User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
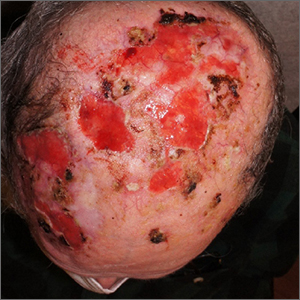
Nonhealing postsurgical scalp ulcers

Two shave biopsies were taken, 1 in the center of a previous SCC site with hyperkeratosis, the other in a site not previously affected by SCC but with the physical features of a pustule. Biopsy results from both sites were consistent with erosive pustular dermatosis, an unusual inflammatory disorder that mimics SCC.
Erosive pustular dermatosis of the scalp is an uncommon dermatitis that usually affects older women but may appear in men and women of all ages. It can mimic many other conditions that can affect the scalp, including seborrheic dermatitis, psoriasis, actinic keratosis, and SCC.
The exact causative mechanism is not understood, and cases may develop spontaneously. Rough papules, pustules, crusts, and ulcers develop and (apart from the pustules) share many features of actinic keratoses, SCCs, and field cancerization. The presence of pustules helps point to the diagnosis.
Triggers include previous surgery or physical trauma, burns, skin or hair grafts, and treatment of actinic keratoses with imiquimod, 5-fluourouracil, or photodynamic therapy. Some autoimmune diseases (including Hashimoto thyroiditis, autoimmune hepatitis, and rheumatoid arthritis) have been linked to disease occurrence and severity.1
Treatment includes potent or super-potent topical steroids such as clobetasol 0.05% ointment. Topical tacrolimus 0.1% ointment and calcipotriene 0.005% cream have been reported as steroid alternatives. Paradoxically, photodynamic therapy, while associated with triggering disease, has also been used therapeutically. Systemic immunomodulators such as cyclosporine 3 mg/kg/d or prednisone 0.5 to 1 mg/kg/d may be needed in severe cases. Antibiotics including topical dapsone 5% gel, systemic dapsone from 50 mg bid to tid, and doxycycline have been helpful due, in part, to their immunomodulatory effects.1,2
This patient was told to apply topical triamcinolone 0.1% ointment around and over ulcers and pustules and to take doxycycline 100 mg twice daily. The patient cleared well after 6 weeks. He continued to apply topical triamcinolone every few days as maintenance therapy.
He had some mild recurrence after discontinuing all topical and oral therapy, so he currently is being maintained on topical clobetasol 0.05% ointment every other day. He comes in for follow-up appointments every 3 months to monitor for control of the erosive pustular dermatosis of the scalp and for skin cancer surveillance.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME
1. Karanfilian KM, Wassef C. Erosive pustular dermatosis of the scalp: causes and treatments. Int J Dermatol. 2021;60:25-32. doi: 10.1111/ijd.14955
2. Sasaki R, Asano Y, Fujimura T. A pediatric case of corticosteroid-resistant erosive pustular dermatosis of scalp-like alopecia treated successfully with oral indomethacin, doxycycline, and topical tacrolimus. J Dermatol. 2022;49: e299-e300. doi: 10.1111/1346-8138.16425

Two shave biopsies were taken, 1 in the center of a previous SCC site with hyperkeratosis, the other in a site not previously affected by SCC but with the physical features of a pustule. Biopsy results from both sites were consistent with erosive pustular dermatosis, an unusual inflammatory disorder that mimics SCC.
Erosive pustular dermatosis of the scalp is an uncommon dermatitis that usually affects older women but may appear in men and women of all ages. It can mimic many other conditions that can affect the scalp, including seborrheic dermatitis, psoriasis, actinic keratosis, and SCC.
The exact causative mechanism is not understood, and cases may develop spontaneously. Rough papules, pustules, crusts, and ulcers develop and (apart from the pustules) share many features of actinic keratoses, SCCs, and field cancerization. The presence of pustules helps point to the diagnosis.
Triggers include previous surgery or physical trauma, burns, skin or hair grafts, and treatment of actinic keratoses with imiquimod, 5-fluourouracil, or photodynamic therapy. Some autoimmune diseases (including Hashimoto thyroiditis, autoimmune hepatitis, and rheumatoid arthritis) have been linked to disease occurrence and severity.1
Treatment includes potent or super-potent topical steroids such as clobetasol 0.05% ointment. Topical tacrolimus 0.1% ointment and calcipotriene 0.005% cream have been reported as steroid alternatives. Paradoxically, photodynamic therapy, while associated with triggering disease, has also been used therapeutically. Systemic immunomodulators such as cyclosporine 3 mg/kg/d or prednisone 0.5 to 1 mg/kg/d may be needed in severe cases. Antibiotics including topical dapsone 5% gel, systemic dapsone from 50 mg bid to tid, and doxycycline have been helpful due, in part, to their immunomodulatory effects.1,2
This patient was told to apply topical triamcinolone 0.1% ointment around and over ulcers and pustules and to take doxycycline 100 mg twice daily. The patient cleared well after 6 weeks. He continued to apply topical triamcinolone every few days as maintenance therapy.
He had some mild recurrence after discontinuing all topical and oral therapy, so he currently is being maintained on topical clobetasol 0.05% ointment every other day. He comes in for follow-up appointments every 3 months to monitor for control of the erosive pustular dermatosis of the scalp and for skin cancer surveillance.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME

Two shave biopsies were taken, 1 in the center of a previous SCC site with hyperkeratosis, the other in a site not previously affected by SCC but with the physical features of a pustule. Biopsy results from both sites were consistent with erosive pustular dermatosis, an unusual inflammatory disorder that mimics SCC.
Erosive pustular dermatosis of the scalp is an uncommon dermatitis that usually affects older women but may appear in men and women of all ages. It can mimic many other conditions that can affect the scalp, including seborrheic dermatitis, psoriasis, actinic keratosis, and SCC.
The exact causative mechanism is not understood, and cases may develop spontaneously. Rough papules, pustules, crusts, and ulcers develop and (apart from the pustules) share many features of actinic keratoses, SCCs, and field cancerization. The presence of pustules helps point to the diagnosis.
Triggers include previous surgery or physical trauma, burns, skin or hair grafts, and treatment of actinic keratoses with imiquimod, 5-fluourouracil, or photodynamic therapy. Some autoimmune diseases (including Hashimoto thyroiditis, autoimmune hepatitis, and rheumatoid arthritis) have been linked to disease occurrence and severity.1
Treatment includes potent or super-potent topical steroids such as clobetasol 0.05% ointment. Topical tacrolimus 0.1% ointment and calcipotriene 0.005% cream have been reported as steroid alternatives. Paradoxically, photodynamic therapy, while associated with triggering disease, has also been used therapeutically. Systemic immunomodulators such as cyclosporine 3 mg/kg/d or prednisone 0.5 to 1 mg/kg/d may be needed in severe cases. Antibiotics including topical dapsone 5% gel, systemic dapsone from 50 mg bid to tid, and doxycycline have been helpful due, in part, to their immunomodulatory effects.1,2
This patient was told to apply topical triamcinolone 0.1% ointment around and over ulcers and pustules and to take doxycycline 100 mg twice daily. The patient cleared well after 6 weeks. He continued to apply topical triamcinolone every few days as maintenance therapy.
He had some mild recurrence after discontinuing all topical and oral therapy, so he currently is being maintained on topical clobetasol 0.05% ointment every other day. He comes in for follow-up appointments every 3 months to monitor for control of the erosive pustular dermatosis of the scalp and for skin cancer surveillance.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME
1. Karanfilian KM, Wassef C. Erosive pustular dermatosis of the scalp: causes and treatments. Int J Dermatol. 2021;60:25-32. doi: 10.1111/ijd.14955
2. Sasaki R, Asano Y, Fujimura T. A pediatric case of corticosteroid-resistant erosive pustular dermatosis of scalp-like alopecia treated successfully with oral indomethacin, doxycycline, and topical tacrolimus. J Dermatol. 2022;49: e299-e300. doi: 10.1111/1346-8138.16425
1. Karanfilian KM, Wassef C. Erosive pustular dermatosis of the scalp: causes and treatments. Int J Dermatol. 2021;60:25-32. doi: 10.1111/ijd.14955
2. Sasaki R, Asano Y, Fujimura T. A pediatric case of corticosteroid-resistant erosive pustular dermatosis of scalp-like alopecia treated successfully with oral indomethacin, doxycycline, and topical tacrolimus. J Dermatol. 2022;49: e299-e300. doi: 10.1111/1346-8138.16425
SGLT2 inhibitors: No benefit or harm in hospitalized COVID-19
A new meta-analysis has shown that SGLT2 inhibitors do not lead to lower 28-day all-cause mortality, compared with usual care or placebo, in patients hospitalized with COVID-19.
However, no major safety issues were identified with the use of SGLT2 inhibitors in these acutely ill patients, the researchers report.
“While these findings do not support the use of SGLT2-inhibitors as standard of care for patients hospitalized with COVID-19, I think the most important take home message here is that the use of these medications appears to be safe even in really acutely ill hospitalized patients,” lead investigator of the meta-analysis, Mikhail Kosiborod, MD, Saint Luke’s Mid America Heart Institute, Kansas City, Mo., concluded.
He said this was important because the list of indications for SGLT2 inhibitors is rapidly growing.
“These medications are being used in more and more patients. And we know that when we discontinue medications in the hospital they frequently don’t get restarted, which can lead to real risks if SGLT2 inhibitors are stopped in patients with heart failure, chronic kidney disease, or diabetes. So, ,” he added.
The new meta-analysis was presented at the recent annual congress of the European Society of Cardiology, held in Amsterdam.
Discussant of the presentation at the ESC Hotline session, Muthiah Vaduganathan, MD, MPH, Brigham and Women’s Hospital, Boston, agreed with Dr. Kosiborod’s interpretation.
“Until today we have had very limited information on the safety of SGLT2-inhibitors in acute illness, as the pivotal trials which established the use of these drugs in diabetes and chronic kidney disease largely excluded patients who were hospitalized,” Dr. Vaduganathan said.
“While the overall results of this meta-analysis are neutral and SGLT2 inhibitors will not be added as drugs to be used in the primary care of patients with COVID-19, it certainly sends a strong message of safety in acutely ill patients,” he added.
Dr. Vaduganathan explained that from the beginning of the COVID-19 pandemic, there was great interest in repurposing established therapies for alternative indications for their use in the management of COVID-19.
“Conditions that strongly predispose to adverse COVID outcomes strongly overlap with established indications for SGLT2-inhibitors. So many wondered whether these drugs may be an ideal treatment candidate for the management of COVID-19. However, there have been many safety concerns about the use of SGLT2-inhibitors in this acute setting, with worries that they may induce hemodynamic changes such an excessive lowering of blood pressure, or metabolic changes such as ketoacidosis in acutely ill patients,” he noted.
The initial DARE-19 study investigating SGLT2-inhibitors in COVID-19, with 1,250 participants, found a 20% reduction in the primary outcome of organ dysfunction or death, but this did not reach statistical significance, and no safety issues were seen. This “intriguing” result led to two further larger trials – the ACTIV-4a and RECOVERY trials, Dr. Vaduganathan reported.
“Those early signals of benefit seen in DARE-19 were largely not substantiated in the ACTIV-4A and RECOVERY trials, or in this new meta-analysis, and now we have this much larger body of evidence and more stable estimates about the efficacy of these drugs in acutely ill COVID-19 patients,” he said.
“But the story that we will all take forward is one of safety. This set of trials was arguably conducted in some of the sickest patients we’ve seen who have been exposed to SGLT2-inhibitors, and they strongly affirm that these agents can be safely continued in the setting of acute illness, with very low rates of ketoacidosis and kidney injury, and there was no prolongation of hospital stay,” he commented.
In his presentation, Dr. Kosiborod explained that treatments targeting COVID-19 pathobiology such as dysregulated immune responses, endothelial damage, microvascular thrombosis, and inflammation have been shown to improve the key outcomes in this patient group.
SGLT2 inhibitors, which modulate similar pathobiology, provide cardiovascular protection and prevent the progression of kidney disease in patients at risk for these events, including those with type 2 diabetes, heart failure, and kidney disease, and may also lead to organ protection in a setting of acute illness such as COVID-19, he noted. However, the role of SGLT2 inhibitors in patients hospitalized with COVID-19 remains uncertain.
To address the need for more definitive efficacy data, the World Health Organization Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group conducted a prospective meta-analysis using data from the three randomized controlled trials, DARE-19, RECOVERY, and ACTIV-4a, evaluating SGLT2 inhibitors in patients hospitalized with COVID-19.
Overall, these trials randomized 6,096 participants: 3,025 to SGLT2 inhibitors and 3,071 to usual care or placebo. The average age of participants ranged between 62 and 73 years across the trials, 39% were women, and 25% had type 2 diabetes.
By 28 days after randomization, all-cause mortality, the primary endpoint, had occurred in 11.6% of the SGLT2-inhibitor patients, compared with 12.4% of those randomized to usual care or placebo, giving an odds ratio of 0.93 (95% confidence interval, 0.79-1.08; P = .33) for SGLT2 inhibitors, with consistency across trials.
Data on in-hospital and 90-day all-cause mortality were only available for two out of three trials (DARE-19 and ACTIV-4a), but the results were similar to the primary endpoint showing nonsignificant trends toward a possible benefit in the SGLT2-inhibitor group.
The results were also similar for the secondary outcomes of progression to acute kidney injury or requirement for dialysis or death, and progression to invasive mechanical ventilation, extracorporeal membrane oxygenation, or death, both assessed at 28 days.
The primary safety outcome of ketoacidosis by 28 days was observed in seven and two patients allocated to SGLT2 inhibitors and usual care or placebo, respectively, and overall, the incidence of reported serious adverse events was balanced between treatment groups.
The RECOVERY trial was supported by grants to the University of Oxford from UK Research and Innovation, the National Institute for Health and Care Research, and Wellcome. The ACTIV-4a platform was sponsored by the National Heart, Lung, and Blood Institute. DARE-19 was an investigator-initiated collaborative trial supported by AstraZeneca. Dr. Kosiborod reported numerous conflicts of interest.
A version of this article first appeared on Medscape.com.
A new meta-analysis has shown that SGLT2 inhibitors do not lead to lower 28-day all-cause mortality, compared with usual care or placebo, in patients hospitalized with COVID-19.
However, no major safety issues were identified with the use of SGLT2 inhibitors in these acutely ill patients, the researchers report.
“While these findings do not support the use of SGLT2-inhibitors as standard of care for patients hospitalized with COVID-19, I think the most important take home message here is that the use of these medications appears to be safe even in really acutely ill hospitalized patients,” lead investigator of the meta-analysis, Mikhail Kosiborod, MD, Saint Luke’s Mid America Heart Institute, Kansas City, Mo., concluded.
He said this was important because the list of indications for SGLT2 inhibitors is rapidly growing.
“These medications are being used in more and more patients. And we know that when we discontinue medications in the hospital they frequently don’t get restarted, which can lead to real risks if SGLT2 inhibitors are stopped in patients with heart failure, chronic kidney disease, or diabetes. So, ,” he added.
The new meta-analysis was presented at the recent annual congress of the European Society of Cardiology, held in Amsterdam.
Discussant of the presentation at the ESC Hotline session, Muthiah Vaduganathan, MD, MPH, Brigham and Women’s Hospital, Boston, agreed with Dr. Kosiborod’s interpretation.
“Until today we have had very limited information on the safety of SGLT2-inhibitors in acute illness, as the pivotal trials which established the use of these drugs in diabetes and chronic kidney disease largely excluded patients who were hospitalized,” Dr. Vaduganathan said.
“While the overall results of this meta-analysis are neutral and SGLT2 inhibitors will not be added as drugs to be used in the primary care of patients with COVID-19, it certainly sends a strong message of safety in acutely ill patients,” he added.
Dr. Vaduganathan explained that from the beginning of the COVID-19 pandemic, there was great interest in repurposing established therapies for alternative indications for their use in the management of COVID-19.
“Conditions that strongly predispose to adverse COVID outcomes strongly overlap with established indications for SGLT2-inhibitors. So many wondered whether these drugs may be an ideal treatment candidate for the management of COVID-19. However, there have been many safety concerns about the use of SGLT2-inhibitors in this acute setting, with worries that they may induce hemodynamic changes such an excessive lowering of blood pressure, or metabolic changes such as ketoacidosis in acutely ill patients,” he noted.
The initial DARE-19 study investigating SGLT2-inhibitors in COVID-19, with 1,250 participants, found a 20% reduction in the primary outcome of organ dysfunction or death, but this did not reach statistical significance, and no safety issues were seen. This “intriguing” result led to two further larger trials – the ACTIV-4a and RECOVERY trials, Dr. Vaduganathan reported.
“Those early signals of benefit seen in DARE-19 were largely not substantiated in the ACTIV-4A and RECOVERY trials, or in this new meta-analysis, and now we have this much larger body of evidence and more stable estimates about the efficacy of these drugs in acutely ill COVID-19 patients,” he said.
“But the story that we will all take forward is one of safety. This set of trials was arguably conducted in some of the sickest patients we’ve seen who have been exposed to SGLT2-inhibitors, and they strongly affirm that these agents can be safely continued in the setting of acute illness, with very low rates of ketoacidosis and kidney injury, and there was no prolongation of hospital stay,” he commented.
In his presentation, Dr. Kosiborod explained that treatments targeting COVID-19 pathobiology such as dysregulated immune responses, endothelial damage, microvascular thrombosis, and inflammation have been shown to improve the key outcomes in this patient group.
SGLT2 inhibitors, which modulate similar pathobiology, provide cardiovascular protection and prevent the progression of kidney disease in patients at risk for these events, including those with type 2 diabetes, heart failure, and kidney disease, and may also lead to organ protection in a setting of acute illness such as COVID-19, he noted. However, the role of SGLT2 inhibitors in patients hospitalized with COVID-19 remains uncertain.
To address the need for more definitive efficacy data, the World Health Organization Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group conducted a prospective meta-analysis using data from the three randomized controlled trials, DARE-19, RECOVERY, and ACTIV-4a, evaluating SGLT2 inhibitors in patients hospitalized with COVID-19.
Overall, these trials randomized 6,096 participants: 3,025 to SGLT2 inhibitors and 3,071 to usual care or placebo. The average age of participants ranged between 62 and 73 years across the trials, 39% were women, and 25% had type 2 diabetes.
By 28 days after randomization, all-cause mortality, the primary endpoint, had occurred in 11.6% of the SGLT2-inhibitor patients, compared with 12.4% of those randomized to usual care or placebo, giving an odds ratio of 0.93 (95% confidence interval, 0.79-1.08; P = .33) for SGLT2 inhibitors, with consistency across trials.
Data on in-hospital and 90-day all-cause mortality were only available for two out of three trials (DARE-19 and ACTIV-4a), but the results were similar to the primary endpoint showing nonsignificant trends toward a possible benefit in the SGLT2-inhibitor group.
The results were also similar for the secondary outcomes of progression to acute kidney injury or requirement for dialysis or death, and progression to invasive mechanical ventilation, extracorporeal membrane oxygenation, or death, both assessed at 28 days.
The primary safety outcome of ketoacidosis by 28 days was observed in seven and two patients allocated to SGLT2 inhibitors and usual care or placebo, respectively, and overall, the incidence of reported serious adverse events was balanced between treatment groups.
The RECOVERY trial was supported by grants to the University of Oxford from UK Research and Innovation, the National Institute for Health and Care Research, and Wellcome. The ACTIV-4a platform was sponsored by the National Heart, Lung, and Blood Institute. DARE-19 was an investigator-initiated collaborative trial supported by AstraZeneca. Dr. Kosiborod reported numerous conflicts of interest.
A version of this article first appeared on Medscape.com.
A new meta-analysis has shown that SGLT2 inhibitors do not lead to lower 28-day all-cause mortality, compared with usual care or placebo, in patients hospitalized with COVID-19.
However, no major safety issues were identified with the use of SGLT2 inhibitors in these acutely ill patients, the researchers report.
“While these findings do not support the use of SGLT2-inhibitors as standard of care for patients hospitalized with COVID-19, I think the most important take home message here is that the use of these medications appears to be safe even in really acutely ill hospitalized patients,” lead investigator of the meta-analysis, Mikhail Kosiborod, MD, Saint Luke’s Mid America Heart Institute, Kansas City, Mo., concluded.
He said this was important because the list of indications for SGLT2 inhibitors is rapidly growing.
“These medications are being used in more and more patients. And we know that when we discontinue medications in the hospital they frequently don’t get restarted, which can lead to real risks if SGLT2 inhibitors are stopped in patients with heart failure, chronic kidney disease, or diabetes. So, ,” he added.
The new meta-analysis was presented at the recent annual congress of the European Society of Cardiology, held in Amsterdam.
Discussant of the presentation at the ESC Hotline session, Muthiah Vaduganathan, MD, MPH, Brigham and Women’s Hospital, Boston, agreed with Dr. Kosiborod’s interpretation.
“Until today we have had very limited information on the safety of SGLT2-inhibitors in acute illness, as the pivotal trials which established the use of these drugs in diabetes and chronic kidney disease largely excluded patients who were hospitalized,” Dr. Vaduganathan said.
“While the overall results of this meta-analysis are neutral and SGLT2 inhibitors will not be added as drugs to be used in the primary care of patients with COVID-19, it certainly sends a strong message of safety in acutely ill patients,” he added.
Dr. Vaduganathan explained that from the beginning of the COVID-19 pandemic, there was great interest in repurposing established therapies for alternative indications for their use in the management of COVID-19.
“Conditions that strongly predispose to adverse COVID outcomes strongly overlap with established indications for SGLT2-inhibitors. So many wondered whether these drugs may be an ideal treatment candidate for the management of COVID-19. However, there have been many safety concerns about the use of SGLT2-inhibitors in this acute setting, with worries that they may induce hemodynamic changes such an excessive lowering of blood pressure, or metabolic changes such as ketoacidosis in acutely ill patients,” he noted.
The initial DARE-19 study investigating SGLT2-inhibitors in COVID-19, with 1,250 participants, found a 20% reduction in the primary outcome of organ dysfunction or death, but this did not reach statistical significance, and no safety issues were seen. This “intriguing” result led to two further larger trials – the ACTIV-4a and RECOVERY trials, Dr. Vaduganathan reported.
“Those early signals of benefit seen in DARE-19 were largely not substantiated in the ACTIV-4A and RECOVERY trials, or in this new meta-analysis, and now we have this much larger body of evidence and more stable estimates about the efficacy of these drugs in acutely ill COVID-19 patients,” he said.
“But the story that we will all take forward is one of safety. This set of trials was arguably conducted in some of the sickest patients we’ve seen who have been exposed to SGLT2-inhibitors, and they strongly affirm that these agents can be safely continued in the setting of acute illness, with very low rates of ketoacidosis and kidney injury, and there was no prolongation of hospital stay,” he commented.
In his presentation, Dr. Kosiborod explained that treatments targeting COVID-19 pathobiology such as dysregulated immune responses, endothelial damage, microvascular thrombosis, and inflammation have been shown to improve the key outcomes in this patient group.
SGLT2 inhibitors, which modulate similar pathobiology, provide cardiovascular protection and prevent the progression of kidney disease in patients at risk for these events, including those with type 2 diabetes, heart failure, and kidney disease, and may also lead to organ protection in a setting of acute illness such as COVID-19, he noted. However, the role of SGLT2 inhibitors in patients hospitalized with COVID-19 remains uncertain.
To address the need for more definitive efficacy data, the World Health Organization Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group conducted a prospective meta-analysis using data from the three randomized controlled trials, DARE-19, RECOVERY, and ACTIV-4a, evaluating SGLT2 inhibitors in patients hospitalized with COVID-19.
Overall, these trials randomized 6,096 participants: 3,025 to SGLT2 inhibitors and 3,071 to usual care or placebo. The average age of participants ranged between 62 and 73 years across the trials, 39% were women, and 25% had type 2 diabetes.
By 28 days after randomization, all-cause mortality, the primary endpoint, had occurred in 11.6% of the SGLT2-inhibitor patients, compared with 12.4% of those randomized to usual care or placebo, giving an odds ratio of 0.93 (95% confidence interval, 0.79-1.08; P = .33) for SGLT2 inhibitors, with consistency across trials.
Data on in-hospital and 90-day all-cause mortality were only available for two out of three trials (DARE-19 and ACTIV-4a), but the results were similar to the primary endpoint showing nonsignificant trends toward a possible benefit in the SGLT2-inhibitor group.
The results were also similar for the secondary outcomes of progression to acute kidney injury or requirement for dialysis or death, and progression to invasive mechanical ventilation, extracorporeal membrane oxygenation, or death, both assessed at 28 days.
The primary safety outcome of ketoacidosis by 28 days was observed in seven and two patients allocated to SGLT2 inhibitors and usual care or placebo, respectively, and overall, the incidence of reported serious adverse events was balanced between treatment groups.
The RECOVERY trial was supported by grants to the University of Oxford from UK Research and Innovation, the National Institute for Health and Care Research, and Wellcome. The ACTIV-4a platform was sponsored by the National Heart, Lung, and Blood Institute. DARE-19 was an investigator-initiated collaborative trial supported by AstraZeneca. Dr. Kosiborod reported numerous conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM ESC CONGRESS 2023
Lead exposure still a global health burden
TOPLINE:
Globally, lead exposure is linked to more than 5.5 million adult cardiovascular deaths in 2019, as well as loss of 765 million intelligence quotient (IQ) points in children younger than 5 years, which cost U.S. $6 trillion in lost productivity, new research suggests.
METHODOLOGY:
- Global lead exposure has declined substantially since leaded gasoline was phased out, but several sources of lead remain, resulting in adverse health and economic effects, particularly in low- and middle-income countries (LMICs).
- Estimates of cardiovascular disease (CVD) deaths from lead exposure have been limited to effects of increased blood pressure, but studies show that lead exposure has cardiovascular impacts through mechanisms other than hypertension.
- Drawing from various sources and studies, researchers estimated global blood lead levels and the impact of lead exposure on CVD mortality in 2019 among adults aged 25 years or older, IQ loss in children younger than 5 years, and the related economic costs.
TAKEAWAY:
- Researchers estimated that there were 5,545,000 (95% confidence interval, 2,305,000-8,271,000) cardiovascular deaths in adults from lead exposure in 2019, with as many as 90.2% of these deaths in LMICs; however, this estimate may be incomplete because it does not include the effect of lead exposure on CVD mortality mediated through hypertension.
- The estimated global IQ loss in children younger than 5 years due to lead exposure was 765 million (95% CI, 443 million-1,098 million) IQ points in 2019, 95.3% of which occurred in LMICs.
- These estimates place lead exposure on a par with ambient particulate matter and household air pollution combined, and ahead of unsafe household drinking water, sanitation, and handwashing, as an environmental risk factor.
- The estimated global cost of lead exposure from CVD mortality and IQ loss combined is U.S. $6.0 trillion (range, $2.6 trillion-9.0 trillion) in 2019, equivalent to 6.9% of the 2019 global gross domestic product.
IN PRACTICE:
Given the magnitude of the estimated health effects of lead exposure, particularly in LMICs, “it is imperative that nationally representative periodic blood lead level measurements be institutionalized,” write the authors, adding that these measurements could be incorporated into existing household surveys.
STUDY DETAILS:
The study was conducted by Bjorn Larsen, PhD, environmental economist and consultant to the World Bank, and Ernesto Sánchez-Triana. It was published online in The Lancet Planetary Health.
LIMITATIONS:
- Global blood lead level estimates may be inaccurate, given that measurements are absent for many countries.
- Certain income projections and income losses are uncertain.
- Because the study does not capture the detrimental effects of lead exposure other than IQ loss and CVD mortality, the estimates of global costs are conservative.
DISCLOSURES:
The study received support from the Korea Green Growth Trust Fund and the World Bank’s Pollution Management and Environmental Health Program. The authors have no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
Globally, lead exposure is linked to more than 5.5 million adult cardiovascular deaths in 2019, as well as loss of 765 million intelligence quotient (IQ) points in children younger than 5 years, which cost U.S. $6 trillion in lost productivity, new research suggests.
METHODOLOGY:
- Global lead exposure has declined substantially since leaded gasoline was phased out, but several sources of lead remain, resulting in adverse health and economic effects, particularly in low- and middle-income countries (LMICs).
- Estimates of cardiovascular disease (CVD) deaths from lead exposure have been limited to effects of increased blood pressure, but studies show that lead exposure has cardiovascular impacts through mechanisms other than hypertension.
- Drawing from various sources and studies, researchers estimated global blood lead levels and the impact of lead exposure on CVD mortality in 2019 among adults aged 25 years or older, IQ loss in children younger than 5 years, and the related economic costs.
TAKEAWAY:
- Researchers estimated that there were 5,545,000 (95% confidence interval, 2,305,000-8,271,000) cardiovascular deaths in adults from lead exposure in 2019, with as many as 90.2% of these deaths in LMICs; however, this estimate may be incomplete because it does not include the effect of lead exposure on CVD mortality mediated through hypertension.
- The estimated global IQ loss in children younger than 5 years due to lead exposure was 765 million (95% CI, 443 million-1,098 million) IQ points in 2019, 95.3% of which occurred in LMICs.
- These estimates place lead exposure on a par with ambient particulate matter and household air pollution combined, and ahead of unsafe household drinking water, sanitation, and handwashing, as an environmental risk factor.
- The estimated global cost of lead exposure from CVD mortality and IQ loss combined is U.S. $6.0 trillion (range, $2.6 trillion-9.0 trillion) in 2019, equivalent to 6.9% of the 2019 global gross domestic product.
IN PRACTICE:
Given the magnitude of the estimated health effects of lead exposure, particularly in LMICs, “it is imperative that nationally representative periodic blood lead level measurements be institutionalized,” write the authors, adding that these measurements could be incorporated into existing household surveys.
STUDY DETAILS:
The study was conducted by Bjorn Larsen, PhD, environmental economist and consultant to the World Bank, and Ernesto Sánchez-Triana. It was published online in The Lancet Planetary Health.
LIMITATIONS:
- Global blood lead level estimates may be inaccurate, given that measurements are absent for many countries.
- Certain income projections and income losses are uncertain.
- Because the study does not capture the detrimental effects of lead exposure other than IQ loss and CVD mortality, the estimates of global costs are conservative.
DISCLOSURES:
The study received support from the Korea Green Growth Trust Fund and the World Bank’s Pollution Management and Environmental Health Program. The authors have no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
Globally, lead exposure is linked to more than 5.5 million adult cardiovascular deaths in 2019, as well as loss of 765 million intelligence quotient (IQ) points in children younger than 5 years, which cost U.S. $6 trillion in lost productivity, new research suggests.
METHODOLOGY:
- Global lead exposure has declined substantially since leaded gasoline was phased out, but several sources of lead remain, resulting in adverse health and economic effects, particularly in low- and middle-income countries (LMICs).
- Estimates of cardiovascular disease (CVD) deaths from lead exposure have been limited to effects of increased blood pressure, but studies show that lead exposure has cardiovascular impacts through mechanisms other than hypertension.
- Drawing from various sources and studies, researchers estimated global blood lead levels and the impact of lead exposure on CVD mortality in 2019 among adults aged 25 years or older, IQ loss in children younger than 5 years, and the related economic costs.
TAKEAWAY:
- Researchers estimated that there were 5,545,000 (95% confidence interval, 2,305,000-8,271,000) cardiovascular deaths in adults from lead exposure in 2019, with as many as 90.2% of these deaths in LMICs; however, this estimate may be incomplete because it does not include the effect of lead exposure on CVD mortality mediated through hypertension.
- The estimated global IQ loss in children younger than 5 years due to lead exposure was 765 million (95% CI, 443 million-1,098 million) IQ points in 2019, 95.3% of which occurred in LMICs.
- These estimates place lead exposure on a par with ambient particulate matter and household air pollution combined, and ahead of unsafe household drinking water, sanitation, and handwashing, as an environmental risk factor.
- The estimated global cost of lead exposure from CVD mortality and IQ loss combined is U.S. $6.0 trillion (range, $2.6 trillion-9.0 trillion) in 2019, equivalent to 6.9% of the 2019 global gross domestic product.
IN PRACTICE:
Given the magnitude of the estimated health effects of lead exposure, particularly in LMICs, “it is imperative that nationally representative periodic blood lead level measurements be institutionalized,” write the authors, adding that these measurements could be incorporated into existing household surveys.
STUDY DETAILS:
The study was conducted by Bjorn Larsen, PhD, environmental economist and consultant to the World Bank, and Ernesto Sánchez-Triana. It was published online in The Lancet Planetary Health.
LIMITATIONS:
- Global blood lead level estimates may be inaccurate, given that measurements are absent for many countries.
- Certain income projections and income losses are uncertain.
- Because the study does not capture the detrimental effects of lead exposure other than IQ loss and CVD mortality, the estimates of global costs are conservative.
DISCLOSURES:
The study received support from the Korea Green Growth Trust Fund and the World Bank’s Pollution Management and Environmental Health Program. The authors have no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Heart attack deaths static in those with type 1 diabetes
, new research shows.
Between 2006 and 2020, the annual incidences of overall mortality and major adverse cardiovascular events after a first-time myocardial infarction dropped significantly for people with type 2 diabetes and those without diabetes (controls).
However, the same trend was not seen for people with type 1 diabetes.
“There is an urgent need for further studies understanding cardiovascular disease in people with type 1 diabetes. Clinicians have to be aware of the absence of the declined mortality trend in people with type 1 diabetes having a first-time myocardial infarction,” lead author Thomas Nyström, MD, professor of medicine at the Karolinska Institute, Stockholm, said in an interview.
The findings are scheduled to be presented Oct. 5, 2023, at the annual meeting of the European Association for the Study of Diabetes.
Discussing potential reasons for the findings, the authors say that the standard care after a heart attack has improved with more availability of, for example, percutaneous coronary intervention and better overall medical treatment. However, this standard of care should have improved in all three groups.
“Although glycemic control and diabetes duration were much different between diabetes groups, in that those with type 1 had been exposed for a longer period of glycemia, the current study cannot tell whether glucose control is behind the association between mortality trends observed. Whether this is the case must be investigated with further studies,” Nyström said.
Data from Swedish health care registry
Among people with a first-time MI recorded in national Swedish health care registries between 2006 and 2020, there were 2,527 individuals with type 1 diabetes, 48,321 with type 2 diabetes, and 243,170 controls with neither form of diabetes.
Those with type 1 diabetes were younger than those with type 2 diabetes and controls (62 years vs. 75 and 73 years, respectively). The type 1 diabetes group also had a higher proportion of females (43.6% vs. 38.1% of both the type 2 diabetes and control groups).
The proportions of people with the most severe type of heart attack, ST-elevation MI (STEMI), versus non-STEMI were 29% versus 71% in the type 1 diabetes group, 30% versus 70% in the type 2 diabetes group, and 39% versus 61% in the control group, respectively.
After adjustment for covariates including age, sex, comorbidities, socioeconomic factors, and medication, there was a significant decreased annual incidence trend for all-cause death among the controls (–1.9%) and persons with type 2 diabetes (–1.3%), but there was no such decrease among those with type 1 diabetes.
For cardiovascular deaths, the annual incidence declines were –2.0% and –1.6% in the control group and the type 2 diabetes group, respectively, versus a nonsignificant –0.5% decline in the type 1 diabetes group. Similarly, for major adverse cardiovascular events, those decreases were –2.0% for controls and –1.6% for those with type 2 diabetes, but –0.6% for those with type 1 diabetes – again, a nonsignificant value.
“During the last 15 years, the risk of death and major cardiovascular events in people without diabetes and with type 2 diabetes after having a first-time heart attack has decreased significantly. In contrast, this decreasing trend was absent in people with type 1 diabetes. Our study highlights the urgent need for understanding the cardiovascular risk in people with type 1 diabetes,” the authors conclude.
Dr. Nyström has received honoraria from AstraZeneca, Merck Sharp & Dohme, Novo Nordisk, Eli Lilly , Boehringer Ingelheim, Abbott, and Amgen. The authors acknowledge the ALF agreement between Stockholm County Council and Karolinska Institutet.
A version of this article appeared on Medscape.com.
, new research shows.
Between 2006 and 2020, the annual incidences of overall mortality and major adverse cardiovascular events after a first-time myocardial infarction dropped significantly for people with type 2 diabetes and those without diabetes (controls).
However, the same trend was not seen for people with type 1 diabetes.
“There is an urgent need for further studies understanding cardiovascular disease in people with type 1 diabetes. Clinicians have to be aware of the absence of the declined mortality trend in people with type 1 diabetes having a first-time myocardial infarction,” lead author Thomas Nyström, MD, professor of medicine at the Karolinska Institute, Stockholm, said in an interview.
The findings are scheduled to be presented Oct. 5, 2023, at the annual meeting of the European Association for the Study of Diabetes.
Discussing potential reasons for the findings, the authors say that the standard care after a heart attack has improved with more availability of, for example, percutaneous coronary intervention and better overall medical treatment. However, this standard of care should have improved in all three groups.
“Although glycemic control and diabetes duration were much different between diabetes groups, in that those with type 1 had been exposed for a longer period of glycemia, the current study cannot tell whether glucose control is behind the association between mortality trends observed. Whether this is the case must be investigated with further studies,” Nyström said.
Data from Swedish health care registry
Among people with a first-time MI recorded in national Swedish health care registries between 2006 and 2020, there were 2,527 individuals with type 1 diabetes, 48,321 with type 2 diabetes, and 243,170 controls with neither form of diabetes.
Those with type 1 diabetes were younger than those with type 2 diabetes and controls (62 years vs. 75 and 73 years, respectively). The type 1 diabetes group also had a higher proportion of females (43.6% vs. 38.1% of both the type 2 diabetes and control groups).
The proportions of people with the most severe type of heart attack, ST-elevation MI (STEMI), versus non-STEMI were 29% versus 71% in the type 1 diabetes group, 30% versus 70% in the type 2 diabetes group, and 39% versus 61% in the control group, respectively.
After adjustment for covariates including age, sex, comorbidities, socioeconomic factors, and medication, there was a significant decreased annual incidence trend for all-cause death among the controls (–1.9%) and persons with type 2 diabetes (–1.3%), but there was no such decrease among those with type 1 diabetes.
For cardiovascular deaths, the annual incidence declines were –2.0% and –1.6% in the control group and the type 2 diabetes group, respectively, versus a nonsignificant –0.5% decline in the type 1 diabetes group. Similarly, for major adverse cardiovascular events, those decreases were –2.0% for controls and –1.6% for those with type 2 diabetes, but –0.6% for those with type 1 diabetes – again, a nonsignificant value.
“During the last 15 years, the risk of death and major cardiovascular events in people without diabetes and with type 2 diabetes after having a first-time heart attack has decreased significantly. In contrast, this decreasing trend was absent in people with type 1 diabetes. Our study highlights the urgent need for understanding the cardiovascular risk in people with type 1 diabetes,” the authors conclude.
Dr. Nyström has received honoraria from AstraZeneca, Merck Sharp & Dohme, Novo Nordisk, Eli Lilly , Boehringer Ingelheim, Abbott, and Amgen. The authors acknowledge the ALF agreement between Stockholm County Council and Karolinska Institutet.
A version of this article appeared on Medscape.com.
, new research shows.
Between 2006 and 2020, the annual incidences of overall mortality and major adverse cardiovascular events after a first-time myocardial infarction dropped significantly for people with type 2 diabetes and those without diabetes (controls).
However, the same trend was not seen for people with type 1 diabetes.
“There is an urgent need for further studies understanding cardiovascular disease in people with type 1 diabetes. Clinicians have to be aware of the absence of the declined mortality trend in people with type 1 diabetes having a first-time myocardial infarction,” lead author Thomas Nyström, MD, professor of medicine at the Karolinska Institute, Stockholm, said in an interview.
The findings are scheduled to be presented Oct. 5, 2023, at the annual meeting of the European Association for the Study of Diabetes.
Discussing potential reasons for the findings, the authors say that the standard care after a heart attack has improved with more availability of, for example, percutaneous coronary intervention and better overall medical treatment. However, this standard of care should have improved in all three groups.
“Although glycemic control and diabetes duration were much different between diabetes groups, in that those with type 1 had been exposed for a longer period of glycemia, the current study cannot tell whether glucose control is behind the association between mortality trends observed. Whether this is the case must be investigated with further studies,” Nyström said.
Data from Swedish health care registry
Among people with a first-time MI recorded in national Swedish health care registries between 2006 and 2020, there were 2,527 individuals with type 1 diabetes, 48,321 with type 2 diabetes, and 243,170 controls with neither form of diabetes.
Those with type 1 diabetes were younger than those with type 2 diabetes and controls (62 years vs. 75 and 73 years, respectively). The type 1 diabetes group also had a higher proportion of females (43.6% vs. 38.1% of both the type 2 diabetes and control groups).
The proportions of people with the most severe type of heart attack, ST-elevation MI (STEMI), versus non-STEMI were 29% versus 71% in the type 1 diabetes group, 30% versus 70% in the type 2 diabetes group, and 39% versus 61% in the control group, respectively.
After adjustment for covariates including age, sex, comorbidities, socioeconomic factors, and medication, there was a significant decreased annual incidence trend for all-cause death among the controls (–1.9%) and persons with type 2 diabetes (–1.3%), but there was no such decrease among those with type 1 diabetes.
For cardiovascular deaths, the annual incidence declines were –2.0% and –1.6% in the control group and the type 2 diabetes group, respectively, versus a nonsignificant –0.5% decline in the type 1 diabetes group. Similarly, for major adverse cardiovascular events, those decreases were –2.0% for controls and –1.6% for those with type 2 diabetes, but –0.6% for those with type 1 diabetes – again, a nonsignificant value.
“During the last 15 years, the risk of death and major cardiovascular events in people without diabetes and with type 2 diabetes after having a first-time heart attack has decreased significantly. In contrast, this decreasing trend was absent in people with type 1 diabetes. Our study highlights the urgent need for understanding the cardiovascular risk in people with type 1 diabetes,” the authors conclude.
Dr. Nyström has received honoraria from AstraZeneca, Merck Sharp & Dohme, Novo Nordisk, Eli Lilly , Boehringer Ingelheim, Abbott, and Amgen. The authors acknowledge the ALF agreement between Stockholm County Council and Karolinska Institutet.
A version of this article appeared on Medscape.com.
FROM EASD 2023
Cold weather may challenge blood pressure control
A review of electronic health records of more than 60,000 U.S. adults being treated for hypertension found that on average, systolic BP rose by up to 1.7 mm Hg in the cold winter months, compared with the hot summer months.
On a population level, BP control rates decreased by up to 5% during the cold winter months, compared with control rates in the warm summer months.
“Some patients may benefit from increased pharmacological intervention to keep blood pressure controlled during the winter,” Robert Barrett, with the American Medical Association, Greenville, S.C., told this news organization.
“Individuals with hypertension or values near the range of hypertension may benefit from periodic blood pressure monitoring and improvements in physical activity and nutritional patterns during winter months to offset adverse effects from seasonal blood pressure changes,” Mr. Barrett added in a news release.
Mr. Barrett presented the study findings at the American Heart Association Hypertension Scientific Sessions 2023 in Boston.
Supportive data
Mr. Barrett explained that seasonal variation in BP has been previously documented, and as part of the evaluation for the AMA MAP Hypertension program, he and colleagues were interested in the effect of this variation on population control rates under standard metrics (visits with BP < 140/90 mm Hg).
They analyzed data from 60,676 men and women (mean age, 62 years) with hypertension from six health care organizations in the southeastern and midwestern United States that were participating in the quality improvement program.
During the roughly 5-year assessment period, none of the patients had changes in their antihypertensive medication, and all had at least one visit in each temperate season. The researchers estimated the seasonal effect on average systolic BP and BP control (defined as < 140/90 mm Hg).
Across a total of 453,787 visits, systolic BP during the winter averaged 0.47 mm Hg higher (95% confidence interval, 0.364-0.573) than the yearly average, with a significantly lower odds ratio for BP control (OR, 0.92; 95% CI, 0.91-0.94), the researchers report.
In contrast, average systolic BP was 0.92 mm Hg lower during the summer, with a higher likelihood of BP control (OR ,1.10; 95% CI, 1.07-1.12).
“Seasonal variation in blood pressure has a substantial effect on hypertension control, often defined as blood pressure < 140/90,” Barrett told this news organization.
“Patients with hypertension are less likely to have their blood pressure controlled during winter than summer months. If the blood pressure is very well controlled, for example to < 130/80, then seasonal variation will have little effect on control to < 140/90,” Mr. Barrett noted.
“However, if blood pressure is not well controlled, then patients near the 140/90 level could benefit from monitoring their blood pressure regularly, closer medical follow-up, and avoiding decreased physical activity and increased weight toward year end,” he added.
Wanpen Vongpatanasin, MD, clinical chair for the conference, said that it’s “well known that BP tends to lower during summer months and patients may be susceptible to dehydration and acute kidney injury when BP is too low, particularly when treated with certain medication such as diuretics.”
On the flip side, “cold weather predisposes to vasoconstriction as our blood vessel constrict to maintain core temperature and it could be challenging to manage BP. That’s why it is important for high BP patients to monitor home BP regularly,” said Dr. Vongpatanasin, professor of internal medicine and director of the hypertension section, cardiology division, UT Southwestern Medical Center, Dallas.
The study had no commercial funding. Mr. Barrett and Dr. Vongpatanasin have no relevant disclosures.
A version of this article first appeared on Medscape.com.
A review of electronic health records of more than 60,000 U.S. adults being treated for hypertension found that on average, systolic BP rose by up to 1.7 mm Hg in the cold winter months, compared with the hot summer months.
On a population level, BP control rates decreased by up to 5% during the cold winter months, compared with control rates in the warm summer months.
“Some patients may benefit from increased pharmacological intervention to keep blood pressure controlled during the winter,” Robert Barrett, with the American Medical Association, Greenville, S.C., told this news organization.
“Individuals with hypertension or values near the range of hypertension may benefit from periodic blood pressure monitoring and improvements in physical activity and nutritional patterns during winter months to offset adverse effects from seasonal blood pressure changes,” Mr. Barrett added in a news release.
Mr. Barrett presented the study findings at the American Heart Association Hypertension Scientific Sessions 2023 in Boston.
Supportive data
Mr. Barrett explained that seasonal variation in BP has been previously documented, and as part of the evaluation for the AMA MAP Hypertension program, he and colleagues were interested in the effect of this variation on population control rates under standard metrics (visits with BP < 140/90 mm Hg).
They analyzed data from 60,676 men and women (mean age, 62 years) with hypertension from six health care organizations in the southeastern and midwestern United States that were participating in the quality improvement program.
During the roughly 5-year assessment period, none of the patients had changes in their antihypertensive medication, and all had at least one visit in each temperate season. The researchers estimated the seasonal effect on average systolic BP and BP control (defined as < 140/90 mm Hg).
Across a total of 453,787 visits, systolic BP during the winter averaged 0.47 mm Hg higher (95% confidence interval, 0.364-0.573) than the yearly average, with a significantly lower odds ratio for BP control (OR, 0.92; 95% CI, 0.91-0.94), the researchers report.
In contrast, average systolic BP was 0.92 mm Hg lower during the summer, with a higher likelihood of BP control (OR ,1.10; 95% CI, 1.07-1.12).
“Seasonal variation in blood pressure has a substantial effect on hypertension control, often defined as blood pressure < 140/90,” Barrett told this news organization.
“Patients with hypertension are less likely to have their blood pressure controlled during winter than summer months. If the blood pressure is very well controlled, for example to < 130/80, then seasonal variation will have little effect on control to < 140/90,” Mr. Barrett noted.
“However, if blood pressure is not well controlled, then patients near the 140/90 level could benefit from monitoring their blood pressure regularly, closer medical follow-up, and avoiding decreased physical activity and increased weight toward year end,” he added.
Wanpen Vongpatanasin, MD, clinical chair for the conference, said that it’s “well known that BP tends to lower during summer months and patients may be susceptible to dehydration and acute kidney injury when BP is too low, particularly when treated with certain medication such as diuretics.”
On the flip side, “cold weather predisposes to vasoconstriction as our blood vessel constrict to maintain core temperature and it could be challenging to manage BP. That’s why it is important for high BP patients to monitor home BP regularly,” said Dr. Vongpatanasin, professor of internal medicine and director of the hypertension section, cardiology division, UT Southwestern Medical Center, Dallas.
The study had no commercial funding. Mr. Barrett and Dr. Vongpatanasin have no relevant disclosures.
A version of this article first appeared on Medscape.com.
A review of electronic health records of more than 60,000 U.S. adults being treated for hypertension found that on average, systolic BP rose by up to 1.7 mm Hg in the cold winter months, compared with the hot summer months.
On a population level, BP control rates decreased by up to 5% during the cold winter months, compared with control rates in the warm summer months.
“Some patients may benefit from increased pharmacological intervention to keep blood pressure controlled during the winter,” Robert Barrett, with the American Medical Association, Greenville, S.C., told this news organization.
“Individuals with hypertension or values near the range of hypertension may benefit from periodic blood pressure monitoring and improvements in physical activity and nutritional patterns during winter months to offset adverse effects from seasonal blood pressure changes,” Mr. Barrett added in a news release.
Mr. Barrett presented the study findings at the American Heart Association Hypertension Scientific Sessions 2023 in Boston.
Supportive data
Mr. Barrett explained that seasonal variation in BP has been previously documented, and as part of the evaluation for the AMA MAP Hypertension program, he and colleagues were interested in the effect of this variation on population control rates under standard metrics (visits with BP < 140/90 mm Hg).
They analyzed data from 60,676 men and women (mean age, 62 years) with hypertension from six health care organizations in the southeastern and midwestern United States that were participating in the quality improvement program.
During the roughly 5-year assessment period, none of the patients had changes in their antihypertensive medication, and all had at least one visit in each temperate season. The researchers estimated the seasonal effect on average systolic BP and BP control (defined as < 140/90 mm Hg).
Across a total of 453,787 visits, systolic BP during the winter averaged 0.47 mm Hg higher (95% confidence interval, 0.364-0.573) than the yearly average, with a significantly lower odds ratio for BP control (OR, 0.92; 95% CI, 0.91-0.94), the researchers report.
In contrast, average systolic BP was 0.92 mm Hg lower during the summer, with a higher likelihood of BP control (OR ,1.10; 95% CI, 1.07-1.12).
“Seasonal variation in blood pressure has a substantial effect on hypertension control, often defined as blood pressure < 140/90,” Barrett told this news organization.
“Patients with hypertension are less likely to have their blood pressure controlled during winter than summer months. If the blood pressure is very well controlled, for example to < 130/80, then seasonal variation will have little effect on control to < 140/90,” Mr. Barrett noted.
“However, if blood pressure is not well controlled, then patients near the 140/90 level could benefit from monitoring their blood pressure regularly, closer medical follow-up, and avoiding decreased physical activity and increased weight toward year end,” he added.
Wanpen Vongpatanasin, MD, clinical chair for the conference, said that it’s “well known that BP tends to lower during summer months and patients may be susceptible to dehydration and acute kidney injury when BP is too low, particularly when treated with certain medication such as diuretics.”
On the flip side, “cold weather predisposes to vasoconstriction as our blood vessel constrict to maintain core temperature and it could be challenging to manage BP. That’s why it is important for high BP patients to monitor home BP regularly,” said Dr. Vongpatanasin, professor of internal medicine and director of the hypertension section, cardiology division, UT Southwestern Medical Center, Dallas.
The study had no commercial funding. Mr. Barrett and Dr. Vongpatanasin have no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM HYPERTENSION 2023
Turmeric may be as effective as omeprazole for dyspepsia
TOPLINE:
METHODOLOGY:
- The researchers randomly assigned 206 patients to receive curcumin – the active ingredient in turmeric – alone; omeprazole alone; or curcumin plus omeprazole for 28 days. A total of 151 patients completed the study.
- Doses were two 250-mg curcumin pills four times daily, plus one placebo pill; one 20-mg omeprazole pill daily, plus two placebo pills four times daily; or two 250-mg curcumin pills four times daily, plus one 20-mg omeprazole pill once daily.
- Symptoms of functional dyspepsia were assessed on days 28 and 56 using the Severity of Dyspepsia Assessment (SODA) score.
TAKEAWAY:
- In the combined group, the curcumin-alone group, and the omeprazole-alone group, SODA scores for pain severity declined significantly by day 28 (–4.83, –5.46, and –6.22, respectively), as did scores for severity of other symptoms (–2.22, –2.32, and –2.31, respectively).
- Symptom improvements were even stronger by day 56 for pain (–7.19, –8.07, –8.85) and other symptoms (–4.09, –4.12, –3.71) in the same groups.
- Curcumin was safe and well tolerated, but satisfaction scores did not change significantly over time among those taking it, suggesting the possible need for improvement in its taste or smell.
- There was no synergistic effect between omeprazole and curcumin.
IN PRACTICE:
“The new findings from our study may justify considering curcumin in clinical practice. This multicenter, randomized, controlled trial provides highly reliable evidence for the treatment of functional dyspepsia,” the authors wrote.
SOURCE:
Pradermchai Kongkam, MD, of Chulalongkorn University, Bangkok, and Wichittra Khongkha of Chao Phraya Abhaibhubejhr Hospital, Prachin Buri, Thailand, are joint first authors. The study was published online in BMJ Evidence-Based Medicine.
LIMITATIONS:
A small number of participants in each group were lost to follow-up, and the follow-up period was short (less than 2 months) for all.
DISCLOSURES:
The study was funded by the Thai Traditional and Alternative Medicine Fund. The authors have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The researchers randomly assigned 206 patients to receive curcumin – the active ingredient in turmeric – alone; omeprazole alone; or curcumin plus omeprazole for 28 days. A total of 151 patients completed the study.
- Doses were two 250-mg curcumin pills four times daily, plus one placebo pill; one 20-mg omeprazole pill daily, plus two placebo pills four times daily; or two 250-mg curcumin pills four times daily, plus one 20-mg omeprazole pill once daily.
- Symptoms of functional dyspepsia were assessed on days 28 and 56 using the Severity of Dyspepsia Assessment (SODA) score.
TAKEAWAY:
- In the combined group, the curcumin-alone group, and the omeprazole-alone group, SODA scores for pain severity declined significantly by day 28 (–4.83, –5.46, and –6.22, respectively), as did scores for severity of other symptoms (–2.22, –2.32, and –2.31, respectively).
- Symptom improvements were even stronger by day 56 for pain (–7.19, –8.07, –8.85) and other symptoms (–4.09, –4.12, –3.71) in the same groups.
- Curcumin was safe and well tolerated, but satisfaction scores did not change significantly over time among those taking it, suggesting the possible need for improvement in its taste or smell.
- There was no synergistic effect between omeprazole and curcumin.
IN PRACTICE:
“The new findings from our study may justify considering curcumin in clinical practice. This multicenter, randomized, controlled trial provides highly reliable evidence for the treatment of functional dyspepsia,” the authors wrote.
SOURCE:
Pradermchai Kongkam, MD, of Chulalongkorn University, Bangkok, and Wichittra Khongkha of Chao Phraya Abhaibhubejhr Hospital, Prachin Buri, Thailand, are joint first authors. The study was published online in BMJ Evidence-Based Medicine.
LIMITATIONS:
A small number of participants in each group were lost to follow-up, and the follow-up period was short (less than 2 months) for all.
DISCLOSURES:
The study was funded by the Thai Traditional and Alternative Medicine Fund. The authors have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The researchers randomly assigned 206 patients to receive curcumin – the active ingredient in turmeric – alone; omeprazole alone; or curcumin plus omeprazole for 28 days. A total of 151 patients completed the study.
- Doses were two 250-mg curcumin pills four times daily, plus one placebo pill; one 20-mg omeprazole pill daily, plus two placebo pills four times daily; or two 250-mg curcumin pills four times daily, plus one 20-mg omeprazole pill once daily.
- Symptoms of functional dyspepsia were assessed on days 28 and 56 using the Severity of Dyspepsia Assessment (SODA) score.
TAKEAWAY:
- In the combined group, the curcumin-alone group, and the omeprazole-alone group, SODA scores for pain severity declined significantly by day 28 (–4.83, –5.46, and –6.22, respectively), as did scores for severity of other symptoms (–2.22, –2.32, and –2.31, respectively).
- Symptom improvements were even stronger by day 56 for pain (–7.19, –8.07, –8.85) and other symptoms (–4.09, –4.12, –3.71) in the same groups.
- Curcumin was safe and well tolerated, but satisfaction scores did not change significantly over time among those taking it, suggesting the possible need for improvement in its taste or smell.
- There was no synergistic effect between omeprazole and curcumin.
IN PRACTICE:
“The new findings from our study may justify considering curcumin in clinical practice. This multicenter, randomized, controlled trial provides highly reliable evidence for the treatment of functional dyspepsia,” the authors wrote.
SOURCE:
Pradermchai Kongkam, MD, of Chulalongkorn University, Bangkok, and Wichittra Khongkha of Chao Phraya Abhaibhubejhr Hospital, Prachin Buri, Thailand, are joint first authors. The study was published online in BMJ Evidence-Based Medicine.
LIMITATIONS:
A small number of participants in each group were lost to follow-up, and the follow-up period was short (less than 2 months) for all.
DISCLOSURES:
The study was funded by the Thai Traditional and Alternative Medicine Fund. The authors have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM BMJ EVIDENCE-BASED MEDICINE
Company submits supplemental NDA for topical atopic dermatitis treatment
in adults and children aged 6 years and older.
Roflumilast cream 0.3% (Zoryve) is currently approved by the FDA for the topical treatment of plaque psoriasis, including intertriginous areas, in patients 12 years of age and older. Submission of the sNDA is based on positive results from the Interventional Trial Evaluating Roflumilast Cream for the Treatment of Atopic Dermatitis (INTEGUMENT-1 and INTEGUMENT-2) trials; two identical Phase 3, vehicle-controlled trials in which roflumilast cream 0.15% or vehicle was applied once daily for 4 weeks to individuals 6 years of age and older with mild to moderate AD involving at least 3% body surface area. Roflumilast is a phosphodiesterase-4 (PDE-4) inhibitor.
According to a press release from Arcutis, both studies met the primary endpoint of IGA Success, which was defined as a validated Investigator Global Assessment – Atopic Dermatitis (vIGA-AD) score of ‘clear’ or ‘almost clear’ plus a 2-grade improvement from baseline at week 4. In INTEGUMENT-1 this endpoint was achieved by 32.0% of subjects in the roflumilast cream group vs. 15.2% of those in the vehicle group (P < .0001). In INTEGUMENT-2, this endpoint was achieved by 28.9% of subjects in the roflumilast cream group vs. 12.0% of those in the vehicle group (P < .0001). The most common adverse reactions based on data from the combined trials were headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
in adults and children aged 6 years and older.
Roflumilast cream 0.3% (Zoryve) is currently approved by the FDA for the topical treatment of plaque psoriasis, including intertriginous areas, in patients 12 years of age and older. Submission of the sNDA is based on positive results from the Interventional Trial Evaluating Roflumilast Cream for the Treatment of Atopic Dermatitis (INTEGUMENT-1 and INTEGUMENT-2) trials; two identical Phase 3, vehicle-controlled trials in which roflumilast cream 0.15% or vehicle was applied once daily for 4 weeks to individuals 6 years of age and older with mild to moderate AD involving at least 3% body surface area. Roflumilast is a phosphodiesterase-4 (PDE-4) inhibitor.
According to a press release from Arcutis, both studies met the primary endpoint of IGA Success, which was defined as a validated Investigator Global Assessment – Atopic Dermatitis (vIGA-AD) score of ‘clear’ or ‘almost clear’ plus a 2-grade improvement from baseline at week 4. In INTEGUMENT-1 this endpoint was achieved by 32.0% of subjects in the roflumilast cream group vs. 15.2% of those in the vehicle group (P < .0001). In INTEGUMENT-2, this endpoint was achieved by 28.9% of subjects in the roflumilast cream group vs. 12.0% of those in the vehicle group (P < .0001). The most common adverse reactions based on data from the combined trials were headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
in adults and children aged 6 years and older.
Roflumilast cream 0.3% (Zoryve) is currently approved by the FDA for the topical treatment of plaque psoriasis, including intertriginous areas, in patients 12 years of age and older. Submission of the sNDA is based on positive results from the Interventional Trial Evaluating Roflumilast Cream for the Treatment of Atopic Dermatitis (INTEGUMENT-1 and INTEGUMENT-2) trials; two identical Phase 3, vehicle-controlled trials in which roflumilast cream 0.15% or vehicle was applied once daily for 4 weeks to individuals 6 years of age and older with mild to moderate AD involving at least 3% body surface area. Roflumilast is a phosphodiesterase-4 (PDE-4) inhibitor.
According to a press release from Arcutis, both studies met the primary endpoint of IGA Success, which was defined as a validated Investigator Global Assessment – Atopic Dermatitis (vIGA-AD) score of ‘clear’ or ‘almost clear’ plus a 2-grade improvement from baseline at week 4. In INTEGUMENT-1 this endpoint was achieved by 32.0% of subjects in the roflumilast cream group vs. 15.2% of those in the vehicle group (P < .0001). In INTEGUMENT-2, this endpoint was achieved by 28.9% of subjects in the roflumilast cream group vs. 12.0% of those in the vehicle group (P < .0001). The most common adverse reactions based on data from the combined trials were headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
Diabetes patients satisfied with continuous glucose monitors
TOPLINE:
However, significant proportions also reported concerns about accuracy under certain circumstances and about skin problems.
METHODOLOGY:
Researchers did an online survey of 504 people with type 1 diabetes from the T1D Exchange and 101 with type 2 diabetes from the Dynata database.
TAKEAWAY:
- The Dexcom G6 device was used by 60.7% of all current CGM users, including 69% of those with type 1 diabetes vs. 12% with type 2 diabetes.
- People with type 2 diabetes were more likely to use older Dexcom versions (G4/G5) (32%) or Abbott’s FreeStyle Libre systems (35%).
- Overall, 90% agreed that most sensors were accurate, but just 79% and 78%, respectively, were satisfied with sensor performance on the first and last day of wear.
- Moreover, 42% suspected variations in accuracy from sensor to sensor, and 32% continue to perform finger-stick monitoring more than six times a week.
- Individuals with type 2 diabetes were more likely than those with type 1 diabetes to be concerned about poor sensor performance affecting confidence in making diabetes management decisions (52% vs. 19%).
- Over half reported skin reactions and/or pain with the sensors (53.7% and 55.4%, respectively).
- Concerns about medications affecting sensor accuracy were more common among those with type 2 vs. type 1 diabetes (65% vs. 29%).
- Among overall concerns about substances or situations affecting sensor accuracy, the top choice (47%) was dehydration (despite a lack of supportive published literature), followed by pain medications (43%), cold/flu medications (32%), and coffee (24%).
- Inaccurate/false alarms negatively affected daily life for 36% of participants and diabetes management for 34%.
IN PRACTICE:
“CGM is a game-changing technology and has evolved in the past decade to overcome many technical and usability obstacles. Our survey suggests that there remain areas for further improvement ... Mistrust in CGM performance was more common than expected.”
SOURCE:
The study was done by Elizabeth Holt, of LifeScan, and colleagues. It was published in Clinical Diabetes.
LIMITATIONS:
- The databases used to recruit study participants may not be representative of the entire respective patient populations.
- Exercise wasn’t given as an option for affecting CGM accuracy, which might partly explain the dehydration finding.
DISCLOSURES:
Funding for this study and preparation of the manuscript were provided by LifeScan Inc. Two authors are LifeScan employees, and two others currently work for the T1D Exchange.
A version of this article first appeared on Medscape.com.
TOPLINE:
However, significant proportions also reported concerns about accuracy under certain circumstances and about skin problems.
METHODOLOGY:
Researchers did an online survey of 504 people with type 1 diabetes from the T1D Exchange and 101 with type 2 diabetes from the Dynata database.
TAKEAWAY:
- The Dexcom G6 device was used by 60.7% of all current CGM users, including 69% of those with type 1 diabetes vs. 12% with type 2 diabetes.
- People with type 2 diabetes were more likely to use older Dexcom versions (G4/G5) (32%) or Abbott’s FreeStyle Libre systems (35%).
- Overall, 90% agreed that most sensors were accurate, but just 79% and 78%, respectively, were satisfied with sensor performance on the first and last day of wear.
- Moreover, 42% suspected variations in accuracy from sensor to sensor, and 32% continue to perform finger-stick monitoring more than six times a week.
- Individuals with type 2 diabetes were more likely than those with type 1 diabetes to be concerned about poor sensor performance affecting confidence in making diabetes management decisions (52% vs. 19%).
- Over half reported skin reactions and/or pain with the sensors (53.7% and 55.4%, respectively).
- Concerns about medications affecting sensor accuracy were more common among those with type 2 vs. type 1 diabetes (65% vs. 29%).
- Among overall concerns about substances or situations affecting sensor accuracy, the top choice (47%) was dehydration (despite a lack of supportive published literature), followed by pain medications (43%), cold/flu medications (32%), and coffee (24%).
- Inaccurate/false alarms negatively affected daily life for 36% of participants and diabetes management for 34%.
IN PRACTICE:
“CGM is a game-changing technology and has evolved in the past decade to overcome many technical and usability obstacles. Our survey suggests that there remain areas for further improvement ... Mistrust in CGM performance was more common than expected.”
SOURCE:
The study was done by Elizabeth Holt, of LifeScan, and colleagues. It was published in Clinical Diabetes.
LIMITATIONS:
- The databases used to recruit study participants may not be representative of the entire respective patient populations.
- Exercise wasn’t given as an option for affecting CGM accuracy, which might partly explain the dehydration finding.
DISCLOSURES:
Funding for this study and preparation of the manuscript were provided by LifeScan Inc. Two authors are LifeScan employees, and two others currently work for the T1D Exchange.
A version of this article first appeared on Medscape.com.
TOPLINE:
However, significant proportions also reported concerns about accuracy under certain circumstances and about skin problems.
METHODOLOGY:
Researchers did an online survey of 504 people with type 1 diabetes from the T1D Exchange and 101 with type 2 diabetes from the Dynata database.
TAKEAWAY:
- The Dexcom G6 device was used by 60.7% of all current CGM users, including 69% of those with type 1 diabetes vs. 12% with type 2 diabetes.
- People with type 2 diabetes were more likely to use older Dexcom versions (G4/G5) (32%) or Abbott’s FreeStyle Libre systems (35%).
- Overall, 90% agreed that most sensors were accurate, but just 79% and 78%, respectively, were satisfied with sensor performance on the first and last day of wear.
- Moreover, 42% suspected variations in accuracy from sensor to sensor, and 32% continue to perform finger-stick monitoring more than six times a week.
- Individuals with type 2 diabetes were more likely than those with type 1 diabetes to be concerned about poor sensor performance affecting confidence in making diabetes management decisions (52% vs. 19%).
- Over half reported skin reactions and/or pain with the sensors (53.7% and 55.4%, respectively).
- Concerns about medications affecting sensor accuracy were more common among those with type 2 vs. type 1 diabetes (65% vs. 29%).
- Among overall concerns about substances or situations affecting sensor accuracy, the top choice (47%) was dehydration (despite a lack of supportive published literature), followed by pain medications (43%), cold/flu medications (32%), and coffee (24%).
- Inaccurate/false alarms negatively affected daily life for 36% of participants and diabetes management for 34%.
IN PRACTICE:
“CGM is a game-changing technology and has evolved in the past decade to overcome many technical and usability obstacles. Our survey suggests that there remain areas for further improvement ... Mistrust in CGM performance was more common than expected.”
SOURCE:
The study was done by Elizabeth Holt, of LifeScan, and colleagues. It was published in Clinical Diabetes.
LIMITATIONS:
- The databases used to recruit study participants may not be representative of the entire respective patient populations.
- Exercise wasn’t given as an option for affecting CGM accuracy, which might partly explain the dehydration finding.
DISCLOSURES:
Funding for this study and preparation of the manuscript were provided by LifeScan Inc. Two authors are LifeScan employees, and two others currently work for the T1D Exchange.
A version of this article first appeared on Medscape.com.
Steady VKA therapy beats switch to NOAC in frail AFib patients: FRAIL-AF
Switching frail patients with atrial fibrillation (AFib) from anticoagulation therapy with vitamin K antagonists (VKAs) to a novel oral anticoagulant (NOAC) resulted in more bleeding without any reduction in thromboembolic complications or all-cause mortality, randomized trial results show.
The study, FRAIL-AF, is the first randomized NOAC trial to exclusively include frail older patients, said lead author Linda P.T. Joosten, MD, Julius Center for Health Sciences and Primary Care in Utrecht, the Netherlands, and these unexpected findings provide evidence that goes beyond what is currently available.
“Data from the FRAIL-AF trial showed that switching from a VKA to a NOAC should not be considered without a clear indication in frail older patients with AF[ib], as switching to a NOAC leads to 69% more bleeding,” she concluded, without any benefit on secondary clinical endpoints, including thromboembolic events and all-cause mortality.
“The results turned out different than we expected,” Dr. Joosten said. “The hypothesis of this superiority trial was that switching from VKA therapy to a NOAC would result in less bleeding. However, we observed the opposite. After the interim analysis, the data and safety monitoring board advised to stop inclusion because switching from a VKA to a NOAC was clearly contraindicated with a hazard ratio of 1.69 and a highly significant P value of .001.”
Results of FRAIL-AF were presented at the annual congress of the European Society of Cardiology and published online in the journal Circulation.
Session moderator Renate B. Schnabel, MD, interventional cardiologist with University Heart & Vascular Center Hamburg (Germany), congratulated the researchers on these “astonishing” data.
“The thing I want to emphasize here is that, in the absence of randomized controlled trial data, we should be very cautious in extrapolating data from the landmark trials to populations not enrolled in those, and to rely on observational data only,” Dr. Schnabel told Dr. Joosten. “We need randomized controlled trials that sometimes give astonishing results.”
Frailty a clinical syndrome
Frailty is “a lot more than just aging, multiple comorbidities and polypharmacy,” Dr. Joosten explained. “It’s really a clinical syndrome, with people with a high biological vulnerability, dependency on significant others, and a reduced capacity to resist stressors, all leading to a reduced homeostatic reserve.”
Frailty is common in the community, with a prevalence of about 12%, she noted, “and even more important, AF[ib] in frail older people is very common, with a prevalence of 18%. And “without any doubt, we have to adequately anticoagulate frail AF[ib] patients, as they have a high stroke risk, with an incidence of 12.4% per year,” Dr. Joosten noted, compared with 3.9% per year among nonfrail AFib patients.
NOACs are preferred over VKAs in nonfrail AFib patients, after four major trials, RE-LY with dabigatran, ROCKET-AF with rivaroxaban, ARISTOTLE with apixaban, and ENGAGE-AF with edoxaban, showed that NOAC treatment resulted in less major bleeding while stroke risk was comparable with treatment with warfarin, she noted.
The 2023 European Heart Rhythm Association consensus document on management of arrhythmias in frailty syndrome concludes that the advantages of NOACs relative to VKAs are “likely consistent” in frail and nonfrail AFib patients, but the level of evidence is low.
So it’s unknown if NOACs are preferred over VKAs in frail AFib patients, “and it’s even more questionable whether patients on VKAs should switch to NOAC therapy,” Dr. Joosten said.
This new trial aimed to answer the question of whether switching frail AFib patients currently managed on a VKA to a NOAC would reduce bleeding. FRAIL-AF was a pragmatic, multicenter, open-label, randomized, controlled superiority trial.
Older AFib patients were deemed frail if they were aged 75 years or older and had a score of 3 or more on the validated Groningen Frailty Indicator (GFI). Patients with a glomerular filtration rate of less than 30 mL/min per 1.73 m2 or with valvular AFib were excluded.
Eligible patients were then assigned randomly to switch from their international normalized ratio (INR)–guided VKA treatment with either 1 mg acenocoumarol or 3 mg phenprocoumon, to a NOAC, or to continue VKA treatment. They were followed for 12 months for the primary outcome – major bleeding or clinically relevant nonmajor bleeding complication, whichever came first – accounting for death as a competing risk.
A total of 1,330 patients were randomly assigned between January 2018 and June 2022. Their mean age was 83 years, and they had a median GFI of 4. After randomization, 6 patients in the switch-to-NOAC arm, and 1 in the continue-VKA arm were found to have exclusion criteria, so in the end, 662 patients were switched from a VKA to NOAC, while 661 continued on VKA therapy. The choice of NOAC was made by the treating physician.
Major bleeding was defined as a fatal bleeding; bleeding in a critical area or organ; bleeding leading to transfusion; and/or bleeding leading to a fall in hemoglobin level of 2 g/dL (1.24 mmol/L) or more. Nonmajor bleeding was bleeding not considered major but requiring face-to-face consultation, hospitalization or increased level of care, or medical intervention.
After a prespecified futility analysis planned after 163 primary outcome events, the trial was halted when it was seen that there were 101 primary outcome events in the switch arm compared to 62 in the continue arm, Dr. Joosten said. The difference appeared to be driven by clinically relevant nonmajor bleeding.
Secondary outcomes of thromboembolic events and all-cause mortality were similar between the groups.
Completely different patients
Discussant at the meeting for the presentation was Isabelle C. Van Gelder, MD, University Medical Centre Groningen (the Netherlands). She said the results are important and relevant because it “provides data on an important gap of knowledge in our AF[ib] guidelines, and a note for all the cardiologists – this study was not done in the hospital. This trial was done in general practitioner practices, so that’s important to consider.”
Comparing FRAIL-AF patients with those of the four previous NOAC trials, “you see that enormous difference in age,” with an average age of 83 years versus 70-73 years in those trials. “These are completely different patients than have been included previously,” she said.
That GFI score of 4 or more includes patients on four or more different types of medication, as well as memory complaints, an inability to walk around the house, and problems with vision or hearing.
The finding of a 69% increase in bleeding with NOACs in FRAIL-AF was “completely unexpected, and I think that we as cardiologists and as NOAC believers did not expect it at all, but it is as clear as it is.” The curves don’t diverge immediately, but rather after 3 months or thereafter, “so it has nothing to do with the switching process. So why did it occur?”
The Netherlands has dedicated thrombosis services that might improve time in therapeutic range for VKA patients, but there is no real difference in TTRs in FRAIL-AF versus the other NOAC trials, Dr. Van Gelder noted.
The most likely suspect in her view is frailty itself, in particular the tendency for patients to be on a high number of medications. A previous study showed, for example, that polypharmacy could be used as a proxy for the effect of frailty on bleeding risk; patients on 10 or more medications had a higher risk for bleeding on treatment with rivaroxaban versus those on 4 or fewer medications.
“Therefore, in my view, why was there such a high risk of bleeding? It’s because these are other patients than we are normally used to treat, we as cardiologists,” although general practitioners see these patients all the time. “It’s all about frailty.”
NOACs are still relatively new drugs, with possible unknown interactions, she added. Because of their frailty and polypharmacy, these patients may benefit from INR control, Dr. Van Gelder speculated. “Therefore, I agree with them that we should be careful; if such old, frail patients survive on VKA, do not change medications and do not switch!”
The study was supported by the Dutch government with additional and unrestricted educational grants from Boehringer Ingelheim, BMS-Pfizer, Bayer, and Daiichi Sankyo. Dr. Joosten reported no relevant financial relationships. Dr. Van Gelder reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Switching frail patients with atrial fibrillation (AFib) from anticoagulation therapy with vitamin K antagonists (VKAs) to a novel oral anticoagulant (NOAC) resulted in more bleeding without any reduction in thromboembolic complications or all-cause mortality, randomized trial results show.
The study, FRAIL-AF, is the first randomized NOAC trial to exclusively include frail older patients, said lead author Linda P.T. Joosten, MD, Julius Center for Health Sciences and Primary Care in Utrecht, the Netherlands, and these unexpected findings provide evidence that goes beyond what is currently available.
“Data from the FRAIL-AF trial showed that switching from a VKA to a NOAC should not be considered without a clear indication in frail older patients with AF[ib], as switching to a NOAC leads to 69% more bleeding,” she concluded, without any benefit on secondary clinical endpoints, including thromboembolic events and all-cause mortality.
“The results turned out different than we expected,” Dr. Joosten said. “The hypothesis of this superiority trial was that switching from VKA therapy to a NOAC would result in less bleeding. However, we observed the opposite. After the interim analysis, the data and safety monitoring board advised to stop inclusion because switching from a VKA to a NOAC was clearly contraindicated with a hazard ratio of 1.69 and a highly significant P value of .001.”
Results of FRAIL-AF were presented at the annual congress of the European Society of Cardiology and published online in the journal Circulation.
Session moderator Renate B. Schnabel, MD, interventional cardiologist with University Heart & Vascular Center Hamburg (Germany), congratulated the researchers on these “astonishing” data.
“The thing I want to emphasize here is that, in the absence of randomized controlled trial data, we should be very cautious in extrapolating data from the landmark trials to populations not enrolled in those, and to rely on observational data only,” Dr. Schnabel told Dr. Joosten. “We need randomized controlled trials that sometimes give astonishing results.”
Frailty a clinical syndrome
Frailty is “a lot more than just aging, multiple comorbidities and polypharmacy,” Dr. Joosten explained. “It’s really a clinical syndrome, with people with a high biological vulnerability, dependency on significant others, and a reduced capacity to resist stressors, all leading to a reduced homeostatic reserve.”
Frailty is common in the community, with a prevalence of about 12%, she noted, “and even more important, AF[ib] in frail older people is very common, with a prevalence of 18%. And “without any doubt, we have to adequately anticoagulate frail AF[ib] patients, as they have a high stroke risk, with an incidence of 12.4% per year,” Dr. Joosten noted, compared with 3.9% per year among nonfrail AFib patients.
NOACs are preferred over VKAs in nonfrail AFib patients, after four major trials, RE-LY with dabigatran, ROCKET-AF with rivaroxaban, ARISTOTLE with apixaban, and ENGAGE-AF with edoxaban, showed that NOAC treatment resulted in less major bleeding while stroke risk was comparable with treatment with warfarin, she noted.
The 2023 European Heart Rhythm Association consensus document on management of arrhythmias in frailty syndrome concludes that the advantages of NOACs relative to VKAs are “likely consistent” in frail and nonfrail AFib patients, but the level of evidence is low.
So it’s unknown if NOACs are preferred over VKAs in frail AFib patients, “and it’s even more questionable whether patients on VKAs should switch to NOAC therapy,” Dr. Joosten said.
This new trial aimed to answer the question of whether switching frail AFib patients currently managed on a VKA to a NOAC would reduce bleeding. FRAIL-AF was a pragmatic, multicenter, open-label, randomized, controlled superiority trial.
Older AFib patients were deemed frail if they were aged 75 years or older and had a score of 3 or more on the validated Groningen Frailty Indicator (GFI). Patients with a glomerular filtration rate of less than 30 mL/min per 1.73 m2 or with valvular AFib were excluded.
Eligible patients were then assigned randomly to switch from their international normalized ratio (INR)–guided VKA treatment with either 1 mg acenocoumarol or 3 mg phenprocoumon, to a NOAC, or to continue VKA treatment. They were followed for 12 months for the primary outcome – major bleeding or clinically relevant nonmajor bleeding complication, whichever came first – accounting for death as a competing risk.
A total of 1,330 patients were randomly assigned between January 2018 and June 2022. Their mean age was 83 years, and they had a median GFI of 4. After randomization, 6 patients in the switch-to-NOAC arm, and 1 in the continue-VKA arm were found to have exclusion criteria, so in the end, 662 patients were switched from a VKA to NOAC, while 661 continued on VKA therapy. The choice of NOAC was made by the treating physician.
Major bleeding was defined as a fatal bleeding; bleeding in a critical area or organ; bleeding leading to transfusion; and/or bleeding leading to a fall in hemoglobin level of 2 g/dL (1.24 mmol/L) or more. Nonmajor bleeding was bleeding not considered major but requiring face-to-face consultation, hospitalization or increased level of care, or medical intervention.
After a prespecified futility analysis planned after 163 primary outcome events, the trial was halted when it was seen that there were 101 primary outcome events in the switch arm compared to 62 in the continue arm, Dr. Joosten said. The difference appeared to be driven by clinically relevant nonmajor bleeding.
Secondary outcomes of thromboembolic events and all-cause mortality were similar between the groups.
Completely different patients
Discussant at the meeting for the presentation was Isabelle C. Van Gelder, MD, University Medical Centre Groningen (the Netherlands). She said the results are important and relevant because it “provides data on an important gap of knowledge in our AF[ib] guidelines, and a note for all the cardiologists – this study was not done in the hospital. This trial was done in general practitioner practices, so that’s important to consider.”
Comparing FRAIL-AF patients with those of the four previous NOAC trials, “you see that enormous difference in age,” with an average age of 83 years versus 70-73 years in those trials. “These are completely different patients than have been included previously,” she said.
That GFI score of 4 or more includes patients on four or more different types of medication, as well as memory complaints, an inability to walk around the house, and problems with vision or hearing.
The finding of a 69% increase in bleeding with NOACs in FRAIL-AF was “completely unexpected, and I think that we as cardiologists and as NOAC believers did not expect it at all, but it is as clear as it is.” The curves don’t diverge immediately, but rather after 3 months or thereafter, “so it has nothing to do with the switching process. So why did it occur?”
The Netherlands has dedicated thrombosis services that might improve time in therapeutic range for VKA patients, but there is no real difference in TTRs in FRAIL-AF versus the other NOAC trials, Dr. Van Gelder noted.
The most likely suspect in her view is frailty itself, in particular the tendency for patients to be on a high number of medications. A previous study showed, for example, that polypharmacy could be used as a proxy for the effect of frailty on bleeding risk; patients on 10 or more medications had a higher risk for bleeding on treatment with rivaroxaban versus those on 4 or fewer medications.
“Therefore, in my view, why was there such a high risk of bleeding? It’s because these are other patients than we are normally used to treat, we as cardiologists,” although general practitioners see these patients all the time. “It’s all about frailty.”
NOACs are still relatively new drugs, with possible unknown interactions, she added. Because of their frailty and polypharmacy, these patients may benefit from INR control, Dr. Van Gelder speculated. “Therefore, I agree with them that we should be careful; if such old, frail patients survive on VKA, do not change medications and do not switch!”
The study was supported by the Dutch government with additional and unrestricted educational grants from Boehringer Ingelheim, BMS-Pfizer, Bayer, and Daiichi Sankyo. Dr. Joosten reported no relevant financial relationships. Dr. Van Gelder reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Switching frail patients with atrial fibrillation (AFib) from anticoagulation therapy with vitamin K antagonists (VKAs) to a novel oral anticoagulant (NOAC) resulted in more bleeding without any reduction in thromboembolic complications or all-cause mortality, randomized trial results show.
The study, FRAIL-AF, is the first randomized NOAC trial to exclusively include frail older patients, said lead author Linda P.T. Joosten, MD, Julius Center for Health Sciences and Primary Care in Utrecht, the Netherlands, and these unexpected findings provide evidence that goes beyond what is currently available.
“Data from the FRAIL-AF trial showed that switching from a VKA to a NOAC should not be considered without a clear indication in frail older patients with AF[ib], as switching to a NOAC leads to 69% more bleeding,” she concluded, without any benefit on secondary clinical endpoints, including thromboembolic events and all-cause mortality.
“The results turned out different than we expected,” Dr. Joosten said. “The hypothesis of this superiority trial was that switching from VKA therapy to a NOAC would result in less bleeding. However, we observed the opposite. After the interim analysis, the data and safety monitoring board advised to stop inclusion because switching from a VKA to a NOAC was clearly contraindicated with a hazard ratio of 1.69 and a highly significant P value of .001.”
Results of FRAIL-AF were presented at the annual congress of the European Society of Cardiology and published online in the journal Circulation.
Session moderator Renate B. Schnabel, MD, interventional cardiologist with University Heart & Vascular Center Hamburg (Germany), congratulated the researchers on these “astonishing” data.
“The thing I want to emphasize here is that, in the absence of randomized controlled trial data, we should be very cautious in extrapolating data from the landmark trials to populations not enrolled in those, and to rely on observational data only,” Dr. Schnabel told Dr. Joosten. “We need randomized controlled trials that sometimes give astonishing results.”
Frailty a clinical syndrome
Frailty is “a lot more than just aging, multiple comorbidities and polypharmacy,” Dr. Joosten explained. “It’s really a clinical syndrome, with people with a high biological vulnerability, dependency on significant others, and a reduced capacity to resist stressors, all leading to a reduced homeostatic reserve.”
Frailty is common in the community, with a prevalence of about 12%, she noted, “and even more important, AF[ib] in frail older people is very common, with a prevalence of 18%. And “without any doubt, we have to adequately anticoagulate frail AF[ib] patients, as they have a high stroke risk, with an incidence of 12.4% per year,” Dr. Joosten noted, compared with 3.9% per year among nonfrail AFib patients.
NOACs are preferred over VKAs in nonfrail AFib patients, after four major trials, RE-LY with dabigatran, ROCKET-AF with rivaroxaban, ARISTOTLE with apixaban, and ENGAGE-AF with edoxaban, showed that NOAC treatment resulted in less major bleeding while stroke risk was comparable with treatment with warfarin, she noted.
The 2023 European Heart Rhythm Association consensus document on management of arrhythmias in frailty syndrome concludes that the advantages of NOACs relative to VKAs are “likely consistent” in frail and nonfrail AFib patients, but the level of evidence is low.
So it’s unknown if NOACs are preferred over VKAs in frail AFib patients, “and it’s even more questionable whether patients on VKAs should switch to NOAC therapy,” Dr. Joosten said.
This new trial aimed to answer the question of whether switching frail AFib patients currently managed on a VKA to a NOAC would reduce bleeding. FRAIL-AF was a pragmatic, multicenter, open-label, randomized, controlled superiority trial.
Older AFib patients were deemed frail if they were aged 75 years or older and had a score of 3 or more on the validated Groningen Frailty Indicator (GFI). Patients with a glomerular filtration rate of less than 30 mL/min per 1.73 m2 or with valvular AFib were excluded.
Eligible patients were then assigned randomly to switch from their international normalized ratio (INR)–guided VKA treatment with either 1 mg acenocoumarol or 3 mg phenprocoumon, to a NOAC, or to continue VKA treatment. They were followed for 12 months for the primary outcome – major bleeding or clinically relevant nonmajor bleeding complication, whichever came first – accounting for death as a competing risk.
A total of 1,330 patients were randomly assigned between January 2018 and June 2022. Their mean age was 83 years, and they had a median GFI of 4. After randomization, 6 patients in the switch-to-NOAC arm, and 1 in the continue-VKA arm were found to have exclusion criteria, so in the end, 662 patients were switched from a VKA to NOAC, while 661 continued on VKA therapy. The choice of NOAC was made by the treating physician.
Major bleeding was defined as a fatal bleeding; bleeding in a critical area or organ; bleeding leading to transfusion; and/or bleeding leading to a fall in hemoglobin level of 2 g/dL (1.24 mmol/L) or more. Nonmajor bleeding was bleeding not considered major but requiring face-to-face consultation, hospitalization or increased level of care, or medical intervention.
After a prespecified futility analysis planned after 163 primary outcome events, the trial was halted when it was seen that there were 101 primary outcome events in the switch arm compared to 62 in the continue arm, Dr. Joosten said. The difference appeared to be driven by clinically relevant nonmajor bleeding.
Secondary outcomes of thromboembolic events and all-cause mortality were similar between the groups.
Completely different patients
Discussant at the meeting for the presentation was Isabelle C. Van Gelder, MD, University Medical Centre Groningen (the Netherlands). She said the results are important and relevant because it “provides data on an important gap of knowledge in our AF[ib] guidelines, and a note for all the cardiologists – this study was not done in the hospital. This trial was done in general practitioner practices, so that’s important to consider.”
Comparing FRAIL-AF patients with those of the four previous NOAC trials, “you see that enormous difference in age,” with an average age of 83 years versus 70-73 years in those trials. “These are completely different patients than have been included previously,” she said.
That GFI score of 4 or more includes patients on four or more different types of medication, as well as memory complaints, an inability to walk around the house, and problems with vision or hearing.
The finding of a 69% increase in bleeding with NOACs in FRAIL-AF was “completely unexpected, and I think that we as cardiologists and as NOAC believers did not expect it at all, but it is as clear as it is.” The curves don’t diverge immediately, but rather after 3 months or thereafter, “so it has nothing to do with the switching process. So why did it occur?”
The Netherlands has dedicated thrombosis services that might improve time in therapeutic range for VKA patients, but there is no real difference in TTRs in FRAIL-AF versus the other NOAC trials, Dr. Van Gelder noted.
The most likely suspect in her view is frailty itself, in particular the tendency for patients to be on a high number of medications. A previous study showed, for example, that polypharmacy could be used as a proxy for the effect of frailty on bleeding risk; patients on 10 or more medications had a higher risk for bleeding on treatment with rivaroxaban versus those on 4 or fewer medications.
“Therefore, in my view, why was there such a high risk of bleeding? It’s because these are other patients than we are normally used to treat, we as cardiologists,” although general practitioners see these patients all the time. “It’s all about frailty.”
NOACs are still relatively new drugs, with possible unknown interactions, she added. Because of their frailty and polypharmacy, these patients may benefit from INR control, Dr. Van Gelder speculated. “Therefore, I agree with them that we should be careful; if such old, frail patients survive on VKA, do not change medications and do not switch!”
The study was supported by the Dutch government with additional and unrestricted educational grants from Boehringer Ingelheim, BMS-Pfizer, Bayer, and Daiichi Sankyo. Dr. Joosten reported no relevant financial relationships. Dr. Van Gelder reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE ESC CONGRESS 2023
‘New dawn’ for aldosterone as drug target in hypertension?
Once-daily treatment with the selective aldosterone synthase inhibitor lorundrostat (Mineralys Therapeutics) safely and significantly reduced blood pressure in adults with uncontrolled hypertension in a phase 2, randomized, controlled trial.
Eight weeks after adding lorundrostat (50 mg or 100 mg once daily) or placebo to background therapy, the medication lowered seated automated office systolic BP significantly more than placebo (−9.6 mm Hg with 50 mg; −7.8 mm Hg with 100 mg), with the greatest effects seen in adults with obesity.
“We need new drugs for treatment-resistant hypertension,” study investigator Steven Nissen, MD, chief academic officer at the Heart Vascular & Thoracic Institute at the Cleveland Clinic, said in an interview. Lorundrostat represents a “new class” of antihypertensive that “looks to be safe and we’re seeing very large reductions in blood pressure.”
Results of the Target-HTN trial were published online in JAMA to coincide with presentation at the Hypertension Scientific Sessions, sponsored by the American Heart Association.
Aldosterone’s contribution ‘vastly underappreciated’
Excess aldosterone production contributes to uncontrolled BP in patients with obesity and other associated diseases, such as obstructive sleep apnea and metabolic syndrome.
“Aldosterone’s contribution to uncontrolled hypertension is vastly underappreciated,” first author and study presenter Luke Laffin, MD, also with the Cleveland Clinic, said in an interview.
Aldosterone synthase inhibitors are a novel class of BP-lowering medications that decrease aldosterone production. Lorundrostat is one of two such agents in advanced clinical development. The other is baxdrostat (CinCor Pharma/AstraZeneca).
The Target-HTN randomized, placebo-controlled, dose-ranging trial enrolled 200 adults (mean age, 66 years; 60% women) with uncontrolled hypertension while taking two or more antihypertensive medications; 42% of participants were taking three or more antihypertensive medications, 48% were obese and 40% had diabetes.
The study population was divided into two cohorts: an initial cohort of 163 adults with suppressed plasma renin activity at baseline (PRA ≤ 1.0 ng/mL per hour) and elevated plasma aldosterone (≥ 1.0 ng/dL) and a second cohort of 37 adults with PRA greater than 1.0 ng/mL per hour.
Participants were randomly assigned to placebo or one of five doses of lorundrostat in the initial cohort (12.5 mg, 50 mg, or 100 mg once daily or 12.5 mg or 25 mg twice daily).
In the second cohort, participants were randomly assigned (1:6) to placebo or lorundrostat 100 mg once daily. The primary endpoint was change in automated office systolic BP from baseline to week 8.
Among participants with suppressed PRA, following 8 weeks of treatment, changes in office systolic BP of −14.1, −13.2, and −6.9 mm Hg were observed with 100 mg, 50 mg, and 12.5 mg once-daily lorundrostat, respectively, compared with a change of −4.1 mm Hg with placebo.
Reductions in systolic BP in individuals receiving twice-daily doses of 25 mg and 12.5 mg of lorundrostat were −10.1 and −13.8 mm Hg, respectively.
Among participants without suppressed PRA, lorundrostat 100 mg once daily decreased systolic BP by 11.4 mm Hg, similar to BP reduction in those with suppressed PRA receiving the same dose.
A prespecified subgroup analysis showed that participants with obesity demonstrated greater BP lowering in response to lorundrostat.
No instances of cortisol insufficiency occurred. Six participants had increases in serum potassium above 6.0 mEq/L (6.0 mmol/L) that corrected with dose reduction or drug discontinuation.
The increase in serum potassium is “expected and manageable,” Dr. Laffin said in an interview. “Anytime you disrupt aldosterone production, you’re going to have to have an increase in serum potassium, but it’s very manageable and not something that is worrisome.”
A phase 2 trial in 300 adults with uncontrolled hypertension is currently underway. The trial will evaluate the BP-lowering effects of lorundrostat, administered on a background of a standardized antihypertensive medication regimen. A larger phase 3 study will start before the end of the year.
‘New dawn’ for therapies targeting aldosterone
The author of an editorial in JAMA noted that more 70 years after the first isolation of aldosterone, then called electrocortin, “there is a new dawn for therapies targeting aldosterone.”
“There is now real potential to provide better-targeted treatment for patients in whom aldosterone excess is known to contribute to their clinical condition and influence their clinical outcome, notably those with difficult-to-control hypertension, obesity, heart failure, chronic kidney disease, and the many with yet-to-be-diagnosed primary aldosteronism,” said Bryan Williams, MD, University College London.
The trial was funded by Mineralys Therapeutics, which is developing lorundrostat. Dr. Laffin reported that the Cleveland Clinic, his employer, was a study site for the Target-HTN trial and that C5Research, the academic research organization of the Cleveland Clinic, receives payment for services related to other Mineralys clinical trials. Dr. Laffin also reported receipt of personal fees from Medtronic, Lilly, and Crispr Therapeutics, grants from AstraZeneca, and stock options for LucidAct Health and Gordy Health. Dr. Nissen reported receipt of grants from Mineralys during the conduct of the study and grants from AbbVie, AstraZeneca, Amgen, Bristol-Myers Squibb, Lilly, Esperion Therapeutics, Medtronic, grants from MyoKardia, New Amsterdam Pharmaceuticals, Novartis, and Silence Therapeutics. Dr. Williams reported being the unremunerated chair of the steering committee designing a phase 3 trial of the aldosterone synthase inhibitor baxdrostat for AstraZeneca.
A version of this article first appeared on Medscape.com.
Once-daily treatment with the selective aldosterone synthase inhibitor lorundrostat (Mineralys Therapeutics) safely and significantly reduced blood pressure in adults with uncontrolled hypertension in a phase 2, randomized, controlled trial.
Eight weeks after adding lorundrostat (50 mg or 100 mg once daily) or placebo to background therapy, the medication lowered seated automated office systolic BP significantly more than placebo (−9.6 mm Hg with 50 mg; −7.8 mm Hg with 100 mg), with the greatest effects seen in adults with obesity.
“We need new drugs for treatment-resistant hypertension,” study investigator Steven Nissen, MD, chief academic officer at the Heart Vascular & Thoracic Institute at the Cleveland Clinic, said in an interview. Lorundrostat represents a “new class” of antihypertensive that “looks to be safe and we’re seeing very large reductions in blood pressure.”
Results of the Target-HTN trial were published online in JAMA to coincide with presentation at the Hypertension Scientific Sessions, sponsored by the American Heart Association.
Aldosterone’s contribution ‘vastly underappreciated’
Excess aldosterone production contributes to uncontrolled BP in patients with obesity and other associated diseases, such as obstructive sleep apnea and metabolic syndrome.
“Aldosterone’s contribution to uncontrolled hypertension is vastly underappreciated,” first author and study presenter Luke Laffin, MD, also with the Cleveland Clinic, said in an interview.
Aldosterone synthase inhibitors are a novel class of BP-lowering medications that decrease aldosterone production. Lorundrostat is one of two such agents in advanced clinical development. The other is baxdrostat (CinCor Pharma/AstraZeneca).
The Target-HTN randomized, placebo-controlled, dose-ranging trial enrolled 200 adults (mean age, 66 years; 60% women) with uncontrolled hypertension while taking two or more antihypertensive medications; 42% of participants were taking three or more antihypertensive medications, 48% were obese and 40% had diabetes.
The study population was divided into two cohorts: an initial cohort of 163 adults with suppressed plasma renin activity at baseline (PRA ≤ 1.0 ng/mL per hour) and elevated plasma aldosterone (≥ 1.0 ng/dL) and a second cohort of 37 adults with PRA greater than 1.0 ng/mL per hour.
Participants were randomly assigned to placebo or one of five doses of lorundrostat in the initial cohort (12.5 mg, 50 mg, or 100 mg once daily or 12.5 mg or 25 mg twice daily).
In the second cohort, participants were randomly assigned (1:6) to placebo or lorundrostat 100 mg once daily. The primary endpoint was change in automated office systolic BP from baseline to week 8.
Among participants with suppressed PRA, following 8 weeks of treatment, changes in office systolic BP of −14.1, −13.2, and −6.9 mm Hg were observed with 100 mg, 50 mg, and 12.5 mg once-daily lorundrostat, respectively, compared with a change of −4.1 mm Hg with placebo.
Reductions in systolic BP in individuals receiving twice-daily doses of 25 mg and 12.5 mg of lorundrostat were −10.1 and −13.8 mm Hg, respectively.
Among participants without suppressed PRA, lorundrostat 100 mg once daily decreased systolic BP by 11.4 mm Hg, similar to BP reduction in those with suppressed PRA receiving the same dose.
A prespecified subgroup analysis showed that participants with obesity demonstrated greater BP lowering in response to lorundrostat.
No instances of cortisol insufficiency occurred. Six participants had increases in serum potassium above 6.0 mEq/L (6.0 mmol/L) that corrected with dose reduction or drug discontinuation.
The increase in serum potassium is “expected and manageable,” Dr. Laffin said in an interview. “Anytime you disrupt aldosterone production, you’re going to have to have an increase in serum potassium, but it’s very manageable and not something that is worrisome.”
A phase 2 trial in 300 adults with uncontrolled hypertension is currently underway. The trial will evaluate the BP-lowering effects of lorundrostat, administered on a background of a standardized antihypertensive medication regimen. A larger phase 3 study will start before the end of the year.
‘New dawn’ for therapies targeting aldosterone
The author of an editorial in JAMA noted that more 70 years after the first isolation of aldosterone, then called electrocortin, “there is a new dawn for therapies targeting aldosterone.”
“There is now real potential to provide better-targeted treatment for patients in whom aldosterone excess is known to contribute to their clinical condition and influence their clinical outcome, notably those with difficult-to-control hypertension, obesity, heart failure, chronic kidney disease, and the many with yet-to-be-diagnosed primary aldosteronism,” said Bryan Williams, MD, University College London.
The trial was funded by Mineralys Therapeutics, which is developing lorundrostat. Dr. Laffin reported that the Cleveland Clinic, his employer, was a study site for the Target-HTN trial and that C5Research, the academic research organization of the Cleveland Clinic, receives payment for services related to other Mineralys clinical trials. Dr. Laffin also reported receipt of personal fees from Medtronic, Lilly, and Crispr Therapeutics, grants from AstraZeneca, and stock options for LucidAct Health and Gordy Health. Dr. Nissen reported receipt of grants from Mineralys during the conduct of the study and grants from AbbVie, AstraZeneca, Amgen, Bristol-Myers Squibb, Lilly, Esperion Therapeutics, Medtronic, grants from MyoKardia, New Amsterdam Pharmaceuticals, Novartis, and Silence Therapeutics. Dr. Williams reported being the unremunerated chair of the steering committee designing a phase 3 trial of the aldosterone synthase inhibitor baxdrostat for AstraZeneca.
A version of this article first appeared on Medscape.com.
Once-daily treatment with the selective aldosterone synthase inhibitor lorundrostat (Mineralys Therapeutics) safely and significantly reduced blood pressure in adults with uncontrolled hypertension in a phase 2, randomized, controlled trial.
Eight weeks after adding lorundrostat (50 mg or 100 mg once daily) or placebo to background therapy, the medication lowered seated automated office systolic BP significantly more than placebo (−9.6 mm Hg with 50 mg; −7.8 mm Hg with 100 mg), with the greatest effects seen in adults with obesity.
“We need new drugs for treatment-resistant hypertension,” study investigator Steven Nissen, MD, chief academic officer at the Heart Vascular & Thoracic Institute at the Cleveland Clinic, said in an interview. Lorundrostat represents a “new class” of antihypertensive that “looks to be safe and we’re seeing very large reductions in blood pressure.”
Results of the Target-HTN trial were published online in JAMA to coincide with presentation at the Hypertension Scientific Sessions, sponsored by the American Heart Association.
Aldosterone’s contribution ‘vastly underappreciated’
Excess aldosterone production contributes to uncontrolled BP in patients with obesity and other associated diseases, such as obstructive sleep apnea and metabolic syndrome.
“Aldosterone’s contribution to uncontrolled hypertension is vastly underappreciated,” first author and study presenter Luke Laffin, MD, also with the Cleveland Clinic, said in an interview.
Aldosterone synthase inhibitors are a novel class of BP-lowering medications that decrease aldosterone production. Lorundrostat is one of two such agents in advanced clinical development. The other is baxdrostat (CinCor Pharma/AstraZeneca).
The Target-HTN randomized, placebo-controlled, dose-ranging trial enrolled 200 adults (mean age, 66 years; 60% women) with uncontrolled hypertension while taking two or more antihypertensive medications; 42% of participants were taking three or more antihypertensive medications, 48% were obese and 40% had diabetes.
The study population was divided into two cohorts: an initial cohort of 163 adults with suppressed plasma renin activity at baseline (PRA ≤ 1.0 ng/mL per hour) and elevated plasma aldosterone (≥ 1.0 ng/dL) and a second cohort of 37 adults with PRA greater than 1.0 ng/mL per hour.
Participants were randomly assigned to placebo or one of five doses of lorundrostat in the initial cohort (12.5 mg, 50 mg, or 100 mg once daily or 12.5 mg or 25 mg twice daily).
In the second cohort, participants were randomly assigned (1:6) to placebo or lorundrostat 100 mg once daily. The primary endpoint was change in automated office systolic BP from baseline to week 8.
Among participants with suppressed PRA, following 8 weeks of treatment, changes in office systolic BP of −14.1, −13.2, and −6.9 mm Hg were observed with 100 mg, 50 mg, and 12.5 mg once-daily lorundrostat, respectively, compared with a change of −4.1 mm Hg with placebo.
Reductions in systolic BP in individuals receiving twice-daily doses of 25 mg and 12.5 mg of lorundrostat were −10.1 and −13.8 mm Hg, respectively.
Among participants without suppressed PRA, lorundrostat 100 mg once daily decreased systolic BP by 11.4 mm Hg, similar to BP reduction in those with suppressed PRA receiving the same dose.
A prespecified subgroup analysis showed that participants with obesity demonstrated greater BP lowering in response to lorundrostat.
No instances of cortisol insufficiency occurred. Six participants had increases in serum potassium above 6.0 mEq/L (6.0 mmol/L) that corrected with dose reduction or drug discontinuation.
The increase in serum potassium is “expected and manageable,” Dr. Laffin said in an interview. “Anytime you disrupt aldosterone production, you’re going to have to have an increase in serum potassium, but it’s very manageable and not something that is worrisome.”
A phase 2 trial in 300 adults with uncontrolled hypertension is currently underway. The trial will evaluate the BP-lowering effects of lorundrostat, administered on a background of a standardized antihypertensive medication regimen. A larger phase 3 study will start before the end of the year.
‘New dawn’ for therapies targeting aldosterone
The author of an editorial in JAMA noted that more 70 years after the first isolation of aldosterone, then called electrocortin, “there is a new dawn for therapies targeting aldosterone.”
“There is now real potential to provide better-targeted treatment for patients in whom aldosterone excess is known to contribute to their clinical condition and influence their clinical outcome, notably those with difficult-to-control hypertension, obesity, heart failure, chronic kidney disease, and the many with yet-to-be-diagnosed primary aldosteronism,” said Bryan Williams, MD, University College London.
The trial was funded by Mineralys Therapeutics, which is developing lorundrostat. Dr. Laffin reported that the Cleveland Clinic, his employer, was a study site for the Target-HTN trial and that C5Research, the academic research organization of the Cleveland Clinic, receives payment for services related to other Mineralys clinical trials. Dr. Laffin also reported receipt of personal fees from Medtronic, Lilly, and Crispr Therapeutics, grants from AstraZeneca, and stock options for LucidAct Health and Gordy Health. Dr. Nissen reported receipt of grants from Mineralys during the conduct of the study and grants from AbbVie, AstraZeneca, Amgen, Bristol-Myers Squibb, Lilly, Esperion Therapeutics, Medtronic, grants from MyoKardia, New Amsterdam Pharmaceuticals, Novartis, and Silence Therapeutics. Dr. Williams reported being the unremunerated chair of the steering committee designing a phase 3 trial of the aldosterone synthase inhibitor baxdrostat for AstraZeneca.
A version of this article first appeared on Medscape.com.
FROM HYPERTENSION 2023