of Teledermatology in Triaging and Diagnosing Skin Malignancies: Case Series
With the increasing utilization of telemedicine since the COVID-19 pandemic, it is critical that clinicians have an appropriate understanding of the application of virtual care resources, including teledermatology. We present a case series of 3 patients to demonstrate the clinical utility of teledermatology in reducing the time to diagnosis of various rare and/or aggressive cutaneous malignancies, including Merkel cell carcinoma, malignant melanoma, and atypical fibroxanthoma. Cases were obtained from one large Midwestern medical center during the month of July 2021. Each case presented includes a description of the initial teledermatology presentation and reviews the clinical timeline from initial consultation submission to in-person clinic visit with lesion biopsy. This case series demonstrates real-world examples of how teledermatology can be utilized to expedite the care of specific vulnerable patient populations.
Teledermatology is a rapidly growing digital resource with specific utility in triaging patients to determine those requiring in-person evaluation for early and accurate detection of skin malignancies. Approximately one-third of teledermatology consultations result in face-to-face clinical encounters, with malignant neoplasms being the leading cause for biopsy.1,2 For specific populations, such as geriatric and immunocompromised patients, teledermatology may serve as a valuable tool, particularly in the wake of the COVID-19 pandemic. Furthermore, telemedicine may aid in addressing health disparities within the field of medicine and ultimately may improve access to care for vulnerable populations.3 Along with increasing access to specific subspecialty expertise, the use of teledermatology may reduce health care costs and improve the overall quality of care delivered to patients.4,5
We describe the clinical utility of teledermatology in triaging and diagnosing skin malignancies through a series of 3 cases obtained from digital image review at one large Midwestern medical center during the month of July 2021. Three unique cases with a final diagnosis of a rare or aggressive skin cancer were selected as examples, including a 75-year-old man with Merkle cell carcinoma, a 55-year-old man with aggressive pT3b malignant melanoma, and a 72-year-old man with an atypical fibroxanthoma. A clinical timeline of each case is presented, including the time intervals from initial image submission to image review, image submission to face-to-face clinical encounter, and image submission to final diagnosis. In all cases, the primary care provider submitted an order for teledermatology, and the teledermatology team obtained the images.
Case Series
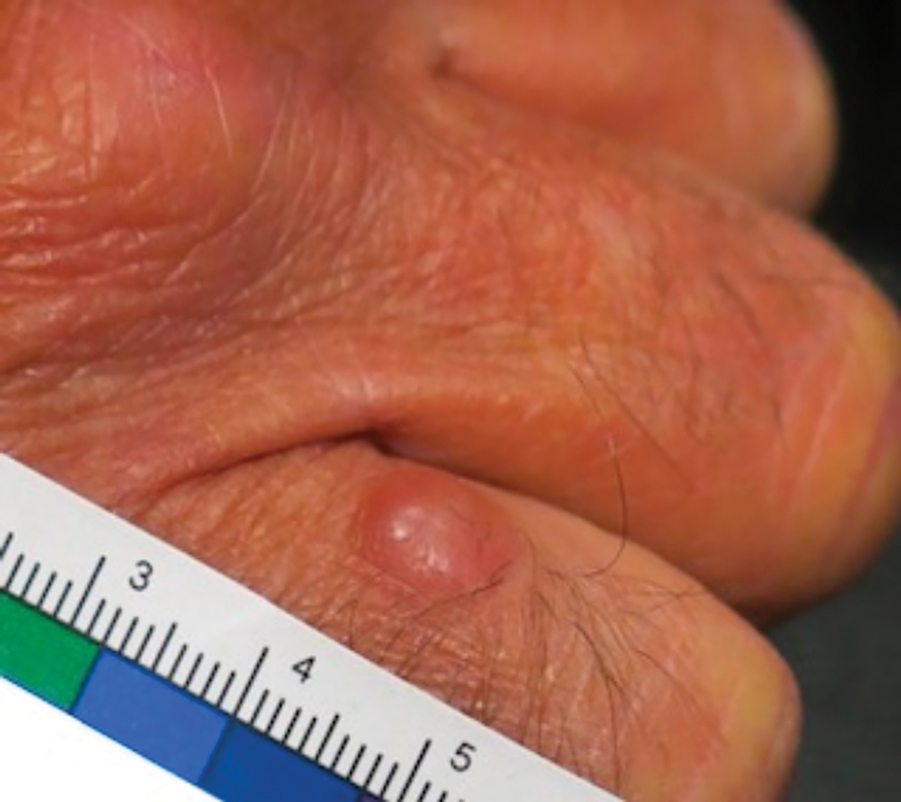
Patient 1—Images of the right hand of a 75-year-old man with a medical history of basal cell carcinoma were submitted for teledermatology consultation utilizing store-and-forward image-capturing technology (day 1). The patient history provided with image submission indicated that the lesion had been present for 6 months and there were no associated symptoms. Clinical imaging demonstrated a pink-red pearly papule located on the proximal fourth digit of the dorsal aspect of the right hand (Figure 1). One day following the teledermatology request (day 2), the patient’s case was reviewed and triaged for an in-person visit. The patient was brought to clinic on day 34, and a biopsy was performed. On day 36, dermatopathology results indicated a diagnosis of Merkel cell carcinoma. On day 37, the patient was referred to surgical oncology, and on day 44, the patient underwent an initial surgical oncology visit with a plan for wide local excision of the right fourth digit with right axillary sentinel lymph node biopsy.
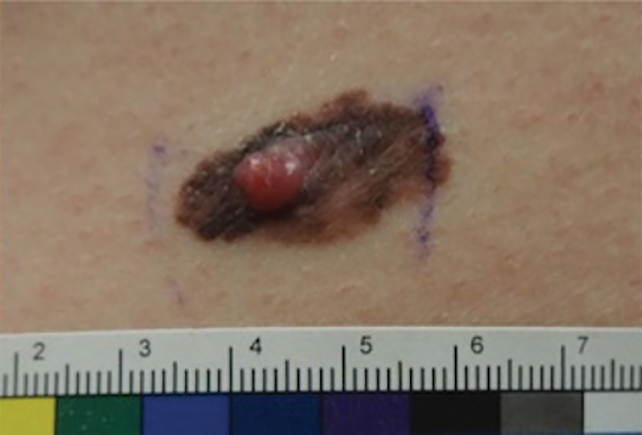
Patient 2—Images of the left flank of a 55-year-old man were submitted for teledermatology consultation via store-and-forward technology (day 1). A patient history provided with the image indicated that the lesion had been present for months to years and there were no associated symptoms, but the lesion recently had changed in color and size. Teledermatology images were reviewed on day 3 and demonstrated a 2- to 3-cm brown plaque on the left flank with color variegation and a prominent red papule protruding centrally (Figure 2). The patient was scheduled for an urgent in-person visit with biopsy. On day 6, the patient presented to clinic and an excision biopsy was performed. Dermatopathology was ordered with a RUSH indication, with results on day 7 revealing a pT3b malignant melanoma. An urgent consultation to surgical oncology was placed on the same day, and the patient underwent an initial surgical oncology visit on day 24 with a plan for wide local excision with left axillary and inguinal sentinel lymph node biopsy.
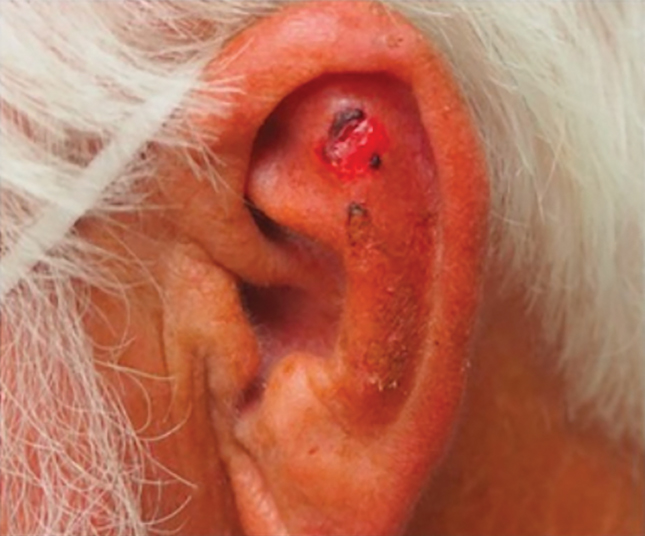
Patient 3—Images of the left ear of a 72-year-old man were submitted for teledermatology consultation utilizing review via store-and-forward technology (day 1). A patient history indicated that the lesion had been present for 3 months with associated bleeding. Image review demonstrated a solitary pearly pink papule located on the crura of the antihelix (Figure 3). Initial teledermatology consultation was reviewed on day 2 with notification of the need for in-person evaluation. The patient presented to clinic on day 33 for a biopsy, with dermatopathology results on day 36 consistent with an atypical fibroxanthoma. The patient was scheduled for Mohs micrographic surgery on day 37 and underwent surgical treatment on day 64.
Comment
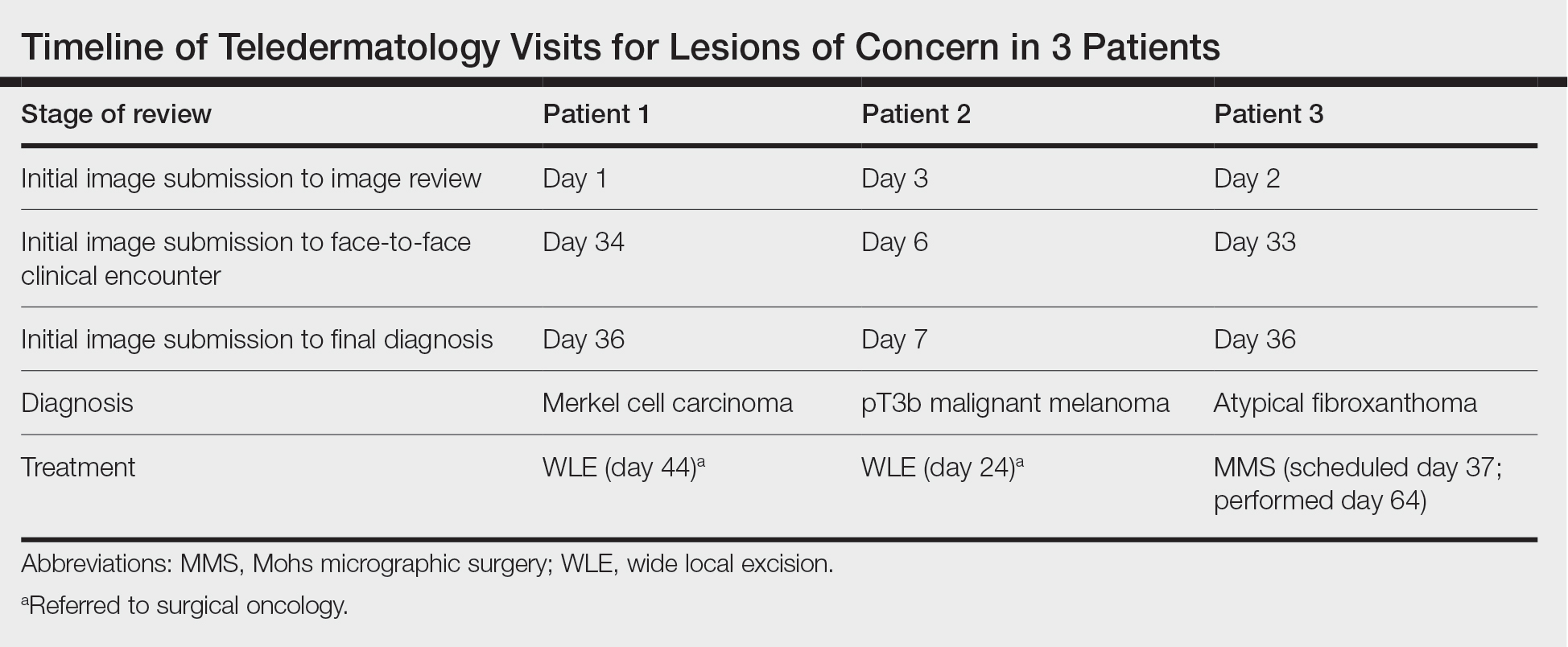
Teledermatology consultations from all patients demonstrated adequate image quality to be able to evaluate the lesion of concern and yielded a request for in-person evaluation with possible biopsy (Table). In this case series, the average time interval from teledermatology consultation placement to teledermatology image report was 2 days (range, 1–3 days). The average time from teledermatology consultation placement to face-to-face encounter with biopsy was 24.3 days for the 3 cases presented in this series (range, 6–34 days). The initial surgical oncology visits took place an average of 34 days after the initial teledermatology consultation was placed for the 2 patients requiring referral (44 days for patient 1; 24 days for patient 2). For patient 3, Mohs micrographic surgery was required for treatment, which was scheduled by day 37 and subsequently performed on day 64.
When specifically looking at the diagnosis of cutaneous malignancies, studies have found that the incidence of skin cancer detection is similar for teledermatology compared to in-person clinic visits.6,7 Creighton-Smith et al6 performed a retrospective cohort study comparing prebiopsy and postbiopsy diagnostic accuracy and detection rates of skin cancer between store-and-forward technology and face-to-face consultation. When adjusting for possible compounding factors including personal and family history of skin cancer, there was no notable difference in detection rates of any skin cancer, including melanoma and nonmelanoma skin cancers. Furthermore, the 2 cohorts of patients were found to have similar prebiopsy and postbiopsy diagnostic concordance, with similar times from consultation being placed to requested biopsy and time from biopsy to final treatment.6
Clarke et al7 similarly analyzed the accuracy of store-and-forward teledermatology and found that there was overall concordance in diagnosis when comparing clinical dermatologists to teledermatologists. Moreover, when melanocytic lesions were excluded from the study, the decision to biopsy did not differ substantially.7
Areas of further study include determining what percentage of teledermatology lesions of concern for malignancy were proven to be skin cancer after in-person evaluation and biopsy, as well as investigating the effectiveness of teledermatology for melanocytic lesions, which frequently are removed from analysis in large-scale teledermatology studies.
Although teledermatology has substantial clinical utility and may serve as a great resource for specific populations, including geriatric patients and those who are immunocompromised, it is important to recognize notable limitations. Specifically, brief history and image review should not serve as replacements for a face-to-face visit with physical examination in cases where the diagnosis remains uncertain or when high-risk skin malignancies are suspected or included in the differential. Certain aggressive cutaneous malignancies such as Merkel cell carcinoma may appear as less aggressive via teledermatology due to restrictions of technology.
Conclusion
Teledermatology has had a major impact on the way health care is delivered to patients and may increase access to care, reducing unnecessary in-person visits and decreasing the number of in-person visit no-shows. With the appropriate use of a brief clinical history and image review, teledermatology can be effective to evaluate specific lesions of concern. We report 3 unique cases identified during a 1-month period at a large Midwestern medical center. These cases serve as important examples of the application of teledermatology in reducing the time to diagnosis of aggressive skin malignancies. Further research on the clinical utility of teledermatology is warranted.
Acknowledgments—The authors thank the additional providers from the University of Wisconsin and William S. Middleton Memorial Veterans Hospital (both in Madison, Wisconsin) involved in the medical care of the patients included in this case series.
- Bianchi MG, Santos A, Cordioli E. Benefits of teledermatology for geriatric patients: population-based cross-sectional study. J Med Internet Res. 2020;22:E16700.
- Mortimer S, Rosin A. A retrospective review of incidental malignancies in veterans seen for face-to-face follow-up after teledermatology consultation. J Am Acad Dermatol. 2021;84:1130-1132.
- Costello CM, Cumsky HJL, Maly CJ, et al. Improving access to care through the establishment of a local, teledermatology network. Telemed J E Health. 2020;26:935-940. doi:10.1089/tmj.2019.0051
- Lee JJ, English JC 3rd. Teledermatology: a review and update. Am J Clin Dermatol. 2018;19:253-260. doi:10.1007/s40257-017-0317-6
- Hadeler E, Beer J, Nouri K. The influence of teledermatology on health care access and equity. J Am Acad Dermatol. 2021;84:E219-E220. doi:10.1016/j.jaad.2020.12.036
- Creighton-Smith M, Murgia RD 3rd, Konnikov N, et al. Incidence of melanoma and keratinocytic carcinomas in patients evaluated by store-and-forward teledermatology vs dermatology clinic. Int J Dermatol. 2017;56:1026-1031. doi:10.1111/ijd.13672
- Clarke EL, Reichenberg JS, Ahmed AM, et al. The utility of teledermatology in the evaluation of skin lesions. J Telemed Telecare. 2023;29:382-389. doi:10.1177/1357633X20987423
With the increasing utilization of telemedicine since the COVID-19 pandemic, it is critical that clinicians have an appropriate understanding of the application of virtual care resources, including teledermatology. We present a case series of 3 patients to demonstrate the clinical utility of teledermatology in reducing the time to diagnosis of various rare and/or aggressive cutaneous malignancies, including Merkel cell carcinoma, malignant melanoma, and atypical fibroxanthoma. Cases were obtained from one large Midwestern medical center during the month of July 2021. Each case presented includes a description of the initial teledermatology presentation and reviews the clinical timeline from initial consultation submission to in-person clinic visit with lesion biopsy. This case series demonstrates real-world examples of how teledermatology can be utilized to expedite the care of specific vulnerable patient populations.
Teledermatology is a rapidly growing digital resource with specific utility in triaging patients to determine those requiring in-person evaluation for early and accurate detection of skin malignancies. Approximately one-third of teledermatology consultations result in face-to-face clinical encounters, with malignant neoplasms being the leading cause for biopsy.1,2 For specific populations, such as geriatric and immunocompromised patients, teledermatology may serve as a valuable tool, particularly in the wake of the COVID-19 pandemic. Furthermore, telemedicine may aid in addressing health disparities within the field of medicine and ultimately may improve access to care for vulnerable populations.3 Along with increasing access to specific subspecialty expertise, the use of teledermatology may reduce health care costs and improve the overall quality of care delivered to patients.4,5
We describe the clinical utility of teledermatology in triaging and diagnosing skin malignancies through a series of 3 cases obtained from digital image review at one large Midwestern medical center during the month of July 2021. Three unique cases with a final diagnosis of a rare or aggressive skin cancer were selected as examples, including a 75-year-old man with Merkle cell carcinoma, a 55-year-old man with aggressive pT3b malignant melanoma, and a 72-year-old man with an atypical fibroxanthoma. A clinical timeline of each case is presented, including the time intervals from initial image submission to image review, image submission to face-to-face clinical encounter, and image submission to final diagnosis. In all cases, the primary care provider submitted an order for teledermatology, and the teledermatology team obtained the images.
Case Series
Patient 1—Images of the right hand of a 75-year-old man with a medical history of basal cell carcinoma were submitted for teledermatology consultation utilizing store-and-forward image-capturing technology (day 1). The patient history provided with image submission indicated that the lesion had been present for 6 months and there were no associated symptoms. Clinical imaging demonstrated a pink-red pearly papule located on the proximal fourth digit of the dorsal aspect of the right hand (Figure 1). One day following the teledermatology request (day 2), the patient’s case was reviewed and triaged for an in-person visit. The patient was brought to clinic on day 34, and a biopsy was performed. On day 36, dermatopathology results indicated a diagnosis of Merkel cell carcinoma. On day 37, the patient was referred to surgical oncology, and on day 44, the patient underwent an initial surgical oncology visit with a plan for wide local excision of the right fourth digit with right axillary sentinel lymph node biopsy.

Patient 2—Images of the left flank of a 55-year-old man were submitted for teledermatology consultation via store-and-forward technology (day 1). A patient history provided with the image indicated that the lesion had been present for months to years and there were no associated symptoms, but the lesion recently had changed in color and size. Teledermatology images were reviewed on day 3 and demonstrated a 2- to 3-cm brown plaque on the left flank with color variegation and a prominent red papule protruding centrally (Figure 2). The patient was scheduled for an urgent in-person visit with biopsy. On day 6, the patient presented to clinic and an excision biopsy was performed. Dermatopathology was ordered with a RUSH indication, with results on day 7 revealing a pT3b malignant melanoma. An urgent consultation to surgical oncology was placed on the same day, and the patient underwent an initial surgical oncology visit on day 24 with a plan for wide local excision with left axillary and inguinal sentinel lymph node biopsy.

Patient 3—Images of the left ear of a 72-year-old man were submitted for teledermatology consultation utilizing review via store-and-forward technology (day 1). A patient history indicated that the lesion had been present for 3 months with associated bleeding. Image review demonstrated a solitary pearly pink papule located on the crura of the antihelix (Figure 3). Initial teledermatology consultation was reviewed on day 2 with notification of the need for in-person evaluation. The patient presented to clinic on day 33 for a biopsy, with dermatopathology results on day 36 consistent with an atypical fibroxanthoma. The patient was scheduled for Mohs micrographic surgery on day 37 and underwent surgical treatment on day 64.

Comment
Teledermatology consultations from all patients demonstrated adequate image quality to be able to evaluate the lesion of concern and yielded a request for in-person evaluation with possible biopsy (Table). In this case series, the average time interval from teledermatology consultation placement to teledermatology image report was 2 days (range, 1–3 days). The average time from teledermatology consultation placement to face-to-face encounter with biopsy was 24.3 days for the 3 cases presented in this series (range, 6–34 days). The initial surgical oncology visits took place an average of 34 days after the initial teledermatology consultation was placed for the 2 patients requiring referral (44 days for patient 1; 24 days for patient 2). For patient 3, Mohs micrographic surgery was required for treatment, which was scheduled by day 37 and subsequently performed on day 64.

When specifically looking at the diagnosis of cutaneous malignancies, studies have found that the incidence of skin cancer detection is similar for teledermatology compared to in-person clinic visits.6,7 Creighton-Smith et al6 performed a retrospective cohort study comparing prebiopsy and postbiopsy diagnostic accuracy and detection rates of skin cancer between store-and-forward technology and face-to-face consultation. When adjusting for possible compounding factors including personal and family history of skin cancer, there was no notable difference in detection rates of any skin cancer, including melanoma and nonmelanoma skin cancers. Furthermore, the 2 cohorts of patients were found to have similar prebiopsy and postbiopsy diagnostic concordance, with similar times from consultation being placed to requested biopsy and time from biopsy to final treatment.6
Clarke et al7 similarly analyzed the accuracy of store-and-forward teledermatology and found that there was overall concordance in diagnosis when comparing clinical dermatologists to teledermatologists. Moreover, when melanocytic lesions were excluded from the study, the decision to biopsy did not differ substantially.7
Areas of further study include determining what percentage of teledermatology lesions of concern for malignancy were proven to be skin cancer after in-person evaluation and biopsy, as well as investigating the effectiveness of teledermatology for melanocytic lesions, which frequently are removed from analysis in large-scale teledermatology studies.
Although teledermatology has substantial clinical utility and may serve as a great resource for specific populations, including geriatric patients and those who are immunocompromised, it is important to recognize notable limitations. Specifically, brief history and image review should not serve as replacements for a face-to-face visit with physical examination in cases where the diagnosis remains uncertain or when high-risk skin malignancies are suspected or included in the differential. Certain aggressive cutaneous malignancies such as Merkel cell carcinoma may appear as less aggressive via teledermatology due to restrictions of technology.
Conclusion
Teledermatology has had a major impact on the way health care is delivered to patients and may increase access to care, reducing unnecessary in-person visits and decreasing the number of in-person visit no-shows. With the appropriate use of a brief clinical history and image review, teledermatology can be effective to evaluate specific lesions of concern. We report 3 unique cases identified during a 1-month period at a large Midwestern medical center. These cases serve as important examples of the application of teledermatology in reducing the time to diagnosis of aggressive skin malignancies. Further research on the clinical utility of teledermatology is warranted.
Acknowledgments—The authors thank the additional providers from the University of Wisconsin and William S. Middleton Memorial Veterans Hospital (both in Madison, Wisconsin) involved in the medical care of the patients included in this case series.
With the increasing utilization of telemedicine since the COVID-19 pandemic, it is critical that clinicians have an appropriate understanding of the application of virtual care resources, including teledermatology. We present a case series of 3 patients to demonstrate the clinical utility of teledermatology in reducing the time to diagnosis of various rare and/or aggressive cutaneous malignancies, including Merkel cell carcinoma, malignant melanoma, and atypical fibroxanthoma. Cases were obtained from one large Midwestern medical center during the month of July 2021. Each case presented includes a description of the initial teledermatology presentation and reviews the clinical timeline from initial consultation submission to in-person clinic visit with lesion biopsy. This case series demonstrates real-world examples of how teledermatology can be utilized to expedite the care of specific vulnerable patient populations.
Teledermatology is a rapidly growing digital resource with specific utility in triaging patients to determine those requiring in-person evaluation for early and accurate detection of skin malignancies. Approximately one-third of teledermatology consultations result in face-to-face clinical encounters, with malignant neoplasms being the leading cause for biopsy.1,2 For specific populations, such as geriatric and immunocompromised patients, teledermatology may serve as a valuable tool, particularly in the wake of the COVID-19 pandemic. Furthermore, telemedicine may aid in addressing health disparities within the field of medicine and ultimately may improve access to care for vulnerable populations.3 Along with increasing access to specific subspecialty expertise, the use of teledermatology may reduce health care costs and improve the overall quality of care delivered to patients.4,5
We describe the clinical utility of teledermatology in triaging and diagnosing skin malignancies through a series of 3 cases obtained from digital image review at one large Midwestern medical center during the month of July 2021. Three unique cases with a final diagnosis of a rare or aggressive skin cancer were selected as examples, including a 75-year-old man with Merkle cell carcinoma, a 55-year-old man with aggressive pT3b malignant melanoma, and a 72-year-old man with an atypical fibroxanthoma. A clinical timeline of each case is presented, including the time intervals from initial image submission to image review, image submission to face-to-face clinical encounter, and image submission to final diagnosis. In all cases, the primary care provider submitted an order for teledermatology, and the teledermatology team obtained the images.
Case Series
Patient 1—Images of the right hand of a 75-year-old man with a medical history of basal cell carcinoma were submitted for teledermatology consultation utilizing store-and-forward image-capturing technology (day 1). The patient history provided with image submission indicated that the lesion had been present for 6 months and there were no associated symptoms. Clinical imaging demonstrated a pink-red pearly papule located on the proximal fourth digit of the dorsal aspect of the right hand (Figure 1). One day following the teledermatology request (day 2), the patient’s case was reviewed and triaged for an in-person visit. The patient was brought to clinic on day 34, and a biopsy was performed. On day 36, dermatopathology results indicated a diagnosis of Merkel cell carcinoma. On day 37, the patient was referred to surgical oncology, and on day 44, the patient underwent an initial surgical oncology visit with a plan for wide local excision of the right fourth digit with right axillary sentinel lymph node biopsy.

Patient 2—Images of the left flank of a 55-year-old man were submitted for teledermatology consultation via store-and-forward technology (day 1). A patient history provided with the image indicated that the lesion had been present for months to years and there were no associated symptoms, but the lesion recently had changed in color and size. Teledermatology images were reviewed on day 3 and demonstrated a 2- to 3-cm brown plaque on the left flank with color variegation and a prominent red papule protruding centrally (Figure 2). The patient was scheduled for an urgent in-person visit with biopsy. On day 6, the patient presented to clinic and an excision biopsy was performed. Dermatopathology was ordered with a RUSH indication, with results on day 7 revealing a pT3b malignant melanoma. An urgent consultation to surgical oncology was placed on the same day, and the patient underwent an initial surgical oncology visit on day 24 with a plan for wide local excision with left axillary and inguinal sentinel lymph node biopsy.

Patient 3—Images of the left ear of a 72-year-old man were submitted for teledermatology consultation utilizing review via store-and-forward technology (day 1). A patient history indicated that the lesion had been present for 3 months with associated bleeding. Image review demonstrated a solitary pearly pink papule located on the crura of the antihelix (Figure 3). Initial teledermatology consultation was reviewed on day 2 with notification of the need for in-person evaluation. The patient presented to clinic on day 33 for a biopsy, with dermatopathology results on day 36 consistent with an atypical fibroxanthoma. The patient was scheduled for Mohs micrographic surgery on day 37 and underwent surgical treatment on day 64.

Comment
Teledermatology consultations from all patients demonstrated adequate image quality to be able to evaluate the lesion of concern and yielded a request for in-person evaluation with possible biopsy (Table). In this case series, the average time interval from teledermatology consultation placement to teledermatology image report was 2 days (range, 1–3 days). The average time from teledermatology consultation placement to face-to-face encounter with biopsy was 24.3 days for the 3 cases presented in this series (range, 6–34 days). The initial surgical oncology visits took place an average of 34 days after the initial teledermatology consultation was placed for the 2 patients requiring referral (44 days for patient 1; 24 days for patient 2). For patient 3, Mohs micrographic surgery was required for treatment, which was scheduled by day 37 and subsequently performed on day 64.

When specifically looking at the diagnosis of cutaneous malignancies, studies have found that the incidence of skin cancer detection is similar for teledermatology compared to in-person clinic visits.6,7 Creighton-Smith et al6 performed a retrospective cohort study comparing prebiopsy and postbiopsy diagnostic accuracy and detection rates of skin cancer between store-and-forward technology and face-to-face consultation. When adjusting for possible compounding factors including personal and family history of skin cancer, there was no notable difference in detection rates of any skin cancer, including melanoma and nonmelanoma skin cancers. Furthermore, the 2 cohorts of patients were found to have similar prebiopsy and postbiopsy diagnostic concordance, with similar times from consultation being placed to requested biopsy and time from biopsy to final treatment.6
Clarke et al7 similarly analyzed the accuracy of store-and-forward teledermatology and found that there was overall concordance in diagnosis when comparing clinical dermatologists to teledermatologists. Moreover, when melanocytic lesions were excluded from the study, the decision to biopsy did not differ substantially.7
Areas of further study include determining what percentage of teledermatology lesions of concern for malignancy were proven to be skin cancer after in-person evaluation and biopsy, as well as investigating the effectiveness of teledermatology for melanocytic lesions, which frequently are removed from analysis in large-scale teledermatology studies.
Although teledermatology has substantial clinical utility and may serve as a great resource for specific populations, including geriatric patients and those who are immunocompromised, it is important to recognize notable limitations. Specifically, brief history and image review should not serve as replacements for a face-to-face visit with physical examination in cases where the diagnosis remains uncertain or when high-risk skin malignancies are suspected or included in the differential. Certain aggressive cutaneous malignancies such as Merkel cell carcinoma may appear as less aggressive via teledermatology due to restrictions of technology.
Conclusion
Teledermatology has had a major impact on the way health care is delivered to patients and may increase access to care, reducing unnecessary in-person visits and decreasing the number of in-person visit no-shows. With the appropriate use of a brief clinical history and image review, teledermatology can be effective to evaluate specific lesions of concern. We report 3 unique cases identified during a 1-month period at a large Midwestern medical center. These cases serve as important examples of the application of teledermatology in reducing the time to diagnosis of aggressive skin malignancies. Further research on the clinical utility of teledermatology is warranted.
Acknowledgments—The authors thank the additional providers from the University of Wisconsin and William S. Middleton Memorial Veterans Hospital (both in Madison, Wisconsin) involved in the medical care of the patients included in this case series.
- Bianchi MG, Santos A, Cordioli E. Benefits of teledermatology for geriatric patients: population-based cross-sectional study. J Med Internet Res. 2020;22:E16700.
- Mortimer S, Rosin A. A retrospective review of incidental malignancies in veterans seen for face-to-face follow-up after teledermatology consultation. J Am Acad Dermatol. 2021;84:1130-1132.
- Costello CM, Cumsky HJL, Maly CJ, et al. Improving access to care through the establishment of a local, teledermatology network. Telemed J E Health. 2020;26:935-940. doi:10.1089/tmj.2019.0051
- Lee JJ, English JC 3rd. Teledermatology: a review and update. Am J Clin Dermatol. 2018;19:253-260. doi:10.1007/s40257-017-0317-6
- Hadeler E, Beer J, Nouri K. The influence of teledermatology on health care access and equity. J Am Acad Dermatol. 2021;84:E219-E220. doi:10.1016/j.jaad.2020.12.036
- Creighton-Smith M, Murgia RD 3rd, Konnikov N, et al. Incidence of melanoma and keratinocytic carcinomas in patients evaluated by store-and-forward teledermatology vs dermatology clinic. Int J Dermatol. 2017;56:1026-1031. doi:10.1111/ijd.13672
- Clarke EL, Reichenberg JS, Ahmed AM, et al. The utility of teledermatology in the evaluation of skin lesions. J Telemed Telecare. 2023;29:382-389. doi:10.1177/1357633X20987423
- Bianchi MG, Santos A, Cordioli E. Benefits of teledermatology for geriatric patients: population-based cross-sectional study. J Med Internet Res. 2020;22:E16700.
- Mortimer S, Rosin A. A retrospective review of incidental malignancies in veterans seen for face-to-face follow-up after teledermatology consultation. J Am Acad Dermatol. 2021;84:1130-1132.
- Costello CM, Cumsky HJL, Maly CJ, et al. Improving access to care through the establishment of a local, teledermatology network. Telemed J E Health. 2020;26:935-940. doi:10.1089/tmj.2019.0051
- Lee JJ, English JC 3rd. Teledermatology: a review and update. Am J Clin Dermatol. 2018;19:253-260. doi:10.1007/s40257-017-0317-6
- Hadeler E, Beer J, Nouri K. The influence of teledermatology on health care access and equity. J Am Acad Dermatol. 2021;84:E219-E220. doi:10.1016/j.jaad.2020.12.036
- Creighton-Smith M, Murgia RD 3rd, Konnikov N, et al. Incidence of melanoma and keratinocytic carcinomas in patients evaluated by store-and-forward teledermatology vs dermatology clinic. Int J Dermatol. 2017;56:1026-1031. doi:10.1111/ijd.13672
- Clarke EL, Reichenberg JS, Ahmed AM, et al. The utility of teledermatology in the evaluation of skin lesions. J Telemed Telecare. 2023;29:382-389. doi:10.1177/1357633X20987423
Practice Points
- Teledermatology via store-and-forward technology has been demonstrated to be effective in assessing and triaging various cutaneous malignancies.
- The use of teledermatology has increased because of the COVID-19 pandemic and may be useful for specific vulnerable populations.
- When used appropriately, teledermatology may function as a useful resource to triage patients requiring in-person evaluation for the diagnosis of aggressive skin malignancies and may aid in reducing the time to diagnosis of various skin cancers.
The Role of Dietary Antioxidants in Melanoma and Nonmelanoma Skin Cancer
Nonmelanoma skin cancer (NMSC) is the most common cancer in the United States, and cutaneous melanoma is projected to be the fifth most common form of cancer in 2022, with increasing incidence and high potential for mortality.1-3 Estimates indicate that 35% to 45% of all cancers in White patients are cutaneous, with 4% to 5% occurring in Hispanic patients, 2% to 4% in Asian patients, and 1% to 2% in Black patients.4 Of the keratinocyte carcinomas, basal cell carcinoma (BCC) is the most prevalent, projected to affect approximately 33% to 39% of White males and 23% to 28% of White females in the United States during their lifetimes. Squamous cell carcinoma (SCC) is the second most common skin malignancy, with a lifetime risk of 9% to 14% for White males and 4% to 9% for White females in the United States.5 The incidence of melanoma continues to increase, with approximately 99,780 new cases expected in the United States in 2022.1
UV-induced DNA damage plays a key role in the pathogenesis and development of various skin malignancies.6 UV radiation from sunlight or tanning devices causes photocarcinogenesis due to molecular and cellular effects, including the generation of reactive oxygen species, DNA damage due to the formation of cyclobutane pyrimidine dimers and pyrimidine-pyrimidone, melanogenesis, apoptosis, and the increased expression of harmful genes and proteins.6 The summation of this damage can result in skin malignancies, including NMSC and melanoma.6,7 Dietary antioxidants theoretically help prevent oxidative reactions from occurring within the body, and it has been suggested that intake of dietary antioxidants may decrease DNA damage and prevent tumorigenesis secondary to UV radiation.8 Antioxidants exist naturally in the body but can be acquired exogenously. Investigators have studied dietary antioxidants in preventing skin cancer formation with promising results in the laboratory setting.8-11 Recently, more robust human studies have been initiated to further delineate this relationship. We present clinical evidence of several frequently utilized antioxidant vitamins and their effects on melanoma and NMSC.
Antioxidants
Vitamin A—Vitamin A is a fat-soluble vitamin found in animal sources, including fish, liver, and eggs. Carotenoids, such as beta carotene, are provitamin A plant derivatives found in fruits and vegetables that are converted into biologically active retinol and retinoic acid.12 Retinols play a key role in cellular growth and differentiation and are thought to be protective against skin cancer via the inactivation of free radicals and immunologic enhancement due to their antiproliferative, antioxidative, and antiapoptotic effects.13-16 Animal studies have demonstrated this protective effect and the ability of retinoids to suppress carcinogenesis; however, human studies reveal conflicting results.17,18
Greenberg et al19 investigated the use of beta carotene in preventing the formation of NMSC. Patients (N=1805) were randomized to receive 50 mg of beta carotene daily or placebo. Over a 5-year period, there was no significant reduction in the occurrence of NMSC (relative risk [RR], 1.05; 95% CI, 0.91-1.22).19 Frieling et al20 conducted a similar randomized, double-blind, placebo-controlled trial investigating beta carotene for primary prevention of NMSC in 22,071 healthy male physicians. The study group received 50 mg of beta carotene every other day for 12 years’ duration, and there was no significant effect on the incidence of first NMSC development (RR, 0.98; 95% CI, 0.92-1.05).20
A case-control study by Naldi et al21 found an inverse association between vitamin A intake and development of melanoma. Study participants were stratified into quartiles based on level of dietary intake and found an odds ratio (OR) of 0.71 for beta carotene (95% CI, 0.50-1.02), 0.57 for retinol (95% CI, 0.39-0.83), and 0.51 for total vitamin A (95% CI, 0.35-0.75) when comparing the upper quartile of vitamin A intake to the lower quartile. Upper-quartile cutoff values of vitamin A intake were 214 µg/d for beta carotene, 149 µg/d for retinol, and 359 µg/d for total vitamin A.21 More recently, a meta-analysis by Zhang et al22 pooled data from 8 case-control studies and 2 prospective studies. Intake of retinol but not total vitamin A or beta carotene was associated with a reduced risk for development of melanoma (retinol: OR, 0.80; 95% CI, 0.69-0.92; total vitamin A: OR, 0.86; 95% CI, 0.59-1.25; beta carotene: OR, 0.87; 95% CI, 0.62-1.20).22 Feskanich et al23 demonstrated similar findings with use of food-frequency questionnaires in White women, suggesting that retinol intake from food combined with supplements may be protective for women who were otherwise at a low risk for melanoma based on nondietary factors. These factors included painful or blistering sunburns during childhood, history of more than 6 sunburns, more than 3 moles on the left arm, having red or blonde hair, and having a parent or sibling with melanoma (P=.01). However, this relationship did not hold true when looking at women at an intermediate or high risk for melanoma (P=.16 and P=.46).23
When looking at high-risk patients, such as transplant patients, oral retinoids have been beneficial in preventing NMSC.24-27 Bavinck et al24 investigated 44 renal transplant patients with a history of more than 10 NMSCs treated with 30 mg of acitretin daily vs placebo. Patients receiving oral retinoid supplementation developed fewer NMSCs over a 6-month treatment period (P=.01).24 Similarly, George et al25 investigated acitretin in renal transplant patients and found a statistically significant decrease in number of SCCs in patients on supplementation (P=.002). Solomon-Cohen et al26 performed a retrospective case-crossover study in solid organ transplant recipients and found that those treated with 10 mg of acitretin daily for 2 years had a significant reduction in the number of new keratinocyte carcinomas (P=.002). Other investigators have demonstrated similar results, and in 2006, Otley et al27 proposed standardized dosing of acitretin for chemoprevention in high-risk patients, including patients developing 5 to 10 NMSCs per year, solid organ transplant recipients, and those with syndromes associated with the development of NMSC.28,29 Overall, in the general population, vitamin A and related compounds have not demonstrated a significant association with decreased development of NMSC; however, oral retinoids have proven useful for high-risk patients. Furthermore, several studies have suggested a negative association between vitamin A levels and the incidence of melanoma, specifically in the retinol formulation.
Vitamin B3—Nicotinamide (also known as niacinamide) is a water-soluble form of vitamin B3 and is obtained from animal-based and plant-based foods, such as meat, fish, and legumes.30 Nicotinamide plays a key role in cellular metabolism, cellular signaling, and DNA repair, including protection from UV damage within keratinocytes.31,32 Early mouse models demonstrated decreased formation of skin tumors in mice treated with topical or oral nicotinamide.32,33 A number of human studies have revealed similar results.34-36
Chen et al34 conducted the ONTRAC study, a phase 3, double-blind, randomized controlled trial (RCT) looking at 386 participants with a history of at least 2 NMSCs in the preceding 5 years. At 12 months, those treated with 500 mg of nicotinamide twice daily demonstrated a statistically significant decreased rate of SCC formation (P=.05). A decreased incidence of BCC development was noted; however, this trend did not reach statistical significance (P=.12). Precancerous skin lesions also were found to be decreased in the treatment group, with 20% lower incidence of actinic keratoses (AKs) after 9 months of treatment (P<.001).34 Drago et al35 specifically studied the incidence of AKs in 38 transplant recipients—8 liver and 30 kidney—and found that previously noted AKs had decreased in size for 18 of 19 patients taking 500 mg of nicotinamide daily when originally photographed AKs were remeasured at 6-month follow-up, with 7 of these 18 patients demonstrating complete clinical regression. Of those on nicotinamide supplementation, no new AKs developed compared to the control group, which demonstrated increased size of AKs or development of new AKs in 91% of patients, with 7 AKs progressing into SCC.35
Nicotinamide has been demonstrated to be useful in preventing skin cancer in high-risk populations, such as transplant patients or those with a high incidence of NMSC.34,36 Despite promising results within the laboratory setting, nicotinamide’s effects on melanoma in humans remains less clear.31,37 Studies suggest that nicotinamide enhances tumor-infiltrating lymphocytes and DNA repair mechanisms in melanocytes, which may translate into nicotinamide, providing chemoprevention for melanoma, but research in human patients is limited.31,37
Vitamin B9—Folate, the natural form of vitamin B9, is a water-soluble compound that is found in many foods, especially green leafy vegetables, and often is supplemented because of its health benefits.38,39 In the skin, folic acid plays a key role in cellular replication and proliferation.38 Controversy exists regarding folate’s effects on cellular growth and turnover with respect to cancer incidence.38,40 Donnenfeld et al41 conducted a prospective study assessing dietary folic acid intake and development of NMSC. A total of 5880 participants completed dietary records throughout the first 2 years of the study. After an average follow-up period of 12.6 years, there was an overall increased incidence of skin cancer in those with increased dietary folate (P=.03). Furthermore, when striating by skin cancer type, there was an increased incidence of NMSC overall as well as BCC when analyzing by type of NMSC (P=.03 for NMSC; P=.05 for BCC). However, when stratifying by gender, these findings only held true for women.41 Similar effects were observed by Fung et al,42 who prospectively studied the intake of various vitamins in relationship to the development of BCC in women. During 12 years of follow-up, a positive association was observed between folate intake and BCC development (OR, 1.2; 95% CI, 1.10-1.31).42 Fung et al43 also investigated the role of several vitamins in the development of SCC and found that folate showed a negative association, which did not reach statistical significance (RR, 0.79; 95% CI, 0.56-1.11). Furthermore, Vollset et al40 conducted a meta-analysis comparing folic acid to placebo in the incidence of various types of cancer. The study excluded NMSC but reported no significant association between the development of melanoma and folic acid supplementation.40 In summary, the effects of folate have diverse consequences, potentially promoting the formation of NMSC, but studies suggest that an individual’s gender and other genetic and environmental factors also may play a role.
Vitamin C—Vitamin C (also known as ascorbic acid) is a water-soluble vitamin with antioxidant immune-mediating effects. It is found in various fruits and vegetables and serves as a cofactor for enzymes within the body playing a key role in immune function and collagen formation.44,45 It has been postulated that ascorbic acid can provide protection from UV radiation damage via its intracellular activity but conversely can contribute to oxidative damage.44 Multiple in vitro laboratory studies and animal models have demonstrated photoprotective effects of ascorbic acid.46-48 Despite these findings, minimal photoprotective effects have been found in the human population.
Kune et al49 performed a case-control study of 88 males with previously diagnosed NMSC undergoing surgical removal and investigated patients’ prior dietary habits. Patients with NMSC had a statistically significantly lower level of vitamin C–containing food in their diet than those without NMSC (P=.004).49 In addition, Vural et al50 analyzed plasma samples and blood cells of patients with AK and BCC and found a significant decrease in ascorbic acid levels in both the AK (P<.001) and BCC (P<.001) groups compared with controls. However, studies have found that consumption of certain dietary compounds can rapidly increase plasma concentration levels, which may serve as a major confounding variable in this study. Plasma concentrations of ascorbic acid and beta carotene were found to be significantly increased following consumption of a high-antioxidant diet for as short a duration as 2 weeks (P<.05).51 More recently, Heinen et al52 performed a prospective study on 1001 adults. In patients without a history of skin cancer, they found that vitamin C from food sources plus dietary supplements was positively associated with the development of BCC (P=.03).52 Similarly, Fung et al42 performed a study in women and found a positive association between vitamin C intake and the development of BCC (OR, 1.13; 95% CI, 1.03-1.23).
The relationship between vitamin C intake—either in dietary or supplemental form—and melanoma remains controversial. Mice-based studies found that high concentrations of orally administered vitamin C induce cytotoxicity in melanoma cell lines, but at low concentrations they promote tumor growth of malignant melanoma.53 Feskanich et al23 examined the relationship between vitamin C intake and melanoma development via food frequency questionnaires in White women and found that vitamin C was associated with a higher risk for melanoma (P=.05), and furthermore, a positive dose response with frequency of orange juice intake was observed (P=.008). Overall, despite promising laboratory studies, there is a lack of RCTs investigating the use of vitamin C supplementation for prevention of NMSC and melanoma in humans, and the oral benefits of vitamin C for chemoprevention remain unclear.
Vitamin D—Vitamin D is a fat-soluble vitamin that is found in fish, liver, egg, and cheese, and is endogenously produced when UV radiation from sun exposure interacts with the skin, triggering the synthesis of vitamin D.54 Vitamin D is biologically inactive and must be converted to its active form 1,25-dihydroxyvitamin D after entering the body. Vitamin D modulates many genes involved in cellular proliferation and differentiation.54 Vitamin D receptors are expressed on keratinocytes and melanocytes.55 Animal studies have demonstrated a potentially protective effect of vitamin D in the development of NMSC.56 In a mouse model, Ellison et al56 found that mice without vitamin D receptors developed skin tumors more rapidly than those with vitamin D receptors.
Unfortunately, these findings have not been demonstrated in humans, and studies have even reported an increased risk for development of NMSC in patients with normal or increased vitamin D levels compared with those with low levels of vitamin D.57-60 Eide et al57 studied 3223 patients seeking advice for low bone density by recording their vitamin D levels at the time of presentation and monitoring development of NMSC. Vitamin D levels greater than 15 ng/mL were positively associated with the development of NMSC (OR, 1.7; 95% CI, 1.04-2.7). This association held true for both SCC and BCC, with a higher risk estimated for SCC (OR, 3.2; 95% CI, 0.4-24.0 for SCC; OR, 1.7; 95% CI, 0.5-5.8 for BCC).57 An increased vitamin D serum level also was found to be significantly associated with a higher risk for BCC and melanoma by van der Pols et al.58 This prospective study looked at the incidence of skin cancer over 11 years. Study participants with vitamin D levels over 75 nmol/L more frequently developed BCC (P=.01) and melanoma (P=.05). In contrast, SCC was less frequently observed in participants with these high levels of vitamin D (P=.07).58 Furthermore, Park et al60 looked at vitamin D and skin cancer risk for men and women in the United States and found no association with risk for SCC or melanoma but a positive association with BCC (P=.05 for total vitamin D; P<.01 for dietary vitamin D). Additional studies have been performed with inconsistent results, and multiple authors suggest the possible confounding relationship between vitamin D levels and UV radiation exposure.59-62 Furthermore, some studies have even demonstrated a negative association between vitamin D and NMSC. Tang et al63 performed a retrospective case-control study in elderly males, investigating serum levels of vitamin D and patients’ self-reported history of NMSC, which demonstrated that higher levels of vitamin D were associated with a decreased risk for NMSC. Overall, the relationship between vitamin D and skin cancer development remains unclear for both melanoma and NMSC.
Vitamin E—Vitamin E is a fat-soluble vitamin that is found in plant-based oils, nuts, seeds, fruits, and vegetables.64 It works as an antioxidant to protect against free radicals and heighten immune function, and it also serves as a pro-oxidant.65,66 Vitamin E naturally exists in 8 chemical forms, of which gamma-tocopherol is the most frequently obtained form in the diet, and alpha-tocopherol is the most abundant form found in the body.64,65
Early animal studies demonstrated the inhibition of UV-induced damage in mice receiving vitamin E supplementation.67,68 Human studies have not consistently shown these effects. Vural et al50 investigated plasma samples and blood cells of patients with AKs and BCCs and reported a significant decrease in alpha-tocopherol levels in both the AK (P<.05) and BCC (P<.001) groups compared with controls. However, studies also have demonstrated a positive association between vitamin E intake and the development of BCC, including one by Fung et al,42 which found a significant association in women (OR, 1.15; 95% CI, 1.06-1.26).
Vitamin E has been found to inhibit melanin synthesis in the laboratory, suggesting a potentially protective effect in melanoma.69,70 However, in the study performed by Feskanich et al23 examining vitamin intake and melanoma incidence via food-frequency questionnaires, vitamin E was not associated with a lower risk for melanoma. Despite promising laboratory studies, the data surrounding the use of a vitamin E supplement for prevention of melanoma and NMSC in humans remains unclear.
Selenium—Selenium is a trace mineral found in plants, meat, and fish. It plays a key role in reproduction, hormone metabolism, DNA synthesis, and protection from oxidative damage.71 In mice studies, lack of selenium-containing proteins resulted in skin abnormalities, including the development of a hyperplastic epidermis and aberrant hair follicle morphogenesis with alopecia after birth, and numerous experimental studies have demonstrated a negative association between selenium intake and cancer.72,73 However, human studies have yielded alternative results.
The Nutritional Prevention of Cancer Study Group analyzed 1312 dermatology patients with a history of NMSC.74 The study population was obtained from 7 dermatology clinics with randomization to control for confounding variables. Study participants received either 200 μg of selenium daily or placebo.74 Baseline characteristics of each study group were overall balanced. Selenium intake was found to have no effect on the development of BCC (hazard ratio [HR], 1.09; 95% CI, 0.94-1.26) but an increased risk for developing SCC (HR, 1.25; 95% CI, 1.03-1.51) and total NMSC (HR, 1.17; 95% CI, 1.02-1.34).74,75 Similarly, Reid et al76 performed an RCT comparing patients treated with 400 μg/d of selenium to those treated with 200 μg/d of selenium. When compared with placebo, those treated with 200 μg/d of selenium had a statistically significantly increased incidence of NMSC (P=.006); however, those treated with 400 μg/d of selenium had no significant change in total incidence of NMSC (P=.51).76 Furthermore, Vinceti et al77 performed a review of 83 studies from the literature investigating the effect of dietary selenium, and from the RCTs, there was no beneficial effect of selenium in reducing cancer risk in general; however, some studies demonstrated an increased incidence of other types of cancer, including melanoma. Of the RCTs included in the study investigating NMSC incidence specifically, it was found that the incidence was not affected by selenium administration (RR, 1.16; 95% CI, 0.30-4.42; 2 studies, 2027 participants).77 Despite data from several studies demonstrating an increased risk for NMSC, the effects of selenium on the risk for NMSC and melanoma remain unclear.
Combination Antioxidant Studies
In addition to investigating the use of single antioxidants in skin cancer prevention, studies utilizing the combination of various antioxidants or other dietary minerals have been conducted. Hercberg et al78 performed a randomized, double-blinded, placebo-controlled trial of 13,017 adults (7876 women and 5141 men) receiving a combination of 120 mg vitamin C, 30 mg vitamin E, 100 μg selenium, 6 mg beta carotene, and 20 mg zinc. Study participants were followed for an average of 7.5 years, and the development of skin cancers were recorded. Overall, the incidence rate of skin cancer did not differ between the 2 treatment groups; however, when segregated by gender, the study found that there was an increased risk for developing skin cancer in women taking the antioxidant supplement combination compared with placebo (P=.03). This difference was not observed in the 2 treatment groups of male patients (P=.11). When looking specifically at NMSC, there was no difference between treatment groups for male or female patients (P=.39 for males; P=.15 for females). In contrast, there was a higher incidence of melanoma identified in female patients taking the combination antioxidant supplement (P=.01), but this was not seen within the male study population (P=.51).78 In addition, Chang et al79 performed a meta-analysis of 10 previously published RCTs. Analysis revealed that treatment with a variety of supplements, including vitamins A, C, E, and beta carotene, were found to have no preventative effects on the incidence of skin cancer development (RR, 0.98; CI, 0.98-1.03). Notable limitations to this study included the variability in protocols of the studies included in this meta-analysis, the limited number of RCTs investigating vitamin supplementation and the risk for skin cancer development, and the influence of dietary intake on study outcomes.79
Other Dietary Agents
Furocoumarins—Furocoumarins are botanical substances found in various fruits and plants, including many citrus products. Furocoumarins are activated by UV light radiation and can lead to development of a phototoxic eruption. Several studies have suggested a pharmacogenetic effect of furocoumarins.80 Sun et al80 collected dietary data from 47,453 men and 75,291 women on furocoumarin intake and correlation with the development of NMSC. Overall, the study suggested that the intake of furocoumarins may lead to an increase in the development of BCC (HR, 1.16; 95% CI, 1.11-1.21; P=.002); however, there was no significant association identified between total intake of furocoumarins in the risk for SCC or melanoma.80 Furthermore, Sakaki et al81 conducted a survey study looking at the consumption of citrus products and the development of NMSC. The group found that there was an increased risk for NMSC in those consuming an increased amount of citrus products (P=.007).81
Conclusion
Dietary antioxidants have been investigated for their potential role in the prevention of tumorigenesis. Specific antioxidant vitamins, such as vitamin A derivatives and niacinamide, have demonstrated clinical utility in the prevention of NMSC in high-risk populations. Retinol also has been associated with a reduced incidence of melanoma. Numerous antioxidants have demonstrated promising data within the laboratory setting; however, inconsistent results have been appreciated in humans. Furthermore, several research studies suggest that folate, vitamin D, and furocoumarins may be associated with an increased risk for skin cancer development; however, these studies are inconclusive, and dietary studies are challenging to conduct. Overall, RCTs investigating the role of antioxidants for chemoprevention are limited. Moreover, the study of dietary antioxidants and vitamins may be affected by various confounding variables that can be difficult to account for because of patients’ potentially poor recall of dietary intake and the effect of dietary intake in supplemental studies. Given the increasing prevalence of skin cancer worldwide, further research into the clinical utility of antioxidants in skin cancer prevention is warranted.
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7-33.
- Global Burden of Disease Cancer Collaboration; Fitzmaurice C, Abate D, Abbasi N, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the Global Burden of Disease Study. JAMA Oncol. 2019;5:1749-1768.
- Leiter U, Keim U, Garbe C. Epidemiology of skin cancer: update 2019. In: Reichrath J, ed. Sunlight, Vitamin D and Skin Cancer. Springer International Publishing; 2020:123-139.
- Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206; quiz 178.
- Miller DL, Weinstock MA. Nonmelanoma skin cancer in the United States: incidence. J Am Acad Dermatol. 1994;30:774-778.
- Young AR, Claveau J, Rossi AB. Ultraviolet radiation and the skin: photobiology and sunscreen photoprotection. J Am Acad Dermatol. 2017;76(3S1):S100-S109.
- Pleasance ED, Cheetham RK, Stephens PJ, et al. A comprehensive catalogue of somatic mutations from a human cancer genome. Nature. 2010;463:191-196.
- Baek J, Lee MG. Oxidative stress and antioxidant strategies in dermatology. Redox Rep. 2016;21:164-169.
- Katta R, Brown DN. Diet and skin cancer: the potential role of dietary antioxidants in nonmelanoma skin cancer prevention. J Skin Cancer. 2015;2015:893149.
- Stoj V, Shahriari N, Shao K, et al. Nutrition and nonmelanoma skin cancers. Clin Dermatol. 2022;40:173-185.
- O’Connor EA, Evans CV, Ivlev I, et al. Vitamin and mineral supplements for the primary prevention of cardiovascular disease and cancer: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2022;327:2334-2347.
- National Institutes of Health Office of Dietary Supplements. Vitamin A and carotenoids. fact sheet for health professionals. Updated June 15, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminA-HealthProfessional/
- Keller KL, Fenske NA. Uses of vitamins A, C, and E and related compounds in dermatology: a review. J Am Acad Dermatol. 1998;39:611-625.
- Wright TI, Spencer JM, Flowers FP. Chemoprevention of nonmelanoma skin cancer. J Am Acad Dermatol. 2006;54:933-946; quiz 947-950.
- Bushue N, Wan YJY. Retinoid pathway and cancer therapeutics. Adv Drug Deliv Rev. 2010;62:1285-1298.
- Stahl W, Sies H. β-Carotene and other carotenoids in protection from sunlight. Am J Clin Nutr. 2012;96:1179S-1184S.
- Bukhari MH, Qureshi SS, Niazi S, et al. Chemotherapeutic/chemopreventive role of retinoids in chemically induced skin carcinogenesis in albino mice. Int J Dermatol. 2007;46:1160-1165.
- Lambert LA, Wamer WG, Wei RR, et al. The protective but nonsynergistic effect of dietary beta-carotene and vitamin E on skin tumorigenesis in Skh mice. Nutr Cancer. 1994;21:1-12.
- Greenberg ER, Baron JA, Stukel TA, et al. A clinical trial of beta carotene to prevent basal-cell and squamous-cell cancers of the skin. The Skin Cancer Prevention Study Group. N Engl J Med. 1990;323:789-795.
- Frieling UM, Schaumberg DA, Kupper TS, et al. A randomized, 12-year primary-prevention trial of beta carotene supplementation for nonmelanoma skin cancer in the physician’s health study. Arch Dermatol. 2000;136:179-184.
- Naldi L, Gallus S, Tavani A, et al; Oncology Study Group of the Italian Group for Epidemiologic Research in Dermatology. Risk of melanoma and vitamin A, coffee and alcohol: a case-control study from Italy. Eur J Cancer Prev. 2004;13:503-508.
- Zhang YP, Chu RX, Liu H. Vitamin A intake and risk of melanoma: a meta-analysis. PloS One. 2014;9:e102527.
- Feskanich D, Willett WC, Hunter DJ, et al. Dietary intakes of vitamins A, C, and E and risk of melanoma in two cohorts of women. Br J Cancer. 2003;88:1381-1387.
- Bavinck JN, Tieben LM, Van der Woude FJ, et al. Prevention of skin cancer and reduction of keratotic skin lesions during acitretin therapy in renal transplant recipients: a double-blind, placebo-controlled study. J Clin Oncol. 1995;13:1933-1938.
- George R, Weightman W, Russ GR, et al. Acitretin for chemoprevention of non-melanoma skin cancers in renal transplant recipients. Australas J Dermatol. 2002;43:269-273.
- Solomon-Cohen E, Reiss-Huss S, Hodak E, et al. Low-dose acitretin for secondary prevention of keratinocyte carcinomas in solid-organ transplant recipients. Dermatology. 2022;238:161-166.
- Otley CC, Stasko T, Tope WD, et al. Chemoprevention of nonmelanoma skin cancer with systemic retinoids: practical dosing and management of adverse effects. Dermatol Surg. 2006;32:562-568.
- Kadakia KC, Barton DL, Loprinzi CL, et al. Randomized controlled trial of acitretin versus placebo in patients at high-risk for basal cell or squamous cell carcinoma of the skin (North Central Cancer Treatment Group Study 969251). Cancer. 2012;118:2128-2137.
- McKenna DB, Murphy GM. Skin cancer chemoprophylaxis in renal transplant recipients: 5 years of experience using low-dose acitretin. Br J Dermatol. 1999;140:656-660.
- National Institutes of Health Office of Dietary Supplements. Niacin: fact sheet for health professionals. Updated August 23, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/Niacin-HealthProfessional/
- Malesu R, Martin AJ, Lyons JG, et al. Nicotinamide for skin cancer chemoprevention: effects of nicotinamide on melanoma in vitro and in vivo. Photochem Photobiol Sci. 2020;19:171-179.
- Gensler HL. Prevention of photoimmunosuppression and photocarcinogenesis by topical nicotinamide. Nutr Cancer. 1997;29:157-162.
- Gensler HL, Williams T, Huang AC, et al. Oral niacin prevents photocarcinogenesis and photoimmunosuppression in mice. Nutr Cancer. 1999;34:36-41.
- Chen AC, Martin AJ, Choy B, et al. A phase 3 randomized trial of nicotinamide for skin-cancer chemoprevention. N Engl J Med. 2015;373:1618-1626.
- Drago F, Ciccarese G, Cogorno L, et al. Prevention of non-melanoma skin cancers with nicotinamide in transplant recipients: a case-control study. Eur J Dermatol. 2017;27:382-385.
- Yélamos O, Halpern AC, Weinstock MA. Reply to “A phase II randomized controlled trial of nicotinamide for skin cancer chemoprevention in renal transplant recipients.” Br J Dermatol. 2017;176:551-552.
- Scatozza F, Moschella F, D’Arcangelo D, et al. Nicotinamide inhibits melanoma in vitro and in vivo. J Exp Clin Cancer Res. 2020;39:211.
- National Institutes of Health Office of Dietary Supplements. Folate: fact sheet for health professionals. Updated November 1, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/Folate-HealthProfessional/
- Butzbach K, Epe B. Photogenotoxicity of folic acid. Free Radic Biol Med. 2013;65:821-827.
- Vollset SE, Clarke R, Lewington S, et al. Effects of folic acid supplementation on overall and site-specific cancer incidence during the randomised trials: meta-analyses of data on 50,000 individuals. Lancet. 2013;381:1029-1036.
- Donnenfeld M, Deschasaux M, Latino-Martel P, et al. Prospective association between dietary folate intake and skin cancer risk: results from the Supplémentation en Vitamines et Minéraux Antioxydants cohort. Am J Clin Nutr. 2015;102:471-478.
- Fung TT, Hunter DJ, Spiegelman D, et al. Vitamins and carotenoids intake and the risk of basal cell carcinoma of the skin in women (United States). Cancer Causes Control. 2002;13:221-230.
- Fung TT, Spiegelman D, Egan KM, et al. Vitamin and carotenoid intake and risk of squamous cell carcinoma of the skin. Int J Cancer. 2003;103:110-115.
- National Institutes of Health Office of Dietary Supplements. Vitamin C: fact sheet for health professionals. Updated March 26, 2021. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminC-HealthProfessional/
- Spoelstra-de Man AME, Elbers PWG, Oudemans-Van Straaten HM. Vitamin C: should we supplement? Curr Opin Crit Care. 2018;24:248-255.
- Moison RMW, Beijersbergen van Henegouwen GMJ. Topical antioxidant vitamins C and E prevent UVB-radiation-induced peroxidation of eicosapentaenoic acid in pig skin. Radiat Res. 2002;157:402-409.
- Lin JY, Selim MA, Shea CR, et al. UV photoprotection by combination topical antioxidants vitamin C and vitamin E. J Am Acad Dermatol. 2003;48:866-874.
- Pauling L, Willoughby R, Reynolds R, et al. Incidence of squamous cell carcinoma in hairless mice irradiated with ultraviolet light in relation to intake of ascorbic acid (vitamin C) and of D, L-alpha-tocopheryl acetate (vitamin E). Int J Vitam Nutr Res Suppl. 1982;23:53-82.
- Kune GA, Bannerman S, Field B, et al. Diet, alcohol, smoking, serum beta-carotene, and vitamin A in male nonmelanocytic skin cancer patients and controls. Nutr Cancer. 1992;18:237-244.
- Vural P, Canbaz M, Selçuki D. Plasma antioxidant defense in actinic keratosis and basal cell carcinoma. J Eur Acad Dermatol Venereol. 1999;13:96-101.
- Record IR, Dreosti IE, McInerney JK. Changes in plasma antioxidant status following consumption of diets high or low in fruit and vegetables or following dietary supplementation with an antioxidant mixture. Br J Nutr. 2001;85:459-464.
- Heinen MM, Hughes MC, Ibiebele TI, et al. Intake of antioxidant nutrients and the risk of skin cancer. Eur J Cancer. 2007;43:2707-2716.
- Yang G, Yan Y, Ma Y, et al. Vitamin C at high concentrations induces cytotoxicity in malignant melanoma but promotes tumor growth at low concentrations. Mol Carcinog. 2017;56:1965-1976.
- National Institutes of Health Office of Dietary Supplements. Vitamin D: fact sheet for health professionals. Updated August 12, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/
- Reichrath J, Saternus R, Vogt T. Endocrine actions of vitamin D in skin: relevance for photocarcinogenesis of non-melanoma skin cancer, and beyond. Mol Cell Endocrinol. 2017;453:96-102.
- Ellison TI, Smith MK, Gilliam AC, et al. Inactivation of the vitamin D receptor enhances susceptibility of murine skin to UV-induced tumorigenesis. J Invest Dermatol. 2008;128:2508-2517.
- Eide MJ, Johnson DA, Jacobsen GR, et al. Vitamin D and nonmelanoma skin cancer in a health maintenance organization cohort. Arch Dermatol. 2011;147:1379-1384.
- van der Pols JC, Russell A, Bauer U, et al. Vitamin D status and skin cancer risk independent of time outdoors: 11-year prospective study in an Australian community. J Invest Dermatol. 2013;133:637-641.
- Caini S, Gnagnarella P, Stanganelli I, et al. Vitamin D and the risk of non-melanoma skin cancer: a systematic literature review and meta-analysis on behalf of the Italian Melanoma Intergroup. Cancers (Basel). 2021;13:4815.
- Park SM, Li T, Wu S, et al. Vitamin D intake and risk of skin cancer in US women and men. PLoS One. 2016;11:e0160308.
- Afzal S, Nordestgaard BG, Bojesen SE. Plasma 25-hydroxyvitamin D and risk of non-melanoma and melanoma skin cancer: a prospective cohort study. J Invest Dermatol. 2013;133:629-636.
- Asgari MM, Tang J, Warton ME, et al. Association of prediagnostic serum vitamin D levels with the development of basal cell carcinoma. J Invest Dermatol. 2010;130:1438-1443.
- Tang JY, Parimi N, Wu A, et al. Inverse association between serum 25(OH) vitamin D levels and non-melanoma skin cancer in elderly men. Cancer Causes Control. 2010;21:387-391.
- Keen MA, Hassan I. Vitamin E in dermatology. Indian Dermatol Online J. 2016;7:311-315.
- National Institutes of Health Office of Dietary Supplements. Vitamin E: fact sheet for health professionals. Updated March 26, 2021. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional/
- Pearson P, Lewis SA, Britton J, et al. The pro-oxidant activity of high-dose vitamin E supplements in vivo. BioDrugs. 2006;20:271-273.
- Gerrish KE, Gensler HL. Prevention of photocarcinogenesis by dietary vitamin E. Nutr Cancer. 1993;19:125-133.
- McVean M, Liebler DC. Prevention of DNA photodamage by vitamin E compounds and sunscreens: roles of ultraviolet absorbance and cellular uptake. Mol Carcinog. 1999;24:169-176.
- Prasad KN, Cohrs RJ, Sharma OK. Decreased expressions of c-myc and H-ras oncogenes in vitamin E succinate induced morphologically differentiated murine B-16 melanoma cells in culture. Biochem Cell Biol. 1990;68:1250-1255.
- Funasaka Y, Komoto M, Ichihashi M. Depigmenting effect of alpha-tocopheryl ferulate on normal human melanocytes. Pigment Cell Res. 2000;13(suppl 8):170-174.
- National Institutes of Health Office of Dietary Supplements. Selenium: fact sheet for health professionals. Updated March 26, 2021. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/Selenium-HealthProfessional/
- Sengupta A, Lichti UF, Carlson BA, et al. Selenoproteins are essential for proper keratinocyte function and skin development. PLoS One. 2010;5:e12249.
- Das RK, Hossain SKU, Bhattacharya S. Diphenylmethyl selenocyanate inhibits DMBA-croton oil induced two-stage mouse skin carcinogenesis by inducing apoptosis and inhibiting cutaneous cell proliferation. Cancer Lett. 2005;230:90-101.
- Clark LC, Combs GF Jr, Turnbull BW, et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA. 1996;276:1957-1963.
- Duffield-Lillico AJ, Slate EH, Reid ME, et al. Selenium supplementation and secondary prevention of nonmelanoma skin cancer in a randomized trial. J Natl Cancer Inst. 2003;95:1477-1481.
- Reid ME, Duffield-Lillico AJ, Slate E, et al. The nutritional prevention of cancer: 400 mcg per day selenium treatment. Nutr Cancer. 2008;60:155-163.
- Vinceti M, Filippini T, Del Giovane C, et al. Selenium for preventing cancer. Cochrane Database Syst Rev. 2018;1:CD005195.
- Hercberg S, Ezzedine K, Guinot C, et al. Antioxidant supplementation increases the risk of skin cancers in women but not in men. J Nutr. 2007;137:2098-2105.
- Chang YJ, Myung SK, Chung ST, et al. Effects of vitamin treatment or supplements with purported antioxidant properties on skin cancer prevention: a meta-analysis of randomized controlled trials. Dermatology. 2011;223:36-44.
- Sun W, Rice MS, Park MK, et al. Intake of furocoumarins and risk of skin cancer in 2 prospective US cohort studies. J Nutr. 2020;150:1535-1544.
- Sakaki JR, Melough MM, Roberts MB, et al. Citrus consumption and the risk of non-melanoma skin cancer in the Women’s Health Initiative. Cancers (Basel). 2021;13:2173.
Nonmelanoma skin cancer (NMSC) is the most common cancer in the United States, and cutaneous melanoma is projected to be the fifth most common form of cancer in 2022, with increasing incidence and high potential for mortality.1-3 Estimates indicate that 35% to 45% of all cancers in White patients are cutaneous, with 4% to 5% occurring in Hispanic patients, 2% to 4% in Asian patients, and 1% to 2% in Black patients.4 Of the keratinocyte carcinomas, basal cell carcinoma (BCC) is the most prevalent, projected to affect approximately 33% to 39% of White males and 23% to 28% of White females in the United States during their lifetimes. Squamous cell carcinoma (SCC) is the second most common skin malignancy, with a lifetime risk of 9% to 14% for White males and 4% to 9% for White females in the United States.5 The incidence of melanoma continues to increase, with approximately 99,780 new cases expected in the United States in 2022.1
UV-induced DNA damage plays a key role in the pathogenesis and development of various skin malignancies.6 UV radiation from sunlight or tanning devices causes photocarcinogenesis due to molecular and cellular effects, including the generation of reactive oxygen species, DNA damage due to the formation of cyclobutane pyrimidine dimers and pyrimidine-pyrimidone, melanogenesis, apoptosis, and the increased expression of harmful genes and proteins.6 The summation of this damage can result in skin malignancies, including NMSC and melanoma.6,7 Dietary antioxidants theoretically help prevent oxidative reactions from occurring within the body, and it has been suggested that intake of dietary antioxidants may decrease DNA damage and prevent tumorigenesis secondary to UV radiation.8 Antioxidants exist naturally in the body but can be acquired exogenously. Investigators have studied dietary antioxidants in preventing skin cancer formation with promising results in the laboratory setting.8-11 Recently, more robust human studies have been initiated to further delineate this relationship. We present clinical evidence of several frequently utilized antioxidant vitamins and their effects on melanoma and NMSC.
Antioxidants
Vitamin A—Vitamin A is a fat-soluble vitamin found in animal sources, including fish, liver, and eggs. Carotenoids, such as beta carotene, are provitamin A plant derivatives found in fruits and vegetables that are converted into biologically active retinol and retinoic acid.12 Retinols play a key role in cellular growth and differentiation and are thought to be protective against skin cancer via the inactivation of free radicals and immunologic enhancement due to their antiproliferative, antioxidative, and antiapoptotic effects.13-16 Animal studies have demonstrated this protective effect and the ability of retinoids to suppress carcinogenesis; however, human studies reveal conflicting results.17,18
Greenberg et al19 investigated the use of beta carotene in preventing the formation of NMSC. Patients (N=1805) were randomized to receive 50 mg of beta carotene daily or placebo. Over a 5-year period, there was no significant reduction in the occurrence of NMSC (relative risk [RR], 1.05; 95% CI, 0.91-1.22).19 Frieling et al20 conducted a similar randomized, double-blind, placebo-controlled trial investigating beta carotene for primary prevention of NMSC in 22,071 healthy male physicians. The study group received 50 mg of beta carotene every other day for 12 years’ duration, and there was no significant effect on the incidence of first NMSC development (RR, 0.98; 95% CI, 0.92-1.05).20
A case-control study by Naldi et al21 found an inverse association between vitamin A intake and development of melanoma. Study participants were stratified into quartiles based on level of dietary intake and found an odds ratio (OR) of 0.71 for beta carotene (95% CI, 0.50-1.02), 0.57 for retinol (95% CI, 0.39-0.83), and 0.51 for total vitamin A (95% CI, 0.35-0.75) when comparing the upper quartile of vitamin A intake to the lower quartile. Upper-quartile cutoff values of vitamin A intake were 214 µg/d for beta carotene, 149 µg/d for retinol, and 359 µg/d for total vitamin A.21 More recently, a meta-analysis by Zhang et al22 pooled data from 8 case-control studies and 2 prospective studies. Intake of retinol but not total vitamin A or beta carotene was associated with a reduced risk for development of melanoma (retinol: OR, 0.80; 95% CI, 0.69-0.92; total vitamin A: OR, 0.86; 95% CI, 0.59-1.25; beta carotene: OR, 0.87; 95% CI, 0.62-1.20).22 Feskanich et al23 demonstrated similar findings with use of food-frequency questionnaires in White women, suggesting that retinol intake from food combined with supplements may be protective for women who were otherwise at a low risk for melanoma based on nondietary factors. These factors included painful or blistering sunburns during childhood, history of more than 6 sunburns, more than 3 moles on the left arm, having red or blonde hair, and having a parent or sibling with melanoma (P=.01). However, this relationship did not hold true when looking at women at an intermediate or high risk for melanoma (P=.16 and P=.46).23
When looking at high-risk patients, such as transplant patients, oral retinoids have been beneficial in preventing NMSC.24-27 Bavinck et al24 investigated 44 renal transplant patients with a history of more than 10 NMSCs treated with 30 mg of acitretin daily vs placebo. Patients receiving oral retinoid supplementation developed fewer NMSCs over a 6-month treatment period (P=.01).24 Similarly, George et al25 investigated acitretin in renal transplant patients and found a statistically significant decrease in number of SCCs in patients on supplementation (P=.002). Solomon-Cohen et al26 performed a retrospective case-crossover study in solid organ transplant recipients and found that those treated with 10 mg of acitretin daily for 2 years had a significant reduction in the number of new keratinocyte carcinomas (P=.002). Other investigators have demonstrated similar results, and in 2006, Otley et al27 proposed standardized dosing of acitretin for chemoprevention in high-risk patients, including patients developing 5 to 10 NMSCs per year, solid organ transplant recipients, and those with syndromes associated with the development of NMSC.28,29 Overall, in the general population, vitamin A and related compounds have not demonstrated a significant association with decreased development of NMSC; however, oral retinoids have proven useful for high-risk patients. Furthermore, several studies have suggested a negative association between vitamin A levels and the incidence of melanoma, specifically in the retinol formulation.
Vitamin B3—Nicotinamide (also known as niacinamide) is a water-soluble form of vitamin B3 and is obtained from animal-based and plant-based foods, such as meat, fish, and legumes.30 Nicotinamide plays a key role in cellular metabolism, cellular signaling, and DNA repair, including protection from UV damage within keratinocytes.31,32 Early mouse models demonstrated decreased formation of skin tumors in mice treated with topical or oral nicotinamide.32,33 A number of human studies have revealed similar results.34-36
Chen et al34 conducted the ONTRAC study, a phase 3, double-blind, randomized controlled trial (RCT) looking at 386 participants with a history of at least 2 NMSCs in the preceding 5 years. At 12 months, those treated with 500 mg of nicotinamide twice daily demonstrated a statistically significant decreased rate of SCC formation (P=.05). A decreased incidence of BCC development was noted; however, this trend did not reach statistical significance (P=.12). Precancerous skin lesions also were found to be decreased in the treatment group, with 20% lower incidence of actinic keratoses (AKs) after 9 months of treatment (P<.001).34 Drago et al35 specifically studied the incidence of AKs in 38 transplant recipients—8 liver and 30 kidney—and found that previously noted AKs had decreased in size for 18 of 19 patients taking 500 mg of nicotinamide daily when originally photographed AKs were remeasured at 6-month follow-up, with 7 of these 18 patients demonstrating complete clinical regression. Of those on nicotinamide supplementation, no new AKs developed compared to the control group, which demonstrated increased size of AKs or development of new AKs in 91% of patients, with 7 AKs progressing into SCC.35
Nicotinamide has been demonstrated to be useful in preventing skin cancer in high-risk populations, such as transplant patients or those with a high incidence of NMSC.34,36 Despite promising results within the laboratory setting, nicotinamide’s effects on melanoma in humans remains less clear.31,37 Studies suggest that nicotinamide enhances tumor-infiltrating lymphocytes and DNA repair mechanisms in melanocytes, which may translate into nicotinamide, providing chemoprevention for melanoma, but research in human patients is limited.31,37
Vitamin B9—Folate, the natural form of vitamin B9, is a water-soluble compound that is found in many foods, especially green leafy vegetables, and often is supplemented because of its health benefits.38,39 In the skin, folic acid plays a key role in cellular replication and proliferation.38 Controversy exists regarding folate’s effects on cellular growth and turnover with respect to cancer incidence.38,40 Donnenfeld et al41 conducted a prospective study assessing dietary folic acid intake and development of NMSC. A total of 5880 participants completed dietary records throughout the first 2 years of the study. After an average follow-up period of 12.6 years, there was an overall increased incidence of skin cancer in those with increased dietary folate (P=.03). Furthermore, when striating by skin cancer type, there was an increased incidence of NMSC overall as well as BCC when analyzing by type of NMSC (P=.03 for NMSC; P=.05 for BCC). However, when stratifying by gender, these findings only held true for women.41 Similar effects were observed by Fung et al,42 who prospectively studied the intake of various vitamins in relationship to the development of BCC in women. During 12 years of follow-up, a positive association was observed between folate intake and BCC development (OR, 1.2; 95% CI, 1.10-1.31).42 Fung et al43 also investigated the role of several vitamins in the development of SCC and found that folate showed a negative association, which did not reach statistical significance (RR, 0.79; 95% CI, 0.56-1.11). Furthermore, Vollset et al40 conducted a meta-analysis comparing folic acid to placebo in the incidence of various types of cancer. The study excluded NMSC but reported no significant association between the development of melanoma and folic acid supplementation.40 In summary, the effects of folate have diverse consequences, potentially promoting the formation of NMSC, but studies suggest that an individual’s gender and other genetic and environmental factors also may play a role.
Vitamin C—Vitamin C (also known as ascorbic acid) is a water-soluble vitamin with antioxidant immune-mediating effects. It is found in various fruits and vegetables and serves as a cofactor for enzymes within the body playing a key role in immune function and collagen formation.44,45 It has been postulated that ascorbic acid can provide protection from UV radiation damage via its intracellular activity but conversely can contribute to oxidative damage.44 Multiple in vitro laboratory studies and animal models have demonstrated photoprotective effects of ascorbic acid.46-48 Despite these findings, minimal photoprotective effects have been found in the human population.
Kune et al49 performed a case-control study of 88 males with previously diagnosed NMSC undergoing surgical removal and investigated patients’ prior dietary habits. Patients with NMSC had a statistically significantly lower level of vitamin C–containing food in their diet than those without NMSC (P=.004).49 In addition, Vural et al50 analyzed plasma samples and blood cells of patients with AK and BCC and found a significant decrease in ascorbic acid levels in both the AK (P<.001) and BCC (P<.001) groups compared with controls. However, studies have found that consumption of certain dietary compounds can rapidly increase plasma concentration levels, which may serve as a major confounding variable in this study. Plasma concentrations of ascorbic acid and beta carotene were found to be significantly increased following consumption of a high-antioxidant diet for as short a duration as 2 weeks (P<.05).51 More recently, Heinen et al52 performed a prospective study on 1001 adults. In patients without a history of skin cancer, they found that vitamin C from food sources plus dietary supplements was positively associated with the development of BCC (P=.03).52 Similarly, Fung et al42 performed a study in women and found a positive association between vitamin C intake and the development of BCC (OR, 1.13; 95% CI, 1.03-1.23).
The relationship between vitamin C intake—either in dietary or supplemental form—and melanoma remains controversial. Mice-based studies found that high concentrations of orally administered vitamin C induce cytotoxicity in melanoma cell lines, but at low concentrations they promote tumor growth of malignant melanoma.53 Feskanich et al23 examined the relationship between vitamin C intake and melanoma development via food frequency questionnaires in White women and found that vitamin C was associated with a higher risk for melanoma (P=.05), and furthermore, a positive dose response with frequency of orange juice intake was observed (P=.008). Overall, despite promising laboratory studies, there is a lack of RCTs investigating the use of vitamin C supplementation for prevention of NMSC and melanoma in humans, and the oral benefits of vitamin C for chemoprevention remain unclear.
Vitamin D—Vitamin D is a fat-soluble vitamin that is found in fish, liver, egg, and cheese, and is endogenously produced when UV radiation from sun exposure interacts with the skin, triggering the synthesis of vitamin D.54 Vitamin D is biologically inactive and must be converted to its active form 1,25-dihydroxyvitamin D after entering the body. Vitamin D modulates many genes involved in cellular proliferation and differentiation.54 Vitamin D receptors are expressed on keratinocytes and melanocytes.55 Animal studies have demonstrated a potentially protective effect of vitamin D in the development of NMSC.56 In a mouse model, Ellison et al56 found that mice without vitamin D receptors developed skin tumors more rapidly than those with vitamin D receptors.
Unfortunately, these findings have not been demonstrated in humans, and studies have even reported an increased risk for development of NMSC in patients with normal or increased vitamin D levels compared with those with low levels of vitamin D.57-60 Eide et al57 studied 3223 patients seeking advice for low bone density by recording their vitamin D levels at the time of presentation and monitoring development of NMSC. Vitamin D levels greater than 15 ng/mL were positively associated with the development of NMSC (OR, 1.7; 95% CI, 1.04-2.7). This association held true for both SCC and BCC, with a higher risk estimated for SCC (OR, 3.2; 95% CI, 0.4-24.0 for SCC; OR, 1.7; 95% CI, 0.5-5.8 for BCC).57 An increased vitamin D serum level also was found to be significantly associated with a higher risk for BCC and melanoma by van der Pols et al.58 This prospective study looked at the incidence of skin cancer over 11 years. Study participants with vitamin D levels over 75 nmol/L more frequently developed BCC (P=.01) and melanoma (P=.05). In contrast, SCC was less frequently observed in participants with these high levels of vitamin D (P=.07).58 Furthermore, Park et al60 looked at vitamin D and skin cancer risk for men and women in the United States and found no association with risk for SCC or melanoma but a positive association with BCC (P=.05 for total vitamin D; P<.01 for dietary vitamin D). Additional studies have been performed with inconsistent results, and multiple authors suggest the possible confounding relationship between vitamin D levels and UV radiation exposure.59-62 Furthermore, some studies have even demonstrated a negative association between vitamin D and NMSC. Tang et al63 performed a retrospective case-control study in elderly males, investigating serum levels of vitamin D and patients’ self-reported history of NMSC, which demonstrated that higher levels of vitamin D were associated with a decreased risk for NMSC. Overall, the relationship between vitamin D and skin cancer development remains unclear for both melanoma and NMSC.
Vitamin E—Vitamin E is a fat-soluble vitamin that is found in plant-based oils, nuts, seeds, fruits, and vegetables.64 It works as an antioxidant to protect against free radicals and heighten immune function, and it also serves as a pro-oxidant.65,66 Vitamin E naturally exists in 8 chemical forms, of which gamma-tocopherol is the most frequently obtained form in the diet, and alpha-tocopherol is the most abundant form found in the body.64,65
Early animal studies demonstrated the inhibition of UV-induced damage in mice receiving vitamin E supplementation.67,68 Human studies have not consistently shown these effects. Vural et al50 investigated plasma samples and blood cells of patients with AKs and BCCs and reported a significant decrease in alpha-tocopherol levels in both the AK (P<.05) and BCC (P<.001) groups compared with controls. However, studies also have demonstrated a positive association between vitamin E intake and the development of BCC, including one by Fung et al,42 which found a significant association in women (OR, 1.15; 95% CI, 1.06-1.26).
Vitamin E has been found to inhibit melanin synthesis in the laboratory, suggesting a potentially protective effect in melanoma.69,70 However, in the study performed by Feskanich et al23 examining vitamin intake and melanoma incidence via food-frequency questionnaires, vitamin E was not associated with a lower risk for melanoma. Despite promising laboratory studies, the data surrounding the use of a vitamin E supplement for prevention of melanoma and NMSC in humans remains unclear.
Selenium—Selenium is a trace mineral found in plants, meat, and fish. It plays a key role in reproduction, hormone metabolism, DNA synthesis, and protection from oxidative damage.71 In mice studies, lack of selenium-containing proteins resulted in skin abnormalities, including the development of a hyperplastic epidermis and aberrant hair follicle morphogenesis with alopecia after birth, and numerous experimental studies have demonstrated a negative association between selenium intake and cancer.72,73 However, human studies have yielded alternative results.
The Nutritional Prevention of Cancer Study Group analyzed 1312 dermatology patients with a history of NMSC.74 The study population was obtained from 7 dermatology clinics with randomization to control for confounding variables. Study participants received either 200 μg of selenium daily or placebo.74 Baseline characteristics of each study group were overall balanced. Selenium intake was found to have no effect on the development of BCC (hazard ratio [HR], 1.09; 95% CI, 0.94-1.26) but an increased risk for developing SCC (HR, 1.25; 95% CI, 1.03-1.51) and total NMSC (HR, 1.17; 95% CI, 1.02-1.34).74,75 Similarly, Reid et al76 performed an RCT comparing patients treated with 400 μg/d of selenium to those treated with 200 μg/d of selenium. When compared with placebo, those treated with 200 μg/d of selenium had a statistically significantly increased incidence of NMSC (P=.006); however, those treated with 400 μg/d of selenium had no significant change in total incidence of NMSC (P=.51).76 Furthermore, Vinceti et al77 performed a review of 83 studies from the literature investigating the effect of dietary selenium, and from the RCTs, there was no beneficial effect of selenium in reducing cancer risk in general; however, some studies demonstrated an increased incidence of other types of cancer, including melanoma. Of the RCTs included in the study investigating NMSC incidence specifically, it was found that the incidence was not affected by selenium administration (RR, 1.16; 95% CI, 0.30-4.42; 2 studies, 2027 participants).77 Despite data from several studies demonstrating an increased risk for NMSC, the effects of selenium on the risk for NMSC and melanoma remain unclear.
Combination Antioxidant Studies
In addition to investigating the use of single antioxidants in skin cancer prevention, studies utilizing the combination of various antioxidants or other dietary minerals have been conducted. Hercberg et al78 performed a randomized, double-blinded, placebo-controlled trial of 13,017 adults (7876 women and 5141 men) receiving a combination of 120 mg vitamin C, 30 mg vitamin E, 100 μg selenium, 6 mg beta carotene, and 20 mg zinc. Study participants were followed for an average of 7.5 years, and the development of skin cancers were recorded. Overall, the incidence rate of skin cancer did not differ between the 2 treatment groups; however, when segregated by gender, the study found that there was an increased risk for developing skin cancer in women taking the antioxidant supplement combination compared with placebo (P=.03). This difference was not observed in the 2 treatment groups of male patients (P=.11). When looking specifically at NMSC, there was no difference between treatment groups for male or female patients (P=.39 for males; P=.15 for females). In contrast, there was a higher incidence of melanoma identified in female patients taking the combination antioxidant supplement (P=.01), but this was not seen within the male study population (P=.51).78 In addition, Chang et al79 performed a meta-analysis of 10 previously published RCTs. Analysis revealed that treatment with a variety of supplements, including vitamins A, C, E, and beta carotene, were found to have no preventative effects on the incidence of skin cancer development (RR, 0.98; CI, 0.98-1.03). Notable limitations to this study included the variability in protocols of the studies included in this meta-analysis, the limited number of RCTs investigating vitamin supplementation and the risk for skin cancer development, and the influence of dietary intake on study outcomes.79
Other Dietary Agents
Furocoumarins—Furocoumarins are botanical substances found in various fruits and plants, including many citrus products. Furocoumarins are activated by UV light radiation and can lead to development of a phototoxic eruption. Several studies have suggested a pharmacogenetic effect of furocoumarins.80 Sun et al80 collected dietary data from 47,453 men and 75,291 women on furocoumarin intake and correlation with the development of NMSC. Overall, the study suggested that the intake of furocoumarins may lead to an increase in the development of BCC (HR, 1.16; 95% CI, 1.11-1.21; P=.002); however, there was no significant association identified between total intake of furocoumarins in the risk for SCC or melanoma.80 Furthermore, Sakaki et al81 conducted a survey study looking at the consumption of citrus products and the development of NMSC. The group found that there was an increased risk for NMSC in those consuming an increased amount of citrus products (P=.007).81
Conclusion
Dietary antioxidants have been investigated for their potential role in the prevention of tumorigenesis. Specific antioxidant vitamins, such as vitamin A derivatives and niacinamide, have demonstrated clinical utility in the prevention of NMSC in high-risk populations. Retinol also has been associated with a reduced incidence of melanoma. Numerous antioxidants have demonstrated promising data within the laboratory setting; however, inconsistent results have been appreciated in humans. Furthermore, several research studies suggest that folate, vitamin D, and furocoumarins may be associated with an increased risk for skin cancer development; however, these studies are inconclusive, and dietary studies are challenging to conduct. Overall, RCTs investigating the role of antioxidants for chemoprevention are limited. Moreover, the study of dietary antioxidants and vitamins may be affected by various confounding variables that can be difficult to account for because of patients’ potentially poor recall of dietary intake and the effect of dietary intake in supplemental studies. Given the increasing prevalence of skin cancer worldwide, further research into the clinical utility of antioxidants in skin cancer prevention is warranted.
Nonmelanoma skin cancer (NMSC) is the most common cancer in the United States, and cutaneous melanoma is projected to be the fifth most common form of cancer in 2022, with increasing incidence and high potential for mortality.1-3 Estimates indicate that 35% to 45% of all cancers in White patients are cutaneous, with 4% to 5% occurring in Hispanic patients, 2% to 4% in Asian patients, and 1% to 2% in Black patients.4 Of the keratinocyte carcinomas, basal cell carcinoma (BCC) is the most prevalent, projected to affect approximately 33% to 39% of White males and 23% to 28% of White females in the United States during their lifetimes. Squamous cell carcinoma (SCC) is the second most common skin malignancy, with a lifetime risk of 9% to 14% for White males and 4% to 9% for White females in the United States.5 The incidence of melanoma continues to increase, with approximately 99,780 new cases expected in the United States in 2022.1
UV-induced DNA damage plays a key role in the pathogenesis and development of various skin malignancies.6 UV radiation from sunlight or tanning devices causes photocarcinogenesis due to molecular and cellular effects, including the generation of reactive oxygen species, DNA damage due to the formation of cyclobutane pyrimidine dimers and pyrimidine-pyrimidone, melanogenesis, apoptosis, and the increased expression of harmful genes and proteins.6 The summation of this damage can result in skin malignancies, including NMSC and melanoma.6,7 Dietary antioxidants theoretically help prevent oxidative reactions from occurring within the body, and it has been suggested that intake of dietary antioxidants may decrease DNA damage and prevent tumorigenesis secondary to UV radiation.8 Antioxidants exist naturally in the body but can be acquired exogenously. Investigators have studied dietary antioxidants in preventing skin cancer formation with promising results in the laboratory setting.8-11 Recently, more robust human studies have been initiated to further delineate this relationship. We present clinical evidence of several frequently utilized antioxidant vitamins and their effects on melanoma and NMSC.
Antioxidants
Vitamin A—Vitamin A is a fat-soluble vitamin found in animal sources, including fish, liver, and eggs. Carotenoids, such as beta carotene, are provitamin A plant derivatives found in fruits and vegetables that are converted into biologically active retinol and retinoic acid.12 Retinols play a key role in cellular growth and differentiation and are thought to be protective against skin cancer via the inactivation of free radicals and immunologic enhancement due to their antiproliferative, antioxidative, and antiapoptotic effects.13-16 Animal studies have demonstrated this protective effect and the ability of retinoids to suppress carcinogenesis; however, human studies reveal conflicting results.17,18
Greenberg et al19 investigated the use of beta carotene in preventing the formation of NMSC. Patients (N=1805) were randomized to receive 50 mg of beta carotene daily or placebo. Over a 5-year period, there was no significant reduction in the occurrence of NMSC (relative risk [RR], 1.05; 95% CI, 0.91-1.22).19 Frieling et al20 conducted a similar randomized, double-blind, placebo-controlled trial investigating beta carotene for primary prevention of NMSC in 22,071 healthy male physicians. The study group received 50 mg of beta carotene every other day for 12 years’ duration, and there was no significant effect on the incidence of first NMSC development (RR, 0.98; 95% CI, 0.92-1.05).20
A case-control study by Naldi et al21 found an inverse association between vitamin A intake and development of melanoma. Study participants were stratified into quartiles based on level of dietary intake and found an odds ratio (OR) of 0.71 for beta carotene (95% CI, 0.50-1.02), 0.57 for retinol (95% CI, 0.39-0.83), and 0.51 for total vitamin A (95% CI, 0.35-0.75) when comparing the upper quartile of vitamin A intake to the lower quartile. Upper-quartile cutoff values of vitamin A intake were 214 µg/d for beta carotene, 149 µg/d for retinol, and 359 µg/d for total vitamin A.21 More recently, a meta-analysis by Zhang et al22 pooled data from 8 case-control studies and 2 prospective studies. Intake of retinol but not total vitamin A or beta carotene was associated with a reduced risk for development of melanoma (retinol: OR, 0.80; 95% CI, 0.69-0.92; total vitamin A: OR, 0.86; 95% CI, 0.59-1.25; beta carotene: OR, 0.87; 95% CI, 0.62-1.20).22 Feskanich et al23 demonstrated similar findings with use of food-frequency questionnaires in White women, suggesting that retinol intake from food combined with supplements may be protective for women who were otherwise at a low risk for melanoma based on nondietary factors. These factors included painful or blistering sunburns during childhood, history of more than 6 sunburns, more than 3 moles on the left arm, having red or blonde hair, and having a parent or sibling with melanoma (P=.01). However, this relationship did not hold true when looking at women at an intermediate or high risk for melanoma (P=.16 and P=.46).23
When looking at high-risk patients, such as transplant patients, oral retinoids have been beneficial in preventing NMSC.24-27 Bavinck et al24 investigated 44 renal transplant patients with a history of more than 10 NMSCs treated with 30 mg of acitretin daily vs placebo. Patients receiving oral retinoid supplementation developed fewer NMSCs over a 6-month treatment period (P=.01).24 Similarly, George et al25 investigated acitretin in renal transplant patients and found a statistically significant decrease in number of SCCs in patients on supplementation (P=.002). Solomon-Cohen et al26 performed a retrospective case-crossover study in solid organ transplant recipients and found that those treated with 10 mg of acitretin daily for 2 years had a significant reduction in the number of new keratinocyte carcinomas (P=.002). Other investigators have demonstrated similar results, and in 2006, Otley et al27 proposed standardized dosing of acitretin for chemoprevention in high-risk patients, including patients developing 5 to 10 NMSCs per year, solid organ transplant recipients, and those with syndromes associated with the development of NMSC.28,29 Overall, in the general population, vitamin A and related compounds have not demonstrated a significant association with decreased development of NMSC; however, oral retinoids have proven useful for high-risk patients. Furthermore, several studies have suggested a negative association between vitamin A levels and the incidence of melanoma, specifically in the retinol formulation.
Vitamin B3—Nicotinamide (also known as niacinamide) is a water-soluble form of vitamin B3 and is obtained from animal-based and plant-based foods, such as meat, fish, and legumes.30 Nicotinamide plays a key role in cellular metabolism, cellular signaling, and DNA repair, including protection from UV damage within keratinocytes.31,32 Early mouse models demonstrated decreased formation of skin tumors in mice treated with topical or oral nicotinamide.32,33 A number of human studies have revealed similar results.34-36
Chen et al34 conducted the ONTRAC study, a phase 3, double-blind, randomized controlled trial (RCT) looking at 386 participants with a history of at least 2 NMSCs in the preceding 5 years. At 12 months, those treated with 500 mg of nicotinamide twice daily demonstrated a statistically significant decreased rate of SCC formation (P=.05). A decreased incidence of BCC development was noted; however, this trend did not reach statistical significance (P=.12). Precancerous skin lesions also were found to be decreased in the treatment group, with 20% lower incidence of actinic keratoses (AKs) after 9 months of treatment (P<.001).34 Drago et al35 specifically studied the incidence of AKs in 38 transplant recipients—8 liver and 30 kidney—and found that previously noted AKs had decreased in size for 18 of 19 patients taking 500 mg of nicotinamide daily when originally photographed AKs were remeasured at 6-month follow-up, with 7 of these 18 patients demonstrating complete clinical regression. Of those on nicotinamide supplementation, no new AKs developed compared to the control group, which demonstrated increased size of AKs or development of new AKs in 91% of patients, with 7 AKs progressing into SCC.35
Nicotinamide has been demonstrated to be useful in preventing skin cancer in high-risk populations, such as transplant patients or those with a high incidence of NMSC.34,36 Despite promising results within the laboratory setting, nicotinamide’s effects on melanoma in humans remains less clear.31,37 Studies suggest that nicotinamide enhances tumor-infiltrating lymphocytes and DNA repair mechanisms in melanocytes, which may translate into nicotinamide, providing chemoprevention for melanoma, but research in human patients is limited.31,37
Vitamin B9—Folate, the natural form of vitamin B9, is a water-soluble compound that is found in many foods, especially green leafy vegetables, and often is supplemented because of its health benefits.38,39 In the skin, folic acid plays a key role in cellular replication and proliferation.38 Controversy exists regarding folate’s effects on cellular growth and turnover with respect to cancer incidence.38,40 Donnenfeld et al41 conducted a prospective study assessing dietary folic acid intake and development of NMSC. A total of 5880 participants completed dietary records throughout the first 2 years of the study. After an average follow-up period of 12.6 years, there was an overall increased incidence of skin cancer in those with increased dietary folate (P=.03). Furthermore, when striating by skin cancer type, there was an increased incidence of NMSC overall as well as BCC when analyzing by type of NMSC (P=.03 for NMSC; P=.05 for BCC). However, when stratifying by gender, these findings only held true for women.41 Similar effects were observed by Fung et al,42 who prospectively studied the intake of various vitamins in relationship to the development of BCC in women. During 12 years of follow-up, a positive association was observed between folate intake and BCC development (OR, 1.2; 95% CI, 1.10-1.31).42 Fung et al43 also investigated the role of several vitamins in the development of SCC and found that folate showed a negative association, which did not reach statistical significance (RR, 0.79; 95% CI, 0.56-1.11). Furthermore, Vollset et al40 conducted a meta-analysis comparing folic acid to placebo in the incidence of various types of cancer. The study excluded NMSC but reported no significant association between the development of melanoma and folic acid supplementation.40 In summary, the effects of folate have diverse consequences, potentially promoting the formation of NMSC, but studies suggest that an individual’s gender and other genetic and environmental factors also may play a role.
Vitamin C—Vitamin C (also known as ascorbic acid) is a water-soluble vitamin with antioxidant immune-mediating effects. It is found in various fruits and vegetables and serves as a cofactor for enzymes within the body playing a key role in immune function and collagen formation.44,45 It has been postulated that ascorbic acid can provide protection from UV radiation damage via its intracellular activity but conversely can contribute to oxidative damage.44 Multiple in vitro laboratory studies and animal models have demonstrated photoprotective effects of ascorbic acid.46-48 Despite these findings, minimal photoprotective effects have been found in the human population.
Kune et al49 performed a case-control study of 88 males with previously diagnosed NMSC undergoing surgical removal and investigated patients’ prior dietary habits. Patients with NMSC had a statistically significantly lower level of vitamin C–containing food in their diet than those without NMSC (P=.004).49 In addition, Vural et al50 analyzed plasma samples and blood cells of patients with AK and BCC and found a significant decrease in ascorbic acid levels in both the AK (P<.001) and BCC (P<.001) groups compared with controls. However, studies have found that consumption of certain dietary compounds can rapidly increase plasma concentration levels, which may serve as a major confounding variable in this study. Plasma concentrations of ascorbic acid and beta carotene were found to be significantly increased following consumption of a high-antioxidant diet for as short a duration as 2 weeks (P<.05).51 More recently, Heinen et al52 performed a prospective study on 1001 adults. In patients without a history of skin cancer, they found that vitamin C from food sources plus dietary supplements was positively associated with the development of BCC (P=.03).52 Similarly, Fung et al42 performed a study in women and found a positive association between vitamin C intake and the development of BCC (OR, 1.13; 95% CI, 1.03-1.23).
The relationship between vitamin C intake—either in dietary or supplemental form—and melanoma remains controversial. Mice-based studies found that high concentrations of orally administered vitamin C induce cytotoxicity in melanoma cell lines, but at low concentrations they promote tumor growth of malignant melanoma.53 Feskanich et al23 examined the relationship between vitamin C intake and melanoma development via food frequency questionnaires in White women and found that vitamin C was associated with a higher risk for melanoma (P=.05), and furthermore, a positive dose response with frequency of orange juice intake was observed (P=.008). Overall, despite promising laboratory studies, there is a lack of RCTs investigating the use of vitamin C supplementation for prevention of NMSC and melanoma in humans, and the oral benefits of vitamin C for chemoprevention remain unclear.
Vitamin D—Vitamin D is a fat-soluble vitamin that is found in fish, liver, egg, and cheese, and is endogenously produced when UV radiation from sun exposure interacts with the skin, triggering the synthesis of vitamin D.54 Vitamin D is biologically inactive and must be converted to its active form 1,25-dihydroxyvitamin D after entering the body. Vitamin D modulates many genes involved in cellular proliferation and differentiation.54 Vitamin D receptors are expressed on keratinocytes and melanocytes.55 Animal studies have demonstrated a potentially protective effect of vitamin D in the development of NMSC.56 In a mouse model, Ellison et al56 found that mice without vitamin D receptors developed skin tumors more rapidly than those with vitamin D receptors.
Unfortunately, these findings have not been demonstrated in humans, and studies have even reported an increased risk for development of NMSC in patients with normal or increased vitamin D levels compared with those with low levels of vitamin D.57-60 Eide et al57 studied 3223 patients seeking advice for low bone density by recording their vitamin D levels at the time of presentation and monitoring development of NMSC. Vitamin D levels greater than 15 ng/mL were positively associated with the development of NMSC (OR, 1.7; 95% CI, 1.04-2.7). This association held true for both SCC and BCC, with a higher risk estimated for SCC (OR, 3.2; 95% CI, 0.4-24.0 for SCC; OR, 1.7; 95% CI, 0.5-5.8 for BCC).57 An increased vitamin D serum level also was found to be significantly associated with a higher risk for BCC and melanoma by van der Pols et al.58 This prospective study looked at the incidence of skin cancer over 11 years. Study participants with vitamin D levels over 75 nmol/L more frequently developed BCC (P=.01) and melanoma (P=.05). In contrast, SCC was less frequently observed in participants with these high levels of vitamin D (P=.07).58 Furthermore, Park et al60 looked at vitamin D and skin cancer risk for men and women in the United States and found no association with risk for SCC or melanoma but a positive association with BCC (P=.05 for total vitamin D; P<.01 for dietary vitamin D). Additional studies have been performed with inconsistent results, and multiple authors suggest the possible confounding relationship between vitamin D levels and UV radiation exposure.59-62 Furthermore, some studies have even demonstrated a negative association between vitamin D and NMSC. Tang et al63 performed a retrospective case-control study in elderly males, investigating serum levels of vitamin D and patients’ self-reported history of NMSC, which demonstrated that higher levels of vitamin D were associated with a decreased risk for NMSC. Overall, the relationship between vitamin D and skin cancer development remains unclear for both melanoma and NMSC.
Vitamin E—Vitamin E is a fat-soluble vitamin that is found in plant-based oils, nuts, seeds, fruits, and vegetables.64 It works as an antioxidant to protect against free radicals and heighten immune function, and it also serves as a pro-oxidant.65,66 Vitamin E naturally exists in 8 chemical forms, of which gamma-tocopherol is the most frequently obtained form in the diet, and alpha-tocopherol is the most abundant form found in the body.64,65
Early animal studies demonstrated the inhibition of UV-induced damage in mice receiving vitamin E supplementation.67,68 Human studies have not consistently shown these effects. Vural et al50 investigated plasma samples and blood cells of patients with AKs and BCCs and reported a significant decrease in alpha-tocopherol levels in both the AK (P<.05) and BCC (P<.001) groups compared with controls. However, studies also have demonstrated a positive association between vitamin E intake and the development of BCC, including one by Fung et al,42 which found a significant association in women (OR, 1.15; 95% CI, 1.06-1.26).
Vitamin E has been found to inhibit melanin synthesis in the laboratory, suggesting a potentially protective effect in melanoma.69,70 However, in the study performed by Feskanich et al23 examining vitamin intake and melanoma incidence via food-frequency questionnaires, vitamin E was not associated with a lower risk for melanoma. Despite promising laboratory studies, the data surrounding the use of a vitamin E supplement for prevention of melanoma and NMSC in humans remains unclear.
Selenium—Selenium is a trace mineral found in plants, meat, and fish. It plays a key role in reproduction, hormone metabolism, DNA synthesis, and protection from oxidative damage.71 In mice studies, lack of selenium-containing proteins resulted in skin abnormalities, including the development of a hyperplastic epidermis and aberrant hair follicle morphogenesis with alopecia after birth, and numerous experimental studies have demonstrated a negative association between selenium intake and cancer.72,73 However, human studies have yielded alternative results.
The Nutritional Prevention of Cancer Study Group analyzed 1312 dermatology patients with a history of NMSC.74 The study population was obtained from 7 dermatology clinics with randomization to control for confounding variables. Study participants received either 200 μg of selenium daily or placebo.74 Baseline characteristics of each study group were overall balanced. Selenium intake was found to have no effect on the development of BCC (hazard ratio [HR], 1.09; 95% CI, 0.94-1.26) but an increased risk for developing SCC (HR, 1.25; 95% CI, 1.03-1.51) and total NMSC (HR, 1.17; 95% CI, 1.02-1.34).74,75 Similarly, Reid et al76 performed an RCT comparing patients treated with 400 μg/d of selenium to those treated with 200 μg/d of selenium. When compared with placebo, those treated with 200 μg/d of selenium had a statistically significantly increased incidence of NMSC (P=.006); however, those treated with 400 μg/d of selenium had no significant change in total incidence of NMSC (P=.51).76 Furthermore, Vinceti et al77 performed a review of 83 studies from the literature investigating the effect of dietary selenium, and from the RCTs, there was no beneficial effect of selenium in reducing cancer risk in general; however, some studies demonstrated an increased incidence of other types of cancer, including melanoma. Of the RCTs included in the study investigating NMSC incidence specifically, it was found that the incidence was not affected by selenium administration (RR, 1.16; 95% CI, 0.30-4.42; 2 studies, 2027 participants).77 Despite data from several studies demonstrating an increased risk for NMSC, the effects of selenium on the risk for NMSC and melanoma remain unclear.
Combination Antioxidant Studies
In addition to investigating the use of single antioxidants in skin cancer prevention, studies utilizing the combination of various antioxidants or other dietary minerals have been conducted. Hercberg et al78 performed a randomized, double-blinded, placebo-controlled trial of 13,017 adults (7876 women and 5141 men) receiving a combination of 120 mg vitamin C, 30 mg vitamin E, 100 μg selenium, 6 mg beta carotene, and 20 mg zinc. Study participants were followed for an average of 7.5 years, and the development of skin cancers were recorded. Overall, the incidence rate of skin cancer did not differ between the 2 treatment groups; however, when segregated by gender, the study found that there was an increased risk for developing skin cancer in women taking the antioxidant supplement combination compared with placebo (P=.03). This difference was not observed in the 2 treatment groups of male patients (P=.11). When looking specifically at NMSC, there was no difference between treatment groups for male or female patients (P=.39 for males; P=.15 for females). In contrast, there was a higher incidence of melanoma identified in female patients taking the combination antioxidant supplement (P=.01), but this was not seen within the male study population (P=.51).78 In addition, Chang et al79 performed a meta-analysis of 10 previously published RCTs. Analysis revealed that treatment with a variety of supplements, including vitamins A, C, E, and beta carotene, were found to have no preventative effects on the incidence of skin cancer development (RR, 0.98; CI, 0.98-1.03). Notable limitations to this study included the variability in protocols of the studies included in this meta-analysis, the limited number of RCTs investigating vitamin supplementation and the risk for skin cancer development, and the influence of dietary intake on study outcomes.79
Other Dietary Agents
Furocoumarins—Furocoumarins are botanical substances found in various fruits and plants, including many citrus products. Furocoumarins are activated by UV light radiation and can lead to development of a phototoxic eruption. Several studies have suggested a pharmacogenetic effect of furocoumarins.80 Sun et al80 collected dietary data from 47,453 men and 75,291 women on furocoumarin intake and correlation with the development of NMSC. Overall, the study suggested that the intake of furocoumarins may lead to an increase in the development of BCC (HR, 1.16; 95% CI, 1.11-1.21; P=.002); however, there was no significant association identified between total intake of furocoumarins in the risk for SCC or melanoma.80 Furthermore, Sakaki et al81 conducted a survey study looking at the consumption of citrus products and the development of NMSC. The group found that there was an increased risk for NMSC in those consuming an increased amount of citrus products (P=.007).81
Conclusion
Dietary antioxidants have been investigated for their potential role in the prevention of tumorigenesis. Specific antioxidant vitamins, such as vitamin A derivatives and niacinamide, have demonstrated clinical utility in the prevention of NMSC in high-risk populations. Retinol also has been associated with a reduced incidence of melanoma. Numerous antioxidants have demonstrated promising data within the laboratory setting; however, inconsistent results have been appreciated in humans. Furthermore, several research studies suggest that folate, vitamin D, and furocoumarins may be associated with an increased risk for skin cancer development; however, these studies are inconclusive, and dietary studies are challenging to conduct. Overall, RCTs investigating the role of antioxidants for chemoprevention are limited. Moreover, the study of dietary antioxidants and vitamins may be affected by various confounding variables that can be difficult to account for because of patients’ potentially poor recall of dietary intake and the effect of dietary intake in supplemental studies. Given the increasing prevalence of skin cancer worldwide, further research into the clinical utility of antioxidants in skin cancer prevention is warranted.
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7-33.
- Global Burden of Disease Cancer Collaboration; Fitzmaurice C, Abate D, Abbasi N, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the Global Burden of Disease Study. JAMA Oncol. 2019;5:1749-1768.
- Leiter U, Keim U, Garbe C. Epidemiology of skin cancer: update 2019. In: Reichrath J, ed. Sunlight, Vitamin D and Skin Cancer. Springer International Publishing; 2020:123-139.
- Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206; quiz 178.
- Miller DL, Weinstock MA. Nonmelanoma skin cancer in the United States: incidence. J Am Acad Dermatol. 1994;30:774-778.
- Young AR, Claveau J, Rossi AB. Ultraviolet radiation and the skin: photobiology and sunscreen photoprotection. J Am Acad Dermatol. 2017;76(3S1):S100-S109.
- Pleasance ED, Cheetham RK, Stephens PJ, et al. A comprehensive catalogue of somatic mutations from a human cancer genome. Nature. 2010;463:191-196.
- Baek J, Lee MG. Oxidative stress and antioxidant strategies in dermatology. Redox Rep. 2016;21:164-169.
- Katta R, Brown DN. Diet and skin cancer: the potential role of dietary antioxidants in nonmelanoma skin cancer prevention. J Skin Cancer. 2015;2015:893149.
- Stoj V, Shahriari N, Shao K, et al. Nutrition and nonmelanoma skin cancers. Clin Dermatol. 2022;40:173-185.
- O’Connor EA, Evans CV, Ivlev I, et al. Vitamin and mineral supplements for the primary prevention of cardiovascular disease and cancer: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2022;327:2334-2347.
- National Institutes of Health Office of Dietary Supplements. Vitamin A and carotenoids. fact sheet for health professionals. Updated June 15, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminA-HealthProfessional/
- Keller KL, Fenske NA. Uses of vitamins A, C, and E and related compounds in dermatology: a review. J Am Acad Dermatol. 1998;39:611-625.
- Wright TI, Spencer JM, Flowers FP. Chemoprevention of nonmelanoma skin cancer. J Am Acad Dermatol. 2006;54:933-946; quiz 947-950.
- Bushue N, Wan YJY. Retinoid pathway and cancer therapeutics. Adv Drug Deliv Rev. 2010;62:1285-1298.
- Stahl W, Sies H. β-Carotene and other carotenoids in protection from sunlight. Am J Clin Nutr. 2012;96:1179S-1184S.
- Bukhari MH, Qureshi SS, Niazi S, et al. Chemotherapeutic/chemopreventive role of retinoids in chemically induced skin carcinogenesis in albino mice. Int J Dermatol. 2007;46:1160-1165.
- Lambert LA, Wamer WG, Wei RR, et al. The protective but nonsynergistic effect of dietary beta-carotene and vitamin E on skin tumorigenesis in Skh mice. Nutr Cancer. 1994;21:1-12.
- Greenberg ER, Baron JA, Stukel TA, et al. A clinical trial of beta carotene to prevent basal-cell and squamous-cell cancers of the skin. The Skin Cancer Prevention Study Group. N Engl J Med. 1990;323:789-795.
- Frieling UM, Schaumberg DA, Kupper TS, et al. A randomized, 12-year primary-prevention trial of beta carotene supplementation for nonmelanoma skin cancer in the physician’s health study. Arch Dermatol. 2000;136:179-184.
- Naldi L, Gallus S, Tavani A, et al; Oncology Study Group of the Italian Group for Epidemiologic Research in Dermatology. Risk of melanoma and vitamin A, coffee and alcohol: a case-control study from Italy. Eur J Cancer Prev. 2004;13:503-508.
- Zhang YP, Chu RX, Liu H. Vitamin A intake and risk of melanoma: a meta-analysis. PloS One. 2014;9:e102527.
- Feskanich D, Willett WC, Hunter DJ, et al. Dietary intakes of vitamins A, C, and E and risk of melanoma in two cohorts of women. Br J Cancer. 2003;88:1381-1387.
- Bavinck JN, Tieben LM, Van der Woude FJ, et al. Prevention of skin cancer and reduction of keratotic skin lesions during acitretin therapy in renal transplant recipients: a double-blind, placebo-controlled study. J Clin Oncol. 1995;13:1933-1938.
- George R, Weightman W, Russ GR, et al. Acitretin for chemoprevention of non-melanoma skin cancers in renal transplant recipients. Australas J Dermatol. 2002;43:269-273.
- Solomon-Cohen E, Reiss-Huss S, Hodak E, et al. Low-dose acitretin for secondary prevention of keratinocyte carcinomas in solid-organ transplant recipients. Dermatology. 2022;238:161-166.
- Otley CC, Stasko T, Tope WD, et al. Chemoprevention of nonmelanoma skin cancer with systemic retinoids: practical dosing and management of adverse effects. Dermatol Surg. 2006;32:562-568.
- Kadakia KC, Barton DL, Loprinzi CL, et al. Randomized controlled trial of acitretin versus placebo in patients at high-risk for basal cell or squamous cell carcinoma of the skin (North Central Cancer Treatment Group Study 969251). Cancer. 2012;118:2128-2137.
- McKenna DB, Murphy GM. Skin cancer chemoprophylaxis in renal transplant recipients: 5 years of experience using low-dose acitretin. Br J Dermatol. 1999;140:656-660.
- National Institutes of Health Office of Dietary Supplements. Niacin: fact sheet for health professionals. Updated August 23, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/Niacin-HealthProfessional/
- Malesu R, Martin AJ, Lyons JG, et al. Nicotinamide for skin cancer chemoprevention: effects of nicotinamide on melanoma in vitro and in vivo. Photochem Photobiol Sci. 2020;19:171-179.
- Gensler HL. Prevention of photoimmunosuppression and photocarcinogenesis by topical nicotinamide. Nutr Cancer. 1997;29:157-162.
- Gensler HL, Williams T, Huang AC, et al. Oral niacin prevents photocarcinogenesis and photoimmunosuppression in mice. Nutr Cancer. 1999;34:36-41.
- Chen AC, Martin AJ, Choy B, et al. A phase 3 randomized trial of nicotinamide for skin-cancer chemoprevention. N Engl J Med. 2015;373:1618-1626.
- Drago F, Ciccarese G, Cogorno L, et al. Prevention of non-melanoma skin cancers with nicotinamide in transplant recipients: a case-control study. Eur J Dermatol. 2017;27:382-385.
- Yélamos O, Halpern AC, Weinstock MA. Reply to “A phase II randomized controlled trial of nicotinamide for skin cancer chemoprevention in renal transplant recipients.” Br J Dermatol. 2017;176:551-552.
- Scatozza F, Moschella F, D’Arcangelo D, et al. Nicotinamide inhibits melanoma in vitro and in vivo. J Exp Clin Cancer Res. 2020;39:211.
- National Institutes of Health Office of Dietary Supplements. Folate: fact sheet for health professionals. Updated November 1, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/Folate-HealthProfessional/
- Butzbach K, Epe B. Photogenotoxicity of folic acid. Free Radic Biol Med. 2013;65:821-827.
- Vollset SE, Clarke R, Lewington S, et al. Effects of folic acid supplementation on overall and site-specific cancer incidence during the randomised trials: meta-analyses of data on 50,000 individuals. Lancet. 2013;381:1029-1036.
- Donnenfeld M, Deschasaux M, Latino-Martel P, et al. Prospective association between dietary folate intake and skin cancer risk: results from the Supplémentation en Vitamines et Minéraux Antioxydants cohort. Am J Clin Nutr. 2015;102:471-478.
- Fung TT, Hunter DJ, Spiegelman D, et al. Vitamins and carotenoids intake and the risk of basal cell carcinoma of the skin in women (United States). Cancer Causes Control. 2002;13:221-230.
- Fung TT, Spiegelman D, Egan KM, et al. Vitamin and carotenoid intake and risk of squamous cell carcinoma of the skin. Int J Cancer. 2003;103:110-115.
- National Institutes of Health Office of Dietary Supplements. Vitamin C: fact sheet for health professionals. Updated March 26, 2021. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminC-HealthProfessional/
- Spoelstra-de Man AME, Elbers PWG, Oudemans-Van Straaten HM. Vitamin C: should we supplement? Curr Opin Crit Care. 2018;24:248-255.
- Moison RMW, Beijersbergen van Henegouwen GMJ. Topical antioxidant vitamins C and E prevent UVB-radiation-induced peroxidation of eicosapentaenoic acid in pig skin. Radiat Res. 2002;157:402-409.
- Lin JY, Selim MA, Shea CR, et al. UV photoprotection by combination topical antioxidants vitamin C and vitamin E. J Am Acad Dermatol. 2003;48:866-874.
- Pauling L, Willoughby R, Reynolds R, et al. Incidence of squamous cell carcinoma in hairless mice irradiated with ultraviolet light in relation to intake of ascorbic acid (vitamin C) and of D, L-alpha-tocopheryl acetate (vitamin E). Int J Vitam Nutr Res Suppl. 1982;23:53-82.
- Kune GA, Bannerman S, Field B, et al. Diet, alcohol, smoking, serum beta-carotene, and vitamin A in male nonmelanocytic skin cancer patients and controls. Nutr Cancer. 1992;18:237-244.
- Vural P, Canbaz M, Selçuki D. Plasma antioxidant defense in actinic keratosis and basal cell carcinoma. J Eur Acad Dermatol Venereol. 1999;13:96-101.
- Record IR, Dreosti IE, McInerney JK. Changes in plasma antioxidant status following consumption of diets high or low in fruit and vegetables or following dietary supplementation with an antioxidant mixture. Br J Nutr. 2001;85:459-464.
- Heinen MM, Hughes MC, Ibiebele TI, et al. Intake of antioxidant nutrients and the risk of skin cancer. Eur J Cancer. 2007;43:2707-2716.
- Yang G, Yan Y, Ma Y, et al. Vitamin C at high concentrations induces cytotoxicity in malignant melanoma but promotes tumor growth at low concentrations. Mol Carcinog. 2017;56:1965-1976.
- National Institutes of Health Office of Dietary Supplements. Vitamin D: fact sheet for health professionals. Updated August 12, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/
- Reichrath J, Saternus R, Vogt T. Endocrine actions of vitamin D in skin: relevance for photocarcinogenesis of non-melanoma skin cancer, and beyond. Mol Cell Endocrinol. 2017;453:96-102.
- Ellison TI, Smith MK, Gilliam AC, et al. Inactivation of the vitamin D receptor enhances susceptibility of murine skin to UV-induced tumorigenesis. J Invest Dermatol. 2008;128:2508-2517.
- Eide MJ, Johnson DA, Jacobsen GR, et al. Vitamin D and nonmelanoma skin cancer in a health maintenance organization cohort. Arch Dermatol. 2011;147:1379-1384.
- van der Pols JC, Russell A, Bauer U, et al. Vitamin D status and skin cancer risk independent of time outdoors: 11-year prospective study in an Australian community. J Invest Dermatol. 2013;133:637-641.
- Caini S, Gnagnarella P, Stanganelli I, et al. Vitamin D and the risk of non-melanoma skin cancer: a systematic literature review and meta-analysis on behalf of the Italian Melanoma Intergroup. Cancers (Basel). 2021;13:4815.
- Park SM, Li T, Wu S, et al. Vitamin D intake and risk of skin cancer in US women and men. PLoS One. 2016;11:e0160308.
- Afzal S, Nordestgaard BG, Bojesen SE. Plasma 25-hydroxyvitamin D and risk of non-melanoma and melanoma skin cancer: a prospective cohort study. J Invest Dermatol. 2013;133:629-636.
- Asgari MM, Tang J, Warton ME, et al. Association of prediagnostic serum vitamin D levels with the development of basal cell carcinoma. J Invest Dermatol. 2010;130:1438-1443.
- Tang JY, Parimi N, Wu A, et al. Inverse association between serum 25(OH) vitamin D levels and non-melanoma skin cancer in elderly men. Cancer Causes Control. 2010;21:387-391.
- Keen MA, Hassan I. Vitamin E in dermatology. Indian Dermatol Online J. 2016;7:311-315.
- National Institutes of Health Office of Dietary Supplements. Vitamin E: fact sheet for health professionals. Updated March 26, 2021. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional/
- Pearson P, Lewis SA, Britton J, et al. The pro-oxidant activity of high-dose vitamin E supplements in vivo. BioDrugs. 2006;20:271-273.
- Gerrish KE, Gensler HL. Prevention of photocarcinogenesis by dietary vitamin E. Nutr Cancer. 1993;19:125-133.
- McVean M, Liebler DC. Prevention of DNA photodamage by vitamin E compounds and sunscreens: roles of ultraviolet absorbance and cellular uptake. Mol Carcinog. 1999;24:169-176.
- Prasad KN, Cohrs RJ, Sharma OK. Decreased expressions of c-myc and H-ras oncogenes in vitamin E succinate induced morphologically differentiated murine B-16 melanoma cells in culture. Biochem Cell Biol. 1990;68:1250-1255.
- Funasaka Y, Komoto M, Ichihashi M. Depigmenting effect of alpha-tocopheryl ferulate on normal human melanocytes. Pigment Cell Res. 2000;13(suppl 8):170-174.
- National Institutes of Health Office of Dietary Supplements. Selenium: fact sheet for health professionals. Updated March 26, 2021. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/Selenium-HealthProfessional/
- Sengupta A, Lichti UF, Carlson BA, et al. Selenoproteins are essential for proper keratinocyte function and skin development. PLoS One. 2010;5:e12249.
- Das RK, Hossain SKU, Bhattacharya S. Diphenylmethyl selenocyanate inhibits DMBA-croton oil induced two-stage mouse skin carcinogenesis by inducing apoptosis and inhibiting cutaneous cell proliferation. Cancer Lett. 2005;230:90-101.
- Clark LC, Combs GF Jr, Turnbull BW, et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA. 1996;276:1957-1963.
- Duffield-Lillico AJ, Slate EH, Reid ME, et al. Selenium supplementation and secondary prevention of nonmelanoma skin cancer in a randomized trial. J Natl Cancer Inst. 2003;95:1477-1481.
- Reid ME, Duffield-Lillico AJ, Slate E, et al. The nutritional prevention of cancer: 400 mcg per day selenium treatment. Nutr Cancer. 2008;60:155-163.
- Vinceti M, Filippini T, Del Giovane C, et al. Selenium for preventing cancer. Cochrane Database Syst Rev. 2018;1:CD005195.
- Hercberg S, Ezzedine K, Guinot C, et al. Antioxidant supplementation increases the risk of skin cancers in women but not in men. J Nutr. 2007;137:2098-2105.
- Chang YJ, Myung SK, Chung ST, et al. Effects of vitamin treatment or supplements with purported antioxidant properties on skin cancer prevention: a meta-analysis of randomized controlled trials. Dermatology. 2011;223:36-44.
- Sun W, Rice MS, Park MK, et al. Intake of furocoumarins and risk of skin cancer in 2 prospective US cohort studies. J Nutr. 2020;150:1535-1544.
- Sakaki JR, Melough MM, Roberts MB, et al. Citrus consumption and the risk of non-melanoma skin cancer in the Women’s Health Initiative. Cancers (Basel). 2021;13:2173.
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7-33.
- Global Burden of Disease Cancer Collaboration; Fitzmaurice C, Abate D, Abbasi N, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the Global Burden of Disease Study. JAMA Oncol. 2019;5:1749-1768.
- Leiter U, Keim U, Garbe C. Epidemiology of skin cancer: update 2019. In: Reichrath J, ed. Sunlight, Vitamin D and Skin Cancer. Springer International Publishing; 2020:123-139.
- Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206; quiz 178.
- Miller DL, Weinstock MA. Nonmelanoma skin cancer in the United States: incidence. J Am Acad Dermatol. 1994;30:774-778.
- Young AR, Claveau J, Rossi AB. Ultraviolet radiation and the skin: photobiology and sunscreen photoprotection. J Am Acad Dermatol. 2017;76(3S1):S100-S109.
- Pleasance ED, Cheetham RK, Stephens PJ, et al. A comprehensive catalogue of somatic mutations from a human cancer genome. Nature. 2010;463:191-196.
- Baek J, Lee MG. Oxidative stress and antioxidant strategies in dermatology. Redox Rep. 2016;21:164-169.
- Katta R, Brown DN. Diet and skin cancer: the potential role of dietary antioxidants in nonmelanoma skin cancer prevention. J Skin Cancer. 2015;2015:893149.
- Stoj V, Shahriari N, Shao K, et al. Nutrition and nonmelanoma skin cancers. Clin Dermatol. 2022;40:173-185.
- O’Connor EA, Evans CV, Ivlev I, et al. Vitamin and mineral supplements for the primary prevention of cardiovascular disease and cancer: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2022;327:2334-2347.
- National Institutes of Health Office of Dietary Supplements. Vitamin A and carotenoids. fact sheet for health professionals. Updated June 15, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminA-HealthProfessional/
- Keller KL, Fenske NA. Uses of vitamins A, C, and E and related compounds in dermatology: a review. J Am Acad Dermatol. 1998;39:611-625.
- Wright TI, Spencer JM, Flowers FP. Chemoprevention of nonmelanoma skin cancer. J Am Acad Dermatol. 2006;54:933-946; quiz 947-950.
- Bushue N, Wan YJY. Retinoid pathway and cancer therapeutics. Adv Drug Deliv Rev. 2010;62:1285-1298.
- Stahl W, Sies H. β-Carotene and other carotenoids in protection from sunlight. Am J Clin Nutr. 2012;96:1179S-1184S.
- Bukhari MH, Qureshi SS, Niazi S, et al. Chemotherapeutic/chemopreventive role of retinoids in chemically induced skin carcinogenesis in albino mice. Int J Dermatol. 2007;46:1160-1165.
- Lambert LA, Wamer WG, Wei RR, et al. The protective but nonsynergistic effect of dietary beta-carotene and vitamin E on skin tumorigenesis in Skh mice. Nutr Cancer. 1994;21:1-12.
- Greenberg ER, Baron JA, Stukel TA, et al. A clinical trial of beta carotene to prevent basal-cell and squamous-cell cancers of the skin. The Skin Cancer Prevention Study Group. N Engl J Med. 1990;323:789-795.
- Frieling UM, Schaumberg DA, Kupper TS, et al. A randomized, 12-year primary-prevention trial of beta carotene supplementation for nonmelanoma skin cancer in the physician’s health study. Arch Dermatol. 2000;136:179-184.
- Naldi L, Gallus S, Tavani A, et al; Oncology Study Group of the Italian Group for Epidemiologic Research in Dermatology. Risk of melanoma and vitamin A, coffee and alcohol: a case-control study from Italy. Eur J Cancer Prev. 2004;13:503-508.
- Zhang YP, Chu RX, Liu H. Vitamin A intake and risk of melanoma: a meta-analysis. PloS One. 2014;9:e102527.
- Feskanich D, Willett WC, Hunter DJ, et al. Dietary intakes of vitamins A, C, and E and risk of melanoma in two cohorts of women. Br J Cancer. 2003;88:1381-1387.
- Bavinck JN, Tieben LM, Van der Woude FJ, et al. Prevention of skin cancer and reduction of keratotic skin lesions during acitretin therapy in renal transplant recipients: a double-blind, placebo-controlled study. J Clin Oncol. 1995;13:1933-1938.
- George R, Weightman W, Russ GR, et al. Acitretin for chemoprevention of non-melanoma skin cancers in renal transplant recipients. Australas J Dermatol. 2002;43:269-273.
- Solomon-Cohen E, Reiss-Huss S, Hodak E, et al. Low-dose acitretin for secondary prevention of keratinocyte carcinomas in solid-organ transplant recipients. Dermatology. 2022;238:161-166.
- Otley CC, Stasko T, Tope WD, et al. Chemoprevention of nonmelanoma skin cancer with systemic retinoids: practical dosing and management of adverse effects. Dermatol Surg. 2006;32:562-568.
- Kadakia KC, Barton DL, Loprinzi CL, et al. Randomized controlled trial of acitretin versus placebo in patients at high-risk for basal cell or squamous cell carcinoma of the skin (North Central Cancer Treatment Group Study 969251). Cancer. 2012;118:2128-2137.
- McKenna DB, Murphy GM. Skin cancer chemoprophylaxis in renal transplant recipients: 5 years of experience using low-dose acitretin. Br J Dermatol. 1999;140:656-660.
- National Institutes of Health Office of Dietary Supplements. Niacin: fact sheet for health professionals. Updated August 23, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/Niacin-HealthProfessional/
- Malesu R, Martin AJ, Lyons JG, et al. Nicotinamide for skin cancer chemoprevention: effects of nicotinamide on melanoma in vitro and in vivo. Photochem Photobiol Sci. 2020;19:171-179.
- Gensler HL. Prevention of photoimmunosuppression and photocarcinogenesis by topical nicotinamide. Nutr Cancer. 1997;29:157-162.
- Gensler HL, Williams T, Huang AC, et al. Oral niacin prevents photocarcinogenesis and photoimmunosuppression in mice. Nutr Cancer. 1999;34:36-41.
- Chen AC, Martin AJ, Choy B, et al. A phase 3 randomized trial of nicotinamide for skin-cancer chemoprevention. N Engl J Med. 2015;373:1618-1626.
- Drago F, Ciccarese G, Cogorno L, et al. Prevention of non-melanoma skin cancers with nicotinamide in transplant recipients: a case-control study. Eur J Dermatol. 2017;27:382-385.
- Yélamos O, Halpern AC, Weinstock MA. Reply to “A phase II randomized controlled trial of nicotinamide for skin cancer chemoprevention in renal transplant recipients.” Br J Dermatol. 2017;176:551-552.
- Scatozza F, Moschella F, D’Arcangelo D, et al. Nicotinamide inhibits melanoma in vitro and in vivo. J Exp Clin Cancer Res. 2020;39:211.
- National Institutes of Health Office of Dietary Supplements. Folate: fact sheet for health professionals. Updated November 1, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/Folate-HealthProfessional/
- Butzbach K, Epe B. Photogenotoxicity of folic acid. Free Radic Biol Med. 2013;65:821-827.
- Vollset SE, Clarke R, Lewington S, et al. Effects of folic acid supplementation on overall and site-specific cancer incidence during the randomised trials: meta-analyses of data on 50,000 individuals. Lancet. 2013;381:1029-1036.
- Donnenfeld M, Deschasaux M, Latino-Martel P, et al. Prospective association between dietary folate intake and skin cancer risk: results from the Supplémentation en Vitamines et Minéraux Antioxydants cohort. Am J Clin Nutr. 2015;102:471-478.
- Fung TT, Hunter DJ, Spiegelman D, et al. Vitamins and carotenoids intake and the risk of basal cell carcinoma of the skin in women (United States). Cancer Causes Control. 2002;13:221-230.
- Fung TT, Spiegelman D, Egan KM, et al. Vitamin and carotenoid intake and risk of squamous cell carcinoma of the skin. Int J Cancer. 2003;103:110-115.
- National Institutes of Health Office of Dietary Supplements. Vitamin C: fact sheet for health professionals. Updated March 26, 2021. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminC-HealthProfessional/
- Spoelstra-de Man AME, Elbers PWG, Oudemans-Van Straaten HM. Vitamin C: should we supplement? Curr Opin Crit Care. 2018;24:248-255.
- Moison RMW, Beijersbergen van Henegouwen GMJ. Topical antioxidant vitamins C and E prevent UVB-radiation-induced peroxidation of eicosapentaenoic acid in pig skin. Radiat Res. 2002;157:402-409.
- Lin JY, Selim MA, Shea CR, et al. UV photoprotection by combination topical antioxidants vitamin C and vitamin E. J Am Acad Dermatol. 2003;48:866-874.
- Pauling L, Willoughby R, Reynolds R, et al. Incidence of squamous cell carcinoma in hairless mice irradiated with ultraviolet light in relation to intake of ascorbic acid (vitamin C) and of D, L-alpha-tocopheryl acetate (vitamin E). Int J Vitam Nutr Res Suppl. 1982;23:53-82.
- Kune GA, Bannerman S, Field B, et al. Diet, alcohol, smoking, serum beta-carotene, and vitamin A in male nonmelanocytic skin cancer patients and controls. Nutr Cancer. 1992;18:237-244.
- Vural P, Canbaz M, Selçuki D. Plasma antioxidant defense in actinic keratosis and basal cell carcinoma. J Eur Acad Dermatol Venereol. 1999;13:96-101.
- Record IR, Dreosti IE, McInerney JK. Changes in plasma antioxidant status following consumption of diets high or low in fruit and vegetables or following dietary supplementation with an antioxidant mixture. Br J Nutr. 2001;85:459-464.
- Heinen MM, Hughes MC, Ibiebele TI, et al. Intake of antioxidant nutrients and the risk of skin cancer. Eur J Cancer. 2007;43:2707-2716.
- Yang G, Yan Y, Ma Y, et al. Vitamin C at high concentrations induces cytotoxicity in malignant melanoma but promotes tumor growth at low concentrations. Mol Carcinog. 2017;56:1965-1976.
- National Institutes of Health Office of Dietary Supplements. Vitamin D: fact sheet for health professionals. Updated August 12, 2022. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/
- Reichrath J, Saternus R, Vogt T. Endocrine actions of vitamin D in skin: relevance for photocarcinogenesis of non-melanoma skin cancer, and beyond. Mol Cell Endocrinol. 2017;453:96-102.
- Ellison TI, Smith MK, Gilliam AC, et al. Inactivation of the vitamin D receptor enhances susceptibility of murine skin to UV-induced tumorigenesis. J Invest Dermatol. 2008;128:2508-2517.
- Eide MJ, Johnson DA, Jacobsen GR, et al. Vitamin D and nonmelanoma skin cancer in a health maintenance organization cohort. Arch Dermatol. 2011;147:1379-1384.
- van der Pols JC, Russell A, Bauer U, et al. Vitamin D status and skin cancer risk independent of time outdoors: 11-year prospective study in an Australian community. J Invest Dermatol. 2013;133:637-641.
- Caini S, Gnagnarella P, Stanganelli I, et al. Vitamin D and the risk of non-melanoma skin cancer: a systematic literature review and meta-analysis on behalf of the Italian Melanoma Intergroup. Cancers (Basel). 2021;13:4815.
- Park SM, Li T, Wu S, et al. Vitamin D intake and risk of skin cancer in US women and men. PLoS One. 2016;11:e0160308.
- Afzal S, Nordestgaard BG, Bojesen SE. Plasma 25-hydroxyvitamin D and risk of non-melanoma and melanoma skin cancer: a prospective cohort study. J Invest Dermatol. 2013;133:629-636.
- Asgari MM, Tang J, Warton ME, et al. Association of prediagnostic serum vitamin D levels with the development of basal cell carcinoma. J Invest Dermatol. 2010;130:1438-1443.
- Tang JY, Parimi N, Wu A, et al. Inverse association between serum 25(OH) vitamin D levels and non-melanoma skin cancer in elderly men. Cancer Causes Control. 2010;21:387-391.
- Keen MA, Hassan I. Vitamin E in dermatology. Indian Dermatol Online J. 2016;7:311-315.
- National Institutes of Health Office of Dietary Supplements. Vitamin E: fact sheet for health professionals. Updated March 26, 2021. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional/
- Pearson P, Lewis SA, Britton J, et al. The pro-oxidant activity of high-dose vitamin E supplements in vivo. BioDrugs. 2006;20:271-273.
- Gerrish KE, Gensler HL. Prevention of photocarcinogenesis by dietary vitamin E. Nutr Cancer. 1993;19:125-133.
- McVean M, Liebler DC. Prevention of DNA photodamage by vitamin E compounds and sunscreens: roles of ultraviolet absorbance and cellular uptake. Mol Carcinog. 1999;24:169-176.
- Prasad KN, Cohrs RJ, Sharma OK. Decreased expressions of c-myc and H-ras oncogenes in vitamin E succinate induced morphologically differentiated murine B-16 melanoma cells in culture. Biochem Cell Biol. 1990;68:1250-1255.
- Funasaka Y, Komoto M, Ichihashi M. Depigmenting effect of alpha-tocopheryl ferulate on normal human melanocytes. Pigment Cell Res. 2000;13(suppl 8):170-174.
- National Institutes of Health Office of Dietary Supplements. Selenium: fact sheet for health professionals. Updated March 26, 2021. Accessed November 14, 2022. https://ods.od.nih.gov/factsheets/Selenium-HealthProfessional/
- Sengupta A, Lichti UF, Carlson BA, et al. Selenoproteins are essential for proper keratinocyte function and skin development. PLoS One. 2010;5:e12249.
- Das RK, Hossain SKU, Bhattacharya S. Diphenylmethyl selenocyanate inhibits DMBA-croton oil induced two-stage mouse skin carcinogenesis by inducing apoptosis and inhibiting cutaneous cell proliferation. Cancer Lett. 2005;230:90-101.
- Clark LC, Combs GF Jr, Turnbull BW, et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA. 1996;276:1957-1963.
- Duffield-Lillico AJ, Slate EH, Reid ME, et al. Selenium supplementation and secondary prevention of nonmelanoma skin cancer in a randomized trial. J Natl Cancer Inst. 2003;95:1477-1481.
- Reid ME, Duffield-Lillico AJ, Slate E, et al. The nutritional prevention of cancer: 400 mcg per day selenium treatment. Nutr Cancer. 2008;60:155-163.
- Vinceti M, Filippini T, Del Giovane C, et al. Selenium for preventing cancer. Cochrane Database Syst Rev. 2018;1:CD005195.
- Hercberg S, Ezzedine K, Guinot C, et al. Antioxidant supplementation increases the risk of skin cancers in women but not in men. J Nutr. 2007;137:2098-2105.
- Chang YJ, Myung SK, Chung ST, et al. Effects of vitamin treatment or supplements with purported antioxidant properties on skin cancer prevention: a meta-analysis of randomized controlled trials. Dermatology. 2011;223:36-44.
- Sun W, Rice MS, Park MK, et al. Intake of furocoumarins and risk of skin cancer in 2 prospective US cohort studies. J Nutr. 2020;150:1535-1544.
- Sakaki JR, Melough MM, Roberts MB, et al. Citrus consumption and the risk of non-melanoma skin cancer in the Women’s Health Initiative. Cancers (Basel). 2021;13:2173.
Practice Points
- Melanoma and nonmelanoma skin cancer (NMSC) are 2 of the most frequently diagnosed cancers in the United States. UV radiation plays a key role in the pathogenesis of both.
- Dietary antioxidants may mechanistically decrease DNA damage caused by UV radiation and could play a potential role in the prevention or development of melanoma and NMSC.
Juvenile Dermatomyositis–Associated Panniculitis
To the Editor:
Juvenile dermatomyositis (JDM) is an autoimmune disorder with childhood onset that predominantly affects the muscles and skin, among other organs. Since the recognition of dermatomyositis (DM) more than 100 years ago, a variety of clinical diagnostic criteria have been utilized. Classically, DM presents with muscle weakness and a pathognomonic cutaneous macular, violaceous, erythematous eruption. The juvenile variant is defined by onset prior to 16 years of age. Histologically, these entities are indistinguishable and demonstrate an interface dermatitis with epidermal atrophy. Clinically, JDM has a higher incidence of calcinosis cutis and is not associated with an increased risk for malignancy in contrast to the adult-onset variant.1 Panniculitis is a rare but serious complication in a subset of patients with DM and may represent a precursor to calcinosis cutis.2 We describe a case of JDM-associated panniculitis that was difficult to control with prednisone and rituximab.
A 21-year-old woman with fever, fatigue, muscle pain, and new-onset swelling of 2 weeks’ duration was admitted to the hospital. She had a 5-year history of intermittent muscle weakness and concomitant rash. Prior to presentation, she had been hospitalized twice for fever of unknown origin, and the source remained undetermined. Physical examination revealed prominent facial and periorbital edema. There was tender nonpitting edema present on all 4 extremities and hyperpigmented indurated nodules on the shins (Figure 1). A full laboratory and imaging workup was performed for autoantibodies and infectious etiologies. The complete blood cell count was notable for pancytopenia, and a thorough infectious workup was negative. Creatine kinase level was within reference range. A biopsy of the right shin was performed, and histopathology revealed a lobular panniculitis with fat necrosis and mixed inflammation with neutrophils with perieccrine involvement as well as an interface dermatitis (Figure 2). Periodic acid–Schiff, Grocott methenamine-silver, and Gram stains were negative. Myositis-specific antibody testing revealed anti-p155/140 autoantibodies, and magnetic resonance imaging did not reveal active myositis within the visualized muscles, consistent with stable nonprogressing DM. A diagnosis of JDM with panniculitis was made. The patient was started on oral prednisone. Subsequently, a trial of rituximab was initiated. Although the patient’s symptoms initially improved, the response was not sustained on rituximab, and the patient was continued on systemic steroids with initiation of cyclosporine.
Juvenile dermatomyositis is an autoimmune disorder with childhood onset that involves systemic inflammation of the muscles, skin, and internal organs. It often can present diagnostic and therapeutic challenges.2,3 Bohan and Peter4,5 clinical criteria may help identify potential patients with JDM, but magnetic resonance imaging, electromyography, and muscle biopsy often are required to confirm the diagnosis.6 Skin manifestations include heliotrope rash; V sign; shawl sign; Gottron papules; periorbital edema; and infrequently panniculitis, the subcutaneous inflammation of adipose tissue.3,7
Although panniculitis is found in approximately 10% of skin biopsies in patients with DM, our patient presented with anti-p155/140 antibodies.8-10 Fat involvement in these patients traditionally manifests as lipodystrophy. Panniculitis also may precede calcinosis cutis, a debilitating skin change that may occur in approximately 46% of patients with JDM and can cause severe morbidity.2,6,9
Subcutaneous edema rarely is described in DM-panniculitis, present in only 6% of 86 DM patients in one study.7 The pathophysiology of DM may be due to antibodies that target endothelial cells and activate complement, resulting in the membranolytic attack complex. This leads to microischemia, and microinfarction of the muscle fibers has been suggested to result in edema of the subcutaneous tissue in severe cases.7,11 Microinfarction has been found to be present 2.3 times more often in edematous DM compared with nonedematous DM.7 Subcutaneous edema may be an isolated presentation of DM that arises more quickly with severe disease activity. As such, recommendations have been made to consider edema in future classification schemes.7
Because of the severity of edematous and/or subcutaneous DM, aggressive therapy may be required. First-line therapy consists of corticosteroids with additional immunosuppressants and immunomodulatory agents if adequate response is not achieved.3,12 The effectiveness of rituximab in DM has been suggested.2,12,13 The Rituximab in Myositis (RIM) trial (N=200) was the first double-blind, placebo-controlled, phase 3 clinical trial to assess rituximab’s efficacy in refractory compared with early-onset inflammatory myopathies. Although outcomes were similar in both groups, 83% of patients overall, including the JDM subset, met the definition of improvement.12 In re-examining the RIM trial data and other cases using rituximab to treat inflammatory myopathies, an overall response rate of 78.3% was observed, with 52.1% of patients with DM reporting improvement in skin lesions (N=458, pooled from 48 studies).13 Further analysis of the RIM data revealed that panniculitis affected 10.4% of patients with JDM at baseline, which decreased to 6.8% at 36 weeks of rituximab therapy (N=48).12
As exhibited in our patient, subcutaneous tissue involvement, including calcinosis cutis and panniculitis, is seen more often in JDM than adult DM.2,6 However, panniculitis in anti-p155/140 patients is rare. Our patient also had antibody positivity, which likely predisposed her to a more severe course. Despite not having sustained improvement on rituximab, initiating aggressive therapy earlier in the disease course may be beneficial, and our patient continues with alternative therapies.
- Jorizzo JL, Vleugels RA. Dermatomyositis. In: Bolognia J, Schaffer J, Cerroni L. Dermatology. 4th ed. Elsevier; 2019:681-687.
- Aggarwal R, Loganathan P, Koontz D, et al. Cutaneous improvement in refractory adult and juvenile dermatomyositis after treatment with rituximab. Rheumatology. 2016;56:247-254.
- Santos-Briz A, Calle A, Linos K, et al. Dermatomyositis panniculitis: a clinicopathological and immunohistochemical study of 18 cases. J Eur Acad Dermatol Venereol. 2018;32:1352-1359.
- Bohan A, Peter JB. Polymyositis and dermatomyositis (first of two parts). N Engl J Med. 1975;292:344-347.
- Bohan A, Peter JB. Polymyositis and dermatomyositis (second of two parts). N Engl J Med. 1975;292:403-407.
- Sakurai N, Hino-Shishikura A, Nozawa T, et al. Clinical significance of subcutaneous fat and fascial involvement in juvenile dermatomyositis. Mod Rheumatol. 2019;29:808-813.
- Milisenda JC, Doti PI, Prieto-Gonzalez S, et al. Dermatomyositis presenting with severe subcutaneous edema: five additional cases and review of the literature. Semin Arthritis Rheum. 2014;44:228-233.
- Janis JF, Winkelmann RK. Histopathology of the skin in dermatomyositis: a histopathologic study of 55 cases. Arch Dermatol. 1968;97:640-650.
- van Dongen HM, van Vugt RM, Stoof TJ. Extensive persistent panniculitis in the context of dermatomyositis. J Clin Rheumatol. 2020;26:e187-e188.
- Gunawardena H, Wedderburn LR, North J, et al. Clinical associations of autoantibodies to a p155/140 kDa doublet protein in juvenile dermatomyositis. Rheumatology. 2008;47:324-328.
- Dalakas MC, Hohlfeld R. Polymyositis and dermatomyositis. Lancet. 2003;362:971-982.
- Oddis CV, Reed AM, Aggarwal R, et al. Rituximab in the treatment of refractory adult and juvenile dermatomyositis and adult polymyositis: a randomized, placebo-phase trial. Arthritis Rheum. 2013;65:314-324.
- Fasano S, Gordon P, Hajji R, et al. Rituximab in the treatment of inflammatory myopathies: a review. Rheumatology. 2016;56:26-36.
To the Editor:
Juvenile dermatomyositis (JDM) is an autoimmune disorder with childhood onset that predominantly affects the muscles and skin, among other organs. Since the recognition of dermatomyositis (DM) more than 100 years ago, a variety of clinical diagnostic criteria have been utilized. Classically, DM presents with muscle weakness and a pathognomonic cutaneous macular, violaceous, erythematous eruption. The juvenile variant is defined by onset prior to 16 years of age. Histologically, these entities are indistinguishable and demonstrate an interface dermatitis with epidermal atrophy. Clinically, JDM has a higher incidence of calcinosis cutis and is not associated with an increased risk for malignancy in contrast to the adult-onset variant.1 Panniculitis is a rare but serious complication in a subset of patients with DM and may represent a precursor to calcinosis cutis.2 We describe a case of JDM-associated panniculitis that was difficult to control with prednisone and rituximab.

A 21-year-old woman with fever, fatigue, muscle pain, and new-onset swelling of 2 weeks’ duration was admitted to the hospital. She had a 5-year history of intermittent muscle weakness and concomitant rash. Prior to presentation, she had been hospitalized twice for fever of unknown origin, and the source remained undetermined. Physical examination revealed prominent facial and periorbital edema. There was tender nonpitting edema present on all 4 extremities and hyperpigmented indurated nodules on the shins (Figure 1). A full laboratory and imaging workup was performed for autoantibodies and infectious etiologies. The complete blood cell count was notable for pancytopenia, and a thorough infectious workup was negative. Creatine kinase level was within reference range. A biopsy of the right shin was performed, and histopathology revealed a lobular panniculitis with fat necrosis and mixed inflammation with neutrophils with perieccrine involvement as well as an interface dermatitis (Figure 2). Periodic acid–Schiff, Grocott methenamine-silver, and Gram stains were negative. Myositis-specific antibody testing revealed anti-p155/140 autoantibodies, and magnetic resonance imaging did not reveal active myositis within the visualized muscles, consistent with stable nonprogressing DM. A diagnosis of JDM with panniculitis was made. The patient was started on oral prednisone. Subsequently, a trial of rituximab was initiated. Although the patient’s symptoms initially improved, the response was not sustained on rituximab, and the patient was continued on systemic steroids with initiation of cyclosporine.

Juvenile dermatomyositis is an autoimmune disorder with childhood onset that involves systemic inflammation of the muscles, skin, and internal organs. It often can present diagnostic and therapeutic challenges.2,3 Bohan and Peter4,5 clinical criteria may help identify potential patients with JDM, but magnetic resonance imaging, electromyography, and muscle biopsy often are required to confirm the diagnosis.6 Skin manifestations include heliotrope rash; V sign; shawl sign; Gottron papules; periorbital edema; and infrequently panniculitis, the subcutaneous inflammation of adipose tissue.3,7
Although panniculitis is found in approximately 10% of skin biopsies in patients with DM, our patient presented with anti-p155/140 antibodies.8-10 Fat involvement in these patients traditionally manifests as lipodystrophy. Panniculitis also may precede calcinosis cutis, a debilitating skin change that may occur in approximately 46% of patients with JDM and can cause severe morbidity.2,6,9
Subcutaneous edema rarely is described in DM-panniculitis, present in only 6% of 86 DM patients in one study.7 The pathophysiology of DM may be due to antibodies that target endothelial cells and activate complement, resulting in the membranolytic attack complex. This leads to microischemia, and microinfarction of the muscle fibers has been suggested to result in edema of the subcutaneous tissue in severe cases.7,11 Microinfarction has been found to be present 2.3 times more often in edematous DM compared with nonedematous DM.7 Subcutaneous edema may be an isolated presentation of DM that arises more quickly with severe disease activity. As such, recommendations have been made to consider edema in future classification schemes.7
Because of the severity of edematous and/or subcutaneous DM, aggressive therapy may be required. First-line therapy consists of corticosteroids with additional immunosuppressants and immunomodulatory agents if adequate response is not achieved.3,12 The effectiveness of rituximab in DM has been suggested.2,12,13 The Rituximab in Myositis (RIM) trial (N=200) was the first double-blind, placebo-controlled, phase 3 clinical trial to assess rituximab’s efficacy in refractory compared with early-onset inflammatory myopathies. Although outcomes were similar in both groups, 83% of patients overall, including the JDM subset, met the definition of improvement.12 In re-examining the RIM trial data and other cases using rituximab to treat inflammatory myopathies, an overall response rate of 78.3% was observed, with 52.1% of patients with DM reporting improvement in skin lesions (N=458, pooled from 48 studies).13 Further analysis of the RIM data revealed that panniculitis affected 10.4% of patients with JDM at baseline, which decreased to 6.8% at 36 weeks of rituximab therapy (N=48).12
As exhibited in our patient, subcutaneous tissue involvement, including calcinosis cutis and panniculitis, is seen more often in JDM than adult DM.2,6 However, panniculitis in anti-p155/140 patients is rare. Our patient also had antibody positivity, which likely predisposed her to a more severe course. Despite not having sustained improvement on rituximab, initiating aggressive therapy earlier in the disease course may be beneficial, and our patient continues with alternative therapies.
To the Editor:
Juvenile dermatomyositis (JDM) is an autoimmune disorder with childhood onset that predominantly affects the muscles and skin, among other organs. Since the recognition of dermatomyositis (DM) more than 100 years ago, a variety of clinical diagnostic criteria have been utilized. Classically, DM presents with muscle weakness and a pathognomonic cutaneous macular, violaceous, erythematous eruption. The juvenile variant is defined by onset prior to 16 years of age. Histologically, these entities are indistinguishable and demonstrate an interface dermatitis with epidermal atrophy. Clinically, JDM has a higher incidence of calcinosis cutis and is not associated with an increased risk for malignancy in contrast to the adult-onset variant.1 Panniculitis is a rare but serious complication in a subset of patients with DM and may represent a precursor to calcinosis cutis.2 We describe a case of JDM-associated panniculitis that was difficult to control with prednisone and rituximab.

A 21-year-old woman with fever, fatigue, muscle pain, and new-onset swelling of 2 weeks’ duration was admitted to the hospital. She had a 5-year history of intermittent muscle weakness and concomitant rash. Prior to presentation, she had been hospitalized twice for fever of unknown origin, and the source remained undetermined. Physical examination revealed prominent facial and periorbital edema. There was tender nonpitting edema present on all 4 extremities and hyperpigmented indurated nodules on the shins (Figure 1). A full laboratory and imaging workup was performed for autoantibodies and infectious etiologies. The complete blood cell count was notable for pancytopenia, and a thorough infectious workup was negative. Creatine kinase level was within reference range. A biopsy of the right shin was performed, and histopathology revealed a lobular panniculitis with fat necrosis and mixed inflammation with neutrophils with perieccrine involvement as well as an interface dermatitis (Figure 2). Periodic acid–Schiff, Grocott methenamine-silver, and Gram stains were negative. Myositis-specific antibody testing revealed anti-p155/140 autoantibodies, and magnetic resonance imaging did not reveal active myositis within the visualized muscles, consistent with stable nonprogressing DM. A diagnosis of JDM with panniculitis was made. The patient was started on oral prednisone. Subsequently, a trial of rituximab was initiated. Although the patient’s symptoms initially improved, the response was not sustained on rituximab, and the patient was continued on systemic steroids with initiation of cyclosporine.

Juvenile dermatomyositis is an autoimmune disorder with childhood onset that involves systemic inflammation of the muscles, skin, and internal organs. It often can present diagnostic and therapeutic challenges.2,3 Bohan and Peter4,5 clinical criteria may help identify potential patients with JDM, but magnetic resonance imaging, electromyography, and muscle biopsy often are required to confirm the diagnosis.6 Skin manifestations include heliotrope rash; V sign; shawl sign; Gottron papules; periorbital edema; and infrequently panniculitis, the subcutaneous inflammation of adipose tissue.3,7
Although panniculitis is found in approximately 10% of skin biopsies in patients with DM, our patient presented with anti-p155/140 antibodies.8-10 Fat involvement in these patients traditionally manifests as lipodystrophy. Panniculitis also may precede calcinosis cutis, a debilitating skin change that may occur in approximately 46% of patients with JDM and can cause severe morbidity.2,6,9
Subcutaneous edema rarely is described in DM-panniculitis, present in only 6% of 86 DM patients in one study.7 The pathophysiology of DM may be due to antibodies that target endothelial cells and activate complement, resulting in the membranolytic attack complex. This leads to microischemia, and microinfarction of the muscle fibers has been suggested to result in edema of the subcutaneous tissue in severe cases.7,11 Microinfarction has been found to be present 2.3 times more often in edematous DM compared with nonedematous DM.7 Subcutaneous edema may be an isolated presentation of DM that arises more quickly with severe disease activity. As such, recommendations have been made to consider edema in future classification schemes.7
Because of the severity of edematous and/or subcutaneous DM, aggressive therapy may be required. First-line therapy consists of corticosteroids with additional immunosuppressants and immunomodulatory agents if adequate response is not achieved.3,12 The effectiveness of rituximab in DM has been suggested.2,12,13 The Rituximab in Myositis (RIM) trial (N=200) was the first double-blind, placebo-controlled, phase 3 clinical trial to assess rituximab’s efficacy in refractory compared with early-onset inflammatory myopathies. Although outcomes were similar in both groups, 83% of patients overall, including the JDM subset, met the definition of improvement.12 In re-examining the RIM trial data and other cases using rituximab to treat inflammatory myopathies, an overall response rate of 78.3% was observed, with 52.1% of patients with DM reporting improvement in skin lesions (N=458, pooled from 48 studies).13 Further analysis of the RIM data revealed that panniculitis affected 10.4% of patients with JDM at baseline, which decreased to 6.8% at 36 weeks of rituximab therapy (N=48).12
As exhibited in our patient, subcutaneous tissue involvement, including calcinosis cutis and panniculitis, is seen more often in JDM than adult DM.2,6 However, panniculitis in anti-p155/140 patients is rare. Our patient also had antibody positivity, which likely predisposed her to a more severe course. Despite not having sustained improvement on rituximab, initiating aggressive therapy earlier in the disease course may be beneficial, and our patient continues with alternative therapies.
- Jorizzo JL, Vleugels RA. Dermatomyositis. In: Bolognia J, Schaffer J, Cerroni L. Dermatology. 4th ed. Elsevier; 2019:681-687.
- Aggarwal R, Loganathan P, Koontz D, et al. Cutaneous improvement in refractory adult and juvenile dermatomyositis after treatment with rituximab. Rheumatology. 2016;56:247-254.
- Santos-Briz A, Calle A, Linos K, et al. Dermatomyositis panniculitis: a clinicopathological and immunohistochemical study of 18 cases. J Eur Acad Dermatol Venereol. 2018;32:1352-1359.
- Bohan A, Peter JB. Polymyositis and dermatomyositis (first of two parts). N Engl J Med. 1975;292:344-347.
- Bohan A, Peter JB. Polymyositis and dermatomyositis (second of two parts). N Engl J Med. 1975;292:403-407.
- Sakurai N, Hino-Shishikura A, Nozawa T, et al. Clinical significance of subcutaneous fat and fascial involvement in juvenile dermatomyositis. Mod Rheumatol. 2019;29:808-813.
- Milisenda JC, Doti PI, Prieto-Gonzalez S, et al. Dermatomyositis presenting with severe subcutaneous edema: five additional cases and review of the literature. Semin Arthritis Rheum. 2014;44:228-233.
- Janis JF, Winkelmann RK. Histopathology of the skin in dermatomyositis: a histopathologic study of 55 cases. Arch Dermatol. 1968;97:640-650.
- van Dongen HM, van Vugt RM, Stoof TJ. Extensive persistent panniculitis in the context of dermatomyositis. J Clin Rheumatol. 2020;26:e187-e188.
- Gunawardena H, Wedderburn LR, North J, et al. Clinical associations of autoantibodies to a p155/140 kDa doublet protein in juvenile dermatomyositis. Rheumatology. 2008;47:324-328.
- Dalakas MC, Hohlfeld R. Polymyositis and dermatomyositis. Lancet. 2003;362:971-982.
- Oddis CV, Reed AM, Aggarwal R, et al. Rituximab in the treatment of refractory adult and juvenile dermatomyositis and adult polymyositis: a randomized, placebo-phase trial. Arthritis Rheum. 2013;65:314-324.
- Fasano S, Gordon P, Hajji R, et al. Rituximab in the treatment of inflammatory myopathies: a review. Rheumatology. 2016;56:26-36.
- Jorizzo JL, Vleugels RA. Dermatomyositis. In: Bolognia J, Schaffer J, Cerroni L. Dermatology. 4th ed. Elsevier; 2019:681-687.
- Aggarwal R, Loganathan P, Koontz D, et al. Cutaneous improvement in refractory adult and juvenile dermatomyositis after treatment with rituximab. Rheumatology. 2016;56:247-254.
- Santos-Briz A, Calle A, Linos K, et al. Dermatomyositis panniculitis: a clinicopathological and immunohistochemical study of 18 cases. J Eur Acad Dermatol Venereol. 2018;32:1352-1359.
- Bohan A, Peter JB. Polymyositis and dermatomyositis (first of two parts). N Engl J Med. 1975;292:344-347.
- Bohan A, Peter JB. Polymyositis and dermatomyositis (second of two parts). N Engl J Med. 1975;292:403-407.
- Sakurai N, Hino-Shishikura A, Nozawa T, et al. Clinical significance of subcutaneous fat and fascial involvement in juvenile dermatomyositis. Mod Rheumatol. 2019;29:808-813.
- Milisenda JC, Doti PI, Prieto-Gonzalez S, et al. Dermatomyositis presenting with severe subcutaneous edema: five additional cases and review of the literature. Semin Arthritis Rheum. 2014;44:228-233.
- Janis JF, Winkelmann RK. Histopathology of the skin in dermatomyositis: a histopathologic study of 55 cases. Arch Dermatol. 1968;97:640-650.
- van Dongen HM, van Vugt RM, Stoof TJ. Extensive persistent panniculitis in the context of dermatomyositis. J Clin Rheumatol. 2020;26:e187-e188.
- Gunawardena H, Wedderburn LR, North J, et al. Clinical associations of autoantibodies to a p155/140 kDa doublet protein in juvenile dermatomyositis. Rheumatology. 2008;47:324-328.
- Dalakas MC, Hohlfeld R. Polymyositis and dermatomyositis. Lancet. 2003;362:971-982.
- Oddis CV, Reed AM, Aggarwal R, et al. Rituximab in the treatment of refractory adult and juvenile dermatomyositis and adult polymyositis: a randomized, placebo-phase trial. Arthritis Rheum. 2013;65:314-324.
- Fasano S, Gordon P, Hajji R, et al. Rituximab in the treatment of inflammatory myopathies: a review. Rheumatology. 2016;56:26-36.
Practice Points
- Juvenile dermatomyositis is an autoimmune disorder with childhood onset that predominantly affects the muscles and skin.
- Juvenile dermatomyositis has a higher incidence of calcinosis cutis and is not associated with an increased risk for malignancy in contrast to the adult-onset variant, dermatomyositis (DM).
- Panniculitis is a rare but severe complication of DM, and this subset of DM may be challenging to treat, requiring aggressive therapy.