User login
Management of the Biconcave (B2) Glenoid in Shoulder Arthroplasty: Technical Considerations
Total shoulder arthroplasty (TSA) has demonstrated excellent long-term clinical outcomes for the treatment of advanced glenohumeral osteoarthritis (OA).1-5 Glenohumeral OA is characterized by a broad spectrum of glenoid pathology. Both the morphology of the glenoid and humeral head subluxation are important preoperative factors to evaluate, as these have been shown to adversely impact shoulder arthroplasty outcomes.6,7
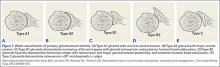
Walch and colleagues8 have previously classified glenoid morphology in cases of advanced glenohumeral arthritis based on the preoperative computed tomography (CT) scans of individuals undergoing shoulder arthroplasty (Figures 1A-1E). The biconcave (B2) glenoid is characterized by asymmetric posterior bone loss and a posterior translated humeral head that is seated in a biconcave glenoid. The degree and extent of bone loss in the B2 glenoid can be highly variable, ranging from the classic interpretation, in which 50% of the native glenoid fossa is preserved, to the more extreme case with little remaining native anterior glenoid. Scalise and colleagues9 have reported that determining the premorbid native glenoid version with a 3-dimensional (3D) glenoid vault model can aid in differentiating a pathologic B2 glenoid from a nonpathologic type C glenoid.
The B2 glenoid in particular has been associated with poor shoulder arthroplasty outcomes and component survivorship.6,10-12 There are many factors that are thought to contribute to this problem, such as glenoid component malposition, or undercorrection of the pathologic retroversion.6,13,14 Walch and colleagues10 reported that if the neoglenoid retroversion was greater than 27°, there was a 44% incidence of loosening and/or instability and 60% of the dislocations were observed when the humeral head subluxation was greater than 80%. Cases with severe posterior glenoid bone deficiency present a unique challenge to the surgeon, and the ability to accurately and securely place an implant in the correct anatomic position can be compromised. Standard TSA has proven excellent outcomes in the setting of typical glenohumeral OA, but in the B2 glenoid with significant posterior bone erosion, additional attention must be given to ensure adequate correction of the bony deformity, soft tissue balancing, and implant stability.
Several strategies that have been proposed to address extreme bone loss in the B2 glenoid will be discussed in this review. These include hemiarthroplasty, TSA with asymmetric reaming of the high side, TSA with bone grafting of the posterior glenoid bone loss, TSA with an augmented glenoid component, and reverse shoulder arthroplasty (RSA). Importantly, while these techniques have been proposed for managing the B2 glenoid, currently there is no gold standard consensus for the treatment of this condition. The purpose of this review is to highlight important characteristics of the B2 glenoid morphology on clinical outcomes and discuss the current surgical management options for this condition.
Preoperative Planning
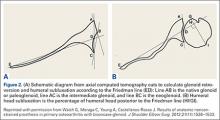
Being able to accurately determine the amount of retroversion is critical for preoperative planning. Friedman and colleagues15 initially described a method to measure glenoid retroversion; however, this is less accurate in B2 glenoids (Figures 2A, 2B). More recently, Rouleau and colleagues16 have validated and published methods to measure glenoid retroversion and subluxation in the B2 glenoid using 3 reference lines: the paleoglenoid (native glenoid surface), intermediate glenoid (line from anterior and posterior edge), and neoglenoid (eroded posterior surface) (Figure 2).
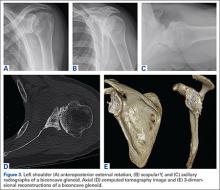
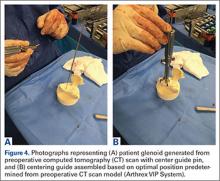
Preoperative evaluation starts with plain radiographs; however, additional imaging is needed, as the axillary view has shown to overestimate retroversion in 86% of patients (Figures 3A-3E).17 For a detailed evaluation of the glenoid retroversion and bone deficiency, CT scans with 3D reconstructions are useful.18,19 The surgical plan should be guided by the location and extent of glenoid bone loss. One tool that has been developed to help in predicting premorbid glenoid version, inclination, and position of the joint line is the 3D virtual glenoid vault model.9,20,21 This helps determine accurate premorbid glenoid anatomy and has been shown to assist in the selection of the optimal implant in an attempt to restore native glenoid anatomy, and avoid peg perforation.21 Patient-specific instrumentation (PSI) for shoulder arthroplasty is being used more frequently and has shown promise for more accurate glenoid component placement, particularly in the complex glenoid with severe bone deficiency. PSI involves creating a custom-fitted guide that is referenced to surface anatomy derived from the preoperative CT scan, which can then direct the surgeon toward optimal implant position with regard to glenoid component location, version and inclination (Figures 4A, 4B). Early reports show that PSI has resulted in a significant reduction in the frequency of malpositioned glenoid implants, with the greatest benefit observed in patients with retroversion in excess of 16°.22
Surgical Management
Hemiarthroplasty
Shoulder hemiarthroplasty has been traditionally described as an option for younger, more active patients in whom longevity of the glenoid component is a concern, or in patients with inadequate glenoid bone stock to tolerate a glenoid component. While there are no reports of hemiarthroplasty specifically for patients with B2 glenoids, one study has examined the effect of glenoid morphology on the outcomes of hemiarthroplasty for shoulder osteoarthritis. Levine and colleagues7 reported inferior clinical outcomes after shoulder hemiarthroplasty in patients with eccentric posterior glenoid wear. Several authors have advocated a “ream-and-run” technique to create a concentric glenoid and re-center the humeral head while still maintaining the native glenoid.23,24 However, in a recent series of 162 ream-and-run procedures, Gilmer and colleagues25 reported that only 23% of patients with B2 glenoid geometry achieved a minimal clinically important change in patient-reported outcome scores and 14% required revision. Furthermore, Lynch and colleagues26 found that progressive medial erosion and recurrent posterior glenoid erosion occur in a significant percentage of patients at early follow-up. Given these recent findings, the use of hemiarthroplasty alone or a ream-and-run procedure for patients with B2 glenoid morphology should be approached with caution.
Total Shoulder Arthroplasty
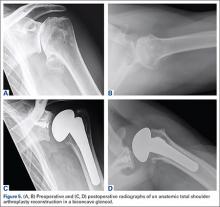
As with any TSA, the primary goals in treating patients with B2 glenoid defects are to provide the patient with a pain-free, stable, and functional shoulder (Figures 5A-5D). There are, however, a few challenges that are unique to TSA in the setting of B2 glenoid defects. Because the humeral head is often subluxated posteriorly into the defect, the anterior capsule and rotator cuff can tighten while the posterior aspect of the joint becomes lax. These soft tissues must be balanced during TSA in order to stabilize the shoulder and restore the appropriate length-tension relationship of the rotator cuff. The other primary concern is restoration of appropriate glenoid version and lateralization. To accomplish this, the most common techniques utilized are asymmetric reaming, bone graft augmentation, and glenoid component augmentation.27,28
Asymmetric Reaming. One of the more readily utilized techniques for addressing the B2 glenoid during TSA is eccentric or asymmetric reaming. During this process, the anterior glenoid is preferentially reamed while little to no bone is removed posteriorly. This technique is generally felt to be sufficient to treat posterior defects up to 5 mm to 8 mm or retroversion up to 15°.28 These upper limits have been confirmed in a number of cadaveric and simulated models.29-31
The success of this technique hinges on excellent glenoid exposure. With appropriate retractors in place, the anterior capsulolabral complex, including the biceps insertion, is resected to improve visualization. The inferior capsule must be resected carefully to ensure exposure and better motion postoperatively. On the other hand, it is imperative to protect the posterior capsulolabral attachments because of the increased risk of posterior instability in patients with B2 glenoids.
Detailed imaging such as CT scans with 3D reconstructions have improved our understanding of the degree of the deformities in all directions, which can better guide the reaming. PSI and planning software developed to improve the surgeon’s ability to place the glenoid component centrally in the best possible position after version correction can be even more helpful. We find that using a burr to provisionally lower the high side (anterior) provides a more en face view, which subsequently makes the eccentric reaming easier. As a guide, we will not ream more than 1 cm of anterior bone or attempt to correct more than ~20° of retroversion. The goal should be to create a glenoid surface that is more neutral and congruent to the posterior surface of the glenoid component while not overmedializing the component.
Although eccentric reaming may be one of the more straightforward methods for addressing posterior glenoid erosion, it is not without a number of potential downsides. When attempting to correct defects >10 mm or retroversion beyond 15°, excessive medialization of the implant can occur. Although increasing the thickness of the glenoid component can compensate for small amounts of medialization, excessive medialization can lead to a number of issues.27,28,32 As reaming progresses medially, the risk of keel penetration increases as the glenoid vault narrows.30,32 Further medialization decreases posterior cortical support for the implant, which increases the risk of component loosening and subsidence.33-35 The more medial the implant is placed, the smaller the surface of available bone for implant fixation. This often requires utilization of a smaller sized glenoid component that may result in component mismatch with the humeral implant. Finally, excessive medialization has the potential to under tension the rotator cuff, leading to decreased shoulder stability, strength, and function.
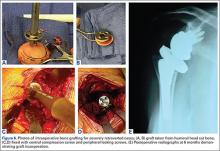
Bone Graft Augmentation. When posterior erosion becomes too excessive to address with eccentric reaming alone, defect augmentation is another option to consider (Figures 6A-6E). While technically more demanding, bone graft also provides the advantage of better re-creating the natural joint line and center of rotation of the glenohumeral joint.
For most defects, the resected humeral head provides the ideal source of graft. After initial reaming of the anterior glenoid, the defect must be sized and measured. We then recommend using a guided, cannulated system to place a central pin, lying perpendicular to the glenoid axis in neutral position. The anterior glenoid is then reamed enough to create a flat surface on which to attach the bone graft. The posterior surface is then gently burred to create a bleeding surface to enhance graft incorporation. The graft is then contoured to the defect and placed flush with the anterior glenoid. Cannulated screws are placed over guidewires to fix the graft. Using an arthroscopic cannula inserted posteriorly allows for easier placement of the guidewires and easier implantation of the screws. Although a reamer or burr can be used to contour the graft once it is fixed in place, this should be minimized to prevent loss of fixation. When the graft is fixed, we then cement the glenoid component into place.
Although good clinical results have been obtained with this technique, there is concern of incomplete graft healing and component loosening in the long term. Even in clinically asymptomatic and well functioning patients, some degree of radiographic lucency may be present in over 50% of cases.31,36,37 Glenoid Component Augmentation. To address the issues related to lucency and nonunion of bone graft augmentation, several augmented glenoid components have been developed. Augmented glenoid components have the benefit of filling posterior defects and stabilizing the shoulder without requiring excessive medialization (as often occurs with eccentric reaming) or union of a bone-to-bone interface (as is required in bone graft augmentation).38 Although many of the metal back designs experienced undesirably high failure rates and have since been recalled,39 more modern all-polyethylene components hold promise. The 2 most commonly utilized designs are the posterior step augment (DePuy) and the posterior wedge (Exactech). Although biomechanical analyses of both designs have demonstrated increased stability during loading in cadaveric and simulation models, the step augment (DePuy) has demonstrated increased stability and resistance to loosening.40,41 Although midterm results are not yet available for this newest generation of augmented components, short-term results with 2 to 3 years of follow-up have demonstrated excellent clinical outcomes.28
Reverse Total Shoulder Arthroplasty
While most commonly indicated for patients with rotator cuff tear arthropathy, RSA has recently been advocated for older patients with osteoarthritis and B2 glenoids in the setting of an intact rotator cuff. The semi-constrained design of the RSA is a potential solution to the static posterior humeral head subluxation seen in patients with B2 glenoid geometry (Figure 6E).
Technically, RSA is often an easier solution than a TSA with bone grafting because there is usually enough glenoid bone stock for fixation. That said, we always get a CT scan with 3D reconstructions to better appreciate the anatomy. Note that in B2 glenoids, the bone loss is typically posterior and inferior. RSA in the setting of a B2 glenoid is one of the ideal indications to use PSI to ensure ideal placement of the central pin, which is the key to glenoid baseplate positioning. Even when using a RSA, eccentric reaming and/or bone grafting allow for more ideal component placement. Using the same eccentric reaming techniques described above, one should try to ream to place the baseplate at 10° of retroversion. In cases where retroversion cannot be corrected to 10°, graft can be taken from the humeral head, iliac crest, or allograft. A benefit to using bone graft with RSA as opposed to TSA is that the graft can be fashioned to the baseplate, impacted/compressed into the B2 glenoid, and then secured with a central compression screw and peripheral locking screws.
Mizuno and colleagues41 reported a retrospective series of 27 RSAs performed for primary glenohumeral osteoarthritis and biconcave glenoid. At a mean follow-up of nearly 5 years, the authors noted significant improvement in Constant scores and shoulder motion with minimal complications. There was no recurrence of posterior instability observed by the time of final follow-up.41
RSA is a promising treatment for primary glenohumeral arthritis with posterior glenoid bone loss and static posterior subluxation in elderly or less active patients, but the longevity of these implants has yet to be established for younger, more active patients and requires further study.
Conclusion
Reconstruction of the B2 glenoid presents a challenging clinical problem that has been associated with poor clinical outcomes and implant survivorship. The high failure rate from glenoid component loosening and subsequent premature implant failure can be substantially decreased with accurate glenoid component positioning and appropriate correction of the pathologic glenoid retroversion. Careful preoperative planning is essential for accurate preparation and execution of the optimal surgical plan. There are many surgical strategies to address the B2 glenoid, but no consensus on the optimal method exists, as the technique should be uniquely customized to the individual’s pathology and surgeon preference (Table). Cases with mild deformity may be corrected with eccentric reaming and TSA, while the more severe deformities may require posterior glenoid bone grafting and/or augmented implants to restore native version. Finally, the RSA is a reliable option to restore stability and address bone deficiency for the severe B2 glenoid in an older, lower demand patient.
1. Barrett WP, Franklin JL, Jackins SE, Wyss CR, Matsen FA 3rd. Total shoulder arthroplasty. J Bone Joint Surg Am. 1987;69(6):865-872.
2. Bryant D, Litchfield R, Sandow M, Gartsman GM, Guyatt G, Kirkley A. A comparison of pain, strength, range of motion, and functional outcomes after hemiarthroplasty and total shoulder arthroplasty in patients with osteoarthritis of the shoulder. A systematic review and meta-analysis. J Bone Joint Surg Am. 2005;87(9):1947-1956.
3. Matsen FA 3rd. Early effectiveness of shoulder arthroplasty for patients who have primary glenohumeral degenerative joint disease. J Bone Joint Surg Am. 1996;78(2):260-264.
4. Fenlin JM Jr, Frieman BG. Indications, technique, and results of total shoulder arthroplasty in osteoarthritis. Orthop Clin North Am. 1998;29(3):423-434.
5. Singh JA, Sperling JW, Cofield RH. Revision surgery following total shoulder arthroplasty: Analysis of 2588 shoulders over three decades (1976 to 2008). J Bone Joint Surg Br. 2011;93(11):1513-1517.
6. Iannotti JP, Norris TR. Influence of preoperative factors on outcome of shoulder arthroplasty for glenohumeral osteoarthritis. J Bone Joint Surg Am. 2003;85-A(2):251-258.
7. Levine WN, Djurasovic M, Glasson JM, Pollock RG, Flatow EL, Bigliani LU. Hemiarthroplasty for glenohumeral osteoarthritis: Results correlated to degree of glenoid wear. J Shoulder Elbow Surg. 1997;6(5):449-454.
8. Walch G, Badet R, Boulahia A, Khoury A. Morphologic study of the glenoid in primary glenohumeral osteoarthritis. J Arthroplasty. 1999;14(6):756-760.
9. Scalise JJ, Codsi MJ, Bryan J, Iannotti JP. The three-dimensional glenoid vault model can estimate normal glenoid version in osteoarthritis. J Shoulder Elbow Surg. 2008;17(3):487-491.
10. Walch G, Moraga C, Young A, Castellanos-Rosas J. Results of anatomic nonconstrained prosthesis in primary osteoarthritis with biconcave glenoid. J Shoulder Elbow Surg. 2012;21(11):1526-1533.
11. Kany J, Katz D. How to deal with glenoid type B2 or C? How to prevent mistakes in implantation of glenoid component? Eur J Orthop Surg Traumatol. 2013;23(4):379-385.
12. Denard PJ, Walch G. Current concepts in the surgical management of primary glenohumeral arthritis with a biconcave glenoid. J Shoulder Elbow Surg. 2013;22(11):1589-1598.
13. Iannotti JP, Greeson C, Downing D, Sabesan V, Bryan JA. Effect of glenoid deformity on glenoid component placement in primary shoulder arthroplasty. J Shoulder Elbow Surg. 2012;21(1):48-55.
14. Ho JC, Sabesan VJ, Iannotti JP. Glenoid component retroversion is associated with osteolysis. J Bone Joint Surg Am. 2013;95(12):e82.
15. Friedman RJ, Hawthorne KB, Genez BM. The use of computerized tomography in the measurement of glenoid version. J Bone Joint Surg Am. 1992;74(7):1032-1037.
16. Rouleau DM, Kidder JF, Pons-Villanueva J, Dynamidis S, Defranco M, Walch G. Glenoid version: How to measure it? Validity of different methods in two-dimensional computed tomography scans. J Shoulder Elbow Surg. 2010;19(8):1230-1237.
17. Nyffeler RW, Jost B, Pfirrmann CW, Gerber C. Measurement of glenoid version: Conventional radiographs versus computed tomography scans. J Shoulder Elbow Surg. 2003;12(5):493-496.
18. Budge MD, Lewis GS, Schaefer E, Coquia S, Flemming DJ, Armstrong AD. Comparison of standard two-dimensional and three-dimensional corrected glenoid version measurements. J Shoulder Elbow Surg. 2011;20(4):577-583.
19. Bokor DJ, O’Sullivan MD, Hazan GJ. Variability of measurement of glenoid version on computed tomography scan. J Shoulder Elbow Surg. 1999;8(6):595-598.
20. Ganapathi A, McCarron JA, Chen X, Iannotti JP. Predicting normal glenoid version from the pathologic scapula: A comparison of 4 methods in 2- and 3-dimensional models. J Shoulder Elbow Surg. 2011;20(2):234-244.
21. Ricchetti ET, Hendel MD, Collins DN, Iannotti JP. Is premorbid glenoid anatomy altered in patients with glenohumeral osteoarthritis? Clin Orthop Relat Res. 2013;471(9):2932-2939.
22. Hendel MD, Bryan JA, Barsoum WK, et al. Comparison of patient-specific instruments with standard surgical instruments in determining glenoid component position: A randomized prospective clinical trial. J Bone Joint Surg Am. 2012;94(23):2167-2175.
23. Matsen FA 3rd, Warme WJ, Jackins SE. Can the ream and run procedure improve glenohumeral relationships and function for shoulders with the arthritic triad? Clin Orthop Relat Res. 2015;473(6):2088-2096.
24. Saltzman MD, Chamberlain AM, Mercer DM, Warme WJ, Bertelsen AL, Matsen FA 3rd. Shoulder hemiarthroplasty with concentric glenoid reaming in patients 55 years old or less. J Shoulder Elbow Surg. 2011;20(4):609-615.
25. Gilmer BB, Comstock BA, Jette JL, Warme WJ, Jackins SE, Matsen FA. The prognosis for improvement in comfort and function after the ream-and-run arthroplasty for glenohumeral arthritis: An analysis of 176 consecutive cases. J Bone Joint Surg Am. 2012;94(14):e102.
26. Lynch JR, Franta AK, Montgomery WH Jr, Lenters TR, Mounce D, Matsen FA 3rd. Self-assessed outcome at two to four years after shoulder hemiarthroplasty with concentric glenoid reaming. J Bone Joint Surg Am. 2007;89(6):1284-1292.
27. Donohue KW, Ricchetti ET, Iannotti JP. Surgical management of the biconcave (B2) glenoid. Curr Rev Musculoskelet Med. 2016;9(1):30-39.
28. Clavert P, Millett PJ, Warner JJ. Glenoid resurfacing: What are the limits to asymmetric reaming for posterior erosion? J Shoulder Elbow Surg. 2007;16(6):843-848.
29. Gillespie R, Lyons R, Lazarus M. Eccentric reaming in total shoulder arthroplasty: A cadaveric study. Orthopedics. 2009;32(1):21.
30. Neer CS 2nd, Morrison DS. Glenoid bone-grafting in total shoulder arthroplasty. J Bone Joint Surg Am. 1988;70(8):1154-1162.
31. Nowak DD, Bahu MJ, Gardner TR, et al. Simulation of surgical glenoid resurfacing using three-dimensional computed tomography of the arthritic glenohumeral joint: The amount of glenoid retroversion that can be corrected. J Shoulder Elbow Surg. 2009;18(5):680-688.
32. Strauss EJ, Roche C, Flurin PH, Wright T, Zuckerman JD. The glenoid in shoulder arthroplasty. J Shoulder Elbow Surg. 2009;18(5):819-833.
33. Walch G, Young AA, Boileau P, Loew M, Gazielly D, Mole D. Patterns of loosening of polyethylene keeled glenoid components after shoulder arthroplasty for primary osteoarthritis: Results of a multicenter study with more than five years of follow-up. J Bone Joint Surg Am. 2012;94(2):145-150.
34. Walch G, Young AA, Melis B, Gazielly D, Loew M, Boileau P. Results of a convex-back cemented keeled glenoid component in primary osteoarthritis: Multicenter study with a follow-up greater than 5 years. J Shoulder Elbow Surg. 2011;20(3):385-394.
35. Klika BJ, Wooten CW, Sperling JW, et al. Structural bone grafting for glenoid deficiency in primary total shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):1066-1072.
36. Sabesan V, Callanan M, Sharma V, Iannotti JP. Correction of acquired glenoid bone loss in osteoarthritis with a standard versus an augmented glenoid component. J Shoulder Elbow Surg. 2014;23(7):964-973.
37. Steinmann SP, Cofield RH. Bone grafting for glenoid deficiency in total shoulder replacement. J Shoulder Elbow Surg. 2000;9(5):361-367.
38. Cil A, Sperling JW, Cofield RH. Nonstandard glenoid components for bone deficiencies in shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):e149-e157.
39. Iannotti JP, Lappin KE, Klotz CL, Reber EW, Swope SW. Liftoff resistance of augmented glenoid components during cyclic fatigue loading in the posterior-superior direction. J Shoulder Elbow Surg. 2013;22(11):1530-1536.
40. Knowles NK, Ferreira LM, Athwal GS. Augmented glenoid component designs for type B2 erosions: A computational comparison by volume of bone removal and quality of remaining bone. J Shoulder Elbow Surg. 2015;24(8):1218-1226.
41. Mizuno N, Denard PJ, Raiss P, Walch G. Reverse total shoulder arthroplasty for primary glenohumeral osteoarthritis in patients with a biconcave glenoid. J Bone Joint Surg Am. 2013;95(14):1297-1304.
Total shoulder arthroplasty (TSA) has demonstrated excellent long-term clinical outcomes for the treatment of advanced glenohumeral osteoarthritis (OA).1-5 Glenohumeral OA is characterized by a broad spectrum of glenoid pathology. Both the morphology of the glenoid and humeral head subluxation are important preoperative factors to evaluate, as these have been shown to adversely impact shoulder arthroplasty outcomes.6,7
Walch and colleagues8 have previously classified glenoid morphology in cases of advanced glenohumeral arthritis based on the preoperative computed tomography (CT) scans of individuals undergoing shoulder arthroplasty (Figures 1A-1E). The biconcave (B2) glenoid is characterized by asymmetric posterior bone loss and a posterior translated humeral head that is seated in a biconcave glenoid. The degree and extent of bone loss in the B2 glenoid can be highly variable, ranging from the classic interpretation, in which 50% of the native glenoid fossa is preserved, to the more extreme case with little remaining native anterior glenoid. Scalise and colleagues9 have reported that determining the premorbid native glenoid version with a 3-dimensional (3D) glenoid vault model can aid in differentiating a pathologic B2 glenoid from a nonpathologic type C glenoid.
The B2 glenoid in particular has been associated with poor shoulder arthroplasty outcomes and component survivorship.6,10-12 There are many factors that are thought to contribute to this problem, such as glenoid component malposition, or undercorrection of the pathologic retroversion.6,13,14 Walch and colleagues10 reported that if the neoglenoid retroversion was greater than 27°, there was a 44% incidence of loosening and/or instability and 60% of the dislocations were observed when the humeral head subluxation was greater than 80%. Cases with severe posterior glenoid bone deficiency present a unique challenge to the surgeon, and the ability to accurately and securely place an implant in the correct anatomic position can be compromised. Standard TSA has proven excellent outcomes in the setting of typical glenohumeral OA, but in the B2 glenoid with significant posterior bone erosion, additional attention must be given to ensure adequate correction of the bony deformity, soft tissue balancing, and implant stability.
Several strategies that have been proposed to address extreme bone loss in the B2 glenoid will be discussed in this review. These include hemiarthroplasty, TSA with asymmetric reaming of the high side, TSA with bone grafting of the posterior glenoid bone loss, TSA with an augmented glenoid component, and reverse shoulder arthroplasty (RSA). Importantly, while these techniques have been proposed for managing the B2 glenoid, currently there is no gold standard consensus for the treatment of this condition. The purpose of this review is to highlight important characteristics of the B2 glenoid morphology on clinical outcomes and discuss the current surgical management options for this condition.
Preoperative Planning
Being able to accurately determine the amount of retroversion is critical for preoperative planning. Friedman and colleagues15 initially described a method to measure glenoid retroversion; however, this is less accurate in B2 glenoids (Figures 2A, 2B). More recently, Rouleau and colleagues16 have validated and published methods to measure glenoid retroversion and subluxation in the B2 glenoid using 3 reference lines: the paleoglenoid (native glenoid surface), intermediate glenoid (line from anterior and posterior edge), and neoglenoid (eroded posterior surface) (Figure 2).
Preoperative evaluation starts with plain radiographs; however, additional imaging is needed, as the axillary view has shown to overestimate retroversion in 86% of patients (Figures 3A-3E).17 For a detailed evaluation of the glenoid retroversion and bone deficiency, CT scans with 3D reconstructions are useful.18,19 The surgical plan should be guided by the location and extent of glenoid bone loss. One tool that has been developed to help in predicting premorbid glenoid version, inclination, and position of the joint line is the 3D virtual glenoid vault model.9,20,21 This helps determine accurate premorbid glenoid anatomy and has been shown to assist in the selection of the optimal implant in an attempt to restore native glenoid anatomy, and avoid peg perforation.21 Patient-specific instrumentation (PSI) for shoulder arthroplasty is being used more frequently and has shown promise for more accurate glenoid component placement, particularly in the complex glenoid with severe bone deficiency. PSI involves creating a custom-fitted guide that is referenced to surface anatomy derived from the preoperative CT scan, which can then direct the surgeon toward optimal implant position with regard to glenoid component location, version and inclination (Figures 4A, 4B). Early reports show that PSI has resulted in a significant reduction in the frequency of malpositioned glenoid implants, with the greatest benefit observed in patients with retroversion in excess of 16°.22
Surgical Management
Hemiarthroplasty
Shoulder hemiarthroplasty has been traditionally described as an option for younger, more active patients in whom longevity of the glenoid component is a concern, or in patients with inadequate glenoid bone stock to tolerate a glenoid component. While there are no reports of hemiarthroplasty specifically for patients with B2 glenoids, one study has examined the effect of glenoid morphology on the outcomes of hemiarthroplasty for shoulder osteoarthritis. Levine and colleagues7 reported inferior clinical outcomes after shoulder hemiarthroplasty in patients with eccentric posterior glenoid wear. Several authors have advocated a “ream-and-run” technique to create a concentric glenoid and re-center the humeral head while still maintaining the native glenoid.23,24 However, in a recent series of 162 ream-and-run procedures, Gilmer and colleagues25 reported that only 23% of patients with B2 glenoid geometry achieved a minimal clinically important change in patient-reported outcome scores and 14% required revision. Furthermore, Lynch and colleagues26 found that progressive medial erosion and recurrent posterior glenoid erosion occur in a significant percentage of patients at early follow-up. Given these recent findings, the use of hemiarthroplasty alone or a ream-and-run procedure for patients with B2 glenoid morphology should be approached with caution.
Total Shoulder Arthroplasty
As with any TSA, the primary goals in treating patients with B2 glenoid defects are to provide the patient with a pain-free, stable, and functional shoulder (Figures 5A-5D). There are, however, a few challenges that are unique to TSA in the setting of B2 glenoid defects. Because the humeral head is often subluxated posteriorly into the defect, the anterior capsule and rotator cuff can tighten while the posterior aspect of the joint becomes lax. These soft tissues must be balanced during TSA in order to stabilize the shoulder and restore the appropriate length-tension relationship of the rotator cuff. The other primary concern is restoration of appropriate glenoid version and lateralization. To accomplish this, the most common techniques utilized are asymmetric reaming, bone graft augmentation, and glenoid component augmentation.27,28
Asymmetric Reaming. One of the more readily utilized techniques for addressing the B2 glenoid during TSA is eccentric or asymmetric reaming. During this process, the anterior glenoid is preferentially reamed while little to no bone is removed posteriorly. This technique is generally felt to be sufficient to treat posterior defects up to 5 mm to 8 mm or retroversion up to 15°.28 These upper limits have been confirmed in a number of cadaveric and simulated models.29-31
The success of this technique hinges on excellent glenoid exposure. With appropriate retractors in place, the anterior capsulolabral complex, including the biceps insertion, is resected to improve visualization. The inferior capsule must be resected carefully to ensure exposure and better motion postoperatively. On the other hand, it is imperative to protect the posterior capsulolabral attachments because of the increased risk of posterior instability in patients with B2 glenoids.
Detailed imaging such as CT scans with 3D reconstructions have improved our understanding of the degree of the deformities in all directions, which can better guide the reaming. PSI and planning software developed to improve the surgeon’s ability to place the glenoid component centrally in the best possible position after version correction can be even more helpful. We find that using a burr to provisionally lower the high side (anterior) provides a more en face view, which subsequently makes the eccentric reaming easier. As a guide, we will not ream more than 1 cm of anterior bone or attempt to correct more than ~20° of retroversion. The goal should be to create a glenoid surface that is more neutral and congruent to the posterior surface of the glenoid component while not overmedializing the component.
Although eccentric reaming may be one of the more straightforward methods for addressing posterior glenoid erosion, it is not without a number of potential downsides. When attempting to correct defects >10 mm or retroversion beyond 15°, excessive medialization of the implant can occur. Although increasing the thickness of the glenoid component can compensate for small amounts of medialization, excessive medialization can lead to a number of issues.27,28,32 As reaming progresses medially, the risk of keel penetration increases as the glenoid vault narrows.30,32 Further medialization decreases posterior cortical support for the implant, which increases the risk of component loosening and subsidence.33-35 The more medial the implant is placed, the smaller the surface of available bone for implant fixation. This often requires utilization of a smaller sized glenoid component that may result in component mismatch with the humeral implant. Finally, excessive medialization has the potential to under tension the rotator cuff, leading to decreased shoulder stability, strength, and function.
Bone Graft Augmentation. When posterior erosion becomes too excessive to address with eccentric reaming alone, defect augmentation is another option to consider (Figures 6A-6E). While technically more demanding, bone graft also provides the advantage of better re-creating the natural joint line and center of rotation of the glenohumeral joint.
For most defects, the resected humeral head provides the ideal source of graft. After initial reaming of the anterior glenoid, the defect must be sized and measured. We then recommend using a guided, cannulated system to place a central pin, lying perpendicular to the glenoid axis in neutral position. The anterior glenoid is then reamed enough to create a flat surface on which to attach the bone graft. The posterior surface is then gently burred to create a bleeding surface to enhance graft incorporation. The graft is then contoured to the defect and placed flush with the anterior glenoid. Cannulated screws are placed over guidewires to fix the graft. Using an arthroscopic cannula inserted posteriorly allows for easier placement of the guidewires and easier implantation of the screws. Although a reamer or burr can be used to contour the graft once it is fixed in place, this should be minimized to prevent loss of fixation. When the graft is fixed, we then cement the glenoid component into place.
Although good clinical results have been obtained with this technique, there is concern of incomplete graft healing and component loosening in the long term. Even in clinically asymptomatic and well functioning patients, some degree of radiographic lucency may be present in over 50% of cases.31,36,37 Glenoid Component Augmentation. To address the issues related to lucency and nonunion of bone graft augmentation, several augmented glenoid components have been developed. Augmented glenoid components have the benefit of filling posterior defects and stabilizing the shoulder without requiring excessive medialization (as often occurs with eccentric reaming) or union of a bone-to-bone interface (as is required in bone graft augmentation).38 Although many of the metal back designs experienced undesirably high failure rates and have since been recalled,39 more modern all-polyethylene components hold promise. The 2 most commonly utilized designs are the posterior step augment (DePuy) and the posterior wedge (Exactech). Although biomechanical analyses of both designs have demonstrated increased stability during loading in cadaveric and simulation models, the step augment (DePuy) has demonstrated increased stability and resistance to loosening.40,41 Although midterm results are not yet available for this newest generation of augmented components, short-term results with 2 to 3 years of follow-up have demonstrated excellent clinical outcomes.28
Reverse Total Shoulder Arthroplasty
While most commonly indicated for patients with rotator cuff tear arthropathy, RSA has recently been advocated for older patients with osteoarthritis and B2 glenoids in the setting of an intact rotator cuff. The semi-constrained design of the RSA is a potential solution to the static posterior humeral head subluxation seen in patients with B2 glenoid geometry (Figure 6E).
Technically, RSA is often an easier solution than a TSA with bone grafting because there is usually enough glenoid bone stock for fixation. That said, we always get a CT scan with 3D reconstructions to better appreciate the anatomy. Note that in B2 glenoids, the bone loss is typically posterior and inferior. RSA in the setting of a B2 glenoid is one of the ideal indications to use PSI to ensure ideal placement of the central pin, which is the key to glenoid baseplate positioning. Even when using a RSA, eccentric reaming and/or bone grafting allow for more ideal component placement. Using the same eccentric reaming techniques described above, one should try to ream to place the baseplate at 10° of retroversion. In cases where retroversion cannot be corrected to 10°, graft can be taken from the humeral head, iliac crest, or allograft. A benefit to using bone graft with RSA as opposed to TSA is that the graft can be fashioned to the baseplate, impacted/compressed into the B2 glenoid, and then secured with a central compression screw and peripheral locking screws.
Mizuno and colleagues41 reported a retrospective series of 27 RSAs performed for primary glenohumeral osteoarthritis and biconcave glenoid. At a mean follow-up of nearly 5 years, the authors noted significant improvement in Constant scores and shoulder motion with minimal complications. There was no recurrence of posterior instability observed by the time of final follow-up.41
RSA is a promising treatment for primary glenohumeral arthritis with posterior glenoid bone loss and static posterior subluxation in elderly or less active patients, but the longevity of these implants has yet to be established for younger, more active patients and requires further study.
Conclusion
Reconstruction of the B2 glenoid presents a challenging clinical problem that has been associated with poor clinical outcomes and implant survivorship. The high failure rate from glenoid component loosening and subsequent premature implant failure can be substantially decreased with accurate glenoid component positioning and appropriate correction of the pathologic glenoid retroversion. Careful preoperative planning is essential for accurate preparation and execution of the optimal surgical plan. There are many surgical strategies to address the B2 glenoid, but no consensus on the optimal method exists, as the technique should be uniquely customized to the individual’s pathology and surgeon preference (Table). Cases with mild deformity may be corrected with eccentric reaming and TSA, while the more severe deformities may require posterior glenoid bone grafting and/or augmented implants to restore native version. Finally, the RSA is a reliable option to restore stability and address bone deficiency for the severe B2 glenoid in an older, lower demand patient.
Total shoulder arthroplasty (TSA) has demonstrated excellent long-term clinical outcomes for the treatment of advanced glenohumeral osteoarthritis (OA).1-5 Glenohumeral OA is characterized by a broad spectrum of glenoid pathology. Both the morphology of the glenoid and humeral head subluxation are important preoperative factors to evaluate, as these have been shown to adversely impact shoulder arthroplasty outcomes.6,7
Walch and colleagues8 have previously classified glenoid morphology in cases of advanced glenohumeral arthritis based on the preoperative computed tomography (CT) scans of individuals undergoing shoulder arthroplasty (Figures 1A-1E). The biconcave (B2) glenoid is characterized by asymmetric posterior bone loss and a posterior translated humeral head that is seated in a biconcave glenoid. The degree and extent of bone loss in the B2 glenoid can be highly variable, ranging from the classic interpretation, in which 50% of the native glenoid fossa is preserved, to the more extreme case with little remaining native anterior glenoid. Scalise and colleagues9 have reported that determining the premorbid native glenoid version with a 3-dimensional (3D) glenoid vault model can aid in differentiating a pathologic B2 glenoid from a nonpathologic type C glenoid.
The B2 glenoid in particular has been associated with poor shoulder arthroplasty outcomes and component survivorship.6,10-12 There are many factors that are thought to contribute to this problem, such as glenoid component malposition, or undercorrection of the pathologic retroversion.6,13,14 Walch and colleagues10 reported that if the neoglenoid retroversion was greater than 27°, there was a 44% incidence of loosening and/or instability and 60% of the dislocations were observed when the humeral head subluxation was greater than 80%. Cases with severe posterior glenoid bone deficiency present a unique challenge to the surgeon, and the ability to accurately and securely place an implant in the correct anatomic position can be compromised. Standard TSA has proven excellent outcomes in the setting of typical glenohumeral OA, but in the B2 glenoid with significant posterior bone erosion, additional attention must be given to ensure adequate correction of the bony deformity, soft tissue balancing, and implant stability.
Several strategies that have been proposed to address extreme bone loss in the B2 glenoid will be discussed in this review. These include hemiarthroplasty, TSA with asymmetric reaming of the high side, TSA with bone grafting of the posterior glenoid bone loss, TSA with an augmented glenoid component, and reverse shoulder arthroplasty (RSA). Importantly, while these techniques have been proposed for managing the B2 glenoid, currently there is no gold standard consensus for the treatment of this condition. The purpose of this review is to highlight important characteristics of the B2 glenoid morphology on clinical outcomes and discuss the current surgical management options for this condition.
Preoperative Planning
Being able to accurately determine the amount of retroversion is critical for preoperative planning. Friedman and colleagues15 initially described a method to measure glenoid retroversion; however, this is less accurate in B2 glenoids (Figures 2A, 2B). More recently, Rouleau and colleagues16 have validated and published methods to measure glenoid retroversion and subluxation in the B2 glenoid using 3 reference lines: the paleoglenoid (native glenoid surface), intermediate glenoid (line from anterior and posterior edge), and neoglenoid (eroded posterior surface) (Figure 2).
Preoperative evaluation starts with plain radiographs; however, additional imaging is needed, as the axillary view has shown to overestimate retroversion in 86% of patients (Figures 3A-3E).17 For a detailed evaluation of the glenoid retroversion and bone deficiency, CT scans with 3D reconstructions are useful.18,19 The surgical plan should be guided by the location and extent of glenoid bone loss. One tool that has been developed to help in predicting premorbid glenoid version, inclination, and position of the joint line is the 3D virtual glenoid vault model.9,20,21 This helps determine accurate premorbid glenoid anatomy and has been shown to assist in the selection of the optimal implant in an attempt to restore native glenoid anatomy, and avoid peg perforation.21 Patient-specific instrumentation (PSI) for shoulder arthroplasty is being used more frequently and has shown promise for more accurate glenoid component placement, particularly in the complex glenoid with severe bone deficiency. PSI involves creating a custom-fitted guide that is referenced to surface anatomy derived from the preoperative CT scan, which can then direct the surgeon toward optimal implant position with regard to glenoid component location, version and inclination (Figures 4A, 4B). Early reports show that PSI has resulted in a significant reduction in the frequency of malpositioned glenoid implants, with the greatest benefit observed in patients with retroversion in excess of 16°.22
Surgical Management
Hemiarthroplasty
Shoulder hemiarthroplasty has been traditionally described as an option for younger, more active patients in whom longevity of the glenoid component is a concern, or in patients with inadequate glenoid bone stock to tolerate a glenoid component. While there are no reports of hemiarthroplasty specifically for patients with B2 glenoids, one study has examined the effect of glenoid morphology on the outcomes of hemiarthroplasty for shoulder osteoarthritis. Levine and colleagues7 reported inferior clinical outcomes after shoulder hemiarthroplasty in patients with eccentric posterior glenoid wear. Several authors have advocated a “ream-and-run” technique to create a concentric glenoid and re-center the humeral head while still maintaining the native glenoid.23,24 However, in a recent series of 162 ream-and-run procedures, Gilmer and colleagues25 reported that only 23% of patients with B2 glenoid geometry achieved a minimal clinically important change in patient-reported outcome scores and 14% required revision. Furthermore, Lynch and colleagues26 found that progressive medial erosion and recurrent posterior glenoid erosion occur in a significant percentage of patients at early follow-up. Given these recent findings, the use of hemiarthroplasty alone or a ream-and-run procedure for patients with B2 glenoid morphology should be approached with caution.
Total Shoulder Arthroplasty
As with any TSA, the primary goals in treating patients with B2 glenoid defects are to provide the patient with a pain-free, stable, and functional shoulder (Figures 5A-5D). There are, however, a few challenges that are unique to TSA in the setting of B2 glenoid defects. Because the humeral head is often subluxated posteriorly into the defect, the anterior capsule and rotator cuff can tighten while the posterior aspect of the joint becomes lax. These soft tissues must be balanced during TSA in order to stabilize the shoulder and restore the appropriate length-tension relationship of the rotator cuff. The other primary concern is restoration of appropriate glenoid version and lateralization. To accomplish this, the most common techniques utilized are asymmetric reaming, bone graft augmentation, and glenoid component augmentation.27,28
Asymmetric Reaming. One of the more readily utilized techniques for addressing the B2 glenoid during TSA is eccentric or asymmetric reaming. During this process, the anterior glenoid is preferentially reamed while little to no bone is removed posteriorly. This technique is generally felt to be sufficient to treat posterior defects up to 5 mm to 8 mm or retroversion up to 15°.28 These upper limits have been confirmed in a number of cadaveric and simulated models.29-31
The success of this technique hinges on excellent glenoid exposure. With appropriate retractors in place, the anterior capsulolabral complex, including the biceps insertion, is resected to improve visualization. The inferior capsule must be resected carefully to ensure exposure and better motion postoperatively. On the other hand, it is imperative to protect the posterior capsulolabral attachments because of the increased risk of posterior instability in patients with B2 glenoids.
Detailed imaging such as CT scans with 3D reconstructions have improved our understanding of the degree of the deformities in all directions, which can better guide the reaming. PSI and planning software developed to improve the surgeon’s ability to place the glenoid component centrally in the best possible position after version correction can be even more helpful. We find that using a burr to provisionally lower the high side (anterior) provides a more en face view, which subsequently makes the eccentric reaming easier. As a guide, we will not ream more than 1 cm of anterior bone or attempt to correct more than ~20° of retroversion. The goal should be to create a glenoid surface that is more neutral and congruent to the posterior surface of the glenoid component while not overmedializing the component.
Although eccentric reaming may be one of the more straightforward methods for addressing posterior glenoid erosion, it is not without a number of potential downsides. When attempting to correct defects >10 mm or retroversion beyond 15°, excessive medialization of the implant can occur. Although increasing the thickness of the glenoid component can compensate for small amounts of medialization, excessive medialization can lead to a number of issues.27,28,32 As reaming progresses medially, the risk of keel penetration increases as the glenoid vault narrows.30,32 Further medialization decreases posterior cortical support for the implant, which increases the risk of component loosening and subsidence.33-35 The more medial the implant is placed, the smaller the surface of available bone for implant fixation. This often requires utilization of a smaller sized glenoid component that may result in component mismatch with the humeral implant. Finally, excessive medialization has the potential to under tension the rotator cuff, leading to decreased shoulder stability, strength, and function.
Bone Graft Augmentation. When posterior erosion becomes too excessive to address with eccentric reaming alone, defect augmentation is another option to consider (Figures 6A-6E). While technically more demanding, bone graft also provides the advantage of better re-creating the natural joint line and center of rotation of the glenohumeral joint.
For most defects, the resected humeral head provides the ideal source of graft. After initial reaming of the anterior glenoid, the defect must be sized and measured. We then recommend using a guided, cannulated system to place a central pin, lying perpendicular to the glenoid axis in neutral position. The anterior glenoid is then reamed enough to create a flat surface on which to attach the bone graft. The posterior surface is then gently burred to create a bleeding surface to enhance graft incorporation. The graft is then contoured to the defect and placed flush with the anterior glenoid. Cannulated screws are placed over guidewires to fix the graft. Using an arthroscopic cannula inserted posteriorly allows for easier placement of the guidewires and easier implantation of the screws. Although a reamer or burr can be used to contour the graft once it is fixed in place, this should be minimized to prevent loss of fixation. When the graft is fixed, we then cement the glenoid component into place.
Although good clinical results have been obtained with this technique, there is concern of incomplete graft healing and component loosening in the long term. Even in clinically asymptomatic and well functioning patients, some degree of radiographic lucency may be present in over 50% of cases.31,36,37 Glenoid Component Augmentation. To address the issues related to lucency and nonunion of bone graft augmentation, several augmented glenoid components have been developed. Augmented glenoid components have the benefit of filling posterior defects and stabilizing the shoulder without requiring excessive medialization (as often occurs with eccentric reaming) or union of a bone-to-bone interface (as is required in bone graft augmentation).38 Although many of the metal back designs experienced undesirably high failure rates and have since been recalled,39 more modern all-polyethylene components hold promise. The 2 most commonly utilized designs are the posterior step augment (DePuy) and the posterior wedge (Exactech). Although biomechanical analyses of both designs have demonstrated increased stability during loading in cadaveric and simulation models, the step augment (DePuy) has demonstrated increased stability and resistance to loosening.40,41 Although midterm results are not yet available for this newest generation of augmented components, short-term results with 2 to 3 years of follow-up have demonstrated excellent clinical outcomes.28
Reverse Total Shoulder Arthroplasty
While most commonly indicated for patients with rotator cuff tear arthropathy, RSA has recently been advocated for older patients with osteoarthritis and B2 glenoids in the setting of an intact rotator cuff. The semi-constrained design of the RSA is a potential solution to the static posterior humeral head subluxation seen in patients with B2 glenoid geometry (Figure 6E).
Technically, RSA is often an easier solution than a TSA with bone grafting because there is usually enough glenoid bone stock for fixation. That said, we always get a CT scan with 3D reconstructions to better appreciate the anatomy. Note that in B2 glenoids, the bone loss is typically posterior and inferior. RSA in the setting of a B2 glenoid is one of the ideal indications to use PSI to ensure ideal placement of the central pin, which is the key to glenoid baseplate positioning. Even when using a RSA, eccentric reaming and/or bone grafting allow for more ideal component placement. Using the same eccentric reaming techniques described above, one should try to ream to place the baseplate at 10° of retroversion. In cases where retroversion cannot be corrected to 10°, graft can be taken from the humeral head, iliac crest, or allograft. A benefit to using bone graft with RSA as opposed to TSA is that the graft can be fashioned to the baseplate, impacted/compressed into the B2 glenoid, and then secured with a central compression screw and peripheral locking screws.
Mizuno and colleagues41 reported a retrospective series of 27 RSAs performed for primary glenohumeral osteoarthritis and biconcave glenoid. At a mean follow-up of nearly 5 years, the authors noted significant improvement in Constant scores and shoulder motion with minimal complications. There was no recurrence of posterior instability observed by the time of final follow-up.41
RSA is a promising treatment for primary glenohumeral arthritis with posterior glenoid bone loss and static posterior subluxation in elderly or less active patients, but the longevity of these implants has yet to be established for younger, more active patients and requires further study.
Conclusion
Reconstruction of the B2 glenoid presents a challenging clinical problem that has been associated with poor clinical outcomes and implant survivorship. The high failure rate from glenoid component loosening and subsequent premature implant failure can be substantially decreased with accurate glenoid component positioning and appropriate correction of the pathologic glenoid retroversion. Careful preoperative planning is essential for accurate preparation and execution of the optimal surgical plan. There are many surgical strategies to address the B2 glenoid, but no consensus on the optimal method exists, as the technique should be uniquely customized to the individual’s pathology and surgeon preference (Table). Cases with mild deformity may be corrected with eccentric reaming and TSA, while the more severe deformities may require posterior glenoid bone grafting and/or augmented implants to restore native version. Finally, the RSA is a reliable option to restore stability and address bone deficiency for the severe B2 glenoid in an older, lower demand patient.
1. Barrett WP, Franklin JL, Jackins SE, Wyss CR, Matsen FA 3rd. Total shoulder arthroplasty. J Bone Joint Surg Am. 1987;69(6):865-872.
2. Bryant D, Litchfield R, Sandow M, Gartsman GM, Guyatt G, Kirkley A. A comparison of pain, strength, range of motion, and functional outcomes after hemiarthroplasty and total shoulder arthroplasty in patients with osteoarthritis of the shoulder. A systematic review and meta-analysis. J Bone Joint Surg Am. 2005;87(9):1947-1956.
3. Matsen FA 3rd. Early effectiveness of shoulder arthroplasty for patients who have primary glenohumeral degenerative joint disease. J Bone Joint Surg Am. 1996;78(2):260-264.
4. Fenlin JM Jr, Frieman BG. Indications, technique, and results of total shoulder arthroplasty in osteoarthritis. Orthop Clin North Am. 1998;29(3):423-434.
5. Singh JA, Sperling JW, Cofield RH. Revision surgery following total shoulder arthroplasty: Analysis of 2588 shoulders over three decades (1976 to 2008). J Bone Joint Surg Br. 2011;93(11):1513-1517.
6. Iannotti JP, Norris TR. Influence of preoperative factors on outcome of shoulder arthroplasty for glenohumeral osteoarthritis. J Bone Joint Surg Am. 2003;85-A(2):251-258.
7. Levine WN, Djurasovic M, Glasson JM, Pollock RG, Flatow EL, Bigliani LU. Hemiarthroplasty for glenohumeral osteoarthritis: Results correlated to degree of glenoid wear. J Shoulder Elbow Surg. 1997;6(5):449-454.
8. Walch G, Badet R, Boulahia A, Khoury A. Morphologic study of the glenoid in primary glenohumeral osteoarthritis. J Arthroplasty. 1999;14(6):756-760.
9. Scalise JJ, Codsi MJ, Bryan J, Iannotti JP. The three-dimensional glenoid vault model can estimate normal glenoid version in osteoarthritis. J Shoulder Elbow Surg. 2008;17(3):487-491.
10. Walch G, Moraga C, Young A, Castellanos-Rosas J. Results of anatomic nonconstrained prosthesis in primary osteoarthritis with biconcave glenoid. J Shoulder Elbow Surg. 2012;21(11):1526-1533.
11. Kany J, Katz D. How to deal with glenoid type B2 or C? How to prevent mistakes in implantation of glenoid component? Eur J Orthop Surg Traumatol. 2013;23(4):379-385.
12. Denard PJ, Walch G. Current concepts in the surgical management of primary glenohumeral arthritis with a biconcave glenoid. J Shoulder Elbow Surg. 2013;22(11):1589-1598.
13. Iannotti JP, Greeson C, Downing D, Sabesan V, Bryan JA. Effect of glenoid deformity on glenoid component placement in primary shoulder arthroplasty. J Shoulder Elbow Surg. 2012;21(1):48-55.
14. Ho JC, Sabesan VJ, Iannotti JP. Glenoid component retroversion is associated with osteolysis. J Bone Joint Surg Am. 2013;95(12):e82.
15. Friedman RJ, Hawthorne KB, Genez BM. The use of computerized tomography in the measurement of glenoid version. J Bone Joint Surg Am. 1992;74(7):1032-1037.
16. Rouleau DM, Kidder JF, Pons-Villanueva J, Dynamidis S, Defranco M, Walch G. Glenoid version: How to measure it? Validity of different methods in two-dimensional computed tomography scans. J Shoulder Elbow Surg. 2010;19(8):1230-1237.
17. Nyffeler RW, Jost B, Pfirrmann CW, Gerber C. Measurement of glenoid version: Conventional radiographs versus computed tomography scans. J Shoulder Elbow Surg. 2003;12(5):493-496.
18. Budge MD, Lewis GS, Schaefer E, Coquia S, Flemming DJ, Armstrong AD. Comparison of standard two-dimensional and three-dimensional corrected glenoid version measurements. J Shoulder Elbow Surg. 2011;20(4):577-583.
19. Bokor DJ, O’Sullivan MD, Hazan GJ. Variability of measurement of glenoid version on computed tomography scan. J Shoulder Elbow Surg. 1999;8(6):595-598.
20. Ganapathi A, McCarron JA, Chen X, Iannotti JP. Predicting normal glenoid version from the pathologic scapula: A comparison of 4 methods in 2- and 3-dimensional models. J Shoulder Elbow Surg. 2011;20(2):234-244.
21. Ricchetti ET, Hendel MD, Collins DN, Iannotti JP. Is premorbid glenoid anatomy altered in patients with glenohumeral osteoarthritis? Clin Orthop Relat Res. 2013;471(9):2932-2939.
22. Hendel MD, Bryan JA, Barsoum WK, et al. Comparison of patient-specific instruments with standard surgical instruments in determining glenoid component position: A randomized prospective clinical trial. J Bone Joint Surg Am. 2012;94(23):2167-2175.
23. Matsen FA 3rd, Warme WJ, Jackins SE. Can the ream and run procedure improve glenohumeral relationships and function for shoulders with the arthritic triad? Clin Orthop Relat Res. 2015;473(6):2088-2096.
24. Saltzman MD, Chamberlain AM, Mercer DM, Warme WJ, Bertelsen AL, Matsen FA 3rd. Shoulder hemiarthroplasty with concentric glenoid reaming in patients 55 years old or less. J Shoulder Elbow Surg. 2011;20(4):609-615.
25. Gilmer BB, Comstock BA, Jette JL, Warme WJ, Jackins SE, Matsen FA. The prognosis for improvement in comfort and function after the ream-and-run arthroplasty for glenohumeral arthritis: An analysis of 176 consecutive cases. J Bone Joint Surg Am. 2012;94(14):e102.
26. Lynch JR, Franta AK, Montgomery WH Jr, Lenters TR, Mounce D, Matsen FA 3rd. Self-assessed outcome at two to four years after shoulder hemiarthroplasty with concentric glenoid reaming. J Bone Joint Surg Am. 2007;89(6):1284-1292.
27. Donohue KW, Ricchetti ET, Iannotti JP. Surgical management of the biconcave (B2) glenoid. Curr Rev Musculoskelet Med. 2016;9(1):30-39.
28. Clavert P, Millett PJ, Warner JJ. Glenoid resurfacing: What are the limits to asymmetric reaming for posterior erosion? J Shoulder Elbow Surg. 2007;16(6):843-848.
29. Gillespie R, Lyons R, Lazarus M. Eccentric reaming in total shoulder arthroplasty: A cadaveric study. Orthopedics. 2009;32(1):21.
30. Neer CS 2nd, Morrison DS. Glenoid bone-grafting in total shoulder arthroplasty. J Bone Joint Surg Am. 1988;70(8):1154-1162.
31. Nowak DD, Bahu MJ, Gardner TR, et al. Simulation of surgical glenoid resurfacing using three-dimensional computed tomography of the arthritic glenohumeral joint: The amount of glenoid retroversion that can be corrected. J Shoulder Elbow Surg. 2009;18(5):680-688.
32. Strauss EJ, Roche C, Flurin PH, Wright T, Zuckerman JD. The glenoid in shoulder arthroplasty. J Shoulder Elbow Surg. 2009;18(5):819-833.
33. Walch G, Young AA, Boileau P, Loew M, Gazielly D, Mole D. Patterns of loosening of polyethylene keeled glenoid components after shoulder arthroplasty for primary osteoarthritis: Results of a multicenter study with more than five years of follow-up. J Bone Joint Surg Am. 2012;94(2):145-150.
34. Walch G, Young AA, Melis B, Gazielly D, Loew M, Boileau P. Results of a convex-back cemented keeled glenoid component in primary osteoarthritis: Multicenter study with a follow-up greater than 5 years. J Shoulder Elbow Surg. 2011;20(3):385-394.
35. Klika BJ, Wooten CW, Sperling JW, et al. Structural bone grafting for glenoid deficiency in primary total shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):1066-1072.
36. Sabesan V, Callanan M, Sharma V, Iannotti JP. Correction of acquired glenoid bone loss in osteoarthritis with a standard versus an augmented glenoid component. J Shoulder Elbow Surg. 2014;23(7):964-973.
37. Steinmann SP, Cofield RH. Bone grafting for glenoid deficiency in total shoulder replacement. J Shoulder Elbow Surg. 2000;9(5):361-367.
38. Cil A, Sperling JW, Cofield RH. Nonstandard glenoid components for bone deficiencies in shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):e149-e157.
39. Iannotti JP, Lappin KE, Klotz CL, Reber EW, Swope SW. Liftoff resistance of augmented glenoid components during cyclic fatigue loading in the posterior-superior direction. J Shoulder Elbow Surg. 2013;22(11):1530-1536.
40. Knowles NK, Ferreira LM, Athwal GS. Augmented glenoid component designs for type B2 erosions: A computational comparison by volume of bone removal and quality of remaining bone. J Shoulder Elbow Surg. 2015;24(8):1218-1226.
41. Mizuno N, Denard PJ, Raiss P, Walch G. Reverse total shoulder arthroplasty for primary glenohumeral osteoarthritis in patients with a biconcave glenoid. J Bone Joint Surg Am. 2013;95(14):1297-1304.
1. Barrett WP, Franklin JL, Jackins SE, Wyss CR, Matsen FA 3rd. Total shoulder arthroplasty. J Bone Joint Surg Am. 1987;69(6):865-872.
2. Bryant D, Litchfield R, Sandow M, Gartsman GM, Guyatt G, Kirkley A. A comparison of pain, strength, range of motion, and functional outcomes after hemiarthroplasty and total shoulder arthroplasty in patients with osteoarthritis of the shoulder. A systematic review and meta-analysis. J Bone Joint Surg Am. 2005;87(9):1947-1956.
3. Matsen FA 3rd. Early effectiveness of shoulder arthroplasty for patients who have primary glenohumeral degenerative joint disease. J Bone Joint Surg Am. 1996;78(2):260-264.
4. Fenlin JM Jr, Frieman BG. Indications, technique, and results of total shoulder arthroplasty in osteoarthritis. Orthop Clin North Am. 1998;29(3):423-434.
5. Singh JA, Sperling JW, Cofield RH. Revision surgery following total shoulder arthroplasty: Analysis of 2588 shoulders over three decades (1976 to 2008). J Bone Joint Surg Br. 2011;93(11):1513-1517.
6. Iannotti JP, Norris TR. Influence of preoperative factors on outcome of shoulder arthroplasty for glenohumeral osteoarthritis. J Bone Joint Surg Am. 2003;85-A(2):251-258.
7. Levine WN, Djurasovic M, Glasson JM, Pollock RG, Flatow EL, Bigliani LU. Hemiarthroplasty for glenohumeral osteoarthritis: Results correlated to degree of glenoid wear. J Shoulder Elbow Surg. 1997;6(5):449-454.
8. Walch G, Badet R, Boulahia A, Khoury A. Morphologic study of the glenoid in primary glenohumeral osteoarthritis. J Arthroplasty. 1999;14(6):756-760.
9. Scalise JJ, Codsi MJ, Bryan J, Iannotti JP. The three-dimensional glenoid vault model can estimate normal glenoid version in osteoarthritis. J Shoulder Elbow Surg. 2008;17(3):487-491.
10. Walch G, Moraga C, Young A, Castellanos-Rosas J. Results of anatomic nonconstrained prosthesis in primary osteoarthritis with biconcave glenoid. J Shoulder Elbow Surg. 2012;21(11):1526-1533.
11. Kany J, Katz D. How to deal with glenoid type B2 or C? How to prevent mistakes in implantation of glenoid component? Eur J Orthop Surg Traumatol. 2013;23(4):379-385.
12. Denard PJ, Walch G. Current concepts in the surgical management of primary glenohumeral arthritis with a biconcave glenoid. J Shoulder Elbow Surg. 2013;22(11):1589-1598.
13. Iannotti JP, Greeson C, Downing D, Sabesan V, Bryan JA. Effect of glenoid deformity on glenoid component placement in primary shoulder arthroplasty. J Shoulder Elbow Surg. 2012;21(1):48-55.
14. Ho JC, Sabesan VJ, Iannotti JP. Glenoid component retroversion is associated with osteolysis. J Bone Joint Surg Am. 2013;95(12):e82.
15. Friedman RJ, Hawthorne KB, Genez BM. The use of computerized tomography in the measurement of glenoid version. J Bone Joint Surg Am. 1992;74(7):1032-1037.
16. Rouleau DM, Kidder JF, Pons-Villanueva J, Dynamidis S, Defranco M, Walch G. Glenoid version: How to measure it? Validity of different methods in two-dimensional computed tomography scans. J Shoulder Elbow Surg. 2010;19(8):1230-1237.
17. Nyffeler RW, Jost B, Pfirrmann CW, Gerber C. Measurement of glenoid version: Conventional radiographs versus computed tomography scans. J Shoulder Elbow Surg. 2003;12(5):493-496.
18. Budge MD, Lewis GS, Schaefer E, Coquia S, Flemming DJ, Armstrong AD. Comparison of standard two-dimensional and three-dimensional corrected glenoid version measurements. J Shoulder Elbow Surg. 2011;20(4):577-583.
19. Bokor DJ, O’Sullivan MD, Hazan GJ. Variability of measurement of glenoid version on computed tomography scan. J Shoulder Elbow Surg. 1999;8(6):595-598.
20. Ganapathi A, McCarron JA, Chen X, Iannotti JP. Predicting normal glenoid version from the pathologic scapula: A comparison of 4 methods in 2- and 3-dimensional models. J Shoulder Elbow Surg. 2011;20(2):234-244.
21. Ricchetti ET, Hendel MD, Collins DN, Iannotti JP. Is premorbid glenoid anatomy altered in patients with glenohumeral osteoarthritis? Clin Orthop Relat Res. 2013;471(9):2932-2939.
22. Hendel MD, Bryan JA, Barsoum WK, et al. Comparison of patient-specific instruments with standard surgical instruments in determining glenoid component position: A randomized prospective clinical trial. J Bone Joint Surg Am. 2012;94(23):2167-2175.
23. Matsen FA 3rd, Warme WJ, Jackins SE. Can the ream and run procedure improve glenohumeral relationships and function for shoulders with the arthritic triad? Clin Orthop Relat Res. 2015;473(6):2088-2096.
24. Saltzman MD, Chamberlain AM, Mercer DM, Warme WJ, Bertelsen AL, Matsen FA 3rd. Shoulder hemiarthroplasty with concentric glenoid reaming in patients 55 years old or less. J Shoulder Elbow Surg. 2011;20(4):609-615.
25. Gilmer BB, Comstock BA, Jette JL, Warme WJ, Jackins SE, Matsen FA. The prognosis for improvement in comfort and function after the ream-and-run arthroplasty for glenohumeral arthritis: An analysis of 176 consecutive cases. J Bone Joint Surg Am. 2012;94(14):e102.
26. Lynch JR, Franta AK, Montgomery WH Jr, Lenters TR, Mounce D, Matsen FA 3rd. Self-assessed outcome at two to four years after shoulder hemiarthroplasty with concentric glenoid reaming. J Bone Joint Surg Am. 2007;89(6):1284-1292.
27. Donohue KW, Ricchetti ET, Iannotti JP. Surgical management of the biconcave (B2) glenoid. Curr Rev Musculoskelet Med. 2016;9(1):30-39.
28. Clavert P, Millett PJ, Warner JJ. Glenoid resurfacing: What are the limits to asymmetric reaming for posterior erosion? J Shoulder Elbow Surg. 2007;16(6):843-848.
29. Gillespie R, Lyons R, Lazarus M. Eccentric reaming in total shoulder arthroplasty: A cadaveric study. Orthopedics. 2009;32(1):21.
30. Neer CS 2nd, Morrison DS. Glenoid bone-grafting in total shoulder arthroplasty. J Bone Joint Surg Am. 1988;70(8):1154-1162.
31. Nowak DD, Bahu MJ, Gardner TR, et al. Simulation of surgical glenoid resurfacing using three-dimensional computed tomography of the arthritic glenohumeral joint: The amount of glenoid retroversion that can be corrected. J Shoulder Elbow Surg. 2009;18(5):680-688.
32. Strauss EJ, Roche C, Flurin PH, Wright T, Zuckerman JD. The glenoid in shoulder arthroplasty. J Shoulder Elbow Surg. 2009;18(5):819-833.
33. Walch G, Young AA, Boileau P, Loew M, Gazielly D, Mole D. Patterns of loosening of polyethylene keeled glenoid components after shoulder arthroplasty for primary osteoarthritis: Results of a multicenter study with more than five years of follow-up. J Bone Joint Surg Am. 2012;94(2):145-150.
34. Walch G, Young AA, Melis B, Gazielly D, Loew M, Boileau P. Results of a convex-back cemented keeled glenoid component in primary osteoarthritis: Multicenter study with a follow-up greater than 5 years. J Shoulder Elbow Surg. 2011;20(3):385-394.
35. Klika BJ, Wooten CW, Sperling JW, et al. Structural bone grafting for glenoid deficiency in primary total shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):1066-1072.
36. Sabesan V, Callanan M, Sharma V, Iannotti JP. Correction of acquired glenoid bone loss in osteoarthritis with a standard versus an augmented glenoid component. J Shoulder Elbow Surg. 2014;23(7):964-973.
37. Steinmann SP, Cofield RH. Bone grafting for glenoid deficiency in total shoulder replacement. J Shoulder Elbow Surg. 2000;9(5):361-367.
38. Cil A, Sperling JW, Cofield RH. Nonstandard glenoid components for bone deficiencies in shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):e149-e157.
39. Iannotti JP, Lappin KE, Klotz CL, Reber EW, Swope SW. Liftoff resistance of augmented glenoid components during cyclic fatigue loading in the posterior-superior direction. J Shoulder Elbow Surg. 2013;22(11):1530-1536.
40. Knowles NK, Ferreira LM, Athwal GS. Augmented glenoid component designs for type B2 erosions: A computational comparison by volume of bone removal and quality of remaining bone. J Shoulder Elbow Surg. 2015;24(8):1218-1226.
41. Mizuno N, Denard PJ, Raiss P, Walch G. Reverse total shoulder arthroplasty for primary glenohumeral osteoarthritis in patients with a biconcave glenoid. J Bone Joint Surg Am. 2013;95(14):1297-1304.
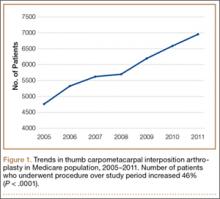
Trends in Thumb Carpometacarpal Interposition Arthroplasty in the United States, 2005–2011
A common entity, osteoarthritis (OA) at the base of the thumb is largely caused by the unique anatomy and biomechanics of the thumb carpometacarpal (CMC) joint.1 Radiographically evident CMC degeneration occurs in 40% of women and 25% of men over age 75 years, making the thumb CMC joint the most common site of surgical reconstruction for upper extremity OA.2,3
Over the past 40 years, numerous surgical techniques for managing thumb CMC-OA have been described. These include volar ligament reconstruction, first metacarpal osteotomy, CMC arthrodesis, CMC joint replacement, and trapeziectomy. Trapeziectomy can be performed in isolation or in combination with tendon interposition, ligament reconstruction, or ligament reconstruction and tendon interposition (LRTI).4-20 The authors of a recent systematic review concluded there is no evidence that any one surgical procedure for CMC-OA is superior to another in terms of pain, function, satisfaction, range of motion, or strength.4 Nevertheless, a recent survey found that 719 (62%) of 1156 US hand surgeons used LRTI as the treatment of choice for advanced CMC-OA.21
Our detailed literature search yielded no other database studies characterizing current trends in the practice patterns of US orthopedic surgeons who perform interposition arthroplasty for CMC arthritis. Analysis of these trends is important not only to patients but also to the broader orthopedic and health care community.22
We conducted a study to investigate current trends in CMC interposition arthroplasty across time, sex, age, and region of the United States; per-patient charges and reimbursements; and the association between this procedure and concomitantly performed carpal tunnel syndrome (CTS) and carpal tunnel release (CTR). In addition, we compared incidence of CMC interposition arthroplasty with that of CMC arthrodesis.
Patients and Methods
All data were derived from the PearlDiver Patient Records Database (PearlDiver Technologies), a publicly available database of patients. The database stores procedure volumes, demographics, and average charge information for patients with International Classification of Diseases, Ninth Revision (ICD-9) diagnoses and procedures or Current Procedural Terminology (CPT) codes. Data for the present study were drawn from the Medicare database within the PearlDiver records, which has a total of 179,094,296 patient records covering the period 2005–2011. This study did not require institutional review board approval, as it used existing, publicly available data without identifiers linked to subjects.
PearlDiver Technologies granted us database access for academic research. The database was stored on a password-protected server maintained by PearlDiver. ICD-9 and CPT codes can be searched in isolation or in combination. Search results yield number of patients with a searched code (or combination of codes) in each year, age group, or region of the United States, as well as mean charge and mean reimbursement for the code or combination of codes.
We used CPT code 25447 (arthroplasty, interposition, intercarpal, or CMC joints) to search the database for patients who underwent thumb CMC interposition arthroplasty. Although this code does not specify thumb, we are unaware of any procedure (other than thumb CMC interposition arthroplasty) typically given this code. Our search yielded procedure volumes, sex distribution, age distribution, region volumes, and mean per-patient charges and reimbursements for each CPT code. We then searched the resulting cohort for CTS (ICD-9 code 354.0), endoscopic CTR (CPT code 29848), and open CTR (CPT code 64721) to find CTR performed concomitantly with CMC interposition arthroplasty. Last, patients were tracked in the database past their surgery date to evaluate for postoperative physical or occupational therapy evaluations within 6 months (using CPT codes appearing in at least 1% of the cohort: 97001, 97003, 97004, 97110, 97112, 97124, 97140, 97150, 97350, 97535) and postoperative thumb, hand, or wrist radiographs within 6 months (using CPT codes appearing in at least 1% of the cohort: 73140, 73130, 73110). To ensure adequacy of 6-month postoperative data, we included in this portion of the study only those patients with surgery dates between 2005 and 2010.
For comparative purposes, we also searched the database for patients who underwent thumb CMC arthrodesis within the same period—using CPT codes 26841 and 26842 (arthrodesis CMC joint thumb, with or without internal fixation; with or without autograft) and CPT code 26820 (fusion in opposition, thumb, with autogenous graft).
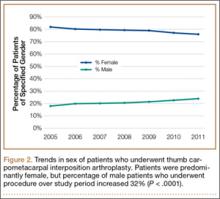
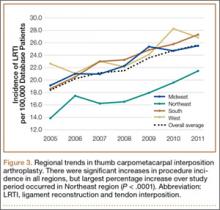
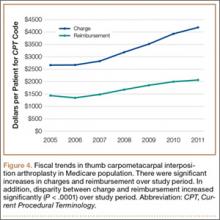
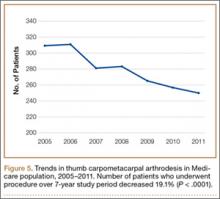
Overall procedure volume data are reported as number of patients with the given CPT code in the database output in a given year. Age-group and sex analyses are reported as number of patients reported in the database output and as percentage of patients who underwent the CPT code of interest that year. Mean charges and reimbursements are reported as results by the database for the code of interest (CPT 25447). Data for the region analysis are presented as an incidence, as there is an uneven distribution of patient volumes among regions. This incidence is calculated as number of patients in a particular region and year normalized to total number of patients in the database for that particular region or year. Regions are defined as Midwest (IA, IL, IN, KS, MI, MN, MO, ND, NE, OH, SD, WI), Northeast (CT, MA, ME, NH, NJ, NY, PA, RI, VT), South (AL, AR, DC, DE, FL, GA, KY, LA, MD, MS, NC, OK, SC, TN, TX, VA, WV), and West (AK, AZ, CA, CO, HI, ID, MT, NM, NV, OR, UT, WA, WY).
Chi-squared linear-by-linear association analysis was used to determine statistical significance with regard to trends over time in procedure volumes, sex, age group, and region. For all statistical comparisons, P < .05 was considered significant.
Results
In the database, we identified 41,171 unique patients who underwent CMC interposition arthroplasty between 2005 and 2011. Over the 7-year study period, number of patients who had CMC interposition arthroplasty increased 46.2%, from 4761 in 2005 to 6960 in 2011 (P < .0001) (Table 1, Figure 1). Throughout this period, females underwent CMC interposition arthroplasty more frequently than males at all time points (P < .0001). Overall ratio of female to male patients, however, changed significantly. In 2005, 18.1% of all CMC interposition arthroplasties were performed on male patients; this increased to 23.9% of all procedures by 2011 (P < .0001) (Figure 2). Table 1 presents an age-group analysis. There were no significant differences in relative percentage of patients in any given age group who underwent CMC interposition arthroplasty over the study period.
Analysis of overall procedure incidence by region revealed significant increases in all regions (P < .0001), ranging from 18.5% (West) to 54.5% (Northeast) (Figure 3). At all time points, the incidence of CMC interposition arthroplasty was significantly lower in the Northeast than in any other region and compared with the overall average.
Between 2005 and 2011, there were significant increases in both per-patient charges and reimbursements for CMC interposition arthroplasty (Figure 4). Mean per-patient charge increased from $2676 in 2005 to $4181 in 2011 (P < .0001), and mean per-patient reimbursement increased from $1445 in 2005 to $2061 in 2011 (P < .0001). The discrepancy between charge and reimbursement increased throughout the study period: Reimbursement in 2005 was 54.0% of the charge; this decreased to 49.3% by 2011 but was not statistically significant (P = .08).
Overall, 40.9% of patients who underwent CMC interposition arthroplasty also had a CTS diagnosis. Between 15.5% and 17.3% of these patients had concomitant open or endoscopic CTR at time of CMC interposition arthroplasty (Table 2). Percentage of patients who underwent concomitant CTR did not change significantly from 2005 to 2011 (P = .139). Use of postoperative occupational and/or physical therapy increased significantly over the study period, from 33.5% of patients in 2005 to 50.7% of patients in 2010 (P < .0001). Use of postoperative thumb, hand, and/or wrist radiography also increased throughout the study period, from 7.4% of patients in 2005 to 18.7% of patients in 2010 (P < .0001).
We identified 1916 unique patients who underwent thumb CMC arthrodesis between 2005 and 2011. Over the 7-year study period, there was a 19.1% decrease in number of patients who underwent CMC arthrodesis, from 309 in 2005 to 250 in 2011 (P < .0001) (Figure 5). Significantly fewer patients had CMC arthrodesis compared with CMC interposition arthroplasty at all time points, ranging from 6.5% (thumb CMC arthrodesis:CMC interposition arthroplasty) in 2005 to 3.6% in 2011 (P < .0001).
Discussion
Our results demonstrated a significant increase in use of thumb CMC interposition arthroplasty in a US Medicare population, with an increase of more than 46% from 2005 to 2011. This finding supports the findings of a recent cross-sectional survey-based study in which 719 (62%) of 1156 surveyed US hand surgeons reported performing trapeziectomy with LRTI for advanced thumb CMC-OA.21 A prior study had similar findings, with 692 (68%) of 1024 American Society for Surgery of the Hand (ASSH) members performing LRTI and 766 (75%) of 1024 performing some type of CMC interposition with trapeziectomy for advanced CMC-OA.23 This preference for CMC interposition arthroplasty prevails despite the fact that numerous studies have shown no superiority of any surgical procedure to another for CMC-OA in terms of pain, function, satisfaction, range of motion, and strength.7,15,18,19,24-34 Our data demonstrated that, not only does CMC interposition arthroplasty remain the most frequently used procedure for thumb CMC-OA, the incidence of CMC interposition arthroplasty continues to increase yearly.
The incidence of thumb CMC-OA is higher in women than in men, with more joint laxity a known contributor and subtle sex differences in trapezium geometry and congruence postulated as additional factors.3,35,36 This trend was confirmed in the present study, as females underwent significantly more CMC interposition arthroplasties at all time points. It is interesting that the overall ratio of female to male patients changed significantly over the study period, with the percentage of patients who were male increasing from 18.1% in 2005 to 23.9% in 2011. No previous studies have captured such a large cross section of the population to establish this trend. Although this trend is not necessarily intuitive, potential theories include increased acceptance of CMC interposition arthroplasty as a surgical option for male patients, and potentially a larger number of male patients seeking medical care for thumb CMC-OA in recent years.
Increases in procedure incidence were noted in all regions of the United States, but the largest percentage increase occurred in the Northeast. Despite this increase, the Northeast also had significantly lower CMC interposition arthroplasty incidence compared with all other regions and with the average procedure incidence throughout the study period—demonstrating some regional bias as to treatment of thumb CMC-OA. Unfortunately, because of database limitations and lack of specific CPT codes for other treatment options for thumb CMC-OA, we cannot ascertain if other types of surgery are more frequently used in the Northeast.
CTS and thumb CMC-OA often coexist.37 The estimated incidence of concomitant CTS in patients with CMC-OA is between 4% and 43%, but the rate of concomitant CTR and CMC interposition arthroplasty was not previously characterized in the literature.38,39 Results of the present study supported these findings; 41% of patients who underwent CMC interposition arthroplasty in our study also had a CTS diagnosis, compared with 43% in the 246-patient study by Florack and colleagues.38 We also found that 16% to 17% of patients who underwent CMC interposition arthroplasty underwent concomitant CTR; this rate remained consistent throughout the study period.
Our study demonstrated that, compared with CMC interposition arthroplasties, significantly fewer thumb CMC arthrodesis procedures were performed in the same Medicare population during the same period. Furthermore, the number of thumb CMC arthrodesis procedures declined yearly, with an overall decrease of 19% from 2005 to 2011. In a recent single-blinded, randomized trial, Vermeulen and colleagues40 compared thumb CMC arthrodesis and trapeziectomy with LRTI. They found superior patient satisfaction and significantly lower complication rates in women who underwent LRTI versus arthrodesis. The study was terminated prematurely because of these complications and thus was underpowered to determine differences in specific outcome measures. Previous studies comparing arthrodesis and interposition arthroplasties reported inconsistent outcomes. Hart and colleagues41 found no significant differences in pain or function between CMC arthrodesis and LRTI at a mean 7-year follow-up in a level II randomized controlled trial. Hartigan and colleagues15 reached similar conclusions in their retrospective comparison of the procedures. Without clear evidence supporting arthrodesis over interposition arthroplasty, the majority of surgeons favor interposition arthroplasty for thumb CMC-OA. Among Medicare patients, use of thumb CMC arthrodesis continues to fall.
This national database study had several limitations, which are common to all studies using the PearlDiver database22,42-47:
1. The power of the analysis depended on the quality of available data. Potential sources of error included accuracy of billing codes, and miscoding or noncoding by physicians.46
2. Although we used this database to try to accurately represent a large population of interest, we cannot guarantee the database represented a true cross section of the United States.
3. For the Medicare population, the PearlDiver database indexes data only in 7-year increments. Although the study period was long enough to detect significant trends, some data may not be accurately captured over a 7-year period.
4. Patients were not randomized to a treatment group.
5. The PearlDiver database does not include any clinical outcome data. Therefore, we cannot comment on the efficacy of the reported evaluations and interventions.
6. There is no specific CPT code for thumb CMC interposition arthroplasty. However, we are unaware of a CMC interposition arthroplasty performed for any area besides the thumb. Theoretically, the study population can include a negligible percentage of patients who had interposition arthroplasty of a CMC joint other than the thumb.
7. The database cannot be searched for use of thumb CMC-OA surgical techniques other than CMC interposition arthroplasty or arthrodesis, as isolated trapeziectomy, volar ligament reconstruction, implant arthroplasty, and metacarpal osteotomy lack specific CPT codes.
Conclusion
Thumb CMC-OA is a common entity among Medicare patients. There are numerous surgical options for cases that have failed conservative treatment. Despite the lack of evidence that thumb CMC interposition arthroplasty is superior to other surgical options, the number of patients who had this procedure increased 46% during the 2005–2011 study period. Although the majority of patients who undergo CMC interposition arthroplasty are female, the percentage of male patients has increased significantly. More than 40% of patients who have CMC interposition arthroplasty are also diagnosed with CTS, and 16% to 17% of patients who have CMC interposition arthroplasty will have a concomitant CTR. CMC arthrodesis is used in significantly fewer patients of Medicare age, and its use has been declining.
1. Hentz VR. Surgical treatment of trapeziometacarpal joint arthritis: a historical perspective. Clin Orthop Relat Res. 2014;472(4):1184-1189.
2. Armstrong AL, Hunter JB, Davis TR. The prevalence of degenerative arthritis of the base of the thumb in post-menopausal women. J Hand Surg Br. 1994;19(3):340-341.
3. Van Heest AE, Kallemeier P. Thumb carpal metacarpal arthritis. J Am Acad Orthop Surg. 2008;16(3):140-151.
4. Vermeulen GM, Slijper H, Feitz R, Hovius SE, Moojen TM, Selles RW. Surgical management of primary thumb carpometacarpal osteoarthritis: a systematic review. J Hand Surg Am. 2011;36(1):157-169.
5. Bodin ND, Spangler R, Thoder JJ. Interposition arthroplasty options for carpometacarpal arthritis of the thumb. Hand Clin. 2010;26(3):339-350, v-vi.
6. Cooney WP, Linscheid RL, Askew LJ. Total arthroplasty of the thumb trapeziometacarpal joint. Clin Orthop Relat Res. 1987;(220):35-45.
7. De Smet L, Vandenberghe L, Degreef I. Long-term outcome of trapeziectomy with ligament reconstruction and tendon interposition (LRTI) versus prosthesis arthroplasty for basal joint osteoarthritis of the thumb. Acta Orthop Belg. 2013;79(2):146-149.
8. Dell PC, Muniz RB. Interposition arthroplasty of the trapeziometacarpal joint for osteoarthritis. Clin Orthop Relat Res. 1987;(220):27-34.
9. Dhar S, Gray IC, Jones WA, Beddow FH. Simple excision of the trapezium for osteoarthritis of the carpometacarpal joint of the thumb. J Hand Surg Br. 1994;19(4):485-488.
10. Eaton RG, Littler JW. Ligament reconstruction for the painful thumb carpometacarpal joint. J Bone Joint Surg Am. 1973;55(8):1655-1666.
11. Eaton RG, Lane LB, Littler JW, Keyser JJ. Ligament reconstruction for the painful thumb carpometacarpal joint: a long-term assessment. J Hand Surg Am. 1984;9(5):692-699.
12. Eaton RG, Glickel SZ, Littler JW. Tendon interposition arthroplasty for degenerative arthritis of the trapeziometacarpal joint of the thumb. J Hand Surg Am. 1985;10(5):645-654.
13. Elfar JC, Burton RI. Ligament reconstruction and tendon interposition for thumb basal arthritis. Hand Clin. 2013;29(1):15-25.
14. Froimson AI. Tendon arthroplasty of the trapeziometacarpal joint. Clin Orthop Relat Res. 1970;70:191-199.
15. Hartigan BJ, Stern PJ, Kiefhaber TR. Thumb carpometacarpal osteoarthritis: arthrodesis compared with ligament reconstruction and tendon interposition. J Bone Joint Surg Am. 2001;83(10):1470-1478.
16. Kenniston JA, Bozentka DJ. Treatment of advanced carpometacarpal joint disease: arthrodesis. Hand Clin. 2008;24(3):285-294, vi-vii.
17. Kokkalis ZT, Zanaros G, Weiser RW, Sotereanos DG. Trapezium resection with suspension and interposition arthroplasty using acellular dermal allograft for thumb carpometacarpal arthritis. J Hand Surg Am. 2009;34(6):1029-1036.
18. Kriegs-Au G, Petje G, Fojtl E, Ganger R, Zachs I. Ligament reconstruction with or without tendon interposition to treat primary thumb carpometacarpal osteoarthritis. Surgical technique. J Bone Joint Surg Am. 2005;87 suppl 1(Pt 1):78-85.
19. Park MJ, Lichtman G, Christian JB, et al. Surgical treatment of thumb carpometacarpal joint arthritis: a single institution experience from 1995–2005. Hand. 2008;3(4):304-310.
20. Park MJ, Lee AT, Yao J. Treatment of thumb carpometacarpal arthritis with arthroscopic hemitrapeziectomy and interposition arthroplasty. Orthopedics. 2012;35(12):e1759-e1764.
21. Wolf JM, Delaronde S. Current trends in nonoperative and operative treatment of trapeziometacarpal osteoarthritis: a survey of US hand surgeons. J Hand Surg Am. 2012;37(1):77-82.
22. Zhang AL, Kreulen C, Ngo SS, Hame SL, Wang JC, Gamradt SC. Demographic trends in arthroscopic SLAP repair in the United States. Am J Sports Med. 2012;40(5):1144-1147.
23. Brunton LM, Wilgis EF. A survey to determine current practice patterns in the surgical treatment of advanced thumb carpometacarpal osteoarthrosis. Hand. 2010;5(4):415-422.
24. Belcher HJ, Nicholl JE. A comparison of trapeziectomy with and without ligament reconstruction and tendon interposition. J Hand Surg Br. 2000;25(4):350-356.
25. Davis TR, Pace A. Trapeziectomy for trapeziometacarpal joint osteoarthritis: is ligament reconstruction and temporary stabilisation of the pseudarthrosis with a Kirschner wire important? J Hand Surg Eur Vol. 2009;34(3):312-321.
26. Davis TR, Brady O, Dias JJ. Excision of the trapezium for osteoarthritis of the trapeziometacarpal joint: a study of the benefit of ligament reconstruction or tendon interposition. J Hand Surg Am. 2004;29(6):1069-1077.
27. De Smet L, Sioen W, Spaepen D, van Ransbeeck H. Treatment of basal joint arthritis of the thumb: trapeziectomy with or without tendon interposition/ligament reconstruction. Hand Surg. 2004;9(1):5-9.
28. Field J, Buchanan D. To suspend or not to suspend: a randomised single blind trial of simple trapeziectomy versus trapeziectomy and flexor carpi radialis suspension. J Hand Surg Eur Vol. 2007;32(4):462-466.
29. Gerwin M, Griffith A, Weiland AJ, Hotchkiss RN, McCormack RR. Ligament reconstruction basal joint arthroplasty without tendon interposition. Clin Orthop Relat Res. 1997;(342):42-45.
30. Jorheim M, Isaxon I, Flondell M, Kalen P, Atroshi I. Short-term outcomes of trapeziometacarpal Artelon implant compared with tendon suspension interposition arthroplasty for osteoarthritis: a matched cohort study. J Hand Surg Am. 2009;34(8):1381-1387.
31. Lehmann O, Herren DB, Simmen BR. Comparison of tendon suspension-interposition and silicon spacers in the treatment of degenerative osteoarthritis of the base of the thumb. Ann Chir Main Memb Super. 1998;17(1):25-30.
32. Nilsson A, Liljensten E, Bergstrom C, Sollerman C. Results from a degradable TMC joint spacer (Artelon) compared with tendon arthroplasty. J Hand Surg Am. 2005;30(2):380-389.
33. Schroder J, Kerkhoffs GM, Voerman HJ, Marti RK. Surgical treatment of basal joint disease of the thumb: comparison between resection-interposition arthroplasty and trapezio-metacarpal arthrodesis. Arch Orthop Trauma Surg. 2002;122(1):35-38.
34. Tagil M, Kopylov P. Swanson versus APL arthroplasty in the treatment of osteoarthritis of the trapeziometacarpal joint: a prospective and randomized study in 26 patients. J Hand Surg Br. 2002;27(5):452-456.
35. North ER, Rutledge WM. The trapezium-thumb metacarpal joint: the relationship of joint shape and degenerative joint disease. Hand. 1983;15(2):201-206.
36. Ateshian GA, Rosenwasser MP, Mow VC. Curvature characteristics and congruence of the thumb carpometacarpal joint: differences between female and male joints. J Biomech. 1992;25(6):591-607.
37. Sless Y, Sampson SP. Experience with transtrapezium approach for transverse carpal ligament release in patients with coexisted trapeziometacarpal joint osteoarthritis and carpal tunnel syndrome. Hand. 2007;2(3):151-154.
38. Florack TM, Miller RJ, Pellegrini VD, Burton RI, Dunn MG. The prevalence of carpal tunnel syndrome in patients with basal joint arthritis of the thumb. J Hand Surg Am. 1992;17(4):624-630.
39. Tsai TM, Laurentin-Perez LA, Wong MS, Tamai M. Ideas and innovations: radial approach to carpal tunnel release in conjunction with thumb carpometacarpal arthroplasty. Hand Surg. 2005;10(1):61-66.
40. Vermeulen GM, Brink SM, Slijper H, et al. Trapeziometacarpal arthrodesis or trapeziectomy with ligament reconstruction in primary trapeziometacarpal osteoarthritis: a randomized controlled trial. J Bone Joint Surg Am. 2014;96(9):726-733.
41. Hart R, Janecek M, Siska V, Kucera B, Stipcak V. Interposition suspension arthroplasty according to Epping versus arthrodesis for trapeziometacarpal osteoarthritis. Eur Surg. 2006;38(6):433-438.
42. Abrams GD, Frank RM, Gupta AK, Harris JD, McCormick FM, Cole BJ. Trends in meniscus repair and meniscectomy in the United States, 2005–2011. Am J Sports Med. 2013;41(10):2333-2339.
43. Montgomery SR, Ngo SS, Hobson T, et al. Trends and demographics in hip arthroscopy in the United States. Arthroscopy. 2013;29(4):661-665.
44. Zhang AL, Montgomery SR, Ngo SS, Hame SL, Wang JC, Gamradt SC. Arthroscopic versus open shoulder stabilization: current practice patterns in the United States. Arthroscopy. 2014;30(4):436-443.
45. Yeranosian MG, Arshi A, Terrell RD, Wang JC, McAllister DR, Petrigliano FA. Incidence of acute postoperative infections requiring reoperation after arthroscopic shoulder surgery. Am J Sports Med. 2014;42(2):437-441.
46. Yeranosian MG, Terrell RD, Wang JC, McAllister DR, Petrigliano FA. The costs associated with the evaluation of rotator cuff tears before surgical repair. J Shoulder Elbow Surg. 2013;22(12):1662-1666.
47. Daffner SD, Hymanson HJ, Wang JC. Cost and use of conservative management of lumbar disc herniation before surgical discectomy. Spine J. 2010;10(6):463-468.
A common entity, osteoarthritis (OA) at the base of the thumb is largely caused by the unique anatomy and biomechanics of the thumb carpometacarpal (CMC) joint.1 Radiographically evident CMC degeneration occurs in 40% of women and 25% of men over age 75 years, making the thumb CMC joint the most common site of surgical reconstruction for upper extremity OA.2,3
Over the past 40 years, numerous surgical techniques for managing thumb CMC-OA have been described. These include volar ligament reconstruction, first metacarpal osteotomy, CMC arthrodesis, CMC joint replacement, and trapeziectomy. Trapeziectomy can be performed in isolation or in combination with tendon interposition, ligament reconstruction, or ligament reconstruction and tendon interposition (LRTI).4-20 The authors of a recent systematic review concluded there is no evidence that any one surgical procedure for CMC-OA is superior to another in terms of pain, function, satisfaction, range of motion, or strength.4 Nevertheless, a recent survey found that 719 (62%) of 1156 US hand surgeons used LRTI as the treatment of choice for advanced CMC-OA.21
Our detailed literature search yielded no other database studies characterizing current trends in the practice patterns of US orthopedic surgeons who perform interposition arthroplasty for CMC arthritis. Analysis of these trends is important not only to patients but also to the broader orthopedic and health care community.22
We conducted a study to investigate current trends in CMC interposition arthroplasty across time, sex, age, and region of the United States; per-patient charges and reimbursements; and the association between this procedure and concomitantly performed carpal tunnel syndrome (CTS) and carpal tunnel release (CTR). In addition, we compared incidence of CMC interposition arthroplasty with that of CMC arthrodesis.
Patients and Methods
All data were derived from the PearlDiver Patient Records Database (PearlDiver Technologies), a publicly available database of patients. The database stores procedure volumes, demographics, and average charge information for patients with International Classification of Diseases, Ninth Revision (ICD-9) diagnoses and procedures or Current Procedural Terminology (CPT) codes. Data for the present study were drawn from the Medicare database within the PearlDiver records, which has a total of 179,094,296 patient records covering the period 2005–2011. This study did not require institutional review board approval, as it used existing, publicly available data without identifiers linked to subjects.
PearlDiver Technologies granted us database access for academic research. The database was stored on a password-protected server maintained by PearlDiver. ICD-9 and CPT codes can be searched in isolation or in combination. Search results yield number of patients with a searched code (or combination of codes) in each year, age group, or region of the United States, as well as mean charge and mean reimbursement for the code or combination of codes.
We used CPT code 25447 (arthroplasty, interposition, intercarpal, or CMC joints) to search the database for patients who underwent thumb CMC interposition arthroplasty. Although this code does not specify thumb, we are unaware of any procedure (other than thumb CMC interposition arthroplasty) typically given this code. Our search yielded procedure volumes, sex distribution, age distribution, region volumes, and mean per-patient charges and reimbursements for each CPT code. We then searched the resulting cohort for CTS (ICD-9 code 354.0), endoscopic CTR (CPT code 29848), and open CTR (CPT code 64721) to find CTR performed concomitantly with CMC interposition arthroplasty. Last, patients were tracked in the database past their surgery date to evaluate for postoperative physical or occupational therapy evaluations within 6 months (using CPT codes appearing in at least 1% of the cohort: 97001, 97003, 97004, 97110, 97112, 97124, 97140, 97150, 97350, 97535) and postoperative thumb, hand, or wrist radiographs within 6 months (using CPT codes appearing in at least 1% of the cohort: 73140, 73130, 73110). To ensure adequacy of 6-month postoperative data, we included in this portion of the study only those patients with surgery dates between 2005 and 2010.
For comparative purposes, we also searched the database for patients who underwent thumb CMC arthrodesis within the same period—using CPT codes 26841 and 26842 (arthrodesis CMC joint thumb, with or without internal fixation; with or without autograft) and CPT code 26820 (fusion in opposition, thumb, with autogenous graft).
Overall procedure volume data are reported as number of patients with the given CPT code in the database output in a given year. Age-group and sex analyses are reported as number of patients reported in the database output and as percentage of patients who underwent the CPT code of interest that year. Mean charges and reimbursements are reported as results by the database for the code of interest (CPT 25447). Data for the region analysis are presented as an incidence, as there is an uneven distribution of patient volumes among regions. This incidence is calculated as number of patients in a particular region and year normalized to total number of patients in the database for that particular region or year. Regions are defined as Midwest (IA, IL, IN, KS, MI, MN, MO, ND, NE, OH, SD, WI), Northeast (CT, MA, ME, NH, NJ, NY, PA, RI, VT), South (AL, AR, DC, DE, FL, GA, KY, LA, MD, MS, NC, OK, SC, TN, TX, VA, WV), and West (AK, AZ, CA, CO, HI, ID, MT, NM, NV, OR, UT, WA, WY).
Chi-squared linear-by-linear association analysis was used to determine statistical significance with regard to trends over time in procedure volumes, sex, age group, and region. For all statistical comparisons, P < .05 was considered significant.
Results
In the database, we identified 41,171 unique patients who underwent CMC interposition arthroplasty between 2005 and 2011. Over the 7-year study period, number of patients who had CMC interposition arthroplasty increased 46.2%, from 4761 in 2005 to 6960 in 2011 (P < .0001) (Table 1, Figure 1). Throughout this period, females underwent CMC interposition arthroplasty more frequently than males at all time points (P < .0001). Overall ratio of female to male patients, however, changed significantly. In 2005, 18.1% of all CMC interposition arthroplasties were performed on male patients; this increased to 23.9% of all procedures by 2011 (P < .0001) (Figure 2). Table 1 presents an age-group analysis. There were no significant differences in relative percentage of patients in any given age group who underwent CMC interposition arthroplasty over the study period.
Analysis of overall procedure incidence by region revealed significant increases in all regions (P < .0001), ranging from 18.5% (West) to 54.5% (Northeast) (Figure 3). At all time points, the incidence of CMC interposition arthroplasty was significantly lower in the Northeast than in any other region and compared with the overall average.
Between 2005 and 2011, there were significant increases in both per-patient charges and reimbursements for CMC interposition arthroplasty (Figure 4). Mean per-patient charge increased from $2676 in 2005 to $4181 in 2011 (P < .0001), and mean per-patient reimbursement increased from $1445 in 2005 to $2061 in 2011 (P < .0001). The discrepancy between charge and reimbursement increased throughout the study period: Reimbursement in 2005 was 54.0% of the charge; this decreased to 49.3% by 2011 but was not statistically significant (P = .08).
Overall, 40.9% of patients who underwent CMC interposition arthroplasty also had a CTS diagnosis. Between 15.5% and 17.3% of these patients had concomitant open or endoscopic CTR at time of CMC interposition arthroplasty (Table 2). Percentage of patients who underwent concomitant CTR did not change significantly from 2005 to 2011 (P = .139). Use of postoperative occupational and/or physical therapy increased significantly over the study period, from 33.5% of patients in 2005 to 50.7% of patients in 2010 (P < .0001). Use of postoperative thumb, hand, and/or wrist radiography also increased throughout the study period, from 7.4% of patients in 2005 to 18.7% of patients in 2010 (P < .0001).
We identified 1916 unique patients who underwent thumb CMC arthrodesis between 2005 and 2011. Over the 7-year study period, there was a 19.1% decrease in number of patients who underwent CMC arthrodesis, from 309 in 2005 to 250 in 2011 (P < .0001) (Figure 5). Significantly fewer patients had CMC arthrodesis compared with CMC interposition arthroplasty at all time points, ranging from 6.5% (thumb CMC arthrodesis:CMC interposition arthroplasty) in 2005 to 3.6% in 2011 (P < .0001).
Discussion
Our results demonstrated a significant increase in use of thumb CMC interposition arthroplasty in a US Medicare population, with an increase of more than 46% from 2005 to 2011. This finding supports the findings of a recent cross-sectional survey-based study in which 719 (62%) of 1156 surveyed US hand surgeons reported performing trapeziectomy with LRTI for advanced thumb CMC-OA.21 A prior study had similar findings, with 692 (68%) of 1024 American Society for Surgery of the Hand (ASSH) members performing LRTI and 766 (75%) of 1024 performing some type of CMC interposition with trapeziectomy for advanced CMC-OA.23 This preference for CMC interposition arthroplasty prevails despite the fact that numerous studies have shown no superiority of any surgical procedure to another for CMC-OA in terms of pain, function, satisfaction, range of motion, and strength.7,15,18,19,24-34 Our data demonstrated that, not only does CMC interposition arthroplasty remain the most frequently used procedure for thumb CMC-OA, the incidence of CMC interposition arthroplasty continues to increase yearly.
The incidence of thumb CMC-OA is higher in women than in men, with more joint laxity a known contributor and subtle sex differences in trapezium geometry and congruence postulated as additional factors.3,35,36 This trend was confirmed in the present study, as females underwent significantly more CMC interposition arthroplasties at all time points. It is interesting that the overall ratio of female to male patients changed significantly over the study period, with the percentage of patients who were male increasing from 18.1% in 2005 to 23.9% in 2011. No previous studies have captured such a large cross section of the population to establish this trend. Although this trend is not necessarily intuitive, potential theories include increased acceptance of CMC interposition arthroplasty as a surgical option for male patients, and potentially a larger number of male patients seeking medical care for thumb CMC-OA in recent years.
Increases in procedure incidence were noted in all regions of the United States, but the largest percentage increase occurred in the Northeast. Despite this increase, the Northeast also had significantly lower CMC interposition arthroplasty incidence compared with all other regions and with the average procedure incidence throughout the study period—demonstrating some regional bias as to treatment of thumb CMC-OA. Unfortunately, because of database limitations and lack of specific CPT codes for other treatment options for thumb CMC-OA, we cannot ascertain if other types of surgery are more frequently used in the Northeast.
CTS and thumb CMC-OA often coexist.37 The estimated incidence of concomitant CTS in patients with CMC-OA is between 4% and 43%, but the rate of concomitant CTR and CMC interposition arthroplasty was not previously characterized in the literature.38,39 Results of the present study supported these findings; 41% of patients who underwent CMC interposition arthroplasty in our study also had a CTS diagnosis, compared with 43% in the 246-patient study by Florack and colleagues.38 We also found that 16% to 17% of patients who underwent CMC interposition arthroplasty underwent concomitant CTR; this rate remained consistent throughout the study period.
Our study demonstrated that, compared with CMC interposition arthroplasties, significantly fewer thumb CMC arthrodesis procedures were performed in the same Medicare population during the same period. Furthermore, the number of thumb CMC arthrodesis procedures declined yearly, with an overall decrease of 19% from 2005 to 2011. In a recent single-blinded, randomized trial, Vermeulen and colleagues40 compared thumb CMC arthrodesis and trapeziectomy with LRTI. They found superior patient satisfaction and significantly lower complication rates in women who underwent LRTI versus arthrodesis. The study was terminated prematurely because of these complications and thus was underpowered to determine differences in specific outcome measures. Previous studies comparing arthrodesis and interposition arthroplasties reported inconsistent outcomes. Hart and colleagues41 found no significant differences in pain or function between CMC arthrodesis and LRTI at a mean 7-year follow-up in a level II randomized controlled trial. Hartigan and colleagues15 reached similar conclusions in their retrospective comparison of the procedures. Without clear evidence supporting arthrodesis over interposition arthroplasty, the majority of surgeons favor interposition arthroplasty for thumb CMC-OA. Among Medicare patients, use of thumb CMC arthrodesis continues to fall.
This national database study had several limitations, which are common to all studies using the PearlDiver database22,42-47:
1. The power of the analysis depended on the quality of available data. Potential sources of error included accuracy of billing codes, and miscoding or noncoding by physicians.46
2. Although we used this database to try to accurately represent a large population of interest, we cannot guarantee the database represented a true cross section of the United States.
3. For the Medicare population, the PearlDiver database indexes data only in 7-year increments. Although the study period was long enough to detect significant trends, some data may not be accurately captured over a 7-year period.
4. Patients were not randomized to a treatment group.
5. The PearlDiver database does not include any clinical outcome data. Therefore, we cannot comment on the efficacy of the reported evaluations and interventions.
6. There is no specific CPT code for thumb CMC interposition arthroplasty. However, we are unaware of a CMC interposition arthroplasty performed for any area besides the thumb. Theoretically, the study population can include a negligible percentage of patients who had interposition arthroplasty of a CMC joint other than the thumb.
7. The database cannot be searched for use of thumb CMC-OA surgical techniques other than CMC interposition arthroplasty or arthrodesis, as isolated trapeziectomy, volar ligament reconstruction, implant arthroplasty, and metacarpal osteotomy lack specific CPT codes.
Conclusion
Thumb CMC-OA is a common entity among Medicare patients. There are numerous surgical options for cases that have failed conservative treatment. Despite the lack of evidence that thumb CMC interposition arthroplasty is superior to other surgical options, the number of patients who had this procedure increased 46% during the 2005–2011 study period. Although the majority of patients who undergo CMC interposition arthroplasty are female, the percentage of male patients has increased significantly. More than 40% of patients who have CMC interposition arthroplasty are also diagnosed with CTS, and 16% to 17% of patients who have CMC interposition arthroplasty will have a concomitant CTR. CMC arthrodesis is used in significantly fewer patients of Medicare age, and its use has been declining.
A common entity, osteoarthritis (OA) at the base of the thumb is largely caused by the unique anatomy and biomechanics of the thumb carpometacarpal (CMC) joint.1 Radiographically evident CMC degeneration occurs in 40% of women and 25% of men over age 75 years, making the thumb CMC joint the most common site of surgical reconstruction for upper extremity OA.2,3
Over the past 40 years, numerous surgical techniques for managing thumb CMC-OA have been described. These include volar ligament reconstruction, first metacarpal osteotomy, CMC arthrodesis, CMC joint replacement, and trapeziectomy. Trapeziectomy can be performed in isolation or in combination with tendon interposition, ligament reconstruction, or ligament reconstruction and tendon interposition (LRTI).4-20 The authors of a recent systematic review concluded there is no evidence that any one surgical procedure for CMC-OA is superior to another in terms of pain, function, satisfaction, range of motion, or strength.4 Nevertheless, a recent survey found that 719 (62%) of 1156 US hand surgeons used LRTI as the treatment of choice for advanced CMC-OA.21
Our detailed literature search yielded no other database studies characterizing current trends in the practice patterns of US orthopedic surgeons who perform interposition arthroplasty for CMC arthritis. Analysis of these trends is important not only to patients but also to the broader orthopedic and health care community.22
We conducted a study to investigate current trends in CMC interposition arthroplasty across time, sex, age, and region of the United States; per-patient charges and reimbursements; and the association between this procedure and concomitantly performed carpal tunnel syndrome (CTS) and carpal tunnel release (CTR). In addition, we compared incidence of CMC interposition arthroplasty with that of CMC arthrodesis.
Patients and Methods
All data were derived from the PearlDiver Patient Records Database (PearlDiver Technologies), a publicly available database of patients. The database stores procedure volumes, demographics, and average charge information for patients with International Classification of Diseases, Ninth Revision (ICD-9) diagnoses and procedures or Current Procedural Terminology (CPT) codes. Data for the present study were drawn from the Medicare database within the PearlDiver records, which has a total of 179,094,296 patient records covering the period 2005–2011. This study did not require institutional review board approval, as it used existing, publicly available data without identifiers linked to subjects.
PearlDiver Technologies granted us database access for academic research. The database was stored on a password-protected server maintained by PearlDiver. ICD-9 and CPT codes can be searched in isolation or in combination. Search results yield number of patients with a searched code (or combination of codes) in each year, age group, or region of the United States, as well as mean charge and mean reimbursement for the code or combination of codes.
We used CPT code 25447 (arthroplasty, interposition, intercarpal, or CMC joints) to search the database for patients who underwent thumb CMC interposition arthroplasty. Although this code does not specify thumb, we are unaware of any procedure (other than thumb CMC interposition arthroplasty) typically given this code. Our search yielded procedure volumes, sex distribution, age distribution, region volumes, and mean per-patient charges and reimbursements for each CPT code. We then searched the resulting cohort for CTS (ICD-9 code 354.0), endoscopic CTR (CPT code 29848), and open CTR (CPT code 64721) to find CTR performed concomitantly with CMC interposition arthroplasty. Last, patients were tracked in the database past their surgery date to evaluate for postoperative physical or occupational therapy evaluations within 6 months (using CPT codes appearing in at least 1% of the cohort: 97001, 97003, 97004, 97110, 97112, 97124, 97140, 97150, 97350, 97535) and postoperative thumb, hand, or wrist radiographs within 6 months (using CPT codes appearing in at least 1% of the cohort: 73140, 73130, 73110). To ensure adequacy of 6-month postoperative data, we included in this portion of the study only those patients with surgery dates between 2005 and 2010.
For comparative purposes, we also searched the database for patients who underwent thumb CMC arthrodesis within the same period—using CPT codes 26841 and 26842 (arthrodesis CMC joint thumb, with or without internal fixation; with or without autograft) and CPT code 26820 (fusion in opposition, thumb, with autogenous graft).
Overall procedure volume data are reported as number of patients with the given CPT code in the database output in a given year. Age-group and sex analyses are reported as number of patients reported in the database output and as percentage of patients who underwent the CPT code of interest that year. Mean charges and reimbursements are reported as results by the database for the code of interest (CPT 25447). Data for the region analysis are presented as an incidence, as there is an uneven distribution of patient volumes among regions. This incidence is calculated as number of patients in a particular region and year normalized to total number of patients in the database for that particular region or year. Regions are defined as Midwest (IA, IL, IN, KS, MI, MN, MO, ND, NE, OH, SD, WI), Northeast (CT, MA, ME, NH, NJ, NY, PA, RI, VT), South (AL, AR, DC, DE, FL, GA, KY, LA, MD, MS, NC, OK, SC, TN, TX, VA, WV), and West (AK, AZ, CA, CO, HI, ID, MT, NM, NV, OR, UT, WA, WY).
Chi-squared linear-by-linear association analysis was used to determine statistical significance with regard to trends over time in procedure volumes, sex, age group, and region. For all statistical comparisons, P < .05 was considered significant.
Results
In the database, we identified 41,171 unique patients who underwent CMC interposition arthroplasty between 2005 and 2011. Over the 7-year study period, number of patients who had CMC interposition arthroplasty increased 46.2%, from 4761 in 2005 to 6960 in 2011 (P < .0001) (Table 1, Figure 1). Throughout this period, females underwent CMC interposition arthroplasty more frequently than males at all time points (P < .0001). Overall ratio of female to male patients, however, changed significantly. In 2005, 18.1% of all CMC interposition arthroplasties were performed on male patients; this increased to 23.9% of all procedures by 2011 (P < .0001) (Figure 2). Table 1 presents an age-group analysis. There were no significant differences in relative percentage of patients in any given age group who underwent CMC interposition arthroplasty over the study period.
Analysis of overall procedure incidence by region revealed significant increases in all regions (P < .0001), ranging from 18.5% (West) to 54.5% (Northeast) (Figure 3). At all time points, the incidence of CMC interposition arthroplasty was significantly lower in the Northeast than in any other region and compared with the overall average.
Between 2005 and 2011, there were significant increases in both per-patient charges and reimbursements for CMC interposition arthroplasty (Figure 4). Mean per-patient charge increased from $2676 in 2005 to $4181 in 2011 (P < .0001), and mean per-patient reimbursement increased from $1445 in 2005 to $2061 in 2011 (P < .0001). The discrepancy between charge and reimbursement increased throughout the study period: Reimbursement in 2005 was 54.0% of the charge; this decreased to 49.3% by 2011 but was not statistically significant (P = .08).
Overall, 40.9% of patients who underwent CMC interposition arthroplasty also had a CTS diagnosis. Between 15.5% and 17.3% of these patients had concomitant open or endoscopic CTR at time of CMC interposition arthroplasty (Table 2). Percentage of patients who underwent concomitant CTR did not change significantly from 2005 to 2011 (P = .139). Use of postoperative occupational and/or physical therapy increased significantly over the study period, from 33.5% of patients in 2005 to 50.7% of patients in 2010 (P < .0001). Use of postoperative thumb, hand, and/or wrist radiography also increased throughout the study period, from 7.4% of patients in 2005 to 18.7% of patients in 2010 (P < .0001).
We identified 1916 unique patients who underwent thumb CMC arthrodesis between 2005 and 2011. Over the 7-year study period, there was a 19.1% decrease in number of patients who underwent CMC arthrodesis, from 309 in 2005 to 250 in 2011 (P < .0001) (Figure 5). Significantly fewer patients had CMC arthrodesis compared with CMC interposition arthroplasty at all time points, ranging from 6.5% (thumb CMC arthrodesis:CMC interposition arthroplasty) in 2005 to 3.6% in 2011 (P < .0001).
Discussion
Our results demonstrated a significant increase in use of thumb CMC interposition arthroplasty in a US Medicare population, with an increase of more than 46% from 2005 to 2011. This finding supports the findings of a recent cross-sectional survey-based study in which 719 (62%) of 1156 surveyed US hand surgeons reported performing trapeziectomy with LRTI for advanced thumb CMC-OA.21 A prior study had similar findings, with 692 (68%) of 1024 American Society for Surgery of the Hand (ASSH) members performing LRTI and 766 (75%) of 1024 performing some type of CMC interposition with trapeziectomy for advanced CMC-OA.23 This preference for CMC interposition arthroplasty prevails despite the fact that numerous studies have shown no superiority of any surgical procedure to another for CMC-OA in terms of pain, function, satisfaction, range of motion, and strength.7,15,18,19,24-34 Our data demonstrated that, not only does CMC interposition arthroplasty remain the most frequently used procedure for thumb CMC-OA, the incidence of CMC interposition arthroplasty continues to increase yearly.
The incidence of thumb CMC-OA is higher in women than in men, with more joint laxity a known contributor and subtle sex differences in trapezium geometry and congruence postulated as additional factors.3,35,36 This trend was confirmed in the present study, as females underwent significantly more CMC interposition arthroplasties at all time points. It is interesting that the overall ratio of female to male patients changed significantly over the study period, with the percentage of patients who were male increasing from 18.1% in 2005 to 23.9% in 2011. No previous studies have captured such a large cross section of the population to establish this trend. Although this trend is not necessarily intuitive, potential theories include increased acceptance of CMC interposition arthroplasty as a surgical option for male patients, and potentially a larger number of male patients seeking medical care for thumb CMC-OA in recent years.
Increases in procedure incidence were noted in all regions of the United States, but the largest percentage increase occurred in the Northeast. Despite this increase, the Northeast also had significantly lower CMC interposition arthroplasty incidence compared with all other regions and with the average procedure incidence throughout the study period—demonstrating some regional bias as to treatment of thumb CMC-OA. Unfortunately, because of database limitations and lack of specific CPT codes for other treatment options for thumb CMC-OA, we cannot ascertain if other types of surgery are more frequently used in the Northeast.
CTS and thumb CMC-OA often coexist.37 The estimated incidence of concomitant CTS in patients with CMC-OA is between 4% and 43%, but the rate of concomitant CTR and CMC interposition arthroplasty was not previously characterized in the literature.38,39 Results of the present study supported these findings; 41% of patients who underwent CMC interposition arthroplasty in our study also had a CTS diagnosis, compared with 43% in the 246-patient study by Florack and colleagues.38 We also found that 16% to 17% of patients who underwent CMC interposition arthroplasty underwent concomitant CTR; this rate remained consistent throughout the study period.
Our study demonstrated that, compared with CMC interposition arthroplasties, significantly fewer thumb CMC arthrodesis procedures were performed in the same Medicare population during the same period. Furthermore, the number of thumb CMC arthrodesis procedures declined yearly, with an overall decrease of 19% from 2005 to 2011. In a recent single-blinded, randomized trial, Vermeulen and colleagues40 compared thumb CMC arthrodesis and trapeziectomy with LRTI. They found superior patient satisfaction and significantly lower complication rates in women who underwent LRTI versus arthrodesis. The study was terminated prematurely because of these complications and thus was underpowered to determine differences in specific outcome measures. Previous studies comparing arthrodesis and interposition arthroplasties reported inconsistent outcomes. Hart and colleagues41 found no significant differences in pain or function between CMC arthrodesis and LRTI at a mean 7-year follow-up in a level II randomized controlled trial. Hartigan and colleagues15 reached similar conclusions in their retrospective comparison of the procedures. Without clear evidence supporting arthrodesis over interposition arthroplasty, the majority of surgeons favor interposition arthroplasty for thumb CMC-OA. Among Medicare patients, use of thumb CMC arthrodesis continues to fall.
This national database study had several limitations, which are common to all studies using the PearlDiver database22,42-47:
1. The power of the analysis depended on the quality of available data. Potential sources of error included accuracy of billing codes, and miscoding or noncoding by physicians.46
2. Although we used this database to try to accurately represent a large population of interest, we cannot guarantee the database represented a true cross section of the United States.
3. For the Medicare population, the PearlDiver database indexes data only in 7-year increments. Although the study period was long enough to detect significant trends, some data may not be accurately captured over a 7-year period.
4. Patients were not randomized to a treatment group.
5. The PearlDiver database does not include any clinical outcome data. Therefore, we cannot comment on the efficacy of the reported evaluations and interventions.
6. There is no specific CPT code for thumb CMC interposition arthroplasty. However, we are unaware of a CMC interposition arthroplasty performed for any area besides the thumb. Theoretically, the study population can include a negligible percentage of patients who had interposition arthroplasty of a CMC joint other than the thumb.
7. The database cannot be searched for use of thumb CMC-OA surgical techniques other than CMC interposition arthroplasty or arthrodesis, as isolated trapeziectomy, volar ligament reconstruction, implant arthroplasty, and metacarpal osteotomy lack specific CPT codes.
Conclusion
Thumb CMC-OA is a common entity among Medicare patients. There are numerous surgical options for cases that have failed conservative treatment. Despite the lack of evidence that thumb CMC interposition arthroplasty is superior to other surgical options, the number of patients who had this procedure increased 46% during the 2005–2011 study period. Although the majority of patients who undergo CMC interposition arthroplasty are female, the percentage of male patients has increased significantly. More than 40% of patients who have CMC interposition arthroplasty are also diagnosed with CTS, and 16% to 17% of patients who have CMC interposition arthroplasty will have a concomitant CTR. CMC arthrodesis is used in significantly fewer patients of Medicare age, and its use has been declining.
1. Hentz VR. Surgical treatment of trapeziometacarpal joint arthritis: a historical perspective. Clin Orthop Relat Res. 2014;472(4):1184-1189.
2. Armstrong AL, Hunter JB, Davis TR. The prevalence of degenerative arthritis of the base of the thumb in post-menopausal women. J Hand Surg Br. 1994;19(3):340-341.
3. Van Heest AE, Kallemeier P. Thumb carpal metacarpal arthritis. J Am Acad Orthop Surg. 2008;16(3):140-151.
4. Vermeulen GM, Slijper H, Feitz R, Hovius SE, Moojen TM, Selles RW. Surgical management of primary thumb carpometacarpal osteoarthritis: a systematic review. J Hand Surg Am. 2011;36(1):157-169.
5. Bodin ND, Spangler R, Thoder JJ. Interposition arthroplasty options for carpometacarpal arthritis of the thumb. Hand Clin. 2010;26(3):339-350, v-vi.
6. Cooney WP, Linscheid RL, Askew LJ. Total arthroplasty of the thumb trapeziometacarpal joint. Clin Orthop Relat Res. 1987;(220):35-45.
7. De Smet L, Vandenberghe L, Degreef I. Long-term outcome of trapeziectomy with ligament reconstruction and tendon interposition (LRTI) versus prosthesis arthroplasty for basal joint osteoarthritis of the thumb. Acta Orthop Belg. 2013;79(2):146-149.
8. Dell PC, Muniz RB. Interposition arthroplasty of the trapeziometacarpal joint for osteoarthritis. Clin Orthop Relat Res. 1987;(220):27-34.
9. Dhar S, Gray IC, Jones WA, Beddow FH. Simple excision of the trapezium for osteoarthritis of the carpometacarpal joint of the thumb. J Hand Surg Br. 1994;19(4):485-488.
10. Eaton RG, Littler JW. Ligament reconstruction for the painful thumb carpometacarpal joint. J Bone Joint Surg Am. 1973;55(8):1655-1666.
11. Eaton RG, Lane LB, Littler JW, Keyser JJ. Ligament reconstruction for the painful thumb carpometacarpal joint: a long-term assessment. J Hand Surg Am. 1984;9(5):692-699.
12. Eaton RG, Glickel SZ, Littler JW. Tendon interposition arthroplasty for degenerative arthritis of the trapeziometacarpal joint of the thumb. J Hand Surg Am. 1985;10(5):645-654.
13. Elfar JC, Burton RI. Ligament reconstruction and tendon interposition for thumb basal arthritis. Hand Clin. 2013;29(1):15-25.
14. Froimson AI. Tendon arthroplasty of the trapeziometacarpal joint. Clin Orthop Relat Res. 1970;70:191-199.
15. Hartigan BJ, Stern PJ, Kiefhaber TR. Thumb carpometacarpal osteoarthritis: arthrodesis compared with ligament reconstruction and tendon interposition. J Bone Joint Surg Am. 2001;83(10):1470-1478.
16. Kenniston JA, Bozentka DJ. Treatment of advanced carpometacarpal joint disease: arthrodesis. Hand Clin. 2008;24(3):285-294, vi-vii.
17. Kokkalis ZT, Zanaros G, Weiser RW, Sotereanos DG. Trapezium resection with suspension and interposition arthroplasty using acellular dermal allograft for thumb carpometacarpal arthritis. J Hand Surg Am. 2009;34(6):1029-1036.
18. Kriegs-Au G, Petje G, Fojtl E, Ganger R, Zachs I. Ligament reconstruction with or without tendon interposition to treat primary thumb carpometacarpal osteoarthritis. Surgical technique. J Bone Joint Surg Am. 2005;87 suppl 1(Pt 1):78-85.
19. Park MJ, Lichtman G, Christian JB, et al. Surgical treatment of thumb carpometacarpal joint arthritis: a single institution experience from 1995–2005. Hand. 2008;3(4):304-310.
20. Park MJ, Lee AT, Yao J. Treatment of thumb carpometacarpal arthritis with arthroscopic hemitrapeziectomy and interposition arthroplasty. Orthopedics. 2012;35(12):e1759-e1764.
21. Wolf JM, Delaronde S. Current trends in nonoperative and operative treatment of trapeziometacarpal osteoarthritis: a survey of US hand surgeons. J Hand Surg Am. 2012;37(1):77-82.
22. Zhang AL, Kreulen C, Ngo SS, Hame SL, Wang JC, Gamradt SC. Demographic trends in arthroscopic SLAP repair in the United States. Am J Sports Med. 2012;40(5):1144-1147.
23. Brunton LM, Wilgis EF. A survey to determine current practice patterns in the surgical treatment of advanced thumb carpometacarpal osteoarthrosis. Hand. 2010;5(4):415-422.
24. Belcher HJ, Nicholl JE. A comparison of trapeziectomy with and without ligament reconstruction and tendon interposition. J Hand Surg Br. 2000;25(4):350-356.
25. Davis TR, Pace A. Trapeziectomy for trapeziometacarpal joint osteoarthritis: is ligament reconstruction and temporary stabilisation of the pseudarthrosis with a Kirschner wire important? J Hand Surg Eur Vol. 2009;34(3):312-321.
26. Davis TR, Brady O, Dias JJ. Excision of the trapezium for osteoarthritis of the trapeziometacarpal joint: a study of the benefit of ligament reconstruction or tendon interposition. J Hand Surg Am. 2004;29(6):1069-1077.
27. De Smet L, Sioen W, Spaepen D, van Ransbeeck H. Treatment of basal joint arthritis of the thumb: trapeziectomy with or without tendon interposition/ligament reconstruction. Hand Surg. 2004;9(1):5-9.
28. Field J, Buchanan D. To suspend or not to suspend: a randomised single blind trial of simple trapeziectomy versus trapeziectomy and flexor carpi radialis suspension. J Hand Surg Eur Vol. 2007;32(4):462-466.
29. Gerwin M, Griffith A, Weiland AJ, Hotchkiss RN, McCormack RR. Ligament reconstruction basal joint arthroplasty without tendon interposition. Clin Orthop Relat Res. 1997;(342):42-45.
30. Jorheim M, Isaxon I, Flondell M, Kalen P, Atroshi I. Short-term outcomes of trapeziometacarpal Artelon implant compared with tendon suspension interposition arthroplasty for osteoarthritis: a matched cohort study. J Hand Surg Am. 2009;34(8):1381-1387.
31. Lehmann O, Herren DB, Simmen BR. Comparison of tendon suspension-interposition and silicon spacers in the treatment of degenerative osteoarthritis of the base of the thumb. Ann Chir Main Memb Super. 1998;17(1):25-30.
32. Nilsson A, Liljensten E, Bergstrom C, Sollerman C. Results from a degradable TMC joint spacer (Artelon) compared with tendon arthroplasty. J Hand Surg Am. 2005;30(2):380-389.
33. Schroder J, Kerkhoffs GM, Voerman HJ, Marti RK. Surgical treatment of basal joint disease of the thumb: comparison between resection-interposition arthroplasty and trapezio-metacarpal arthrodesis. Arch Orthop Trauma Surg. 2002;122(1):35-38.
34. Tagil M, Kopylov P. Swanson versus APL arthroplasty in the treatment of osteoarthritis of the trapeziometacarpal joint: a prospective and randomized study in 26 patients. J Hand Surg Br. 2002;27(5):452-456.
35. North ER, Rutledge WM. The trapezium-thumb metacarpal joint: the relationship of joint shape and degenerative joint disease. Hand. 1983;15(2):201-206.
36. Ateshian GA, Rosenwasser MP, Mow VC. Curvature characteristics and congruence of the thumb carpometacarpal joint: differences between female and male joints. J Biomech. 1992;25(6):591-607.
37. Sless Y, Sampson SP. Experience with transtrapezium approach for transverse carpal ligament release in patients with coexisted trapeziometacarpal joint osteoarthritis and carpal tunnel syndrome. Hand. 2007;2(3):151-154.
38. Florack TM, Miller RJ, Pellegrini VD, Burton RI, Dunn MG. The prevalence of carpal tunnel syndrome in patients with basal joint arthritis of the thumb. J Hand Surg Am. 1992;17(4):624-630.
39. Tsai TM, Laurentin-Perez LA, Wong MS, Tamai M. Ideas and innovations: radial approach to carpal tunnel release in conjunction with thumb carpometacarpal arthroplasty. Hand Surg. 2005;10(1):61-66.
40. Vermeulen GM, Brink SM, Slijper H, et al. Trapeziometacarpal arthrodesis or trapeziectomy with ligament reconstruction in primary trapeziometacarpal osteoarthritis: a randomized controlled trial. J Bone Joint Surg Am. 2014;96(9):726-733.
41. Hart R, Janecek M, Siska V, Kucera B, Stipcak V. Interposition suspension arthroplasty according to Epping versus arthrodesis for trapeziometacarpal osteoarthritis. Eur Surg. 2006;38(6):433-438.
42. Abrams GD, Frank RM, Gupta AK, Harris JD, McCormick FM, Cole BJ. Trends in meniscus repair and meniscectomy in the United States, 2005–2011. Am J Sports Med. 2013;41(10):2333-2339.
43. Montgomery SR, Ngo SS, Hobson T, et al. Trends and demographics in hip arthroscopy in the United States. Arthroscopy. 2013;29(4):661-665.
44. Zhang AL, Montgomery SR, Ngo SS, Hame SL, Wang JC, Gamradt SC. Arthroscopic versus open shoulder stabilization: current practice patterns in the United States. Arthroscopy. 2014;30(4):436-443.
45. Yeranosian MG, Arshi A, Terrell RD, Wang JC, McAllister DR, Petrigliano FA. Incidence of acute postoperative infections requiring reoperation after arthroscopic shoulder surgery. Am J Sports Med. 2014;42(2):437-441.
46. Yeranosian MG, Terrell RD, Wang JC, McAllister DR, Petrigliano FA. The costs associated with the evaluation of rotator cuff tears before surgical repair. J Shoulder Elbow Surg. 2013;22(12):1662-1666.
47. Daffner SD, Hymanson HJ, Wang JC. Cost and use of conservative management of lumbar disc herniation before surgical discectomy. Spine J. 2010;10(6):463-468.
1. Hentz VR. Surgical treatment of trapeziometacarpal joint arthritis: a historical perspective. Clin Orthop Relat Res. 2014;472(4):1184-1189.
2. Armstrong AL, Hunter JB, Davis TR. The prevalence of degenerative arthritis of the base of the thumb in post-menopausal women. J Hand Surg Br. 1994;19(3):340-341.
3. Van Heest AE, Kallemeier P. Thumb carpal metacarpal arthritis. J Am Acad Orthop Surg. 2008;16(3):140-151.
4. Vermeulen GM, Slijper H, Feitz R, Hovius SE, Moojen TM, Selles RW. Surgical management of primary thumb carpometacarpal osteoarthritis: a systematic review. J Hand Surg Am. 2011;36(1):157-169.
5. Bodin ND, Spangler R, Thoder JJ. Interposition arthroplasty options for carpometacarpal arthritis of the thumb. Hand Clin. 2010;26(3):339-350, v-vi.
6. Cooney WP, Linscheid RL, Askew LJ. Total arthroplasty of the thumb trapeziometacarpal joint. Clin Orthop Relat Res. 1987;(220):35-45.
7. De Smet L, Vandenberghe L, Degreef I. Long-term outcome of trapeziectomy with ligament reconstruction and tendon interposition (LRTI) versus prosthesis arthroplasty for basal joint osteoarthritis of the thumb. Acta Orthop Belg. 2013;79(2):146-149.
8. Dell PC, Muniz RB. Interposition arthroplasty of the trapeziometacarpal joint for osteoarthritis. Clin Orthop Relat Res. 1987;(220):27-34.
9. Dhar S, Gray IC, Jones WA, Beddow FH. Simple excision of the trapezium for osteoarthritis of the carpometacarpal joint of the thumb. J Hand Surg Br. 1994;19(4):485-488.
10. Eaton RG, Littler JW. Ligament reconstruction for the painful thumb carpometacarpal joint. J Bone Joint Surg Am. 1973;55(8):1655-1666.
11. Eaton RG, Lane LB, Littler JW, Keyser JJ. Ligament reconstruction for the painful thumb carpometacarpal joint: a long-term assessment. J Hand Surg Am. 1984;9(5):692-699.
12. Eaton RG, Glickel SZ, Littler JW. Tendon interposition arthroplasty for degenerative arthritis of the trapeziometacarpal joint of the thumb. J Hand Surg Am. 1985;10(5):645-654.
13. Elfar JC, Burton RI. Ligament reconstruction and tendon interposition for thumb basal arthritis. Hand Clin. 2013;29(1):15-25.
14. Froimson AI. Tendon arthroplasty of the trapeziometacarpal joint. Clin Orthop Relat Res. 1970;70:191-199.
15. Hartigan BJ, Stern PJ, Kiefhaber TR. Thumb carpometacarpal osteoarthritis: arthrodesis compared with ligament reconstruction and tendon interposition. J Bone Joint Surg Am. 2001;83(10):1470-1478.
16. Kenniston JA, Bozentka DJ. Treatment of advanced carpometacarpal joint disease: arthrodesis. Hand Clin. 2008;24(3):285-294, vi-vii.
17. Kokkalis ZT, Zanaros G, Weiser RW, Sotereanos DG. Trapezium resection with suspension and interposition arthroplasty using acellular dermal allograft for thumb carpometacarpal arthritis. J Hand Surg Am. 2009;34(6):1029-1036.
18. Kriegs-Au G, Petje G, Fojtl E, Ganger R, Zachs I. Ligament reconstruction with or without tendon interposition to treat primary thumb carpometacarpal osteoarthritis. Surgical technique. J Bone Joint Surg Am. 2005;87 suppl 1(Pt 1):78-85.
19. Park MJ, Lichtman G, Christian JB, et al. Surgical treatment of thumb carpometacarpal joint arthritis: a single institution experience from 1995–2005. Hand. 2008;3(4):304-310.
20. Park MJ, Lee AT, Yao J. Treatment of thumb carpometacarpal arthritis with arthroscopic hemitrapeziectomy and interposition arthroplasty. Orthopedics. 2012;35(12):e1759-e1764.
21. Wolf JM, Delaronde S. Current trends in nonoperative and operative treatment of trapeziometacarpal osteoarthritis: a survey of US hand surgeons. J Hand Surg Am. 2012;37(1):77-82.
22. Zhang AL, Kreulen C, Ngo SS, Hame SL, Wang JC, Gamradt SC. Demographic trends in arthroscopic SLAP repair in the United States. Am J Sports Med. 2012;40(5):1144-1147.
23. Brunton LM, Wilgis EF. A survey to determine current practice patterns in the surgical treatment of advanced thumb carpometacarpal osteoarthrosis. Hand. 2010;5(4):415-422.
24. Belcher HJ, Nicholl JE. A comparison of trapeziectomy with and without ligament reconstruction and tendon interposition. J Hand Surg Br. 2000;25(4):350-356.
25. Davis TR, Pace A. Trapeziectomy for trapeziometacarpal joint osteoarthritis: is ligament reconstruction and temporary stabilisation of the pseudarthrosis with a Kirschner wire important? J Hand Surg Eur Vol. 2009;34(3):312-321.
26. Davis TR, Brady O, Dias JJ. Excision of the trapezium for osteoarthritis of the trapeziometacarpal joint: a study of the benefit of ligament reconstruction or tendon interposition. J Hand Surg Am. 2004;29(6):1069-1077.
27. De Smet L, Sioen W, Spaepen D, van Ransbeeck H. Treatment of basal joint arthritis of the thumb: trapeziectomy with or without tendon interposition/ligament reconstruction. Hand Surg. 2004;9(1):5-9.
28. Field J, Buchanan D. To suspend or not to suspend: a randomised single blind trial of simple trapeziectomy versus trapeziectomy and flexor carpi radialis suspension. J Hand Surg Eur Vol. 2007;32(4):462-466.
29. Gerwin M, Griffith A, Weiland AJ, Hotchkiss RN, McCormack RR. Ligament reconstruction basal joint arthroplasty without tendon interposition. Clin Orthop Relat Res. 1997;(342):42-45.
30. Jorheim M, Isaxon I, Flondell M, Kalen P, Atroshi I. Short-term outcomes of trapeziometacarpal Artelon implant compared with tendon suspension interposition arthroplasty for osteoarthritis: a matched cohort study. J Hand Surg Am. 2009;34(8):1381-1387.
31. Lehmann O, Herren DB, Simmen BR. Comparison of tendon suspension-interposition and silicon spacers in the treatment of degenerative osteoarthritis of the base of the thumb. Ann Chir Main Memb Super. 1998;17(1):25-30.
32. Nilsson A, Liljensten E, Bergstrom C, Sollerman C. Results from a degradable TMC joint spacer (Artelon) compared with tendon arthroplasty. J Hand Surg Am. 2005;30(2):380-389.
33. Schroder J, Kerkhoffs GM, Voerman HJ, Marti RK. Surgical treatment of basal joint disease of the thumb: comparison between resection-interposition arthroplasty and trapezio-metacarpal arthrodesis. Arch Orthop Trauma Surg. 2002;122(1):35-38.
34. Tagil M, Kopylov P. Swanson versus APL arthroplasty in the treatment of osteoarthritis of the trapeziometacarpal joint: a prospective and randomized study in 26 patients. J Hand Surg Br. 2002;27(5):452-456.
35. North ER, Rutledge WM. The trapezium-thumb metacarpal joint: the relationship of joint shape and degenerative joint disease. Hand. 1983;15(2):201-206.
36. Ateshian GA, Rosenwasser MP, Mow VC. Curvature characteristics and congruence of the thumb carpometacarpal joint: differences between female and male joints. J Biomech. 1992;25(6):591-607.
37. Sless Y, Sampson SP. Experience with transtrapezium approach for transverse carpal ligament release in patients with coexisted trapeziometacarpal joint osteoarthritis and carpal tunnel syndrome. Hand. 2007;2(3):151-154.
38. Florack TM, Miller RJ, Pellegrini VD, Burton RI, Dunn MG. The prevalence of carpal tunnel syndrome in patients with basal joint arthritis of the thumb. J Hand Surg Am. 1992;17(4):624-630.
39. Tsai TM, Laurentin-Perez LA, Wong MS, Tamai M. Ideas and innovations: radial approach to carpal tunnel release in conjunction with thumb carpometacarpal arthroplasty. Hand Surg. 2005;10(1):61-66.
40. Vermeulen GM, Brink SM, Slijper H, et al. Trapeziometacarpal arthrodesis or trapeziectomy with ligament reconstruction in primary trapeziometacarpal osteoarthritis: a randomized controlled trial. J Bone Joint Surg Am. 2014;96(9):726-733.
41. Hart R, Janecek M, Siska V, Kucera B, Stipcak V. Interposition suspension arthroplasty according to Epping versus arthrodesis for trapeziometacarpal osteoarthritis. Eur Surg. 2006;38(6):433-438.
42. Abrams GD, Frank RM, Gupta AK, Harris JD, McCormick FM, Cole BJ. Trends in meniscus repair and meniscectomy in the United States, 2005–2011. Am J Sports Med. 2013;41(10):2333-2339.
43. Montgomery SR, Ngo SS, Hobson T, et al. Trends and demographics in hip arthroscopy in the United States. Arthroscopy. 2013;29(4):661-665.
44. Zhang AL, Montgomery SR, Ngo SS, Hame SL, Wang JC, Gamradt SC. Arthroscopic versus open shoulder stabilization: current practice patterns in the United States. Arthroscopy. 2014;30(4):436-443.
45. Yeranosian MG, Arshi A, Terrell RD, Wang JC, McAllister DR, Petrigliano FA. Incidence of acute postoperative infections requiring reoperation after arthroscopic shoulder surgery. Am J Sports Med. 2014;42(2):437-441.
46. Yeranosian MG, Terrell RD, Wang JC, McAllister DR, Petrigliano FA. The costs associated with the evaluation of rotator cuff tears before surgical repair. J Shoulder Elbow Surg. 2013;22(12):1662-1666.
47. Daffner SD, Hymanson HJ, Wang JC. Cost and use of conservative management of lumbar disc herniation before surgical discectomy. Spine J. 2010;10(6):463-468.