User login
When Should You Suspect Kawasaki Disease as the Cause of Fever in an Infant?
Case
A seven-week-old Hispanic female with a history of prematurity (born at 35 weeks by C-section) presents to the ED with four days of fever as high as 102°F and new-onset cyanotic spells. Cultures of blood, urine, and cerebrospinal fluid obtained 48 hours prior to admission were negative, but she continued to have intermittent fevers and developed a macular, non-pruritic rash on her hands and feet, with associated non-bilious emesis. One day prior to admission, she began to have episodes of apnea, with color change and cyanosis of her lips and eyelids. In the ED, her vital signs include a rectal temperature of 38.4°C, heart rate of 178/min, respiratory rate of 27/min, and blood pressure of 79/66. Examination reveals a non-toxic-appearing infant, with no conjunctival or oropharyngeal abnormalities, unremarkable heart and lung exam, and a blanching, erythematous macular rash on her hands, lower legs, and feet.
When should you suspect Kawasaki disease (KD) as the cause of fever in an infant?
Background
KD is an acute systemic vasculitis of unknown etiology that occurs in children. Affecting the small- and medium-sized arteries, with a striking predilection for coronary arteries, it is the leading cause of acquired pediatric heart disease in Japan and the U.S.1 Occurring predominantly in children younger than five years, KD has been diagnosed in infants and in young adults.2 The incidence of KD is lowest among white children and highest among Asians and Pacific Islanders, with the highest incidence in children of Japanese descent.
A recent epidemiologic study performed in Taiwan showed an incidence of 69 cases per 100,000 per year among children younger than five years, with a male/female ratio of 1.62:1.3 The peak of mortality occurs 15-45 days after onset of fever, although sudden cardiac death may occur many years later. Recurrence rate is approximately 3%. In the U.S., the estimated incidence ranges from nine to 18 per 100,000 children younger than five years per year.4
Review of Data
Because there is no specific diagnostic test or pathognomonic clinical feature, clinical diagnostic criteria have been established to guide physicians. KD diagnosis traditionally requires fever for at least five days and the presence of at least four of the following five principal features:
- bilateral conjunctival injection;
- changes in the mucous membranes of the upper respiratory tract (injected pharynx, infected, fissured lips, strawberry tongue);
- polymorphous rash;
- changes of the extremities (peripheral edema, erythema, periungual desquamation); and
- cervical lymphadenopathy.5
The fever, which is remittent, typically peaks at 39ºC to 40ºC. The mean duration of untreated fever is 11 days; with prompt treatment, fever typically subsides in two days. Bilateral painless non-exudative conjunctival injection begins shortly after onset of fever, involves typically bulbar conjunctiva, and is not associated with edema.
Erythematous rash usually appears within five days of onset of fever and is often a diffuse, nonspecific maculopapular eruption that is commonly pronounced in the perineal region. The appearance might be urticarial, micropustular, or erythema multiforme-like. Changes in extremities include erythema of palms and soles and tender induration of the hands and feet. Subsequently, desquamation begins in the periungual area within two to three weeks after the onset of fever. Typically, peeling begins around the nail folds of fingers, followed by the toes. The least common of the principal clinical features is tender unilateral anterior cervical lymphadenopathy (1.5 cm or greater in diameter).
When a patient presents with a history, examination, and laboratory findings consistent with KD without meeting the typical diagnostic standard, incomplete KD should be considered. The term “incomplete” is favored over “atypical” for this pre-sentation, because these patients are otherwise similar to other patients with KD. Patients with fever for five or fewer days and fewer than four principal features can be diagnosed as having KD when coronary artery disease is detected by two-dimensional echocardiography or coronary angiography (see Figure 1, p. 10). In the presence of four or more principal criteria, KD can be diagnosed before day four of the illness by an experienced clinician.6 Features less consistent with KD include the presence of exudative conjunctivitis, exudative pharyngitis, discrete intraoral lesions, bullous or vesicular rash, or generalized adenopathy.
If clinical features are consistent with KD, further risk stratification with erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) will determine whether patients are followed daily (if low) or if supplementary laboratory tests should be done (see Figure 1, p. 10). If three or more of supplementary laboratory criteria are present (albumin ≤3.0 g/dL, anemia for age, elevation of alanine aminotransferase (ALT), platelet count after seven days is 450 000/mm3 or greater, white blood cell count is 15,000/mm3 or greater, and urinary sediment containing 10 white blood cells/high-power field or more), echocardiogram should be performed and treatment initiated if abnormal.6
Young infants are more likely to manifest an incomplete presentation of KD, with a polymorphous rash being the most common symptom other than fever in this age group.7 Acute phase symptoms were also more likely to progress rapidly in this age group, with a higher risk of developing cardiac sequelae.8 As a result, any infant under the age of six months with fever for more than seven days and no other clear etiology should be evaluated for KD even in the absence of other diagnostic criteria.9
Other clinical manifestations of KD may include:
- Irritability: more notable in KD than in other febrile illnesses;
- Arthralgia and arthritis: may occur in the first week;
- Gastrointestinal complaints and findings: hepatomegaly, jaundice; and
- Abnormal chest X-ray findings: may be present in as many as 15% of patients.
Cardiovascular manifestations can be prominent in the acute phase of KD and are the leading cause of long-term morbidity and mortality. Coronary artery aneurysms occur in 20% of affected children with KD. Other cardiovascular complications include myocardial ischemia and ensuing depressed contractility and arrhythmias, as well as vascular obstruction in peripheral arteries.
A subset of KD patients develops hemodynamic instability requiring management in a critical care setting. This phenomenon has been named Kawasaki disease shock syndrome, where hemodynamic instability is not related to administration of intravenous immunoglobulin (IVIG). Patients are more likely to be female, to have laboratory findings consistent with greater inflammation, and to have impaired systolic and diastolic function. They also exhibit resistance to IVIG more often and have higher rates of coronary artery dilation and aneurysm formation.10
Differential diagnoses for KD may include viral infections, scarlet fever, staphylococcal scalded skin syndrome, toxic shock syndrome, Rocky Mountain spotted fever, cervical lymphadenitis, drug hypersensitivity, Stevens-Johnson syndrome, systemic idiopathic arthritis, leptospirosis, and mercury hypersensitivity reaction.11
Work-Up
Laboratory evaluation of a patient with suspected KD should include:
- Complete blood count (CBC) with differential: leukocytosis, anemia, thrombocytosis that peaks in the third week is characteristic. A manual differential may reveal an increase in band forms.
- Acute phase reactants: If C-reactive protein (CRP) is 3 mg/dL or greater and erythrocyte sedimentation rate (ESR) is 40 mm/hr or greater, supplementary laboratory work-up should be done. Make sure not to cloud classic with incomplete KD; the stepwise lab evaluation only pertains to the latter.
- Liver panel: Elevated ALT and gamma-glutamyl transferase (GGT), mild hyperbilirubinemia, or hypoalbuminemia may be present.
- Urinalysis: Sterile pyuria may be present; if present, it may be of urethral origin, and catheterized samples could miss this finding.12
Lack of elevated inflammatory markers (CRP is less than 3 mg/dl and ESR is less than 40 mm/hr) and the presence of two or three principal clinical features warrant ongoing daily monitoring of ESR, CRP, and fever until day seven of illness. If the fever resolves but is followed by peeling of extremities, an echocardiogram should be done. Lumbar puncture might help differentiate from CNS infectious etiologies, but about 50% of KD patients have a cerebrospinal fluid pleocytosis.
Echocardiography is the preferred imaging modality for the initial cardiovascular evaluation and follow-up.1 It has a sensitivity of 100% and specificity of 96% for the detection of proximal coronary aneurysms.13 Coronary aneurysms are clinically silent in most cases and can manifest with delayed complications, such as myocardial infarction or sudden death. Imaging plays an important role in the early diagnosis of these aneurysms and in estimating their number, size, and location, important elements in making a therapeutic decision.14
Although the echocardiography should be done as soon as KD is suspected, definitive treatment must not be delayed. Evaluation of all coronary artery segments, as well as cardiac contractility and presence of effusion, should be noted on echocardiography. In the absence of complications, echocardiography is performed at the time of diagnosis and at two weeks and six to eight weeks after disease onset.11
Treatment
Treatment goals for Kawasaki disease in the acute phase are reduction of systemic and coronary arterial inflammation and prevention of coronary thrombosis. The long-term therapy in individuals who develop coronary aneurysms is aimed at preventing myocardial ischemia or infarction.6 The current standard of care for the treatment of children in the U.S. is anti-inflammatory therapy with:
- immunoglobulin (IVIG) in a single 2 g/kg/dose infused over 10–12 hours, accompanied by;
- high-dose aspirin (80–100 mg/kg/day orally in four divided doses).6,15
IVIG administration within 10 days of the onset of fever results in more favorable outcomes. Live virus vaccines should be delayed to 11 months after administration of IVIG. Both aspirin and IVIG have anti-inflammatory effects. This regimen applies to patients without abnormalities on initial echocardiography. High-dose aspirin typically is continued for 48-72 hours after the child becomes afebrile. Thereafter, low-dose aspirin (3-5 mg/kg/day) is prescribed until patient shows no evidence of coronary changes, typically by six to eight weeks after onset of illness. Children with coronary abnormalities should continue aspirin indefinitely.
Approximately 10% of patients are IVIG-resistant and have persistent or recurrent fever for at least 36 hours after completion of the infusion. The current recommendation is to re-treat with IVIG at the same dose. If the patient has fever 36 hours after the second dose of IVIG, this is considered true treatment failure.
Other possible treatments for KD refractory to IVIG include IV methylprednisolone (30 mg/kg over two to three hours daily for three days) or infliximab.16 Even with prompt treatment, 5% of children who have KD develop coronary artery dilation, and 1% develop giant aneurysms.
Back to the Case
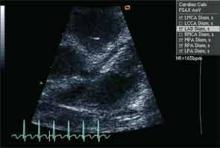
Initial laboratory evaluation revealed white blood cell count of 19.0×103 cells/mm3, hemoglobin of 8.9 gm/dL, CRP of 17.9 mg/dL, and ESR of 73 mm/hr. Because of persistent fevers for 48 hours after admission in the absence of another cause to explain the illness, the KD service was consulted. Echocardiography revealed dilatation of the left main (z-score 4.23) and proximal right (z-score 2.59), confirming the diagnosis of KD. Ejection fraction was read as qualitatively normal.
The infant received infliximab and IVIG, as well as high-dose aspirin, clopidogrel, and propranolol. This treatment regimen was directed by a KD expert and was more aggressive than typical therapy due to the severity of presentation. She received blood transfusions for worsening symptomatic anemia (hemoglobin 7.0 gm/dL) with hypoxia.
Following her IVIG infusion, she remained afebrile with progressive reduction in her CRP. She was discharged on hospital day seven on aspirin until her next follow-up, with propranolol for three days to limit potential tachycardia. At her three-week follow-up visit, her ESR had improved to 8 mm/hr. Her echocardiogram revealed a normal ejection fraction. Echocardiography revealed resolution of all abnormalities except for a borderline prominence of the right coronary artery (z-score 2.11). At this time it was recommended that her aspirin be discontinued.
She continues to be followed by the KD service as an outpatient and has done well without cardiovascular symptoms four months after her diagnosis.
Bottom Line
KD can manifest an incomplete presentation, especially in infants under the age of six months. Clinicians should maintain a high level of suspicion for KD in young infants with unexplained fevers lasting more than seven days.
Dr. Gurevich-Panigrahi is a fellow in pediatric hospital medicine at Cleveland Clinic Children’s Hospital. Dr. Kanegaye is a clinical professor of pediatrics at the University of California San Diego (UCSD) School of Medicine and attending physician in the emergency care center at Rady Children’s Hospital San Diego. Dr. Chang is associate clinical professor of pediatrics and medicine at UCSD School of Medicine, a pediatric hospitalist at Rady Children’s, and pediatric editor of The Hospitalist.
References
- Hendaoui L, Stanson AW, Habib Bouhaouala M, Joffre F, eds. Systemic Vasculitis: Imaging Features. New York: Springer; 2012.
- Manlhiot C, Yeung RS, Clarizia NA, Chahal N, McCrindle BW. Kawasaki disease at the extremes of the age spectrum. Pediatrics. 2009;124(3):e410-e415.
- Huang WC, Huang LM, Chang IS, et al. Epidemiologic features of Kawasaki disease in Taiwan, 2003-2006. Pediatrics. 2009;123(3):e401-405.
- Holman RC, Belay ED, Christensen KY, Folkema AM, Steiner CA, Schonberger LB. Hospitalizations for Kawasaki syndrome among children in the United States, 1997-2007. Pediatr Infect Dis J. 2010;29(6):483-438.
- Council on Cardiovascular Disease in the Young, Committee on Rheumatic Fever Endocarditis, Kawasaki Disease, American Heart Association. Diagnostic guidelines for Kawasaki disease. Circulation. 2001;103:335-336.
- Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 2004;110(17):2747-2771.
- Shiozawa Y, Inuzuka R, Harita Y, Kagawa J. Age-related differences in the course of the acute phase symptoms of Kawasaki disease. Pediatr Infect Dis J. 2013;32(9):e365-369.
- Genizi J, Miron D, Spiegel R, Fink D, Horowitz Y. Kawasaki disease in very young infants: high prevalence of atypical presentation and coronary arteritis. Clin Pediatr (Phila.). 2003;42(3):263-267.
- Sundel R. Incomplete (atypical) Kawasaki disease. UpToDate. Available at: http://www.uptodate.com/contents/incomplete-atypical-kawasaki-disease. Accessed June 9, 2014.
- Kanegaye JT, Wilder MS, Molkara D, et al. Recognition of a Kawasaki disease shock syndrome. Pediatrics. 2009;123(5):e783-e789.
- Fimbres AM, Shulman ST. Kawasaki disease. Pediatr Rev. 2008;29(9):308-315.
- Shike H, Kanegaye JT, Best BM, Pancheri J, Burns JC. Pyuria associated with acute Kawasaki disease and fever from other causes. Pediatr Infect Dis J. 2009;28(5):440-443.
- Capannari TE, Daniels SR, Meyer RA, Schwartz DC, Kaplan S. Sensitivity, specificity and predictive value of two-dimensional echocardiography in detecting coronary artery aneurysms in patients with Kawasaki disease. J Am Coll Cardiol. 1986;7(2):355-360.
- Mavrogeni S, Papadopoulos G, Karanasios E, Cokkinos DV. How to image Kawasaki disease: a validation of different imaging techniques. Int J Cardiol. 2008;124(1):27-31.
- Burns JC, Glodé MP. Kawasaki syndrome. Lancet. 2004;364(9433):533-544.
- Ghelani SJ, Pastor W, Parikh K. Demographic and treatment variability of refractory Kawasaki Disease: a multicenter analysis from 2005 to 2009. Hosp Pediatr. 2012;2(2):71-76.
Case
A seven-week-old Hispanic female with a history of prematurity (born at 35 weeks by C-section) presents to the ED with four days of fever as high as 102°F and new-onset cyanotic spells. Cultures of blood, urine, and cerebrospinal fluid obtained 48 hours prior to admission were negative, but she continued to have intermittent fevers and developed a macular, non-pruritic rash on her hands and feet, with associated non-bilious emesis. One day prior to admission, she began to have episodes of apnea, with color change and cyanosis of her lips and eyelids. In the ED, her vital signs include a rectal temperature of 38.4°C, heart rate of 178/min, respiratory rate of 27/min, and blood pressure of 79/66. Examination reveals a non-toxic-appearing infant, with no conjunctival or oropharyngeal abnormalities, unremarkable heart and lung exam, and a blanching, erythematous macular rash on her hands, lower legs, and feet.
When should you suspect Kawasaki disease (KD) as the cause of fever in an infant?
Background
KD is an acute systemic vasculitis of unknown etiology that occurs in children. Affecting the small- and medium-sized arteries, with a striking predilection for coronary arteries, it is the leading cause of acquired pediatric heart disease in Japan and the U.S.1 Occurring predominantly in children younger than five years, KD has been diagnosed in infants and in young adults.2 The incidence of KD is lowest among white children and highest among Asians and Pacific Islanders, with the highest incidence in children of Japanese descent.
A recent epidemiologic study performed in Taiwan showed an incidence of 69 cases per 100,000 per year among children younger than five years, with a male/female ratio of 1.62:1.3 The peak of mortality occurs 15-45 days after onset of fever, although sudden cardiac death may occur many years later. Recurrence rate is approximately 3%. In the U.S., the estimated incidence ranges from nine to 18 per 100,000 children younger than five years per year.4
Review of Data
Because there is no specific diagnostic test or pathognomonic clinical feature, clinical diagnostic criteria have been established to guide physicians. KD diagnosis traditionally requires fever for at least five days and the presence of at least four of the following five principal features:
- bilateral conjunctival injection;
- changes in the mucous membranes of the upper respiratory tract (injected pharynx, infected, fissured lips, strawberry tongue);
- polymorphous rash;
- changes of the extremities (peripheral edema, erythema, periungual desquamation); and
- cervical lymphadenopathy.5
The fever, which is remittent, typically peaks at 39ºC to 40ºC. The mean duration of untreated fever is 11 days; with prompt treatment, fever typically subsides in two days. Bilateral painless non-exudative conjunctival injection begins shortly after onset of fever, involves typically bulbar conjunctiva, and is not associated with edema.
Erythematous rash usually appears within five days of onset of fever and is often a diffuse, nonspecific maculopapular eruption that is commonly pronounced in the perineal region. The appearance might be urticarial, micropustular, or erythema multiforme-like. Changes in extremities include erythema of palms and soles and tender induration of the hands and feet. Subsequently, desquamation begins in the periungual area within two to three weeks after the onset of fever. Typically, peeling begins around the nail folds of fingers, followed by the toes. The least common of the principal clinical features is tender unilateral anterior cervical lymphadenopathy (1.5 cm or greater in diameter).
When a patient presents with a history, examination, and laboratory findings consistent with KD without meeting the typical diagnostic standard, incomplete KD should be considered. The term “incomplete” is favored over “atypical” for this pre-sentation, because these patients are otherwise similar to other patients with KD. Patients with fever for five or fewer days and fewer than four principal features can be diagnosed as having KD when coronary artery disease is detected by two-dimensional echocardiography or coronary angiography (see Figure 1, p. 10). In the presence of four or more principal criteria, KD can be diagnosed before day four of the illness by an experienced clinician.6 Features less consistent with KD include the presence of exudative conjunctivitis, exudative pharyngitis, discrete intraoral lesions, bullous or vesicular rash, or generalized adenopathy.
If clinical features are consistent with KD, further risk stratification with erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) will determine whether patients are followed daily (if low) or if supplementary laboratory tests should be done (see Figure 1, p. 10). If three or more of supplementary laboratory criteria are present (albumin ≤3.0 g/dL, anemia for age, elevation of alanine aminotransferase (ALT), platelet count after seven days is 450 000/mm3 or greater, white blood cell count is 15,000/mm3 or greater, and urinary sediment containing 10 white blood cells/high-power field or more), echocardiogram should be performed and treatment initiated if abnormal.6
Young infants are more likely to manifest an incomplete presentation of KD, with a polymorphous rash being the most common symptom other than fever in this age group.7 Acute phase symptoms were also more likely to progress rapidly in this age group, with a higher risk of developing cardiac sequelae.8 As a result, any infant under the age of six months with fever for more than seven days and no other clear etiology should be evaluated for KD even in the absence of other diagnostic criteria.9
Other clinical manifestations of KD may include:
- Irritability: more notable in KD than in other febrile illnesses;
- Arthralgia and arthritis: may occur in the first week;
- Gastrointestinal complaints and findings: hepatomegaly, jaundice; and
- Abnormal chest X-ray findings: may be present in as many as 15% of patients.
Cardiovascular manifestations can be prominent in the acute phase of KD and are the leading cause of long-term morbidity and mortality. Coronary artery aneurysms occur in 20% of affected children with KD. Other cardiovascular complications include myocardial ischemia and ensuing depressed contractility and arrhythmias, as well as vascular obstruction in peripheral arteries.
A subset of KD patients develops hemodynamic instability requiring management in a critical care setting. This phenomenon has been named Kawasaki disease shock syndrome, where hemodynamic instability is not related to administration of intravenous immunoglobulin (IVIG). Patients are more likely to be female, to have laboratory findings consistent with greater inflammation, and to have impaired systolic and diastolic function. They also exhibit resistance to IVIG more often and have higher rates of coronary artery dilation and aneurysm formation.10
Differential diagnoses for KD may include viral infections, scarlet fever, staphylococcal scalded skin syndrome, toxic shock syndrome, Rocky Mountain spotted fever, cervical lymphadenitis, drug hypersensitivity, Stevens-Johnson syndrome, systemic idiopathic arthritis, leptospirosis, and mercury hypersensitivity reaction.11
Work-Up
Laboratory evaluation of a patient with suspected KD should include:
- Complete blood count (CBC) with differential: leukocytosis, anemia, thrombocytosis that peaks in the third week is characteristic. A manual differential may reveal an increase in band forms.
- Acute phase reactants: If C-reactive protein (CRP) is 3 mg/dL or greater and erythrocyte sedimentation rate (ESR) is 40 mm/hr or greater, supplementary laboratory work-up should be done. Make sure not to cloud classic with incomplete KD; the stepwise lab evaluation only pertains to the latter.
- Liver panel: Elevated ALT and gamma-glutamyl transferase (GGT), mild hyperbilirubinemia, or hypoalbuminemia may be present.
- Urinalysis: Sterile pyuria may be present; if present, it may be of urethral origin, and catheterized samples could miss this finding.12
Lack of elevated inflammatory markers (CRP is less than 3 mg/dl and ESR is less than 40 mm/hr) and the presence of two or three principal clinical features warrant ongoing daily monitoring of ESR, CRP, and fever until day seven of illness. If the fever resolves but is followed by peeling of extremities, an echocardiogram should be done. Lumbar puncture might help differentiate from CNS infectious etiologies, but about 50% of KD patients have a cerebrospinal fluid pleocytosis.
Echocardiography is the preferred imaging modality for the initial cardiovascular evaluation and follow-up.1 It has a sensitivity of 100% and specificity of 96% for the detection of proximal coronary aneurysms.13 Coronary aneurysms are clinically silent in most cases and can manifest with delayed complications, such as myocardial infarction or sudden death. Imaging plays an important role in the early diagnosis of these aneurysms and in estimating their number, size, and location, important elements in making a therapeutic decision.14
Although the echocardiography should be done as soon as KD is suspected, definitive treatment must not be delayed. Evaluation of all coronary artery segments, as well as cardiac contractility and presence of effusion, should be noted on echocardiography. In the absence of complications, echocardiography is performed at the time of diagnosis and at two weeks and six to eight weeks after disease onset.11
Treatment
Treatment goals for Kawasaki disease in the acute phase are reduction of systemic and coronary arterial inflammation and prevention of coronary thrombosis. The long-term therapy in individuals who develop coronary aneurysms is aimed at preventing myocardial ischemia or infarction.6 The current standard of care for the treatment of children in the U.S. is anti-inflammatory therapy with:
- immunoglobulin (IVIG) in a single 2 g/kg/dose infused over 10–12 hours, accompanied by;
- high-dose aspirin (80–100 mg/kg/day orally in four divided doses).6,15
IVIG administration within 10 days of the onset of fever results in more favorable outcomes. Live virus vaccines should be delayed to 11 months after administration of IVIG. Both aspirin and IVIG have anti-inflammatory effects. This regimen applies to patients without abnormalities on initial echocardiography. High-dose aspirin typically is continued for 48-72 hours after the child becomes afebrile. Thereafter, low-dose aspirin (3-5 mg/kg/day) is prescribed until patient shows no evidence of coronary changes, typically by six to eight weeks after onset of illness. Children with coronary abnormalities should continue aspirin indefinitely.
Approximately 10% of patients are IVIG-resistant and have persistent or recurrent fever for at least 36 hours after completion of the infusion. The current recommendation is to re-treat with IVIG at the same dose. If the patient has fever 36 hours after the second dose of IVIG, this is considered true treatment failure.
Other possible treatments for KD refractory to IVIG include IV methylprednisolone (30 mg/kg over two to three hours daily for three days) or infliximab.16 Even with prompt treatment, 5% of children who have KD develop coronary artery dilation, and 1% develop giant aneurysms.
Back to the Case
Initial laboratory evaluation revealed white blood cell count of 19.0×103 cells/mm3, hemoglobin of 8.9 gm/dL, CRP of 17.9 mg/dL, and ESR of 73 mm/hr. Because of persistent fevers for 48 hours after admission in the absence of another cause to explain the illness, the KD service was consulted. Echocardiography revealed dilatation of the left main (z-score 4.23) and proximal right (z-score 2.59), confirming the diagnosis of KD. Ejection fraction was read as qualitatively normal.
The infant received infliximab and IVIG, as well as high-dose aspirin, clopidogrel, and propranolol. This treatment regimen was directed by a KD expert and was more aggressive than typical therapy due to the severity of presentation. She received blood transfusions for worsening symptomatic anemia (hemoglobin 7.0 gm/dL) with hypoxia.
Following her IVIG infusion, she remained afebrile with progressive reduction in her CRP. She was discharged on hospital day seven on aspirin until her next follow-up, with propranolol for three days to limit potential tachycardia. At her three-week follow-up visit, her ESR had improved to 8 mm/hr. Her echocardiogram revealed a normal ejection fraction. Echocardiography revealed resolution of all abnormalities except for a borderline prominence of the right coronary artery (z-score 2.11). At this time it was recommended that her aspirin be discontinued.
She continues to be followed by the KD service as an outpatient and has done well without cardiovascular symptoms four months after her diagnosis.
Bottom Line
KD can manifest an incomplete presentation, especially in infants under the age of six months. Clinicians should maintain a high level of suspicion for KD in young infants with unexplained fevers lasting more than seven days.
Dr. Gurevich-Panigrahi is a fellow in pediatric hospital medicine at Cleveland Clinic Children’s Hospital. Dr. Kanegaye is a clinical professor of pediatrics at the University of California San Diego (UCSD) School of Medicine and attending physician in the emergency care center at Rady Children’s Hospital San Diego. Dr. Chang is associate clinical professor of pediatrics and medicine at UCSD School of Medicine, a pediatric hospitalist at Rady Children’s, and pediatric editor of The Hospitalist.
References
- Hendaoui L, Stanson AW, Habib Bouhaouala M, Joffre F, eds. Systemic Vasculitis: Imaging Features. New York: Springer; 2012.
- Manlhiot C, Yeung RS, Clarizia NA, Chahal N, McCrindle BW. Kawasaki disease at the extremes of the age spectrum. Pediatrics. 2009;124(3):e410-e415.
- Huang WC, Huang LM, Chang IS, et al. Epidemiologic features of Kawasaki disease in Taiwan, 2003-2006. Pediatrics. 2009;123(3):e401-405.
- Holman RC, Belay ED, Christensen KY, Folkema AM, Steiner CA, Schonberger LB. Hospitalizations for Kawasaki syndrome among children in the United States, 1997-2007. Pediatr Infect Dis J. 2010;29(6):483-438.
- Council on Cardiovascular Disease in the Young, Committee on Rheumatic Fever Endocarditis, Kawasaki Disease, American Heart Association. Diagnostic guidelines for Kawasaki disease. Circulation. 2001;103:335-336.
- Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 2004;110(17):2747-2771.
- Shiozawa Y, Inuzuka R, Harita Y, Kagawa J. Age-related differences in the course of the acute phase symptoms of Kawasaki disease. Pediatr Infect Dis J. 2013;32(9):e365-369.
- Genizi J, Miron D, Spiegel R, Fink D, Horowitz Y. Kawasaki disease in very young infants: high prevalence of atypical presentation and coronary arteritis. Clin Pediatr (Phila.). 2003;42(3):263-267.
- Sundel R. Incomplete (atypical) Kawasaki disease. UpToDate. Available at: http://www.uptodate.com/contents/incomplete-atypical-kawasaki-disease. Accessed June 9, 2014.
- Kanegaye JT, Wilder MS, Molkara D, et al. Recognition of a Kawasaki disease shock syndrome. Pediatrics. 2009;123(5):e783-e789.
- Fimbres AM, Shulman ST. Kawasaki disease. Pediatr Rev. 2008;29(9):308-315.
- Shike H, Kanegaye JT, Best BM, Pancheri J, Burns JC. Pyuria associated with acute Kawasaki disease and fever from other causes. Pediatr Infect Dis J. 2009;28(5):440-443.
- Capannari TE, Daniels SR, Meyer RA, Schwartz DC, Kaplan S. Sensitivity, specificity and predictive value of two-dimensional echocardiography in detecting coronary artery aneurysms in patients with Kawasaki disease. J Am Coll Cardiol. 1986;7(2):355-360.
- Mavrogeni S, Papadopoulos G, Karanasios E, Cokkinos DV. How to image Kawasaki disease: a validation of different imaging techniques. Int J Cardiol. 2008;124(1):27-31.
- Burns JC, Glodé MP. Kawasaki syndrome. Lancet. 2004;364(9433):533-544.
- Ghelani SJ, Pastor W, Parikh K. Demographic and treatment variability of refractory Kawasaki Disease: a multicenter analysis from 2005 to 2009. Hosp Pediatr. 2012;2(2):71-76.
Case
A seven-week-old Hispanic female with a history of prematurity (born at 35 weeks by C-section) presents to the ED with four days of fever as high as 102°F and new-onset cyanotic spells. Cultures of blood, urine, and cerebrospinal fluid obtained 48 hours prior to admission were negative, but she continued to have intermittent fevers and developed a macular, non-pruritic rash on her hands and feet, with associated non-bilious emesis. One day prior to admission, she began to have episodes of apnea, with color change and cyanosis of her lips and eyelids. In the ED, her vital signs include a rectal temperature of 38.4°C, heart rate of 178/min, respiratory rate of 27/min, and blood pressure of 79/66. Examination reveals a non-toxic-appearing infant, with no conjunctival or oropharyngeal abnormalities, unremarkable heart and lung exam, and a blanching, erythematous macular rash on her hands, lower legs, and feet.
When should you suspect Kawasaki disease (KD) as the cause of fever in an infant?
Background
KD is an acute systemic vasculitis of unknown etiology that occurs in children. Affecting the small- and medium-sized arteries, with a striking predilection for coronary arteries, it is the leading cause of acquired pediatric heart disease in Japan and the U.S.1 Occurring predominantly in children younger than five years, KD has been diagnosed in infants and in young adults.2 The incidence of KD is lowest among white children and highest among Asians and Pacific Islanders, with the highest incidence in children of Japanese descent.
A recent epidemiologic study performed in Taiwan showed an incidence of 69 cases per 100,000 per year among children younger than five years, with a male/female ratio of 1.62:1.3 The peak of mortality occurs 15-45 days after onset of fever, although sudden cardiac death may occur many years later. Recurrence rate is approximately 3%. In the U.S., the estimated incidence ranges from nine to 18 per 100,000 children younger than five years per year.4
Review of Data
Because there is no specific diagnostic test or pathognomonic clinical feature, clinical diagnostic criteria have been established to guide physicians. KD diagnosis traditionally requires fever for at least five days and the presence of at least four of the following five principal features:
- bilateral conjunctival injection;
- changes in the mucous membranes of the upper respiratory tract (injected pharynx, infected, fissured lips, strawberry tongue);
- polymorphous rash;
- changes of the extremities (peripheral edema, erythema, periungual desquamation); and
- cervical lymphadenopathy.5
The fever, which is remittent, typically peaks at 39ºC to 40ºC. The mean duration of untreated fever is 11 days; with prompt treatment, fever typically subsides in two days. Bilateral painless non-exudative conjunctival injection begins shortly after onset of fever, involves typically bulbar conjunctiva, and is not associated with edema.
Erythematous rash usually appears within five days of onset of fever and is often a diffuse, nonspecific maculopapular eruption that is commonly pronounced in the perineal region. The appearance might be urticarial, micropustular, or erythema multiforme-like. Changes in extremities include erythema of palms and soles and tender induration of the hands and feet. Subsequently, desquamation begins in the periungual area within two to three weeks after the onset of fever. Typically, peeling begins around the nail folds of fingers, followed by the toes. The least common of the principal clinical features is tender unilateral anterior cervical lymphadenopathy (1.5 cm or greater in diameter).
When a patient presents with a history, examination, and laboratory findings consistent with KD without meeting the typical diagnostic standard, incomplete KD should be considered. The term “incomplete” is favored over “atypical” for this pre-sentation, because these patients are otherwise similar to other patients with KD. Patients with fever for five or fewer days and fewer than four principal features can be diagnosed as having KD when coronary artery disease is detected by two-dimensional echocardiography or coronary angiography (see Figure 1, p. 10). In the presence of four or more principal criteria, KD can be diagnosed before day four of the illness by an experienced clinician.6 Features less consistent with KD include the presence of exudative conjunctivitis, exudative pharyngitis, discrete intraoral lesions, bullous or vesicular rash, or generalized adenopathy.
If clinical features are consistent with KD, further risk stratification with erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) will determine whether patients are followed daily (if low) or if supplementary laboratory tests should be done (see Figure 1, p. 10). If three or more of supplementary laboratory criteria are present (albumin ≤3.0 g/dL, anemia for age, elevation of alanine aminotransferase (ALT), platelet count after seven days is 450 000/mm3 or greater, white blood cell count is 15,000/mm3 or greater, and urinary sediment containing 10 white blood cells/high-power field or more), echocardiogram should be performed and treatment initiated if abnormal.6
Young infants are more likely to manifest an incomplete presentation of KD, with a polymorphous rash being the most common symptom other than fever in this age group.7 Acute phase symptoms were also more likely to progress rapidly in this age group, with a higher risk of developing cardiac sequelae.8 As a result, any infant under the age of six months with fever for more than seven days and no other clear etiology should be evaluated for KD even in the absence of other diagnostic criteria.9
Other clinical manifestations of KD may include:
- Irritability: more notable in KD than in other febrile illnesses;
- Arthralgia and arthritis: may occur in the first week;
- Gastrointestinal complaints and findings: hepatomegaly, jaundice; and
- Abnormal chest X-ray findings: may be present in as many as 15% of patients.
Cardiovascular manifestations can be prominent in the acute phase of KD and are the leading cause of long-term morbidity and mortality. Coronary artery aneurysms occur in 20% of affected children with KD. Other cardiovascular complications include myocardial ischemia and ensuing depressed contractility and arrhythmias, as well as vascular obstruction in peripheral arteries.
A subset of KD patients develops hemodynamic instability requiring management in a critical care setting. This phenomenon has been named Kawasaki disease shock syndrome, where hemodynamic instability is not related to administration of intravenous immunoglobulin (IVIG). Patients are more likely to be female, to have laboratory findings consistent with greater inflammation, and to have impaired systolic and diastolic function. They also exhibit resistance to IVIG more often and have higher rates of coronary artery dilation and aneurysm formation.10
Differential diagnoses for KD may include viral infections, scarlet fever, staphylococcal scalded skin syndrome, toxic shock syndrome, Rocky Mountain spotted fever, cervical lymphadenitis, drug hypersensitivity, Stevens-Johnson syndrome, systemic idiopathic arthritis, leptospirosis, and mercury hypersensitivity reaction.11
Work-Up
Laboratory evaluation of a patient with suspected KD should include:
- Complete blood count (CBC) with differential: leukocytosis, anemia, thrombocytosis that peaks in the third week is characteristic. A manual differential may reveal an increase in band forms.
- Acute phase reactants: If C-reactive protein (CRP) is 3 mg/dL or greater and erythrocyte sedimentation rate (ESR) is 40 mm/hr or greater, supplementary laboratory work-up should be done. Make sure not to cloud classic with incomplete KD; the stepwise lab evaluation only pertains to the latter.
- Liver panel: Elevated ALT and gamma-glutamyl transferase (GGT), mild hyperbilirubinemia, or hypoalbuminemia may be present.
- Urinalysis: Sterile pyuria may be present; if present, it may be of urethral origin, and catheterized samples could miss this finding.12
Lack of elevated inflammatory markers (CRP is less than 3 mg/dl and ESR is less than 40 mm/hr) and the presence of two or three principal clinical features warrant ongoing daily monitoring of ESR, CRP, and fever until day seven of illness. If the fever resolves but is followed by peeling of extremities, an echocardiogram should be done. Lumbar puncture might help differentiate from CNS infectious etiologies, but about 50% of KD patients have a cerebrospinal fluid pleocytosis.
Echocardiography is the preferred imaging modality for the initial cardiovascular evaluation and follow-up.1 It has a sensitivity of 100% and specificity of 96% for the detection of proximal coronary aneurysms.13 Coronary aneurysms are clinically silent in most cases and can manifest with delayed complications, such as myocardial infarction or sudden death. Imaging plays an important role in the early diagnosis of these aneurysms and in estimating their number, size, and location, important elements in making a therapeutic decision.14
Although the echocardiography should be done as soon as KD is suspected, definitive treatment must not be delayed. Evaluation of all coronary artery segments, as well as cardiac contractility and presence of effusion, should be noted on echocardiography. In the absence of complications, echocardiography is performed at the time of diagnosis and at two weeks and six to eight weeks after disease onset.11
Treatment
Treatment goals for Kawasaki disease in the acute phase are reduction of systemic and coronary arterial inflammation and prevention of coronary thrombosis. The long-term therapy in individuals who develop coronary aneurysms is aimed at preventing myocardial ischemia or infarction.6 The current standard of care for the treatment of children in the U.S. is anti-inflammatory therapy with:
- immunoglobulin (IVIG) in a single 2 g/kg/dose infused over 10–12 hours, accompanied by;
- high-dose aspirin (80–100 mg/kg/day orally in four divided doses).6,15
IVIG administration within 10 days of the onset of fever results in more favorable outcomes. Live virus vaccines should be delayed to 11 months after administration of IVIG. Both aspirin and IVIG have anti-inflammatory effects. This regimen applies to patients without abnormalities on initial echocardiography. High-dose aspirin typically is continued for 48-72 hours after the child becomes afebrile. Thereafter, low-dose aspirin (3-5 mg/kg/day) is prescribed until patient shows no evidence of coronary changes, typically by six to eight weeks after onset of illness. Children with coronary abnormalities should continue aspirin indefinitely.
Approximately 10% of patients are IVIG-resistant and have persistent or recurrent fever for at least 36 hours after completion of the infusion. The current recommendation is to re-treat with IVIG at the same dose. If the patient has fever 36 hours after the second dose of IVIG, this is considered true treatment failure.
Other possible treatments for KD refractory to IVIG include IV methylprednisolone (30 mg/kg over two to three hours daily for three days) or infliximab.16 Even with prompt treatment, 5% of children who have KD develop coronary artery dilation, and 1% develop giant aneurysms.
Back to the Case
Initial laboratory evaluation revealed white blood cell count of 19.0×103 cells/mm3, hemoglobin of 8.9 gm/dL, CRP of 17.9 mg/dL, and ESR of 73 mm/hr. Because of persistent fevers for 48 hours after admission in the absence of another cause to explain the illness, the KD service was consulted. Echocardiography revealed dilatation of the left main (z-score 4.23) and proximal right (z-score 2.59), confirming the diagnosis of KD. Ejection fraction was read as qualitatively normal.
The infant received infliximab and IVIG, as well as high-dose aspirin, clopidogrel, and propranolol. This treatment regimen was directed by a KD expert and was more aggressive than typical therapy due to the severity of presentation. She received blood transfusions for worsening symptomatic anemia (hemoglobin 7.0 gm/dL) with hypoxia.
Following her IVIG infusion, she remained afebrile with progressive reduction in her CRP. She was discharged on hospital day seven on aspirin until her next follow-up, with propranolol for three days to limit potential tachycardia. At her three-week follow-up visit, her ESR had improved to 8 mm/hr. Her echocardiogram revealed a normal ejection fraction. Echocardiography revealed resolution of all abnormalities except for a borderline prominence of the right coronary artery (z-score 2.11). At this time it was recommended that her aspirin be discontinued.
She continues to be followed by the KD service as an outpatient and has done well without cardiovascular symptoms four months after her diagnosis.
Bottom Line
KD can manifest an incomplete presentation, especially in infants under the age of six months. Clinicians should maintain a high level of suspicion for KD in young infants with unexplained fevers lasting more than seven days.
Dr. Gurevich-Panigrahi is a fellow in pediatric hospital medicine at Cleveland Clinic Children’s Hospital. Dr. Kanegaye is a clinical professor of pediatrics at the University of California San Diego (UCSD) School of Medicine and attending physician in the emergency care center at Rady Children’s Hospital San Diego. Dr. Chang is associate clinical professor of pediatrics and medicine at UCSD School of Medicine, a pediatric hospitalist at Rady Children’s, and pediatric editor of The Hospitalist.
References
- Hendaoui L, Stanson AW, Habib Bouhaouala M, Joffre F, eds. Systemic Vasculitis: Imaging Features. New York: Springer; 2012.
- Manlhiot C, Yeung RS, Clarizia NA, Chahal N, McCrindle BW. Kawasaki disease at the extremes of the age spectrum. Pediatrics. 2009;124(3):e410-e415.
- Huang WC, Huang LM, Chang IS, et al. Epidemiologic features of Kawasaki disease in Taiwan, 2003-2006. Pediatrics. 2009;123(3):e401-405.
- Holman RC, Belay ED, Christensen KY, Folkema AM, Steiner CA, Schonberger LB. Hospitalizations for Kawasaki syndrome among children in the United States, 1997-2007. Pediatr Infect Dis J. 2010;29(6):483-438.
- Council on Cardiovascular Disease in the Young, Committee on Rheumatic Fever Endocarditis, Kawasaki Disease, American Heart Association. Diagnostic guidelines for Kawasaki disease. Circulation. 2001;103:335-336.
- Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 2004;110(17):2747-2771.
- Shiozawa Y, Inuzuka R, Harita Y, Kagawa J. Age-related differences in the course of the acute phase symptoms of Kawasaki disease. Pediatr Infect Dis J. 2013;32(9):e365-369.
- Genizi J, Miron D, Spiegel R, Fink D, Horowitz Y. Kawasaki disease in very young infants: high prevalence of atypical presentation and coronary arteritis. Clin Pediatr (Phila.). 2003;42(3):263-267.
- Sundel R. Incomplete (atypical) Kawasaki disease. UpToDate. Available at: http://www.uptodate.com/contents/incomplete-atypical-kawasaki-disease. Accessed June 9, 2014.
- Kanegaye JT, Wilder MS, Molkara D, et al. Recognition of a Kawasaki disease shock syndrome. Pediatrics. 2009;123(5):e783-e789.
- Fimbres AM, Shulman ST. Kawasaki disease. Pediatr Rev. 2008;29(9):308-315.
- Shike H, Kanegaye JT, Best BM, Pancheri J, Burns JC. Pyuria associated with acute Kawasaki disease and fever from other causes. Pediatr Infect Dis J. 2009;28(5):440-443.
- Capannari TE, Daniels SR, Meyer RA, Schwartz DC, Kaplan S. Sensitivity, specificity and predictive value of two-dimensional echocardiography in detecting coronary artery aneurysms in patients with Kawasaki disease. J Am Coll Cardiol. 1986;7(2):355-360.
- Mavrogeni S, Papadopoulos G, Karanasios E, Cokkinos DV. How to image Kawasaki disease: a validation of different imaging techniques. Int J Cardiol. 2008;124(1):27-31.
- Burns JC, Glodé MP. Kawasaki syndrome. Lancet. 2004;364(9433):533-544.
- Ghelani SJ, Pastor W, Parikh K. Demographic and treatment variability of refractory Kawasaki Disease: a multicenter analysis from 2005 to 2009. Hosp Pediatr. 2012;2(2):71-76.